 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Mechanisms of ozone effects on plant stress in soybean across growing season: from leaf to regional perspective
Luka
Mamić
 *abc,
Mj
Riches
*abc,
Mj
Riches
 bd,
Rose K.
Rossell
bd,
Rose K.
Rossell
 b and
Delphine K.
Farmer
b and
Delphine K.
Farmer
 *b
*b
aDepartment of Civil, Building and Environmental Engineering, Sapienza University of Rome, Via Eudossiana 18, 00184, Rome, Italy. E-mail: luka.mamic@uniroma1.it
bDepartment of Chemistry, Colorado State University, 1301 Center Ave Mall, 80523-1872, Fort Collins, Colorado, USA. E-mail: delphine.farmer@colostate.edu
cInterdepartmental Research Centre in Geomatics (CIRGEO), University of Padua, Corte Benedettina - Via Roma 34, 35020, Legnaro, Italy
dDepartment of Agricultural Biology, Colorado State University, 307 University Ave, 80523-1101, Fort Collins, Colorado, USA
First published on 3rd February 2026
Abstract
Ground-level ozone (O3) is a major constraint on agricultural productivity, yet most knowledge comes from controlled fumigation experiments using chronic exposures that differ from the episodic conditions crops experience in the field. Here, we combine a five-week chamber experiment with multi-year satellite observations (2018–2021, Arkansas, U.S.) to investigate how O3 affects photosynthesis, efficiency, and growth across scales of soybean plants (Glycine max). At the leaf level, initial O3 fumigation (80 ppb for 4 h) caused the strongest suppression of CO2 assimilation (A), stomatal conductance (Gs), and photosystem II efficiency (ΦPSII), indicating entry into a physiological strain phase. Recovery between exposures was incomplete, leading to sustained growth reductions despite moderate O3 levels. At the regional scale, analysis of solar-induced fluorescence (SIF) and MODIS productivity metrics revealed parallel patterns. Early-season O3 episodes produced greater suppression of SIF, GPP, and Gs compared to equivalent late-season events, and recovery lagged for several weeks. Seasonal yield proxies were best explained not by total O3 accumulation, but by early- and peak-season exposures, which accounted for up to 98% of variance across four growing seasons. Our findings highlight that the timing of O3 episodes is more consequential than cumulative dose, and that functional indicators such as SIF can detect strain-phase stress before structural indices diverge. By linking controlled experiments with regional-scale satellite monitoring, this study advances mechanistic understanding of O3 impacts on soybean and supports the development of remote sensing-based early warning tools for crop management.
Environmental significanceGround-level ozone (O3) is a pervasive air pollutant that threatens food security, but most risk assessments are based on chronic, elevated exposures that poorly represent real agroecosystems. By linking controlled fumigation experiments with multi-year satellite observations, this study shows that the timing of O3 exposure, particularly during early and peak growth stages, is more damaging to soybean productivity than total seasonal dose. Even moderate, short-term O3 episodes, under current regulations, suppressed photosynthesis, slowed canopy development, and reduced yield proxies under otherwise healthy field conditions. We further demonstrate that solar-induced chlorophyll fluorescence can detect O3 stress before visible damage occurs, offering a scalable tool for monitoring crop vulnerability. |
1. Introduction
Air pollution threatens agricultural productivity and food security around the globe.1,2 Among air pollutants, ground-level ozone (O3) is one of the most important stressors to global crop production.3,4 O3 is a secondary air pollutant formed via photochemical reactions between nitrogen oxides and volatile organic compounds. The terrestrial biosphere is a key sink for O3, which may reduce atmospheric concentrations of this pollutant, but means that O3 deposition can directly impact crop yield and productivity.5,6 Even short-term periods of elevated O3 can significantly decrease yields,2,7 with soybean being one of the most sensitive – yet economically important – crops.8,9While there is a broad understanding of O3-plant interactions, many details remain unclear, such as species-specific physiological responses,10 the mechanisms underlying photosynthetic and metabolic impairments,11 and interactions with climatic and agronomic factors.12 Plants primarily regulate O3 entry through stomatal control, yet stomatal uptake accounts for only ∼45% of total ecosystem-level O3 deposition, with the remainder occurring via non-stomatal pathways such as uptake by cuticular surfaces, soil, and wet leaf films.5 These non-stomatal routes are especially important in dry conditions when stomata are partially closed, as harmful oxidative stress may persist internally,13 eventually impairing photosynthesis and accelerating senescence which leads to yield losses in sensitive crops.9,14 Felzer et al.15 used a coupled biosphere-atmosphere model to estimate that, under extreme conditions, surface O3 could reduce global crop yields by up to 20% for wheat and 60% for soybeans by 2100. These reductions stem from the chronic suppression of photosynthesis and biomass accumulation based on dose–response functions from chamber and field experiments built into their model. When these losses were integrated over time and scaled to global agricultural output, the cumulative economic loss was estimated to be up to $8 trillion. This loss would primarily affect major food-producing regions in North America and Asia. Despite recognition of this scale, most large-scale estimates still rely on simplified exposure-response functions,12,16–19 while the mechanistic pathways of O3 damage across growth stages and environments remain poorly resolved. In particular, little is known about how repeated, realistic O3 exposures interact with climatic variability to shape the transition from hidden physiological strain to visible damage and yield loss.
Plant stress can be generally described by three successive phases: (i) an initial state of applied force, (ii) a strain phase in which stress is expressed before visible damage occurs, and (iii) the damage phase, in which acute and chronic injury become visible on the leaf surface.20 These phases are particularly challenging to study under O3 exposure because they require carefully controlled fumigation with a highly reactive and toxic gas. Many experiments have relied on either a single acute pulse of unrealistically high O3 (ref. 21 and 22) or long-term chronic exposures.9,23 In contrast, O3 in the field fluctuates with the diurnal cycle and is punctuated by episodic high-O3 days.5,24 The physiological effects of repeated exposures to realistic, fluctuating concentrations may therefore differ substantially from those observed under strictly acute or chronic treatments.
Soybean, the second most widely grown crop in the United States (U.S.) and a supplier of more than one-third of global production,28 is highly susceptible to elevated O3, which significantly reduces photosynthesis, stomatal conductance (Gs), biomass accumulation, and seed yield.8,9,21 Meta-analyses report average shoot biomass reductions of up to 34% and yield reductions of ∼24% under chronic exposures near 70 parts per billion by volume (ppb) O3.9 The main physiological processes affected by O3 include carboxylation efficiency (the capacity to fix CO2 during photosynthesis), photosystem II efficiency (ΦPSII, the effectiveness of light-driven electron transport), and carbon allocation patterns (the distribution of assimilated carbon between growth and storage).8,22 Variations among cultivars and climatic conditions further complicate these responses.23
At the field scale, the SoyFACE (Soybean Free Air Concentration Enrichment) project provided critical evidence on the effects of chronic O3 under open-air, agronomic conditions. Experiments at SoyFACE demonstrated significant reductions in canopy photosynthesis, earlier leaf senescence, and lower harvest index values in soybeans exposed to elevated O3 (∼60–70 ppb average daytime concentration, sustained across full growing seasons).16,17,25–27 These physiological effects were often undetectable with canopy greenness-based indices such as the normalized difference vegetation index (NDVI), which primarily captures structural leaf area and chlorophyll content. By contrast, solar-induced chlorophyll fluorescence (SIF), a direct proxy for photochemical efficiency, and gas exchange measurements of photosynthesis and Gs provide more sensitive indicators of early stress. Using these approaches, SoyFACE studies identified the strain phase of O3 stress.26 Furthermore, SoyFACE results confirmed reproductive growth stages are especially vulnerable, showing reduced maximum rate of carboxylation (Vcmax), maximum rate of electron transport (Jmax), and faster canopy decline under elevated O3 conditions.25 While SoyFACE and similar experiments provide key insight into soybean responses, they simulated future chronic air quality scenarios by maintaining elevated O3 for entire growing seasons. In contrast, agricultural ecosystems today are often exposed to O3 in shorter, episodic bursts, such as a few hours on high-O3 afternoons, or multiple exceedance events across a season depending on location.28–30 Defining what constitutes “high O3” in agronomic terms, and understanding how repeated realistic exposures differ from strictly acute or strictly chronic treatments, remains an unresolved challenge. These differences are critical for establishing thresholds for O3 damage in soybean12,17 and for clarifying the mechanisms by which plants defend against and recover from varying O3 stress regimes.22,23
Remote sensing offers a useful pathway to investigate these knowledge gaps across agroecosystems. However, traditional remote sensing indices (e.g., NDVI) can only detect the damage phase of stress after visible morphological changes have occurred.31,32 At that stage, damage is often irreversible, as photosynthetic machinery and leaf area are already degraded and yield potential cannot be recovered.9,25 However, if the stressor is removed during the strain phase, the plant can partially recover and reestablish a new physiological standard.20 SIF is a promising parameter for detecting the strain phase earlier. Ground-based SIF measurements can monitor photosynthetic efficiency before visible symptoms develop.26 Recently-available satellite SIF data from, for example, the TROPOspheric Monitoring Instrument (TROPOMI) onboard Sentinel-5P, provides an opportunity to identify crop stress across larger scales and in near-real time. Such analyses, however, require a mechanistic understanding of how crop fluorescence responds to O3 exposure.
This study addresses how repeated, realistic O3 exposures interact with environmental conditions to affect soybean physiology, and the need to evaluate when and how such stress becomes detectable with remote sensing. We combine a five-week plant-level exposure experiment with multi-year satellite observations of soybean fields in Arkansas, U.S. (2018–2021) to investigate O3 stress across spatial and temporal scales. Specifically, we aim to (i) identify the strain phase of O3 stress, when physiological impairment occurs before visible damage; (ii) determine when during the growing season O3 stress is most consequential for soybean development; (iii) assess the potential for recovery following episodic O3 exposure and how this differs between controlled and field conditions; and (iv) evaluate the capacity of remote sensing indicators, particularly SIF, to detect these processes at regional scale. By integrating experimental and remote sensing perspectives, our goal is to improve mechanistic understanding of O3 stress in soybean and contribute to the development of remote sensing-based early warning systems and decision-support tools for farmers and agronomists.
2. Methodology
Our study integrates experiments and observations across spatial and temporal scales. We began with controlled, leaf-level measurements using a portable photosynthesis system (PPS) and extended the analysis to regional-scale using satellite remote sensing data. Conditions in the chamber experiment were designed to replicate the average temperature and relative humidity (RH) observed over four soybean growing seasons (2018–2021) in Crittenden County, Arkansas (Section 2.2) (Fig. S1). The same environmental settings were applied during PPS measurements (Section 2.1) to ensure consistency across scales. This approach allowed us to compare chamber findings with multi-year satellite observations from TROPOMI and the Moderate Resolution Imaging Spectroradiometer (MODIS).We emphasize that chamber and satellite results are not directly comparable because they represent very different spatial scales (leaf vs. field). Instead, our aim was to test whether the mechanisms of O3 stress observed under controlled conditions also emerge at the regional scale, linking mechanistic understanding with applied monitoring. Based on the remote sensing data, soybean fields in Crittenden County did not experience numerous extreme weather or pollution events during the observed period (Fig. S1 and S2), representing relatively healthy agroecosystems – making this study both more challenging and more relevant, as it examines O3 effects under moderate, yet realistic, conditions.
2.1. Plant-level chamber experiments
Soybeans (Glycine max) were grown from seed. Seeds were sown in small containers on 06 February 2025, and germination began on 12 February 2025. Plants grew under controlled laboratory conditions with a standard 12 hours light–dark cycle. Other than during fumigation periods (conducted in a chamber and described below), plants were exposed only to ambient laboratory O3 levels, with daily means of ∼30 ppb.The fumigation chamber (0.61 m × 0.61 m × 0.61 m) was made of transparent, chemical-resistant, polytetrafluoroethylene film with perfluoroalkoxy and polytetrafluoroethylene Swagelok fittings coupled with clear fluorinated ethylene propylene tubing (Fig. 1). A 60 L food-grade CO2 cylinder (Sodastream) maintained mixing ratios in the chamber at ∼400 ppm. Lab air was pushed by a Gast compressor/vacuum pump (DOA-P704-AA) through a bubbler system with deionized water into the chamber to maintain RH at 65 ± 10% throughout the fumigation. Internal chamber temperature was 25 ± 2 °C. A 300 W LED grow light (MARS HYDRO) controlled light intensity at 1000 µmol m−2 s−1, measured at the top of the chamber. A Li-COR CO2/H2O gas analyzer (LI-840A) and a 2B-Technologies O3 monitor (Model 202) continuously measured chamber CO2 and O3 mixing ratios.
The five-week plant-level experiment started on 03 March 2025. During this period, 1–3 leaves from each selected plant (five leaves in total) were measured weekly using a Li-Cor LI-6800 PPS. Parameters included CO2 assimilation (A, rate of CO2 movement through the stomata) stomatal conductance (Gs, movement of CO2 and H2O through the stomata).33 We also measured maximum light-adapted fluorescence 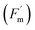 and steady-state fluorescence (Fs) which were used to calculate ΦPSII. PPS leaf chamber conditions were set to 26 ± 0.2 °C and 63 ± 0.5% RH, equivalent to 1.36 ± 0.04 kPa VPD. These conditions matched the growth chamber environment and were representative of field conditions in Crittenden County, Arkansas (see Section 2.2, Fig. S1).
and steady-state fluorescence (Fs) which were used to calculate ΦPSII. PPS leaf chamber conditions were set to 26 ± 0.2 °C and 63 ± 0.5% RH, equivalent to 1.36 ± 0.04 kPa VPD. These conditions matched the growth chamber environment and were representative of field conditions in Crittenden County, Arkansas (see Section 2.2, Fig. S1).
Plants were divided into two groups: three controls and three O3 fumigation plants. Both groups followed the same measurement schedule and chamber conditions. Each week, fumigated plants were exposed for four hours to 80 ± 10 ppb O3 generated by a pen-ray lamp (Analytik Jena AG) connected to the dry air inlet. This exposure corresponds approximately to an 8 hours mean O3 concentration of ∼40 ppb, which is representative of typical daytime O3 levels observed during the soybean growing season (Fig. S2). PPS measurements were taken on five leaves immediately before each exposure (pre-treatment) following a 20 minutes acclimation period, and again immediately after exposure (post-treatment). These measurements were taken immediately before and after weekly O3 exposure in the fumigation chamber; plants were otherwise continuously maintained under identical laboratory conditions. Control plants underwent the same procedure, but without O3 fumigation. This cycle was repeated weekly for five consecutive weeks, during which plants advanced through vegetative growth and began pod development (Fig. 1). The five-week chamber experiment spans early vegetative development and is designed to probe physiological responses to initial and repeated episodic O3 exposure, rather than to represent the full seasonal progression of soybean growth.
Being aware of the limited leaf-level dataset and the importance of distinguishing biological outliers from systematic ones,34,35 we applied the stricter approach of Leys et al.,36 excluding values outside three median absolute deviations (MADs) (Fig. S3). This approach minimizes the impact of errors in measurements while maintaining the most representative physiological responses. We also note that measurements of A in week five may be confounded by plants nearing the end of their life cycle or by limitations to root growth from the small containers.
The results of the chamber experiment are presented in Section 3, including analyses of (i) soybean leaf responses to initial O3 exposure, (ii) recovery potential between repeated exposures, (iii) effects of fumigation on plant growth, and (iv) the behavior of photosynthetic fluorescence parameters under subtle O3 stress.
2.2. Remote sensing analysis
The chosen study area, shown on Fig. 2, located in Crittenden County (Arkansas, U.S.), is a significant soybean-producing region in a state that ranks among the top soybean producers nationally.37 Due to the coarse spatial resolution of the TROPOMI SIF product (7 × 3.5 km), two points of interest were selected over a cluster of large soybean fields (AR1 and AR2).To evaluate the impact of O3 on soybean fields at the regional scale, we integrated multiple datasets (Table 1) (Fig. 3 and S4). Ground-level O3 data from a nearby Environmental Protection Agency (EPA) monitoring station in Crittenden County provided daily 8 hours means and hourly concentrations, the latter used to calculate weekly AOT40 by summing exceedances of O3 above 40 ppb during daylight hours (08:00 to 20:00). Daily meteorological variables, including maximum temperature (Tmax), RH, VPD, grass reference evapotranspiration (ETo), and precipitation (PREC) were obtained from the University of Idaho GRIDMET dataset.38 MODIS datasets provided vegetation and productivity indicators, including 8 days cumulative gross primary production (GPP, the total carbon fixed by plants through photosynthesis summed over 8 days period),39 4 days composite fraction of photosynthetically active radiation (fPAR, the portion of incoming sunlight absorbed by the canopy), 4 days composite leaf area index (LAI, one-sided green leaf area per unit ground area), daily NDVI (derived from surface reflectance, as a measure of canopy greenness), and 3-hourly total photosynthetically active radiation (PAR, incident solar radiation in the visible spectrum (400–700 nanometers)).
| Data | Description | Units | Temporal resolution | Spatial resolution | Source |
|---|---|---|---|---|---|
| O3 | In situ ozone | ppb | Daily | — | EPA |
| SIF | Solar-induced fluorescence at 743 nm | mW m−2 sr−1 nm−1 | Daily | 7 × 3.5 km | ESA-TROPOSIF |
| T max | Maximum temperature | °C | Daily | 4.6 km | GRIDMET |
| RH | Relative humidity | % | Daily | 4.6 km | GRIDMET |
| VPD | Vapor pressure deficit | kPa | Daily | 4.6 km | GRIDMET |
| PREC | Precipitation amount | mm | Daily | 4.6 km | GRIDMET |
| ETo | Grass reference evapotranspiration | mm | Daily | 4.6 km | GRIDMET |
| GPP | Gross primary production | kgC m−2 8 days−1 | 8 day | 0.3 km | MODIS |
| PAR | Photosynthetically active radiation | W m−2 | 3 hour | 0.5 km | MODIS |
| fPAR | Fraction of photosynthetically active radiation | — | 4 day | 0.5 km | MODIS |
| LAI | Leaf area index | — | 4 day | 0.5 km | MODIS |
| NDVI | Normalized difference vegetation index | — | Daily | 0.5 km | MODIS |
SIF 743 nm data were derived from the ESA-TROPOSIF project,40 which uses daily Sentinel-5P TROPOMI measurements. Data from May 2018 to December 2021 were extracted over selected points, filtered to remove negative and extreme values, and averaged between fields to maximize signal coherence. The analysis period covered four soybean growing seasons, each defined as the period from April to September. Within each season, we further subdivided the timeframe into early, peak, and late stages based on SIF dynamics. The peak season range was defined as the four weeks before and after the maximum SIF value. The four weeks prior to the peak period were defined as the early season, and the four weeks after the peak period were defined as the late season, corresponding with senescence and harvesting (Fig. 3).
We aggregated all datasets to a weekly scale for analysis. Weekly water balance (WB) was calculated as the difference between accumulated PREC and ETo. Each of the eight 3-hourly PAR bands per day (in W m−2) was converted to photons using the standard factor for sunlight (1 W m−2 ≈ 4.57 µmol m−2 s−1) and integrated over 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 s (3 hours), summed across the day and then across seven days to obtain weekly incident PAR (mol m−2 weeks−1).41,42 8 day GPP was first divided by 8 to get approximate daily values and then summed across week. We calculated weekly absorbed photosynthetically active radiation (APAR) as APAR = PAR × fPAR, which was used to calculate weekly light use deficiency (LUE) as LUE = GPP/APAR, and SIFyield = SIF/APAR (efficiency of energy conversion from absorbed light into fluorescence).43,44 Following the framework of Massmann et al.,45 we further estimated Gs as a function of GPP and VPD (eqn (1)):
800 s (3 hours), summed across the day and then across seven days to obtain weekly incident PAR (mol m−2 weeks−1).41,42 8 day GPP was first divided by 8 to get approximate daily values and then summed across week. We calculated weekly absorbed photosynthetically active radiation (APAR) as APAR = PAR × fPAR, which was used to calculate weekly light use deficiency (LUE) as LUE = GPP/APAR, and SIFyield = SIF/APAR (efficiency of energy conversion from absorbed light into fluorescence).43,44 Following the framework of Massmann et al.,45 we further estimated Gs as a function of GPP and VPD (eqn (1)):
 | (1) |
This proxy eqn (1) assumes that plants open their stomata during photosynthesis (higher GPP) and close them when the atmosphere becomes drier (higher VPD), helping estimate potential O3 uptake, rather than as a direct measure of stomatal conductance.
The remote sensing allowed us to assess O3 impacts on soybean physiology at the regional scale across four growing seasons. In Section 4, we present these results, including analyses of (i) the effects of early versus late O3 episodes, (ii) the potential for recovery in the weeks following O3 episodes, (iii) the relationship between seasonal O3 accumulation and yield proxies, and (iv) changes in the coupling between vegetation health indicators under different O3 conditions. Comparisons with plant-level chamber experiments are made throughout the section to evaluate whether the mechanisms observed at the leaf level can also be detected across larger spatial and temporal scales.
3. Plant-level O3 fumigation experiment
3.1. Impact of initial O3 exposure on photosynthesis
Initial exposure (Week 1) to O3 had the most pronounced immediate effect on all measured physiological parameters (Fig. 4). In the first week of fumigation, treated plants showed a stronger decrease in the ratio of post-to pre-treatment values for both A and Gs compared to controls, indicating early and acute stress response to O3. This immediate response, which was not observed in following exposures, suggests that plants were not acclimated to the elevated O3 and had not yet activated defense mechanisms, consistent with earlier reports that initial O3 events trigger strong physiological reactions in soybean.9,21 These findings highlight the acute sensitivity of soybean plants during the early vegetative stage and support the idea that timing of exposure is as important as intensity. That is, elevated O3 early in the growing season is likely to cause more lasting damage on photosynthesis than equivalent exposure later.In the following weeks, the pattern was reversed: control plants often showed stronger negative post-treatment reactions than fumigated plants. This finding suggests that the chamber environment itself is a stressor, confounding immediate post-fumigation responses and complicating the separation of O3 effects from chamber effects. Adding O3 as an additional stressor may have altered or masked immediate post-fumigation signals, leading to inconsistent ratios. For this reason, we interpret pre-treatment measurements in subsequent weeks as more reliable indicators of long-term O3 impact, revealing the potential for recovery, which we explore in the next section.
3.2. O3 exposure limits plants' ability to recover photosynthesis and efficiency
Focusing only on pre-treatment values allows us to assess how much plants recovered between exposures. Soybean plants showed limited resilience in photosynthetic function (Fig. 5). A brief decline in pre-treatment values observed in control plants in Week 2 likely reflects short-term acclimation to handling and repeated chamber placement; control plants subsequently stabilized or increased physiological parameters, indicating recovery and continued development under constant growth conditions. After the first fumigation, A and Gs never fully returned to pre-fumigation levels in O3-treated plants – in clear contrast to control plants, which maintained stable or increasing values across the same period. Similarly, ΦPSII declined and remained suppressed in fumigated plants, as opposed to controls. This trend indicates that O3 not only limits carbon assimilation and gas exchange, but also impairs photochemical efficiency, further constraining recovery potential. Thus, while the impact of the first O3 treatment may not have been extreme relative to the control on immediate, post-treatment parameters, plant damage was clear within a week, reflecting interrupted development from the modest four-hour O3 fumigation at 80 ppb. Thus, O3 exposure not only causes an immediate – albeit subtle – stress on the time scale of hours, but also causes a longer-term response of suppressed growth on the time scale of days to weeks. These findings align with other controlled studies showing that repeated O3 exposures compromise recovery processes such as stomatal reopening and enzymatic function in the Calvin cycle.9,23 Suppression of ΦPSII is consistent with reports of O3-induced damage at the chloroplast level,22 as well as with declines in canopy-scale fluorescence under elevated O3.26Pre-treatment measurements show that O3-treated plants failed to recover photosynthetic function between exposures, with sustained suppression of A, Gs, and ΦPSII indicating an ongoing physiological burden that extends beyond short-term inhibition and is reflected in the slower growth patterns described in the following section.
3.3. Subtle O3 exposure suppresses soybean growth and biomass
O3 exposure impacted morphological development. Weekly measurements showed stalk height increasing more slowly in O3-treated plants than in control plants (Fig. 6). By the end of the five-week experiment, average plant height in the fumigated group was consistently lower, even though no visible foliar damage was apparent. Final dry weight (DW) measurements supported this pattern, with non-fumigated plants accumulating more biomass (0.26 ± 0.04 g DW) than fumigated plants (0.21 ± 0.03 g DW). This growth suppression under ambient O3 aligns with prior findings that O3 reduces biomass accumulation even in the absence of necrotic symptoms.9 This effect is likely caused by reduced carbon uptake due to the observed lower assimilation rates (see Section 3.2), combined with a shift in resource allocation from growth processes to antioxidant defense mechanisms.23 | ||
| Fig. 6 Weekly stalk height measurements of soybean plants under O3 fumigation (red) and control conditions (blue) over a five-week period. Bars represent mean stalk height ± SE (n = 5 stalks). | ||
The impact of fumigation on growth persisted despite some recovery of gas exchange parameters in later weeks (see Section 3.2), suggesting that the effect of O3 on developmental processes extends beyond short-term photosynthetic inhibition. This reinforces the concept that chronic, sub-lethal O3 exposure slows physiological development, especially when plants are exposed early in their growth cycle. These results are consistent with SoyFACE observations, where long-term O3 exposure reduced canopy height and biomass even when visual damage was absent.25,26 However, our results further demonstrate that even more subtle and occasional changes in O3, consistent with realistic scenarios currently occurring in agricultural regions, are sufficient to suppress plant growth and reduce biomass.
3.4. Long-term O3 exposure decouples photosynthetic efficiency from fluorescence signals
Further analysis of pre-treatment values, discussed in Section 3.2, revealed that a relationship between ΦPSII and its fluorescence components (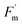 and Fs) weakened and differed between treatments under repeated O3 exposure (Fig. 7). Because ΦPSII is calculated as
and Fs) weakened and differed between treatments under repeated O3 exposure (Fig. 7). Because ΦPSII is calculated as 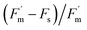 , these variables are not independent and nonlinear behavior is expected; however, the treatment-dependent reduction in explained variance and altered fitted relationships suggests increased variability in fluorescence-efficiency coupling under O3 stress. ΦPSII is usually tightly linked to its fluorescence components, reflecting a balance between absorbed light energy, photochemistry, and non-photochemical quenching.46
, these variables are not independent and nonlinear behavior is expected; however, the treatment-dependent reduction in explained variance and altered fitted relationships suggests increased variability in fluorescence-efficiency coupling under O3 stress. ΦPSII is usually tightly linked to its fluorescence components, reflecting a balance between absorbed light energy, photochemistry, and non-photochemical quenching.46
Observed decoupling indicates an imbalance between electron transport and energy dissipation, characteristic of strain-phase stress responses,20 which implies that plants under chronic O3 exposure may dissipate light energy in ways that no longer track with photochemical efficiency, an early-warning signal of impaired photosynthetic regulation.47 Furthermore, Betzelberger et al.8 argue that ΦPSII is among the most sensitive fluorescence-derived parameters to O3 stress in soybean, even when gas exchange traits partially recover and that indices based solely on Fs may fail to detect strain, highlighting why ΦPSII offers a more mechanistic indicator of early stress.
Note that discussed decoupling was not observed in immediate post-treatment measurements, where 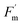 and Fs maintained similar slopes and R2 with ΦPSII for fumigated and non-fumigated plants (Fig. S5). This close relationship reinforces the idea, emphasized in previous sections, that the impact of O3 is not immediately visible after exposure, but emerges over longer timescales and disrupts the functional link between fluorescence signals and ΦPSII efficiency.
and Fs maintained similar slopes and R2 with ΦPSII for fumigated and non-fumigated plants (Fig. S5). This close relationship reinforces the idea, emphasized in previous sections, that the impact of O3 is not immediately visible after exposure, but emerges over longer timescales and disrupts the functional link between fluorescence signals and ΦPSII efficiency.
4. Remote sensing analysis
4.1. Effect of first O3 episode in the growing season is more visible than equivalent exposure later
To test whether the timing of O3 stress modulates crop responses, we identified O3 episodes as weeks when AOT40 increased by more than 350 ppb h relative to the previous week. This threshold was selected to capture episodic O3 anomalies above local background conditions that occur regularly in the study region. For each of the four growing seasons (2018–2021), the earliest such episode was paired with a later episode of equivalent magnitude within the same season (n = 4, Table S1). To minimize influence of other environmental variables, the later episode was chosen to match the first one to have similar PAR, WB, RH, VPD, and Tmax values. Remote sensing vegetation indicators are shown as absolute weekly values (units given in Table 1) and are compared across first versus later O3 episodes (Fig. 8). Here, a negative effect is defined as a reduction in vegetation indicators during the first seasonal O3 episode relative to a later episode of equivalent O3 magnitude (ΔAOT40 > 350 ppb h) within the same growing season, rather than relative to an absolute ambient O3 baseline. Another approach to minimizing seasonality trends was residualizing vegetation indicators against same meteorological covariates; results from the two approaches were consistent (Fig. S6). Comparisons between early and later O3 episodes in the remote sensing analysis are based on seasonal timing at the canopy scale and are not intended to imply direct phenological equivalence with the chamber experiment. | ||
| Fig. 8 Vegetation health indicators values compared between the first and later O3 episodes of equivalent AOT40 increase (>350 ppb h) within each growing season (2018–2021). Panels show (A) SIF, (B) fPAR, (C) Gs, (D) GPP, (E) NDVI, and (F) LAI. Bars represent absolute mean weekly values of each indicator (units as in Table 1) ± SE across four paired episodes; no baseline normalization or temporal smoothing beyond weekly aggregation was applied. Asterisks indicate significance levels (*p < 0.05). | ||
Across all four years, the first O3 episode of the season had a significantly stronger negative impact on functional indicators compared to equivalent exposures later in the season. SIF, GPP, fPAR, and Gs all showed significantly lower values during early-season episodes, while NDVI and LAI, which follow structural canopy development were not significantly different between two episodes. Notably, in 2020 and 2021 the first major O3 episodes occurred later within the early growing period (Table S1), yet the relative suppression during first versus later episodes was comparable to that observed in 2018 and 2019, suggesting that the response is not solely driven by absolute seasonal timing. These findings mirror the leaf-level results (Section 3.1), where the first O3 fumigation caused the largest drop in A and Gs, while following exposures triggered more muted responses as plants initiated partial defense mechanisms. Together, our results emphasize that the soybean is particularly vulnerable to O3 early in its vegetative cycle, when photosynthetic capacity and resource allocation patterns are still being established.8
Physiologically, early vulnerability could be the result of the rapid development of sink capacity, which promotes O3 uptake and amplifies oxidative stress.9,21 At this stage, antioxidant defenses and photoprotective processes have not yet fully developed, leaving ΦPSII particularly vulnerable to disruption.22,23 This seasonal sensitivity is consistent with FACE experiments, in which chronic O3 exposure accelerated senescence and reduced photosynthetic capacity, particularly during reproductive development.25,26 Our results extend these insights by demonstrating that short-term, episodic O3 events, found in agricultural regions – well below regulatory thresholds and chronic FACE exposures – can induce measurable strain when they occur early in the season. The strong suppression of SIF (A) and GPP (D) during these episodes underscores their value as functional indicators for detecting O3 stress before visible canopy changes occur (E and F), in line with regional studies reporting stronger SIF–O3 coupling during early vegetative stages.16,17
4.2. Real-world O3 episodes slow down plant recovery
Using the O3 episodes identified in Section 4.1 (ΔAOT40 > 350 ppb h), we observe how soybean vegetation health indicators recover from their first seasonal O3 event compared to a later event (Fig. 9). Recovery trajectories are based on absolute weekly values of vegetation indicators following identified O3 episodes, without normalization to a baseline. We are aware that values of our vegetation indices could in principle be influenced by differences in canopy development between early and later episodes. However, as shown in Section 4.1, structural parameters such as NDVI and LAI did not differ significantly between the paired events, suggesting that canopy development was not a major confounding factor. | ||
| Fig. 9 Recovery of functional vegetation indicators (GPP, Gs, and SIF) following the first (dark bars) vs. later (light bars) seasonal O3 episodes (ΔAOT40 > 350 ppb h). Bars show absolute mean weekly values of each indicator (units as in Table 1) ± SE across four growing seasons (2018–2021); no baseline normalization or temporal smoothing beyond weekly aggregation was applied. Asterisks indicate significance levels (*p < 0.05). | ||
Results show that functional indicators (GPP, Gs, and SIF) were consistently more suppressed in the week following the first seasonal O3 episode, compared to later episodes. In subsequent weeks, the gap between early and later episodes narrowed, suggesting canopy-scale recovery. This recovery contrasts with the chamber experiment (Section 3.2), where fumigated plants failed to recover fully between weekly exposures. The consistent stability of GPP, Gs, and SIF after later-season O3 episodes suggests that plants may adapt to previously experienced strain. Structural indicators such as LAI, fPAR, and APAR (Fig. S7) showed similar patterns – significant suppression after early episodes followed by recovery. Notably, LAI indicates that canopy development slowed after the first O3 episode, compared to later ones, consistent with chamber findings on growth suppression (Section 3.3).
The multi-week recovery lag observed after early episodes aligns with leaf-level evidence from Section 3.2. As the season progresses, acclimation and phenological shifts likely shorten this lag by increasing sink strength, enhancing antioxidant defences, and adjusting stomatal sensitivity, buffering transient O3 pulses.8,16,23 These processes, and acclimation to an earlier strain phase,20 may explain the reduced sensitivity to later O3 episodes.
4.3. Higher O3 accumulation in early and peak growing season reduces final soybean yield
We used cumulative GPP as a proxy for soybean yield48–50 to examine how seasonal accumulations of AOT40 translate into productivity losses (Fig. 10). Results show that total accumulated O3 across the full season did not correlate with yield (R2 = 0.04, p = 0.81). Instead, exposure during the early growing season alone explained 47% of variance in cumulative GPP, and when early and peak seasons were combined, the relationship strengthened dramatically (R2 = 0.98, p = 0.01). The strong predictive power of early and peak season exposure for final yield also aligns with meta-analyses showing that reproductive and vegetative transitions are the most sensitive phases for soybean yield determination16,17 Given the limited and small number of seasons observed, these cumulative relationships are interpreted as descriptive associations that motivate the within-season analyses, rather than as causal attribution of yield responses to O3 alone.This finding reinforces our plant-level results (Section 3.3), where growth suppression occurred even in the absence of visible foliar damage. Furthermore, we connect findings from Sections 3.1 and 4.1, which showed that early-season exposures leave lasting impacts while later episodes have weaker effects, highlighting that it is not the total seasonal O3 dose that determines yield losses, but rather when an elevated exposure event occurs. Early and peak stages, when plants are expanding canopies and establishing reproductive sinks, are the most consequential for O3 impacts.9,25
For real-world application, we show that monitoring and mitigating O3 during the early and peak growing season is critical for protecting yields, shedding light on potential mechanisms behind regional yield losses reported in the U.S. and Asia, where seasonal O3 episodes often cluster in late spring and early summer.18,19 Early strain detection using SIF and other functional indicators, as demonstrated in previous sections, could allow agronomists and farmers to take adaptive actions before the damage phase occurs and losses become irreversible. The seasonal distribution of O3 further supports this conclusion. For example, 2019 had the highest total accumulated AOT40 but yielded relatively well because much of the exposure occurred late in the season (Fig. S8). By contrast, the high combined early and peak season exposure of 2018 had the lowest yield. These results emphasize that policies and crop management strategies must consider seasonal timing of O3 exposure, not just seasonal totals or regulatory exceedances.
4.4. Accumulated O3 affects relationships between remote sensing vegetation health indicators
We split the early and peak dataset into AOT40 tertiles (≤311, 311–508, ≥508 ppb h), comprising n = 26, 22, and 25 weekly observations, respectively, and within each class fit standardized regressions. We define these tertiles as ‘lower’, ‘middle’, and ‘higher’ O3 exposure. Relationships between GPP-SIF, Gs-SIF, LAI-SIF, fPAR-GPP, and fPAR-SIF, stayed statistically strong across all classes (Fig. S9); the main changes were modest shifts in explained variance rather than large slope breaks. The fact that all of the relationships remained significant across classes suggests that basic canopy coordination is maintained, even if stress reduces its strength. Accordingly, changes in coupling are interpreted as O3-associated patterns under realistic meteorological covariance, rather than as O3-only effects.Moving beyond GPP–SIF–fPAR–Gs relationships, we also examined how O3 alters broader coordination between light absorption, fluorescence, and efficiency. Fig. 11 shows six additional relationships (APAR–GPP, APAR–SIF, LUE–Gs, LUE–SIF, SIFyield–Gs, and SIFyield–LUE) across AOT40 tertiles. These metrics explicitly link absorbed energy, stomatal control, and realized carbon gain, and therefore allow us to assess whether O3 disrupts not just single pairings but the broader chain of processes from light capture to photosynthesis.
Relationships linking absorbed light to productivity (APAR–GPP) and fluorescence (APAR–SIF) remained strong across all classes but weakened under high O3 which indicates that while canopies continued absorbing and re-emitting light, their ability to convert light into carbon gain declined, reflecting reduced LUE.8,23 Similarly, the already weak LUE–Gs relationship eroded with higher O3, consistent with additional biochemical limitations beyond stomatal control.5
Relationships between efficiency metrics collapsed at high O3. The LUE–SIF correlation, low, but present under lower exposures, disappeared in the highest class, suggesting that fluorescence no longer tracks photosynthetic efficiency under O3 stress. This canopy-scale pattern mirrors the leaf-level decoupling observed between ΦPSII and 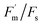 in Section 3.4, reinforcing that O3 disrupts the coherence between photochemical signals and productivity across scales.51 On the other hand, SIFyield–LUE was almost zero under all O3 classes, indicating a full decoupling between fluorescence efficiency and realized carbon gain.
in Section 3.4, reinforcing that O3 disrupts the coherence between photochemical signals and productivity across scales.51 On the other hand, SIFyield–LUE was almost zero under all O3 classes, indicating a full decoupling between fluorescence efficiency and realized carbon gain.
Intermediate relationships, such as SIFyield–Gs, also weakened with rising O3, suggesting that stomatal regulation of fluorescence efficiency is partly maintained but insufficient to buffer stress. These findings highlight that accumulated O3 not only weakens individual linkages but also fragments the entire chain from absorbed light → stomata → fluorescence → carbon gain.
By comparing these canopy-scale patterns with chamber experiments, we see consistent evidence that O3 drives a progressive decoupling of functional indicators. While chambers revealed leaf-level strain through impaired ΦPSII relationships, the remote sensing analysis demonstrates that the same disruption propagates across multiple efficiency metrics at the regional scale. This underscores the value of SIF and derived quantities like SIFyield for identifying when stress pushes canopies from coordinated functioning into a decoupled strain phase.
A sensitivity analysis refitting the within-tertile regressions with meteorological covariates (Fig. S10 and S11) shows that while some relationships remain similar, others change in magnitude and, in some cases, strengthen at higher O3 exposure, indicating non-monotonic responses and changes in functional coupling. We emphasize that although this sensitivity analysis is useful for accounting for key meteorological covariates, it may also mask aspects of O3 influence, as O3 is never isolated from meteorology in real-world systems. For this reason, we do not attribute the observed relationships solely to O3; instead, the tertile-based approach is used to capture changes and non-linear patterns across increasing O3 levels, rather than relying on a binary or fully detrended framework.
5. Conclusions
This study links leaf-level chamber experiments with regional satellite observations to advance mechanistic understanding of how O3 affects soybean physiology under realistic exposure conditions. Across both scales, we find that the timing of O3 stress is more important than its cumulative dose. In chambers, the first fumigation event caused the strongest suppression of photosynthesis (A and Gs) and efficiency (ΦPSII), initiating a strain phase from which plants did not fully recover. Recovery between weekly exposures was incomplete, leading to sustained growth suppression – despite the quite moderate levels of air pollution used herein. A limitation of the plant-level experiment is the small number of samples per treatment, which may limit statistical power and generalizability. However, the purpose of this experiment is to elucidate mechanistic responses to episodic O3 exposure that can be evaluated and contextualized using independent regional-scale remote sensing observations. Accordingly, chamber and satellite analyses are linked through shared response mechanisms (e.g., strain-phase behavior and recovery limitations) rather than through direct alignment of phenological stages.The controlled chamber experiments applied repeated exposures to mildly elevated O3, consistent with conditions typical of North American agroecosystems. We note that fumigation levels were below the thresholds set by the U.S. EPA for human health. The National Ambient Air Quality Standard is based on an 8 hours average of 70 ppb, while we show that exposures equivalent to a weekly 8 hours average of 40 ppb, or a 4 hours average of 80 ppb, were sufficient to cause significant stress and clear reductions in crop productivity. Our findings highlight the need for regulators to consider agricultural impacts when managing O3 precursors, which is particularly urgent given that wildfire smoke, rising temperatures, and other climate-related drivers are likely to increase regional O3 episodes in the future.52,53
At the regional scale, multi-year satellite observations confirmed that early-season O3 episodes produced stronger declines in SIF, GPP, and Gs than equivalent late-season exposures, with recovery lagging for several weeks. Seasonal yield proxies were best explained not by total O3 accumulation, but instead by exposures during early and peak growth phases, when canopies are expanding and reproductive sinks are being established. At this scale, O3 effects are evaluated under realistic environmental covariance, and results highlight changes in functional coordination rather than O3-only attribution. These findings bridge controlled and field conditions, demonstrating that even subtle, episodic O3 events can constrain soybean productivity when they occur at vulnerable stages – highlighting the potential of functional indicators such as SIF to detect stress before irreversible damage occurs.
A limitation of this study is the coarse resolution of the TROPOMI SIF product (∼7 × 3.5 km), which restricts application to small fields. Nevertheless, the physiological mechanisms identified, and detection strategies developed herein, are broadly transferable across agricultural systems. The upcoming ESA FLEX mission, dedicated to fluorescence monitoring at finer spatial scales, carries potential to enhance the applicability of these approaches, enabling earlier and more precise detection of O3 stress. Additionally, while stricter regulation of O3 precursors in agricultural regions could help protect crop productivity, such measures are difficult to implement as high O3 events often coincide with heat waves and drought, which themselves increase precursor emissions. We therefore emphasize the need to reduce ground-level O3 as part of integrated strategies for crop health and global food security in a changing climate.
Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting the findings of this study are available within the article and its supplementary information (SI). Chamber experiment data and processed remote sensing datasets used in this study are available from the corresponding author upon reasonable request. Satellite remote sensing datasets are publicly accessible from ESA-TROPOSIF (SIF), MODIS (NDVI, LAI, fPAR, GPP), and GRIDMET (meteorology). Ground-level O3 observations were obtained from the U.S. Environmental Protection Agency (EPA) Air Quality System (AQS). Supplementary information is available. See DOI: https://doi.org/10.1039/d5em00785b.Acknowledgements
This paper and related research have been conducted within the framework of the Italian National PhD Program in Earth Observation funded by the European Union – Next Generation EU through the Project of Italian Recovery and Resilience Plan (NRRP) (B53C22004370006). The TROPOSIF products were generated by the TROPOSIF team conducted by NOVELTIS under the European Space Agency (ESA) Sentinel-5p+ Innovation activity Contract No. 4000127461/19/I-NS. We acknowledge funding from a National Science Foundation AGS Postdoctoral Research Fellowship (MR; 2306215). We would like to acknowledge the help of Lydia Tonnesen and Alyssa Belanger, who assisted with planting and caring for our soybean plants throughout the entire plant-level experiment. We thank the three anonymous reviewers for their comments, which helped improve this manuscript.References
- D. Dong and J. Wang, Air pollution as a substantial threat to the improvement of agricultural total factor productivity: Global evidence, Environ. Int., 2023, 173, 107842 CrossRef CAS PubMed.
- A. P. K. Tai, M. V. Martin and C. L. Heald, Threat to future global food security from climate change and ozone air pollution, Nat. Clim. Change, 2014, 4(9), 817–9821 CrossRef CAS.
- M. R. Ashmore, Assessing the future global impacts of ozone on vegetation, Plant Cell Environ., 2005, 28(8), 949–964, DOI:10.1111/j.1365-3040.2005.01341.x.
- L. D. Emberson, P. Büker, M. R. Ashmore, G. Mills, L. S. Jackson and M. Agrawal, et al., A comparison of North American and Asian exposure–response data for ozone effects on crop yields, Atmos. Environ., 2009, 43(12), 1945–1953 CrossRef CAS.
- O. E. Clifton, A. M. Fiore, W. J. Massman, C. B. Baublitz, M. Coyle, and L. Emberson, et al., Dry Deposition of Ozone Over Land: Processes, Measurement, and Modeling, in Reviews of Geophysics, Blackwell Publishing Ltd, vol. 58, 2020 Search PubMed.
- S. Fares, F. Savi, J. Muller, G. Matteucci and E. Paoletti, Simultaneous measurements of above and below canopy ozone fluxes help partitioning ozone deposition between its various sinks in a Mediterranean Oak Forest, Agric. For. Meteorol., 2014, 198–199, 181–191 CrossRef.
- G. Mills, H. Pleijel, C. S. Malley, B. Sinha, O. R. Cooper and M. G. Schultz, et al., Tropospheric ozone assessment report: Present-day tropospheric ozone distribution and trends relevant to vegetation, Elementa, 2018, 6, 47 Search PubMed.
- A. M. Betzelberger, C. R. Yendrek, J. Sun, C. P. Leisner, R. L. Nelson and D. R. Ort, et al., Ozone exposure response for U.S. soybean cultivars: Linear reductions in photosynthetic potential, biomass, and yield, Plant Physiol., 2012, 160(4), 1827–1839 CrossRef CAS PubMed.
- P. B. Morgan, E. A. Ainsworth and S. P. Long, How does elevated ozone impact soybean? A meta-analysis of photosynthesis, growth and yield, Plant Cell Environ., 2003, 26(8), 1317–1328 CrossRef CAS.
- Z. Feng, E. Hu, X. Wang, L. Jiang and X. Liu, Ground-level O3 pollution and its impacts on food crops in China: A review, Environ. Pollut., 2015, 199, 42–48 CrossRef CAS PubMed.
- E. A. Ainsworth, Understanding and improving global crop response to ozone pollution, Plant J., 2017, 90(5), 886–897 CrossRef CAS PubMed.
- G. Mills, K. Sharps, D. Simpson, H. Pleijel, M. Broberg and J. Uddling, et al., Ozone pollution will compromise efforts to increase global wheat production, Glob. Change Biol., 2018, 24(8), 3560–3574 CrossRef PubMed.
- A. Y. H. Wong, J. A. Geddes, J. A. Ducker, C. D. Holmes, S. Fares and A. H. Goldstein, et al., New Evidence for the Importance of Non-Stomatal Pathways in Ozone Deposition During Extreme Heat and Dry Anomalies, Geophys. Res. Lett., 2022, 49(8), e2021GL095717 CrossRef CAS.
- J. M. McGrath, A. M. Betzelberger, S. Wang, E. Shook, X. G. Zhu and S. P. Long, et al., An analysis of ozone damage to historical maize and soybean yields in the United States, Proc. Natl. Acad. Sci. U. S. A., 2015, 112(46), 14390–14395 CrossRef CAS PubMed.
- B. S. Felzer, T. Cronin, J. M. Reilly, J. M. Melillo and X. Wang, Impacts of ozone on trees and crops, C. R. Geosci., 2007, 339(11–12), 784–798 CrossRef CAS.
- C. M. Montes, H. J. Demler, S. Li, D. G. Martin and E. A. Ainsworth, Approaches to investigate crop responses to ozone pollution: from O3-FACE to satellite-enabled modeling, Plant J., 2022, 109(2), 432–446, DOI:10.1111/tpj.15501.
- Z. Feng and K. Kobayashi, Assessing the impacts of current and future concentrations of surface ozone on crop yield with meta-analysis, Atmos. Environ., 2009, 43(8), 1510–1519 CrossRef CAS.
- A. P. K. Tai, M. Sadiq, J. Y. S. Pang, D. H. Y. Yung and Z. Feng, Impacts of Surface Ozone Pollution on Global Crop Yields: Comparing Different Ozone Exposure Metrics and Incorporating Co-effects of CO2, Front. Sustain. Food Syst., 2021, 5, 534616 CrossRef.
- F. Leung, S. Sitch, A. P. K. Tai, A. J. Wiltshire, J. L. Gornall and G. A. Folberth, et al., CO2 fertilization of crops offsets yield losses due to future surface ozone damage and climate change, Environ. Res. Lett., 2022, 17(7), 074007 CrossRef.
- M. Meroni, C. Panigada, M. Rossini, V. Picchi, S. Cogliati and R. Colombo, Using optical remote sensing techniques to track the development of ozone-induced stress, Environ. Pollut., 2009, 157(5), 1413–1420 CrossRef CAS PubMed.
- E. Singh, S. Tiwari and M. Agrawal, Effects of elevated ozone on photosynthesis and stomatal conductance of two soybean varieties: A case study to assess impacts of one component of predicted global climate change, Plant Biol., 2009, 11(suppl.1), 101–108 CrossRef CAS PubMed.
- C. P. Chen, T. D. Frank and S. P. Long, Is a short, sharp shock equivalent to long-term punishment? Contrasting the spatial pattern of acute and chronic ozone damage to soybean leaves via chlorophyll fluorescence imaging, Plant Cell Environ., 2009, 32(4), 327–335 CrossRef CAS PubMed.
- S. A. Osborne, G. Mills, F. Hayes, E. A. Ainsworth, P. Büker and L. Emberson, Has the sensitivity of soybean cultivars to ozone pollution increased with time? An analysis of published dose-response data, Glob. Change Biol., 2016, 22(9), 3097–3111 CrossRef.
- S. C. Kavassalis and J. G. Murphy, Understanding ozone-meteorology correlations: A role for dry deposition, Geophys. Res. Lett., 2017, 44(6), 2922–2931 CrossRef.
- E. A. Ainsworth, S. P. Serbin, J. A. Skoneczka and P. A. Townsend, Using leaf optical properties to detect ozone effects on foliar biochemistry, Photosynth. Res., 2014, 119(1–2), 65–76 CrossRef CAS PubMed.
- G. Wu, K. Guan, E. A. Ainsworth, D. G. Martin, H. Kimm and X. Yang, Solar-induced chlorophyll fluorescence captures the effects of elevated ozone on canopy structure and acceleration of senescence in soybean, J. Exp. Bot., 2024, 75(1), 350–363 CrossRef CAS PubMed.
- D. M. Eastburn, M. M. Degennaro, E. H. Delucia, O. Dermody and A. J. Mcelrone, Elevated atmospheric carbon dioxide and ozone alter soybean diseases at SoyFACE, Glob. Change Biol., 2010, 16(1), 320–330 CrossRef.
- X. Liu and A. R. Desai, Significant Reductions in Crop Yields From Air Pollution and Heat Stress in the United States, Earths Future, 2021, 9(8), e2021EF002000, DOI:10.1029/2021EF002000.
- O. R. Cooper, K. L. Chang, K. Bates, S. S. Brown, W. S. Chace and M. M. Coggon, et al., Early Season 2023 Wildfires Generated Record-Breaking Surface Ozone Anomalies Across the U.S. Upper Midwest, Geophys. Res. Lett., 2024, 51(22), e2024GL111481, DOI:10.1029/2024GL111481.
- K. L. Chang, B. C. McDonald, C. Harkins and O. R. Cooper, Surface ozone trend variability across the United States and the impact of heat waves (1990–2023), Atmos. Chem. Phys., 2025, 25(10), 5101–5132 CrossRef CAS.
- A. K. Mahlein, E. C. Oerke, U. Steiner and H. W. Dehne, Recent advances in sensing plant diseases for precision crop protection, Eur. J. Plant Pathol., 2012, 133(1), 197–209, DOI:10.1007/s10658-011-9878-z.
- P. J. Pinter, J. L. Hatfield, J. S. Schepers, E. M. Barnes, M. S. Moran and C. S. T. Daughtry, et al., Remote Sensing for Crop Management, Photogramm. Eng. Rem. Sens., 2003, 69(6), 647–664 CrossRef.
- M. Riches, D. Lee and D. K. Farmer, Simultaneous leaf-level measurement of trace gas emissions and photosynthesis with a portable photosynthesis system, Atmos. Meas. Tech., 2020, 13(8), 4123–4139 CrossRef CAS.
- C. Julián, S. Villadangos, L. Jené, O. Pasques, M. Pintó-Marijuan and S. Munné-Bosch, Biological outliers: essential elements to understand the causes and consequences of reductions in maximum photochemical efficiency of PSII in plants, Planta, 2024, 260(1), 32 CrossRef PubMed.
- S. Alvarez Prado, I. Sanchez, L. Cabrera-Bosquet, A. Grau, C. Welcker and F. Tardieu, et al., To clean or not to clean phenotypic datasets for outlier plants in genetic analyses?, J. Exp. Bot., 2019, 70(15), 3693 CrossRef.
- C. Leys, C. Ley, O. Klein, P. Bernard and L. Licata, Detecting outliers: Do not use standard deviation around the mean, use absolute deviation around the median, J. Exp. Soc. Psychol., 2013, 49(4), 764–766 CrossRef.
- J. Ross, Arkansas Soybean Research Studies 2019, Arkansas Agricultural Experiment Station Research Series, 2020, https://scholarworks.uark.edu/aaesser/166 Search PubMed.
- J. T. Abatzoglou, Development of gridded surface meteorological data for ecological applications and modelling, Int. J. Climatol., 2013, 33(1), 121–131, DOI:10.1002/joc.3413.
- N. P. Robinson, B. W. Allred, W. K. Smith, M. O. Jones, A. Moreno and T. A. Erickson, et al., Terrestrial primary production for the conterminous United States derived from Landsat 30 m and MODIS 250 m, Remote Sens. Ecol. Conserv., 2018, 4(3), 264–280, DOI:10.1002/rse2.74.
- L. Guanter, C. Bacour, A. Schneider, I. Aben, T. A. Van Kempen and F. Maignan, et al., The TROPOSIF global sun-induced fluorescence dataset from the Sentinel-5P TROPOMI mission, Earth Syst. Sci. Data, 2021, 13(11), 5423–5440 CrossRef.
- D. Wang, S. Liang, Y. Zhang, X. Gao, M. G. L. Brown and A. Jia, A New Set of MODIS Land Products (MCD18): Downward Shortwave Radiation and Photosynthetically Active Radiation, Remote Sens., 2020, 12(1), 168 CrossRef.
- I. Lozano, I. Alados, G. Sánchez-Hernández, J. L. Guerrero-Rascado and I. Foyo-Moreno, Improving the Estimation of the Diffuse Component of Photosynthetically Active Radiation (PAR), J. Geophys. Res. Atmos., 2023, 128(22), e2023JD039256, DOI:10.1029/2023JD039256.
- T. S. Magney, D. R. Bowling, B. A. Logan, K. Grossmann, J. Stutz and P. D. Blanken, et al., Mechanistic evidence for tracking the seasonality of photosynthesis with solar-induced fluorescence, Proc. Natl. Acad. Sci., 2019, 116(24), 11640–11645, DOI:10.1073/pnas.1900278116.
- G. Miao, K. Guan, X. Yang, C. J. Bernacchi, J. A. Berry and E. H. DeLucia, et al., Sun-Induced Chlorophyll Fluorescence, Photosynthesis, and Light Use Efficiency of a Soybean Field from Seasonally Continuous Measurements, J. Geophys. Res.:Biogeosci., 2018, 123(2), 610–623, DOI:10.1002/2017JG004180.
- A. Massmann, P. Gentine and C. Lin, When Does Vapor Pressure Deficit Drive or Reduce Evapotranspiration?, J. Adv. Model. Earth Syst., 2019, 11(10), 3305–3320, DOI:10.1029/2019MS001790.
- K. Oxborough and N. R. Baker, Resolving chlorophyll a fluorescence images of photosynthetic efficiency into photochemical and non-photochemical components - Calculation of qP and Fv’/Fm’ without measuring Fo’, Photosynth. Res., 1997, 54(2), 135–142, DOI:10.1023/A:1005936823310.
- A. V. Ruban and E. H. Murchie, Assessing the photoprotective effectiveness of non-photochemical chlorophyll fluorescence quenching: A new approach, Biochim. Biophys. Acta, Bioenerg., 2012, 1817(7), 977–982 CrossRef CAS PubMed.
- P. M. Wurster, M. Maneta, J. S. Kimball, K. A. Endsley and S. Beguería, Monitoring Crop Status in the Continental United States Using the SMAP Level-4 Carbon Product, Front. Big Data, 2021, 3, 597720 CrossRef PubMed.
- C. Dold, J. L. Hatfield, J. H. Prueger, T. B. Moorman, T. J. Sauer and M. H. Cosh, et al., Upscaling Gross Primary Production in Corn-Soybean Rotation Systems in the Midwest, Remote Sens., 2019, 11(14), 1688 CrossRef.
- M. Marshall, K. Tu and J. Brown, Optimizing a remote sensing production efficiency model for macro-scale GPP and yield estimation in agroecosystems, Remote Sens. Environ., 2018, 217, 258–271 CrossRef.
- J. Chen, S. Wang, K. Zhu, B. Chen, Q. Wang and L. Zhang, et al., Tropospheric ozone alters solar-induced chlorophyll fluorescence and its relationship with gross primary production in a subtropical evergreen forest, Agric. For. Meteorol., 2025, 372, 110695 CrossRef.
- L. Xu, J. D. Crounse, K. T. Vasquez, H. Allen, P. O. Wennberg and I. Bourgeois, et al., Ozone chemistry in western U.S. wildfire plumes, Sci. Adv., 2021, 7(50), 3648 CrossRef PubMed.
- S. E. Pusede, A. L. Steiner and R. C. Cohen, Temperature and Recent Trends in the Chemistry of Continental Surface Ozone, Chem. Rev., 2015, 115(10), 3898–3918, DOI:10.1021/cr5006815.
| This journal is © The Royal Society of Chemistry 2026 |









