Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Quantification, diel variation and photochemistry of inorganic chlorine trace gases in continental Germany
Lasse
Moormann
 *a,
John N.
Crowley
*a,
John N.
Crowley
 b,
Friederike
Fachinger
a and
Frank
Drewnick
a
b,
Friederike
Fachinger
a and
Frank
Drewnick
a
aMultiphase Chemistry Department, Max Planck Institute for Chemistry, Mainz, 55128, Germany. E-mail: lasse-moormann@mpic.de
bAtmospheric Chemistry Department, Max Planck Institute for Chemistry, Mainz, 55128, Germany
First published on 23rd February 2026
Abstract
Understanding the sources, distribution, and lifetime of inorganic chlorine-containing trace gases is crucial to assessing their tropospheric impacts. We report in situ measurements of Cl2, HOCl, ClNO2, and ClONO2 using iodide chemical ionization mass spectrometry during a 2.5-week campaign in June 2024 at a rural continental site in central Germany. Air masses that had passed over “marine-anthropogenic” regions (≈400 km distant) showed significantly higher mixing ratios of chlorine-containing gases than “continental-unpolluted” air masses. From the marine-anthropogenic period, we provide the first quantitative observations of ClONO2 in the lower troposphere (up to 59.8 pptv during daytime). Persistent nonzero ClONO2 at night implies a non-photochemical source of ClONO2 or its precursor ClO, and/or that heterogeneous loss is slower than laboratory uptake coefficients suggest. ClNO2 levels were consistent with production via N2O5 uptake on chloride-containing particles; both ClNO2 and Cl2 were enhanced when O3-/N2O5-rich air entrained into the nocturnal boundary layer. Photolysis of ClNO2, Cl2, and HOCl yielded mean maximum Cl atom production rates of 1.0 × 106 cm−3 s−1 under marine-anthropogenic influenced air and 1.6 × 105 cm−3 s−1 under continental-unpolluted conditions. In the early morning, Cl production (due to ClNO2 photolysis) exceeded primary–OH production from O3 photolysis, while after noon HOCl photolysis was the dominant Cl source. At low solar zenith angles, HOCl photolysis contributed up to 40% of primary OH. These measurements indicate that Cl atoms can strongly influence hydrocarbon oxidation in similar rural regions, with potential regional and global implications (of up to 15%) for the methane lifetime.
Environmental significanceTropospheric, inorganic chlorine-containing trace gases (Cl-species) provide a source of chlorine atoms that impact e.g. on the rates of hydrocarbon oxidation and thus photochemical O3 formation. We report measurements of Cl2, ClNO2, ClONO2 and HOCl in central Germany, distant from coastal regions and thus not expected to be influenced by marine-emissions of sea-salt. The measurement of ClONO2, the first ever made in the lower troposphere, enabled estimation of nighttime ClO and Cl radical mixing ratios. The impact of Cl2, ClNO2, ClONO2 and HOCl on Cl and OH radical production is shown to be significant in rural continental regions, when air masses have passed marine and anthropogenically polluted regions. Further estimations suggest a significant influence on the methane lifetime. |
1 Introduction
The role of inorganic chlorine trace-gases in atmospheric science is well established for the stratosphere, where molecules such as ClONO2, HOCl, HCl and thermally unstable Cl2O2 play a central role in ozone depletion, most noticeably in polar regions where the catalytic recycling of Cl atoms through a mixture of heterogeneous and photochemical processes leads to the formation of the “ozone-hole”.1–3 In the stratosphere, the source of reactive chlorine trace gases is the photolysis of organic chlorine-containing species such as CH3Cl and CCl2F2, which were emitted into the lower atmosphere in large quantities prior to their ban in the Montreal Protocol in 1989.4–7Chlorine species are highly reactive when in radical forms such as Cl or ClO, resulting in a short lifetime (seconds) and significant chemical effects on their immediate environment. The lifetime of the inorganic radical reservoirs, including photolabile Cl2, ClNO2, ClONO2, and HOCl depends on the actinic flux,8,9 while the lifetime of HCl depends on its reaction with OH and deposition. While there are several observations of ClNO2, HOCl, and Cl2 in the troposphere e.g.,10–14 ClONO2 is considered as a stratospheric trace gas, which is only present in the upper troposphere as a result of intrusion from stratospheric air masses.15–17 In the troposphere, chlorine atoms are important as they contribute significantly to the oxidative capacity and especially the oxidation of hydrocarbons.18–21
The presence of chlorine radicals derived from inorganic reservoirs such as ClNO2 and HOCl enhances the rate of oxidation of hydrocarbons (forming HCl and organic peroxyl radicals), which influences air composition and especially photochemical ozone production.22–24 Understanding the sources, distribution and lifetime of inorganic chlorine trace-gases is thus crucial for evaluating their impact on chemical processes in the troposphere.
ClNO2 and Cl2, HOCl and HCl (in both gas and particle phases) are generally believed to be the most abundant, inorganic chlorine-containing gas phase species in the troposphere.25 Various routes to their formation in the atmosphere are illustrated in Fig. 1 and described below.
The main source of gaseous inorganic chlorine in the troposphere is the release in the form of HCl, e.g., via acid displacement of chloride from sea-salt (mainly NaCl)26–28 and gaseous emission from biomass burning, waste disposal, and fossil fuel extraction.29–33 Additionally, HCl can be formed through H-abstraction from volatile organic compounds (VOCs) by Cl atoms that originate from the mechanisms discussed below.25 In a continental atmosphere, NH3 and HCl react to form NH4Cl, which partially partitions into the particle phase. The heterogenous uptake of N2O5 on chlorinated particles, liberates chloride as ClNO2 into the gas phase.34
| N2O5 + Cl− → ClNO2 + NO3− | (R1) |
N2O5 is formed in the reaction between NO2 and NO3 (the latter formed in the oxidation of NO2 by O3) and, due to its thermal equilibrium with photolabile NO3(R2), is only present in significant concentrations at night:35
| NO2 + NO3 + M ↔ N2O5 + M | (R2) |
The abundance of N2O5 (and thus ClNO2) is closely tied to the availability of NOX and the lifetime of NO3 and is therefore maximized in nocturnal air masses that are either impacted by anthropogenic emissions with high levels of NOX or in which the NO3 reactivity towards unsaturated hydrocarbons is low. NO3 and thus N2O5 lifetimes in forested regions are often very short36,37 as the abundance of terpenoids, which react rapidly with NO3, is high. In this case, activation of reactive chlorine from its particle reservoirs is inefficient and the formation of ClNO2 is suppressed. High levels of ClNO2 are typically found at night in polluted marine areas such as Asian megacities and industrial metropolitan areas.38–41 ClNO2 has also been observed in air masses which have been transported long distances from polluted and marine source regions to continental sites.10,13,14
Once formed in the multiphase reaction between N2O5 and particle-chloride, most of the ClNO2, which has a low solubility,42 is released into the gas phase where it is photolyzed to Cl and NO2(R3).43–45 Other loss processes, such as reaction with OH are negligible by comparison.46
| ClNO2 + hv → Cl + NO2 | (R3) |
ClNO2 can also react with acidified (pH < 2), chloride-containing particles to generate gas-phase Cl2:
| ClNO2 + Cl− → Cl2 + NO2− | (R4) |
Cl2 is also formed through the uptake of O3 onto acidic, chloride-containing particles30 and (as shown below) through other heterogeneous processes.47 During daytime, Cl2 is rapidly photolyzed in the troposphere (lifetime ∼10 min) to form two Cl atoms:
| Cl2 + hv → 2Cl | (R5) |
The ClO radical is formed when Cl atoms react with O3(R6).34 ClO radicals then react with HO2 and NO2 to form HOCl and ClONO2, respectively ((R7) and (R8)).
| Cl + O3 → ClO + O2 | (R6) |
| ClO + HO2 → HOCl + O2 | (R7) |
| ClO + NO2 + M → ClONO2 + M | (R8) |
As HOX radicals are ubiquitous in the atmosphere, the ClO radical is the limiting trace gas for formation of HOCl. Consequently, larger mixing ratios of the latter have been measured near source regions with high chloride load such as marine sites or industrial facilities.11,48 The formation of ClONO2 requires the presence of NO2 and is thus expected to be formed at largest rates in regions with both anthropogenic influence and particulate chloride as precursor for Cl-atoms and thus ClO.
HOCl and ClONO2 are lost in both gas- and multiphase processes. Similar to ClNO2, the uptake of HOCl or ClONO2 to chloride-containing, acidic particles results in the generation of Cl2 ((R9) and (R10)),49,50 while heterogenous hydrolysis of ClONO2 forms HOCl (R11).51 The photolysis of HOCl leads to the production of Cl atoms and OH radicals (R12), whereas the photolysis of ClONO2 leads mainly to Cl atoms (R13a) with a minor contribution to ClO formation (R13b).34
| HOCl + Cl− → Cl2 + OH− | (R9) |
| ClONO2 + Cl− → Cl2 + NO3− | (R10) |
| ClONO2 + H2O → HOCl + HNO3 | (R11) |
| HOCl + hv → Cl + OH | (R12) |
| ClONO2 + hv → Cl + NO3 | (R13a) |
| ClONO2 + hv → ClO + NO2 | (R13b) |
Measurements of chlorine-containing trace-gases in the troposphere date back to the mid-1980s, whereby chromatographic methods were used to monitor e.g., HCl and HOCl.26,52 Recent developments in mass spectrometry have enabled sensitive and selective detection of several chlorinated trace gases with chemical-ionization mass spectrometry (CIMS) using iodide as the reagent ion for a wide variety of inorganic chlorine species.11,23,53,54 The recent development of high-resolution (e.g., Time-of-Flight, ToF) analysers allows identification of non-fragmented chlorine species and therefore offers high selectivity as well as excellent sensitivity. However, the number of studies using such devices to examine tropospheric chlorine chemistry is still limited and those available mostly report measurements in polluted marine regions. The presence of chlorine-species in rural continental sites, where the source of chlorine is obscure, remains largely unexplored.
In this study we present the quantitative measurement of Cl2, HOCl, ClNO2 and ClONO2 using VUV-Iodide-ToF-CIMS during field measurements in central Germany. The formation and loss of inorganic chlorine-containing species throughout the diel cycle are discussed, including the first quantitative measurements of ClONO2 in the troposphere and potential limitations by interfering secondary ion reactions. The goal of our analysis is to assess the influence of chlorine species on local Cl and OH production for air masses with marine-anthropogenic and continental-unpolluted origin and to estimate the impact on e.g., the lifetimes of methane and non-methane hydrocarbons.
2 Methods: CIMS operation and calibration
The inorganic chlorine trace-gases ClNO2, ClONO2, HOCl and Cl2 as well as N2O5 were measured using an Iodide-ToF-CIMS (Aerodyne Research Inc., USA55) during the BISTUM24 campaign (Section 2.3) and in further laboratory studies (Section 2.2 and Supplement). A vacuum ultraviolet (VUV) lamp and 20 ppmv CH3I in N2 were used to generate iodide primary ions, which form clusters with the inorganic chlorine-containing compounds in the ion molecular reactor (IMR). The IMR was maintained at 170 mbar, heated to 60 °C and continuously humidified (>7000 ppm H2O). The chlorine-containing trace gases and N2O5 were detected as clusters with iodide at the mass-to-charge ratios (m/z) listed in Table 1. During all measurements, the CIMS resolution was m/z/Δm/z = 5000 at m/z 250. Peak assignment and data processing was conducted with Tofware V4.0.3, ensuring that isotopic ratios are in accordance with their natural abundance. Iodide clusters with Cl and ClO were also observed but were minor in magnitude (<3%) of the signal from ClNO2. These were excluded from further analysis as they most likely originate from the fragmentation of larger chlorine-containing species39 as indicated by high correlations with ClNO2 (R2 = 0.96). The unambiguous first in-field detection of ClONO2 is shown in the mass spectra in Fig. S2.| Species | I−-cluster/m/z | LoD lab/pptv | LoD campaign/pptv | Calibration method | Uncertainty/% |
|---|---|---|---|---|---|
| a Uncertainties calculated via error-propagation considering e.g. calibration methods and the linear regression of the calibration curves (Fig. S3 and S4): a<7% (mainly uncertainty in Cl2 cross-sections56 and humidity correction). b ∼8% (measurement uncertainty in Cl2 and humidity correction, assumes 100% conversion of HOCl to Cl2.) c 28% (same as N2O5, assumes 100% conversion of N2O5 to ClNO2). d12% (uncertainty in ClONO2 IR-cross-sections57 and humidity correction). e28% (uncertainty in NO2 cross-sections and conversion of N2O5 to NO2.58 Uncertainty in dilution factor is based on a flow controller uncertainty of 0.6%. The uncertainty associated with the humidity dependence function is: HOCl: 3.7%, Cl2: 5.3%, ClNO2: 0.9%, ClONO2: 6.1% and N2O5: 1.5%. | |||||
| Cl2 | 196.843 | 0.05 | 0.03 | Direct (Cl2 gas), CRDS | 7a |
| HOCl | 178.877 | 3.50 | 2.21 | Indirect (HOCl-to-Cl2 conversion) | 8b |
| ClNO2 | 207.867 | 0.19 | 0.10 | Indirect (N2O5-to-ClNO2 conversion), TD-CRDS | 28c |
| ClONO2 | 223.862 | 0.20 | 0.36 | Direct (synthesis), FT-IR | 12d |
| N2O5 | 234.886 | 0.12 | 0.03 | Direct (synthesis), TD-CRDS | 28e |
2.1 VUV lamp as reagent ion source in CIMS
Iodide reagent ions in the CIMS were generated using a VUV lamp mounted in front of the IMR. The VUV lamp (10.6 eV photon energy, PKR 106, Heraeus, Germany) was powered with a current of 2 mA at 1500 V DC. An electronic pre-filter was used to reduce noise from the switching power supply (Fig. S1).Two major changes were implemented in our setup in contrast to the VUV lamp assembly described by Ji et al.59 First, an acceleration voltage of 5 V was used for the ions to guide them into the IMR, which doubled the reagent ion yield. Second, the reagent ion flow was directed towards the VUV lamp before leaving the photo-ionization region. This has the advantage of maximizing the irradiance of the reagent gas shortly before the ions enter the IMR and requires less accurate alignment of the VUV lamp in its stainless-steel tube. A detailed overview of the differences between the VUV lamp and 210Po ion sources with regard to the characteristics of Iodide-CIMS spectra can be found in Ji et al.59
2.2 Calibration of chlorine-containing species
Relative humidity dependent calibrations of Cl2, ClNO2, ClONO2, HOCl and N2O5 were conducted post-campaign. In addition, we examined the effect of adding O3 to flows of ClNO2 and HOCl to assess potential secondary ion chemistry including the formation of IClONO2− and IHO2Cl− (presented in SI). A summary of calibration curves for ClNO2, ClONO2, Cl2 and HOCl can be found in Fig. S3. Higher mixing ratios than found in the atmosphere were used to enable simultaneous detection with less sensitive but absolute methods e.g., CRDS and FT-IR with dynamic dilution for the CIMS.An expression for the humidity-dependence of the calibration signal (for constant trace-gas mixing ratio) is given by polynomial fits (Fig. S4) that cover the full ambient humidity, i.e., IH2O−/(I− + IH2O−) range so that no extrapolation beyond these humidities was necessary. Uncertainties associated with the humidity-dependent calibrations and the detection limits during the campaign and laboratory calibration are presented in Table 1.
2.3 BISTUM24 field campaign: measurement site and instrumentation
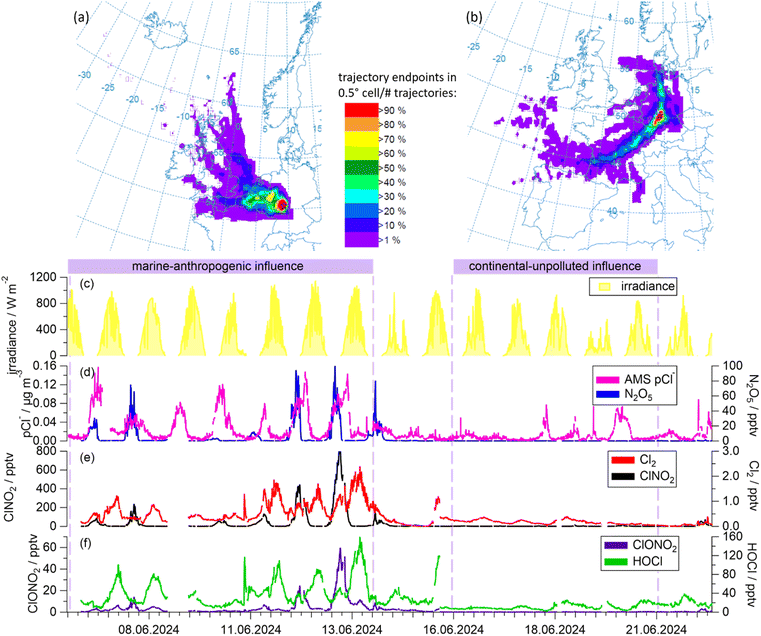
The BISTUM24 field campaign took place from June 6 to 22, 2024 in Spielberg, central Germany (50°19′N, 9°15′E, Fig. S6). The rural measurement site, which is located 391 m above mean sea level on top of a hill in a treeless clearing, was selected because air masses encountered at the site originate mainly from rural regions and are rarely influenced by urban pollution despite the proximity to the Frankfurt metropolitan area (∼45 km). With a distance of ∼400 km to the next coastline, the North and Baltic Seas, the site may be described as continental with no direct marine influence (Fig. 2a and b).The instruments used in this study sampled from inlets at a height of 6 m (Fig. S7) and were housed in the “Mobile Laboratory”.63 These included the Iodide-ToF-CIMS and an Aerosol Mass Spectrometer (AMS, Aerodyne Research Inc., USA), as well as an O3 monitor (TB205, Monitor 205 Dual Beam Ozone Monitor, 2B Technologies, Inc., USA) and a pyranometer (CMP3 Pyranometer Sensor, Campbell Scientific Inc., UK). The surface area and volume of aerosol particles were calculated from a merged size distribution from different particle sizers (Fast Mobility Particle Sizer Model 3091, TSI, Inc., USA and Optical Particle Counter Model 1.109, Grimm Aerosoltechnik GmbH, Germany).64 Local meteorological information was provided by a meteorological station (WXT520, Vaisala Oyj, Finland).
The CIMS was connected to the inlet via 6 m of PTFE tubing for which the residence time was ∼1 s (corresponding to a total flow of 32 L min−1). The CIMS sub-sampled 2 L min−1 from this flow. The inlet line also included a PTFE-particle filter, which was changed after every 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 liters of sampled air. While heterogeneous conversion of chloride to chlorine trace gases on filters or inlet lines can be an issue in coastal regions with high sea-salt loadings,11,65 this is not expected to occur at the present location where chloride is not associated with sea-salt. The non-observation of ClNO2 formation despite the presence of high concentration of N2O5 and the use of “aged” inlet lines supports this assumption. Zero measurements were performed every 8 h for 20 min during the campaign using humidified N2. The CIMS data were zero-corrected, and the signal at each mass analyzed was normalized to the sum of the most abundant reagent ions, I− and IH2O−. Calibration factors were applied, determined as described in Section. 2.2.
000 liters of sampled air. While heterogeneous conversion of chloride to chlorine trace gases on filters or inlet lines can be an issue in coastal regions with high sea-salt loadings,11,65 this is not expected to occur at the present location where chloride is not associated with sea-salt. The non-observation of ClNO2 formation despite the presence of high concentration of N2O5 and the use of “aged” inlet lines supports this assumption. Zero measurements were performed every 8 h for 20 min during the campaign using humidified N2. The CIMS data were zero-corrected, and the signal at each mass analyzed was normalized to the sum of the most abundant reagent ions, I− and IH2O−. Calibration factors were applied, determined as described in Section. 2.2.
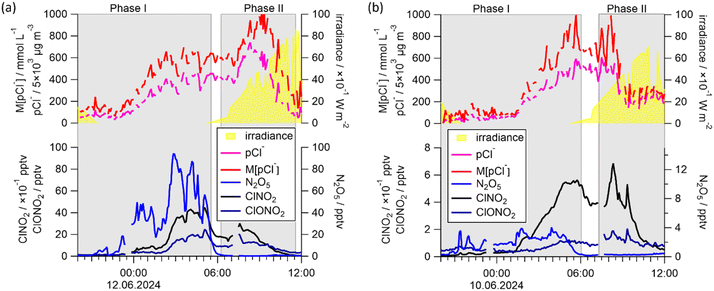
3 Chlorine-containing compounds and air mass origin
Fig. 2 provides a time series of the chlorine-containing trace gases ClNO2, ClONO2, HOCl, Cl2 and N2O5, measured by the CIMS, as well as particulate chloride (pCl−) as measured by the AMS. Although the AMS does not efficiently detect pCl− from sea salt, it can detect pCl− from, e.g., ammonium chloride. For this reason, pCl− does not necessarily represent the total amount of particulate chloride present in the particle phase. Information on airmass origin was gained using HySplit66,67 with which 48-h backward trajectories ending 30 m above ground level at the measurement site were calculated.In the first phase (June 6 to 14), back-trajectories showed that the air passed over the English Channel and industrialized regions in Belgium and the Netherlands, as well as the German Ruhr metropolitan area before reaching the measurement site. The transport times were 12 to 36 h for UK and Benelux and 4 to 6 h for the Ruhr region, respectively. During transport, trajectories rarely exceeded altitudes of 500 m above mean sea level, indicating that the air was transported within the boundary layer. We refer to the air masses in this period as having a “marine-anthropogenic” influence as evidenced by the enhanced levels of pCl− and N2O5 (Fig. 2d).
In contrast, backward trajectories for the period June 16 to 21 indicate that the air sampled during this period travelled at altitudes > 1000 m above mean sea level for several days before descending close to the measurement site. The vast majority of these trajectories passed over the relatively sparsely populated, non-industrialized center of France and rural south-west Germany (Fig. 2b and S8b). We designate these air masses as “continental-unpolluted”. In these air masses, N2O5 was generally below the detection limit and concentrations of pCl− were much lower than those of the other air masses.
As expected, the mixing ratios of Cl2 and ClNO2 were largest during the “marine-anthropogenic” period, with ClNO2 exceeding 70 pptv every night and Cl2 exceeding 0.5 pptv every day (Fig. 2e). HOCl and ClONO2 were less abundant than ClNO2, with mixing ratios of up to 60 pptv (Fig. 2f). During this period, mean mixing ratios of 56.2 pptv ClNO2, 0.64 pptv Cl2, 41.3 pptv HOCl and 4.8 pptv ClONO2 were observed, whereas during the “continental-unpolluted” period, 2.25 pptv ClNO2, 0.17 pptv Cl2 and 11.1 pptv HOCl were measured and ClONO2 levels were below the detection limit.
Worldwide, the mixing ratios of inorganic chlorine species vary considerably depending on the air masses sampled and the geographical location. Our ClNO2 mixing ratios are consistent with those observed at continental sites with similar (indirect) marine-influenced air masses, which show peak ClNO2 concentrations of 300–700 pptv.13,14 These mixing ratios are higher than those measured in maritime air,22,48 which results from a longer residence time over landmasses with a greater anthropogenic influence, which is crucial for the formation of ClNO2 (see Section. 1). Consequently, for highly polluted sites like in Hong Kong or Los Angeles up to 3.5 ppbv of ClNO2 have been reported.68,69
During both periods of the BISTUM24 campaign, the Cl2 levels were generally low compared to those reported in other studies, especially those influenced by highly polluted urban regions.48,70 Cl2 mixing ratios in the marine boundary layer vary from ∼2 to ∼500 pptv11,48,69,71,72 and are enhanced compared to rural continental sites ranging from ∼2 to ∼33 pptv,22,73 depend heavily on the agricultural land use in region. There are no previous reports of ClONO2 measurements in lower troposphere with which to compare our data.
Results from our HOCl measurements are broadly consistent with the 2–120 pptv HOCl range reported for continental and marine air masses.11,48 The large variability is related to air mass history, even when only marine regions are considered.
Fig. 2 reveals substantial differences in the diel profiles of the trace gases measured. N2O5 and ClNO2 were observed predominantly at nighttime whereas HOCl and Cl2 were observed predominantly during the day. The absence of N2O5 during the day is expected ((R2), Section 1) and, as N2O5 is the precursor of ClNO2, the nighttime dominance of the latter is also consistent with present knowledge of its production and loss terms (Section 1). pCl− has a less clear day–night variation which is related to the presence of multiphase equilibria, whereby chloride is present as a dissolved ion in the particle phase or degassed from aerosols as HCl. For the mixing ratios of ClONO2, the two strongest features were observed after midnight on the nights of 13th and 14th June with strong co-variance with ClNO2. This was unexpected, as ClONO2 is believed to be formed exclusively in the reaction of ClO with NO2, with the ClO-radical being present at significant levels only during the daytime when photochemically generated Cl atoms react with O3.
While many studies have reported ClNO2 and Cl2 in coastal regions39,48,54,74 or in highly polluted regions where non-marine chloride sources exist,12,32,33,75 only few10,13,22 have identified reactive chlorine trace-gases in air masses that may be described both as non-marine (i.e., several hundred km from coastal regions) and also not impacted by local pollution. Of these, only Phillips et al.13 investigated chlorine chemistry in mainland Europe. They argued that, as coarse mode sea-salt particles are not efficiently transported hundreds of km, the source of particle chloride was HCl that was released from coarse-mode sea-salt particles close to coastal regions (by, e.g., acid displacement) and subsequently transported as gas phase HCl or particulate ammonium chloride. As Phillips et al.13 did not measure other chlorine-containing trace-gases apart from ClNO2, they did not consider the potential role of HOCl, Cl2 or ClONO2 in, e.g., the production rate of Cl atoms or OH radicals.
4 The diel cycle of Cl2, HOCl, ClNO2 and ClONO2
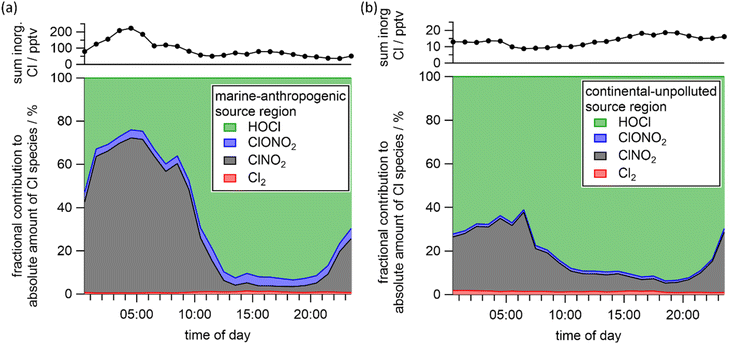
Fig. 3 displays the mean, fractional contribution of each individual chlorine-containing trace gas to the total inorganic chloride mixing ratios during BISTUM24. For the “marine-anthropogenic” influenced phase (a), ClNO2 is dominant during the night (max. ∼70%), with HOCl being dominant during the day (max. ∼90%) and ClONO2 and Cl2 representing only a few percent of overall chlorine-containing species throughout the diel cycle. During the “continental-unpolluted” phase (b), HOCl is the dominant chlorine-containing trace gas both day and night, accounting for > 60% during the night and >80% during the day. Both ClONO2 and Cl2 are minor contributors with ∼1% each, while the nocturnal ClNO2 contribution is relatively low due to N2O5-poor conditions (∼30%). The diel profile of ClNO2 is understood in terms of its generation through multiphase chemistry involving N2O5 during the night, followed by its daytime photolysis to release Cl atoms that (via reactions of HO2 and ClO) lead to daytime HOCl. The significant nighttime contribution of HOCl is likely related to incomplete loss from the previous day rather than nocturnal formation, for which a non-radical mechanism is unknown. The heterogenous loss rate coefficient (khet) for HOCl with respect to uptake on aerosol particles was calculated with eqn (1):| khet = 0.25γAcHOCl | (1) |
with the mean molecular velocity cHOCl = 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 cm s−1, derived from the molecular weight of the gas species for 298 K, the mean aerosol particle surface during the campaign A = 130 µm2 cm−3, which is representative also for nighttime (Fig. S10), and the uptake coefficient γ < 2 × 10−4 that is available for particles with pH = 1.34
700 cm s−1, derived from the molecular weight of the gas species for 298 K, the mean aerosol particle surface during the campaign A = 130 µm2 cm−3, which is representative also for nighttime (Fig. S10), and the uptake coefficient γ < 2 × 10−4 that is available for particles with pH = 1.34
The calculated lifetime of HOCl due to uptake on acidic particles (khet−1 ≈ 5 days) is much longer than the lifetime with respect to photolysis (∼1 h, Table 2). Hence, the observed nighttime HOCl was most likely generated photochemically the previous day.
| Species | J s−1 | Lifetime min |
|---|---|---|
| a Thermolabile N2O5 forms NO3 during daytime in an equilibrium. Fast photolysis of NO3 significantly decreases the N2O5-lifetime. | ||
| O3(1D) | 3.52 × 10−5 | 473 |
| ClNO2 | 4.97 × 10−4 | 33.5 |
| ClONO2 | 4.97 × 10−5 | 335 |
| Cl2 | 2.43 × 10−3 | 6.9 |
| HOCl | 2.86 × 10−4 | 58.3 |
| NO3 | 2.37 × 10−2 | 0.7 |
| N2O5 | 4.78 × 10−5 | 349a |
4.1 ClNO2
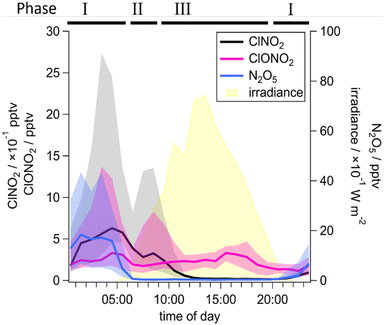
In the following, we investigate the formation and loss of ClNO2 through the diel cycle during the “marine-anthropogenic” period and highlight the influence of air mass history and vertical mixing on the ClNO2 abundance during two individual night–day transitions.In Fig. 4 we present the median, diel cycles of the mixing ratios of ClNO2, ClONO2 and N2O5 over the entire “marine-anthropogenic” time period. The diel cycle is divided into three phases which represent (I) nighttime, when N2O5 is present, (II) the period just after sunrise when N2O5 is rapidly depleted and the mixing ratios of ClNO2 and ClONO2 are either constant or increasing, and phase III when irradiance is stronger and ClNO2 mixing ratios decrease monotonically to zero.
The formation of ClNO2 depends on the presence of N2O5 and pCl−(R1) and thus we expect net-production only when N2O5 and pCl-are present. The data in phase I and II from June 10 and 12, 2024 show that this is not always reflected in the ClNO2 mixing ratios which remain constant or sometimes even increase during the early morning when N2O5 is absent, although the photolytic loss of ClNO2 increases with intensifying irradiance. Note that, subsequent to a post-dawn peak, the ClNO2 mixing ratios decrease with a decay constant of 5 × 10−4 s−1 in accord with JClNO2 (Table 2).
At the beginning of the night from June 12, from 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00 to 01
00 to 01![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 (Fig. 5a), an approximately linear increase in ClNO2 of (19 ± 1) pptv h−1 was observed. The ClNO2 production rate can be calculated from khet, the N2O5 mixing ratio and the fraction α of N2O5 that forms ClNO2:
30 (Fig. 5a), an approximately linear increase in ClNO2 of (19 ± 1) pptv h−1 was observed. The ClNO2 production rate can be calculated from khet, the N2O5 mixing ratio and the fraction α of N2O5 that forms ClNO2:
| P(ClNO2) = [N2O5]αkhet | (2) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 cm s−1. Considering a mean [N2O5] = 18 pptv, and α = 0.8 given for a 0.22 M chloride in particles,62 a ClNO2 production rate of 7.7 pptv h−1 is obtained. Given the factor 3 uncertainty in γ34,77 and the AMS uncertainty in pCl− and thus,78 the calculated P(ClNO2) is consistent with the observed, net ClNO2 formation rates.
800 cm s−1. Considering a mean [N2O5] = 18 pptv, and α = 0.8 given for a 0.22 M chloride in particles,62 a ClNO2 production rate of 7.7 pptv h−1 is obtained. Given the factor 3 uncertainty in γ34,77 and the AMS uncertainty in pCl− and thus,78 the calculated P(ClNO2) is consistent with the observed, net ClNO2 formation rates.
 | ||
| Fig. 5 Mixing ratios of ClNO2 (black), ClONO2 (dark blue) and pCl− (as mass (pink) and molar (red) concentration) through the diel cycle on June 10 (a) and 12 (b), 2025. | ||
After 01 : 30 the ClNO2-to-N2O5 ratio switches from ∼2 to ∼6, and the high ClNO2 levels in the next 4 h cannot be explained by local production from N2O5. This trend continues in phase II in Fig. 5a, where an increase in pCl− (up to 1 M Cl−) was accompanied by an increase in ClNO2 levels although N2O5 levels were close to zero. A similar picture emerges from the data in Fig. 5b, in which post-dawn peaks in ClNO2 (∼55 pptv between 05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00 and 06
00 and 06![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00 and ∼69 pptv between 08
00 and ∼69 pptv between 08![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 and 09
20 and 09![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20) are observed despite the production term (in the absence of N2O5) being zero. In both cases, the in situ production of ClNO2 from N2O5 reaction with pCl− can be ruled out and the increase in ClNO2 after dawn is attributed to the sampling of air masses in which N2O5 had been previously converted to ClNO2. This simply reflects the fact that ClNO2 is substantially longer-lived during the day than N2O5 as JClNO2 is much lower than JNO3 (which controls the N2O5 lifetime due to its equilibrium with N2O5).
20) are observed despite the production term (in the absence of N2O5) being zero. In both cases, the in situ production of ClNO2 from N2O5 reaction with pCl− can be ruled out and the increase in ClNO2 after dawn is attributed to the sampling of air masses in which N2O5 had been previously converted to ClNO2. This simply reflects the fact that ClNO2 is substantially longer-lived during the day than N2O5 as JClNO2 is much lower than JNO3 (which controls the N2O5 lifetime due to its equilibrium with N2O5).
The phase II, post-dawn peaks in ClNO2 were found to be enhanced for those days when backward trajectories showed subsiding air masses 1–3 h before the air reached the measurement site (see interquartile range in Fig. 4). The air from higher altitudes entrains O3-rich residual layer air into the nocturnal boundary layer,79 which results in enhanced formation of N2O5 and thus of ClNO2. Additionally, at dawn when the nocturnal boundary layer dissipates through radiative heating and convective mixing of air from the residual layer, O3-rich air may be transported to the surface. The daily development and breakdown of the nocturnal boundary layer can also cause variability in ClNO2 levels due to vertical mixing.
4.2 ClONO2
While ClNO2, has been measured in the lowermost troposphere on many occasions,10,12–14 the detection of ClONO2 during BISTUM24 was unexpected. Previous studies have demonstrated the potential of using CIMS methods with SF6− and I− to measure ClONO2 as FClNO3− and IClNO3−,15,75 and airborne SF6−-CIMS measurements have quantified ClONO2 in the upper troposphere where its presence (at levels up to 10 pptv) was explained by the entrainment of stratospheric air.15Our study presents the first quantitative ambient ground-level ClONO2 measurements using Iodide-CIMS. As already mentioned, the accepted mechanism for formation of atmospheric ClONO2 is (R8), which requires the presence of ClO and NO2. ClO is a radical that is formed as a result of photochemical processes and expected to be present predominantly during daytime; we therefore expect reaction (R8) to be responsible for the net increase in ClONO2 observed during the day (Phase III in Fig. 5). However, following this line of reasoning ClONO2 is not expected to be formed in significant rates during the night, which we nonetheless observe (Phase I in Fig. 5). In the following, we first calculate the ClONO2 lifetime and then estimate the production rate that would be required to maintain the observed levels. The photolysis frequency of ClONO2 is 4.97 × 10−5 s−1, resulting in a lifetime of ∼5–6 h with respect to this loss process ((R13), Table 2). The heterogeneous loss frequency of ClONO2 was calculated from eqn (1) with the uptake coefficient on aqueous particles γ = 0.027 and cClONO2 = 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 590 cm s−1.34 Using a campaign mean particle surface area density A of 130 µm2 cm−3 results in a loss rate constant of khet = 2.25 × 10−4 s−1, or a lifetime of ∼75 min. Given this short lifetime with respect to heterogeneous uptake, ClONO2 cannot survive from the previous day and, in the absence of sources, its mixing ratios should tend to zero in the first hours during the night. In order to explain the observed mean ClONO2 mixing ratio of 4.3 pptv at 05
590 cm s−1.34 Using a campaign mean particle surface area density A of 130 µm2 cm−3 results in a loss rate constant of khet = 2.25 × 10−4 s−1, or a lifetime of ∼75 min. Given this short lifetime with respect to heterogeneous uptake, ClONO2 cannot survive from the previous day and, in the absence of sources, its mixing ratios should tend to zero in the first hours during the night. In order to explain the observed mean ClONO2 mixing ratio of 4.3 pptv at 05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00 during the night, we require a nocturnal production rate of ∼2.4 × 104 molecules cm−3 s−1, which can be equated to P(ClONO2) = kNO2+ClO × [ClO] × [NO2]. Assuming an NO2 mixing ratio of 1–2 ppbv during the “marine-anthropogenic” period as measured at a similar site with similar prevailing wind direction80 and that the sole source of ClONO2 is the reaction between ClO and NO2 (with a rate coefficient, kNO2+ClO, at 298 K and 1 bar of 2.4 × 10−12 cm3 molecule−1 s−1, IUPAC) we calculate that the ClO concentration necessary to maintain this level of ClONO2 is [ClO] = (2–4) × 105 molecules cm−3, where the spread in values results from the range of NO2 mixing ratios assumed for the calculation.
00 during the night, we require a nocturnal production rate of ∼2.4 × 104 molecules cm−3 s−1, which can be equated to P(ClONO2) = kNO2+ClO × [ClO] × [NO2]. Assuming an NO2 mixing ratio of 1–2 ppbv during the “marine-anthropogenic” period as measured at a similar site with similar prevailing wind direction80 and that the sole source of ClONO2 is the reaction between ClO and NO2 (with a rate coefficient, kNO2+ClO, at 298 K and 1 bar of 2.4 × 10−12 cm3 molecule−1 s−1, IUPAC) we calculate that the ClO concentration necessary to maintain this level of ClONO2 is [ClO] = (2–4) × 105 molecules cm−3, where the spread in values results from the range of NO2 mixing ratios assumed for the calculation.
A well-known source of gas phase ClO is the reaction between Cl atoms and O3(R6). Thus, the presence of nighttime ClONO2 would require a reaction that generates either Cl atoms or ClO directly, both in the absence of sunlight. The conversion of Cl to ClO is directly influenced by O3 levels and competing Cl reactions with, e.g., hydrocarbons and NOX trace gases. The loss rate constant of Cl through its reaction with O3 is given as kCl+O3 × [O3] = 11.8 s−1 (calculated from the mean average O3 concentration (40 ppbv) and the rate coefficient kCl+O3 = 1.2 × 10−11 cm3 molecule−1 s−1)34.
The first-order loss rate constant of Cl atoms reacting with the most abundant alkanes can be calculated from kCl+CH4 × [CH4] + kCl+C2H6 × [C2H6] + kCl+C3H8 × [C3H8]. As concentrations of these hydrocarbons were not measured during the campaign, we use reference mixing ratios of 1.9 ppm CH4, 0.8 ppbv C2H6, and 0.3 ppbv C3H8 that are typical for rural continental conditions and the corresponding rate coefficients with Cl.34,81 The calculated total loss rate constant for reaction with these hydrocarbons is ∼6.8 s−1, with the largest single contributions from methane (4.6 s−1, i.e., ∼25% of Cl). We acknowledge that loss of Cl atoms due to terpenoids can be significant, however, their concentration is highly variable (even in rural continental regions) and their contribution is highly uncertain (considering 1 ppbv terpenoids with kCl+isoprene = 4.6 × 10−10 molecules cm−3 s−1 would result in a loss rate to terpenoids of ∼11.2 s−1,82i.e., the same as the loss rate to O3 adding a uncertainty factor of 2 to the calculation). Cl atoms react with NO2 with a rate coefficient of 6.7 × 10−12 cm3 molecule−1 s−1 campaign conditions.83 Assuming (as above) that NO2 mixing ratios were between 1 and 2 ppbv at night results in a Cl atom loss rate constant of ∼0.3 s−1.
Taking the formation rate of ClO from Cl (11.8 s−1) and the loss rates of Cl through other pathways (6.8 s−1 and 0.3 s−1), we can conclude that approximately 60% of the Cl atoms generated react to form ClO. We can estimate the production rate of ClO by assuming that its concentration is in steady state with respect to its gas phase formation and loss processes at night. In the absence of NO, the dominant loss process for ClO at night is its reaction with NO2, with a loss rate of 2.4 × 104 molecules cm−3 s−1. Using the 60% ClO yield from Cl, this in turn can be converted into a Cl atom production rate P(Cl) = kNO2+ClO × [ClO] × [NO2]/0.6 = (2.0–3.9) × 104 atoms cm−3 s−1.
We now examine potential reactions that, at nighttime, could lead to Cl atom production rates of this magnitude. The dominant radical at nighttime is often considered to be NO3, which initiates the oxidation of many biogenic hydrocarbons. However, reactions of NO3 radicals with sufficiently abundant (non-radical) chlorine species to generate ClO or Cl atoms are either unknown or expected to be too slow to contribute significantly. For example, the rate coefficient for the reaction between NO3 and HCl (R14) is < 5 × 10−17 cm3 molecule−1 s−1,34 which would result in a maximal Cl atom production rate of 1.2 × 102 atoms cm−3 s−1, if we assume typical mixing ratios of 100 pptv HCl84,85 and 10 to 40 pptv NO3,80 which is a factor ∼350 lower than that required.
| HCl + NO3 → Cl + HNO3 | (R14) |
As the reaction between NO3 and HCl cannot explain the nighttime production of Cl atoms, we consider the potential role of OH radicals, which react much more rapidly with HCl ((R15), kOH+HCl = 7.8 × 10−13 cm3 molecule−1 s−1)34.
| HCl + OH →Cl + H2O | (R15) |
OH radicals are present at night owing to their formation in the ozonolysis of unsaturated hydrocarbons,86,87 and their abundance has been the subject of much research, with concentrations found to be varying between 9.1 × 104 and 7 × 106 molecules cm−3 at night time.88–91 Due to the absence of OH measurements during BISTUM24, we assume a nighttime concentration of 1 × 105 molecules cm−3 that was determined in a similar agricultural rural environment.88 For the HCl mixing ratio, we assume a value of 100 pptv HCl (as measured over the continent85) and calculate a Cl production rate of 1.9 × 102 atoms cm−3 s−1, which is a factor 100–200 smaller than the value of (2.0–3.9) × 104 atoms cm−3 s−1 derived above.
The nocturnal ozonolysis of unsaturated hydrocarbons also leads to the formation of Crigee-intermediates.87,92 Studies of the reaction of HCl with Crigee-intermediates appears however not to form Cl or ClO but R1R2OOCl compounds, where R1 and R2 are organic groups.93
In addition, we also consider the direct formation of ClO through the reaction of OH with HOCl (R16):
| HOCl + OH → ClO + H2O | (R16) |
for which the rate coefficient at 298 K is kOH+HOCl = 5.0 × 10−13 cm3 molecule−1 s−1.34 Assuming again an OH concentration of 1.0 × 105 molecules cm−3 during night88 and nocturnal campaign mean mixing ratio of 50 pptv for HOCl we derive a ClO production rate of 1.2 × 103 ClO cm−3 s−1 which is a factor ten larger than the rate of formation of Cl from OH + HCl but falls well short of the value required to explain the observations of nighttime ClONO2.
We thus conclude that the presence of ClONO2 at nighttime, despite its rapid heterogeneous loss to particles, cannot be explained by the OH-induced conversion of HCl and HOCl to Cl and ClO, even within the major uncertainties related to the estimations of the HCl and OH levels. We note however, that nighttime Cl levels of 2.0 × 104 atoms cm−3 have been reported in the urban UK,91 although the authors were not able to name a Cl source. Such levels of Cl result in a ClO production rate of 2.4 × 105 molecules cm−3 s−1 which is a factor 10 larger than at required to explain our observations.
At this point, we examine the possibility that ClONO2 is actually significantly longer lived at night than so far calculated based on an uptake coefficient of γ = 0.027,34 which is the result of laboratory experiments performed on inorganic aqueous solutions containing sulphate, halides etc. We now draw analogy to the heterogeneous uptake to aqueous particles of another di-acid anhydride, N2O5 for which γ from laboratory studies is also very similar (∼0.02).34 In contrast to ClONO2, the tropospheric, multiphase chemistry of N2O5 has been the subject of many studies and the rate of uptake to “real” atmospheric particles that are internal mixtures of organics, sulphates, nitrates etc. is a strong function of composition, where the presence of organic has been observed to reduce γ significantly.94 As the mechanism of uptake of N2O5 and ClONO2 to aqueous particles is expected to be similar (both hydrolyze/ionize on/at the surface) it is very possible that γ and the rate of uptake of ClONO2 is greatly reduced compared to the values calculated above, and that the source term required to generate the observed mixing ratios of ClONO2 is actually lower. In the absence of laboratory studies investigating the dependence of γ(ClONO2) on e.g., particle organic content, this remains speculative. In addition, while no appropriate mechanism is known to us, we cannot rule out alternative (potentially multiphase) routes to ClONO2 formation at nighttime may be more important than the gas phase reaction between ClO and NO2.
However, to illustrate the potential importance of nocturnal Cl production we calculate its effect on the methane lifetime. Based on the measurement of ClONO2, we derived a nocturnal Cl production rate of (2.0–3.9) × 104 atoms cm−3 s−1 (see above). We can combine this with the fact that ∼25% of the Cl atoms formed will react with CH4 (see above) to calculate a CH4 lifetime of ∼45 years with respect to nocturnal loss via reaction with Cl atoms. Given that the lifetime (i.e., the inverse reaction rate) of the important climate gas methane is of the order of 10 years (predominantly through reaction with OH) this translates to a ∼15% fractional contribution to CH4 loss. We note that this calculation is based on the assumption that ClONO2 is formed solely via gas phase reaction of ClO and NO2. A heterogeneous process forming ClONO2 that does not require the formation of Cl atoms would have no effect on hydrocarbon oxidation rates. A more rigorous assessment of the global impact of regional chlorine activation, for example regarding the methane lifetime, requires further related observations, inclusion of realistic chlorine activation scenarios and chlorine photochemistry in global models.
4.3 Formation and diel cycle of Cl2 and HOCl
Median diel cycles of HOCl and Cl2 are depicted in Fig. 6 and show maxima of 60–70 pptv HOCl and 0.8–1.0 pptv Cl2 in the afternoon (Fig. 6), implying that both are largely of photochemical origin. Strong correlations of Cl2 (R2 = 0.88) and HOCl (R2 = 0.85) with O3 (Fig. S11) indicate a photochemical source.Cl2 is known to be formed in the reaction between gaseous chlorine species such as ClNO2, ClONO2 and HOCl with pCl− in acidified aerosol, which may help to explain the timing of the peak Cl2 mixing ratio in the afternoon (phase III) after maximum irradiance ((R4), (R9), (R10), and Oum et al.30). In addition to the daytime peak, a slight increase in Cl2 levels was monitored between 04![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00 to 07
00 to 07![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00, i.e., between phase I and II. This observation may also be linked to the entrainment of O3/N2O5-rich air from the residual layer into the nocturnal boundary layer (Section. 4.1) in agreement with Xia et al.,95 so that particle chloride can be converted into chlorine-containing gases such as Cl2 and ClNO2. Cl2 is also formed in other multiphase processes involving chloride and the intermediary of NO3−, O(3P), Fe(III) and TiO2.47,96–99
00, i.e., between phase I and II. This observation may also be linked to the entrainment of O3/N2O5-rich air from the residual layer into the nocturnal boundary layer (Section. 4.1) in agreement with Xia et al.,95 so that particle chloride can be converted into chlorine-containing gases such as Cl2 and ClNO2. Cl2 is also formed in other multiphase processes involving chloride and the intermediary of NO3−, O(3P), Fe(III) and TiO2.47,96–99
For HOCl, the recognized and only atmospherically relevant route to its generation in the gas phase is through the reaction between two radical species, ClO and HO2(R7) which takes place predominantly during the daytime when active photochemistry leads to high radical concentrations. The particle phase hydrolysis of ClONO2(R11) also leads to the formation of HOCl, which can either degas from the particles or react with particle chloride to form Cl2. As Cl2 is weakly soluble, this will likely transfer to the gas phase where it is rapidly photolyzed (Table 2) to reform Cl atoms that, in turn, reform HCl and the HOCl precursor ClO. As HOCl is also photolabile, its gas phase mixing ratios are determined by chemical cycles that are strongly dependent on actinic flux and the availability of, e.g., O3.
5 Daytime Cl-atom production rates
While the OH radical is the primary driver of oxidation of the vast majority of atmospheric trace gases, the oxidation rates of certain trace gases can be augmented by the presence of Cl atoms. In this section, we examine Cl-atom production rates P(Cl) from the photolysis of ClNO2, ClONO2, HOCl and Cl2 ((R3), (R5), (R12) and (R13)) over the diel cycle:| P(Cl) = JClNO2[ClNO2] + JClONO2[ClONO2] + 2JCl2[Cl2] + JHOCl[HOCl] | (3) |
The photolysis frequencies J of ClNO2, ClONO2, HOCl and Cl2 were derived for each trace gas through the diel cycle (data provided by the TUV calculator for cloudless conditions for the BISTUM24 location76), corrected for cloud-reduced irradiance, measured using a local on-site pyranometer. We recognize that clouds do not impact irradiance in the same way for all wavelengths; however, considering cloudiness provides a more accurate estimate of real photolysis rates.
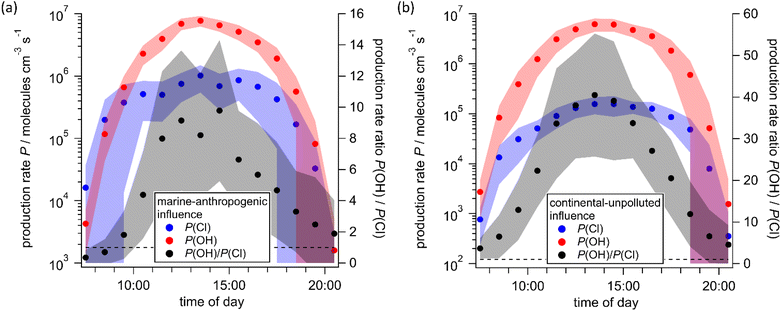
The results of the calculations are summarized in Fig. 7 which shows that Cl production follows the actinic flux, reaching a maximum of (1.0 ± 0.47) × 106 atoms cm−3 s−1 in the period in which we sampled “marine-anthropogenic” air. The maximum for “continental-unpolluted” air is a factor ∼6 lower (1.6 ± 0.7) × 105 atoms cm−3 s−1. The daytime maximum Cl production rates are broadly consistent with summertime observations from Los Angeles100 (6.2 × 105 atoms cm−3 s−1), Houston22 (4.7 × 105 atoms cm−3 s−1), Beijing75 (1.6 × 105 atoms cm−3 s−1) and wintertime Manchester-UK39 (3.0 × 105 atoms cm−3 s−1). Note however, that the values for Houston considered only Cl2 and ClNO2 as Cl sources,22 while additionally, for Los Angeles HCl + OH contributes significantly.100 In the Beijing-study a minor role was attributed to HOCl and ClONO2 (not quantified and considered in Le Breton et al.75) and ClNO2 was considered to be the only Cl sources in marine urban Manchester.39 The fact that Cl production rates were observed in polluted and marine influenced regions, as well as in rural continental areas (this work) indicates the role of Cl chemistry could be more widespread than currently assumed.
The fractional contribution of each chlorine-containing trace gas to Cl atom production rates is highlighted in Fig. 8. HOCl is the major contributor to Cl production from photolysis, accounting in the morning for 10% for “marine-anthropogenic” air masses and 49% for air masses from “continental-unpolluted” source regions. After ∼12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00, the HOCl contribution is in both cases between 70% and 80% (see Fig. 8a and b).
00, the HOCl contribution is in both cases between 70% and 80% (see Fig. 8a and b).
Contrary to HOCl, the contribution of ClNO2 to Cl production decreases from 85% at sunrise (07![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00) to 3% after 13
00) to 3% after 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00 if the air masses were transported from the “marine-anthropogenic” source region. The contribution of ClNO2 in continental unpolluted air, meanwhile, is significantly lower, ranging from 6% to 33%. This observation emphasizes the importance of ClNO2 as a nighttime reservoir species, particularly for air masses affected by polluted areas.
00 if the air masses were transported from the “marine-anthropogenic” source region. The contribution of ClNO2 in continental unpolluted air, meanwhile, is significantly lower, ranging from 6% to 33%. This observation emphasizes the importance of ClNO2 as a nighttime reservoir species, particularly for air masses affected by polluted areas.
For air masses with marine-anthropogenic signature, Cl2 contributes approximately 5% to Cl production rate in the morning hours, increasing to 20% from 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00 on. Despite its much lower mixing ratios, after 11
00 on. Despite its much lower mixing ratios, after 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00 the contribution of Cl2 is larger than that of ClNO2, which is related to the absorption cross section of Cl2 which overlaps much more with the actinic flux (Fig. S12). The contribution of Cl2 to Cl production rates from continental unpolluted air remains around 20% throughout the day.
00 the contribution of Cl2 is larger than that of ClNO2, which is related to the absorption cross section of Cl2 which overlaps much more with the actinic flux (Fig. S12). The contribution of Cl2 to Cl production rates from continental unpolluted air remains around 20% throughout the day.
Low daytime concentrations of maximum 3.3 pptv, coupled with low absorption cross-sections at the wavelengths of the actinic flux, mean that ClONO2 plays a minor role in Cl production. Fig. 8a indicates that ClONO2 contributes only 0.5% to Cl production, independent of the time of day, which is consistent with conclusions of Le Breton et al.75
The impact of a summed, maximum daytime Cl production rate of ∼1 × 106 atoms cm−3 s−1 (at ∼14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00 local time) can also be illustrated by estimating its impact on CH4 oxidation. As discussed in Sect. 4.2, ∼25% of the Cl atoms generated under these conditions will react with CH4. The CH4 loss due to Cl during one hour at noon is ∼40 pptv. This can be compared to the CH4 loss in the same period owing to reaction with OH, which is ∼400 pptv, assuming OH concentrations of 1 × 107 molecules cm−3 using 1.9 ppmv CH4. While these estimations highlight the potential of Cl compared to OH regarding CH4 oxidation, rigorous assessment of the impact of chlorine chemistry on a long-lived climate gas such as CH4 requires implementing appropriate chemistry into global chemistry-transport models, which is beyond the scope of this manuscript.
00 local time) can also be illustrated by estimating its impact on CH4 oxidation. As discussed in Sect. 4.2, ∼25% of the Cl atoms generated under these conditions will react with CH4. The CH4 loss due to Cl during one hour at noon is ∼40 pptv. This can be compared to the CH4 loss in the same period owing to reaction with OH, which is ∼400 pptv, assuming OH concentrations of 1 × 107 molecules cm−3 using 1.9 ppmv CH4. While these estimations highlight the potential of Cl compared to OH regarding CH4 oxidation, rigorous assessment of the impact of chlorine chemistry on a long-lived climate gas such as CH4 requires implementing appropriate chemistry into global chemistry-transport models, which is beyond the scope of this manuscript.
6 Photolysis of HOCl as source of OH
As shown in reaction (R12), the photolysis of HOCl generates both a Cl atom and an OH radical. In the following, we compare the rates of formation of OH through the diel cycle airing from HOCl photolysis with the rate of OH formation from the photolysis of O3 in the presence of water-vapor (R19), traditionally considered the major primary source of atmospheric OH.| O3 + hv → O(1D) + O2 | (R19a) |
| O(1D) + H2O → 2OH | (R19b) |
The primary photochemical production rate of OH from O3, PO3(OH) was calculated from the O3 photolysis frequency JO(1D) modified using the rate coefficients for the O(1D) reaction with H2O or quenching by N2 and O2 in eqn (4). Rate coefficients k were taken from the IUPAC evaluation.34
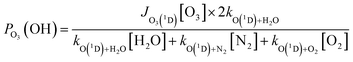 | (4) |
The diurnal cycle of the Cl and OH production rates during the “marine-anthropogenic” and “continental-unpolluted” periods (Fig. 7) show that for both periods the OH production from O3 reaches a maximum of ∼7 × 106 molecules cm−3 s−1 around 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00. For comparison, the maximum Cl (and thus OH from photolysis of HOCl) production rate is a factor of seven lower for the “marine-anthropogenic” case (Fig. 7a) and a factor 40 smaller for the “continental-unpolluted” case (Fig. 7b). However, in the early morning, in marine-anthropogenically influenced air the Cl production rate is more rapid than that of OH, which results from the red-shifted absorption spectra of Cl2, HOCl and ClNO2 compared to O3 and thus better overlap with the early morning actinic flux (Fig. 7a). This is entirely consistent with the findings of Phillips et al.13 at a measurement site 40 km away from our site when marine-influenced air was encountered. In this early morning period, the generation of Cl atoms results in the oxidation of hydrocarbons and thus increases the rate of formation of organic peroxyl radicals that play an important role in daytime, photochemical ozone formation.
00. For comparison, the maximum Cl (and thus OH from photolysis of HOCl) production rate is a factor of seven lower for the “marine-anthropogenic” case (Fig. 7a) and a factor 40 smaller for the “continental-unpolluted” case (Fig. 7b). However, in the early morning, in marine-anthropogenically influenced air the Cl production rate is more rapid than that of OH, which results from the red-shifted absorption spectra of Cl2, HOCl and ClNO2 compared to O3 and thus better overlap with the early morning actinic flux (Fig. 7a). This is entirely consistent with the findings of Phillips et al.13 at a measurement site 40 km away from our site when marine-influenced air was encountered. In this early morning period, the generation of Cl atoms results in the oxidation of hydrocarbons and thus increases the rate of formation of organic peroxyl radicals that play an important role in daytime, photochemical ozone formation.
Tropospheric OH formation occurs via primary formation from O3 photolysis as well as by recycling through reaction of HO2 with NO and (generally less importantly) the photolysis of peroxides and HONO.101,102 HOCl is generally not considered as a potential contributor to OH production due to a lack of HOCl data. However, Fig. 9a shows that while OH formation from photolysis of O3 is dominant, OH formation from HOCl (compare eqn (3)) is still significant at some times of the day when the actinic flux at the shorter wavelengths needed to photolyze O3 is relatively weak (see Fig. 9b). For marine-anthropogenically influenced air masses, the contribution of HOCl photolysis to OH formation approaches 40% at sunrise and sunset, compared to 8% at noon. For the “continental-unpolluted” air masses, the contribution of HOCl to OH ranges from 2% at noon to 17% at sunrise and sunset. This analysis shows that HOCl should be considered as a source of primary OH formation, especially during the morning and the evening hours.
7 Conclusions
Several inorganic chlorine species (Cl2, HOCl, ClNO2 and ClONO2) were measured with an Iodide-CIMS at a continental measurement site in rural central Germany, ∼400 km from the nearest coastline. The largest mixing ratios were detected when 48 h-backward trajectories indicated that the air mass had passed over marine and polluted regions. The dominant chlorine-containing trace gases were ClNO2 during the night and early-morning and HOCl throughout the day. A consistent feature of the ClNO2 diel profile was an early morning peak, which was associated with vertical mixing. In unpolluted air, the levels of HOCl and Cl2 were strongly enhanced compared to ClNO2, while ClONO2 was not observed.In air masses with marine-anthropogenic influence we observed not only ClNO2 but also ClONO2 along with enhanced particulate chlorine during the nighttime. ClONO2 was thus observed for the first time in tropospheric air masses, albeit at low mixing ratios of 2–3 pptv. Laboratory experiments helped rule out that this observation was not an artefact resulting, e.g., from reaction of ClNO2 with IOX− in the IMR of the instrument. Calculations of the total chlorine production rate from the photolysis of the observed species reveal values of 1.0 × 106 atoms cm−3 s−1 for air masses with marine polluted source regions, and 1.6 × 105 atoms cm−3 s−1 for continental clean ones. Depending on the air mass source region, ClNO2 contributed 30–90% to Cl atom production up to three hours after sunrise and 5–10% throughout the rest of the day. Additionally, throughout the day, HOCl contributed 10–85% to Cl production, while Cl2 was responsible for 5–20%.
Cl formation contributed significantly to radical generation (and thus oxidation rates) in the early morning implying significant peroxyl radical and ClO formation rates. The formation of OH radicals from photolysis of HOCl was compared to that by photolysis of O3 and shown to make a significant contribution for air masses that had passed over maritime and polluted regions before reaching the site with a maximum contribution of 40% of the total primary OH formation at sunrise and sunset.
Consequentially, we show that N2O5-induced activation of chlorine from its particle reservoir into photolabile, gas phase forms has repercussions for the lifetimes of many hydrocarbons, highlighting methane in remote continental areas distant to source regions where it affects local chemical processes.
Author contributions
L. M. performed CIMS measurements, trajectory calculation, analyzed the data and drafted the manuscript. F. F. and F. D. performed ground-based measurements with the MoLa and data post-processing. J. N. C. performed CRDS, TD-CRDS and FT-IR measurements, synthesized the ClONO2 and helped with calibration. All authors discussed the data and the presented results. All co-authors commented on the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: discussions regarding secondary ion chemistry and supporting figures. See DOI: https://doi.org/10.1039/d5ea00142k.Data for this article, including data from all figures are available at Zenodo at https://doi.org/10.5281/zenodo.18670872.
Acknowledgements
This work was supported by internal funding from the Max Planck Society. L. M. is funded by the Deutsche Forschungs-gemeinschaft (DFG, German Research Foundation)–TRR 301 “TPChange” (Project-ID 428312742). The authors thank Thomas Böttger and Philipp Schuhmann for support during the BISTUM24 campaign, and Laura Wüst for preparation of N2O5 crystals (all Max-Planck-Institute for Chemistry). Additionally, the authors thank Yijing Chen (Tsinghua University), Lewis Marden and Jake Willow (both from University of York), Cheng Wu (University of Gothenburg) and Cora Young (York University) for discussions about secondary ion chemistry. Open Access funding provided by the Max Planck Society.References
- M. J. Molina and F. S. Rowland, Nature, 1974, 249, 810–812 CrossRef CAS.
- F. S. Rowland, Environ. Conserv., 1988, 15, 101–115 CrossRef CAS.
- S. Solomon, Rev. Geophys., 1999, 37, 275–316 CrossRef CAS.
- J. W. Elkins, T. M. Thompson, T. H. Swanson, J. H. Butler, B. D. Hall, S. O. Cummings, D. A. Fishers and A. G. Raffo, Nature, 1993, 364, 780–783 CrossRef CAS.
- A. Engel, U. Schmidt and D. McKenna, Geophys. Res. Lett., 1998, 25, 3319–3322 Search PubMed.
- R. G. Prinn, R. F. Weiss, P. J. Fraser, P. G. Simmonds, D. M. Cunnold, F. N. Alyea, S. O'Doherty, P. Salameh, B. R. Miller, J. Huang, R. H. J. Wang, D. E. Hartley, C. Harth, L. P. Steele, G. Sturrock, P. M. Midgley and A. McCulloch, J. Geophys. Res. Atmos., 2000, 105, 17751–17792 Search PubMed.
- A. Engel, M. Strunk, M. Müller, H.-P. Haase, C. Poss, I. Levin and U. Schmidt, J. Geophys. Res. Atmos., 2002, 25(17), 3319–3322 Search PubMed.
- M. K. W. Ko and N. D. Sze, J. Geophys. Res. Atmos., 1984, 89, 11619–11632 CrossRef CAS.
- T. Von Clarmann and S. Johansson, Atmos. Chem. Phys., 2018, 18, 15363–15386 CrossRef.
- J. A. Thornton, J. P. Kercher, T. P. Riedel, N. L. Wagner, J. Cozic, J. S. Holloway, W. P. Dubé, G. M. Wolfe, P. K. Quinn, A. M. Middlebrook, B. Alexander and S. S. Brown, Nature, 2010, 464, 271–274 Search PubMed.
- M. J. Lawler, R. Sander, L. J. Carpenter, J. D. Lee, R. Von Glasow, R. Sommariva and E. S. Saltzman, Atmos. Chem. Phys., 2011, 11, 7617–7628 CrossRef CAS.
- L. H. Mielke, A. Furgeson and H. D. Osthoff, Environ. Sci. Technol., 2011, 45, 8889–8896 CrossRef CAS PubMed.
- G. J. Phillips, M. J. Tang, J. Thieser, B. Brickwedde, G. Schuster, B. Bohn, J. Lelieveld and J. N. Crowley, Geophys. Res. Lett., 2012, 39, L10811 Search PubMed.
- T. J. Bannan, A. M. Booth, A. Bacak, J. B. A. Muller, K. E. Leather, M. Le Breton, B. Jones, D. Young, H. Coe, J. Allan, S. Visser, J. G. Slowik, M. Furger, A. S. H. Prévôt, J. Lee, R. E. Dunmore, J. R. Hopkins, J. F. Hamilton, A. C. Lewis, L. K. Whalley, T. Sharp, D. Stone, D. E. Heard, Z. L. Fleming, R. Leigh, D. E. Shallcross and C. J. Percival, J. Geophys. Res. Atmos., 2015, 120, 5638–5657 Search PubMed.
- T. P. Marcy, R. S. Gao, M. J. Northway, P. J. Popp, H. Stark and D. W. Fahey, Int. J. Mass Spectrom., 2005, 243, 63–70 CrossRef CAS.
- M. Von Hobe, J. U. Grooß, R. Müller, S. Hrechanyy, U. Winkler and F. Stroh, Atmos. Chem. Phys., 2005, 5, 693–702 CrossRef CAS.
- R. Atkinson, D. L. Baulch, R. A. Cox, J. N. Crowley, R. F. Hampson, R. G. Hynes, M. E. Jenkin, M. J. Rossi and J. Troe, Atmos. Chem. Phys., 2007, 7, 981–1191 CrossRef CAS.
- R. Sander and P. J. Crutzen, J. Geophys. Res. Atmos., 1996, 101, 9121–9138 CrossRef CAS.
- R. von Glasow and P. J. Crutzen, in Treatise on Geochemistry, ed. H. D. Holland and K. K. Turekian, Pergamon, Oxford, 2003, pp. 1–67, DOI:10.1016/B0-08-043751-6/04141-4.
- C. Arsene, A. Bougiatioti, M. Kanakidou, B. Bonsang and N. Mihalopoulos, Atmos. Chem. Phys., 2007, 7, 4661–4673 CrossRef CAS.
- R. Von Glasow, R. Sander, A. Bott and P. J. Crutzen, J. Geophys. Res. Atmos., 2002, 107(D17), ACH 9 Search PubMed.
- C. Faxon, J. Bean and L. Ruiz, Atmosphere, 2015, 6, 1487–1506 CrossRef CAS.
- N. Bhattacharyya, M. Modi, L. G. Jahn and L. Hildebrandt Ruiz, Environ. Sci.: Atmos., 2023, 3, 1174–1185 CAS.
- Q. Li, D. Meidan, P. Hess, J. A. Añel, C. A. Cuevas, S. Doney, R. P. Fernandez, M. van Herpen, L. Höglund-Isaksson, M. S. Johnson, D. E. Kinnison, J.-F. Lamarque, T. Röckmann, N. M. Mahowald, A. Saiz-Lopez, Q. Li, D. Meidan, P. Hess, J. A. Añel, C. A. Cuevas, S. Doney, R. P. Fernandez, M. van Herpen, L. Höglund-Isaksson, M. S. Johnson, D. E. Kinnison, J.-F. Lamarque, T. Röckmann, N. M. Mahowald and A. Saiz-Lopez, Nat. Commun., 2023, 14(1), 4045 CrossRef CAS PubMed.
- X. Wang, D. J. Jacob, S. D. Eastham, M. P. Sulprizio, L. Zhu, Q. Chen, B. Alexander, T. Sherwen, M. J. Evans, B. H. Lee, J. D. Haskins, F. D. Lopez-Hilfiker, J. A. Thornton, G. L. Huey and H. Liao, Atmos. Chem. Phys., 2019, 19, 3981–4003 CrossRef CAS.
- W. C. Keene, A. A. P. Pszenny, D. J. Jacob, R. A. Duce, J. N. Galloway, J. J. Schultz-Tokos, H. Sievering and J. F. Boatman, Global Biogeochem. Cycles, 1990, 4, 407–430 CrossRef CAS.
- W. C. Keene, R. Sander, A. A. P. Pszenny, R. Vogt, P. J. Crutzen and J. N. Galloway, J. Aerosol Sci., 1998, 29, 339–356 CrossRef CAS.
- C. Volpe, M. Wahlen, A. A. P. Pszenny and A. J. Spivack, Geophys. Res. Lett., 1998, 25, 3831–3834 CrossRef.
- T. A. Cahill, K. Wilkinson and R. Schnell, J. Geophys. Res. Atmos., 1992, 97(D13), 14513–14520 CrossRef.
- K. W. Oum, M. J. Lakin, D. O. DeHaan, T. Brauers and B. J. Finlayson-Pitts, Science, 1998, 279 Search PubMed.
- J. M. Lobert, W. C. Keene, J. A. Logan and R. Yevich, J. Geophys. Res. Atmos., 1999, 104, 8373–8389 Search PubMed.
- T. J. Bannan, M. A. H. Khan, M. Le Breton, M. Priestley, S. D. Worrall, A. Bacak, N. A. Marsden, D. Lowe, J. Pitt, G. Allen, D. Topping, H. Coe, G. McFiggans, D. E. Shallcross and C. J. Percival, Geophys. Res. Lett., 2019, 46, 8508–8516 Search PubMed.
- C. G. Masoud, M. Modi, N. Bhattacharyya, L. G. Jahn, K. N. McPherson, P. Abue, K. Patel, D. T. Allen and L. Hildebrandt Ruiz, Environ. Sci. Technol., 2023, 57, 15454–15464 CrossRef CAS PubMed.
- M. Ammann, R. A. Cox, J. N. Crowley, H. Herrmann, M. E. Jenkin, V. F. McNeill, A. Mellouki, M. J. Rossi, J. Troe and T. J. Wallington, Evaluated Kinetic and Photochemical Data Foratmospheric Chemistry, 2025, https://iupac.aeris-data.fr/.
- S. S. Brown and J. Stutz, Chem. Soc. Rev., 2012, 41, 6405 Search PubMed.
- J. Liebmann, E. Karu, N. Sobanski, J. Schuladen, M. Ehn, S. Schallhart, L. Quéléver, H. Hellen, H. Hakola, T. Hoffmann, J. Williams, H. Fischer, J. Lelieveld and J. N. Crowley, Atmos. Chem. Phys., 2018, 18, 3799–3815 CrossRef CAS.
- P. Dewald, T. Seubert, S. T. Andersen, G. N. T. E. Türk, J. Schuladen, M. R. McGillen, C. Denjean, J.-C. Etienne, O. Garrouste, M. Jamar, S. Harb, M. Cirtog, V. Michoud, M. Cazaunau, A. Bergé, C. Cantrell, S. Dusanter, B. Picquet-Varrault, A. Kukui, C. Xue, A. Mellouki, J. Lelieveld and J. N. Crowley, Atmos. Chem. Phys., 2024, 24, 8983–8997 CrossRef CAS.
- X. Wang, H. Wang, L. Xue, T. Wang, L. Wang, R. Gu, W. Wang, Y. J. Tham, Z. Wang, L. Yang, J. Chen and W. Wang, Atmos. Environ., 2017, 156, 125–134 CrossRef CAS.
- M. Priestley, M. Le Breton, T. J. Bannan, S. D. Worrall, A. Bacak, A. R. D. Smedley, E. Reyes-Villegas, A. Mehra, J. Allan, A. R. Webb, D. E. Shallcross, H. Coe and C. J. Percival, Atmos. Chem. Phys., 2018, 18, 13481–13493 CrossRef CAS.
- J. D. Haskins, B. H. Lee, F. D. Lopez-Hilifiker, Q. Peng, L. Jaeglé, J. M. Reeves, J. C. Schroder, P. Campuzano-Jost, D. Fibiger, E. E. McDuffie, J. L. Jiménez, S. S. Brown and J. A. Thornton, J. Geophys. Res. Atmos., 2019, 124, 8851–8869 CrossRef CAS.
- G. Chen, X. Fan, H. Wang, Y. J. Tham, Z. Lin, X. Ji, L. Xu, B. Hu and J. Chen, Atmos. Chem. Phys., 2025, 25, 7815–7828 CrossRef CAS.
- F. Schweitzer, P. Mirabel and C. George, J. Phys. Chem. A, 1998, 102, 3942–3952 CrossRef CAS.
- B. J. Finlayson-Pitts, M. J. Ezell and J. N. Pitts, Nature, 1989, 337, 241–244 CrossRef CAS.
- M.-T. Leu, R. S. Timonen, L. F. Keyser and Y. L. Yung, J. Phys. Chem., 1995, 99, 13203–13212 CrossRef CAS.
- G. J. Phillips, J. Thieser, M. Tang, N. Sobanski, G. Schuster, J. Fachinger, F. Drewnick, S. Borrmann, H. Bingemer, J. Lelieveld and J. N. Crowley, Atmos. Chem. Phys., 2016, 16, 13231–13249 CrossRef CAS.
- J. A. Ganske, M. J. Ezell, H. N. Berko and B. J. Finlayson-Pitts, Chem. Phys. Lett., 1991, 179, 204–210 CrossRef CAS.
- X. Peng, T. Wang, W. Wang, A. R. Ravishankara, C. George, M. Xia, M. Cai, Q. Li, C. M. Salvador, C. Lau, X. Lyu, C. N. Poon, A. Mellouki, Y. Mu, M. Hallquist, A. Saiz-Lopez, H. Guo, H. Herrmann, C. Yu, J. Dai, Y. Wang, X. Wang, A. Yu, K. Leung, S. Lee and J. Chen, Nat. Commun., 2022, 13, 939 Search PubMed.
- G. Chen, X. Fan, S. Yu, Y. J. Tham, Z. Lin, X. Ji, L. Xu and J. Chen, Environ. Sci. Technol., 2025, 59(10), 5164–5171 Search PubMed.
- M. Eigen and K. Kustin, J. Am. Chem. Soc., 1962, 84, 1355–1361 Search PubMed.
- G. Deiber, C. George, S. Le Calvé, F. Schweitzer and P. Mirabel, Atmos. Chem. Phys., 2004, 4, 1291–1299 Search PubMed.
- M. A. Tolbert, M. J. Rossi, R. Malhotra and D. M. Golden, Science, 1987, 238, 1258–1260 Search PubMed.
- R. J. Cicerone, Rev. Geophys., 1981, 19, 123–139 Search PubMed.
- T. E. Caldwell, K. L. Foster, T. Benter, S. Langer, J. C. Hemminger and B. J. Finlayson-Pitts, J. Phys. Chem. A, 1999, 103, 8231–8238 Search PubMed.
- J. P. Kercher, T. P. Riedel and J. A. Thornton, Atmos. Meas. Tech., 2009, 2, 193–204 Search PubMed.
- B. H. Lee, F. D. Lopez-Hilfiker, C. Mohr, T. Kurtén, D. R. Worsnop and J. A. Thornton, Environ. Sci. Technol., 2014, 48, 6309–6317 Search PubMed.
- D. Maric, J. P. Burrows, R. Meller and G. K. Moortgat, J. Photochem. Photobiol., A, 1993, 70, 205–214 CrossRef CAS.
- J. A. Davidson, C. A. Cantrell, R. E. Shetter, A. H. McDaniel and J. G. Calvert, J. Geophys. Res. Atmos., 1987, 92, 10921–10925 CrossRef.
- N. Sobanski, J. Schuladen, G. Schuster, J. Lelieveld and J. N. Crowley, Atmos. Meas. Tech., 2016, 9, 5103–5118 Search PubMed.
- Y. Ji, L. G. Huey, D. J. Tanner, Y. R. Lee, P. R. Veres, J. A. Neuman, Y. Wang and X. Wang, Atmos. Meas. Tech., 2020, 13, 3683–3696 Search PubMed.
- J. Thieser, G. Schuster, J. Schuladen, G. J. Phillips, A. Reiffs, U. Parchatka, D. Pöhler, J. Lelieveld and J. N. Crowley, Atmos. Meas. Tech., 2016, 9, 553–576 Search PubMed.
- J. A. Davidson, A. A. Viggiano, C. J. Howard, I. Dotan, F. C. Fehsenfeld, D. L. Albritton and E. E. Ferguson, J. Chem. Phys., 1978, 68, 2085–2087 Search PubMed.
- J. M. Roberts, H. D. Osthoff, S. S. Brown, A. R. Ravishankara, D. Coffman, P. Quinn and T. Bates, Geophys. Res. Lett., 2009, 36, L20808 Search PubMed.
- F. Drewnick, T. Böttger, S. L. von der Weiden-Reinmüller, S. R. Zorn, T. Klimach, J. Schneider and S. Borrmann, Atmos. Meas. Tech., 2012, 5, 1443–1457 Search PubMed.
- J. Pikmann, F. Drewnick, F. Fachinger and S. Borrmann, Atmos. Chem. Phys., 2024, 24, 12295–12321 CrossRef CAS.
- G. C. Roberts, M. V. Ramana, C. Corrigan, D. Kim and V. Ramanathan, Proc. Natl. Acad. Sci. U. S. A., 2008, 105, 7370–7375 CrossRef CAS PubMed.
- A. F. Stein, R. R. Draxler, G. D. Rolph, B. J. B. Stunder, M. D. Cohen and F. Ngan, Bull. Am. Meteorol. Soc., 2015, 96, 2059–2077 CrossRef.
- G. Rolph, A. Stein and B. Stunder, Environ. Model. Software, 2017, 95, 210–228 CrossRef.
- Y. J. Tham, C. Yan, L. Xue, Q. Zha, X. Wang and T. Wang, Chin. Sci. Bull., 2014, 59, 356–359 CrossRef CAS.
- T. P. Riedel, G. M. Wolfe, K. T. Danas, J. B. Gilman, W. C. Kuster, D. M. Bon, A. Vlasenko, S. M. Li, E. J. Williams, B. M. Lerner, P. R. Veres, J. M. Roberts, J. S. Holloway, B. Lefer, S. S. Brown and J. A. Thornton, Atmos. Chem. Phys., 2014, 14, 3789–3800 CrossRef.
- B. D. Finley and E. S. Saltzman, Geophys. Res. Lett., 2006, 33, L11809 CrossRef.
- A. A. P. Pszenny, W. C. Keene, D. J. Jacob, S. Fan, J. R. Maben, M. P. Zetwo, M. Springer-Young and J. N. Galloway, Geophys. Res. Lett., 1993, 20, 699–702 CrossRef CAS.
- C. W. Spicer, E. G. Chapman, B. J. Finlayson-Pitts, R. A. Plastridge, J. M. Hubbe, J. D. Fast and C. M. Berkowitz, Nature, 1998, 394, 353–356 CrossRef CAS.
- X. Chen, Y. Jiang, Z. Zong, Y. Wang, W. Sun, Y. Wang, M. Xia, L. Guan, P. Liu, C. Zhang, J. Chen, Y. Mu and T. Wang, Environ. Sci. Technol., 2025, 59, 12775–12785 CrossRef CAS PubMed.
- H. D. Osthoff, J. M. Roberts, A. R. Ravishankara, E. J. Williams, B. M. Lerner, R. Sommariva, T. S. Bates, D. Coffman, P. K. Quinn, J. E. Dibb, H. Stark, J. B. Burkholder, R. K. Talukdar, J. Meagher, F. C. Fehsenfeld and S. S. Brown, Nat. Geosci., 2008, 1, 324–328 CrossRef CAS.
- M. Le Breton, Å. M. Hallquist, R. K. Pathak, D. Simpson, Y. Wang, J. Johansson, J. Zheng, Y. Yang, D. Shang, H. Wang, Q. Liu, C. Chan, T. Wang, T. J. Bannan, M. Priestley, C. J. Percival, D. E. Shallcross, K. Lu, S. Guo, M. Hu and M. Hallquist, Atmos. Chem. Phys., 2018, 18, 13013–13030 CrossRef CAS.
- J. Zhang and D. Kinnison, TUV Calculator, NCAR (National Center for Atmospheric Research), Boulder, Colorado, 2025, https://www.acom.ucar.edu/Models/TUV/Interactive_TUV/, accessed 15.06.2025.
- T. H. Bertram and J. A. Thornton, Atmos. Chem. Phys., 2009, 9, 8351–8363 CrossRef CAS.
- S. R. Zorn, F. Drewnick, M. Schott, T. Hoffmann and S. Borrmann, Atmos. Chem. Phys., 2008, 8, 4711–4728 CrossRef CAS.
- L. Moormann, F. Fachinger, F. Drewnick and H. Tost, EGUsphere, 2025, pp. 1–23, DOI:10.5194/egusphere-2025-3862.
- J. N. Crowley, G. Schuster, N. Pouvesle, U. Parchatka, H. Fischer, B. Bonn, H. Bingemer and J. Lelieveld, Atmos. Chem. Phys., 2010, 10, 2795–2812 CrossRef CAS.
- Y. Ge, S. Solberg, M. R. Heal, S. Reimann, W. Van Caspel, B. Hellack, T. Salameh and D. Simpson, Atmos. Chem. Phys., 2024, 24, 7699–7729 CrossRef CAS.
- M. Ragains and B. Finlayson-Pitts, J. Phys. Chem. A, 1997, 101, 1509–1517 CrossRef CAS.
- J. Burkholder, S. Sander, J. Abbatt, J. Barker, C. Cappa, J. Crounse, T. Dibble, R. Huie, C. Kolb and M. Kurylo, Chemical Kinetics and Photochemical Data for Use in Atmospheric Studies; Evaluation Number 19, Jet Propulsion Laboratory, National Aeronautics and Space, Pasadena, CA, 2020 Search PubMed.
- J. D. Haskins, L. Jaeglé, V. Shah, B. H. Lee, F. D. Lopez-Hilfiker, P. Campuzano-Jost, J. C. Schroder, D. A. Day, H. Guo, A. P. Sullivan, R. Weber, J. Dibb, T. Campos, J. L. Jimenez, S. S. Brown and J. A. Thornton, J. Geophys. Res. Atmos., 2018, 123, 12897–12916 CAS.
- A. A. Angelucci, T. C. Furlani, X. Wang, D. J. Jacob, T. C. Vandenboer and C. J. Young, ACS Earth Space Chem., 2021, 5, 2507–2516 CrossRef CAS.
- R. Atkinson, S. M. Aschmann, J. Arey and B. Shorees, J. Geophys. Res. Atmos., 1992, 97, 6065–6073 CrossRef CAS.
- R. A. Cox, M. Ammann, J. N. Crowley, H. Herrmann, M. E. Jenkin, V. F. McNeill, A. Mellouki, J. Troe and T. J. Wallington, Atmos. Chem. Phys., 2020, 20, 13497–13519 CrossRef CAS.
- I. Bey, B. Aumont and G. Toupance, Geophys. Res. Lett., 1997, 24, 1067–1070 CrossRef CAS.
- I. Faloona, D. Tan, W. Brune, J. Hurst, D. Barket, T. L. Couch, P. Shepson, E. Apel, D. Riemer, T. Thornberry, M. A. Carroll, S. Sillman, G. J. Keeler, J. Sagady, D. Hooper and K. Paterson, J. Geophys. Res. Atmos., 2001, 106, 24315–24333 CrossRef CAS.
- X. Ren, H. Harder, M. Martinez, R. L. Lesher, A. Oliger, J. B. Simpas, W. H. Brune, J. J. Schwab, K. L. Demerjian, Y. He, X. Zhou and H. Gao, Atmos. Environ., 2003, 37, 3639–3651 CrossRef CAS.
- M. A. H. Khan, M. J. Ashfold, G. Nickless, D. Martin, L. A. Watson, P. D. Hamer, R. P. Wayne, C. E. Canosa-Mas and D. E. Shallcross, Atmos. Sci. Lett., 2008, 9, 140–146 CrossRef.
- R. L. Caravan, M. F. Vansco and M. I. Lester, Commun. Chem., 2021, 4, 44 CrossRef CAS PubMed.
- H. Wang and Y.-P. Lee, J. Phys. Chem. A, 2024, 128, 8690–8698 CrossRef CAS PubMed.
- T. H. Bertram, J. A. Thornton, T. P. Riedel, A. M. Middlebrook, R. Bahreini, T. S. Bates, P. K. Quinn and D. J. Coffman, Geophys. Res. Lett., 2009, 36, L19803 CrossRef.
- M. Xia, X. Peng, W. Wang, C. Yu, P. Sun, Y. Li, Y. Liu, Z. Xu, Z. Wang, Z. Xu, W. Nie, A. Ding and T. Wang, Atmos. Chem. Phys., 2020, 20, 6147–6158 CrossRef CAS.
- Y. Li, W. Nie, Y. Liu, D. Huang, Z. Xu, X. Peng, C. George, C. Yan, Y. J. Tham, C. Yu, M. Xia, X. Fu, X. Wang, L. Xue, Z. Wang, Z. Xu, X. Chi, T. Wang and A. Ding, Environ. Sci. Technol. Lett., 2020, 7, 70–75 CrossRef CAS.
- E. Z. Dalton, E. H. Hoffmann, T. Schaefer, A. Tilgner, H. Herrmann and J. D. Raff, J. Am. Chem. Soc., 2023, 145, 15652–15657 CrossRef CAS PubMed.
- G. Chen, L. Xu, S. Yu, L. Xue, Z. Lin, C. Yang, X. Ji, X. Fan, Y. J. Tham, H. Wang, Y. Hong, M. Li, J. H. Seinfeld and J. Chen, Environ. Sci. Technol., 2024, 58, 22714–22721 CrossRef CAS PubMed.
- J. Dai, T. Wang, H. Shen, M. Xia, W. Sun and G. P. Brasseur, Environ. Sci. Technol., 2025, 59, 2169–2180 CrossRef CAS PubMed.
- T. P. Riedel, T. H. Bertram, T. A. Crisp, E. J. Williams, B. M. Lerner, A. Vlasenko, S.-M. Li, J. Gilman, J. de Gouw, D. M. Bon, N. L. Wagner, S. S. Brown and J. A. Thornton, Environ. Sci. Technol., 2012, 46, 10463–10470 CrossRef CAS PubMed.
- J. Lelieveld, F. J. Dentener, W. Peters and M. C. Krol, Atmos. Chem. Phys., 2004, 4, 2337–2344 CrossRef CAS.
- G. Chua, V. Naik and L. W. Horowitz, Atmos. Chem. Phys., 2023, 23, 4955–4975 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |