 Open Access Article
Open Access ArticleA review on chlorophyll-based active and intelligent packaging: chemistry, stability and applications in food freshness monitoring
Harshita Ranjana,
Manisha Joshia and
Swarup Roy *bc
*bc
aSchool of Bioengineering and Food Technology, Shoolini University, Bajhol, Solan 173229, India
bDepartment of Food Technology and Nutrition, School of Agriculture, Lovely Professional University, Phagwara, Punjab 144411, India. E-mail: swaruproy2013@gmail.com
cDepartment of Food and Nutrition, Kyung Hee University, Seoul 02447, Republic of Korea
First published on 16th March 2026
Abstract
Chlorophylls, which are natural bioactive pigments, have recently gained significant attention as multifunctional components in sustainable food packaging due to their antioxidant, antimicrobial, and pH-sensitive properties. The incorporation of chlorophylls into biodegradable films, coatings and encapsulated systems provides dual functionality, acting as active agents that extend shelf life by reducing oxidative damage and microbial spoilage and serving as intelligent indicators that signal freshness through colorimetric or fluorescence changes. Despite these advantages, chlorophylls are inherently unstable and degrade rapidly under light, heat, and oxygen. Additional challenges, including large-scale processing limitations and the high cost of advanced extraction and stabilization techniques, restrict their practical implementation. This review highlights the chemical properties, conventional and non-conventional extraction methods, stabilization strategies, and encapsulation approaches of chlorophylls, along with their integration into biopolymeric matrices within scalable and regulatory frameworks. Packaging based on chlorophylls represents a sustainable approach with strong potential to reduce microbial spoilage, enable real-time quality monitoring, and contribute to food-waste reduction.
Sustainability spotlightChlorophylls are the most abundant natural pigment that can be extracted and utilized in making functional packaging. This review investigates recent advances in packaging that incorporate chlorophylls and discusses their use in improving and monitoring the shelf life of packed food. The area of this study is directly aligned with Sustainability Development Goal 3, good health and well-being. The presence of chlorophylls in packaging could be beneficial for developing smart packaging materials. |
1 Introduction
Packaging is a crucial aspect in the food industry for the protection of food from external conditions,1 such as temperature, humidity, and light, while maintaining quality, integrity, freshness, and safety during its shelf life. This is achieved by controlling gas and vapor exchange with the atmosphere, preventing degradation while preserving sensory and nutritional qualities. Beyond protection, packaging significantly influences consumer perception, as the appearance of the product is often associated with its freshness and overall quality, and ultimately affects consumption patterns. Traditionally, food packaging has used materials such as paper and glass. Paper is lightweight, recyclable, and cost-effective, while glass is inert and provides excellent barriers to oxygen and moisture, making it suitable for acidic or fatty products. Furthermore, plastics are versatile and exhibit excellent barrier properties that are ideal for a wide range of foods.2 In recent years, there has been a growing emphasis on environmentally friendly and sustainable packaging alternatives. This shift has driven research and innovations in biomaterials for food packaging, leading to the rise in the application of advanced techniques such as coatings and antimicrobial and antioxidant packaging, in addition to modified atmosphere packaging.3,4Active and intelligent packaging has garnered attention for its ability to extend functionality beyond passive containment. Active packaging interacts with the food component to extend the shelf life, mitigating microbial spoilage,5 while intelligent packaging reflects environmental factors6 through visible color changes or variations in fluorescence to provide non-invasive information on food freshness, spoilage,7 or changes in the package microenvironment.8 Addressing issues such as temperature fluctuations, microbial contamination, and package integrity, these packaging systems contribute to reduced food waste, improved traceability, and decreased risk of food-borne illness.9–11
Chlorophylls are naturally derived pigments with antioxidant properties and nutritional value. They eliminate free radicals and protect cells from oxidative damage. Several studies have reported the promising antioxidant, anti-inflammatory and anti-cancer properties of chlorophylls and their derivatives, but pigment loss through oxidation of chlorophylls' phenolic moieties presents a challenge. Although chlorophylls are sensitive to light and oxidation, advanced techniques, such as encapsulation with different carriers, including carboxymethylcellulose, can increase their stability and performance.12–14
There have been recent advancements in biodegradable films and smart packaging, showing the potential of natural extracts and nanoparticles as functional additives. Numerous studies have examined the efficiency of packaging based on chlorophylls, highlighting the pigment's amphiphilic structure, pH sensitivity and antioxidant activity, which have potential advantages in both active and intelligent applications. For example, sodium iron chlorophyllin incorporated into chitosan/gelatin films enhanced antimicrobial and antioxidant activity, improved UV barrier properties, and effectively delayed spoilage in fresh-cut chilli peppers, demonstrating its potential as an active packaging agent.15 Similarly, chlorophyllin incorporated into photoactive coatings generated reactive oxygen species (ROS), enabling effective inactivation of L. monocytogenes, demonstrating both antimicrobial activity and light-responsive packaging behavior.16 Further, another study reported that cornstarch chlorophyllin composite films generated ROS under light exposure, resulting in lower microbial and nitrogen levels compared to control samples, while maintaining acceptable quality of shrimp for up to 4 days.17 Moreover, chitosan-based films incorporating chlorophylls with curcumin exhibited enhanced barrier and mechanical properties and significant antimicrobial activity, and extended the shelf life of cherries and pork.18 The integration of chlorophyll pigments has significantly expanded the functionality of food packaging.19 These natural components are especially found in plant extracts, such as green tea and basil, and are explored for smart packaging due to their unique chemical and functional properties.
The aim of this review is to comprehensively analyze recent advancements in chlorophyll-based smart packaging for maintaining and monitoring food quality and freshness, focusing on key properties of chlorophylls and their relevance in active and intelligent packaging. It also discusses the development and characterization of chlorophyll-incorporated films and coatings, while analyzing their applications in both active and intelligent packaging. Furthermore, the review highlights current knowledge gaps and areas that require further development for commercial optimization and broader adoption.
2 Chlorophyll chemistry and properties
Chlorophylls have unique chemical structures and properties that are fundamentally responsible for their characteristic color and potent antioxidant activity, and make them suitable for packaging technologies aimed at maintaining and monitoring food freshness. Fig. 1 shows the molecular structure of chlorophyll, highlighting the porphyrin ring with the central magnesium ion and the phytol tail.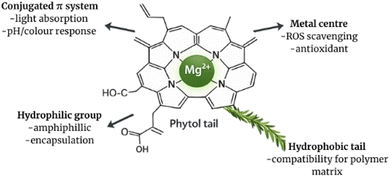 | ||
| Fig. 1 Molecular structure of chlorophyll.20,21 | ||
2.1 Molecular structure of chlorophylls
Chlorophylls are complex organic molecules made of a hydrophilic porphyrin ring, a central magnesium ion, and a strongly hydrophobic phytol side chain. The porphyrin ring, a macrocyclic conjugated tetrapyrrole structure that contains four nitrogen atoms in the pyrrole groups, is responsible for absorbing light energy, while the magnesium ion acts as an electron acceptor. Various conjugated double bonds enhance the ability of chlorophylls to absorb visible light.14,22,23There are several forms of chlorophyll, including chlorophyll a, b, c, d, and e, with chlorophyll a being the most common in plants. Chlorophylls efficiently absorb light in the red and blue regions of the spectrum, exhibiting peak absorption around 430 and 662 nanometers, respectively. Chlorophyll b, which mainly functions to protect chlorophyll a from excessive light, absorbs light optimally near 453 nanometers. Other chlorophyll forms, such as chlorophyll c, d, and e, are found in diverse organisms, such as algae, and they differ in their absorption spectra and biological roles. For instance, chlorophyll c absorbs primarily in the blue-green region, chlorophyll d in the red region, and chlorophyll e in the far-red region of the spectrum.14 Structurally, chlorophyll molecules vary chiefly in the degree of saturation in their pyrrolic rings and in functional groups attached to the macrocycle. For example, chlorophyll b differs from chlorophyll a by having an aldehyde group instead of a methyl group at the C7 position, which results from enzymatic oxidation of the methyl group catalyzed by oxygenase. Chlorophyll c contains a fully unsaturated phytoporphyrin system (with a double bond between C17 and C18), whereas chlorophylls a, d, and f contain partially saturated phytochlorins. These structural differences significantly affect the absorption properties: chlorophylls a, d, and f show approximately balanced absorption intensities in blue, red, and green regions, while chlorophyll c absorbs weakly in the red and more strongly around 450 nm. This variety in the absorption spectra is mirrored in their distinctive green hues: chlorophyll a appears bluish-green, chlorophyll b bright green, chlorophyll c yellowish-green, chlorophyll d bright forest green, and chlorophyll f emerald green. This diversity makes these pigments promising candidates as natural colorants.14 Chlorophyll molecules possess two distinct regions: a hydrophilic macrocycle and a hydrophobic phytol tail. The most hydrophilic segments of the macrocycle include the cyclopentanone ring and a propionic acid ester group at the C17 position. This amphiphilic nature influences solvent selection for chlorophyll extraction, impacting yield and purity, which is important for technological applications.23
Chlorophylls' distinctive structures positively influence their function in packaging. For example, the hydrophilic porphyrin macrocycle promotes antioxidant activity, allowing the chlorophyll and its derivatives to act as a singlet oxygen quencher and free radical scavenger. This protective effect against lipid oxidation is attributed not only to their free radical scavenging capacity but also to their ability to chelate pro-oxidant metal ions, such as Fe2+ and Cu2+, which retard lipid peroxidation in food.5 On the other hand, chlorophylls act as photosensitizers, absorbing visible light and transferring the energy to molecular oxygen to produce singlet oxygen and other reactive oxygen species.24
2.2 Extraction methods
Chlorophylls are widely extracted for industrial applications from stinging nettle, spinach, algae, silkworm excreta, alfalfa, pine needles, pasture grasses, and plant-harvest by-products.25 Several methods have been used to extract chlorophylls from plants, which include aqueous extraction, enzyme-assisted extraction, alcoholic/organic solvent extraction, and supercritical/subcritical extraction,26 as summarized in Table 1. Extraction can be carried out in various ways, involving different solvents and preparation methods. Variables such as pressure, temperature, contact efficiency, and time are critical. Since chlorophylls are sensitive to extreme light exposure, pH, and temperature, solvent selection must consider factors such as density, viscosity, heat of evaporation, and cost, in addition to environmental and health effects. The solvent should not react with or damage the extracted compound and must not be corrosive. Different methods are tailored for different microalgae species.27| Extraction method | Raw material/food | Yield | Purity | Conditions | References |
|---|---|---|---|---|---|
| Solid-liquid extraction | Spinach (solid phase) | NA | Chlorophyll a/b ratio 4.8 in ethyl acetate phase | 25 °C, 30 min, 600 rpm agitation; mild energy (centrifugation + vacuum drying); solvent recovery-condenser | 28 |
Extraction by methanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50) atmospheric 50) atmospheric |
Cucumber | Chlorophyll a: 480.14 mg/100 g, chlorophyll b: 342 mg/100 g | Chlorophyll a/b ratio ≈ 1.4 (480/342); CV: 52% chlorophyll a, 48% chlorophyll b | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 ratio (1 g 5 ratio (1 g![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 mL methanol); centrifugation 3000 rpm, 10 min; room temp 50 mL methanol); centrifugation 3000 rpm, 10 min; room temp |
29 |
| Enzyme-assisted extraction (Pectinex ultra SP-L) | Spinach pulp | 50.747 mg TCC/100 g spinach pulp (optimized); 39% higher Zn-chlorophylls derivative yield vs. non-enzymatic | NA | 8% enzyme (Pectinex ultra SP-L), 45 °C, 30 min; followed by ethanol extraction (2.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio, 60 °C, 45 min) 1 ratio, 60 °C, 45 min) |
5 |
| Subcritical fluid extraction (R134a + ethanol cosolvent) | Laminaria japonica Aresch (seaweed) | Chlorophyll a: 2.326 g kg−1 (optimized) | NA | Optimized: 324.13 K (51.13 °C), 17 MPa, 4.73% ethanol cosolvent; RSM Box–Behnken design | 30 |
| Subcritical CO2 extraction | Nigella sativa seeds | Volatile oil: 66.6 wt% of oleoresin | NA | 70 bar, 30 °C, 2 hours | 31 |
| Supercritical CO2 extraction with a static modifier | Scenedesmus obliquus (microalga) | Chlorophyll a: 0.848 mg g−1 biomass; chlorophyll b: 0.356 mg g−1 biomass; chlorophyll c: 0.018 mg g−1 biomass | NA | 200–250 bar; 40–60 °C; CO2 flow rate varied; 7.7% v/v ethanol cosolvent | 32 |
| Ultrasound-assisted green solvent extraction | Scenedesmus obliquus, Arthrospira platensis (microalgae) | Optimized ultrasound + ethanol/ionic liquid solutions; carotenoid/chlorophyll extracts potent peroxyl scavengers (5.94–26.08 × α-tocopherol) | NA | Ultrasound extraction; ethanol + ethanolic ionic liquid solutions; optimized extraction time, repetitions | 33 |
Over the years, several approaches have been reported to remove chlorophylls from botanical crude extracts (CEs). Most rely on solid-phase extraction (SPE) with stationary phases, such as Diaion HP-20, for initial fractionation. Charcoal has also been used to remove chlorophylls or other pigments entirely from CEs without fractionation of other constituents. However, these methods often eliminate other unspecified, potentially bioactive compounds along with chlorophylls. Detailed protocols vary by author, and preservation of the original phytochemical profiles is generally not assessed.34
Liquid–liquid or countercurrent separation (CCS) methods are effective for isolating large quantities of target compounds from complex matrices. CCS offers ease of use, affordable cost, reduced solvent consumption, and efficient equipment performance, making it valuable in natural product research. CCS has been applied to isolate plant pigments such as anthocyanins and carotenoids from chlorophylls-enriched extracts obtained from grass, spinach, and other plant materials. However, CCS's suitability for the removal of chlorophylls remains under evaluation. In CCS, both the stationary and mobile phases are liquids, forming a biphasic solvent mixture. This mixture solubilizes extracts entirely while balancing the partitioning of target compounds. Phytochemicals in both phases can be recovered with solvent evaporation, making CCS effectively loss-free when analytes are non-volatile. A recent study implemented CCS to selectively remove chlorophylls from botanical CEs to produce chlorophylls-free “degreened” Knock-Out Extracts (chlorophylls-KOEs). This method enables the subtraction of assay interference compounds while optimally recovering other phytochemicals. The study produced chlorophylls-KOEs from three botanicals: Epimedium sagittatum, Senna alexandrina L., and Trifolium pratense L., with the reproducibility and selectivity evaluated using HPLC, UHPLC-UV/MS, LC-MS, and 1H NMR spectroscopy.34
Supercritical/subcritical CO2 extraction selectively isolates components and is suitable for heat-sensitive products. Traditionally, the extraction of chlorophylls involves liquid solvents followed by evaporation and drying at high temperatures, risking degradation. Supercritical/subcritical CO2 extraction operates at low temperatures, preventing thermal degradation. CO2 is inert, non-toxic, non-flammable, inexpensive, and easily separated from extracts without heating, producing solvent-free extracts. Stability tests on chlorophylls from katuk leaves showed decreased content at high temperatures, indicating supercritical/subcritical CO2 as the preferred extraction method to preserve chlorophylls.26 Enzyme-assisted extraction is gaining attention for eco-friendly processing. Targeted enzymes such as pectinases, cellulases, and hemicellulases degrade cell walls, increasing solvent pre-treatment efficiency, reducing solvent use, and increasing bioactive compound yield. Used widely in juice processing and beer clarification, enzymatic pretreatment enhances extractability. Enzyme-assisted extraction of Zn-chlorophyll derivatives from spinach pulp under optimized conditions (8% enzyme concentration, 45 °C, 30 min) increased the yield by 39%.5
2.3 Key properties of chlorophylls relevant to packaging
The chemical structure of chlorophylls determines their bioactivity and influences their potential health benefits.14 As mentioned earlier, chlorophylls are among the most prominent bioactive compounds, having positive health impacts and high antioxidant activity. They are used as natural food coloring agents and have wound-healing and anti-mutagenic properties.35Emerging stabilization technologies, such as core–shell nanoparticle encapsulation using zein, casein, or whey protein isolate as wall materials, offer enhanced protection for chlorophylls in food packaging applications. Encapsulation efficiencies of chlorophylls' retention after 10 days were 83.6–96.3% and 39–97.8% at pH 3.0 under light and acidic stress, respectively, compared with 40% for free chlorophylls. These technologies, therefore, offer superior photostability and controlled delivery for active packaging applications.42
3 Chlorophyll-based active and intelligent packaging development and key properties
