Open Access Article
Open Access ArticlePhysicochemical and bioactive characterization of microencapsulated Phyllanthus emblica kombucha powder through foam mat drying
Nur Haniza
Ewandi Jong
 ab,
Norhayati
Muhammad
ab,
Norhayati
Muhammad
 *a,
Norazlin
Abdullah
*a,
Norazlin
Abdullah
 a and
Dayang Norulfairuz
Abang Zaidel
a and
Dayang Norulfairuz
Abang Zaidel
 cd
cd
aDepartment of Technology and Natural Resources, Faculty of Applied Sciences and Technology, Universiti Tun Hussein Onn Malaysia, Pagoh Educational Hub, 84600 Pagoh, Johor Darul Takzim, Malaysia. E-mail: norhayatim@uthm.edu.my
bPoliteknik Tun Syed Nasir Syed Ismail, Hab Pendidikan Tinggi Pagoh, KM 1 Jalan Panchor 84600 Pagoh, Johor, Malaysia
cMalaysia – Japan International Institute of Technology (MJIIT), Universiti Teknologi Malaysia, Jalan Sultan Yahya Petra, 54100 Kuala Lumpur, Malaysia
dInnovation Centre in Agritechnology for Advanced Bioprocessing, Universiti Teknologi Malaysia Pagoh Campus, 84600 Pagoh, Johor Darul Takzim, Malaysia
First published on 15th December 2025
Abstract
Interest in botanical kombucha enriched with bioactive compounds has expanded, especially those that integrate Phyllanthus emblica. However, its sour taste and astringency limit its acceptability. This study aims to examine the physicochemical and bioactive properties of a microencapsulated foam mat-dried kombucha powder developed from Phyllanthus emblica extract and nipa palm sugar. The liquid kombucha was encapsulated through foam mat drying at 65 °C for 5 hours using orange, pomegranate and P. emblica pomace powder, which were then tested for encapsulation efficiency. Characterization of the quality attributes involved the total phenolic content, 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging activity, pH, moisture content, total soluble solid, ethanol content, powder properties, proximate analysis and toxicity test. The powdered kombucha encapsulated with P. emblica pomace powder demonstrated the highest encapsulation efficiency of 86.10% with a total phenolic content of 998 ± 35.41 mg GAE per g. It showed a DPPH radical scavenging activity value of 63.62% ± 0.78%, pH of 3.09 ± 0.04, moisture content of 1.67 ± 0.40, and a total soluble solid of 7.80 ± 0.30 (Brix°) and met the halal regulatory limit with an ethanol content of 0.31% ± 0.02%. Proximate analysis revealed 391.10 ± 0.15 kcal/100 g, 96.60 ± 0.01 g carbohydrates, 0.87 ± 0.02 g protein, 0.63 ± 0.00 g fibre, and 0 g fat. A toxicity evaluation using the brine shrimp lethality assay yielded an LC50 of 1043.87 µg mL−1, confirming its nontoxicity. Overall, the foam mat-dried P. emblica kombucha powder demonstrated promising physicochemical stability and bioactivity, underscoring its potential as a safe and value-added ingredient in functional beverages.
Sustainability spotlightThis study advances sustainable food innovation by transforming underutilized tropical resources; Phyllanthus emblica and Nypa fruticans into a functional, shelf-stable kombucha powder using energy-efficient foam mat drying. The integration of natural carrier materials from fruit pomace promotes waste valorization and supports circular bioeconomy principles. By reducing product perishability and enhancing nutritional value, this research aligns with the UN Sustainable Development Goals (SDGs), particularly SDG 2 (Zero Hunger), SDG 3 (Good Health and Well-Being), and SDG 12 (Responsible Consumption and Production). The findings contribute to sustainable nutraceutical development and local resource utilization, fostering healthier diets and reducing environmental impact in functional beverage production. |
1 Introduction
In recent years, demand for natural, nutrient-dense foods has risen due to increasing emphasis on health and well-being. Among these, fruit-based beverages have gained popularity, particularly those enhanced with functional ingredients that promote health beyond basic nutrition.1 Thirst quenching is no longer the sole focus of modern beverage development, as there is also an emphasis on delivering physiological benefits, improving mental and physical health, and reducing the risk of nutrition-related diseases.2 Within this context, by aligning with the current economic and health trends, kombucha has emerged as one of the most promising functional beverages.3 Traditionally, kombucha is produced using a symbiotic culture of bacteria and yeast (SCOBY) through the fermentation of sweetened black or green tea.4 The nutritional value, bioavailability, sensory quality and shelf stability are enhanced by fermentation while preserving the thermolabile nutrients.5 Studies report that consumer interest in kombucha is mostly driven by its purported health benefits, including immunomodulatory, cardioprotective, anti-inflammatory, anti-microbial and anticancer properties.6 The physicochemical composition, bioactivities, and sensory traits of kombucha are largely influenced by the type of substrate. Thus, incorporating novel substrates often enriches the product with additional bioactive compounds such as phenolics.7Fruits and botanicals have been recently explored in studies as alternatives, although tea remains the conventional substrate.8 Among these, Phyllanthus emblica is especially noteworthy. P. emblica is valued for its exceptionally high vitamin C content and strong antioxidant activity. This fruit has diverse biofunctional properties and is therefore widely used in food, cosmetics, and healthcare industries.9 Despite these advantages, its astringent and sour taste limit its consumption and reduce consumer acceptance.1 In Malaysia, the tree is commonly cultivated as an ornamental plant, particularly in the state of Melaka, where its historical significance is linked to the naming of the state. However, much of the fruit goes unused and is wasted annually.10,11 This highlights a missed opportunity for valorisation of P. emblica into functional food applications that could increase its dietary relevance.12
In order to improve palatability and consumer acceptance, the use of natural sweeteners is essential. Nipa palm sugar has emerged as a desirable alternative to refined sugar due to its unique flavour, nutritional value, and low glycaemic index.13 Unlike highly processed cane sugar, nipa palm sugar undergoes minimal processing without chemical additives and is rich in minerals and vitamins.14 Research indicates that the flavour of nipa palm sugar is influenced by volatile compounds formed during heat processing. It has a nutty, roasted flavour due to the high concentration of pyrazines, whilst a larger ratio of furan derivatives results in a sweet, burnt, caramelized flavour. The phenolic compounds and brown pigments produced during the Maillard reaction and caramelization are also the main sources of antioxidants in nipa palm sugar.15 However, despite its potential, nipa palm sugar remains a niche commodity and is underutilised in food innovation. Younger generations are less familiar with its distinct taste, contributing to a gradual decline in its cultural appreciation.16 Incorporating nipa palm sugar into kombucha not only provides a healthier sweetener alternative, but also supports the revival of traditional ingredients.
At the same time, functional foods and beverages are increasingly being transformed into powdered forms to enhance their stability, portability, and shelf life. In this domain, foam mat drying (FMD) stands out as an efficient and cost-effective drying method that is particularly suited for heat-sensitive bioactive-rich liquids.17 Although FMD has been studied with various fruit juices and vegetable matrices, its application to kombucha powders (especially those based on innovative substrates like P. emblica) remains unexplored. P. emblica is a rich source of vitamin C and polyphenols, and exhibits potent antioxidant activity, but its high astringency and sensitivity to thermal degradation pose formulation challenges.9 For this reason, an effective encapsulation method is essential for preserving its functional properties in powdered form.
Hence, this study aims to apply FMD as an encapsulation technique to produce a stable, bioactive-rich, and potentially stable kombucha powder made from an alternative substrate of P. emblica extract with the use of fruit pomace powder as an encapsulating agent. The investigation focuses on how FMD preserves the physicochemical properties, bioactivity of the phenolic content and antioxidant activity, and the safety profile of the final powder, establishing it as a viable functional and halal-compliant beverage ingredient.
2 Materials and methods
2.1 Chemicals and materials
P. emblica fruits were collected in Merlimau, Melaka, while nipa palm sugar was procured from SPL Food Industries Sdn Bhd (Sarawak) with halal certification. The kombucha starter culture (SCOBY) was purchased from Spoon Health (M) Sdn Bhd (Wondrous, Negeri Sembilan). Pomegranate and orange pomace powders were purchased from Raison Ayurvedic. Maltodextrin and egg albumin powder were purchased from BisChem Technology Sdn Bhd. Gallic acid, 2,2-diphenyl-1-picrylhydrazyl, ascorbic acid and Folin–Ciocalteu's phenol reagent were purchased from Sigma Aldrich, USA. Sodium carbonate, methanol, and ethanol were purchased from Bendosen, Malaysia. All utilized chemicals were of analytical grade and procured locally.2.2 Formulation of the P. emblica kombucha powder
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (w/w).19 Bioactive compounds are mostly carried by maltodextrin (MD). Owing to its exceptional functional properties, water solubility, high glass transition temperature, and low cost, MD finds extensive applications in food items.21 The homogenized kombucha-carrier agent underwent encapsulation via foam mat drying, utilizing egg albumin as the foaming agent according to Saentaweesuk.22 This involved a 37.25% egg albumin solution at a 1% weight concentration. Egg albumin was dissolved in filtered water to achieve the desired concentration.
1 (w/w).19 Bioactive compounds are mostly carried by maltodextrin (MD). Owing to its exceptional functional properties, water solubility, high glass transition temperature, and low cost, MD finds extensive applications in food items.21 The homogenized kombucha-carrier agent underwent encapsulation via foam mat drying, utilizing egg albumin as the foaming agent according to Saentaweesuk.22 This involved a 37.25% egg albumin solution at a 1% weight concentration. Egg albumin was dissolved in filtered water to achieve the desired concentration.
The kombucha-encapsulation agent combination was then gradually stirred with a speed of 1500 rpm using IKA RW20. The mixing process continued for 5 minutes. This drying process included spreading the homogenized mixture onto a foam mat to create a uniform layer. It was then subjected to controlled drying conditions at a temperature of around 65 °C for a duration of 5 hours using a food dehydrator (Excalibur, USA).22,23 The drying temperature of 65 °C was selected based on a previous foam-mat drying study, which reported that temperatures between 60–70 °C effectively reduce moisture while minimizing the degradation of heat-sensitive phenolics and ascorbic acid.22 In addition, foam-mat drying requires 4 to 6 hours for complete moisture removal in fruit-based foams.23 The dried material was ground into a fine powder using a high-speed blender (Panasonic MX-151SG). The encapsulated kombucha particles were stored in PE bags at 4 °C until further analysis. Table 1 shows the formulation ratio for the encapsulation and production of the encapsulated kombucha powder using different carrier materials.
| Ingredients | Ratio of formulation (per 500 mL) (%) | ||
|---|---|---|---|
| Sample PPP | Sample OPP | Sample EPP | |
| Kombucha-like beverage liquid | 26.37 | 26.37 | 26.37 |
| Carrier agent | 26.37 | 26.37 | 26.37 |
| Maltodextrin | 10 | 10 | 10 |
| 1% egg albumin | 37.25 | 37.25 | 37.25 |
| Total | 100 | 100 | 100 |
Following encapsulation and foam mat drying, the resulting powders were analysed for their total phenolic content to determine the extent of bioactive retention post-processing.
2.3 Determination of the total phenolic content (TPC)
The total phenolic content (TPC) was determined using the Folin–Ciocalteu colorimetric method.24 A diluted sample of 20 µL (using a dilution factor of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) was mixed with 100 µL of 10% (v/v) Folin–Ciocalteu reagent in a 96-well plate and allowed to rest in the dark at 25 °C for 8 minutes. Then, 80 µL of 7.5% (w/v) sodium carbonate was added to the mixture and allowed to rest in the dark at 24 °C for 30 minutes. The absorbance was measured at 765 nm. Gallic acid was used as a standard. The results were presented in milligrams of gallic acid equivalent per millilitre (mg GAE per mL). The obtained TPC values were used to further calculate the encapsulation efficiency, providing insight into the effectiveness of the carrier materials.
