 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Regulation and reformulation: how the EU Paints Directive shaped volatile organic compound emissions from UK decorative paints
Lidia Kristia Alfanti *a,
Amber May Yeoman
*a,
Amber May Yeoman ab,
Andrew Robert Rickard
ab,
Andrew Robert Rickard ab,
David Shaw
ab,
David Shaw bc,
Martyn William Wardab,
Sarah Julia Moller
bc,
Martyn William Wardab,
Sarah Julia Moller ab and
Alastair Charles Lewis
ab and
Alastair Charles Lewis ab
ab
aWolfson Atmospheric Chemistry Laboratories, University of York, York, YO10 5DD, UK. E-mail: lidia.alfanti@york.ac.uk
bNational Centre for Atmospheric Science, University of York, York, YO10 5DD, UK
cDepartment of Environment and Geography, University of York, York, YO10 5DD, UK
First published on 20th April 2026
Abstract
Decorative paints have undergone substantial solvent reformulation in response to air quality regulatory pressure and advances in materials science, but they remain an important contributor to national VOC emissions. Paint-derived VOCs currently represent 9.6% of total United Kingdom VOC emissions. The changes in UK emissions from decorative paints (1990–2024) have been quantified using a combination of national emissions inventory data, laboratory analysis using headspace GC/MS, and atmospheric modelling. A major decline in total mass of VOC emissions from paints followed the implementation of the EU Paints Directive, with per capita emissions falling by approximately 60% between 2004 and 2019 from 0.532 kg per person per year to 0.210 kg per person per year. The reduction in mass of paint VOC emissions coincided with a transition away from hydrocarbon solvents, notably alkanes such as decane, and other alkane isomers in the C8–C12 range. Paints in 2024 contain virtually no alkane content, instead comprising a wide range of chemical functionalities, including alcohols, ketones, esters, and siloxanes. The VOC speciation in 2024 varied significantly by manufacturer, but rather little across manufacturers' product range for different end-uses. Reformulated 2024 paints exhibited substantially lower SOA formation potential, approximately six times lower than older alkane-based paint formulations and had reduced total hydroxyl radical reactivity. Atmospheric model simulations comparing decane (reflective of 1990 paints) and t-butanol (reflecting 2024 paints) indicated that modern VOC emission mixtures likely degrade more slowly and generate less formaldehyde as a by-product. This study provides an updated VOC speciation for paints, suitable for use in emissions inventories and air pollution assessments.
Environmental significanceThis study provides the first systematic characterisation of volatile organic compounds (VOCs) in modern wall paint formulations following regulatory implementation. Older paints were dominated by alkanes, whereas contemporary products contain more oxygenated VOCs that are less reactive and degrade more slowly in the atmosphere. Although these new formulations show reduced potential for ozone and secondary organic aerosol formation, current emission inventories still rely on outdated 1990s profiles that no longer reflect market realities. By delivering an updated VOC profile, this work demonstrates the effectiveness of regulation and provides essential data to improve emission inventories and support understanding of air quality impacts. |
1 Introduction
Paints and coatings are a well-established and substantial emission source of both indoor and outdoor air pollution. Freshly applied paints release substantial amounts of volatile organic compounds (VOCs), making them an active emission source.1 Unlike other indoor sources of VOCs, such as combustion,2 aerosols,3 and non-aerosol products,4 paint emissions are often underestimated5 despite having a long-term impact on indoor air quality. Even after drying, paints continue to outgas VOCs passively for long periods.6Individuals spend over 90% of their time indoors,7 and since most homes and offices have painted walls and ceilings, paint is a widespread contributor to indoor VOC concentrations. A change in recent years has been a move to low-VOC and ‘eco-friendly’ paints,8 although there has been limited evaluation of whether these alternatives genuinely reduce emissions and indoor impacts over time.
Defining VOCs is not straightforward, as different regulations apply different standards.9 For example, the 2004/42/EC Paints Directive defines VOCs as organic compounds with a boiling point below 250 °C at a standard pressure of 101.3 kPa.10 In contrast, organisations such as the World Health Organization (WHO) classify VOCs based on their volatility,11 which distinguishes between very volatile organic compounds (VVOCs), volatile organic compounds (VOCs), and semi-volatile organic compounds (SVOCs). These differences in classification affect the framework of regulations and can influence how individual compounds are assessed or regulated in indoor environments. The lack of standardisation in VOC classifications complicates the challenge of evaluating their impact on indoor air quality. In contrast to outdoor air, where VOCs are more easily dispersed,12 indoor environments can trap and accumulate VOCs to higher concentrations.13 VOCs indoors have been linked to acute or chronic illness for decades,14,15 while long-term exposure may be linked to carcinogenic effects even at moderate exposure levels.16 Previous literature has found VOCs in human urine17 and blood;18 once in the bloodstream, there are reported impacts on vital organs.19 Awareness of indoor air pollution has increased notably over recent years,20 with VOCs being one of the major pollutants of concern.21
Among numerous VOC emissions sources, paints have long been recognised as a major contributor, given their high solvent content and extended off-gassing period.22 By the 1980s, the United States Environmental Protection Agency (US EPA) had identified VOC emissions from paints as a major precursor to ground-level ozone (O3), and showed that in some chemical regimes, controlling VOC emissions would be an effective way to reduce photochemical ozone formation.23 The US EPA began to issue regulations specifically targeting VOC emissions shortly after, including paints, which led to the implementation of the Clean Air Act Amendments of 1990 (CAAA). The establishment of CAAA signified that paint VOCs were formally acknowledged as contributors to ground-level ozone. Moreover, it provided a blueprint for the regulations of the VOC content in paints. This, in turn, prompted global implementation to introduce updated guidelines on paint formulation. The European Parliament implemented Directive 2004/42/CE of the European Parliament and of the Council24 to restrict VOC content in paints and varnishes, with the objective of reducing ozone formation. This was built on the earlier VOC Solvents Emission Directive (1999/13/EC),25 which targeted industrial sources of VOCs, marking the first step toward broader VOC regulation in the EU. Subsequent EU regulations, including Commission Decisions 2009/543/EC26 and 2009/544/EC,27 established criteria for awarding the EU ecolabel to paints and varnishes, which were further updated by Commission Decision 2014/312/EU28 to introduce explicit limits on semi-volatile organic compounds (SVOCs). The successful implementation of VOC limits under the CAAA showed that regulating VOCs in paints was feasible, paving the way for similar measures like the Paint Directive.29
Regulatory pressures arising from the Paints Directive in 2004, and growing consumer awareness, drove a major shift in paint formulations by the European paint industry, including major reformulations to reduce VOC content.30 A rising concern around indoor VOC exposure drove consumer demand for healthier, safer, and eco-friendly products, in the development of “VOC-free” formulations.31 Paint manufacturers define “low-VOC” as products having VOC content ranging from 50 g L−1 to 250 g L−1, whilst “zero-VOC” indicates a VOC level below 5 g L−1.22 The paint reformulation was enabled by a shift from VOC-based to water-based solvents.32 With the Paint Directive becoming active to limit VOC emissions, indoor air quality is anticipated to have significantly improved. However, evaluating the comprehensive consequences of paint reformulation must include a complete analysis of not only direct emissions but also any secondary chemistry arising, for example capacity to form other harmful air pollutants such as formaldehyde or particulate matter.
Giampieri et al.33 undertook an analysis of the impacts of automotive paints, exploring historical reductions in air pollutants from manufacturing and how regulations influenced emission trends. The paper highlighted that water-based automotive paints had around half the VOC emissions of solvent-based automotive paints. Improvements in spray application technology led to reductions in automotive paint VOC emissions from 500 g m−2 in the 1970s to under 35 g m−2 in 2020. This re-evaluation indicated that paint reformulations could play an important role in emissions reduction from the automotive sector. Pilon et al.34 evaluated the performance of 77 different commonly used solvents in the paints and coatings industries, highlighting their crucial functional roles, while identifying their substantial role in VOC emissions. While overall tonnages of VOCs released by the paint industry are believed to have decreased as a consequence of domestic paint reformulation, a comprehensive analysis of impacts on the indoor environment is lacking.
This study investigates the impact of paint reformulation on the current paint formulation, particularly focusing on specific VOC emission compounds and the formation of their secondary products. Thirty-three commercially available wall paint samples in the UK were analysed for their detailed VOC speciation. The novelty of this study is the quantification of modern paint speciation and an investigation of whether newer alternative components in reformulated paints introduce new air quality concerns, such as secondary pollutant formation, since indoor VOCs can take part in reactions to produce secondary organic aerosol, ozone, and other harmful air pollutants such as formaldehyde.35 The long-term trends in VOC emissions from paint are evaluated using the UK National Atmospheric Emissions Inventory, with a comparison between the inventory speciation (based on 1990s data) and present-day composition derived from new laboratory measurements reported here.
2 Experimental
2.1. Sample selection process
Thirty-three paint samples from nineteen different brands were acquired through various UK retail channels, with the selection focused on products intended for residential/domestic use. Samples were chosen to represent the diversity of paint products commonly available to UK consumers. All samples were standard liquid wall paints, with no aerosolised paints included, since for emissions inventories these are categorised differently. The paints tested included a range of product categories and formulations, including paints marketed with specific functional claims, such as anti-mould, anti-condensation, and vegan formulations. The samples were classified into eleven categories based on their product type and functional claims, shown in Fig. 1.2.2. Sample preparation and VOC identification
Approximately 2 grams of paint were weighed and transferred into 20 mL clear glass round-bottom headspace vials and subsequently sealed with magnetic stainless-steel screw caps equipped with a 1.6 mm PTFE-faced butyl septum.VOCs present in the headspace of 33 paint samples were first separated on a BPX5 capillary column (50 m × 0.32 mm ID, 1 µm film thickness) using helium as the carrier gas at a constant flow rate of 2 mL min−1. A 2 mL gas aliquot of the headspace was injected into the GC inlet held at 330 °C in split mode (split ratio 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, split flow 15 mL min−1). The headspace syringe needle was maintained at 70 °C. Prior to injection, samples were incubated at 35 °C and agitated for 5 minutes using a Gerstel agitator. The system was coupled with a Gerstel MultiPurpose Autosampler (Gerstel, Germany) fitted with a 2.5 mL headspace syringe. The oven was initially held at 40 °C for 5 minutes before being ramped at 10 °C min−1 to a final temperature of 340 °C, giving a total runtime of 40 minutes. Eluting compounds were identified using a 7200 Accurate-Mass Quadrupole Time-of-Flight Gas Chromatography/Mass Spectrometer (Q-TOF GC/MS; Agilent Technologies, Santa Clara, CA, USA). The transfer line temperature was held at 350 °C, and mass spectra were collected over the range of 35–350 amu. A nitrogen collision gas was introduced in the mass spectrometer at a flow rate of 1.5 mL min−1, and spectra were recorded at 5 Hz.
1, split flow 15 mL min−1). The headspace syringe needle was maintained at 70 °C. Prior to injection, samples were incubated at 35 °C and agitated for 5 minutes using a Gerstel agitator. The system was coupled with a Gerstel MultiPurpose Autosampler (Gerstel, Germany) fitted with a 2.5 mL headspace syringe. The oven was initially held at 40 °C for 5 minutes before being ramped at 10 °C min−1 to a final temperature of 340 °C, giving a total runtime of 40 minutes. Eluting compounds were identified using a 7200 Accurate-Mass Quadrupole Time-of-Flight Gas Chromatography/Mass Spectrometer (Q-TOF GC/MS; Agilent Technologies, Santa Clara, CA, USA). The transfer line temperature was held at 350 °C, and mass spectra were collected over the range of 35–350 amu. A nitrogen collision gas was introduced in the mass spectrometer at a flow rate of 1.5 mL min−1, and spectra were recorded at 5 Hz.
GC/MS Q-TOF data processed with Agilent MassHunter Qualitative Analysis (vB.07.00) and the NIST Library (MS Search v3.0). Chromatographic peaks were assigned based on spectral similarity and retention time. Only peaks with a signal-to-noise ratio of ≥3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 were considered for identification.
1 were considered for identification.
Peak areas obtained from GC-MS headspace analysis are unitless values representing the relative abundance of each VOC. To estimate concentrations, a normalisation procedure was applied whereby the peak area of each compound was adjusted based on its frequency of occurrence across samples and expressed as a proportion of the total peak area for all detected compounds. This yielded a percentage contribution for each VOC. For the purposes of later modelling, the internally normalised distribution was then scaled to an estimated total VOC in room gas phase concentration of 300 ppb, selected as a representative value for older housing stock with lower ventilation rates, and based on previous indoor air quality studies.36,37 Final results are expressed in ppb, representing the gas-phase mixing ratio of VOCs under standard atmospheric conditions. This method provides semi-quantitative data suitable for comparative analysis between compounds and samples.
2.3. Estimation of photochemical ozone creation potential (POCPE)
POCP is defined as the ability of an individual VOC to produce ozone depends upon the amount of VOC emitted, its reactivity, and its propensity to form ozone. The potential for a given VOC to form ozone is related to the number of relative C–C and C–H bonds it contains.38 POCP values were estimated for each VOC following the approach adapted from Jenkin et al.39 Here, POCP refers to values derived from full 5 days photochemical trajectory model simulations reported in the literature,40 whereas POCPE denotes estimated values for each VOC, based on OH reactivity and molecular structure, derived using the parametric equation described in eqn (1). Both POCP and POCPE are dimensionless, expressed relative to ethene as a reference compound (POCP for ethene = 100).| POCPE = (A × γs × R × S × F) + P + RO3 − Q | (1) |
All parameters presented in eqn (1) are unitless.39 The parameter A acts as a general multiplier in the expression, and γs is the structural reactivity parameter. This latter term captures the potential for ozone formation based on the number of reactive sites per unit mass. R is the OH reactivity factor, defined by the relative rate coefficient of the VOC with hydroxyl (OH) radicals compared to ethene. The gas-phase OH rate constants (kOH) for the investigated compounds were obtained from the EUROCHAMP-2020 Database for the Kinetics of the Gas-Phase Atmospheric Reactions of Organic Compounds (https://data.eurochamp.org/).41 For each species, the recommended kOH value (cm3 molecule−1 s−1) under standard conditions (298 K, 1 atm) was used. The size-related parameter S adjusts for the decreasing ozone-forming efficiency of larger VOCs due to factors such as increased organic nitrate formation and extended degradation pathways.
In addition to these core parameters, four correction factors are included for specific compound classes. F accounts for the formation of unreactive carbonyl products that can interrupt radical propagation and reduce ozone yield. P is an optional term used to correct for additional compound-specific reactivity factors. RO3 represents the contribution of free radicals generated directly from VOC–ozone reactions. Lastly, Q is applied to selected aromatic VOCs that form high yields of nitrophenol products, which act as sinks for radicals and NO2, further limiting ozone formation. More details on the applications of the factors defined in eqn (1) can be found in Jenkin et al.39
2.4. Calculation of primary OH reactivity
Primary OH reactivity was calculated using VOC concentrations under standard atmospheric conditions (temperature of 298 K and pressure of 1 atm), applying the following general formula adapted from:42| OH reactivity (s−1) = [VOC (ppb)] × 10−9 × [M] × kOH(298 K) | (2) |
2.5. Analysis of secondary organic aerosol formation potential (SOAP)
A simplified mass-based approach43 was applied to estimate the secondary organic aerosol (SOA) formation potential of individual VOCs. This approach is consistent with methods used in prior studies, such as,44 which provides SOAP values based on modelled mass yields under UK atmospheric conditions. In this analysis, SOAP was calculated using VOC-specific SOA yield values (µg m−3 SOA µg m−3 VOC, defined as the mass of SOA formed per unit mass of VOC reacted), multiplied by the ambient concentration of each VOC compound.VOC mole fractions obtained from the primary OH reactivity calculation were then converted to µg m−3 using the ideal gas law equation:
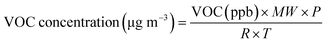 | (3) |
The atmospheric pressure was assumed to be 1 atm (101![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 325 Pa). The ideal gas constant (R) used in the calculation was 8.314 J mol−1 K−1. The temperature was set at 298 K to represent typical ambient conditions. Following this conversion, the potential SOA mass was then estimated as:
325 Pa). The ideal gas constant (R) used in the calculation was 8.314 J mol−1 K−1. The temperature was set at 298 K to represent typical ambient conditions. Following this conversion, the potential SOA mass was then estimated as:
| SOA mass (µg m−3) = VOC concentration (µg m−3) × SOA yield | (4) |
The SOA yield values used in this analysis were compiled from various studies.45–50 This SOA yield represents the mass fraction of VOC that is converted into secondary organic aerosol under typical atmospheric conditions. Where yield values were reported as a range, the upper limit was selected to represent a worst-case scenario for SOAP.
2.6. Atmospheric modelling of different VOC speciations
Atmospheric modelling was performed using the Indoor Chemical Model in Python (INCHEM-Py)51,52 to investigate the atmospheric decay of individual VOC species, as well as the formation of formaldehyde (HCHO) as a secondary pollutant. The model was specifically run for decane and t-butanol, which were chosen to represent the major compounds in the 1990 and 2024 paint formulations. The simulation settings were configured with no artificial or natural light. Hydroxyl radicals (OH) acted as the only oxidant reacting directly with t-butanol and decane, while background concentrations of ozone (O3) and nitrate radicals (NO3) were also included to capture reactions of secondary products leading to formaldehyde formation. The default configuration in INCHEM-Py was used with no surface deposition processes considered. The simulation files are provided alongside the paper for reference. Both compounds were simulated over a period of seven days in order to capture their decay dynamics and the associated secondary pollutant formation. Key atmospheric oxidants were introduced to initiate chemical reactions, based on typical atmospheric conditions: OH at 1.00 × 106 molecules cm−3,53 O3 at 1.00 × 1012 molecules cm−3,54 NO3 at 1.00 × 108 molecules cm−3,55 and NOx at 1 ppb. All oxidant concentrations were held constant throughout the simulations. The simulations were performed under the assumption that 1 L of paint is applied to a UK minimum room size of 17.25 m3, with each compound present at 20 mg per 100 mL of paint, in order to represent realistic emission scenarios in indoor environments. Full model input parameters are summarised in Table S2 of the SI.3 Results and discussion
3.1. Trends in indoor VOC emissions
Fig. 2 presents the change over time in total UK VOC emissions from various domestic sources of relevance to indoor air quality, between 1990 and 2019, based on data from the UK national Atmospheric Emissions Inventory. The data is adjusted to provide a per-capita estimate of emissions, correcting for changes in the overall UK population. The UK National Atmospheric Emissions Inventory (NAEI) provides annual emission data for numerous VOCs from various air pollutant sources (https://naei.energysecurity.gov.uk/air-pollutants). This study uses filtered data from category 2D3d – coating applications, specifically retail decorative paints, which are primarily used in domestic settings. Data for this category are derived from UK government statistics, trade bodies like the British Coatings Federation, industrial operators, and regulators. Where direct data are unavailable, modelled estimates using proxies such as plant capacity ensure consistency over time. Emission factors are sourced from operator reports, industry data, UK research, and internationally recognised guidance, including the European Monitoring and Evaluation Programme (EMEP) and European Environment Agency (EEA) Air Pollutant Emission Inventory Guidebook. This multi-source methodology underpins a robust and current inventory of VOC emissions from coating applications in the UK. Indoor VOC emissions per person were estimated by combining national VOC inventory data with household occupancy statistics from the Office for National Statistics (https://www.ons.gov.uk/). The data used was from the People, population and community section – population estimates, showing the annual population estimates across the UK. Total VOC emissions by source and year were first adjusted to account for the fraction estimated to be released indoors, using the Indoor Air Quality report from the Air Quality Expert Group on indoor air quality in the UK.56 These indoor emissions were then normalised by the UK population to obtain per-capita VOC exposure. Finally, average household occupancy data were used to convert per-household emissions into per-person estimates.Emissions from aerosols – cosmetics and toiletries ranged from 0.56 kg per person per year in 1990 to 0.68 kg per person per year in 2019, making it the largest contributor to VOC emissions for most of this period. The only exception was between 2001 and 2004, when decorative paints briefly became the largest source. Aerosols represent a significant source of VOC emissions due to high VOC content and have widespread and stable consumption patterns in high-income countries such as the UK.57 Some fraction of aerosols are paint-related; however, the majority are for personal-care, including deodorants, perfumes, and hairsprays.58 To better interpret the fluctuating pattern in decorative paint emissions, the trend is divided into three distinct phases: A (1990–1996), B (1996–2004), and C (2004–2019), as labelled in Fig. 2.
The decline in VOC emissions from decorative paints between 1990 and 1996, from 0.44 kg per person annually to 0.31 kg per person annually, was potentially driven by economic factors that reduced the demand. Based on a European Commission report on VOC emissions from decorative paints and varnishes, economic conditions play an important role in shaping paint demand, as periods of economic slowdown were associated with decreased paint consumption and subsequently lower emissions.59 Between 1996 and 2004, the resurgence in VOC emissions of decorative paints from 0.31 kg per person annually to 0.53 kg per person annually may be attributed to a significant rise in do-it-yourself (DIY) home improvement activities in the UK. Watson and Shove60 reported that since the late 1990s, the UK DIY market has experienced steady growth driven by a buoyant housing market and increasing consumer interest in home improvement and renovation projects. They further identified that product innovations during this period redistributed skills from professional tradespeople to amateur consumers, enabling a wider range of DIY tasks to be undertaken at home. This shift contributed to expanding DIY participation and increased consumption of associated products, including paints, which has implications for rising indoor paint emissions.
The most significant reduction in paint VOC emissions occurred between 2004 and 2019, with amounts decreasing from approximately 0.53 kg per person annually to 0.21 kg per person. This decline coincided with the implementation of the EU Paints Directive (2004/42/EC), which set limits on VOC content in decorative paints and varnishes, driving a widespread shift to lower-VOC and water-based formulations.24 Emissions of VOCs from paints decreased by approximately 60% during this period. To comply with the Directive, manufacturers reformulated products, transitioning from hydrocarbon-based paints to water-based or lower-VOC content alternatives.61 Water-based paints have significantly lower VOC emissions compared to traditional hydrocarbon solvent-based paints, primarily because water replaces a substantial portion of the organic solvents used as carriers in the formulation.62 Older solvent-based paints had a VOC content of ∼300–500 g L−1,63 whilst regulatory standards in the EU and UK limit VOC concentrations in water-based interior wall paints to a maximum of 30 g L−1 UK.64 This regulatory implementation has been a key driver behind the decline in VOC emissions from decorative paints observed over the past two decades.
3.2. Comparative VOC profiling of wall paints
VOC speciation in contemporary wall paints was quantified using GC/MS to assess the impact of paint reformulation. This analytical technique enables detailed profiling of volatile compounds, providing insights into the composition of current formulations. Fig. 3 shows the VOC speciation profiles of 33 decorative wall paint samples from various manufacturers, based on peak areas obtained from Q-TOF GC/MS analysis. The samples are grouped by manufacturer, and the identified compounds are categorised by chemical class. Initially, paint samples were grouped by functional type to assess similarities in their chemical profiles. However, this grouping did not reveal common patterns in VOC composition. In contrast, categorising samples by manufacturer revealed a reasonably consistent chemical profile within each brand, as shown in Fig. 3. This pattern indicates that formulation practices are largely manufacturer-specific rather than functional type-dependent. It suggests that each manufacturer adheres to their own formulation strategies, likely driven by production methods,65 cost considerations,66 and varying degrees of commitment to low-emission technologies.67 When paints are marketed under the same functional category, such as waterproof, emulsion, or acrylic, notable differences in chemical profiles can be observed between manufacturers. As an example, three different bathroom paints that were all labelled for waterproof use exhibited unique VOC fingerprints. Interestingly, each of these bathroom paints showed greater compositional similarity to other products from the same manufacturer, such as kitchen paints, than to other waterproof bathroom paints produced by different manufacturers.The results presented in Fig. 3 show that the most frequently detected VOC across the 2024 decorative wall paint samples is acetone, followed by t-butanol. Acetone was identified in 29 out of 33 samples, while t-butanol was present in 27 out of 33. Acetone is primarily used in waterborne paints as a solvent because it helps create a uniform and homogeneous reaction mixture.68 Additionally, it has a low boiling point and high volatility69 allowing it to evaporate quickly, facilitating rapid drying after application. t-Butanol is likewise used as a cosolvent in modern paint systems.70 When mixed with water, t-butanol can enhance the solubility of hydrophobic substances,71 making it beneficial for use in waterborne paint and coating formulations.
Paint formulations rely on a range of chemical classes, each serving distinct functions that collectively enhance performance. Alcohols and ketones are commonly used as solvents in paint formulations due to their rapid evaporation rates, which facilitate quick drying and efficient film formation.72 Furthermore, esters can contribute to a stable solvent system and help optimise film-forming properties when blended with other solvents.73 Another relevant class is ethers, particularly glycol ethers, which are commonly used in waterborne paints as effective coupling agents, enabling the solubilisation of otherwise water-insoluble components and promoting uniform film formation.74 Alongside these, hydrocarbons, including aromatic hydrocarbons, serve as solvents in paint formulations.75 They are used for their solvency power, which facilitates resin dissolution and enhances coating performance; however, compared to oxygenated solvents, they tend to be more atmospherically reactive, leading to greater potential for photochemical and aerosol formation.76 In addition, acrylate polymers commonly act as binders in paint, forming a strong, flexible, and water-resistant film upon drying.77 These compounds typically represent the primary functional ingredients in a wide range of paint formulations, especially waterborne paints, and play critical roles in achieving desired solvent properties, film formation, and overall coating performance.
Beyond the primary chemical components, paint formulations commonly incorporate various additives and modifiers designed to improve functional performance and maintain formulation stability. Amines function as a pH adjuster and co-dispersant, aiding in emulsion stabilisation and enhancing resin solubility.78 Additionally, siloxanes are used in paints to modify surfaces, predominantly improving slip properties and water repellence.79 Meanwhile, isobutyraldehyde, an aldehyde compound detected in the formulation, is generally used as a chemical intermediate in the synthesis of other compounds rather than as a direct functional additive.80 Among the various compounds identified, chlorinated species were also present, prompting consideration of their potential role and regulatory status. Chlorinated compounds are now largely avoided in mainstream formulations due to environmental and health risks, but they are still used in certain specifications, such as anti-mould applications.81 In addition to the main functional groups, several other lower-abundance compounds were detected that influence paint performance or that may represent unintended secondary byproducts formed during the manufacturing processes. These include neopentyl glycol, texanol, tetrabutyl titanate, and diisobutyl oxalate, each associated with distinct functions in paint formulations. Neopentyl glycol is commonly used as a building block in the synthesis of alkyd and polyester resins, enhancing the durability and weather resistance of coatings, which makes it an important component in many paint formulations.82 Another notable compound is texanol, a widely used coalescing agent in waterborne paints that improves film formation by temporarily plasticising the binder, thereby enhancing scrub resistance and overall appearance.83 Meanwhile, tetrabutyl titanate commonly acts as a catalyst or cross-linking agent, contributing to improved coating hardness and chemical resistance.84 The last compound to consider is diisobutyl oxalate. Although chemically an ester, it is rarely classified alongside commonly used ester solvents or coalescents in paint formulations, likely due to its niche or uncertain function. This compound was detected exclusively in one paint sample, an acrylic eggshell formulation, suggesting its specific or limited use in certain types of paints. Overall, these preliminary results provide a foundational understanding of the chemical compounds and their roles in new paint formulations and highlight that there is still much to explore in this area.
3.3. Shift in VOC composition
To understand how paint formulations have evolved, it is important to first examine the baseline VOC profile of paints from 1990. Fig. 4 shows the VOC speciation of a typical 1990 paint formulation in the UK based on the UK National Atmospheric Emissions Inventory (NAEI-2D3d), focusing on the compounds that contributed to the top 50% of the total VOC emissions by mass, expressed in kilotonnes. Notably, only 19 out of 180 reported VOCs were responsible for 50% of the total emissions. The top-emitting species in the 1990 speciation primarily consisted of long-chain and branched alkanes, cycloalkanes, and aromatic hydrocarbons, where formulations relied heavily on high-volatility hydrocarbon solvents to achieve desired performance characteristics such as rapid drying and good film formation.62 | ||
| Fig. 4 VOC speciation in the 2019 NAEI, although based on a much earlier VOC speciation, dating back to the 1990s. The figure shows the top 50% emission-contributing species by mass. | ||
Decane was reported in the NAEI as the single largest individual VOC emitted from paints in 1990 formulations, contributing approximately 2.24 kilotonnes of emissions. As a straight-chain alkane, decane is a high-volatility, non-polar solvent known for its rapid evaporation rate and ability to dissolve hydrophobic components such as resins and binders.85 Based on Fig. 4, alkanes constituted a major proportion of VOC emissions in the 1990 paint formulation. Their relatively high volatility facilitated rapid solvent evaporation after application, leaving behind a solid film of pigment and binder.86
Fig. 5 illustrates the significant shift in VOC chemical class distribution between those included in the NAEI (2019) and present-day (2024) paint formulations. Alkanes, which comprised nearly half (48.31%) of total paint VOC emissions in 1990, are completely eliminated in 2024 paints, reflecting major changes in solvent use and formulation chemistry. There has been a substantial increase in relative alcohol content in paints, rising from 2.71% in 1990 to 20.78% in 2024, although the overall amount has decreased in line with the reduction in total VOC emissions. Aromatic hydrocarbons have also increased modestly, while esters and ketones have emerged as major components. These compositional changes are a consequence of regulatory measures and the Paints Directive, which mandated reductions in VOC content and prompted widespread reformulation to lower emissions European.24 The use of water-based paints necessitated the increased use of more polar solvents and a switch away from hydrocarbons.87
The primary objective of the Paints Directive was to reduce the overall amount of VOC emitted. The compositional shift to oxygenated solvents facilitated that, allowing higher water content in paints. However, a consequence of the Paints Directive has been not merely a matter of overall emissions reduction, but one of functional replacement. Alkanes, once prevalent for their volatility and solvency, have been replaced by oxygenated compounds, including alcohols, esters, and ketones. These solvents facilitate waterborne formulations due to their polarity, but also generally lead to lower environmental, air quality and health hazards.22,88,89 The emergence of new chemical classes, including acrylates and siloxanes, signals an increase in formulation sophistication, while the decline in the undefined 'Others' category reflects improved chemical specificity and more refined analytical techniques. Altogether, Fig. 5 encapsulates the trajectory of paint formulation over three decades, shaped by regulatory pressure and the transition from solvent-based to water-based formulations.
Fig. 5 also reveals notable differences in VOC chemical class distribution between present-day emulsion and decorative-finish paints, reflecting formulation divergence within the modern product landscape. Emulsion paints are dominated by alcohols, followed by esters and siloxanes, consistent with their water-based nature and reliance on polar, water miscible solvents (Pieters and Mekonnen, 2024). In contrast, decorative-finish paints exhibit a markedly different profile, with aromatic hydrocarbons comprising the largest share (40%), followed by alcohols, esters, ethers, and ketones. The lowest abundance classes also differ, with acrylates least represented in emulsion paints and amines in decorative paints, highlighting variation in additive and coalescent strategies. These findings emphasise that the 2024 formulation does not represent a single, uniform VOC composition, but rather reflects a spectrum of chemical profiles that vary according to paint type, functional requirements, and manufacturer-specific formulation strategies.
3.4. Evaluating the implications of VOC reformulation in paints
To assess the atmospheric implications of VOC reformulation in paints beyond overall mass emission reductions, we evaluated the potential for secondary pollutant formation associated with VOC species present in original NAEI and 2024 formulations. This assessment focused on five key indicators: photochemical ozone creation potential (POCP), photochemical ozone creation potential estimation (POCPE), ozone formation potential (OFP), secondary organic aerosol potential (SOAP), secondary organic aerosol formation potential (SFP) and OH reactivity (OHR). POCP values were taken from Derwent et al. (2007),40 derived using a photochemical trajectory model with the Master Chemical Mechanism. POCPE values, which represent the intrinsic ozone-forming potential of individual VOCs, were calculated using eqn (1) as described in Section 2.3. A normalisation approach, shown as OFP, was obtained by multiplying concentrations with their POCPE value. This value was applied to reflect the real-world impact of VOC profiles better, as each compound contributes differently to the overall composition. Relying on POCPE alone does not account for these varying proportions; therefore, a weighted average called OFP, which is based on emission share was necessary to allow fair comparison across compounds.90 For the 1990 formulation, OFP values were obtained by weighting the POCPE of each compound according to its concentration share, derived from compound-specific VOC data reported in the NAEI inventory mentioned in Section 3.1. In contrast, for the 2024 formulation, where direct concentration data were unavailable, normalisation was based on estimated concentrations using peak area as a proxy. This OFP value was then calculated by weighting the POCPE of each compound by its relative contribution, derived from normalised peak area data. This approach enables a more nuanced evaluation of reformulation effectiveness, accounting not only for changes in the chemical nature of solvents used but also their role in generating secondary pollutants such as ozone and aerosols. As a result, we can robustly compare the potential air quality impacts of historic and modern paint formulations. Secondary organic aerosols formation potential (SFP) was derived by multiplying the concentration by its secondary organic aerosols potential (SOAP) value acquired from Section 2.5, using the same method as for OFP.90 Importantly, this study provides the first known estimation of SFP from modern paint VOC emissions, offering a novel dimension of assessment not previously addressed in paint research.The comparison of secondary pollutant formation potential between 1990 and 2024 paint VOC speciation in Table 1 reveals clear differences in modern formulations. The SOAP of the 2024 speciation is approximately six times lower than that of the 1990 profile, with the average SOAP in 1990 at 10.1 mg m−3 compared to 1.7 mg m−3 in 2024. This reflects a substantial reduction in the aerosol formation potential from paint-related VOCs over paint reformulation. Furthermore, POCPE values in the 2024 speciation are lower than in 1990, with averages of 40.3 and 34.9, respectively. As POCPE values are compound-specific rather than normalised, this decrease reflects a shift in the underlying chemical composition. In particular, oxygenated compounds such as acetone and t-butanol, which exhibit substantially lower POCP values than alkanes, now contribute more prominently to paint formulations. This contrasts with earlier formulations, where highly reactive species like alkanes dominated and contributed strongly to ozone formation. As a result, the reformulation of paints has not only reduced total VOC mass but has also lowered their SOAP and POCPE, contributing to improved air quality.
| VOC species | POCP | POCPE | OFP | SOAP (mg m−3) | SFP (mg m−3) | OHR (s−1) |
|---|---|---|---|---|---|---|
| Speciation 1990 | ||||||
| Decane | 36.0 | 31.9 | 90.4 | 16.5 | 46.7 | 15.3 |
| Nonane | 34.0 | 33.6 | 55.0 | 8.6 | 14.0 | 7.8 |
| 2-Methylnonane | 35.0 | 26.9 | 16.5 | 3.6 | 2.2 | 2.4 |
| 3-Methylnonane | 39.0 | 26.9 | 19.1 | 4.1 | 3.0 | 2.8 |
| 3-Methyldecane | 36.0 | 31.7 | 16.3 | 3.3 | 1.7 | 3.3 |
| 4-Methyldecane | 36.0 | 30.7 | 26.5 | 5.5 | 4.8 | 5.2 |
| 3-Ethyl-2-methylheptane | 34.0 | 26.9 | 18.7 | 0.8 | 0.6 | 2.7 |
| 2,6-Dimethyloctane | 36.0 | 38.2 | 22.0 | 3.0 | 1.7 | 3.7 |
| Undecane | 36.0 | 30.7 | 45.5 | 9.5 | 14.0 | 9.0 |
| C11 alkanes | 35.0 | 30.2 | 15.0 | 3.2 | 1.6 | 3.0 |
| Propylcyclohexane | 60.0 | 38.7 | 22.8 | 6.1 | 3.6 | 3.9 |
| C10 cycloalkanes | 59.0 | 38.3 | 20.6 | 6.2 | 3.3 | 2.6 |
| (1-Methylpropyl)cyclohexane | 60.0 | 22.6 | 11.2 | 5.7 | 2.8 | 1.5 |
| Limonene | 71.0 | 106.8 | 228.1 | 69.0 | 147.3 | 173.3 |
| 1,2,4-Trimethylbenzene | 110.0 | 90.8 | 73.8 | 6.3 | 5.1 | 12.8 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Speciation 2024 | ||||||
| Acetone | 6.0 | 3.7 | 14.4 | 1.6 | 6.3 | 0.3 |
| t-Butanol | 2.0 | 10.2 | 25.7 | 1.4 | 3.5 | 1.4 |
| Isobutanol | 36.0 | 49.2 | 26.9 | 0.3 | 0.2 | 2.6 |
| Cyclohexanol | 45.0 | 50.9 | 9.0 | 0.1 | 0.0 | 1.5 |
| Texanol | — | 17.9 | 13.7 | 6.8 | 5.2 | 3.8 |
| Isobutyl lactate | — | 23.3 | 28.3 | 1.3 | 1.6 | 4.2 |
| Methyl methacrylate | — | 48.4 | 18.5 | 0.3 | 0.1 | 7.6 |
| Methyl isobutyrate | — | 13.9 | 3.5 | 0.2 | 0.0 | 0.2 |
| Butyl acetate | 26.0 | 27.6 | 5.0 | 0.2 | 0.0 | 0.5 |
| Butyl cellosolve | 45.0 | 46.6 | 50.9 | 0.9 | 1.0 | 16.1 |
| o-Xylene | 78.0 | 66.8 | 21.4 | 1.8 | 0.6 | 2.1 |
| Ethylbenzene | 46.0 | 38.6 | 8.7 | 5.9 | 1.3 | 0.8 |
| Propylbenzene | 38.0 | 30.9 | 1.8 | 0.9 | 0.0 | 0.2 |
| Isopropylbenzene | 32.0 | 32.4 | 5.8 | 2.7 | 0.5 | 0.6 |
| Isopropylamine | — | 63.9 | 206.2 | 1.4 | 4.5 | 63.5 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Average value | ||||||
| Speciation 1990 | 40.3 | 45.4 | 10.1 | 16.8 | 16.6 | |
| Speciation 2024 | 34.9 | 29.3 | 1.7 | 1.7 | 7.0 | |
3.5. Atmospheric modelling of speciation impacts
Atmospheric modelling was used to compare the oxidation behaviour of decane and t-butanol, representing two key solvents commonly found in the 1990 and 2024 speciation profiles, respectively. Initial concentrations of 4.92 × 1010 molecules cm−3 for decane and 9.45 × 1010 molecules cm−3 for t-butanol were used, derived from the assumptions detailed in the methodology section. The modelling was performed using INCHEM-Py, with settings listed in detail in the methodology section. In these simulations, no indoor lighting or sunlight was present, so photolytic processes did not contribute to the chemical transformations. The absence of photolytic processes in the model configuration means these results are not directly transferable to outdoor urban conditions, where sunlight drives additional oxidation pathways, and oxidant levels vary diurnally. These results should therefore be interpreted as indicating relative differences in oxidation behaviour between the two compounds, rather than as absolute predictions of ambient atmospheric concentrations. However, atmospheric oxidants were introduced in a constant setting throughout the model run. Fig. 6 shows the decline in ambient concentrations of both compounds over a seven-day simulation under identical initial conditions. Decane decays much more rapidly, reaching zero just before six days, while t-butanol remains around 6.8 µg m−3 at the same time point. The sharp decline in decane reflects its higher atmospheric reactivity, due to efficient oxidation by hydroxyl radicals.91 Although rapid degradation may imply a short atmospheric lifetime,92 it also indicates a greater potential for contributing to ozone and radical formation,93 highlighting its role in driving photochemical smog.94 In contrast, t-butanol exhibits slower degradation, suggesting lower reactivity and a reduced contribution to short-term ozone production,95 despite its longer persistence in the atmosphere. Additionally, the lower reactivity of t-butanol96 can lead to less formation of reactive intermediates and secondary pollutants, which may reduce its overall impact on air quality and human health compared to more reactive solvents like decane.97 | ||
| Fig. 6 Atmospheric decay profiles of decane (as 1990 solvent) and t-butanol (as 2024 solvent) over 7 days under identical conditions. | ||
Fig. 7 compares the secondary formation of formaldehyde (HCHO) as a result of the atmospheric oxidation of decane and t-butanol by OH, O3, NO3, and NOx. HCHO formation from decane begins approximately 20 hours into the simulation and subsequently exceeds that from t-butanol. Decane reaches its maximum HCHO concentration slightly later than t-butanol, peaking after approximately 4 days compared with t-butanol on day 3. The peak HCHO concentration from decane is 2.22 µg m−3, approximately five times higher than that produced from t-butanol, which reaches 0.46 µg m−3. Detailed model run outputs are provided in Table S3 of the SI. This difference highlights distinct oxidation behaviours: t-butanol likely follows a more direct and faster pathway to HCHO formation due to its oxygenated structure and smaller carbon number, whereas the longer carbon chain in decane leads to extended reaction chains before significant HCHO formation. Although decane ultimately yields more HCHO, the earlier formation and peak from t-butanol may influence the timing and location of secondary pollutant formation. These variations underscore how both the magnitude and kinetics of secondary chemistry differ by solvent type,98 potentially impacting exposure peaks. These findings suggest that the shift toward solvents like t-butanol, commonly used in newer waterborne paint formulations, may offer some atmospheric advantages over traditional solvents such as decane. In particular, t-butanol exhibits slower reactivity and lower total HCHO production, with a more gradual and earlier onset of secondary chemistry. This could reduce the peak intensity and spatial concentration of secondary pollutants such as formaldehyde. However, given the complexity of atmospheric chemistry and the limited scope of this comparison, broader evaluations across more compounds are necessary before definitive conclusions can be made regarding the overall atmospheric benefit of waterborne paint solvents.
 | ||
| Fig. 7 Simulated secondary formaldehyde (HCHO) formation from decane (as 1990 solvent) and t-butanol (as 2024 solvent) under atmospheric conditions. | ||
4 Conclusions
This study presents evidence that decorative paints have undergone substantial reformulation over the past three decades, both in terms of total VOC emissions and chemical composition. Compared to other domestic sources, the emission trajectory of paints is unique, highlighting this sector as both a major historical contributor and a clear example of successful emission mitigation. Hydrocarbon-based solvents such as alkanes, which dominated formulations in the 1990s, have been largely replaced by more oxygenated compounds, including alcohols, ketones, and esters. These changes have been primarily driven by regulatory measures, particularly the European Union Paints Directive, and have resulted in an estimated 60% reduction in mass of paint-related VOC emissions since 2004.A key contribution of this work is the quantification of secondary organic aerosol formation potential from paint VOC emissions, an aspect not previously addressed in paint research, and not a policy motivation at the time of the Paint Directive. The SOA formation potential of modern paint formulations is approximately six times lower than that of the 1990 profile, indicating a substantial reduction in the likelihood of fine particle formation from modern paint use. This assessment provides an important advancement in understanding the wider air quality implications of product reformulation.
Despite these advances, modern formulations also include a more diverse range of oxygenated and nitrogen-containing compounds, some of which exhibit higher ozone formation potential. Although total VOC emissions have declined, the increase in photochemical reactivity observed in certain formulations suggests that regulatory targets based solely on VOC mass may not fully capture their atmospheric or health impacts. This highlights the need for evaluation frameworks that consider both emission quantity and secondary pollutant formation potential. Importantly, the findings presented here are based on standardised comparative analysis of paint compositions and associated reactivity metrics. In practice, the real-world impact of VOC emissions will depend strongly on which specific paint formulations are most widely used. At present, such paint-use data are not publicly available, making it difficult to accurately scale theoretical estimates to market-level exposure or air quality impacts. Addressing this gap would enable more representative assessments and should be considered a key area for future research. One particularly practical approach would be for manufacturers to report their sales and production data directly to the NAEI, which would be both more cost-effective and more reliable than market surveys, consumer behaviour studies, or third-party sales data analysis. Future work could also look at long-term monitoring of VOC emissions from modern paints under real-world application conditions, and explore how usage data could be incorporated into emission inventories to give a more accurate picture of paint-related air quality impacts.
Overall, these results emphasise the need for continued monitoring of modern paint formulation and the incorporation of secondary pollutant metrics into both regulatory and scientific evaluations. The implementation of the EU Paints Directive, originally designed to reduce VOC emissions impacting outdoor air quality, has been particularly effective in driving reformulation, leading to substantial reductions in VOC emissions from decorative paints over the past three decades. Given the evolving composition of modern paint formulations, it is important to assess not only their outdoor air quality benefits but also their potential impacts on indoor air. As a highly used product with a history of rapid and widespread reformulation, decorative paints remain a valuable case study for examining the combined influence of policy, industrial innovation, and compositional change in improving air quality while maintaining product performance. Crucially, the updated VOC profiles presented here provide a more accurate basis for improving emission inventories, which currently rely on outdated speciation data, and will enable more representative assessments of paint-related air quality impacts in future.
Author contributions
Lidia Kristia Alfanti: writing – original draft, writing – review & editing, investigation, formal analysis, visualization, validation, resources, project administration, methodology. Amber May Yeoman: methodology, writing – review & editing, resources. Andrew Robert Rickard: validation, writing – review & editing. David Shaw: software, validation, writing – review & editing. Martyn William Ward: methodology, validation. Sarah Julia Moller: supervision, writing – review & editing, funding acquisition. Alastair Charles Lewis: conceptualization, writing – original draft, writing – review & editing, supervision, funding acquisition.Conflicts of interest
There are no declarations of interest to disclose.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ea00134j.Acknowledgements
This work was supported by the NERC Panorama Doctoral Training Partnership (DTP) under grant NE/S007458/1, which funded the studentship of Lidia Kristia Alfanti. Additional CASE funding for her studentship was provided by Defra. Amber May Yeoman was supported by NCAS (grant no. R8/H12/83/010). Sarah Julia Moller acknowledges Defra for funding. Andrew Robert Rickard, Alastair Charles Lewis, David Shaw, and Sarah Julia Moller acknowledge funding from NCAS.References
- J.-L. Mai, W.-W. Yang, Y. Zeng, Y.-F. Guan and S.-J. Chen, Volatile organic compounds (VOCs) in residential indoor air during interior finish period: Sources, variations, and health risks, Hyg. Environ. Health Adv., 2024, 9, 100087 CrossRef CAS.
- E. D. Vicente, A. M. Vicente, M. Evtyugina, F. I. Oduber, F. Amato, X. Querol and C. Alves, Impact of wood combustion on indoor air quality, Sci. Total Environ., 2020, 705, 135769 CrossRef CAS PubMed.
- N. Hobeika, C. García-Sánchez and P. M. Bluyssen, Assessing Indoor Air Quality and Ventilation to Limit Aerosol Dispersion—Literature Review, 2023, p. 13 Search PubMed.
- A. M. Yeoman, M. Shaw, N. Carslaw, T. Murrells, N. Passant and A. C. Lewis, Simplified speciation and atmospheric volatile organic compound emission rates from non-aerosol personal care products, Indoor Air, 2020, 30, 459–472 CrossRef CAS PubMed.
- A. Turner, Paint particles in the marine environment: An overlooked component of microplastics, Water Res.:X, 2021, 12, 100110 CAS.
- R. Tanzer-Gruener, P. E. Rajan, L. D. Dugan, M. E. Bier, A. L. Robinson and A. A. Presto, Watching Paint Dry: Organic Vapor Emissions from Architectural Coatings and their Impact on Secondary Organic Aerosol Formation, Environ. Sci. Technol., 2022, 56, 11236–11245 CrossRef CAS PubMed.
- S. Dimitroulopoulou, M. R. Dudzińska, L. Gunnarsen, L. Hägerhed, H. Maula, R. Singh, O. Toyinbo and U. Haverinen-Shaughnessy, Indoor air quality guidelines from across the world: An appraisal considering energy saving, health, productivity, and comfort, Environ. Int., 2023, 178, 108127 CrossRef CAS PubMed.
- A. M. Jiménez-López and G. A. Hincapié-Llanos, Identification of factors affecting the reduction of VOC emissions in the paint industry: Systematic literature review - SLR, Prog. Org. Coat., 2022, 170, 106945 CrossRef.
- S. Barry and B. O'Regan, A refined method for the calculation of the Non-Methane Volatile Organic Compound emission estimate from Domestic Solvent Usage in Ireland from 1992 to 2014 – A case study for Ireland, Atmos. Environ., 2016, 138, 15–21 CrossRef CAS.
- A. C. Lewis, J. R. Hopkins, D. C. Carslaw, J. F. Hamilton, B. S. Nelson, G. Stewart, J. Dernie, N. Passant and T. Murrells, An increasing role for solvent emissions and implications for future measurements of volatile organic compounds, Philos. Trans. R. Soc., A, 2020, 378, 20190328 CrossRef CAS PubMed.
- A. Karwasz, F. Osiński, W. Kaczmarek, K. Furmaniak and I. Rojek, The Influence of Polylactic Acid Filament Moisture Content on Dust Emissions in 3D Printing Process, 2024, vol. 24 Search PubMed.
- E. David and V. C. Niculescu, Volatile Organic Compounds (VOCs) as Environmental Pollutants: Occurrence and Mitigation Using Nanomaterials, Int. J. Environ. Res. Public Health, 2021, 18, 13147 CrossRef CAS PubMed.
- G. Hernandez, S. L. Wallis, I. Graves, S. Narain, R. Birchmore and T.-A. Berry, The effect of ventilation on volatile organic compounds produced by new furnishings in residential buildings, Atmos. Environ.:X, 2020, 6, 100069 CAS.
- P. J. Irga, G. Mullen, R. Fleck, S. Matheson, S. J. Wilkinson and F. R. Torpy, Volatile organic compounds emitted by humans indoors– A review on the measurement, test conditions, and analysis techniques, Build. Environ., 2024, 255, 111442 CrossRef.
- K. Rumchev, H. Brown and J. Spickett, Volatile organic compounds: do they present a risk to our health?, Rev. Environ. Health, 2007, 22, 39–55 CAS.
- Z. Mo, S. Lu and M. Shao, Volatile organic compound (VOC) emissions and health risk assessment in paint and coatings industry in the Yangtze River Delta, China, Environ. Pollut., 2021, 269, 115740 CrossRef CAS PubMed.
- M. Li, Y. Wan, X. Qian, A. Wang, G. Mahai, Z. He, Y. Li, S. Xu and W. Xia, Urinary metabolites of multiple volatile organic compounds among pregnant women across pregnancy: Variability, exposure characteristics, and associations with selected oxidative stress biomarkers, Environ. Int., 2023, 173, 107816 CrossRef CAS PubMed.
- K. Toma, S. Suzuki, T. Arakawa, Y. Iwasaki and K. Mitsubayashi, External ears for non-invasive and stable monitoring of volatile organic compounds in human blood, Sci. Rep., 2021, 11, 10415 CrossRef CAS PubMed.
- S. Cakmak, C. Cole, C. Hebbern, J. Andrade and R. Dales, Associations between blood volatile organic compounds, and changes in hematologic and biochemical profiles, in a population-based study, Environ. Int., 2020, 145, 106121 CrossRef CAS PubMed.
- N. Carslaw, G. Bekö, S. Langer, C. Schoemaecker, V. G. Mihucz, M. Dudzinska, P. Wiesen, S. Nehr, K. Huttunen, X. Querol and D. Shaw, A new framework for indoor air chemistry measurements: Towards a better understanding of indoor air pollution, Indoor Environ., 2024, 1, 100001 CrossRef.
- S. Wang, H. M. Ang and M. O. Tade, Volatile organic compounds in indoor environment and photocatalytic oxidation: State of the art, Environ. Int., 2007, 33, 694–705 CrossRef CAS PubMed.
- A. Schieweck and M.-C. Bock, Emissions from low-VOC and zero-VOC paints – Valuable alternatives to conventional formulations also for use in sensitive environments?, Build. Environ., 2015, 85, 243–252 CrossRef.
- W. F. Hunt and E. J. Lillis, 1980 Ambient Assessment-Air Portion, US Environmental Protection, Office of Air Quality Planning and Standards, 1981 Search PubMed.
- E. Parliament, Directive 2004/42/Ce Of The European Parliament And Of The Council of 21 april 2004 on the limitation of emissions of volatile organic compounds due to the use of organic solvents in certain paints and varnishes and vehicle refinishing products and amending directive 1999/13/EC, 2004.
- T. C. o. t. E. Union, Council Directive 1999/13/EC of 11 March 1999 on the Limitation of Emissions of Volatile Organic Compounds due to the Use of Organic Solvents in Certain Activities and Installations, 1999.
- T. C. o. t. E. Communities, Commission Decision of 13 August 2008 Establishing the Ecological Criteria for the Award of the Community Eco-Label to Outdoor Paints and Varnishes (Notified Under Document Number C(2008) 4452) (Text With EEA Relevance) (2009/543/EC), 2009.
- T. C. o. t. E. Communities, Commission Decision of 13 August 2008 Establishing the Ecological Criteria for the Award of the Community Eco-Label to Indoor Paints and Varnishes (Notified Under Document Number C(2008) 4453) (Text With EEA Relevance) (2009/544/EC), 2009.
- T. E. commission, Commission Decision of 28 May 2014 Establishing the Ecological Criteria for the Award of the EU Ecolabel for Indoor and Outdoor Paints and Varnishes (Notified Under Document C(2014) 3429) (Text With EEA Relevance) (2014/312/EU), 2014.
- K. Ross, J. F. Chmiel and T. Ferkol, The impact of the Clean Air Act, J. Pediatr., 2012, 161, 781–786 CrossRef PubMed.
- J. Geldermann, H. Schollenberger, O. Rentz, G. Huppes, L. van Oers, C. France, B. Nebel, R. Clift, A. Lipkova, S. Saetta, U. Desideri and T. May, An integrated scenario analysis for the metal coating sector in Europe, Technol. Forecast. Soc. Change, 2007, 74, 1482–1507 CrossRef.
- J. S. Kougoulis, R. Kaps, B. Walsh, K. Bojczuk and T. Crichton, Revision of EU European Ecolabel and Development of EU Green Public Procurement Criteria for Indoor and Outdoor Paints and Varnishes, Green Public Procurement Background Report, European Commission, Joint Research Centre (JRC); Seville: Institute for Prospective Technological Studies (IPTS), 2012 Search PubMed.
- K. Pieters and T. H. Mekonnen, Progress in waterborne polymer dispersions for coating applications: commercialized systems and new trends, RSC Sustain., 2024, 2, 3704–3729 RSC.
- A. Giampieri, J. Ling-Chin, Z. Ma, A. Smallbone and A. P. Roskilly, A review of the current automotive manufacturing practice from an energy perspective, Appl. Energy, 2020, 261, 114074 CrossRef.
- L. Pilon, D. Day, H. Maslen, O. P. J. Stevens, N. Carslaw, D. R. Shaw and H. F. Sneddon, Development of a solvent sustainability guide for the paints and coatings industry, Green Chem., 2024, 26, 9697–9711 RSC.
- J. Palmisani, A. W. Nørgaard, V. Kofoed-Sørensen, P. A. Clausen, G. de Gennaro and P. Wolkoff, Formation of ozone-initiated VOCs and secondary organic aerosol following application of a carpet deodorizer, Atmos. Environ., 2020, 222, 117149 CrossRef CAS.
- P. N. Grenga, M. J. Gallagher, M. E. McGahan, D. M. Raymond and R. Priefer, Assessment of Airborne Total Volatile Organic Compounds of Niagara Falls Residences as Compared to Resident Lifestyle, Indoor Built Environ., 2010, 20, 226–231 CrossRef.
- A. C. Heeley-Hill, S. K. Grange, M. W. Ward, A. C. Lewis, N. Owen, C. Jordan, G. Hodgson and G. Adamson, Frequency of use of household products containing VOCs and indoor atmospheric concentrations in homes, Environ. Sci.: Processes Impacts, 2021, 23, 699–713 RSC.
- R. G. Derwent, M. E. Jenkin and S. M. Saunders, Photochemical ozone creation potentials for a large number of reactive hydrocarbons under European conditions, Atmos. Environ., 1996, 30, 181–199 CrossRef CAS.
- M. E. Jenkin, R. G. Derwent and T. J. Wallington, Photochemical ozone creation potentials for volatile organic compounds: Rationalization and estimation, Atmos. Environ., 2017, 163, 128–137 CrossRef CAS.
- R. G. Derwent, M. E. Jenkin, N. R. Passant and M. J. Pilling, Reactivity-based strategies for photochemical ozone control in Europe, Environ. Sci. Pol., 2007, 10, 445–453 CrossRef CAS.
- M. R. McGillen, W. P. L. Carter, A. Mellouki, J. J. Orlando, B. Picquet-Varrault, and T. J. Wallington, Database for the kinetics of the gas-phase atmospheric reactions of organic compounds, https://data.eurochamp.org/sar-database/, accessed 21 September, 2025.
- R. E. Dunmore, J. R. Hopkins, R. T. Lidster, J. D. Lee, M. J. Evans, A. R. Rickard, A. C. Lewis and J. F. Hamilton, Diesel-related hydrocarbons can dominate gas phase reactive carbon in megacities, Atmos. Chem. Phys., 2015, 15, 9983–9996 CrossRef CAS.
- J. R. Odum, T. Hoffmann, F. Bowman, D. Collins, R. C. Flagan and J. H. Seinfeld, Gas/Particle Partitioning and Secondary Organic Aerosol Yields, Environ. Sci. Technol., 1996, 30, 2580–2585 CrossRef CAS.
- R. G. Derwent, M. E. Jenkin, S. R. Utembe, D. E. Shallcross, T. P. Murrells and N. R. Passant, Secondary organic aerosol formation from a large number of reactive man-made organic compounds, Sci. Total Environ., 2010, 408, 3374–3381 CrossRef CAS PubMed.
- M. B. Humes, M. Wang, S. Kim, J. E. Machesky, D. R. Gentner, A. L. Robinson, N. M. Donahue and A. A. Presto, Limited Secondary Organic Aerosol Production from Acyclic Oxygenated Volatile Chemical Products, Environ. Sci. Technol., 2022, 56, 4806–4815 CrossRef CAS PubMed.
- A. Mutzel, Y. Zhang, O. Böge, M. Rodigast, A. Kolodziejczyk, X. Wang and H. Herrmann, Importance of secondary organic aerosol formation of α-pinene, limonene, and m-cresol comparing day- and nighttime radical chemistry, Atmos. Chem. Phys., 2021, 21, 8479–8498 CrossRef CAS.
- L. Li, L. Dong, X. Liu, Y. Guo and Y. Wang, Selective production of ethylbenzene from lignin oil over FeOx modified Ru/Nb2O5 catalyst, Appl. Catal., B, 2020, 260, 118143 CrossRef CAS.
- L. Zhao, Z. Cheng, L. Ye, F. Zhang, L. Zhang, F. Qi and Y. Li, Experimental and kinetic modeling study of premixed o-xylene flames, Proc. Combust. Inst., 2015, 35, 1745–1752 CrossRef CAS.
- J. F. Hunter, A. J. Carrasquillo, K. E. Daumit and J. H. Kroll, Secondary organic aerosol formation from acyclic, monocyclic, and polycyclic alkanes, Environ. Sci. Technol., 2014, 48, 10227–10234 CrossRef CAS PubMed.
- D. R. Cocker Iii, B. T. Mader, M. Kalberer, R. C. Flagan and J. H. Seinfeld, The effect of water on gas–particle partitioning of secondary organic aerosol: II. m-xylene and 1,3,5-trimethylbenzene photooxidation systems, Atmos. Environ., 2001, 35, 6073–6085 CrossRef.
- D. R. Shaw, T. J. Carter, H. L. Davies, E. Harding-Smith, E. C. Crocker, G. Beel, Z. Wang and N. Carslaw, INCHEM-Py v1.2: a community box model for indoor air chemistry, Geosci. Model Dev., 2023, 16, 7411–7431 CrossRef CAS.
- D. Shaw and N. Carslaw, INCHEM-Py: An open source Python box model for indoor air chemistry, J. Open Source Softw., 2021, 6, 3224 CrossRef.
- R. Atkinson, Atmospheric chemistry of VOCs and NOx, Atmos. Environ., 2000, 34, 2063–2101 CrossRef CAS.
- J. H. Kroll, N. L. Ng, S. M. Murphy, R. C. Flagan and J. H. Seinfeld, Secondary organic aerosol formation from isoprene photooxidation under high-NOx conditions, Geophys. Res. Lett., 2005, 32, L18808 CrossRef.
- B. Yang, H. Zhang, Y. Wang, P. Zhang, J. Shu, W. Sun and P. Ma, Experimental and theoretical studies on gas-phase reactions of NO3 radicals with three methoxyphenols: Guaiacol, creosol, and syringol, Atmos. Environ., 2016, 125, 243–251 CrossRef CAS.
- A. C. Lewis, J. D. Allan, D. Carruthers, D. Carslaw, G. W. Fuller, R. M. Harrison, M. R. Heal, E. Nemitz, C. E. Reeves and N. Carslaw, Indoor Air Qual., 2022, 142 Search PubMed.
- A. M. Yeoman and A. C. Lewis, Global emissions of VOCs from compressed aerosol products, Elementa: Sci. Anthropocene, 2021, 9, 00177 Search PubMed.
- C. H. Halios, C. Landeg-Cox, S. D. Lowther, A. Middleton, T. Marczylo and S. Dimitroulopoulou, Chemicals in European residences – Part I: A review of emissions, concentrations and health effects of volatile organic compounds (VOCs), Sci. Total Environ., 2022, 839, 156201 CrossRef CAS PubMed.
- E. Chemiewinkel, Ireland and WIMM, Study on the Potential for Reducing Emissions of Volatile Organic Compounds (VOC) Due to the Use of Decorative Paints and Varnishes for Professional and Non-professional Use, 2000 Search PubMed.
- M. Watson and E. Shove, Doing it yourself? Products, competence and meaning in the practices of DIY, European Sociological Association Conference, 2005 Search PubMed.
- J. E. Bugaj and R. B. Dorshow, Absence of developmental or reproductive toxicity in rats for MB-102, a fluorescent tracer agent for point-of-care measurement of glomerular filtration rate, Regul. Toxicol. Pharmacol., 2022, 131, 105158 CrossRef CAS PubMed.
- C. E. Stockwell, M. M. Coggon, G. I. Gkatzelis, J. Ortega, B. C. McDonald, J. Peischl, K. Aikin, J. B. Gilman, M. Trainer and C. Warneke, Volatile organic compound emissions from solvent- and water-borne coatings – compositional differences and tracer compound identifications, Atmos. Chem. Phys., 2021, 21, 6005–6022 CrossRef CAS.
- L. Feng, H. Yan, C. Dai, W. Xu, F. Gu, F. Zhang, T. Li, J. Xian, X. He, Y. Yu, M. Ma, F. Wang and Z. He, The systematic exploration of cadmium-accumulation characteristics of maize kernel in acidic soil with different pollution levels in China, Sci. Total Environ., 2020, 729, 138972 CrossRef CAS PubMed.
- U. Government, The Volatile Organic Compounds in Paints, Varnishes and Vehicle Refinishing Products Regulations 2012, 2012.
- T. W. S. Panjaitan, P. Dargusch, D. Wadley and A. A. Aziz, A study of management decisions to adopt emission reduction measures in heavy industry in an emerging economy, Sci. Rep., 2023, 13, 1413 CrossRef CAS PubMed.
- Q. Wan, X. Zhao, H. Liu, H. Dinçer and S. Yüksel, Assessing the New Product Development Process for the Industrial Decarbonization of Sustainable Economies, Sage Open, 2022, 12, 21582440211067231 CrossRef.
- J. Ma, Carbon emissions and low-carbon innovation in firms, PLoS One, 2024, 19, e0312759 CrossRef CAS PubMed.
- H. Honarkar, Waterborne Polyurethanes: A Review, J. Dispersion Sci. Technol., 2017, 39, 507–516 CrossRef.
- H. Moradi, H. Bahmanyar and H. Azizpour, Molecular simulation of liquid-liquid extraction of acetic acid and acetone from water in the presence of nanoparticles based on prediction of solubility parameters, Heliyon, 2024, 10, e38086 CrossRef CAS PubMed.
- A. S. Bale and J. S. Lee, An overview of butanol-induced developmental neurotoxicity and the potential mechanisms related to these observed effects, Neurotoxicol. Teratol., 2016, 53, 33–40 CrossRef CAS PubMed.
- B. S. Bhatnagar, J. Sonje, E. Shalaev, S. W. H. Martin, D. L. Teagarden and R. Suryanarayanan, A refined phase diagram of the tert-butanol–water system and implications on lyophilization process optimization of pharmaceuticals, Phys. Chem. Chem. Phys., 2020, 22, 1583–1590 RSC.
- R. Talbert, Paint Technology Handbook, CRC press, 2007 Search PubMed.
- A. A. Tracton, Coatings Technology Handbook, CRC press, 2005 Search PubMed.
- C. L. Rodriguez, J. Weathers, B. Corujo and P. Peterson, Formulating water-based systems with propylene-oxide-based glycol ethers, J. Coating Technol., 2000, 72, 67–72 CrossRef CAS.
- Z. He, Y. Sun, C. Zhang, J. Zhang, S. Liu, K. Zhang and M. Lan, Recent advances of solvent-engineered carbon dots: A review, Carbon, 2023, 204, 76–93 CrossRef CAS.
- T. Paterson, Hydrocarbon solvents for the future, Surf. Coating. Int., 1999, 82, 177–180 CrossRef CAS.
- C. Gao, S. Han, D. Zhang, B. Wang, C. Wang, Y. Wu and Y. Liu, A facile preparation of UV-cured films from waterborne unsaturated polyester via click reaction, Prog. Org. Coat., 2018, 124, 232–239 CrossRef CAS.
- J. R. Fried, Polymer Science and Technology, Pearson Education, 2014 Search PubMed.
- K. Kumari, A. Singh and D. Marathe, Cyclic volatile methyl siloxanes (D4, D5, and D6) as the emerging pollutants in environment: environmental distribution, fate, and toxicological assessments, Environ. Sci. Pollut. Res., 2024, 31, 38681–38709 CrossRef CAS PubMed.
- L. Guo, L. Sun and Y.-X. Huo, Toward bioproduction of oxo chemicals from C1 feedstocks using isobutyraldehyde as an example, Biotechnol. Biofuels Bioprod., 2022, 15, 80 CrossRef CAS PubMed.
- S. Rogawansamy, S. Gaskin, M. Taylor and D. Pisaniello, An evaluation of antifungal agents for the treatment of fungal contamination in indoor air environments, Int. J. Environ. Res. Public Health, 2015, 12, 6319–6332 CrossRef CAS PubMed.
- J. van Haveren, E. A. Oostveen, F. Miccichè, B. A. J. Noordover, C. E. Koning, R. A. T. M. van Benthem, A. E. Frissen and J. G. J. Weijnen, Resins and additives for powder coatings and alkyd paints, based on renewable resources, J. Coat. Technol. Res., 2007, 4, 177–186 CrossRef CAS.
- V. Divry, L. Jacomine, V. Le Houérou, D. Collin, C. Gauthier and Y. Holl, Influence of formulation on friction properties of latex films, Prog. Org. Coat., 2017, 113, 189–199 CrossRef CAS.
- L. Xu, Z. Chen, F. Huang, Y. Zuo, X. Shi and X. Zhou, Corrosion Resistance of Waterborne Epoxy Resin Coating Cross-Linked by Modified Tetrabutyl Titanate, Scanning, 2020, 2020, 1392385 Search PubMed.
- E. Uhde and T. Salthammer, Impact of reaction products from building materials and furnishings on indoor air quality—A review of recent advances in indoor chemistry, Atmos. Environ., 2007, 41, 3111–3128 CrossRef CAS.
- P. C. Painter and M. M. Coleman, Essentials of Polymer Science and Engineering, DEStech Publications, Inc, 2009 Search PubMed.
- L. A. Brown, G. Fisk, M. A. Cohen, A. Crollard and S. G. Whittaker, Using an alternatives assessment framework to evaluate waterborne versus solventborne basecoats used in automotive refinishing, Integr. Environ. Assess. Manage., 2022, 18, 1101–1113 CrossRef CAS PubMed.
- L. Ernstgård, A. Löf, G. Wieslander, D. Norbäck and G. Johanson, Acute effects of some volatile organic compounds emitted from water-based paints, J. Occup. Environ. Med., 2007, 49, 880–889 CrossRef PubMed.
- J. C. S. Chang, R. Fortmann, N. Roache and H.-C. Lao, Evaluation of Low-VOC Latex Paints, Indoor Air, 1999, 9, 253–258 CrossRef CAS PubMed.
- P. Chen, X. Zhao, O. Wang, M. Shao, X. Xiao, S. Wang and Q. g. Wang, Characteristics of VOCs and their Potentials for O3 and SOA Formation in a Medium-sized City in Eastern China, Aerosol Air Qual. Res., 2022, 22, 210239 CrossRef CAS.
- R. L. Thompson, S. A. Montzka, M. K. Vollmer, J. Arduini, M. Crotwell, P. B. Krummel, C. Lunder, J. Mühle, S. O'Doherty, R. G. Prinn, S. Reimann, I. Vimont, H. Wang, R. F. Weiss and D. Young, Estimation of the atmospheric hydroxyl radical oxidative capacity using multiple hydrofluorocarbons (HFCs), Atmos. Chem. Phys., 2024, 24, 1415–1427 CrossRef CAS.
- N. J. Bunce, J. S. Nakai and M. Yawching, Estimates of the tropospheric lifetimes of short- and long-lived atmospheric pollutants, J. Photochem. Photobiol., A, 1991, 57, 429–439 CrossRef CAS.
- W. Ma, Z. Feng, J. Zhan, Y. Liu, P. Liu, C. Liu, Q. Ma, K. Yang, Y. Wang, H. He, M. Kulmala, Y. Mu and J. Liu, Influence of photochemical loss of volatile organic compounds on understanding ozone formation mechanism, Atmos. Chem. Phys., 2022, 22, 4841–4851 CrossRef CAS.
- A. Shiroudi, T. Kurtén and J. Czub, Mechanistic insights and atmospheric implications of the degradation reaction of 3-methoxy-1-propanol by reaction with hydroxyl radicals and identification of the end products in the presence of O2/NO, Sci. Rep., 2025, 15, 1038 CrossRef CAS PubMed.
- L. I. Kleinman, P. H. Daum, Y.-N. Lee, L. J. Nunnermacker, S. R. Springston, J. Weinstein-Lloyd and J. Rudolph, Ozone production efficiency in an urban area, J. Geophys. Res.:Atmos., 2002, 107, 2321 CrossRef.
- R. L. Brainard and R. J. Madix, Enhanced stability of t-butanol reaction intermediates on oxygen covered Cu (110): Cleavage of unactivated C–H bonds on metal surfaces, Surf. Sci., 1989, 214, 396–406 CrossRef CAS.
- O. Herbinet, B. Husson, M. Ferrari, P.-A. Glaude and F. Battin-Leclerc, Low temperature oxidation of benzene and toluene in mixture with n-decane, Proc. Combust. Inst., 2013, 34, 297–305 CrossRef CAS PubMed.
- C. Mapelli, J. V. Schleicher, A. Hawtin, C. D. Rankine, F. C. Whiting, F. Byrne, C. R. McElroy, C. Roman, C. Arsene, R. I. Olariu, I. G. Bejan and T. J. Dillon, Atmospheric breakdown chemistry of the new “green” solvent 2,2,5,5-tetramethyloxolane via gas-phase reactions with OH and Cl radicals, Atmos. Chem. Phys., 2022, 22, 14589–14602 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |




