Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Tuning reactivity through implementation of the HSAB concept in oxygen- and sulphur-bridged Al/P and Ga/P FLPs
Julian
Buth
 a,
Yury V.
Vishnevskiy
a,
Yury V.
Vishnevskiy
 ab,
Jan-Hendrik
Lamm
ab,
Jan-Hendrik
Lamm
 a,
Beate
Neumann
a,
Hans-Georg
Stammler
a,
Beate
Neumann
a,
Hans-Georg
Stammler
 a and
Norbert W.
Mitzel
a and
Norbert W.
Mitzel
 *a
*a
aLehrstuhl für Anorganische Chemie und Strukturchemie, Centrum für Molekulare Materialen CM2, Fakultät für Chemie, Universität Bielefeld, Universitätsstraße 25, Bielefeld 33615, Germany. E-mail: mitzel@uni-bielefeld.de
bDepartment of Chemistry, M. V. Lomonosov Moscow State University, Leninskie Gory 1–3, Moscow 119991, Russia
First published on 22nd January 2026
Abstract
Sulphur-bridged frustrated Lewis pairs (FLPs) of the type Bis2E-S-PtBu2 (ESP; E = Al, Ga) were synthesised in analogy to their oxygen-bridged E–O–P analogues (EOP). An exchange reaction between AlSP and GaOP affords selectively and in accordance with the concept of hard and soft acids and bases (HSAB) the inverted systems AlOP and GaSP. Reactivity studies towards small molecules, such as CO2, CS2, SO2, N2O, and propylene sulphide, revealed differences in adduct formations. The adducts EXP·CX2 and EXP·SO2 (X = O, S) consist of five-membered heterocylces. The oxidation products EXP·X are four-membered rings; they result from the reaction of the EXP with N2O and/or propylene sulfide (under loss of propene), except the reaction of AlSP with propylene sulfide that forms a six-membered ring with the whole substrate molecule. The FLP GaSP is exceptional because the formation of its CO2 adduct is temperature-dependent, confirmed by variable-temperature NMR studies, and its adduct GaSP·CS2 has two structural isomers. All CS2 adducts impress with different colours in solution.
Introduction
With the concept of hard and soft acids and bases (HSAB) Ralph G. Pearson defined simple, but reliable rules for interactions between different categories of Lewis acids (LA) and bases (LB).1 The classification into ‘hard’ and ‘soft’ particles, depending on their orbital characteristics, charge density, polarizability, and electronegativity adds a qualitative, predictive power to Lewis theory, especially for stability and direction of reactions. For instance, palladium recycling has recently been improved by applying this principle and exploiting the strong HSAB affinity of Pd2+ ions for S2− species in the active site of a capture material.2In view of the increased demand for metal-free catalysts and the discovery of frustrated Lewis pairs (FLPs) with unique reactivity by Douglas W. Stephan in 2006, research in this area has continuously attracted interest.3–5 Different intra-, intermolecular and hidden FLPs have been developed, featuring various LA/LB element combinations and substituents.6–16 As limited reversibility has hindered broader catalytic applications, careful tuning of Lewis acidity and basicity is crucial to achieve the necessary balance between reactivity and turnover.
Modulation of the linker moiety in intramolecular systems allows adjusting the strength of the Lewis acidity.17,18 Comparing CO2 adducts of methylene- and oxygen-bridged (C2F5)3SiX-PtBu2 FLPs (X = CH2, O), shows that the electron density donated by the oxygen atom entails a temperature-dependent equilibrium, while the CH2-spacer leads to an irreversible adduct.13,19 Other donating linker units consist of nitrogen atoms, for example Al- and Ga-hydrazides, which are easily accessible via hydrometallation of hydrazones.20,21 Further, we synthesised chalcogen-bridged Sb/P systems and their formal HCl adducts.16
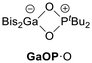
Recently, we described oxygen-bridged FLPs of the type Bis2EOPtBu2 (EOP, Bis = CH(SiMe3)2, E = Al, Ga) based on aluminium and gallium with a focus on their reactivity towards hydrogen (Scheme 1).22 While both systems split hydrogen, AlOP is capable of reducing CO2 to the formate stage in two successive steps.23 For the gallium analogue we found a dynamic temperature-dependent equilibrium of the hydrogen adduct Bis2Ga(H)OP(H)tBu2 (GaOP·H2), separating to free Bis2GaH and phosphane oxide at higher temperatures.22 Beyond small molecule activation and adduct formation, the controlled release of bound species remains essential for enabling catalysis with FLPs. In this work, we implemented the HSAB concept to tune reactivity towards small molecules of oxygen- and sulphur-bridged Al/P and Ga/P FLPs.
Results and discussion
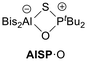
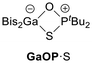
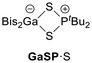
In analogy to the synthesis of Bis2AlOPtBu2 (AlOP),23 we reacted Bis2AlH with the phosphane sulphide tBu2P(S)H to afford the sulphur-bridged Bis2AlSPtBu2 (AlSP) in a dehydrogenation reaction (Scheme 2). Synthetic access to Bis2GaSPtBu2 (GaSP) was achieved by reacting the gallium bromide Bis2GaBr with tBu2P(S)H followed by deprotonation and salt elimination reaction with potassium hexamethyldisilazanide (KHMDS) – also in analogy to its oxygen-bridged counterpart GaOP. | ||
| Scheme 2 Synthesis of the sulphur-bridged systems AlSPvia dehydrogenation and GaSPvia deprotonation and salt elimination from di-tert-butylphosphane sulphide. | ||
The free ESP systems were isolated (AlSP: quantitative; GaSP: 96%) and fully characterised using NMR spectroscopy, elemental analysis, and single crystal X-ray diffraction experiments. The 1H NMR spectra show that both FLPs exhibit similar shifts and coupling constants for the doublet of the tert-butyl groups: 1.24 ppm (3JP,H = 11.6 Hz) for AlSP and 1.27 ppm (3JP,H = 11.5 Hz) for GaSP. The methine protons were detected at 0.27 ppm (AlSP) and 1.06 ppm (GaSP), respectively, both strongly downfield-shifted compared to their oxygen-bridged FLP analogues (AlOP: −0.38 ppm; GaOP: 0.53 ppm). Likewise, the 31P{1H} NMR spectra display equal singlets at 69.2 ppm (AlSP) and 68.5 ppm (GaSP), whereas the corresponding EOP systems exhibit far greater downfield shifts (AlOP: 142.6 ppm; GaOP: 143.5 ppm).
The solid-state structures of the free FLPs ESP show trigonal planar coordinated aluminium or gallium atoms, respectively (Fig. 1). Both structures have similar E⋯P distances as well as E–S and S–P bond lengths, while the latter are found to be longer than in tBu2P(S)H (1.967(1) Å).24 The E–S–P angles are identical (AlSP: 106.3(1)°; GaSP: 106.1(1)°) and vastly narrower than the E–O–P angles (AlOP: 138.1(1)°; GaOP: 126.2(2)°).
In contrast to the synthesis of AlOP, the dehydrogenation to AlSP is fast and irreversible. No signals for the H2 adduct AlSP·H2 were detected, even under hydrogen atmosphere. Contrary to the established procedure for the oxygen-bridged systems EOP, no adduct formation to a formal intermediate HBr adduct Bis2Ga(Br)·SP(H)tBu2 (GaSP·HBr) was observed. Even though an interaction should be favoured, according to the HSAB concept, the signals of Bis2GaBr and tBu2P(S)H are both present in the 1H NMR spectrum. Presumably the relatively low Lewis acidity and basicity of both components prevent the adduct formation. Using the more Lewis acidic Bis2AlBr instead, the formal HBr adduct AlSP·HBr is afforded in quantitative yield. The proton NMR spectrum shows a doublet at 6.02 ppm with a 1JP,H coupling constant of 445 Hz, which is downfield-shifted and wider than in free tBu2P(S)H (5.51 ppm; 1JP,H = 415 Hz). Similarly, the methine proton resonance at −0.41 ppm indicates a tetra-coordinated aluminium atom compared to the tri-coordinate one in the free AlSP system (0.27 ppm). The shifts and coupling constants are in line with those of the oxygen-bridged HBr adducts EOP·HBr.22
The determination of the molecular structure of AlSP·HBr in the crystalline state confirmed the tetrahedral environment at the aluminium atom (Fig. 1). The Al⋯P distance and the Al–S bond are longer, while the S–P bond is shorter than in the free FLP AlSP. In addition to a wider Al–S–P angle for AlSP·HBr, these trends are congruent to the EOP/EOP·HBr systems. However, in contrast to the oxygen-bridged HBr adducts, it is noticeable that the H⋯Br distance with 2.75(2) Å is less than the sum of the van der Waals radii (3.06 Å).25 Furthermore, the torsion angle τ(Br–Al–P–H) of AlSP·HBr is 3.7(8)° and suggests an attractive H⋯Br interaction as well, which is significantly lower than in the EOP·HBr adducts (AlOP·HBr τ = 56.1(5)°; GaOP·HBr τ = 71.6(8)°). Possibly, this HBr-contact stabilises the AlSP·HBr adduct and is a further factor, why the aluminium containing system forms this adduct in contrast to its gallium analogue.
Strikingly, the reaction of AlSP and GaOP yielded an exchange to AlOP and GaSP, which is in accordance with the HSAB concept (Scheme 3). Classifying aluminium and oxygen atoms as ‘hard’ and gallium and sulphur atoms as ‘soft’ in this context, this exchange benefits from the favoured ‘hard–hard’ and ‘soft–soft’ interactions. Monitoring this selective exchange 31P{1H} NMR spectroscopically, full conversion was achieved within 48 h (Fig. 2). Furthermore, the same tendency was observed, when adding tBu2P(S)H to GaOP, which results in the formation of tBu2P(O)H and the GaSP system. We calculated the thermodynamics of both exchange reactions and confirmed the experimental outcome. GaOP + AlSP → GaSP + AlOP results in an energy gain of −10.0 kcal mol−1 and GaOP + tBu2P(S)H → GaSP + tBu2P(O)H yields −1.9 kcal mol−1 (for more details see Table S5 in the SI).
 | ||
| Scheme 3 Exchange reactions of the oxygen- and sulphur-bridged systems GaOP/AlSP to AlOP/GaSP as well as GaOP/tBu2P(S)H to GaSP/tBu2P(O)H. | ||
 | ||
| Fig. 2 Sections of the 31P{1H} NMR spectra of the exchange reaction between the oxygen- and sulphur-bridged systems GaOP/AlSP and AlOP/GaSP in C6D6. | ||
A series of investigations were conducted with the objective of ascertaining the reactivity of the free FLPs towards a range of small molecules, including CO2, CS2, and SO2 (Scheme 4). All isolated adducts were fully characterised by means of NMR spectroscopy, elemental analyses and single crystal X-ray diffraction experiments.
 | ||
| Scheme 4 Reactions of EXP with carbon dioxide, carbon disulfide, and sulphur dioxide to their corresponding adducts with five-membered rings. | ||
Starting with carbon dioxide, adduct formation with CO2 to EXP·CO2 was observed for all systems. This was confirmed spectroscopically by 13C{1H} NMR measurements, with the characteristic doublet for the CO2 unit at 167.2 (1JP,C = 98.5 Hz) for GaOP·CO2, 163.3 (1JP,C = 79.0 Hz) for AlSP·CO2 and 164.2 (1JP,C = 75.1 Hz) for GaSP·CO2, respectively. In all cases the methine proton resonance and the 31P{1H} NMR signal shift strongly towards higher field and the coupling constants of the tert-butyl group become larger compared to the free EXP systems (Table 1). Whereas GaOP·CO2 and AlSP·CO2 were isolable solids, like the previously reported AlOP·CO2,23 the GaSP·CO2 adduct is in an equilibrium with free GaSP plus CO2 and only the free FLP was obtained in the residue after drying under reduced pressure. Under CO2 atmosphere (1 bar) and 298 K in a closed Young NMR tube, the ratio of free GaSP and adduct GaSP·CO2 is about 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8, as determined by integration of the 1H NMR signals. Variable temperature (VT) NMR measurements in the range between 253 and 373 K confirm the reversibility (Fig. 3). At 273 K and below, the equilibrium shifts completely to the side of the adduct GaSP·CO2, and no observable signal for GaSP remains in the 31P{1H} NMR spectrum. Heating to 373 K, results in the release of CO2 and the recovery of the free GaSP. With higher temperatures, also a gradual downfield shift is observed. Minor impurities with tBu2P(S)H, due to hydrolysis, are subject to the same trend. Additionally, quantum chemical calculations of the Gibbs free energy (
8, as determined by integration of the 1H NMR signals. Variable temperature (VT) NMR measurements in the range between 253 and 373 K confirm the reversibility (Fig. 3). At 273 K and below, the equilibrium shifts completely to the side of the adduct GaSP·CO2, and no observable signal for GaSP remains in the 31P{1H} NMR spectrum. Heating to 373 K, results in the release of CO2 and the recovery of the free GaSP. With higher temperatures, also a gradual downfield shift is observed. Minor impurities with tBu2P(S)H, due to hydrolysis, are subject to the same trend. Additionally, quantum chemical calculations of the Gibbs free energy ( ) verifies the experimental observed equilibrium.
) verifies the experimental observed equilibrium.
 | ||
| Fig. 3 Details of the 31P{1H} VT NMR spectra of GaSP under CO2 atmosphere (1 bar) recorded in the range between 273 and 373 K in toluene-d8. | ||
| 1H (ECH)/ppm | 1H (tBu)/ppm | 13C{1H} (PCX2)/ppm | 31P{1H}/ppm | |
|---|---|---|---|---|
| GaOP·CO2 | −0.35 | 1.06 (3JP,H = 15.1 Hz) | 167.2 (1JP,C = 98.5 Hz) | 57.1 |
| AlSP·CO2 | −0.79 | 1.05 (3JP,H = 16.8 Hz) | 163.3 (1JP,C = 79.0 Hz) | 73.3 |
| GaSP·CO2 | −0.27 | 1.13 (3JP,H = 16.4 Hz) | 164.2 (1JP,C = 75.1 Hz) | 67.9 |
| GaOP·CS2 | −0.17 | 1.14 (3JP,H = 14.9 Hz) | 237.8 (1JP,C = 42.4 Hz) | 63.5 |
| AlSP·CS2 | −0.59 | 1.14 (3JP,H = 16.5 Hz) | 232.7 (1JP,C = 18.3 Hz) | 91.9 |
| GaSP·CS2 | −0.12 | 1.19 (3JP,H = 16.3 Hz) | 233.0 (1JP,C = 16.2 Hz) | 91.7 |
| GaOP·SO2 | −0.31, −0.03 | 0.87/1.21 (3JP,H = 14.5/15.1 Hz) | — | 88.1 |
| AlSP·SO2 | −0.69, −0.57 | 0.89/1.18 (3JP,H = 16.3/16.4 Hz) | — | 117.0 |
| GaSP·SO2 | −0.08 | 1.07 (br. s) | — | 114.9 |
Using carbon disulfide instead, an adduct formation to EXP·CS2 was achieved. Similar to the CO2 adducts EXP·CO2, the carbon atom of the CS2 unit in EXP·CS2 show a low field shift in the 13C{1H} NMR spectra (232.7–237.8 ppm) compared to free CS2 (192.7 ppm)26 and is split into a doublet due to the 1JP,C-coupling (Table 1). Expectedly, the methine proton resonances of EXP·CS2 show a weaker downfield shift (Δδ = 0.15–0.20 ppm) than in EXP·CO2.
Interestingly, the colours of the adduct solutions range from blue (GaOP·CS2) over green (AlSP·CS2) to yellow (GaSP·CS2), while the obtained crystals were all violet (see Fig. S1 in the SI). UV/vis spectroscopic analyses show single, isolated absorption bands for GaOP·CS2 (λmax = 589 nm) and AlSP·CS2 (λmax = 613 nm), while for GaSP·CS2 two shoulders indicate two species (Fig. S90/S91, SI). TD-DFT calculations suggest that mainly HOMO → LUMO transitions induce the colouring, while in the CO2 adducts this transition is shifted into the non-visible area (see SI).
Moreover, the reactivity of GaSP towards CS2 is special and results in a mixture of two species. Beside the already mentioned ‘classical’ adduct with Ga–S–C–P connectivity, another similar set of signals is observed. Due to the further low-field shift of the CS2 unit (13C{1H} NMR: 255.8 ppm), the absence of a P,C-coupling, a signal at 97.1 ppm in the 31P{1H} NMR spectrum, and the results of the elemental analyses we suggest it to be an isomer. The ratio of ‘classical’ to ‘inverted’ adduct is approximately 2.6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0. The ‘inverted’ adduct with Ga–C–S–P connectivity matches with the NMR data. Although the calculated thermodynamics for this adduct GaSP·S2C is +22.2 kcal mol−1 higher than the ‘classical’ adduct GaSP·CS2. Further, we optimised acyclic structures and those of a hypothetical six-membered motif plus its dimerisation, whereas the latter is indeed energetically favoured (see SI). However, diffusion-ordered spectroscopy (DOSY) NMR measurements show that both species have similar diffusion coefficients, which contradicts these considerations. Additionally, attempts to calculate a hypothetical four-membered ring adduct failed to converge.
1.0. The ‘inverted’ adduct with Ga–C–S–P connectivity matches with the NMR data. Although the calculated thermodynamics for this adduct GaSP·S2C is +22.2 kcal mol−1 higher than the ‘classical’ adduct GaSP·CS2. Further, we optimised acyclic structures and those of a hypothetical six-membered motif plus its dimerisation, whereas the latter is indeed energetically favoured (see SI). However, diffusion-ordered spectroscopy (DOSY) NMR measurements show that both species have similar diffusion coefficients, which contradicts these considerations. Additionally, attempts to calculate a hypothetical four-membered ring adduct failed to converge.
By reacting the EXP systems with sulphur dioxide, and contrary to the partial decomposition with AlOP,19 stable adducts were isolated for all three analogues EXP·SO2. All resonances in the 1H NMR spectra for GaOP·SO2 and AlSP·SO2 are split, presumably due to the chirality at the sulphur atom. For GaSP·SO2 extremely broad resonances were detected, especially in the 13C{1H} NMR spectrum for the methine and the tert-butyl group.
Crystal structure determinations of the isolated CO2, CS2, and SO2 adducts of the EXP FLPs, confirm five-membered rings with an exocyclic oxygen or sulphur atom, respectively (Fig. 4). In all cases the environment of the Lewis acidic atom, aluminium or gallium, is tetrahedral. For an easier comparison of the structures, we listed relevant distances, bond lengths and angles in Table 2. The E⋯P distance for all adducts increases from oxygen-bridged EOP to sulphur-bridged ESP systems and from aluminium to gallium.
| Compound | AlOP | GaOP | AlSP | GaSP | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adduct | CO2 | CS2 | SO2 | CO2 | CS2 | SO2 | CO2 | CS2 | SO2 | CO2 | CS2 | SO2 |
| d(E⋯P)/Å | 2.901(2) | 3.033(1) | — | 3.005(1) | 3.116(1) | 3.095(1) | 3.255(1) | 3.465(1) | 3.427(1) | — | 3.513(1) | 3.481(1) |
| d(E–X)/Å | 1.855(2) | 1.827(1) | — | 1.989(2) | 1.959(1) | 1.981(1) | 2.477(2) | 2.336(1) | 2.414(1) | — | 2.383(1) | 2.451(1) |
| d(X–P)/Å | 1.540(2) | 1.544(1) | — | 1.537(2) | 1.532(1) | 1.536(1) | 1.979(2) | 2.017(1) | 2.008(1) | — | 2.015(1) | 2.007(1) |
| E–X–P/° | 117.1(1) | 128.0(1) | — | 116.4(1) | 126.0(1) | 122.8(1) | 93.2(1) | 105.3(1) | 101.2(1) | — | 105.8(1) | 102.2(1) |
| C–E–C/° | 108.8(2) | 119.6(1) | — | 131.2(2) | 123.8(1) | 125.0(1) | 123.3(1) | 122.4(1) | 123.0(1) | — | 125.2(1) | 127.3(1) |
It is interesting to compare the structural parameters of the EXP adducts with those of the free EXP molecules. Similar to the examples of five-membered heterocyclic adducts of AlOP,19,23,27 the E–X bonds are longer and the X–P bonds are shorter than in the free FLPs. For the E–X–P angle of the different adducts, the smallest changes are observed for the CS2-adducts EXP·CS2, while the carbon dioxide adducts EXP·CO2 show the largest distortions.
While all atoms of the five-membered ring in the EXP·CO2 and EXP·CS2 adducts lie almost in the same plane (including the exocyclic atoms) the SO2 adducts show a substantial distortion from planarity. As expected, due to the presence of the lone electron pair at the sulphur atom, the oxygen atom bound to the aluminium or gallium atom, respectively, as well as the exocyclic oxygen atom deviate from this plane.
Further attempts to generate strained four-membered rings by transferring single oxygen or sulphur atoms were conducted using nitrogen oxide (N2O) and propylene sulphide, respectively (Scheme 5). Interestingly, the EXP FLPs show different behaviours for the reaction with N2O. Accompanying with the release of molecular nitrogen, the formal oxygen adducts Bis2GaOPtBu2·O (GaOP·O) and Bis2AlSPtBu2·O (AlSP·O) were formed, while GaSP showed no reaction even after heating to 70 °C for several days. 1H NMR spectroscopic analyses showed similar high-field shifts for the tert-butyl groups and the methine protons in comparison to EXP for both oxygen adducts. The singlets in the 31P{1H} NMR spectra at 85.2 (GaOP·O) and 116.4 ppm (AlSP·O) also suggest a cooperative bonding of the oxygen atom by the LA and the LB function. For AlSP·O the four-membered heterocyclic motif was verified by X-ray diffraction measurements (Fig. 5). However, due to the limited quality of the crystals, only the connectivity can be discussed.
 | ||
| Scheme 5 Transfer of oxygen- and sulphur atoms by reaction of EXP with nitrogen dioxide and propylene sulphide under the loss of nitrogen and propene, respectively. | ||
Previously reported by Uhl et al., the formation of a sulphide adduct of a benzylidene bridged Ga/P FLP was achieved using propylene sulphide under the release of propene when heated.28 In analogy, propylene sulphide and GaOP led to the formation of propene and the corresponding sulphide adduct Bis2GaOPtBu2·S (GaOP·S) upon heating to 80 °C. The signals of GaOP·S in the 1H NMR spectrum are slightly shifted towards higher field compared to those of GaOP·O. In contrast, the 31P NMR signal of GaOP·S at 117.8 ppm is extremely broad, significantly shifted towards lower field and is between the ones of the free FLPs GaOP (143.5 ppm) and GaSP (68.5 ppm). Although the GaSP system reacts in an unselective way with N2O, the reaction with propylene sulphide afforded Bis2GaSPtBu2·S (GaSP·S) and it was possible to determine its molecular structure in the solid-state, by picking crystals, suitable for X-ray diffraction measurements, from the concentrated solution (Fig. 5).
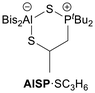
Interestingly, the AlSP system showed a different behaviour (Table 3). In this case, a ring opening of propylene sulphide leads to the formation of a six-membered heterocycle without the outgassing of propene, despite heating to 80 °C. The connectivity was enlightened with the help of NMR spectroscopy: the aluminium atom binds to the sulphur atom, while the methylene unit is linked to the phosphorus atom. In the 1H NMR spectrum the protons of the CH2 unit are diastereotopic and therefore induce two different multiplets (1.62/1.82 ppm). Additionally, the multiplet at 3.12 ppm belongs to the methine proton of the propylene unit, as it is the case for the methyl-group at 1.46 ppm. The methine protons of the Bis-groups are detected at −0.54 ppm. Likewise to the adduct AlSP·SO2, the tert-butyl resonance is split into two doublets at 0.80/0.91 ppm with 3JP,H-couplings constants of 15.3/16.0 Hz. The 31P NMR spectrum displays a broad singlet at 77.3 ppm.
The molecular structure in the solid-state of AlSP·SC3H6 validates the six-membered heterocycle. With 3.707(1) Å the Al⋯P distance is significantly larger than in the above-mentioned adducts of AlSP. The Al–S–P angle is also the widest with 112.2(1)°, probably resulting from the lower ring strain. The exact opposite trends are observed for the GaSP·S system: the Ga⋯P distance (3.034(2) Å) as well as the Ga–S–P angles (85.2(9)°) are drastically smaller than in the free GaSP (3.482(1) Å; 106.1(1)°).
Conclusions
We expanded the range of geminal Al/P and Ga/P FLPs with the synthesis and full characterisation of the sulphur-bridged FLPs Bis2ESPtBu2 (ESP, E = Al, Ga). In contrast to the oxygen-bridged systems EOP, no H2 activation is observed for ESP. Likewise, no formal HBr adduct is formed for the GaSP FLP in the synthetic procedure, whereas AlSP·HBr was isolated and the molecular structure in the solid-state shows a stabilising intramolecular H⋯Br contact. According to the HSAB concept, a selective exchange reaction of AlSP and GaOP leads to AlOP and GaSP.In combination with the oxygen-bridged GaOP system, the reactivity of sulphur-bridged ESP FLPs towards a series of small molecules (CO2, CS2, SO2, N2O, propylene sulphide) was tested. As expected, the adducts EXP·CO2, EXP·CS2, and EXP·SO2 (X = O, S) comprise five-membered rings, and the reaction with N2O results in the oxygen adduct EXP·O, with a four-membered heterocycle. The GaSP FLP is an exception and shows slightly different reactivity: a temperature-dependent CO2 adduct, two structural isomers for GaSP·CS2 and no reaction with N2O.
The interesting colouration of the EXP·CS2 adducts were analysed by UV/vis spectroscopy supported by quantum chemical calculations, which suggests that mainly the HOMO → LUMO transition produces the colouring. Furthermore, activation of propylene sulphide results in different adducts, for both gallium FLPs into the sulphide adduct GaXP·S with outgassing of propene. On the other hand, AlSP forms the addition product, a six-membered heterocycle.
This comparative study demonstrates how minor changes in Lewis acidity and basicity, involving different elements and linker units in intramolecular FLPs, affect reactivity, particularly with regard to the HSAB concept.
Experimental section
General methods
All reactions and manipulations with air and moisture sensitive compounds were carried out under conventional Schlenk techniques or in a glove box using argon as inert gas. Volatile compounds were handled in a vacuum line. The solvents n-hexane, toluene, toluene-d8 and benzene-d6 were dried over a Na/K alloy, dichloromethane over CaH2, and were also distilled and degassed prior to use. Bis2AlH,1 Bis2GaBr,2 Bis2GaOPtBu2,2 and tBu2P(S)H3 were prepared according to literature procedures. CO2 (99.5%, Linde), SO2 (99.98%, Air Liquid), and N2O (extracted from a capsule for cream whipping) were used without further purification. CS2 (99.9%, J.T. Baker) was dried over P4O10, distilled and degassed prior to use. Propylene sulphide (98%, TCI) was distilled and degassed prior to use. NMR spectra were recorded using a Bruker Avance III 500, Avance III 500 HD, Ascend 500 neo2K or Ascend 500 neo3K spectrometer at ambient temperature unless otherwise stated. Chemical shifts were referenced to the residual proton or carbon signal of the solvent (benzene-d6: 1H: 7.16 ppm, 13C: 128.1 ppm; toluene-d8: 1H: 2.09 ppm) or externally (29Si: SiMe4, 31P: 85% H3PO4 in H2O). Elemental analyses were carried out using a HEKATECH EURO Elemental Analyzer.Synthetic procedures
Bis 2 GaOP t Bu 2 ·CO2 (GaOP·CO2) was obtained as a colourless solid (42 mg, 71 μmol, quant.). 1H NMR (500 MHz, C6D6): δ [ppm] = −0.35 (s, 2H, GaCH), 0.37 (s, 18H, Si(CH3)3), 0.39 (s, 18H, Si(CH3)3), 1.06 (d, 3JP,H = 15.1 Hz, 18H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 4.6 (s, Si(CH3)3), 4.8 (s, GaCH), 26.7 (s, C(CH3)3), 34.7 (d, 1JP,C = 45.4 Hz, C(CH3)3), 167.2 (d, 1JP,C = 98.5 Hz, PCO2). 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −1.2 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 57.1 (s). Elemental analysis calcd (%) for C23H56GaO3PSi4 (Mr = 593.73): C 46.53, H 9.51; found C 46.30, H 9.78.
Bis 2 AlSP t Bu 2 ·CO2 (AlSP·CO2) was obtained as a colourless solid (39 mg, 69 μmol, quant.). 1H NMR (500 MHz, C6D6): δ [ppm] = −0.79 (s, 2H, AlCH), 0.41 (s, 18H, Si(CH3)3), 0.42 (s, 18H, Si(CH3)3), 1.05 (d, 3JP,H = 16.8 Hz, 18H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 5.1 (s, Si(CH3)3), 5.3 (s, Si(CH3)3), 5.7 (s, AlCH), 27.3 (s, C(CH3)3), 38.3 (d, 1JP,C = 26.4 Hz, C(CH3)3), 163.3 (d, 1JP,C = 79.0 Hz, PCO2). 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −1.7 (s), −1.3 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 73.3 (s). Elemental analysis calcd (%) for C23H56AlO2PSSi4 (Mr = 567.05): C 48.72, H 9.95, S 5.65; found C 48.93, H 10.23, S 5.48.
Bis 2 GaSP t Bu 2 ·CO2 (GaSP·CO2): GaSP forms a temperature-dependent equilibrium with GaSP·CO2 under an atmosphere of CO2. VT NMR studies were conducted in the range between 253–373 K. 1H NMR (500 MHz, C6D6): δ [ppm] = −0.27 (s, 2H, GaCH), 0.40 (s, 36H, Si(CH3)3), 1.13 (d, 3JP,H = 16.4 Hz, 18H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 4.9 (s, Si(CH3)3), 11.8 (s, GaCH), 27.6 (s, C(CH3)3), 38.1 (d, 1JP,C = 27.0 Hz, C(CH3)3), 164.2 (d, 1JP,C = 75.1 Hz, PCO2). 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −0.6 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 67.9 (s).
Bis2GaOPtBu2·CS2 (GaOP·CS2) crystallises as wide, violet needles (11 mg, 18 μmol, 31%) from a concentrated n-hexane solution at −18 °C. 1H NMR (500 MHz, C6D6): δ [ppm] = −0.17 (s, 2H, GaCH), 0.36 (s, 36H, Si(CH3)3), 1.14 (d, 3JP,H = 14.9 Hz, 18H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 4.7 (s, Si(CH3)3), 4.8 (s, Si(CH3)3), 11.4 (s, GaCH), 27.3 (s, C(CH3)3), 36.6 (d, 1JP,C = 51.0 Hz, C(CH3)3), 237.8 (d, 1JP,C = 42.4 Hz, PCS2). 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −1.5 (s), −0.9 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 63.5 (s). Elemental analysis calcd (%) for C23H56GaOPS2Si4 (Mr = 625.86): C 44.14, H 9.02, S 10.25; found C 44.35, H 8.93, S 10.17.
Bis 2 AlSP t Bu 2 ·CS2 (AlSP·CS2) crystallises as small violet blocks (22 mg, 37 μmol, 47%) from a concentrated n-hexane solution at −18 °C. 1H NMR (500 MHz, C6D6): δ [ppm] = −0.59 (s, 2H, AlCH), 0.40 (s, 18H, Si(CH3)3), 0.42 (s, 18H, Si(CH3)3), 1.14 (d, 3JP,H = 16.5 Hz, 18H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 5.5 (s, Si(CH3)3), 7.0 (s, AlCH), 28.4 (s, C(CH3)3), 41.7 (d, 1JP,C = 29.5 Hz, C(CH3)3), 232.7 (d, 1JP,C = 18.3 Hz, PCS2). 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −1.5 (s), −1.4 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 91.9 (s). Elemental analysis calcd (%) for C23H56AlPS3Si4 (Mr = 599.18): C 46.11, H 9.42, S 16.05; found C 46.44, H 9.80, S 15.69.
Bis 2 GaSP t Bu 2 ·CS2 (GaSP·CS2) crystallises as small violet blocks (56 mg, 87 μmol, 46%) from a concentrated n-hexane solution at −18 °C. ‘Classical’ adduct (GaSCP-connectivity): 1H NMR (500 MHz, C6D6): δ [ppm] = −0.12 (s, 2H, GaCH), 0.38 (s, 18H, Si(CH3)3), 0.39 (s, 18H, Si(CH3)3), 1.19 (d, 3JP,H = 16.3 Hz, 18H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 5.0 (s, Si(CH3)3), 5.1 (s, Si(CH3)3), 12.6 (s, GaCH), 28.6 (s, C(CH3)3), 41.7 (d, 1JP,C = 30.1 Hz, C(CH3)3), 233.0 (d, 1JP,C = 16.2 Hz, PCS2). 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −0.6 (s), −0.5 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 91.7 (s). ‘Inverted’ adduct (GaCSP-connectivity): 1H NMR (500 MHz, C6D6): δ [ppm] = −0.13 (s, 2H, GaCH), 0.24 (s, 36H, Si(CH3)3), 1.44 (d, 3JP,H = 16.0 Hz, 18H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 4.2 (s, Si(CH3)3), 13.2 (s, GaCH), 28.6 (s, C(CH3)3), 40.7 (d, 1JP,C = 36.4 Hz, C(CH3)3), 255.8 (s, GaCS2). 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −0.3 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 97.1 (s). Elemental analysis calcd (%) for C23H56GaPS3Si4 (Mr = 641.92): C 43.04, H 8.79, S 14.98; found C 43.28, H 9.05, S 14.87.
Bis 2 GaOP t Bu 2 ·SO2 (GaOP·SO2) was obtained as a colourless solid (23 mg, 38 μmol, quant.). 1H NMR (500 MHz, C6D6): δ [ppm] = −0.31 (br. s, 1H, GaCH), −0.03 (br. s, 1H, GaCH), 0.37–0.49 (m, 36H, Si(CH3)3), 0.87 (d, 3JP,H = 14.5 Hz, 9H, C(CH3)3), 1.21 (d, 3JP,H = 15.1 Hz, 9H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 4.8 (s, Si(CH3)3), 5.0 (s, Si(CH3)3), 12.2 (br. s, GaCH), 26.7 (s, C(CH3)3), 27.1 (s, C(CH3)3), 37.0 (d, 1JP,C = 21.1 Hz, C(CH3)3), 39.3 (d, 1JP,C = 21.2 Hz, C(CH3)3). 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −1.3 (s), −0.9 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 88.1 (s). Elemental analysis calcd (%) for C22H56GaO3PSSi4 (Mr = 613.78): C 43.05, H 9.20, S 5.22; found C 42.51, H 9.03, S 5.30.
Bis 2 AlSP t Bu 2 ·SO2 (AlSP·SO2) was obtained as a colourless solid (21 mg, 37 μmol, quant.). 1H NMR (500 MHz, C6D6): δ [ppm] = −0.69 (br. s, 1H, AlCH), −0.57 (br. s, 1H, AlCH), 0.31–0.59 (m, 36H, Si(CH3)3), 0.89 (d, 3JP,H = 16.3 Hz, 9H, C(CH3)3), 1.18 (d, 3JP,H = 16.4 Hz, 9H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 5.0 (s, Si(CH3)3), 5.2 (s, Si(CH3)3), 6.1 (s, AlCH), 8.5 (s, AlCH), 27.5 (s, C(CH3)3), 27.9 (s, C(CH3)3), 39.2 (d, 1JP,C = 21.3 Hz, C(CH3)3), 42.4 (d, 1JP,C = 18.9 Hz, C(CH3)3). 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −1.5 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 117.0 (s). Elemental analysis calcd (%) for C22H56AlO2PS2Si4 (Mr = 587.10): C 45.01, H 9.61, S 10.92; found C 45.11, H 9.59, S 10.92.
Bis 2 GaSP t Bu 2 ·SO2 (GaSP·SO2) was obtained as a colourless solid (25 mg, 39 μmol, 95%). 1H NMR (500 MHz, C6D6): δ [ppm] = −0.08 (br. s, 2H, GaCH), 0.44 (s, 36H, Si(CH3)3), 1.07 (br. s, 18H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 5.1 (s, Si(CH3)3), 28.3 (s, C(CH3)3), carbon atoms (GaCH, C(CH3)3) not detected due to broadening. 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −1.1 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 114.9 (s). Elemental analysis calcd (%) for C23H56GaO2PS2Si4 (Mr = 629.84): C 41.95, H 8.96, S 10.18; found C 42.38, H 9.07, S 9.99.
Bis 2 GaOP t Bu 2 ·O (GaOP·O) was obtained as a colourless solid (23 mg, 40 μmol, quant.). 1H NMR (500 MHz, C6D6): δ [ppm] = 0.18 (s, 2H, GaCH), 0.38 (s, 36H, Si(CH3)3), 1.18 (d, 3JP,H = 14.0 Hz, 18H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 4.6 (s, Si(CH3)3), 13.3 (s, GaCH), 27.5 (s, C(CH3)3), 35.7 (d, 1JP,C = 73.5 Hz, C(CH3)3). 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −1.3 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 85.2 (s). Elemental analysis calcd (%) for C22H56GaO2PSi4 (Mr = 565.72): C 46.71, H 9.98; found C 46.76, H 10.13.
Bis 2 AlSP t Bu 2 ·O (AlSP·O) was obtained as a colourless solid (16 mg, 30 μmol, 92%). 1H NMR (500 MHz, C6D6): δ [ppm] = −0.54 (br. s, 1H, AlCH), −0.47 (br. s, 1H, AlCH), 0.42 (s, 36H, Si(CH3)3), 1.09 (d, 3JP,H = 16.3 Hz, 18H, C(CH3)3). 13C{1H} NMR (126 MHz, C6D6): δ [ppm] = 5.1 (s, Si(CH3)3), 5.5 (s, Si(CH3)3), 9.5 (s, AlCH), 27.0 (d, 2JP,C = 2.8 Hz, C(CH3)3), 35.7 (d, 1JP,C = 50.1 Hz, C(CH3)3). 29Si{1H} NMR (99 MHz, C6D6): δ [ppm] = −2.0 (s). 31P{1H} NMR (202 MHz, C6D6): δ [ppm] = 116.4 (s). Elemental analysis calcd (%) for C22H56AlOPSSi4 (Mr = 539.04): C 49.02, H 10.47, S 5.95; found C 49.20, H 10.64, S 5.83.
Author contributions
J. Buth: investigation, methodology, validation, visualisation, writing (original draft), Y. V. Vishnevskiy (quantum chemical calculations), J.-H. Lamm, B. Neumann, and H.-G. Stammler: investigation (SCXRD), N. W. Mitzel: funding acquisition, project administration, supervision, writing, reviewing and editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data that supports the findings of this study are available in the supplementary information (SI). Supplementary information: NMR spectra, crystallographic data and computational details. See DOI: https://doi.org/10.1039/d6dt00151c.CCDC 2503479–2503493 contain the supplementary crystallographic data for this paper.29a–o
Acknowledgements
The authors thank Marco Wißbrock and Dr Andreas Mix for recording VT and DOSY NMR spectra, Barbara Teichner for performing elemental analyses, Erik Niekamp, Marvin Waschipki, Ece Ayhan, and Hannah Koch for support with syntheses.This work was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation, grant MI 477/44-1, project no. 461833739, and grant VI 713/3-1, project no. 243500032). We acknowledge support by the Paderborn Center for Parallel Computing (PC2, HPC system Noctua 2) and by the Regional Computing Centre of the University of Cologne (RRZK, HPC system RAMSES) for providing computing time.
References
- R. G. Pearson, J. Am. Chem. Soc., 1963, 85, 3533–3539 CrossRef CAS
.
- A. D. Pournara, J.-H. Tang, L. Yang, J.-T. Liu, X.-Y. Huang, M.-L. Feng and M. G. Kanatzidis, Chem. Mater., 2024, 36, 3013–3021 CrossRef CAS
.
- D. W. Stephan, G. C. Welch, R. R. S. Juan and J. D. Masuda, Science, 2006, 314, 1124–1126 CrossRef PubMed
.
- D. W. Stephan, J. Am. Chem. Soc., 2015, 137, 10018–10032 CrossRef CAS PubMed
.
- D. W. Stephan, Science, 2016, 354, aaf7229 CrossRef PubMed
.
- P. Spies, G. Erker, G. Kehr, K. Bergander, R. Fröhlich, S. Grimme and D. W. Stephan, Chem. Commun., 2007, 5072 RSC
.
- G. C. Welch and D. W. Stephan, J. Am. Chem. Soc., 2007, 129, 1880–1881 CrossRef CAS PubMed
.
- S. Styra, M. Radius, E. Moos, A. Bihlmeier and F. Breher, Chem. –Eur. J., 2016, 22, 9508–9512 CrossRef CAS PubMed
.
- C. Appelt, H. Westenberg, F. Bertini, A. W. Ehlers, J. C. Slootweg, K. Lammertsma and W. Uhl, Angew. Chem., Int. Ed., 2011, 50, 3925–3928 CrossRef CAS PubMed
.
- J. Possart and W. Uhl, Organometallics, 2018, 37, 1314–1323 CrossRef CAS
.
- K. Chang, X. Wang, Z. Fan and X. Xu, Inorg. Chem., 2018, 57, 8568–8580 CrossRef CAS PubMed
.
- M. Pieper, J.-H. Lamm, B. Neumann, H.-G. Stammler and N. W. Mitzel, Dalton Trans., 2017, 46, 5326–5336 RSC
.
- B. Waerder, M. Pieper, L. A. Körte, T. A. Kinder, A. Mix, B. Neumann, H.-G. Stammler and N. W. Mitzel, Angew. Chem., Int. Ed., 2015, 54, 13416–13419 CrossRef CAS PubMed
.
- P. Holtkamp, F. Friedrich, E. Stratmann, A. Mix, B. Neumann, H.-G. Stammler and N. W. Mitzel, Angew. Chem., Int. Ed., 2019, 58, 5114–5118 CrossRef CAS PubMed
.
- F. Breher, F. Krämer, J. Paradies and I. Fernández, Nat. Chem., 2024, 16, 63–69 CrossRef PubMed
.
- J. Krieft, H. Koch, B. Neumann, H.-G. Stammler, J.-H. Lamm, A. Mix and N. W. Mitzel, Dalton Trans., 2024, 53, 16280–16286 RSC
.
- D. Zhu, Z.-W. Qu and D. W. Stephan, Dalton Trans., 2020, 49, 901–910 RSC
.
- Y. Wang, Z. H. Li and H. Wang, RSC Adv., 2018, 8, 26271–26276 RSC
.
- L. Wickemeyer, P. C. Trapp, N. Aders, B. Neumann, H. Stammler and N. W. Mitzel, Chem. –Eur. J., 2023, 29, e202203685 CrossRef CAS PubMed
.
- W. Uhl, M. Willeke, F. Hengesbach, A. Hepp and M. Layh, Organometallics, 2016, 35, 3701–3712 CrossRef CAS
.
- W. Uhl, M. Willeke, A. Hepp, D. Pleschka and M. Layh, Z. Anorg. Allg. Chem., 2017, 643, 387–397 CrossRef CAS
.
- J. Buth, M. J. Klingsiek, Y. V. Vishnevskiy, A. Mix, J.-H. Lamm, B. Neumann, H.-G. Stammler and N. W. Mitzel, ChemistryEurope, 2025, e20250030723 Search PubMed
.
- L. Wickemeyer, N. Aders, A. Mix, B. Neumann, H.-G. Stammler, J. J. Cabrera-Trujillo, I. Fernández and N. W. Mitzel, Chem. Sci., 2022, 13, 8088–8094 RSC
.
- G. Y. Li and W. J. Marshall, Organometallics, 2002, 21, 590–591 CrossRef CAS
.
- S. Alvarez, Dalton Trans., 2013, 42, 8617 RSC
.
- G. R. Fulmer, A. J. M. Miller, N. H. Sherden, H. E. Gottlieb, A. Nudelman, B. M. Stoltz, J. E. Bercaw and K. I. Goldberg, Organometallics, 2010, 29, 2176–2179 CrossRef CAS
.
- L. Wickemeyer, L. Hartmann, B. Neumann, H. Stammler and N. W. Mitzel, Chem. –Eur. J., 2023, 29, e202202842 CrossRef CAS PubMed
.
- W. Uhl, J. Possart, A. Hepp and M. Layh, Z. Anorg. Allg. Chem., 2017, 643, 1016–1029 CrossRef CAS
.
-
(a)
CCDC 2503479: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12d8
; (b) CCDC 2503480: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12f9
; (c) CCDC 2503481: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12gb
; (d) CCDC 2503482: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12hc
; (e) CCDC 2503483: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12jd
; (f) CCDC 2503484: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12kf
; (g) CCDC 2503485: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12lg
; (h) CCDC 2503486: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12mh
; (i) CCDC 2503487: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12nj
; (j) CCDC 2503488: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12pk
; (k) CCDC 2503489: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12ql
; (l) CCDC 2503490: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12rm
; (m) CCDC 2503491: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12sn
; (n) CCDC 2503492: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12tp
; (o) CCDC 2503493: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q12vq
.
| This journal is © The Royal Society of Chemistry 2026 |