Open Access Article
Open Access ArticleBoron nitride nanotubes for next-generation battery systems
Thang Quoc Huynh *a and
Tan-Lien Pham*b
*a and
Tan-Lien Pham*b
aDepartment of Chemical Engineering, Pohang University of Science and Technology, Pohang, 37673 Republic of Korea. E-mail: quocthangdr@postech.ac.kr
bDepartment of Chemistry, College of Natural Science, Ulsan National Institute of Science and Technology, Ulsan, 44919, Republic of Korea. E-mail: lienpham@unist.ac.kr
First published on 20th April 2026
Abstract
Despite possessing an exceptional combination of electrical insulation, ultrahigh thermal conductivity, and mechanical stiffness, boron nitride nanotubes (BNNTs) have remained largely peripheral to battery research. To date, only a limited number of studies have demonstrated BNNT integration in electrochemical systems, often treating them as passive fillers rather than functional components. Here, we argue that this narrow view obscures the true potential of BNNTs. Owing to their one-dimensional geometry and intrinsically polarized B–N lattice, BNNTs operate as active interfacial regulators capable of directing ion transport, redistributing mechanical stress, and homogenizing thermal gradients. By reframing BNNTs as architectural elements that stabilize electrochemical interfaces under coupled ionic, mechanical, and thermal fields, this work establishes a new design paradigm for robust and safe next-generation energy storage systems.
Broader contextElectrochemical energy storage is central to renewable energy integration and electrified transportation, yet progress is increasingly limited by safety, lifetime, and thermal management challenges. Most nanomaterial strategies in batteries target isolated functions, such as mechanical reinforcement or ionic conductivity, often neglecting their coupled thermal and interfacial effects. Boron nitride nanotubes (BNNTs) offer a distinct opportunity as electrically insulating yet highly thermally conductive nanomaterials that can simultaneously regulate ion transport, reinforce mechanically fragile interfaces, and dissipate localized heat. This multifunctional behavior enables a shift from component-level materials optimization toward integrated, system-level battery design. The concepts demonstrated here are broadly applicable across battery chemistries and operating conditions, providing a general framework for safer, longer-lasting, and more sustainable energy storage technologies. |
1. Reframing BNNTs through the lens of battery instability
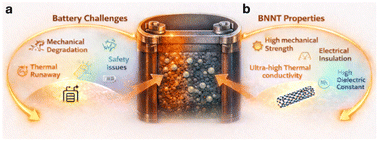
Electrochemical energy storage is dominated by failure modes that arise not from the lack of active-material capacity, but from the inability to stabilize interfaces under mechanical, thermal, chemical, and ionic stress (Fig. 1a). Lithium metal suffers from uncontrolled electrodeposition and dendrite formation. High-voltage cathodes undergo parasitic oxygen release, surface reconstruction, and electrolyte oxidation. Polymer and solid electrolytes fracture or deform under cycling. Nearly all these problems stem from field gradients – gradients of ion concentration, temperature, stress, and electron density that self-amplify during operation.1–3The most striking aspect of boron nitride nanotubes (BNNTs) is how precisely their intrinsic properties map onto these field instabilities. BNNTs possess a near-theoretical axial modulus (∼1 TPa), a wide bandgap (∼5–6 eV) that prevents electron leakage, a thermal conductivity that rivals or exceeds that of carbon nanotubes, and an exceptional resistance to oxidation and chemical decomposition across a broad voltage window (Fig. 1b). Few materials combine these attributes so naturally.2,4 To more clearly contextualize these properties, it is instructive to compare BNNTs quantitatively with other nanofillers commonly explored in battery systems, including carbon nanotubes (CNTs), hexagonal boron nitride (h-BN), MXenes, transition metal dichalcogenide nanotubes (e.g., MoS2), and conventional oxide particles (Table 1). While CNTs share comparable mechanical stiffness and even higher thermal conductivity, their metallic or semimetallic nature introduces continuous electron pathways that can accelerate parasitic reactions and increase short-circuit risks.5 In contrast, MXenes offer high electrical conductivity and tunable surface chemistry but suffer from oxidative instability and similarly promote undesired electronic leakage.6,7 Two-dimensional h-BN provides excellent thermal conductivity and electrical insulation, yet lacks the one-dimensional geometry required for directional stress transfer and guided transport.8 It is important to note that the advantages of BNNTs are not simply a result of increased aspect ratio compared to 2D h-BN. Their one-dimensional cylindrical geometry enables the formation of continuous, anisotropic networks with lower percolation thresholds and more effective interfacial connectivity. In contrast, h-BN typically forms stacked or randomly oriented platelets with limited directional transport. This geometric distinction underpins the unique functionality of BNNTs as interfacial regulators. MoS2 nanotubes present structural similarity to BNNTs but exhibit significantly lower mechanical stiffness and thermal transport capability, limiting their effectiveness as multifunctional regulators.9 Conventional oxide fillers such as Al2O3 or SiO2, while chemically stable, typically possess low aspect ratios and limited thermal conductivity, restricting their ability to simultaneously address multiple failure modes.10,11 Within this broader materials landscape, BNNTs uniquely integrate high mechanical strength, efficient thermal transport, and electrical insulation into a one-dimensional architecture. This combination enables them to interact with electrochemical systems not merely as passive additives, but as structural elements capable of modulating coupled ionic, thermal, and mechanical fields. As such, BNNTs are more appropriately viewed as interfacial regulators rather than conventional filler materials.
| Material | Young's modulus (TPa) | Thermal conductivity (W m−1 K−1) | Electrical property | Key limitation in batteries |
|---|---|---|---|---|
| BNNT | ∼1–1.2 | ∼200–300 | Insulator (∼5–6 eV bandgap) | Limited availability, cost |
| CNT | ∼1 | ∼300–3000 | Highly conductive | Promotes side reactions, short-circuit risk |
| h-BN (2D) | ∼0.8 | ∼200–400 (in-plane) | Insulator | Lacks 1D transport pathways |
| MXene (Ti3C2Tx) | ∼0.3–0.5 | ∼50–150 | Highly conductive | Oxidation instability, parasitic reactions |
| MoS2 nanotube | ∼0.2–0.3 | ∼30–80 | Semiconducting | Lower mechanical/thermal performance |
| Al2O3/SiO2 | ∼100–300 GPa | <50 | Insulator | Low aspect ratio, weak multifunctionality |
Recent studies further reinforce this reframing of BNNTs as interfacial stability regulators rather than passive fillers. Kim et al. demonstrated that incorporating purified BNNTs onto polypropylene (PP) separators in Li–S batteries dramatically suppresses polysulfide shuttling, homogenizes ion transport, and stabilizes lithium metal deposition, all achieved without modifying cathode chemistry. Their purified BNNTs (p-BNNT) form a three-dimensional spider net-like coating with partial ionic character originating from the B–N lattice, which increases ionic conductivity, enhances Li+ diffusivity, and creates a physical-electrostatic barrier to polysulfide migration. As a result, the p-BNNT separator delivered a high discharge capacity of 1429 mAh g−1 and maintained stable cycling for more than 200 cycles, significantly outperforming the conventional PP separator. These improvements arise from reduced dendrite formation, improved electrolyte wettability, and controlled Li+ flux enabled by the BNNT network's random stacking and polarized surface. This study illustrates a fundamental paradigm shift: a material that is electrically insulating and nominally electrochemically “silent” can still reshape battery behavior by governing fields, transport, and interfacial reactions at the separator-electrode boundary.12
Similarly, He and co-workers developed a multifunctional composite solid electrolyte (CSE) by incorporating BNNTs into a polyvinylidene fluoride-hexafluoropropylene (PVDF-HFP) matrix, achieving a room-temperature ionic conductivity of 8.0 × 10−4 S cm−1 – approximately three times that of the pristine PVDF-HFP (2.7 × 10−4 S cm−1) – along with a Li+ transference number of 0.60. This improvement arises not from direct ion conduction through the BNNTs but from their high-aspect-ratio structure, which promotes lithium salt dissociation, reduces polymer crystallinity, enhances segmental motion, and establishes long-range Li+ transport pathways, thereby optimizing the ionic microenvironment. Additionally, BNNTs impart superior thermal conductivity (in-plane: 4.0 W m−1 K−1; out-of-plane: 0.12 W m−1 K−1) and flame retardancy, with a reduced peak heat release rate of 30.1 W g−1 compared to 36.4 W g−1 for pristine PVDF-HFP. The CSE also forms a thin, stable solid electrolyte interphase (SEI) layer rich in Li3N, Li2S, and LiF at the lithium anode interface, suppressing dendrite growth. Electrochemical testing demonstrated exceptional stability: Li//Li symmetric cells cycled for 3400 h at 0.2 mA cm−2 and 25 °C without short-circuiting, LiFePO4/Li cells retained 93.2% capacity after 850 cycles at 0.5C (and 96.8% after 300 cycles at 1C), and high-voltage LiCoO2/Li cells achieved 96.4% retention after 400 cycles at 1C. Pouch cells based on this CSE exhibited robust safety, maintaining functionality under mechanical abuse such as puncturing and cutting. Overall, this work highlights BNNTs as effective fillers for advancing flexible, safe, and high-performance solid polymer electrolytes in all-solid-state lithium metal batteries.13
Yadav et al. dispersed BNNTs into conventional liquid Li-ion battery electrolytes (1 M LiPF6 in EC/DMC) at <1 wt% loading, observing enhanced ionic conductivity, higher coulombic efficiency, and reduced parasitic reactions through electrode–electrolyte interface modulation. At optimal 0.9 wt%, BNNTs’ Lewis acid sites promoted Li+ desolvation, anion immobilization at defects, and exo-/endohedral ion transport, yielding a 30% conductivity boost (0.87 mS cm−1 from 0.61 mS cm−1) and a Li+ transference number of 0.73 (from 0.49). In NCM622//graphite full cells, this enabled reversible capacities of 153 mAh g−1 at 1C (vs. 125 mAh g−1 without), superior rate performance across −10 to 60 °C (e.g., 88.6 mAh g−1 at 0.5C/−10 °C vs. 63.3 mAh g−1), and 71.5 mAh g−1 with 99.6% coulombic efficiency over 500 cycles at 10C (vs. 40 mAh g−1, 97.5%). Further benefits include uniform SEI formation, reduced separator thermal shrinkage (∼4.2% vs. 10.5%), and compatibility with Li metal anodes and LCO//graphite pouch cells (97% retention after 300 cycles at 0.5C), highlighting BNNTs’ multifunctionality for safer, high-performance LIBs.14
Despite the broad set of attractive physicochemical properties that BNNTs possess, their use as electrode materials remains extremely limited. In contrast to carbon nanotubes, which are widely adopted as conductive scaffolds in both anodes and cathodes, BNNTs have not been established as electroactive or electronically percolating components, resulting in very few reports exploring them within electrode architectures. To date, one of the only studies demonstrating BNNT-containing battery electrodes is the work of Abass et al., who incorporated trace amounts of BNNTs (0.25–0.5 wt%) into a polymer-derived silicon oxycarbide (SiOC) composite anode, producing a mechanically reinforced electrode that exhibited an initial lithiation capacity of ∼812 mAh g−1 and a stabilized reversible capacity of 238 mAh g−1.15 Importantly, BNNTs in this system functioned not as active redox centers but as a structural and thermal stabilizer within the SiOC matrix, highlighting that their role in electrodes has so far been auxiliary rather than intrinsic. This scarcity of literature underscores a significant knowledge gap and invites further investigation into whether BNNTs, when engineered through defect control, heteroatom doping, or hybrid nano construction, could evolve into functional electrode frameworks for next-generation batteries.
Additional evidence comes from theoretical DFT studies, where BNNTs show promise as anode materials for beyond-lithium systems. For instance, Najafi (2017) reported that F-functionalized BNNTs enhance voltage cell and adsorption energy for Li-ion anodes,4 with average Vcell of 0.405 V and Ead of 5.25 kcal mol−1 for BNNT(8,0) and (10,0), increasing to 0.182 V and 8.89 kcal mol−1 upon F functionalization.16 Saadh et al. (2024) extended this to Ca-ion batteries, highlighting BNNT's superior adhesion energy, demonstrating that Ca can be used as an anode due to its high Vcell, with Ead order BNNT > BNNC.17 In multi-ion contexts, Al-Seady et al. (2023)18 found BNNTs generate higher gain voltages for Na/Li/K-ion batteries compared to BeONTs, providing theoretical insights into their potential as anodes.19 These computational insights reinforce BNNTs’ role in stabilizing ion adsorption at electrode interfaces.
Importantly, this framework is most applicable to battery systems where interfacial instabilities dominate performance, including lithium metal anodes, polymer and solid-state electrolytes, and separator-electrode interfaces. In such systems, the ability to regulate coupled ionic, thermal, and mechanical fields is critical. Taken together, these examples reveal a critical truth: BNNTs are not passive fillers. They are architectural regulators that restructure the physical fields governing battery behavior.
2. Mechanistic origins: how BNNTs regulate ion transport, mechanical stress, and thermal gradients
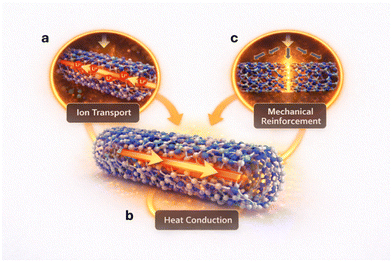
While several studies have demonstrated improved electrochemical performance upon BNNTs incorporation, the underlying mechanisms remain only partially understood. In this section, we explicitly distinguish between experimentally observed phenomena, insights derived from analogous material systems, and forward-looking mechanistic interpretations. This framework enables a physically grounded, yet evolving, understanding of how BNNTs regulate electrochemical behavior. BNNTs exert their influence on battery systems through three interconnected mechanisms: facilitating ion transport, mechanical reinforcement of interfaces, and homogenizing thermal gradients. These mechanisms are rooted in the unique atomic structure of BNNTs, where alternating boron and nitrogen atoms create a polarized lattice with partial ionic character, leading to anisotropic properties that can be harnessed for electrochemical applications. Below, we dissect each mechanism with scientific rigor, incorporating theoretical models, experimental data, and computational insights.First, experimentally, the incorporation of BNNTs into polymer and liquid electrolytes has been shown to enhance ionic conductivity and increase Li+ transference number (Fig. 2a). Building on these observations, BNNTs are proposed to regulate ion transport through a combination of polarized surface chemistry, interfacial coordination, and nanoscale transport geometry. The electron-deficient boron sites are expected to interact with anions (e.g., TFSI−, PF6−), as suggested by studies on structurally related h-BN systems and supported by limited theoretical analyses. However, direct experimental evidence of such coordination behavior in BNNT-based battery environments remains limited. This anion-adsorption effect, previously demonstrated for two-dimensional h-BN by both DFT and MD simulations, suppresses anion mobility and enhances Li-salt dissociation, thereby increasing the concentration of free Li+ and elevating the Li+ transference number.20 Similar anion-trapping phenomena have been observed in other ceramic nano-fillers used for solid polymer electrolytes.21 The high-aspect-ratio morphology of BNNTs further introduces percolated ion-conduction pathways that bypass the inherent segmental-relaxation limits of polymer hosts. When incorporated into PVDF-HFP matrices, BNNTs create fast, continuous Li+ transport channels, increasing room-temperature ionic conductivity to the 8 × 10−4 S cm−1 regime and achieving Li+ transference numbers up to ∼0.6.13 In addition, the strong BNNT–polymer interfacial interactions locally modulate chain mobility and free volume, altering the dynamic ion-solvation environment similar to other polar inorganic-polymer hybrid electrolytes.2 Collectively, BNNTs function as multifunctional ion-transport regulators-promoting selective Li+ conduction, suppressing anion diffusion, enabling homogenized ionic flux, and improving the fundamental electrochemical stability needed for next-generation solid-state or lithium-metal batteries.
Second, thermal gradient homogenization: BNNTs further contribute to battery stability by homogenizing thermal gradients through their intrinsically high axial thermal conductivity, reported in the 103 W m−1 K−1 range for well-crystallized nanotubes. This exceptional thermal transport arises from strong B–N covalent bonding, low defect densities, and phonon-dominated heat conduction, enabling BNNTs to dissipate localized heat generated at electrode interfaces, hotspots, and regions of high current density (Fig. 2b).4 When integrated into polymer electrolytes or composite electrodes, BNNTs form thermally conductive yet electrically insulating percolation pathways, analogous to the thermal management benefits demonstrated for h-BN nanosheets in solid-state electrolytes.22 This homogenization of thermal fields minimizes local overheating, reduces thermal runaway risk, and stabilizes electrochemical kinetics by maintaining uniform ionic mobility across the electrolyte. Similar studies on thermally conductive ceramic additives in batteries have shown suppressed dendrite growth at elevated currents due to improved heat dissipation and lowered interfacial overpotential.23 Unlike CNTs, whose high electrical conductivity may introduce parasitic pathways, BNNTs uniquely provide high thermal conductivity while preserving electrical insulation, making them ideal for managing heat without compromising safety. Therefore, BNNTs function as nanoscale thermal regulators that flatten temperature gradients, delay hotspot formation, and enhance both safety and uniformity during high-rate cycling. Although BNNTs are often described as chemically and thermally stable, their electrochemical stability under battery operating conditions remains insufficiently characterized. Direct measurements of the stability window of BNNTs, particularly versus Li/Li+, are scarce. Existing assumptions are largely inferred from the wide bandgap of BN-based materials and studies on h-BN, which suggest stability over a broad potential range. However, the exact oxidation and reduction limits of BNNTs, especially in contact with reactive electrodes and electrolytes, have not been systematically established. This represents a critical knowledge gap for their implementation at electrode–electrolyte interfaces.
Third, mechanical reinforcement: BNNTs also play a critical role in regulating mechanical stress within battery architectures by serving as nanoscale reinforcement elements that stabilize fragile electrolyte and electrode interfaces (Fig. 2c). Their exceptional axial Young's modulus (∼1.0–1.2 TPa) and high tensile strength, comparable to or exceeding CNTs, allow BNNTs to effectively distribute and dissipate mechanical loads generated during Li plating/stripping, volumetric changes of active materials, and thermally induced expansion.24 When embedded in polymer electrolytes, BNNTs establish a mechanically robust percolating framework that increases modulus without compromising ionic mobility – an effect analogous to ceramic nanowire reinforcement reported in solid-state battery electrolytes.25,26 This reinforcement suppresses interfacial stress accumulation and inhibits crack propagation, which is crucial for preventing filamentary lithium dendrite penetration. Experimental studies on nanoceramic-reinforced solvent-free polymeric electrolytes have shown significant increases in critical current density and suppression of dendrite initiation through the creation of mechanically stiff yet ion-permeable networks.27 Due to their electrically insulating but mechanically strong structure, BNNTs uniquely provide dendrite-blocking capability without introducing parasitic electronic pathways, an inherent advantage over conductive CNT fillers. Thus, BNNTs act as multifunctional mechanical regulators, reinforcing the electrolyte matrix, stabilizing the electrode–electrolyte interface, and enabling safer long-term cycling under high current densities. While these effects are consistent with established physical principles, direct mechanistic verification in BNNT-based battery systems remains limited.
These three mechanisms-ion-transport regulations, mechanical stress redistribution, and thermal-gradient homogenization-demonstrate that BNNTs act as multifunctional nanoscale regulators within lithium battery systems. Their polarized surfaces selectively promote Li+ mobility, their exceptional mechanical stiffness stabilizes fragile interfaces, and their high thermal conductivity dissipates localized heat without introducing electronic pathways. By simultaneously addressing ionic, mechanical, and thermal bottlenecks, BNNTs offer a promising materials platform for addressing coupled instabilities that enhance safety, uniformity, and high-rate performance, positioning them as a strategically valuable component in next-generation solid-state and lithium-metal batteries. We emphasize that these mechanisms remain hypotheses that require direct experimental validation in BNNT-based electrochemical systems. Moreover, BNNTs are not universally optimal; in applications where electronic conductivity is required, alternative materials such as CNTs or MXenes may be more appropriate.
3. Why BNNT battery research has stalled: a deep structural misalignment
Despite their exceptional combination of thermal conductivity, electrical insulation, mechanical stiffness, and chemical robustness, BNNTs have not yet achieved widespread adoption in battery systems. This limited penetration is not due to a lack of intrinsic merit, but rather arises from four interrelated challenges spanning synthesis, structural control, materials quality, and conceptual deployment.The first and most fundamental limitation is synthesis scalability. High-quality BNNTs are primarily produced via arc-discharge, laser ablation, or chemical vapor deposition routes. While these methods yield nanotubes with excellent crystallinity, long aspect ratios, and minimal intrinsic defects, their production rates remain extremely low, typically on the order of grams per hour or less. In parallel, the energy-intensive nature of these synthesis techniques, coupled with complex post-processing requirements, results in costs exceeding $1000 per gram, several orders of magnitude higher than those of carbon nanotubes. As a result, BNNT availability remains restricted to laboratory-scale studies, severely limiting systematic optimization and industrial translation. Numerous review articles have explicitly identified scalable and economically viable BNNT synthesis as a prerequisite for meaningful technological deployment, yet practical solutions remain elusive.19,28 As a result, limited production throughput and energy-intensive synthesis directly translate into high material costs, which remain a primary barrier to large-scale adoption in battery technologies.
The second challenge concerns defect control and chemical functionality. Pristine BNNTs possess a highly stable hexagonal boron nitride lattice with strong B–N bonds and minimal electronic states near the Fermi level. While this chemical inertness underpins their outstanding stability, it also limits interactions with ions, polymer matrices, and host materials. For BNNTs to actively participate in composite architectures, controlled defects, such as boron or nitrogen vacancies, substitutional dopants, or localized functional sites, are often required to enable anchoring, interfacial coupling, or local field modulation. However, current synthesis methods offer limited control over defect density, type, and spatial distribution. As a result, defect introduction is frequently stochastic, leading to inconsistent interfacial behavior and irreproducible performance across different batches. This lack of precision contrasts sharply with the growing demand for deterministic interface design in advanced battery materials.19,29 This lack of control over defect chemistry further complicates reproducibility, making it difficult to establish consistent structure–property relationships at scale.
Purity represents the third, and arguably most critical, bottleneck. As-produced BNNTs powders are rarely phase-pure and typically contain significant fractions of hexagonal boron nitride platelets, amorphous BN, unreacted precursors, and residual catalyst particles. In addition, BNNT populations often exhibit broad distributions in tube diameter, wall number, and length. These heterogeneities profoundly affect composite behavior: h-BN platelets disrupt percolation pathways, amorphous phases degrade mechanical reinforcement, and metallic residues introduce parasitic reactions. Consequently, impurities distort thermal transport, mechanical stress distribution, and interfacial uniformity, obscuring the intrinsic contributions of BNNTs. Importantly, every major successful demonstration of BNNT-enabled battery components, most notably those reported by Kim, He, and Yadav, relied on rigorously purified BNNTs. This recurring pattern highlights purity not as a secondary processing detail, but as a foundational requirement for reproducibility, mechanistic clarity, and performance optimization.12–14 Consequently, variability in purity and morphology not only affects performance but also poses significant challenges for process standardization and industrial quality control.
Finally, and more subtly, progress has been hindered by conceptual misalignment in how BNNTs are incorporated into battery architectures. Battery failure is inherently an interfacial phenomenon, originating from localized gradients in concentration, mechanical stress, electric field, and temperature. These instabilities emerge at lithium metal surfaces, at grain boundaries in brittle solid electrolytes, at cathode–electrolyte interfaces, and within mechanically constrained polymer hosts. BNNTs, by virtue of their one-dimensional geometry, anisotropic transport properties, and exceptional stiffness, are uniquely suited to regulate such interfacial fields. Yet in many studies, BNNTs are dispersed as general fillers within bulk materials, where their directional and interfacial advantages are diluted. This filler-centric paradigm overlooks the architectural nature of BNNTs. Until BNNTs are deliberately positioned at failure-prone interfaces and oriented to control stress, heat, and ion flux, their role will remain underestimated and their impact marginal.19,28 From a practical standpoint, this conceptual misalignment also limits effective material utilization, as randomly dispersed BNNTs fail to leverage their anisotropic properties, reducing their cost-effectiveness in real devices.
Taken together, these factors highlight that the challenges associated with BNNTs are not solely material limitations, but system-level integration issues that must be addressed collectively. Future progress will depend on the convergence of scalable synthesis, controlled purification, and architecture-aware processing strategies. In particular, approaches that maximize functional efficiency-such as targeted interfacial placement or alignment—may enable the use of minimal BNNT loadings while preserving performance benefits, thereby improving economic feasibility for practical battery applications.
4. A forward framework: BNNTs as engineered interfacial architectures
To unlock the full potential of BNNTs in battery systems, the field must adopt an architectural mindset – one driven by interface programming rather than additive blending.The first requirement is alignment. BNNTs cannot function as regulators when randomly dispersed. Alignment transforms BNNTs from passive inclusions into directional mechanical beams, ion flux guides, and thermal channels. Techniques such as shear-induced alignment, magnetic-field alignment, freeze casting, and electrospinning are not exotic research tools – they are manufacturable processes that can be directly adapted to electrolyte films and separator coatings. The separator-coating approach used by Kim et al. provides the simplest evidence: even partial alignment produced substantial performance gains.12 Emerging work on BNNT porins suggests aligned nanochannels for selective ion transport.30
A second requirement is the deliberate programming of BNNT defect chemistry. Defect engineering has already been explored in 2D boron nitride to tune catalytic behavior, hydrogen adsorption, and electronic properties. Applying similar strategies to BNNTs would allow researchers to tune the intensity of BNNT–ion interactions, control polymer anchoring behavior, and potentially stabilize solid-electrolyte interphases.19,29
A third requirement is strategic spatial placement. BNNTs should be incorporated precisely where field gradients are most harmful. At the Li metal interface, BNNTs could serve as both electron blockers and mechanical stabilizers, producing more uniform Li plating. At the cathode interface, they could suppress high-voltage electrolyte oxidation and mitigate oxygen release. In solid-state batteries, BNNTs could reinforce grain boundaries, preventing catastrophic cracking. These are not speculative roles; they directly extend the successes reported in the existing literature.13,31,32
A fourth requirement is viewing BNNTs as frameworks rather than particles. BNNT scaffolds or networks – whether constructed by freeze casting, templating, or interfacial self-assembly – could act as hybrid architectures that integrate thermal conduction, mechanical reinforcement, and ionic transport tuning into a single physical platform.19,28 With recent advances in scalable synthesis and defect control, BNNTs could enable batteries with unprecedented safety and longevity by 2030.
A final requirement is chemistry selection; the suitability of BNNTs is heavily dependent on the chemistry of the battery and the dominant failure mechanisms. BNNTs are most suitable for lithium metal batteries, lithium–sulfur batteries, and solid-state or polymer electrolytes, where interfacial instability, dendrite formation, and thermal gradients are dominant. In these cases, the ability of BNNTs to regulate ionic flux, redistribute mechanical stress, and dissipate heat is directly beneficial for overcoming major performance challenges in these systems.33–36 On the contrary, BNNTs are not so suitable for cases where high electronic conductivity is a requirement, such as a conductive scaffold for conventional lithium-ion batteries. In these cases, materials such as CNTs and MXenes are far better for electronic percolation.37–39 In cases where cost is a major factor and the ability of materials to regulate is not required, simple ceramics could be considered for use. This again points to the fact that BNNTs have to be considered for use in cases where a combination of properties is required and could be fully leveraged.
Author contributions
Both authors contributed equally to this work. Each author has the right to list themselves as the first author in their own professional profile.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software, or code have been included, and no new data were generated or analyzed as part of this perspective.References
- Y. Xiao, Y. Wang, S.-H. Bo, J. C. Kim, L. J. Miara and G. Ceder, Nat. Rev. Mater., 2019, 5, 105–126 CrossRef.
- D. Kim, J. Ko, Y.-K. Kim, S. S. Lee, S. Ahn, S. Lee, S. G. Jang and Y. Joo, Small Struct., 2024, 5, 2400259 CrossRef CAS.
- H. Xu, X. Song, Y. Gu, J. Fan, J. Liu and S. Wang, J. Mater. Chem. A, 2025, 13, 10388–10414 RSC.
- C. Zhi, Y. Bando, C. Tang and D. Golberg, Mater. Sci. Eng., R, 2010, 70, 92–111 CrossRef.
- A. M. Marconnet, M. A. Panzer and K. E. Goodson, Rev. Mod. Phys., 2013, 85, 1295–1326 CrossRef CAS.
- A. K. Worku, M. A. Alemu, D. W. Ayele, M. Z. Getie and M. A. Teshager, Green Chem. Lett. Rev., 2024, 17, 2325983 CrossRef.
- S. G. Eswaran, M. Rashad, A. S. K. Kumar and A. F. M. El-Mahdy, Chem. – Asian J., 2025, 20, e202401181 CrossRef PubMed.
- J. Chen, X. Huang, B. Sun and P. Jiang, ACS Nano, 2019, 13, 337–345 CrossRef CAS PubMed.
- J. Liu, M. Fang, E.-H. Yang and X. Zhang, Sci. Rep., 2025, 15, 1976 CrossRef CAS PubMed.
- Y. Ouyang, X. Li, H. Tian, L. Bai and F. Yuan, Nanomaterials, 2021, 11, 2654 CrossRef CAS PubMed.
- Z. Ahmed, L. S. Nasrat and M. Rihan, Electrical Engineering Electromechanics, 2025, 84–89 CrossRef.
- H.-S. Kim, H.-J. Kang, H. Lim, H. J. Hwang, J.-W. Park, T.-G. Lee, S. Y. Cho, S. G. Jang and Y.-S. Jun, Nanomaterials, 2022, 12, 11 CrossRef CAS PubMed.
- Q. He, X. Liu, G. Xiao, X. He, W. Gong, L. Tang, Q. Chen, Q. Zhang and Y. Yao, Small, 2024, 20, 2403660 CrossRef CAS PubMed.
- D. Yadav, J.-H. Jung, Y. Lee, T. Y.-S. Kim, E. Park, K.-I. Choi, J. Cha, W.-J. Song, J.-H. Choi, S. Doo and J. Kim, ACS Mater. Lett., 2023, 5, 2648–2655 CrossRef CAS.
- M. A. Abass, A. A. Syed, C. Gervais and G. Singh, RSC Adv., 2017, 7, 21576–21584 RSC.
- M. Najafi, Can. J. Chem., 2017, 95, 687–690 CrossRef CAS.
- M. J. Saadh, S. F. Jawad, S. Ballal, G. V. S. Prasad, A. I. Saber, M. Singh, M. N. Shakir, A. Elawady and R. Zainul, J. Nanopart. Res., 2024, 26, 173 CrossRef CAS.
- M. A. Al-Seady, H. H. Abed and S. M. Alghazaly, et al. Prospective utilization of boron nitride and beryllium oxide nanotubes for Na, Li, and K-ion batteries: a DFT-based analysis, J. Mol. Model., 2023, 29, 348 CrossRef CAS PubMed.
- S. Angizi, S. A. A. Alem, M. Torabian, M. Khalaj, D. Golberg and A. Pakdel, Energy Environ. Mater., 2024, 7, e12777 CrossRef CAS.
- Y. Li, L. Zhang, Z. Sun, G. Gao, S. Lu, M. Zhu, Y. Zhang, Z. Jia, C. Xiao, H. Bu, K. Xi and S. Ding, J. Mater. Chem. A, 2020, 8, 9579–9589 RSC.
- Z. Shen, Y. Cheng, S. Sun, X. Ke, L. Liu and Z. Shi, Carbon Energy, 2021, 3, 482–508 CrossRef CAS.
- G. Y. Uzunoglu, S. Coskun and R. Yuksel, ChemElectroChem, 2025, 12, e202500011 CrossRef.
- X. Feng, D. Ren, X. He and M. Ouyang, Joule, 2020, 4, 743–770 CrossRef CAS.
- D. Golberg, Y. Bando, Y. Huang, T. Terao, M. Mitome, C. Tang and C. Zhi, ACS Nano, 2010, 4, 2979–2993 CrossRef CAS PubMed.
- G. Dong, H. Zhang, Y. Cheng and L. Xu, Nano Trends, 2024, 7, 100043 CrossRef CAS.
- W. Liu, N. Liu, J. Sun, P.-C. Hsu, Y. Li, H.-W. Lee and Y. Cui, Nano Lett., 2015, 15, 2740–2745 CrossRef CAS PubMed.
- T. Subhani, S. Khademolqorani, S. N. Banitaba, M. Ramadan, A. Khaliq, I. A. Chaudhry and A. I. Osman, ACS Appl. Mater. Interfaces, 2024, 16, 63089–63108 CrossRef CAS PubMed.
- D. Sun, Z. Sun, D. Yang, X. Jiang, J. Tang and X. Wang, EcoEnergy, 2023, 1, 375–404 CrossRef.
- J. Pu, K. Zhang, Z. Wang, C. Li, K. Zhu, Y. Yao and G. Hong, Adv. Funct. Mater., 2021, 31, 2106315 CrossRef CAS.
- Z. Li, A. T. Hall, Y. Wang, Y. Li, D. O. Byrne, L. R. Scammell, R. R. Whitney, F. I. Allen, J. Cumings and A. Noy, Sci. Adv., 2024, 10, eado8081 CrossRef CAS PubMed.
- J.-H. Jung, N. Yanar, M.-J. Yang, S. Kandula, D. Yadav, T. Y.-S. Kim, J.-H. Shim, S. Doo and J. Kim, J. Power Sources, 2025, 628, 235938 CrossRef CAS.
- M. M. Rahman, S. Mateti, Q. Cai, I. Sultana, Y. Fan, X. Wang, C. Hou and Y. Chen, Energy Storage Mater., 2019, 19, 352–359 CrossRef.
- H.-S. Kim, H.-J. Kang, H. Lim, H. J. Hwang, J.-W. Park, T.-G. Lee, S. Y. Cho, S. G. Jang and Y.-S. Jun, Nanomaterials, 2022, 12, 11 CrossRef CAS PubMed.
- Y. Guo, Q. Niu, F. Pei, Q. Wang, Y. Zhang, L. Du, Y. Zhang, Y. Zhang, Y. Zhang, L. Fan, Q. Zhang, L. Yuan and Y. Huang, Energy Environ. Sci., 2024, 17, 1330–1367 RSC.
- Z. Wang, S. Qin, F. Chen, S. Chen, D. Liu, D. Jiang, P. Zhang, P. Mota-Santiago, D. Hegh, P. Lynch, A. S. Alotabi, G. G. Andersson, P. C. Howlett, M. Forsyth, W. Lei and J. M. Razal, ACS Nano, 2024, 18, 3531–3541 CrossRef CAS PubMed.
- H. Lim, H. Jeong, Y.-C. Song, S.-B. Han, J. Lee, J. B. Park and Y.-W. Lee, Korean J. Chem. Eng., 2026, 43, 389–401 CrossRef CAS.
- D. Sunkari, K. Deshmukh, S. Panda and S. K. K. Pasha, J. Energy Storage, 2024, 92, 112017 CrossRef CAS.
- Z. Hong, H. Tian, Z. Fang, H. Wu, F. Zhao, Q. Li, S. Fan, J. Wang and P. Liu, ACS Appl. Energy Mater., 2024, 7, 8004–8013 CrossRef CAS.
- G.-H. Dong, Y.-Q. Mao, Y.-Q. Li, P. Huang and S.-Y. Fu, Electrochim. Acta, 2022, 420, 140464 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |