Open Access Article
Open Access ArticleCovalent triazine framework@silica core–shell spheres decorated with ultrafine platinum nanoparticles: a robust catalyst for selective aqueous phase hydrogenation of butane-2,3-dione and pyruvic acid
Claudio
Evangelisti
 *a,
Werner
Oberhauser
*b,
Francesco
Poggialini
a,
Emanuela
Pitzalis
a,
Xuan Trung
Nguyen
a,
Esther
Punzi
a,
Serena
Coiai
*a,
Werner
Oberhauser
*b,
Francesco
Poggialini
a,
Emanuela
Pitzalis
a,
Xuan Trung
Nguyen
a,
Esther
Punzi
a,
Serena
Coiai
 a,
Nicola
Scotti
a,
Nicola
Scotti
 c,
Lorenzo
Poggini
bd and
Alessandro
Mandoli
c,
Lorenzo
Poggini
bd and
Alessandro
Mandoli
 e
e
aInstitute of Chemistry of OrganoMetallic Compounds, ICCOM-CNR, Via G. Moruzzi 1, 56124 Pisa, Italy. E-mail: claudio.evangelisti@cnnr.it
bInstitute of Chemistry of OrganoMetallic Compounds, ICCOM-CNR, Via Madonna del Piano 10, 50019 Sesto Fiorentino, FI, Italy. E-mail: werner.oberhauser@cnr.it
cInstitute of Science and Chemical Technologies “Giulio Natta”, SCITEC-CNR, Via C. Golgi 19, 20133 Milano, Italy
dDepartment of Chemistry “U. Schiff” – DICUS – and INSTM Research Unit, University of Florence, Via della Lastruccia 3-13, 50019 Sesto Fiorentino, FI, Italy
eDepartment of Chemistry and Industrial Chemistry, University of Pisa, Via G. Moruzzi 13, 56124 Pisa, Italy
First published on 9th January 2026
Abstract
Sub-nanometric Pt nanoparticles, generated by the metal vapor synthesis technique, were supported on covalent triazine framework@silica (CTF@SiO2) core–shell hybrid microspheres obtained through polycondensation reactions at the SiO2 surface. The latter catalyst and a reference system (i.e. Pt nanoparticles deposited on bare SiO2 microspheres) were screened under identical experimental conditions for the hydrogenation of butane-2,3-dione and pyruvic acid in water, yielding butane-2,3-diol and lactic acid with high selectivity. A combination of catalytic recycling experiments was conducted with both catalysts, along with transmission electron microscopy and X-ray diffraction analyses performed on both the as-synthesized and recovered catalysts, which confirmed the remarkable role of the CTF shell layer in the stabilization of ultrafine Pt NPs against their aggregation under catalysis as well as in enhancing their catalytic activity.
1. Introduction
Since their first report in 2005, covalent organic frameworks (COFs) have received increasing interest in catalysis.1,2 The ordered structure and the light-element composition of COF-based materials lead to a high specific surface area and adjustable pore sizes. Moreover, their customizable structure allows for the incorporation of various functional groups, making them promising materials for different catalytic applications such as photocatalysis, electrocatalysis, energy storage, and fine chemical production.1,3,4 Among COFs, covalent triazine frameworks (CTFs) stand out due to their unique porous structure, which is attributed to the 1,3,5-triazine units with π-conjugation properties that enhance their thermal and chemical stability.5–7 Their high nitrogen content, high surface area, and high porosity make them versatile hosts for catalytically active metal species, ranging from single metal atoms to nanoparticles (NPs).8,9 In fact, the porosity of the polymeric support layer, along with the favourable electron donor properties of the support, is a prerequisite for high catalytic activity in metal-based heterogeneous catalysis.10–12 Recently, both noble and non-noble metal NPs like Pd, Pt and Ni13–15 have been successfully immobilized on CTFs, exhibiting elevated dispersion even at high metal loadings and improved catalytic efficiency and recyclability/durability for selective hydrogenation reactions compared to conventional supports (e.g. carbon).Although different synthesis approaches for CTFs have been developed,16 the large-scale production of these materials still remains a challenge.17
Hybrid particles with a silica core and a CTF porous shell combine the advantages of the shell material and the mechanical stability of silica, significantly reducing the required amount of CTF material. As a consequence, the synthesis of the catalytic support can be easily scaled up. In this work, we present a facile approach to fabricate morphology-controllable CTF shells through polycondensation reactions using SiO2 core spheres as a hard template. Sub-nanometer Pt NPs were generated by metal vapour synthesis (MVS),18,19 which allows the synthesis of size-tailored metal NPs regardless of the nature of the support. The CTF@SiO2 system was selected as a carrier for the ultrafine Pt NPs to obtain the Pt/CTF@SiO2 composite (1). Pure silica spheres were also used to obtain the Pt/SiO2 system (2) as a reference catalytic material. Both heterogeneous catalysts were thoroughly characterized by microscopic, spectroscopic, diffraction and thermogravimetric techniques and then used to promote the hydrogenation of butane-2,3-dione (2,3-BDO) and pyruvic acid (PyA) in water. The chemoselective conversion of both substrates into butane-2,3-diol and lactic acid, respectively, which are recognized as bio-based platform molecules,20,21 is challenging, since both substrates undergo undesired side reactions due to their reactivity (i.e. dimerization of 2,3-BDO due to the keto–enol tautomerization22 and decarboxylation of PyA23). Hence, the development of an efficient and recyclable hydrogenation catalyst, which operates efficiently at relatively low reaction temperature, thereby preventing high temperature-induced side reactions, would be highly desirable. To this aim, we propose a heterogeneous nitrogen-atom stabilizing support material that efficiently stabilizes Pt NPs, in which Pt–N interfacial interactions promote heterolytic hydrogen splitting, making it ideal for the hydrogenation of polar functional groups.24,25
2. Experimental
2.1 Materials
Mesitylene (98%, Merck®) was distilled over metallic Na and then stored under a dry argon atmosphere. The following reagents were purchased from Merck® and used as received without any further purification: ethanol (purity 99.0%), ammonia solution (25–28%), tetraethoxysilane (TEOS, purity 98.0%), (3-aminopropyl)triethoxysilane (APTES, purity 99.0%), dimethyl sulfoxide (purity 99.7%), terephthalaldehyde (TPA, purity 99.0%), cesium carbonate (purity 99.0%), tetrahydrofuran (THF, purity 99.9% inhibitor free), n-pentane (purity 99.0%), 2,3-BDO and PyA were purchased from Aldrich and used as received. Water used for catalytic reactions was bidistilled prior to use.2.2 Synthesis
2.3 Characterization
Inductively coupled plasma–optical emission spectroscopy (ICP-OES) analyses were carried out with an ICP-optical emission dual view PerkinElmer OPTIMA 8000 apparatus. The Pt/mes solution (0.5 mL) (MVS) was heated over a heating plate in a porcelain crucible to remove the solvent, and the solid residue was digested in aqua regia (2.0 mL) for 6 h. The latter solution was then diluted with ultrapure water (PureLab Pro, 18.2 MΩ cm). For the determination of the catalysts’ metal loading, the catalyst (10 mg) was treated with aqua regia (4 mL) and refluxed for 6 h. The suspension was then diluted with ultrapure water and centrifuged to remove the silica residue. Quantification of Pt was obtained by means of a calibration curve obtained with suitably diluted Pt stock solutions. The limit of detection (L.O.D.) calculated for platinum was 2 ppb. In all cases, the results of MVS solution and catalyst loading were highly consistent.Fourier transform infrared (FT-IR) spectra were recorded in ATR mode on sample powder using a Frontiers FTIR spectrometer (PerkinElmer, Milan, Italy), equipped with a diamond-attenuated total reflectance (ATR) sampling accessory. For each sample, three spectra were recorded in the 4000–600 cm−1 spectral range with a 4 cm−1 resolution, with 32 scans for the background and the sample.
Nitrogen physisorption isotherms were collected with a Micromeritics ASAP2020 instrument. Prior to the analysis carried out at 77 K, the sample was treated under high vacuum at 200 °C for 2 h.
Powder X-ray diffraction (PXRD) was carried out with a PANalytical powder diffractometer equipped with a copper anode (CuKα-radiation, λ = 1.541874 Å) and a PIXcel detector. Samples were prepared on a Si wafer (zero background) and spectra were acquired at 2Θ intervals from 10.0 to 80.0°, with a step size of 0.0263° and a counting time of 167.8 s.
Transmission Electron Microscopy (TEM) characterization was performed using a Talos™ F200X G2 TEM microscope (Thermo Scientific). Energy dispersive X-ray spectra (EDS) and elemental maps were collected along with HAADF-STEM (high angular annular dark field scanning transmission electron microscopy) micrographs using a four-detector Super-X Energy Dispersive system. Samples for analysis were ultrasonically dispersed in iso-propyl alcohol and a drop of the suspension was deposited on a holey carbon copper grid (300 mesh).
X-ray photoelectron spectroscopy (XPS) analyses were carried out in a UHV chamber (10−9/10−10 mbar) equipped with non-monochromatized Al radiation (hν = 1486.6 eV, VSW-TA1) combined with a hemispherical electron/ion energy analyzer (VSW-HA100 with a 16-channel detector). The operating power of the X-ray source was 144 W (12 kV and 12 mA), and photoelectrons were collected normal to the sample surface, maintaining the angle between the analyzer axis and the X-ray source fixed at 54.5°. All samples were adsorbed on carbon tape, and the XPS spectra were acquired in a fixed analyzer transmission mode with a pass energy of 44.0 eV. CasaXPS software was used to analyze the spectra and a linear or Shirley function was used for background correction. The deconvolution of XPS spectra has been performed by applying a combination of Gaussian and Lorentzian functions (70/30 ratio) for N 1s and C 1s and Lorentzian asymmetric lineshapes for Al 2p and Pt 4f. All the binding energies (B.E.) were calibrated upon fixing C 1s of the carbon tape at 285.1 eV.28
Thermogravimetric analyses (TGA) were carried out using a SEIKO SII TG/DTA 7200 EXSTAR instrument (Chiba, Japan). Samples with masses between 3 and 5 mg were heated from 30 to 900 °C under a nitrogen flow of 200 mL min−1, at a constant heating rate of 10 °C min−1. The rate inflection temperatures corresponding to the different degradation steps were determined from the first-derivative thermogravimetric (DTG) curves. The final residue was evaluated at 900 °C.
High performance liquid chromatography (HPLC) analyses were carried out on an HPLC apparatus (Shimadzu), equipped with an Aminex HPX-87H chromatographic column (300 × 7.8 mm) (BIO-RAD) and an RID 10A detector. All analyses were carried out using sulfuric acid (0.005 M) as the eluent and applying a flow rate of 0.4 mL min−1 and a column temperature of 35 °C. All organic compounds were quantified by means of calibration curves.
1H NMR spectra were recorded with a Bruker AVANCE 400 spectrometer at room temperature using an Evans tube filled with D2O.
Gas chromatography-mass spectrometry (GC-MS) was carried out with a GC-MS 2010SE apparatus (Shimadzu) equipped with a capillary column (VF WAXms, 30.0 m × 0.25 mm × 0.25 μm) and He as the carrier gas.
2.4 Catalysis
Catalytic hydrogenation reactions were conducted in a Teflon-lined stainless steel autoclave (70 mL) equipped with a magnetic stirrer, an electronic pressure controller and a thermocouple. The autoclave was heated by means of an external temperature-controlled oil bath. Typically, catalytic reactions were conducted as follows: the autoclave was charged with the catalyst (20 mg) and then sealed and evacuated. Afterwards, a deaerated water solution of the substrate (20 mL, 57.5 mM) was introduced into the autoclave by suction, followed by heating it to the desired temperature. Once the reaction temperature was reached, the autoclave was pressurized with hydrogen (25 bar) and magnetically stirred for the desired reaction time. The autoclave was then cooled to 10 °C by means of a water–ice bath, excess gas was vented off and the solid catalyst was separated from water solution by centrifugation. The clear water solution was analysed by HPLC, 1H NMR spectroscopy, GC-MS spectrometry and ICP-OES. Recycling experiments were carried out upon washing the separated catalyst with water, followed by centrifugation. The wet catalyst was then redispersed in water (20 mL), which contained the substrate (1.15 mmol). The obtained suspension was transferred to the evacuated autoclave by suction. The autoclave was then successively sealed, slowly flushed with hydrogen for 2 minutes, heated to the desired temperature and then pressurized with hydrogen (25 bar) followed by magnetic stirring of the reaction suspension. Catalysts 1 and 2, recovered and subjected to TEM, XPS and PXRD analyses were extensively rinsed with water and then vacuum-dried at 50 °C.3. Results and discussion
3.1 Synthesis and characterization of the materials
The synthesis approach of SiO2 spheres and CTF@SiO2 spheres is presented in Fig. 1. Following a previously reported procedure,27 uniform SiO2 spheres with diameters in the range of 240–260 nm were prepared. The SiO2 spheres were treated with (3-aminopropyl)triethoxysilane (APTES) and the amino groups present at their surface were further used to anchor terephthalaldehyde by a Schiff base reaction (see FT-IR spectra reported in Fig. S1). The aldehyde monomers were then reacted with 1,4-terephthalamidine dihydrochloride at the surface of the material and finally, both precursors were added simultaneously to obtain the final CTF@SiO2.Ultrafine Pt NPs were synthesized as ligand-free particles in their reduced form by the MVS approach19,29 and supported on either CTF@SiO2 or SiO2 to obtain composites 1 and 2, respectively, that were then stored at room temperature, under a dry air atmosphere. The quantitative deposition of Pt NPs on the supports was verified by ICP-OES analysis, which gave a final Pt content of 2.0 wt% for both 1 and 2.
Transmission electron microscopy analysis was performed to investigate the structural and morphological features of both the pristine supports CTF@SiO2 and SiO2 (Fig. 2(a)–(c), and Fig. S2, respectively) and of the corresponding derived catalysts 1 and 2 (Fig. 3 and S3, and Fig. S4, respectively). TEM analysis of the CTF@SiO2 and SiO2 support showed in both samples the presence of SiO2 spheres with a very homogeneous size distribution, ranging from 240 to 260 nm in diameter. In addition, CTF@SiO2 exhibited the additional presence of a rough organic shell (CTF) 25 to 30 nm thick covering the entire surface of the SiO2 spheres. HAADF-STEM/EDS element map analysis confirmed the presence of both carbon and nitrogen atoms at the outer shell of the CTF-SiO2 spheres. Interestingly, no formation of segregated organic aggregates outside of the SiO2 surface was observed. Moreover, FT-IR spectra of the CTF@SiO2 support (Fig. 2(d)), as well as of the SiO2 functionalization intermediates (i.e. SiO2 surface reaction with APTES and the further modification with TPA, Fig S4), confirmed that the growth of the organic framework successfully occurred at the surface of the silica spheres. The obtained CTF@SiO2 exhibited characteristic vibrations of the triazine units at 1523 and 1367 cm−1, ascribed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching vibrations that are identical to those observed for the bare CTF bulk material. Moreover, as expected, the Si–O stretching frequency at 1078 cm−1 was observed in the examined samples (Fig. 2(d) and S1).
N stretching vibrations that are identical to those observed for the bare CTF bulk material. Moreover, as expected, the Si–O stretching frequency at 1078 cm−1 was observed in the examined samples (Fig. 2(d) and S1).
To get further insight into the structural features of CTF@SiO2, X-ray Photoelectron Spectroscopy (XPS) analyses were carried out (Fig. 2e and S5). Survey spectra of the support showed the presence of the expected elemental composition (i.e. Si, C, N and O). High-resolution N 1s XPS spectra reported in Fig. 2(e) revealed the main component centred at a binding energy (B.E.) of 399.0 eV, which proves the presence of pyridinic nitrogen (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N−C) stemming from CTF triazine moieties. In addition, a second N 1s photoelectron contribution at B.E. of 400.7 eV and a shakeup component B.E. of 402.5 eV also confirmed the presence of oxidized nitrogen atoms in the CTF framework.30–32 The Si 2p photoelectron spectrum of both supports was characterized by a single contribution at a B.E. of ca. 103.7 eV, which corroborated the presence of SiO2.33 While bare SiO2 microspheres exhibited a specific surface area (SSA) of 16 m2 g−1, the presence of CTF on the surface of the SiO2 spheres (CTF@SiO2) significantly increased the SSA to 33 m2 g−1 (Table S2). Very low porosity was observed for both materials (Fig. S6) and the H3 type hysteresis loop indicates slit-shaped pores, which are typical of aggregates of plate-like particles.
N−C) stemming from CTF triazine moieties. In addition, a second N 1s photoelectron contribution at B.E. of 400.7 eV and a shakeup component B.E. of 402.5 eV also confirmed the presence of oxidized nitrogen atoms in the CTF framework.30–32 The Si 2p photoelectron spectrum of both supports was characterized by a single contribution at a B.E. of ca. 103.7 eV, which corroborated the presence of SiO2.33 While bare SiO2 microspheres exhibited a specific surface area (SSA) of 16 m2 g−1, the presence of CTF on the surface of the SiO2 spheres (CTF@SiO2) significantly increased the SSA to 33 m2 g−1 (Table S2). Very low porosity was observed for both materials (Fig. S6) and the H3 type hysteresis loop indicates slit-shaped pores, which are typical of aggregates of plate-like particles.
TEM analysis of both MVS-derived 1 (Fig. 3a and b, and Fig. S3) and 2 (Fig. S4) showed no appreciable alterations in the structural and morphological properties of the corresponding pristine supports. TEM analysis at high magnifications along with HAADF-STEM/EDS platinum maps showed the presence of highly dispersed Pt-NPs along the support surface, falling in the sub-nanometer range (<1.3 nm) for both 1 and 2 (Fig. 3(b), S3, and S4, respectively). Similar results were previously reported by depositing pre-formed MVS-derived Pt/mesitylene NPs on different carbon-based supports.19,29
The FT-IR spectrum of 1 (Fig. 3(c)) was very similar to that recorded for pristine CTF@SiO2, indicating the absence of any significant support modification during the immobilization of Pt NPs onto CTF-SiO2.
XPS analysis of 1 showed the presence of the expected elemental composition (i.e. Si, C, N, O and Pt) (Fig. S7) and the corresponding N 1s photoelectron spectrum (Fig. 2(e)) showed the same feature as that found for CFT@SiO2. In addition, the B.E. for the pyridinic nitrogen atom at a B.E. of 399.1 eV is almost identical to that found for CTF@SiO2 (399.0 eV), indicating that no Pt–N interaction was detected between the surface Pt and nitrogen atoms of CTF, likely due to the low Pt loading (2 wt% Pt, see section 2.2). Interestingly, the Si 2p photoelectron peak of 1 and 2 exhibited the same characteristic B.E. of 103.7 eV, but differed notably in intensity, which clearly suggests the presence of a CTF layer entirely covering the SiO2 nanospheres (Fig. S5). Pt 4f XPS spectrum shown for 1 in Fig. 3(d) contains two contributions that correspond to Pt(II), which is the major fraction (83.3 at%), at a B.E. of 72.3 eV (Pt 4f7/2) and Pt(0) at a B.E. of 71.1 eV (Pt 4f7/2).34,35 Interestingly, the Pt 4f photoelectron spectrum of 2 is characterized by the presence of a lower percentage of surface Pt(II) atoms (i.e. 68.5 at%, Table S1), which might indicate easier oxidation of surface Pt atoms in 1 due to electron donation originating from CTF nitrogen atoms, compared to 2 (i.e. Pt(II) 68.5 at%, Table S1, Fig. S9).
The thermal behaviour of 1 and of the corresponding support was investigated by TGA under a nitrogen atmosphere (Fig. 4).
The TGA curve of pristine silica (SiO2) showed a minor weight loss of about 6 wt% up to 900 °C mainly due to the desorption of physically adsorbed water below 150 °C (about 2%) and the condensation of surface hydroxyl groups above 200 °C.36 In contrast, silica coated with the covalent triazine-based framework (CTF@SiO2) showed an initial weight loss starting at around 200 °C and continuing up to 600 °C, followed by a second step between 600 and 900 °C, consistent with the decomposition of the organic framework as observed for the bulk CTF reference. The overall mass loss of this sample (≈18% at 900 °C) corresponds to a CTF content of about 20–25 wt%. 1 showed a similar decomposition profile to that of CTF@SiO2, with a slightly higher total weight loss (≈19% at 900 °C), confirming a comparable amount of CTF.
3.2 Catalysis in water
Catalysts 1 and 2 have been screened in water for the hydrogenation of 2,3-BDO and PyA by applying a substrate to a Pt molar ratio of 561. The reaction products were analyzed by means of HPLC, 1H NMR spectroscopy and GC-MS spectrometry. All obtained products were quantified by HPLC using calibration curves for each compound. All catalytic reactions were carried out twice and the reported results, compiled in Table 1, are average values. To avoid diffusion-controlled reactions, the stirring speed of the autoclave (i.e. magnetic stirring) was optimized to 1000 rpm. Also, the hydrogen pressure used in catalyst screening (i.e. from 5 to 30 bar) was optimized to 25 bar (Table 1). The relatively high hydrogen pressure needed to obtain a rational conversion of intermediate A into B and C is due to the low solubility of hydrogen in water.37 In contrast, the conversion of 2,3-BDO in intermediate A occurs at high conversion even at low hydrogen pressure (5 bar) (Table 1).| Entrya | Catalyst | t (h) | T (°C) | Conv. substrate (%) | Ab (%) | B (%) | C (%) [meso/rac] | D (%) | E (%) | F (%) | G (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | — | 1 | 85 | — | — | — | — | ||||
| 2 | 1 | 0.5 | 85 | 93.1 | 65.9 | 33.6 | 0.5 | ||||
| 3 | 1 | 1 | 85 | 96.5 | 52.2 | 45.9 | 1.9 | ||||
| 4c | 1 | 1 | 85 | — | — | — | — | ||||
| 5d | 1 | 1 | 85 | 95.0 | 75.2 | 24.8 | — | ||||
| 6e | 1 | 1 | 85 | 97.0 | 68.2 | 31.8 | — | ||||
| 7f | 1 | 1 | 85 | 98.0 | 60.0 | 39.0 | 1.0 | ||||
| 8g | 1 | 1 | 85 | 97.5 | 51.0 | 47.9 | 1.1 | ||||
| 9 | 1 | 3 | 85 | 100.0 | 35.2 | 62.6 | 2.2 | ||||
| 10 | 2 | 1 | 85 | 96.5 | 76.8 | 23.2 | - | ||||
| 11 | 2 | 3 | 85 | 99.5 | 44.5 | 55.5 | - | ||||
| 12 | 1 | 3 | 105 | 100.0 | — | 76.8 | 23.2 | ||||
| 13 | 1 | 15 | 105 | 100.0 | — | 0.9 | 99.1/[3/4] | ||||
| 14 | 2 | 3 | 105 | 100.0 | 2.1 | 95.5 | 2.4 | ||||
| 15 | 2 | 15 | 105 | 100.0 | — | 80.9 | 19.1/[3/4] | ||||
| 16h | 2 | 3 | 105 | 89.0 | 20.0 | 79.0 | 1.0 | ||||
| 17h | 1 | 3 | 105 | 100.0 | 77.0 | 23.0 | |||||
| 18i | 1 | 3 | 105 | 100.0 | 77.3 | 22.7 | |||||
| 19j | 1 | 3 | 105 | 100.0 | 77.8 | 22.2 | |||||
| a Catalytic conditions: catalyst (20.0 mg, 2.05 μmol of Pt), substrate (1.15 mmol), water (20.0 mL), p(H2) (25 bar). b (rac/meso ratio close to 1). c Without H2. d p(H2) (5 bar). e p(H2) (10 bar). f p(H2) (20 bar). g p(H2) (30 bar). h 1st recycling. i 2nd recycling. j 3rd recycling. | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 20 | — | 1 | 105 | — | — | — | — | — | — | — | — |
| 21c | 1 | 1 | 105 | — | — | — | — | — | |||
| 22f | 1 | 1 | 105 | 77.2 | 92.9 | 2.2 | 1.9 | 3.0 | |||
| 23 | 1 | 1 | 105 | 85.6 | — | — | — | 90.9 | 2.9 | 2.2 | 4.0 |
| 24g | 1 | 1 | 105 | 86.5 | 89.2 | 2.9 | 3.9 | 4.0 | |||
| 25 | 2 | 1 | 105 | 32.4 | — | — | — | 83.7 | 5.5 | 6.9 | 3.9 |
| 26 | 2 | 2 | 105 | 52.8 | 91.6 | 2.9 | 2.1 | 3.4 | |||
| 27h | 1 | 1 | 105 | 86.7 | 90.9 | 5.9 | 2.7 | 0.5 | |||
| 28i | 1 | 1 | 105 | 85.6 | 90.7 | 5.5 | 3.8 | — | |||
| 29j | 1 | 1 | 105 | 85.1 | 89.9 | 5.0 | 5.1 | — | |||
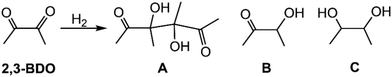
Irrespective of the catalyst used, the hydrogenation of 2,3-BDO was found to give three reaction products, namely 3,4-dihydroxy-3,4-dimethylhexa-2,5-dione (A), 3-hydroxy-butan-2-one (B) and butane-2,3-diol (C), whereas in the absence of the Pt-based catalyst, no substrate conversion occurred (Table 1, entry 1). Upon carrying out catalytic reactions with either catalyst at two different reaction temperatures (i.e. 85 and 105 °C) the stepwise hydrogenation reaction from 2,3-BDO to butane-2,3-diol was obvious.38–42 At 85 °C 2,3-BDO was converted first into dimer A, which was successively converted into B, as confirmed by catalytic reactions conducted at the latter temperature at different reaction times (Table 1, entries 2, 3 and 9). The chemical nature of dimer A has been proved by 1H and 13C{1H} NMR spectroscopy (Fig. S10 and S11) as well as by MS analysis (Fig. S12). This dimer formation contrasts with that reported for 2,3-BDO occurring in dichloromethane in the presence of Pt NPs decorated with the nitrogen atom containing ligands.12 The formation of compound A occurred by reductive coupling of two molecules of 2,3-BDO in the presence of NPs’ surface Pt(0) atoms, which act as a reducing agent, while water is needed to protonate the deprotonated diol intermediate, yielding A (Scheme S1).43,44 The experimental fact that in the absence of hydrogen, 2,3-BDO was not converted into A, whereas in the presence of a relatively low hydrogen pressure (5 bar) a high 2,3-BDO conversion into A (95%, Table 1, entry 5) was observed, indicates that hydrogen is needed to regenerate Pt(0) NPs’ surface atoms.
At 85 °C, only small amounts of C (2.2%) were obtained with the most active catalyst 1 after a reaction time of 3 h (Table 1, entry 9) (Fig. S13). Upon increasing the reaction temperature to 105 °C, a notable conversion of 2,3-BDO into C (23.2%) was achieved for 1 (Table 1, entry 12), while an almost quantitative formation of C (99.1%) was achieved after a reaction lasting 15 h (Table 1, entry 13) (Fig. S14 and S15). In contrast, 2 led to only 19.1% of C under these conditions (Table 1, entry 15 vs. 13). Irrespective of the catalyst, both stereoisomers of C (meso/rac) were obtained in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 molar ratio (Table 1).
4 molar ratio (Table 1).
