 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Evaluating sustainable techniques for the extraction of oils from processed sea bass and sea bream aquaculture side streams
Federica
Grasso
a,
Federica
Turrini
 *ab,
Marte
Jenssen
c,
Valentina
Orlandi
d,
Aseel
Swaidan
a,
Filippo
Falco
a,
Kjersti
Lian
c and
Raffaella
Boggia
ae
*ab,
Marte
Jenssen
c,
Valentina
Orlandi
d,
Aseel
Swaidan
a,
Filippo
Falco
a,
Kjersti
Lian
c and
Raffaella
Boggia
ae
aDepartment of Pharmacy, University of Genova, Viale Cembrano 4, 16148 Genova, Italy. E-mail: Federica.turrini@unige.it
bNational Center for the Development of New Technologies in Agriculture (Agritech), 80121 Napoli, Italy
cDepartment of Marine Biotechnology, Norwegian Institute of Food, Fisheries and Aquaculture Research (Nofima), Tromsø, Norway
dAimplas, Asociación de Investigación de Materiales Plásticos Y Conexas, Carrer de Gustave Eiffel, 4, 46980 Valencia, Spain
eNational Biodiversity Future Center (NBFC), 90133 Palermo, Italy
First published on 27th January 2026
Abstract
Global fish production has risen significantly to meet the growing demand for protein- and oil-rich food sources, yet this expansion has also led to increased waste generation, raising environmental concerns. Within the Horizon2020 EcoeFISHent project, this study investigates the valorization of unsorted, dehydrated side-streams from the filleting of farmed sea bream and sea bass. At laboratory scale, three environmentally sustainable methods for crude oil extraction were compared: green solvent extraction, Microwave-Assisted Extraction coupled to green solvents, and Enzyme-Assisted Extraction. All approaches yielded satisfactory oil recovery (33–42%) and maintained lipid oxidation parameters within the acceptable limits defined by regulatory standards (Codex Alimentarius, CXS 329-2017), confirming the oxidative stability of the extracted oils under the tested conditions. Fatty acid methyl ester profiling revealed consistent lipid composition across methods, with all oils exhibiting a beneficial omega-6/omega-3 ratio (1.7–2.6), supporting their potential for nutraceutical applications. Enzyme-assisted extraction emerged as a particularly efficient and scalable approach enabling the simultaneous recovery of oil and protein hydrolysates. This method was selected for semi-pilot scale-up in a 30 L reactor, achieving yields of 25% (w/w) for oil and 16.5% (w/w) for protein hydrolysates, whose protein content is 84.6 ± 1.44% (w/w). These findings highlight the feasibility of recovering two high-value fractions suitable for use in nutraceuticals, cosmetics, and biomaterials, contributing to the sustainable valorization of fishery side-streams.
Sustainability SpotlightThis study, developed within the EU Horizon 2020 EcoeFISHent project (call: Green Deal), addresses the growing environmental and economic challenges linked to aquaculture waste. This study investigates the sustainable valorisation of side-streams from farmed sea bass and sea bream through eco-friendly oil extraction methods. Enzyme-assisted extraction (EAE) proved to be the most efficient and scalable strategy, enabling the recovery of oil and protein hydrolysates for potential use in nutraceuticals, cosmetics, and biomaterials. The approach supports circular economy principles and aligns with UN SDGs: SDG 12 (Responsible Consumption and Production), SDG 9 (Industry, Innovation and Infrastructure), SDG 14 (Life Below Water), and SDG 3 (Good Health and Well-Being). |
1 Introduction
The fishery and aquaculture food supply chain generates increasing amounts of side-streams each year, including consumer waste, low-value industrial by-products, and by-catch that are undersized, damaged, or of limited economic value. Specifically, fish processing industries produce between 50–80% (w/w) of organic biomass such as heads, scales, skin, viscera and bones, which are usually only partially recycled (e.g., for animal feed) and more often disposed of in landfills, incinerated, or often released back into the sea, creating both economic and environmental concerns.1,2 Moreover, “destructive fishing”—defined as any fishing practice that depletes fish and invertebrate populations in an unsustainable manner while damaging habitats and ecosystems, thereby impairing their ecological functions3—poses significant ecological and economic challenges. One such practice involves discarding unwanted catches back into the ocean, which contributes to ecosystem degradation and resource inefficiency.2 In response, recent European Union regulations have prohibited this practice, mandating that all unwanted catches be brought ashore for proper management and utilization.4,5According to recent data from the Food and Agriculture Organization (FAO), global aquaculture production surpassed capture fisheries in 2022, reaching 130.9 million tons. In Italy, the aquaculture sector cultivates 25 species and is valued at approximately 400 million euros.6 To prevent overfishing and protect marine ecosystems, it is essential to manage undesired catches or by catches,-which include undersized, surplus, endangered, threatened, and protected species. This underscores the importance of adopting efficient and sustainable strategies for the utilization of aquaculture side streams.
To address these problems, the EU Commission has introduced the “Blue Growth” strategy, which aims for sustainable growth in marine and maritime sectors and reducing waste to meet the UN Sustainable Development Goals.4 The scientific community is actively working to valorize biomass that is typically discarded, aiming to isolate biomolecules that can be used for, among other applications, green and safe food ingredients or for other industrial applications. The EcoeFISHent project (Horizon 2020) aims to create sustainable regional clusters based on a multi-circular economy and industrial symbiosis.7 Its main goal is to boost financial returns for fisheries and aquaculture by utilizing underused biomass. In this context, the EcoeFISHent project explores valorization strategies, focusing on extracting valuable products from wild and aquaculture side-streams. In fact, fishery leftovers are usually rich in proteins, minerals, and lipids, and given the current global scenario regarding food security and availability, the loss of these important biomolecules is no longer acceptable.8
Among the various high-value compounds that can be extracted from fishery side-streams, fish oils are the most important. They are particularly rich in omega-3 polyunsaturated fatty acids (PUFAs), especially eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA).9,10 These bioactive compounds are widely utilized for nutraceutical and pharmaceutical applications due to their well-documented benefits in preventing and managing cardiovascular diseases, neurological disorders, and infant developmental issues.11,12 Wild caught and farmed fish – responsible for 90% of the global fish oil supply-have long been the primary sources of omega-3. Recent estimates indicate that the current global production of EPA and DHA is approximately 160![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons per year, with over 90% sourced from fisheries and aquaculture activities.13 However, as demand continues to surge, the search for sustainable alternatives has become increasingly urgent. For this reason, a promising economic and environmental strategy is to consider fish side-stream as valuable sources of bioactive compounds rich in PUFAs.14 At the same time, the development of eco-friendly and advanced technologies for efficient utilization of these side-streams is essential to address the growing challenges associated with the improper use of fish discards. The application of ultra-modern bio-intensified technologies such as enzymatic hydrolysis or Enzymatic Assisted Extraction (EAE), Supercritical Fluid Extraction (SFE), Pulsed Electric Field (PEF), High-Pressure Processing (HPP), extrusion, Microwave (MAE), and Ultrasound-Assisted Extraction (UAE) has been extensively researched.15,16
000 tons per year, with over 90% sourced from fisheries and aquaculture activities.13 However, as demand continues to surge, the search for sustainable alternatives has become increasingly urgent. For this reason, a promising economic and environmental strategy is to consider fish side-stream as valuable sources of bioactive compounds rich in PUFAs.14 At the same time, the development of eco-friendly and advanced technologies for efficient utilization of these side-streams is essential to address the growing challenges associated with the improper use of fish discards. The application of ultra-modern bio-intensified technologies such as enzymatic hydrolysis or Enzymatic Assisted Extraction (EAE), Supercritical Fluid Extraction (SFE), Pulsed Electric Field (PEF), High-Pressure Processing (HPP), extrusion, Microwave (MAE), and Ultrasound-Assisted Extraction (UAE) has been extensively researched.15,16
Recent advances in SFE have demonstrated improved selectivity and recovery of polyunsaturated fatty acids from marine sources, with optimized pressure and temperature conditions enhancing extract quality.17 Similarly, UAE has gained attention for its ability to intensify lipid extraction through cavitation effects.18
EAE has emerged as a green and selective method for lipid recovery, leveraging specific enzymes to disrupt cell walls and enhance solvent penetration, thereby increasing yield and preserving bioactivity.19
In MAE, microwave irradiation causes dipole rotation and ionic conduction within the sample, leading to rapid internal heating. This disrupts hydrogen bonds and weakens cell wall integrity, accelerating lipid diffusion into the solvent. The technique is especially efficient for extracting lipids from microalgae and fish tissues, preserving polyunsaturated fatty acids thanks to short exposure times.20 In parallel, the use of green solvents such as ethanol, aqueous ethanol, and deep eutectic solvents is gaining traction as sustainable alternatives to traditional organic solvents like hexane, due to their lower toxicity, biodegradability, and compatibility with food-grade applications.21
This study, summarized in Fig. 1, explores the potential of using fish biomass as an innovative source of bioactive compounds. The biomass consists of dehydrated, mixed side-streams from seabream (Sparus aurata) and seabass (Dicentrarchus labrax) filleting—including skin, head, tail, bones, and bowels—supplied by the Italian aquaculture company Aqua de Mâ, located in the Gulf of Tigullio, near Lavagna (Genova, Italy). The novelty of this study lies in the upcycling of unsorted fish biomass to extract lipids using green technologies. Several extraction methods were investigated for the recovery of crude oil from side streams of sea bass and sea bream. Conventional solid–liquid (S/L) extraction was performed using green solvents such as ethyl acetate and ethanol (a). These solvents were also employed in microwave-assisted extraction, initially based on parameters reported in the literature (b_1) and subsequently optimized (b_2). In addition, enzymatic extraction approaches were explored using two food-grade proteases: alcalase 3G PBN-66 L (c_1), a serine hydrolase of the subtilisin family produced via controlled fermentation of Bacillus licheniformis, and Corolase® 8000 (c_2), a fungal alkaline protease commonly applied in the hydrolysis of animal proteins to assess any possible differences in terms of oil recovery. Conditions where enzymes were omitted (c_3) were also evaluated to assess their impact on oil yield and quality.
Unlike conventional approaches that rely on sorted side-streams like fish skins or fish viscera, this study utilizes mixed filleting leftovers and undersized fish as the starting material. One of the key innovations is the cost-effective valorization of fish side-streams. Sorting individual parts of the fish is one of the most resource-intensive steps for small-scale fisheries. This makes recovery of side-stream particularly challenging in countries like Italy, where many small aquaculture farms discard or underutilize these resources due to the high costs of side-streams classification. Moreover, this method can be also applied to unwanted catches, enabling full utilization of these resources without the need for sorting.22
To address the logistical and utilization issues linked to the rapid perishability of fish side-streams, the biomass is first homogenized using a knife mill, ensuring uniform particle size and texture. This preparatory step facilitates subsequent industrial dehydration, which is carried out through a patented process operating under mild temperature and pressure conditions, preventing biomass degradation, such as lipid oxidation.23 The method guarantees microbiological stabilization and allows for the treatment of up to 120 kg h−1 of side streams. This step is crucial for improving logistics, as it eliminates the need for a costly cold chain, allowing unconventional biomass to be stored and handled at room temperature.
This innovative biomass provides an additional source for obtaining fish oil. When extracted using green technologies under mild conditions, it yields a high-quality product that, in many cases, can be used as is, eliminating the need for the costly refining process. Thus, three different green extraction methods were compared at laboratory scale to select a favorable process for upscaling at semi-pilot scale (30 L), providing good results in terms of yield, quality parameters, and fatty acid composition.
2 Materials and methods
2.1 Samples
Crude lipids were extracted from processed fish biomass composed of the filleting leftovers produced by the fish farm Aqua De Mâ (Lavagna, Italy) during the filleting operation of both seabream (Sparus aurata) and seabass (Dicentrarchus labrax). These side streams were left unsorted, comprising a heterogenous mix of heads, scales, bones, skin, viscera, etc., collectively referred to as Fish Mix (FM). The FM was further dried by the Italian company Themis S.p.A. (Legnano, Italy) with an innovative, patented industrial process (Patent No WO2015181769) using high vacuum conditions and low temperature.23 The resulting material, termed Dehydrated Fish Mix (DFM) is illustrated in Fig. 2. | ||
| Fig. 2 Samples of unsorted side streams of seabream and seabass before (FM) and after stabilization with a patented dehydration procedure (DFM). From left to right: unground fresh leftovers, ground fresh leftovers (FM), and a dehydrated fish mix (DFM) exhibiting an oily/creamy texture with almost no water content (see Table 2). | ||
For comparative purposes, two conventional sorted biomasses, fish skin and viscera, from the same aquaculture company were also analyzed for their proximate composition. The two samples, namely Dehydrated Fish Skin (DFS) and Dehydrated Fish Viscera (DFV) (Fig. 3) were processed using the same patented dehydration process described above.
 | ||
| Fig. 3 Samples of sorted side streams of skin and viscera of seabream and seabass after stabilization with a patented dehydration procedure (DFS and DFV). From left to right: dehydrated fish skin (DFS), exhibiting a solid texture, and dehydrated fish viscera (DFV), exhibiting an oily/creamy texture (see Table 2). | ||
2.2 Chemicals
All the chemicals and reagents used in this research were of analytical grade, furthermore ultrapure Milli-Q water (18 MΩ), produced by a Millipore Milli-Q system (Bedford, MA, USA), was employed. Ethyl acetate, ethanol 96°, hexane, sulfuric acid, sodium hydroxide, sodium sulphate, hydrochloric acid, thiobarbituric acid, trichloroacetic acid, 1-butanol, isopropanol and 1,1,3,3-tetramethoxypropane were purchased from Sigma-Aldrich Chemical Company (Steinheim, Germany). Büchi Labortechnik AG (Flawil, Switzerland) supplied the catalyst tablets and boric acid (2%) with Sher indicator for the Kjeldahl analysis. The enzyme 3G PBN-66 L was provided by 3G SINCE 2000 (Barcelona, Spain). Alcalase 3G PBN-66 L is a food-grade serine hydrolase of the subtilisin family, produced through controlled fermentation of Bacillus licheniformis. The enzyme exhibits broad proteolytic activity with low substrate specificity, enabling the hydrolysis of internal peptide bonds in various denatured proteins, including casein, hemoglobin, gelatin, soy, fish, and other proteins of both animal and plant origin. The hydrolysis products are low molecular weight peptides that are soluble in trichloroacetic acid. 3G PBN-66 L maintains its activity and stability at elevated temperatures. The optimal temperature for enzymatic activity is approximately 60 °C, with an effective activity range between 30 °C and 65 °C. The enzyme remains stable up to 70 °C, beyond which thermal inactivation occurs.24–26 The enzyme Corolase® 8000 was purchased from AB Enzymes GmbH (Darmstadt, Germany). It is a food-grade fungal alkaline protease used primarily for the hydrolysis of animal proteins. The enzyme is thermo-tolerant and exhibits high activity across a broad pH and temperature range, with optimal performance under neutral to alkaline conditions. It is typically applied in processes such as gelatin extraction, collagen hydrolysis, recycling of meat cut-offs, and recovery of fish proteins from by-products of the fishing industry. It supports cost-efficient hydrolysis processes. Its thermal stability—maintaining activity at temperatures up to 80 °C—contributes to streamlined industrial operations and helps reduce microbial risks during processing.27,28 CDR Srl (Firenze, Italy) supplied the CDR FoodLab® disposable pre-vialed reagents for the analyses of acidity, peroxides and p-anisidine.2.3 Proximate composition of starting material
The initial raw material was examined for its proximate composition using established official methods.29 The residual moisture content was analyzed both with official procedures (AOAC 950.46B) and with a Sartorius thermo-gravimetric analyzer (Massachusetts, USA). The determination of the ashes followed official methods (AOAC 942.05).29 The protein fraction was determined by Kjeldahl method (AOAC 981.10). The lipid fraction was extracted with a defatting proposed by Hara and Radin30 using a mixture of hexane/isopropanol and water.2.4 Detection of oxidative stress in starting material using TBARS (thiobarbituric acid reactive substances) assay
Following the same approach proposed by Grasso et al.,25 the starting materials DFM, DFS, and DFV were analyzed by evaluating the oxidative stress after a preliminary step of homogenization. The oxidation products were assessed with the TBARS analysis proposed by Hu and Zhong31 with minor differences. Briefly, to prepare the TBARS solution, 15 g of trichloroacetic acid and 0.75 g of thiobarbituric acid were dissolved in 100 mL of a mixture of 2-butanol, isopropanol, and 0.5 M HCl (in water) in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2:1 ratio (v/v/v). The samples were directly weighed into Pyrex tubes, the TBARS solution was added, and the tubes were incubated for 2 hours at 95 °C in a water bath (E200, LAUDA, Germany). The tubes were then quickly cooled with tap water. A calibration curve was created in the range of 0.61–6.10 µM using MDA (1,1,3,3-tetramethoxypropane) as the standard, and the absorbances were measured at 532 nm using a UV-Visible spectrophotometer (Agilent Cary 100 Varian Co., Santa Clara, CA, USA).
2:1 ratio (v/v/v). The samples were directly weighed into Pyrex tubes, the TBARS solution was added, and the tubes were incubated for 2 hours at 95 °C in a water bath (E200, LAUDA, Germany). The tubes were then quickly cooled with tap water. A calibration curve was created in the range of 0.61–6.10 µM using MDA (1,1,3,3-tetramethoxypropane) as the standard, and the absorbances were measured at 532 nm using a UV-Visible spectrophotometer (Agilent Cary 100 Varian Co., Santa Clara, CA, USA).
2.5 Crude oil extraction
Three different green extraction methods were evaluated for extracting crude lipids from DFM: Solid/Liquid (S/L) extraction using green solvents (i.e., ethyl acetate and ethanol) (a); MAE coupled with green solvents (b_1, b_2); EAE (c_1, c_2) compared with aqueous extraction without enzymes (c_3) as summarized in Fig. 4.The mass of the initial biomass used for extraction and the total lipid content determined by the Hara–Radin defatting method (ref)30 were considered when calculating yield and recovery (eqn (1) and (2) provided below). The oil was stored in a dark dryer until constant weight.
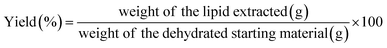 | (1) |
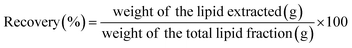 | (2) |
The recovery (%) was considered as the response variable. 14 experiments were carried out, plus three experiments corresponding to the central point in the model to estimate the experimental variability, for a total of 17 experiments (2k + 2k + N; k = 3, N = 3) planned to be performed randomly (refer to Table 3 for the experimental matrix/plan).
The postulated mathematical model is reported in eqn (3) below:
| Y = b0 + b1X1 + b2X2 + b3X3 + b12X1X2 + b13X1X3 + b23X2X3 + b11X12 + b22X22 + b33X32 | (3) |
The extraction was performed, according to the experimental plan, in an orbital shaking incubator (SKI 8R, Argo Lab, Kunshan, China) at 22 °C, protecting the flasks from light during the whole extraction to prevent oxidation. The extraction temperature of 22 °C was selected as a representative value for ambient laboratory conditions, reflecting the intention to perform the extraction under mild, non-thermal conditions, minimizing the risk of lipid oxidation or degradation while ensuring reproducibility across experiments. Therefore, a centrifugation step (4 °C, 15 min, 4200 rpm) followed and the upper phase was filtered with sodium sulphate and recovered; finally, a Rotavapor R-100 (BÜCHI Labortechnik AG, Switzerland), with a rotating bath heated up to 37 °C and away from light, was employed to vaporize the solvents mixture. The oil was placed in a dark dryer until constant weight, and the results were expressed in terms of yield and recovery (eqn (1) and (2)).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (w/v) and 25% ethanol. This latter used a solvent mixture of ethyl acetate/ethanol in a 3
10 (w/v) and 25% ethanol. This latter used a solvent mixture of ethyl acetate/ethanol in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (v/v), and a solid/solvent ratio of 1
1 ratio (v/v), and a solid/solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (w/v). Using a maximum extraction power of 1000 W, the samples were initially pre-heated for 2 minutes, then they were heated to 60 °C for 15 minutes, followed by a cooling period to 25 °C for an additional 15 minutes. After the extraction, a filtration with filter paper (4–12 µm) using a drying agent as sodium sulphate was performed to recover the extracts. At the end, a Multivapor system (Multivapor P-12, BÜCHI Labortechnik AG, Switzerland) with a rotating bath heated up to 37 °C and away from light, was used to evaporate the solvent mixture. The oil was placed in a dark dryer until constant weight, and the results were expressed in terms of yield and recovery (eqn (1) and (2)).
10 (w/v). Using a maximum extraction power of 1000 W, the samples were initially pre-heated for 2 minutes, then they were heated to 60 °C for 15 minutes, followed by a cooling period to 25 °C for an additional 15 minutes. After the extraction, a filtration with filter paper (4–12 µm) using a drying agent as sodium sulphate was performed to recover the extracts. At the end, a Multivapor system (Multivapor P-12, BÜCHI Labortechnik AG, Switzerland) with a rotating bath heated up to 37 °C and away from light, was used to evaporate the solvent mixture. The oil was placed in a dark dryer until constant weight, and the results were expressed in terms of yield and recovery (eqn (1) and (2)).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio (w/v) avoiding the increase of pH. Enzyme concentration of 0.25% was tested, and reactions were conducted for 1 h at 60 °C by constantly stirring the flasks with an orbital shaking incubator (SKI 8R, Argo Lab, Kunshan, China). At the end of the extraction, the enzyme was inactivated in a water thermostatic bath (E200, LAUDA, Germany) for 15 minutes at 90 °C. Subsequently, the mixture was stored at 4 °C overnight to allow separation of the liquid and solid phases. Therefore, a centrifugation step (25 °C, 20 minutes, 4200 rpm) occurred to separate the liquid from the solid: the upper phase, containing the oil was manually recovered, while the emulsion and the aqueous phases were centrifugated again (4 °C, 20 minutes, 4200 rpm) in small vials to isolate the remaining lipids still present in the emulsion. The corresponding yields and recoveries were calculated as previously reported.
2 ratio (w/v) avoiding the increase of pH. Enzyme concentration of 0.25% was tested, and reactions were conducted for 1 h at 60 °C by constantly stirring the flasks with an orbital shaking incubator (SKI 8R, Argo Lab, Kunshan, China). At the end of the extraction, the enzyme was inactivated in a water thermostatic bath (E200, LAUDA, Germany) for 15 minutes at 90 °C. Subsequently, the mixture was stored at 4 °C overnight to allow separation of the liquid and solid phases. Therefore, a centrifugation step (25 °C, 20 minutes, 4200 rpm) occurred to separate the liquid from the solid: the upper phase, containing the oil was manually recovered, while the emulsion and the aqueous phases were centrifugated again (4 °C, 20 minutes, 4200 rpm) in small vials to isolate the remaining lipids still present in the emulsion. The corresponding yields and recoveries were calculated as previously reported.
For comparison, the same protocol was followed, omitting only the addition of the enzyme, to evaluate any differences in oil recovery (aqueous extraction, c_3).
2.6 Evaluation of the quality of the extracted oil
The crude oil was analyzed with photometer CDR FoodLab ® Fat system (Firenze, Italy) using fast and low toxic analytical procedures that allow to obtain results comparable to the ISO/AOCS method as reported by the results obtained by titration methods Campden BRI Method TES-AC-211 based on ISO 660 or AOCS Cd 3d-63 or AOCS Ca 5a-40 or AOAC 940.28 for the analysis of the acidity, Campden BRI Method TES-AC-511 based on ISO 3960 or AOCS Cd 8b-90 or AOAC 965.33 for the analysis of peroxides, and Campden BRI Method TES-AC-360 for the analysis of p-anisidine.35,36 The tests aimed to evaluate the quality of the crude oils performing the analysis of the acidity (free fatty acids), the primary oxidation products (Peroxide Value, PV) assay and the p-Anisidine Value (AV) one. Therefore, the Total oxidation index (TOTOX index) was calculated as indicated in eqn (4) below.37| TOTOX = 2PV + AV | (4) |
2.7 Fatty acids analysis
Total lipids were extracted from the samples using different methods evaluated in this study, including cold extraction (Hara–Radin method),30 green solvent extraction, microwave-assisted extraction (MAE), enzymatic-assisted extraction (EAE), and water extraction.Fatty acid methyl esters (FAME) were prepared from the lipid extracts following the sequential alkaline–acid transmethylation procedure according to ISO 12966-2:2017.38 Briefly, 1 g of lipid extract was reacted with methanolic sodium methoxide to trans-esterify glycerides. Subsequently, a methanolic acid catalyst (methanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sulfuric acid 9
sulfuric acid 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) was added to esterify free fatty acids and residual acyl groups. The resulting FAME were recovered in hexane.
1, v/v) was added to esterify free fatty acids and residual acyl groups. The resulting FAME were recovered in hexane.
FAME analysis was performed using an Agilent 7890A GC system in accordance with ISO 12966-4:2015.39 Separation was achieved on a highly polar RTX-2330 capillary column (90% biscyanopropyl/10% phenylcyanopropyl polysiloxane; 60 m × 0.25 mm i.d., 0.20 µm film thickness; Restek Corp., Bellefonte, PA, USA). FAMEs were identified by comparison of retention times with a standard mixture (SUPELCO® 37 Component FAME Mix, Merck/Sigma-Aldrich, Darmstadt, Germany). The GC operating conditions were oven temperature from 50 °C (2 min) to 240 °C (15 min) at 2 °C min; injector and detector temperatures at 250 °C; helium carrier gas at a constant linear velocity of 30 cm s; injection volume 1 µL (in duplicate); split ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50.
50.
2.8 Upscaling of enzymatic extraction
A pilot scale experiment was conducted using a total of 8 kg of DFM suspended in 16 L of tap water. Enzymatic hydrolysis was performed at 60 °C for 60 minutes using 0.25% (w/w, based on wet biomass weight) Corolase® 8000 as the proteolytic enzyme. The hydrolysis was performed in a Chemglass 30 L reactor with Optichem stirring system (ChemGlass Life Sciences, Vineland, New Jersey, USA). Upon completion, the enzyme was inactivated by heating the mixture to 85 °C for 20 minutes. Large insoluble fractions (e.g., bones) were separated using a rough filter and an emulsion phase was manually removed. The remaining material was stored overnight at 4 °C, before further handling. Overnight storage led to a separation of the liquid phase and the emulsion phase. The emulsion phase was manually removed before the remaining material was subjected to centrifugation using an Avanti JXN-26 centrifuge (Beckman Coulter, Brea, California, USA), resulting in a liquid protein hydrolysate, an oil/emulsion phase, and some remaining insoluble material. The protein hydrolysate was kept at 4 °C overnight to remove any potential remaining oil. This was further concentrated via rotary evaporation at 50 °C, using a Hei_VAP industrial rotavapor (Heidolph, Schwabach, Germany) under a vacuum of 20 mbar and a rotation speed of 50 rpm. The concentrated protein hydrolysate was finally lyophilized, yielding a dry protein hydrolysate. The oil/emulsion layer was centrifuged to recover the oil phase, to which 0.5% (w/v) tocopherol was added as an antioxidant. The oil was stored at −20 °C and the solid fraction, collected after filtration and centrifugation, was lyophilized using a Labconco FreeZone Plus freeze dryer (Labconco Corporation, Kansas City, MO, USA). Further analyses regarding the quality of the oil, i.e. acidity, peroxide value, p-anisidine value together with TOTOX index were carried out as previously described.2.9 Statistical analysis
Every measurement was carried out at least two times, and results are presented as the mean value ± standard deviation (SD). Both the Excel Data Analysis Tool and the Chemometric Agil Tool (CAT) were employed to analyze data.32 Statistical significance was assessed using the One-way ANOVA and Tukey's post hoc test. Differences were considered statistically significant at p < 0.05. Principal component analysis (PCA) was applied as a common multivariate statistical method of unsupervised pattern recognition.3 Results and discussion
3.1 Proximate analysis and detection of oxidative stress in starting material
The samples were represented by an unusual type of fish biomass, composed of all the side streams generated by an Italian aquaculture company after the process of seabream and seabass filleting. These side streams of both fishes include different fractions (heads, scales, bones, skin, viscera etc.) all mixed, in contrast to what can be found in most articles dealing with the subject of extraction of lipids or proteins; in fact, traditional methods use sorted biomasses like fish skin40 or viscera.41 It is important to note that, for small aquaculture farms aiming to valorize their side streams, the sorting process—required to separate components such as heads, fins, and bones—remains prohibitively expensive, both in terms of money and time. As a result, it is frequently omitted, with significant negative implications for the environmental sustainability of aquaculture products. Moreover, due to the high perishability of this organic matrix, the biomass was pre-processed using an industrially dehydration technique under patent developed by Themis S.p.A. (Legnano, Italy)23 to avoid the costs of storage linked to the cold chain, extend the shelf life and to enhance the logistics lowering the volume up to 2/3 of the starting weight.25 This pretreatment step enhances the efficiency of downstream valorization processes, which are frequently conducted away from the point of side-stream generation.The proximate analysis of the unconventional biomass after dehydration (DFM) was compared to the proximate analysis of two sorted biomasses—skin and viscera—from the same fishery side-streams namely DFS and DFV, respectively. Due to the high heterogeneity observed among batches—each consisting of varying fractions of industrial fish by-products—sampling and related analyses were conducted repeatedly across numerous batches throughout one year of production (year 2022). The results of the average proximate composition of the DFM are presented as ranges and mean values, reflecting the significant variability inherent to the different batch composition, while for DFS and DFV, the results were reported as average and standard deviation (Table 2).
| DFM (g/100 g) | DFS (g/100 g) | DFV (g/100 g) | |
|---|---|---|---|
| a Results are expressed as range and mean value (in brackets). b Results are expressed as mean ± SD (n = 2). | |||
| Residual moisturea,b | 1–3 (1.2) | 5.0 ± 0.4 | 4.5 ± 0.4 |
| Crude proteinsa,b | 39–48 (42.3) | 59.9 ± 4.2 | 15.2 ± 0.1 |
| Ashesa,b | 10–17 (14.8) | 17.8 ± 0.1 | 2.5 ± 0.1 |
| Lipidsa,b | 27–46 (34) | 17.4 ± 1.1 | 55.8 ± 6.8 |
| TBARS (mmol MDA/kg)b | 1.4 ± 0.5 | 0.8 ± 0.1 | 2.3 ± 0.4 |
Thanks to the dehydration process, fats, proteins and ashes were concentrated in all the samples and the water was barely absent, resulting in an improvement in the extractability of the lipids. Based on the proximate analysis results, although the viscera naturally contain the highest lipid content, the unsorted sample (DFM) exhibited a particularly intriguing composition for lipid extraction, facilitating their upcycling. Moreover, the TBARS test was conducted directly on the samples (DFM, DFS, DFV), confirming the high quality of the starting biomass. The short supply chain between the aquaculture farm—crucial for preserving freshness—and the mild dehydration facility likely play a key role in limiting oxidative stress. This hypothesis is further supported by a comparative analysis with similar, unsorted tuna side-streams processed using the same dehydration method, which exhibited higher TBARS values.25,42 These differences are likely attributable not only to species-specific and compositional factors, but also to the longer and more complex supply chain typically associated with tuna processing.
3.2 Crude oil extraction
A Design of Experiment (DoE) approach was utilized to optimize oil extraction using green solvents (ethanol and ethyl acetate mixture) from DFM biomass. The experiments were conducted randomly, and the results are presented in Table 3, expressed as both yield (%) and recovery (%), relative to a chosen reference method, to account for the intrinsic variability of different batches of organic biomass, as previously discussed. The reference method selected for determining the total lipid content in fish samples was the Hara–Radin protocol,30 a cold solid/liquid extraction using a hexane/isopropanol mixture, as recommended by Ramalhosa et al.50 and confirmed by numerous experiments carried out with similar fish matrices by the authors.42 The method was preferred to the traditional Folch,44,45 to avoid the use of halogenated solvents, i.e. chloroform, and to the official Soxhlet, which is considered too highly energy-consuming and highly impactful on thermosensitive compounds.51
| X 1 solid/solvent (w/v) | X 2 time (min) | X 3 EtOH (%) | Yield (%) | Recovery (%)a | |
|---|---|---|---|---|---|
| a Recovery was calculated based on a 34% yield of total lipids, as determined using the extraction protocol described by Hara and Radin.30 | |||||
| 1 | −1 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
−1 (30) | −1 (0% EtOH) | 31.3 | 92.1 |
| 2 | 1 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) 10) |
−1 (30) | −1 (0% EtOH) | 32.7 | 96.0 |
| 3 | 0 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) 5) |
0 (105) | −1 (0% EtOH) | 31.7 | 93.1 |
| 4 | −1 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
1 (180) | −1 (0% EtOH) | 20.8 | 61.3 |
| 5 | 1 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) 10) |
1 (180) | −1 (0% EtOH) | 30.9 | 91.0 |
| 6 | 0 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) 5) |
−1 (30) | 0 (25% EtOH) | 31.9 | 93.8 |
| 7 | −1 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
0 (105) | 0 (25% EtOH) | 26.7 | 78.5 |
| 8 | 0 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) 5) |
0 (105) | 0 (25% EtOH) | 31.1 | 91.4 |
| 9 | 1 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) 10) |
0 (105) | 0 (25% EtOH) | 33.5 | 98.4 |
| 10 | 0 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) 5) |
1 (180) | 0 (25% EtOH) | 30.8 | 90.5 |
| 11 | −1 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
−1 (30) | 1 (50% EtOH) | 26.7 | 78.4 |
| 12 | 1 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) 10) |
−1 (30) | 1 (50% EtOH) | 33.4 | 98.2 |
| 13 | 0 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) 5) |
0 (105) | 1 (50% EtOH) | 31.8 | 93.4 |
| 14 | −1 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
1 (180) | 1 (50% EtOH) | 20.7 | 60.9 |
| 15 | 1 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) 10) |
1 (180) | 1 (50% EtOH) | 33.2 | 97.7 |
| 16 | 0 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) 5) |
0 (105) | 0 (25% EtOH) | 32.4 | 95.2 |
| 17 | 0 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) 5) |
0 (105) | 0 (25% EtOH) | 31.3 | 92.1 |
Table 3 shows data from a single batch. Later analyses were done on other batches, which had slightly different lipid contents (though still within the range reported in Table 3). To make results comparable across batches, results are expressed as recovery percentages.
The analysis of the results enabled the quantification of the following model coefficients using recovery (%) as the response variable (eqn (5)):
| y = 93.7 (***) + 11.0 x1 (***) − 5.7 x2 (***) − 0.5 x3 + 5.3 x1x2 (**) + 2.9 x1x3 (*) + 2.2 x2x3 − 5.9 x12 (*) − 2.2 x22 − 1.1 x32 | (5) |
They are graphically shown in Fig. 5. Stars represent the statistical significance (p-value; * = 0.05, ** = 0.01, *** = 0.001).
 | ||
| Fig. 5 The coefficients of the models obtained by the FCCCD (p-value * = 0.05, ** = 0.01, *** = 0.001). | ||
From the coefficient plot (Fig. 5), it is evident that the linear terms X1 (ratio: solid-to-solvent ratio) and X2 (time) (***: p < 0.005), the first two interactions, namely X1X2 (ratio-time; **: p < 0.01) and X1X3 (ratio-ethanol; *: p < 0.05), and the quadratic term X12 (ratio–ratio; *: p < 0.05) significantly influence the response. Specifically, regarding the linear terms, the variable ratio (X1) positively influences the response, meaning the yield of extracted fish oil increases with the amount of solvent used, while the extraction time (X2) is inversely proportional to the extraction yield.
The results indicate that the optimal conditions are represented by a solid/solvent ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (w/v), a solvent ratio of 25% EtOH/75% EtOAc, and an extraction time of 105 minutes. As highlighted by the Response Surface Recovery (Fig. 6), the DoE approach revealed several favorable conditions yielding over 90%, demonstrating the robustness and flexibility of the process. Among these optimal scenarios evaluated, the selected condition was the one that ensured the highest yield. However, other configurations also proved to be optimal, as they offered advantages such as reduced processing time and/or solvent consumption, with only a moderate compromise in terms of yield. This choice aligns with the core principles of DoE, which emphasize not only performance maximization but also practical applicability and balance among multiple factors.52 The yield could achieve more than 90% while saving time and solvent if the extraction time was shortened to 30 minutes and the solid/solvent (w/v) ratio was lowered to 1
10 (w/v), a solvent ratio of 25% EtOH/75% EtOAc, and an extraction time of 105 minutes. As highlighted by the Response Surface Recovery (Fig. 6), the DoE approach revealed several favorable conditions yielding over 90%, demonstrating the robustness and flexibility of the process. Among these optimal scenarios evaluated, the selected condition was the one that ensured the highest yield. However, other configurations also proved to be optimal, as they offered advantages such as reduced processing time and/or solvent consumption, with only a moderate compromise in terms of yield. This choice aligns with the core principles of DoE, which emphasize not only performance maximization but also practical applicability and balance among multiple factors.52 The yield could achieve more than 90% while saving time and solvent if the extraction time was shortened to 30 minutes and the solid/solvent (w/v) ratio was lowered to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 but avoiding the 1
5 but avoiding the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (w/v) one. It is worth considering the high cost of solvents, too, especially in anticipation of upscaling the process at an industrial level. Furthermore, it is important to consider that prolonged processing times significantly influence oxidative processes. Similarly, the solid/solvent ratio (w/v) plays a critical role, as larger volumes require extended evaporation times under vacuum at mild temperatures (e.g., 37 °C). Although vacuum evaporation at such temperatures helps preserve thermolabile compounds and mitigates oxidation, it inherently demands longer durations, particularly when handling greater solvent volumes.
2 (w/v) one. It is worth considering the high cost of solvents, too, especially in anticipation of upscaling the process at an industrial level. Furthermore, it is important to consider that prolonged processing times significantly influence oxidative processes. Similarly, the solid/solvent ratio (w/v) plays a critical role, as larger volumes require extended evaporation times under vacuum at mild temperatures (e.g., 37 °C). Although vacuum evaporation at such temperatures helps preserve thermolabile compounds and mitigates oxidation, it inherently demands longer durations, particularly when handling greater solvent volumes.
| Yield (%)a | Recovery (%) | |
|---|---|---|
| a Results are reported as mean ± SD (n ≥ 2). | ||
| ref_S/L cold extraction with Hara–Radin method30 | 42.1 ± 3.0a | 100 |
| a_S/L green solvents | 34.4 ± 4.2ab | 81.6 |
b_1 MAE lit34(ethyl acetate![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol 2 ethanol 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) 1, v/v) |
34.8 ± 0.3ab | 82.7 |
b_2 MAE opt (ethyl acetate![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol 3 ethanol 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) 1, v/v) |
41.8 ± 1.6ab | 99.3 |
| c_1 EAE, 3G PBN-66 L enzyme | 32.6 ± 0.2b | 77.4 |
| c_2 EAE, Corolase® 8000 enzyme | 34.0 ± 1.9ab | 80.7 |
| c_3 water | 35.2 ± 2.4ab | 83.6 |
Table 4 shows both the yields and the recovery (%) of each oil obtained using the conventional S/L cold extraction using ethyl acetate![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol (3
ethanol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) (a), as well as MAE using ethyl acetate
1, v/v) (a), as well as MAE using ethyl acetate![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol (2
ethanol (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) (b_1), ethyl acetate
1, v/v) (b_1), ethyl acetate![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol (3
ethanol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) (b_2), EAE (c_1, c_2) and water (c_3). As shown in the table, the extraction method, the type of solvent and the ratio (w/v) used for extraction influenced the oil yield (%). The One-Way ANOVA analysis (F (6, 7) = 5.476, p = 0.021), followed by Tukey's post hoc test, reveal statistically significant differences in lipid yield among the reference method (ref_S/L cold extraction with Hara–Radin method) and the EAE method (c_1). As previously mentioned, another batch of the same DFM biomass was used, and all the extraction methods were repeated in replicates for consistency. A yield of 34.4 ± 4.2% was achieved using the S/L cold extraction method under conditions determined by the previous DoE (a),53 (1
1, v/v) (b_2), EAE (c_1, c_2) and water (c_3). As shown in the table, the extraction method, the type of solvent and the ratio (w/v) used for extraction influenced the oil yield (%). The One-Way ANOVA analysis (F (6, 7) = 5.476, p = 0.021), followed by Tukey's post hoc test, reveal statistically significant differences in lipid yield among the reference method (ref_S/L cold extraction with Hara–Radin method) and the EAE method (c_1). As previously mentioned, another batch of the same DFM biomass was used, and all the extraction methods were repeated in replicates for consistency. A yield of 34.4 ± 4.2% was achieved using the S/L cold extraction method under conditions determined by the previous DoE (a),53 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 w/v, 105 min and 25% EtOH), corresponding to a recovery of about 82%. To enhance the yield, MAE was performed using the same green solvents (ethanol
10 w/v, 105 min and 25% EtOH), corresponding to a recovery of about 82%. To enhance the yield, MAE was performed using the same green solvents (ethanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethyl acetate) but following the conditions recommended by Costa et al.,34 with a solvent mixture ratio of 2
ethyl acetate) but following the conditions recommended by Costa et al.,34 with a solvent mixture ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) (b_1). This resulted in a yield of 34.8 ± 0.3%, showing no significant improvement in yield compared to extraction without microwaves, although it did offer a substantial time-saving advantage. The conventional extraction method takes approximately 105 minutes per sample, whereas MAE reduces this to 17 minutes of extraction followed by 15 minutes of cooling. Additionally, the ETHOS-X microwave system (Milestone S.r.l., Sorisole, Bergamo, Italy) allows for simultaneous processing of up to 15 samples, increasing overall efficiency.
1 (v/v) (b_1). This resulted in a yield of 34.8 ± 0.3%, showing no significant improvement in yield compared to extraction without microwaves, although it did offer a substantial time-saving advantage. The conventional extraction method takes approximately 105 minutes per sample, whereas MAE reduces this to 17 minutes of extraction followed by 15 minutes of cooling. Additionally, the ETHOS-X microwave system (Milestone S.r.l., Sorisole, Bergamo, Italy) allows for simultaneous processing of up to 15 samples, increasing overall efficiency.
The highest yield (41.8 ± 1.6%) was obtained using MAE with a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) ethyl acetate/ethanol ratio, which corresponds to the optimized condition identified in the previous design of experiments (DoE, condition a) When comparing the same solvent system and ratio (3
1 (v/v) ethyl acetate/ethanol ratio, which corresponds to the optimized condition identified in the previous design of experiments (DoE, condition a) When comparing the same solvent system and ratio (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), MAE proved more effective than conventional solid–liquid (S/L) extraction, (41.8 ± 1.6% vs. 34.4 ± 4.2%).
1), MAE proved more effective than conventional solid–liquid (S/L) extraction, (41.8 ± 1.6% vs. 34.4 ± 4.2%).
Unlike traditional extraction techniques, MAE employs electromagnetic waves to enhance solvent penetration and facilitate the release of target analytes from the matrix. This mechanism increases extraction efficiency while significantly reducing processing time.54 These findings support the observed improvement in oil yield with MAE, even when the solvent composition and ratio were kept constant.
Furthermore, the increased oil yield observed when shifting the solvent ratio from 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 3
1 to 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) is attributed to the higher proportion of ethyl acetate, a more non-polar solvent than ethanol. Ethyl acetate exhibits greater affinity for lipophilic compounds, enhancing their solubility and interaction with the matrix. This results in more efficient extraction of non-polar constituents at higher ratios.
1 (v/v) is attributed to the higher proportion of ethyl acetate, a more non-polar solvent than ethanol. Ethyl acetate exhibits greater affinity for lipophilic compounds, enhancing their solubility and interaction with the matrix. This results in more efficient extraction of non-polar constituents at higher ratios.
To enable microwave transmission in the apolar solvent system, a button made of Milestone's patented Weflon™ material was inserted as a coupling agent. Weflon™, a proprietary dielectric material, absorbs microwave energy and transfers heat to the surrounding apolar medium, which otherwise would not interact effectively with microwave radiation. This setup ensures efficient energy transfer and enables the successful application of MAE in non-polar environments. Although MAE using Weflon™ has demonstrated promising laboratory-scale results, its translation to industrial settings faces notable limitations. Weflon™ is a proprietary dielectric material, and its restricted availability and potentially high cost may hinder large-scale implementation, particularly in continuous processing environments. Furthermore, its long-term durability under sustained microwave exposure and in contact with volatile solvent mixtures remains to be thoroughly assessed. Industrial integration would necessitate custom-designed microwave reactors compatible with Weflon™, which could significantly increase capital expenditure and operational complexity. Additionally, the use of low-polar solvents such as ethyl acetate in microwave-assisted systems introduces safety concerns due to their flammability and volatility, requiring stringent process controls and specialized equipment. Therefore, while MAE with Weflon™ offers high extraction efficiency at laboratory scale, its industrial applicability depends on further engineering validation and cost-benefit analysis.
EAE is one of the most remarkable green lipid extraction methods available.55–57 EAE minimizes the use of toxic solvents, thereby lowering environmental impact and improving safety for both operators and end-users. From an economic perspective, while enzyme costs may be non-negligible, the reduction in solvent handling, disposal requirements, and potential regulatory burdens can offset these expenses, especially in large-scale or food-grade applications.58 This kind of extraction has been shown to improve the extraction efficiency of lipids and lipophilic substances such as vitamins.59 It also separates the protein fraction, yielding low molecular weight peptides,42,60 and the lipid fraction using the aqueous phase–organic phase separation principle.
Despite its environmental advantages, EAE demonstrated slightly lower performance compared to organic solvent extraction in several cases, including the present study (see Table 4). Organic solvents, due to their stronger affinity for lipophilic compounds and more aggressive extraction capabilities, often result in a little higher yield.
Nevertheless, EAE offers several advantages over organic solvent-based methods, both in terms of cost-effectiveness and extraction versatility. In particular, EAE enables the simultaneous recovery of hydrolyzed peptides along with lipids, thanks to enzymatic breakdown of the protein matrix and the use of aqueous–organic phase separation (Fig. 7). This dual recovery makes EAE a valuable approach when both lipid and protein fractions are of interest, especially in food and nutraceutical applications.
EAE is indeed regarded as the most scalable method due to its low costs, use of simple equipment, and the ability to simultaneously valorize the protein fraction.25,42 Although the hexane/isopropanol method (ref) and optimized microwave extraction (b_2) have produced higher yields (42.1 ± 3.0 and 41.8 ± 1.6, respectively), the lipid yields obtained through EAE were considered good: approximately 33% using 3G-PBN-66L (c_1) and 34% with Corolase® 8000 (c_2). These values are comparable to those obtained using water alone (c_3) (around 35%), as well as those achieved through conventional solid–liquid extraction with green solvents (a) (34.4%) and MAE (b_1) (approximately 35%).
Although the extraction yields obtained with enzymatic hydrolysis and water-only treatment were comparable, the use of enzymes was preferred due to their ability to promote the release of low molecular weight peptides. These peptides are particularly relevant for applications in the nutraceutical and cosmetic fields, where bioactivity and absorption are often enhanced by reduced molecular size.61 Thus, enzymatic treatment was selected not solely for efficiency, but for its capacity to generate functionally valuable compounds.
3.3 Evaluation of the quality of the oil
To evaluate the quality of oils, Codex Alimentarius (CXS 329-2017)62 recommends three analytical measurements: acidity, PV, and AV. The TOTOX index is a combination of PV and AV, which are thought to be responsible for primary and secondary oxidation products, respectively, and acidity, which specifically indicates the free fatty acids (FFA) as a percentage of oleic acid. These parameters must coincide with those that Codex recommends acidity ≤ 3 mg KOH per g, PV ≤ 5 meqO2 per kg oil, AV ≤ 20, and ToTox ≤ 26. The same limits are recommended by the International Fish Oil Standards (IFOS),63 while the European Pharmacopeia Standard, recommends higher values: PV ≤ 10 meqO2 per kg oil, AV ≤ 30, and TOTOX ≤50, but lower levels for Acidity ≤ 2.64 The acidity, PV, AV, and TOTOX results from the analyses of crude oils extracted using different methods are presented in Table 5 below.| Acidity (% oleic Acid)a | PVa | AVa | TOTOXa | |
|---|---|---|---|---|
| a Results are expressed as mean ± SD (n = 2). | ||||
| ref_S/L cold extraction with Hara-Radin method30 | 2.8 ± 0.1b | 6.91 ± 0.1a | 1.0 ± 0.3a | 14.82 ± 0.30a |
| a_S/L green solvents | 3.2 ± 0.1a | 1.77 ± 0.04b | 1.6 ± 0.1a | 5.08 ± 0.10b |
b_1 MAE lit34 (ethyl acetate![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ethanol 2 Ethanol 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) 1, v/v) |
3.4 ± 0.1a | 1.36 ± 0.20b | 2.1 ± 0.2a | 4.77 ± 0.61b |
b_2 MAE opt (ethyl acetate![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ethanol 3 Ethanol 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) 1, v/v) |
3.2 ± 0.1a | 2.11 ± 0.88b | 1.7 ± 0.2a | 5.86 ± 1.98b |
| c_1 EAE, 3 G PBN 66L enzyme | 3.3 ± 0.1a | 2.57 ± 1.66b | 1.6 ± 0.2a | 6.68 ± 3.11b |
| c_2 EAE, Corolase® 8000 enzyme | 3.4 ± 0.1a | 1.71 ± 1.12b | 1.8 ± 0.1a | 5.17 ± 2.16b |
| c_3 water | 3.4 ± 0.1a | 0.55 ± 0.26b | 1.2 ± 0.7a | 2.29 ± 0.18b |
All the extracted oils exhibited exceptionally low oxidation parameters, fully compliant with Codex Alimentarius standards, with TOTOX values significantly below the maximum permitted limit of 26. Therefore, the results confirmed their excellent oxidative stability and high overall quality. The One-Way ANOVA analysis (F (6, 7) = 11.697, p = 0.002), followed by Tukey's post hoc test, reveals statistically significant differences in TOTOX among the reference method (ref_S/L cold extraction with Hara–Radin method) and all the other extraction methods. No statistically significant differences were observed among the six green extraction methods (One-Way ANOVA analysis F (5,6) = 1.412, p = 0.340). With respect to acidity, the measured values were slightly above the Codex Alimentarius threshold yet remained consistent across the various extraction methods evaluated. The One-Way ANOVA analysis (F (6,7) = 12.037, p = 0.002), followed by Tukey's post-hoc test, reveals statistically significant differences in acidity among the reference method (ref_S/L cold extraction with Hara–Radin method) and all the other extraction methods. No statistically significant differences were observed among the six green extraction methods (One-Way ANOVA analysis F (5,6) = 3.720, p = 0.070).
All the crude oils demonstrated superior oxidative stability compared to values reported by Šimat et al. for oils obtained through industrial tricanter-type decanter centrifuge.65 Both primary and secondary oxidation indicators were consistently lower, resulting in a more favorable TOTOX index. These findings suggest that the applied extraction method may better preserve the oxidative integrity of the oil, despite its less industrialized nature. The only parameter in which the tricanter-derived oil showed an advantage was free fatty acid content (FFA), with a reported value of 2.0%. This difference is likely due to the post-extraction drying step commonly implemented in industrial processes. Drying reduces residual moisture, thereby limiting hydrolytic degradation of triglycerides and lowering FFA levels.
3.4 Fatty acids profiles
The fatty acid compositions of the extracted oils are presented in Fig. 8 and details are reported in SI (Table S1). Statistical differences among extraction methods were evaluated by One-Way ANOVA followed by Tukey's honestly significant difference (HSD) post hoc test (p < 0.05). As reported in Table S1, values within the same row followed by different superscript letters indicate significant differences among methods, whereas values sharing at least one common letter are not significantly different. Overall, the extraction method significantly affected the fatty acid profile of the samples. In particular, the use of S/L green solvents generally resulted in higher proportions of several saturated and monounsaturated fatty acids (e.g., C14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0, C16
0, C16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0, C18
0, C18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0, C18
0, C18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), as indicated by the exclusive assignment of the letter “a” compared with the reference and other extraction approaches. Conversely, the reference method and some alternative techniques (MAE and EAE) often exhibited intermediate values, sharing statistical groupings with treatments showing either higher or lower results, thus indicating partial overlap and the absence of statistically significant differences.
1), as indicated by the exclusive assignment of the letter “a” compared with the reference and other extraction approaches. Conversely, the reference method and some alternative techniques (MAE and EAE) often exhibited intermediate values, sharing statistical groupings with treatments showing either higher or lower results, thus indicating partial overlap and the absence of statistically significant differences.
For polyunsaturated fatty acids, including the omega-3 and omega-6 series, significant differences among extraction methods were also observed. In particular, the reference method (ref) and aqueous extraction (c_3) consistently showed higher contents of long-chain omega-3 fatty acids, such as EPA (C20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) and DHA (C22
5) and DHA (C22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6), as indicated by shared superscript letters and significantly higher values for these compounds. Despite these differences, the overall fatty acid profiles remained largely comparable among samples, as confirmed by PCA analysis (Section 4.1). Consistently, the ω6/ω3 ratio was significantly lower in oils obtained by the reference and aqueous extraction methods, suggesting a more favourable nutritional profile.
6), as indicated by shared superscript letters and significantly higher values for these compounds. Despite these differences, the overall fatty acid profiles remained largely comparable among samples, as confirmed by PCA analysis (Section 4.1). Consistently, the ω6/ω3 ratio was significantly lower in oils obtained by the reference and aqueous extraction methods, suggesting a more favourable nutritional profile.
Oleic acid (C18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, omega-9) was the predominant fatty acid in all samples, ranging from 39.1% to 43.2%, reflecting the typical monounsaturated fatty acids (MUFAs)-rich nature of fish oil. Linoleic acid (C18
1, omega-9) was the predominant fatty acid in all samples, ranging from 39.1% to 43.2%, reflecting the typical monounsaturated fatty acids (MUFAs)-rich nature of fish oil. Linoleic acid (C18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, omega-6) was consistently present across all extracts, with the highest levels observed in the reference method (ref) (18.7%), followed closely by the EAE (c_2) (18.5%) and aqueous (c_3) (18.4%) samples. DHA (C22
2, omega-6) was consistently present across all extracts, with the highest levels observed in the reference method (ref) (18.7%), followed closely by the EAE (c_2) (18.5%) and aqueous (c_3) (18.4%) samples. DHA (C22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6) showed the highest recovery in the aqueous extract (c_3), (4.7%), followed closely by the reference method (ref) (4.6%), indicating that both water-based and conventional solvent-based extractions are effective in preserving this highly unsaturated fatty acid, with no significant difference between the two methods. Intermediate DHA levels were observed in c_1 (EAE, 3.5%), MAE lit. (b_1) (3.2%), and MAE opt. (b_2) (3.0%). EPA (C20
6) showed the highest recovery in the aqueous extract (c_3), (4.7%), followed closely by the reference method (ref) (4.6%), indicating that both water-based and conventional solvent-based extractions are effective in preserving this highly unsaturated fatty acid, with no significant difference between the two methods. Intermediate DHA levels were observed in c_1 (EAE, 3.5%), MAE lit. (b_1) (3.2%), and MAE opt. (b_2) (3.0%). EPA (C20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) followed a similar trend, with the highest concentrations detected in the reference (ref) (2.3%) and aqueous (c_3) (2.3%) extracts. Lower EPA levels were observed in the green solvent extract (a) (1.2%), and modest recovery was noted in the MAE and EAE samples (ranging from 1.6% to 1.8%). The fatty acid profiles obtained from different extraction methods revealed that aqueous extraction (c_3) and the reference method (ref) yielded the highest concentrations of PUFAs, especially EPA and DHA, while green solvent-based methods (particularly a and b_2) showed enrichment in MUFAs, especially oleic acid (C18
5) followed a similar trend, with the highest concentrations detected in the reference (ref) (2.3%) and aqueous (c_3) (2.3%) extracts. Lower EPA levels were observed in the green solvent extract (a) (1.2%), and modest recovery was noted in the MAE and EAE samples (ranging from 1.6% to 1.8%). The fatty acid profiles obtained from different extraction methods revealed that aqueous extraction (c_3) and the reference method (ref) yielded the highest concentrations of PUFAs, especially EPA and DHA, while green solvent-based methods (particularly a and b_2) showed enrichment in MUFAs, especially oleic acid (C18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1).
1).
This trend can be explained by the partitioning behavior of lipids during extraction, influenced by both solvent polarity and lipid localization in the tissue matrix. PUFA-rich lipids, such as phospholipids and free fatty acids, are often associated with membrane structures and may be more accessible in aqueous environments, especially when aided by enzymatic hydrolysis (c_1, c_2) or simple water extraction (c_3). These methods promote the release of membrane–bound lipids without disrupting their structure, and the mild conditions (60 °C, no organic solvents) help preserve thermolabile PUFAs.
In contrast, green solvents like ethyl acetate and ethanol, particularly in mixtures with higher ethyl acetate content (a and b_2), preferentially extract neutral lipids such as triglycerides, which are typically richer in MUFAs. Ethyl acetate's lower polarity enhances solubilization of non-polar compounds, while ethanol contributes to intermediate polarity, allowing partial extraction of more polar lipids. However, the overall solvent system remains less effective at extracting highly unsaturated, polar lipids like DHA and EPA.
Additionally, MAE may enhance lipid release through rapid heating and cell disruption, but the short exposure time and solvent composition still favor the extraction of neutral lipids.
These observations suggest that aqueous and enzymatic methods are more suitable for PUFAs recovery, especially when targeting omega-3 fatty acids for nutraceutical applications, while green solvent-based methods may be more appropriate for MUFAs-rich oils.
In general, the comparative evaluation of Fatty Acid Methyl Ester (FAME) profiles revealed a largely similar lipid composition among the oils extracted using different techniques, as can be observed in Fig. 9 where the fatty acids have been grouped into macro-classes.
 | ||
| Fig. 9 Saturated, monounsaturated, and polyunsaturated FAMEs composition of oils extracted using different techniques. The Hara–Radin lipid extraction method was used as the reference method (ref).30 | ||
Total saturated fatty acids (SFA) reached their highest concentration in the sample extracted with S/L green solvents (a), accounting for 24.94% of the total lipid profile. Similarly, this extraction method yielded the highest proportion of MUFAs at 51.45%. In contrast, the highest PUFAs content was observed in the aqueous extract (c_3), with 32.87%, indicating a potentially more favorable composition for nutritional and functional applications.
The complexity and heterogeneity of the starting material used in this study pose challenges for establishing direct comparisons with previously published data. The lipids analyzed were extracted from a composite mixture of aquaculture side streams, specifically filleting by-products of both sea bream and sea bass, which include various tissue types and anatomical regions. This complexity contrasts with more defined matrices such as fish heads, livers, guts or skin.66
The fatty acids profiles obtained from these samples revealed SFA ranging from 21% to 25%, MUFA between 45% and 51%, and PUFA spanning 24% to 33%. These lipids were extracted using environmentally sustainable techniques, including MAE, EAE, and the use of green solvents. In contrast, literature data on sea bream and sea bass heads extracted via Soxhlet and microwave methods using hexane report SFA contents between 20% and 21%, MUFA between 41% and 45%, and PUFA between 35% and 38%.67 Although the SFA levels are broadly comparable, the MUFA content in the present samples is notably higher. This difference may reflect both the composition of the raw material and the influence of the extraction methodology. The use of green techniques appears to favor the recovery of MUFAs, suggesting a selective affinity or preservation effect associated with these methods. These findings highlight the relevance of matrix complexity and extraction strategy in determining the final fatty acid profile. Similar results in terms of SFA, MUFA, and PUFA composition have been reported by Korkmaz et al., who investigated oils extracted from rainbow trout by-products using EAE.68
Many studies focus on the fatty acid profiles of fish fillets, particularly for species such as sea bream and sea bass, which are widely consumed. These studies often emphasize the influence of dietary regimes and production systems, providing detailed comparisons between farmed and wild specimens.69–72 In contrast, data on lipid profiles from side streams remain relatively limited, underscoring the relevance of the present findings in expanding the scope of valorization strategies for aquaculture by-products.
In recent decades, the dietary ratio of omega-6/omega-3 in Western populations has shifted dramatically from an estimated 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 in ancestral diets to values exceeding 15
1 in ancestral diets to values exceeding 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.73 Such disproportionate intake has been associated with an increased prevalence of chronic inflammatory conditions, including cardiovascular disease, metabolic syndrome, and autoimmune disorders.74 Emerging evidence underscores the health-promoting potential of omega-3 PUFAs, particularly EPA and DHA, which exhibit anti-inflammatory, cardioprotective, and neurodevelopmental benefits.75 Consequently, optimizing the omega-6/omega-3 ratio through dietary interventions or supplementation is considered a key strategy in improving public health outcomes.
1.73 Such disproportionate intake has been associated with an increased prevalence of chronic inflammatory conditions, including cardiovascular disease, metabolic syndrome, and autoimmune disorders.74 Emerging evidence underscores the health-promoting potential of omega-3 PUFAs, particularly EPA and DHA, which exhibit anti-inflammatory, cardioprotective, and neurodevelopmental benefits.75 Consequently, optimizing the omega-6/omega-3 ratio through dietary interventions or supplementation is considered a key strategy in improving public health outcomes.
Studies suggest that an omega-6/omega-3 ratio below 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and ideally around 2–3
1, and ideally around 2–3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 is generally regarded as nutritionally advantageous, as they reflect a higher proportion of anti-inflammatory omega-3 fatty acids relative to pro-inflammatory omega-6 species.76,77 All the extraction methods yielded highly favorable lipid profile in terms of omega-6/omega-3 balance, suggesting their potential for producing fish oil with enhanced health-promoting properties.
1 is generally regarded as nutritionally advantageous, as they reflect a higher proportion of anti-inflammatory omega-3 fatty acids relative to pro-inflammatory omega-6 species.76,77 All the extraction methods yielded highly favorable lipid profile in terms of omega-6/omega-3 balance, suggesting their potential for producing fish oil with enhanced health-promoting properties.
| SFA | MUFA | PUFA | EPA | DHA | TOT_OMEGA_6 | TOT_OMEGA_3 | OMEGA_6/OMEGA_3 | Recovery | Acidity | TOTOX | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| ref | 21.48 | 46.5 | 32.47 | 2.3 | 1.1 | 20.01 | 11.6 | 1.73 | 100 | 2.8 | 14.82 |
| a | 24.94 | 51.45 | 24.09 | 1.2 | 0.59 | 16.83 | 6.39 | 2.63 | 81.6 | 3.2 | 5.08 |
| b_1 | 23.46 | 48.19 | 28.74 | 1.7 | 0.85 | 19.19 | 8.85 | 2.17 | 82.7 | 3.4 | 4.77 |
| b_2 | 23.41 | 49.78 | 27.13 | 1.6 | 0.77 | 18.31 | 8.17 | 2.24 | 99.3 | 3.2 | 5.86 |
| c_1 | 23 | 49.01 | 28.4 | 1.8 | 0.88 | 18.28 | 9.18 | 1.99 | 77.4 | 3.3 | 6.68 |
| c_2 | 23.7 | 45.43 | 27.75 | 1.8 | 0.5 | 19.66 | 7.5 | 2.62 | 80.7 | 3.4 | 5.17 |
| c_3 | 21.06 | 46.45 | 32.87 | 2.3 | 1.1 | 19.67 | 11.6 | 1.70 | 83.6 | 3.4 | 2.29 |
Fig. 10 presents the PCA results performed on the autoscaled data matrix D7,11, highlighting the biplot (score and loading plots) of the first two principal components, which together explain 86.9% of the total variance.
The biplot (Fig. 10) and the analysis of the loading plots (Fig. 11) reveal that the first principal component is primarily associated with variables related to the fatty acid profile, while the second component is mainly influenced by variables concerning both the recovery and the quality of the lipid fraction (acidity and TOTOX).
Analysis of the PCA scores indicates that the reference method (ref) and the water-based extraction (c_3) are closely positioned along PC1, reflecting a comparable fatty acid profile, with slightly higher PUFA and ω-3 contents. In contrast, the conventional solid/liquid cold extraction using ethyl acetate:ethanol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) (a_S/L) is clearly separated along PC1, showing higher SFA and MUFA levels and a concomitant reduction in PUFA content.
1, v/v) (a_S/L) is clearly separated along PC1, showing higher SFA and MUFA levels and a concomitant reduction in PUFA content.
Green organic solvent-based extractions, including conventional and microwave-assisted methods, tend to preferentially solubilize neutral storage lipids, resulting in higher MUFA and SFA contents. Enzymatic-assisted extractions cluster with microwave-assisted methods, suggesting that enzymatic hydrolysis primarily enhances lipid release through matrix degradation rather than significantly altering lipid selectivity. This indicates that the extraction mechanism and solvent–matrix interactions, rather than the mere presence of water, are the main factors driving the observed differences in fatty acid profiles. Microwave-assisted extractions performed with ethyl acetate![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol at ratios of 2
ethanol at ratios of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) (b_1) and 3
1 (v/v) (b_1) and 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) (b_2), together with enzymatic-assisted extractions (c_1 and c_2), display a similar trend along PC1, confirming their comparable fatty acid compositions. The enzymatic methods cluster closely and show a fatty acid profile resembling that of b_1, while offering the additional advantage of protein hydrolysate recovery, making them particularly attractive for process upscaling. Between c_1 and c_2, method c_2 was selected due to its higher extraction yield.
1 (v/v) (b_2), together with enzymatic-assisted extractions (c_1 and c_2), display a similar trend along PC1, confirming their comparable fatty acid compositions. The enzymatic methods cluster closely and show a fatty acid profile resembling that of b_1, while offering the additional advantage of protein hydrolysate recovery, making them particularly attractive for process upscaling. Between c_1 and c_2, method c_2 was selected due to its higher extraction yield.
Although ref and c_3 exhibit similar behavior along PC1, they are clearly separated along PC2. This separation reflects differences in technological and quality-related parameters: c_3 shows lower recovery and TOTOX values but higher acidity, likely due to prolonged contact with the aqueous phase, which may promote hydrolytic reactions and increase free fatty acid formation.
Overall, PC1 is mainly associated with fatty acid composition (PUFA, MUFA, and SFA), while PC2 is driven by recovery and oxidative stability indicators (TOTOX and acidity). PCA therefore confirms that fatty acid composition accounts for most of the variance among extraction methods, while technological factors related to oil quality and process efficiency further contribute to sample discrimination. Nevertheless, differences in fatty acid composition among the various extraction methods are subtle but consistent. PCA highlights these subtle variations by projecting the autoscaled data (mean-centered and scaled to unit variance) into a reduced-dimensional space.
3.5 Upscaling of EAE
EAE emerged as the most suitable method for the industrial upscaling of fishery side-streams. Its mild operating conditions, namely moderate temperature and neutral Ph, reduce energy consumption and simplify equipment requirements.78,79 Moreover, the use of enzymatic reactions in aqueous media, already well-established in industrial biotechnology applications, significantly minimizes environmental impact, while maintaining high extraction yields and selectivity.80 The ability to recover valuable co-products, such as proteins and hydrolysed peptides, further enhances the overall economic viability of the process, making EAE the ideal choice for large-scale extraction aligned with environmental sustainability and industrial efficiency goals. For these reasons, a pilot scale EAE experiment was performed in a 30 L reactor, using 8 kg DFM and 16 L water. The enzymatic hydrolysis of DFM (schematized in Fig. 12) and the following separation yielded 10.5 L of protein hydrolysate. The subsequent concentration step reduced the volume to 3.2 L. The overnight storage at 4 °C facilitated phase separation, revealing the presence of an emulsion layer, which was effectively removed. This step proved critical for improving the purity of the hydrolysate and recovering the lipid fraction. The oil recovery process yielded 2 L of oil (25% yield), which was stabilized with tocopherol to prevent oxidative degradation. Lyophilization of the hydrolysate yielded 1.324 g dried hydrolysate, representing a yield of 16.5%. The remaining solid residue amounted to 4.67 kg wet weight, with a dry matter content of 48.2%, representing the non-solubilized fraction. Calculated based on dry matter, the remaining solid phase amounts to approximately 28% of the initial biomass input. The total yield of oil, hydrolysate, and solid phase equals approximately 70% of the starting material. The remaining approximately 30% represents the substantial emulsion phase, which was removed, and minor losses during handling, transfer, and processing. In the current study, the oil phase is the focus. In previous studies,60,81 it was shown that EAE of this biomass yielded protein hydrolysates with high protein contents (>80%), and with interesting bioactivities and functionalities.Regarding the quality of the extracted oil, preliminary analyses revealed good oxidative stability, as indicated by a TOTOX index of 15.2 ± 0.6. However, the acidity value (8.8 ± 0.9) was higher than those typically observed at laboratory scale. To address this issue, several strategies are currently under investigation, including the integration of an oil drying system following separation. This solution is planned to be implemented in the pilot-scale system currently being designed within the EcoeFISHent project. In an industrial setting, long incubation steps would usually be avoided, as the process progresses continuously, without the long incubation steps, such as the overnight storage performed in this pilot experiment. This could positively affect the oil quality and the economic sustainability of the process.
4 Conclusions
This study highlights the feasibility of sustainably valorizing fishery side-streams through the recovery of oxidatively stable, health-promoting fish oils. By addressing the environmental impact of increasing fish production and offering scalable extraction solutions, it contributes to the broader scientific discourse on circular bioeconomy, sustainable food systems, and the upcycling of agri-food by-products. The approach exemplifies how circular economy principles can be effectively applied to the seafood sector, transforming waste into valuable resources and promoting long-term environmental and economic sustainability. Indeed, the significant global increase in fish production, driven by both aquaculture and wild capture fisheries, has led to a parallel rise in the generation of fishery side streams. This trend poses considerable economic and environmental challenges, underscoring the urgent need for sustainable valorization strategies for these underutilized by-products.Within the Horizon2020 EcoeFISHent framework, efforts have been directed toward fostering a sustainable blue circular economy by developing innovative value chains for the extraction and application of bioactive and functional compounds from fishery side streams. A key innovation lies in the valorization of unsorted and non-separated residues, thus avoiding preliminary sorting, namely the separation of different by-products (e.g., skin, bones, tails…), which often proves economically unsustainable for small and medium-sized enterprises and is typically excluded from conventional processing workflows.
Side streams generated during the filleting of sea bream and sea bass were recovered in an unsorted form, milled, and dehydrated using a patented mild processing technique. This process yielded a concentrated biomass with high oil content (∼40%), minimal oxidative deterioration, and low residual moisture (1–3%), ensuring extended shelf life.
The upcycling of this biomass focused on crude oil extraction using three environmentally friendly methods at laboratory scale: green solvent extraction, microwave-assisted extraction (MAE), and enzyme-assisted extraction (EAE). All methods yielded satisfactory oil recovery (33–42%) and low oxidation levels. Comparative analysis of FAME profiles revealed consistent lipid composition across the different techniques, with all oils exhibiting a beneficial omega-6/omega-3 ratio, supporting their potential for nutraceutical applications.
Among the tested methods, EAE emerged as the most promising for industrial upscaling of fishery side-streams, offering a cost-effective and sustainable alternative to other advanced techniques like microwave-assisted extraction (MAE). Its operation under mild conditions, namely moderate temperature and neutral pH, significantly reduces energy consumption and simplifies equipment requirements. From an engineering standpoint, EAE is highly scalable, leveraging well-established aqueous enzymatic processes commonly used in industrial biotechnology. The use of eco-friendly solvents, such as water, further lowers environmental impact and operational costs. Additionally, the ability to recover valuable co-products like proteins and hydrolyzed peptides enhances the overall economic viability of the process. These combined benefits make EAE the ideal choice for large-scale extraction, aligning both environmental sustainability and industrial efficiency goals.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included within the article and the supplementary information (SI). Supplementary information on the fatty acids profiles (%) and ratio omega-6/omega-3 (ω6/ω3) of oils, is available. See DOI: https://doi.org/10.1039/d5fb00589b.Acknowledgements
This work originates from the EcoeFISHent project (“Demonstrable and replicable cluster implementing systemic solutions through multilevel circular value chains for eco-efficient valorisation of fishing and fish industry side-streams”), funded under the Horizon 2020 Program – Green Deal (Innovation Action, Grant Agreement ID: 101036428). We gratefully acknowledge AQUA Società Agricola Srl, (@Aqua de Mâ) for providing the side streams, and Themis S.p.A. for carrying out the preliminary stabilization treatment of these biomasses. We gratefully acknowledge Micamo Lab Srl, ARPAL Liguria, and ANFACO-CECOPESCA for their support in assessing the compliance of both the biomasses and the extracted products through comprehensive microbiological and chemical contaminant analyses. We also thank Fkv Srl and Milestone Srl for the technical support provided in optimizing the Microwave-Assisted Extraction processes.References
- I. S. Arvanitoyannis and A. Kassaveti, Int. J. Food Sci. Technol., 2008, 43, 726–745 CrossRef CAS.
- D. Zeller, T. Cashion, M. Palomares and D. Pauly, Fish Fish., 2018, 19, 30–39 Search PubMed.
- M. Carneiro and R. Martins, in Life below Water, W. Leal Filho, A. M. Azul, L. Brandli, A. Lange Salvia and T. Wall, Springer International Publishing, Cham, 2022, pp. 295–304 Search PubMed.
- European Commission Joint Research Centre, Blue Growth and Smart Specialisation: How to Catch Maritime Growth through ‘Value Nets’., Publications Office, LU, 2016 Search PubMed.
- European Union, Regulation (EU) No 1380/2013 of the European Parliament and of the Council of 11 December 2013 on the Common Fisheries Policy, 2013 Search PubMed.
- The State of World Fisheries and Aquaculture 2024, FAO, 2024, https://openknowledge.fao.org/handle/20.500.14283/cd0683en Search PubMed.
- European Commission, EcoeFISHent Project, 2021 Search PubMed.
- A.-L. Välimaa, S. Mäkinen, P. Mattila, P. Marnila, A. Pihlanto, M. Mäki and J. Hiidenhovi, Food Qual. Saf., 2019, 3, 209–226 CrossRef.
- R. Song, W. Li, S. Deng, Y. Zhao and N. Tao, Front. Nutr., 2023, 10, 1136490 CrossRef PubMed.
- M. Pateiro, R. Domínguez, T. Varzakas, P. E. S. Munekata, E. Movilla Fierro and J. M. Lorenzo, Mar. Drugs, 2021, 19, 233 Search PubMed.
- S. C. Dyall, Front. Aging Neurosci., 2015, 7, 52 Search PubMed.
- S. C. Cottin, T. A. Sanders and W. L. Hall, Proc. Nutr. Soc., 2011, 70, 215–231 CrossRef CAS PubMed.
- H. A. Hamilton, R. Newton, N. A. Auchterlonie and D. B. Müller, Nat Food, 2020, 1, 59–62 Search PubMed.
- A. Sasidharan, T. Rustad and G. M. Cusimano, Environ. Sci. Pollut. Res., 2024, 31, 62230–62248 CrossRef PubMed.
- S. Naseem, A. Imam, A. S. Rayadurga, A. Ray and S. K. Suman, Crit. Rev. Food Sci. Nutr., 2024, 64, 9240–9260 Search PubMed.
- M. Usman, M. Nakagawa and S. Cheng, Processes, 2023, 11, 3444 Search PubMed.
- S. Tzima, I. Georgiopoulou, V. Louli and K. Magoulas, Molecules, 2023, 28, 1410 CrossRef CAS PubMed.
- O. Ciftci and H. Keskin Cavdar, Eur. J. Lipid Sci. Technol., 2025, 127, e202400131 Search PubMed.
- K. K. A. Sanjeewa, K. H. I. N. M. Herath, Y.-S. Kim, Y.-J. Jeon and S.-K. Kim, TrAC, Trends Anal. Chem., 2023, 167, 117266 CrossRef CAS.
- S. N. A. Papadopoulou, T. Adamantidi, D. Kranas, P. Cholidis, C. Anastasiadou and A. Tsoupras, Mar. Drugs, 2025, 23, 299 CrossRef CAS PubMed.
- K. Bouizgma, N. Rabbah, Z. Abbas and A. Abourriche, Sep. Sci. Technol., 2025, 60, 657–683 Search PubMed.
- V. Orlandi, L. Dondero, F. Turrini, G. De Negri Atanasio, F. Grasso, E. Grasselli and R. Boggia, Molecules, 2023, 28, 7637 CrossRef CAS PubMed.
- M. Romanò, Method for Transforming Waste and System for Performing Said Method, WO2015181769A1, 2015, available online: https://patents.google.com/patent/WO2015181769A1 Search PubMed.
- J. Butler, G. G. Jayson and A. J. Swallow, Biochim. Biophys. Acta, 1975, 408, 215–222 CrossRef CAS PubMed.
- F. Grasso, D. Méndez-Paz, R. Vázquez Sobrado, V. Orlandi, F. Turrini, G. De Negri Atanasio, E. Grasselli, M. Tiso and R. Boggia, Gels, 2023, 9, 760 CrossRef CAS PubMed.
- F. Grasso, D. Méndez Paz, R. Vázquez Sobrado, V. Orlandi, F. Turrini, L. Agostinis, A. Morandini, M. Jenssen, K. Lian and R. Boggia, Gels, 2024, 10, 246 CrossRef CAS PubMed.
- K. Lian, I. Maribu, T. M. Rode, M. Jenssen, B. Vang and R. G. Solstad, Front. Sustain. Food Syst., 2024, 8, 1346548 Search PubMed.
- COROLASE® 8000 product information, https://www.abenzymes.com/en/product-repository-food/corolase-8000/, accessed 7 November 2025 Search PubMed.
- AOAC International, Official Methods of Analysis of AOAC International, Association of Official Analytical Chemists, Gaithersburg, MD, USA, 19th edn, 2012 Search PubMed.
- A. Hara and N. S. Radin, Anal. Biochem., 1978, 90, 420–426 CrossRef CAS PubMed.
- Z. Hu and Q. Zhong, Food Chem., 2010, 123, 794–799 CrossRef CAS.
- R. Leardi, C. Melzi and G. Polotti, CAT (Chemometric Agile Tool), freely downloadable from http://gruppochemiometria.it/index.php/software Search PubMed.
- C. M. Alder, J. D. Hayler, R. K. Henderson, A. M. Redman, L. Shukla, L. E. Shuster and H. F. Sneddon, Green Chem., 2016, 18, 3879–3890 RSC.
- D. D. S. V. Costa and N. Bragagnolo, Eur. J. Lipid Sci. Technol., 2017, 119, 1600108 Search PubMed.
- CDR FoodLab®, Validations of the CDR FoodLab® testing equipment, https://www.cdrfoodlab.com/cdrfoodlab/validations, accessed 21 October 2025 Search PubMed.
- C. W. Kwon, K.-M. Park, J. W. Park, J. Lee, S. J. Choi and P.-S. Chang, J. Chem., 2016, 2016, 1–6 CrossRef.
- S. Koohikamali and M. S. Alam, J. Food Sci. Technol., 2019, 56, 5063–5073 Search PubMed.
- International Organization for Standardization, ISO 12966-2:2017 — Animal and Vegetable Fats and Oils — Gas Chromatography of Fatty Acid Methyl Esters — Part 2: Preparation of Methyl Esters of Fatty Acids, Geneva, Switzerland, 2017 Search PubMed.
- International Organization for Standardization, ISO 12966-4:2015 — Animal and Vegetable Fats and Oils — Gas Chromatography of Fatty Acid Methyl Esters — Part 4: Determination by Capillary Gas Chromatography, Geneva, Switzerland, 2015 Search PubMed.
- J. M. Joy, A. Padmaprakashan, A. Pradeep, P. T. Paul, R. J. Mannuthy and S. Mathew, Foods, 2024, 13, 2793 CrossRef CAS PubMed.
- C. He, J. Cao, Y. Bao, Z. Sun, Z. Liu and C. Li, Food Chem., 2021, 347, 129057 Search PubMed.
- F. Grasso, M. M. A. Martínez, F. Turrini, D. Méndez Paz, R. Vázquez Sobrado, V. Orlandi, M. Jenssen, K. Lian, J. Rombi, M. Tiso, E. Razzuoli, C. Costas and R. Boggia, Antioxidants, 2024, 13, 1011 CrossRef CAS PubMed.
- P. Manirakiza, A. Covaci and P. Schepens, J. Food Compos. Anal., 2001, 14, 93–100 CrossRef CAS.
- J. Folch, I. Ascoli, M. Lees, J. A. Meath and F. N. LeBaron, J. Biol. Chem., 1951, 191, 833–841 Search PubMed.
- J. Folch, M. Lees and G. H. S. Stanley, J. Biol. Chem., 1957, 226, 497–509 CrossRef CAS PubMed.
- E. G. Bligh and W. J. Dyer, Can. J. Biochem. Physiol., 1959, 37, 911–917 CrossRef CAS PubMed.
- F. Smedes, Analyst, 1999, 124, 1711–1718 RSC.
- M. J. Zarrinmehr, E. Daneshvar, S. Nigam, K. P. Gopinath, J. K. Biswas, E. E. Kwon, H. Wang, O. Farhadian and A. Bhatnagar, Bioresour. Technol., 2022, 344, 126303 CrossRef CAS PubMed.
- C. Breil, M. Abert Vian, T. Zemb, W. Kunz and F. Chemat, IJMS, 2017, 18, 708 Search PubMed.
- M. J. Ramalhosa, P. Paíga, S. Morais, M. Rui Alves, C. Delerue-Matos and M. B. P. P. Oliveira, Food Chem., 2012, 131, 328–336 CrossRef CAS.
- Hielscher Ultrasonics GmbH, Soxhlet Extraction Guide for Academic and Professional Domains, https://www.hielscher.com/soxhlet Search PubMed.
- R. Leardi, Anal. Chim. Acta, 2009, 652, 161–172 Search PubMed.
- V. Orlandi, Formulation of a new nutraceutical ingredient using green technologies, PhD thesis, Università di Genova, 2025.
- D. E. S. Souza, J. J. C. D. Melo, F. F. D. Santos, A. L. D. S. Vasconcelos, A. D. S. D. Jesus, L. D. S. Freitas, R. L. D. Souza and C. M. F. Soares, Foods, 2024, 13, 3141 CrossRef CAS PubMed.
- Z. Yuan, X. Ye, Z. Hou and S. Chen, Trends Food Sci. Technol., 2024, 143, 104276 CrossRef CAS.
- S. Ghalamara, C. Brazinha, S. Silva and M. Pintado, Curr. Food Sci. Tech. Rep., 2024, 2, 377–391 Search PubMed.
- V. Venugopal, Curr. Res. Environ. Sustain., 2022, 4, 100164 Search PubMed.
- A. Łubek-Nguyen, W. Ziemichód and M. Olech, Appl. Sci., 2022, 12, 3232 Search PubMed.
- X. Fang, X. Fei, H. Sun and Y. Jin, Eur. J. Lipid Sci. Technol., 2016, 118, 244–251 Search PubMed.
- M. Jenssen, J. Matic, I. Sone, N. T. Solberg, S. Wubshet and K. Lian, Front. Mar. Sci., 2025, 12, 1572015 Search PubMed.
- M. Jenssen, I. Sone, F. Grasso, F. Turrini, F. Tardanico, G. D. N. Atanasio, D. M. Paz, R. V. Sobrado, M. A. Martínez, R. Boggia, E. Grasselli and K. Lian, Front. Nutr., 2025, 12, 1663294 CrossRef PubMed.
- Codex Alimentarius Commission, Food and Agriculture Organization of the United Nations, Codex Standard for Fish Oils (CXS 329-2017), 2017 Search PubMed.
- International Fish Oil Standards (IFOS) and IFFO, Codex Standard for Fish Oils, 2017 Search PubMed.
- Council of Europe, European Pharmacopoeia, 11th ed.; Fish Oil Monograph, Council of Europe, Strasbourg, France, 11th edn, 2025 Search PubMed.
- V. Šimat, J. Vlahović, B. Soldo, I. Generalić Mekinić, M. Čagalj, I. Hamed and D. Skroza, Food Biosci., 2020, 33, 100484 CrossRef.
- P. E. S. Munekata, M. Pateiro, R. Domínguez, J. Zhou, F. J. Barba and J. M. Lorenzo, Biomolecules, 2020, 10, 232 CrossRef CAS PubMed.
- B. De La Fuente, J. Pinela, R. C. Calhelha, S. A. Heleno, I. C. F. R. Ferreira, F. J. Barba, H. Berrada, C. Caleja and L. Barros, Food Bioprod. Process., 2022, 136, 97–105 CrossRef CAS.
- K. Korkmaz and S. Öztürk, Foods, 2025, 14, 3227 CrossRef CAS PubMed.
- E. Orban, T. Nevigato, G. D. Lena, I. Casini and A. Marzetti, J. Food Sci., 2003, 68, 128–132 CrossRef CAS.
- A. C. R. Parpoura and M. N. Alexis, Aquac. Int., 2001, 9, 463–476 CrossRef CAS.
- M. Yildiz, E. Şener and M. Timur, Int. J. Food Sci. Technol., 2008, 43, 853–858 CrossRef CAS.
- T. Šegvić-Bubić, S. Zrnčić, E. Tibaldi, J. Pleadin, D. Oraić, I. Balenović, T. Lešić, N. Kudumija, I. Cvitić, I. G. Zupičić, I. Lepen Pleić, G. Cardinaletti and A. Vulić, Future Foods, 2025, 12, 100747 CrossRef.
- M. Sala-Climent, E. Lal, F. Cedola, M. Alharthi, M. Fernandez-Bustamante, M. Agustin-Perez, A. Singh, S.-I. Choi, T. Rivera, K. Nguyen, S. Lee, S. Golshan, T. Holt, O. Quehenberger, R. Coras and M. Guma, Clin. Nutr. ESPEN, 2025, 68, 389–402 Search PubMed.
- I. J. Malesza, M. Malesza, J. Walkowiak, N. Mussin, D. Walkowiak, R. Aringazina, J. Bartkowiak-Wieczorek and E. Mądry, Cells, 2021, 10, 3164 CrossRef CAS PubMed.
- M. Mititelu, D. Lupuliasa, S. M. Neacşu, G. Olteanu, Ș. S. Busnatu, A. Mihai, V. Popovici, N. Măru, S. C. Boroghină, S. Mihai, C.-B. Ioniță-Mîndrican and A. Scafa-Udrişte, Foods, 2024, 14, 46 Search PubMed.
- A. P. Simopoulos, Biomed. Pharmacother., 2002, 56, 365–379 Search PubMed.
- J. J. DiNicolantonio and J. O’ Keefe, Mo. Med., 2021, 453–459 Search PubMed.
- E. Quitério, C. Grosso, R. Ferraz, C. Delerue-Matos and C. Soares, Mar. Drugs, 2022, 20, 677 CrossRef PubMed.
- S. J. Marathe, S. B. Jadhav, S. B. Bankar and R. S. Singhal, in Food Bioactives, M. Puri, Springer International Publishing, Cham, 2017, pp. 171–201 Search PubMed.
- J. A. Herrera Barragán, G. Olivieri, I. Boboescu, M. Eppink, R. Wijffels and A. Kazbar, Front. Mar. Sci., 2022, 9, 948086 CrossRef.
- M. Jenssen, I. Sone, F. Grasso, F. Turrini, F. Tardanico, G. D. N. Atanasio, D. M. Paz, R. V. Sobrado, M. A. Martínez, R. Boggia, E. Grasselli and K. Lian, Front. Nutr., 2025, 12, 1663294 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |








