Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Introducing BPA-equivalents: assessing mixture toxicity and substitution of BPA in environmental exposure scenarios
Vanessa
Srebny
a,
Georg
Braun
a,
Niklas
Wojtysiak
a and
Beate. I.
Escher
 *ab
*ab
aDepartment of Cell Toxicology, Helmholtz Centre for Environmental Research – UFZ, Permoser Str. 15, 04318 Leipzig, Germany. E-mail: beate.escher@ufz.de
bEnvironmental Toxicology, Department of Geosciences, Eberhard Karls University Tübingen, Schnarrenberger Str. 94-96, 72076, Tübingen, Germany
First published on 13th February 2026
Abstract
Bisphenol A (BPA) is a plastic chemical that has been phased out from many applications because it acts as endocrine disruptor. Numerous structurally similar replacements are already in use, and environmental monitoring data indicates exposure of aquatic ecosystems to mixtures of BPA and its alternatives. As many replacement chemicals elicit similar modes of action and some even have higher relative effect potencies than BPA, it is likely that BPA and its alternatives act together in mixtures. Mixtures of BPA alternatives in concentration ratios as they were detected in the surface water across Europe showed effects that were consistent with the mixture prediction model of concentration addition in in vitro bioassays for cytotoxicity, estrogenicity and mitochondrial toxicity. Even partial agonists contributed to estrogenic effects. In case of the activation of the aryl hydrocarbon receptor, most of the mixture components were not specifically active but rather acted as baseline toxicants and the AhR activation was identified as a cytotoxic burst effect. Simulations showed that the apparently AhR-inactive constituents also contributed to the experimental mixture effect at baseline toxic concentrations. We introduced BPA equivalent concentrations (BPA-EQ) or short BPA-equivalents as a simple communication tool to evaluate the contribution of different alternatives to the mixture effects. BPA-EQ enabled comparison of toxicity data of different bisphenol mixtures, and simulation of different replacement scenarios. For instance, when adding five frequently detected BPA alternatives (BPS, BPF, BPAF, BPE and BPB) to BPA, the total surface water concentrations were ten times higher than of BPA alone, but BPA-EQ for cytotoxicity were 24 times and BPA-EQ for estrogenicity were 12 times higher than BPA alone. Further, BPPH dominated the mixture cytotoxicity and BPZ and BPAF the estrogenicity under this real-world scenario. This and other realistic mixtures comprising three to ten bisphenols indicate that structural analogues of BPA should be evaluated as mixture rather than as individual chemicals. Merely changing substituents on the bisphenol core is likely a “regrettable substitution”.
Environmental significanceThe need for mixture risk assessment in chemical regulation is a highly relevant topic. We observed mixture effects under environmentally realistic scenarios, and they can be reliably predicted. When chemicals are phased out due to their hazard properties, many replacement products enter the market immediately. On the example of bisphenol A and nine of its alternatives, we demonstrated that these structurally and toxicologically similar chemicals act together in mixtures. Combined evaluation of similar chemicals is a more realistic predictor for environmental impacts than assessing each chemical alone. Our approach can be extended to other exposure contexts and provides a practical tool supporting the regulation of mixture risks of substitution products as currently discussed in the EU. |
1 Introduction
Bisphenol A is a high production volume chemical that has been banned in many consumer applications in the European Union,1 such as paper receipts2 and food contact materials3 due to its classification as endocrine disruptor for the environment, human health4,5 and subsequent categorisation as substance of very high concern. In response, numerous alternatives such as bisphenol F (BPF) and bisphenol S (BPS) have entered the market to replace BPA. While BPS and BPF have already been recognized as “toxic for reproduction”,6 much less is known about the toxicological profiles of other replacement products, toxicokinetic and toxicodynamic behaviour and long-term risks for the environment and human health (as reviewed in ref. 7–9). Due to their close structural resemblance, many alternatives are expected to display similar endocrine-disrupting properties, which has already been confirmed for several compounds such as BPAF, BPB and BPS.10,11Recent monitoring data suggest that (co-)exposure to BPA and its alternatives already occurs in humans and the environment.7,12,13 However, current risk assessment frameworks overlook the reality of mixture exposures, which results in a systematic underestimation of risk if chemicals act together.14 It is well established that endocrine disruptors can contribute to mixture effects additively15–17 – even below their individual detectable effect levels – a phenomenon often referred to as the “something from nothing” effect.18,19
Studies assessing mixture effects of BPA and multiple BPA alternatives are scarce, often limited in number of components investigated and primarily focused on estrogenicity.20,21 We tested several multi-component mixtures of BPA and up to nine BPA alternatives. The mixtures were designed in the concentration ratios detected in surface water7 in five in vitro bioassays covering diverse modes of action: activation of the estrogen receptor alpha (ERα) and the aryl hydrocarbon receptor (AhR), oxidative stress response (ARE), inhibition of the mitochondrial membrane potential (MMP), and neurite outgrowth inhibition (NOI). These modes of action had been identified as relevant for BPA alternatives,7 and we have previously published the effect data of BPA and 26 alternatives in the same assays.11 These in vitro assays were chosen to test for mixture interaction of diverse modes of action as part of hazard assessment and not to extrapolate to in vivo effects.
To design mixture experiments, and predict potential effects, modelling approaches are required. The two most widely used concepts are concentration addition (CA) introduced by Loewe22 and independent action (IA) described by Bliss.23 Both models assume that the components of the mixture do not interact with each other.
While IA is based on probabilistic response addition where each component contributes via a different mechanism at different target sites to the observed effect, CA assumes a common mode of action and can be interpreted that each component can be seen as a diluted or more concentrated version of the other components. In general, CA has been established as a suitable “default” model for mixture toxicity, as it is often more conservative than IA24 and differences between the two models are typically decreasing as the number of mixture components increases. Deviations from CA and IA in form of synergism or antagonism have been rarely observed,25 but depending on the respective mixture scenario should also not be neglected.26
A practical application of the CA concept is the use of bioanalytical equivalent concentrations (BEQ), also referred to as toxic equivalent concentrations (TEQ), which express the effect of a mixture as concentrations of a reference compound that has the same effect as the mixture by scaling detected concentrations with relative effect potencies (REP).27 Introduced as a special case of CA – also described as “simple similar action”28 – the BEQ concept was developed and continuously applied to facilitate communication and simplify mixture assessments. This approach requires that the log-concentration–response curves be parallel. This implies that the REP is constant across all effect levels and between the mixture components.27
However, in practice, dose–response curves often differ in slope and maximum effect,29 which restricts the applicability of the BEQ concept. This is especially true for the BPA alternatives, which show partial agonism for the estrogen receptor.10,11 Yet, it has been shown that most dose–response curves can be approximated as linear in the low-effect range (<30%), and the REPs are then a ratio of the slopes and not dependent on the effect level.30 With this simplification, the BEQ concept could in theory be applied up to effect levels of 30% for most compounds and endpoints, including BPA and its alternatives, but at higher effect levels more sophisticated mixture models are needed.
Experimental mixture data can be affected by artefacts, and their interpretation is often complicated. False conclusions can result from issues such as missing activity of individual mixture components, solubility limitations or poor single-chemical fits, as well as from specific features of BPA alternatives such as their partial agonism at ERα.
To address these issues, we tested different variants of the CA model and compared their predictive performance with the linear CA approach. The applicability of CA for partial agonism is inherently limited, because predictions are undefined beyond the lowest maximum effect observed for any component. Only a few extended CA frameworks have been proposed to account for such cases,31,32 enabling predictions for mixtures of compounds with varying efficacy.
In this study we demonstrate the applicability of the linear CA model and the BEQ concept for BPA and its alternatives using realistic environmental concentrations to design the mixtures. We compare the prediction quality of different mixture models with varying complexity. We then demonstrate potential applications of the BEQ-based mixture assessment for BPA and its alternatives.
2 Materials and methods
2.1 Chemicals and cells
Full chemical names, abbreviations are given in Table 1 with details in Table S1. The BPA alternatives were all dissolved in methanol as 10 g per L stock solutions (LC-MS grade, Chemsolute, TH Geyer, Renningen Germany) and all sourced from Chiron AS (Trondheim, Norway). All components of the bioassay media, as well as the ERα-UAS-bla GripTite were purchased from Thermo Fisher Scientific (Waltham, MA, U.S.). AREc32 cells were kindly provided by C. Roland Wolf from Cancer Research, UK,33 and AhR CALUX H4L7.5c2 cells34 by Michael Denison, UC Davis, U.S. SH-SY5Y was purchased from Sigma-Aldrich (St. Louis, MO, U.S.) Chemicals needed for cell culture, bioassay detection and reference compounds are listed in Table S2 (reprinted from Srebny et al.11).| Chemical name | Abbr. | Median surface water concentration Ci (µg L−1) | Molar fraction pi in mixture | IC10,median (µM) | EC10,ERα (µM) | EC10,MMP (µM) | EC10,NOI (µM) | EC10,AhR (µM) | ||
|---|---|---|---|---|---|---|---|---|---|---|
| I | II | III | ||||||||
| a >IC10 means that no EC10 could be derived because cytotoxicity masked the effect, or in case of reporter gene assays, a cytotoxicity burst35 could have falsified the effect data. | ||||||||||
| 4,4′-(Propane-2,2-diyl)diphenol | BPA | 0.35 | 0.29 | 70.0 | 0.370 | 28.0 | 50.8 | 264.1 | ||
| 4,4′-Sulfonyldiphenol | BPS | 0.82 | 0.61 | 378.6 | 1.430 | 117.0 | >IC10a | >IC10 | ||
| Bis(4-hydroxyphenyl)methane | BPF | 0.11 | 0.10 | 430.5 | 0.995 | 164.7 | >IC10 | >IC10 | ||
| 4,4′-(1,1,1,3,3,3-Hexafluoropropane-2,2-diyl)diphenol | BPAF | 0.41 | 0.14 | 24.8 | 0.024 | 5.32 | 16.2 | >IC10 | ||
| 4,4′-Ethylidenbisphenol | BPE | 0.52 | 0.28 | 81.9 | 0.401 | 32.1 | 113.8 | 61.9 | ||
| 4,4′-(Butane-2,2-diyl)diphenol | BPB | 1.18 | 0.57 | 53.6 | 0.135 | 16.4 | 28.0 | >IC10 | ||
| 1,1′-Bis(4-hydroxyphenyl)cyclohexane | BPZ | n.d. | 0.25 | 75.8 | 0.044 | 22.1 | 30.5 | 80.4 | ||
| 4,4′-(1-Phenylethylidene)bisphenol | BPAP | n.d. | 0.25 | 63.7 | 0.194 | >IC10 | 51.2 | >IC10 | ||
| 2,2-Bis(2-hydroxy-5-biphenylyl)propane | BPPH | n.d. | 0.25 | 10.6 | >IC10 | 1.96 | >IC10 | 9.5 | ||
| 4-[[4-(Allyloxy)phenyl]sulfonyl]phenol | BPS-MAE | n.d. | 0.25 | 190.0 | 14.1 | 78.5 | 25.2 | >IC10 | ||
2.2 Physicochemical properties and baseline toxicity predictions
All physicochemical data of BPA and 9 alternatives were taken from Srebny et al.11 and, where possible, based on experimentally measured values (Table S1). The baseline toxicity predictions36 and single chemical effect and cytotoxicity data were reprinted from Srebny et al.11 (Table S1). The effect concentrations EC10 represent the absolute 10% effect levels and the inhibitory concentrations IC10 represent absolute 10% cytotoxicity.372.3 Environmental occurrence data
Environmental monitoring data for surface water occurrence of seven BPA alternatives were compiled in the review by Adamovsky et al.7 (Fig. S1). For six substances (BPA, BPS, BPF, BPE, BPB, BPAF), the available data were sufficiently abundant to derive median environmental concentrations (Cmedian, Table S2). For compounds with limited or no occurrence data (BPPH, BPAP, BPS-MAE, BPZ), concentrations were theoretically set to 2.31 nM, corresponding to a fixed share of 10% in a hypothetical mixture containing all ten bisphenols.2.4 Mixture design
The grouping of the bisphenols for mixture toxicity testing (Fig. 1) was done based on the availability of occurrence and bioactivity data (Table 1).7–9 Mixture I included BPA, BPF, and BPS, for which both occurrence and in vitro toxicity data are readily available (Fig. 1a). Mixture II comprises BPAF, BPB, and BPE, which have been detected in surface waters but lack comprehensive in vitro toxicity characterization. Mixture III contains alternatives (BPZ, BPPH, BPAP, BPS-MAE) that have neither been detected in environmental monitoring. The mixtures were also combined to cover all possible unique combinations (I + II, I + III, II + III) as well as a 10-component mixture (I + II + III, Fig. 1a).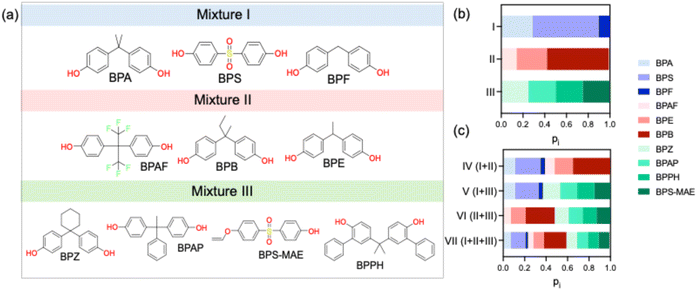 | ||
| Fig. 1 (a) Schematic illustration of the three base mixtures I, II, III with chemical structures of the 10 mixture components (Table S3). (b) Representation of the mixture design and as stacked bar plots for mixture I with BPA, BPS and BPF coloured in blue, mixture II with BPAF, BPE, and BPB coloured in red, mixture III with BPZ, BPAP, BPPH and BPS-MAE in green. (c) The other mixtures I + II, I + III, II + III are combinations of the first three. Mixture I + II + III is a 10-component mixture. Data from Tables 1 and S3. | ||
Molar fractions pi (eqn (1)) were calculated based the concentration Ci of component i and total concentration Ctot in each mixture (eqn (2)) and are reported in Tables 1, S3, Fig. 1b and c.
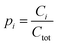 | (1) |
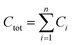 | (2) |
2.5 Preparation of mixture and dosing solutions
The mixtures were prepared by combining the 10 g per L methanolic stock solutions of each bisphenol in proportions defined by the predetermined fraction pi (Table S3). This mixture stock was transferred to conical dosing vials, evaporated, and reconstituted in the respective assay medium. The final concentration in each well corresponded to three times the concentration predicted by the preliminary CA model to elicit a 10% effect. A detailed description of the mixture preparation is provided in Table S4.2.6 Bioassays in 384 well plate format
Bioassay and cell culture procedures for ERα-UAS-bla GripTite, AhR-CALUX, neurite outgrowth inhibition and MitoOxTox have been described extensively in previous studies,11,34,35,38,39 more details are provided in Table S2 and Text S2. The experiments of the single chemicals from Srebny et al.11 (reprinted in Table S1) and the designed mixtures were performed with the same cell lines under identical experimental conditions.Briefly, 2650 to 5000 cells were seeded into 384-well plates, followed by 24 h incubation at 37 °C and 5% CO2. Then, the chemical mixtures were dosed to the cells dissolved in the appropriate medium and incubated again for 24 hours at 37 °C and 5% CO2. Cell viability was monitored both before and after 24 h chemical exposure via cell confluency with an IncuCyte S3 Live-Cell Analysis System (Sartorius AG, Essen, Germany), except for the neurite outgrowth assay, where a live–dead staining with Nuclear Green LcS1 (138904, Abcam, Cambridge, U.K.) and propidium iodide (Abcam, Cambridge, U.K.) was performed. Only concentrations with cell viability >90% (i.e., concentrations < IC10) were included for evaluation of the reporter gene assays ERα, AhR and ARE to avoid artefacts from the cytotoxicity burst.35
2.7 Data evaluation
| y = slope × C | (3) |
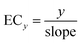 | (4) |
Although this low effect-range is relevant for risk assessment and for mixture effect evaluation of realistic mixtures, we additionally evaluated the full CRC up to the maximum effect (Emax) using a full-range log-logistic model, also called Hill model (eqn (5) with a minimum effect = 0, y = effect, b = Hill slope, EC50,relative median effect concentration). These CRCs were fitted with the function “log(agonist) vs. response – variable slope (four parameters)” in GraphPad Prism, V10.6.1. Since the EC50,relative are not comparable between each different CRCs with different Emax, we converted the EC50,relative to absolute 10% effect concentration, EC10, with eqn (6). In an ideal CRC, the EC10 derived from the linear portion and the full logistic CRC would be identical. However, in practice, they may differ slightly.
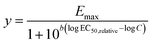 | (5) |
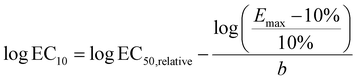 | (6) |
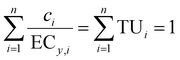 | (7) |
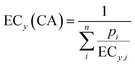 | (8) |
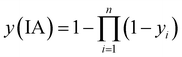 | (9) |
If effects do not exceed 10% and for the linear model with IR, CA and IA converge to the same model (eqn (10)),30 where the slopei refers to the slope of the linear CRC of each component i and slopeCA is the predicted slope of the mixture, which is the sum of the product of pi and the individual slopes slopei. ECy(CA/IA) does not require the distinction between CA and IA and avoids the complication with Emax, because Emax must be at least 10%, else the chemical/mixture is considered inactive.
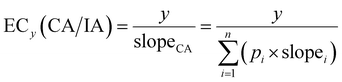 | (10) |
This low effect-level CA/IA model is relevant when looking at realistic environmental concentrations that are typically occurring at much lower effect levels, but not if we want to analyse mixture interactions at high effect levels and high concentrations.
To explore mixture effects and potential interactions at high concentrations, we fitted the raw CRCs of Srebny et al.11 again with the full-range log-logistic model (eqn (5)) and then applied the general CA prediction model (eqn (8)). NOI and MMP were fitted with (eqn (5)) and the Emax fixed to 100% and hence the general CA prediction model (eqn (8)) could be applied incrementally from 0% to 100%. However, in case of ERα activation, the Emax did not reach 100% and was very variable for different single chemicals, which meant that the CA prediction (eqn (8)) can only be performed up to the lowest Emax of the mixture components i.
There are two alternative models that can deal with this issue encountered only for ERα activation: First, the “general concentration addition” model (GCA), which was first proposed by Howard and Webster,31 and adapted by Scholze et al.,32 can be applied for a fixed slope b = 1 and variable Emax (eqn (11)). The predicted Emax that the mixture can reach is defined by eqn (11).
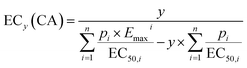 | (11) |
 | (12) |
Second, Scholze et al.32 proposed a model that can deal with variable slopes b and variable Emax, which they called “toxic unit extrapolation” (TUE) approach. This model assumes that once a mixture component reaches 70% of its maximum effect (Emax), the corresponding effect concentration is fixed for all higher effect levels and eqn (7) expands to eqn (13). This model extrapolates the contribution of this component to the overall mixture effect beyond its effect maximum Emax. Beyond this point, i.e., at higher mixture concentrations, its relative contribution is held constant. As a result, the component becomes progressively less influential at higher effect levels but still contributes to the mixture effect y. The extrapolated TUk(0.7Emaxk) can be calculated from the ratio of the fraction pk, total concentration ECy(CA) and the effect concentration at 0.7 Emax. The estimation of ECy(CA) is given in eqn (14).
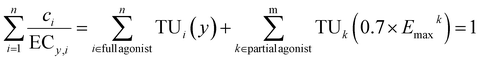 | (13) |
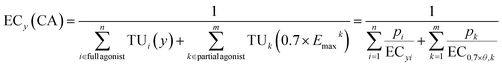 | (14) |
BPA-EQmix of the experimental mixture was derived from the experimental effect concentration EC10,exp of the tested mixture by eqn (15). For easier communication and comparison, we expressed BPA-EQ in mass concentrations. The molecular weight of BPA (MWBPA) is 228.29 g mol−1.
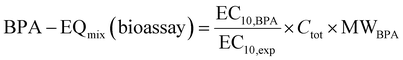 | (15) |
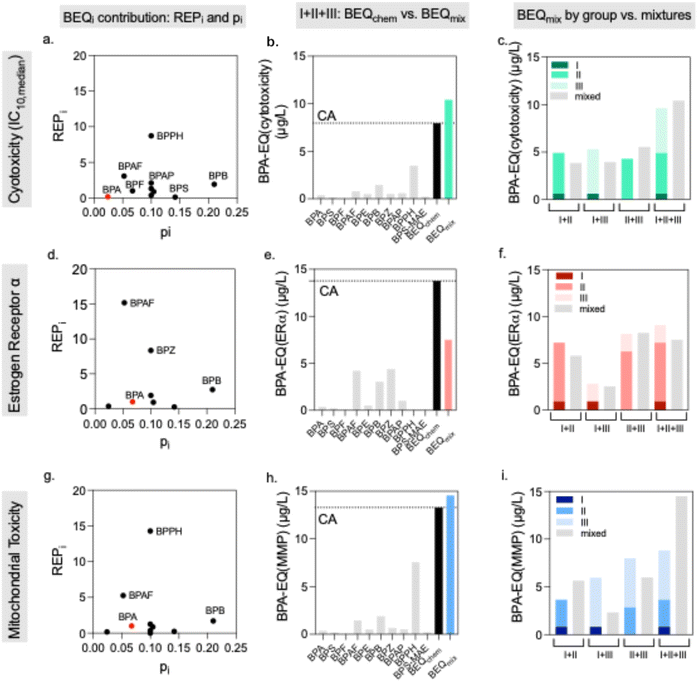
Typically, each bioassay has its specific reference compound. As we used BPA as reference for all bioassays, we indicate the name of the bioassay (abbreviated as cytotoxicity (for the median cytotoxicity IC10,median), ERα, ARE, MMP, NOI, and AhR).
The BPA equivalent concentration BPA-EQchem of the mixture can be predicted from the BPA-EQi of all mixture components i if CA can be confirmed as mixture model and if the slopes of the log CRCs are similar or if the CRC can be fit with the linear CRC model at low effect levels (eqn (6)).30 We used the latter in this study. Then the BPA-EQi simply add up (eqn (16)).37
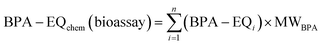 | (16) |
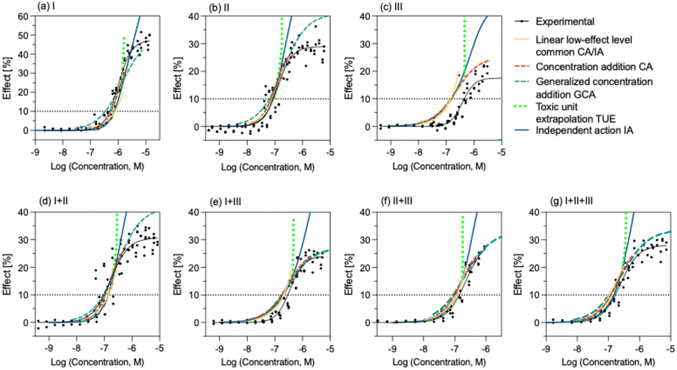
The BPA-EQi was calculated based on the relative effect potency as compared to BPA (REPi, eqn (17))27 and its concentration Ci in the mixture.
| BPA-EQi (bioassay) = REPi × Ci | (17) |
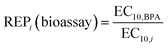 | (18) |
3 Results and discussion
3.1 Cytotoxicity
As for single compounds,11 the IC10 of the mixtures varied little between cell lines (Table S5 and Fig. S2) and therefore we used IC10,median for the following analysis. IC10,median(exp) agreed within a factor of two with the predicted IC10,median(CA/IA) (Fig. 2b and Table S5).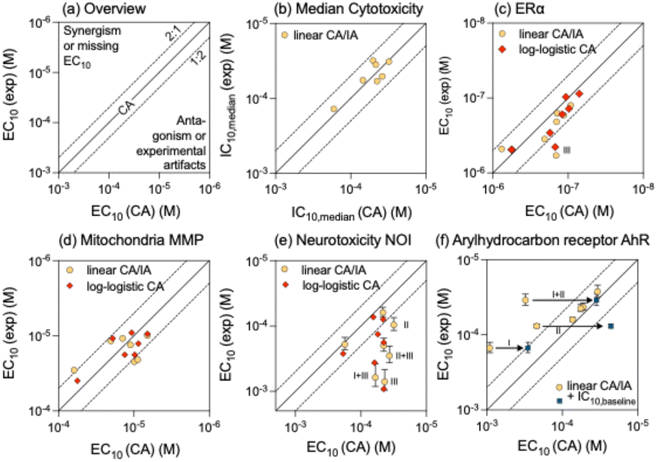 | ||
| Fig. 2 Comparison between predicted and experimental effect and inhibitory concentrations for different endpoints and median cytotoxicity. (a) Schematic representation of prediction outcomes: solid diagonal line indicates perfect agreement with concentration addition modeling (CA), dashed lines mark the range of twofold deviation in both directions. Data points above the range indicate apparent synergism or missing single chemical values, data points below the diagonal line hint towards antagonism or experimental artefacts. (b) Cytotoxicity IC10,median(exp) vs. IC10,median(CA) (Table S5). (c) ERα-UAS-bla GripTite (ERα) (Table S7). (d) Mitochondrial membrane potential inhibition (MMP) (Table S9). (e) Neurite outgrowth inhibition (NOI) (Table S10). (f) Aryl hydrocarbon receptor activation (AhR) (Table S11). Orange circles represent IC10(exp) and EC10(exp) vs. IC10(CA) or EC10(CA), respectively, predicted with the low-effect linear-slope model (abbreviated with linear CA/IA in the figure (eqn (10))), the red diamonds the CA predicted with the full CA model and log-logistic CRCs as input data (abbreviated with log-logistic CA in the figure (eqn (10))) and the blue squares the predictions for AhR (eqn (10)) with missing EC10 replaced by IC10,baseline (Table S1). | ||
3.2 Estrogen receptor α (ERα)
For ERα, EC10(exp) of the mixtures agreed well with the CA-predictions using the linear CA/IA model (Fig. 2c, S3 and Table S7). We observed no synergism or antagonism for any mixture at 10% effect level. BPPH in mixture III was inactive and one would expect a deviation towards “synergy or missing EC10”, however the deviation from the CA model prediction by a factor of four for mixture III with the most hydrophobic components would rather suggest solubility problems (Fig. 2c and Table S7).Although environmentally relevant mixtures generally occur at low concentrations, for which the linear low-effect model is applicable, we extended the analysis for ERα to four more complex mixture models (log-logistic CRC–CA, log-logistic IA, GCA, and TUE). This additional evaluation was conducted to better understand whether the partial agonists interact in mixtures at higher effect levels (>30%, Fig. 3). All these predictions were based on the full log CRC (eqn (5)) of the individual mixture components (Fig. S4 and Table S7). The experimental CRCs were well predicted by all models up to approximately 20% (Fig. 3c) with exception of mixture III.
 | ||
| Fig. 3 Full concentration–effect curves for estrogen receptor ERα activation in ERα-UAS-bla GripTite. (a) Mixtures I, (b) II, (c) III, (d) I + II, (e) I + III, (f) II + III, (g) I + II + III. Black dots are the experimental data points and black thin line fit with the full-range log-logistic model (eqn (5)). Linear CA&IA (mango line) refers to the linear low-effect model for prediction of concentration addition (CA)/independent action (IA) (eqn (10)). CA (red dashed line) prediction with eqn (6) from the full log-logistic CRC of the single chemicals; IA (blue solid line) refers to IA prediction with eqn (7) from the full log-logistic CRC of the single chemicals; GCA (dashed teal line) refers to general concentration addition model (eqn (11)) and TUE (light green dotted line) to toxic unit extrapolation model (eqn (14)). All data from Table S7. | ||
By definition the log-logistic CRC–CA model only predicts up to the Emax of the mixture component with lowest maximum and hence the predicted CRCs (red dashed lines in Fig. 3) ended often between 20 and 30% effect, but below this model overlapped with the linear CA model. The log-logistic IA could not predict Emax but predicted the shape of the low-effect level portion of the CRCs very well.
The CGA model adequately predicted the maximum efficacy of ERα activation (Emax, Table S7), but was performing poorer regarding the predicted effect levels <10% as already observed by Scholze et al.32 This is due to very similar log-logistic fits of the CRCs of the mixture components (Fig. S3) that agreed well between adjustable slope b and b = 1 but sometimes underperformed at low effect levels.
The TUE represents a worst-case scenario that implies that partial agonists keep activating the receptor beyond their maximum effect level. This model clearly overestimated the observed mixture effect (Fig. 3). We hypothesize that the partial agonists still contribute beyond their individual maximum effect levels to the mixture effect, most likely by remaining bound in the ERα binding pocket and not being displaced by other alternatives, thereby decreasing Emax of the mixture.
The confirmation of CA for low effects in this study agreed well with those reported in literature for BPA and alternatives. Skledar et al.20 investigated binary, ternary, and up to six component mixtures of BPA and five alternatives, including BPF, BPAF, BPS, Bisphenol C (BPC), and BPZ in HeLa 9903 cells. Their study included equimolar and equipotent mixtures, as well as mixture scenarios based on a “something from nothing” scenario. They observed good agreement between predicted and experimentally determined estrogenicity when using the concentration addition model (Fig. S5a and Table S8). Similarly, Lee et al.21 demonstrated CA for binary and tertiary combinations of BPA and eight other alternatives when comparing benchmarks at 50% transactivation activity (Fig. S5b and Table S8). These studies, along with our own findings, strengthen the evidence that concentration addition is a reliable model for predicting combined estrogenic effects of structurally related compounds as the BPA alternatives.
Synergistic and antagonistic effects occur typically at high doses, if at all. While synergy is rare, it cannot be neglected entirely, especially in in vivo studies.26,42In vitro studies on mixtures of estrogens and xenoestrogens have mainly reported CA,15,17,18,32 but there exist also reports on how non- or low-estrogenic modulators impacted the estrogenic effect in mixtures.43 Our experiments of mixtures of low-potency xenoestrogens at high doses essentially concurred with CA, thereby refuting any synergy or antagonism.
3.3 Oxidative stress response (ARE)
None of the selected BPA alternatives or BPA activated ARE in our previous study (Table S1).11 However, we were still able to detect a response for three of the mixtures (ECIR1.5(I) = 146 µM, ECIR1.5(II) = 68 µM and ECIR1.5(I + II) = 188 µM). The binary mixture of I with II had an ECIR1.5(CA) = 87 µM, which is less than a twofold deviation. We previously made similar observations for the endpoint ARE previously in designed mixtures of diverse organic micropollutants, where equipotent mixtures showed CA,44 while mixtures representative for surface water that contained inactive and active components showed higher mixture effects than predicted by CA, indicating that inactive mixture components can contribute to mixture effects.45 In contrast, when five or ten potent activators of ARE were combined with five inactive pharmaceuticals, we did not see any contribution of the inactive mixture.463.4 Mitochondrial membrane potential (MMP) inhibition
For MMP, we applied two approaches: first we fitted single chemical and mixture data with the linear CRC model (eqn (3)), and derived EC10 values from these fits (eqn (4) and Fig. S6). The experimental EC10 agreed with the predicted EC10(CA/IA) within a factor of two (Fig. 2d and Table S9).The log-logistic fits yielded Emax between 84% and 102% for single chemicals and mixtures (Table S6). As in theory 100% effect should be attained for this endpoint (unlike for the partial agonists of the hormone receptors), all CRCs were additionally fitted with the Emax set to 100% to simplify the mixture prediction for CA and IA (Table S6 and Fig. S7 for the individual components) and to predict the full CRCs for CA and IA (Fig. S7).
As the slopes of the single chemicals were rather steep, ranging from b = 1.8 to 5.0, the predictions for CA were separated clearly from the IA predictions for all mixtures containing II (Fig. S7). Typically, the steeper the slopes of the components' CRCs, the more the CA and IA predictions diverge. Mixture I was more potent than its CA and IA predictions (Fig. S7a), for mixture II the experimental CRC lay exactly between CA and IA predictions (Fig. S7b), and mixture III was less potent than CA and IA predictions (Fig. S7c) and for the remaining mixtures we observed the same pattern (Fig. S7d–f).
The linear predictions at low effect levels crossed the CA curve at 10% effect level, which indicates that the linear model performed well, which is also confirmed by Fig. 2d that visualises that all predictions agreed with CA. The linear model is very relevant for environmentally realistic mixtures, where chemicals occur at very low concentrations that would trigger mixture effects in MMP of only up to 0.02% (for I + II + III) and even less for the other mixtures. We have argued previously that CA and IA overlap at low effect levels,30 but this only holds for strictly linear CRCs at low effect levels and only if the slopes b of the log CRCs are close to 1.37 If they are larger than 2, the linearity of the linear CRCs is compromised (Fig. S8). The steep log CRCs for mitochondrial toxicity will not only lead to a separation of the CA and IA prediction as discussed above but also to imperfect fit of the linear CRCs. However, as Fig. S7 demonstrates, in practice there is a very good consistency of the linear CA/IA model with the CA model derived from the log CRCs at the 10% effect level. This justifies the derivation of BPA-EQ(MMP), which will be further pursued below.
Mitochondrial toxicity data for BPA and its alternatives have been less frequently reported than for estrogenicity.7,9 A binary mixture of BPA and BPAF induced mitochondrial dysfunction in mitochondrial dysfunction in human granulosa cells,47 but concentrations in the mixtures were the same as in the single chemical experiments, which means that the mixture effect could not be compared with CA or IA.
3.5 Neurite outgrowth inhibition (NOI)
In our previous study, we could not derive EC10 of NOI for BPS, BPF and BPPH (Table S1)11 and the linear fit was rather poor for the mixtures (Fig. S10). Nevertheless, even with data for two chemicals missing, mixture I still agreed within a factor of 2 with the linear CA/IA model (Table S10), which means that BPA would have dominated the mixture effect on its own, but mixture III, where BPPH was missing, did not agree with the prediction (Table S10 and Fig. 2e). All mixtures that included mixture III deviated from CA in the direction of “antagonism or experimental artefacts” (Fig. 2e). This anomaly is likely due BPAP and BPPH being the most hydrophobic chemicals (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kow 6.67 and 5.06) that might not be well solubilized. This would suggest an experimental artefact rather than an antagonistic effect.
Kow 6.67 and 5.06) that might not be well solubilized. This would suggest an experimental artefact rather than an antagonistic effect.
When analysing the whole CRCs of the single chemicals (Fig. S11) and mixtures (Fig. S12) and compare with the CA and IA predictions, it becomes evident that all mixtures containing III predict a much higher effect than observed, while all mixtures with I and II agreed perfectly with the CA and IA predictions and there was little difference between CA and IA (Fig. S12). The experimental data also scatter more than for other mixtures. This further supports our theory that for NOI solubility issues highly influenced the experimental results.
3.6 Activation of aryl hydrocarbon receptor (AhR)
All mixtures activated AhR but the activation was relatively weak and occurred close to cytotoxicity. Due to the high cytotoxicity, only the linear portion of the CRCs could be evaluated and hence only the linear CA/IA prediction could be performed (Table S11). It is noteworthy that all mixtures were active even though the EC10 for AhR activation could only be derived for BPA, BPE, BPZ and BPPH (Table S1).11 The toxic ratio (TR = IC10,baseline/IC10) of the single chemicals ranged from 0.09 to 2.67 and the specificity ratio (SR =IC10/EC10) of the four active ones from 1.04 to 1.36, which indicates that AhR activation was measurable but so weak that it occurred close to baseline cytotoxicity and is due to the cytotoxicity burst, which is the nonspecific activation of any reporter gene at concentrations close to cell death.35,48 For single chemicals we followed a strict cytotoxicity cutoff at 10% cytotoxicity, which means that all concentrations that led to more than 10% cytotoxicity were omitted for the evaluation of the CRC of AhR activation,37 but we relaxed this criterion for mixtures where specific and non-specific effects can be relevant for different mixture components. For prediction of the mixture effects including the inactive mixture components, we filled these data gaps by their predicted IC10,baseline (Table S1).11 The mixture simulations resulted in greatly improved predictions for mixtures I, II and I + II, while all others did not vary (Fig. 2f). These simulations demonstrate that (a) apparently inactive chemicals might contribute to mixture effects and (b) that the activation of AhR by bisphenols is so weak that we essentially just measured cytotoxicity. This case demonstrates the challenge we often encounter when applying reporter gene assays. In many studies, cytotoxicity is not reported alongside the activation of the receptor or pathway. These effects are often erroneously interpreted as specific, when they are actually non-specific and indirectly caused by the cytotoxicity burst. Thus, the mixture effects reported for AhR activation are, in effect, mixture effects triggered by cytotoxicity, and in this particular example even baseline toxicity because not only the SRs were close to 1 but also the TRs.3.7 Evaluating BPA alternative mixtures with the BPA-EQ concept
For those assays for which we could successfully demonstrate concentration additivity, and which also represent relevant effects for BPA and the alternatives we applied a modified BEQ-model – coined as BPA-EQ. We included median cytotoxicity (IC10,median), estrogen receptor alpha activation, and mitochondrial membrane potential inhibition (MMP).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kow of 6.67 (Fig. 4a). Predicted and experimental BPA-EQs of the 10-component mixture agreed well (Fig. 4b and Table S12) with BPA-EQchem of 11.2 µg L−1 and BPA-EQmix of 10.4 µg L−1. The BPA-EQchem was 7% higher than BPA-EQmix. BEQs of the combined mixtures corresponded well with those of the individual mixtures I to III (Fig. 4c), within a range of −22% to +8% (ΔMixture I + II: −22%, ΔMixture I + III: −25%, ΔMixture II + III: −38%, ΔMixture I + II + III: +8%).
Kow of 6.67 (Fig. 4a). Predicted and experimental BPA-EQs of the 10-component mixture agreed well (Fig. 4b and Table S12) with BPA-EQchem of 11.2 µg L−1 and BPA-EQmix of 10.4 µg L−1. The BPA-EQchem was 7% higher than BPA-EQmix. BEQs of the combined mixtures corresponded well with those of the individual mixtures I to III (Fig. 4c), within a range of −22% to +8% (ΔMixture I + II: −22%, ΔMixture I + III: −25%, ΔMixture II + III: −38%, ΔMixture I + II + III: +8%).
When comparing BPA-EQchem and BPA-EQmix for the 10-component mixture I + II + III (Fig. 4e). BPA-EQmix was approximately 37% lower than the predicted BPA-EQchem. Differences between the sum of individual mixtures I–III and their binary and tertiary mixtures were slightly smaller but otherwise corresponded well (Fig. 4f) (ΔMixture I + II = −19.3%), (ΔMixture I + III −9.4%), (ΔMixture II + III: +1.6%) and (ΔMixture I + II + III: −17%).
3.8 Evaluation of simulated substitution scenarios using the BPA-EQ concept
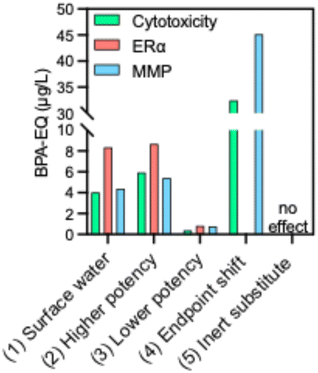
To demonstrate the practical relevance and applicability of the BPA-EQ for the validated endpoints, we simulated four replacement scenarios in addition to the reconstructed environmental mixtures. These scenarios were designed to evaluate the potential consequences of substituting BPA and to illustrate how the BPA-EQ can facilitate communication between different scientific disciplines.The scenarios encompass: (1) “surface water”, (2) “high-potency”, (3) “low-potency”, (4) “endpoint shift” and (5) “inert behavior”. For each scenario, all currently detected bisphenols in mixture I + II (scenario (1)) were substituted by a single replacement compound in the same molar concentration: (2) BPB, (3) BPS, (4) BPPH, or (5) 2,2,4,4-tetramethyl-1,3-cyclobutanediol (TMCD) (Table S13). The outcomes were then benchmarked against the results of the real-world surface water mixture I + II.
In the high-potency scenario (2), we assumed that all detected bisphenol-like chemicals in the mixture were replaced by BPB, which is roughly twice as cytotoxic, 8.6-fold more potent than BPA in eliciting ERα agonism and able to cause mitochondrial toxicity. Applying this substitution to mixture I + II resulted in BPA-EQ of 5.4, 8.7, and 5.9 µg L−1 for cytotoxicity, ERα agonism and mitochondrial toxicity, respectively (Fig. 5 and Table S13). Replacing BPA and other alternatives with BPB—or with any other alternative of similar or higher potency—would therefore not mitigate but rather exacerbate the overall toxicity burden in the environment.
In the low-potency scenario (3), all BPA and other bisphenol alternatives were replaced by bisphenol S (BPS), which is about ten times less cytotoxic compared to BPA and approximately five times less potent as an ERα agonist. With this simulation, the overall toxicity burden of the mixture would be markedly reduced by a factor of ten (BPA-EQ(cytotoxicity) = 0.25 µg L−1, BPA-EQ(ERα) = 0.82 µg L−1, BPA-EQ(MMP) = 0.76 µg L−1).
However, this benefit would only hold if environmental exposure levels remained constant or decreased, since a substantial increase in BPS concentrations, e.g., due to higher overall production/consumption could offset the lower potency. Moreover, BPS is still considered an estrogenic and reproduction toxic,4,9,11,49 even if higher concentrations are required to elicit effects, and its physicochemical properties differ from BPA. For example, BPS was more resistant to degradation in the environment.50 Consequently, long-term exposure could accumulate and counteract the apparent advantage of lower potency. It is also listed as a substance of very concern (SVHC) like BPA and received a CLP categorization as reproduction toxic 2B.51
Regarding the endpoint shift scenario (4), BPPH is a bisphenol compound that has not yet been detected in surface waters but is used as a substitute for BPA. While BPPH shows no measurable activity as an ERα agonist, it exhibits markedly higher toxicity on other endpoints: approximately 10 times increased cytotoxicity compared to BPA and 14-fold higher mitochondrial toxicity, consistent with its greater hydrophobicity. The BPA-EQs changed accordingly to BPA-EQ(cytotoxicity) = 32.5 µg L−1 and BPA-EQ(MMP) = 45.2 µg L−1. This scenario illustrates the importance of considering a broad spectrum of toxicological endpoints before introducing a new compound that may appear “safer” when judged solely by its estrogenicity. Such substitution strategy risks shifting the toxicity burden from one endpoint to another rather than reducing it overall.
The inert substitution scenario (5) with TMCD represents the only case in which replacing all bisphenols would substantially reduce the mixture's overall toxic contribution. TMCD has extremely low potency, and even in case of a 1000-fold increase in environmental concentrations would be lower as the current range of mixture toxicity, irrespective which endpoint considered. Its negligible toxicological activity means that substitution with such an inert compound would effectively eliminate mixture toxicity.
Taken together, these thought experiments illustrate three key points. First, it is inherently difficult to replace a high-volume industrial chemical with vital economic functions while ensuring that the substitute truly has fewer environmental impacts. Second, successful risk reduction requires limiting the overall release of chemicals into the environment, not just exchanging one compound for another. Third, the long-term solution lies in designing chemicals with intrinsically lower hazard profiles and reduced environmental persistence, thereby preventing the need for problematic substitutions in the first place.
4 Conclusion
BPA alternative mixtures followed the concentration addition model not only for estrogenicity but also for mitochondrial toxicity and cytotoxicity. In cases where the linear CA model at low effect levels was applicable, BPA-EQs could be derived as a robust and communicable metric to predict mixture effects of BPA alternatives across these endpoints, with potential applications ranging from environmental monitoring to human biomonitoring, and replacement scenario analyses. Most importantly, BPA-EQs enable an early quantitative comparison of potential replacement compounds and their mixture contributions, thereby supporting prioritisation and informed substitution decisions.Meaningful risk reduction can only be achieved if alternatives are less potent than BPA, ideally inactive, and do not introduce additional toxicological endpoints.11 Ultimately, the use of BPA and its alternatives should be limited to essential applications to minimize both environmental burden and human exposure.
The freshwater environmental quality standard (EQS) of BPA compliant with the Water Framework Directive of the European Union is presently proposed as 129 µg L−1 and the corresponding EQS for surface water used for abstraction of drinking water is presently at 2.5 µg L−1 but recent revisions of the tolerable daily intake for humans might trigger a lowering of the EQS to 0.034 ng L−1.52 The median surface water concentration of BPA of 0.35 µg L−1 is below the existing EQS for surface water. However, if we compare the BPA-EQ of the 6-component mixture (I + II) in all bioassays, they would exceed the EQS of the single BPA. The BPA-EQ of the 10-component mixture (I + II + III) would exceed the EQS of the single BPA even by a large margin. EQS are derived for single chemicals but using them as a threshold for BEQs seems intuitively convincing.
This study provides a robust and communicable framework to better capture mixture effects of BPA alternatives, which may ultimately simplify the reporting and prediction of mixture effects, and in the long-term support regulatory discussions such as the inclusion of the mixture allocation factor (MAF)53,54 in the current revision of REACH.
Author contributions
Vanessa Srebny: formal analysis, visualization, writing –original draft. Georg Braun: formal analysis, visualization, methodology and investigation, review & editing. Niklas Wojtysiak: methodology and investigation. Beate Escher: conceptualization, formal analysis, visualization, supervision, writing – review & editing.Conflicts of interest
All authors have given approval to the final version of the article.Data availability
All effect concentrations derived from CRCs are available in the supplementary information (SI) ESI_B EXCEL file. Raw data of the CRCs can be obtained from the authors. Supplementary information: ESI_A_Mixtures BPA.docx is a word file with additional text and figures. ESI_B_Mixtures BPA.xlsx is an excel file containing all data (Tables S1 to S13). See DOI: https://doi.org/10.1039/d5em00802f.Acknowledgements
This work was carried out in the framework of the European Partnership for the Assessment of Risks from Chemicals (PARC) and has received partial funding from the European Union's Horizon Europe research and innovation program under Grant Agreement No. 101057014. Views and opinions expressed are, however, those of the author(s) only and do not necessarily reflect those of the European Union or the Health and Digital Executive Agency. Neither the European Union nor the granting authority can be held responsible. We gratefully acknowledge access to the platform CITEPro (Chemicals in the Environment Profiler) funded by the Helmholtz Association for bioassay and analytical measurements. The authors were supported by the Helmholtz Association under the recruiting initiative scheme, which is funded by the German Ministry of Education and Research, within the Helmholtz POF IV Topic 9 and the Integrated Project “Healthy Planet-towards a non-toxic environment”. We also thank the Helmholtz Initiative and Networking Fund for financial support of the project Co-Creation process for the modernization of chemical hazard indicators CoModHaz (KA-TCP-20). We extend our gratitude to Julia Huchthausen and Jenny Braasch of UFZ for support of the bioassay measurements and thank Martin Scholze of Brunel University for helpful discussions. We used DeepL for improving <5% of the sentences.References
- European Chemical Agency (ECHA), Restriction List. Substances restricted under REACHhttps://chem.echa.europa.eu/obligation-lists/restrictionList?searchText=, https://www.echa.europa.eu/web/guest/candidate-list-table, accessed 2025/09/27.
- European Chemical Agency (ECHA), ANNEX XVII TO REACH – Conditions of restriction, Entry 66, Bisphenol A https://echa.europa.eu/documents/10162/1451b2a8-117e-e179-cfcd-c57214bb4adf, https://www.echa.europa.eu/web/guest/candidate-list-table, accessed 2025/09/27.
- European Food Safety Authority (EFSA), Commission adopts ban of Bisphenol A in food contact materials (19 Dec 2024). https://food.ec.europa.eu/food-safety-news-0/commission-adopts-ban-bisphenol-food-contact-materials-2024-12-19_en, 2024.
- J. R. Rochester, Bisphenol A and human health: a review of the literature, Reprod. Toxicol., 2013, 42, 132–155, DOI:10.1016/j.reprotox.2013.08.008
.
- F. S. Vom Saal and L. N. Vandenberg, Update on the Health Effects of Bisphenol A: Overwhelming Evidence of Harm, Endocrinology, 2021, 162(3), bqaa171, DOI:10.1210/endocr/bqaa171
.
- European Chemical Agency (ECHA), Candidate List of Substances of Very High Concern for Authorization, https://www.echa.europa.eu/web/guest/candidate-list-table, accessed 2025/09/27.
- O. Adamovsky, K. J. Groh, A. Bialk-Bielinska, B. I. Escher, R. Beaudouin, L. Mora Lagares, K. E. Tollefsen, M. Fenske, E. Mulkiewicz, N. Creusot, A. Sosnowska, S. Loureiro, J. Beyer, G. Repetto, A. Stern, I. Lopes, M. Monteiro, A. Zikova-Kloas, T. Elersek and K. Kyriakopoulou,
et al., Exploring BPA alternatives - Environmental levels and toxicity review, Environ. Int., 2024, 189, 108728, DOI:10.1016/j.envint.2024.108728
.
- S. Mhaouty-Kodja, D. Zalko, S. Tait, E. Testai, C. Viguie, E. Corsini, N. Grova, F. M. Buratti, N. J. Cabaton, L. Coppola, A. De la Vieja, M. Dusinska, N. El Yamani, V. Galbiati, P. Iglesias-Hernandez, Y. Kohl, A. Maddalon, F. Marcon, L. Naule and M. J. Silva,
et al., A critical review to identify data gaps and improve risk assessment of bisphenol A alternatives for human health, Crit. Rev. Toxicol., 2024, 54, 696–753, DOI:10.1080/10408444.2024.2388712
.
- K. Pelch, J. A. Wignall, A. E. Goldstone, P. K. Ross, R. B. Blain, A. J. Shapiro, S. D. Holmgren, J. H. Hsieh, D. Svoboda, S. S. Auerbach, F. M. Parham, S. A. Masten, V. Walker, A. Rooney and K. A. Thayer, A scoping review of the health and toxicological activity of bisphenol A (BPA) structural analogues and functional alternatives, Toxicology, 2019, 424, 152235, DOI:10.1016/j.tox.2019.06.006
.
- K. E. Pelch, Y. Li, L. Perera, K. A. Thayer and K. S. Korach, Characterization of Estrogenic and Androgenic Activities for Bisphenol A-like Chemicals (BPs): In Vitro Estrogen and Androgen Receptors Transcriptional Activation, Gene Regulation, and Binding Profiles, Toxicol. Sci., 2019, 172, 23–37, DOI:10.1093/toxsci/kfz173
.
- V. Srebny, L. Henneberger, M. Konig, J. Huchthausen, J. Braasch and B. I. Escher, Beyond Estrogenicity: A Comparative Assessment of Bisphenol A and Its Alternatives in In Vitro Assays Questions Safety of Replacements, Environ. Sci. Technol., 2025, 59, 17457–17470, DOI:10.1021/acs.est.5c07018
.
- D. Chen, K. Kannan, H. Tan, Z. Zheng, Y. L. Feng, Y. Wu and M. Widelka, Bisphenol Analogues Other Than BPA: Environmental Occurrence, Human Exposure, and Toxicity-A Review, Environ. Sci. Technol., 2016, 50, 5438–5453, DOI:10.1021/acs.est.5b05387
.
- C. Swansborough, J. Soto, I. Shapland, C. B. Hughes, E. Jennings, M. McArdle, E. Mihaich, E. Majoris and M. Bunge, Trend analysis of environmental concentrations of Bisphenol A in European waters and sediments, Environ. Res., 2025, 282, 121946, DOI:10.1016/j.envres.2025.121946
.
- A. Kortenkamp and M. Faust, Regulate to reduce chemical mixture risk, Science, 2018, 361, 224–226, DOI:10.1126/science.aat9219
.
- J. Payne, M. Scholze and A. Kortenkamp, Mixtures of four organochlorines enhance human breast cancer cell proliferation, Environ. Health Perspect., 2001, 109, 391–397 CrossRef CAS PubMed
.
- N. Rajapakse, E. Silva and A. Kortenkamp, Combining xenoestrogens at levels below individual no-observed-effect concentrations dramatically enhances steroid hormone action, Environ. Health Perspect., 2002, 110, 917–921 CrossRef CAS PubMed
.
- E. Silva, N. Rajapakse, M. Scholze, T. Backhaus, S. Ermler and A. Kortenkamp, Joint Effects of Heterogeneous Estrogenic Chemicals in the E-Screen-Exploring the Applicability of Concentration Addition, Toxicol. Sci., 2011, 122, 383–394, DOI:10.1093/toxsci/kfr103
.
- E. Silva, N. Rajapakse and A. Kortenkamp, Something from "nothing" – eight weak estrogenic chemicals combined at concentrations below NOECs produce significant mixture effects, Environ. Sci. Technol., 2002, 36, 1751–1756, DOI:10.1021/es0101227
.
- A. Kortenkamp, Low dose mixture effects of endocrine disrupters and their implications for regulatory thresholds in chemical risk assessment, Curr. Opin. Pharmacol., 2014, 19, 105–111, DOI:10.1016/j.coph.2014.08.006
.
- D. G. Skledar and L. P. Masic, In vitro estrogenic activity of binary and multicomponent mixtures with bisphenol A, Sci. Total Environ., 2020, 707, 135211, DOI:10.1016/j.scitotenv.2019.135211
.
- H. Lee, J. Park and K. Park, Mixture Effects of Bisphenol A and Its Structural Analogs on Estrogen Receptor Transcriptional Activation, Toxics, 2023, 11(12), 986, DOI:10.3390/toxics11120986
.
- S. Loewe, The problem of synergism and antagonism of combined drugs, Arzneimittelforschung, 1953, 3, 285–290 CAS
.
- C. I. Bliss, The toxicity of poisons applied jointly, Ann. Appl. Biol., 1939, 26, 585–615, DOI:10.1111/j.1744-7348.1939.tb06990.x
.
- T. Backhaus, Å. Arrhenius and H. Blanck, Toxicity of a Mixture of Dissimilarly Acting Substances to Natural Algal Communities: Predictive Power and Limitations of Independent Action and Concentration Addition, Environ. Sci. Technol., 2004, 38, 6363–6370, DOI:10.1021/es0497678
.
- N. Cedergreen, Quantifying synergy: A systematic review of mixture toxicity studies within environmental toxicology, PLoS One, 2014, 9, e96580, DOI:10.1371/journal.pone.0096580
.
- O. V. Martin, Synergistic effects of chemical mixtures: How frequent is rare?, Curr. Opin Toxicol., 2023, 36, 100424, DOI:10.1016/j.cotox.2023.100424
.
- D. L. Villeneuve, A. L. Blankenship and J. P. Giesy, Derivation and application of relative potency estimates based on in vitro bioassay results, Environ. Toxicol. Chem., 2000, 19, 2835–2843 CrossRef CAS
.
- P. S. Hewlett and R. L. Plackett, A Unified Theory for Quantal Responses to Mixtures of Drugs: Non-Interactive Action, Biometrics, 1959, 15, 591–610, DOI:10.2307/2527657
.
- M. Scholze, W. Boedeker, M. Faust, T. Backhaus, R. Altenburger and L. H. Grimme, A general best-fit method for concentration-response curves and the estimation of low-effect concentrations, Environ. Toxicol. Chem., 2001, 20, 448–457, DOI:10.1002/etc.5620200228
.
- B. Escher, G. Braun and C. Zarfl, Exploring the Concepts of Concentration Addition and Independent Action Using a Linear Low-Effect Mixture Model, Environ. Toxicol. Chem., 2020, 39, 2552–2559, DOI:10.1002/etc.4868
.
- G. J. Howard and T. F. Webster, Generalized concentration addition: a method for examining mixtures containing partial agonists, J. Theor. Biol., 2009, 259, 469–477, DOI:10.1016/j.jtbi.2009.03.030
.
- M. Scholze, E. Silva and A. Kortenkamp, Extending the applicability of the dose addition model to the assessment of chemical mixtures of partial agonists by using a novel toxic unit extrapolation method, PLoS One, 2014, 9, e88808, DOI:10.1371/journal.pone.0088808
.
- X. J. Wang, J. D. Hayes and C. R. Wolf, Generation of a stable antioxidant response element–driven reporter gene cell line and its use to show redox-dependent activation of Nrf2 by cancer chemotherapeutic agents, Cancer Res., 2006, 66, 10983–10994, DOI:10.1158/0008-5472.can-06-2298
.
- J. C. Brennan, G. He, T. Tsutsumi, J. Zhao, E. Wirth, M. H. Fulton and M. S. Denison, Development of Species-Specific Ah Receptor-Responsive Third Generation CALUX Cell Lines with Enhanced Responsiveness and Improved Detection Limits, Environ. Sci. Technol., 2015, 49, 11903–11912, DOI:10.1021/acs.est.5b02906
.
- B. I. Escher, L. Henneberger, M. Konig, R. Schlichting and F. C. Fischer, Cytotoxicity Burst? Differentiating Specific from Nonspecific Effects in Tox21 in Vitro Reporter Gene Assays, Environ. Health Perspect., 2020, 128, 77007, DOI:10.1289/EHP6664
.
- J. Huchthausen, J. Braasch, B. I. Escher, M. Konig and L. Henneberger, Effects of Chemicals in Reporter Gene Bioassays with Different Metabolic Activities Compared to Baseline Toxicity, Chem. Res. Toxicol., 2024, 37, 744–756, DOI:10.1021/acs.chemrestox.4c00017
.
- B. I. Escher, P. A. Neale and D. L. Villeneuve, The advantages of linear concentration-response curves for in vitro bioassays with environmental samples, Environ. Toxicol. Chem., 2018, 37, 2273–2280, DOI:10.1002/etc.4178
.
- P. A. Neale, R. Altenburger, S. Ait-Aissa, F. Brion, W. Busch, G. de Aragão Umbuzeiro, M. S. Denison, D. Du Pasquier, K. Hilscherova, H. Hollert, D. A. Morales, J. Novac, R. Schlichting, T.-B. Seiler, H. Serra, Y. Shao, A. J. Tindall, K. E. Tollefsen, T. D. Williams and B. I. Escher, Development of a bioanalytical test battery for water quality monitoring: Fingerprinting identified micropollutants and their contribution to effects in surface water, Water Res., 2017, 123, 734–750, DOI:10.1016/j.watres.2017.07.016
.
- J. Lee, J. Huchthausen, R. Schlichting, S. Scholz, L. Henneberger and B. I. Escher, Validation of an SH-SY5Y Cell-Based Acetylcholinesterase Inhibition Assay for Water Quality Assessment, Environ. Toxicol. Chem., 2022, 41, 3046–3057, DOI:10.1002/etc.5490
.
- B. I. Escher, M. Dutt, E. Maylin, J. Y. Tang, S. Toze, C. R. Wolf and M. Lang, Water quality assessment using the AREc32 reporter gene assay indicative of the oxidative stress response pathway, J. Environ. Monit., 2012, 14, 2877–2885, 10.1039/c2em30506b
.
- R. Altenburger, T. Backhaus, W. Boedeker, M. Faust, M. Scholze and L. H. Grimme, Predictability of the toxicity of multiple chemical mixtures to Vibrio fischeri: Mixtures composed of similarly acting chemicals, Environ. Toxicol. Chem., 2000, 19, 2341–2347, DOI:10.1002/etc.5620190926
.
- A. Boobis, R. Budinsky, S. Collie, K. Crofton, M. Embry, S. Felter, R. Hertzberg, D. Kopp, G. Mihlan, M. Mumtaz, P. Price, K. Solomon, L. Teuschler, R. Yang and R. Zaleski, Critical analysis of literature on low-dose synergy for use in screening chemical mixtures for risk assessment, Crit. Rev. Toxicol., 2011, 41, 369–383, DOI:10.3109/10408444.2010.543655
.
- R. M. Evans, M. Scholze and A. Kortenkamp, Additive Mixture Effects of Estrogenic Chemicals in Human Cell-Based Assays Can Be Influenced by Inclusion of Chemicals with Differing Effect Profiles, PLoS One, 2012, 7(8), e43606, DOI:10.1371/journal.pone.0043606
.
- P. A. Neale, G. Braun, W. Brack, E. Carmona, R. Gunold, M. Konig, M. Krauss, L. Liebmann, M. Liess, M. Link, R. B. Schafer, R. Schlichting, V. C. Schreiner, T. Schulze, P. Vormeier, O. Weisner and B. I. Escher, Assessing the Mixture Effects in In Vitro Bioassays of Chemicals Occurring in Small Agricultural Streams during Rain Events, Environ. Sci. Technol., 2020, 54, 8280–8290, DOI:10.1021/acs.est.0c02235
.
- R. Altenburger, M. Scholze, W. Busch, B. I. Escher, G. Jakobs, M. Krauss, J. Krüger, P. A. Neale, S. Aït-Aïssa, A. C. Almeida, T.-B. Seiler, F. Brion, K. Hilscherová, H. Hollert, J. Novák, R. Schlichting, H. Serra, Y. Shao, A. J. Tindall, K. E. Tolefsen, G. de Aragão Umbuzeiro, T. D. Williams and A. Kortenkamp, Mixture effects in samples of multiple contaminants – an inter-laboratory study with manifold bioassays, Environ. Int., 2018, 114, 95–116, DOI:10.1016/j.envint.2018.02.013
.
- B. I. Escher, C. van Daele, M. Dutt, J. Y. M. Tang and R. Altenburger, Most oxidative stress response in water samples comes from unknown chemicals: the need for effect-based water quality trigger values, Environ. Sci. Technol., 2013, 47, 7002–7011, DOI:10.1021/es304793h
.
- M. Huang, G. Xu, M. Li and M. Yang, Bisphenol A and bisphenol AF co-exposure induced apoptosis of human ovarian granulosa cells via mitochondrial dysfunction, Food Chem. Toxicol., 2024, 191, 114894, DOI:10.1016/j.fct.2024.114894
.
- K. A. Fay, D. L. Villeneuve, J. Swintek, S. W. Edwards, M. D. Nelms, B. R. Blackwell and G. T. Ankley, Differentiating Pathway-Specific From Nonspecific Effects in High-Throughput Toxicity Data: A Foundation for Prioritizing Adverse Outcome Pathway Development, Toxicol. Sci., 2018, 163, 500–515, DOI:10.1093/toxsci/kfy049
.
- M. K. Björnsdotter, J. de Boer and A. Ballesteros-Gómez, Bisphenol A and replacements in thermal paper: A review, Chemosphere, 2017, 182, 691–706, DOI:10.1016/j.chemosphere.2017.05.070
.
- R. Frankowski, A. Zgoła-Grześkowiak, W. Smułek and T. Grześkowiak, Removal of Bisphenol A and Its Potential Substitutes by Biodegradation, Appl. Biochem. Biotechnol., 2020, 191, 1100–1110, DOI:10.1007/s12010-020-03247-4
.
- European Chemical Agency (ECHA), 4,4’-Sulphonyldiphenol – ECHA Chemical Database, https://chem.echa.europa.eu/100.001.137/overview?searchText=BisphenolS, accessed 2025/07/04, 2025.
- Scientific Committee on Health Environmental and Emerging Risks (SCHEER), Scientific Opinion on "Draft Environmental Quality Standards for Priority Substances under the Water Framework Directive" - Bisphenol-A, 2022, https://health.ec.europa.eu/publications/scheer–scientific–opinion–draft–environmental–quality–standards–priority–substances–under–water–3_en, https://health.ec.europa.eu/document/download/037b602d–3102–3104b3106f–8928–3192de3106fcb3107fc3107_en?filename=scheer_o_3042.pdf.
- T. Backhaus, The mixture assessment or allocation factor: conceptual background, estimation algorithms and a case study example, Environ. Sci. Eur., 2023, 35, 55, DOI:10.1186/s12302-023-00757-w
.
- T. Backhaus, M. Scholze, W. Brack, O. Martin, D. Slunge, M. Ågerstrand, A. Kortenkamp and B. Escher, Include a mixture allocation factor to improve EU chemical risk management, Science, 2025, 390, 678–680, DOI:10.1126/science.aeb6374
.
| This journal is © The Royal Society of Chemistry 2026 |