Active and intelligent smart packaging based on chlorophylls is a promising method that leverages the natural bioactivity and colorimetric responsiveness of chlorophyll extracts from various sources, including plants and algae. Active compounds as intelligent indicators enhance the stability and function of packaging or coating matrices. These pigments can change color because of environmental stimuli, such as temperature, pH, oxygen, light or microbial activity, providing visual cues to consumers about the freshness, spoilage, ripeness, or contamination of food.43 The packaging is typically developed by incorporating chlorophylls or microencapsulated chlorophylls into biodegradable polymer matrices such as alginate, chitosan, gelatin, or pectin. Common fabrication methods include solution casting and extrusion,44 which are designed to preserve the functional and colorimetric properties of chlorophylls during processing.3.1 Film/coating fabrication using chlorophylls
As mentioned in the introduction, chlorophylls have recently emerged as promising bioactive compounds for the development of active and intelligent packaging films and coatings because of their unique chemical, antioxidant, and colorimetric properties. For example, edible coatings are revolutionizing food preservation by offering a sustainable and effective solution to key industry challenges. They are produced from natural biopolymers, such as proteins (e.g., gelatin, zein), polysaccharides (e.g., starch, chitosan, alginate), and lipids, and the coatings form a thin, edible layer on food surfaces. These biodegradable and edible matrices reduce moisture loss, protect against oxidative damage, and limit microbial growth, thereby extending the shelf life while preserving food quality. Enhanced with natural additives such as essential oils and antioxidants, these coatings offer antimicrobial benefits and contribute to health.45 Fig. 2 shows the overall process for chlorophylls' extraction, incorporation into the matrix, and subsequent analysis, leading to the development of chlorophyll-integrated film and food packaging applications. Lv et al. (2023)19 developed a film containing chlorophylls and chitosan having the potential to generate color within the colorimetric temperature range of 50–75 °C. The system underwent an irreversible color change from green to yellow when exposed to this temperature range.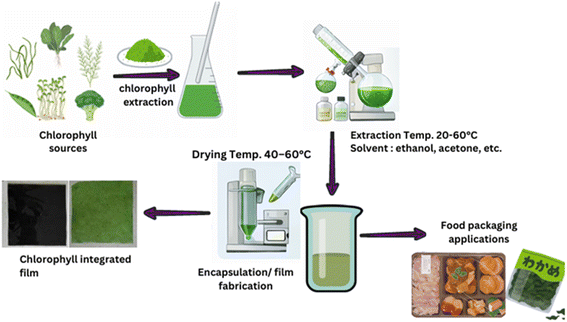 | ||
| Fig. 2 Schematic of chlorophyll-based active film synthesis.46,47 | ||
In comparison to the widely used solvent casting method, extrusion of corn starch and poly (butylene adipate-co-terephthalate) (PBAT) blends with intact Chlorella pyrenoidosa biomass utilizes the amphiphilic nature of chlorophylls and algal lipids to improve compatibility for hydrophilic starch and hydrophobic PBAT phases. The results indicate that films containing a higher content (5.0%) of intact Chlorella pyrenoidosa biomass exhibit superior tensile strength (4.37 ± 0.24 MPa) and elongation (88.43% ± 6.8%) compared to films with disrupted biomass. This suggests better dispersion and interactions between the phases, leading to enhanced homogeneity because of the increased amphiphilic compatibility, as further confirmed by SEM analysis. Furthermore, these films display lower water vapor permeability (5.19 × 10−11 g m−1 s−1 Pa−1), enhancing their barrier properties. Films produced by blown extrusion from starch, PBAT, and Chlorella pyrenoidosa microalgae biomass have the technological potential to be used as packaging for food products. Starch and PBAT blends are widely studied.52 Overall, films produced by blown extrusion, blending starch or biodegradable polymers incorporating microalgae biomass containing chlorophylls, have shown good mechanical and antioxidant properties.
Another study examined the encapsulation of chlorophylls with MD and WPI as carriers applying both spray-drying and freeze-drying methods; the study was conducted to increase the stability of chlorophylls extracted from Ulva intestinalis algae. The optimum combination of wall and core materials to achieve the highest response, including encapsulation efficiency (EE) and chlorophyll content (CC), was obtained by using a response surface methodology and central composite design. The optimal chlorophyll microcapsule obtained was chosen for subsequent tests that examined the solubility, moisture content, and antioxidant properties. The results showed that the highest EE and CC were 90.27% ± 0.21%, 55.36 ± 0.36 µg mL−1, and 90.46% ± 0.62%, 85.85 ± 0.43 µg mL−1, respectively, for the SD and FD methods. The microcapsules produced by freeze drying (FD) had higher antioxidant activity (79.1 ± 0.24)% than the microcapsules produced by spray drying (SD) (67.5 ± 0.16)%. The highest solubility (95.32%) and the lowest moisture content (3.7 ± 0.05)% were found for SD. The freeze-drying method (FDM) had the highest EE (91.2%), CC (89.67 µg mL−1), and antioxidant properties (79.1%).55
In modern packaging, embedded sensors not only interact with the food product but also assess its surrounding conditions, providing stakeholders with real-time information and insights, detecting freshness, pathogens, pH levels and other environmental changes, offering more insight than traditional measures such as weight or appearance. Compared to other natural food colorants, the colors of chlorophylls are relatively stable, making them less sensitive and observable colorimetric indicators for intelligent biodegradable packaging. This suggests that they may be less applicable for providing real-time information.56 A study by Chavoshizadeh et al. (2020)12 introduced a wheat gluten-based biodegradable film incorporating chlorophylls, highlighting its role in enhancing the shelf life of sesame oil and indicating expiration dates. The film reduced oil oxidation, as evident from the halved peroxide value, and changed color from green to yellow in response to oil quality after a prolonged storage period. Integrating chlorophylls into edible films or coatings has a significant effect, enhancing their physical and functional properties. For example, chlorophyll microcapsules incorporated into an alginate-based edible film showed enhanced film thickness and improved surface texture, which ultimately resulted in a smoother, more homogeneous film without agglomeration54 (discussed in Table 2). This resulted in enhanced surface morphology and improved barrier properties of the film while protecting the food product. Moreover, scanning electron microscopy (SEM) evaluation of the microstructure showed that the chlorophyll-enriched films exhibited a clearly different and smoother surface compared to control films without chlorophylls. Also, Fourier transform infrared spectroscopy (FTIR) analyses showed interactions between the chlorophyll microcapsules and the film matrix. The functional films demonstrated the enhanced antimicrobial activity by slowly releasing chlorophylls compounds that inhibited the growth of molds such as Rhizopus sp.; and bacteria such as Escherichia coli on food products, thereby extending shelf life.54
| Biomaterial | Food product | Functional properties | Packaging | Physical properties | Limitations | References |
|---|---|---|---|---|---|---|
| Cornstarch + chlorophylls in + lipids (coconut oil, oregano essential oil, beeswax) | Shrimp | Photoactive antibacterial; ROS generation; shelf-life extension | Active | Increased tensile strength; improved water vapour permeability; rough surface | Starch's susceptibility to moisture; lipid composition affects performance | 57 |
| Pectin + chlorophylls (leaf extract) + CMC/silica nanoparticles | — | Antioxidant; antimicrobial (against E. coli and S. aureus); Thermally stable | Active | Increased tensile strength, increased flexibility, thickness, improved | Decreased film flexibility; enhanced opacity by the addition of nanoparticles | 13 |
| Chlorophylls microcapsules (gum Arabic/maltodextrin) | — | Antioxidant activity; UV protection | Active | Improved thermal stability, high encapsulation efficiency (77.19%) | Spray-drying-induced chlorophylls degradation; high surface oil with gum Arabic blends | 58 |
| Wheat gluten + chlorophylls extract-based film | Sesame oil | UV blocking; color change (indicator) | Active/Intelligent | Increased thickness; increased water vapor permeability (WVP) | Weak mechanical properties; uneven nanocomposite dispersion affecting functional uniformity; higher water vapor permeability and moisture sensitivity | 12 |
| Polyester-based film (INZEA) + chlorophylls hybrid nanopigments | — | Strong antioxidant potential; pigment stabilization; UV resistance | Active | Improved Young's modulus, and increased thermal stability | Limited biodegradability; reduced transparency | 59 |
| Chlorophylls orella-k-carrageenan composite films | — | Rapid biodegradation; UV barrier | Active | High opacity | High opacity affects in consumer visibility | 60 |
| WPI + chlorophylls microcapsules | — | High antioxidant potential; pH-responsive indicator | Active | Low moisture content, enhanced water solubility | — | 61 |
| Chitosan + chlorophylls a + 2-HP-β-cyclodextrin | — | High antioxidant potential; generates reactive oxygen species (ROS) under light; antimicrobial activity | Active | Amorphous structure, modified surface roughness, good light absorbance | Absence of controlled release of bioactive compounds analysis; light-dependent ROS | 62 |
| PVA + ZnO + AgI + chlorophylls (from spinach) | — | Photocatalytic pollutants degradation; strong activity | Active | — | — | 63 |
3.2 Key properties of chlorophyll-based smart packaging
Chlorophylls' pH-driven color change occurs because of the loss of Mg2+ from the porphyrin ring, whereupon the bright green color of the chlorophylls becomes olive brown64 and the absorption bands shift from ∼430/662 nm (green) to ∼410/650 nm (yellow/brown).65 Koca et al. (2006)40 observed that chlorophyll a degraded faster than chlorophyll b across pH 5.5–7.5 at higher temperatures, and, due to reduced pheophytinization, the green color loss rate constants decreased at higher pH.
4 Applications of chlorophyll-based packaging films and coatings
Chlorophylls, a naturally occurring green pigment essential for the photosynthetic process in plants, have garnered attention in sustainable packaging research due to their biocompatibility, eco-friendly characteristics, and functional versatility.35 They exist in several forms, primarily classified into six types, namely: chlorophyll a, b, c, d, e, and f. Among these, chlorophyll a and b are predominant in higher terrestrial plants, with chlorophyll a serving as the principal photosynthetic pigment found across plants, algae, cyanobacteria, and other phototrophic organisms, while chlorophyll b functions as an accessory pigment predominantly located in green algae and higher plants.67 The presence of extensive conjugated double bonds in these molecules contributes to their significant antioxidant capacity.374.1 Active packaging
Chlorophylls are widely distributed across various plant-based sources, particularly in green vegetables and fruits. Its distinctive physicochemical and biological properties, including strong light absorption, pH sensitivity, antioxidant activity, and chromatic responsiveness, render it a valuable component in the formulation of both active and intelligent biodegradable packaging systems.68 Furthermore, its molecular structure, characterized by a porphyrin ring coordinated with a central magnesium ion (notably in chlorophyll a and b), is inherently organic and biodegradable.69 Natural pigments or dyes are suitable for sustainable intelligent food packaging application. This structural composition allows for environmental degradation through natural enzymatic and microbial pathways, enhancing its appeal for environmentally sustainable applications. Fig. 3 illustrates a multilayer, smart packaging system that consists of a barrier and an active layer. The active layer has dual functions: as an active releasing system that delivers antimicrobial agents and antioxidants to the food, and as an active scavenging system regulating and responding to key environmental factors, such as oxygen, carbon dioxide, moisture, ethylene, and odor. This combination can help to maintain food quality while extending shelf life by actively interacting with the packaged food environment.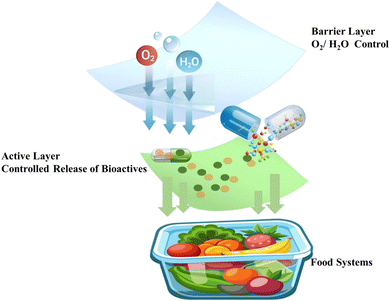 | ||
| Fig. 3 Schematic of a multilayer, smart packaging system consisting of a barrier and an active layer. | ||
The incorporation of chlorophylls into biopolymer-based packaging films has been shown to markedly influence their physical (e.g., thickness, opacity, and ultraviolet light-blocking capability), mechanical (e.g., tensile strength and elongation at break), and barrier properties (including resistance to water vapor and gas transmission). Insights into the edible and biodegradable ulvan-based films and coatings for food packaging was discussed in a previous report and the addition of chlorophyll improved the antioxidant activity of the film.70 These alterations are strongly dependent on the nature of the polymer matrix as well as the method of film fabrication. Numerous studies have explored such formulations to enhance the functional attributes of environmentally sustainable packaging materials. For instance, Dewi et al. (2022)54 developed an alginate-based film embedded with chlorophyll microcapsules derived from Caulerpa racemosa, and reported improved barrier performance, particularly in reducing moisture and gas permeability, which consequently extended the shelf life of packaged fish products. Similarly, Ukwatta et al. (2025)57 investigated chlorophyllin-doped corn-starch films combined with different lipid additives and observed that the addition of chlorophylls enhanced the water vapor barrier properties. This enhancement was attributed to increased matrix density, making the films more effective for packaging applications involving light- and moisture-sensitive food items.
Chlorophyll-based packaging materials exhibit high environmental degradability, as their constituent compounds, including chlorophyll pigments and biopolymeric matrices, such as starch and chitosan, are susceptible to enzymatic breakdown by naturally occurring microbial populations, particularly bacteria and fungi.71 The degradation processes typically involve the breakdown of the porphyrin ring structure in chlorophylls, resulting in simpler intermediates such as pheophytin and chlorins, which are eventually mineralized into carbon dioxide, water, and biomass.72 These degradation products are non-toxic to ecosystems and may even serve as beneficial nutrients within the soil. Hence, the unique physicochemical and biological attributes of chlorophylls can be effectively leveraged for the development of active and intelligent packaging systems. Such packaging systems offer a sustainable and innovative approach to enhancing food preservation, monitoring product quality, and reducing environmental impact within the packaging sector.73
Recent developments have underscored the effectiveness of chlorophyll-derived compounds as antimicrobial agents within active packaging systems, emphasizing their potential for extending shelf life and improving food safety. These packaging films are specifically designed to suppress or delay the growth of spoilage and pathogenic microorganisms that compromise food quality and safety.74 In a study conducted by Dewi et al. (2022),54 an alginate-based edible film was formulated using chlorophylls extracted from Caulerpa racemosa, a species of green seaweed. The antimicrobial efficacy of the film was evaluated by wrapping fish snacks, and the results indicated a notable reduction in microbial proliferation, particularly targeting spoilage organisms during storage. The observed antimicrobial effect was attributed to the chlorophyll component, which exhibited both antioxidant and antimicrobial functionalities, thereby positioning it as a promising natural preservative for use in biodegradable packaging systems. The inclusion of antioxidants such as chlorophylls in packaging materials plays a critical role in inhibiting oxidative degradation in food products, especially those rich in lipids and proteins.24 These compounds function through various mechanisms, including neutralization of singlet oxygen, scavenging of free radicals, reduction of hydrogen peroxide, and chelation of pro-oxidant metal ions.37 Such multifunctional activities contribute to the stabilization of food quality during storage and further enhance the utility of chlorophyll-based systems in active packaging applications. Plant-derived botanical extracts have been widely utilized as natural antioxidants in the development of active packaging materials.75 In a study by Micó-Vicent et al. (2020),59 chlorophyll-containing hybrid nano pigments sourced from broccoli processing residues were integrated into polyester-based bio-nanocomposite films. This incorporation led to a marked improvement in the antioxidant functionality of the resulting packaging films. Additionally, the films exhibited improved thermal stability, color uniformity, and mechanical performance, thereby indicating their suitability for active food packaging applications. This study underscores the dual advantage of valorizing agricultural waste while integrating naturally occurring antioxidant compounds into packaging matrices to prolong food shelf life and mitigate environmental burden. Chlorophyll-based materials thus offer a promising and sustainable alternative for active packaging technologies. Beyond packaging, chlorophyll pigments are currently utilized across multiple sectors, including cosmetics, pharmaceuticals, and the food industry.76 Within the food sector, chlorophylls (designated as E-140) are primarily employed as natural colorants.56 However, their functionality may be further extended to serve as natural pH-responsive indicators for monitoring food freshness, presenting additional value in the development of intelligent packaging systems.
4.2 Intelligent packaging
Intelligent packaging in the food sector integrates inherent biofunctional properties with real-time monitoring capabilities to enhance food quality and safety management.77 Chlorophylls, owing to their chemical and optical sensitivity, serve a dual role in such systems, functioning not only as a packaging component but also as a responsive indicator capable of providing visual or quantifiable feedback on product freshness, quality, and safety.78 One of the most widely employed mechanisms in this context is colorimetric sensing, where chlorophyll-based compounds exhibit perceptible color changes in response to environmental factors, such as pH fluctuations, temperature shifts, or the presence of spoilage-associated gases like ammonia or oxygen.79The pH-induced chemical transformation of the chlorophylls' macrocycle determines the colorimetric properties of the chlorophyll-based intelligent indicators. Chlorophylls consist of a porphyrin (tetrapyrrole) ring complexed with a central Mg2+ ion, giving these pigments their green coloration and visible-region absorption.80 Moreover, in acidic environments, the protonation of the porphyrin nitrogen atoms results in the removal of Mg2+ and formation of pheophytin and a visual color change from green to olive or yellow-brown.81 Under alkaline conditions, deprotonation occurs, and changes in the porphyrin structure result in a color shift, and the electron delocalization further modifies the optical behavior.82 These structural modifications impact absorption directly, i.e., native chlorophylls exhibit strong UV-Vis absorptions near 430 nm (Soret band) and 660–665 nm (Q band).83 Moreover, the peak broadening, lower intensity, and shift in wavelengths caused by the pheophytin produced in acidic environments strengthen the physicochemical basis concerning the use of chlorophylls as a halochromic indicator in smart packaging devices.
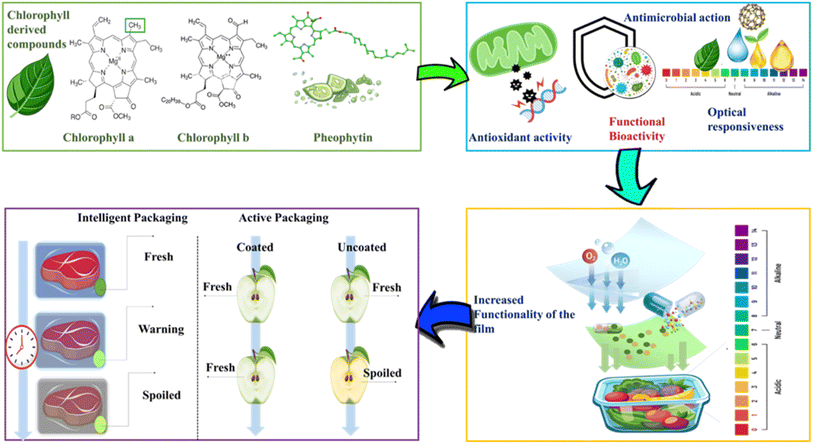
These alterations enable visual detection of food spoilage without needing to open the package, offering a user-friendly freshness assessment tool for both manufacturers and consumers. For example, Yu et al. (2024)84 developed a chlorophylls-infused colorimetric film that demonstrated distinct color transitions under varying pH conditions. These pH-responsive films were effectively applied to monitor spoilage in actual food systems. The chlorophylls functioned as the primary halochromic pigment, undergoing progressive discoloration in response to acidic or alkaline environments, typically resulting from microbial activity, thereby serving as a reliable indicator of food degradation.85 Similarly, in a study by Mohammadian et al. (2020),86 chlorophylls were incorporated as natural pigments in packaging films designed to function as temperature-sensitive indicators. While the primary focus was pH and gas sensitivity, the study observed visible color variations at elevated temperatures, correlating with the microbial spoilage activity. In another study, Kılıç (2024)87 fabricated a thermoplastic starch-based film enriched with chlorophyll-rich Aronia extract, which demonstrated gas-responsive colorimetric behavior. Exposure to ammonia, a volatile compound typically generated during the initial stages of spoilage in protein-rich food products, such as fish, induces a noticeable color change in the film, shifting its hue from green to brown.60 Fig. 4 illustrates the utilization of extracted chlorophylls from plants to develop bioactive compounds that are further incorporated into intelligent and active packaging systems. In intelligent packaging, these chlorophyll-based indicators visually signal food freshness by changing color, whereas in active packaging, chlorophyll-based coatings help maintain food freshness by slowing spoilage compared to uncoated packaging, thereby extending shelf life through bioactive protection. Collectively, these studies confirm the efficacy of chlorophylls as a functional indicator for intelligent packaging systems aimed at real-time spoilage detection.88
 | ||
| Fig. 4 Schematic of the extraction of chlorophylls from plants and their incorporation into intelligent and active food packaging systems. | ||
Chlorophylls also possess intrinsic fluorescence properties that can be exploited in the design of fluorescent sensing systems for intelligent packaging. Their fluorescence is highly responsive to environmental variations, making them viable noninvasive indicators for detecting food spoilage and microbial contamination.89 Upon exposure to light, chlorophylls emit red fluorescence in the spectral range of approximately 680 to 740 nm.90 However, during food degradation, metabolic by-products, such as organic acids, ammonia, and reactive oxygen species, alter local pH and oxidative conditions, leading to structural modifications in chlorophylls that reduce the fluorescence intensity or shift the emission wavelength. Such fluorescence variations provide measurable signals corresponding to the degree of food spoilage.91 For instance, Xue et al. (2025)92 designed a dual-channel pH-responsive fluorescent bio-ink system that integrated chlorophylls' natural fluorescence to generate visual cues corresponding to pH fluctuations during spoilage. Similarly, Herppich (2021)93 evaluated chlorophylls' fluorescence imaging (CFI) as a non-destructive tool for assessing the physiological status and quality of fresh produce, demonstrating its potential for real-time contactless evaluation in intelligent packaging.
When incorporated into biopolymer-based films and coatings, chlorophylls' intrinsic properties, including pH sensitivity, antioxidant capacity, and color responsiveness, can modulate physical parameters, such as tensile strength, flexibility, and barrier performance, which are all critical for packaging functionality and product protection. Table 2 summarizes the diverse range of biomaterials that incorporate chlorophylls for use in active and intelligent food packaging systems, highlighting applications across various food products, functional properties, such as antioxidant, antimicrobial, photoactivity, and freshness indicators, alongside their physical attributes (e.g., barrier performance, mechanical strength, biodegradability). Empirical studies have reported outcomes such as prolonged shelf life, minimized food spoilage, and visual cues for product freshness. Nevertheless, broader commercial applications require challenges related to physicochemical stability and cost-effective extraction methods to be addressed. Future advances in microencapsulation techniques, polymer matrix optimization, and integration with sensing technologies are expected to enhance the functional performance, facilitating sustainable, intelligent packaging solutions.
5 Conclusion, challenges, and future perspective
Chlorophyll-based active and intelligent packaging has emerged as a sustainable, multi-functional food packaging technology. Chlorophylls' molecular structures impart high antioxidant activity, photo-responsiveness, pH sensitivity, and spontaneous biodegradability. These naturally occurring properties enable chlorophylls to perform dual functions in packaging: they are active ingredients that slow food spoilage through antioxidant and antimicrobial activities, and they act as active indicators that provide immediate visual cues about food freshness through colorimetric and fluorescent signals. The addition of chlorophylls or microencapsulated chlorophylls to biodegradable polymer matrices enhances the physical and functional characteristics of films. Some examples include improved thickness, improved barrier properties towards moisture and gases, and changed mechanical behavior, typically resulting in smoother and more homogeneous film microstructures. Active packaging films incorporating chlorophylls have been shown to be effective scavengers of free radicals and inhibitors of spoilage bacteria, which results in extended shelf lives for various foods, including seafood and meat. Concurrently, the pH, temperature, and gas sensitivity (such as ammonia) of chlorophylls provide a measurable and often observable color transition from green to yellow, brown, or other colors characteristic of spoilage, thus arming consumers and supply chains with non-invasive freshness sensors.Chlorophylls are inherently unstable during conventional processing and storage conditions as they are sensitive and degrade rapidly when exposed to light, heat, oxygen, and fluctuations in pH, which damages their functional shelf life and color stability. Enhancing their stability through the application of advanced encapsulation methods such as micro- and nano-encapsulation by biopolymer carriers is critical; however, these approaches currently make manufacturing more complex and costly. Moreover, cost-effective large-scale extraction and purification procedures that preserve the functionality of chlorophylls must be optimized. Furthermore, compatibility with various polymer matrices can affect film homogeneity, mechanical resistance, and barrier properties, which requires proper formulation and control of processing.
Future advancements should focus more on encapsulation to preserve chlorophylls' bioactivity and enable controlled release of antimicrobial and antioxidant molecules, thereby optimizing shelf-life extension potential. Refining fabrication processes (extrusion, solution casting) may increase film robustness and scalability without compromising functionality. Combinations with digital sensing technology, such as fluorescence-based biosensors and IoT-based smart labels, can potentially advance real-time monitoring of food quality. As such, future research efforts should focus on developing active and intelligent packaging from chlorophylls to offer sustainable alternatives that enable both food preservation and transparency of freshness. This will ultimately result in safer, fresher, and more sustainable global food supply chains with reduced food waste and environmental impact.
Author contributions
Harshita Ranjan: writing – original draft, software, methodology, investigation, data curation, formal analysis. Manisha Joshi: writing – original draft, investigation, software. Swarup Roy: conceptualization, writing – review & editing, visualization, validation, supervision.Conflicts of interest
The authors declare that there is no conflicts of interest in this work.Data availability
No primary research results, software or code have been included, and no new data were generated or analyzed as part of this review.References
- D. Turan, B. M. Keukens and H. N. Schifferstein, Food packaging technology considerations for designers: Attending to food, consumer, manufacturer, and environmental issues, Compr. Rev. Food Sci. Food Saf., 2024, 23(6), e70058, DOI:10.1111/1541-4337.70058.
- M. E. González-López, S. D. J. Calva-Estrada, M. S. Gradilla-Hernández and P. Barajas-Álvarez, Current trends in biopolymers for food packaging: a review, Front. Sustain. Food Syst., 2023, 7, 1225371, DOI:10.3389/fsufs.2023.1225371.
- M. Z. Al Mahmud, M. H. Mobarak and N. Hossain, Emerging trends in biomaterials for sustainable food packaging: A comprehensive review, Heliyon, 2024, 10(1), 24122, DOI:10.1016/j.heliyon.2024.e24122.
- K. B. Biji, C. N. Ravishankar, C. O. Mohan and T. K. Srinivasa Gopal, Smart packaging systems for food applications: a review, J. Food Sci. Technol., 2015, 52(10), 6125–6135, DOI:10.1007/s13197-015-1766-7.
- G. Özkan and S. E. Bilek, Enzyme-assisted extraction of stabilized chlorophyll from spinach, Food Chem., 2015, 176, 152–157, DOI:10.1016/j.foodchem.2014.12.059.
- B. Park, S. Wi, H. Chung and H. Lee, Chlorophyll fluorescence imaging for environmental stress diagnosis in crops, Sensors, 2024, 24(5), 1442, DOI:10.3390/s24051442.
- H. Liu, Y. Zhou, Z. Ji, Z. Wang and X. Yang, Development of a fluorescence imaging system for real-time assessment and monitoring of the quality deterioration in Lapins cherries during refrigerated storage, Food Chem., 2026, 147890, DOI:10.1016/j.foodchem.2026.147890.
- M. Singh, K. Wang, C. Chen and M. G. Corradini, Luminescence-based techniques to monitor pre-, post-harvest and processing changes in fresh produce and derivatives: Principles, applications and trends, Trends Food Sci. Technol., 2025, 162, 105077, DOI:10.1016/j.tifs.2025.105077.
- A. Dodero, A. Escher, S. Bertucci, M. Castellano and P. Lova, Intelligent packaging for real-time monitoring of food-quality: Current and future developments, Appl. Sci., 2021, 11(8), 3532, DOI:10.3390/app11083532.
- T. Mkhari, J. O. Adeyemi and O. A. Fawole, Recent advances in the fabrication of intelligent packaging for food preservation: a review, Processes, 2025, 13(2), 539, DOI:10.3390/pr13020539.
- Y. Palanisamy, V. Kadirvel and N. D. Ganesan, Recent technological advances in food packaging: sensors, automation, and application, Sustain. Food Technol., 2025, 3(1), 161–180, 10.1039/d4fb00296b.
- S. Chavoshizadeh, S. Pirsa and F. Mohtarami, Conducting/smart color film based on wheat gluten/chlorophyll/polypyrrole nanocomposite, Food Packag. Shelf Life, 2020, 24, 100501, DOI:10.1016/j.fpsl.2020.100501.
- K. A. Sharifi and S. Pirsa, Biodegradable film of black mulberry pulp pectin/chlorophyll of black mulberry leaf encapsulated with carboxymethylcellulose/silica nanoparticles: Investigation of physicochemical and antimicrobial properties, Mater. Chem. Phys., 2021, 267, 124580, DOI:10.1016/j.matchemphys.2021.124580.
- T. Martins, A. N. Barros, E. Rosa and L. Antunes, Enhancing health benefits through chlorophylls and chlorophyll-rich agro-food: A comprehensive review, Molecules, 2023, 28(14), 5344, DOI:10.3390/molecules28145344.
- B. Yan, J. Huang, X. Yin, X. Guan, Y. Tang, J. Wen and X. Li, Sodium iron chlorophyllin-reinforced chitosan/gelatin films for fresh-cut vegetable packaging, Food Packag. Shelf Life, 2025, 52, 101608, DOI:10.1016/j.fpsl.2025.101608.
- G. López-Carballo, P. Hernández-Muñoz and R. Gavara, Photoactivated self-sanitizing chlorophyllin-containing coatings to prevent microbial contamination in packaged food, Coatings, 2018, 8(9), 328, DOI:10.3390/coatings8090328.
- R. H. Ukwatta, Y. Zheng, Y. Ma, F. Xue, X. Xiong and C. Li, The characterisation of cornstarch-based chlorophyllin composite film for preservation of shrimp under photodynamic irradiation, Int. J. Food Sci. Technol., 2024, 59(6), 3950–3966, DOI:10.1111/ijfs.17147.
- Y. Ni, Y. Li, M. Wang, H. Li, W. Zhang, L. Tan and B. Xu, Chitosan-based packaging films with antibacterial-sterilization integrated continuous activity for extending the shelf life of perishable foods, Int. J. Biol. Macromol., 2024, 275, 133351, DOI:10.1016/j.ijbiomac.2024.133351.
- Y. Lv, Y. Ai, F. Fang and H. Liao, Development of active packaging films utilized natural colorants derived from plants and their diverse applications in protein-rich food products, Food Innov. Adv., 2023, 2(3), 203–216, DOI:10.48130/FIA-2023-0022.
- R. Tanaka and A. Tanaka, Chlorophyll cycle regulates the construction and destruction of the light-harvesting complexes, Biochim. Biophys. Acta Bioenerg., 2011, 1807(8), 968–976, DOI:10.1016/j.bbabio.2011.01.002.
- R. Mandal and G. Dutta, From photosynthesis to biosensing: Chlorophyll proves to be a versatile molecule, Sens. Int., 2020, 1, 100058, DOI:10.1016/j.sintl.2020.100058.
- S. Ghosh, T. Sarkar, A. Das and R. Chakraborty, Natural colorants from plant pigments and their encapsulation: An emerging window for the food industry, Lwt, 2022, 153, 112527, DOI:10.1016/j.lwt.2021.112527.
- S. Pareek, N. A. Sagar, S. Sharma, V. Kumar, T. Agarwal, G. A. González-Aguilar, & E. M. Yahia. Chlorophylls: Chemistry and biological functions. Fruit and Vegetable Phytochemicals: Chemistry and Human Health, 2nd edn, 2017, 269–284. DOI:10.1002/9781119158042.ch14.
- B. K. Ndwandwe, S. P. Malinga, E. Kayitesi and B. C. Dlamini, Recent developments in the application of natural pigments as pH-sensitive food freshness indicators in biopolymer-based smart packaging: Challenges and opportunities, Int. J. Food Sci. Technol., 2024, 59(4), 2148–2161, DOI:10.1111/ijfs.16990.
- M. A. M. Jinasena, A. D. U. S. Amarasinghe, B. M. W. P. Amarasinghe and M. A. B. Prashantha, Extraction and degradation of chlorophyll a and b from Alternanthera sessilis, J. Natl. Sci. Found. Sri Lanka, 2016, 44(1), 11–21, DOI:10.4038/jnsfsr.v44i1.7977.
- E. Kwartiningsih, A. N. Ramadhani, N. G. A. Putri and V. C. J. Damara, Chlorophyll extraction methods review and chlorophyll stability of katuk leaves (Sauropus androgynous), J. Phys.: Conf. Ser., 2021, 1858(1), 012015, DOI:10.1088/1742-6596/1858/1/012015.
- É. Molnár, D. Rippel-Pethő and R. Bocsi, Solid-liquid extraction of chlorophyll from microalgae from photoautotroph open-air cultivation, Hungar. J. Ind. Chem., 2013, 119–122, DOI:10.1515/511.
- A. M. Ferreira, A. C. Leite, J. A. Coutinho and M. G. Freire, Chlorophylls extraction from spinach leaves using aqueous solutions of surface-active ionic liquids, Chem. Sustain. Dev., 2021, 2(4), 764–777, DOI:10.3390/suschem2040040.
- M. A. Costache, G. H. E. O. Campeanu and G. Neata, Studies concerning the extraction of chlorophyll and total carotenoids from vegetables, Rom. Biotechnol. Lett., 2012, 17(5), 7702–7708 CAS.
- J. Lu, X. Feng, Y. Han and C. Xue, Optimization of subcritical fluid extraction of carotenoids and chlorophyll a from Laminaria japonica Aresch by response surface methodology, J. Sci. Food Agric., 2014, 94(1), 139–145, DOI:10.1002/jsfa.6224.
- A. E. Edris, P. Wawrzyniak and D. Kalemba, Subcritical CO2 extraction of a volatile oil-rich fraction from the seeds of Nigella sativa for potential pharmaceutical and nutraceutical applications, J. Essent. Oil Res., 2018, 30(2), 84–91, DOI:10.1080/10412905.2017.1391721.
- A. C. Guedes, M. S. Gião, A. A. Matias, A. V. Nunes, M. E. Pintado, C. M. Duarte and F. X. Malcata, Supercritical fluid extraction of carotenoids and chlorophylls a, b and c, from a wild strain of Scenedesmus obliquus for use in food processing, J. Food Eng., 2013, 116(2), 478–482, DOI:10.1016/j.jfoodeng.2012.12.015.
- A. S. Fernandes, P. A. Caetano, E. Jacob-Lopes, L. Q. Zepka and V. V. de Rosso, Alternative green solvents associated with ultrasound-assisted extraction: A green chemistry approach for the extraction of carotenoids and chlorophylls from microalgae, Food Chem., 2024, 455, 139939, DOI:10.1016/j.foodchem.2024.139939.
- S. B. Kim, J. Bisson, J. B. Friesen, G. F. Pauli and C. Simmler, Selective chlorophyll removal method to “degreen” botanical extracts, J. Nat. Prod., 2020, 83(6), 1846–1858, DOI:10.1021/acs.jnatprod.0c00005.
- P. Ebrahimi, Z. Shokramraji, S. Tavakkoli, D. Mihaylova and A. Lante, Chlorophylls as natural bioactive compounds existing in food by-products: A critical review, Plants, 2023, 12(7), 1533, DOI:10.3390/plants12071533.
- U. M. Lanfer-Marquez, R. M. Barros and P. Sinnecker, Antioxidant activity of chlorophylls and their derivatives, Food Res. Int., 2005, 38(8–9), 885–891, DOI:10.1016/j.foodres.2005.02.012.
- A. Pérez-Gálvez, I. Viera and M. Roca, Carotenoids and chlorophylls as antioxidants, Antioxidants, 2020, 9(6), 505, DOI:10.3390/antiox9060505.
- C. Y. Hsu, P. Y. Chao, S. P. Hu and C. M. Yang, The antioxidant and free radical scavenging activities of chlorophylls and pheophytins, Food Nutr. Sci., 2013, 4(8A), 1, DOI:10.4236/fns.2013.48A001.
- I. Nurafifah, M. A. Hardianto, T. Erfianti, R. Amelia, D. Kurnianto and E. A. Suyono, The effect of acidic pH on chlorophyll, carotenoids, and carotenoid derivatives of Euglena sp. as antioxidants, AACL Bioflux, 2023, 16(4), 2391–2401 Search PubMed.
- N. Koca, F. Karadeniz and H. S. Burdurlu, Effect of pH on chlorophyll degradation and colour loss in blanched green peas, Food Chem., 2007, 100(2), 609–615, DOI:10.1016/j.foodchem.2005.09.079.
- S. A. Ledari, J. M. Milani, S. A. Shahidi and A. Golkar, Comparative analysis of freeze drying and spray drying methods for encapsulation of chlorophyll with maltodextrin and whey protein isolate, Food Chem., 2024, 21, 101156, DOI:10.1016/j.fochx.2024.101156.
- I. E. Agarry, Z. Wang, T. Cai, Z. Wu, J. Kan and K. Chen, Utilization of different carrier agents for chlorophyll encapsulation: Characterization and kinetic stability study, Food Res. Int., 2022, 160, 111650, DOI:10.1016/j.foodres.2022.111650.
- S. Sheibani, S. Jafarzadeh, Z. Qazanfarzadeh, M. J. O. Wijekoon, N. H. M. Rozalli and A. M. Nafchi, Sustainable strategies for using natural extracts in smart food packaging, Int. J. Biol. Macromol., 2024, 267, 131537, DOI:10.1016/j.ijbiomac.2024.131537.
- K. Huang and Y. Wang, Advances in bio-based smart food packaging for enhanced food safety, Trends Food Sci. Technol., 2025, 159, 104960, DOI:10.1016/j.tifs.2025.104960.
- A. Karnwal, G. Kumar, R. Singh, M. Selvaraj, T. Malik and A. R. M. Al Tawaha, Natural biopolymers in edible coatings: Applications in food preservation, Food Chem., 2025, 25, 102171, DOI:10.1016/j.fochx.2025.102171.
- S. Soiklom, W. Siri-Anusornsak, K. Petchpoung, S. Soiklom and T. Maneeboon, Development of bioactive edible film and coating obtained from Spirogyra sp. Extract applied for enhancing shelf life of fresh products, Foods, 2025, 14(5), 804, DOI:10.3390/foods14050804.
- S. Roshanak, M. Rahimmalek and S. A. H. Goli, Evaluation of seven different drying treatments in respect to total flavonoid, phenolic, vitamin C content, chlorophyll, antioxidant activity and color of green tea (Camellia sinensis or C. assamica) leaves, J. Food Sci. Technol., 2016, 53(1), 721–729, DOI:10.1007/s13197-015-2030-x.
- S. Punia Bangar, V. Chaudhary, N. Thakur, P. Kajla, M. Kumar and M. Trif, Natural antimicrobials as additives for edible food packaging applications: A review, Foods, 2021, 10(10), 2282, DOI:10.3390/foods10102282.
- D. Gupta, A. Lall, S. Kumar, T. D. Patil and K. K. Gaikwad, Plant-based edible films and coatings for food-packaging applications: recent advances, applications, and trends, Sustain. Food Technol., 2024, 2(5), 1428–1455, 10.1039/D4FB00110A.
- G. C. Pedro, F. D. S. Gorza, N. C. De Souza and J. R. Silva, Fractal structures in casting films from chlorophyll, J. Phys.: Conf. Ser., 2014, 480(1), 012011, DOI:10.1088/1742-6596/480/1/012011.
- S. Kumar, A. Mukherjee and J. Dutta, Chitosan based nanocomposite films and coatings: Emerging antimicrobial food packaging alternatives, Trends Food Sci. Technol., 2020, 97, 196–209, DOI:10.1016/j.tifs.2020.01.002.
- L. A. Gallo-García, B. Peron-Schlosser, D. Carpiné, R. M. de Oliveira, B. M. Simões, A. P. Dias and M. R. Spier, Feasibility of production starch/poly (butylene adipate-co-terephthalate) biodegradable materials with microalgal biomass by blown film extrusion, J. Food Process. Eng., 2022, 45(12), e14181, DOI:10.1111/jfpe.14181.
- Y. Xu, X. Yan, H. Zheng, J. Li, X. Wu, J. Xu and C. Du, The application of encapsulation technology in the food Industry: Classifications, recent Advances, and perspectives, Food Chem., 2024, 21, 101240, DOI:10.1016/j.fochx.2024.101240.
- E. N. Dewi, A. C. M. A. Tassakka, M. Yuwono, E. A. Suyono, L. Purnamayati and J. F. Alam, Effect of chlorophyll in alginate-based edible film in inhibiting spoilage of fish snacks, Canrea J. Food Technol. Nutr. Culin., 2022, 57–68, DOI:10.20956/canrea.v5i1.571.
- S. A. Ledari, J. M. Milani, S. A. Shahidi and A. Golkar, Fabrication and optimization of ultra-long stable microencapsulated chlorophyll using combinations of wall material via response surface methodology, Heliyon, 2024, 10(22), 40161, DOI:10.1016/j.heliyon.2024.e40161.
- I. Chiu and T. Yang, Biopolymer-based intelligent packaging integrated with natural colourimetric sensors for food safety and sustainability, Anal. Sci. Adv., 2024, 5(5–6), e2300065, DOI:10.1002/ansa.202300065.
- R. H. Ukwatta, R. Yuan, Y. Ma, X. Xiong, Y. Hu, C. Li and F. Xue, Effect of lipid addition on the physiochemical, structural, and photoactive antibacterial properties of cornstarch-chlorophyllin composite film, Food Res. Int., 2025, 202, 115699, DOI:10.1016/j.foodres.2025.115699.
- Y. R. Kang, Y. K. Lee, Y. J. Kim and Y. H. Chang, Characterization and storage stability of chlorophylls microencapsulated in different combination of gum Arabic and maltodextrin, Food Chem., 2019, 272, 337–346, DOI:10.1016/j.foodchem.2018.08.063.
- B. Micó-Vicent, M. Ramos, F. Luzi, F. Dominici, V. Viqueira, L. Torre and M. C. Garrigós, Effect of chlorophyll hybrid nanopigments from broccoli waste on thermomechanical and colour behaviour of polyester-based bionanocomposites, Polymers, 2020, 12(11), 2508, DOI:10.3390/polym12112508.
- G. Marappan, H. E. Tahir, N. Karim, A. Lakshmanan, M. R. I. Shishir, S. B. Hashim and X. Zou, Natural pigment-based pH/gas-sensitive intelligent packaging film for freshness monitoring of meat and seafood: Influencing factors, technological advances, and future perspectives, Food Rev. Int., 2026, 42(1), 58–95, DOI:10.1080/87559129.2025.2473026.
- Z. H. Zhang, H. Peng, M. W. Woo, X. A. Zeng, M. Brennan and C. S. Brennan, Preparation and characterization of whey protein isolate-chlorophyll microcapsules by spray drying: Effect of WPI ratios on the physicochemical and antioxidant properties, J. Food Eng., 2020, 267, 109729, DOI:10.1016/j.jfoodeng.2019.109729.
- V. Rizzi, P. Fini, F. Fanelli, T. Placido, P. Semeraro, T. Sibillano and P. Cosma, Molecular interactions, characterization and photoactivity of Chlorophyll a/chitosan/2-HP-β-cyclodextrin composite films as functional and active surfaces for ROS production, Food Hydrocolloids, 2016, 58, 98–112, DOI:10.1016/j.foodhyd.2016.02.012.
- H. Soltaninejad, H. Zare-Zardini, M. Ordooei, Y. Ghelmani, A. Ghadiri-Anari, S. Mojahedi and A. A. Hamidieh, Antimicrobial peptides from amphibian innate immune system as potent antidiabetic agents: a literature review and bioinformatics analysis, J. Diabetes Res., 2021, 2021(1), 2894722, DOI:10.1155/2021/2894722.
- R. Ambra, G. Pastore and F. Natella, The fate of chlorophylls in alkali-treated green table olives: A review, Molecules, 2023, 28(18), 6673, DOI:10.3390/molecules28186673.
- S. M. Milenković, J. B. Zvezdanović, T. D. Anđelković and D. Z. Marković, The identification of chlorophyll and its derivatives in the pigment mixtures: HPLC-chromatography, visible and mass spectroscopy studies, Adv. Technol, 2012, 1(1), 16–24 Search PubMed.
- G. Lopez-Carballo, P. Hernández-Muñoz, R. Gavara and M. J. Ocio, Photoactivated chlorophyllin-based gelatin films and coatings to prevent microbial contamination of food products, Int. J. Food Microbiol., 2008, 126(1–2), 65–70, DOI:10.1016/j.ijfoodmicro.2008.05.002.
- I. Viera, A. Pérez-Gálvez and M. Roca, Green natural colorants, Molecules, 2019, 24(1), 154, DOI:10.3390/molecules24010154.
- Y. Kumar, Y. Bist, D. Thakur, M. Nagar and D. C. Saxena, A review on the role of pH-sensitive natural pigments in biopolymers based intelligent food packaging films, Int. J. Biol. Macromol., 2024, 276, 133869, DOI:10.1016/j.ijbiomac.2024.133869.
- S. Bisht and K. K. Gaikwad, Natural pigments or dyes for sustainable food packaging application, Food Bioprocess Technol., 2025, 18(5), 4301–4325, DOI:10.1007/s11947-025-03756-2.
- H. Wang, Z. Cao, L. Yao, T. Feng, S. Song and M. Sun, Insights into the edible and biodegradable ulvan-based films and coatings for food packaging, Foods, 2023, 12(8), 1622, DOI:10.3390/foods12081622.
- D. Magalhães, R. Gonçalves, C. V. Rodrigues, H. R. Rocha, M. Pintado and M. C. Coelho, Natural pigments recovery from food by-products: Health benefits towards the food industry, Foods, 2024, 13(14), 2276, DOI:10.3390/foods13142276.
- A. Tanaka and H. Ito, Chlorophyll degradation and its physiological function, Plant Cell Physiol., 2025, 66(2), 139–152, DOI:10.1093/pcp/pcae093.
- G. Ghoshal, Recent trends in active, smart, and intelligent packaging for food products. In Food packaging and preservation, Academic Press, 2018, pp. 343–374. DOI:10.1016/b978-0-12-811516-9.00010-5.
- J. Jiang, W. Zhang, X. Yi, Q. Lei, Y. Liao, Y. Tan and W. Yu, Recent progress in properties and application of antibacterial food packaging materials based on polyvinyl alcohol, e-Polymers, 2024, 24(1), 20230097, DOI:10.1515/epoly-2023-0097.
- R. U. Islam, M. A. Khan and S. U. Islam, Plant derivatives as promising materials for processing and packaging of meat-based products–focus on antioxidant and antimicrobial effects, J. Food Process. Preserv., 2017, 41(2), e12862, DOI:10.1111/jfpp.12862.
- M. Latos-Brozio and A. Masek, The application of natural food colorants as indicator substances in intelligent biodegradable packaging materials, Food Chem. Toxicol., 2020, 135, 110975, DOI:10.1016/j.fct.2019.110975.
- A. Sobhan, A. Hossain, L. Wei, K. Muthukumarappan and M. Ahmed, IoT-enabled biosensors in food packaging: A breakthrough in food safety for monitoring risks in real time, Foods, 2025, 14(8), 1403, DOI:10.3390/foods14081403.
- X. Luo, A. Zaitoon and L. T. Lim, A review on colorimetric indicators for monitoring product freshness in intelligent food packaging: Indicator dyes, preparation methods, and applications, Compr. Rev. Food Sci. Food Saf., 2022, 21(3), 2489–2519, DOI:10.1111/1541-4337.12942.
- B. Liu, J. Zhuang and G. Wei, Recent advances in the design of colorimetric sensors for environmental monitoring, Environ. Sci.: Nano, 2020, 7(8), 2195–2213, 10.1039/D0EN00449A.
- K. D. Borah and J. Bhuyan, Magnesium porphyrins with relevance to chlorophylls, Dalton Trans., 2017, 46(20), 6497–6509, 10.1039/C7DT00823F.
- O. Długosz and M. Banach, Synthesis of metalloporphyrin complexes based on chlorophyllin, J. Mol. Struct., 2022, 1260, 132841, DOI:10.1016/j.molstruc.2022.132841.
- X. Li, R. Zhou, K. Xu, J. Xu, J. Jin, H. Fang and Y. He, Rapid determination of chlorophyll and pheophytin in green tea using fourier transform infrared spectroscopy, Molecules, 2018, 23(5), 1010, DOI:10.3390/molecules23051010.
- B. Seifert, M. Pflanz and M. Zude, Spectral shift as advanced index for fruit chlorophyll breakdown, Food Bioprocess Technol., 2014, 7(7), 2050–2059, DOI:10.1007/s11947-013-1218-1.
- D. Yu, S. Cheng, Y. Li, W. Su and M. Tan, Recent advances on natural colorants-based intelligent colorimetric food freshness indicators: fabrication, multifunctional applications and optimization strategies, Crit. Rev. Food Sci. Nutr., 2024, 64(33), 12448–12472, DOI:10.1080/10408398.2023.2252904.
- H. Eghbaljoo, M. Alizadeh Sani, I. K. Sani, S. M. Maragheh, D. K. Sain, Z. H. Jawhar and S. M. Jafari, Development of smart packaging halochromic films embedded with anthocyanin pigments; recent advances, Crit. Rev. Food Sci. Nutr., 2025, 65(4), 770–786, DOI:10.1080/10408398.2023.2280769.
- E. Mohammadian, M. Alizadeh-Sani and S. M. Jafari, Smart monitoring of gas/temperature changes within food packaging based on natural colorants, Compr. Rev. Food Sci. Food Saf., 2020, 19(6), 2885–2931, DOI:10.1111/1541-4337.12635.
- N. N. Kılıç, Thermoplastic Starch Films Doped with Cellulose Nanocrystals and Aronia Extract for Food Spoilage Detection Master's Thesis, Middle East Technical University, 2024 Search PubMed.
- O. S. Ingale, P. P. Bora, P. A. Pawase, O. Bashir, R. Shams, S. R. Patharkar and S. Roy, A review on intelligent packaging systems using betalain-rich biobased composite films in monitoring freshness of fish, shrimp, and meat, Food Bioprocess Technol., 2025, 18(10), 8154–8183, DOI:10.1007/s11947-025-03946-y.
- C. Chen, N. Gong, F. Qu, Y. Gao, W. Fang, C. Sun and Z. Men, Effects of carotenoids on the absorption and fluorescence spectral properties and fluorescence quenching of Chlorophyll a, Spectrochim. Acta, Part A, 2018, 204, 440–445, DOI:10.1016/j.saa.2018.06.061.
- E. Janeeshma, R. Johnson, M. S. Amritha, L. Noble, K. R. Aswathi, A. Telesiński and J. T. Puthur, Modulations in chlorophyll a fluorescence based on intensity and spectral variations of light, Int. J. Mol. Sci., 2022, 23(10), 5599, DOI:10.3390/ijms23105599.
- A. N. Misra, M. Misra and R. Singh, Chlorophyll fluorescence in plant biology, Biophysics, 2012, 7, 171–192, DOI:10.5772/35111.
- W. Xue, Q. Fan, Z. Wei, J. Zhu, H. Wu and K. Xu, Smart fluorescent bio-inks for DIW 3D printing: real-time food freshness sensors with dual-channel pH sensitivity, Adv. Compos. Hybrid Mater., 2025, 8(3), 262, DOI:10.1007/s42114-025-01322-0.
- W. B. Herppich, Chlorophylls fluorescence imaging for process optimisation in horticulture and fresh food production, Photosynthetica, 2021, 59, 422–437, DOI:10.32615/ps.2021.033.
| This journal is © The Royal Society of Chemistry 2026 |
