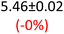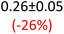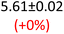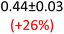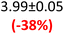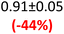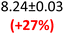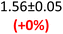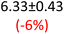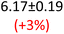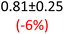Open Access Article
Open Access ArticleSub-picosecond permittivity of carbon nitrides probed with terahertz spectroscopy: revealing high dielectric response and conductivity
Reehab
Jahangir†
 ab,
Filip
Podjaski†
ab,
Filip
Podjaski†
 *ac,
Paransa
Alimard
*ac,
Paransa
Alimard
 a,
Sam A. J.
Hillman
a,
Sam A. J.
Hillman
 a,
Stuart
Davidson
a,
Stuart
Davidson
 b,
Stefan
Stoica
b,
Stefan
Stoica
 b,
Andreas
Kafizas
b,
Andreas
Kafizas
 a,
Mira
Naftaly
a,
Mira
Naftaly
 b and
James R.
Durrant
b and
James R.
Durrant
 ac
ac
aDepartment of Chemistry and Centre for Processable Electronics, Imperial College London, UK. E-mail: filip.podjaski@chem.ox.ac.uk
bNational Physical Laboratory, Teddington, UK
cDepartment of Chemistry, University of Oxford, UK
First published on 6th February 2026
Abstract
Organic based semiconductor materials offer emerging and sustainable solutions for solar energy conversion technologies and electronics. However, knowledge of their intrinsic (photo)physical properties is often limited, especially the effect of dielectric properties (εr = ε′) on exciton separation and charge generation at sub-picosecond timescales, which corresponds to THz frequencies. Thus, THz Time Domain Spectroscopy (THz-TDS) is used to directly and accurately extract the complex permittivity (ε = ε′ + iε″) and THz conductivity (σTHz) of organic Carbon Nitrides (CNx) and other polymers and elucidate the influence of environmental humidities. Overall, the THz dielectric response ε′ of CNx surpasses other organic and even glycolated materials, and water. For the ionic and 2D carbon nitride K-PHI, complex permittivity ε was observed to be strongly humidity dependent, with both ε′ and σTHz doubling from dry to humid conditions (ε′ from ∼4 to 8, σTHz 75 to 150 S/m, respectively). When compared to other photocatalysts, the THz dielectric response of CNx and especially humid K-PHI is within range of well-known oxides such as TiO2 that can efficiently generate charges from excitons, due to low exciton binding energies resulting from high ε′. The importance of dielectric property characterization on functionally relevant frequencies is thus highlighted, especially in the THz gap (0.1 – 10 THz), to understand the photophysical behaviour of organic semiconductors, even in the presence of water and hydrated ions. Such THz complex permittivity determination may also be beneficial for exploring next generation photo(electro)catalysts, electronics or ionotronics, and for computational property predictions that often require knowledge of ultrafast photophysical properties.
Broader ContextSolar energy conversion technologies are widely explored for renewable energy supply in the form of electricity and fuels, for sensors etc. Most rely on photovoltaic effects, where light absorption generates excitons that are subsequently separated into functional charges. In organic systems, this transition must overcome the exciton binding energy, which is inversely proportional to the materials’ dielectric response εr. But its highly frequency dependent values are typically unknown in the corresponding fs-ps regime for organic semiconductors (THz gap). Hence, they are approximated from static or low frequency values, or those at much higher frequency. Using THz time domain spectroscopy, we show that the full complex permittivity, i.e. the dielectric response εr and loss factors related to THz conductivity, can be measured directly and non-invasively, on the corresponding time scales of exciton separation and charge generation. Our study focussed on various polymers and Carbon Nitrides (CNx), and the influence of environmental effects in wetting materials. It reveals that glycolation, being used in polymers to enhance permittivity in the µs-s regime, has minor effects in the THz regime. CNx however have unexpectedly high THz dielectric values, being larger than water. The highest values are observed for the ionic CNx K-PHI in ambient or humid conditions, reflecting their application conditions. These are linked to synergistic ion-humidity effects, giving also rise to high THz conductivity. This highlights the crucial importance of permittivity probing in relevant conditions. |
Introduction
Organic semiconductor materials are increasingly used for optoelectronic and energy conversion applications since they are often easily processable and offer bottom-up tunability for desired properties and functions.1 A potential pathway for their exploitation in a sustainable manner, addressing increasing environmental challenges, lies in their ability to absorb visible light and to utilise this energy for electricity generation in solar cells, as photodetectors, or for the direct photocatalytic production of fuels and chemicals. In all these processes, the absorption of photons typically generates bound excitons, that are separated into charges, driving the desired function. However, in many organic based semiconductors, such as carbon nitrides (CNx), covalent organic frameworks, or polymer systems, light energy conversion is still less efficient than in inorganic materials or the much-studied hybrid perovskites. In particular, the generation of free charges from excitons in these materials is often a key challenge for efficient device performance.2,3 The fundamental reason for the strong exciton recombination often observed in organic based semiconductors is linked to their relatively high exciton binding energy ΔEEx_BE (compared with inorganic semiconductors), which is related to their intrinsic real permittivity εr:4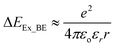 | (1) |
With low εr, the Coulomb attraction of excitons is poorly screened by the materials' backbones' polarizability, which affects the excitons' ability to dissociate, and thus hinders the generation, stabilization, and extraction of useful charges.6
The complex permittivity ε = ε′ + iε″ of materials, from which static real permittivity is typically deduced in the low frequency limit (also known as dielectric constant εr = ε′; a term to be avoided nowadays), actually is a highly frequency dependent property, as depicted in Fig. 1a. Crucially, often little is known about the complex permittivity properties of materials on the relevant timescale for exciton generation and separation – this typically being in the fs-ps regime, corresponding to THz frequencies. A further challenge is that the influence of environmental factors such as water exposure on the complex permittivity of the material is typically disregarded, with exciton separation often assumed to be an intrinsic material property. This environmental factor, however, is especially relevant in wetting or porous materials that have high volume interactions with their environment, and hence also in applications such as photocatalytic water splitting.7
 | ||
| Fig. 1 Frequency-dependent complex permittivity and CNx materials investigated. (a) Different polarisation mechanisms are activated with increasing frequency of an electric field E, leading to frequency dependent (dispersive) complex permittivity ε responses. The top scale shows the correlation of the timescale to the frequency at which these polarisation events may influence (opto)electronic properties (reproduced from 31 under the CC BY 4.0 license). (b) Schematics of atomic structures of the carbon nitrides (CNx) investigated: the one-dimensional graphitic CNx melon,21 and the 2D poly(heptazine imide) PHI (here with K+ and H+ ions as K-PHI and H-PHI, respectively).24 (c) Schematic of the transmission-mode Terahertz Time-Domain Spectroscopy (THz-TDS) set-up used and pictured, a processed sample of a melon pellet used for the study. | ||
Dielectric properties are typically probed from the mHz up to the MHz regime by electrochemical impedance spectroscopy (EIS), with free space microwave dielectric measurements with Vector Network Analyzers (∼0.5–300 GHz),8–11 and by optical means (e.g. Spectroscopic Ellipsometry (SE)) at 1014–1015 Hz (Peta–Hz), hence leaving out the important THz timescales, often known as the ‘THz-gap’ (Fig. 1a).12–14 Water has a very high DC real permittivity (ε′H2O ≈ 74), which has been shown to affect the intrinsic charge generation and stabilization of semiconductors.15 However, at THz regimes, its value is very moderate (ε′H2O, 1.5 THz ≈ 4.15).16 Both studies of the THz dielectric response of organic semiconductors, and the impact of water exposure on their response, have been very limited to date. Our study shows how to access the dielectric response of various organic semiconductors and its modification by humidity exposure, on time scales relevant for solar energy conversion and photocatalysis, by using terahertz time-domain spectroscopy (THz-TDS).
THz-TDS is an increasingly commonly available technique that allows to probe the complex permittivity ε of materials in their bulk (averaged over the volume of the probing beam interacting with the sample, no approximations required) and that have been exposed to different environments, thus providing important insights into their photophysical properties on the ps timescale and below (1 THz ∼1 ps) (Fig. 1a).17,18 For this study, different insulating polymers and the broadly employed organic semiconductor poly(3-hexylthiophene) = P3HT were chosen as references,19 while focussing on heptazine-based CNx materials, which are among the most widely used model organic semiconductors for various light driven energy conversion applications.4 The materials are hence broadly studied benchmark materials, and have modifiable wetting properties that are key to probe interactions with water – while also being sufficiently processable for the characterization. CNx are easy to synthesise and to post-functionalise, highly stable, and have visible light bandgaps (typically 2.7 to 3 eV) that are suitably positioned to drive various photo–redox reactions, such as photocatalytic water splitting. Their application areas also include environmental remediation, photodetectors and sensors, ion pumps, light driven microswimmers, photocharging and dark photocatalysis, as well as photobatteries. The latter are often linked to ionic interactions with photogenerated charges.20
The first THz complex permittivity measurements on these layered materials are now presented, comparing different graphitic carbon nitrides (also called gC3N4, which are also discussed to be the same as a 1D melon-type CNx)20,21 and 2D-linked poly(heptazine imide) PHI, which can contain different solvated ions in their pores (called M-PHI then), that affect their function and performance (see Fig. 1b).22–25 The influence of exposure to humidity (water) is further studied, which commonly has a strong influence on ionic mobility, and show its potential to synergistically affect a material's real and imaginary permittivity at THz frequencies, in cases where water is strongly adsorbed.26–30 The influence of an increase in intrinsic real permittivity and THz conductivity we describe herein is mirrored by more pronounced pre-charge signals of PHI in humid conditions, indicating modified exciton-to-charge relaxation mechanisms, measured by transient absorption spectroscopy (TAS) in the fs-ns regime, underlining the important influence of humidity on intrinsic photophysical properties. The findings are discussed in detail and especially in the context of photocatalytic reactions in water.
Results
Materials and preparation
The different CNx were synthesized following previously reported procedures (see Experimental methods for details). Three batches of g-C3N4 -type CNx were synthesised at slightly different conditions (g-C3N4 # 1 by annealing urea at 550 °C in air followed by an acid/base wash; g-C3N4 #2 by annealing urea in air at 500 °C; and g-C3N4 #3 by annealing urea in nitrogen at 550 °C); two batches of K-PHI (derived from melamine and KSCN, K-PHI #1 by annealing g-C3N4 in argon at 500 °C; and K-PHI #2 by annealing g-C3N4 in air at 550 °C); two batches of the protonated form of PHI (H-PHI #1, #2) were obtained from the K-PHI's by acid washing.24 Powder XRD data (Fig. S1) shows characteristic peaks at 2Θ = 13.1° and 27.8° for g-C3N4, each corresponding to (100) and (002) the periodic arrangement of the tri-s-triazine rings and the interlayer stacking planes respectively,32–34 and the prominent ∼10° and ∼28° peaks for K-PHI and H-PHI, corresponding to the (010) and (001) planes respectively.25 The optical band gaps, extracted from Tauc plots and diffuse reflectance measurements, are of approx. 3 eV for the melon samples and H-PHI, and ∼2.8 eV for the K-PHI samples (Fig. S2), as reported.To study their complex permittivity by THz-TDS (see Fig. 1c), thin pellets (d = 10 mm) were pressed from the materials (see SI, Tables S1–S3). Since THz probes the entire sample volume, precise knowledge of porosity is required.35 For this, the material density was measured by a He-pycnometer, resulting in 1.88(1) g cm−3 for melon #1 and K-PHI in ambient conditions, and 1.85(1) g cm−3 for H-PHI. Upon drying using conventional methods such as convection oven and desiccator, the amount of water adsorbed was reduced and stable densities of 1.72(1), 1.84(1), and 1.81(1) g cm−3 were obtained respectively (see Experimental Methods for more details). These were subsequently used to calculate an effective pellet porosity (see Fig. S3 and SI Note 1).
THz-TDS data was collected directly from these pellets in a custom-built chamber purged with dry compressed air to avoid the parasitic absorption of ambient water vapour (Fig. 1c). The terahertz pulse transmitted through the sample is Fourier-transformed from the time to frequency domain. The measured change in phase and amplitude relative to the reference beam allows calculation of the complex refractive index n. The complex permittivity εis extracted via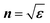 , including error propagation (SI Note 2). ε′(ω) depicts the frequency(ω)-dependend real permittivity (= dielectric polarisation reponse), and ε″(ω) the imaginary permittivity, which is a measure of energy dissipation due to loss mechanisms arising from polarisation, phonon contributions and accelaration of charge carriers due to the alternating electric field, relating linearly to THz conductivity (σTHz, eqn S11).
, including error propagation (SI Note 2). ε′(ω) depicts the frequency(ω)-dependend real permittivity (= dielectric polarisation reponse), and ε″(ω) the imaginary permittivity, which is a measure of energy dissipation due to loss mechanisms arising from polarisation, phonon contributions and accelaration of charge carriers due to the alternating electric field, relating linearly to THz conductivity (σTHz, eqn S11).
Ambient measurements
First, data acquisition was veryfied on high density polyethylene (HDPE) and hygroscopic poly(ethyleneglycol) = PEG, confirming literature values and expected dispersion trends (Fig. 2 and SI Note 3). Their real permittivity at 1.5 THz is ε′1.5 THz = 2.39 and 2.76, respectively (Table S4). New measurements on the semiconductor P3HT show a flat dispersion profile in the range probed, with ε′1.5 THz = 3.44. All g-C3N4 samples show very similar complex permittivity dispersion (Fig. S4, S6 and SI Note 4), with ε′1.5 THz = 5.5 in average being significantly higher than our reference material (+57% vs. P3HT, which is widely considered a the workhorse in the conjugated polymer family). This elevation in ε′ could be affected by the pores of g-C3N4 containing trapped water molecules (ε′1.5 THz ∼4.15), as these were measured in ambient conditions with 50% relative humidity (RH), which motivates further studying these materials by decoupling them from water (vide infra).Overall, from Fig. 3a,d and g, one can see that the 2D PHI based materials have increased ambient real and complex permittivity values compared to 1D melon/g-C3N4, and that the presence of alkali metal ions in the pores (here, K+) of PHI seems to affect the dispersion profile of the complex permittivity spectra. The average ε′ of K-PHI is 6.75, which is 22% higher than the average melon value, with a less flat dispersion profile compared to all other materials. H-PHI has ε′1.5 THz = 6.51 (similar to K-PHI), but with 40% reduced ε″1.5 THz (see Table S4 and SI Note 3).
 | ||
| Fig. 3 Real permittivity (ε′), imaginary permittivity (ε″) and THz conductivity (σ) spectra of melon # 1 (a–c), K-PHI # 1 (d–f), H-PHI # 2 (g–i) measured in vacuum dried, ambient and 100% RH conditions, respectively. The acquired frequency range for the samples varies due to different absorption.36 Shaded areas show the standard deviation for the respective material batch. (j–l): overview comparing the three CNx materials at 1.5 THz across different humidity environments, error bars represent standard deviation of all respective sample types. | ||
Since the ions in PHI are not bound to the material backbone,24,25 it appears that they are freely moving and contributing to the dielectric response, perhaps with a different (saturating) behavior than the PHI− backbone at frequencies above 1.5 THz, giving rise to stronger levelling-off (decreasing) for these samples due to increasingly fast polarization. Although the proton mobility on H-PHI is not yet clarified, it is likely that these hydrated ions are mobile within the pore structure.24 However, these much smaller H+ and probably mobile ions (via H3O+) appear to result in less pronounced interactions on the THz scale (akin to water itself), which has a lower ε′ than all CNx measured herein. In the ε″ spectra, the 2D structure of PHI appears to enable a stronger response than the 1D melon, and the presence of K+ ions seem to increase the THz response and conductivity (vide infra) most strongly (see SI Note 3 and 4).
Humidity dependent THz measurements
Terahertz spectroscopy is widely used to measure water contents in materials, making use of its high absorbance in the THz regime.26–28 The complex permittivity of water is distinct and well characterised in the THz regime (ε′1.5 THz ≈ 4.1 and ε″1.5 THz ≈ 1.9).16,37 While water has much higher values of ε″ than the materials in this study, its ε′ is smaller than that measured for CNx in ambient conditions, but higher than that of many common polymers like P3HT reported above. Thanks to this difference in ε′ with respect to CNx, the impact of adsorbed or structural water in these materials can also be resolved at THz frequencies. This makes THz-TDS measurements highly relevant for studying CNx, as the practical applications of these materials typically take place in aqueous conditions. Thus, the studied materials were measured not only in their ambient state, but also when vacuum-dried and at 100% relative humidity (RH), to generate more intrinsic and application relevant insights.As evidenced from density measurements (Fig. S3), the CNx samples studied herein are susceptible to water uptake, with g-C3N4 #1 changing in density more strongly and taking longer to stabilise than K-PHI # 1 or H-PHI upon drying (ambient density: 1.88 g.cm−3; dry: −8.5% for g-C3N4, –2.1% for K-PHI). This hydrophilic behaviour enabling water uptake is also qualitatively confirmed by contact angle measurements, with the melon samples being partially wetting (contact angles between 24° and 59°), and K-PHI & H-PHI being fully wetting (Fig. S5).
The full (dry, ambient, humid) THz-TDS datasets for g-C3N4, K-PHI and H-PHI are shown in Fig. 3. For g-C3N4 # 1 (first row, contact angle of 41°) the ε′ dispersion profiles vary little as the sample is vacuum dried or exposed to 100% RH for 4 h, whereas the imaginary permittivity ε″fluctuates slightly (Fig. 2a and b respectively). Similar trends are observed for the other samples melon #2 and melon #3 (Fig. S6), which are also slightly wetting, but with different contact angles (Fig. S5a, c and d). The THz conductivity of melon changes accordingly to its ε″ (Fig. 3c), i.e. dropping when dried.
For K-PHI on the other hand, which is fully wetting (Fig. S5b), the water content has a strong influence on the real permittivity (Fig. 3d and Table 1), yielding an increase of more than 100% from dry (ε′1.5 THz = 3.99) to humid (ε′1.5 THz = 8.24). The behaviour of ε″ (Fig. 3e) is more complex with the ambient and wet samples showing similar ε″ above 1 THz, and hence conductivity σTHz, being ∼50% higher than the dry sample. It suggests that the THz absorption and thus conductivity properties saturate with ambient water content and do not improve further. Below 1 THz, vacuum drying seems to affect the slope of ε″ – a process that is not yet understood – and potentially affected by measurement sensitivity in this range, as well as the fact that our K-PHI pellets become more fragile when dried under vacuum.
H-PHI shows effects in-between the other two CNx samples. ε′ is constant with respect to changes in humidity, akin to g-C3N4, but with a 25% higher value (Table 1), being close to ambient K-PHI (within error, Fig. 3g). ε″follows a behaviour like K-PHI, also approximately doubling upon exposure to ambient conditions, and not changing any more at 100% RH (Fig. 3h). The values for H-PHI are approximately 47% lower than for ambient K-PHI, indicating amplification effects by K+ ions.
Fig. 3j–l summarize the humidity dependent data for ε′, ε″and σ. Overall, the structurally porous 2D PHI materials have elevated ε′ values compared to g-C3N4 in ambient or wet conditions, and K-PHI's ε′ response significantly depends on the humid environment. The ε″ of g-C3N4 is constantly and significantly lower than of the PHI materials. It also increases at 100% RH, which may be related to the water loss or uptake observed in density measurements (Fig. S3) and related proton mobility. Both H-PHI and K-PHI approximately double their ε″ upon exposure to ambient humidity. K-PHI shows slightly elevated values and stronger dispersion than H-PHI. σTHz reflects this ε″ behaviour: g-C3N4 has moderate conductivity values that are slightly affected by humidity (32–40 S m−1 at 1.5 THz), being much lower than of the PHIs. The fact that H-PHI has elevated σTHz compared to g-C3N4, while increasing more strongly from dry to ambient (47 to 69 S m−1), points to two factors: an inherently increased ultrafast complex permittivity response with the 2D molecular backbone forming pore channels, and water playing a different, more pronounced role when adsorbed inside of PHI's confined 1D channels that do not dry out fully.24 This is even more pronounced in K-PHI, where σTHz is larger than in all humid cases of the other materials when being dry (75 S m−1, >2× higher than g-C3N4), and doubling again in humid conditions (150 S m−1), where it is > 4× higher melon and >2× than H-PHI.
Since for THz conductivity, literature values are available, where σTHz was obtained directly from THz amplitude changes and using thin-film approximations, these can be put into context of other reported materials for further comparison most easily:38 triazine based carbon nitrides, poly(triazine imide) = PTI, are structurally similar to PHI and can also contain ions in smaller pores. The ion free PTI-IF, and PTI-LiBr show σ ∼7 and 35 S m−1 at 1.5 THz in ambient conditions, measured in free-standing pellet form.39 The conductivity on THz timescales is hence also strongly improved by the presence of ions (x3.7 in PTI, compared to ∼x2 in H– vs. K-PHI), albeit at lower absolute values, being only 10% for PTI-IF vs. H-PHI (7 vs. 70 S m−1), and 17% in the ionic case (26 vs. 150 S m−1 for PTI-LiBr vs. K-PHI, albeit with different ions). A recent report on ion-free tuneable porous covalent organic framework (COF) membranes showed σ1.5 THz between of 5.45 and 75.59 S m−1 for membrane samples denoted TAM-DBD and TBP-TFP respectively,40 hence being overall in the range of CNx, but still lower than ambient K-PHI.
These findings suggest that hydrated ion mobility contributes to much higher THz dielectric properties than expected – and that ε′, ε″ and σ are hence strongly dependent on the environmental conditions the material is probed in (in dry form as neat material, or humid; more akin to application conditions). Thus it is also anticipated that processes occurring on the timescale of THz, i.e. the binding of excitons or charge transfer excitons, and the generation of charges, are positively influenced by the real permittivity in K-PHI in terms of solar energy conversion applications, and especially those in the presence of humidity and water. We hence investigated these materials using fs-ns TAS.
fs-ps transient absorption spectroscopy (TAS) measurements
The real THz permittivity, which corresponds to the picosecond timescale, is an important factor for the Coulomb binding of excitons and the generation of charges by exciton or charge transfer exciton dissociation and their relaxation (eqn (1)). To provide further evidence of the influence of humidity on the dissociation of (bound) excitons to free charges, the charge carrier generation properties of K-PHI samples in ambient and humid conditions were studied using fs-ns TAS. The results are shown in Fig. S8. Panels a) and b) show the spectra of a vacuum-dried K-PHI film in ambient and humid conditions from 0.2 ps to 5 ns, with the signal normalized at each delay time to illustrate temporal shape changes (see Fig. S9 a and c for raw data). After excitation with 355 nm light pulses, the material has a pronounced broad absorption signal which appears to red shift from 600 to 670 nm over 5 ns. The latter-timescale 670 nm feature is assigned to electron absorbance in the K-PHI following bandgap excitation.41–45 From normalized data at each time snapshot, it can be observed that this red-shift begins within the first ps, and hence has a 2nd contribution before stable charge formation. The shape of the 500–650 nm shoulder is much more pronounced in humid conditions than in dry conditions (Fig. S8 b vs. a), clearly indicating the impact of humidity on ultrafast dynamics of K-PHI. Using global analysis, or simple subtraction of early (sub-ps) and late (ns) signals, the spectra can be deconvoluted into two distinct parts (Fig. S8c), with the red part displaying the shoulder component. This allows to compare the relative amplitude and decay (Fig S9–S11) for ambient and humid conditions, respectively. It is possible that the shoulder in the 500–650 nm region is linked to an excitonic or exciton-related state.44 Since the true nature cannot be clarified yet, it is termed as unrelaxed (pre-charge) signal. This signal peaks at times <1 ps and decays while the charge signal rises (peaking at 30–50 ps), consistent with a typical (charge transfer) exciton dissociation into charges, or simply charge related relaxation process (see Fig S10 and S11 for fluence dependent data). It is found that this unrelaxed signal is consistently more pronounced in the wet samples comparted to the ambient ones (∼+20%, Fig. S9e). This observation hence suggests that the more pronounced, unrelaxed pre-charge feature existing on the sub-ps timescale is a consequence of water-induced backbone polarization increasing the dielectric screening and hence impacting the photophysics and decay of excited states. Although the details of spectral assignment for the nature of such excitonic/unrelaxed states requires further dedicated efforts, this measurement underlines an important role of humidity for modifying the K-PHI real permittivity response to photoexcitation in the 0.5 to 5 ns regime, corresponding to the THz frequency range probed. This analysis also directly shows that the yield of this ps-signal can be modified in the material by the presence of water, which is linked to increasing real permittivity in K-PHI, enabling the stabilization of light-induced species that subsequently relax into charges.Discussion
Using THz-TDS, it is shown that the g-C3N4 and PHIs have a relatively high complex permittivity in the THz regime compared with HDPE, PEG and P3HT, qualifying CNx as High Refractive Index Polymers (HRIP), a class of materials that could be useful for many advanced flexible optoelectronic applications such as anti-reflective coatings and encapsulants for OLEDs,46 along with photovoltaic and photocatalytic applications. The real permittivity of proton-terminated g-C3N4 and H-PHI does not vary much with humidity. As a result, it can be concluded that water cannot fully account for the relatively higher complex permittivity values we see in g-C3N4 but is rather a result of its unique chemical structure when compared to P3HT. The heteroaromatic rings with the –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–C– bonds forming an extended conjugated network likely contribute to the molecular polarizability α of g-C3N4, thus elevating its real permittivity ε′ (described by the Clausius Mossoti relation (eqn (2))) beyond that of a conjugated polymer such as P3HT:47
N–C– bonds forming an extended conjugated network likely contribute to the molecular polarizability α of g-C3N4, thus elevating its real permittivity ε′ (described by the Clausius Mossoti relation (eqn (2))) beyond that of a conjugated polymer such as P3HT:47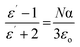 | (2) |
Free water (ε′1.5 THZ ∼4.15), which may be the reason for slight changes in ε′ in presence of humidity (see Table 1, PEG e.g.), can also be ruled out from K-PHI's increased permittivity, since the measured ε spectra cannot be explained by a linear contribution of water adding to complex permittivity (see SI Note 5 and Fig. S11).
So far, it can also be summarized that K-PHI in a dry, intrinsic state has a lower real THz-permittivity than g-C3N4, but with the ions present in the 2D structure and responding to water, ε′ can be doubled, far beyond g-C3N4, H-PHI and water alone (Fig. 2i).
ε′THz in context to other materials
Fig. 4 shows the real permittivity of typical inorganic semiconductors used for solar energy conversion and compares them with organic materials and this study. Indeed, it is found that slightly elevated values exist for solar cell materials like Si or GaAs (ε′1.5THz >10), but many others show only modest values being in the range of humidity variations of K-PHI, i.e. ε′1.5THz = 3–8: e.g. the hybrid perovskite CH3NH3PbI3, II–VI semiconductors like CdTe, and oxide semiconductors like TiO2 or Fe2O3 used frequently for photo(electro)catalysis. The often-made generalization that inorganic materials have higher dielectric response than organic materials, enabling better exciton separation,4 may therefore not be universally true in the THz regime. It can also be noted that exchange of the mobile A-site cation in the lead halide perovskites from methylammonium (CH3NH3) to Cs also show drastic changes in THz permittivity (ε′1.5THz changes from ∼7.7 (ref. 4) (ref. 48) to ∼20.5 (ref. 4) (ref. 49)) despite only a fraction (cation) of the material being modified. | ||
| Fig. 4 Real permittivity at 1.5 THz (ε′1.5THz) for common semiconductor materials used for solar energy conversion applications, relative to measured CNx and water.48–58 | ||
Influence of structure and ions
In K-PHI, pore ions (K+) are in a hydrated state and loosely bound to the 2D PHI− backbone.24,25 It is therefore suggested that these ions, confined in 1D pore PHI channels, have an important impact on the photophysics of K-PHI in the THz regime. A closer look shows K-PHI's ambient and humid ε′ profile rises slightly initially and drops beyond 0.75 THz (Fig. 3d). This indicates that the lower frequency range (0.25–0.75 THz) might capture a snapshot of the ionic (pore confined, hydrated K+) polarisation contributing initially, and then becoming less active as the K+ ions become unable to respond to the alternating electric field above 1 THz (maximum response time is exceeded). H-PHI on the other hand maintains a relatively constant dispersion profile across the frequency spectrum, possibly due to the much lighter H+ ions having a more constant response across the frequencies probed (Fig. 3g). The monotonous dispersion trend in H-PHI and its ε′ values close to K-PHI in ambient conditions suggest that the dominant differences between 1D g-C3N4 and 2D PHI arises from the covalent 2D structure with 1D pore channels in PHI itself, and by the presence of water affecting hydrated ion motion (of H+ and K+) on THz timescales therein. In particular, K-PHI's significantly larger ε′ in humid or aqueous conditions (+100% vs. dry) should in theory faciliate the separation of excitons and charges through decreased Coulomb attraction, whilst more agile polarization should increase charge generation and transport on a fs-ps-timescale. The TAS data in this study provides preliminary evidence for this, with additional (probably excitonic) signals on the timescale between 0.2 and 100 ps arising more pronouncedly in conditions with higher permittivity.Relation to complex permittivity and conductivity
Upon drying, a decrease in ε′ is observed in K-PHI, likely related to lower hydrated K+ ion motion due to a reduction in the water content within the 1D pore channel. However, some water is still expected to be trapped in the pores.24,25 In H-PHI, the residual water may still contribute sufficiently to polarization inducing proton mobility, and thus explain the unchanged ε′ values upon vacuum drying. This can be further rationalized when discussing conductivity, σTHz (being linearly related to ε″, eqn S11): an earlier report on the dominantly ionic conductivity of metal-containing PHI (M-PHI) with different ions, including EIS and NMR probing ionic mobilities on different length scales, indicated that the ionic mobility of M-PHI, contributing to the conductivity in the probed 10−3-106 Hz range, drops significantly when the samples are dried – as in the THz case. On the other hand, the enhancement effect of hydrated ion motion showed an onset of saturation at ambient RH values. This is consistent with the findings in this study, which now extend the frequency regime to the THz timescale: the value of ε″1.5 THz the σTHz are similar for both ambient and 100% RH conditions for each, H– and K-PHI respectively, and halve in dry conditions. Akin to EIS measurements, the influence of adsorbed structural water on material properties saturates in ambient conditions, and the conductivity effects are stronger with metallic ions. In the EIS regime, H-PHI has conductivities order of magnitude below K-PHI – probably since aqueous protons do not have dominant effects in this mHz–MHz frequency range. In the THz regime, H-PHI shows comparably high conductivity values, but K-PHI has σ1.5 THz 67% higher than H-PHI (see Fig. S4), indicating that the larger and heavier K+ ions experience stronger acceleration and resistance within the THz alternating electric field, leading to stronger wave dissipation – but that protons also respond similarly in the 2D H-PHI network on fs-ps timescales.The high ε′ values of humid K-PHI cannot be explained solely by the presence of water in isolation. The humidity data in this study instead suggests that water affects the ε′ of K-PHI through synergistic interactions with ions – likely caused inside structural pore channels. It is suggested that this synergy may occur also in other porous, hydrophilic materials. Such an enhancement in ε′THz (for exciton separation and for charge accumulation inside the material) and in ε″THz (resulting in good conductivity effects on ultrafast fs-ps timescales) seems a key and highly desirable feature for efficient direct conversion of solar energy to fuels in presence of water, as aqueous conditions can apparently boost ε′ responsible for exciton dissociation and charge accumulation in the inner volume of the material, on timescales at which these processes occur. At the same timescale, when charges are just formed, boosted σTHz improves charge transport and hence their extraction. These findings are likely a key factor for the enhanced activity for photocatalytic hydrogen evolution reported for PHI-materials compared to g-C3N4,24,59 although the overall photocatalytic process is more complex and contains also other factors that need to be considered for efficiencies.60
Context to other real permittivity techniques
As explained in the introduction, the dielectric properties of materials are strongly dispersive across the frequency range from 1 Hz up to ∼1015 Hz. The techniques available, each probing a fraction of the frequency spectrum, hence give different values and information.THz-TDS measures complex permittivity at frequencies between 0.3–5 THz. It is a pulsed technique, with the pulse duration being about 1 ps and repetition rate about 80 MHz. Therefore, it observes dielectric response at THz frequencies on timescales of a few ps. EIS measures steady-state impedance at a range of frequences, typically up to a few MHz, with the possibility of a few GHz. At these frequencies, dielectric response differs significantly from its values at THz (Fig. 1a); and moreover, complex permittivity cannot be obtained straightforwardly from impedance, requiring complex equivalent circuit models. SE measures the changes in amplitude and phase at specific photon energies in the visible and near-infrared at differing angles of incidence, from which constants such as refractive index and absorption coefficient are extracted through an optical model, thus rendering it an indirect method of complex permittivity characterisation. At these frequencies, using light in the UV-vis-NIR, dielectric response differs significantly from its values at THz, especially so in materials possessing a Reststrahlen band.17
Given the novelty of the terahertz dielectric property measurements for carbon nitride polymorphs reported herein, we can now place our results in the context of current literature available on these materials, measured with standard techniques such as EIS and Spectroscopic Ellipsometry (SE). These probe the low-mid frequency (10−3 to 106 Hz), and very high frequency range accessible by UV-NIR ellipsometry (1014 to 1016 Hz), respectively and provide the DC and ultrafast ε′ values often referred to as extremes, since other data is missing. The trends in real permittivity ε′, summarized in Fig. 5, illustrate that the THz measurements indeed show intermediate values following the expected trend with frequency: EIS estimated ε′ to have values at ∼15 and 7 at 10−3-105 Hz for K-PHI and g-C3N4 in ambient conditions, respectively.25,61 Both drop significantly to ∼7 and ∼5 at 1.5 THz (−53% and −29%). In the 1014–1015 Hz SE-regime (also ambient conditions), melon stays constant (∼5),62 while K-PHI further decreases to ∼3, 63 which corresponds to its dry value at ∼2 THz.
 | ||
| Fig. 5 Real permittivity comparison over the frequency spectrum for melon and K-PHI, with data extracted from Electrochemical Impedance Spectroscopy (EIS), THz (data from Fig. 2) and Spectroscopic Ellipsometry (SE) (literature values reproduced under CC by 4.0 license),47,62,63 confirming the predicted dispersive trends in real permittivity (Fig. 1a). | ||
The parallel trend between EIS and THz-TDS, with K-PHI retaining its higher dielectric response compared with melon in ambient conditions up to the THz regime, indicates that both EIS and THz-TDS are suitable for a relative comparison of materials in their real permittivity response, including ambient & humidity effects. SE may capture contributions from electronic polarisation at ultrafast timescales (several fs), as these optical excitations are too fast for atomic or molecular rearrangements in either water or CNx materials. Consequently, we observe a similarity between THz permittivity in the dry state, and the one extracted from SE in both melon and K-PHI.
This shows that drawing conclusions about intermediate and especially THz processes from EIS & SE measurements alone may be insufficient, and that separate THz analysis is crucial for capturing different phenomena on its timescales. This THz-characterization is even more relevant if materials are involved where illumination generates carriers that are key to function on the picosecond timescale. The drop from ε′DC (static dielectric response) or EIS to ε′THz is hard to extrapolate, and may be different even for similar materials, as shown here for g-C3N4vs. PHI. While SE may enable estimation of the THz permittivity response in dry conditions, the result may be strongly modified in reality – by ambient effects, ionic or other phenomena in the THz regime, and on slower times. As such, the real permittivity at the low or high frequency limit provides insufficient information on the functionality of materials used for energy conversion.
Water and wetting groups
The presence of water, or its inclusion by wetting groups with high static real permittivity ε′DC (e.g. ethylene glycol),15,64 was discussed earlier in relation to its ability to boost photocatalytic efficiency of polymers e.g. through enhanced permittivity (measured by EIS).65–67 While water has a high dielectric response in the low frequency regime, this is not the case in the THz regime. Hence, it can increase the response of materials with lower ε′ values than itself (ε′ ∼2.77 for ambient PEG-8000 vs. 4.1 for water at 1.5 THz) from a linear combination of water + material, especially in wetting materials, but this effect may be smaller than expected (2–5% changes observed for poly(ethylene glycol)). However, it was not previously clear a priori that ε′ can be increased for materials that have larger ε′ values than water, like CNx,27 where we now revealed synergistic effects beyond those expected from a linear combination of the material and water alone. Hence, when charge stabilizing effects through wetting groups are observed on in ultrafast (fs-ns) spectroscopy measurements (and not only on sub-µs time scales corresponding to EIS analysis) in solar cell or photocatalyst materials, they may likely be linked also to modified THz properties, which can in future be studied separately by THz-TDS – giving a better approximation to applications also involving water, or other environments in principle.Solar energy conversion
Despite the importance of understanding (modified) permittivity on THz timescales vs. mHz–MHz timescales (EIS) or SE, it remains an open question as to which timescale is most relevant for enhancing light-energy conversion function. The result likely depends on the application scenario and on the bottleneck in the process, i.e. on morphological factors (e.g. particle size, grain boundary density and their effects), on the function and density of intrinsic traps, and on transport properties – i.e. the ability to make use of light generated charges after collecting them.4,60,68,69 While the fast timescales are crucial for exciton dissociation and charge carrier generation, they don't have to be limiting (see hybrid perovskites, or SrTiO3 enabling 100% photocatalytic quantum efficiency as perfectly engineered cases70), and slower (µs-s) times scales may also be highly relevant for photocatalytic function, since surface catalytic processes are often slow, requiring charge stabilization.71,72A common method to increase the low frequency permittivity is to add functional groups like ethylene glycol with high ε′ values at low frequences to polymers with intrinsically lower ε′ values.15,64 These functionalizations are reported to increase the conversion efficiency of sunlight to hydrogen in photocatalysis due to various effects occurring on different timescales including the ns-regime,15 but their influence is most clear in the lifetime of charges extending to the µs-s range (corresponding to EIS frequencies, i.e. 1 Hz–1 MHz) due to enhanced polarizability of the material. However, less is known about the functional groups' direct influence on exciton dissociation to charges, which still is a major bottleneck in photocatalysis with organic materials and related solar energy conversion technologies. THz-TDS complex permittivity analysis, carried out herein, can in principle provide insights on the origin of photophysical interaction effects expected in the sub-ps timescale, and thus directly support description of exciton stability (lifetime) and charge generation. At present, such phenomena are typically measured using fs-ns TAS, which is unable to provide direct photophysical material properties like ε′ and can be difficult to interpret due to the high photon fluxes used in the excitation pulse experiments. Further, for the purposes of material modelling and property prediction, knowledge of the complex permittivity at given timescales is key to correctly elucidate corresponding properties and behaviour of functional interest. This is especially key in the context of Artificial Intelligence (AI) and Machine Learning, e.g. if targeting improved synthetic (bottom-up) design, which is particularly promising with organic materials. THz measurements easily provide such valuable insights, without complex models or assumptions being required.
Potential for light-driven ionics
Enhanced THz ε′ relative to melon is herein clearly visible. Further, the presence of aqueous alkali metal ions contributes to THz polarization of the material, and to differently pronounced relaxation mechanisms on the ps-scale. Since permittivity increases because of higher polarizability, the enhanced ε′ in K-PHI indirectly show that also light-driven generation of charges may in turn affect structural hydrated K+ ions on picosecond timescales. As such, these insights also possibly point to efficient optoionic or photocharging interactions, where ions stabilize charge carriers on ultrafast time scales.73,74 In K-PHI, such optoionic stabilization (being mediated by water) occurs on the fs-ps regime – where light generated charge carriers can interact with internal or external (solvated) ions, modifying the material's stoichiometry and with that, photophysical properties. The presence of these alkali metal ions is key to enable photocharging, i.e. the intrinsic combination of light absorption (solar cell function) and charge storage (battery function) properties. Besides photobatteries, this can be used for effects like dark photocatalysis, for powering microparticles, and for sensing etc.20,41–43,63,75 An ionically enhanced permittivity on the THz timescale is expected to be beneficial for generating and/or stabilizing photogenerated charges quickly and efficiently. We therefore suggest that the overall strength optoionic effects and the efficiency of photocharging materials and light-driven ionotronic devices may benefit from enhanced THz permittivities.76–78Summary & outlook
This work presents THz-TDS is a powerful, non-destructive, and non-contact tool for directly characterizing the complex permittivity ε of organic semiconductors. It provides valuable insights into their dielectric response and related properties (e.g. THz conductivity). This characterization on the sub-ps timescale closes the “THz gap” (Fig. 1a) resulting from limitations to EIS (mHz to 106 Hz), microwave dielectric measurements (GHz timescales) and SE (1014 to 1016 Hz). Different polarization processes occur on respective timescales, resulting in timescale-dependent trends and environmental effects that are difficult to extrapolate based on measurements made on non-corresponding timescales. The THz regime encompasses processes on time scales involving exciton generation and separation as well as fast charge relaxation effects. Hence, the characterization of complex permittivity by THz-TDS can provide a broader understanding of functional materials used for light conversion applications, including photovoltaics, photodetectors and photocatalysts, and enable more accurate modelling from knowledge of (photo)physical properties. For example, Fig. 4 compares the ε′1.5THz of materials used for such applications, pointing out that inorganics used for solar energy conversion are not necessarily very high dielectrics in the THz regime, and that CNx has comparable dielectric properties on this timescale. Hence, their photophysical properties for solar energy conversion are probably less limited by exciton dissociation, compared to traditionally used semiconducting polymers.This study further highlights the importance of structurally adsorbed water being able to synergistically enhance the complex permittivity in the THz-regime. Here, a more straightforward and unambiguous characterization of contributions from the backbone, water and ions become possible when comparing to probing techniques like EIS and SE. Hence, care must be taken when comparing measurements acquired in different conditions, and when materials are employed in humid environments, it is important bear in mind that the properties of dry materials may be significantly different from those occurring in real application scenarios (such as photocatalytic reactions in water or other electrolytes). This is also relevant for modelling purposes, which often neglect environmental factors, or might use dielectric properties as input parameters, which cannot be generalized from other frequency domains, and would neglect synergistic effects seen here in the THz regime.
The use of THz-TDS, being increasingly available, is hence highly recommended for characterization of functional materials used in solar energy conversion, or with crucial function in the (sub–)ps regime. In particular, the surprising findings of structural water or hydrated ions apparently contributing to the real dielectric permittivity response even on ps timescales (promoted via ambient humidity, increasing the response beyond the material's backbone alone) sheds further light on material analysis and selection for future ionotronic materials operating on THz timescales – for which K-PHI or other porous framework materials containing ions, like COFs, MOFs or porous (ionic) polymers may be good candidates. THz-TDS also enables direct extraction of their conductivity σTHz without further approximations. Further, direct light-driven ionic effects (optoionics) may potentially be characterized more directly by this technique in the future. The ability of soft ionic materials to exhibit strongly enhanced permittivities in the presence of water may help to combat the often-limiting strong exciton recombination seen in apparently low dielectric materials – and often targeted by wetting – although without knowledge of its fs-ps time scale influence. THz-TDS may hence be a key component for addressing fundamental challenges of organic semiconductors and their applicability for solar energy conversion and thus be an enabler in the transition to a more sustainable energy economy and the development of related materials infrastructure.
Author contributions
J. D., M. N. and F. P. designed and supervised the project. F. P. and P. A. synthesized and prepared the CNx materials. D. S. and S. S. performed and analysed density measurements, P. A the XRD measurements. R. J. and F. P. prepared CNx pellets for characterisation. R. J. and F. P. measured contact angles. R. J. measured and analysed the THz data, with assistance and supervision of M.N. F. P. and S. H. prepared, performed, and analysed fs-ns TAS measurements. F. P. and R. J. wrote the manuscript draft. All authors incl. A. K. contributed to manuscript revisions and discussions.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data reported in this study will be shared on the research repository zenodo following acceptance. See DOI: 10.5281/zenodo.18551232.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5el00170f.
Acknowledgements
The authors acknowledge Dr Carolina Pulignani and Prof. Erwin Reisner (Dep. Of Chemistry, University of Cambridge) for providing carbon nitrides for this study, Dr Nuria Tapia-Ruiz and Sarah McKinney for use of their labs and assistance in pellet preparation, and Dr Andrew Gregory for use of his in-house NPL THz-TDS software for thickness optimisation. Dr Julia Kröger (EnBW) and Prof. Ji-Seon Kim (Imperial) are acknowledged for fruitful discussions. F. P. and J. D. acknowledge funding from UKRI (grant reference EP/X027449/1) and EPSRC (grant reference EP/Z536258/1). F. P. further acknowledges funding from The Royal Society (URF\R1\251843), and J. D. from EPSRC (grant reference EP/T028513/1). P. A. thanks the Science and Solutions for a Changing Planet DTP (NE/S007415/1), hosted at the Grantham Institute for Climate Change and the Environment for funding this research. A. K. thanks the EPSRC for a Programme Grant (EP/W017075/1). Work at the National Physical Laboratory was supported by the UK Government's Department for Science, Innovation and Technology (DSIT) through the UK's National Measurement System programs.References
- H. Naito, Organic semiconductors for optoelectronics, Wiley, 2021 Search PubMed.
- M. T. Sajjad, A. Ruseckas and I. D. W. Samuel, Matter, 2020, 3, 341 CrossRef.
- H. Bronstein, C. B. Nielsen, B. C. Schroeder and I. McCulloch, Nat. Rev. Chem., 2020, 4, 66 Search PubMed.
- T. Banerjee, F. Podjaski, J. Kröger, B. P. Biswal and B. V. Lotsch, Nat. Rev., 2021, 6, 168 Search PubMed.
- O. V Mikhnenko, P. W. M. Blom and T.-Q. Nguyen, Energy Environ. Sci., 2015, 8, 1867 RSC.
- Y.-X. Li and W. Choi, Chem Catal., 2022, 2, 1517 Search PubMed.
- Q. Zhou, Y. Guo and Y. Zhu, Nat. Catal., 2023, 6, 574 CrossRef.
- J. W. Schultz, Focused beam methods: measuring microwave materials in free space, CreateSpace Independent Publishing Platform, Atlanta GA, USA, 2012 Search PubMed.
- R. A. Alahnomi, Z. Zakaria, Z. M. Yussof, A. A. Althuwayb, A. Alhegazi, H. Alsariera and N. A. Rahman, Sensors, 2021, 21, 2267 CrossRef PubMed.
- J. T. Kindt and C. A. Schmuttenmaer, J. Phys. Chem., 1996, 100, 10373 CrossRef.
- T. Mosavirik, M. Soleimani, V. Nayyeri, S. Hossein Mirjahanmardi, G. Student Member, M. Soleimani, S. Member and O. M. Ramahi, IEEE Trans. Instrum. Meas., 2021, 70, 1 Search PubMed.
- A. Armin, D. M. Stoltzfus, J. E. Donaghey, A. J. Clulow, R. C. R. Nagiri, P. L. Burn, I. R. Gentle and P. Meredith, J. Mater. Chem. C, 2017, 5, 3736 RSC.
- N. Mallo, S. McAnally, R. Chu, M. Babazadeh, H. Jin, P. L. Burn, I. R. Gentle and P. E. Shaw, J. Mater. Chem. C, 2023, 11, 14382 RSC.
- Z. Fu, X. Zhang, H. Zhang, Y. Li, H. Zhou and Y. Zhang, Chin. J. Chem., 2021, 39, 381 CrossRef.
- J. Kosco, S. Gonzalez-Carrero, C. T. Howells, W. Zhang, M. Moser, R. Sheelamanthula, L. Zhao, B. Willner, T. C. Hidalgo and H. Faber, Adv. Mater., 2022, 34, 2105007 CrossRef PubMed.
- W. J. Ellison, J. Phys. Chem. Ref. Data, 2007, 36, 1 CrossRef.
- M. Koch, D. M. Mittleman, J. Ornik and E. Castro-Camus, Nat. Rev. Methods Primers, 2023, 3, 48 CrossRef.
- J. Neu and C. A. Schmuttenmaer, J. Appl. Phys., 2018, 124(23), 124 CrossRef.
- X. Huang, X. Wang, Y. Zou, M. An and Y. Wang, Small, 2024, 20, 2400874 CrossRef.
- O. Savateev, M. Antonietti and X. Wang, Carbon nitrides : structure, properties and applications in science and technology, De Gruyter, Berlin, 2023 Search PubMed.
- B. V. Lotsch, M. Döblinger, J. Sehnert, L. Seyfarth, J. Senker, O. Oeckler and W. Schnick, Chem. Eur J., 2007, 13, 4969 CrossRef PubMed.
- O. Ostroverkhova, Chem. Rev., 2016, 116, 13279 CrossRef.
- A. Savateev, S. Pronkin, M. G. Willinger, M. Antonietti and D. Dontsova, Chem.–Asian J., 2017, 12, 1517 Search PubMed.
- H. Schlomberg, J. Kröger, G. Savasci, M. W. Terban, S. Bette, I. Moudrakovski, V. Duppel, F. Podjaski, R. Siegel, J. Senker, R. E. Dinnebier, C. Ochsenfeld and B. V. Lotsch, Chem. Mater., 2019, 31, 7478 CrossRef.
- J. Kröger, F. Podjaski, G. Savasci, I. Moudrakovski, A. Jiménez-Solano, M. W. Terban, S. Bette, V. Duppel, M. Joos, A. Senocrate, R. Dinnebier, C. Ochsenfeld and B. V. Lotsch, Adv. Mater., 2022, 34, e2107061 CrossRef.
- J. F. Federici, J. Infrared Millim. Terahertz Waves, 2012, 33, 97 Search PubMed.
- Z. Zang, Z. Li, X. Lu, J. Liang, J. Wang, H.-L. Cui and S. Yan, Comput. Electron. Agric., 2021, 191, 106515 CrossRef.
- L. Quancheng, L. Xiaoxia, D. Hu, S. Jingxin, G. Deyun, L. Lichuan, Z. Yuzhe and S. Liping, Opt. Eng., 2020, 59, 84102 Search PubMed.
- X. Chen, D. Cao, H. Chen, X. He, G. Fang and X. Chen, Photonics Res., 2025, 14, 230 CrossRef.
- G.-H. Oh, H.-S. Kim, D.-W. Park and H.-S. Kim, Opt Laser. Eng., 2020, 128, 106036 CrossRef.
- W. Si, Q. Liao, W. Hou, L. Chen, X. Li, Z. Zhang, M. Sun, Y. Song and L. Qin, Coatings, 2022, 12, 766 CrossRef CAS.
- D. J. Martin, K. Qiu, S. A. Shevlin, A. D. Handoko, X. Chen, Z. Guo and J. Tang, Angew. Chem., 2014, 126, 9394 Search PubMed.
- X. Wang, Y. Ren, Y. Li and G. Zhang, Chemosphere, 2022, 287, 132098 CrossRef CAS PubMed.
- G. J. Hutchings, P. R. Davies, S. Pattisson, T. E. Davies, D. J. Morgan and M. W. Dlamini, Catal. Commun., 2022, 169, 106480 CrossRef CAS.
- M. Naftaly, I. Tikhomirov, P. Hou and D. Markl, Sensors, 2020, 20, 3120 CrossRef CAS.
- P. U. Jepsen and B. M. Fischer, Opt. Lett., 2005, 30, 29 CrossRef.
- A. Bergner, U. Heugen, E. Bründermann, G. Schwaab, M. Havenith, D. R. Chamberlin and E. E. Haller, Rev. Sci. Instrum., 2005, 76, 63110 CrossRef.
- A. M. Ulatowski, L. M. Herz and M. B. Johnston, J. Infrared Millim. Terahertz Waves, 2020, 41, 1431 CrossRef CAS.
- D. Burmeister, A. Eljarrat, M. Guerrini, E. Röck, J. Plaickner, C. T. Koch, N. Banerji, C. Cocchi, E. List-Kratochvil and M. J. Bojdys, Chem. Sci., 2023, 14, 6269 Search PubMed.
- A. Puthukkudi, S. Nath, P. Shee, A. Dutta, C. V Rajput, S. Bommakanti, J. Mohapatra, M. Samal, S. Anwar and S. Pal, Adv. Mater., 2024, 36, 2312960 Search PubMed.
- H. Kasap, C. A. Caputo, B. C. M. Martindale, R. Godin, V.-H. Lau, B. V. Lotsch, J. R. Durrant and E. Reisner, J. Am. Chem. Soc., 2016, 138(29), 9183–9192 CrossRef CAS.
- V. W. Lau, D. Klose, H. Kasap, F. Podjaski, M. Pignié, E. Reisner, G. Jeschke and B. V. Lotsch, Angew. Chem., 2017, 129, 525 Search PubMed.
- F. Podjaski, J. Kröger and B. V. Lotsch, Adv. Mater., 2018, 30, 1705477 Search PubMed.
- W. Yang, R. Godin, H. Kasap, B. Moss, Y. Dong, S. A. J. Hillman, L. Steier, E. Reisner and J. R. Durrant, J. Am. Chem. Soc., 2019, 141(28), 11219–11229 Search PubMed.
- A. Gouder, A. Jiménez-Solano, N. M. Vargas-Barbosa, F. Podjaski and B. V. Lotsch, Mater. Horiz., 2022, 9, 1866 RSC.
- J. Zhang, T. Bai, W. Liu, M. Li, Q. Zang, C. Ye, J. Z. Sun, Y. Shi, J. Ling and A. Qin, Nat. Commun., 2023, 14, 3524 CrossRef CAS.
- P. Giusto, D. Cruz, T. Heil, H. Arazoe, P. Lova, T. Aida, D. Comoretto, M. Patrini and M. Antonietti, Adv. Mater., 2020, 32, e1908140 Search PubMed.
- S. R. Konda, Y. Lin, R. A. Rajan, W. Yu and W. Li, Materials, 2023, 16, 610 CrossRef CAS PubMed.
- I. Maeng, S. Chen, S. Lee, S. Wang, Y.-K. Kwon and M.-C. Jung, Mater. Today Phys., 2023, 30, 100960 CrossRef CAS.
- J. Calvo-de La Rosa, A. Locquet, D. Bouscaud, S. Berveiller and D. S. Citrin, IEEE Trans. Terahertz Sci. Technol., 2021, 11, 402 Search PubMed.
- D. Grischkowsky, S. Keiding, M. van Exter and C. Fattinger, J. Opt. Soc. Am. B, 1990, 7, 2006 CrossRef CAS.
- A. Deneuville, D. Tanner and P. H. Holloway, Phys. Rev. B, 1991, 43, 6544 CrossRef CAS PubMed.
- C. M. Randall and R. D. Rawcliffe, Appl. Opt., 1968, 7, 213 CrossRef CAS PubMed.
- M. T. Hibberd, V. Frey, B. F. Spencer, P. W. Mitchell, P. Dawson, M. J. Kappers, R. A. Oliver, C. J. Humphreys and D. M. Graham, Solid State Commun., 2016, 247, 68 CrossRef CAS.
- I. D. Vugmeyster, J. F. Whitaker and R. Merlin, Appl. Phys. Lett., 2012, 101, 181101 Search PubMed.
- M. Mumtaz, M. A. Mahmood, S. D. Khan, M. A. Zia, M. Ahmed and I. Ahmad, Opt. Mater., 2019, 96, 109357 Search PubMed.
- G. B. Netto and J. D. P. Siqueira, F. C. Da Cruz, in 2023 Int. Conf. Opt. MEMS Nanophotonics Sbfot. Int. Opt. Photonics Conf. (Sbfot. IOPC), IEEE, 2023, pp. 1–2 Search PubMed.
- A. K. Azad, J. Han and W. Zhang, Appl. Phys. Lett., 2006, 88, 021103 CrossRef.
- G. Zhang, G. Li, Z. Lan, L. Lin, A. Savateev, T. Heil, S. Zafeiratos, X. Wang and M. Antonietti, Angew. Chem., 2017, 129, 13630 CrossRef.
- J. Kröger, A. Jiménez-Solano, G. Savasci, V. W. h. Lau, V. Duppel, I. Moudrakovski, K. Küster, T. Scholz, A. Gouder, M. Schreiber, F. Podjaski, C. Ochsenfeld and B. V. Lotsch, Adv. Funct. Mater., 2021, 31, 2102468 CrossRef.
- P. C. Patra and Y. N. Mohapatra, Appl. Phys. Lett., 2021, 118, 103501 CrossRef CAS.
- T. Berlind, A. Furlan, Z. Czigany, J. Neidhardt, L. Hultman and H. Arwin, Thin Solid Films, 2009, 517, 6652 Search PubMed.
- A. Gouder, L. Yao, Y. Wang, F. Podjaski, K. S. Rabinovich, A. Jiménez-Solano and B. V. Lotsch, Adv. Energy Mater., 2023, 13, 2300245 CrossRef CAS.
- D. J. Woods, S. A. J. Hillman, D. Pearce, L. Wilbraham, L. Q. Flagg, W. Duffy, I. McCulloch, J. R. Durrant, A. A. Y. Guilbert, M. A. Zwijnenburg, R. S. Sprick, J. Nelson and A. I. Cooper, Energy Environ. Sci., 2020, 13, 1843 RSC.
- M. Sachs, R. S. Sprick, D. Pearce, S. A. J. Hillman, A. Monti, A. A. Y. Guilbert, N. J. Brownbill, S. Dimitrov, X. Shi, F. Blanc, M. A. Zwijnenburg, J. Nelson, J. R. Durrant and A. I. Cooper, Nat. Commun., 2018, 9, 4911 Search PubMed.
- J. Choi, W. Jung, S. Gonzalez-Carrero, J. R. Durrant, H. Cha and T. Park, Energy Environ. Sci., 2024, 17(21), 7999–8018 Search PubMed.
- M. J. Zouaoui, B. Nait-Ali, N. Glandut and D. S. Smith, J. Eur. Ceram. Soc., 2016, 36, 163 CrossRef CAS.
- Y. Wang, A. Vogel, M. Sachs, R. S. Sprick, L. Wilbraham, S. J. A. Moniz, R. Godin, M. A. Zwijnenburg, J. R. Durrant, A. I. Cooper and J. Tang, Nat. Energy, 2019, 4, 746 CrossRef CAS.
- S. Corby, R. R. Rao, L. Steier and J. R. Durrant, Nat. Rev. Mater., 2021, 6, 1136 CrossRef CAS.
- T. Takata, J. Jiang, Y. Sakata, M. Nakabayashi, N. Shibata, V. Nandal, K. Seki, T. Hisatomi and K. Domen, Nature, 2020, 581, 411 CrossRef CAS PubMed.
- R. Godin and J. Durrant, Chem. Soc. Rev., 2021, 50, 13372 RSC.
- J. Kosco, S. Gonzalez-Carrero, C. T. Howells, T. Fei, Y. Dong, R. Sougrat, G. T. Harrison, Y. Firdaus, R. Sheelamanthula and B. Purushothaman, Nat. Energy, 2022, 7, 340 CrossRef CAS.
- A. Senocrate, E. Kotomin and J. Maier, Helv. Chim. Acta, 2020, 103, e2000073 CrossRef CAS.
- F. Podjaski and B. V. Lotsch, Adv. Energy Mater., 2021, 11, 2003049 CrossRef CAS.
- V. Sridhar, F. Podjaski, Y. Alapan, J. Kröger, L. Grunenberg, V. Kishore, B. V. Lotsch and M. Sitti, Sci. Robot., 2024, 7, eabm1421 CrossRef.
- A. Rogolino and O. Savateev, Adv. Funct. Mater., 2023, 33, 2305028 Search PubMed.
- C. Wan, K. Xiao, A. Angelin, M. Antonietti and X. Chen, Adv. Intell. Syst., 2019, 1, 1900073 CrossRef.
- V. Druet, D. Ohayon, C. E. Petoukhoff, Y. Zhong, N. Alshehri, A. Koklu, P. D. Nayak, L. Salvigni, L. Almulla and J. Surgailis, Nat. Commun., 2023, 14, 5481 CrossRef CAS PubMed.
Footnote |
| † equally contributed. |
| This journal is © The Royal Society of Chemistry 2026 |