 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Transient interference-based spectroscopy for a molecular-bond-sensitive probe of lithium ion batteries
Mengyun
Hu†
abc,
Enlai
Wan†
ac,
Yu
Qiao
abc,
Shupeng
Xu
abc,
Shiwen
Li
ac,
Rui
Zhu
ac,
Jiamin
Guo
ac,
Xihao
Chen
 d and
Heping
Zeng†
d and
Heping
Zeng†
 *abc
*abc
aState Key Laboratory of Precision Spectroscopy, and Hainan Institute, East China Normal University, Shanghai 200062, China. E-mail: hpzeng@phy.ecnu.edu.cn
bChongqing Key Laboratory of Precision Optics, Chongqing Institute of East China Normal University, Chongqing 401120, China
cInstitute of Precision Optical Equipment for Marine Surveys (POEMS), Yazhou Bay Science and Technology City, Sanya 572000, Hainan, China
dSchool of Materials Science and Engineering, Chongqing University of Arts and Sciences, Chongqing 402160, China
First published on 8th April 2026
Abstract
The lack of effective methods to track in situ lithium-ion distribution and structural evolution remains a bottleneck in the battery industry, yet no techniques currently allow in situ dynamic detection of detrimental Li2O under realistic conditions. Here, a molecular-bond-sensitive probe technique is developed by leveraging transient interference-based spectroscopy (TIBS). By creating structured plasma channels through nonlinear interference of multiple femtosecond laser filaments, we achieve extreme spatial confinement of laser pulses, enabling peak intensities above 1014 W cm−2. This field directly ruptures molecular bonds through tunneling ionization and triggers Coulomb explosion, disintegrating materials into constituent elements and fragments while preserving molecular signature information. The resulting element-specific emissions allow intrinsic probing of the original chemical states and local environments. When coupled with machine learning algorithms, this technique enables precise identification of lithium concentration changes as low as 0.3%, making it particularly valuable for in situ monitoring of atomic-scale structural changes and distinguishing the Li2O side product from active materials.
Broader contextAdvancing next-generation batteries with higher energy density and longer lifespan is critical for the widespread electrification of transportation and grid-scale renewable energy storage. A fundamental obstacle in this pursuit is the inability to directly observe, in real time and under operating conditions, the intricate internal dynamics—such as lithium-ion migration and the formation of detrimental side products like Li2O—that ultimately govern battery performance and degradation. This lack of effective in operando diagnostics at the molecular level hinders the rational design of new materials and the optimization of device engineering. Here, we develop a novel molecular-bond-sensitive probe technique that overcomes this barrier. By enabling the precise, dynamic tracking of lithium distribution and the unambiguous identification of Li2O formation at the atomic scale, our method provides an unprecedented window into the “black box” of battery operation. This breakthrough not only offers a direct path to deconvolute complex degradation mechanisms but also establishes a generalizable platform for probing ultrafast chemical and structural evolution in a vast array of materials beyond energy storage, from catalysts to semiconductors. |
Introduction
The accelerating global transition towards carbon neutrality has positioned lithium-ion battery technology as a cornerstone of the future energy landscape,1–3 driving a profound transformation in energy utilization.4–7 Its applications have expanded beyond consumer electronics and electric vehicles to critical sectors such as aerospace and grid-scale energy storage.8–11 The overall performance and safety of a lithium-ion battery system are primarily dictated by the electrode material, including its chemical composition, crystal structure, surface morphology, and elemental distribution.12–14 However, we still lack in situ probes to track the microscopic movement and spatial distribution of lithium ions, or to capture the dynamic structural evolution of electrode materials, such as phase transitions and lattice rearrangements during lithium (de)intercalation. The continuous generation of Li2O permanently sequesters active lithium and electrolyte components, leading directly to irreversible capacity loss. To date, no techniques are able to effectively monitor Li2O nucleation, growth, distribution, or decomposition. Current mainstream analytical techniques for battery electrodes, such as X-ray diffraction,15 scanning electron microscopy,16 transmission electron microscopy,17 X-ray photoelectron spectroscopy,18 and Auger electron spectroscopy,19 face challenges in accurately capturing the distribution of light elements like lithium or tracking multi-scale structural evolution. These methods involve destructive sampling and complicated preparation procedures, rendering them unsuitable for industrial applications.While laser-induced breakdown spectroscopy (LIBS) has been widely used for elemental analysis and depth profiling in battery materials, it faces fundamental limitations in molecular-level characterization. Conventional laser ablation requires microsecond-scale plasma cooling periods, resulting in the loss of molecular bond specificity. Furthermore, its analytical sensitivity and quantitative accuracy are severely compromised by complex electrode matrix effects, making it particularly challenging to distinguish critical chemical states such as Li2O formation from active lithium compounds or to track elemental distribution with molecular signature preservation. Although femtosecond laser ablation could minimize plasma background fluorescence, the inherent plasma defocusing effect imposes a peak intensity limit of approximately 5 × 1013 W cm−2 in air—insufficient to significantly distort molecular potential barriers or induce observable bond breaking. The electron density in single filaments rarely exceeds 1017 cm−3,20 falling short of the threshold required to initiate detectable avalanche ionization of battery materials.
To overcome these fundamental constraints, we have developed a multi-filament interference approach, termed Transient Interference-Based Spectroscopy (TIBS), that creates transient plasma grating structures. This configuration enables substantial enhancement of peak laser intensity and electron density, achieving the necessary conditions for efficient molecular bond breaking through tunnelling ionization and Coulomb explosion processes. The resulting spectral signatures maintain molecular-level specificity while providing the sensitivity required for precise material characterization, particularly in complex electrochemical systems where traditional methods prove inadequate. When integrated with spectral reconstruction and backpropagation artificial neural networks, the method achieves accurate identification of molecular bonds across different concentration ranges. This advancement represents a significant step forward in analytical spectroscopy, bridging the gap between conventional elemental analysis and intrinsic molecular-level detection capabilities.
Results and discussion
The TIBS approach employs nonlinear interference of three high-intensity femtosecond laser filaments to establish plasma grating channels. As illustrated in Fig. 1(a), during femtosecond laser propagation in air, a dynamic balance between Kerr self-focusing and plasma defocusing effects generates stable femtosecond filaments.21 When two such filaments intersect, their interference creates periodic constructive fringes, where nonlinear self-focusing and plasma defocusing reach equilibrium at significantly enhanced peak intensities, forming a plasma grating22 with a modulation period of λ/[2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin(θ/2)], where λ represents the central laser wavelength of the femtosecond laser pulses and θ denotes the crossing angle between non-collinear filaments. In our experimental configuration with θ = 20°, this yields a plasma grating period of 2.3 µm, as shown in the photos in Fig. 1(a). Below the photo is the calculated spatial intensity distribution of the plasma channel. Notably, these plasma grating channels exhibit exceptional confinement characteristics with widths below 1 µm—significantly narrower than the ∼100 µm diameter typical of single filaments in air—enabling tight self-guiding of laser pulses. The introduction of a third femtosecond filament creates a scaly plasma grating structure that further confines pulses within channels approximately 10 µm long and <1 µm wide. This extreme confinement elevates the peak laser intensity, exceeding 1014 W cm−2,23 generating electric field strengths comparable to intramolecular fields. At these extreme field strengths, the laser–matter interaction proceeds through a cascade of ultrafast processes: initial tunneling ionization liberates electrons and ruptures molecular bonds; these freed electrons subsequently gain energy from the confined laser field, triggering avalanche ionization through collisions with atoms and molecular fragments. The resulting ensemble of highly charged positive ions after the instantaneous electron loss undergoes violent Coulomb repulsion—a Coulomb explosion—that disintegrates the molecular architecture into constituent species. In particular, these molecular species undergo repeated collisions with high-density electrons within the plasma grating channels, which initiate and sustain avalanche ionization for hundreds of nanoseconds post-irradiation, while their dynamic evolution preserves distinctive fingerprints of the original molecular structure. Different molecular bond excitations exhibit distinct characteristics and remain unaffected by matrix effects. Such laser-plasma synergistic excitations enable efficient molecular bond breaking and generate bond-sensitive spectra.
sin(θ/2)], where λ represents the central laser wavelength of the femtosecond laser pulses and θ denotes the crossing angle between non-collinear filaments. In our experimental configuration with θ = 20°, this yields a plasma grating period of 2.3 µm, as shown in the photos in Fig. 1(a). Below the photo is the calculated spatial intensity distribution of the plasma channel. Notably, these plasma grating channels exhibit exceptional confinement characteristics with widths below 1 µm—significantly narrower than the ∼100 µm diameter typical of single filaments in air—enabling tight self-guiding of laser pulses. The introduction of a third femtosecond filament creates a scaly plasma grating structure that further confines pulses within channels approximately 10 µm long and <1 µm wide. This extreme confinement elevates the peak laser intensity, exceeding 1014 W cm−2,23 generating electric field strengths comparable to intramolecular fields. At these extreme field strengths, the laser–matter interaction proceeds through a cascade of ultrafast processes: initial tunneling ionization liberates electrons and ruptures molecular bonds; these freed electrons subsequently gain energy from the confined laser field, triggering avalanche ionization through collisions with atoms and molecular fragments. The resulting ensemble of highly charged positive ions after the instantaneous electron loss undergoes violent Coulomb repulsion—a Coulomb explosion—that disintegrates the molecular architecture into constituent species. In particular, these molecular species undergo repeated collisions with high-density electrons within the plasma grating channels, which initiate and sustain avalanche ionization for hundreds of nanoseconds post-irradiation, while their dynamic evolution preserves distinctive fingerprints of the original molecular structure. Different molecular bond excitations exhibit distinct characteristics and remain unaffected by matrix effects. Such laser-plasma synergistic excitations enable efficient molecular bond breaking and generate bond-sensitive spectra.
During the interaction between laser and materials, inelastic collisions dominate the excitation process. Tunnel ionization induced by extreme electric fields is a field-driven, non-collisional process. Electrons undergo reverse bremsstrahlung radiation and inelastic collisions with atoms and ions to obtain energy from the laser field and trigger avalanche ionization. During the plasma expansion stage, inelastic collisions mainly involve electron collision excitation, resulting in characteristic emission spectral lines.
TIBS technology possesses depth profiling capabilities. It has sufficient thickness resolution, enabling single-pulse measurement and allowing for layer-by-layer analysis. Under conditions of low laser energy and a limited number of pulses, the surface of the material can be detected. By adjusting the pulse energy and the number of pulses, under the excitation of multiple femtosecond pulses, each laser pulse ablates a thin and localized volume, rather than just the outermost layer of the material. This means that the signal collected by each pulse represents chemical information from a specific depth range, rather than just information from the outermost surface. By accumulating consecutive laser pulses, TIBS is able to detect from the electrode surface to the interior of the material, thus sequentially acquiring depth-resolved spectroscopic information. Therefore, TIBS technology enables the monitoring of compositional evolution along the thickness direction of the electrode, which is crucial for characterizing non-uniform delithiation and interfacial side reactions.
Considering the lithium-ion distribution characteristics of batteries during charging and discharging processes, we employed Vesta software to simulate structural changes in the LiCoO2 cathode material at different lithium concentrations.24 As shown in Fig. 1(b), atomic-scale analysis reveals noticeable changes in Co–O and Li–O bond lengths with varying lithium-ion concentration. In LiCoO2 cathode materials with different atomic ratios, the lengths of the Co–O bond are 1.90, 1.92, and 1.94 Å, and those of Li–O bond are 2.19, 2.13, and 2.11 Å, respectively. The specific atomic ratios are marked in Fig. 1(b). The results indicate that lithium-ion concentration directly influences the local bonding environment, inducing subtle adjustments in the electrode material structure. Therefore, developing high-precision quantitative analysis methods can facilitate the accurate identification of atomic-scale structural features and provide new pathways for highly sensitive diagnosis of battery states. TIBS technology, with its unique mechanism of breaking chemical bonds, is highly suitable for detecting differences in chemical bonds.
Lithium-ion battery technology utilizes various cathode material systems, with prominent examples including LiFePO4, LiCoO2, LiMn2O4, and LiNiCoAlO2. We selected olivine-structured LiFePO4 and layered LiCoO2 as the representative cathode materials to assess TIBS performance for different chemical structures and elemental compositions. Intense femtosecond laser pulses were directed to interact with the electrode materials under ambient atmospheric conditions. Laser-induced emissions were analyzed using an on-chip integration mode of an ICCD camera (iStar, Andor Technology) integrated with a spectrometer (Mechelle 5000, Andor Technology). The transient dynamics of plasma evolution were resolved by varying the detection delay time while maintaining a fixed exposure duration of 0.2 seconds per acquisition. Each measurement accumulated 200 consecutive spectra to enhance the signal-to-noise ratio (SNR). TIBS technology does not require a specific gas environment. All measurements were performed in ambient air. Fig. 2(a) and (b) present the time-resolved spectra of LiFePO4 and LiCoO2, revealing distinct temporal characteristics in their plasma emission behavior. At zero delay time, the immediate laser–matter interaction, dominated by tunneling ionization and molecular bond rupture, produces the highest spectral line intensities. TIBS can generate atomic emissions from Li, Fe, and Co simultaneously—the process liberates all atomic species from the lattice in one explosive event, allowing for their simultaneous detection. Crucially, the absence of significant plasma background interference enables clear resolution of element-specific emissions—a notable advantage over nanosecond LIBS, where strong plasma fluorescence typically masks early-stage spectral features. As shown in Fig. 2(c) and (d), the temporal profiles of characteristic peak intensity and SNR reveal element-specific decay features. The Fe and Co spectral lines exhibit rapid decay, diminishing to near-background levels after 300 ns, while the Li signal remains detectable with up to 600 ns delay. These decays exhibit much longer equivalent lifetimes than the corresponding electronic emissions, indicating that these excited species experience significant impact excitations due to collision with high-density electrons in the plasma channels.
Across all delays, the Li signal exhibits consistently higher intensity than both Fe and Co. During laser interaction with both electrode materials, Li+ ions are primarily ejected during Coulomb explosion, and their collision with electrons are enhanced within the plasma grating channels. Fe or Co ions were excited quite differently during the laser interaction period. In olivine-structured LiFePO4, the Fe2+ ion has less tightly bound electrons compared to the Li+ ion. The highly localized Fe–O bonds are broken first by the direct removal of electrons, and the Li–O bonds are broken during the resulting Coulomb explosion, while the covalently bonded phosphate framework behaves as a rigid disintegrating unit during Coulomb explosion. This was validated by the fact that negligible P emission lines were observed in LiFePO4. In layered LiCoO2, the laser field preferentially removes electrons from the cobalt and oxygen ions in the covalent-ionic network of the CoO2 layer. The Co–O bonds are broken first by the direct removal of electrons, triggering a Coulomb explosion that dismantles the entire structure, including the ionic Li sites. All the excitations preserve the molecular bond signatures, whose characteristic features—including wavelength, intensity, line width, and temporal evolution—provide critical structural fingerprinting information for the electrode materials. An optimal acquisition delay was set at 300 ns for LiFePO4 and 200 ns for LiCoO2, providing maximum sensitivity for their respective elemental signatures while maintaining excellent spectral quality for discrimination of different material states.
The spectral performance was compared for three different excitation methods: single femtosecond filaments, plasma gratings, and the TIBS configuration. As demonstrated by the characteristic Li I 610.4 nm emission line shown in Fig. 2(e) and (f), the spectral intensities obtained through these three approaches show remarkable differences. The TIBS technique yields the strongest spectral signal, with intensity approximately four times greater than that achieved with single-filament excitation and about double that obtained through plasma-grating excitation. This confirms that TIBS plasma grating channels had enhanced laser electric fields and elevated electron density, thus giving the strongest molecular rupture and explosion for precise differentiation of material compositions across diverse concentration ranges, structural configurations, and chemical states.
As shown in Fig. 3(a) and (b), LiFePO4 and LiCoO2 dominate, with Li, Fe, and Co characteristic peaks.25–27 As the electrode materials are coated onto an aluminum foil substrate, characteristic aluminum signals are detected in both spectra. It is noteworthy that TIBS provides powerful bond-breaking specificity to distinguish the detrimental side product Li2O28–30 from other active lithium compounds. Fig. 3(c–f) present spectra obtained under the TIBS configuration for the mixture of LiFePO4 and LiCoO2, and for Li2O, Fe2O3, and CoO, respectively. The bond-sensitive nature of TIBS was evidenced by the strong Li I emission lines at 460.3 and 497.2 nm from Li2O, which were much stronger than those from LiFePO4 and LiCoO2. The bond rupture in Li2O is initiated by the direct ionization of the oxygen anion, leading to an instantaneous Coulomb explosion that violently disassembles Li2O into its atomic ions, allowing for precise detection and differentiation from other lithium compounds, which, as aforementioned, undergo quite different bond rupture and Coulomb explosion.
As a battery electrode comprises composite materials, such as a blend of LiFePO4 or LiCoO2, conductive carbon, and a binder, the illuminating intense femtosecond laser pulse interacts with all components, and those with lower ionization thresholds or more localized electrons ionize more readily. Each distinct chemical phase in the mixture undergoes its own localized Coulomb explosion. In the TIBS configuration, intensified bond rupture and compound-specific Coulomb explosions produce observable atomic and molecular fragments of each individual component, which are ejected into plasma grating channels of enhanced electron density, resulting in dramatic enhancement of impact excitation, emitting a composite spectrum that contains quantifiable information about each component. When a composite battery electrode is subjected to an intense femtosecond laser pulse, as shown in Fig. 3(c), elemental signals for Li, Co, and Fe are detected simultaneously. The corresponding oxides show characteristic peaks identical to those in the electrodes, indicating that mechanical mixing and high-energy ball milling—key precursor processing steps in electrode material synthesis—could partially replicate the physicochemical properties of electrode materials, resulting in spectral consistency.
Subsequently, guided by the ion migration behavior during the charging and discharging process, we conducted a quantitative analysis of the lithium battery electrode materials. First, we analyzed standard pellet samples of Li2O, Fe2O3, and CoO with different concentrations. For each concentration, ten sets of spectra were collected under identical conditions. The average spectral line intensity served as the quantitative basis for establishing calibration curves for Li, Fe, and Co, as shown in Fig. 4(a–c). Each calibration curve exhibits good linearity, with R2 values of 0.98 (Li), 0.98 (Fe), and 0.96 (Co), respectively. To simulate the complex component environment of actual electrode materials, mixtures of LiFePO4 and LiCoO2 were used as detection targets. Samples of different concentrations were prepared through standardized sample preparation procedures. Across different concentrations, the corresponding structural differences were successfully captured, as shown in Fig. 4(d–f). The characteristic spectral lines for each element show discernible changes across different compositions. Using lithium as an example, the main peak positions for the three concentrations are 610.20, 610.08, and 610.19 nm, with full width at half-maximum (FWHM) values of 0.95, 0.94, and 0.60 nm, respectively. This indicates that the characteristic spectral lines are profoundly sensitive reporters of the local chemical and physical environment. There exists a very close correlation between spectral features and chemical structures, demonstrating that the discernible changes in spectral intensity, peak wavelength, line width, and spectral shape could be used to monitor atomic-scale structural differences.
Quantitative analysis was then performed on the mixed electrode materials. After standardized sample preparation via ball milling and pelletizing, calibration curves were established. As shown in Fig. 4(g–i), the spectral line intensities of Li, Fe, and Co maintain a good linear relationship with mass fractions, with R2 values of 0.93 (Li), 0.95 (Fe), and 0.94 (Co), meeting quantitative analysis requirements, although the linearity is slightly inferior to that of the oxide standard samples. This difference primarily stems from the complex composition of the actual electrode materials: beyond the active materials LiFePO4 and LiCoO2, inactive components such as binders and conductive agents interfere with the plasma excitation process, affecting the quality of the calibration curves. Additionally, both sample types exhibit relatively large error ranges in their calibration curves.
The observed spectra contain discernible features, including spectral intensities, central wavelengths, and temporal evolutions, which reveal the physicochemical signatures of concentration, chemical states, and fragment dynamics, respectively. The entire rupture of the mixture, from the microscopic bond breaking and Coulomb explosion to the impact excitation in the macroscopic plasma grating structures, is encoded into the complex spectrum, which could be set as a high-dimensional fingerprinting pattern for machine-learning to decode the intrinsic molecular structure information.
The observed spectral lines were then processed by means of the so-called spectral reconstruction (SR) method.31 This method establishes projection operations based on the intrinsic spectral characteristics, utilizing time-frequency features to map the observed data onto a low-dimensional feature subspace, thereby enhancing source feature identifiability while effectively suppressing noise. Using the highest concentration sample as the reference spectrum, spectral reconstruction was performed on the other five concentration samples. Fig. 5(a) compares the original and reconstructed spectra, showing significantly reduced background noise and more prominent signal features. When the reconstructed spectra were used for quantitative modeling, the resulting calibration curve exhibited markedly improved linearity, with the R2 value increasing to 0.99, and the error margins were substantially narrowed, as shown in Fig. 5(b). Furthermore, based on the reconstructed spectral data, a backpropagation artificial neural network (BP-ANN) model was developed to identify electrode materials with different lithium concentrations. This model computes outputs through forward propagation and utilizes an error backpropagation mechanism with a gradient descent algorithm to iteratively adjust network weights and biases layer by layer, minimizing prediction error and achieving high-precision fitting of complex nonlinear mapping relationships. Given that lithium characteristic peaks vary in position, FWHM, and intensity across concentrations, the 604–617 nm wavelength range (comprising 362 data points), which encompasses the complete spectral profile, was selected as the input feature. The dataset includes five concentrations, with 40 spectra per concentration; 70% was allocated as the training set and 30% as the test set. The prediction results on the test set are shown in Fig. 5(c). If the test value is consistent with the true value, the recognition is correct, as shown in the dark purple square in the figure. The confusion matrix indicates that a total of 60 sets of data were used for validation, of which 56 were correctly identified and only four were incorrectly identified, yielding an identification accuracy of 93.3%. To assess model stability, ten independent replicate experiments were conducted, resulting in an average accuracy of approximately 94.7%, demonstrating good consistency, as shown in Fig. 5(d). TIBS reinforced by machine learning algorithms enables in situ and real-time monitoring of the microscopic chemical dynamics during the charging and discharging processes, capable of accurately detecting concentration changes as low as 0.3%. The successful recognition of different concentrations is equivalent to the identification of different bond lengths and atomic-scale structural differences.
The spectroscopic signature of the battery electrode is fundamentally determined by its local chemical environment and bonding, which could be mimicked through different processing routes. Specifically, mechanical mixing and high-energy ball milling could activate solid-state reactions of powder mixtures, such as Li2O, Fe2O3, and CoO, to form compounds with requisite stoichiometries, mimicking chemical environments of a real complex electrode. Moreover, mechanical processing continually fractures particles and alters the physicochemical states, creating surface defects or disordered states that can replicate the local bonding environments found in materials that have been cycled in a battery operation. These processing steps effectively create model systems that allow for more accurate machine learning model training.
Conclusions
In summary, we have demonstrated the bond-sensitive nature of TIBS that provides a powerful tool for state recognition and in situ performance monitoring of battery electrodes. Experimental results demonstrate that compared to femtosecond filament excitation, TIBS enhances the characteristic spectral line intensity by approximately fourfold while effectively suppressing background noise. Quantitative analysis of electrode samples with precisely controlled lithium content successfully captured spectral differences corresponding to distinct structures, indicating the method's capability to identify atomic-scale structural variations and validating the exceptional linear response and high-precision compositional analysis of TIBS for element identification. Further application of spectral reconstruction methods to process the original spectra improved the calibration curve fitting coefficient to 0.99 and significantly reduced error ranges. Integrated with machine learning algorithms, the approach achieved precise identification of concentration differences as low as 0.3%, with an identification accuracy of 94.7%. By integrating theoretical modeling, experimental diagnostics, and machine learning, this study proposes a rapid, high-precision comprehensive detection method for lithium battery electrode materials. This method not only enables quality and safety control of battery components but also facilitates rapid monitoring of lithium battery performance and identification of battery health status. Furthermore, it provides atomic-scale structural analysis capabilities, delivering key technical support for the development of next-generation energy storage systems.TIBS technology is not only applicable to the Li2O, LiFePO4 and LiCoO2 used here, but also usually applicable to various materials, including soft materials, organic components, polymers, etc. TIBS is a universal approach that can detect any element in principle. TIBS can analyze substances in almost all states (solid, gas, liquid). For batteries, it is very suitable for detecting various forms of samples such as thin films, thick electrodes, and powders (pressed into tablets), etc. The TIBS technique proposed in this work, with its unique bond-sensitive characteristics, is applicable to more complex cathode materials involving anionic redox reactions.
Methods
Experiment
The experimental setup is shown in Fig. 6. The light source used for laser ablation was a Ti:sapphire regenerative laser (Coherent Inc.) operated at a fundamental wavelength of 800 nm. It delivered 50 fs pulses at 1 kHz repetition rate, with a maximum single-pulse energy of 3 mJ. The laser beam first passed through two beam splitters to split into three beams of equal energy. Each beam passed through a focusing lens (f = 200 mm) to form a filament. Stepper motors were used to regulate the spatiotemporal synchronization of the three filaments. This ensured the formation of periodic plasma channels—plasma gratings. Unlike traditional pulsed lasers, this plasma grating was used to excite samples and generate plasma. The electrode material was fixed on an electric displacement platform, and excitation at different positions of the electrode material was achieved by adjusting the position of the displacement platform through a controller. The fluorescence emitted by the plasma was collected and focused onto an optical fiber via two lenses (f = 30 mm), which was connected to an Echelle spectrometer (Mechelle 5000, Andor Technology). The spectrometer covered the spectral range of 200–850 nm with a resolution of 0.1 nm and was equipped with an intensified charge coupled device (ICCD) detector.Differences in sample surface and volume do not affect the detection capability of TIBS. The minimum sample thickness required for reliable analysis of the current experimental setup can reach the micrometer level, and detecting thinner samples requires further optimization. There is no upper thickness limit, so long as the sampling space allows it. The area for signal extraction is controllable. In this experiment, the distance from the sample to the detection system is 12 mm. The signal is collected by two short-focus lenses (f = 30 mm) positioned at a 45° angle above the laser focal spot. The distance between the two lenses is twice the focal length, which can be adjusted as needed.
Sample preparations
When conducting quantitative analysis, Li2O, Fe2O3, and CoO were initially used as the standard materials. These oxides were added to a 1.0 g standard soil matrix in increasing amounts: 1.0, 1.5, 2.0, 2.5, 3.0, and 3.5 g. The mass fractions of Li, Fe, and Co were then calculated for each concentration. After homogenizing the mixture through planetary ball milling, standardized tablets were prepared for TIBS analysis. Then, to simulate the complex characteristics of electrode materials in actual detection scenarios, mixed samples of LiCoO2 and LiFePO4 were prepared. Six sets of mixed samples with different mass ratios (total mass is 0.2 g) were designed. Standardized tablets were prepared using the same operating procedure for experimentation.Spectral reconstruction method
This method comprises two main components: the establishment of a mapping matrix from standard spectra and the reconstruction of measured spectra based on this matrix. The mapping matrix is derived by projecting standard spectra onto a sinusoidal spectral basis. The mapping matrix Em×n containing the positional transformation information is obtained by choosing the nearest “neighbor” rule from the set standard sine spectrum. The reconstructed spectrum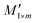 in this method is calculated by:
in this method is calculated by:| R1×n = M1×n·Em×n | (1) |
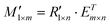 | (2) |
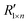 is obtained after fitting the mapping spectrum R1×n.
is obtained after fitting the mapping spectrum R1×n. 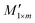 is the reconstructed spectrum obtained through the transpose matrix
is the reconstructed spectrum obtained through the transpose matrix 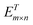 of the mapping matrix Em×n.
of the mapping matrix Em×n.
Backpropagation artificial neural network
BP-ANN is a multi-layer feedforward network based on the gradient descent principle. It computes outputs through forward propagation and subsequently adjusts network parameters by backpropagating prediction errors. In classification tasks, input data undergo nonlinear transformations through hidden layers, ultimately generating class probability distributions at the output layer. During training, the system calculates error gradients by comparing predictions with ground-truth labels. These gradients are propagated backward through the network to iteratively update weights and biases, enabling the model to progressively approximate an optimal mapping relationship and achieve accurate classification. Here, given that lithium characteristic peaks vary in position, FWHM, and intensity across concentrations, the 604–617 nm wavelength range (comprising 362 data points), which encompasses the complete spectral profile, was selected as the input feature. By associating different spectral features with different battery states, high-precision state recognition of lithium batteries can be achieved.Author contributions
M. Hu contributed to methodology, investigation, resources, project administration, supervision, and writing – review & editing. E. Wan contributed to methodology, investigation, visualization, and writing. Y. Qiao contributed to methodology, investigation, and resources. S. Xu contributed to methodology and investigation. S. Li contributed to methodology and investigation. R. Zhu contributed to methodology and investigation. J. Guo contributed to methodology. X. Chen contributed to methodology. H. Zeng contributed to methodology, investigation, funding acquisition, project administration, supervision, writing – original draft, and writing – review & editing. All authors discussed the results and reviewed the manuscript.Conflicts of interest
All authors disclosed no financial/commercial conflict of interest.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request. Correspondence and requests for materials should be addressed to H. Zeng.Acknowledgements
This work was supported by Quantum Science and Technology-National Science and Technology Major Project (2023ZD0301000); National Natural Science Foundation of China (12404360); Hainan Province Science and Technology Special Fund (ZDYF2025GXJS012); National Key Laboratory on Electromagnetic Environmental Effects and Electro-optical Engineering (No. JCKYS2024LD1); Chongqing Special Major Project for Technological Innovation and Application Development (CSTB2025TIAD-STX0024); New Chongqing Youth Innovation Talent Program (CSTB2025YITP-QCRCX0029); Research Project of Shanghai Science and Technology Commission (22560730400).Notes and references
- E. Pomerantseva, et al., Energy storage: The future enabled by nanomaterials, Science, 2019, 366, eaan8285 CrossRef CAS PubMed.
- J. Q. He, et al., Scalable production of high-performing woven lithium-ion fibre batteries, Nature, 2021, 597, 57–63 CrossRef CAS PubMed.
- J. Xiao, et al., From laboratory innovations to materials manufacturing for lithium-based batteries, Nat. Energy, 2023, 8, 329–339 CrossRef.
- Y. Chen, et al., Advances in Lithium-Sulfur Batteries: From Academic Research to Commercial Viability, Adv. Mater., 2021, 33, 2003666 CrossRef CAS PubMed.
- G. Offer, et al., Cool metric for lithium-ion batteries could spur progress, Nature, 2020, 582, 485–487 CrossRef CAS PubMed.
- J. M. Turner, The matter of a clean energy future, Science, 2022, 376, 1361 CrossRef.
- C. Yuan, Sustainable battery manufacturing in the future, Nat. Energy, 2023, 8, 1180–1181 CrossRef.
- S. T. Han, et al., Sequencing polymers to enable solid-state lithium batteries, Nat. Mater., 2023, 22, 1515–1522 CrossRef CAS PubMed.
- G. X. Lu, et al., High-voltage electrosynthesis of organic–inorganic hybrid with ultrahigh fluorine content toward fast Li-ion transport, Sci. Adv., 2023, 10, eado7348 CrossRef PubMed.
- S. Kalnaus, et al., Solid-state batteries: The critical role of mechanics, Science, 2023, 381, eabg5998 CrossRef CAS PubMed.
- Z. Y. Wang, et al., Lithium anode interlayer design for all-solid-state lithium-metal batteries, Nat. Energy, 2024, 9, 251–262 CrossRef CAS.
- S. Kim, et al., A complex hydride lithium superionic conductor for high-energy-density all-solid-state lithium metal batteries, Nat. Commun., 2019, 10, 1081 CrossRef PubMed.
- Y. N. Sun, et al., Solvent co-intercalation in layered cathode active materials for sodium-ion batteries, Nat. Mater., 2025, 24(205), 1441–1449 CrossRef CAS PubMed.
- Y. N. Gao, et al., Design lithium exchanged zeolite based multifunctional electrode additive for ultra-high loading electrode toward high energy density lithium metal battery, Adv. Energy Mater., 2024, 15, 231703 Search PubMed.
- D. W. Choi, et al., Thermal stability and phase transformation of electrochemically charged/discharged LiMnPO4 cathode for Li-ion batteries, Energy Environ. Sci., 2011, 4, 4560–4566 RSC.
- K. J. Hayworth, et al., Gas cluster ion beam SEM for imaging of large tissue samples with 10 nm isotropic resolution, Nat. Methods, 2020, 17, 68–71 CrossRef CAS PubMed.
- Z. F. Huang, et al., Grain rotation and lattice deformation during photoinduced chemical reactions revealed by in situ X-ray nanodiffraction, Nat. Mater., 2015, 14, 691–695 CrossRef CAS PubMed.
- G. Greczynski, et al., X-ray photoelectron spectroscopy of thin films, Nat. Rev. Methods Primers, 2023, 3, 40 CrossRef CAS.
- A. Hans, et al., Experimental realization of Auger decay in the field of a positive elementary charge, Phys. Rev. Lett., 2024, 132, 203002 CrossRef CAS PubMed.
- S. Eisenmann, A. Pukhov and A. Zigler, Fine structure of a laser-plasma filament in air, Phys. Rev. Lett., 2007, 98, 155002 CrossRef PubMed.
- J. H. Odhner, D. A. Romanov and R. J. Levis, Rovibrational wave-packet dispersion during femtosecond laser filamentation in air, Phys. Rev. Lett., 2009, 103, 075005 Search PubMed.
- M. Y. Hu, et al., Plasma-grating-induced breakdown spectroscopy, Adv. Photon., 2020, 2, 065001 CAS.
- A. Couairon and A. Mysyrowicz, Femtosecond filamentation in transparent media, Phys. Rep., 2007, 441, 47–189 CrossRef CAS.
- K. Momma and F. Izumi, VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data, J. Appl. Crystallogr., 2011, 44, 1272–1276 CrossRef CAS.
- S. Imashuku, et al., Quantitative lithium mapping of lithium-ion battery cathode using laser- induced breakdown spectroscopy, J. Power Sources, 2018, 399, 186–191 CrossRef CAS.
- L. Yang, et al., The detection of elemental content changes in electrode coating using laser-induced breakdown spectroscopy, Spectrochim. Acta, Part B, 2024, 214, 106896 CrossRef CAS.
- R. Pamu, et al., Calibration-Free quantitative analysis of lithium-ion battery (LiB) electrode materials using laser-induced breakdown spectroscopy (LIBS), ACS Appl. Energy Mater., 2021, 4, 7259–7267 CrossRef CAS.
- T. C. Liu, et al., In situ quantification of interphasial chemistry in Li-ion battery, Nat. Nanotechnol., 2019, 14, 50–56 CrossRef CAS PubMed.
- Z. A. Yu, Y. Cui and Z. N. Bao, Design principles of artificial solid electrolyte interphases for lithium-metal anodes, Cell Rep. Phys. Sci., 2020, 1, 100119 CrossRef.
- M. Asadi, et al., A lithium-oxygen battery with a long cycle life in an air-like atmosphere, Nature, 2018, 555, 502–506 CrossRef CAS PubMed.
- R. Zhu, et al., Ppb-Level ammonia sensor for exhaled breath diagnosis based on UV-DOAS combined with spectral reconstruction fitting neural network, ACS Sens., 2024, 9, 4286–4294 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |






