A Pd(II) catecholato complex bearing 5,5′-divinyl-2,2′-bipyridine: synthesis, characterization, and electrochemical disproportionation in solutions and electropolymerized films
Rizu
Tahara
a,
Kohei
Matsuura
b and
Keishiro
Tahara
 *bc
*bc
aGraduate School of Science for Creative Emergence, Kagawa University, 2217-20, Hayashi-cho, Takamatsu, Kagawa 761-0396, Japan
bFaculty of Engineering and Design, Kagawa University, 2217-20, Hayashi-cho, Takamatsu, Kagawa 761-0396, Japan. E-mail: tahara.keishiro@kagawa-u.ac.jp
cHealth and Medical Research Institute, National Institute of Advanced Industrial Science and Technology, 2217-14 Hayashi-cho, Takamatsu, Kagawa 761-0395, Japan
First published on 2nd February 2026
Abstract
A new Pd(II) catecholato (Cat) complex bearing 5,5′-divinyl-2,2′-bipyridine as an ancillary ligand (complex 2) was synthesized, and its electrochemical behavior in solutions and electropolymerized films was characterized. A comparison of 2 with an analog containing tBu groups (complex 1) revealed that the introduced vinyl groups positively shifted the reduction potential of the bipyridine (Bpy) moiety, in line with the lowered lowest unoccupied molecular orbital, thus redshifting the ligand-to-ligand charge transfer (LL'CT) transition absorption from the Cat to Bpy ligands across the Pd(II) center. Complex 2 exhibits a reversible redox wave for the semiquinone (SQ)/Cat couple in DMF/nBu4NPF6. On the other hand, the addition of Cl− substantially changed the voltametric properties, indicating disproportionation between the electrogenerated SQ complexes to the regenerated 2 and ligand dissociation products. In contrast to previous reports on the disproportionation of chemically isolated SQ complexes, we found electrochemical features of the repeated anodic oxidation of regenerated 2 followed by disproportionation. The electropolymerized films of 2 on indium tin oxide electrodes exhibited a widely separated pair of anodic and cathodic peaks, similar to those of monomer 2 in solution. The disproportionation of the constituent complexes in the metallopolymers was spectroelectrochemically monitored by the disappearance of the characteristic LL'CT absorption, indicative of ligand dissociation. These findings provide new insights into the fabrication of redox-active metallopolymer films and their applications, which exploit their electrochemical reactivity and bistability in molecular devices.
Introduction
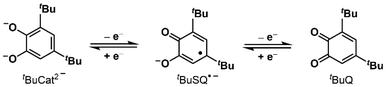
The chemistry of quinone and quinol pairs has continued to generate attention from the viewpoint of basic research and applications.1 In the case of 3,5-di-tert-butyl-1,2-benzoquinone (tBuQ), one-electron-reduction of tBuQ affords the semiquinone species (tBuSQ˙−), which is further converted into the catecholate species (tBuCat2−) (Scheme 1).2 Such two-electron redox processes in quinone/quinol pairs are coupled with protonation and deprotonation, resulting in diverse chemical species, proton-coupled electron-transfer phenomena,3 and mediator functions for electrical energy storage.4 Among quinol derivatives, catechol (CatH2) is the most investigated redox-active ligand5,6 for transition metal complexes. Catecholate (Cat2−), the deprotonated form of CatH2, coordinates with transition metal centers to afford metal catecholato (Cat) complexes. Cat complexes with redox-inactive metal centers form a certain group, such as those with group 10 d8 metal centers (i.e., Ni(II),7 Pd(II),8 and Pt(II)9). For example, a Pd(II) Cat complex with an ancillary bipyridine (Bpy) ligand 1 (Fig. 1) and its derivatives are recognized as important compounds because of their multifunctionality; they can undergo ambipolar (oxidative and reductive) electron transfer and exhibit visible-light absorption arising from the ligand-to-ligand charge transfer (LL'CT) transition from the Cat to Bpy ligands. Complex 1 is electrochemically converted to the semiquinonato (SQ) complex 1˙+via electrode reactions (eqn (1)) while the complex 1˙+PF6− can be isolated via chemical oxidation reactions with ferrocenium hexafluorophosphate (Fc+PF6−). In a notable example, Chang et al. reported that the bimolecular electron transfer of 1˙+ results in disproportionation to the neutral species (1), free ligand tBuQ, and solvated complex in some polar solvents (eqn (2)).10 This finding indicates the potential utility of the redox- and coordination-rich Pd(II) Cat complexes to acquire both Cat- and tBuQ-based forms as bistable molecular materials for the development of switching devices. However, despite the possibility of combining such behavior with above-mentioned multifunctionality, there have been no succeeding reports of the Pd(II) Cat complexes which deepens insight into the disproportionation reaction.| 1 − e− ⇄ 1˙+ | (1) |
| 1˙+ + 1˙+ + 2 L → 1 + [Pd(tBuBpy)(L)2]2+ + tBuQ | (2) |
The methodology of constructing metallopolymeric films allows the redox functions of transition metal complexes in homogeneous solutions to be installed onto electrode surfaces and provides redox functions with a unique stage of the electrode surface. Such a methodology is exemplified by layer-by-layer assembly,11 fabrication of metallosupramolecular polymers12 and coordination nanosheets,13 and electropolymerization.14 The resultant metallopolymeric films are useful in a wide range of applications, including electrochromism, which is the reversible color change of materials in response to electrochemical redox stimuli.11–14 In general, electrochromic metallopolymeric films utilize the electron-transfer reaction between the electrode and transition metal complex moieties, while the resulting reduced or oxidized state is maintained by holding the external electric stimulus. In recent years, bistable electrochromic organic materials have been reported, allowing the maintenance of each redox state without consuming electricity, assisted by proton transfer following electrode electron transfer.15 The basic investigation of the succeeding chemical reactions of transition metal complexes is advantageous for exploring bistable electrochromic systems with a mechanism other than that based on proton transfer.
In this study, we report the synthesis, characterization, and detailed electrochemical investigation of Pd(II) Cat complex 2 containing 5,5′-divinyl-2,2′-bipyridine (vinylBpy) as an ancillary ligand (Fig. 1). In our previous studies, we chemically modified the Pd(II) Cat complex at 4-position of Cat and developed covalent dimers and conjugates for varying intramolecular charge transfer absorption properties in the SQ state.16 Here, we introduce vinyl groups at the ancillary ligand moiety to perform reductive electropolymerization. The solution properties of monomer 2 were characterized using nuclear magnetic resonance (NMR) and UV-vis spectroscopy, electrochemical measurements, and density functional theory (DFT) calculations. In addition, we found the electrochemical conditions under which the electrogenerated SQ complex 2˙+ disproportionated. Furthermore, we report the preparation and characterization of electropolymerized films of 2 on indium tin oxide (ITO) electrodes, revealing the electrochemical reactivity of the Pd(II) Cat complex in vinyl polymers covalently connected at the ancillary ligand moieties.
Results and discussion
Synthesis and characterization of 2
Target product 2 was synthesized in two steps. A Pd(II) acetato complex with two vinyl groups was synthesized by the reaction of Pd(OAc)2 with vinylBpy (Fig. S1), and the resulting precursor complex, Pd(OAc)2(vinylBpy), was reacted with tBuCat in tetrahydrofuran (THF) to afford 2 in moderate yield. These synthesis procedures were the same as for the reference complex 1, except 4,4′-di-tert-butyl-2,2′-bipyridine (tBuBpy) was replaced with vinylBpy. The new complex 2 was characterized using 1H NMR spectroscopy (Fig. S2), and matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF-MS).DFT calculations were performed to optimize the structure by considering solvent effects (CH2Cl2) (Fig. S3 and Table S1). In the optimized structure of complex 2, the Pd(II) center adopts a distorted square-planar geometry with N1–Pd–N2 and O1–Pd–O2 angles of 79.36° and 84.39°, respectively, indicating that chelation leads to structural distortion. Similar distortions were also observed in the optimized structure of 1 (N1–Pd–N2 and O1–Pd–O2 angles of 80.90° and 87.48°, respectively) as well as in the previously reported crystal structures of 1 (N1–Pd–N2 and O1–Pd–O2 angles of 80.18° and 85.44°, respectively).10b
The effects of the vinyl groups attached at 5,5′-positions of bipyridine on frontier molecular orbital properties were investigated by DFT calculations. In 2, the lowest unoccupied molecular orbital (LUMO) was distributed mostly at the vinylBpy ligand (95%) and the highest occupied molecular orbital (HOMO) of the tBuCat ligand (92%) (Fig. 2 and Table 1). The contributions of the Pd center to the frontier orbitals were barely perceptible, confirming the non-innocent ligand properties of vinylBpy and tBuCat in 2. The fragmented contributions of the Bpy (tBuCat) ligands to the LUMOs (HOMOs) were comparable between 1 and 2. The replacement of the tBu groups with vinyl groups decreased the Bpy-based LUMO level by 0.45 eV. This decrease was accompanied by π-extension of Bpy, as indicated by the contribution of the vinyl groups to the LUMO at 2. By contrast, the difference in the tBuCat-based HOMO levels was small (0.05 eV) between 1 and 2. Accordingly, the HOMO–LUMO gaps decreased by 0.40 eV upon changing the tBu groups to vinyl groups in the Bpy ligand.
| Compound | MO | E/eV | Pd | Bpy ligand | t Bu2Cat |
|---|---|---|---|---|---|
| 1 | LUMO | −2.28 | 3 | 95 | 2 |
| HOMO | −4.35 | 5 | 2 | 92 | |
| 2 | LUMO | −2.73 | 2 | 95 | 2 |
| HOMO | −4.40 | 5 | 3 | 92 |
Using cyclic voltammetry (CV), the electrochemical behavior of 2 was investigated in CH2Cl2 containing nBu4NPF6 as the electrolyte at 298 K. On the cathodic side, 2 exhibited a reversible wave at a half-wave potential of Ered1/2 = −1671 mV vs. Fc+/Fc (Fig. 3a and Table 2), which was assigned to the reduction of the Bpy moiety. This assignment agreed well with the DFT calculations, which showed that the LUMO was distributed exclusively at the Bpy ligand (Fig. 2). For the reduction wave of the Bpy moiety, the low ratio of the cathodic peak current Ipc to the anodic peak current Ipa in magnitude (i.e., Ipa/Ipc = 0.487) indicated the high reactivity of the one-electron-reduction species 2˙− for electropolymerization as described later. A pre-peak at ca. −1432 mV vs. Fc+/Fc implied that some reduced species were involved in chemical reactions in the nonpolar medium. The half-wave potential for the first reduction Ered1/2 was 237 mV lower for 2 than that for 1. These results agree well with the decrease in the LUMO level accompanied by the π-extension of Bpy with vinyl groups, as discussed above.
| Compound | E red1/2 | (Ipa/Ipc) | E ox1/2 | (Ipc/Ipa) | Δ |
|---|---|---|---|---|---|
| a Potentials in mV vs. Fc+/Fc in CH2Cl2 (1.0 mM) containing 0.10 M nBu4NPF6. The ratios of absolute peak current values are also shown in parentheses. | |||||
| 1 | −1908 | (0.900) | −306 | (0.967) | 1602 |
| 2 | −1671 | (0.487) | −260 | (0.935) | 1412 |
On the anodic side, complex 2 exhibited a well-defined reversible wave at Eox1/2 = −254 mV vs. Fc+/Fc in CH2Cl2/nBu4NPF6 (Fig. 3a), assigned to the oxidation of the Cat moiety. This assignment agreed well with the DFT calculations, which showed that the HOMO was distributed exclusively on the Cat moiety (Fig. 2). For the oxidation wave of the Cat moiety, the peak current ratio Ipc/Ipa was 0.935, indicating that the one-electron oxidation species, 2˙+, was relatively stable in CH2Cl2/nBu4NPF6. In addition, the peak currents of 2 increased in proportion to the square root of the scan rate over a range of scan rates (10 to 1000 mV s−1), as shown in Fig. 3b and c. Thus, we confirmed that the anodic oxidation of 2 proceeds via the usual diffusion-controlled processes CH2Cl2/nBu4NPF6.
Complex 2 exhibited a visible absorption band at 652 nm, derived from the LL'CT transition from the Cat to Bpy ligands across the Pd(II) center in CH2Cl2, showing a blue color (Fig. 4a). Time-dependent DFT (TD-DFT) calculations predicted the lowest-energy transition from HOMO to LUMO with IEFPCM (CH2Cl2) (Table S2, #1). As described above, the HOMO is predominantly Cat-based (92%) whereas the LUMO is predominantly Bpy-based (95%). Thus, the TD-DFT-predicted transition can be assigned as having LL'CT character. The LL'CT transition absorption for 2 was 1727 cm−1 lower in energy than for 1 (586 nm), corresponding to the decrease of the HOMO–LUMO gaps upon changing the tBu to vinyl groups in the Bpy ligand. The LL'CT transition absorption for 2 showed negative solvatochromic behavior; the absorption maxima depended linearly on the solvent polarity parameters (Fig. 4b). The slopes of the line were almost comparable between 1 and 2, indicating that the attached vinyl groups did not have an impact on alternating the intrinsic solvation properties associated with the optical transition in the Pd(II) Cat complex system. To obtain insight into the solvent effects on the electronic structure of 2, additional DFT calculations were performed by considering effects of EtOH and toluene (Table S3). Upon change of the modeled solvents from toluene through CH2Cl2 to EtOH, the LUMO levels increased by 0.09 eV while the HOMO levels decreased by 0.34 eV, increasing the HOMO–LUMO gaps by 0.43 eV with increasing the solvent polarity. On the other hand, the frontier molecular orbital compositions of 2 remained almost unchanged upon change of the solvents. TD-DFT calculations reproduced the experimentally observed negative solvatochromic behavior of 2; the precited LL'CT lowest-energy transition energy increased with increasing the polarity of the model solvents (Table S2).
 | ||
| Fig. 4 (a) UV-vis spectra of 2 in several solvents (inset: photograph of solutions of 2). (b) Absorption maximum against the solvent polarity parameter for Pd complexes. | ||
Disproportionation of 2˙+ generated by anodic oxidation
Strikingly, drastic changes were observed in the anodic oxidation behavior of complex 2 when Cl− was added to the medium. In the absence of dodecyltrimethylammonium chloride (DTAC), 2 showed a reversible redox wave at Eox1/2 = −159 mV vs. Fc+/Fc in DMF/nBu4NPF6, which was assigned to the SQ/Cat couple (Fig. 5). The Ipc/Ipa value was 0.857, indicating the high stability of the SQ complex 2˙+ generated by anodic oxidation in DMF/nBu4NPF6. On the other hand, the Ipa values gradually increased upon increasing the DTAC concentration to 1.0 mM, reaching a 1.76-fold increase compared with the original value in the absence of DTAC (labelled I in Fig. 5). These changes were accompanied by a decrease in Ipc values, resulting in the complete disappearance of the reduction peak in the reverse scan. When keeping scanning to the more negative side, a small irreversible wave (labelled II) was observed at a cathodic peak potential of Epc = −966 mV vs. Fc+/Fc (Fig. 5). This Epc value was comparable with that for tBuQ (−963 mV vs. Fc+/Fc), which was independently determined in the reference experiment. For wave II with 2, the Ipc values gradually increased with increasing DTAC concentration. Importantly, the voltammograms were scan-path-dependent; no small reduction wave corresponding to wave II was observed when scanning only the negative side (Fig. S5). These results indicate that the electrochemical events corresponding to waves I and II were coupled. In the scan range, no redox waves were observed for DTAC (Fig. 5), ruling out the possibility of oxidation of Cl− during the electrochemical events. | ||
| Fig. 5 Cyclic voltammograms of (top) 2 (1.0 mM) upon the sequential addition of DTAC (0 mM to 1.0 mM) and (bottom) DTAC (1.0 mM) in DMF/nBu4NPF6. Scan rate: 100 mV s−1. | ||
These experimental results are attributed to the anodic oxidation of 2 (eqn (3)), followed by disproportionation of the electrogenerated SQ complex 2˙+ into 2, Pd(vinylBpy)(Cl)2, and tBuQ (eqn (4)).
| 2 − e− ⇄ 2˙+ | (3) |
| 2˙+ + 2˙+ + 2 Cl− → 2 + Pd(vinylBpy)(Cl)2 + tBuQ | (4) |
Wave II was assigned to the first reduction of tBuQ dissociated from the Pd center. These interpretations are consistent with the report by Chang et al. that isolated 1˙+PF6− disproportioned, as shown in eqn (2).10 Although they did not detail the role of Cl− on disproportionation monitored by the UV-vis spectral change of 1˙+PF6−, 1˙+PF6− disproportionated into a mixture of 1, Pd(tBuBpy)(Cl)2, and tBuQ in the presence of nBu4NCl in DMF-d7 as determined by 1H NMR spectroscopy. In our preliminary investigations of the reactivity of 2˙+ in homogeneous solutions, 2 and tBuQ were also detected from the disproportionation reaction solution with nBu4NCl by 1H NMR (Fig. S4). Under the present electrochemical conditions, the regenerated Cat complex 2 immediately underwent anodic oxidation to afford 2˙+ (eqn (3)), followed by disproportionation (eqn (4)). The series of reactions in eqn (3) and (4) were repeated until 2 was transported to the electrode via diffusion. The Ipa value of 2 increased by approximately twofold upon the addition of one equivalent of DTAC.
Preparation and characterization of electropolymerized films on Pt-disk electrodes
Reductive electropolymerization was performed on a solution of 2 in DMF/nBu4NPF6 using Pt-disk electrodes through repeated potential scanning (Fig. 6). For the first scan, the first and second reduction waves were observed at E1pc = −1031 mV and E2pc = −1589 mV vs. Ag/AgCl, respectively. In the second scan, the corresponding currents for both the reduction waves decreased significantly. Then, the current increased gradually and continuously at the more negative potential than E1pc of 2 in the first scan, resulting in the appearance of a new reduction peak with Ered11/2 = −1468 mV vs. Ag/AgCl. These results suggest that intermolecular radical–radical coupling proceeded at the vinyl groups, achieving the in situ deposition of polymeric films on the Pt electrode surface. The Ered11/2 value of the polymer is more negative than the E1pc value for the monomer. This negative shift was reasonable because the conversion of the vinyl groups to saturated butyl linkers increased the electron-donating properties of Bpy.Pt electrodes modified with a polymer composed of 2 (polymer 2) were used as the working electrodes in CV in CH2Cl2 containing nBu4NPF6. Polymer 2 on the Pt electrode retains the reversible SQ/Cat couple at Eox1/2 = 334 mV vs. Ag/AgCl (Fig. 7a). The voltammograms of the polymers were more symmetric than those of monomer 2; the potential difference between Epa and Epc was smaller for the polymers (58 mV) than for 2 (109 mV). These results indicate that electropolymerization of 2 accompanied by deposition on the Pt electrodes resulted in a surface-confined system.17 This was also confirmed by the peak currents of the polymeric film increasing in proportion to the scan rate, which was varied from 10 to 500 mV s−1 (Fig. 7a and b). The surface density of the constituent Cat complex was determined to be 9.3 × 10−11 mol cm−2 using the peak current values. This low density was improved when the electropolymerization of 2 was conducted on chemically pretreated ITO electrodes as described in the next section.
Preparation and characterization of electropolymerized films on ITO electrodes
The reductive electropolymerization of 2 was also performed in DMF/nBu4NPF6 with ITO electrodes. The initial trials resulted in brittle thin films that could easily be peeled off the ITO electrode. Referring to a study on the surface modification of TiO2 with a Ru complex,18 we pretreated the ITO electrodes with vinyltrimethoxysilane (Si(vinyl)(OMe)3) (Fig. 8a). Surface modification with silane coupling reagents was confirmed by a reduction in the wettability of the modified ITO electrodes, and the water contact angle increased from 14.8° to 56.8°. Through repeated potential scanning for a modified ITO electrode between 0 and −2000 mV vs. Ag/Ag+ (Fig. S6), the current increased gradually and continuously, similar to the scanning of the Pt-disk electrodes. A new reduction peak of Bpy was observed with Ered11/2 = −1314 mV vs. Ag/Ag+, which was also shifted to the negative side compared with the initial reduction peak for 2 in solution, indicating that the vinyl groups coupled to afford saturated butyl linkers. Robust ITO electrodes modified with polymer 2 were obtained by repeated potential scanning from 50 to 100 mV s−1. Under these typical preparation conditions, the electropolymerized thin film had a thickness of 50–60 μm and relatively low density, as confirmed by scanning electron microscopy (SEM) (Fig. 8b). The assembly of particular objects was observed on the thin film surface, resulting in high roughness. Because the polymers were deposited on Pt-disk electrodes without surface modification with a silane coupling reagent, the main mechanism of film formation was the electropolymerization of 2 followed by deposition on the ITO electrode surface. Some electrogenerated polymers were grafted onto the surface through coupling between the surface vinyl groups and the ends of the polymers (Fig. 8a), improving the contact between the substrate and thin film.The optical properties of the monomeric Cat complex were successfully transferred to ITO electrode surfaces. The electropolymerized film prepared under typical conditions exhibited a visible absorption at 546 nm, assignable to the LL'CT transition of the constituent Cat complexes (Fig. 8c), displaying a purple color. The LL'CT absorption bands of the electropolymerized film blueshifted relative to those of 2 in various solvents (584–746 nm). This blueshift for the polymeric film was rationalized by the elevation of the LUMO level of the Bpy-based accepting orbital in the LL'CT transition, agreeing well with the above-mentioned reduction potential shift to the negative side in the cyclic voltammogram.
Polymer 2 on ITO electrodes retained the reversible SQ/cat couple at Eox1/2 = 217 mV vs. Ag/AgCl during CV in CH2Cl2 containing nBu4NPF6 (Fig. 9a). For the modified ITO electrodes, the separation between the anodic and cathodic peaks was relatively large, in contrast to the more symmetric voltammograms of the modified Pt electrodes (Fig. 7a). In self-assembled monolayers containing Fc units on Au electrodes, a large separation was also observed between the anodic and cathodic peaks owing to the prevention of the diffusion of counterions by densely packed azobenzene units.19 For the modified ITO electrodes, polymer 2 was grafted from the electrode surface through the silane coupling reagent. Thus, the diffusion of the counteranion (PF6−) should be restricted for constituent Cat complexes within the proximity of the electrode, ultimately leading to the peak separation. The modified ITO electrodes represented the surface-confined system; the peak currents of the polymeric film increased in proportion to the scan rate, which was varied from 10 to 1000 mV s−1 (Fig. 9b). The surface density of the constituent Cat complex on ITO was determined to be 1.32 × 10−9 mol cm−2. This density was of the same order as those of previously reported metallopolymers prepared by reductive electropolymerization of Ru(III) polypyridyl complexes containing a vinylBpy ligand.20
The reversible absorption spectral change of polymer 2 on ITO was achieved in CH2Cl2/nBu4NPF6 (Fig. 9c). Upon applying a potential of 0.80 V vs. Ag/Ag+, a new absorption was observed at 427 nm, assignable to the π–π* transition absorption of the Bpy moiety in the constituent SQ complex, accompanied by the decrease in intensity of the LL'CT absorption band. Indeed, similar spectral properties were reported for the SQ complex 1˙+; the π–π* transition absorption of the Bpy moiety at 433 nm was strong in intensity while the LL'CT absorption at 560 nm was quite low.10 Upon switching the applied potential to −0.50 V vs. Ag/Ag+, the original spectral properties were recovered (Fig. 9c), indicating that the electrogenerated SQ complexes in the metallopolymers were converted into the Cat complexes by the cathodic reduction. The charge transport was supposed to proceed by self electron exchange between the proximal sites of the Cat and SQ complexes although the associated diffusion of PF6− should be somewhat restricted by the above-mentioned steric hindrance in the metallopolymers.
Disproportionation in electropolymerized films on ITO
Remarkable solvent effects were observed in the electrochemical behavior of polymer 2 on ITO. In DMF containing nBu4NPF6, the electropolymerized film on ITO exhibited an irreversible SQ/Cat couple with a high anodic peak current at Epa = 704 mV vs. Ag/Ag+ (labelled I′) (Fig. S7a). When scanning the more negative side, a small irreversible reduction wave with an onset potential of ca. −200 mV (labelled II′) was observed. These voltammetric properties, with a widely separated pair of anodic and cathodic peaks, closely resemble those of monomer 2 in the presence of DTAC (waves I and II in Fig. 5). These results suggest that the SQ complex electrogenerated in the polymerized film disproportionated. Wave II′ was assigned to the reduction of tBuQ dissociated from the polymeric chains. In the polymerized film, dissociated tBuQ diffused from the electrode surface into the bulk solution. Indeed, the reduction wave corresponding to wave II′ was not observed in the second scan for the polymeric film (Fig. S7a). The broad reduction waves on the more negative side were derived from the reduction of the Bpy moieties in the butyl-linked polymers composed of [Pd(Bpy)(L)2]2+ retained on ITO. This assignment agrees well with the fact that the monomeric [Pd(tBuBpy)(L)2]2+ species showed similar broad reduction waves in solution. In general, disproportionation is a bimolecular reaction and its reaction rate is highly dependent on the substrate concentration. The high local concentration of the constituent Cat complex in the electropolymerized film would assist the bimolecular electron transfer between the electrogenerated SQ complexes. In the independent CV experiment for polymer 2 on ITO in the presence of DTCA (1.0 mM), the reduction peak for wave I′ completely disappeared in the reverse scan (Fig. 10a), indicating that Cl− facilitated disproportionation in the film.To gain insight into the species formed during the anodic oxidation process, controlled potential electrolysis was performed for polymer 2 on ITO. Upon applying a potential of 1.3 V vs. Ag/Ag+ in the presence of DTAC (1.0 mM), the intensity of the LL'CT absorption band decreased (Fig. 10b). This spectral change indicated that the electrogenerated SQ complex was immediately converted into a species lacking visible absorption, confirming the above interpretation of the voltammetric results as the disproportionation of the SQ complex. Upon switching the potential to 0 V vs. Ag/Ag+, no further spectral changes were not observed, indicating an irreversible, one-directional transformation of the metallopolymers. Furthermore, even after the addition of tBuQ to the electrolyte solutions, controlled potential electrolysis at −0.8 V vs. Ag/Ag+ did not induce any spectral changes. This suggests that the reduced tBuSQ˙− did not coordinate to the [Pd(Bpy)(Cl)2]2+ polymer retained on the ITO electrode, and thus the original Cat complex could not be regenerated. The spectral change upon applying a potential of 1.3 V vs. Ag/Ag+ was slower in the absence of DTCA (Fig. S7b), confirming the enhancement of disproportionation by Cl− in line with above-mentioned CV results. This work demonstrates a unique alternation of absorption properties in metallopolymeric films under electrochemical conditions associated with ligand-dissociation-induced disappearance of the characteristic LL'CT absorption of the original complex. The resultant bistability of polymer (i.e., its capability to acquire both Cat and Cat-dissociated forms without external electricity or at low applied potentials) is a key prerequisite for write-once-read-many-times (WORM) memories.
The electropolymerization of transition metal complexes containing polymerizable organic fragments has been well studied,14 particularly Ru(III) polypyridyl complexes containing multiple vinyl groups.20 However, we are unaware of previous studies that applied electropolymerization to polypyridyl transition complexes of group 10 d8 metal centers containing vinyl grips. A key advantage of the Pd(II) Cat complex-based electropolymerized films presented here lies in the redox-dependent lability of the Pd center, which enables subsequent chemical transformations (i.e., disproportionation), within the polymer film. In the context of molecular assemblies of the Cat complexes of group 10 d8 metal centers, Chang and co-workers elaborately fabricated columnar liquid crystals of Pt(II) complexes responsible to redox-stimuli,21 as well as self-assembled monolayers of Pt(II) complexes on ITO electrodes through the phosphonate anchors of Cat ligands.22 Importantly, the significance of this work lies in combining multiple functionalities of a single metal complex to achieve electrochemical bistability. The ambipolar redox properties of the Pd(II) Cat complex enable both reductive electropolymerization and anodic oxidation of the constituent complexes, while the redox- and coordination-rich natures allow access to two distinct states that can be discriminated by the appearance and disappearance of ligand-based absorptions.
Conclusions
A new Pd(II) Cat complex containing a Bpy ligand bearing vinyl groups at 5,5′-positions (2) was synthesized and characterized using 1H NMR and UV-vis spectroscopies, MALDI-TOF-MS, and DFT calculations. A detailed comparison of 2 with an analog containing tBu groups (1) revealed that the introduced vinyl groups lowered the Bpy-based LUMO, positively shifted the reduction potential of the Bpy moiety, and redshifted the LL'CT transition absorption. Complex 2 exhibits a reversible redox wave for the SQ/Cat couple in DMF/nBu4NPF6. On the other hand, the addition of Cl− substantially changed the voltametric properties, indicative of the disproportionation of the electrogenerated SQ complex to the regenerated 2 and ligand dissociation products (Pd(vinylBpy)(Cl)2 and tBuQ). These results were consistent with previous reports on the disproportionation of the chemically isolated SQ complex by Chang and co-workers.10 In this study, we determined the electrochemical conditions that induce disproportionation. The regenerated neutral complex 2 was re-oxidized, repeating the anodic oxidation of 2 and subsequent disproportionation of the SQ complex. Electropolymerized films of 2 on ITO retained the absorption properties derived from monomer 2. Furthermore, the metallopolymers on ITO exhibit a widely separated pair of anodic and cathodic peaks, similar to those of monomer 2 in solution. The absorption spectral change of the metallopolymer films was captured using a spectroelectrochemical method, indicating disproportionation of the constituent complexes accompanied by ligand dissociation. These findings provide new insights into the fabrication of redox-active metallopolymer films and their applications, exploiting electrochemical reactivity involving dynamic alternations in coordination modes rather than being limited to simple electron or proton transfer in molecular devices.Experimental
Materials and general measurements
All solvents and chemicals used in the syntheses were of reagent grade and were used without further purification. 1 was synthesized according to a previously reported method.10b The 1H NMR spectra were recorded on a Bruker AVANCE NEO400N spectrometer; tetramethylsilane (TMS) was used as an internal standard (0 ppm). The UV-vis absorption spectra were measured on a JASCO V-670 spectrometers. The MALDI-TOF-MS was obtained by JEOL JMS-S3000 spectrometer. SEM images were obtained by a field-emission microscope (JEOL JSM-7001F) operated at an acceleration of 10 kV. Prior to the SEM measurements, the polymeric film on ITO was coated with Au by sputtering at low vacuum conditions.Synthesis of 2
To a CHCl3 solution (10 mL) of Pd(OAc)2 (108 mg, 0.480 mmol) a CHCl3 solution (4 mL) of vinylBpy (100 mg, 0.480 mmol) was added. The reaction mixture was stirred for 2 h at room temperature. The reaction mixture was evaporated and n-hexane was added to the residue. A crude product of Pd(OAc)2(vinylBpy) was obtained as a light yellow powder (208 mg) by recrystallization from CHCl3/ethyl acetate. 1H NMR (400 MHz, CDCl3, ppm) δ = 8.30 (d, 2H, J = 8.4 Hz), 8.12 (d, 2H, J = 1.9 Hz), 8.04 (dd, 2H, J = 2.0, 8.4 Hz), 6.59 (dd, 2H, J = 11.0, 17.6 Hz), 5.92 (d, 2H, J = 17.6 Hz), 5.60 (d, 2H, J = 11.0 Hz), 2.17 (s, 6H) (Fig. S1). To a dry THF solution (4.8 mL) of Pd(OAc)2(vinylBpy) a dry THF solution (5.4 mL) of 3,5-di-tert-butylcatechol (61.3 mg, 0.276 mmol) was added. The reaction mixture was stirred for 2 h at room temperature. The reaction mixture was evaporated and H2O was added to the residue. A dark green powder was collected as the target compound. Yield: 120 mg (81%). 1H NMR (400 MHz, CDCl3, ppm): δ = 1.31 (s, 9H), 1.54 (s, 9H), 5.60–5.64 (m, 2H), 5.98–6.07 (m, 2H), 6.48 (d, 1H, J = 2.04 Hz), 6.58 (d, 1H, J = 2.04 Hz), 6.78–6.65 (m, 2H), 7.77–7.83 (m, 2H), 7.87–7.90 (m, 2H), 8.71 (m, 2H), 8.91 (m, 2H) (Fig. S2). HR-MALDI-TOF-MS (m/z): calcd for C28H32N2O2Pd ([M]+), 533.1512; found for 533.1518.DFT calculation data
The DFT calculations were carried out using the Gaussian09 program package.23 The three-parameterized Becke–Lee–Yang–Parr (B3LYP) hybrid exchange–correlation functional24 was used with the Lanl2DZ (Hay–Wadt ECP) basis set25 for the Pd atom and the 6-31G(d) basis set26 for the other atoms with considering the solvent effects of CH2Cl2 using a polarizable continuum model with integral equation formalism (IEFPCM).27 The stability of the optimized structures was confirmed by calculating the molecular vibrational frequencies, in which no imaginary frequencies were observed. Molecular orbital composition analysis was conducted using the GaussSum Program. The TD-DFT method was used to calculate the transition energies and oscillator strengths for the lowest 30 singlet excited states.Electrochemical measurements
All voltammetric experiments were carried out using a BAS electrochemical analyzer (Bioanalytical Systems Inc., West Lafayette, IN, USA). All experiments were performed using a conventional three-electrode system at 298 K. A platinum wire (1.6 mm diameter) was employed as the counter electrode, and a glassy carbon electrode (3.0 mm diameter) as the working electrode. For the reference electrodes, an Ag/AgCl (3.0 M NaCl) electrode was used in CH2Cl2 containing nBu4NPF6 (0.1 M) while an Ag wire electrode in DMF containing nBu4NPF6 (0.1 M). Solutions containing 2 were deaerated prior to each measurement, and a N2 atmosphere was maintained inside the cell throughout each measurement. Each experiment was first performed in the absence of any internal standard and then repeated in the presence of Fc (ferrocene). The potentials are quoted relative to Fc+/Fc couple for solutions of monomer 2. In this setup, the Fc+/Fc couple in CH2Cl2/nBu4NPF6 were typically observed at 453 mV vs. Ag/AgCl while that in DMF/nBu4NPF6 at 577 mV vs. Ag/Ag+.ITO electrodes (5 × 40 × 1.1 mm or 7 × 50 × 1.1 mm) were cleaned by the UV ozone treatment prior to the electropolymerization of 2. An Ag/AgCl electrode was used for the measurements of the modified ITO electrodes in CH2Cl2/nBu4NPF6 while an Ag wire electrode for DMF/nBu4NPF6. The controlled potential electrolysis of the electropolymerized films on ITO electrodes was carried out in a quartz cell followed by UV-vis spectroscopy. The applied potential between the working and reference electrodes during the electrolysis was maintained constant with the BAS electrochemical analyzer.
Conflicts of interest
There are no conflicts to declare.Data availability
The additional data supporting this article have been included as part of the supplementary information (SI). Supplementary information: NMR spectra, DFT calculation data, and cyclic voltammograms. See DOI: https://doi.org/10.1039/d5dt03022f.Acknowledgements
This work was partially supported by JSPS KAKENHI JP24K01299 and the Shorai Foundation for Science & Technology. We would like to thank Ms. Ayami Nishioka for assisting SEM measurements.References
- (a) N. Gupta and H. Linschitz, J. Am. Chem. Soc., 1997, 119, 6384–6391 CrossRef CAS; (b) M. W. Lehmann and D. H. Evans, J. Electroanal. Chem., 2001, 500, 1–2 Search PubMed , 12–20.
- (a) M. W. Lehmann and D. H. Evans, J. Phys. Chem. B, 2001, 105, 8877–8884 CrossRef CAS; (b) D. H. Evans and A. René, Phys. Chem. Chem. Phys., 2012, 14, 4844–4848 RSC; (c) A. René and D. H. Evans, J. Phys. Chem. C, 2012, 116, 14454–14460 CrossRef.
- C. Costentin, M. Robert and J.-M. Savéant, Chem. Rev., 2010, 110, PR1–PR40 CrossRef PubMed.
- (a) L. Yan, X. Zeng, S. Zhao, W. Jiang, Z. Li, X. Gao, T. Liu, Z. Ji, T. Ma, M. Ling and C. Liang, ACS Appl. Mater. Interfaces, 2021, 13, 8353–8360 CrossRef CAS PubMed; (b) L. Yan, X. Zeng, Z. Li, X. Meng, D. Wei, T. Liu, M. Ling, Z. Lin and C. Liang, Mater. Today Energy, 2019, 13, 323–330 CrossRef; (c) N. Patil, A. Mavrandonakis, C. Jérôme, C. Detrembleur, J. Palma and R. Marcilla, ACS Appl. Energy Mater., 2019, 2, 3035–3041 CrossRef CAS.
- (a) W. Kaim, Inorg. Chem., 2011, 50, 9752–9765 CrossRef CAS PubMed; (b) V. Lyaskovskyy and B. de Bruin, ACS Catal., 2012, 2, 270–279 CrossRef CAS; (c) F. Thomas, Dalton Trans., 2016, 45, 10866–10877 RSC.
- (a) J. Yuasa, T. Suenobu and S. Fukuzumi, ChemPhysChem, 2006, 7, 942–954 CrossRef CAS PubMed; (b) D. Kumar, V. Gueskine, Z. Khan, R. Crispin and M. Vagin, ACS Omega, 2024, 9, 38498–38505 CrossRef CAS PubMed.
- For selected examples of Ni complexes, see: (a) W. W. Kramer, L. A. Cameron, R. A. Zarkesh, J. W. Ziller and A. F. Heyduk, Inorg. Chem., 2014, 53, 8825–8837 CrossRef CAS PubMed; (b) E. Seraya, Z. Luan, M. Law and A. F. Heyduk, Inorg. Chem., 2015, 54, 7571–7578 CrossRef CAS PubMed; (c) L. A. Cameron, J. W. Ziller and A. F. Heyduk, Chem. Sci., 2016, 7, 1807–1814 RSC.
- For selected examples of Pd complexes, see: (a) P. Ghosh, A. Begum, D. Herebian, E. Bothe, K. Hildenbrand, T. Weyhermüller and K. Wieghardt, Angew. Chem., Int. Ed., 2003, 42, 563–566 CrossRef CAS PubMed; (b) S. Kamath, V. Uma and T. S. Srivastava, Inorg. Chim. Acta, 1989, 166, 91–98 CrossRef CAS; (c) K. H. Puthraya and T. S. Srivastava, Polyhedron, 1985, 4, 1579–1584 CrossRef CAS; (d) S. G. Fox and R. D. Gillard, Polyhedron, 1988, 7, 349–1584 CrossRef CAS; (e) B. Sarkar, R. Hübner, R. Pattacini and I. Hartenbach, Dalton Trans., 2009, 4653–4655 Search PubMed; (f) N. Deibel, D. Schweinfurth, J. Fiedler, S. Záliš and B. Sarkar, Dalton Trans., 2011, 40, 9925–9934 RSC; (g) K. Tahara, Y. Ashihara, T. Higashino, Y. Ozawa, T. Kadoya, K. Sugimoto, A. Ueda, H. Mori and M. Abe, Dalton Trans., 2019, 48, 7367–7377 RSC.
- For selected examples of Pt complexes, see: (a) S. S. Kamath, V. Uma and T. S. Srivastava, Inorg. Chim. Acta, 1989, 166, 91–98 CrossRef CAS; (b) J. A. Weinstein, M. T. Tierney, E. S. Davies, K. Base, A. A. Robeiro and M. W. Grinstaff, Inorg. Chem., 2006, 45, 4544–4555 Search PubMed; (c) N. M. Shavaleev, E. S. Davies, H. Adams, J. Best and J. A. Weinstein, Inorg. Chem., 2008, 47, 1532–1547 Search PubMed; (d) H.-C. Chang, K. Komasaka, K. Kishida, T. Shiozaki, T. Ohmori, T. Matsumoto, A. Kobayashi, M. Kato and S. Kitagawa, Inorg. Chem., 2011, 50, 4279–4288 CrossRef CAS PubMed; (e) S. Yamada, T. Matsumoto, M. Wakizaka and H.-C. Chang, Dalton Trans., 2016, 45, 4974–4977 RSC; (f) J. Best, I. V. Sazanovich, H. Adams, R. D. Bennett, E. S. Davies, A. J. H. M. Meijer, M. Towrie, S. A. Tikhomirov, O. V. Bouganov, M. D. Ward and J. A. Weinstein, Inorg. Chem., 2010, 49, 10041–10056 CrossRef CAS PubMed; (g) K. Heinze and S. Reinhardt, Chem. – Eur. J., 2008, 14, 9482–9486 CrossRef CAS PubMed; (h) K. Heinze and S. Reinhardt, Organometallics, 2007, 26, 5406–5414 Search PubMed; (i) J. Moussa, L.-M. Chamoreau, A. D. Esposti, M. P. Gullo, A. Barbieri and H. Amouri, Inorg. Chem., 2014, 53, 6624–6633 CrossRef CAS PubMed; (j) K. Tahara, S. Akehi, T. Akita, S. Katao, J. Kikuchi and K. Tokunaga, Dalton Trans., 2015, 44, 14635–14645 Search PubMed; (k) K. Tahara, Y. Ashihara, T. Ikeda, T. Kadoya, J. Fujisawa, Y. Ozawa, H. Tajima, N. Toyoda, Y. Haruyama and M. Abe, Inorg. Chem., 2020, 59, 17945–17957 CrossRef CAS PubMed.
- (a) S. Yamada, T. Matsumoto, M. Wakizaka and H.-C. Chang, Dalton Trans., 2016, 45, 4974–4977 RSC; (b) S. Yamada, T. Matsumoto and H.-C. Chang, Chem. – Eur. J., 2019, 25, 8268–8278 CrossRef CAS PubMed.
- (a) R. Sakamoto, S. Katagiri, H. Maeda and H. Nishihara, Coord. Chem. Rev., 2013, 257, 1493–1506 CrossRef CAS; (b) G. de Ruiter, M. Lahav and M. E. van der Boom, Acc. Chem. Res., 2014, 47, 3407–3416 CrossRef CAS PubMed; (c) P. C. Mondal, V. Singh and M. Zharnikov, Acc. Chem. Res., 2017, 50, 2128–2138 CrossRef CAS PubMed; (d) T. Nagashima, H. Ozawa, T. Suzuki, T. Nakabayashi, K. Kanaizuka and M. Haga, Chem. – Eur. J., 2016, 22, 1658–1667 CrossRef CAS PubMed; (e) S. J. Higgins, R. J. Nichols, S. Martin, P. Cea, H. S. J. van der Zant, M. M. Richter and P. J. Low, Organometallics, 2011, 30, 7–12 Search PubMed; (f) D. Bu, Y. Xiong, Y. N. Tan, M. Meng, P. J. Low, D.-B. Kuang and C. Y. Liu, Chem. Sci., 2018, 9, 3438–3450 RSC.
- (a) F. S. Han, M. Higuchi and D. G. Kurth, J. Am. Chem. Soc., 2008, 130, 2073–2081 Search PubMed; (b) A. Wild, A. Winter, F. Schlütter and U. S. Schubert, Chem. Soc. Rev., 2011, 40, 1459–1511 Search PubMed; (c) A. Winter and U. S. Schubert, Chem. Soc. Rev., 2016, 45, 5311–5357 Search PubMed; (d) R. Banasz and M. Wałęsa-Chorab, Coord. Chem. Rev., 2019, 389, 1–18 CrossRef CAS; (e) M. K. Bera, S. Sarmah, D. C. Santra and M. Higuchi, Coord. Chem. Rev., 2024, 501, 215573 CrossRef CAS.
- (a) K. Takada, R. Sakamoto, S.-T. Yi, S. Katagiri, T. Kambe and H. Nishihara, J. Am. Chem. Soc., 2015, 137, 4681–4689 Search PubMed; (b) R. Sakamoto, K. Takada, X. Sun, T. Pal, T. Tsukamoto, E. J. H. Phua, A. Rapakousiou, K. Hoshiko and H. Nishihara, Coord. Chem. Rev., 2016, 320–321, 118–128 CrossRef CAS; (c) M. Wang, R. Dong and X. Feng, Chem. Soc. Rev., 2021, 50, 2764–2793 RSC; (d) M. K. Bera, S. Mohanty, S. S. Kashyap and S. Sarmah, Coord. Chem. Rev., 2022, 454, 214353 CrossRef CAS; (e) S. Roy, S. Halder and C. Chakraborty, Coord. Chem. Rev., 2024, 519, 216088 Search PubMed.
- (a) C. Friebe, M. D. Hager, A. Winter and U. S. Schuber, Adv. Mater., 2012, 24, 332–345 CrossRef CAS PubMed; (b) Y.-W. Zhong, C.-J. Yao and H.-J. Nie, Coord. Chem. Rev., 2013, 257, 1357–1372 Search PubMed; (c) Y. Liang, D. Strohecker, V. Lynch, B. J. Holliday and R. A. Jones, ACS Appl. Mater. Interfaces, 2016, 8, 34568–34580 Search PubMed.
- (a) Y. Wang, S. Wang, X. Wang, W. Zhang, W. Zheng, Y.-M. Zhang and S. X.-A. Zhang, Nat. Mater., 2019, 18, 1335–1342 CrossRef CAS PubMed; (b) Y. Wang, Y.-M. Zhang and S. X.-A. Zhang, Acc. Chem. Res., 2021, 54, 2216–2226 Search PubMed; (c) E. Zhang, L. Bai, Z. Chen, A. Zhang, Y. Tang, J. Ran, S. Wang, J. Wang, C. Jia and X. Guo, Precis. Chem., 2025, 3, 233–260 CrossRef CAS PubMed.
- (a) K. Tahara, T. Kadowaki, S. Akehi and J. Kikuchi, Bull. Chem. Soc. Jpn., 2016, 89, 1041–1047 Search PubMed; (b) K. Tahara, T. Kadowaki, J. Kikuchi, Y. Ozawa, S. Yoshimoto and M. Abe, Bull. Chem. Soc. Jpn., 2018, 91, 1630–1639 CrossRef CAS.
- (a) A. J. Bard and L. R. Faulkner, Electrochemical Methods: Fundamentals and Applications, John Wiley and Sons, New York, 2nd edn, 2000 Search PubMed; (b) T. Ikeda, K. Tahara, T. Kadoya, H. Tajima, N. Toyoda, S. Yasuno, Y. Ozawa and M. Abe, Langmuir, 2020, 36, 5809–5819 Search PubMed.
- L. Wu, M. K. Brennaman, A. Nayak, M. Eberhart, A. J. M. Miller and T. J. Meyer, ACS Cent. Sci., 2019, 5, 506–514 Search PubMed.
- T. Kondo, T. Kanai and K. Uosaki, Langmuir, 2001, 17, 6317–6324 CrossRef CAS.
- (a) H.-J. Nie, J.-Y. Shao, J. Wu, J. Yao and Y.-W. Zhong, Organometallics, 2012, 31, 6952–6959 Search PubMed; (b) H.-J. Nie and Y.-W. Zhong, Inorg. Chem., 2014, 53, 11316–11322 Search PubMed; (c) Y. Zhou, N. Shida, Y. Koizumi, K. Endo, I. Tomita and S. Inagi, Macromolecules, 2020, 53, 8123–8130 CrossRef CAS.
- H.-C. Chang, T. Shiozaki, A. Kamata, K. Kishida, T. Ohmori, D. Kiriya, T. Yamauchi, H. Furukawa and S. Kitagawa, J. Mater. Chem., 2007, 17, 4136–4138 Search PubMed.
- H. Honda, T. Matsumoto, R. Tamura, K. Kanaizuka, A. Kobayashi, M. Kato, M. Haga and H.-C. Chang, Chem. Lett., 2014, 43, 1189–1191 Search PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Gaussian, Inc., Wallingford, CT, 2009 Search PubMed.
- D. Becke, J. Chem. Phys., 1993, 98, 5648–5652 Search PubMed.
- P. Hay and W. R. Wadt, J. Chem. Phys., 1985, 82, 270–273 Search PubMed.
- P. C. Hariharan and J. A. Pople, Theor. Chim. Acta, 1973, 28, 213–222 CrossRef CAS.
- (a) B. Mennucci, E. Cancès and J. Tomasi, J. Phys. Chem. B, 1997, 101, 10506–10517 CrossRef CAS; (b) J. Tomasi, B. Mennucci and R. Cammi, Chem. Rev., 2005, 105, 2999–3093 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |