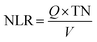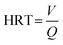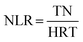Effect of PAC-embedded fillers coupled with electrochemical systems on nitrogen transfer pathways and removal
Received
1st July 2025
, Accepted 11th November 2025
First published on 18th November 2025
Abstract
To enhance electron donor utilization efficiency for advanced nitrogen removal from low carbon-to-nitrogen (C/N) ratio wastewater, polyvinyl alcohol–sodium acetate (PVA–SA) fillers embedded with powdered activated carbon (PAC) were developed and implemented in four lab-scale biofilters treating synthetic effluents from grit chamber and secondary clarifier outputs. Nitrogen removal performance and microbial community dynamics were systematically investigated. The electrochemical biofilter with embedded PAC fillers (EB1) significantly enhanced electron transfer, with nitrogen conversion pathways influenced by the concentrations and types of electron acceptors and donors. Nitrification was suppressed when electron donors (NH4+–N and COD) were present, while denitrification was inhibited under excess NO3−–N conditions. Although non-embedded PAC fillers favored enriching nitrifiers including Nitrosomonas and Nitrospira, and denitrifiers, such as Thauera, Comamonadaceae and Dechloromonas, the electrochemical biofilter facilitated greater accumulation of electrochemically active bacteria on anode plates, including Geobacter (11.64–14.10%), Desulfuromonas (5.88–10.85%) and Pseudomonas (15.15–17.53%). When the influent contained 13 mg L−1 NH4+–N, 8 mg L−1 NO3−–N and 77 mg L−1 COD, Candidatus_Brocadia (0.49–0.61%) was enriched in EB1, and the average contribution of nitrogen conversion via anaerobic ammonia oxidation (anammox) was 10.79% higher than in the non-embedded PAC biofilter. This study offered theoretical insights into optimizing nitrogen removal in low C/N wastewater treatment by enhancing electron donor utilization and promoting functional microbial populations.
Water impact
The C/N ratio of real sewage is enough for the biological nitrogen removal process, but some C will be wasted by other processes. This study explored the effect of influent quality on electron transfer of nitrogen conversion processes under an electrochemical system, aiming to enhance electron donor utilization and propose a strategy for low C/N ratio wastewater treatment.
|
1. Introduction
Excessive nitrogen discharged from urban wastewater treatment plants (WWTPs) is a primary driver of aquatic eutrophication. To restore aquatic ecosystems, it is necessary to implement stricter effluent discharge standards, particularly for total nitrogen (TN). Typical domestic wastewater in China is carbon-limited, with a low carbon-to-nitrogen (C/N) ratio of 3–6. Therefore, an external carbon source, such as sodium acetate, is commonly required to enhance nitrogen removal. Although nitrogen removal could be improved by external carbon source addition, it is frequently accompanied by high operating costs and excess sludge. In theory, 2.86 g of COD is sufficient to reduce 1 g of NO3−–N to N2. Therefore, nitrogen removal in domestic wastewater is not necessarily constrained by carbon deficiency, but rather by the complexity of nitrogen transformation pathways. Enhancing the efficiency of electron donor utilization is considered a promising strategy to improve nitrogen removal performance under low C/N conditions.
Bioelectrochemical systems (BES), particularly microbial fuel cells (MFCs), accelerated electron transfer during biological treatment processes while generating electricity, concomitantly improving pollutant removal.1,2 However, nitrogen conversion consists of complex oxidation and reduction processes, such as nitrification, denitrification, and anammox. Although the MFC system enhanced electron transfer, nitrogen removal represents only a few of the numerous redox biological reaction processes.3,4 Therefore, the actual nitrogen removal performance is highly dependent on the influent water quality.
The growth rates of autotrophic microorganisms involved in biological nitrogen removal, such as nitrifying bacteria and anammox bacteria, are relatively low. To address this limitation, fillers are frequently added to establish an integrated fixed-film activated sludge (IFAS) system, which has been widely applied in retrofitting and upgrading WWTPs.5 The anoxic environment within the dense biofilm facilitated denitrification, thereby often enhancing nitrogen removal performance in IFAS systems.6 Furthermore, some studies developed nitrogen removal fillers by embedding microorganisms. Compared with inorganic fillers, organic fillers have been shown to increase the activity of the attached microorganisms and typically composed of PVA and SA. Adsorbents such as bentonite and PAC were added to prepare composite fillers.7,8 The three-dimensional network structure of these composites not only increased the available surface area for microbial attachment, but also optimized internal mass transfer. Although these approaches offer promising strategies to improve wastewater treatment, whether advanced nitrogen removal can be achieved under low C/N conditions, especially under various influent water qualities, remains unclear.
To address the questions above, this study utilized PVA and SA to prepare two types of fillers, with/without PAC embedding. Four types of biofilters were constructed: an electrochemical biofilter with PAC-embedded fillers (EB1), electrochemical biofilter with non-embedded fillers (EB2), conventional biofilter with PAC-embedded fillers (B1), and conventional biofilter with non-embedded fillers (B2). These systems were operated to treat simulated low C/N wastewater. Nitrogen removal performance and the contribution of various nitrogen conversion pathways were systematically analyzed. Furthermore, the relationship between influent water quality and nitrogen transformation pathways in electrochemical systems was thoroughly evaluated. These findings were expected to provide theoretical support and technical guidance for advanced nitrogen removal in low C/N wastewater treatment.
2. Materials and methods
2.1 Experimental equipment
Filler preparation.
PVA, SA and PAC were soaked in deionized water at concentrations of 12%, 0.5%, and 4%, respectively. The mixture was then heated to 95 °C for 2 h to form a uniform gel. After cooling to room temperature, the gel was injected into spherical molds (3 mm) using a syringe, and frozen at −20 °C for 12 h. The detached microspheres were submerged in a saturated solution of boric acid solution (5% CaCl2) at 4 °C for 24 h to induce cross-linking. The resulting spheres were subsequently washed with deionized water. Non-embedded PAC fillers were prepared using a similar procedure without adding PAC. The average compressive strengths of the PAC-embedded and non-embedded fillers were 2.50 MPa and 1.50 MPa, respectively, while their electrical resistivities were measured at 4.40 Ω cm and 12.13 Ω cm, respectively.
Biofilm attachment.
The prepared fillers were placed in four hollow mesh plastic frames (8 cm) at a packing ratio of 50%, respectively. Then, these frames were suspended in the aerobic tank of a full-scale WWTP (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 m3 d−1, modified AAO process) and anchored with bricks to the bottom. The frames were inspected weekly to monitor biofilm development and cleaned to remove surface impurities. Mature biofilms were successfully formed on all fillers after three months.
000 m3 d−1, modified AAO process) and anchored with bricks to the bottom. The frames were inspected weekly to monitor biofilm development and cleaned to remove surface impurities. Mature biofilms were successfully formed on all fillers after three months.
Experimental setup.
Four cylindrical reactors were constructed to represent EB1, EB2, B1, and B2. Each cylinder had a working volume of 1 L, with a diameter of 6.6 cm and a height of 33.0 cm. The inflow and outflow ports were positioned at the bottom and at a height of 26.5 cm. The embedded PAC fillers were added to EB1 and B1, while non-embedded PAC fillers were added to EB2 and B2. Carbon felts (5 cm) were positioned at the bottom and top of EB1 and EB2 and connected by titanium wires to form a closed loop (Fig. 1). An adjustable 0–9999 Ω resistance box (1000 Ω) was linked into the loop. A data collector (DAQM-4200) was connected to a computer for real-time current and voltage acquisition. The hydraulic retention time (HRT) was controlled at 14 h using a peristaltic pump (LHZW006). All reactors were wrapped in black plastic sheets to prevent algal growth.
 |
| | Fig. 1 Experimental setup. | |
2.2 Simulated wastewater
The experiment was divided into four phases to simulate the effluent mixture of the grit chamber and secondary clarifier. The synthetic wastewater contained CH3COONa·3H2O as the carbon source, NH4Cl and KNO3 as the nitrogen sources, and KH2PO4 as the phosphorus source. The solution was supplemented with 40 mg L−1 CaCl2·2H2O, 375 mg L−1 NaHCO3, 80 mg L−1 MgSO4, and 0.3 mL L−1 of trace element solution. The trace element solution was formulated as 1.5 g L−1 FeCl3·6H2O, 0.15 g L−1 H3BO3, 0.03 g L−1 CuSO4·5H2O, 0.18 g L−1 KI, 0.12 g L−1 MnCl2·4H2O, 0.06 g L−1 Na2MoO4·2H2O, 0.12 g L−1 ZnSO4·7H2O, 0.15 g L−1 CoCl2·6H2O and 10 g L−1 EDTA. The detailed composition is shown in Table 1.
Table 1 Wastewater composition and experimental operation
| Experimental phase |
NH4+–N (mg L−1) |
NO3−–N (mg L−1) |
COD (mg L−1) |
C/N |
Time (d) |
VHL (mg N L−1 h−1) |
| I |
10 |
8 |
60 |
3.33 |
1–45 |
1.29 |
| II |
12 |
0 |
40 |
3.33 |
46–69 |
0.86 |
| III |
0.24 |
10 |
30 |
2.93 |
70–94 |
0.73 |
| IV |
13 |
8 |
77 |
3.67 |
95–120 |
1.50 |
2.3 Analytical methods
Samples were collected daily and filtered through 0.45 μm membranes prior to analysis. Concentrations of NH4+–N, NO2−–N, NO3−–N, COD, and MLSS were determined using standard methods.9 pH and dissolved oxygen (DO) were measured using a portable multi-parameter water quality analyzer (Bante900P). Morphological observation was conducted using an optical microscope (BX51TF) and a scanning electron microscope (SEM) (Model ULTRA 55). At the end of the experiment, biofilms were harvested from the filler surface of each reactor, as well as from the anode surface of EB1 and EB2. High-throughput sequencing was carried out to investigate the distribution of microbial species by the Bio-Engineering (Shanghai) Co., Ltd. The volumetric nitrogen loading rate (NLR) calculation was as follows:| | 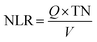 | (1) |
| | 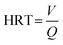 | (2) |
| | 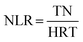 | (3) |
where NLR is the volumetric nitrogen loading rate (mg N L−1 h−1); Q is the inlet flow rate (m3 h−1); V is the reactor volume (m3); TN is the total nitrogen concentration (mg L−1); HRT is the hydraulic retention time (h).
2.4 Kinetic test
Forty grams (wet weight) of fillers were collected, rinsed with deionized water and added to three 250 mL conical flasks labeled A, B and C. Flask A was filled with 150 mL of a solution containing NH4Cl (NH4+–N 20 mg L−1) and NaHCO3 (375 mg L−1), and then aerated for 48 h. Flask B was filled with 150 mL solution of CH3COONa (COD 200 mg L−1) and KNO3 (NO3−–N 20 mg L−1). N2 was purged for 5 min to create anaerobic conditions, and then the flask was sealed and placed in a water bath oscillator (THZ-92C) for 48 h. Flask C contained 150 mL of a mixed solution of NH4Cl, KNO3, and NaNO2 (each at 50 mg L−1) and was operated identically to flask B. The addition of NO3−–N might minimally cause endogenous partial denitrification. Samples were taken at 0, 4, 8, 24 and 48 h. The mixtures were filtered to measure MLSS. The specific nitrification rate (SNR) and specific denitrification rate (SDNR) were calculated based on the slope of the concentration–time curves divided by MLSS. The specific anaerobic ammonia oxidation rate (SAAOR) was calculated to represent the anammox activity.10
2.5 Nitrogen conversion pathways
Five primary nitrogen conversion pathways were considered in the biological wastewater treatment process, including anammox, partial denitrification (PD) (NO3−–N → NO2−–N), partial nitrification (PN) (NH4+–N → NO2−–N), the second step of denitrification (SD) (NO2−–N → N2) and the second step of nitrification (SN) (NO2−–N → NO3−–N). The progress of each conversion pathway was calculated as follows:11| | | NH4+ + 1.32NO2− + 0.66HCO3− + 0.13H+ → 0.66CH2O0.5N0.15 + 1.02N2 + 0.263NO3− + 2.03H2O | (4) |
| | | NO3− + 0.083C6H12O6 → NO2− + 0.5CO2 + 0.5H2O | (5) |
| | | NH4+ + 1.5O2 → NO2− + H2O + 2H+ | (6) |
| | | NO2− + 0.125C6H12O6 + H+ → 0.5N2 + 0.75CO2 + 1.75H2O | (7) |
The final nitrogen conversion pathways were determined by combining the experimental data with reasonable calculation results.
where
x,
y and
z are the reaction progress of the three selected conversion pathways, mmol;
a,
b, and
c are the stoichiometric coefficients of NH
4+–N in each selected equation;
d,
e, and
f are the stoichiometric coefficients of NO
2−–N in each selected equation;
g,
h, and
i are the stoichiometric coefficients of NO
3−–N in each selected equation; and A, B, and C are the changes in NH
4+–N, NO
2−–N and NO
3−–N in each layer of the biofilter, mg L
−1.
3. Results and discussion
3.1 Pollutant removal efficiency
Nitrogen removal performance throughout the experiment is illustrated in Fig. 2. During the initial 25 days, at an influent NH4+–N concentration of 10 mg L−1, the effluent NH4+–N fluctuated significantly and occasionally exceeded the influent levels, resulting in the average effluent NH4+–N during the first phase being higher than the influent. This instability was likely caused by endogenous NH4+–N release from the preexisting biofilm.12,13 With the adaptation of the biofilm to the electrochemical environment, as indicated by the output voltages of EB1 and EB2, the initiation of the electrochemical system coincided with an improvement in NH4+–N removal, highlighting the role of electrochemically active bacteria in microbial niche competition via selective enrichment.14 Specifically, EB1 exhibited a higher stable voltage (325 mV) compared to EB2 (275 mV), which was attributed to the lower internal resistance of PAC-embedded fillers.15 During phase II, NH4+–N was the sole nitrogen source, and effluent NH4+–N exhibited a decreasing trend across all reactors. Since denitrification relied on NO2−–N and NO3−–N produced by nitrification, and nitrification was inhibited during the initial second phase without aeration, denitrification was also inhibited. The TN removal efficiency was initially low but gradually recovered to 44.11–78.85%. During phase III, NO3−–N was the dominant nitrogen source. Considering assimilation, approximately 8.58 g COD was required to completely reduce 1 g of NO3−–N to N2;16 therefore, denitrification was suppressed due to insufficient COD. During phase IV, the TN removal efficiencies of both EB1 and EB2 exceeded those of B1 and B2. Although influent COD remained the highest among the four phases, NH4+–N removal performance was consistently maintained. This was attributed to anammox activity, as NO2−–N accumulation was observed in all reactors, and its TN removal efficiency was also the highest among all four phases.
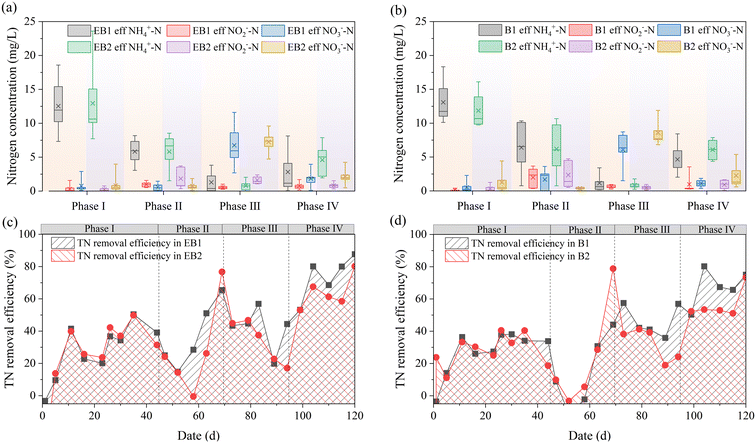 |
| | Fig. 2 Nitrogen removal performance of each reactor: (a) EB1; (b) EB2; (c) B1; (d) B2. | |
The COD removal performance is illustrated in Fig. 3. During the first ten days, COD utilization remained low across all reactors, with removal efficiencies of 28.70–36.45%. Specifically, the PAC-embedded reactors (EB1/B1) exhibited an earlier peak in COD removal. This improvement could be attributed to the high surface area of PAC that facilitated rapid biofilm formation and retained COD for subsequent microbial degradation.17,18 During phase II, the average COD removal efficiencies were 57.92 ± 15.84% and 62.70 ± 10.18% for EB1 and EB2, and 56.06 ± 8.99% and 63.00 ± 8.74% for B1 and B2, respectively. The embedded PAC did not influence COD removal significantly. The third phase was characterized by limited organic carbon and high NO3−–N. Consequently, COD utilization rates declined compared with those in other phases. During phase IV (115–120 d), all reactors achieved COD removal efficiencies of over 80%, with minimal inter-reactor variation. This was due to the abundant carbon sources and the enrichment of diverse microorganisms.
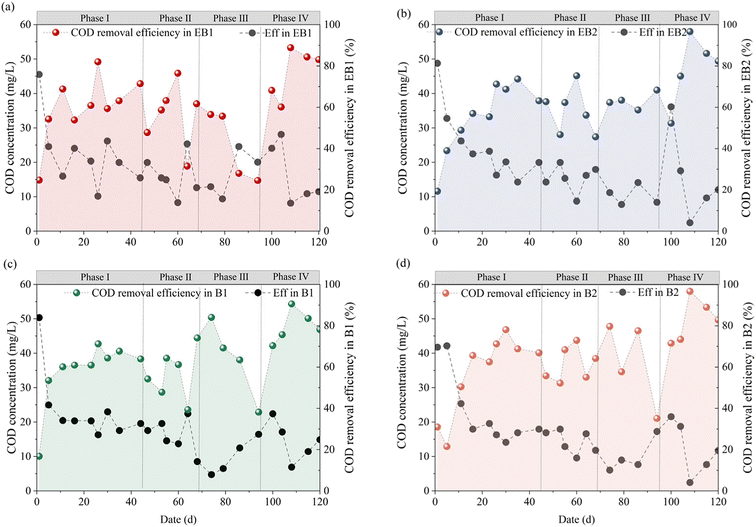 |
| | Fig. 3 COD removal efficiency of each reactor: (a) EB1; (b) EB2; (c) B1; (d) B2. | |
3.2 Nitrogen removal kinetics under different influent water qualities
To further investigate the influence of water quality on nitrogen conversion pathways, nitrogen removal kinetic tests were conducted at the end of each phase (Fig. 3). During phase I, nitrification activity was primarily influenced by the filler type and also the electrochemical system. Specifically, the embedded PAC fillers exhibited significantly higher nitrification activity. The SNRs of EB1 and B1 fillers increased by 84.85% and 48.72%, respectively, compared to EB2 and B2. Notably, EB1 and B1 exhibited similar SNRs, as did EB2 and B2. Denitrification was stable with the SDNR of 0.86–0.97 mg (g d)−1 in all fillers. During phase II, the SNR of all reactors increased under reduced influent COD. Compared to phase I, the SNR improved despite the reduced NLR, which was attributed to increased NH4+–N loading rates.19 During phase III, the SDNRs of EB1 and B1 were 161.90% and 120.00% higher than those of EB2 and B2. This was attributed to the conductive carbon matrix of the embedded PAC, which established a three-dimensional electron transfer network that facilitated inter-bacterial electron transfer and provided micro-environments conducive to localized carbon retention and microbial colonization.20 However, the electrochemical system suppressed denitrification, with the SDNRs of EB1 and EB2 being 28.57% and 40.00% lower than those of B1 and B2, respectively. This was because NO3−–N was directly reduced at the cathode, thereby inhibiting heterotrophic denitrification on the biofilm.21
The surface and cross-sectional morphology of the embedded PAC fillers is depicted in Fig. 4. Irregular fragments were observed on the surface, which were probably residual tiny PAC (Fig. 4(a)). The embedded PAC fillers exhibited a cross-linked network structure, which facilitated the transport of the substrate and oxygen (Fig. 4(b)). During phase IV, the SNR of all reactors increased to 0.73–1.01 mg (g d)−1 under high influent NH4+–N. The SNRs of EB2 and B2 were 19.76% and 15.51% higher than those of EB1 and B1, respectively (Fig. 5). Similar to phase I, the electrochemical system had little influence on nitrification activity, while the embedded PAC fillers exhibited superior denitrification performance. The SDNRs of EB1 and B1 fillers were 23.99% and 32.56% higher than those of EB2 and B2, respectively. The NLR (1.50 mg N L−1 h−1) and C/N (3.67) during this phase were the highest among all four phases, resulting in a higher SNR, SDNR and SAAOR. Interestingly, anammox contributed more to nitrogen removal during phase IV with the embedded PAC filler exhibiting better anammox performance. Specifically, the SAAORs of EB1 and B1 fillers were 81.39% and 476.36% higher than those of EB2 and B2, respectively. Moreover, the electrochemical system enhanced the anammox performance significantly,22 the SAAORs of EB1 and B1 fillers were 204.63% and 867.93% higher than those of EB2 and B2, respectively.
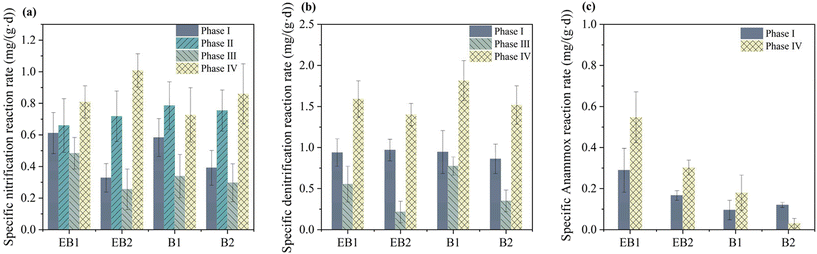 |
| | Fig. 4 Specific nitrogen reaction rate during each phase: (a) nitrification; (b) denitrification; (c) anammox. | |
 |
| | Fig. 5 Morphology of the embedded PAC filler: (a) surface; (b) cross section. | |
3.3 Contribution of nitrogen conversion pathways
The major nitrogen conversion pathways were identified as described in section 2.5, and their contributions to the overall nitrogen conversion progress are illustrated in (Fig. 6). During phase I, PN, PD, and SD were the dominating pathways. The PAC-embedded fillers exhibited higher PN contributions of 13.72% and 9.91% in EB1 and B1, respectively, compared to 10.94% and 4.08% in EB2 and B2. This trend was consistent with the variation of the SNR described in section 3.2. Since there were COD and abundant NO3−–N in the influent, PD was the predominant pathway, although its contribution varied minimally among all reactors (38.06–47.71%). The ΔCOD/ΔTIN (4.4 to 6.2 mg COD/mg N) exhibited higher COD demand in phase I, indicating that nitrogen removal was driven primarily by heterotrophic denitrification. The PAC and electrodes reduced COD demand to varying degrees.
 |
| | Fig. 6 The contribution of major nitrogen conversion pathways: (a) phase I; (b) phase II; (c) phase III; (d) phase IV. | |
During phase II, the dominant conversion pathways were PN, SN and SD. The average contributions of both PN and SN in B1 and B2 fillers were slightly higher than those in EB1 and EB2. Although the electrochemical system slightly inhibited the oxidation pathways,23 it enhanced reduction pathways like SD. EB2 exhibited the highest ΔCOD/ΔTIN value, while B1 showed the lowest value (Fig. 7). Under NH4+–N dominant conditions, NO3−–N was primarily generated through nitrification. The relatively high ΔCOD/ΔTIN in EB2 was associated with weaker coupling of nitrification and denitrification, or greater conversion of COD by assimilation or loss by less efficient heterotrophic pathways.
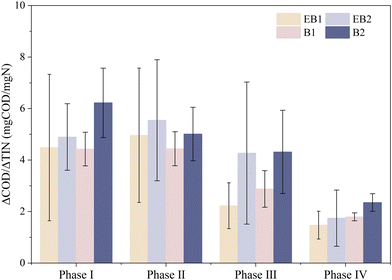 |
| | Fig. 7 ΔCOD/ΔTIN during each phase. | |
During phase III, NO3−–N was the sole nitrogen source, and the primary nitrogen conversion pathways were PD, SD and SN. Embedded PAC fillers improved COD utilization, and the SD contributions of EB1 and B1 (44.73% and 46.08%) were slightly higher than those of EB2 and B2 (40.82% and 45.68%). Although the electrochemical system inhibited denitrification, the SD contributions of EB1 and EB2 were lower than those of B1 and B2. This was attributed to the abundant electron acceptors in the influent, which enhanced oxidation processes. The ΔCOD/ΔTIN values of EB1 (2.23 mg COD/mg N) and B1 (2.88 mg COD/mg N) were significantly lower than those of EB2/B2 (4.27–4.32 mg COD/mg N), with EB2 and B2 being 91.7% and 29.2% higher than EB1 and B1, respectively. This indicated the coupling of PD and anammox, along with electrode/PAC-assisted electron transfer.
During phase IV, the main nitrogen conversion pathways were PD, SD and anammox. The average PD contributions of EB1 and EB2 were slightly lower than those of B1 and B2, respectively, which was consistent with the results of phase I. Following the enrichment over the three phases, the average anammox contribution reached 43.56%, 39.18%, 36.82% and 32.77% for EB1, EB2, B1, and B2, respectively, which was consistent with that of SAAOR, as described in section 3.2. During phase IV, the influent electron donors and acceptors were more balanced in the influent, enhancing both oxidation and reduction processes.24 The anammox contribution in EB1 was 10.79% higher than that in B2, indicating that the electrochemical system with the embedded PAC filler could effectively promote anammox activity. Under relatively high COD and extended operating duration, a large number of electrochemically active bacteria and anammox were enriched, with multiple pathways coexisting. EB1 maintained the lowest ΔCOD/ΔTIN ratio at approximately 1.47. This indicated that under mature conditions, EB1 achieved optimal carbon economy while removing nitrogen, which further demonstrated the enrichment effect of the electrochemical system and embedded PAC on anammox under specific influent water quality.
3.4 Microbial community structure
To elucidate the high anammox contribution during phase IV, microbial communities on different fillers and anode plates were further analyzed (Fig. 8). At the phylum level, the dominant microorganisms on all fillers were Chloroflexi (6.18–11.96%), Bacteroidota (29.84–36.57%), Proteobacteria (23.73–38.02%), Acidobacteriota (2.36–3.45%), Planctomycetota (2.29–3.53%), and Desulfobacterota (0.54–2.58%). Chloroflexi was potentially involved in partial denitrification, exhibiting a relative abundance of 11.95% in EB1 compared to 17.03% in B1, aligning with the results described in section 3.2. Furthermore, more anammox bacteria (AnAOB) were enriched on the PAC-embedded fillers. The relative abundance of Candidatus_Brocadia was 0.42% and 0.61% in EB1 and B1, respectively, compared to those of 0.25% and 0.49% in EB2 and B2.
 |
| | Fig. 8 Distribution of microbial communities: (a) phylum level of fillers; (b) genus level of fillers; (c) genus level of anode plates. | |
At the genus level, the predominant ammonia oxidizing bacterium (AOB) of each filler was Nitrosomonas (0.001–0.10%), while the main nitrite oxidizing bacterium (NOB) was Nitrospira (0.05–0.21%). The abundance of nitrifying bacteria on the non-embedded PAC fillers was higher, which was associated with the higher SNR of EB2 and B2. For the PAC embedded fillers, the electrochemical system enriched more Nitrosomonas (0.03% for EB1 and 0.001% for B1) and Nitrospira (0.07% for EB1 and 0.05% for B1). The dominant denitrifying genera on each filler were Thauera (1.07–4.76%), Comamonadaceae (0.08–0.63%), and Dechloromonas (0.02–0.30%). Similar to nitrifiers, denitrifiers were more abundant on non-embedded PAC fillers (EB2 and B2). However, this contrasted with the SDNR of EB2 and B2, indicating that denitrification was influenced not only by denitrifying bacterial abundance but also environmental factors such as water quality, activity of the denitrifying bacteria, pH, DO, and so on.25,26 Additionally, for the PAC-embedded fillers, the electrochemical system enriched more Thauera, Comamonadaceae and Dechloromonas. Thauera was essential in the PD process,27 and its high relative abundance in all fillers indicated the good potential of PD performance, which was consistent with the contribution to nitrogen conversion pathways. Furthermore, NO2−–N was produced via PD, which was a critical substrate for anammox.28 Both Candidatus_Anammoximicrobium and Candidatus_Brocadia were found in all fillers; the relative abundance of Candidatus_Anammoximicrobium remained below 0.01% in all samples and Candidatus_Brocadia was 0.49%, 0.61%, 0.25% and 0.42% in EB1, EB2, B1 and B2, respectively. Similar to nitrifying and denitrifying bacteria, the embedded PAC fillers also inhibited AnAOB, and the electrochemical system enriched more AnAOB on the embedded PAC fillers.
The microbial communities on EB1 and EB2 anodes at the genus level were further analyzed. Common electrochemically active bacteria, including Geobacter, Desulfuromonas and Pseudomonas, were successfully enriched on the anodes,29 with the anode paired with embedded PAC fillers exhibiting superior enrichment. The relative abundances of Geobacter, Desulfuromonas and Pseudomonas were 14.10%, 10.85%, 17.53% in EB1-A and 11.64%, 5.88%, 15.15% in EB2-A, respectively. Geobacter was capable of producing a ciliated nanowire to transfer electrons from microbial cells to the anode surface.30Desulfuromonas could utilize acetate as the electron donor to generate a potential difference in a short period.31Pseudomonas was involved in electron transfer during NO3−–N reduction.32 The total relative abundance of electrochemically active bacteria in EB1-A (42.48%) was higher than that in EB2-A (32.67%),32 and they were also enriched in both EB1-A (2.20%) and EB2-A (4.74%). Candidatus_Brocadia was significantly enriched on both EB1-A and EB2-A (2.82% and 1.03%, respectively).
These findings revealed the influence of both filler composition and the electrochemical system on the microbial community structure and nitrogen removal performance. The experiments were conducted under stable water quality conditions during long-term operation. However, the robustness of the electrochemical system under short-term water quality shocks or flow fluctuations and strategies to guarantee stable treatment performance remained unclear. Additionally, the simulated wastewater in this study was based on different mixing ratios of the effluents from the grit chamber and secondary clarifier. Whether adjusting the mixing ratio could regulate nitrogen conversion pathways and optimize nitrogen removal performance also requires further in-depth investigation.
4. Conclusion
This study developed an electrochemical system incorporating PVA–SA fillers embedded with PAC to treat low C/N wastewater and systematically evaluated nitrogen conversion pathways under varying nitrogen sources. The influent NH4+–N (C/N = 3.33) enhanced PN and SD, while the influent NO3−–N (C/N = 2.93) promoted SD. When the influent was 13 mg L−1 NH4+–N, 8 mg L−1 NO3−–N and 77 mg L−1 COD, the electrochemical system exhibited greater contribution of anammox to nitrogen removal, with the enrichment of Candidatus_Brocadia on both fillers and the anode. The sum relative abundance of electrochemically active bacteria like Geobacter, Desulfuromonas and Pseudomonas on the anode was 32.67–42.48%. This study provided theoretical and technical guidance for enhancing electron transfer to improve nitrogen removal under low C/N conditions. Further research should focus on controlling the nitrogen conversion pathways by regulating the influent concentrations of electron acceptors/donors, reducing operation time through optimizing influent mixing ratios to accelerate microbial succession, and developing fillers with enhanced mass transfer and electrical conductivity to improve biofilm attachment.
Conflicts of interest
The authors declare no known competing financial interests or personal relationships that could have influenced the work reported in this paper.
Data availability
Data sets generated during the current study are available from the corresponding author on reasonable request.
Acknowledgements
This research was supported by the National Natural Science Foundation of China (42377375) and the Science and Technology Foundation of Henan Province (232102321051).
References
- S. J. You, N. Q. Ren, Q. L. Zhao, P. D. Kiely, J. Y. Wang and F. L. Yang,
et al., Improving phosphate buffer-free cathode performance of microbial fuel cell based on biological nitrification, Biosens. Bioelectron., 2009, 24(12), 3698–3701 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/pii/S0956566309002772.
- F. Ling, Y. Lu, C. Wang, Z. Yuan, R. Yu and G. Zhu, Electron transfer pathways and kinetic analysis of cathodic simultaneous nitrification and denitrification process in microbial fuel cell system, Environ. Res., 2020, 186, 109505 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/pii/S0013935120303984.
- C. Zhao, Y. Wang, S. Meng, W. Zhang, X. Zhang and L. Yan,
et al., Solid slow-release carbon source assembled microbial fuel cell for promoting superior nitrogen removal in an aerobic granular sludge bioreactor, J. Environ. Manage., 2023, 325, 116430 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/pii/S0301479722020035.
- J. Zhang, Z. Zhang, K. Rong, H. Guo, J. Cai and Y. Xing,
et al., Simultaneous Anaerobic Ammonium Oxidation and Electricity Generation in Microbial Fuel Cell: Performance and Electrochemical Characteristics, Processes, 2022, 10(11), 2379 CrossRef CAS , Available from: https://www.mdpi.com/2227-9717/10/11/2379.
- C. Wang, Y. Liu, W. Lv, S. Xia, J. Han and Z. Wang,
et al., Enhancement of nitrogen removal by supplementing fluidized-carriers into the aerobic tank in a full-scale A2/O system, Sci. Total Environ., 2019, 660, 817–825 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/pii/S004896971930052X.
- T. Lou, Z. Peng, K. Jiang, N. Niu, J. Wang and A. Liu, Nitrogen removal characteristics of biofilms in each area of a full-scale AAO oxidation ditch process, Chemosphere, 2022, 302, 134871 CrossRef CAS , Available from: https://www.sciencedirect.com/science/article/pii/S0045653522013649.
- A. R. Massoompour, S. M. Borghei and M. Raie, Enhancement of biological nitrogen removal performance using novel carriers based on the recycling of waste materials, Water Res., 2020, 170, 115340 CrossRef CAS , Available from: https://www.sciencedirect.com/science/article/pii/S0043135419311145.
-
A. A. Rohmah and A. S. Purnomo, The Addition of Different Adsorbents In SA-PVA Matrix For Bacillus Subtilis Immobilization on Methylene Blue Decolorization, in IOP Conference Series: Earth and Environmental Science, IOP Publishing, 2022, p. 012046, Available from: https://iopscience.iop.org/article/10.1088/1755-1315/995/1/012046/meta Search PubMed.
- A. D. Eaton, L. S. Clesceri, A. E. Greenberg and M. A. H. Franson, Standard methods for the examination of water and wastewater, Am. J. Public Health, 1966, 56(3), 387–388 CrossRef.
- T. Lotti, W. R. L. Van Der Star, R. Kleerebezem, C. Lubello and M. C. M. Van Loosdrecht, The effect of nitrite inhibition on the anammox process, Water Res., 2012, 46(8), 2559–2569 CrossRef CAS , Available from: https://www.sciencedirect.com/science/article/pii/S0043135412000991.
- Z. Peng, M. Liu, T. Li, W. Zhang, Y. Wang and L. Yu,
et al., A Novel Layered and Advanced Nitrogen Removal Filter with Gravel and Embedded Bio-Organic Carrier Based on Autotrophic and Heterotrophic Pathways, Water, 2023, 15(10), 1919 CrossRef CAS , Available from: https://www.mdpi.com/2073-4441/15/10/1919.
- C. Duan, Q. Zhang, J. Li, W. Feng, L. Zhang and Y. Peng, Partial nitrification response to dissolved oxygen variation and aerobic starvation: Kinetics and microbial community analyses, Chem. Eng. J., 2024, 481, 148621 CrossRef CAS , Available from: https://www.sciencedirect.com/science/article/pii/S1385894724001062.
- G. Liu and J. Wang, Long-Term Low DO Enriches and Shifts Nitrifier Community in Activated Sludge, Environ. Sci. Technol., 2013, 47(10), 5109–5117, DOI:10.1021/es304647y.
- R. K. Goud and S. V. Mohan, Prolonged applied potential to anode facilitate selective enrichment of bio-electrochemically active Proteobacteria for mediating electron transfer: microbial dynamics and bio-catalytic analysis, Bioresour. Technol., 2013, 137, 160–170 CrossRef , Available from: https://www.sciencedirect.com/science/article/pii/S0960852413004276.
- A. ElMekawy, H. M. Hegab, X. Dominguez-Benetton and D. Pant, Internal resistance of microfluidic microbial fuel cell: challenges and potential opportunities, Bioresour. Technol., 2013, 142, 672–682 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/pii/S0960852413008213.
- Y. Chen, B. Li, L. Ye and Y. Peng, The combined effects of COD/N ratio and nitrate recycling ratio on nitrogen and phosphorus removal in anaerobic/anoxic/aerobic (A2/O)-biological aerated filter (BAF) systems, Biochem. Eng. J., 2015, 93, 235–242 CrossRef CAS , Available from: https://www.sciencedirect.com/science/article/pii/S1369703X14002897.
- O. K. Choi, M. S. Kim and J. W. Lee, Synergistic role of powdered activated carbon (PAC) for aerobic granular sludge (AGS) formation and organic micropollutant (OMPs) removal, Process Saf. Environ. Prot., 2024, 189, 80–88 CrossRef CAS , Available from: https://www.sciencedirect.com/science/article/pii/S0957582024007067.
- S. Ozmihci and F. Kargi, Utilization of powdered waste sludge (PWS) for removal of textile dyestuffs from wastewater by adsorption, J. Environ. Manage., 2006, 81(3), 307–314 CrossRef CAS PubMed.
- W. Lin, J. Tang, L. Zheng, H. Qin and X. Cai, Influence of C/N ratio on biological denitrification in the context of electro-anammox, J. Environ. Chem. Eng., 2025, 13(5), 117474 CrossRef CAS.
- Y. Dong, H. Yuan, D. Ge and N. Zhu, A novel conditioning approach for amelioration of sludge dewaterability using activated carbon strengthening electrochemical oxidation and realized mechanism, Water Res., 2022, 220, 118704 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/pii/S0043135422006571.
- A. Gurung, B. S. Thapa, S. Y. Ko, E. Ashun, U. A. Toor and S. E. Oh, Denitrification in microbial fuel cells using granular activated carbon as an effective biocathode, Energies, 2023, 16(2), 709 CrossRef CAS , Available from: https://www.mdpi.com/1996-1073/16/2/709.
- T. Wang, M. Chen, J. Zhu, N. Li and X. Wang, Anodic ammonium oxidation in microbial electrolysis cell: Towards nitrogen removal in low C/N environment, Water Res., 2023, 242, 120276 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/pii/S0043135423007121.
- L. Zhong, S. S. Yang, J. Ding, G. Y. Wang, C. X. Chen and G. J. Xie,
et al., Enhanced nitrogen removal in an electrochemically coupled biochar-amended constructed wetland microcosms: The interactive effects of biochar and electrochemistry, Sci. Total Environ., 2021, 789, 147761 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/pii/S0048969721028321.
- J. Zhen, M. Zheng, W. Wei, S. Q. Ni and B. J. Ni, Extracellular electron transfer (EET) enhanced anammox process for progressive nitrogen removal: A review, Chem. Eng. J., 2024, 482, 148886 CrossRef CAS , Available from: https://www.sciencedirect.com/science/article/pii/S1385894724003711.
- H. Rasouli-Kenari, M. H. Sarrafzadeh and M. R. Mehrnia, Factors affecting the biological nitrogen removal from wastewater in simultaneous nitrification–denitrification process, J. Biotechnol., 2008, 136, S670–S671 CrossRef , Available from: https://www.sciencedirect.com/science/article/pii/S016816560801821X.
- T. Sirivedhin and K. A. Gray, Factors affecting denitrification rates in experimental wetlands: Field and laboratory studies, Ecol. Eng., 2006, 26(2), 167–181 CrossRef , Available from: https://www.sciencedirect.com/science/article/pii/S0925857405001850.
- R. Du, S. Cao, B. Li, M. Niu, S. Wang and Y. Peng, Performance and microbial community analysis of a novel DEAMOX based on partial-denitrification and anammox treating ammonia and nitrate wastewaters, Water Res., 2017, 108, 46–56 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/pii/S0043135416308041.
- Y. Zhu, D. Li, S. Qi, S. Yang, J. Zhang and W. Wang,
et al., Anticipating on potential electron acceptors for Anammox, Chem. Eng. J., 2023, 467, 143458 CrossRef CAS , Available from: https://www.sciencedirect.com/science/article/pii/S1385894723021897.
- L. Huang and G. L. Puma, Physiological response of electroactive bacteria via secretion of extracellular polymeric substances in microbial electrochemical processes: A review, Curr. Opin. Electrochem., 2022, 36, 101168 CrossRef CAS , Available from: https://www.sciencedirect.com/science/article/pii/S2451910322002332.
- N. S. Malvankar and D. R. Lovley, Microbial nanowires for bioenergy applications, Curr. Opin. Biotechnol., 2014, 27, 88–95 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/pii/S0958166913007180.
- K. P. Katuri, S. Kamireddy, P. Kavanagh, A. Muhammad, P. Ó. Conghaile and A. Kumar,
et al., Electroactive biofilms on surface functionalized anodes: The anode respiring behavior of a novel electroactive bacterium, Desulfuromonas acetexigens, Water Res., 2020, 185, 116284 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/science/article/pii/S0043135420308204.
- J. Xu, H. Pang, J. He, J. Nan, M. Wang and L. Li, Start-up of aerobic granular biofilm at low temperature: Performance and microbial community dynamics, Sci. Total Environ., 2020, 698, 134311 CrossRef CAS PubMed , Available from: https://www.sciencedirect.com/sciensce/article/pii/S0048969719343013.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  *a,
Meiqi
He
a,
Yingjian
Zhang
a,
Likun
Gu
b,
Yan
Wang
b,
Liyun
Sun
a and
Lulu
Cheng
c
*a,
Meiqi
He
a,
Yingjian
Zhang
a,
Likun
Gu
b,
Yan
Wang
b,
Liyun
Sun
a and
Lulu
Cheng
c
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 m3 d−1, modified AAO process) and anchored with bricks to the bottom. The frames were inspected weekly to monitor biofilm development and cleaned to remove surface impurities. Mature biofilms were successfully formed on all fillers after three months.
000 m3 d−1, modified AAO process) and anchored with bricks to the bottom. The frames were inspected weekly to monitor biofilm development and cleaned to remove surface impurities. Mature biofilms were successfully formed on all fillers after three months.