10) was mixed with 100 µL of 10% (v/v) Folin–Ciocalteu reagent in a 96-well plate and allowed to rest in the dark at 25 °C for 8 minutes. Then, 80 µL of 7.5% (w/v) sodium carbonate was added to the mixture and allowed to rest in the dark at 24 °C for 30 minutes. The absorbance was measured at 765 nm. Gallic acid was used as a standard. The results were presented in milligrams of gallic acid equivalent per millilitre (mg GAE per mL). The obtained TPC values were used to further calculate the encapsulation efficiency, providing insight into the effectiveness of the carrier materials.
2.4 Determination of the encapsulation efficiency
The Folin–Ciocalteu method was utilised to ascertain the phenolic content (PC) of the encapsulated powder. A single gram of the sample was mixed with 10 mL of distilled water, shaken well, and then run through a 0.45 µm filter. Distilled water (10 mL volume) was used to wash 1 g of powder onto filter paper to measure the surface phenolic content (SPC). The weight of the encapsulated solid mass was divided by the entire amount of solid mass that needed to be dried and the encapsulation yield was computed.19 The encapsulation efficiency (% EE) was calculated as follows (eqn (1)):| %EE = ((TPC − SPC))/TPC × 100 | (1) |
The obtained EE values provided insights into the effectiveness of the carrier material and were thus used to determine the final encapsulating agent for the kombucha powder.
2.5 Determination of 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging activity
The DPPH assay was conducted based on the procedure written by Gao et al.25 Briefly, 1.5 mL of each sample and 3 mL of DPPH solution (200 µM) were mixed. After incubation for 30 min under dark conditions at 25 °C, the decrease in absorbance at 515 nm was measured, and the free-radical scavenging activity was calculated as follows (eqn (2)):| Scavenging activity (%) = ((Absorbance of control − Absorbance of sample)/(Absorbance of control)) × 100 | (2) |
2.6 Determination of the physicochemical properties
2.7 Proximate analysis
Adapting a method described by Masitlha et al., the powdered sample moisture and ash contents were ascertained using AOAC (1996) procedures 930.15 and 942.05, respectively.29 The micro-Kjeldahl method, as detailed in AOAC (1996) (method 960.5), was used to calculate the crude protein (CP) content in percentage. The Soxhlet method (method 920.39C), as published by AOAC (1996), was used to determine the natural fat content. The amount of crude fibre was measured using the non-enzymatic gravimetric method, and the difference was then used to estimate the total amount of carbohydrates, as per AOAC (1996).2.8 Toxicity test using the brine shrimp lethality assay (BSLA)
As described by Asaduzzaman et al., the brine shrimp lethality assay (BSLA) was used for toxicity testing with minor modifications and applied to the kombucha powder sample.30 Artificial seawater was created by dissolving 19 grams of sea salt in 500 mL of distilled water, serving as a medium for hatching brine shrimp. The shrimp eggs were placed in a partitioned container with dark and light areas for hatching. The eggs were transferred to the dark side for oxygen during hatching, with a lamp on the light side attracting the hatched shrimp. After two days, the nauplii were transferred to fresh, clear artificial seawater for visibility. Ten nauplii were carefully placed in test tubes using a micropipette. A stock solution was prepared by dissolving 10 g of the implant sample in 100 mL of distilled water. Serial dilutions with concentrations of 1000, 500, 250, 100, and 10 ppm were prepared from the stock solution. Each concentration was transferred into labelled test tubes containing ten nauplii and 1 mL of seawater. After 24 hours, the number of dead nauplii was counted against a black background using a magnifying glass, and the death percentage was calculated as in (eqn(3)).| Death (%) = (Number of dead nauplii)/(Number of dead nauplii + Number of live nauplii) × 100 | (3) |
2.9 Analysis of data
The data underwent analysis utilizing IBM SPSS Statistics (Version 27.0, Chicago). The findings were displayed as the mean plus or minus the standard deviation (SD), and analysed statistically by analysis of variance (ANOVA). A P value <0.05 was used to determine the statistical significance. The data were obtained in triplicate.3 Results and discussion
3.1 Total phenolic content (TPC), surface phenolic content (SPC), encapsulation efficiency (EE) and DPPH radical scavenging activity
In this study, the P. emblica kombucha-like beverage was successfully encapsulated using a combination of maltodextrin (MD) and fruit pomace powders (namely, pomegranate pomace powder (PPP), orange pomace powder (OPP) and P. emblica pomace powder (EPP)) through the foam mat drying technique.Among the encapsulated powders, the EPP formulation exhibited the highest encapsulation efficiency (86.15% ± 0.10%), significantly different from both PPP and OPP samples (p < 0.05). This was attributed to EPP's smaller SPC value (227.96 ± 0.97 mg GAE per g), suggesting less phenolic loss on the surface and better retention within the matrix. Lower SPC values are desirable because they indicate that bioactive compounds are better protected from oxidation, light, and heat exposure.19 While all three powders showed a significant drop in TPC compared to the original liquid kombucha-like sample (1633.2 ± 85.31 mg GAE per g), this decrease is expected due to heat exposure during the drying and partial degradation of phenolics.31 The highest retained TPC among the powders was found in EPP encapsulated powder (998.0 ± 35.40 mg GAE per g), followed closely by OPP and PPP. These values reflect the effectiveness of the EPP matrix in preserving the antioxidant compounds of kombucha through foam mat drying.
The variation in SPC values also reflects the quality of encapsulation. EPP showed the lowest SPC, indicating that more polyphenols were successfully embedded within the matrix rather than loosely adhered on the surface.32 Furthermore, the differences in EE values among the carrier agents may be attributed to the physicochemical characteristics of the wall materials used. For example, maltodextrin, used in all formulations at 10% in combination with the respective pomace powders, is widely reported to enhance the encapsulation of polyphenols due to its low viscosity, good solubility, and high glass transition temperature.22 Meanwhile, the unique fibrous composition and polyphenol content of P. emblica pomace may have synergistically improved the matrix's barrier properties, enhancing entrapment and reducing the oxidation of phenolics.22 These findings are supported by M. Saifullah et al., who reported similar trends where maltodextrin-based wall systems in combination with citrus and plant-based pomaces improved both polyphenol retention and flow properties.19 The total phenolic content (TPC), surface phenolic content (SPC), encapsulation efficiency (EE) of all the encapsulated kombucha powder samples were evaluated and are presented in Table 2.
| Analysis | P. emblica liquid kombucha | PPP encapsulated | OPP encapsulated | EPP encapsulated | Positive control (ascorbic acid standard) |
|---|---|---|---|---|---|
| a Values are expressed as mean ± standard deviation (n = 3). Different superscript letters within the same row indicate significant differences (p-value < 0.05). | |||||
| TPC (mg GAE per g) | 1633.20 ± 85.31a | 773.90 ± 42.52b | 951.06 ± 23.21b | 998.00 ± 35.40b | |
| SPC (mg GAE per g) | NA | 260.17 ± 0.81b | 291.15 ± 1.99c | 227.96 ± 0.97a | |
| EE (%) | NA | 84.02 ± 0.01b | 82.04 ± 0.10c | 86.15 ± 0.10a | |
| DPPH radical scavenging activity | 89.68 ± 0.75b | NA | NA | 63.62 ± 0.78c | 95.05 ± 0.30a |
In their study, when used with maltodextrin, lemon pomace powder (LPP) showed not only high EE but also enhanced powder handling and bioactive release in simulated gastrointestinal conditions.
In this study, P. emblica pomace powder was successfully applied as a carrier material for the foam mat drying of the P. emblica kombucha liquid. The pomace powder aids in minimizing surface phenolic loss, and retained a high total phenolic content. These findings align with the known antioxidant capacity of P. emblica derived matrices and their potential for protecting sensitive compounds during thermal processing.32
Following the TPC, SPC and EE value, the kombucha powder encapsulated with EPP was selected for detailed physicochemical analysis and proceeded as the final formulation for the kombucha-like beverage powder. The DPPH radical scavenging activity of encapsulated powder kombucha was recorded at 63.62% ± 0.78%. This value still reflects a moderate antioxidant capacity.33 The relatively lower scavenging rate in the powdered kombucha can be attributed to the dilution of active compounds due to the addition of carrier agents and possible degradation of some polyphenolic compounds during the foam mat drying process.34
A study by Antolak et al. discovered that liquid kombucha typically maintains a higher concentration of active compounds due to the fermentation process, which is less effective in powdered forms.4 Similar observations have been reported by Jayabalan et al., who noted that the antioxidant activity in kombucha tends to slightly decrease when subjected to heat or prolonged drying, even though some polyphenols remain bioavailable.34 Furthermore, since the main ingredient was P. emblica extract which is known for its potent hydrophilic antioxidant compounds, these compounds may perform more effectively in aqueous radical environments.18 This result is also in line with a study by Saifullah et al., where the choice of encapsulation carrier and drying method can influence not just the total phenolic content, but also the chemical form and solubility of antioxidants, thereby affecting their scavenging behavior differently across assays.19
These findings also underscore the fact that although the encapsulated kombucha powder may have slightly lower radical scavenging performance than the liquid kombucha, it still retains significant antioxidant functionality. This behaviour is consistent with findings in other natural antioxidant systems, where processing leads to partial degradation or dilution of active constituents.35 When compared conceptually with high-performance antioxidant systems such as metal–organic complexes containing copper, gold, or silver, the activity of natural P. emblica kombucha is expectedly milder but exhibits superior biocompatibility and safety.9,36 These metal-containing complexes often demonstrate stronger DPPH inhibition but may also show reduced chemical purity and potential cytotoxic effects, as reported in recent studies.36 Therefore, while the antioxidant strength of P. emblica kombucha powder does not match that of metal-enhanced hybrid materials, its natural origin, safety, and functional food compatibility position it as a valuable nutraceutical ingredient.
The differentiation between fermented and unfermented extracts of P. emblica is pivotal in ascertaining their functional and antimicrobial characteristics. The fermentation of kombucha, facilitated by the symbiotic culture of bacteria and yeast (SCOBY), results in an elevation of organic acid levels (including acetic, gluconic, and lactic acids), bioavailable polyphenols, and ascorbic acid, all of which collectively enhance the antimicrobial efficacy in comparison to the unfermented extract.9 Jayabalan et al. and Villarreal-Soto et al. have documented that the fermentation process modifies intrinsic phenolic compounds into smaller, more bioactive metabolites that exhibit heightened antibacterial activity, while concurrently enhancing the antioxidant capacity.34,35
Notably, kombucha displays a selective antimicrobial effect. The organic acids and polyphenols preferentially inhibit pathogenic and opportunistic bacteria, such as Escherichia coli, Staphylococcus aureus, and Salmonella spp., whilst the beneficial gut microbiota (including species of Lactobacillus and Bifidobacterium) demonstrate enhanced tolerance to these compounds due to their acid-resistant physiological adaptations and their ability to metabolize specific phenolics.8,37 This selective inhibition indicates that P. emblica kombucha may serve as a microbiome-supportive beverage, exerting antibacterial effects in vitro while preserving compatibility with probiotic microorganisms in vivo. Given that fermentation markedly enhances the release and transformation of bioactive compounds, it is anticipated that fermented P. emblica kombucha will exhibit superior functional properties compared to its unfermented variant, thus reinforcing its prospective applications within nutraceutical frameworks.8
Moreover, given its organic ingredients, halal compliance, and botanical origin, this kombucha powder remains a promising candidate for functional beverage powders targeting health-conscious markets and convenience.
3.2 Physicochemical analysis of the encapsulated kombucha powder
Evaluating physicochemical properties is crucial for determining the commercial potential of the encapsulated P. emblica kombucha powder (EKP) as a functional beverage.7 For comparative purposes, a commercial black tea kombucha powder (CBTK) was also included in the analysis, serving as a benchmark for standard quality attributes. The pH value of the reconstituted EKP was slightly lower (3.09 ± 0.04) compared to CBTK (3.21 ± 0.02), indicating a higher acidity which may contribute to improved microbial safety and shelf stability.35 Both values fall within the acceptable range for kombucha beverages, as reported by Villarreal-Soto et al., where typical kombucha pH values range from 2.5 to 3.5.35It is also important to acknowledge that the CBTK product included for comparison is not foam-mat dried. Foam-mat drying is an emerging technology for kombucha processing and has been predominantly documented in experimental studies, with no known large-scale commercial applications to date. As a result, the physicochemical variations identified between EKP and CBTK may, in part, be indicative of disparities in the processing methodologies, rather than solely reflecting the inherent quality of the products themselves. Nevertheless, the utilization of CBTK serves as a significant benchmark for comprehending the extent to which EKP aligns with or diverges from prevailing market standards.
The moisture content in EKP was significantly lower (1.67% ± 0.4%) than that in CBTK (3.95% ± 0.5%), suggesting enhanced shelf stability and reduced susceptibility to microbial spoilage.17 The low moisture content of both samples, especially the EKP, contributes to the low bulk density of the sample.33 EKP showed a higher total soluble solid (7.8 ± 0.3 °Brix) compared to the commercial sample (6.5 ± 0.4 °Brix), likely due to residual sugars from the nipa palm sugar or incomplete sugar utilization during a shorter fermentation period. The use of maltodextrin in the formulation may also contribute to this value. Table 3 represents a summary of results obtained for both the encapsulated kombucha powder (EKP) and commercial black tea kombucha (CBTK).
| Parameter | Encapsulated kombucha powder (EKP) | Commercial black tea kombucha powder (CBTK) |
|---|---|---|
| a Different superscripts (a, b) indicate significant differences among samples (p-value < 0.05) according to the post hoc test. | ||
| pH value | 3.09 ± 0.04a | 3.21 ± 0.02b |
| Total soluble solids (°Brix) | 7.80 ± 0.30a | 6.50 ± 0.40b |
| Moisture content (%) | 1.67 ± 0.40a | 3.95 ± 0.50b |
| Ethanol content (% v/v) | 0.31 ± 0.02a | 0.41 ± 0.03b |
| Bulk density (g mL−1) | 0.37 ± 0.05a | 0.43 ± 0.01b |
| Tap density (g mL−1) | 0.47 ± 0.01a | 0.55 ± 0.02b |
On the other hand, the ethanol content in EKP was 0.31% ± 0.02% v/v, well below the 1% ABV threshold for halal compliance as stipulated by JAKIM (Food Act 1983), making it suitable for Muslim consumers.38 In contrast, the commercial powder CBTK had a higher ethanol content of 0.41% ± 0.03% v/v. Moreover, the use of foam mat drying and careful temperature control during the drying process (65 °C for 5 hours) of EKP likely contributed to the further reduction of residual ethanol, as ethanol is volatile and can partially evaporate during drying.39
This additional step helped enhance the product's halal suitability, while preserving its functional properties. For functional food products intended for commercialization in Malaysia and other Muslim-majority countries, the halal status is a critical factor influencing consumer acceptance.40 Halal certification ensures that the product complies with Islamic dietary laws, which strictly prohibit the consumption of alcohol. According to the Special Muzakarah of the Fatwa Committee of the National Council for Malaysian Islamic Religious Affairs (JAKIM, 2011), beverages are considered halal if they are not intentionally fermented to produce alcohol and the alcohol content remains below 1% v/v, if for the purpose of commercialization.37 Any product exceeding this threshold may be deemed questionable or non-compliant, potentially excluding a large segment of health-conscious Muslim consumers.
The ability of a powder to flow smoothly plays an important role in how easily it can be processed, packaged, and handled, both in production and by consumers. In this study, two key flow-related characteristics (which are bulk density and tap density) were assessed to better understand how the properties of the encapsulated kombucha powder compare to those of a commercial black tea kombucha. The results showed that both bulk and tap densities were significantly lower in EKP (p-value = 0.004 and 0.003, respectively). This suggests that EKP has a lighter, more aerated structure, which is likely due to the foam mat drying technique used and the presence of botanical fiber from the carrier material.19,41 These lower density values are beneficial, especially for instant beverage powders, as they tend to dissolve and disperse more readily when reconstituted.
Overall, the physicochemical profile of the encapsulated kombucha powder demonstrated promising stability, acceptable flow properties, and halal-compliant ethanol levels, supporting its potential for market readiness as a functional botanical kombucha powder.
3.3 Field emission scanning electron microscopy (FESEM)
Field emission scanning electron microscopy (FESEM) was used to visualize the surface morphology of the encapsulated kombucha powder at 500× and 1000× magnification. The micrographs in Fig. 1 reveal that the powder sample had an irregular particle shape and microstructure, indicating a broad size distribution with an average particle size of between 12.9–54.31 µm. The particles in the samples were unstructured, and lacked a defined cellular structure and internal porosity. This is likely contributed by the fibrous structure of the P. emblica pomace powder that was used to encapsulate the kombucha.32 Moreover, after the drying procedure of the foam mat, the resultant powder particles were acquired through the mechanical grinding of the desiccated kombucha.40 The mechanical disintegration associated with the drying and grinding procedures resulted in challenges of accurately identifying the cellular characteristics within the powder.19 | ||
| Fig. 1 Surface morphology and particle distribution at (a) 500× magnification and (b) 1000× magnification. | ||
Even though the powdered kombucha does not demonstrate uniform encapsulation, the particle dimensions achieved are adequate for powder formulations that necessitate optimal dispersibility and solubility, while concurrently minimizing the generation of dust.42 This is supported by a previous study by Shekunov et al., where they discovered that smaller particles (such as those in the 50–60 µm range) manage to disperse more uniformly in a medium, enhancing the solubility and ensuring consistent performance in formulations.42
Furthermore, M. Saifullah et al. and K. Muzaffar et al. reported analogous findings in their investigations concerning lemon-scented pomace and amla pomace powder.19,32 The identified porous architecture is beneficial for facilitating rapid rehydration and efficient dissolution, characteristics that augment the applicability of the powder as an instant functional beverage.43
In addition, sustaining particle dimensions above the conventional 50 µm threshold mitigates dustiness and enhances bulk management, given that larger particles are less susceptible to the formation of powder dust. In a separate investigation, Kaderides and Goula discovered that polyphenolic extracts encapsulated within orange pomace dietary fibre exhibited an irregular morphology and were amorphous in nature.44 The encapsulation of the powder was accomplished through a spray drying technique. When it comes to food items like kombucha powder, the particle sizes greatly affect the uniformity and dissolvability of the product. A size range of 50–60 µm is ideal for ensuring a smooth texture and quick dissolution in beverages.19,42,43
3.4 Proximate analysis for nutrition composition
The proximate compositions of the kombucha-like powder encapsulated with PPP were evaluated to understand its basic nutritional profile. The powder displayed a substantial carbohydrate concentration of 96.60 ± 0.02 g/100 g, a minimal protein content (0.87 ± 0.03 g/100 g), and no detectable fat, culminating in a total caloric value of 391.10 ± 0.15 kcal/100 g. These results are in alignment with prior investigations concerning powdered kombucha products derived from fruit-based substrates, which characteristically exhibit elevated carbohydrate density attributable to residual sugars and polysaccharides originating from both the fermentation medium and fruit matrix.8Notably, the product included 0.63 ± 0.01 g/100 g of dietary fiber, mainly derived from the P. emblica pomace used as a carrier material. Although the dietary fibre content is minimal on a per 100 g basis, its incorporation elevates the nutritional profile of the beverage powder, particularly when compared to traditional kombucha powders that generally lack fibre due to filtration methods employed prior to drying.33 Dietary fibre, particularly from fruit pomace sources, has been extensively documented to support gastrointestinal health, promote feelings of fullness, and assist in the regulation of blood glucose levels.34Table 4 summarizes the nutritional composition of the botanical kombucha-like powder.
| Nutrient | Per 100 g | Per serving (15 g) |
|---|---|---|
| Energy | 391.10 ± 0.15 kcal | 58.67 ± 0.15 kcal |
| Carbohydrate | 96.60 ± 0.02 g | 14.50 ± 0.02 g |
| Protein | 0.87 ± 0.03 g | 0.13 ± 0.03 g |
| Fat | 0 g | 0 g |
| Dietary fibre | 0.63 ± 0.01 g | 0.09 ± 0.01 g |
3.5 Toxicity test using the brine shrimp lethality assay (BSLA)
The brine shrimp lethality assay (BSLA) was employed to assess the preliminary toxicity of the kombucha powder encapsulated with P. emblica pomace. This assay is recognized for its simplicity, cost-effectiveness, and reliability in evaluating the bioactivity of natural products, including plant extracts and fermented beverages.45The BSLA assay utility in kombucha research is supported by prior findings indicating that toxicity levels may fluctuate depending on fermentation duration, substrate composition, and the concentration of bioactive compounds.34,35 While kombucha is classically regarded as safe, the accumulation of organic acids, ethanol, microbial metabolites or concentrated phenolics after the drying process could potentially modify its toxicity, thereby necessitating safety verification for powdered formulations.
A study by Murphy et al. highlighted the importance of safety verification for kombucha, particularly in powdered formulations, due to the potential toxicity from accumulated organic acids, ethanol, microbial metabolites, or concentrated phenolics, which may arise during the drying process.46
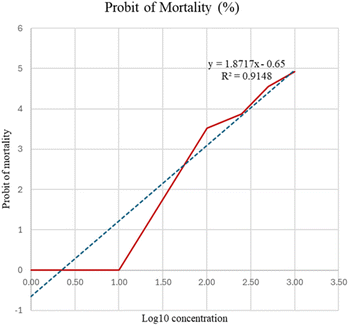
In this study, varying concentrations of the encapsulated kombucha powder were tested against Artemia salina nauplii. Mortality rates were recorded after 24 hours of exposure. The data were analysed using probit analysis. The probit of mortality for the encapsulated kombucha powder is presented in Fig. 2.
This approach involves plotting the logarithm of extract concentrations against the probit-transformed percentage mortality using the Finney table of probit mortality.47 The regression model is represented by y as the probit of mortality and x as the log10 of concentration (µg mL−1). The high R2 value (0.9148) indicates a strong linear relationship between the concentration and mortality, confirming the validity of the test. Using this model, the median lethal concentration (LC50) was calculated to be 1043.87 µg mL−1.
According to the toxicity classification by Meyer et al., substances with LC50 values greater than 1000 µg mL−1 are considered to be nontoxic.45,47 In one study, P. emblica fruit extract showed no acute or chronic toxicity in rats administered with up to 5000 mg kg−1.48 These findings support that as antioxidants are abundantly present in P. emblica and kombucha, even chronic administration did not result in any toxicological changes, thus validating the high safety margin of the kombucha-like beverage powder. Moreover, P. emblica has been approved for its anti-cytotoxic, antioxidant, antimicrobial, anti-inflammatory and chemoprotective properties.9,32 The obtained regression equation is presented below in Table 5.
| Sample | Concentration (µg mL−1) | Log10 concentration | Mortality (%) | Probit of mortality | LC50 (µg mL−1) | Regression |
|---|---|---|---|---|---|---|
| Encapsulated kombucha powder (EKP) | 1 | 0.00 | 0 | 0 | 1043.87 | y = 1.87× − 0.65, R2 = 0.9148 |
| 10 | 1.00 | 0 | 0 | |||
| 100 | 2.00 | 6.67 | 3.52 | |||
| 250 | 2.40 | 13.33 | 3.87 | |||
| 500 | 2.70 | 33.33 | 4.56 | |||
| 1000 | 3.00 | 46.67 | 4.92 |
Although antibacterial assays were not included in the present study, it is well established that kombucha possesses selective antimicrobial properties, wherein organic acids and phenolics inhibit pathogenic bacteria such as E. coli, S. aureus, and Salmonella, while beneficial gut microorganisms typically exhibit resilience to acidic and polyphenolic conditions.9,37 These compounds inhibit pathogenic bacteria more strongly than beneficial gut flora, supporting kombucha's classification as both antimicrobial and microbiome-supportive.
Recent scholarly investigations have also underscored the significance of dormant, metabolically inactive persister cell subpopulations that endure environmental stressors, including acidic pH, oxidative stress, nutrient scarcity, and even bactericidal antibiotics.37 Given that kombucha inherently contains organic acids and stress-inducing phenolics, elucidating the interactions between unfermented and fermented P. emblica kombucha and both active pathogens and persister cell subpopulations represents an emerging area of research inquiry.9 Future studies will therefore focus on evaluating its selective antimicrobial potential and exploring its application as a functional carrier in nutraceutical systems.
In summary, the P. emblica kombucha powder exhibited an LC50 value indicative of its nontoxicity, suggesting its safety for potential nutraceutical applications. However, it is recommended that further in vitro and in vivo studies be conducted to comprehensively evaluate its safety profile.
4 Conclusions
In conclusion, the foam mat drying process effectively produced P. emblica kombucha powder with a high phenolic content of 998 ± 35.41 mg GAE per g, encapsulation efficiency of 86.10% and DPPH radical scavenging activity of 63.62% ± 0.78%. The powder showed stable physicochemical properties, complied with halal ethanol limits, and was confirmed to be nontoxic. These results highlight its potential as a safe and value-added ingredient for functional beverages. Its low ethanol level of 0.31% ± 0.02% complied with halal regulatory limits, while its nutritional composition, favourable pH, soluble solids, and bulk properties confirmed its stability and suitability for storage and product formulation. Moreover, the brine shrimp lethality assay confirmed its nontoxic nature, further supporting its safety for functional applications.Collectively, these findings underscore the potential of foam mat-dried P. emblica kombucha powder as a safe, stable, and value-added ingredient for functional beverages and nutraceutical development. Future studies should focus on its storage stability, sensory acceptance, and the bioavailability of phenolic compounds, and an anti-microbial study to strengthen its commercial prospects.
Author contributions
Nur Haniza Ewandi Jong: conceptualization, formal analysis, writing – original draft; Norhayati Muhammad: writing – review & editing, methodology, resource, supervision; Norazlin Abdullah: methodology, resource, supervision; and Dayang Norulfairuz Abang Zaidel: methodology, laboratory analysis, supervision.Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting the findings of this study are included within the article.Acknowledgements
This research is supported by the Ministry of Higher Education, Malaysia, under the Hadiah Latihan Persekutuan (HLP) program, and is partially funded by Universiti Tun Hussein Onn Malaysia.References
- R. Sharma, G. Burang and S. Kumar, J. Food Sci. Technol., 2022, 59, 2013–2024 CrossRef CAS.
- S. Palamthodi, D. Kadam and S. S. Lele, J. Food Sci. Technol., 2019, 56, 473–482 CrossRef CAS PubMed.
- J. F. de Miranda, L. F. Ruiz, C. B. Silva, T. M. Uekane, K. A. Silva, F. F. Fernandes and A. R. Lima, J. Food Sci., 2022, 87(2), 503–527 CrossRef CAS PubMed.
- H. Antolak, D. Piechota and A. Kucharska, Antioxidants, 2021, 10(10), 1541 CrossRef CAS.
- L. Ayed, S. M'Hir and M. Hamdi, J. Chem., 2020, 2020(1), 1–12 Search PubMed.
- J. Kim and K. Adhikari, Beverages, 2020, 6(1), 1–18 CrossRef.
- Y. Liu, S. Wu, Y. Tao, J. M. Regenstein and P. Zhou, Trends Food Sci. Technol., 2022, 129, 608–616 CrossRef CAS.
- K. E. Emiljanowicz and E. Malinowska-Pańczyk, Crit. Rev. Food Sci. Nutr., 2020, 60(19), 3185–3194 CrossRef CAS.
- F. F. Pria and M. S. Islam, J. Dis. Med. Plants, 2019, 5(1), 1–9 Search PubMed.
- M. A Hussain, M. Y. M. Yunos, M. A. O Mydin, N. Utaberta and N. A. Ismail, J. Teknol., 2015, 75(9), 39–45 Search PubMed.
- Practo.com, https://www.practo.com/healthfeed/5-super-healthbenefits-of-amla-indian-gooseberry-24447/post, accessed September 2024.
- G. Ruhela, P. Dhama, K. Shanker, X. Ding and A. Sharma, Food Chem. Adv., 2023, 3, 100330 CrossRef.
- M. S. Abdullah, A. F. Ab Razak, M. S. Sulaiman, M. Z. Abidin, M. S. Osman, A. A. Razak and M. H. Salleh, Int. J. Mech. Eng., 2022, 7(6), 158–166 Search PubMed.
- T. Sarkar, M. Mukherjee, S. Roy and R. Chakraborty, Heliyon, 2023, 9(4), e14788 CrossRef CAS PubMed.
- N. I Medihi, E. T. T. Tan, M. A. Zabidi and S. A. A. Mutallib, presented in part at Inaugural Symposium of Research and Innovation for Food (Sorif) 2021, Virtual, 2021 Search PubMed.
- N. H. Zakaria, R. A. Ramli and W. R. W. Ismail, presented in part at Conference on Hospitality and Tourism 2021, Virtual, 2021 Search PubMed.
- B. Khatri, N. Hamid, R. Shams, K. K. Dash, A. M. Shaikh and B. Kovács, Discover Food, 2024, 4, 166 CrossRef.
- H. N. M Yusoh, N. H. E Jong, N. M Isa, N. Sulaiman and W. H. M. Yusof, Int. J. Eng. Adv. Res., 2020, 1(2), 7–17 Search PubMed.
- M. Saifullah, R. McCullum, T. O Akanbi and Q. V. Vuong, Food Hydrocolloids Health, 2023, 4(1), 100157 CrossRef CAS.
- E. Leonarski, A. C. Guimarães, K. Cesca and P. Poletto, Food Biosci., 2022, 49, 101841 CrossRef CAS.
- Z. Xiao, J. Xia, Q. Zhao, Y. Niu and D. Zhao, Carbohydr. Polym., 2022, 298, 120113 CrossRef CAS PubMed.
- S. Saentaweesuk, Agric. Sci., 2014, 6, 33–40 Search PubMed.
- P. Kandasamy, N. Varadharaju and S. Kalemullah, Int. J. Food Eng., 2014, 10(1), 163–172 Search PubMed.
- T. Klawpiyapamornkun, T. Uttarotai, S. Wangkarn, P. Sirisa-ard, S. Kiatkarun, Y. Tragoolpua and S. Bovonsombut, Fermentation, 2023, 9(3), 291 CrossRef CAS.
- Y. Gao, J.-Q. Wang, Y.-Q. Fu, J. F. Yin, J. Shi and Y. Q. Shu, Food Chem., 2020, 316, 126370 CrossRef CAS.
- C. D. Barbosa, A. P. T. Uetanabaro and W. C. R. Santos, et al. , LWT Food Sci. Technol., 2021, 148, 111788 CrossRef CAS.
- Y. Plugatar, R. G. Timofeev, V. Korzin, A. N. Kazak and D. V. Nekhaychuk, Beverages, 2023, 9(2), 31 CrossRef CAS.
- G. W. Latimer, Official Methods of Analysis: 22nd Edition, AOAC Publications, New York, 2023 Search PubMed.
- E. P. Masitlha, E. Seifu and D. Teketay, Food Prod. Process. Nutr., 2024, 6(1), 3 CrossRef CAS.
- M. Asaduzzaman, S. Rana, S. M. R. Hasan, M. Hossain and N. Das, Int. J. Pharma Sci. Res., 2015, 6(8), 1179–1185 CAS.
- N. Phuyal, P. K. Jha, P. P. Raturi and S. Rajbhandary, Sci. World J., 2020, 2020, 1–7 CrossRef.
- K. Muzaffar, S. A. S. Rafiq, J. A. Rather, F. Allai, H. A. Makroo, D. Majid and B. N. Dar, Int. J. Food Sci. Technol., 2023, 58(10), 5357–5365 CrossRef CAS.
- S. Fatima and V. Kumar, Sustainable Food Technol., 2025, 3(5), 1542–1555 RSC; R. P. Sari, Z. Ikawati, R. Danarti and T. Hertiani, Arabian J. Chem., 2023, 16(9), 1–8 CrossRef CAS.
- R. Jayabalan, P. N. Chen, Y. S. Hsieh and K. Prabhakaran, Indian J. Biotechnol., 2011, 10(1), 75–82 CAS.
- S. A. Villarreal-Soto, S. Beaufort, J. Bouajila, J. Souchard and P. Taillandier, J. Food Sci., 2018, 83, 580–588 CrossRef CAS PubMed.
- S. Hameed, Y. Wang, L. Zhao, L. Xie and Y. Ying, Mater. Sci. Eng., C, 2020, 108, 110338 CrossRef CAS.
- S. Hameed, J. Nan, X. Zeng and H. Xiong, Colloids Surf., B, 2026, 257, 115173 CrossRef CAS.
- F. W. Farouk, Int. J. Res. Innovations Appl. Sci., 2023, VIII(XI), 72–78 CrossRef.
- A. Nemati, A. Motamedzadegan and J. Mohammadzadeh Milani, J. Food Process. Preserv., 2022, 46(6), e16539 CAS.
- T. Alsaleem, T. Alsaleem, R. Al-Dhelaan and G. Allaheeb, Int. J. Halal Res., 2022, 4(1), 19–28 Search PubMed.
- Y. Yikilkan, A. A. Redha, B. Kaba, H. Pasazadeh and I. Koca, Sustainable Food Technol., 2025, 3, 1329–1340 RSC.
- B. Y. Shekunov, P. Chattopadhyay, H. H. Y. Tong and A. H. L. Chow, Pharm. Res., 2007, 24(2), 203–227 CrossRef CAS.
- P. Mishra, G. K. Rai and C. L. Mahanta, J. Food Process. Preserv., 2015, 39(6), 1216–1228 CrossRef CAS.
- K. Kaderides and A. M. Goula, Food Res. Int., 2017, 100, 612–622 CrossRef CAS.
- B. N. Meyer, N. R. Ferrigni, J. E. Putnam and L. B. Jacobsen, Planta Med., 1982, 45(5), 31–34 CrossRef CAS.
- T. E. Murphy, K. Walia and J. M. Farber, Food Protect. Trends, 2018, 38(5), 329–337 Search PubMed.
- N. F. A. Rahim, N. Muhammad, N. Abdullah, B. A. Talip and K. H. Poh, Food Res., 2020, 4(6), 2042–2048 Search PubMed.
- K. Jaijoy, N. Soonthornchareonnon, N. Lertprasertsuke and A. Panthong, Int. J. Appl. Res. Nat. Prod., 2010, 3(1), 48–58 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |