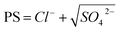Sources, geochemical characteristics, and control mechanisms of groundwater salinity: a case study of the Ulungur River Basin in northwest China
Chenyang
Tian
a and
Hua
Tian
 *ab
*ab
aCollege of Geology and Environment, Xi'an University of Science and Technology, Xi'an, 710054, China. E-mail: tianhua@xust.edu.cn; Fax: +86 180 6663 5829
bShaanxi Provincial Key Laboratory of Geological Support for Coal Green Exploitation, Xi'an, 710054, China
First published on 31st October 2025
Abstract
Groundwater salinisation in inland water-scarce areas exacerbates various ecological, environmental, and social issues. To obtain a comprehensive understanding of the primary mechanisms driving groundwater salinisation and the baseline water quality in the Ulungur River Basin (URB), this study integrated hydrochemical analysis, geostatistical methods, and multivariate statistical techniques. In addition, rational recommendations for the sustainable exploitation and protection of water resources were proposed. Analysis of the dissolved components revealed that groundwater chemistry was predominantly influenced by Na+ (365.62 mg L−1), Ca2+ (205.91 mg L−1), Mg2+ (62.18 mg L−1), Cl− (276.96 mg L−1), and SO42− (817.45 mg L−1). Groundwater in the Low Mountain region was categorised as freshwater, which gradually turned to saline water in the Lacustrine Plain along the flow direction, with hydrochemical types evolving from HCO3·SO4–Na·Ca and SO4·HCO3–Na·Ca (Mg) to SO4–Na·Ca and SO4·Cl–Na·Ca. Principal component analysis identified four principal components (PCs) that collectively accounted for 80.53% of the total cumulative variance in the key determinants of groundwater salinisation. PC1 represented water–rock interactions (which included carbonate and evaporite dissolution or precipitation and cation exchange). PC2 represented the degradation of organic matter and the application of farm manure. PC3 was associated with the return flow of irrigation water and lateral recharge. PC4 involved domestic sewage discharge and fertiliser application. The calculated values of the water quality index indicated that 47% of the samples, classified as having either excellent or good water quality, were suitable for drinking. Furthermore, the results of the permeability index, sodium adsorption ratio, residual sodium carbonate, and potential salinity indicated that both river water and groundwater within the riparian zone were safe and suitable for irrigation purposes. Overall, reducing river water extraction, upgrading agricultural production technologies, and enhancing domestic sewage treatment capacities are key strategies for protecting water resources in the URB.
Environmental significanceThis study provides immediate insights into groundwater salinization, a critical issue in water-scarce regions facing dual threats to water quality and quantity. By integrating multivariate statistical methods and ion ratio analysis, we elucidate the mechanisms of groundwater salinization across different geomorphological types. Our results highlight key drivers, including water–rock interactions, CO2 and pH changes, groundwater depth, and human activities. This work directly informs water resource protection and environmental risk management, offering valuable strategies to mitigate salinization and promote sustainable water use in the Ulungur River Basin. Overall, our findings contribute to advancing knowledge of environmental processes and guiding effective environmental management. |
1 Introduction
In arid and semi-arid areas, groundwater plays a critical role in supplying water for habitation and agricultural production and maintaining ecosystem balance.1 In recent decades, rapid population and economic growth, along with climate change, have gradually exacerbated the salinisation and pollution of groundwater.2,3 Consequently, water quality has deteriorated, biodiversity and bio-functionality have declined, and ecosystem stability and sustainability are threatened.4–6 Furthermore, the systematic characterisation of groundwater hydrochemical evolution, quantitative identification of the primary controlling factors of salinisation, and establishment of regional water quality baselines in the Ulungur River Basin (URB) of northwestern China remain relatively unexplored. This knowledge gap hinders the development of scientifically consistent water resource management strategies in this water-scarce region. Therefore, understanding the hydrochemical features, mineralisation mechanisms, and background water quality is crucial for protecting groundwater resources and maintaining regional ecological balance.7,8Numerous studies have demonstrated that groundwater salinisation is due to the interaction of natural and human factors.9,10 Long-term hydrogeological and topographical conditions influence natural factors, which ultimately results in various processes such as mineral dissolution/precipitation, evaporation, cation exchange, saline water mixing, and deep saline water invasion.11–22 These processes ultimately increase groundwater salinity and establish specific hydrochemical distribution patterns at local and regional scales. Human factors, which significantly influence the degree of groundwater salinisation within a relatively short time, mainly refer to the impacts of human activities superimposed on natural evolution.23 These influences include the emission of nitrates and organic contaminants from domestic sewage, industrial wastewater, and over-fertilisation in agriculture,24–27 alterations in natural runoff patterns and hydraulic connections owing to overexploitation or water diversion projects,28–31 irrigation water return flow,32–34 and the expansion of urbanisation and industrialisation.35,36 Exceeding the carrying capacity of the local or regional water environment can lead to significant alterations in the groundwater chemical environment across multiple spatial scales, which eventually affect the ecological environment.37–40
Given the complexity and variability of the groundwater chemical environment,41 numerous studies have employed multivariate mathematical analysis, hydrochemistry, and geochemical simulation approaches to elucidate the hydrochemical characteristics and the evolution processes of groundwater salinisation and contamination.9,36,42–44 Notably, the primary anions and cations influencing the hydrochemical features of groundwater and the mineral sources of these ions have been identified using Pearson correlation coefficients (PCCs).45,46 The principal natural and/or anthropogenic factors controlling groundwater salinisation have been comprehensively deduced via principal component analysis (PCA) dimension reduction.1,47,48 Furthermore, the types of water–rock interactions, the sources of pollutants, and the impacts of various human activities have been determined using ion ratios.49–51 The chemical reaction pathways and the dissolution and precipitation states of relevant minerals have been delineated through hydrogeochemical simulation.52,53 To effectively protect groundwater resources and optimise management strategies, several studies have established various approaches and indicators for evaluating water quality. These methods are as follows: (1) the single factor assessment method, which focuses solely on the most impactful water quality parameters while neglecting others;54,55 (2) the comprehensive water quality index (WQI) method, which integrates multiple physicochemical parameters into a single index to determine the overall water quality level;56,57 and (3) other assessment indicators such as the chlorinity index, percent sodium, total hardness, permeability index (PI), and sodium adsorption ratio (SAR).58,59 Notably, the ecological environment in water-scarce regions is highly sensitive and fragile, as even minor changes in the chemical characteristics of groundwater can significantly disrupt essential ecological processes.60,61 This sensitivity underscores the need for understanding groundwater origins, flow patterns, hydrogeochemical evolution, and water quality to maintain the systematic functioning of ecological processes. However, the absence of systematic and comprehensive research in this area poses substantial challenges for the protection of groundwater resources.
The URB, located in Xinjiang, encompasses arid and semi-arid regions in northwest China and is classified as an inland river basin (Fig. 1a). Over the past three decades, the area of cultivated land and construction land has significantly increased by 867.61 km2 and 13.63 km2, respectively.59 Conversely, grassland area has decreased by 113.05 km2. This reduction has led to a substantial increase in groundwater resource consumption and a corresponding decline in groundwater levels. These conditions have resulted in reduced river flow, soil salinisation, desertification, and soil erosion.62 Furthermore, the basin is situated at the ecotone between desert and oasis, characterised by an extremely vulnerable and sensitive ecological environment. The depletion of groundwater resources, crucial for maintaining ecological equilibrium, poses substantial risks to the ecosystem of the URB.59,63 Despite previous research addressing fundamental ecological issues in the URB, studies focused on the groundwater quality of this region are scarce. This scarcity severely limits current efforts to improve the ecological environment. Thus, the main objectives of this study are to (1) assess the hydrochemical characteristics of groundwater, (2) identify groundwater circulation patterns, (3) determine the primary mechanisms controlling groundwater salinisation, (4) delineate the quality of groundwater and surface water for drinking and irrigation, and (5) propose rational and effective recommendations for the protection of groundwater resources.
2 Study area
2.1 Location, climate and major surface water
The URB is located within the Altay region (Fig. 1b), 86°59′–90°05′E, 46°06′–47°38′N, with a total area of ∼38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 426 km2. The region features a temperate continental arid climate characterised by short summers and relatively long, cold winters. The mean annual temperature is 5.5 °C, with the lowest temperatures typically recorded between January and February (−20.25 °C) and the highest between July and August (28.3 °C). Average annual precipitation is 224.3 mm in the eastern low mountain areas. This amount gradually decreases to 117.06 mm in the western plains. The annual average evaporation rate is 1725.1 mm.64
426 km2. The region features a temperate continental arid climate characterised by short summers and relatively long, cold winters. The mean annual temperature is 5.5 °C, with the lowest temperatures typically recorded between January and February (−20.25 °C) and the highest between July and August (28.3 °C). Average annual precipitation is 224.3 mm in the eastern low mountain areas. This amount gradually decreases to 117.06 mm in the western plains. The annual average evaporation rate is 1725.1 mm.64
The Ulungur River (R), Jili Lake (J), and Ulungur Lake (U) are the major surface waters of the URB. The R flows from the Altay Mountains and is fed by snowmelt and precipitation. The flow of R is determined by the volume of precipitation, as it exhibits seasonal variations. The R flows through Qinghe and Fuyun Counties and eventually discharges into J before returning to U in the southwest corner of Fuhai County. Originally, U served as the terminal lake for the R. However, since the Oligocene epoch, continuous tectonic activity, which includes tilted uplift and right-lateral strike–slip shearing in the Altay Mountains from the north to the southwest and anticlinal uplift in the Derun Mountains during the middle Quaternary, has led to the collapse of the lake basin. This collapse results in R flowing into J.65,66
2.2 Geological and hydrogeological settings
The URB is associated with the Piedmont peneplain region located at the northeastern edge of the Junggar Basin and the southwestern foothills of the Altay Mountains (Fig. 1). The overall terrain exhibits an irregular distribution from northeast to southwest, with altitudes gradually decreasing. Owing to Meso–Cenozoic endogenetic geological processes, such as crustal uplift and decline, alongside external forces including prolonged weathering and denudation, surface runoff, and biological growth, the area has evolved into a low plain peneplain that connects mountains with contemporary geomorphic features. In this study, the surface morphology and lithologic combinations were classified into seven landforms from east to west based on the genesis of the URB, as follows: Low Mountain (LM), Hilly Region (HR), Erosion Plain (EP), River Valley (RV), Southern Alluvial Plain (SAP), Northern Alluvial Plain (NAP), and Lacustrine Plain (LP) (Fig. 1c). The main lithologic profile is shown in Fig. 2.Field investigations and hydrochemical analyses of groundwater reveal that the tertiary uplift between the SAP and NAP disrupts the hydraulic connection between these two landforms, while the near east–west syncline structure to the north of the NAP serves as a natural watershed separating the NAP from BP.
The exposure strata of the URB range from the Devonian to the Quaternary periods. The Devonian strata are predominantly located in the LM and HR regions, characterised by significant fissure development, with groundwater classified as bedrock fissure water. Field investigations reveal that the spring water in the HR is relatively well-developed and concentrated between the HR and the northwestern boundary of the LM. The Quaternary sedimentary layer predominantly occupies the RV, SAP, NAP, LP, and the river valley bottom of the LM. Its lithology mainly consists of sand, gravel, sandy soil, carbonate rocks, and calcareous cement. Notably, particle sizes decrease from east to west, with field drilling revealing a thin gypsum layer in the RV. This sedimentary layer serves as the primary burial area for phreatic water, with thicknesses varying from <1 m in the LM to >20 m in the LP. Traditional and outdated flooding irrigation practices in the NAP and LP have increased groundwater levels, accompanied by the formation of extensive salt crusts on the surface in these areas. The Paleogene and Neogene strata are widely exposed in the EP, while the Quaternary layer at the surface is permeable but lacks water. Field investigations indicate that water yield properties are relatively abundant in the SAP and LP, ranging from 500 to 1000 m3 day−1, whereas the RV and NAP exhibit poor yields of 100–500 m3 day−1. Notably, the middle section of the RV (DuRe) exhibits abundant water yields, ranging from 1000 to 5000 m3 day−1. The water yield properties in the LM and HR are moderate and poor, measuring 0.1–1 L s−1 and <0.1 L s−1, respectively (Fig. 1c). The groundwater in the URB originates from the eastern mountains, with its flow direction aligning with that of the river. The groundwater ultimately flows into terminal lake areas, where its availability is affected by excessive exploitation and intense evaporation along the route.64,67
3 Materials and methods
3.1 Sampling and analysis
For this study, 103 water samples were collected in July and August 2018. The collected samples comprised 24 surface water samples (12 from the R, 6 from J, and 6 from U) and 79 groundwater samples (Fig. 1c). All groundwater samples were collected in polyethene bottles (150 mL) from civil wells (phreatic aquifers), with each bottle pre-washed at least three times using the respective water samples before sampling. For the sampling of surface water, the sampler was positioned 50 cm below the water table to ensure adequate sample collection. Groundwater wells were pumped until the electrical conductivity (EC) stabilised. Furthermore, all collected samples were stored in sealed containers at a low temperature (4 °C) after on-site filtering using a 0.45 µm filter membrane.68,69 To elucidate the conversion relationship between the R and the groundwater, 15 points were selected for the measurement of the cross-sectional flow of R (Q).Water temperature, total dissolved solids (TDS), pH, and EC were measured in the field using a portable multi-parameter device (Hach HQ40d). The depth of groundwater (D) was also recorded. Chemical analyses of all samples were performed at the Institute of Hydrogeology and Environmental Geology, Chinese Academy of Geological Sciences, located in Shijiazhuang, Hebei Province, China. Key cations, such as potassium (K+), sodium (Na+), calcium (Ca2+), and magnesium (Mg2+), were quantified via inductively coupled plasma atomic emission spectrometry (ICAP-6300). Chloride (Cl−) and sulphate (SO42−) concentrations were determined via ion chromatography (ICS-1100). Nitrate (NO3−) concentration was measured via ultraviolet spectrophotometry. Bicarbonate (HCO3−) and free carbon dioxide (CO2) concentrations were measured via titration using 0.05 mol L−1 hydrochloric acid (HCl) and 0.05 mol L−1 sodium hydroxide (NaOH), respectively. The detection limit for all ions was 0.05 mg L−1, and the ionic electrical balance was maintained within ±5% for all water samples.
Fig. 3 shows the main methodological protocols for data collection, pre-processing, and analysis, which include descriptive and multivariate statistics, geostatistics, and ionic ratios. Except for pH, which exhibited a normal distribution, all other parameters (K+, Na+, Ca2+, Mg2+, HCO3−, Cl−, SO42−, NO3−, CO2, TDS, and H) were positively skewed, with a mean skewness value of 2.19. To ensure the reliability of the mathematical statistical analysis outcomes and their interpretations, a logarithmic transformation was applied to all skewed parameters.70 Furthermore, all parameters were standardised before multivariate statistical analysis to eliminate dimensional constraints.71 Data pre-processing and analysis were conducted using SPSS version 18.0, while ArcGIS version 10.6 was utilised for geostatistical analysis.
3.2 Multivariate statistical analysis
Multivariate statistical analysis is an indispensable tool for investigating complex systems characterised by interconnected multiple indices. This approach simplifies data and extracts key relationships along with underlying statistical laws. Correlation analysis evaluates the strength of the relationships between different datasets using the magnitude of PCCs. The absolute value of the correlation coefficient is typically categorised into three grades, as follows:72 weak (0 < r < 0.3), moderate (0.3 < r < 0.5), and strong (0.5 < r < 1.0). Furthermore, PCCs are frequently employed to trace the potential mineral sources of each ion, which ultimately provides critical insights for further research on the evolution of groundwater hydrogeochemistry.PCA is a statistical method that recombines multiple complex variables into a smaller set of new variables while retaining as much of the original information as possible.73 This method utilises an orthogonal transformation to eliminate secondary factors and extracts principal components (PCs) with eigenvalues > 1 via variance maximisation and rotation processing.36 Considering the complexity of hydrogeochemical systems, which are influenced by multiple variables and are vulnerable to external environmental factors, PCA is widely utilised to identify the primary factors—both natural and anthropogenic—influencing hydrogeochemical evolution.
3.3 Geostatistical analysis
Conventional statistical analysis mainly focuses on the relationships between sample values while neglecting critical information about the spatial positioning of samples and the distances between them.74 To elucidate the spatial distribution of groundwater with varying salinity and pollution levels and to provide essential preliminary data for subsequent analyses of the mechanisms controlling salinisation and rational recommendations for groundwater resource protection, this study employed the kriging analysis method within the ArcGIS 10.6 geostatistical analysis module. In addition, a visual analysis of the spatial evolution of the concentrations of groundwater TDS and NO3− was conducted.3.4 Water applicability evaluation for different purposes
(a). Each parameter was assigned a weight (wi) ranging from 1 to 5, which reflected the perceived severity of its effects on human health and its relative importance for drinking water quality. In this study, 10 groundwater parameters (K+, Na+, Ca2+, Mg2+, Cl−, SO42−, HCO3−, TDS, pH, and NO3−) and 9 surface water parameters (excluding NO3−) were utilised to compute the WQI. The World Health Organization (WHO) standard limits were adopted for each parameter.76Table 1 details the assigned weights (wi).
| WHO standard (2011) | Groundwater | Parameters | Surface water | |||
|---|---|---|---|---|---|---|
| Parameters | Weight (wi) | Relative weight (Wi) | Weight (wi) | Relative weight (Wi) | ||
| a The higher the weight (wi), the greater the influence on water quality. | ||||||
| TDS: 1000 | TDS | 5 | 0.1316 | TDS | 5 | 0.1471 |
| Na+: 200 | Na+ | 5 | 0.1316 | Na+ | 5 | 0.1471 |
| SO42−: 250 | SO42− | 5 | 0.1316 | SO42− | 5 | 0.1471 |
| NO3−: 50 | NO3− | 4 | 0.1081 | pH | 4 | 0.1176 |
| pH: 7∼8.5 | pH | 4 | 0.1053 | Ca2+ | 4 | 0.1176 |
| Cl−: 250 | Cl− | 4 | 0.1053 | HCO3− | 4 | 0.1176 |
| Ca2+: 200 | Ca2+ | 4 | 0.1053 | Mg2+ | 3 | 0.0882 |
| Mg2+: 150 | Mg2+ | 3 | 0.0789 | Cl− | 3 | 0.0882 |
| HCO3−: 380 | HCO3− | 3 | 0.0789 | K+ | 1 | 0.0294 |
| K+: 3 | K+ | 1 | 0.0263 | |||
| ∑wi = 38 | ∑Wi = 1 | ∑wi = 34 | ∑Wi = 1 | |||
(b). The weights of these parameters were incorporated into eqn (1) to obtain the relative weights (Wi). Table 1 presents the results for all parameters.
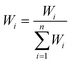 | (1) |
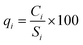 | (2) |
(d). Eqn (3) was utilised to calculate the WQI. The calculated WQI values were categorised into the following five ranks:57 excellent water (<50, suitable for drinking), good water (50–100, safe for drinking), poor water (100–200, not safe for drinking without treatment), very Poor water (200–300, unsafe for drinking), and unsuitable water (>300, unsuitable for drinking).
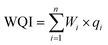 | (3) |
For eqn (1)–(3), i represents the chemical parameters for water, n denotes the total number of parameters, and Ci and Si represent the test concentration of each parameter and the WHO standard values for drinking water, respectively.
4 Results
4.1 Hydrochemistry features
Table 3 presents the statistical results, which include the mean, maximum, and minimum values of the physicochemical parameters for surface water (n = 24) and groundwater (n = 73). The Piper diagram (Fig. 4) shows the hydrochemical types of all water samples.| Source | Value | pH | TDS | K+ | Na+ | Ca2+ | Mg2+ | Cl− | SO42− | HCO3− | CO2 | NO3− | H |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mg L−1 | m | ||||||||||||
| Groundwater samples (LM, HR, RV, SAP, NAP and LP); surface water samples (U, J and R). “-”: No detection; CO2: free carbon dioxide; H (m): groundwater depth. | |||||||||||||
| LM n = 8 | Mean | 7.26 | 701.09 | 3.66 | 139.33 | 68.75 | 21.18 | 60.28 | 247.96 | 299.28 | 18.95 | 6.46 | 4.78 |
| Max | 7.51 | 1127.30 | 5.86 | 232.70 | 125.20 | 33.94 | 112.80 | 560.00 | 390.50 | 32.00 | 9.80 | 7.10 | |
| Min | 6.86 | 494.10 | 1.86 | 69.40 | 33.32 | 5.42 | 28.40 | 144.10 | 181.40 | 15.00 | 3.30 | 3.60 | |
| HR n = 7 | Mean | 7.27 | 2746.15 | 5.02 | 533.29 | 274.77 | 87.22 | 492.06 | 1187.47 | 288.81 | 21.92 | 9.39 | 4.78 |
| Max | 7.67 | 5413.40 | 14.20 | 639.70 | 801.70 | 243.00 | 799.40 | 3073.90 | 414.90 | 30.00 | 17.90 | 12.10 | |
| Min | 7.10 | 1821.50 | 1.09 | 430.90 | 44.10 | 24.30 | 321.70 | 576.40 | 116.60 | 7.50 | 1.70 | 0.00 | |
| RV n = 9 | Mean | 7.31 | 829.62 | 8.73 | 118.68 | 130.15 | 36.34 | 90.38 | 315.38 | 278.38 | 22.39 | 10.92 | 2.35 |
| Max | 7.56 | 1616.20 | 46.98 | 360.60 | 255.00 | 99.00 | 183.70 | 736.70 | 583.20 | 52.40 | 39.40 | 5.60 | |
| Min | 6.69 | 425.40 | 1.90 | 40.70 | 48.10 | 14.60 | 28.40 | 99.90 | 109.80 | 3.70 | 1.40 | 1.00 | |
| SAP n = 9 | Mean | 7.13 | 1168.17 | 7.23 | 171.06 | 137.81 | 55.43 | 142.42 | 466.03 | 294.66 | 21.71 | 26.63 | 2.61 |
| Max | 7.38 | 1478.70 | 22.80 | 252.30 | 206.10 | 94.80 | 198.50 | 672.40 | 353.90 | 37.33 | 46.40 | 4.30 | |
| Min | 6.89 | 885.10 | 3.60 | 112.90 | 87.92 | 29.57 | 85.10 | 307.40 | 200.90 | 11.20 | 4.00 | 1.30 | |
| NAP n = 19 | Mean | 7.26 | 2516.99 | 9.55 | 465.46 | 278.35 | 77.89 | 377.03 | 1112.75 | 341.82 | 31.16 | 21.11 | 1.89 |
| Max | 7.60 | 5621.80 | 34.25 | 1153.00 | 609.90 | 182.60 | 1028.00 | 3038.00 | 695.60 | 67.40 | 64.80 | 3.52 | |
| Min | 6.88 | 565.85 | 2.20 | 78.10 | 64.10 | 11.63 | 56.49 | 193.30 | 200.90 | 7.50 | 1.20 | 0.96 | |
| LP n = 11 | Mean | 7.17 | 3969.65 | 19.46 | 836.79 | 323.28 | 99.05 | 557.15 | 1640.82 | 472.34 | 116.41 | 12.82 | 2.30 |
| Max | 8.14 | 8553.31 | 40.76 | 2866.00 | 624.10 | 169.80 | 1083.00 | 3729.00 | 764.70 | 240.70 | 68.80 | 3.44 | |
| Min | 6.82 | 1522.15 | 5.31 | 246.90 | 56.35 | 44.33 | 222.70 | 592.80 | 256.30 | 3.70 | 3.40 | 0.80 | |
| BP n = 6 | Mean | 7.60 | 517.73 | 3.22 | 64.75 | 58.08 | 24.90 | 41.33 | 188.00 | 184.60 | — | — | — |
| Max | 7.93 | 660.20 | 7.81 | 82.30 | 82.10 | 43.70 | 66.40 | 286.20 | 229.20 | — | — | — | |
| Min | 7.23 | 305.40 | 1.48 | 36.02 | 32.10 | 5.28 | 17.52 | 128.10 | 116.60 | — | — | ||
| U n = 6 | Mean | 8.38 | 2174.98 | 49.11 | 613.50 | 44.62 | 70.81 | 417.70 | 745.40 | 412.97 | 3.83 | — | — |
| Max | 8.60 | 2249.20 | 56.70 | 636.00 | 52.94 | 82.60 | 433.50 | 777.10 | 447.10 | 4.00 | — | — | |
| Min | 8.12 | 2051.50 | 44.58 | 572.00 | 32.10 | 65.66 | 389.20 | 691.60 | 383.50 | 3.00 | — | — | |
| J n = 6 | Mean | 7.68 | 1052.61 | 19.43 | 269.50 | 59.60 | 34.52 | 191.63 | 391.13 | 260.27 | 8.17 | — | — |
| Max | 8.17 | 1254.30 | 23.23 | 321.00 | 80.10 | 38.96 | 225.20 | 460.70 | 288.40 | 16.00 | — | — | |
| Min | 7.45 | 976.40 | 17.04 | 245.00 | 48.10 | 31.11 | 155.70 | 309.80 | 237.00 | 4.00 | — | — | |
| R n = 12 | Mean | 7.67 | 363.37 | 3.77 | 55.10 | 60.96 | 11.93 | 29.42 | 128.03 | 147.08 | 11.42 | — | — |
| Max | 8.11 | 683.80 | 7.14 | 154.00 | 100.60 | 24.94 | 63.20 | 236.60 | 213.90 | 16.00 | — | — | |
| Min | 7.03 | 37.90 | 1.13 | 2.10 | 6.77 | 0.99 | 2.60 | 11.10 | 25.90 | 5.00 | — | — | |
The surface water samples exhibited slight alkalinity, with pH ranging from 7.03 to 8.11. Specifically, the pH for R, J, and U were 7.67, 7.68, and 8.38, respectively. Similarly, the groundwater samples also displayed slight alkalinity, with pH ranging from 6.69 to 8.14. The samples from all landforms included both slightly acidic and slightly alkaline water, excluding the slightly alkaline samples collected in the HR.
The TDS of surface water was 37.90–2249.20 mg L−1, with an upward trend from the R (363.37 mg L−1) to the J (1052.61 mg L−1) and to the U (2174.98 mg L−1) (Fig. 5a). The prevalent ions in the R were HCO3−, SO42−, Na+, and Ca2+, with concentrations of 25.90–213.90 mg L−1, 11.10–236.60 mg L−1, 2.10–154.00 mg L−1, and 6.77–100.60 mg L−1, respectively. The prevalent ions in J were Na+, SO42−, Cl−, and HCO3−, with concentrations of 245.00–321.00 mg L−1, 309.80–460.70 mg L−1, 155.70–225.20 mg L−1, and 237.00–288.40 mg L−1, respectively. The U had the same prevalent cations as J, but with higher concentrations (572.00–636.00 mg L−1). The prevalent anions were SO42− and Cl−, with concentrations of 691.60–777.10 mg L−1 and 389.20–433.50 mg L−1, respectively. Overall, the main hydrochemical types of surface water progressed from HCO3·SO4–Ca·Na and SO4·HCO3–Na·Ca for the R to SO4·Cl HCO3–Na for the J and SO4·Cl–Na for the U.
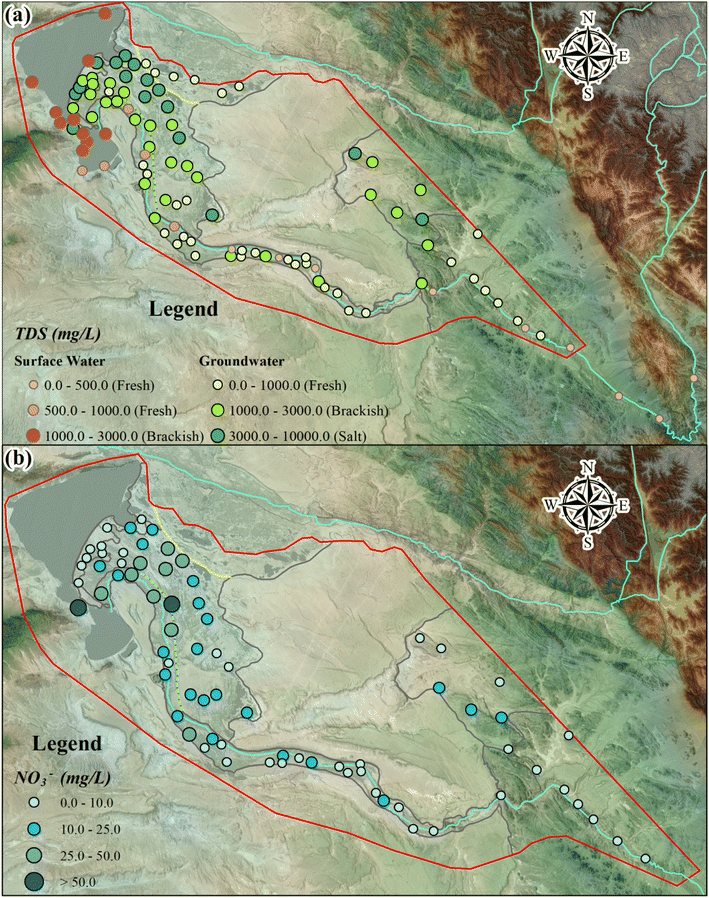 | ||
| Fig. 5 The spatial evolution of TDS and NO3− in the URB. (a) The TDS of surface water and groundwater and (b) the NO3− content of groundwater. | ||
Groundwater from the LM to the LP exhibited similar hydrochemical features to surface water. The TDS exhibited high variation, and the sequence was as follows: LM (701.09 mg L−1) < RV (829.62 mg L−1) < SAP (1168.17 mg L−1) < NAP (2516.99 mg L−1) < HR (2746.15 mg L−1) < LP (3969.65 mg L−1). The prevalent ions in groundwater within the LM were HCO3−, SO42−, Na+, and Ca2+, with concentrations of 181.40–390.50 mg L−1, 144.10–560.00 mg L−1, 69.40–232.70 mg L−1, and 33.32–125.20 mg L−1, respectively. The groundwater in the RV had the same prevalent ions as that in the LM, but with higher concentrations along the extension of the runoff path. For the SAP, in addition to the same prevalent cations as the LM and RV, Mg2+ was also prevalent, with a concentration of 29.57–94.80 mg L−1. The samples in the NAP were dominated by SO42−, Na+, and Ca2+, with concentrations of 193.30–3038.00 mg L−1, 78.10–1153.00 mg L−1, and 64.10–609.90 mg L−1, respectively. The samples in the HR and LP had the same prevalent cations as the NAP. However, the values were higher with concentrations of 430.90–639.70 mg L−1 for Na+ and 41.10–801.70 mg L−1 for Ca2+ in the HR. In the LP, the corresponding concentrations were 246.90–2866.00 mg L−1 and 56.35–624.10 mg L−1. Furthermore, the prevalent anions were SO42− and Cl−, with concentrations of 576.40–3073.90 mg L−1 and 321.10–799.40 mg L−1, respectively, in the HR. In the LP, the corresponding concentrations were 592.80–3729.00 mg L−1 and 222.70–1083.00 mg L−1. Overall, the hydrochemical types of groundwater transformed from HCO3·SO4–Na·Ca in the LM and SO4·HCO3–Ca·Na in the RV into SO4·HCO3–Na·Ca·Mg in the SAP, SO4–Na·Ca in the NAP, and SO4·Cl–Na·Ca in the LP and HR.
4.2 Distribution of groundwater salinity and nitrogen pollution
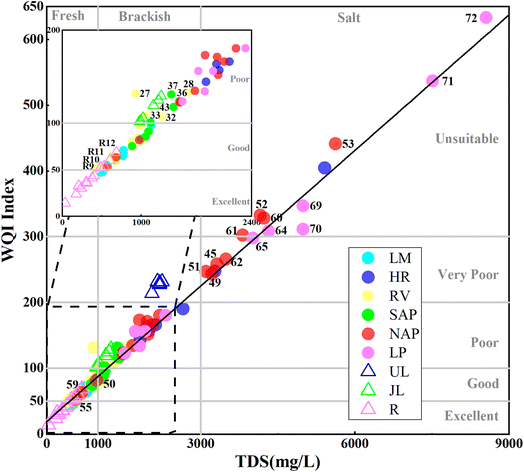
Considering the TDS value, the salinity of water can be categorised into six grades, as follows: freshwater (TDS < 1000 mg L−1), brackish (1000 mg L−1 < TDS < 5000 mg L−1), highly brackish (5000 mg L−1 < TDS < 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg L−1), saline (15
000 mg L−1), saline (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg L−1 < TDS < 30
000 mg L−1 < TDS < 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg L−1), sea water (30
000 mg L−1), sea water (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg L−1 < TDS < 40
000 mg L−1 < TDS < 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg L−1), and brine (TDS > 40
000 mg L−1), and brine (TDS > 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg L−1) (Water Quality Association [WQA, https://wqa.org/]). To streamline classification, we consulted international standards and implemented a four-category TDS-based framework: fresh water (TDS < 1000 mg L−1), brackish water (1000 < TDS < 3000 mg L−1), saline water (3000 < TDS < 10
000 mg L−1) (Water Quality Association [WQA, https://wqa.org/]). To streamline classification, we consulted international standards and implemented a four-category TDS-based framework: fresh water (TDS < 1000 mg L−1), brackish water (1000 < TDS < 3000 mg L−1), saline water (3000 < TDS < 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg L−1), and highly saline water (TDS > 10
000 mg L−1), and highly saline water (TDS > 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg L−1).9
000 mg L−1).9
Notably, groundwater salinity transitioned from freshwater in the LM and RV to brackish water in the SAP and NAP (52% of samples) and saline water in the NAP (42% of samples) and LP (Fig. 5a). The groundwater in the HR predominantly consisted of brackish water (71% of samples). Samples with high TDS were predominantly from the northern region of NPA and near the LP, close to U. Considering that these areas are situated within agricultural zones and near human habitation, groundwater salinisation may be attributed to anthropogenic activities such as domestic sewage discharge and agricultural runoff. In addition, salinity levels in the riparian zones (LM, RV, and SAP) were lower than those in other regions. Although the SAP contained brackish water, the maximum TDS recorded was only 1478.70 mg L−1, with a mean value of 1168.17 mg L−1. The reduced salinity in the riparian zones might be attributed to the dilution effect from R, where the TDS was < 1000 mg L−1. In the study area, ∼38% of samples were freshwater, 39% were brackish water, and 22% were saline water.
The concentration of NO3− in groundwater exhibited an initial increase followed by an overall decrease from the LM to the LP (Fig. 5b), with values ranging from 1.2 to 68.8 mg L−1 and a mean concentration of 14.6 mg L−1 (Table 1). Only two samples from the NAP and LP exceeded the WHO limit for NO3− (50 mg L−1), with concentrations of 64.80 mg L−1 (No. 47) and 68.80 mg L−1 (No. 65), respectively. However, considering the quality standard for groundwater limit (GB/T 14848-2017) for NO3− in China (25 mg L−1), the over-standard rate was 19.2%. Most of these samples were located in the NAP and SAP, while a few were dispersed in the RV and LP.
4.3 Groundwater provenance and conversion relation with river water
The slope of the local meteoric water line (LMWL: δ2H = 6.49δ18O −8.63 [R2 = 0.9869]) in the URB was lower than that of the global meteoric water line (GMWL: δ2H = 8δ18O + 10). Most of the groundwater samples were plotted near both the GMWL and LMWL. This result indicated that the groundwater was recharged via precipitation and snowmelt (Fig. 4). Furthermore, the groundwater and river water points were mixed in the δ2H–δ18O plot (Fig. 5). This finding indicated a hydraulic connection between the groundwater and river water.65 Considering that the previous study did not investigate the recharge relationship, this study integrated the cross-sectional flow of R (Q), hydrogeological background, and hydrochemical features to elucidate the interrelation between R and groundwater.R1–R5: notably, the Q underwent minimal change within R1–R5 (Fig. 6). However, the TDS values displayed a significant growth trend, and the hydrochemical type transitioned from HCO3·SO4–Ca to SO4·HCO3–Ca·Na. Owing to the significant variation in the terrain gradient and the presence of well-developed fissures in the mountainous area, combined with the river's predominantly vertical erosion that lowers the riverbed elevation in this section, groundwater discharges into the river, which makes this segment a ground-fed water reach. Furthermore, given the arid to semi-arid climate, intense evaporation–concentration processes served as a critical mechanism controlling the hydrochemical evolution of shallow groundwater in the region. Evaporation led to continuous water loss and relative enrichment of dissolved salts, which resulted in a significant increase in groundwater TDS.
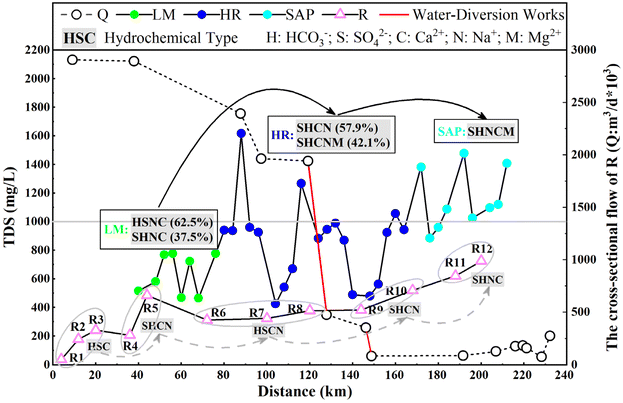 | ||
| Fig. 6 The Q and the hydrochemical features of river water and groundwater along the flow direction. | ||
R5–R6: the hydrochemical type within the R transitioned from SO4·HCO3–Ca·Na to HCO3·SO4–Ca·Na, and the Q and TDS decreased. However, the TDS of groundwater fluctuated within the LM, which exhibited complex hydrochemical types (HCO3·SO4–Na·Ca and SO4·HCO3–Na·Ca). This finding indicated that the R was supplied by groundwater.
R6–R9: the TDS of the R increased with the decline in Q, as the hydrochemical type transitioned from HCO3·SO4–Ca·Na to SO4·HCO3–Na·Ca. Furthermore, the groundwater TDS fluctuated, as it first increased and then decreased. The main hydrochemical types were SO4·HCO3–Ca·Na and SO4·HCO3–Ca·Na·Mg. Owing to the impact of human activities (water diversion works), the Q and TDS of groundwater declined sharply in the R6–R7 and R8–R9 sections. This finding indicated that the R was recharging the aquifer and possibly reflected lateral leakage from the river channel into the adjacent groundwater. However, the Q exhibited only a minor change within the R7–R8 reach, while the TDS of the R increased steadily. This finding indicated that groundwater discharged into the river and revealed intense water–rock interactions in this sector. The described hydrochemical evolution was co-driven by multiple environmental mechanisms. The evaporation–concentration process remains a key natural factor responsible for increased TDS. In addition, the dissolution of carbonate minerals (e.g., calcite and dolomite) and sulphate minerals (e.g., gypsum) within the aquifer might further contribute ions such as Ca2+, Mg2+, HCO3−, and SO42−. Cation exchange was also considered a significant process potentially responsible for the observed increase in the relative proportion of Na+. Furthermore, the operation of water diversion projects altered local hydraulic gradients and directly influenced the recharge–discharge relationship between surface water and groundwater.
R9–R12: the Q first increased and then fluctuated. The TDS growth trend for the R was more substantial compared with that for R6–R9, which exhibited hydrochemical types of SO4·HCO3–Ca·Na and SO4·HCO3–Na·Ca. Subsequently, the TDS of groundwater increased, and the main hydrochemical types were SO4·HCO3–Na·Ca·Mg. Owing to the slight shift in the terrain of the SAP, the lateral erosion activity of the R increased. This observation indicated that the groundwater recharged the R. The fluctuation of Q beyond R12 reflected the mutual transformation of the groundwater and the R near the lake inlet region. Similarly, considering the other processes inferred from the figures above, the observed variations in this region were most likely not driven by a single factor and might also be attributed to the additional mechanisms discussed earlier.
5 Discussion
The findings of this study on the hydrochemical evolution and salinisation mechanisms in the URB offer crucial insights into groundwater dynamics in arid regions. Within a global context, the results align with patterns observed in other inland basins and demonstrate distinct local characteristics. For example, the shift in hydrochemical facies from bicarbonate-dominated to sulphate–chloride types corresponded to the evaporation–concentration-driven processes identified in the Paradox Basin of the Colorado Plateau, where Kr-81 dating revealed that tectonic activity over the past million years reactivated groundwater flow systems.77 Similarly, the dominant role of evaporation and mineral dissolution (e.g., of gypsum and halite) in regulating TDS and ion enrichment aligned with previous findings in the region.78 Furthermore, the spatial heterogeneity of salinity factors observed in the study area was consistent with the conclusions from global machine learning models,79 which identified arid climatic conditions—such as a high aridity index and low recharge—as key predictors of fluoride and TDS enrichment in groundwater across Africa and Asia. This finding highlights the universal significance of climate-driven evaporation and water–rock interactions in arid-zone hydrochemistry. The Discussion section is structured into the following sub-sections.5.1 Main factors controlling groundwater salinization
In the groundwater chemical environment, influenced by both natural and anthropogenic factors, the PCA method can effectively elucidate the primary control mechanisms underlying physicochemical parameters and reveal latent patterns.36 In this study, 12 parameters (TDS, Na+, K+, Ca2+, Mg2+, Cl−, HCO3−, SO42−, CO2, pH, NO3−, and H) were included in the PCA to determine the main factors controlling the hydrogeochemical processes associated with groundwater salinisation. Notably, four PCs were extracted via the maximum variance method, with the extracted PCs accounting for a cumulative variance contribution of 80.53% (Table 4). Specifically, PC1, PC2, PC3, and PC4 accounted for 38.93%, 18.81%, 12.87%, and 9.92% of the cumulative variance, respectively.| Parameters | PC1 | PC2 | PC3 | PC4 |
|---|---|---|---|---|
| TDS | 0.95 | 0.25 | 0.07 | 0.05 |
| SO42− | 0.95 | 0.25 | 0.03 | 0.09 |
| Na+ | 0.95 | 0.06 | −0.01 | −0.01 |
| Cl− | 0.89 | 0.05 | 0.03 | 0.06 |
| Mg2+ | 0.78 | 0.09 | 0.25 | 0.08 |
| Ca2+ | 0.70 | 0.59 | 0.04 | 0.19 |
| pH | −0.02 | −0.83 | 0.11 | −0.12 |
| CO2 | 0.24 | 0.82 | 0.13 | −0.12 |
| H | 0.01 | 0.21 | −0.86 | 0.08 |
| HCO3− | 0.18 | 0.47 | 0.59 | −0.27 |
| K+ | 0.23 | 0.37 | 0.57 | 0.63 |
| NO3− | 0.11 | 0.01 | −0.10 | 0.92 |
| Eigenvalue | 4.67 | 2.26 | 1.54 | 1.19 |
| % of variance | 38.93 | 18.81 | 12.87 | 9.92 |
| Cumulative % | 38.93 | 57.74 | 70.61 | 80.53 |
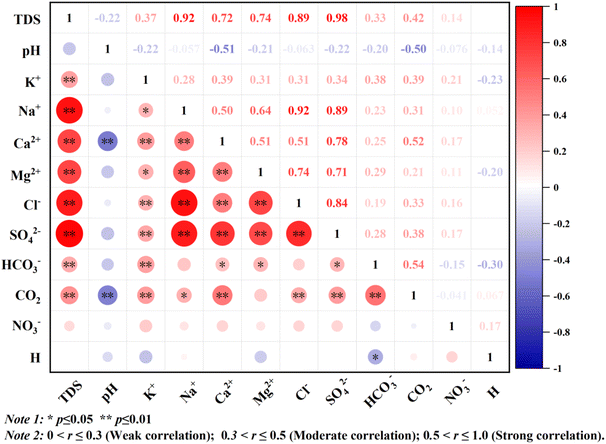 | ||
| Fig. 7 The heat diagram of correlation analysis of different components of groundwater.72 | ||
Notably, along the flow path, the main mechanisms influencing groundwater chemical composition were water–rock interactions and evaporation (Fig. 8).80
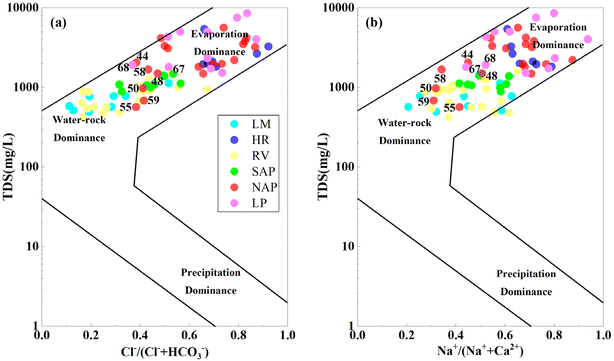 | ||
| Fig. 8 Gibbs diagram representing the major mechanisms controlling hydrogeochemical evolution of groundwater salinization. (a) TDS vs. Cl−/(Cl− + HCO3−) and (b) TDS vs. Na+/(Na++ Ca2+). | ||
Groundwater in the riparian zone (LM, RV, and SAP) was mainly influenced by water–rock interactions owing to the large buried depth and mixing of the R. The mechanisms in the NAP and LP transitioned from water–rock interactions to evaporation–crystallisation processes. This transition could be attributed to the decrease in groundwater depth and the oversaturation of relevant minerals (e.g., calcite and dolomite), which ultimately results in mineral deposition (Fig. 10c and d). Moreover, water–rock interactions dominated no. 44, 48, 50, 55, and 58–59 in the NAP and no. 67–68 samples in the LP with lower TDS values. This condition could be attributed to the mixing of ancient channels.65 Most of the samples in the HR with greater groundwater depth were located at the evaporation–crystallisation end member. This finding might be attributed to the rapid evaporation of spring water and refluxing into the aquifer.
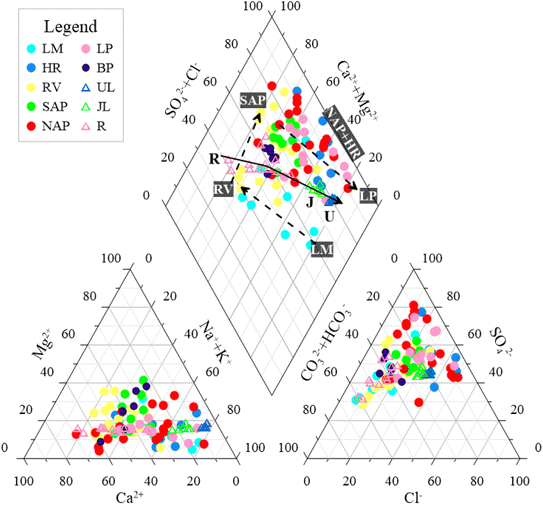
To further identify the main mineral sources of these ions, the relationships among the ions were compared.52 Notably, the main dissolved minerals in groundwater gradually transitioned from carbonates to evaporates (Fig. 9a and b).81 This condition indicated that these two types of minerals were mainly responsible for the ions in the groundwater within the study area. However, several samples in the NAP and LP were shifted towards the silicate end member. This finding indicated that some of the ions in these areas were derived from the dissolution of silicate minerals. The Cl−/Na+ ratio of almost all samples was below the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 line (halite dissolution line), with a mean value of 0.49 (Fig. 9c). This condition indicated that part of the Na+ in the groundwater did not originate solely from the dissolution of halite.52 However, Na+ exhibited strong correlations with Cl− (0.93) and SO42− (0.88) (Fig. 7). The substantial correlation between Na+ and Cl− mainly indicated that halite (NaCl) dissolution was a major source.82 The strong correlation with SO42− further indicated that the dissolution of sodium sulphate salts (e.g., thenardite) might also contribute to Na+ enrichment. Furthermore, Na+ exhibited a positive correlation with Ca2+ (0.50) and Mg2+ (0.62). All the samples between (Ca2+ + Mg2+ − SO42− − HCO3−) and (Na+ + K+ − Cl−) exhibited a linear relationship with a slope close to −1 (y = −0.9844x − 0.0637, R2 = 0.9333) (Fig. 9d). This condition indicated that the concentration of Na+ (K+) and other major cations (Ca2+ and Mg2+) in groundwater were also influenced by the cation exchange reaction.50 Meanwhile, most of the samples were located in the fourth quadrant (Fig. 9d). This finding indicated that the reverse cation exchange reaction (eqn. (5)) was the main factor controlling the concentration of cations in groundwater.50 These findings confirmed that the dominant cations in groundwater transformed from Ca2+ > Na+ into Na+ > Ca2+. Moreover, the individual samples in the RV (no. 16–19 and 26) were located in the second quadrant with the hydrochemical types of SO4·HCO3–Ca · Mg·Na. This result indicated that a different cation exchange type (eqn (4)) also contributed to groundwater composition, where Na+ (K+) was replaced by Ca2+ (Mg2+) on the rock surface. The samples in the LM (excluding no. 3 and 5–6), RV (no. 17–19, 23–26, and 29), and NAP (no. 55 and 59) were plotted near the 1
1 line (halite dissolution line), with a mean value of 0.49 (Fig. 9c). This condition indicated that part of the Na+ in the groundwater did not originate solely from the dissolution of halite.52 However, Na+ exhibited strong correlations with Cl− (0.93) and SO42− (0.88) (Fig. 7). The substantial correlation between Na+ and Cl− mainly indicated that halite (NaCl) dissolution was a major source.82 The strong correlation with SO42− further indicated that the dissolution of sodium sulphate salts (e.g., thenardite) might also contribute to Na+ enrichment. Furthermore, Na+ exhibited a positive correlation with Ca2+ (0.50) and Mg2+ (0.62). All the samples between (Ca2+ + Mg2+ − SO42− − HCO3−) and (Na+ + K+ − Cl−) exhibited a linear relationship with a slope close to −1 (y = −0.9844x − 0.0637, R2 = 0.9333) (Fig. 9d). This condition indicated that the concentration of Na+ (K+) and other major cations (Ca2+ and Mg2+) in groundwater were also influenced by the cation exchange reaction.50 Meanwhile, most of the samples were located in the fourth quadrant (Fig. 9d). This finding indicated that the reverse cation exchange reaction (eqn. (5)) was the main factor controlling the concentration of cations in groundwater.50 These findings confirmed that the dominant cations in groundwater transformed from Ca2+ > Na+ into Na+ > Ca2+. Moreover, the individual samples in the RV (no. 16–19 and 26) were located in the second quadrant with the hydrochemical types of SO4·HCO3–Ca · Mg·Na. This result indicated that a different cation exchange type (eqn (4)) also contributed to groundwater composition, where Na+ (K+) was replaced by Ca2+ (Mg2+) on the rock surface. The samples in the LM (excluding no. 3 and 5–6), RV (no. 17–19, 23–26, and 29), and NAP (no. 55 and 59) were plotted near the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 line (calcite or dolomite dissolution line), and the remaining samples (∼82%) were located above the 1
1 line (calcite or dolomite dissolution line), and the remaining samples (∼82%) were located above the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 line with a Ca2+ + Mg2+vs. HCO3− ratio of 1.38–13.59 (Fig. 9e). Notably, the dissolution of these minerals contributed minimally to Ca2+, Mg2+, and HCO3− in the groundwater.54 This outcome was confirmed by the weak correlation of HCO3− with Ca2+ (0.26) and Mg2+ (0.27) and a saturation index of > 0 (Fig. 10c and d). However, the excess Ca2+ and Mg2+ compared with HCO3− might be attributed to other sources such as magnesium sulphate dissolution (SO42− strong correlation with Mg2+ [0.71]), calcium–magnesium silicates, or gypsum, in addition to the cation exchange reaction. According to the scatter plot of SO42−vs. Ca2+ (Fig. 9f), ∼34% of the samples (most of which were from the RV) were plotted along the 1
1 line with a Ca2+ + Mg2+vs. HCO3− ratio of 1.38–13.59 (Fig. 9e). Notably, the dissolution of these minerals contributed minimally to Ca2+, Mg2+, and HCO3− in the groundwater.54 This outcome was confirmed by the weak correlation of HCO3− with Ca2+ (0.26) and Mg2+ (0.27) and a saturation index of > 0 (Fig. 10c and d). However, the excess Ca2+ and Mg2+ compared with HCO3− might be attributed to other sources such as magnesium sulphate dissolution (SO42− strong correlation with Mg2+ [0.71]), calcium–magnesium silicates, or gypsum, in addition to the cation exchange reaction. According to the scatter plot of SO42−vs. Ca2+ (Fig. 9f), ∼34% of the samples (most of which were from the RV) were plotted along the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 line (gypsum dissolution line), with ratio values of 0.8–1.2. Ca2+ exhibited a strong correlation with SO42− (0.78). This finding indicated that the dissolution of gypsum in the groundwater was an important source of Ca2+.83 Notably, drilling within the RV significantly exposed the gypsum layer, which mainly supplied SO42− and Ca2+ in the groundwater.35 In addition, most of the samples (∼60% samples) in all landforms were above the 1
1 line (gypsum dissolution line), with ratio values of 0.8–1.2. Ca2+ exhibited a strong correlation with SO42− (0.78). This finding indicated that the dissolution of gypsum in the groundwater was an important source of Ca2+.83 Notably, drilling within the RV significantly exposed the gypsum layer, which mainly supplied SO42− and Ca2+ in the groundwater.35 In addition, most of the samples (∼60% samples) in all landforms were above the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 line, with a range of 1.28–9.92. This result revealed that in addition to gypsum dissolution and the reverse cation exchange reaction, other mineral sources such as polyhalite (SO42− moderate correlation with K+ [0.34]) and anhydrite might be present. Furthermore, some samples in the RV were below the 1
1 line, with a range of 1.28–9.92. This result revealed that in addition to gypsum dissolution and the reverse cation exchange reaction, other mineral sources such as polyhalite (SO42− moderate correlation with K+ [0.34]) and anhydrite might be present. Furthermore, some samples in the RV were below the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 line with the hydrochemical types of HCO3·SO4–Ca·Na, which might be attributed to the mixing of the R in R6–R9. The reaction pathway can be described using the following equations:
1 line with the hydrochemical types of HCO3·SO4–Ca·Na, which might be attributed to the mixing of the R in R6–R9. The reaction pathway can be described using the following equations:
| Ca(Mg) − Y2 + 2Na+ ↔ Ca2+(Mg2+) + 2Na − Y | (4) |
| 2Na − Y + Ca2+(Mg2+) ↔ Ca(Mg) − Y2 + Na+ | (5) |
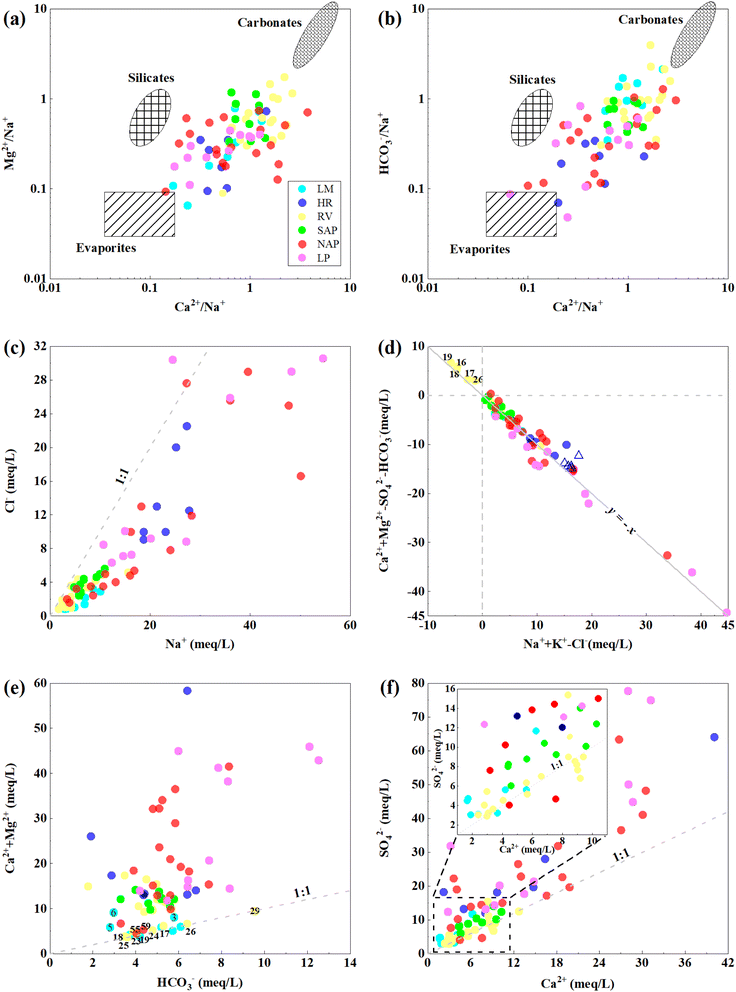
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pco2) in groundwater varied from −2.51 to −1.01 with a mean of −1.71 (Fig. 10a), which was significantly higher than that in the atmosphere (−3.5).84 As indicated in Fig. 10a, the log
Pco2) in groundwater varied from −2.51 to −1.01 with a mean of −1.71 (Fig. 10a), which was significantly higher than that in the atmosphere (−3.5).84 As indicated in Fig. 10a, the log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pco2 value increased from the LM to the LP, and the pH value gradually transformed from slightly acidic to slightly alkaline, which indicates that the carbon dioxide in groundwater mainly originated from the respiration of plant roots and the degradation of organic matter in the soil.85 In addition, the samples with a log
Pco2 value increased from the LM to the LP, and the pH value gradually transformed from slightly acidic to slightly alkaline, which indicates that the carbon dioxide in groundwater mainly originated from the respiration of plant roots and the degradation of organic matter in the soil.85 In addition, the samples with a log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pco2 close to or greater than that in soil (−1.5) are mainly located in the SAP (all samples), NAP (excluding no. 46, 49–50, 54 and 59) and LP (excluding no. 73 and 65). The groundwater samples are all from a phreatic aquifer that belongs to an open system and could freely exchange matter or energy with the atmosphere, indicating that it might be caused by the excessive application of farm manure for agricultural production, as the rural district was widely distributed in the study area. The samples in the LM (no. 2–3 and 8), HR (no. 9 and 13) and RV (no. 19, 23–24, 27 and 29) has high log
Pco2 close to or greater than that in soil (−1.5) are mainly located in the SAP (all samples), NAP (excluding no. 46, 49–50, 54 and 59) and LP (excluding no. 73 and 65). The groundwater samples are all from a phreatic aquifer that belongs to an open system and could freely exchange matter or energy with the atmosphere, indicating that it might be caused by the excessive application of farm manure for agricultural production, as the rural district was widely distributed in the study area. The samples in the LM (no. 2–3 and 8), HR (no. 9 and 13) and RV (no. 19, 23–24, 27 and 29) has high log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pco2 values near the human settlements, which might be related to the discharge of organic waste. The HCO3− concentration of samples increased with the increases in log
Pco2 values near the human settlements, which might be related to the discharge of organic waste. The HCO3− concentration of samples increased with the increases in log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pco2 (Fig. 10b). Considering the positive correlation between CO2 and HCO3− (0.54) and the negative linear relationship between logPco2 and pH, the trend was found to be a result of chemical reaction eqn (6) and (7) moving to the right due to the increase in the concentration of CO2 in groundwater. On the other hand, the high concentration of CO2 could promote the solubility of groundwater in carbonate minerals.54 As shown in Fig. 10c and d, the SIC and SID from the LM to LP first indicated an increasing trend and then a stable trend with the increase in logPco2. This shows that the groundwater chemistry in the study area was significantly affected by CO2, especially in the SAP, NAP and LP. This was evident from the aforesaid regulation change among log
Pco2 (Fig. 10b). Considering the positive correlation between CO2 and HCO3− (0.54) and the negative linear relationship between logPco2 and pH, the trend was found to be a result of chemical reaction eqn (6) and (7) moving to the right due to the increase in the concentration of CO2 in groundwater. On the other hand, the high concentration of CO2 could promote the solubility of groundwater in carbonate minerals.54 As shown in Fig. 10c and d, the SIC and SID from the LM to LP first indicated an increasing trend and then a stable trend with the increase in logPco2. This shows that the groundwater chemistry in the study area was significantly affected by CO2, especially in the SAP, NAP and LP. This was evident from the aforesaid regulation change among log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Pco2, pH and HCO3−, and the concentration of Ca+ and Mg2+ gradually increased along the flow path.
Pco2, pH and HCO3−, and the concentration of Ca+ and Mg2+ gradually increased along the flow path.| CO2 + H20 ↔ H2CO3 | (6) |
| H2CO3 ↔ H+ + HCO3− | (7) |
In the first trend, the samples transitioned from fresh to brackish water from the LM to the SAP, with some samples also occurring in the NAP and LP. D varied significantly in the LM, with values of 0.6–12.1 m. However, groundwater salinity exhibited less variation, which indicated that this pattern might be associated with the development of stratum fissures in the LM, along with significant changes in the hydraulic gradient and lateral recharge. The average of D decreased from 2.5 m in the RV to 1.6 m in the SAP. Water diversion activities reduced the volume of river water along the flow path. This effect altered the interaction between river water and groundwater—from river water recharging groundwater in the RV to a state of mutual exchange in the SAP (subsection 4.3). Furthermore, salinity increased from fresh water (mean TDS value of 852.25 mg L−1) to brackish water (mean TDS value of 1174.83 mg L−1). The samples in the NAP (no. 54, 55, and 59) were classified as freshwater, possibly owing to mixing with the ancient channel. Other brackish water samples in the NAP (no. 45–47 and 56–58) and LP (no. 63 and 66) might have been slightly influenced by agricultural activities, with a mean TDS value of 2169.69 mg L−1.
The samples for the second trend (no. 45, 51–53, and 60–62 in the NAP; 64, 65, 69, and 70–72 in the LP; and 9 in the HR) were classified as saline water, with H between 1.0 and 2.0 m. Most of the samples in the NAP and LP (except no. 65) were located within agricultural land. Owing to widespread flood irrigation methods, irrigation water was more likely to infiltrate into the aquifer, ultimately dissolving soluble salts along the flow paths and further increasing groundwater salinity. Considering the high nitrate concentration (68.80 mg L−1) of the no. 65 samples (Section 4.2), the corresponding high TDS value might be attributed to sanitary sewage. In the HR, sample no. 9 also exhibited saline water characteristics, and mining activities were mainly concentrated around samples no. 9, 14, and 15. Therefore, intensive mining operations were possibly the primary factor contributing to the salinisation of sample no. 9, which had a TDS value of 5413.40 mg L−1.
The samples for the third trend were all located in the HR, where salinity transitioned from brackish to saline. Spring water in the HR was mainly represented by samples no. 12 and 10. Groundwater chemistry in this area was controlled by evaporation–crystallisation processes (Fig. 8). This finding indicated that groundwater salinisation in the HR was due to the mixing of groundwater, combined with the effects of evaporation and mining activities. Consequently, TDS values increased from 1887.85 to 3273.71 mg L−1.
Domestic sewage easily seeped into the aquifer and contaminated the groundwater owing to the widespread distribution of rural settlements in the riparian zones and NAP, coupled with poor sewage discharge management and inadequate collection and treatment facilities. However, the NO3− concentration of sample no. 65 in the LP was 68.80 mg L−1, which might be attributed to the large number of restaurants catering to the tourism industry. The NO3−/Cl− ratio exhibited only slight variation with the increase in Cl− concentration in the remaining samples (HR, LP, and NAP). The NO3−/Cl− ratio was <0.05 in the LP (no. 64, with a NO3− concentration of 19.62 mg L−1) and NAP (no. 45, 49, 53, and 60–62, with a mean value of 24.05 mg L−1). These high nitrate concentrations could be attributed to the application of nitrogen fertiliser for agricultural production. Irrigation water transported NO3− and soluble salts (such as halite and polyhalite) from the soil back into the aquifer, a pattern consistent with the second trend associated with increasing groundwater salinity (Section 5.1.3). The samples from the HR (no. 11 and 14–15) might have been influenced by mining activities, as the NO3− concentration gradually decreased from 17.91 mg L−1 in sample no. 15 to 12.82 mg L−1 in sample no. 11. Samples from the NAP (no. 47 and 51–52) were positioned between the two end members, with NO3−/Cl− ratios > 0.05. This result indicated that the NO3− concentrations were jointly influenced by the infiltration of domestic sewage and agricultural fertilisation.
5.2 Assessment availability for drinking and irrigation water
In addition, 30% of the samples were classified as poor quality and must be treated safely before drinking. This set included all the J samples, some samples located away from cultivated land in the NAP and LP, and individual samples less affected by mining activities in the HR. Meanwhile, another 30% of the samples were classified as unsuitable for drinking. This category included all the U samples and some samples from the NAP (no. 45, 49–53, and 60–62) and LP (no. 64–65 and 70–72) that were contaminated by agricultural runoff and domestic sewage. These samples were categorised as very poor and unsuitable for drinking water and might pose health risks if consumed without treatment. Meanwhile, the TDS and WQI values of all water samples exhibited a significant positive linear relationship (y = 0.0687x + 19.12, R2 = 0.9811), as follows: the higher the TDS value in the study area, the poorer the quality of drinking water. This finding indicated that the primary factor controlling groundwater salinisation was also the key driver of drinking water quality deterioration. Furthermore, TDS exhibited the strongest correlation with the anion SO42− (0.98) and the cation Na+ (0.92) (Fig. 7). This finding further indicated that hydrogeochemical processes such as cation exchange, evaporite dissolution, and irrigation return flow could influence the sources of Na+ and SO42− and affect the drinking water quality in the URB.
In summary, (1) the R and groundwater in the riparian zone were suitable for drinking. However, attention should be given to the increasing impact of human activities in these areas. (2) The J and groundwater in the NAP, LP, and HR were moderately affected by human activities (domestic sewage and agricultural runoff) and require appropriate treatment before drinking. (3) The U and groundwater in the NAP, LP, and HR were significantly affected by human activities and were not suitable for drinking. Policies should be implemented to prevent further deterioration and the expansion of poor water quality in these areas.
5.2.2.1 PI (permeability index). The PI is an indicator of the effect of irrigation water on soil permeability. Long-term irrigation gradually enriches the soil with Ca2+, Mg2+, and Na+, which lead to variations in the physicochemical properties and permeability of soil.86 Notably, both groundwater and surface water used for irrigation were classified as excellent and good (Fig. 14a). Furthermore, ∼16% of the samples (including all J and U, R1, and no. 1, 5, and 7 in the LM) were within the excellent category (PI > 75%) and were suitable for irrigation. Meanwhile, 84% of the remaining water samples were classified as good quality (PI between 25% and 75%). Notably, these samples might reduce soil permeability. Considering the lithological composition of sand gravel, pebble, coarse sand, and medium–coarse sand in the riparian zone and fine sand, silt, clay, and mudstone in the NAP and LP, irrigation in the NAP and LP might reduce soil permeability. The lithological composition of the HR was similar to that of the LM, which predominantly consisted of sand gravel.
 | ||
| Fig. 14 Scatter plots of (a) PI (%) vs. TDS, (b) SAR vs. TDS, (c) RSC vs. TDS and (d) PS vs. TDS for surface water and groundwater. | ||
5.2.2.2 SAR (sodium adsorption ratio). The SAR is a parameter for evaluating the effect of exchangeable sodium in irrigation water on soil properties and plant growth.87 Generally, a high sodium content in irrigation water can lead to the formation of alkaline soils and inhibit plant growth. The classification results based on the SAR indicated that 83% of the samples were of excellent quality and suitable for irrigation. This category included all the R and J samples, all water samples from the riparian zone, and some samples from the HR, NAP, and LP (Fig. 14b). Meanwhile, ∼9% of the samples were saline water within the excellent water quality category (no. 15 and 9 in the HR; 45, 49, and 51–52 in the NAP; and 64 and 69–70 in the LP) These samples might increase the risk of soil salinisation and thus are unfit for irrigation. Furthermore, ∼14% of the samples (all the U, no. 53–54 and 60–62 in the NAP, and 65, 71, and 73 in the LP) were classified as good quality. These samples were only suitable for soils with excellent permeability. However, soil permeability in the NAP and LP is generally poor. Therefore, these samples were unsuitable for irrigation. The no. 72 sample, categorised as doubtful quality in the LP, could damage all soil types if used for irrigation.
5.2.2.3 RSC (residual sodium carbonate). The RSC index is commonly used to assess the effect of carbonate and bicarbonate concentrations in irrigation water.88 Generally, excessive carbonate and bicarbonate levels in irrigation water can promote the dissolution of organic substances in the soil and leave black stains on the soil surface upon drying.86 The RSC values of all water samples were < 1.5, with values between −53.93 and 1.38. These values were categorised as good quality (Fig. 14c), which indicated that all samples were safe for irrigation.
5.2.2.4 PS (potential salinity). The suitability of irrigation water cannot be determined solely by the concentration of soluble salts in the soil.89 With prolonged irrigation, soil salinity tends to increase owing to the continuous accumulation of low-solubility salts within the soil profile.86 Therefore, the PS is an important indicator for assessing the suitability of irrigation water. Notably, ∼32% of the samples (including all the R samples and some samples from the LM, RV, and NAP) were classified as excellent and good quality, which indicated that they were safe for irrigation (Fig. 14d). Furthermore, ∼32% of the samples (including all the J samples, some samples from SAR, and individual samples from the NAP, RV, and LM) were classified as good to injurious quality and could contribute to increased soil salinity. Moreover, all the U samples and some samples from the HR and LP exhibited high PS values between 10.12 and 39.32. These samples were categorised as injurious to unsatisfactory quality, which indicated that they were unsuitable for irrigation.
Overall, the salinisation degree of surface water and groundwater, as well as the evaluation results for irrigation water suitability, indicated that the R and the groundwater in the riparian areas (LM, RV, and SAP) exhibited low TDS values ranging from 39.7 to 1616.20 mg L−1, with a mean value of 755.11 mg L−1. These water samples were suitable for irrigation. The J and U, as well as the groundwater in the NAP, LP, and HR, were unsuitable for irrigation owing to the influence of human activities (fertilisation, irrigation, and mining activities). These anthropogenic impacts might lead to regional soil salinisation and alkalisation, plant toxicity, and increased groundwater salinity. Moreover, these groundwater samples were classified as brackish or saline water (with mean TDS values > 2000 mg L−1), characterised by dominant anionic and cationic constituents of SO42− and Na+, respectively (with major hydrochemical types of SO4–Na·Ca or SO4·Cl–Na·Ca). This finding indicated that the dissolution of evaporite minerals, cation exchange reactions, and irrigation return flow might also influence the quality of irrigation water in these areas.
5.3 Recommendations to protect water resources
Considering the analysis presented in the previous chapters, the human activities contributing to the deterioration of surface water and groundwater quality in the URB can be summarised as follows:(1) Water diversion and storage projects: the water quality of the R was better than that of the groundwater in the riparian zones (LM, RV, and SAP). However, long-term water diversion activities reduced the flow of R and altered the interaction between groundwater and the R. This condition resulted in increased groundwater salinity along the flow paths. Furthermore, considering that the R eventually joined the J, the reduction in river runoff might decrease the discharge from J and U into the J. This condition degrades the water quality of J and its surrounding aquatic environment.
(2) Leakage of domestic sewage: groundwater nitrate pollution, particularly in the SAP and NAP, was exacerbated by the indiscriminate discharge of domestic sewage and inadequate sewage collection and treatment systems.
(3) Obsolete agricultural production technology: long-term flood irrigation exacerbated groundwater salinisation, particularly in the NAP and LP. The widespread application of nitrogen fertiliser in agricultural production intensified groundwater nitrate pollution.
The stability of the oasis–desert ecosystem in the study area is under serious threat owing to the deterioration of both surface water and groundwater quality. Soil salinisation directly inhibits plant survival by degrading the soil structure. Salts from groundwater rise to the topsoil through capillary action. This process inhibits seed germination and root development and leads to the degradation of native drought–tolerant vegetation. Furthermore, salt accumulation alters the chemical environment of groundwater. Thus, the salinity of the oasis–desert transition zone exceeds the salt tolerance thresholds of key plant species. This transition zone serves as a natural barrier against desert encroachment, with its vegetation relying on microscale yet good-quality groundwater for survival. As the transition zone shrinks owing to salinisation, the oasis becomes directly exposed to the forces of sand and wind erosion, which in turn accelerate desertification. This process can trigger an irreversible shift of the entire system from a stable ‘oasis-transition zone-desert’ state to a completely arid condition.
Under the combined pressures of over-extraction and pollution, the URB may have already exceeded the critical groundwater depth threshold. Consequently, vegetation dependent on shallow groundwater can no longer access sufficient water. This condition leads to widespread vegetation growth decline. The compounded effects of this hydrological imbalance, salinisation, and nitrate pollution further undermine the self-regulating capacity of the oasis ecosystem. Salinisation and nitrate pollution are not merely water quality issues. They are key drivers of cascading ecosystem degradation. Their interaction continuously weakens ecosystem resilience, which forces the oasis–desert ecosystem towards an irreversible trajectory of deterioration.
To prevent further deterioration of water quality in the URB, and based on the groundwater circulation patterns, hydrochemical evolution processes, and the spatial distribution characteristics of water quality for both drinking and irrigation, the following recommendations are proposed:
(1) Reducing the diversion volume of the R from water diversion projects and moderately increasing groundwater extraction in the riparian zone to maintain hydrological balance.
(2) Enhancing sewage infrastructure by expanding sewage pipe networks and wastewater treatment facilities, and improving the management and monitoring of sewage discharge throughout the URB.
(3) Replacing flood irrigation with drip irrigation, implementing land improvement policies, and standardising the application of agricultural fertilisers, especially in the NAP and LP.
(4) Mitigating the impact of mining activities and enhancing vegetation diversity and spatial structure to improve water conservation capacity and water quality in the HR.
(5) Establishing a database of background values for the geological and ecological environment in the URB.
However, this study has several limitations, as follows:
(1) The temporal variability of groundwater sampling was limited to a single season, and seasonal fluctuations in recharge and ion concentrations were neglected.
(2) The statistical power of the study may be constrained by the sample size (n = 103) for detecting subtle correlations. This limitation potentially affects the precision of the findings.
(3) In groundwater numerical simulations and analytical studies, parameter uncertainty (e.g., hydraulic conductivity) and the structure of the conceptual model can significantly impact the accuracy of the results.
Future studies should incorporate high-resolution temporal monitoring and extend sampling across both temporal (e.g., dry and normal years) and spatial scales. This approach would reduce epistemic uncertainties through the adoption of multi-model frameworks that integrate machine learning with process-based modelling.
6 Conclusions
This study employed a combination of multivariate statistical analysis, hydrogeochemistry, and water quality assessment for both drinking and irrigation purposes to elucidate the primary mechanisms controlling groundwater salinisation and provide recommendations for the protection of water resources in the URB.The TDS values of surface water exhibited an increasing trend along the flow path, with hydrochemical types evolving from HCO3·SO4–Na·Ca and SO4·HCO3–Ca·Na (R) to SO4·Cl HCO3–Na (J) and SO4·Cl–Na (U). Groundwater transformed from freshwater to saline water from the LM to the LP, with a TDS sequence of LM < RV < SAP < NAP < HR < LP. The dominant hydrochemical types evolved from HCO3·SO4–Na·Ca and SO4·HCO3–Ca·Na (Mg) in the LM, RV, and SAP to SO4–Na·Ca and SO4·Cl–Na·Ca in the NAP, LP, and HR. Nitrate concentrations in groundwater initially increased from the LM to the NAP and then decreased in the LP, within a range of 1.2–68.8 mg L−1.
The salinity and hydrochemical characteristics of groundwater exhibited significant spatial variability, which could be attributed to both natural factors and human activities. PCA revealed four main factors driving the hydrogeochemical evolution of groundwater salinisation, as follows: PC1, water–rock interactions and evaporation–crystallisation processes; PC2, organic matter degradation and the application of farm manure; PC3, lateral recharge, river water infiltration, and irrigation water flow; and PC4, excessive fertiliser application and domestic sewage infiltration.
The WQI results indicated that 47% of water samples were suitable for drinking, with excellent or good water quality. All samples satisfied the RSC criteria for irrigation. However, according to the PI, SAR, and PS indices, only 16%, 74%, and 32% of samples, respectively, were suitable for irrigation, corresponding to excellent quality for the PI and SAR and excellent to good quality for PS. Overall, the R and groundwater in the riparian zones (RV, NAP, and LP) were suitable for both drinking and irrigation.
The water quality of the R was superior to that of groundwater in the riparian zone. However, long-term water diversion activities reduced the flow of the R, altered the dynamic interaction between the groundwater and the river, and introduced two potential risks, as follows:
(1) The deterioration of water quality in both the R and groundwater in the riparian zone.
(2) The degradation of the aquatic environment in the J area owing to reduced flow in the R, which was attributed to the U.
Additionally, the leakage of domestic sewage and outdated agricultural practices exacerbated the depletion of groundwater resources in the URB. Reducing extraction from the R, moderately increasing groundwater exploitation in the riparian zone, promoting the adoption of drip irrigation systems, and improving domestic sewage disposal facilities are key strategies for protecting water resources in the URB. Furthermore, to ensure long-term water security, targeted investigations and remedial policies should be implemented in the next phase.
Conflicts of interest
There are no conflicts to declare.Data availability
The primary data supporting this study were obtained from three sources: (1) experimental records collected in laboratory settings; (2) restricted datasets provided by local government agencies under data-sharing agreements; (3) field data compiled and processed by the authors. These integrated datasets contain sensitive information that could compromise participant privacy and confidentiality. Therefore, in compliance with ethical regulations and data protection agreements, the raw data cannot be made publicly available. Researchers seeking specific non-identifiable subsets of data for verification purposes may contact the corresponding author to discuss feasibility. Any shared data will require execution of a data access agreement and approval from relevant ethics committees.Supplementary information is available. See DOI: https://doi.org/10.1039/d5em00587f.
Acknowledgements
This study was supported by the Geological Exploration Fund (S17-2-XJ07), Key R&D Projects in Shaanxi Province (2021SF-445 and 2021ZDLSF05-09) and the National Natural Science Foundation of China (41202176 and 42177076).References
- Y. Jiang, China’s water security: Current status, emerging challenges and future prospects, Environ. Sci. Pol., 2015, 54, 129–135 CrossRef.
- C. Li, X. Gao, Y. Liu and Y. Wang, Impact of anthropogenic activities on the enrichment of fluoride and salinity in groundwater in the Yuncheng Basin constrained by Cl/Br ratio, δ18O, δ2H, δ13C and δ7Li isotopes, J. Hydrol., 2019, 579, 124211 CrossRef CAS.
- D. Russo, A. Laufer, G. Bardhan and G. J. Levy, Salinity control in a clay soil beneath an orchard irrigated with treated waste water in the presence of a high water table: A numerical study, J. Hydrol., 2015, 531, 198–213 CrossRef.
- Z. Hui, B. Jya, W. Ping, C. Tw and E. Yld, Groundwater-fed oasis in arid Northwest China: insights into hydrological and hydrochemical processes, J. Hydrol., 2021, 603, 126927 Search PubMed.
- Y. Tian, Y. Jiang, Q. Liu, M. Dong, D. Xu and Y. Liu, et al., Using a water quality index to assess the water quality of the upper and middle streams of the Luanhe River, northern China, Sci. Total Environ., 2019, 667, 142–151 CrossRef CAS PubMed.
- X. Yin, Q. Feng, Y. Li, R. C. Deo, W. Liu and M. Zhu, et al., An interplay of soil salinization and groundwater degradation threatening coexistence of oasis-desert ecosystems, Sci. Total Environ., 2022, 806, 150599 CrossRef CAS PubMed.
- J. Li, Y. Wang and X. Xie, Cl/Br ratios and chlorine isotope evidences for groundwater salinization and its impact on groundwater arsenic, fluoride and iodine enrichment in the Datong basin, China, Sci. Total Environ., 2016, 544, 158–167 CrossRef CAS.
- F. Telahigue, F. Souid, B. Agoubi, A. Chahlaoui and A. Kharroubi, Hydrogeochemical and isotopic evidence of groundwater salinization in a coastal aquifer: A case study in Jerba Island, southeastern Tunisia, Phys. Chem. Earth, Parts A/B/C, 2020, 118–119, 102886 CrossRef.
- M. F. Abu-alnaeem, I. Yusoff, T. F. Ng, Y. Alias and M. Raksmey, Assessment of groundwater salinity and quality in Gaza coastal aquifer, Gaza Strip, Palestine: An integrated statistical, geostatistical and hydrogeochemical approaches study, Sci. Total Environ., 2018, 615, 972–989 CrossRef CAS PubMed.
- J. Yan, J. Chen and W. Zhang, Study on the groundwater quality and its influencing factor in Songyuan City, Northeast China, using integrated hydrogeochemical method, Sci. Total Environ., 2021, 773, 144958 CrossRef CAS.
- T. Cruz-Fuentes, MdC. Cabrera, J. Heredia and E. Custodio, Groundwater salinity and hydrochemical processes in the volcano-sedimentary aquifer of La Aldea, Gran Canaria, Canary Islands, Spain, Sci. Total Environ., 2014, 484, 154–166 CrossRef CAS.
- W. M. Edmunds, A. H. Bath and D. L. Miles, Hydrochemical evolution of the East Midlands Triassic sandstone aquifer, England, Geochim. Cosmochim. Acta, 1982, 46, 2069–2081 CrossRef CAS.
- H. Jia, H. Qian, L. Zheng, W. Feng, H. Wang and Y. Gao, Alterations to groundwater chemistry due to modern water transfer for irrigation over decades, Sci. Total Environ., 2020, 717, 137170 CrossRef CAS PubMed.
- J. Chen, H. Qian, Y. Gao, H. Wang and M. Zhang, Insights into hydrological and hydrochemical processes in response to water replenishment for lakes in arid regions, J. Hydrol., 2020, 581, 124386 CrossRef CAS.
- Y. Jia, H. Guo, B. Xi, Y. Jiang, Z. Zhang and R. Yuan, et al., Sources of groundwater salinity and potential impact on arsenic mobility in the western Hetao Basin, Inner Mongolia, Sci. Total Environ., 2017, 601–602, 691–702 CrossRef CAS PubMed.
- K. Jin, W. Rao, H. Tan, Y. Song, B. Yong and F. Zheng, et al., H-O isotopic and chemical characteristics of a precipitation-lake water-groundwater system in a desert area, J. Hydrol., 2018, 559, 848–860 CrossRef CAS.
- J. Li, Z. Wu, G. Tian, C. Ruan, G. Sagoe and X. Wang, Processes controlling the hydrochemical composition of geothermal fluids in the sandstone and dolostone reservoirs beneath the sedimentary basin in north China, Appl. Geochem., 2022, 138, 105211 CrossRef CAS.
- S. A. Lone, G. Jeelani, A. Mukherjee and P. Coomar, Arsenic fate in upper Indus river basin (UIRB) aquifers: Controls of hydrochemical processes, provenances and water-aquifer matrix interaction, Sci. Total Environ., 2021, 795, 148734 CrossRef CAS PubMed.
- I. Mukherjee and U. K. Singh, Hydrogeochemical characterizations and quality evaluation of groundwater in the major river basins of a geologically and anthropogenically driven semi-arid tract of India, Sci. Total Environ., 2022, 805, 150323 CrossRef CAS.
- N. Nasri, R. Ahmed and R. Bouhlila, Hydrogeochemical characteristics and sources of mirabilite in the high saline system of Sabkha Oum El Khialate, Southern Tunisia, Appl. Geochem., 2022, 143, 105294 CrossRef CAS.
- N. Rajmohan and L. Elango, Identification and evolution of hydrogeochemical processes in the groundwater environment in an area of the Palar and Cheyyar River Basins, Southern India, Environ. Geol., 2004, 46, 47–61 CAS.
- A. Vengosh and E. Rosenthal, Saline groundwater in Israel: Its bearing on the water crisis in the country, J. Hydrol., 1994, 156, 389–430 CrossRef CAS.
- Y. Zhai, J. Xiao and R. Zuo, et al., Groundwater nitrate pollution and human health risk assessment by using HHRA model in an agricultural area, NE China, Ecotoxicol. Environ. Saf., 2017, 142, 421–428 Search PubMed.
- M. J. Currell, P. Dahlhaus and H. Ii, Stable isotopes as indicators of water and salinity sources in a southeast Australian coastal wetland: identifying relict marine water, and implications for future change, Hydrogeol. J., 2015, 23, 235–248 CrossRef CAS.
- G. Nogueira, T. Y. Stigter, Y. Zhou, F. Mussa and D. Juizo, Understanding groundwater salinization mechanisms to secure freshwater resources in the water-scarce city of Maputo, Mozambique, Sci. Total Environ., 2019, 661, 723–736 CrossRef CAS PubMed.
- A. Tm, B. Am, C. Pskk, A. Os and A. Jm, Tracking nitrate and sulfate sources in groundwater of an urbanized valley using a multi-tracer approach combined with a Bayesian isotope mixing model, Water Res., 2020, 182, 115962 CrossRef.
- Y. Xiao, Q. Hao, Y. Zhang, Y. Zhu and X. Li, Investigating sources, driving forces and potential health risks of nitrate and fluoride in groundwater of a typical alluvial fan plain, Sci. Total Environ., 2021, 802, 149909 CrossRef PubMed.
- J. Li, Y. Wang, C. Zhu, X. Xue, K. Qian and X. Xie, et al., Hydrogeochemical processes controlling the mobilization and enrichment of fluoride in groundwater of the North China Plain, Sci. Total Environ., 2020, 730, 138877 CrossRef CAS PubMed.
- W. Qi, J. Wu, Y. Xia, X. Zhang, Z. Li and J. Chang, et al., Influence of ionic composition on minerals and source rocks: An investigation between carbonate-type and sulfate-type lacustrine sediments based on hydrochemical classification, Mar. Petrol. Geol., 2021, 130, 105099 CrossRef CAS.
- L. Xing, H. Guo and Y. Zhan, Groundwater hydrochemical characteristics and processes along flow paths in the North China Plain, Asian J. Earth Sci., 2013, 70–71, 250–264 CrossRef.
- C. Zhang, Q. Duan, P. J.-F. Yeh, Y. Pan, H. Gong and H. Moradkhani, et al., Sub-regional groundwater storage recovery in North China Plain after the South-to-North water diversion project, J. Hydrol., 2021, 597, 126156 CrossRef.
- T. Y. Stigter, S. P. J. van Ooijen, V. E. A. Post, C. A. J. Appelo and A. M. M. Carvalho Dill, A hydrogeological and hydrochemical explanation of the groundwater composition under irrigated land in a Mediterranean environment, Algarve, Portugal, J. Hydrol., 1998, 208, 262–279 CrossRef CAS.
- M. Yamanaka, S. H. Bottrell, J. Wu, Y. Kumagai, K. Mori and H. Satake, Chlorine stable isotope evidence for salinization processes of confined groundwater in southwestern Nobi Plain aquifer system, central Japan, J. Hydrol., 2014, 519, 295–306 CrossRef CAS.
- A. Zghibi, J. Tarhouni and L. Zouhri, Assessment of seawater intrusion and nitrate contamination on the groundwater quality in the Korba coastal plain of Cap-Bon (North-east of Tunisia), J. Afr. Earth Sci., 2013, 87, 1–12 CrossRef CAS.
- S. Huang, L. Zhao, G. Huang, J. Chen and Y. Wang, Dissolution characteristics of gypsum salt in the red beds of central Yunnan Province and their effects on groundwater quality: a case study of Xiejiahe valley in Chuxiong City, Carsol. Sin., 2022, 41, 610–622 CAS.
- G. Huang, J. Sun, Y. Zhang, Z. Chen and F. Liu, Impact of anthropogenic and natural processes on the evolution of groundwater chemistry in a rapidly urbanized coastal area, South China, Sci. Total Environ., 2013, 463–464, 209–221 CrossRef CAS PubMed.
- S. He, P. Li, F. Su, D. Wang and X. Ren, Identification and apportionment of shallow groundwater nitrate pollution in Weining Plain, northwest China, using hydrochemical indices, nitrate stable isotopes, and the new Bayesian stable isotope mixing model (MixSIAR), Environ. Pollut., 2022, 298, 118852 CrossRef CAS PubMed.
- X. Wang, W. Zheng, W. Tian, Y. Gao, X. Wang and Y. Tian, et al., Groundwater hydrogeochemical characterization and quality assessment based on integrated weight matter-element extension analysis in Ningxia, upper Yellow River, northwest China, Ecol. Indic., 2022, 135, 108525 CrossRef CAS.
- X. Yin, Q. Feng, Y. Li, R. C. Deo, W. Liu and M. Zhu, et al., An interplay of soil salinization and groundwater degradation threatening coexistence of oasis-desert ecosystems, Sci. Total Environ., 2022, 806, 150599 CrossRef CAS PubMed.
- H. Zhang, J. Yu, P. Wang, T. Wang and Y. Li, Groundwater-fed oasis in arid Northwest China: Insights into hydrological and hydrochemical processes, J. Hydrol., 2021, 597, 126154 CrossRef CAS.
- X. Chen, C. Jiang, L. Zheng, L. Zhang, X. Fu and S. Chen, et al., Evaluating the genesis and dominant processes of groundwater salinization by using hydrochemistry and multiple isotopes in a mining city, Environ. Pollut., 2021, 283, 117381 CrossRef CAS PubMed.
- M. Argamasilla, J. A. Barberá and B. Andreo, Factors controlling groundwater salinization and hydrogeochemical processes in coastal aquifers from southern Spain, Sci. Total Environ., 2017, 580, 50–68 CrossRef CAS.
- J. L. Martinez, M. Raiber and D. I. Cendón, Using 3D geological modelling and geochemical mixing models to characterise alluvial aquifer recharge sources in the upper Condamine River catchment, Queensland, Australia, Sci. Total Environ., 2017, 574, 1–18 CrossRef CAS PubMed.
- J. Yan, J. Chen and W. Zhang, Impact of land use and cover on shallow groundwater quality in Songyuan city, China: A multivariate statistical analysis, Environ. Pollut., 2022, 307, 119532 CrossRef CAS.
- R. Duan, P. Li, L. Wang, X. He and L. Zhang, Hydrochemical characteristics, hydrochemical processes and recharge sources of the geothermal systems in Lanzhou City, northwestern China, Urban Clim., 2022, 43, 101152 CrossRef.
- I. Matiatos, A. Alexopoulos and A. Godelitsas, Multivariate statistical analysis of the hydrogeochemical and isotopic composition of the groundwater resources in northeastern Peloponnesus (Greece), Sci. Total Environ., 2014, 476–477, 577–590 CrossRef CAS PubMed.
- M. Ujević Bošnjak, K. Capak, A. Jazbec, C. Casiot, L. Sipos and V. Poljak, et al., Hydrochemical characterization of arsenic contaminated alluvial aquifers in Eastern Croatia using multivariate statistical techniques and arsenic risk assessment, Sci. Total Environ., 2012, 420, 100–110 CrossRef PubMed.
- J. Wu, P. Li, H. Qian, Z. Duan and X. Zhang, Using correlation and multivariate statistical analysis to identify hydrogeochemical processes affecting the major ion chemistry of waters: a case study in Laoheba phosphorite mine in Sichuan, China, Arabian J. Geosci., 2014, 7, 3973–3982 CrossRef CAS.
- I. Farid, K. Zouari, A. Rigane and R. Beji, Origin of the groundwater salinity and geochemical processes in detrital and carbonate aquifers: Case of Chougafiya basin (Central Tunisia), J. Hydrol., 2015, 530, 508–532 CrossRef CAS.
- J. N. Jannat, M. S. I. Khan, H. M. T. Islam, M. S. Islam, R. Khan and M. A. B. Siddique, et al., Hydro-chemical assessment of fluoride and nitrate in groundwater from east and west coasts of Bangladesh and India, J. Clean. Prod., 2022, 372, 133675 CrossRef CAS.
- Y. Xiao, J. Shao, S. K. Frape, Y. Cui, X. Dang and S. Wang, et al., Groundwater origin, flow regime and geochemical evolution in arid endorheic watersheds: a case study from the Qaidam Basin, northwestern China, Hydrol. Earth Syst. Sci., 2018, 22, 4381–4400 CrossRef CAS.
- J. Jean-Baptiste, C. Le Gal La Salle and P. Verdoux, Use of mixing models to explain groundwater quality time and space variation in a narrowed fluctuating alluvial aquifer, Appl. Geochem., 2020, 121, 104700 CrossRef CAS.
- F. Liu, J. Zou, J. Liu, J. Zhang and P. Zhen, Factors controlling groundwater chemical evolution with the impact of reduced exploitation, Catena, 2022, 214, 106261 CrossRef CAS.
- P. Debels, R. Figueroa, R. Urrutia, R. Barra and X. Niell, Evaluation of Water Quality in the Chillán River (Central Chile) Using Physicochemical Parameters and a Modified Water Quality Index, Environ. Monit. Assess., 2005, 110, 301–322 CrossRef CAS.
- R. K. Horton and J. Uddin, An index-number system for rating water quality, J. Water Pollut. Control Fed., 1965, 37, 300–306 Search PubMed.
- Ş. Şener, E. Şener and A. Davraz, Evaluation of water quality using water quality index (WQI) method and GIS in Aksu River (SW-Turkey), Sci. Total Environ., 2017, 584–585, 131–144 CrossRef PubMed.
- Y. Tian, Y. Jiang, Q. Liu, M. Dong, D. Xu and Y. Liu, et al., Using a water quality index to assess the water quality of the upper and middle streams of the Luanhe River, northern China, Sci. Total Environ., 2019, 667, 142–151 CrossRef CAS PubMed.
- K. Khmila, R. Trabelsi, K. Zouari and U. S. Kumar, Application of geochemical and isotopic tracers for the evaluation of groundwater quality in the irrigated area of the Sbiba plain (Central West Tunisia), Agric. Ecosyst. Environ., 2021, 313, 107298 CrossRef CAS.
- S. Xue, F. Gao, B. He and Z. Yan, Hydrochemistry and evaluation of groundwater suitability for irrigation and drinking purposes in the Markandeya River basin, Belgaum District, Karnataka State, India, Environ. Monit. Assess., 2011, 173, 459–487 CrossRef.
- C. Güler, M. A. Kurt, M. Alpaslan and C. Akbulut, Assessment of the impact of anthropogenic activities on the groundwater hydrology and chemistry in Tarsus coastal plain (Mersin, SE Turkey) using fuzzy clustering, multivariate statistics and GIS techniques, J. Hydrol., 2012, 414–415, 435–451 CrossRef.
- R. Murgai and A. D. Byerlee, Productivity Growth and Sustainability in Post-Green Revolution Agriculture: The Case of the Indian and Pakistan Punjabs, World Bank Res. Obs., 2001, 16, 199–218 CrossRef.
- W. Zhang, Analysis of land use dynamic change in oasis area of Wulungu river basin based on GIS, Prod. Eng. Res. Dev., 2019, 4, 14–18 Search PubMed.
- S. Xue, F. Gao, B. He and Z. Yan, Land pattern evolution and driving force analysis in Ulungur River Basin from 1989 to 2017, Ecol. Sci., 2021, 40, 33–41 Search PubMed.
- F. Gao, B. He, S. Xue and Y. Li, Impact of landscape pattern change on runoff processes in catchment area of the Ulungur River Basin, Water Sci. Technol.: Water Supply, 2020, 20(8), 3359–3370 Search PubMed.
- S. Pan, W. Zhang, G. Zhou and W. Zhang, Effect of Tilted Uplift and Strike-Slip Shear-Structure of Altai Mountains on Erqisi and WulunguRiver-Basin Environment, Bull. Geol. Sci. Technol., 2007, 02, 19–24 Search PubMed.
- Z. Wang, T. Cheng, K. Liu and H. Jiang, Water resources and its features in ulungur river watershed, Arid Land Geogr., 2000, 23(2), 141–147 Search PubMed.
- H. Tian, T. Xin, J. Li, J. Yang and Z. Xie, Characteristics and indication of hydrochemistry and environmental isotopes of different water sources in the Ulungur River basin, Arid Zone Res., 2021, 38, 1497–1505 Search PubMed.
- Z. Chen, W. Wei, J. Liu, Y. Wang and J. Chen, Identifying the recharge sources and age of groundwater in the Songnen Plain (Northeast China) using environmental isotopes, Hydrogeol. J., 2011, 19, 163–176 CrossRef CAS.
- G. Huang, Z. Chen and J. Sun, et al., Groundwater quality in aquifers affected by the anthropogenic and natural processes in an urbanized area, south China, Environ. Forensics, 2015, 17, 41–53 Search PubMed.
- H. Wang, X.-W. Jiang, L. Wan, G. Han and H. Guo, Hydrogeochemical characterization of groundwater flow systems in the discharge area of a river basin, J. Hydrol., 2015, 527, 433–441 CrossRef CAS.
- C. Güler, G. D. Thyne, J. E. Mccray and K. A. Turner, Evaluation of graphical and multivariate statistical methods for classification of water chemistry data, Hydrogeol. J., 2002, 10, 455–474 CrossRef.
- D. J. Rumsey, Statistics for Dummies, Wiley, Hoboken, 2nd edn, 2016 Search PubMed.
- J. Wu, P. Li, D. Wang, X. Ren and M. Wei, Statistical and multivariate statistical techniques to trace the sources and affecting factors of groundwater pollution in a rapidly growing city on the Chinese Loess Plateau, Earth Sci. Environ., 2020, 79, 1–16 CrossRef.
- L. Dai, L. Wang, L. Li, T. Liang, Y. Zhang and C. Ma, et al., Multivariate geostatistical analysis and source identification of heavy metals in the sediment of Poyang Lake in China, Sci. Total Environ., 2018, 621, 1433–1444 CrossRef CAS.
- P. Debels, R. Figueroa, R. Urrutia, R. Barra and X. Niell, Evaluation of water quality in the Chillán River (Central Chile) using physicochemical parameters and a modified water quality index, Environ. Monit. Assess., 2005, 110, 301–322 CrossRef CAS PubMed.
- WHO World Health Organization, Guidelines for Drinking-Water Quality, 4th edn, 2017, p. 564 Search PubMed.
- Ji-H. Kim, Krypton-81 Dating Constrains Timing of Deep Groundwater Flow Activation, Geophys. Res. Lett., 2022, 49, 11 Search PubMed.
- L. Tong, Y. Liu, S. Zhang, X. Liu, Q. Jia, Y. Huang, G. Ni and W. Lu, Temporal variation simulation of salinity and fluoride concentration in Ulungur Lake, Xinjiang, J. Lake Sci., 2022, 34(1), 134–141 CAS.
- J. Podgorski and M. Berg, A global machine learning model to identify potent predictors of fluoride and salinity in groundwater, Environ. Sci. Technol., 2023, 57, 1234–1245 Search PubMed.
- J. H. Feth and R. J. Gibbs, Mechanisms Controlling World Water Chemistry: Evaporation-Crystallization Process, Science, 1971, 172, 870–872 CrossRef CAS.
- J. Gaillardet, B. Dupré, P. Louvat and C. J. Allègre, Global silicate weathering and CO2 consumption rates deduced from the chemistry of large rivers, Chem. Geol., 1999, 159, 3–30 CrossRef CAS.
- Y. Jia, H. Guo, B. Xi, Y. Jiang, Z. Zhang and R. Yuan, et al., Sources of groundwater salinity and potential impact on arsenic mobility in the western Hetao Basin, Inner Mongolia, Sci. Total Environ., 2017, 601–602, 691–702 CrossRef CAS PubMed.
- G. Nogueira, T. Y. Stigter, Y. Zhou, F. Mussa and D. Juizo, Understanding groundwater salinization mechanisms to secure freshwater resources in the water-scarce city of Maputo, Mozambique, Sci. Total Environ., 2019, 661, 723–736 CrossRef CAS PubMed.
- C. Appelo and D. Postma, Geochemistry, Groundwater and Pollution, 2nd edn, 2005, p. 649 Search PubMed.
- W. Baijjali, I. D. Clark and P. Fritz, The artesian thermal groundwaters of northern Jordan: insights into their recharge history and age, J. Hydrol., 1997, 192, 355–382 CrossRef.
- P. Ravikumar, R. K. Somashekar and M. Angami, Hydrochemistry and evaluation of groundwater suitability for irrigation and drinking purposes in the Markandeya River basin, Belgaum District, Karnataka State, India, Environ. Monit. Assess., 2011, 173, 459–487 CrossRef CAS PubMed.
- K. Khmila, R. Trabelsi, K. Zouari and U. S. Kumar, Application of geochemical and isotopic tracers for the evaluation of groundwater quality in the irrigated area of the Sbiba plain (Central West Tunisia), Agric. Ecosyst. Environ., 2021, 313, 107298 CrossRef CAS.
- F. M. Eaton, Significance Of Carbonates In Irrigation Waters, Soil Sci., 1950, 69, 123–134 CrossRef CAS.
- L. D. Doneen, The influence of crop and soil on percolating water, InProceedings of the 1961 biennial conference on groundwater recharge, 1962, pp. 156–163 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |

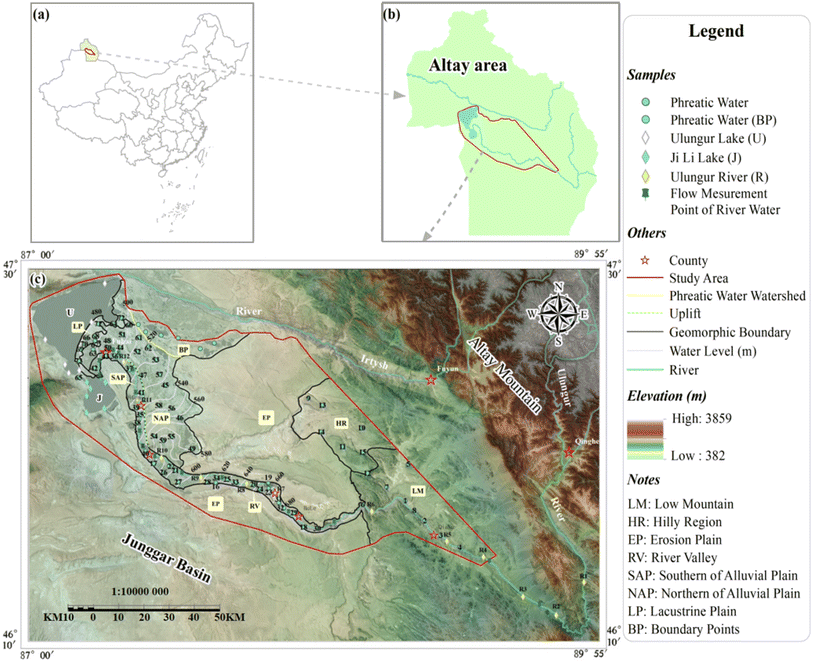
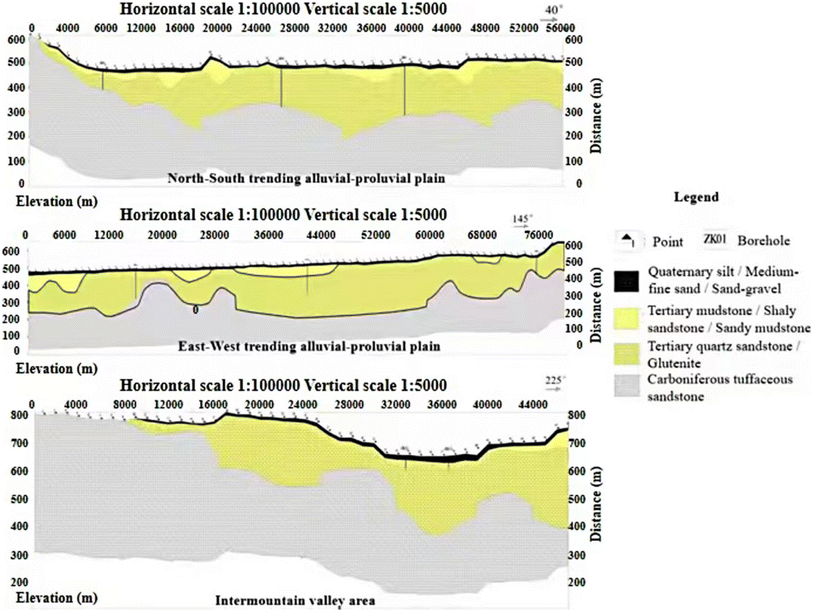
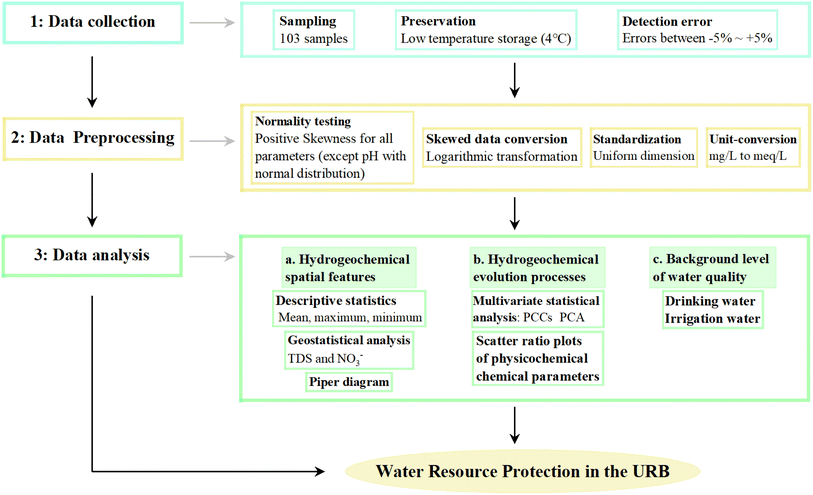
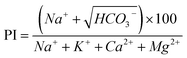
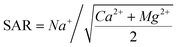
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) (HCO3− + CO32−) − (Ca2+ + Mg2+)
(HCO3− + CO32−) − (Ca2+ + Mg2+)