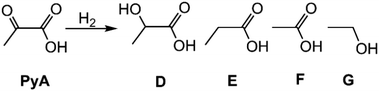
The hydrogenation reactions of PyA45,46 conducted in water at 105 °C and with a hydrogen pressure of 25 bar gave lactic acid (D) as the major compound, irrespective of the catalyst used (Fig. S16 and S17) (Table 1, entries 20–29). Along with lactic acid, propionic (E) and acetic acid (F), as well as trace amounts of ethanol (G) were formed when fresh catalysts were used. Propionic acid was obtained from lactic acid by a dehydration/hydrogenation step.47–51 Acetic acid and ethanol are mainly obtained upon decarboxylation of PyA.52,53 Analogously to the results obtained in the hydrogenation of 2,3-BDO, 1 showed significantly higher catalytic activity compared to 2 (Table 1, entry 23 vs. 25) along with a higher lactic acid selectivity (90.9 (1) vs. 83.7 (2)), due to the formation of lower amounts of propionic and acetic acid.
The significantly higher catalytic hydrogenation activity of 1 compared to 2 is the consequence of efficient surface Pt–N interactions between NPs’ surface Pt(0) atoms (Lewis acid sites) and support-nitrogen atoms (Lewis base sites). These Lewis acid–base couples foster the heterolytic hydrogen splitting, which leads to the formation of surface Pt-hydride species, which are the real catalytic species, operative in hydrogenation reactions when polar functional groups, such as C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds, are converted to the corresponding alcohol functionality.54
O bonds, are converted to the corresponding alcohol functionality.54
The stability of both catalysts during hydrogenation reactions in water was verified by carrying out recycling experiments, ICP-OES analyses of the reaction solution after hot filtration, and the structural characterization of the recovered catalysts. As a result, recycling experiments conducted with 1 and 2 under identical catalytic conditions proved that 1 gave almost identical substrate conversion for three consecutive catalytic reactions, using 2,3-BDO as the substrate (Table 1, entries 17–19), which is in stark contrast to the drop in catalytic activity found for 2, already after the first recycling experiment (Table 1, entry 16 vs. 14). In addition, in consecutive catalytic reactions using PyA as the substrate, 1 also showed an almost identical conversion with high lactic acid selectivity of roughly 90% maintained throughout four consecutive catalytic runs (Table 1, entries 23 vs. 27–29). ICP-OES analyses of the hot filtered solutions resulted in low leaching for both catalysts, which was close to the detection limit of the instrument (<2 ppb). TEM analyses of recovered 1 and 2 (i.e. after 15 h reaction time at 105 °C (1) and after 3 h reaction time at 105 °C (2)) are shown in Fig. 5a and b, respectively, and confirm the high stability of 1 against sintering (Pt NPs of the recovered catalyst showed a comparable size to the as-synthesized one). On the other hand, strong sintering of the Pt NPs was observed in the case of 2 (Fig. 5(b)). In agreement with the TEM results, PXRD spectra acquired for recovered 1 and 2 showed the presence of the characteristic diffraction peaks only in the case of 2 assigned to fcc Pt (i.e. Pt (111) at 39.6 (2Θ) and Pt (200) at 46.4 (2Θ)) (Fig. 5(c)), (Fig. 5c), trace d; the PXRD pattern of pristine Pt-NPs is shown in Fig. 5c, trace (e). This experimental result confirms strong sintering of Pt NPs in 2 (i.e. average NPs’ size of 2.8 nm has been determined by the Debye–Scherrer method46 based on the Pt(111) Bragg reflex). The TGA curve of recovered 1 (i.e. after four catalytic cycles) (Fig. S18) is almost identical to that of as-synthesized 1, confirming the high stability of the organic CTF phase under real catalytic conditions. The temperature corresponding to the maximum degradation rate (DTG peak) was observed at approximately 700 °C, which is comparable to that of pristine CTF, but shifted relative to CTF@SiO2, where the peak appeared at around 640 °C. The high stability of SiO2-supported CTF under the chosen catalytic conditions is further confirmed by the identity of the FT-IR spectra of recovered 1 (after 3 catalytic cycles) and as-synthesized 1 (Fig. 5(d)).
 | ||
| Fig. 5 Representative TEM micrograph of recovered 1 (after 15 h reaction time at 105 °C (a) and recovered 2 (after 3 h reaction time at 105 °C (b)); PXRD spectra acquired at room temperature for 1 and 2 (c)): as-synthesized 1 (a); recovered 1 (b); as-synthesized 2 (c), recovered 2 (d) and pristine Pt-NPs (e); and FT-IR analysis of as-synthesized 1 (green) and recovered 1 after three catalytic cycles (orange) (Table 1 entry 19). | ||
4. Conclusion
We showed that sub-nanometric-sized Pt NPs (<1.3 nm), synthesized by the MVS technique and supported onto a covalent triazine (CTF) framework, which entirely covers SiO2 nanospheres (1), were highly active in the hydrogenation of 2,3-BDO and PyA in water. Butane-2,3-diol and lactic acid were selectively obtained under relatively mild reaction conditions (i.e. T, 105 °C and p(H2), 25 bar). Related catalytic systems hydrogenate 2,3-BDO mainly to 3-hydroxy-2-butanone using ethanol, toluene, iso-propanol or dichloromethane as the reaction medium40–42 and PyA is hydrogenated in water to lactic acid using Ru-based catalysts.45,54 The crucial role of CTF in electron donation to surface Pt(0) centers, favouring the formation of Pt–H species, which are the dominant catalytic species for C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond hydrogenation and in stabilizing the Pt-NPs under real catalytic reaction conditions, has been proved by: (i) comparison of the catalytic activity of Pt NPs on SiO2 (2) (i.e. Pt NPs synthesized by the same method as 1, thus showing the same Pt dispersion on the support surface): catalyst 1 exhibited higher catalytic activity than 2, irrespective of the substrate, but also higher chemoselectivity mainly in the hydrogenation of PyA to lactic acid (Table 1, <90%); (ii) TEM and PXRD analyses conducted on recovered 1 and 2, which confirmed that in contrast to 1, 2 experienced a notable NP sintering even after relatively short reaction times (3 h); and (iii) recycling experiments conducted with 1, which exhibited almost identical catalytic performance in four successive catalytic reactions, regardless of the substrate used.
O bond hydrogenation and in stabilizing the Pt-NPs under real catalytic reaction conditions, has been proved by: (i) comparison of the catalytic activity of Pt NPs on SiO2 (2) (i.e. Pt NPs synthesized by the same method as 1, thus showing the same Pt dispersion on the support surface): catalyst 1 exhibited higher catalytic activity than 2, irrespective of the substrate, but also higher chemoselectivity mainly in the hydrogenation of PyA to lactic acid (Table 1, <90%); (ii) TEM and PXRD analyses conducted on recovered 1 and 2, which confirmed that in contrast to 1, 2 experienced a notable NP sintering even after relatively short reaction times (3 h); and (iii) recycling experiments conducted with 1, which exhibited almost identical catalytic performance in four successive catalytic reactions, regardless of the substrate used.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5dt02604k.Acknowledgements
The authors thank the Italian MUR through the PRIN2022 project “HYPOCOF: Hybrid Porous Materials for Eco-sustainable Catalytic Organic Processes” (20222H43S2; CUP B53C24005950006).References
- Y. Yusran, H. Li, X. Guan, Q. Fang and S. Qiu, EnergyChem, 2020, 2, 100035 CrossRef.
- H. El-Kaderi, J. R. Hunt, J. L. Mendoza-Cortez, A. P. Cote, R. E. Taylor, M. O'Keeffe and O. M. Yaghi, Science, 2007, 316, 268–272 CrossRef CAS PubMed.
- S. S. A. Shah, M. S. Javed, T. Najam, M. A. Nazir, A. ur Rehman, A. Rauf, M. Sohail, F. Verpoort and S. J. Bao, Mater. Today, 2023, 67, 229–255 Search PubMed.
- V. Sharma, M. Nemiwal and D. Kumar, Mini-Rev. Org. Chem., 2022, 19, 815–825 CrossRef CAS.
- N. Tahir, C. Krishnaraj, K. Leus and P. Van Der Voort, Polymers, 2019, 11, 1326 CrossRef CAS PubMed.
- M. Liu, L. Guo, S. Jin and B. Tan, J. Mater. Chem. A, 2019, 7, 5153–5172 RSC.
- P. Puthiaraj, Y. R. Lee, S. Zhang and W. S. Ahn, J. Mater. Chem. A, 2016, 4, 16288–16311 RSC.
- A. Imehoff, M. Vennewald and R. Palkovits, Angew. Chem., Int. Ed., 2023, 62, e202212015 Search PubMed.
- Y. Li, C. Lai, S. Liu, Y. Fu, L. Qin, M. Xu, D. Ma, X. Zhou, F. Xu, H. Liu, L. Li, Q. Sun and N. Wang, J. Mater. Chem. A, 2023, 11, 2070–2091 Search PubMed.
- D. Ma, X. Huo, C. Lai, H. Yi, L. Zeng, F. Xu, H. Yan, X. Zhou, X. Fan, L. Tang and M. Yan, Appl. Catal., B, 2026, 381, 125894 CrossRef CAS.
- D. Ma, C. Lai, H. Yi, X. Huo, L. Li, M. Zhang, F. Xu, H. Yan, S. Hu and Y. Luo, Coord. Chem. Rev., 2025, 522, 216241 CrossRef CAS.
- X. Zhu, F. Guo, Q. Yang, H. Mi, C. Yang and J. Qui, J. Power Sources, 2021, 506, 230224 Search PubMed.
- M. Gao, G. He, X. Long, S. Wang, Z. Guo, Z. Dong and K. Yuan, ACS Sustainable Chem. Eng., 2024, 12, 14732–14746 CrossRef CAS.
- T. He, L. Liu, G. Wu and P. Chen, J. Mater. Chem. A, 2015, 3, 16235–16241 RSC.
- J. Zhang, G. Zhang, L. He, Y. Shi, R. Miao, Y. Zhu and Q. Guan, Appl. Surf. Sci., 2021, 570, 150881 CrossRef CAS.
- L. Liao, M. Li, Y. Yin, J. Chen, Q. Zhong, R. Du, S. Liu, Y. He, W. Fu and F. Zheng, ACS Omega, 2023, 8, 4527–4542 CrossRef CAS PubMed.
- H. Vardhan, G. Rummer, A. Deng and S. Ma, Membranes, 2023, 13, 696 CrossRef CAS PubMed.
- E. Pitzalis, R. Psaro and C. Evangelisti, Inorg. Chim. Acta, 2022, 533, 120782 Search PubMed.
- X. T. Nguyen, E. Kitching, T. Slater, E. Pitzalis, J. Filippi, W. Oberhauser and C. Evangelisti, Catalysts, 2024, 14, 798 Search PubMed.
- Y. Bai, H. Feng, N. Liu and X. Zhao, Energies, 2023, 16, 5802 Search PubMed.
- M. Dusselier, P. Van Wouwe, A. Dewaele, E. Makshina and B. F. Sels, Energy Environ. Sci., 2013, 6, 1415–1442 RSC.
- J. A. Slipszenko, S. P. Griffiths, P. Johnston, K. E. Simons, W. A. H. Vermeer and P. B. Wells, J. Catal., 1998, 179, 267–276 Search PubMed.
- Y. Nagai, S. Morooka, N. Matsubayasi and M. Nakahara, J. Phys. Chem. A, 2004, 108, 11635–11643 Search PubMed.
- D. R. Aireddy and K. Ding, ACS Catal., 2022, 12, 4707–4723 Search PubMed.
- E. A. Redina, K. V. Vikanova, G. I. Kapustin, I. V. Mishin, O. P. Tkachenko and L. M. Kustov, Eur. J. Org. Chem., 2019, 4159–4170 CrossRef CAS.
- N. Wang, G. Cheng, L. Guo, B. Tan and S. Jin, Adv. Funct. Mater., 2019, 29, 1904781 Search PubMed.
- K. Wang, L.-M. Yang, X. Wang, L. Guo, G. Cheng, C. Zhang, S. Jin, B. Tan and A. Cooper, Angew. Chem., Int. Ed., 2017, 56, 14149 Search PubMed.
- A. J. Barlow, S. Popescu, K. Artyushkova, O. Scott, N. Sano, J. Hedley and P. J. Cumpson, Carbon, 2016, 107, 190–197 Search PubMed.
- W. Oberhauser, C. Evangelisti, R. P. Jumde, R. Psaro, F. Vizza, M. Bevilacqua, J. Filippi, B. F. Machado and P. Serp, J. Catal., 2015, 325, 111–117 CrossRef CAS.
- X. Zhu, C. Tian, S. M. Mahurin, S. Chai, C. Wang, S. Brown, G. M. Veith, H. Luo, H. Liu and S. Dai, J. Am. Chem. Soc., 2012, 134, 10478–10484 Search PubMed.
- Z. Luo, S. Lim, Z. Tian, J. Shang, L. Lai, B. MacDonald, C. Fu, Z. Shen, T. Yu and J. Lin, J. Mater. Chem., 2011, 21, 8038–8044 Search PubMed.
- A. Mohtasebi, T. Chowdhury, L. H. Hsu, M. C. Biesinger and P. Kruse, J. Phys. Chem. C, 2016, 120, 29248–29263 CrossRef CAS.
- A. Y. Lee, C. J. Powell, J. M. Gorham, A. Morey, J. H. J. Scott and R. J. Hanisch, Data Sci. J., 2024, 23, 45 Search PubMed.
- M. G. Bancroft, I. Adams, L. L. Coatswirth, D. C. Bennewitz, J. D. Brown and W. D. Westwood, Anal. Chem., 1975, 47, 586–588 CrossRef.
- W. Oberhauser, C. Evangelisti, X. T. Nguyen, J. Filippi, L. Poggini, L. Capozzoli, G. Manca, E. A. Kitching, T. J. A. Slater and M. Danale, Inorg. Chem., 2024, 63, 22912–22922 Search PubMed.
- F. Kunc, V. Balhara, Y. Sun, M. Daroszewska, Z. J. Jakubek, M. Hill, A. Brinkmann and L. J. Johnston, Analyst, 2019, 144, 5589–5599 RSC.
- Z. Zhu, Y. Cao, Z. Zheng and D. Chen, Energies, 2022, 15, 5021 CrossRef CAS.
- H. Duan, Y. Yamada and S. Sato, Catal. Commun., 2017, 99, 53–56 CrossRef CAS.
- N. Carrara, J. Badano, N. Bertero, G. Torres, C. Betti, L. Martinez-Bovier, M. Quiroga and C. Vera, J. Chem. Technol. Biotechnol., 2014, 89, 265–275 Search PubMed.
- N. Carrara, J. M. Badano, F. Coloma-Pascual, C. Vera and M. Quiroga, Chem. Pap., 2017, 71, 1669–1683 Search PubMed.
- R. P. K. Wells, N. R. McGuire, X. Li, R. L. Jenkins, P. J. Collier, R. Whyman and G. J. Hutchings, Phys. Chem. Chem. Phys., 2002, 4, 2839–2845 Search PubMed.
- M. Studer, V. Okafor and H.-U. Blaser, Chem. Commun., 1998, 1053–1054 RSC.
- Y.-S. Yang, Z.-L. Shen and T.-P. Loh, Org. Lett., 2009, 11, 2213–2215 Search PubMed.
- B. Cao, S. Li, W. Kong, J. Guo, Z. Tian and G. Zhang, Inorg. Chem. Commun., 2020, 121, 108227 Search PubMed.
- R. Luque and J. H. Clark, Catal. Commun., 2010, 11, 928–931 CrossRef CAS.
- G. Jones and S. J. Jenkins, J. Am. Chem. Soc., 2008, 130, 14483–14492 CrossRef CAS PubMed.
- X. Li, J. Pang, J. Zhang, C. Yin, W. Zou, C. Tang and L. Dong, Ind. Eng. Chem. Res., 2019, 58, 101–109 CrossRef CAS.
- X. Li, Z. Zhai, C. Tang, L. Sun, Y. Zhang and W. Bai, RSC Adv., 2016, 6, 62252–62262 Search PubMed.
- Z. Huo, J. Xiao, D. Ren, F. Jin, T. Wang and G. Yao, Green Chem., 2017, 19, 1308–1314 RSC.
- S. Liu, H. Feng, T. Li, Y. Wang, N. Rong and W. Yang, Green Chem., 2020, 22, 7468–7475 RSC.
- Y. Nagai, C. Wakai, N. Matubayasi and M. Nakahara, Chem. Lett., 2003, 32, 310–311 CrossRef CAS.
- D. R. Aireddy and K. Ding, ACS Catal., 2022, 12, 4707–4723 CrossRef CAS.
- U. Holzwarth and N. Gibson, Nat. Nanotechnol., 2011, 6, 534 Search PubMed.
- W. Oberhauser, L. Poggini, L. Capozzoli, M. V. Pagliaro, S. Coiai and C. Evangelisti, Inorg. Chem., 2025, 64, 14382–14394 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |