 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
The impact of faecal sludge-derived biochar as an additive on anaerobic degradation of synthetic human excreta
Mengru Qiu *,
Maria E. Koulouri
*,
Maria E. Koulouri ,
Laure Sioné and
Michael R. Templeton
,
Laure Sioné and
Michael R. Templeton
Department of Civil and Environmental Engineering, Imperial College London, London, SW7 2AZ, UK. E-mail: mengru.qiu21@imperial.ac.uk
First published on 14th April 2026
Abstract
Pit latrine filling poses a major challenge in developing countries. This study investigated the effect of faecal sludge-derived biochar (FSB) on anaerobic degradation, the dominant process in pit latrines, using synthetic human excreta under laboratory conditions simulating wet pit environments. Results show that FSB addition significantly increased hydrolysis rate and shortened lag phase by up to 1.5 days. However, this enhancement did not translate into improved overall degradation, as volatile solids (VS) reduction decreased under FSB addition, while only minor changes were observed in methane and total gas production. This decoupling is attributed to redistribution of organic carbon from gaseous products toward microbial biomass and extracellular matrices, as reflected by elevated extracellular polymeric substance (EPS) carbohydrate concentrations. Microbial community analysis revealed a targeted restructuring toward biofilm-associated hydrolytic and methanogenic populations under FSB amendment, characterised by enrichment of metabolically versatile methanogens (Methanosarcina) and proteolytic fermenters. Overall, these findings indicate that FSB promotes microbial aggregation and stabilised anaerobic processes but does not enhance net solids degradation, and therefore the findings do not support the recommendation of FSB as a pit latrine additive for reducing pit filling.
Water impactPit filling rate continues posing challenges in developing countries. This study provides evidence that faecal sludge-derived biochar accelerated anaerobic degradation without enhancing overall solids breakdown. These findings conclude that faecal sludge-derived biochar may not necessarily reduce pit filling rates and highlight the importance of evaluating different types of biochar as pit additives. |
1. Introduction
Pit latrines are sanitation technology used by over 1.8 billion people worldwide and are important to eliminate open defecation, which is still practiced by more than 400 million people globally.1,2 As countries work towards the Sustainable Development Goal of access to adequate and equitable sanitation for all by 2030, reliance on pit latrines is expected to increase.3 However, the long-term effectiveness of pit latrines is fundamentally limited by pit filling, which is often associated with unsafe emptying practices, increases household costs, and undermines their long-term public health benefits.4,5The persistence of solids within pit latrines reflects the inherently slow degradation of faecal organic matter in situ. Although pit environments are largely anaerobic, the conversion of complex particulate substrates into soluble intermediates is inefficient, with hydrolysis widely identified as the dominant rate-limiting step.6
Attempts to enhance pit sludge degradation have traditionally relied on commercially available additives which mainly contain biologically active materials, e.g. enzymes or microorganisms. However, both laboratory and field evaluations have repeatedly demonstrated negligible impacts on pit performance.7–10 These outcomes are generally attributed to fundamental ecological limitations, including dilution of added biomass relative to indigenous communities and poor persistence of introduced organisms in competitive, nutrient-limited environments.11 As a result, it could be argued that attention should be shifted away from biological inoculation towards alternative, readily-available additives, such as biochar, which have the potential to modify the biochemical conditions in anaerobic degradation.
Rather than introducing new microorganisms, biochar influences the degradation process by altering local pH, substrate availability, and the accumulation of inhibitory intermediates, thereby indirectly supporting anaerobic metabolic activity.12–16 These properties have contributed to growing interest in biochar for faecal sludge management (FSM), where it has been considered for soil improvement, heavy metal (HM) immobilisation, water and nutrient retention, and carbon sequestration.17–20 For instance, faecal-sludge-derived biochar (FSB) has been reported to reduce the leaching of heavy metals from faecal sludge and surrounding soil because of its adsorption capacity.17 Previous research on the use of biochar for faecal sludge degradation enhancement, however, has focused on dry composting toilets. For example, it has been reported that biochar (from rice husk) was able to help degrade total organic carbon (TOC) from composting of the source-separated faeces up to 40%, twice higher compared to other traditional additives, and similar effects were found for non-source separated faecal sludge degradation.21 This beneficial effects have been attributed to the higher C/N ratio, provision of microbial attachment sites via its high surface area and porous structure and nutrients supplementation by biochar.12 Although rice husk biochar is among the few feedstocks reported directly for faecal sludge degradation-related applications, biochar derived from other materials should present the similar mechanisms, including wood residues, crop straw, manure, sewage sludge, and faecal sludge itself.19,22–24 Nevertheless, the above findings are largely derived from aerobic or semi-aerobic dry composting systems, leaving the role and potential benefits of biochar in wet pit latrines unrevealed, where anaerobic degradation dominates.
In parallel, the use of additives to enhance anaerobic digestion (AD) performance of organic matters has attracted increasing research interest, including the investigation of biochar addition effects. For instance, under mesophilic AD conditions, the addition of biochar has been reported to enhance methane production across a range of substrates, including animal manure, waste activated sludge, and food waste.15,25–29 Beyond improved methane yields, biochar has also been shown to shorten the lag phase of AD and to facilitate direct interspecies electron transfer (DIET) between syntrophic bacteria and methanogenic archaea, thereby accelerating electron flow and stabilising microbial metabolism.30,31 However, contrasting outcomes have also been reported, with inhibitory or negligible effects attributed to variations in biochar physicochemical properties arising from differences in feedstock type, pyrolysis temperature, and retention time.32,33 In particular, biochar with elevated heavy metal contents may adversely affect AD performance, as metal ions can disrupt enzymatic structure and function through interactions with protein functional groups.30
Despite extensive investigation of biochar as an additive for AD, very little is known about using FSB, even though faecal sludge pyrolysis has already been used in developing countries such as Uganda and Rwanda.34–36 Such biochar exhibits distinct physicochemical characteristics, including high ash content and enriched inorganic fractions,18,24,36 which may alter anaerobic microbial processes relative to conventional biomass-derived biochar. Furthermore, biochar production from faecal sludge involves high-temperature thermochemical treatment, which enables effective pathogen inactivation and thereby makes it safe to handle.37 In addition, the reuse of FSB as a pit additive represents a circular sanitation approach, whereby a treated faecal sludge product is returned to the system to improve degradation processes.
Therefore, this study, for the first time, aimed to investigate the potential of using FSB as an additive to facilitate degradation in wet pit latrine conditions by assessing AD performance of human excreta under laboratory conditions, specifically by: 1) monitoring gas yield and its kinetics, digestate properties and particularly, volatile solid (VS) reduction, and 2) exploring the microbial communities shifts due to biochar addition and DIET performance.
2. Materials and methods
2.1 Simulation of human faeces and urine
To provide controlled and reproducible substrate characteristics suitable for repetitive experiments and avoid health and safety risks from the use of real human excreta in the laboratory, this research uses synthetic human excreta that is adopted from previous research.38,39 The recipe and preparation process are summarised in Table S1. The physicochemical properties of the synthetic faeces and urine were comparable to those of published simulants and real human excreta (Table 1), indicating that the simulant provides a valid representation for the present study. According to the reported daily excreta production per capita, a ratio of faeces to urine simulants of 120 g![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 ml was used to simulate human excreta.38 The characteristics of the synthetic human excreta can be found in Table 2.
300 ml was used to simulate human excreta.38 The characteristics of the synthetic human excreta can be found in Table 2.
| Parameters | Simulant in this study | Simulant from previous studies38,39 | Real human excreta | |
|---|---|---|---|---|
| Faeces | TS (%) | 16.0 ± 0.2 | 18.4 | 15–35 (ref. 40) |
| VS (% of TS) | 86.8 ± 0.2 | 88.5 | 87 (ref. 41) | |
| pH | 6.6 | 5.2 | 5.0–8.0 (ref. 42) | |
| TN (%/TS) | 5.3 ± 0.3 | 4.1±0.2 | 4.7 ± 0.7 (ref. 43) | |
| N–NH4 (mg l−1 N−1) | 660.1 ± 14.9 | — | 719 ± 8.7 (ref. 44) | |
| TP (mg l−1 P) | 1711.3 ± 59.3 | — | — | |
| C/N | 7.8 | 17.3 | 9.6 (ref. 43) | |
| Urine | pH | 6.0 | 6.0 | 6–8.2 (ref. 45) |
| TS (%) | 1.8 ± 0.1 | 2.4 | 2.5–3.7 (ref. 45) | |
| VS (% of TS) | 44.0 ± 0.3 | 49.5 | 60–75 (ref. 45) | |
| pH | 6.0 | 6.0 | 6–8.2 (ref. 45) | |
| TN (mg l−1 N−1) | 6751.0 ± 293.5 | 5200 | 5000–8000 (ref. 46) | |
| N–NH4 (mg N−1) | 202.0 ± 11.7 | 197 | 126–603 (ref. 47, 48) | |
| TP (mg l−1 P) | 462.5 ± 7.4 | 400 | 800–2500 (ref. 40) | |
| C/N | 1.4 | 0.6 | — |
| Characteristics | Synthetic human excreta | FSB | Inoculum |
|---|---|---|---|
| TS (%) | 5.7 ± 0.1 | 75.38 ± 0.0 | 22.3 ± 0.3 |
| VS (%) | 4.4 ± 0.1 | 20.2 ± 0.3 | 14.0 ± 0.3 |
| VS/TS (%) | 76.3 ± 0.5 | 26.8 ± 0.4 | 62.8 ± 0.6 |
| pH | 6.3 | 7.9 | 9.3 |
| C/N | 5.3 | 9.02 | N/A |
2.2 Inoculum and FSB
Centrifuged sludge cake was collected from the anaerobic digester in a sewage treatment plant near London as inoculum for the AD experiments. Fresh inoculum was obtained before each batch experiment and pre-incubated for 7 days to degas and degrade any residual organic matter. Although this inoculum does not perfectly replicate the microbial community of pit latrines, microbial characterisation indicates that it provides a reasonable community-level representation, as it shares dominant phyla commonly reported in pit latrines and taxa associated with human gut and faecal microbiota, as discussed in 3.6.2. Moreover, microbial communities in pit latrines are known to vary substantially due to differences in local environmental conditions, pit design, usage patterns, and user behaviours,49–52 making it inherently challenging to define or reproduce a single “representative” pit latrine microbiome. In this context, the use of a well-established anaerobic digester inoculum offers a controlled and reproducible microbial consortium suitable for mechanistic evaluation of biochar effects, while maintaining relevance to faecal sludge degradation processes.The faecal-sludge-derived biochar (FSB) used in this study was produced from dried faecal sludge collected at Lubigi Sewage Treatment Plant in Kampala, Uganda, as described in ref. 53. Carbonisation (i.e. slow pyrolysis intended for solid char production) was conducted using traditional batch-operated retort kilns consisting of modified air-locked metallic drums.53 The process was initiated using available solid start-up fuels (e.g. wood, charcoal, briquettes), which were ignited prior to loading the dried faecal sludge. Once loaded, the kiln was operated under air-locked conditions and left to carbonise overnight (highest heating temperature recorded via thermometer readings: 400 °C), after which the charred material was collected the following day. The char samples were then ground, sieved (<2 mm) and homogenised, before being shipped to Imperial College London for storage, analysis and experimental use.
The main characteristics of the feedstock materials relevant to AD are presented in Table 2 and the proximate and ultimate analysis results of FSB are shown in Table S2. Total concentrations of the inorganic elements (i.e. macronutrients, trace elements, heavy metals) of all the materials are shown in Table S3.
2.3 Design of the AD experiments
Batch AD experiments were conducted at mesophilic temperature (35 °C) using 500 mL serum bottles with working volume of 400 mL, equipped with airtight caps. 1 L Tedlar bags were connected to the bottles for gas collection, from which a gas-tight syringe was used to measure the gas volume every other day except for the weekends. N2 was flushed by a tube connecting to a N2 cylinder for at least 5 minutes in each bottle before digestion to create anaerobic conditions, where the flushing time was tested to be long enough to flush out air. A detailed equipment set up is included in Fig. S1. The inoculum to substrate ratio (ISR) was set to 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 gVS and total solids (TS) of the mixture was adjusted to approximately 4%. This condition was found to be the optimal condition for degradation from the preliminary experiment56 and represent the average moisture content for wet pit latrines.57 Based on previous studies,29,54,55 FSB was added to the synthetic human excreta at doses of 0, 3, 6, 9, and 12 g L−1 and labelled as B0 (control), B3, B6, B9 and B12, respectively. Higher dose implicitly assumes an unlimited supply of FSB, which is unrealistic for on-site sanitation systems. The TS content after FSB addition ranged from 4.38–5.19% (Table S4). The AD experiments were finished when the daily methane production for three consecutive days is less than 1% of the cumulative methane production according to the biomethane potential tests (BMP) standard,58 which was 18 days in the presented study. All treatments were in triplicated, and the batch AD experiment was also repeated for three times to reduce variability introduced from the inoculum.
1 gVS and total solids (TS) of the mixture was adjusted to approximately 4%. This condition was found to be the optimal condition for degradation from the preliminary experiment56 and represent the average moisture content for wet pit latrines.57 Based on previous studies,29,54,55 FSB was added to the synthetic human excreta at doses of 0, 3, 6, 9, and 12 g L−1 and labelled as B0 (control), B3, B6, B9 and B12, respectively. Higher dose implicitly assumes an unlimited supply of FSB, which is unrealistic for on-site sanitation systems. The TS content after FSB addition ranged from 4.38–5.19% (Table S4). The AD experiments were finished when the daily methane production for three consecutive days is less than 1% of the cumulative methane production according to the biomethane potential tests (BMP) standard,58 which was 18 days in the presented study. All treatments were in triplicated, and the batch AD experiment was also repeated for three times to reduce variability introduced from the inoculum.
2.4 Analytical analysis
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 ratio (biochar
20 ratio (biochar![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O [g
H2O [g![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mL]) after mechanical shaking for 1 h.61
mL]) after mechanical shaking for 1 h.61Carbon and nitrogen content that is reported for C/N ratio were analysed using a Flash Smart CHNS/O Elemental analyser (Thermo Fisher Scientific, Germany).
 | (1) |
For total ammonia nitrogen, soluble chemical oxygen demand (sCOD), and volatile fatty acid (VFA) measurements, samples were filtered by 0.45 um membrane to obtain the liquid form. NH4–N and total nitrogen were determined by Skalar San++ (Skalar Analytical B.V., The Netherlands), where the digestion method for the latter is described in the SI. sCOD was measured according to ref. 64 and VFA were determined by HACH test kit (LCK365) and a spectrophotometer (UV-2401PC). Extracellular polymeric substances (EPS) were extracted from homogenized digestate following the formaldehyde–NaOH method.65 Briefly, 5 mL of sample was treated with 0.03 mL of 38% (w/w) formaldehyde at 4 °C for 1 h, followed by the addition of 2 mL of 1 N NaOH and incubation at 4 °C for 3 h. The mixture was then centrifuged at 20000 × g and 4 °C for 20 min, and the supernatant was collected as the EPS extract. Protein and carbohydrate concentrations in the EPS were quantified using the Pierce™ BCA Protein Assay Kit (Thermo Scientific™) and the phenol–sulfuric acid method,66 respectively.
The inorganic composition of feedstock material and the AD digestate were quantified using an inductively coupled plasma optical emission spectrometer (ICP-OES) (AVIO 500, Perkin Elmer, USA). Prior to the measurement, 6 ml hydrochloric acid and 2 ml nitric acid were into 0.4 g samples and the mixture was heated for 2 h at 95 °C and diluted using 2% nitric acid.67
2.5 Kinetics model
Of the various anaerobic digestion models, the first-order model and the modified Gompertz model are the most widely recognised and have been extensively applied in previous AD research to describe the kinetic behaviour of the system.68 The first-order model (eqn (2)) is based on the assumption that hydrolysis is the rate-limiting step in AD under pit latrine conditions.| B(t) = Bmax[1 − e−kt] | (2) |
The modified Gompertz model (eqn (3)) can be used to represent bacterial growth with the assumption that the methane production rate is corresponding to the growth rate of methanogenic microorganisms.
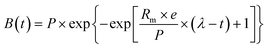 | (3) |
2.6 Microbial analysis
Microbial community analysis was conducted at the end of the experiment for the control (B0) and the 12 g L−1 biochar-amended reactors (B12) from two of the three batches, all are in triplicated. DNA extraction was performed on the digestate samples using DNeasy PowerSoil Pro Kits according to the manufacturer's protocol and the DNA quality and concentration were assessed via Agarose Gel Electrophoresis and NanoDrop2000. The V3-V4 hypervariable region of the bacterial 16S rRNA gene was amplified by PCR using the primer pair 341F (5′-CCTAYGGGRBGCASCAG-3′) and 806R (5′-GACTACNNGGGTATCTAAT-3′). PCR amplicons were purified and quantified prior to paired-end sequencing on an Illumina MiSeq platform (PE250). Sequence processing and taxonomic assignment were performed using QIIME2. Alpha and beta diversity metrics were calculated based on the QIIME2 outputs.2.7 Data analysis
To account for potential batch-to-batch variability in inoculum characteristics, gas and methane yields, as well as other digestate parameters, were normalised to their respective control reactors. Relative values were derived from three independent experimental batches, each consisting of triplicate treatment reactors, and are reported as mean values. Prior to statistical analysis, data quality was assessed through outlier screening. For gas yield measurements, outliers were identified and removed based on a relative standard deviation threshold of <10%, in accordance with the BMP test standard.69 For other physicochemical parameters, Grubbs' test was applied (two-sided, α = 0.05), as the datasets were normally distributed and comprised nine observations. Due to inherent batch-to-batch variability in AD microbiomes driven by operational and inoculum differences, microbial metrics were analysed within each batch rather than pooled across all samples.Differences in alpha-diversity indices between two independent groups were assessed using the Wilcoxon rank-sum test due to the small sample size and non-normal distribution, while two-sample t-tests were used for other measured parameters. Statistical significance was defined at p < 0.05. All statistical analyses were conducted in R (v4.5.2).
3. Results and discussion
3.1 Gas and methane yield
The relative cumulative methane and gas yield compared with the control (B0) for AD of synthetic human excreta with different doses of FSB added are presented in Fig. 1 and the absolute values expressed at Nml per g VSadded rom each batch are shown in Fig. S2. Across all the FSB doses, FSB addition showed a distinct early-stage enhancement compared to the control, as the relative values are all above 1. Peak methane and biogas production stimulation ranged from 1.48–1.60 and 1.32–1.68-fold, respectively, on day 3. This early response indicates that FSB primarily promotes upstream processes, particularly hydrolysis and acidogenesis, thereby accelerating the solubilisation of particulate organic matter and the formation of gaseous intermediates (e.g. CO2) and methanogenic precursors.70 This can be attributed to the macroporous structure (as is produced at low temperature) and mineral-rich surface of FSB that promote microbial attachment, improve enzyme–substrate contact, and stabilise local microenvironments during rapid acidogenesis.13,71 Similar enhancement has been reported during AD of lipid-rich substrates with sewage-sludge-derived biochar (5 g L−1) and during anaerobic co-digestion of activated sludge and food waste amended with sawdust-derived biochar (2–15 g L−1), which both applied similar doses as in this study.14,72Cumulative methane and gas production showed only minor differences between FSB-added reactors and the control as relative methane and gas production in all treatments gradually converged toward the control by the end of digestion on day 18. Conversely, biochar addition has been shown to increase methane yields when using biochar derived from bagasse biomass, wood, digested sludge, etc.28,29,73 One of the reasons for the observations in this study could be the lack of additional bioavailable carbon source compared to the other types of biochar. Unlike plant-derived biochar, which typically contains higher carbon content (40–71% (ref. 74)), FSB has been reported to contain less than 45% carbon17 or even lower as 8–24%.75 The FSB used in this study contains only 17.28 ± 0.21% carbon (Table S2), which could be due to the extensive biological degradation and the high ash content of faecal sludge prior to pyrolysis.75 Consequently, FSB is unlikely to act as a supplementary carbon source for methanogenesis.
Furthermore, since the preliminary experiments helped build the optimal condition by adjusting ISR and TS content, the system likely operated close to its biochemical methane potential and FSB addition would not promote additional gas production. A similar conclusion was also reported by ref. 76 who found that a oak sawdust-derived biochar addition failed to affect methane production form AD of dog food under the optimal ISR (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), while it enhanced methane production when the substrate was overloaded (ISR 1 and 0.5). In contrast, real pit latrine conditions are characterised by continuous faecal input and consequently lower effective ISR, under which FSB addition may be more likely to improve gas production. Therefore, further studies are needed to investigate the FSB performance under lower ISR and higher TS content conditions.
1), while it enhanced methane production when the substrate was overloaded (ISR 1 and 0.5). In contrast, real pit latrine conditions are characterised by continuous faecal input and consequently lower effective ISR, under which FSB addition may be more likely to improve gas production. Therefore, further studies are needed to investigate the FSB performance under lower ISR and higher TS content conditions.
Some studies also suggest that the high surface area of biochar generated by high pyrolysis temperature (>500 °C) could enhance absorption of the produced CH4,77,78 which is less likely to be the main reason for the unchanged methane and biogas yields observed here, as the present FSB was produced at 400 °C which normally has a surface area low to 2.1 m2 g−1,79 and there was no decrease in gas yield as FSB dose increased.
3.2 Kinetic models analysis
Unlike qualitative curve comparisons, kinetic simulations provide process-specific parameters that allow direct assessment of hydrolysis and methane production kinetics, offering a more mechanistic and transferable interpretation of biochar effects on AD performance. Model-derived kinetic parameters showed batch-to-batch variability (e.g. first-order rate k ranging from ∼0.04 to ∼0.14 d−1), reflecting differences in inoculum activity between experimental batches. In contrast to gas yields, kinetic parameters represent fitted process rates and are therefore not directly normalized across batches. Accordingly, model outputs are interpreted based on consistent within-batch trends for comparison with literature in Table 3. A fitted model for each batch is presented in Fig. S3.| First-order model | Modified Gompertz model | |||||||
|---|---|---|---|---|---|---|---|---|
| Bmax | K | R2 | P | Rm | λ | R2 | ||
| Batch 1 | B0 | 329.40 | 0.04 | 0.954 | 194.94 | 14.21 | 2.52 | 0.993 |
| B3 | 150.85 | 0.10 | 0.982 | 122.10 | 13.45 | 0.98 | 0.998 | |
| B6 | 220.70 | 0.10 | 0.986 | 177.86 | 17.95 | 0.73 | 0.996 | |
| B9 | 248.66 | 0.09 | 0.976 | 197.96 | 21.20 | 1.04 | 0.994 | |
| B12 | 199.47 | 0.11 | 0.978 | 165.88 | 18.77 | 0.94 | 0.995 | |
| Batch 2 | B0 | 243.22 | 0.04 | 0.927 | 125.37 | 17.52 | 3.33 | 0.999 |
| B3 | 206.03 | 0.05 | 0.924 | 116.88 | 17.12 | 3.14 | 0.997 | |
| B6 | 269.04 | 0.04 | 0.940 | 140.24 | 17.46 | 2.96 | 0.998 | |
| B9 | 175.40 | 0.07 | 0.929 | 113.63 | 17.00 | 2.86 | 0.997 | |
| B12 | 129.53 | 0.09 | 0.947 | 95.87 | 13.93 | 2.21 | 0.998 | |
| Batch 3 | B0 | 196.12 | 0.14 | 0.990 | 177.13 | 19.07 | 0.04 | 0.986 |
| B3 | 206.52 | 0.13 | 0.981 | 184.08 | 18.23 | 0.00 | 0.977 | |
| B6 | 196.36 | 0.14 | 0.988 | 179.10 | 19.54 | 0.00 | 0.983 | |
| B9 | 203.84 | 0.15 | 0.985 | 187.00 | 21.20 | 0.05 | 0.982 | |
| B12 | 200.29 | 0.14 | 0.988 | 181.52 | 20.59 | 0.18 | 0.987 | |
In all three batches, FSB-amended reactors generally exhibit higher hydrolysis rate constant k and higher maximum methane production rates Rm compared to the corresponding batch controls, further indicating enhanced early-stage digestion kinetics. This is consistent with a previous study28 which reported that biochar addition resulted in 1.2–1.4-fold increases in the hydrolysis efficiencies of major organic components. In parallel, lag phase λ was reduced by up to 1.5 days in FSB-amended treatments, particularly in B3–B9, which aligns with the experimental observation of accelerated early methane and gas production. This shortened lag phase was also consistent with the higher first-order hydrolysis rate constant (k) observed in the FSB treatments, suggesting faster initial substrate conversion. This could be resulting from the enhanced biofilm manufacture,80 which is further confirmed by the increased EPS carbohydrate content following FSB addition (see 3.3). A previous study81 also observed shortened lag phase by 10% and 27% with 1 and 10 g L−1, respectively, of dairy manure-derived biochar added to AD of the same feedstock, providing a relatively relevant comparison with another manure-based substrate. These results suggest that, from a practical sanitation perspective, FSB addition may be able to accelerate the pit latrine content degradation and reduce solids accumulation rates, since pit latrines are operated under continuous feeding mode.
Moreover, the methane production potential parameters Bmax and P fitted by the two models are both reduced by FSB addition, which reflects changes in methane production dynamics rather than a reduction in actual biodegradation potential. This could be because by increasing hydrolysis rate and shortening the lag phase, FSB accelerated early methane production, causing earlier curve saturation and a lower fitted methane potential due to parameter coupling.
3.3 VS reduction and digestate properties
VS, VFAs, NH4–N, sCOD and EPS (protein and carbohydrates) were measured at the end of the AD of synthetic human excreta, as shown in Fig. 2. VS reduction decreased following FSB addition lower to 0.6-fold with the highest dose of FSB, even using the Van Kleeck mass-corrected approach which accounts for the solid contribution of biochar (Fig. 2a). This confirms that although FSB increased the first-order rate constant and reduced the lag phase, it did not enhance the overall extent of organic matter mineralisation in this study. Instead, by providing higher surface area and more favourable attachment sites than the control, biochar promotes biofilm formation and stimulates EPS production, diverting a fraction of degradable carbon into biomass-associated VS rather than CH4 and CO2.82 This reflects in the elevated carbon-rich carbohydrates in EPS with FSB added (Fig. 2f). Protein content in EPS showed insignificant changes among all treatment, consistent with the absence of significant changes in NH4–N concentration (Fig. 2c). Evidence from the literature indicates that biochar mitigates the ammonia inhibition during AD, particularly in ammonia-stressed systems. For example, fruitwood-derived biochar reduced NH4–N by approximately 25% during AD of chicken manure, while corn stover and pine wood derived biochar reduced NH4–N concentration by up to 13% and 11% in sewage sludge digestion.83,32 In the present study, although NH4–N concentrations across three batches did not differ significantly with FSB addition, modest reductions (up to 22%) were observed in individual batches (Fig. S4). These results suggest that ammonia mitigation by FSB was not a consistent effect under the tested conditions.Fig. 2b shows a significant decrease in relative total VFA concentration with FSB added, achieving approximately 0.75-fold for B3-B12 compared to the control. A similar reduction in VFA accumulation has been reported by a previous study84 which applied dairy manure-derived biochar into AD of dairy manure at different temperatures. Biochar has been widely reported to enhance VFA turnover during anaerobic digestion by stimulating early-stage hydrolysis and acidogenesis while accelerating downstream VFA consumption through improved syntrophic interactions and methanogenic activity.16,72 This indicates faster intermediate conversion rather than suppressed acidogenesis in this study. Despite this, sCOD remained stable in FSB-amended reactors, indicating that faster VFA consumption was balanced by the formation and release of other soluble components, such as soluble microbial products and EPS-derived carbohydrates associated with biochar-enhanced biofilm activity.
3.4 Heavy metals concentrations and environmental implications
Heavy metals are among the key parameters considered in evaluating the environmental safety of pit latrine contents, particularly with respect to soil and groundwater contamination.85 Fig. 3 shows the total heavy metal concentrations in the AD digestate from one of the batches (batch 2), with higher values observed in the FSB-amended reactors, attributable to the addition of biochar (see Table S3). Several reference limits were used to contextualise the measured heavy metal concentrations and labelled as lines in Fig. 3: 1) regulatory thresholds for agricultural land application in South Africa and EU,85 to assess potential environmental and public health risks, and 2) IC50 values for AD processes,86,87 representing a 50% reduction in biological activity, to evaluate possible impacts on AD performance, given the sensitivity of key microbial processes to heavy metals.88,87 The heavy metal concentrations ranged between 2.9–18.8 mg L−1 (Cd), 6.3–39.6 mg L−1 (Cr), 17.5–158.8 mg L−1 (Pb), and 103.3–147.5 mg L−1 (Zn), 26.9–67.5 mg L−1 (Cu), and 8.1–22.5 mg L−1 (Ni). Most metals complied with the regulatory limits for agricultural land application, except for Cd and Cu in the B3 and B6 treatments. Lower total heavy metal concentrations were observed in batch 1 (Table S5), in which all measured metals were below the corresponding limits. | ||
| Fig. 3 Heavy metal concentrations in AD digestate, limits for spreading for agriculture in South Africa (blue solid line) and the European Union (red solid line),85 and the inhibitory concentrations of HM that reduces biological activities rate by 50% (IC50) for acidogenesis (green dash line) and methanogenesis (purple dash line).86,87 For Cr, Cu and Ni, the y-axis is displayed with a break to accommodate the large separation between measured concentrations and regulatory or inhibitory threshold values. For Pb and Zn, there is no corresponding limit from the same reference. Boxplots are plotted on median, first quartile, third quartile and minimum/maximum values. | ||
Generally, the solubility and mobility of heavy metals in aqueous environments are strongly pH dependent. Since digestate produced by AD typically exhibits a neutral to alkaline pH (approximately 8–8.5)89 and biochar addition increases soil pH,90 conditions under which the solubility of many heavy metals is substantially reduced. For example, Cu exhibits its highest leachability under acidic conditions (pH 4–5) and decreases progressively as pH increases.91 This limits metal dissolution when digestate is in contact with water and consequently lowers the potential for leaching and soil contamination. Furthermore, heavy metals associated with FSB are predominantly retained within the internal structure of biochar by adsorption and formation as complex with biochar functional groups, further constraining their release during percolation or leaching processes.30,92 These results indicate that, despite elevated total metal concentrations in some FSB-amended treatments, the resulting digestate generally remains suitable for land application under existing regulatory frameworks.
On the other hand, Fig. 3 shows that heavy metal concentrations in FSB-added reactors have exceeded the theoretical inhibitory limits except for Ni, while the absence of delayed methane onset or reduced production rates suggests that metal-related inhibition was unlikely to be responsible for the lack of enhanced cumulative methane production. This discrepancy can be attributed to the fact that the heavy metal concentrations reported here represent total metal contents, including both dissolved and solid-bound (inert) fractions, whereas inhibitory limits are primarily defined based on the bioavailable dissolved fraction that directly influences microbial activity. Finally, the reported heavy metal values should not be applied indiscriminately, as heavy metal behaviour and trace element requirements are highly dependent on process conditions, substrate composition, biochar properties, and site-specific soil and hydrogeological characteristics.87
3.5 Shifts in microbial communities
AD microbial communities are known to vary between experimental batches due to subtle differences in microbial assembly and inoculum state.93 Therefore, microbial responses were evaluated within each batch and interpreted based on consistent trends, rather than pooled statistical significance alone.In contrast, Wilcoxon test shows a clear difference between batches (p < 0.01), with higher richness and diversity observed in batch 1. This suggests a batch effect and reinforces that within-batch analyses are necessary. Same conclusion can be gained from Principal Coordinates Analysis (PCoA) (Fig. 4d) that batch 1 and batch 2 separated clearly along PC1 (60.44% variance). Regarding FSB effect, B0 and B12 formed distinguishable clusters in Batch 1 whereas overlapped in batch 2, suggesting that the biochar treatment could influence microbial community structure, though inconsistently between batches.
Twenty-two taxa were selected from the top 30 genera based on their functional relevance to anaerobic digestion and are presented in Fig. 5b. The full annotation table can be found in Table S7. At genus level, hydrolysis, acidogenesis and methanogenesis related taxa account for most of the microbial communities while acetogenesis related taxa remain at low relative abundance (<5%) (Fig. 5b), which is typical for a stable mesophilic AD system.100 Methanosarcina was the dominant genus detected in both batches, accounting for approximately 20–30% of the total microbial community, with a higher relative abundance observed in B12. As a metabolically versatile methanogen, Methanosarcina is capable of acetoclastic, hydrogenotrophic, and methylotrophic methanogenesis, and has been reported to engage in syntrophic interactions with Syntrophomonas via DIET.101,102 The higher relative abundance of Methanosarcina was accompanied by a consistent decrease in the hydrogenotrophic methanogen Methanobacterium, which was detected at much lower relative abundance following FSB addition. This opposing trend may suggest that FSB amendment influenced methanogen-associated community patterns, potentially favoring alternative methanogenic routes beyond strict hydrogen-mediated pathways, including acetoclastic methanogenesis. Similar shifts in methanogen-associated community structure have been reported in biochar-amended anaerobic co-digestion systems treating raw straw and cow manure.103 In contrast, Methanothrix, a strictly acetoclastic methanogen that has also been reported to participate in DIET-based electron uptake,101 remained stable in relative abundance under FSB amendment. Taken together, the enrichment of Methanosarcina, the stability of Methanothrix, and the decline of hydrogenotrophic Methanobacterium indicate a reduced reliance on hydrogen-mediated methanogenesis and are consistent with a possible biochar-mediated modulation of methanogenic electron transfer pathways. Moreover, although Methanosarcina increased in relative abundance, this does not necessarily translate into higher methane or biogas production, as methanogenic activity is primarily governed by substrate availability rather than methanogen abundance. However, because the 341F/806R primer set was not specifically optimized for archaeal profiling, these archaeal patterns should be interpreted with caution.
FSB addition consistently enriched the proteolytic genus Proteiniphilum, indicating that protein hydrolysis was maintained or enhanced under biochar addition.104 In contrast, several fast acidogenic and amino-acid-fermenting taxa, including Romboutsia and Intestinibacter, decreased across both batches, suggesting a restructuring of the acidogenic consortium rather than inhibition of upstream digestion.105,106 This shift implies that FSB favoured aggregate associated proteolytic fermenters, which is consistent with the observed increase in carbohydrate-rich EPS and altered carbon partitioning. In addition to AD-related microorganisms, Candidatus_Microthrix consistently increased in all batches. This genus has been widely associated with sludge bulking in wastewater treatment plants, which are primarily attributed to its filamentous morphology embedded within a carbohydrate-rich EPS matrix,107,108 which is consistent with the measured increase in EPS carbohydrates. Furthermore, the slight decrease in Syntrophomonas and Propionicimonas likely reflects reduced demand for H2-dependent syntrophic oxidation under FSB amendment, consistent with stable VFA profiles and a shift in methanogenic electron transfer pathways.
Overall, FSB reshaped the microbial community structure toward biofilm-associated hydrolytic and methanogenic populations, supporting stable methanogenesis while promoting EPS-mediated carbon retention.
4. Implications, limitations, and future work
According to the findings in this study, the primary role of FSB is more likely associated with accelerating early-stage hydrolysis rather than enhancing overall mineralisation. Under continuously fed pit conditions with low effective ISR ratios, increased hydrolysis may facilitate the breakdown and disintegration of freshly added faecal solids, potentially influencing physical accumulation patterns within the pit. However, the absence of increased VS reduction in this study indicates that such hydrolytic enhancement does not necessarily translate into greater long-term stabilisation or volume reduction of pit contents. Consequently, its application as a pit additive may not, on its own, provide sufficient benefit to justify its use.A further consideration is that the limited methane response to FSB may reflect FSB-specific properties, including comparatively lower carbon content, lower accessible surface area/porosity, and potentially weaker buffering or adsorption capacity relative to lignocellulosic biochar.17,75,79 By contrast, lignocellulosic biochar often exhibits higher surface area and alkalinity and generally lower intrinsic HM contents,109,110 which may increase their capacity to adsorb inhibitory intermediates and support syntrophic metabolism, thereby more readily enhancing methane production under stressed or overloaded conditions. Nevertheless, FSB retains a unique advantage in circularity, as it valorises faecal sludge into a reusable pit additive with potential operational and resource-recovery benefits.
Extrapolation of the findings of this study, conducted under controlled, idealised conditions, to real pit latrine systems remains uncertain, as pits operate under non-optimised and highly dynamic conditions characterised by continuous faecal input, lower effective ISR, variable moisture content, and long solids retention times. Under such conditions, the effects of FSB on hydrolysis, solids stabilisation, and gas production may differ substantially from those observed in batch laboratory assays. Future research should therefore prioritise long-term and semi-continuous experiments, field-scale pit simulations, and dissolved-phase metal speciation analyses to better resolve the role of FSB in regulating degradation pathways and pit-filling kinetics.
5. Conclusions
This study demonstrated that FSB addition to the anaerobic digestion of synthetic human excreta accelerates early-stage solubilisation of particulate organics while decreased the VS reduction. This was accompanied by a significant increase in carbohydrate-rich EPS, indicating a shift in carbon partitioning from mineralisation toward retention within extracellular polymeric matrices and microbial biomass. Microbial community analysis revealed insignificant changes in the diversity and evenness of microorganisms while suggesting targeted restructuring toward aggregate-associated populations, including enrichment of metabolically versatile methanogens (Methanosarcina) and proteolytic fermenters (Proteiniphilum). Although FSB offers advantages in circularity and sanitation safety, the findings of this study suggest that its application as a pit additive is unlikely to deliver substantial performance benefits.Author contributions
Mengru Qiu- conceptualization, formal analysis, methodology, validation, visualization, writing – original draft. Maria E. Koulouri- writing – review and editing. Laure Sioné- supervision. Michael R. Templeton- conceptualization, funding acquisition, supervision, writing – review and editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The microbial sequencing data were uploaded to NLM-NCBI, see http://www.ncbi.nlm.nih.gov/bioproject/1415342. The supporting data has been provided as part of the supplementary information (SI), including description of human excreta synthesis, proximate and ultimate analysis of FSB, total element concentrations of all materials, digestion method for TN, batch methane, gas, properties data, kinetic model fitting results, HM concentrations in other batches, alpha diversity results, and relative abundance of all top 30 genus. Supplementary information is available. See DOI: https://doi.org/10.1039/d6ew00095a.Acknowledgements
The authors would like to thank the Department of Civil and Environmental Engineering of Imperial College London for a Skempton scholarship. This project was supported by the Royal Academy of Engineering under the Research Chairs and Senior Research Fellowships programme. The authors would also like to thank Water For People Uganda team for their help in acquiring the biochar.References
- WHO and UNICEF, State of the World's Sanitation: An urgent call to transform sanitation for better health, environments, economies and societies, New York, USA, 2020, https://www.who.int/publications/i/item/9789240014473 Search PubMed.
- W. Gwenzi, J. Marumure, Z. Makuvara, T. T. Simbanegavi, E. L. Njomou-Ngounou and E. L. Nya, et al., The pit latrine paradox in low-income settings: A sanitation technology of choice or a pollution hotspot?, Sci. Total Environ., 2023, 879, 163179 CrossRef CAS PubMed.
- R. G. K. Hinton, R. M. Kalin, M. B. Kanjaye, P. Mleta, C. J. A. Macleod and M. Troldborg, Spatial model of groundwater contamination risks from pit-latrines in a low-income country, Water Res., 2024, 267, 122734 CrossRef CAS PubMed.
- E. Tilley, L. Ulrich, C. Luthi, P. Reymond and C. Zurbrügg, Compendium of Sanitation Systems and Technologies, Swiss Federal Institute of Aquatic Science and technology (Eawag), Dübendorf, Switzerland, 2014 Search PubMed.
- Z. Burt, R. Sklar and A. Murray, Costs and Willingness to Pay for Pit Latrine Emptying Services in Kigali, Rwanda, Int. J. Environ. Res. Public Health, 2019, 16, 4738 CrossRef PubMed.
- M. H. A. van Eekert, W. T. Gibson, B. Torondel, F. Abilahi, B. Liseki and E. Schuman, et al., Anaerobic digestion is the dominant pathway for pit latrine decomposition and is limited by intrinsic factors, Water Sci. Technol., 2019, 79, 2242–2250 CrossRef CAS PubMed.
- L. Taljaard, A. Venter, D. Gorton and Commission SAWR, An Evaluation of Different Commercial Microbial Or Microbially-derived Products for the Treatment of Organic Waste in Pit Latrines, Water Research Commission, 2005 Search PubMed, https://books.google.co.uk/books?id=eBhDtwAACAAJ.
- J. N. Bhagwan, D. Still, C. Buckley and K. Foxon, Challenges with up-scaling dry sanitation technologies, Water Sci. Technol., 2008, 58, 21–27 CrossRef CAS PubMed.
- B. F. Bakare, C. F. Nwaneri, K. M. Foxon, C. J. Brouckaert, D. Still and C. A. Buckley, PIT LATRINE, ADDITIVES, LABORATORY AND FIELD TRIALS, 2010 Search PubMed.
- E. Kemboi, J. van de Vossenberg, C. Hooijmans and G. Mamani, Impacts of Pit Latrine Additives on Volatile Solids and E. coli in Faecal Sludge, 2018, pp. 445–464 Search PubMed.
- C. Buckley, K. Foxon, C. Brouckaert, N. Rodda, C. Nwaneri and E. Balboni et al., Scientific support for the design and operation of ventilated improved pitlatrines and the efficacy of pit latrine additives, 2008, https://www.researchgate.net/publication/323748720 Search PubMed.
- J. Pan, J. Ma, L. Zhai, T. Luo, Z. Mei and H. Liu, Achievements of biochar application for enhanced anaerobic digestion: A review, Bioresour. Technol., 2019, 292, 122058 CrossRef CAS PubMed.
- S. Tang, Z. Wang, Z. Liu, Y. Zhang and B. Si, The Role of Biochar to Enhance Anaerobic Digestion: A Review, J. Renewable Mater., 2020, 8, 1033–1052 CAS.
- Q. Xu, Y. Liao, E. Cho and J. H. Ko, Effects of biochar addition on the anaerobic digestion of carbohydrate-rich, protein-rich, and lipid-rich substrates, J. Air Waste Manage. Assoc., 2020, 70, 455–467 CrossRef CAS PubMed.
- D. Li, Q. Ping, W. Guo, Y. Chen, L. Wang and Y. Li, Evaluating effects of biochar on anaerobic digestion of dewatered waste activated sludge: Digester performance, microbial co-metabolism and underlying mechanism, Chemosphere, 2023, 341, 140139 CrossRef CAS PubMed.
- Y. Xiao, H. Yang, X. Jiang, W. Wang and L. Deng, Mitigation of ammonia and volatile fatty acids inhibition in dry anaerobic digestion of chicken manure by biochar prepared at varying pyrolysis temperatures, Bioresour. Technol., 2025, 428, 132465 CrossRef CAS PubMed.
- B. C. Krueger, G. D. Fowler, M. R. Templeton and B. Moya, Resource recovery and biochar characteristics from full-scale faecal sludge treatment and co-treatment with agricultural waste, Water Res., 2020, 169, 115253 CrossRef CAS PubMed.
- M. Mamera, J. J. van Tol, M. P. Aghoghovwia, A. B. J. C. Nhantumbo, L. M. Chabala and A. Cambule, et al., Potential Use of Biochar in Pit Latrines as a Faecal Sludge Management Strategy to Reduce Water Resource Contaminations: A Review, Appl. Sci., 2021, 11, 11772 CrossRef CAS.
- M. E. Koulouri, M. Qiu, M. R. Templeton and G. D. Fowler, Carbon flows and biochar stability during co-pyrolysis of human faeces with wood biomass, Environ. Sci.: Water Res. Technol., 2024, 10, 2709–2722 RSC.
- L. Li, C. Zhu, B. Geng and L. Luo, Biochar-enhanced ex situ fermentation for rural toilet waste treatment: GHG mitigation and humification, Environ. Technol. Innovation, 2025, 40, 104479 CrossRef CAS.
- S. Hashemi, S. Boudaghpour and M. Han, Evaluation of different natural additives effects on the composting process of source separated feces in resource-oriented sanitation systems, Ecotoxicol. Environ. Saf., 2019, 185, 109667 CrossRef CAS PubMed.
- A. E. Erdogdu, R. Polat and G. Ozbay, Pyrolysis of goat manure to produce bio-oil, Eng. Sci. Technol. Int. J., 2019, 22, 452–457 Search PubMed.
- K. B. Cantrell, P. G. Hunt, M. Uchimiya, J. M. Novak and K. S. Ro, Impact of pyrolysis temperature and manure source on physicochemical characteristics of biochar, Bioresour. Technol., 2012, 107, 419–428 CrossRef CAS PubMed.
- B. C. Krueger, G. D. Fowler, M. R. Templeton and B. Moya, Resource recovery and biochar characteristics from full-scale faecal sludge treatment and co-treatment with agricultural waste, Water Res., 2020, 169, 115253 CrossRef CAS PubMed.
- J. T. E. Lee, E. Y. Lim, L. Zhang, T.-H. Tsui, H. Tian and M. Yan, et al., Methanosarcina thermophila bioaugmentation and its synergy with biochar growth support particles versus polypropylene microplastics in thermophilic food waste anaerobic digestion, Bioresour. Technol., 2022, 360, 127531 CrossRef CAS PubMed.
- B. A. Collins, C. H. Birzer, S. P. Kidd, T. Hall and P. R. Medwell, The influence of biochar position in a leach bed system anaerobically digesting chicken litter, J. Environ. Manage., 2023, 344, 118404 CrossRef CAS PubMed.
- T. Ngo, L. S. Khudur, C. Krohn, S. Hassan, K. Jansriphibul and I. G. Hakeem, et al., Wood biochar enhances methanogenesis in the anaerobic digestion of chicken manure under ammonia inhibition conditions, Heliyon, 2023, 9, e21100 CrossRef CAS PubMed.
- S. Duan, J. He, X. Xin, L. Li, X. Zou and Y. Zhong, et al., Characteristics of digested sludge-derived biochar for promoting methane production during anaerobic digestion of waste activated sludge, Bioresour. Technol., 2023, 384, 129245 CrossRef CAS PubMed.
- S. Xu, J. Bu, C. Li, Y. W. Tiong, P. Sharma and K. Liu, et al., Biochar enhanced methane yield on anaerobic digestion of shell waste and the synergistic effects of anaerobic co-digestion of shell and food waste, Fuel, 2024, 357, 129933 CrossRef CAS.
- P. Devi and C. Eskicioglu, Effects of biochar on anaerobic digestion: a review, Environ. Chem. Lett., 2024, 22, 2845–2886 CrossRef CAS.
- R. Shen, Y. Jing, J. Feng, L. Zhao, Z. Yao and J. Yu, et al., Simultaneous carbon dioxide reduction and enhancement of methane production in biogas via anaerobic digestion of cornstalk in continuous stirred-tank reactors: The influences of biochar, environmental parameters, and microorganisms, Bioresour. Technol., 2021, 319, 124146 CrossRef CAS PubMed.
- J. Pan, J. Ma, X. Liu, L. Zhai, X. Ouyang and H. Liu, Effects of different types of biochar on the anaerobic digestion of chicken manure, Bioresour. Technol., 2019, 275, 258–265 CrossRef CAS PubMed.
- C. Cimon, P. Kadota and C. Eskicioglu, Effect of biochar and wood ash amendment on biochemical methane production of wastewater sludge from a temperature phase anaerobic digestion process, Bioresour. Technol., 2020, 297, 122440 CrossRef CAS PubMed.
- D. Roobroeck, R. Hood-Nowotny, D. Nakubulwa, J.-B. Tumuhairwe, M. J. G. Mwanjalolo and I. Ndawula, et al., Biophysical potential of crop residues for biochar carbon sequestration, and co-benefits, in Uganda, Ecol. Appl., 2019, 29, e01984 CrossRef PubMed.
- A. Uwase, Biochar: Properties and Potential Benefits for Agricultural Soil in Rwanda, Honors Program, Theses, 2022, https://digitalcommons.unl.edu/honorstheses/465 Search PubMed.
- M. E. Koulouri, L. Owomuhangi, Y. Lugali, M. R. Templeton and G. D. Fowler, Pyrolysis of faecal sludge and biomass waste for resource recovery in Kampala, Uganda, Environ. Sci.: Water Res. Technol., 2025, 11, 2542–2554 RSC.
- T. Bond, Q. Tse, C. L. Chambon, P. Fennell, G. D. Fowler and B. C. Krueger, et al., The feasibility of char and bio-oil production from pyrolysis of pit latrine sludge, Environ. Sci.: Water Res. Technol., 2018, 4, 253–264 RSC.
- J. Colón, A. A. Forbis-Stokes and M. A. Deshusses, Anaerobic digestion of undiluted simulant human excreta for sanitation and energy recovery in less-developed countries, Energy Sustainable Dev., 2015, 29, 57–64 CrossRef.
- R. Penn, B. J. Ward, L. Strande and M. Maurer, Review of synthetic human faeces and faecal sludge for sanitation and wastewater research, Water Res., 2018, 132, 222–240 CrossRef CAS PubMed.
- K. Wignarajah, E. Litwiller, J. W. Fisher and J. Hogan, Simulated Human Feces for Testing Human Waste Processing Technologies in Space Systems, SAE Trans., 2006, 115, 424–430 Search PubMed.
- R. Rajagopal, J. W. Lim, Y. Mao, C.-L. Chen and J.-Y. Wang, Anaerobic co-digestion of source segregated brown water (feces-without-urine) and food waste: For Singapore context, Sci. Total Environ., 2013, 443, 877–886 CrossRef CAS PubMed.
- C. Rose, A. Parker, B. Jefferson and E. Cartmell, The characterization of feces and urine: A review of the literature to inform advanced treatment technology, Crit. Rev. Environ. Sci. Technol., 2015, 45, 1827–1879 CrossRef CAS PubMed.
- Y. Cai, Z. Zheng, L. Wei, H. Zhang and X. Wang, The characteristics of multi-substrates (low and high C/N) anaerobic digestion: focus on energy recovery and the succession of methanogenic pathway, Bioresour. Technol., 2022, 343, 125976 CrossRef CAS PubMed.
- S. Singh, N. Hariteja, S. Sharma, N. J. Raju and T. J. R. Prasad, Production of biogas from human faeces mixed with the co-substrate poultry litter & cow dung, Environ. Technol. Innovation, 2021, 23, 101551 CrossRef CAS.
- D. F. Putnam, Composition and concentrative properties of human urine, 1971, https://ntrs.nasa.gov/citations/19710023044 (accessed 24 July2023) Search PubMed.
- Z. S. Ban and G. Dave, Laboratory studies on recovery of n and p from human urine through struvite crystallisation and zeolite adsorption, Environ. Technol., 2004, 25, 111–121 CrossRef CAS PubMed.
- B. B. Jana, S. Rana and S. K. Bag, Use of human urine in phytoplankton production as a tool for ecological sanitation, Water Sci. Technol., 2012, 65, 1350–1356 CrossRef CAS PubMed.
- B. Beler-Baykal, A. D. Allar and S. Bayram, Nitrogen recovery from source-separated human urine using clinoptilolite and preliminary results of its use as fertilizer, Water Sci. Technol., 2011, 63, 811–817 CrossRef CAS PubMed.
- B. Torondel, J. H. J. Ensink, O. Gundogdu, U. Z. Ijaz, J. Parkhill and F. Abdelahi, et al., Assessment of the influence of intrinsic environmental and geographical factors on the bacterial ecology of pit latrines, Microb. Biotechnol., 2016, 9, 209–223 CrossRef PubMed.
- A. Byrne, R. Sindall, L. Wang, F. De los Reyes and C. Buckley, What happens inside a pour-flush pit? Insights from comprehensive characterization, 2017, https://repository.lboro.ac.uk/articles/conference_contribution/What_happens_inside_a_pour-flush_pit_Insights_from_comprehensive_characterization/9589328/1 (accessed 26 Aug2023) Search PubMed.
- M. C. Changara, W. T. Sanyika, C. Bangira and S. Misi, Physico-chemical properties and bacterial community structure dynamics during the mesophilic anaerobic digestion of pit latrine faecal sludge, Water SA, 2019, 45, 338–348 CrossRef CAS.
- S. B. Sam, S. K. Smith, R. Niederdorfer, A. Scheidegger, B. J. Ward and J. M. Tembo, et al., Microbial community composition reflects water usage and storage conditions in a city-wide study of non-sewered wastewater (fecal sludge), PLOS Water, 2025, 4, e0000386 CrossRef.
- M. E. Koulouri, L. Owomuhangi, Y. Lugali, M. R. Templeton and G. D. Fowler, Pyrolysis of faecal sludge and biomass waste for resource recovery in Kampala, Uganda, Environ. Sci.: Water Res. Technol., 2025, 11, 2542–2554 RSC.
- N. M. S. Sunyoto, M. Zhu, Z. Zhang and D. Zhang, Effect of biochar addition on hydrogen and methane production in two-phase anaerobic digestion of aqueous carbohydrates food waste, Bioresour. Technol., 2016, 219, 29–36 CrossRef CAS PubMed.
- S. Basumatary, P. Goswami and P. Kalita, Impact of coconut husk biochar on methane production rate in batch type anaerobic digester fed with cattle dung and cooked kitchen waste, Biomass Bioenergy, 2024, 187, 107300 CrossRef CAS.
- M. Qiu, L. Sioné and M. R. Templeton, Orthophosphate addition to enhance degradation of synthetic human excreta in simulated wet pit latrines, J. Environ. Manage., 2026, 400, 128792 CrossRef CAS PubMed.
- A. Fanyin-Martin, W. Tamakloe, E. Antwi, J. Ami, E. Awarikabey and J. Apatti, Chemical characterization of faecal sludge in the Kumasi metropolis, Ghana, Gates Open Research, 2017, 1, 12 Search PubMed.
- C. Holliger, M. Alves, D. Andrade, I. Angelidaki, S. Astals and U. Baier, et al., Towards a standardization of biomethane potential tests, Water Sci. Technol., 2016, 74, 2515–2522 CrossRef CAS PubMed.
- ASTM, Standard Test Methods for Proximate Analysis of Coal and Coke by Macro Thermogravimetric Analysis, 2023, https://store.astm.org/d7582-15.html (accessed 26 Jan2026).
- B. C. Krueger, G. D. Fowler, M. R. Templeton and S. Septien, Faecal sludge pyrolysis: Understanding the relationships between organic composition and thermal decomposition, J. Environ. Manage., 2021, 298, 113456 CrossRef CAS PubMed.
- B. Singh, M. Camps-Arbestain and J. Lehmann, Biochar: A Guide to Analytical Methods, Csiro Publishing, 2017 Search PubMed.
- APHA and 2540 SOLIDS, in Standard Methods For the Examination of Water and Wastewater, American Public Health Association, 2017, DOI:10.2105/SMWW.2882.030.
- M. S. Switzenbaum, J. B. Farrell and A. B. Pincince, Relationship between the Van Kleeck and mass-balance calculation of volatile solids loss, Water Environ. Res., 2003, 75, 377–380 CrossRef CAS PubMed.
- APHA, 5220 CHEMICAL OXYGEN DEMAND (COD), in Standard Methods For the Examination of Water and Wastewater, 2017, DOI:10.2105/SMWW.2882.103.
- H. Liu and H. H. P. Fang, Extraction of extracellular polymeric substances (EPS) of sludges, J. Biotechnol., 2002, 95, 249–256 CrossRef CAS PubMed.
- A. A. Albalasmeh, A. A. Berhe and T. A. Ghezzehei, A new method for rapid determination of carbohydrate and total carbon concentrations using UV spectrophotometry, Carbohydr. Polym., 2013, 97, 253–261 CrossRef CAS PubMed.
- US EPA O. EPA Method 3050B: Acid Digestion of Sediments, Sludges, and Soils, 2019, https://www.epa.gov/esam/epa-method-3050b-acid-digestion-sediments-sludges-and-soils (accessed 26 Jan2026).
- S. Xie, F. I. Hai, X. Zhan, W. Guo, H. H. Ngo and W. E. Price, et al., Anaerobic co-digestion: A critical review of mathematical modelling for performance optimization, Bioresour. Technol., 2016, 222, 498–512 CrossRef CAS PubMed.
- S. D. Hafner, H. Fruteau de Laclos, K. Koch and C. Holliger, Improving Inter-Laboratory Reproducibility in Measurement of Biochemical Methane Potential (BMP), Water, 2020, 12, 1752 CrossRef CAS.
- M. Krishania, V. K. Vijay and R. Chandra, Methane fermentation and kinetics of wheat straw pretreated substrates co-digested with cattle manure in batch assay, Energy, 2013, 57, 359–367 CrossRef CAS.
- R. Sirohi, M. Kumar, V. Vivekanand, A. Shakya, A. Tarafdar and R. Singh, et al., Integrating biochar in anaerobic digestion: Insights into diverse feedstocks and algal biochar, Environ. Technol. Innovation, 2024, 36, 103814 CrossRef CAS.
- G. Wang, Q. Li, X. Gao and X. C. Wang, Synergetic promotion of syntrophic methane production from anaerobic digestion of complex organic wastes by biochar: Performance and associated mechanisms, Bioresour. Technol., 2018, 250, 812–820 CrossRef CAS PubMed.
- T. Ngo, L. S. Khudur, S. Hassan, K. Jansriphibul and A. S. Ball, Enhancing microbial viability with biochar for increased methane production during the anaerobic digestion of chicken manure, Fuel, 2024, 368, 131603 CrossRef CAS.
- B. K. Biswal and R. Balasubramanian, Use of biomass-derived biochar as a sustainable material for carbon sequestration in soil: recent advancements and future perspectives, npj Mater. Sustainability, 2025, 3, 26 CrossRef CAS.
- H. L. Nicholas, I. Mabbett, H. Apsey and I. Robertson, Physico-chemical properties of waste derived biochar from community scale faecal sludge treatment plants, Gates Open Research, 2022, 6, 96 Search PubMed.
- J. Hu, K. Stenchly, W. Gwenzi, M. Wachendorf and K. Kaetzl, Critical evaluation of biochar effects on methane production and process stability in anaerobic digestion, Front. Energy Res., 2023, 11, 1205818 CrossRef.
- B. Zhou, Y. Feng, Y. Wang, L. Yang, L. Xue and B. Xing, Impact of hydrochar on rice paddy CH4 and N2O emissions: A comparative study with pyrochar, Chemosphere, 2018, 204, 474–482 CrossRef CAS PubMed.
- B. Wu, Q. Yang, F. Yao, S. Chen, L. He and K. Hou, et al., Evaluating the effect of biochar on mesophilic anaerobic digestion of waste activated sludge and microbial diversity, Bioresour. Technol., 2019, 294, 122235 CrossRef CAS PubMed.
- M. E. Koulouri, M. R. Templeton and G. D. Fowler, Enhancing the nitrogen and phosphorus content of faecal-derived biochar via adsorption and precipitation from human urine, J. Environ. Manage., 2024, 352, 119981 CrossRef CAS PubMed.
- R. Kundu, B. Kunnoth, S. Pilli, V. R. Polisetty and R. D. Tyagi, Biochar symbiosis in anaerobic digestion to enhance biogas production: A comprehensive review, J. Environ. Manage., 2023, 344, 118743 CrossRef CAS PubMed.
- H. M. Jang, J. Brady and E. Kan, Succession of microbial community in anaerobic digestion of dairy manure induced by manure-derived biochar, Environ. Eng. Res., 2021, 26, DOI:10.4491/eer.2020.001.
- M. Kayoumu, H. Wang and G. Duan, Interactions between microbial extracellular polymeric substances and biochar, and their potential applications: a review, Biochar, 2025, 7, 62 CrossRef CAS.
- Y. Shen, S. Forrester, J. Koval and M. Urgun-Demirtas, Yearlong semi-continuous operation of thermophilic two-stage anaerobic digesters amended with biochar for enhanced biomethane production, J. Cleaner Prod., 2017, 167, 863–874 CrossRef CAS.
- H. M. Jang, Y.-K. Choi and E. Kan, Effects of dairy manure-derived biochar on psychrophilic, mesophilic and thermophilic anaerobic digestions of dairy manure, Bioresour. Technol., 2018, 250, 927–931 CrossRef CAS PubMed.
- P. T. Odirile, V. C. Obuseng, M. Moshoeshoe, L. Tshenyego and B. Mbongwe, Assessment of faecal sludge quality, heavy metal contamination, and ecological risk: implications for sustainable agriculture, Environ. Monit. Assess., 2024, 196, 1270 CrossRef CAS PubMed.
- C.-Y. Lin, Effect of heavy metals on volatile fatty acid degradation in anaerobic digestion, Water Res., 1992, 26, 177–183 CrossRef CAS.
- R. Kadam, K. Khanthong, H. Jang, J. Lee, J. Park and R. Kadam, et al., Occurrence, Fate, and Implications of Heavy Metals during Anaerobic Digestion A Review, Energies, 2022, 15, 8618 CrossRef CAS.
- J. Glass and V. J. Orphan, Trace Metal Requirements for Microbial Enzymes Involved in the Production and Consumption of Methane and Nitrous Oxide, Front. Microbiol., 2012, 3, 00061 Search PubMed.
- M. Makádi, A. Tomócsik, V. Orosz, M. Makádi, A. Tomócsik and V. Orosz, Digestate: A New Nutrient Source - Review, in Biogas, IntechOpen, 2012, DOI:10.5772/31355.
- Y. Tang, C. Wang, P. E. Holm, H. C. B. Hansen and K. K. Brandt, Impacts of biochar materials on copper speciation, bioavailability, and toxicity in chromated copper arsenate polluted soil, J. Hazard. Mater., 2023, 459, 132067 CrossRef CAS PubMed.
- M. Izquierdo and X. Querol, Leaching behaviour of elements from coal combustion fly ash: An overview, Int. J. Coal Geol., 2012, 94, 54–66 CrossRef CAS.
- J. Liu, J. Zheng, Y. Niu, Z. Zuo, J. Zhang and Y. Wei, Effect of zero-valent iron combined with carbon-based materials on the mitigation of ammonia inhibition during anaerobic digestion, Bioresour. Technol., 2020, 311, 123503 CrossRef CAS PubMed.
- A. R. Perrotta, R. Kumaraswamy, J. R. Bastidas-Oyanedel, E. J. Alm and J. Rodríguez, Inoculum composition determines microbial community and function in an anaerobic sequential batch reactor, PLoS One, 2017, 12, e0171369 CrossRef PubMed.
- N. Maqbool, S. Sam, S. Jamal Khan and L. Strande, Relation of organic fractions in fresh and stored fecal sludge and foodwaste to biogas production, J. Water, Sanit. Hyg. Dev., 2024, 14, 277–290 CrossRef.
- S. B. Sam, B. J. Ward, R. Niederdorfer, E. Morgenroth and L. Strande, Elucidating the role of extracellular polymeric substances (EPS) in dewaterability of fecal sludge from onsite sanitation systems, and changes during anaerobic storage, Water Res., 2022, 222, 118915 CrossRef CAS PubMed.
- S. B. Sam, E. Morgenroth and L. Strande, Changes in organic fractions, cations, and stabilization from feces to fecal sludge: implications for dewatering performance and management solutions, J. Water, Sanit. Hyg. Dev., 2023, 13, 699–710 CrossRef.
- Q. Fan, X. Fan, P. Fu, Y. Sun, Y. Li and S. Long, et al., Microbial community evolution, interaction, and functional genes prediction during anaerobic digestion in the presence of refractory organics, J. Environ. Chem. Eng., 2022, 10, 107789 CrossRef CAS.
- A. García Álvaro, C. Ruiz Palomar, E. I. Valenzuela, D. Hermosilla Redondo, R. Muñoz Torre and C. I. de Godos, Microbial analysis of anaerobic digester reveals prevalence of manure microbiota, J. Water Process Eng., 2024, 60, 105162 CrossRef.
- Q. Yang, H. Liu, L. Liu, Z. Yan, C. Chui and N. Yang, et al., Enhancing Methane Production in Anaerobic Digestion of Food Waste Using Co-Pyrolysis Biochar Derived from Digestate and Rice Straw, Molecules, 2025, 30, 1766 CrossRef CAS PubMed.
- K. Xu, H. Liu, G. Du and J. Chen, Real-time PCR assays targeting formyltetrahydrofolate synthetase gene to enumerate acetogens in natural and engineered environments, Anaerobe, 2009, 15, 204–213 CrossRef CAS PubMed.
- Z. Wang, T. Wang, B. Si, J. Watson and Y. Zhang, Accelerating anaerobic digestion for methane production: Potential role of direct interspecies electron transfer, Renewable Sustainable Energy Rev., 2021, 145, 111069 CrossRef CAS.
- A.-E. Rotaru, G. Gharib, A. Jabaley, K. Anestis and R. Kotoky, Cell surface differences within the genus Methanosarcina shape interactions with the extracellular environment, J. Bacteriol., 2025, 207, e00112–e00125 CrossRef PubMed.
- R. Shen, Y. Jing, J. Feng, J. Luo, J. Yu and L. Zhao, Performance of enhanced anaerobic digestion with different pyrolysis biochars and microbial communities, Bioresour. Technol., 2020, 296, 122354 CrossRef CAS PubMed.
- Z. Wu, D. Nguyen, T. Y. C. Lam, H. Zhuang, S. Shrestha and L. Raskin, et al., Synergistic association between cytochrome bd-encoded Proteiniphilum and reactive oxygen species (ROS)-scavenging methanogens in microaerobic-anaerobic digestion of lignocellulosic biomass, Water Res., 2021, 190, 116721 CrossRef CAS PubMed.
- M. Abid, J. Wu, Y. Yuanyuan, Z. Ajmal, T. Mehmood and S. N. Husnain, et al., Enhanced anaerobic digestion of freezing and thawing pretreated cow manure with increasing solid content: kinetics and microbial community dynamics, Sci. Rep., 2024, 14, 25579 CrossRef CAS PubMed.
- Y. L. Song, C. X. Liu, M. McTeague, P. Summanen and S. M. Finegold, Clostridium bartlettii sp. nov., isolated from human faeces, Anaerobe, 2004, 10, 179–184 CrossRef CAS PubMed.
- M. C. Collivignarelli, M. Baldi, A. Abbà, F. M. Caccamo, M. C. Miino and E. C. Rada, et al., Foams in Wastewater Treatment Plants: From Causes to Control Methods, Appl. Sci., 2020, 10, 2716 CrossRef CAS.
- M. Nierychlo, C. M. Singleton, F. Petriglieri, L. Thomsen, J. F. Petersen and M. Peces, et al., Low Global Diversity of Candidatus Microthrix, a Troublesome Filamentous Organism in Full-Scale WWTPs, Front. Microbiol., 2021, 12, 690215 Search PubMed.
- L. Wang, Y. S. Ok, D. C. W. Tsang, D. S. Alessi, J. Rinklebe and H. Wang, et al., New trends in biochar pyrolysis and modification strategies: feedstock, pyrolysis conditions, sustainability concerns and implications for soil amendment, Soil Use Manage., 2020, 36, 358–386 CrossRef.
- A. B. Duwiejuah, A. H. Abubakari, A. K. Quainoo and Y. Amadu, Review of Biochar Properties and Remediation of Metal Pollution of Water and Soil, J. health pollut., 2020, 10, 200902 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |




