Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Quantifying sensitivity of PM2.5 mass to ammonia and nitrate availability in Hong Kong based on four-year hourly measurements
Zijing
Zhang
 a and
Jian Zhen
Yu
a and
Jian Zhen
Yu
 *ab
*ab
aDivision of Environment and Sustainability, Hong Kong University of Science and Technology, Clearwater Bay, Kowloon, Hong Kong. E-mail: jian.yu@ust.hk
bDepartment of Chemistry, Hong Kong University of Science and Technology, Clearwater Bay, Kowloon, Hong Kong
First published on 6th February 2026
Abstract
Ammonium and nitrate are major components of PM2.5, and their fractional contributions to urban PM2.5 in China have increased in recent years, largely due to successful sulfate reductions. Owing to their semi-volatile nature, ammonium and nitrate in PM2.5 are strongly affected by gas–particle partitioning, which depends on temperature, relative humidity, and the ionic composition of PM2.5. Quantifying the sensitivity of PM2.5 mass to precursor availability under locale-specific atmospheric conditions is therefore essential for air quality management. In this study, we analyzed four years (2013–2017) of hourly concentrations of inorganic ions and their gaseous precursors at an urban site in Hong Kong. We estimated aerosol water content (AWC) and aerosol pH and conducted a detailed seasonal analysis. Our results revealed distinct sensitivities of PM2.5 to ammonia and nitrate availability, modulated by aerosol pH and AWC. In fall, sulfate-dominated PM2.5 exhibited low sensitivity to HNO3, while in winter, increased partitioning ratios led to higher PM2.5 sensitivity to HNO3. Quantitative assessment showed that reductions in water-soluble inorganic PM were proportional to decreases in total nitrate (TNO3, including gaseous HNO3 and particulate nitrate) and sulfate, but exhibited a parabolic relationship with reductions in total ammonia (TNH3, including gaseous NH3 and particulate ammonium). As TNH3 levels are further reduced, the sensitivity of PM2.5 to NH3 would increase. Our findings highlight the importance of synergistic reduction of NH3 and NOx emissions in effectively mitigating PM2.5 pollution. This analysis provides valuable insights to inform the development of targeted, integrated air quality management strategies for urban environments.
Environmental significanceUrban PM2.5 is increasingly dominated by semi-volatile nitrate and ammonium, challenging one-size-fits-all precursor control. We show why local thermodynamics matter using four years of hourly observations in Hong Kong. We constrain aerosol pH and water and quantify PM2.5 sensitivity to precursor availability. PM2.5 composition is under “ammonium-rich” regime with substantial NH4NO3, and the sensitivity to TNO3 is higher in winter than fall. Water-soluble inorganic PM falls linearly with total nitrate and sulfate but nonlinearly with total ammonia, yielding larger marginal benefits as NH3 declines. Therefore, synergistic NH3–NOx controls, tuned to season, would offer efficient PM2.5 reductions in Hong Kong and analogous urban environments. |
1. Introduction
Fine particulate matter (PM2.5) adversely affects human health, contributing to respiratory diseases and cardiovascular diseases. Secondary inorganic aerosols (SIAs), including sulfate, ammonium, and nitrate, constitute a major portion of PM2.5 worldwide and are therefore central to air quality management and public health. In Hong Kong, SIA species account for over 40% of ambient PM2.5 mass.1–3 Over the years, the composition of PM2.5 in Hong Kong has changed markedly in response to emission controls targeting SIA precursor gases. For example, SO2 emissions from public electricity generation and navigation decreased by around 40% and 50%, respectively, from 2013 to 2017.4 Consistent with these reductions, annual PM2.5 and sulfate concentrations at the urban Tsuen Wan site in Hong Kong showed a clear decline from 2013 to 2017, as documented by Chow et al.2 In comparison, nitrate concentrations based on filter samples remained roughly stable from 2013 to 2016 and increased in 2017, while ammonium concentrations showed minimal interannual variability. Consequently, NH4+ and NO3− have taken up an increasing mass fraction of PM2.5 in Hong Kong. Nitrate forms via atmospheric oxidation of NOx; following stricter vehicle-emission controls, nitrate levels declined notably from 2008 to 2013 in Hong Kong.2 Recent studies further suggest that ammonia abatement can be a cost-effective lever for PM reduction, with benefit-to-cost ratios as high as 4.6.5,6 Given ongoing NOx controls and the substantial potential for NH3 emission reductions—particularly from agriculture in mainland China—both NOx and NH3 remain key considerations for formulating strategies to further lower PM2.5 in Hong Kong.In Hong Kong, major NOx sources in 2017 included public electricity generation (37%), marine navigation (27%), and road transport (20%).4 Ammonia emissions are dominated globally by agriculture (livestock and nitrogen fertilizer use),7–9 and China is a recognized hotspot due to intensive agricultural activities.8,10 Additional NH3 sources include urban transport and certain industrial processes.8,9 Vehicular NH3—produced by catalytic aftertreatment chemistry—can be important locally in Hong Kong given high traffic volumes, with emission rates dependent on vehicle technology, operating conditions, and catalyst age.11,12 Ammonia readily partitions into the particle phase as ammonium sulfate and ammonium nitrate; the abundant NH3 often yields a strong correlation between sulfate and ammonium in Hong Kong.2 Although sulfate remains the dominant inorganic PM2.5 component on average, nitrate frequently dominates during pollution episodes, especially in fall and winter. Suburban observations indicate that nitrate episodes often occur under “ammonium-rich” conditions, highlighting the increasing role of ammonium during such events.13 These findings imply that reducing the availability of both HNO3 and NH3 would help curb episodic PM2.5, especially during fall and winter.
The abundance of ammonium and nitrate in the particle phase is governed by their thermodynamic equilibrium with gaseous NH3 and HNO3 and is strongly modulated by aerosol acidity (pH). As a result, equal total ammonia (TNH3 = NH3 + NH4+, i.e., combined gaseous NH3 and PM NH4+) or total nitrate (TNO3 = HNO3 + NO3−, combined gaseous HNO3 and PM NO3−) can yield different particle-phase NH4+ and NO3− concentrations depending on aerosol pH. Therefore, the effectiveness of reducing PM mass per unit decrease in TNH3 or TNO3 depends on acidity; quantifying this effectiveness, hereafter referred to as PM mass sensitivity, is essential for designing efficient control strategies.
Aerosol pH exhibits significant spatial and temporal variability, influenced by PM composition and mass concentration, aerosol water content (AWC), relative humidity (RH), and temperature. Reported pH during pollution episodes in northern China (e.g., Tianjin and Beijing) typically ranges from ∼4 to 5.4,14–17 decreasing to below 3 during cleaner periods with reduced dust, alkaline species, and AWC.17 In the Yangtze River Delta (YRD) region, Shanghai's aerosol pH decreased from 3.30 ± 0.58 in 2011 to 3.06 ± 0.58 in 2019.18 In the Pearl River Delta region, for example Hong Kong, average aerosol pH is far more acidic (e.g., −0.03 ± 0.77).19 Similarly low pH (∼0–1) has been observed in the southeastern United States despite >60% sulfate reductions from 1999 to 2014.20 Given the central role of pH in controlling gas–particle partitioning and its variability, updated assessments for recent years are needed to inform control policy.
The effect of aerosol pH, temperature and relative humidity on the gas–particle partitioning ratio of nitrate and ammonium was intensively investigated and established in earlier studies.21–25 As the partitioning ratios ε(NO3−) and ε(NH4+) are functions of pH, temperature and AWC, Nenes et al.26 recently developed a thermodynamic regime framework that classifies PM response to NH3 and HNO3 availability. Four regimes—Insensitive, HNO3-Sensitive, NH3-Sensitive, and Both-Sensitive—are defined by threshold partitioning ratios in the framework. For example, when ε(NO3−) exceeds a threshold (e.g., 0.1, indicating >10% of TNO3 in the particle phase), the conditions are classified as “HNO3-Sensitive”; if both ε(NO3−) and ε(NH4+) exceed the threshold vale, the system is Both-Sensitive. Applying this framework to observations enables identification of dominant controlling precursors. For instance, analysis in Tianjin by Zhao et al.27 indicated predominant HNO3 sensitivity, with NH3 sensitivity increasing with sulfate due to ammonium sulfate formation.
Here, using four years of hourly SIA and related measurements (2013–2017), we quantified trends and variability in aerosol pH in Hong Kong and applied the Nenes framework to assess how PM2.5 responds to changes in NH3 and HNO3 availability under varying PM loadings and across seasons. We further examine nitrate formation under both ammonium-poor and ammonium-rich regimes. More importantly, we quantified PM2.5 mass responses to incremental reductions in TNH3 and TNO3 in a real-world urban environment through simulations, providing a predictive framework for designing effective control strategies. Our results inform control strategies targeting NH3 and HNO3 to mitigate PM2.5 in Hong Kong and offer broader insights for urban regions facing analogous challenges.
2. Methods
2.1 Data sources
Sampling was conducted at the Yuen Long Air Quality Monitoring Station (YL AQMS; 22°26′48″ N, 114°01′21.7″ E), an urban site within the Hong Kong Air Quality Monitoring Network. Measurements spanned July 2013 to June 2017, with a suspension from September 2015 to August 2016. The YL site is characterized by heavy nearby traffic and is therefore considered to be under notable influence of vehicular emissions. Supporting evidence includes distinct diurnal patterns of NH3 and NH4+ at YL compared with suburban sites (Fig. S1 and S2).Hourly concentrations of water-soluble inorganic ions in PM2.5 (Na+, NH4+, K+, Mg2+, Ca2+, Cl−, NO3−, and SO42−) and corresponding semi-volatile gases (NH3, HNO3, and HCl) were measured using a Monitor for AeRosols and Gases in ambient Air (MARGA 2S ADI2080, Metrohm Applikon). Gaseous species were collected in a wet rotating denuder (WRD) using H2O2 as the absorbent, while particles were dissolved in a steam-jet aerosol collector (SJAC). The resulting solutions were analyzed by ion chromatography, with each sample calibrated using an internal LiBr standard. Data validation procedures for MARGA are provided in Section S1. Concurrently, hourly PM2.5 mass was measured with a Synchronized Hybrid Ambient Real-time Particulate Monitor (SHARP 5030, Thermo Scientific), and NOx data were obtained from the Hong Kong Environmental Protection Department (HKEPD) air quality database.
2.2 Calculation of aerosol pH and aerosol water content
Because aerosol pH is not directly measurable, we estimated it using ISORROPIA-II,28 which calculates the equilibrium partitioning of inorganic gases and aerosols and outputs aerosol water content of inorganic species (Wi) and hydrogen ion concentration c(H+). We ran ISORROPIA-II in forward mode using measured total concentrations of semi-volatile species (HNO3 + NO3−, NH3 + NH4+, HCl + Cl−) as inputs, which reduces sensitivity to measurement errors compared with reverse mode.29,30 Because the model computes the equilibrium partitioning of NH3/NH4+ and HNO3/NO3−, its predications were compared with MARGA measurements to assess the performance of model calculation. Overall, the model reproduced the measurements from MARGA, indicating reliable model predictions in our study (Fig. S4). When the RH decreases below the deliquescence RH, the aerosol would not crystalize immediately but instead constitute a supersaturated aqueous solution.15,31 Considering Hong Kong's wet climate with average RH as high as 64% ± 14% in our study, the calculation was based on metastable state assumption (i.e., all salts in the aqueous phase without precipitated salt). Aerosol pH was computed as: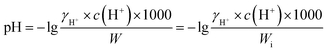 | (1) |
2.3 Determining PM sensitivity to TNO3 and TNH3
PM2.5 responses to precursor availability are governed by gas–particle partitioning thermodynamics. For a given TNO3, a higher nitrate partitioning ratio ε(NO3−) yields a larger fraction of TNO3 in the particle phase and thus a greater PM mass response to incremental HNO3. Analogously, PM sensitivity to TNH3 scales with the ammonium partitioning ratio ε(NH4+). To maintain comparability with prior studies, we adopt a partitioning threshold of 0.1. For example, ε(NO3−) > 0.1 indicates that, for a unit increase in HNO3, 10% of it partitions to the particle phase, producing a meaningful PM mass response; we therefore classify ε(NO3−) > 0.1 as HNO3-Sensitive and ε(NH4+) > 0.1 as NH3-Sensitive.26,27Gaseous HNO3 is in equilibrium with particulate nitrate through dissolution of HNO3 and acid dissociation. With these two processes of equilibrium, the theoretical ε(NO3−) can be calculated using eqn (2):26,29
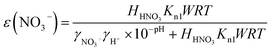 | (2) |
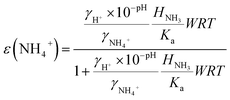 | (3) |
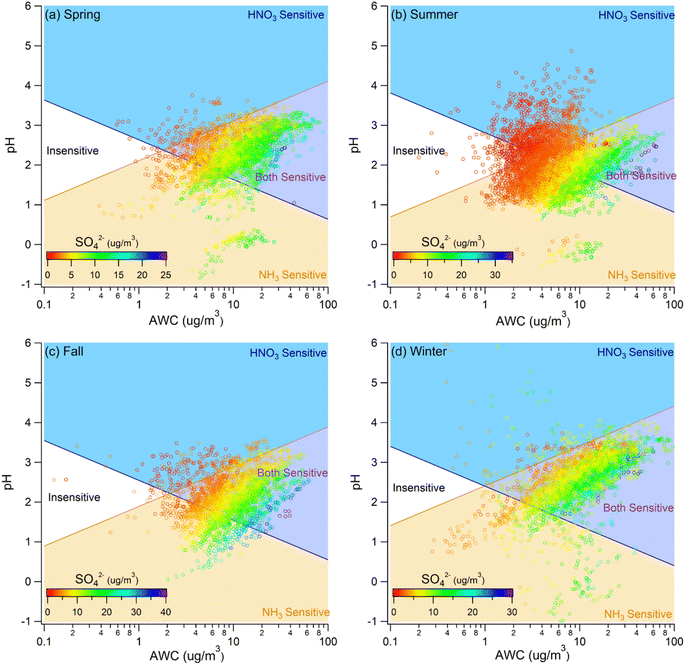
We applied the thermodynamic regime framework of Nenes et al.26 to classify each hourly observation into four sensitivity regimes, namely “HNO3-Sensitive”, “NH3-Sensitive”, “Both-Sensitive”, and “Insensitive”, based on ε(NO3−) and ε(NH4+), which are functions of pH, W, and T. To examine how PM loadings and meteorology modulate sensitivity, we further stratified the data by PM2.5 concentration and by season. This stratification enables evaluation of regime frequency and PM response across contrasting conditions.
3. Results & discussion
3.1 Interannual and seasonal variability of pollutant concentrations
Monthly averages (Fig. 1) show consistent seasonality for PM2.5, TNO3, and TNH3, with maxima in fall–winter and minima in summer. From 2013 to 2016, wintertime PM2.5 and NOx decreased by approximately 40% and 33%, respectively (Table 1). TNO3 declined by 37% over the same period, consistent with reductions in NOx emissions. However, TNH3 concentration only decreased by 7.1%, much less than other compounds. Even in summer (the least polluted season), PM2.5, NOx, TNO3, and SO42− all exhibited year-on-year decreases, while TNH3 was even higher in 2016 than 2013. This divergence indicates that while controls on SO2 and NOx were effective, ammonia emissions have not been effectively controlled, and its availability remained relatively stable. Notably, as PM2.5 concentrations declined, the fractional contribution of SIA to PM2.5 increased, highlighting a growing relative importance of ammonium and nitrate. This shift underscores the need to complement ongoing SO2 and NOx controls with strategies targeting NH3 and HNO3 precursors, particularly to address semi-volatile SIA components that become more influential at lower PM loadings.| PM2.5 (µg m−3) | NOx (ppb) | TNO3 (µg m−3) | TNH3 (µg m−3) | SO42−(µg m−3) | |
|---|---|---|---|---|---|
| 2013-Summer | 22.7 | 42.0 | 4.5 | 6.4 | 6.2 |
| 2013-Winter | 54.2 | 59.3 | 10.2 | 9.9 | 11.2 |
| 2014-Summer | 23.2 | 37.8 | 3.0 | 5.8 | 6.6 |
| 2014-Winter | 58.3 | 56.6 | 9.2 | 10.9 | 10.8 |
| 2015-Summer | 16.3 | 32.1 | 2.3 | 5.8 | 4.4 |
| 2015-Winter | — | — | — | — | — |
| 2016-Summer | 19.6 | 34.2 | 3.3 | 7.1 | 5.6 |
| 2016-Winter | 32.7 | 41.4 | 6.3 | 9.2 | 7.7 |
3.2 Aerosol pH in Hong Kong
Aerosol pH is a central thermodynamic parameter that influences multiple atmospheric processes. For example, stronger acidity accelerates acid-catalyzed pathways for secondary organic aerosol (SOA) formation from volatile organic compounds (VOCs), and controls the solubility of SO2, NH3, and HNO3, thereby regulating aqueous-phase sulfate production and the gas–particle partitioning of nitrate and ammonium.32 Because in Hong Kong both SOA and SIA constitute substantial fractions of PM2.5, aerosol pH fundamentally modulates secondary aerosol formation rates and mechanisms and is a key factor in understanding variability in the city's PM levels.33,34Fig. 2 shows a pronounced diurnal cycle in aerosol pH during all seasons. The method of dividing the four seasons was based on upper-level wind, dew point and sea level pressure.35 Aerosol pH was the lowest in the afternoon and the highest around dawn; afternoon values were typically 0.6–0.8 units lower than the values at dawn. This diurnal variability was comparable to interannual differences despite substantial compositional changes over the years (Fig. 1), indicating that meteorology (RH and temperature) dominates pH variability. Consistently, the pH diurnal cycle tracked RH and was anticorrelated with temperature (Fig. S5 and S6). Seasonally, winter exhibited the highest pH despite having the lowest RH, which would ordinarily depress pH by reducing aerosol water. This contrast suggests that temperatures dominate the seasonal control on aerosol pH in Hong Kong, consistent with observations in Shanghai.18
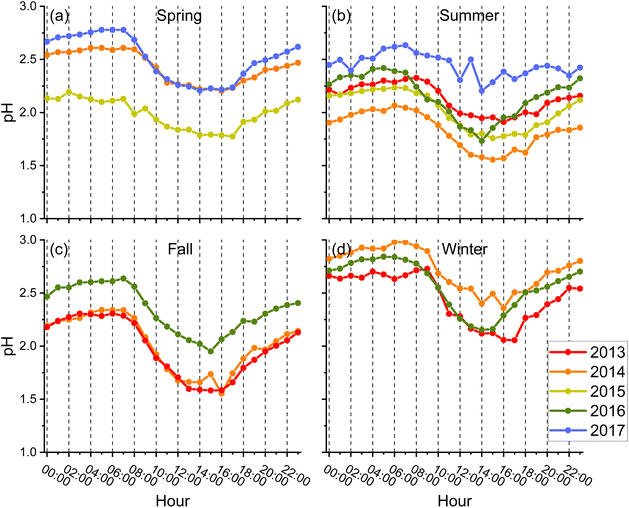 | ||
| Fig. 2 Season-specific diurnal variation of aerosol pH: (a) spring, (b) summer, (c) fall, and (d) winter. | ||
Over the five-year period, aerosol pH remained relatively stable even as sulfate and NOx concentrations declined markedly, paralleling trends observed in the southeastern United States.20 However, summertime pH in Hong Kong (1.5–3.0) was higher than that typically observed in the southeastern U.S. (0–1). The comparatively lower acidity in Hong Kong implies weaker acid-catalyzed uptake of certain VOC oxidation products (e.g., IEPOX from isoprene oxidation) and slower acid-driven heterogeneous reactions,36,37 while favoring faster aqueous-phase sulfate production from SO2. Consequently, Hong Kong's higher pH environment likely enhances inorganic sulfate formation but suppresses SOA yields from acid-dependent pathways, increasing the relative contribution of inorganic aerosol compared with regions characterized by more acidic particles.
3.3 Impact of key factors on PM sensitivity across seasons
The thermodynamic regime framework indicates that PM mass sensitivity to TNO3 and TNH3 is governed by AWC and aerosol pH. Because higher PM loadings enhance water uptake and shift composition, both AWC and pH are indirectly modulated by PM mass and species such as sulfate and ammonium. We therefore examined how PM2.5 mass, sulfate, and TNH3 influence sensitivity across seasons.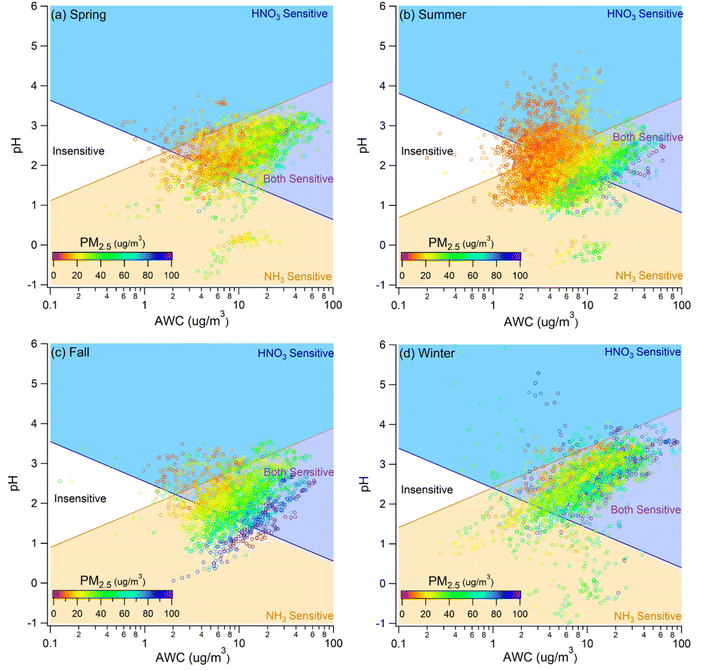 | ||
| Fig. 3 Sensitivity regime plots for (a) spring, (b) summer, (c) fall, and (d) winter. The data points are colored by PM concentration using a common scale. | ||
To contextualize high-loading conditions, we adopted the 24-hour PM2.5 standard of 50 µg m−3 (WHO interim target-2),38 as used in Hong Kong's Air Quality Objectives for 2022–2025, to define polluted conditions. Exceedances occurred more frequently in fall and winter (Fig. 3), so subsequent analyses of polluted cases focus on these seasons (Fig. S7). Fall polluted cases exhibited stronger sensitivity to TNH3 than to TNO3 (Fig. S8). In winter, sensitivity to TNO3 intensified relative to fall, while NH3 sensitivity remained. Elevated sulfate was associated with increased NH3 sensitivity in both fall and winter. This mirrors the effect of higher PM in fall but contrasts with winter, where increases in PM did not shift regimes as distinctly. Overall, these patterns suggest that sulfate is a primary driver of NH3 sensitivity in fall, whereas nitrate assumes a comparatively greater role in winter, consistent with colder temperatures favoring partitioning of HNO3 and NH3 to the particle phase.
 | ||
| Fig. 5 Sensitivity regime plots colored by TNH3 concentration for (a) spring, (b) summer, (c) fall, and (d) winter. | ||
These seasonal differences are consistent with composition-dependent thermodynamics. During winter, elevated TNO3 facilitates NH4NO3 formation when TNH3 is abundant, so PM mass becomes more responsive to additional HNO3. In fall, comparatively lower TNO3 constrains NH4NO3 formation, limiting the influence of TNH3 on HNO3 sensitivity. Nevertheless, higher TNH3 increases AWC and reduces particle-phase ammonia scarcity, both of which favor NH3 partitioning and elevate ε(NH4+). Consequently, regardless of season, increases in TNH3 enhance NH3 sensitivity, while the concurrent effect on HNO3 sensitivity depends on the availability of total nitrate and seasonal thermodynamic conditions.
3.4 Nitrate formation under ammonium-rich and ammonium-poor regimes
In the thermodynamic equilibrium state, the particulate ammonium concentration constrains the simultaneous formation of ammonium sulfate and ammonium nitrate. Significant concentrations of particulate nitrate (as NH4NO3) are thermodynamically sustainable only when the molar amount of aerosol-phase ammonium exceeds that required to associate with sulfate.40 The “excess ammonium” is defined as [NH4+] − K × [SO42−], where K is an empirical criterion molar ratio of ammonium to sulfate.41 Following established practice,42,43K is obtained as the x-intercept of the correlation between [NO3−]/[SO42−] and [NH4+]/[SO42−]. Conditions with [NH4+]/[SO42−] > K, which means that the excess ammonium is greater than zero, are classified as “NH4+-rich”, and those with [NH4+]/[SO42−] < K as “NH4+-poor”. This particle-phase classification provides a diagnostic for the thermodynamic state resulting from the partitioning of TNH3 and TNO3. The “NH4+-rich” regime indicates that, given the ambient TNH3, TNO3, and thermodynamic conditions (AWC, pH, T), the system favors the formation of particulate nitrate. Its occurrence is therefore modulated by factors such as AWC, which shifts the partitioning equilibrium.The classification of regimes is primarily controlled by the relative emission strengths and transport of precursors, and the regime shift is a more fundamental change in aerosol composition that typically occurs over longer timescales. Therefore, here we separate the data by seasons. Season-resolved correlation plots indicate that NH4+-rich conditions prevail in spring, fall, and winter, whereas NH4+-poor conditions occur more frequently in summer (Fig. S9). Linear regressions of [NO3−]/[SO42−] against [NH4+]/[SO42−], along with the derived K values for each season, are summarized in Table 2. Note that the value of K is not a universal constant but is influenced by local thermodynamic environment. Its value ranges from 1.58 to 1.78 across seasons, and the seasonal variation in K could be attributed to the variation in non-volatile cations, temperature and RH. For example, temperature can influence the activity coefficients and the equilibrium constant for NH4NO3 dissociation, subtly shifting the K value defined by the [NO3−]/[SO42−] vs. [NH4+]/[SO42−] correlation. The range of K values in our study is consistent with that reported in prior studies.41,43,44 Importantly, AWC is a key factor influencing this local thermodynamic environment. By affecting the activity coefficients and the equilibrium constant for semi-volatile species such as NH4NO3, variations in AWC (driven by RH and inorganic mass) contribute to the observed seasonal shifts in the empirical K value.
| Season | Regression equation | K value |
|---|---|---|
| Spring | Y = 0.83X − 1.31 | 1.58 ± 0.025 |
| Summer | Y = 0.91X − 1.62 | 1.78 ± 0.030 |
| Fall | Y = 0.80X − 1.35 | 1.69 ± 0.034 |
| Winter | Y = 0.85X − 1.38 | 1.62 ± 0.020 |
Using the season-specific K, we calculated “excess ammonium” and examined its relationship with particulate nitrate. Under “NH4+-rich” conditions, excess ammonium strongly correlates with nitrate concentration in all seasons (Fig. 6), supporting the interpretation that NH4NO3 forms efficiently when ammonium is available beyond that required to neutralize sulfate. The seasonal slopes of nitrate versus excess ammonium ranged from 0.71 to 0.84, indicating that the majority of excess ammonium is present as NH4NO3. By contrast, nitrate concentrations remained very low under “NH4+ poor” conditions, reflecting the constraint imposed by insufficient ammonium to neutralize nitric acid. Consequently, nitrate-dominated aerosol states primarily occurred in “NH4+-rich” regimes, while “NH4+-poor” conditions at the YL site were generally associated with cleaner periods. Aerosol pH ranged from 0.5 to 4 across all seasons. In the “NH4+-rich” regime, aerosol pH increased with nitrate concentration, consistent with the thermodynamic expectation that higher pH favors nitrate partitioning into the particle phase. Differently, aerosol pH was more influenced by crustal cations under “NH4+-poor” regime. On average, aerosol pH was 0.2 to 0.6 units higher in the “NH4+-rich” regime due to the higher ammonia abundance (Table 3). This analysis underscores that substantial nitrate formation requires not only high total precursor concentrations but also a thermodynamic state (often characterized by sufficient AWC and higher aerosol pH) that drives TNO3 into the particle phase.
 | ||
| Fig. 6 Correlation plots between nitrate concentration and excess ammonium concentration for (a) spring, (b) summer, (c) fall, and (d) winter with data points colored by aerosol pH. | ||
| Season | NH4+-rich | NH4+-poor |
|---|---|---|
| Spring | 2.4 | 2.0 |
| Summer | 2.1 | 1.9 |
| Fall | 2.2 | 1.6 |
| Winter | 2.6 | 2.0 |
3.5 Simulation of water-soluble inorganic component (WSIC) reduction in fall and winter
| TNO3 | TNH3 | TCl | SO42− | Na+ | K+ | Mg2+ | Ca2+ | Temp. | RH | |
|---|---|---|---|---|---|---|---|---|---|---|
| a Unit: µg m−3 for WSICs, °C for temp. and % for RH. | ||||||||||
| Fall | 5.0 | 9.9 | 0.66 | 10.9 | 0.23 | 0.26 | 0.04 | 0.16 | 25.1 | 59.8 |
| Winter | 8.5 | 10.0 | 0.94 | 9.8 | 0.32 | 0.43 | 0.05 | 0.18 | 17.6 | 55.9 |
In fall, the base state lies in the “Both-Sensitive” regime. Reducing TNH3 rapidly induces rapid decline in HNO3 sensitivity; an ∼40% reduction in TNH3 shifts the system to the “NH3-Sensitive” regime. Decreasing sulfate lowers NH3 sensitivity, consistent with sulfate's role in enhancing acidity and AWC and thereby promoting NH3 partitioning. An ∼60% reduction in sulfate drives the system toward the “HNO3-Sensitive” regime. Notably, sulfate reductions have little effect on the degree of HNO3 sensitivity itself, as points translate largely parallel to the “HNO3-Sensitive” boundary, confirming limited NH4NO3 formation in fall because of constrained TNO3. In contrast, changes in TNO3 have negligible influence on pH, AWC, or regime position, indicating weak leverage of nitrate on sensitivity under fall conditions.
Winter shows a similar baseline in the “Both-Sensitive” regime. As in fall, cutting TNH3 quickly diminishes HNO3 sensitivity, and pH and AWC remain insensitive to TNO3 changes. However, sulfate reductions in winter increase HNO3 sensitivity, with points moving away from the “HNO3-Sensitive” boundary and penetrating deeper into the “HNO3-Sensitive” regime. This pattern indicates that ammonium nitrate contributes substantially to PM in winter alongside ammonium sulfate, so lowering sulfate frees ammonium for nitrate formation and accentuates responsiveness to HNO3.
Using the simulated regime outputs, we further quantified the reductions in WSIC concentrations resulting from decreases in TNO3, TNH3, or sulfate and compared the relative efficiencies of each control pathway in fall and winter (Fig. 8, red curve). Sulfate abatement is the most effective strategy for reducing WSICs because sulfate is non-volatile and its reductions directly diminish particulate SO42− while simultaneously lowering the partitioning of NH3 to NH4+, thereby reducing particle-phase ammonium. Consistent with the regime analysis (Fig. S10 and S11), sulfate controls thus exert compounding benefits on WSICs.
In contrast, TNO3 reductions have minimal influence on aerosol pH and AWC, so they do not substantially alter ammonia partitioning. Under these conditions, the decrease in SIA attributable to nitrate controls is approximately the reduction in TNO3 multiplied by the particle-phase nitrate partitioning ratio (Fig. 8, blue curve). TNH3 reduction would lead to a great increase in ε(NH4+) because of the lowered aerosol pH (Fig. S11). Consequently, TNH3 reduction would be increasingly efficient as more TNH3 is reduced, because a larger fraction of TNH3 would be in the particle phase, producing a characteristic parabolic relationship between WSIC reduction and TNH3 reduction (Fig. 8, pale yellow curve).
In fall, the effectiveness of reducing TNO3 or TNH3 is limited by the low abundance of NH4NO3. For example, a 60% decrease in TNH3 yields only about a 2 µg m−3 reduction in WSICs, and a 60% cut in TNO3 reduces WSICs by roughly 1.4 µg m−3. By comparison, sulfate control is markedly more efficient: a 60% reduction in sulfate lowers WSICs by approximately 9.5 µg m−3. In winter, higher NH4NO3 abundance increases the leverage of both TNO3 and TNH3 controls. The marginal efficiency of TNO3 control rises to about 0.08 µg m−3 WSIC reduction per 1% TNO3 decrease, or roughly 0.85 µg m−3 WSIC reduction per 1 µg m−3 decrease in TNO3 (Fig. S12). Sulfate control remains effective but is somewhat less efficient than in fall due to the lower fraction of ammonium sulfate; only about 0.14 µg m−3 of WSICs is reduced per 1% decrease in sulfate. For modest reductions (<20% or <2.5 µg m−3), TNH3 control is less effective than TNO3 control for lowering WSICs in winter. However, at larger reductions (>45% or ≥4.3 µg m−3), TNH3 abatement becomes the most efficient of the two semi-volatile pathways (Fig. S12). These results support the feasibility of controlling semi-volatile precursors, TNH3 and TNO3, for wintertime PM mitigation, while highlighting sulfate abatement as the dominant WSIC-reduction lever in fall.
Although our single-factor perturbation analyses provide quantitative insights into the individual sensitivity of PM2.5 mass to these components, it is important to acknowledge their limitation in capturing the full complexity of real-world emission scenarios. In practice, controls on TNO3, sulfate and TNH3 are likely to be implemented simultaneously, and the thermodynamic system response may be non-additive due to strong non-linear interactions. For instance, the ε(NO3−) level is strongly modulated by the ambient NH3 levels (Fig. S10 and S11), so a coordinated reduction of TNH3 and TNO3 would not be additive. Instead, it would drive the system into a regime where the marginal PM2.5 reduction per unit of NH3 cut is greatly amplified, representing a synergistic effect. Quantification of this synergistic effect is analyzed in the following section.
 | ||
| Fig. 9 Simulated WSIC concentrations as functions of TNO3 and TNH3 in the range of 0 to 20 µg m−3 during (a) fall and (b) winter. The star in each plot denotes seasonal average conditions. | ||
Baseline conditions, marked according to seasonal averages in Table 4, lie near the ridge line of the WSIC response surface in both seasons. This placement indicates that reducing either TNO3 or TNH3 from current levels is effective in cutting down WSIC concentration, and that concurrent reductions in both precursors provide an even larger benefit. Accordingly, coordinated TNO3 and TNH3 controls represent a thermodynamically efficient strategy for mitigating PM2.5 during high-pollution seasons, outperforming approaches that target only one of the two semi-volatile precursors.
4. Conclusions
We combined hourly water-soluble ionic species measurements with meteorological data and ISORROPIA-II thermodynamic modeling to characterize aerosol pH and quantify PM2.5 mass sensitivity to ammonia and nitrate availability in urban Hong Kong. Using the sensitivity regime framework of Nenes et al.,26 we mapped seasonal regime behaviors and further quantified sensitivities through simulations that reduced total ammonia (TNH3), total nitrate (TNO3), and sulfate. Aerosol pH typically ranged from 1.5 to 3 throughout the year. Its diurnal amplitude exceeded seasonal and interannual variability, indicating that meteorology, particularly temperature, exerts stronger control on aerosol acidity than compositional changes under Hong Kong conditions. Winter consistently showed higher pH than other seasons, attributable to lower temperatures.Pollution episodes were concentrated in fall and winter, but seasonal differences in composition produced distinct sensitivity patterns. In fall, sulfate dominated WSICs, and PM mass was more sensitive to TNH3 than to TNO3 availability. In winter, nitrate and ammonium contributed larger mass fractions, yielding enhanced sensitivity to both TNH3 and TNO3. Component-wise perturbation analyses clarified these mechanisms. Reducing TNO3 had negligible effects on aerosol pH or AWC and therefore did not shift PM sensitivity regimes. Reducing TNH3 increased NH3 sensitivity while diminishing TNO3 sensitivity in both seasons by limiting ammonium availability for NH4NO3 formation. Sulfate reductions strengthened TNH3 sensitivity across seasons by lowering acidity and AWC, with a more pronounced enhancement of TNO3 sensitivity in winter than in fall, consistent with greater wintertime NH4NO3 formation.
Nitrate was primarily present as ammonium nitrate. Most observations fell within the “NH4+ rich” regime, where nitrate is neutralized by excess ammonium, and more than 70% of excess ammonium was present as NH4NO3. These findings support TNH3 control as an effective lever for PM2.5 mitigation. Efficiency analyses further showed that sulfate abatement reduces WSICs slightly less efficiently in winter than in fall (approximately 0.14 versus 0.16 µg m−3 per 1% sulfate reduction). By contrast, TNO3 controls were substantially more effective in winter than in fall due to higher NH4NO3 abundance (approximately 0.08 versus 0.03 µg m−3 WSIC reduction per 1% TNO3 reduction). Reductions in WSICs exhibited a nonlinear (parabolic) response to TNH3 abatement; as TNH3 decreases, the marginal efficiency of control increases. Sulfate always had the highest marginal efficiency in cutting down PM mass during fall, followed by TNH3 and TNO3. In winter, the marginal efficiency of TNH3 reduction would be higher than TNO3 with a reduction rate of 20%; once TNH3 reductions exceed about 45%, ammonia control becomes more effective than sulfate in lowering WSICs.
Overall, coordinated reductions of TNH3 and TNO3 provide a thermodynamically efficient pathway for PM2.5 mitigation during high-pollution seasons, while sulfate abatement remains a robust year-round strategy, especially in fall when sulfate dominates WSICs and nitrate formation is constrained. These results highlight the importance of season-specific, composition-aware control portfolios that target both non-volatile and semi-volatile precursors to optimize PM2.5 reductions in urban areas that face similar challenges to those encountered in Hong Kong.
Conflicts of interest
There are no conflicts to declare.Data availability
Data for this article, including raw sampling data and simulation results, are available at: https://dataspace.hkust.edu.hk/bib/JKV87K.Supplementary information (SI): figures. See DOI: https://doi.org/10.1039/d5ea00114e.
Acknowledgements
This work was partly supported by the Research Grants Council of Hong Kong (R6011-18). We thank the Hong Kong Environmental Protection Department for provision of the real-time PM2.5 and MARGA datasets. The content of this paper does not necessarily reflect the views and policies of the HKSAR Government, nor does mention of trade names or commercial products constitute an endorsement or recommendation of their use.References
- X. H. H. Huang, Q. Bian, W. M. Ng, P. K. K. Louie and J. Z. Yu, Characterization of PM2.5 Major Components and Source Investigation in Suburban Hong Kong: A One Year Monitoring Study, Aerosol Air Qual. Res., 2014, 14(1), 237–250 CrossRef.
- W. S. Chow, K. Liao, X. Huang, K. F. Leung, A. K. Lau and J. Z. Yu, Measurement Report: Ten-year trend of PM 2.5 major components and source tracers from 2008 to 2017 in an urban site of Hong Kong, China, Atmos. Chem. Phys., 2022, 1–24 Search PubMed.
- G. John J. C. C. Watson, X. Wang and D. Steven, Kohl Hong Kong Fine Particulate Matter (PM2.5) Chemical Compositions, 2021 Search PubMed.
- J. Zhen Yu and T. Zhang, Final Report for Chemical Speciation of PM2.5 Filter Samples, January 1 through December 31, 2017, 2018 Search PubMed.
- B. J. Gu, L. Zhang, R. Van Dingenen, M. Vieno, H. J. M. Van Grinsven, X. M. Zhang, S. H. Zhang, Y. F. Chen, S. T. Wang, C. C. Ren, S. Rao, M. Holland, W. Winiwarter, D. L. Chen, J. M. Xu and M. A. Sutton, Abating ammonia is more cost-effective than nitrogen oxides for mitigating PM2.5 air pollution, Science, 2021, 374(6568), 758 CrossRef PubMed.
- M. Liu, X. Huang, Y. Song, J. Tang, J. Cao, X. Zhang, Q. Zhang, S. Wang, T. Xu, L. Kang, X. Cai, H. Zhang, F. Yang, H. Wang, J. Z. Yu, A. K. H. Lau, L. He, X. Huang, L. Duan, A. Ding, L. Xue, J. Gao, B. Liu and T. Zhu, Ammonia emission control in China would mitigate haze pollution and nitrogen deposition, but worsen acid rain, Proc. Natl. Acad. Sci. U. S. A., 2019, 116(16), 7760–7765 CrossRef PubMed.
- A. Pozzer, A. P. Tsimpidi, V. A. Karydis, A. de Meij and J. Lelieveld, Impact of agricultural emission reductions on fine-particulate matter and public health, Atmos. Chem. Phys., 2017, 17(20), 12813–12826 CrossRef.
- A. Ismaeel, A. P. K. Tai and J. Wu, Understanding the spatial patterns of atmospheric ammonia trends in South Asia, Sci. Total Environ., 2024, 954, 176118 CrossRef PubMed.
- K. E. Wyer, D. B. Kelleghan, V. Blanes-Vidal, G. Schauberger and T. P. Curran, Ammonia emissions from agriculture and their contribution to fine particulate matter: A review of implications for human health, J. Environ. Manage., 2022, 323, 116285 CrossRef PubMed.
- L. Liu, W. Xu, X. Lu, B. Zhong, Y. Guo, X. Lu, Y. Zhao, W. He, S. Wang, X. Zhang, X. Liu and P. Vitousek, Exploring global changes in agricultural ammonia emissions and their contribution to nitrogen deposition since 1980, Proc. Natl. Acad. Sci. U. S. A., 2022, 119(14), e2121998119 CrossRef PubMed.
- T. Huai, T. D. Durbin, J. W. Miller, J. T. Pisano, C. G. Sauer, S. H. Rhee and J. M. Norbeck, Investigation of NH3 emissions from new technology vehicles as a function of vehicle operating conditions, Environ. Sci. Technol., 2003, 37(21), 4841–4847 Search PubMed.
- D. C. Carslaw and G. Rhys-Tyler, New insights from comprehensive on-road measurements of NOx, NO2 and NH3 from vehicle emission remote sensing in London, UK, Atmos. Environ., 2013, 81, 339–347 CrossRef.
- S. M. Griffith, X. H. H. Huang, P. K. K. Louie and J. Z. Yu, Characterizing the thermodynamic and chemical composition factors controlling PM2.5 nitrate: Insights gained from two years of online measurements in Hong Kong, Atmos. Environ., 2015, 122, 864–875 CrossRef.
- Y. Cheng, G. Zheng, C. Wei, Q. Mu, B. Zheng, Z. Wang, M. Gao, Q. Zhang, K. He and G. Carmichael, Reactive nitrogen chemistry in aerosol water as a source of sulfate during haze events in China, Sci. Adv., 2016, 2(12), e1601530 CrossRef PubMed.
- M. Liu, Y. Song, T. Zhou, Z. Xu, C. Yan, M. Zheng, Z. Wu, M. Hu, Y. Wu and T. Zhu, Fine particle pH during severe haze episodes in northern China, Geophys. Res. Lett., 2017, 44(10), 5213–5221 Search PubMed.
- T. Tan, M. Hu, M. Li, Q. Guo, Y. Wu, X. Fang, F. Gu, Y. Wang and Z. Wu, New insight into PM2.5 pollution patterns in Beijing based on one-year measurement of chemical compositions, Sci. Total Environ., 2018, 621, 734–743 Search PubMed.
- G. Shi, J. Xu, X. Peng, Z. Xiao, K. Chen, Y. Tian, X. Guan, Y. Feng, H. Yu, A. Nenes and A. G. Russell, pH of Aerosols in a Polluted Atmosphere: Source Contributions to Highly Acidic Aerosol, Environ. Sci. Technol., 2017, 51(8), 4289–4296 Search PubMed.
- M. Zhou, G. Zheng, H. Wang, L. Qiao, S. Zhu, D. Huang, J. An, S. Lou, S. Tao and Q. Wang, Long-term trends and drivers of aerosol pH in eastern China, Atmos. Chem. Phys., 2021, 1–21 Search PubMed.
- J. Xue, A. K. H. Lau and J. Z. Yu, A study of acidity on PM2.5 in Hong Kong using online ionic chemical composition measurements, Atmos. Environ., 2011, 45(39), 7081–7088 Search PubMed.
- R. J. Weber, H. Guo, A. G. Russell and A. Nenes, High aerosol acidity despite declining atmospheric sulfate concentrations over the past 15 years, Nat. Geosci., 2016, 9(4), 282–285 Search PubMed.
- A. S. Wexler and S. L. Clegg, Atmospheric aerosol models for systems including the ions H+, NH4+, Na+, SO42−, NO3−, Cl−, Br−, and H2O, J. Geophys. Res.: Atmos., 2002, 107(D14), ACH 14 Search PubMed.
- A. Stelson and J. Seinfeld, Relative humidity and temperature dependence of the ammonium nitrate dissociation constant, Atmos. Environ., 2007, 41, 126–135 Search PubMed.
- M. Michael, The dissociation constant of ammonium nitrate and its dependence on temperature, relative humidity and particle size, Atmos. Environ., 1993, 27(2), 261–270 Search PubMed.
- Z. Meng and J. H. Seinfeld, Time scales to achieve atmospheric gas-aerosol equilibrium for volatile species, Atmos. Environ., 1996, 30(16), 2889–2900 Search PubMed.
- A. Stelson and J. Seinfeld, Relative humidity and pH dependence of the vapor pressure of ammonium nitrate-nitric acid solutions at 25 C, Atmos. Environ., 1982, 16(5), 993–1000 Search PubMed.
- A. Nenes, S. N. Pandis, R. J. Weber and A. Russell, Aerosol pH and liquid water content determine when particulate matter is sensitive to ammonia and nitrate availability, Atmos. Chem. Phys., 2020, 20(5), 3249–3258 CrossRef.
- Q. Zhao, A. Nenes, H. Yu, S. Song, Z. Xiao, K. Chen, G. Shi, Y. Feng and A. G. Russell, Using High-Temporal-Resolution Ambient Data to Investigate Gas-Particle Partitioning of Ammonium over Different Seasons, Environ. Sci. Technol., 2020, 54(16), 9834–9843 CrossRef PubMed.
- C. Fountoukis and A. Nenes, ISORROPIA II: a computationally efficient thermodynamic equilibrium model for K+–Ca2+–Mg2+–NH4+–Na+–SO42−–NO3−–Cl−–H2O aerosols, Atmos. Chem. Phys., 2007, 7(17), 4639–4659 Search PubMed.
- H. Guo, A. P. Sullivan, P. Campuzano-Jost, J. C. Schroder, F. D. Lopez-Hilfiker, J. E. Dibb, J. L. Jimenez, J. A. Thornton, S. S. Brown, A. Nenes and R. J. Weber, Fine particle pH and the partitioning of nitric acid during winter in the northeastern United States, J. Geophys. Res.: Atmos., 2016, 121(17), 10355–10376 Search PubMed.
- C. J. Hennigan, J. Izumi, A. P. Sullivan, R. J. Weber and A. Nenes, A critical evaluation of proxy methods used to estimate the acidity of atmospheric particles, Atmos. Chem. Phys., 2015, 15(5), 2775–2790 CrossRef.
- H. Guo, L. Xu, A. Bougiatioti, K. M. Cerully, S. L. Capps, J. R. Hite, A. G. Carlton, S. H. Lee, M. H. Bergin, N. L. Ng, A. Nenes and R. J. Weber, Fine-particle water and pH in the southeastern United States, Atmos. Chem. Phys., 2015, 15(9), 5211–5228 CrossRef.
- J. I. Steinfeld, Atmospheric chemistry and physics: from air pollution to climate change, Environ.: Sci. Policy Sustainable Dev., 1998, 40(7), 26 CrossRef.
- W. S. Chow, X. H. H. Huang, K. F. Leung, L. Huang, X. Wu and J. Z. Yu, Molecular and elemental marker-based source apportionment of fine particulate matter at six sites in Hong Kong, China, Sci. Total Environ., 2022, 813, 152652 CrossRef PubMed.
- J. Li, Q. Liu, Y. Li, T. Liu, D. Huang, J. Zheng, W. Zhu, M. Hu, Y. Wu and S. Lou, Characterization of aerosol aging potentials at suburban sites in northern and southern China utilizing a potential aerosol mass (Go: PAM) reactor and an aerosol mass spectrometer, J. Geophys. Res.: Atmos., 2019, 124(10), 5629–5649 Search PubMed.
- Y. K. Wong, K. M. Liu, C. Yeung, K. K. Leung and J. Z. Yu, Measurement report: Characterization and source apportionment of coarse particulate matter in Hong Kong: insights into the constituents of unidentified mass and source origins in a coastal city in southern China, Atmos. Chem. Phys., 2022, 22(7), 5017–5031 CrossRef CAS.
- Q. Ying, J. Li and S. H. Kota, Significant Contributions of Isoprene to Summertime Secondary Organic Aerosol in Eastern United States, Environ. Sci. Technol., 2015, 49(13), 7834–7842 CrossRef CAS PubMed.
- H. O. Pye, R. W. Pinder, I. R. Piletic, Y. Xie, S. L. Capps, Y. H. Lin, J. D. Surratt, Z. Zhang, A. Gold, D. J. Luecken, W. T. Hutzell, M. Jaoui, J. H. Offenberg, T. E. Kleindienst, M. Lewandowski and E. O. Edney, Epoxide pathways improve model predictions of isoprene markers and reveal key role of acidity in aerosol formation, Environ. Sci. Technol., 2013, 47(19), 11056–11064 Search PubMed.
- World Health Organization, WHO Global Air Quality Guidelines: Particulate Matter (PM2.5 and PM10), Ozone, Nitrogen Dioxide, Sulfur Dioxide and Carbon Monoxide, World Health Organization, Geneva, p. 2021 Search PubMed.
- J. P. S. Wong, Y. Yang, T. Fang, J. A. Mulholland, A. G. Russell, S. Ebelt, A. Nenes and R. J. Weber, Fine Particle Iron in Soils and Road Dust Is Modulated by Coal-Fired Power Plant Sulfur, Environ. Sci. Technol., 2020, 54(12), 7088–7096 CrossRef CAS PubMed.
- Y. Tao, X. Ye, Z. Ma, Y. Xie, R. Wang, J. Chen, X. Yang and S. Jiang, Insights into different nitrate formation mechanisms from seasonal variations of secondary inorganic aerosols in Shanghai, Atmos. Environ., 2016, 145, 1–9 CrossRef CAS.
- R. K. Pathak, W. S. Wu and T. Wang, Summertime PM 2.5 ionic species in four major cities of China: nitrate formation in an ammonia-deficient atmosphere, Atmos. Chem. Phys., 2009, 9(5), 1711–1722 CrossRef CAS.
- K. He, Q. Zhao, Y. Ma, F. Duan, F. Yang, Z. Shi and G. Chen, Spatial and seasonal variability of PM2.5 acidity at two Chinese megacities: insights into the formation of secondary inorganic aerosols, Atmos. Chem. Phys., 2012, 12(3), 1377–1395 Search PubMed.
- Y.-C. Lin, Y.-L. Zhang, M.-Y. Fan and M. Bao, Heterogeneous formation of particulate nitrate under ammonium-rich regimes during the high-PM2.5 events in Nanjing, China, Atmos. Chem. Phys., 2020, 20(6), 3999–4011 CrossRef CAS.
- X. Huang, R. Qiu, C. K. Chan and P. Ravi Kant, Evidence of high PM2.5 strong acidity in ammonia-rich atmosphere of Guangzhou, China: Transition in pathways of ambient ammonia to form aerosol ammonium at [NH4+]/[SO42–]=1.5, Atmos. Res., 2011, 99(3–4), 488–495 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |