 Open Access Article
Open Access ArticleEmissions of volatile organic compounds from industrial sources and their effects on ozone formation in Wuhan, China
Yuhan
Xiang
a,
Tianqi
Zhang
a,
Yanfen
Hao
 *a,
Beirou
Yang
a,
Tiantian
Han
a,
Jing
Xu
c,
Wenjuan
Li
a,
Pu
Wang
ab and
Yong
Liang
*a,
Beirou
Yang
a,
Tiantian
Han
a,
Jing
Xu
c,
Wenjuan
Li
a,
Pu
Wang
ab and
Yong
Liang
 a
a
aHubei Key Laboratory of Environmental and Health Effects of Persistent Toxic Substances, School of Environment and Health, Jianghan University, Wuhan, 430056, China. E-mail: Hao_YF@jhun.edu.cn
bHubei Key Laboratory of Industrial Fume & Dust Pollution Control, Jianghan University, Wuhan, 430056, China
cAdvanced Interdisciplinary Institute of Environment and Ecology, Beijing Normal University, Zhuha, 519087, China
First published on 26th November 2025
Abstract
Emissions of typical forty-eight volatile organic compounds (VOCs) were investigated inside sixteen industries, including the vehicle-related industries and commodity manufacturing industries located in Wuhan, China. The total concentrations of the twenty-seven detectable VOCs ranged from 0.4 to 99.8 µg m−3, with vehicle-related industries (1.3–99.8 µg m−3) emitting more VOCs than the commodity manufacturing industry (0.4–20.0 µg m−3). Data comparison between organized and fugitive emission groups revealed that the VOCs emitted from organized vehicle-related industries implementing the zeolite rotary concentrator system coupled with the regenerative thermal oxidizer catalytic combustion technology (0.04–31.5 µg m−3) could significantly meet the national emission standards (500–40000 µg m−3). This technology was proven to have a remarkable effect on reducing benzene-based VOCs, which are important precursors of ozone (O3). A significant decreasing trend of O3 levels was observed with the increase in the distance around these vehicle-related industries based on the data from the monitoring and control platform of small air quality monitoring stations of Wuhan. The total O3 formation potential (OFP) values calculated based on these twenty-seven VOCs in the vehicle-related industries were 5.3–646.8 µg m−3, in which aromatic hydrocarbons were the most reactive analogues, with a cumulative share of 93.0% of the O3 formation potential. 1,3,5-Trimethylbenzene and o-xylene, which together account for 52.3% of the total OFP values, should be identified as the priority pollutants to be controlled in the vehicle-related industry. This study emphasizes the contribution of VOCs emitted from vehicle-related industries to ozone pollution and further provides ideas for prioritizing industrial VOCs for improving regional O3 control strategies.
Environmental significanceThe increasingly severe ozone (O3) pollution poses a serious threat to ecosystems and public health. Volatile organic compounds (VOCs), released from industrial emission sources, are the main precursors for ozone formation, so it is crucial to identify key VOC emission sources and implement effective control measures. This study has precisely observed the concentrations and analogue profiles of forty-eight VOCs emitted from sixteen typical vehicle-related and commodity manufacturing industries in Wuhan, central China. The impact of vehicle-related industries on the regional O3 pollution has been stressed, and O3 formation potential values have been further evaluated to identify the crucial VOC analogues. These findings provide direct scientific evidence for feasibly controlling VOC emissions and O3 pollution to alleviate the ecological hazards. |
1. Introduction
Ozone (O3) is an important trace gas that regulates temperature and climate in the lower atmosphere.1 However, excessive atmospheric O3 can adversely affect human health and vegetation due to its strong oxidizing properties.1,2 Previous studies have assessed the association between long-term O3 exposure and mortality, particularly cardiovascular disease mortality, which caused 17.8 million deaths in 2017, accounting for 31.8% of all-cause deaths globally.2 In addition, excessive atmospheric O3 can trigger plant oxidation, thereby disrupting photosynthesis1,3 and reducing gas exchange within plants, further retarding natural vegetation and crop growth.1,4,5 Therefore, there is an increasing concern about the concentration of O3 in the air. It has been observed that the industrialization of society is one of the main reasons for the increasing number of O3 pollution incidents.5 Volatile organic compounds (VOCs), defined as a class of compounds with high vapor pressure, low water solubility, and photochemical reactivity, have been reported to be the key precursors of O3 in most industrial regions.6 Increasing VOCs emitted in industrial areas might cause excessive O3 in the air,5 and the effect of VOC emission control will directly affect the control and prevention of O3 pollution.The emission sources of atmospheric VOCs have been divided into two categories: natural sources and anthropogenic sources.7 Anthropogenic sources are generally regarded as the main source of atmospheric VOC emissions, with their increasing emissions and high potential in forming O3. Previous studies have shown that in typical industrial cities, the concentration level of VOCs from anthropogenic sources can reach 3.4 times higher than that of natural sources, and industries are the main part of the anthropogenic sources.8 The emission characteristics of these VOCs from anthropogenic sources show obvious industry-specificity due to the significant differences in the production processes, energy structures, and product characteristics of different industries.9 A previous study reported the compositions of VOCs emitted from various industries in Zhengzhou City, China. It was observed that VOCs emitted by packaging and printing enterprises are mainly composed of oxygenated VOCs (OVOCs), of which ethyl acetate and isopropanol together account for more than 93.1%; whereas, aromatic hydrocarbons dominate the VOCs emitted from the automotive, furniture, and paint manufacturing enterprises, accounting for 33.5–90.0%, with m/p-xylene, o-xylene and ethylbenzene being the main constituents. Dominant analogues were halogenated hydrocarbons (52.3%), alkanes (28.8%), and aromatic hydrocarbons (28.5%) in food processing enterprises, rubber enterprises, and graphite and carbon enterprises, respectively.10 Given the different O3 formation potentials (OFP) of various VOC analogues, the diverse analogue profiles of VOCs in different industries might have different contributions to O3 pollution. This indicates that conducting precise research on VOC emissions in typical industries and further evaluating their OPF is of vital importance.
Previous research on VOCs mainly focused on the emission inventory and treatment methods of VOCs.11–13 Studies on the impact of industrial source VOC emissions on atmospheric O3 pollution mainly concentrated on simulation analysis based on large-scale environmental monitoring data.14 There were a few domestic studies on the emission profiles of industrial VOCs in specific cities, including Beijing,15 Shanghai,16 Nanjing,16 Chengdu,17 Zhengzhou,10 Hangzhou,18 Yichang19 and Chongqing.20 Given the significant differences in the pillar industries in different cities, there is limited data on the VOC emission profiles in Wuhan, which is one of the typical industrial cities in China.21 This could hinder the evaluation of VOC emissions in China, which would further have a negative impact on the formulation of legislation controlling regional O3 pollution.18 Therefore, this study aims to investigate the emission patterns and potential O3 formation of VOCs in typical industries in Wuhan. Wuhan is located in the central part of China, where O3 pollution might cause adverse effects on a large scale. Sixteen typical VOC-emitting enterprises were selected in the key industrial area of Wuhan, including vehicle-related industries, as well as commodity manufacturing industries. VOC emission data were collected by measurement inside these enterprises, emission profiles between organized and fugitive emission groups were observed, and OFP values were further calculated to evaluate the impacts of these enterprises on O3 pollution. The objectives of this study were as follows: (1) analyze the concentrations and analogue compositions of VOCs emitted from typical industries in Wuhan; (2) identify the effects of VOC treatment equipment in organized emission groups; (3) reveal the impacts of various industries on the regional O3 pollution and specify the specific VOC analogues that need to be strictly controlled. Therefore, this study is of great significance for controlling VOC emissions and improving O3 pollution in megacities.
2. Materials and methods
2.1. Sample collection
The industrial development of Hubei Province has been rapid,22 with the industrial growth increasing by 14.8% in 2021. This growth rate is ranked first in central China and second overall in China.23 The sixteen selected industries in this study were located in a typical industrial region in Wuhan, Hubei Province, including nine vehicle-related industries and seven commodity manufacturing industries. Table S1 shows the basic information from the nine vehicle-related and seven commodity manufacturing industries. The sampling campaign was carried out under the HJ 644-2013 Chinese standard method during the period of July 19-21, 2021. The detailed information for the 16 sampling points is shown in Table S2. In brief, each adsorption tube was aged at 350 °C and 40 mL min−1 for 15 minutes to remove interfering substances before sampling. A TH-110E air sampler (Tianhong Inc, Wuhan, China) was used to collect atmospheric VOCs inside the industries, and the sampling rate and duration were set at 100 mL min−1 and 20 minutes, respectively. Two parallel samples, as well as one field blank sample, were collected in each sampling site, and forty-eight sampling tubes in total were collected in sixteen industries. After sampling, the adsorption tube was sealed in a cleaned sleeve, wrapped with aluminum foil and transported to the laboratory for instrumental analysis.2.2. Instrumental analysis
There are currently over a thousand known analogues of VOCs,24 which could be classified into four major categories: aromatic hydrocarbons, halogenated aromatic hydrocarbons, haloalkanes, and halogenated olefins. The selection of target analogues in this study comprehensively considered these four major categories, the China national standards for VOCs emissions,25 as well as the local VOC emissions characteristics of Wuhan.26 In addition, the production raw materials, process features, and VOC emission characteristics of these representative industries were also taken into consideration. Therefore, forty-eight VOC analogues were selected as the target substances, which cover all four major categories (aromatic hydrocarbons, halogenated aromatic hydrocarbons, haloalkanes, and halogenated olefins) and the main emitted analogues from these industries. It was supposed that the observation in this study could represent the emission characteristics of VOCs from related industries to some extent. The concentrations of forty-eight typical VOCs in the sampling tubes were measured based on the EPA method 8260D.27 The individual compounds are as follows: trans-1,2-dichloroethene, 1,1-dichloroethene, cis-1,2-dichloroethane, bromochloromethane, chloroform, 1,1,1-trichloroethene, 1,1-dichloropropene, 1,2-dichloroethene, benzene, trichloroethene, 1,2-dichloropropane, methylene bromide, bromodichloromethane, cis-1,3-dichloropropene, toluene, trans-1,3-dichloropropene, 1,1,2-trichloroethane, 1,3-dichloropropane, tetrachloroethene, chlorodibromomethane, 1,2-dibromoethane, chlorobenzene, 1,1,1,2-tetrachloroethane, ethylbenzene, m + p-xylene, o-xylene, styrene, bromoform, isopropylbenzene, 1,1,2,2-tetracholoethane, bromobenzene, 1,2,3-trichloropropane, propylbenzene, 2-chlorotoluene, 1,3,5-trimethylbenzene, tert-butylbenzene, 1,2,4-trimethylbenzene, sec-butylbenzene, 1,3-dichlorobenzene, p-cymene, 1,4-dichlorobenzene, 1,2-dichlorobenzene, butylbenzene, 1,2-dibromo-3-chloropropane, 1,2,4-trichlorobenzene, hexachloro-1, 3-butadiene, naphthalene, and 1,2,3-trichlorobenzene. All these forty-eight compounds are regarded as the common VOCs emitted from industries and have been identified as contributors to the O3 pollution.The internal standard (4-bromofluorobenzene, BFB) was injected into the adsorption tube before the thermal desorption. All target VOCs were then desorbed by a multi-tube thermal desorber (TD100, Makers International, UK) and analyzed by the gas chromatography-mass spectrometer (TSQ8000Evo, Thermo Fisher Scientific). DB-624 capillary chromatography columns were used for the separation of VOCs (30 m × 0.25 mm i.d. × 1.4 µm film thickness). The detailed temperature programming was provided in the EPA method 8260D.27
2.3. QA & QC
The concentrations of VOCs in the samples were calculated based on the internal standard method. The calibration curves were developed by using five standard samples (5, 10, 25, 50, 100 mg L−1) comprising forty-eight VOCs. One laboratory blank was added after every ten samples. The R2 values of the curve for each VOC were ≥0.99, and the deviation of BFB concentration in both standard and collected samples was less than 6%. The method detection limit (MDL) was defined as the field or laboratory blank sample when the targets were detectable, and was triple the signal-to-noise ratio values when the targets were undetectable. If all VOCs were detected in the blanks and were less than 10% of those in the collected samples, the final measurement results were obtained by subtracting the blank samples from the parallel samples.2.4. Data analysis
The OFP value is an important index for evaluating the contribution of VOC analogues to O3 generation. It is calculated based on the concentrations and maximum incremental reactivity (MIR) of each VOC. The MIR values used for the calculation in this study were obtained in a previous study;28 the calculation is as follows:| OFPi = MIRi × Con.i |
OFPi is the OFP of a specific VOC analogue (i, µg m−3), MIRi is the MIR coefficient of a specific VOC analogue, Con.i is the measured concentration of a specific VOC analogue (µg m−3).
3. Results and discussion
3.1. Concentration and characterization of VOCs
Forty-eight VOCs emitted from operating vehicle-related industries and other commodity manufacturing industries in the typical industrial area of Wuhan were analyzed, in which twenty-seven analogues were detectable. These detectable VOC analogues were classified into four major categories: aromatic hydrocarbons, halogenated aromatic hydrocarbons, haloalkanes and halogenated olefins (Fig. 1). The concentrations of twenty-seven VOCs (CΣ27VOCs) ranged from 0.4 µg m−3 to 100.0 µg m−3. Among them, the CΣ27VOCs observed from the vehicle-related industries (1.3–100.0 µg m−3) are nearly an order of magnitude higher than those from the commodity manufacturing industry (0.4–20.0 µg m−3) (Fig. 1 and Table 1). Within these typical nine vehicle-related industries, the CΣ27VOCs were dominated by aromatic hydrocarbons, with an average concentration of 30.8 µg m−3, accounting for 72.4% of CΣ27VOCs. Haloalkanes were the second largest group, with an average concentration of 6.6 µg m−3 (15.5%), followed by halogenated aromatic hydrocarbons (4.9 µg m−3, 11.4%) and halogenated olefins (0.3 µg m−3, 0.7%). o-Xylene (7.2 µg m−3) was the most predominant analogue emitted from vehicle-related industries, accounting for 18.2% of the CΣ27VOCs, followed by sec-butylbenzene (6.6 µg m−3), 1,3,5-trimethylbenzene (5.1 µg m−3), 1,2,3-trichloropropane (5.1 µg m−3) and 2-chlorotoluene (4.8 µg m−3), which respectively account for 11.7%, 11.1%, 11.0, and 11.64% of CΣ27VOCs. Previous studies have shown that coating and spraying are the crucial processes that release considerable VOCs.22 The solvent-based materials used in these two processes are mainly constituted of C7–C8 aromatic hydrocarbons.11,29 Therefore, the analogue profiles investigated in the vehicle-related industries were mainly attributed to the coating and spraying processes.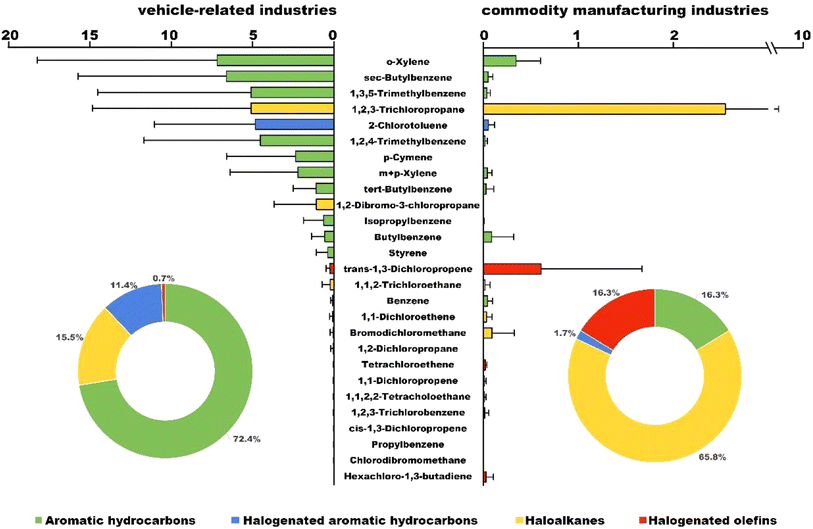 | ||
| Fig. 1 Concentrations (µg m−3) of individual analogues and the proportions (%) of major categories emitted from vehicle-related industries and commodity manufacturing industries. | ||
| Sampling sites | Vehicle-related industries | Commodity manufacturing industries | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DFFS | DFSC | DFYC | SLSC | LY | DFADT | DFEC | GM | VSKT | MXDZ | ZL | JC | BD | CLBL | HB | HJL | |
| Organized (O) or fugitive (F) emissions | O | O | O | O | O | F | F | F | F | O | F | F | F | F | F | F |
| a n.d. = not detected. | ||||||||||||||||
| o-Xylene | 0.6 | 3.7 | 1.6 | 4.7 | 23.2 | 0.3 | 0.7 | 29.4 | 0.5 | 0.2 | 0.8 | 0.1 | 0.2 | 0.2 | 0.31 | 0.6 |
| 1,2,3-Trichloropropane | 0.4 | 0.4 | 19.0 | 25.2 | 0.7 | n.d. | n.d. | 0.2 | 0.03 | 0.3 | 17.5 | n.d. | 0.02 | n.d. | n.d. | 0.01 |
| sec-butylbenzene | 0.5 | 16.5 | 1.9 | 12.3 | 25.0 | 0.04 | 0.2 | 3.0 | 0.2 | 0.1 | 0.03 | 0.02 | n.d. | 0.1 | 0.04 | 0.1 |
| 1,3,5-Trimethylbenzene | 1.9 | 0.4 | 0.03 | 0.1 | 21.2 | 0.1 | 0.1 | 22.2 | 0.1 | 0.01 | 0.1 | n.d. | 0.1 | n.d. | 0.04 | 0.1 |
| 2-Chlorotoluene | 0.5 | 18.4 | 2.7 | 10.8 | 5.5 | 0.2 | 0.2 | 4.9 | 0.4 | n.d. | 0.2 | n.d. | 0.1 | 0.01 | n.d. | 0.1 |
| 1,2,4-Trimethylbenzene | 0.7 | 19.2 | 2.4 | 14.3 | 0.1 | 0.01 | 0.2 | 3.6 | 0.3 | 0.01 | n.d. | n.d. | n.d. | 0.1 | 0.05 | 0.02 |
| P-cymene | 0.2 | 12.1 | 0.9 | 6.7 | n.d. | n.d. | 0.1 | 1.1 | 0.1 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Between + paraxylene | 0.1 | 0.5 | 0.4 | 1.0 | 5.5 | 0.03 | 0.2 | 12.3 | 0.04 | 0.02 | 0.1 | <0.01 | 0.01 | 0.02 | 0.04 | 0.1 |
| Tert-butylbenzene | 0.1 | 3.1 | 0.4 | 3.3 | 2.4 | n.d. | n.d. | 0.7 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.2 |
| 1,2-Dibromo-3-chloropropane | n.d. | 0.9 | n.d. | 0.7 | 7.9 | n.d. | n.d. | 0.2 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| trans-1,3-Dichloropropene | 0.1 | 0.1 | 0.2 | 0.1 | 0.5 | 0.1 | 0.1 | 0.3 | 0.8 | n.d. | 1.0 | 0.1 | 2.9 | 0.1 | 0.08 | 0.7 |
| Isopropylbenzene | 0.02 | 0.3 | 0.1 | 0.4 | 3.8 | 0.1 | 0.03 | 1.1 | n.d. | <0.01 | 0.01 | 0.01 | n.d. | n.d. | <0.01 | n.d. |
| Butylbenzene | 0.02 | 2.2 | 0.2 | 1.3 | 1.3 | n.d. | n.d. | 0.2 | n.d. | 0.6 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Styrene | <0.01 | 0.1 | 0.04 | 0.2 | 2.0 | n.d. | 0.01 | 1.1 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| 1,1,2-Trichloroethane | 0.01 | 0.02 | n.d. | 1.5 | n.d. | 0.1 | 0.3 | 0.2 | n.d. | 0.1 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Bromodichloromethane | n.d. | n.d. | <0.01 | n.d. | n.d. | n.d. | n.d. | 0.5 | n.d. | 0.6 | n.d. | n.d. | 0.01 | n.d. | n.d. | n.d. |
| Benzene | 0.1 | 0.01 | <0.01 | 0.1 | 0.03 | 0.4 | 0.1 | 0.03 | 0.02 | 0.02 | 0.1 | 0.1 | n.d. | 0.01 | 0.01 | 0.1 |
| 1,1-Dichloroethylene | n.d. | n.d. | <0.01 | 0.01 | 0.6 | n.d. | n.d. | n.d. | n.d. | 0.01 | n.d. | n.d. | n.d. | 0.04 | 0.03 | 0.2 |
| 1,2-Dichloropropane | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.4 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Tetrachloroethylene | 0.01 | 0.02 | 0.01 | 0.02 | 0.02 | n.d. | 0.02 | 0.02 | 0.03 | 0.01 | 0.03 | 0.03 | 0.1 | 0.03 | 0.03 | 0.02 |
| Hexachloro-1,3-butadiene | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.2 | n.d. | n.d. | n.d. | n.d. | n.d. |
| 1,2,3-Trichlorobenzene | n.d. | n.d. | n.d. | n.d. | 0.1 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.1 |
| 1,1-Dichloropropene | n.d. | n.d. | n.d. | 0.01 | n.d. | n.d. | n.d. | 0.1 | n.d. | n.d. | n.d. | n.d. | 0.1 | n.d. | n.d. | n.d. |
| 1,1,2,2-Tetracholaneethane | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.01 | 0.03 | n.d. | n.d. | n.d. | 0.1 | n.d. | n.d. | n.d. | n.d. |
| cis-1,3-Dichloropropene | n.d. | n.d. | n.d. | n.d. | 0.03 | n.d. | 0.04 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Propylbenzene | n.d. | 0.05 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Chlorodibromomethane | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.04 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| C Σ27VOCs | 5.0 | 77.8 | 29.8 | 82.6 | 99.8 | 1.3 | 2.3 | 81.4 | 2.5 | 2.0 | 20.0 | 0.4 | 3.4 | 0.5 | 0.6 | 1.8 |
For the commodity manufacturing industries, different emission profiles were observed. The CΣ27VOCs of four major categories were in the order of haloalkanes (2.7 µg m−3, 65.80%) > aromatic hydrocarbons (0.7 µg m−3, 16.3%) = halogenated olefins (0.7 µg m−3, 16.3%) > halogenated aromatic hydrocarbons (0.1 µg m−3, 1.7%). Moreover, the 1,2,3-trichloropropane (2.6 µg m−3, 14.6%), trans-1,3-dichloropropene (0.6 µg m−3, 20.4%), o-xylene (0.4 µg m−3, 23.9%), bromodichloromethane (0.09 µg m−3, 4.6%), and butylbenzene (0.1 µg m−3, 4.4%) were the top five VOC analogues emitted from other commodity manufacturing industries. Different analogue profiles of VOCs might be associated with the different raw materials used in these industries. Among them, the dominant analogue in the packaging enterprise was 1,2,3-trichloropropane (17.5 µg m−3, 87.9%), whereas that in the clothing enterprise was trans-1,3-dichloropropene (2.9 µg m−3, 84.2%). Previous studies have shown that the main emission source of VOCs in the packaging enterprise was related to the use of printing inks,30 whereas that in the clothing enterprise was the polyester products,31 which are supposed to be related to the processes of printing and dyeing, respectively.
It was found that there were relatively limited studies on the analogue profiles of VOCs emitted from typical industries in Wuhan, which hindered the data comparison. Therefore, previous monitoring data19 in other industrial areas was applied for comparison. It was observed that the VOC emission profiles previously reported in Yichang (Hubei Province) were similar to those in this study, whose sampling sites were also set in Hubei Province. VOC emission profiles obtained in Yichang were based on monitoring data, which were affected by various industries. It was previously observed that the percentage values of halogenated hydrocarbons (32.6% to 80.7%) were higher than those for aromatic hydrocarbons (33.1% to 55.1%) from commodity manufacturing industries in Yichang.19 These emission profiles were similar to those from commodity manufacturing industries in this study, with higher halogenated hydrocarbons (19.3% to 94.8%) than aromatic hydrocarbons (5.2% to 69.8%). For the VOC emission profiles observed in industrial areas in other cities, similar high percentage values of aromatic hydrocarbons from vehicle-related industries were also observed in Chongqing,20 Nanjing,16 Zhengzhou,10 Beijing,15 and Shanghai16 (27.1–90%). The significant commonalities in the emission profiles of industrial VOCs in different regions not only verified the validity of the data in this study, but also stressed the importance of providing a scientific basis for formulating regional coordinated VOC control strategies. In addition, aromatic hydrocarbons, especially the benzene series of compounds, were previously identified to have a significant contribution to the O3 pollution and adverse impact on public health.32 The higher percentage values of aromatic hydrocarbons in vehicle-related industries observed in this study attract attention to the impact of these industries on the regional air pollution in Wuhan. The adverse impacts of vehicle-related industries on the air pollution in a large-scale region, including the North China Plain, Central China, and the Pearl River Delta regions,24 were also previously reported based on the large-scale environmental monitoring data, which further stressed the importance of investigating the emission profiles of VOCs from vehicle-related industries.
3.2. Comparison of emission profiles between organized and fugitive emission groups
From the on-site investigation, VOC treatment devices were installed in some of the vehicle-related industries in this study, aiming to reduce the emission of VOC analogues. The industries equipped with VOC treatment devices were defined as the organized emission group (O), whereas the others as the fugitive emission group (F). As shown in Table 1, the average CΣ27VOCs in the organized emission group was 21.9 µg m−3, which is two times lower than that of the fugitive emission group (59.0 µg m−3), implying the validity of VOC treatment devices. According to Chinese emission standards (DB42/1539-2019, China),25 the twenty-seven detectable VOCs in this study were classified into four major categories: benzene, toluene and xylene, the benzene series, and total non-methane hydrocarbons (NMHs). A comparison of the concentrations (in logarithmic value) of these four categories from the organized emission group and their corresponding highest acceptable values25 was made, as presented in Fig. 2. The specific detection data showed that the average concentrations of benzene, toluene and xylene, the benzene series, and NMHs were 0.04 µg m−3, 15.8 µg m−3, 31.5 µg m−3, and 11.7 µg m−3, which are lower than the highest national emission standards of 500 µg m−3, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 µg m−3, 20
000 µg m−3, 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 µg m−3, and 40
000 µg m−3, and 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 µg m−3, respectively. Among them, the difference in value between NMHs and the standard value is the largest, which is related to the relatively lenient national standard for NMHs. This result suggests that current VOC treatment devices can enable the VOCs released from the industries to meet the Chinese emission standards.
000 µg m−3, respectively. Among them, the difference in value between NMHs and the standard value is the largest, which is related to the relatively lenient national standard for NMHs. This result suggests that current VOC treatment devices can enable the VOCs released from the industries to meet the Chinese emission standards.
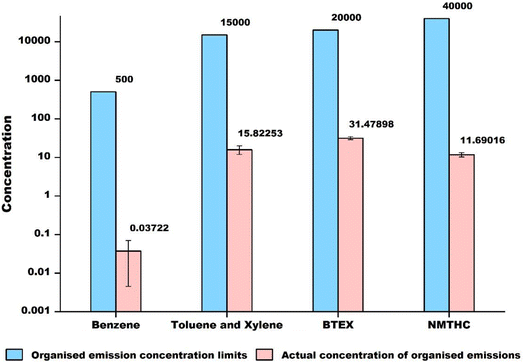 | ||
| Fig. 2 Comparison of the concentrations (in logarithmic value) of VOCs from the organized emission group and emission standard (DB42/1539—2019, China).25 | ||
The VOC treatment device installed in the organized emission group in this study is based on the technology of the zeolite rotary (ZR) concentrator system coupled with regenerative thermal oxidizer (RTO) catalytic combustion. ZR adsorption is a physical adsorption technology aiming to concentrate VOCs emitted from industries by using hydrophobic zeolite materials. The RTO catalytic combustion is an oxidative reaction technology used for the removal of these concentrated VOCs, and it transforms them into CO2 and H2O, especially for the removal of aromatic VOCs.33 Therefore, the VOCs treatment device installed in the organized emission group mainly contains three stages: adsorption, catalytic combustion, and cooling regeneration. Firstly, the VOCs emitted from the industries were adsorbed and concentrated by the hydrophobic zeolite material. Then, these absorbed VOCs were desorbed into gaseous VOCs through thermal desorption. These gaseous VOCs were further treated by the RTO catalytic combustion and converted into CO2 and H2O. Finally, the non-toxic CO2 and H2O were emitted. The hydrophobic zeolite material, after thermal desorption, was rotated and cooled to restore its adsorption capacity, and could be reused in the adsorption stage. Previous research has reported a strong removal effect on aromatic hydrocarbon VOCs through a Pt catalyst in the thermal desorption regeneration stage. It was observed that the removal rate can reach over 97% under the conditions of a reactor inlet temperature of 200 °C and space velocity of 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h−1.34 Aromatic hydrocarbons were observed to be the dominant VOC categories from vehicle-related industries in this study, with seven of the top ten VOC belonging to aromatic hydrocarbon analogues. Given the lack of data about VOC emission profiles before and after the VOC treatment device, the comparison of the VOC emission profiles of organized and fugitive emission groups from vehicle-related industries was carried out to verify the efficiency of this device in actual scenarios (Fig. 3). The percentage values of four major categories of CΣ27VOCs in the organized emission group were in the order of aromatic hydrocarbons (67.3%) > haloalkanes (19.4%) > halogenated aromatic hydrocarbons (12.9%) > halogenated olefins (0.4%), whereas those in fugitive emission group were as follows: aromatic hydrocarbons (89.6%) > halogenated aromatic hydrocarbons (6.5%) > haloalkanes (2.2%) > halogenated olefins (1.8%). Previous studies have proven that the aromatic hydrocarbons are more toxic than other types of VOCs.35 Therefore, reducing the proportion of highly toxic aromatic hydrocarbons35 is essential for eliminating the adverse impact on the public. Moreover, the variation of the proportions is significant in controlling VOC emissions. It should be noted that the proportion of aromatic hydrocarbons in the organized emission group was almost one-third lower than that in the fugitive emission group, implying the validity of the VOC treatment device in reducing aromatic hydrocarbons and its necessity in the vehicle-related industries. Given that the concentrations of emitted VOCs were highly related to the scale of the industries, and the scale of the organized emission group was commonly larger than that of the fugitive emission group, based on the survey. It is no surprise that the total of the aromatic VOCs in the organized emission group (198.7 µg m−3) was much higher than that of the fugitive emission group (78.3 µg m−3). However, a slight increase in the percentage values for halogenated aromatic hydrocarbons in the organized emission group was observed, compared with the fugitive emission group. The impact of halogenated aromatic hydrocarbons on public health should not be ignored due to their potential toxicity. Therefore, the optimization of the VOC treatment device in controlling halogenated aromatic hydrocarbons was also applied in actual scenarios.
000 h−1.34 Aromatic hydrocarbons were observed to be the dominant VOC categories from vehicle-related industries in this study, with seven of the top ten VOC belonging to aromatic hydrocarbon analogues. Given the lack of data about VOC emission profiles before and after the VOC treatment device, the comparison of the VOC emission profiles of organized and fugitive emission groups from vehicle-related industries was carried out to verify the efficiency of this device in actual scenarios (Fig. 3). The percentage values of four major categories of CΣ27VOCs in the organized emission group were in the order of aromatic hydrocarbons (67.3%) > haloalkanes (19.4%) > halogenated aromatic hydrocarbons (12.9%) > halogenated olefins (0.4%), whereas those in fugitive emission group were as follows: aromatic hydrocarbons (89.6%) > halogenated aromatic hydrocarbons (6.5%) > haloalkanes (2.2%) > halogenated olefins (1.8%). Previous studies have proven that the aromatic hydrocarbons are more toxic than other types of VOCs.35 Therefore, reducing the proportion of highly toxic aromatic hydrocarbons35 is essential for eliminating the adverse impact on the public. Moreover, the variation of the proportions is significant in controlling VOC emissions. It should be noted that the proportion of aromatic hydrocarbons in the organized emission group was almost one-third lower than that in the fugitive emission group, implying the validity of the VOC treatment device in reducing aromatic hydrocarbons and its necessity in the vehicle-related industries. Given that the concentrations of emitted VOCs were highly related to the scale of the industries, and the scale of the organized emission group was commonly larger than that of the fugitive emission group, based on the survey. It is no surprise that the total of the aromatic VOCs in the organized emission group (198.7 µg m−3) was much higher than that of the fugitive emission group (78.3 µg m−3). However, a slight increase in the percentage values for halogenated aromatic hydrocarbons in the organized emission group was observed, compared with the fugitive emission group. The impact of halogenated aromatic hydrocarbons on public health should not be ignored due to their potential toxicity. Therefore, the optimization of the VOC treatment device in controlling halogenated aromatic hydrocarbons was also applied in actual scenarios.
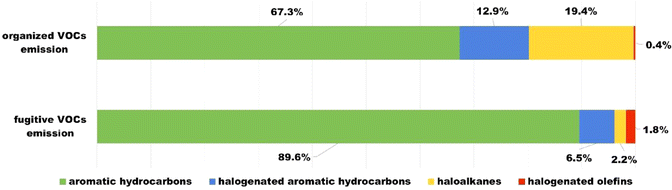 | ||
| Fig. 3 Comparison of the VOC emission profiles of organized and fugitive emission groups from vehicle-related industries. | ||
3.3. Calculated ozone formation potentials
VOCs are crucial precursors of O3, and different VOC analogues have different potential for O3 formation. Therefore, identifying the main VOC analogues that generate O3 in specific regions and reducing their emissions is essential to prevent atmospheric O3 pollution. Based on the real-time data collected from the monitoring and control platform of small air quality monitoring stations of Wuhan, a decreasing trend of O3 concentrations was observed as the distance from vehicle-related industries increased (Fig. 4). The O3 concentrations at monitoring stations within a 6 km radius around vehicle-related industries ranged from 58.3 µg m−3 to 61.4 µg m−3. It decreased to 46.5–56.5 µg m−3 within the range of 6–7 km, and further dropped to 38.8–51.7 µg m−3 beyond 7 km. The percentage value decreased to 13.3–36.5%, indicating the significant impact of VOCs emitted from vehicle-related industries on the regional O3 pollution, whereas the trend of O3 concentrations around commodity manufacturing industries was different. The O3 concentration within a 28 km radius remained consistently at a certain level, with the fluctuation being only ±3 µg m−3 (Fig. 4). Thus, although an overall higher O3 concentration around commodity manufacturing industries than those around vehicle-related industries was observed, the contribution of VOCs emitted from these industries on regional O3 pollution could be ignored. Further attention should be paid to the impacts of VOCs released from vehicle-related industries on the generation of O3.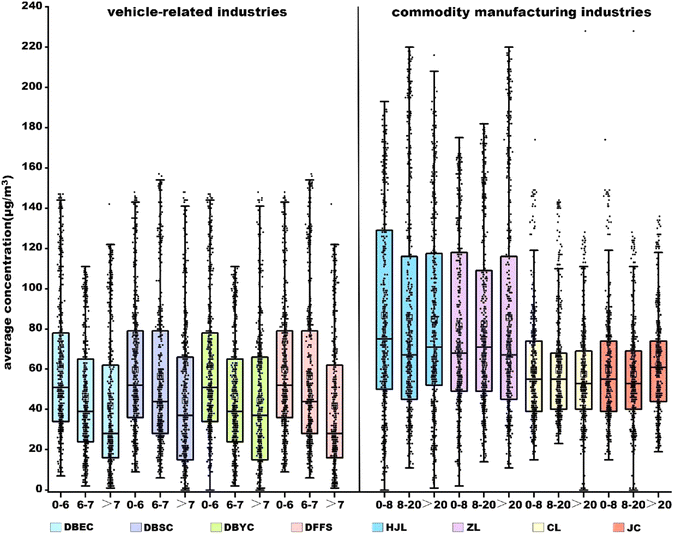 | ||
| Fig. 4 Concentrations of atmospheric ozone around vehicle-related and commodity manufacturing industries. | ||
The calculated OFP values of VOC analogues (ΣOFP) emitted from sixteen factories were 1.1–646.8 µg m−3, with an average value of 126.7 µg m−3. The ΣOFP from vehicle-related industries ranged from 5.3 µg m−3 to 646.8 µg m−3, with an average value of 219.7 µg m−3 (Table S1), whereas the ΣOFP from commodity manufacturing industries ranged from 1.1 µg m−3 to 17.4 µg m−3, with an average value of 7.3 µg m−3 (Table S2). It was observed that aromatic hydrocarbons were dominant categories for ΣOFP from the vehicle-related industry (Table S3). Their OFP values ranged from 4.4 µg m−3 to 630.7 µg m−3, with an average percentage value of 93.0% to ΣOFP. The top five VOC analogues with high percentage values with respect to ΣOFP were 1,3,5-trimethylbenzene (27.3%), o-xylene (25.0%), 1,2,4-trimethylbenzene (18.3%), m-xylene (7.9%), and sec-butylbenzene (7.1%). As for commodity manufacturing industries, the contributions of aromatic hydrocarbons and halogenated olefins to ΣOFP were comparable, with average percentage values of 55.6% and 42.2%, respectively. The VOC analogues with dominant ΣOFP were o-xylene (36.4%) and trans-1,3-dichloropropene (42.2%).
Given the significant impact of VOCs emitted from vehicle-related industries on the regional O3 pollution as mentioned above, the emission sources of crucial VOC analogues generating O3 were further investigated. 1,3,5-Trimethylbenzene and o-xylene emitted from vehicle-related industries have been observed as the most important VOC analogues to the generation of O3 in this study. It is worth noting that the concentrations of these two analogues (22.2 and 29.4 µg m−3) emitted from the spraying process were two to three magnitudes higher than those from the process of sand core baking (0.08 and 0.5 µg m−3), resulting in higher ΣOFP (646.8 µg m−3) calculated in samples close to the spraying process. Previous studies also stressed the importance of the spraying stage on the emission of aromatic hydrocarbons, including m + p-xylene, o-xylene, 1,2,4-trimethylbenzene and benzene,36,37 which were identified as the crucial analogues for O3 generation. Therefore, optimization of the spraying process in vehicle-related industries is the key to controlling VOC emissions and O3 pollution. The usage of solvent-based coatings in the spraying process has been reported as the main reason for a considerable amount of VOC emissions, and powder-type coatings were suggested as substitutes to reduce the emission of VOCs in this process.6 Moreover, a novel car body coating cleaning production method was also proposed to improve production efficiency and further reduce the emission of VOCs.14 It has been previously reported that the whale optimization algorithm can increase the production efficiency by 42.1%, and achieve an exhaust gas purification rate of over 98.0%.14
4. Impacts
This study focused on the emissions of VOCs from industrial sources and their effects on regional O3 pollution in Wuhan, China. Higher concentrations and different analogue profiles of VOCs emitted from vehicle-related industries were observed, compared with those from commodity manufacturing industries. Aromatic hydrocarbons with high toxicity were identified as the main categories from vehicle-related industries, and the VOC treatment device was proven to be efficient for them based on actual scenarios. Monitoring data further indicated the adverse impact of vehicle-related industries on regional O3 pollution, and spraying was the main process releasing VOCs in vehicle-related industries. This is the first study reporting the emission of industrial VOCs in Wuhan and exploring their contribution to O3 pollution. The practical significance of this research is manifested in multiple aspects. From the perspective of China, this study helps the public better understand the hazardous VOC analogues in the regional air, thereby enhancing their awareness of daily protection. Managers who determine whether to install treatment facilities or how to select raw materials in the related industries could refer to the results in this study, which could facilitate effective self-regulation by enterprises. As for the government, the results can provide hints for formulating key VOC control policies, promoting the establishment of an efficient O3 pollution prevention and control regulatory system. From an international perspective, China, as the world's second-largest economy,38 plays an important role in international affairs. Meanwhile, Wuhan is one of the typical cities in the significant Yangtze River Economic Belt in China,39 whose environmental governance experience has a widespread effect. The international community can precisely formulate corresponding control strategies and jointly promote global green and sustainable development, based on the emission characteristics of VOCs in the Wuhan industrial area revealed by this study.Author contributions
Conceptualization: Hao Yanfen. Data curation: Yang Beirou, Han Tiantian, Xu Jing. Funding acquisition: Hao Yanfen. Investigation: Xiang Yuhan, Zhang Tianqi. Methodology: Yang Beirou, Han Tiantian, Xu Jing, Li Wenjuan. Project administration: Hao Yanfen. Supervision: Wang Pu, Liang Yong. Visualization: Xiang Yuhan, Zhang Tianqi. Writing – original draft: Xiang Yuhan, Zhang Tianqi. Writing – review & editing: Hao Yanfen, Wang Pu, Liang Yong.Conflicts of interest
There are no conflicts to declare.Data availability
(1) Data described in [Fig. 4] for this article are available at the monitoring and control platform of small air quality monitoring stations of Wuhan at http://119.96.213.169:8083/index.html#/map/HomeTianMap. It is not accessible to the public.Supplementary information (SI): sampling sites, OFP values and concentrations of VOCs. See DOI: https://doi.org/10.1039/d5ea00109a.
Acknowledgements
This study was supported by National Natural Science Foundation of China (22406067, 42476261), China Scholarship Council (202508420136), Key Research and Development Program of Hubei Province of China (2023BBB067), and the Graduate Scientific Research Foundation of Jianghan University.References
- T. Wang, L. Xue, Z. Feng, J. Dai, Y. Zhang and Y. Tan, Ground-level ozone pollution in China: a synthesis of recent findings on influencing factors and impacts, Environ. Res. Lett., 2022, 17(6), 063003 CrossRef
.
- S. Liu, Y. Zhang, R. Ma, X. Liu, J. Liang, H. Lin, P. Shen, J. Zhang, P. Lu and X. Tang,
et al., Long-term exposure to ozone and cardiovascular mortality in a large Chinese cohort, Environ. Int., 2022, 165, 107280, DOI:10.1016/j.envint.2022.107280FromNLM
.
- S. Safiri, K. Carson-Chahhoud, M. Noori, S. A. Nejadghaderi, M. J. M. Sullman, J. Ahmadian Heris, K. Ansarin, M. A. Mansournia, G. S. Collins and A. A. Kolahi,
et al., Burden of chronic obstructive pulmonary disease and its attributable risk factors in 204 countries and territories, 1990-2019: results from the Global Burden of Disease Study 2019, Bmj, 2022, 378, e069679, DOI:10.1136/bmj-2021-069679FromNLM
.
- Y. Wang, O. Wild, X. Chen, Q. Wu, M. Gao, H. Chen, Y. Qi and Z. Wang, Health impacts of long-term ozone exposure in China over 2013-2017, Environ. Int., 2020, 144, 106030, DOI:10.1016/j.envint.2020.106030FromNLM
.
- W. Guo, Q. Chen, Y. Yang, Y. Zhang, X. Liu, R. Zhang, Y. Zhu, G. Li, P. Liu and M. Chen, Investigating the mechanism of morning ozone concentration peaks in a petrochemical industrial city, Atmos. Environ., 2022, 270, 118897, DOI:10.1016/j.atmosenv.2021.118897
.
- W. J. Liang, J. Z. Wang, C. H. Zhang and J. Liu, Synergetic emission reduction of VOCs and CO2 in the automotive coating industry using life cycle assessment (LCA) technology, J. Environ. Chem. Eng., 2025, 13(2), 115534, DOI:10.1016/j.jece.2025.115534
.
- A. Garg, M. Desservettaz, A. Christodoulou, T. Christoudias, V. P. Kanawade, C. Savvides, M. Vrekoussis, S. Naqui, T. Jokinen, J. Byron, J. Williams, N. Mihalopoulos, E. Liakakou, J. Sciare and E. Bourtsoukidis, Heat and continental transport shape the variability of volatile organic compounds in the Eastern Mediterranean: Insights from multi-year observations and regional modeling, EGUsphere., 2025, 5124, DOI:10.5194/egusphere-2025-5124
.
- L. J. Wang Xinhui and L. Jiang,
et al., Identification of High-VOC Areas in Urban Areas Based on Satellites and Long-Term Time Series Changes, China Environ. Sci., 2025, 45(1), 66–77, DOI:10.19674/j.cnki.issn1000-6923.2025.0006
.
- Z. Zhonghua, L. Q. Kai and T. Xiang, Emission Characteristics and Risk Assessment of VOCs Sources in Typical Industries in Jiangxi Province, Environ. Sci., 2024, 1–15, DOI:10.13227/j.hjkx.202406185FromCnki
.
- N. J. Qi Yijin, Z. Dongxu, Y. Yi, L. Han and Li Bowei, Characteristics and Risk Assessment of VOCs Emissions from Typical Industrial Enterprises in Zhengzhou City, Environ. Sci., 2020, 41, 3056–3065, DOI:10.13227/j.hjkx.201911106
.
- H. Xiao, J. Zhang, Y. Hou, Y. Wang, Y. Qiu, P. Chen and D. Ye, Process-specified emission factors and characteristics of VOCs from the auto-repair painting industry, J. Hazard. Mater., 2024, 467, 133666 CrossRef CAS PubMed
.
- Y. Zhang, J. Liu, Y. Liu, J. Wang, T. Wang, L. Zhong and W.-P. Pan, Producing Pd single site on cerium oxides using ball milling process to accelerate catalytic VOCs removal efficiency: experimental and DFT+ U study, J. Therm. Anal. Calorim., 2025, 1–26 Search PubMed
.
- P. Rybarczyk, Removal of Volatile Organic Compounds (VOCs) from Air: Focus on Biotrickling Filtration and Process Modeling, Processes, 2022, 10(12), 2531, DOI:10.3390/pr10122531PubliclyAvailableContentDatabase
.
- Q. Yang, X. N. Li, T. Yang, H. Wu and L. W. Zhang, An Improved Whale Optimization Algorithm for the Clean Production Transformation of Automotive Body Painting, Biomimetics, 2025, 10(5), 273, DOI:10.3390/biomimetics10050273
.
- X. Liu, H. Zhang, Z. Lv, H. Bai and G. Li, Characterization identification and speciated emission inventory construction of anthropogenic volatile organic compounds (VOCs) in Beijing, China, Atmos. Pollut. Res., 2025, 16(4), 102452 CrossRef CAS
.
- Z. Yue, Analysis of Variation Characteristics and Source of Atmospheric Ozone Pollution in the Yangtze River Delta Region, Res. Environ. Sci., 2024, 37(7), 1500–1512, DOI:10.13198/j.issn.1001-6929.2024.05.09
.
- Y. S. Bao and Y. Deng,
et al., Source profiles and ozone formation potential of VOCs emitted from typical industries using organic solvents in Chengdu[J], Acta Sci. Circumstantiae, 2020, 40(1), 76–82 CAS
.
- W. Tang, X. Yang, P. He, T. Shenwei, Q. Zhang, D. Wang and J. Lin, VOC Emission Spectrum and Industry-Specific Analysis in the Industrial Coating Industry of Hangzhou, China, Coatings, 2025, 15(4), 429, DOI:10.3390/coatings15040429PubliclyAvailableContentDatabase
.
-
N. Zhenzhen, Characteristics of VOCs and Species-Categorized Emission Inventory from Typical Evaporation Emission Sources, China University of Geosciences, 2021 Search PubMed
.
- L. Li, Z. L. Li, W. K. Fang, X. C. Wang, X. Pu, L. T. Wang, R. Yuan, W. D. Zhang and C. Z. Zhai, [Speciated Emission Inventory of VOCs from Industrial Sources and Their Ozone Formation Potential in Chongqing], Huanjing Kexue, 2022, 43(4), 1756–1765, DOI:10.13227/j.hjkx.202108015FromNLM
.
- K. S.-f. SU Wei-feng, Z. H. E. Huang, C. H. E. N. Nan, Bo ZHU, Q. U. A. N. Ji-hong and S.-hua QI, Real-time Composition and Sources of VOCs in Summer in Wuhan, Environ. Sci., 2022, 43(6), 2966–2978 Search PubMed
.
- Y. Liu, J. Xu, Y. Zhou, A. Muhtar and L. Wang, Spatiotemporal Differentiation of the Coupling and Coordination of Production-Living-Ecology Functions in Hubei Province Based on the Global Entropy Value Method, Int. J. Environ. Res. Public Health, 2022, 19(23), 16062, DOI:10.3390/ijerph192316062FromNLM
.
-
Department of Economy and Information Technology of Hubei Province, Concentrate Efforts to Open a New Situation and Work with All Our Might to Continue the Splendor - Part Four: The Operation of the Industrial Economy in the Province in 2021, Department of Economy and Information Technology of Hubei Province, 2022, vol. 4, https://jxt.hubei.gov.cn/bmdt/ztzl/wqzt/jjjl/202201/t20220126_3982058.shtml Search PubMed
.
- A. Mozaffar and Y.-L. Zhang, Atmospheric Volatile Organic Compounds (VOCs) in China: a Review, Curr. Pollut. Rep., 2020, 6(3), 250–263, DOI:10.1007/s40726-020-00149-1
.
- DB42/1539—2019, Emission standard of volatile organic compounds for surface coating of automobile manufacturing industry, 2019 Search PubMed.
- L. Hui, X. Liu, Q. Tan, M. Feng, J. An, Y. Qu, Y. Zhang, Y. Deng, R. Zhai and Z. Wang, VOC characteristics, chemical reactivity and sources in urban Wuhan, central China, Atmos. Environ., 2020, 224, 117340 Search PubMed
.
- EPA, U. S. Detection of volatile organic compounds by gas chromatography/mass spectrometry, 2018 Search PubMed.
- M. A. Venecek, W. P. Carter and M. J. Kleeman, Updating the SAPRC Maximum Incremental Reactivity (MIR) scale for the United States from 1988 to 2010, J. Air Waste Manage. Assoc., 2018, 68(12), 1301–1316 Search PubMed
.
- M. Song and H. Chun, Species and characteristics of volatile organic compounds emitted from an auto-repair painting workshop, Sci. Rep., 2021, 11(1), 16586 CrossRef CAS PubMed
.
- L. D. Zhu, X. Guan, J. Li, Y. B. Peng, X. Zhang, A. B. Gong, M. M. Li, H. Xie, S. R. Chen and J. Li,
et al., Characterization of VOCs emissions and associated health risks inherent to the packaging and printing industries in Shandong Province, China, Sci. Total Environ., 2024, 946, 174108, DOI:10.1016/j.scitotenv.2024.174108
.
- W. R. Qian, Y. Q. Guo, X. P. Wang, X. X. Qiu, X. Ji, L. L. Wang and Y. Li, Quantification and assessment of chemical footprint of VOCs in polyester fabric production, J. Cleaner Prod., 2022, 339, 130628, DOI:10.1016/j.jclepro.2022.130628
.
- A. Tamrakar, S. Pervez, M. Verma, D. Majumdar, Y. F. Pervez, C. Candeias, P. Dugga, A. Mishra, S. R. Verma and M. K. Deb,
et al., BTEX in Ambient Air of India: a Scoping Review of their Concentrations, Sources, and impact, Water, Air, Soil Pollut., 2022, 233(10), 411, DOI:10.1007/s11270-022-05863-8
.
- G. hao, The Application of Zeolite rotor + Regenerative thermal Oxidizer in VOCs Treatment, Zhejiang Chem. Ind., 2021, 52(4), 37–41 Search PubMed
.
- J. R. Zhang Yufan, Y. Qiao, Z. He and F. Xingqiao, Resaerch Progress in the Treatment Method of Atmospheric Benzene Series Pollution, J. North Univ. China (Natural Sci. Ed.), 2021, 42(6), 530–541 Search PubMed
.
- M. Shi, R. Zhang, Y. Wang, Y. Zhang, Y. Zhang and Y. Zhang, Health risk assessments of polycyclic aromatic hydrocarbons and chlorinated/brominated polycyclic aromatic hydrocarbons in urban air particles in a haze frequent area in China, Emerging Contam., 2020, 6, 172–178 CrossRef
.
- Z. J. HUANG Yu and P. QIU, Emission Levels and Composition Characteristics of VOCs from Different Process Links in Automobile Maintenance Industry in Wuhan City, Res. Environ. Sci., 2021, 34(8), 1825–1834 Search PubMed
.
- Y. Wu, Research on the Emission Characteristics and Control Standard System of VOCs in Key Industrial Coating Industries in Shanghai, J. East China Univ. Sci. Technol., 2020, 1–131 Search PubMed
.
-
H. Liping, China as the world's second largest economy: Qualifications and implications, China and East Asia: After the Wall Street crisis, 2013, vol. 33 Search PubMed
.
- X. Ke, A. Mougharbel, H. Guo, D. Wang, Y. Wang and F. Min, Early warning simulation of urban ecological security in the Yangtze River Economic Belt: a case study of Chongqing, Wuhan, and Shanghai, J. Environ. Plann. Manag., 2020, 63(10), 1811–1833 CrossRef
.
| This journal is © The Royal Society of Chemistry 2026 |
