Hydroboration of carbonyl compounds catalysed by a molybdenum germylyne complex: metal–ligand cooperative mechanism
Christof
Fontanilla
a,
Yuto
Shimizu
b,
Koichi
Nagata
 a,
Sakura
Iwatsuki
a,
Aoi
Tanamura
a,
Seiji
Mori
a,
Sakura
Iwatsuki
a,
Aoi
Tanamura
a,
Seiji
Mori
 *b,
Tara Prasad
Dhungana
a and
Hisako
Hashimoto
*b,
Tara Prasad
Dhungana
a and
Hisako
Hashimoto
 *a
*a
aDepartment of Chemistry, Graduate School of Science, Tohoku University, 6-3 Aoba, Sendai, Miyagi 980-8578, Japan. E-mail: hisako.hashimoto.b7@tohoku.ac.jp
bInstitute of Quantum Beam Science, Graduate School of Science and Engineering, Ibaraki University, 2-1-1 Bunkyo, Mito, Ibaraki 310-8512, Japan
First published on 15th December 2025
Abstract
A molybdenum germylyne complex with a Mo![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Ge triple bond promotes the catalytic hydroboration of aldehydes and ketones with HBpin. Hydroboration of aldehydes proceeded efficiently at ambient temperature, affording the corresponding borate esters in 92–99% NMR yields, whereas hydroboration of ketones required a slight elevated temperature (70 °C), affording the products in 90–96% NMR yields. Mechanistic investigations, including KIE experiments, kinetic analyses and DFT calculations suggest a unique metal–ligand cooperative mechanism at the Mo
Ge triple bond promotes the catalytic hydroboration of aldehydes and ketones with HBpin. Hydroboration of aldehydes proceeded efficiently at ambient temperature, affording the corresponding borate esters in 92–99% NMR yields, whereas hydroboration of ketones required a slight elevated temperature (70 °C), affording the products in 90–96% NMR yields. Mechanistic investigations, including KIE experiments, kinetic analyses and DFT calculations suggest a unique metal–ligand cooperative mechanism at the Mo![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Ge bond that promotes hydroboration of substrates through a six-membered-ring transition state during the rate-determining step.
Ge bond that promotes hydroboration of substrates through a six-membered-ring transition state during the rate-determining step.
Introduction
Catalytic hydroboration of unsaturated compounds using boranes has emerged as a powerful synthetic strategy, enabling reductions without the need for stoichiometric metal hydride reagents such as NaBH4 or LiAlH4.1–6 The resulting organoboron products serve as versatile intermediates for a broad range of synthetic transformations. In particular, the catalytic hydroboration of carbonyl compounds has been established as an efficient route to functionalized alcohols, thereby stimulating the development of diverse molecular catalysts. Transition-metal-based systems represent a major class and continue to expand in scope, while increasing attention has recently been directed toward main-group catalysts, including those derived from s-block and p-block elements.1,4,7 Within this expanding landscape of catalyst design, mechanistic studies have also advanced, with notable contributions from frustrated Lewis pairs,8 bifunctional architectures and metal–ligand cooperative systems.9,10From the bifunctional and cooperative perspectives,11 low-valent main-group species, such as tetrylenes and tetrylene transition-metal complexes featuring divalent heavier group 14 elements, have attracted considerable attention as a new class of molecular activation platforms.7,11–13 Recent intensive studies have revealed their remarkable ability to activate small molecules, including σ-bonds and unsaturated bonds, which is ascribed to their low-coordinate and unique electronic structures (bearing both Lewis base and Lewis acid moieties), ultimately leading to the discovery of their catalytic performance. Tetrylene species7,13–17 such as germylene A,9a stannylene B![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16a and silylene C,17a illustrated in Fig. 1, have been reported to act as precatalysts or catalysts for the hydroboration of aldehydes and ketones. Although detailed mechanistic studies remain limited, tetrylene hydride species are widely regarded as key intermediates initiating carbonyl activation at the tetrel centre, whereas the germylene A system is proposed to operate via metal–ligand cooperation.
16a and silylene C,17a illustrated in Fig. 1, have been reported to act as precatalysts or catalysts for the hydroboration of aldehydes and ketones. Although detailed mechanistic studies remain limited, tetrylene hydride species are widely regarded as key intermediates initiating carbonyl activation at the tetrel centre, whereas the germylene A system is proposed to operate via metal–ligand cooperation.
 | ||
| Fig. 1 (a) Carbonyl hydroboration and (b) selected examples of catalysts involving low-valent group 14 species. | ||
In the case of tetrylene complexes, catalytic performance has also been demonstrated. For example, several silylene complexes featuring M![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Si bonds have been shown to catalyse the hydrosilylation of carbonyl compounds, as reported by Tilley et al. using iridium silylene complex D,18c and by Muraoka and Ueno's group using iron silylene complex E.19 In these cases, the electrophilic tetrel centre is also regarded to act as the initial activator of the carbonyls rather than the metal centre, with subsequent catalysis achieved through the cooperative action of both the metal and the tetrel centres, namely, metal–ligand cooperation is operative. In these systems, however, catalytic hydroboration reactions have not yet been reported.
Si bonds have been shown to catalyse the hydrosilylation of carbonyl compounds, as reported by Tilley et al. using iridium silylene complex D,18c and by Muraoka and Ueno's group using iron silylene complex E.19 In these cases, the electrophilic tetrel centre is also regarded to act as the initial activator of the carbonyls rather than the metal centre, with subsequent catalysis achieved through the cooperative action of both the metal and the tetrel centres, namely, metal–ligand cooperation is operative. In these systems, however, catalytic hydroboration reactions have not yet been reported.
Furthermore, monovalent tetrylyne complexes remain far less explored despite their distinct electronic structures and promising reactivity,20 and thus represent an emerging frontier in catalyst design. Recently, our group discovered that the germylyne complex Cp*(OC)2Mo![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) GeC(SiMe3)3 (1),21 featuring a monovalent germanium ligand, can catalyse the hydrosilylation of aldehydes and ketones.22,23 We further found that the same complex can catalyse hydroboration. Herein, we present experimental results of hydroboration reactions catalysed by the molybdenum germylyne complex 1, alongside mechanistic investigations combining kinetic experiments and DFT calculations. These studies elucidate the cooperative roles of the Mo and Ge centres in substrate activation and transformation, providing a comprehensive understanding of the reaction pathway in this new system.
GeC(SiMe3)3 (1),21 featuring a monovalent germanium ligand, can catalyse the hydrosilylation of aldehydes and ketones.22,23 We further found that the same complex can catalyse hydroboration. Herein, we present experimental results of hydroboration reactions catalysed by the molybdenum germylyne complex 1, alongside mechanistic investigations combining kinetic experiments and DFT calculations. These studies elucidate the cooperative roles of the Mo and Ge centres in substrate activation and transformation, providing a comprehensive understanding of the reaction pathway in this new system.
Results and discussion
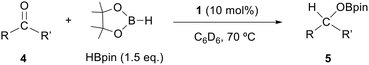
Catalytic hydroboration of aldehydes and ketones by complex 1
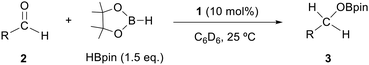
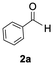
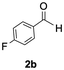
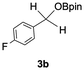
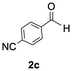
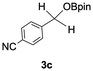
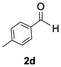
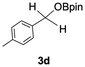
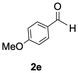
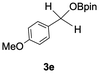
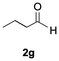
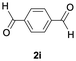
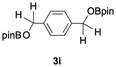
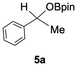
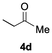
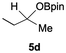
The catalytic activity of the molybdenum germylyne complex 1 was first examined in the hydroboration of benzaldehyde 2a with HBpin (pin = pinacolate). Prior to this, we benchmarked the transformation of 2a with HBpin under otherwise identical conditions, both in the absence and presence of complex 1, since aldehydes are known to undergo hydroboration with HBpin under neat conditions even in the absence of a catalyst.24 In the absence of the catalyst, the reaction proceeded only slightly (Fig. S1 in SI), consistent with the reported uncatalysed reactivity.24 When 2a (1.0 eq.) and HBpin (1.5 eq.) were treated with 10 mol% of complex 1 in toluene under an inert atmosphere at 25 °C, the corresponding hydroboration product PhCH2OBpin (3a) was obtained in 98% NMR yield (Table 1, entry 1). These results indicate that complex 1 serves as an effective catalyst for the hydroboration, significantly enhancing the reaction efficiency.To further explore the scope of this catalytic system, we next examined the hydroboration of various aldehydes under similar conditions (0.10 M solutions, 10 mol% 1, 1.5 eq. HBpin). The hydroboration products 3a–3i were identified by comparing their NMR data with previously reported 1H, 13C{1H} and 11B{1H} NMR spectra (see SI for details). As summarised in Table 1, the reaction proceeded at room temperature (25 °C) smoothly across a broad range of substrates, affording the corresponding hydroboration products in high yields. Both aryl-substituted (entries 1–5) and alkyl-substituted (entries 6–8) aldehydes were well tolerated, highlighting the generality of 1 as a catalyst. Notably, the catalyst was also competent in effecting double hydroboration of dialdehyde substrates (entry 9), further demonstrating its synthetic versatility. In the comparison of para-substituted arylaldehydes, all derivatives tested afforded the corresponding products in high yields (entries 1–5), regardless of whether electron-withdrawing or electron-donating groups were introduced. These results indicate that the effect of para-substituents is relatively minor under the present reaction conditions (vide infra). On the other hand, steric effects were observed for alkyl-substituted aldehydes. Aldehydes bearing linear or moderately branched alkyl groups (entries 7 and 8) reacted at significantly faster rates, whereas the presence of a bulky tert-butyl group (entry 6) led to a marked decrease in reactivity.
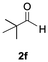
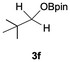
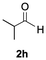
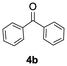
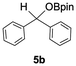
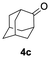
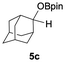
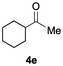
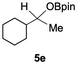
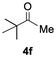
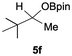
Having confirmed that complex 1 effectively catalyses the hydroboration of aldehydes, we next investigated its activity toward ketones (Table 2), using the same conditions as in Table 1 but at an elevated temperature of 70 °C. These ketones include monoaryl, diaryl, polycyclic, linear, monocyclic, and branched aliphatic groups; acetophenone 4a, benzophenone 4b, 2-adamantanone 4c, 2-butanone 4d, acetylcyclohexane 4e and pinacolone 4f. Hydroboration of these ketones afforded the corresponding products in 90–96% yields. Steric effects, as observed for aldehydes, strongly affect the reaction rate, as indicated by the comparisons between entries 1 and 2 and entries 5 and 6; bulkier substrates require much longer reaction times. This is likely due to steric hindrance from the bulky Tsi substituent on the germanium centre.
Compared to other catalytic hydroboration systems, complex 1 can still be regarded as a catalyst in its early stage of development. The TOFs (h−1) for aldehydes listed in Table 1 range from 0.4 to 2.0, whereas those for ketones in Table 2 range from 0.02 to 0.20. For example, in the hydroboration of benzaldehyde, the TOF for complex 1 is 0.5 h−1, which is much lower than that of stannylene B (198 h−1),16a but comparable to or slightly below that of germylene A (2.1 h−1)9a,25 and silylene C (3.3 h−1).17a Although its current performance is limited, complex 1 effectively promotes hydroboration across a broad range of carbonyl substrates, providing a basis for a new catalytic platform for this transformation.
KIE and kinetic studies
To gain mechanistic insights into the new catalytic system, we carried out isotope effect26 and kinetic studies.27 The kinetic isotope effect (KIE = kH/kD) was examined using benzaldehyde 2a with either HBpin or DBpin under the same conditions as in Table 1, with a carbonyl concentration of 0.05 M. A comparison of the initial rate plots (kD) for HBpin and DBpin (Fig. S3) indicates a slower reaction with the deuterated borane, giving an observed KIE of 1.8(1) (1.5 by DFT calculations, vide infra). This KIE value is interpreted as a primary isotope effect, albeit modest, suggesting that the B–H bond is substantially weakened in the rate-determining step. Notably, this value is close to the KIE of 1.62 ± 0.13 reported by Hartwig et al. for B–H bond activation of catecholborane (HBcat) by a ruthenium complex CpRu(PPh3)2(Me).26a In that system, the reaction is proposed to proceed via a four-centred transition state featuring a Ru–H–B(cat)–Me linkage.Kinetic studies were also performed using benzaldehyde 2a and HBpin under conditions similar to Table 1, with an aldehyde concentration of 0.05 M. Eyring plots were constructed based on the net initial rate constants, which were obtained by subtracting the contribution of the uncatalysed reaction background from the observed rates in the presence of 1 (see SI for details). Initial rate measurements were carried out at five temperatures ranging from 305 to 320 K. The Gibbs activation energy (ΔG‡) for 2a was determined to be 24.9(3) [kcal mol−1], consistent with the reaction proceeding at room temperature (Table 3). The enthalpy of activation (ΔH‡) was estimated to be 6.0(3) kcal mol−1. The large negative entropy of activation [ΔS‡ = −63(1) cal mol−1 K−1] is indicative of an associative mechanism, suggesting the formation of a highly ordered transition state (vide infra).
Possible pathways
Based on these experimental results and the fundamental reactivity of germylyne complexes, where the substrate heteroatom consistently interacts with the tetrel centre, previously investigated by us,28 we considered four possible pathways for this catalytic system (Fig. 2). The first pathway (1) involves CO dissociation from the 18-electron saturated complex 1, generating a 16-electron unsaturated species I, which could undergo B–H oxidative addition of HBpin or allow the aldehyde to coordinate to the metal centre. However, our previous calculations suggest that species II is significantly higher in energy (ΔG = 51.1 kcal mol−1) than complex 1 and is therefore thermally inaccessible.23 The second pathway (2) involves B–H oxidative addition and aldehyde coordination through the formation of metallogermylene species II. Our DFT calculations, however, did not identify such a species as either an intermediate or a transition state. Instead, species III, featuring a six-membered ring, was identified as a transition state, suggesting that pathway (3) proceeds via a concerted H-migration mechanism, which will be discussed in detail in the following section. This mechanism is in line with our recent proposal for the hydrosilylation mechanism catalysed by complex 1.23 We also examined a direct B–H addition pathway (4) involving species IV, which resembles mechanisms occasionally proposed for other systems involving 1,2-addition of B–H to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond.1,5,6 However, this route appears unlikely in this system because the calculated activation barrier is prohibitively high (39.3 kcal mol−1; see Fig. S53 in the SI). Furthermore, the conventional mechanisms, such as those involving a metal hydride species4 or a tetrylene hydride species7 as the active catalyst, are unlikely in this case, as no in situ formation of either species was detected. Based on these results, we focused our detailed analysis on pathway (3).
O bond.1,5,6 However, this route appears unlikely in this system because the calculated activation barrier is prohibitively high (39.3 kcal mol−1; see Fig. S53 in the SI). Furthermore, the conventional mechanisms, such as those involving a metal hydride species4 or a tetrylene hydride species7 as the active catalyst, are unlikely in this case, as no in situ formation of either species was detected. Based on these results, we focused our detailed analysis on pathway (3).
 | ||
| Fig. 2 Possible pathways for the catalytic mechanism via (1) CO dissociation pathway, (2) B–H oxidative addition pathway, (3), concerted H-migration and (4) 1,2-addition pathway. | ||
Computational studies
DFT calculations were carried out for the hydroboration of benzaldehyde 2a with HBpin as a representative case (Fig. 3, see also Table S3 in the SI). The optimised geometry of 1 was in good agreement with its crystallographically determined solid-state structure.21 At the initial stage, we hypothesised that a Lewis acid–base adduct forms between HBpin and the aldehyde,24 given that a measurable uncatalysed reaction occurs under these conditions.Computational analysis
Identified an initial state (denoted as a) in which the borane–aldehyde adduct is formed and weakly interacts with complex 1. Based on this model, the mechanistic study examined the Gibbs energy profile starting from state a, as shown in Fig. 3. The profile comprises two transition states (TS-1 and TS-2, see also Fig. 4). The activation barrier associated with TS-1 (ΔG‡298.15 K = 25.0 kcal mol−1) is higher than that of TS-2 (21.9 kcal mol−1), making TS-1 the rate-determining step. The calculated parameters for this step (ΔG‡298.15 K = 25.0 kcal mol−1, ΔH‡ = 6.7 kcal mol−1, ΔS‡ = −53.1 cal mol−1 K−1) are in very good agreement with the experimental values (ΔG‡298.15 K = 24.9(3) kcal mol−1, ΔH‡ = 6.0(3) kcal mol−1, ΔS‡ = −63(1) cal mol−1 K−1) (Table 1), supporting that the computed rate-determining step reliably reproduces the reaction energetics.In the initial state a, the Mo–Ge distance (2.30 Å) of a remains essentially unchanged compared to that in free catalyst 1 (2.29 Å), while the borane and the aldehyde are significantly interacting (B⋯O distance, 2.90 Å). From state a, the borane–aldehyde adduct dissociates to give b, where the electrophilic Ge(I) centre interacts with the aldehyde oxygen.29 This intermediate b represents shallow minima (ΔG = −8.3 kcal mol−1) between the state a and the subsequent state c. In b, the Ge⋯O distance in b (2.46 Å) is longer than typical Ge ← O coordination bonds (1.8–2.1 Å),30 indicating only weak coordination. Re-interaction of HBpin with the coordinated aldehyde in b gives c, where the borane hydride weakly interacts with the aldehyde carbon (H⋯C distance: 2.72 Å), accompanied by a slightly stronger Ge⋯O interaction (2.43 Å).
Next, a hydride migration occurs from the borane to the carbonyl carbon via transition state TS-1 (Fig. 4), producing intermediate d. This phase can be considered the rate-determining step, as TS-1 exhibits the highest activation barrier in the entire catalytic cycle. At TS-1, relative to intermediate c, the Mo–Ge bond elongates from 2.33 Å to 2.42 Å, which is close to the Mo![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge bond length [2.4224(9) Å] of the synthetic precursor of 1:21 and falls within the typical range of Mo
Ge bond length [2.4224(9) Å] of the synthetic precursor of 1:21 and falls within the typical range of Mo![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge double bonds (2.38–2.56 Å).31 The ∠(Mo–Ge–C(Tsi)) angle decreases only slightly from 155° to 151° by 4°. The resulting intermediate d exhibits a further weakened Mo
Ge double bonds (2.38–2.56 Å).31 The ∠(Mo–Ge–C(Tsi)) angle decreases only slightly from 155° to 151° by 4°. The resulting intermediate d exhibits a further weakened Mo![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge bond (2.47 Å) while remaining within the typical double-bond range, and a bent Mo–Ge–C angle of 145°. These structural changes indicate that, in TS-1, both the borane and the aldehyde are cooperatively activated through concurrent Mo–B and Ge–O bond formation, while the Mo–Ge multiple bond is preserved yet flexibly modulated to facilitate this unique three-component interaction. Notably, the persistence of Mo–Ge multiple bonding in TS-1 is further supported by molecular orbital analysis, revealing the presence of π-type orbitals (Fig. S52 in the SI). Intermediate d exists in a rapidly interconvertible conformation (d′), separated by only 0.2 kcal mol−1, which is attributable to rotation around the Ge–O bond. Subsequent B–O bond formation proceeds through TS-2, yielding the product dissociation state e. In this step, B–O bond formation is accompanied by cleavage of the Mo–B bond and weakening of the Ge–O interaction, ultimately restoring the Mo
Ge bond (2.47 Å) while remaining within the typical double-bond range, and a bent Mo–Ge–C angle of 145°. These structural changes indicate that, in TS-1, both the borane and the aldehyde are cooperatively activated through concurrent Mo–B and Ge–O bond formation, while the Mo–Ge multiple bond is preserved yet flexibly modulated to facilitate this unique three-component interaction. Notably, the persistence of Mo–Ge multiple bonding in TS-1 is further supported by molecular orbital analysis, revealing the presence of π-type orbitals (Fig. S52 in the SI). Intermediate d exists in a rapidly interconvertible conformation (d′), separated by only 0.2 kcal mol−1, which is attributable to rotation around the Ge–O bond. Subsequent B–O bond formation proceeds through TS-2, yielding the product dissociation state e. In this step, B–O bond formation is accompanied by cleavage of the Mo–B bond and weakening of the Ge–O interaction, ultimately restoring the Mo![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Ge bond (2.39 Å).
Ge bond (2.39 Å).
It should be noted here that the electron density in TS-1 is redistributed over the entire six-membered-ring framework, with negligible charge development on the aromatic ring (Ph), thereby minimizing the influence of para-substituents of aryl carbonyl substrates in this system, which may explain the experimental observation (vide supra). It should also be noted that the lower reactivity of aromatic aldehydes/ketones compared with aliphatic substrates has also been reported in other hydroboration systems.13,17d In our system, the rate-determining TS-1 involves a six-membered-ring transition state, which could be affected by the steric bulk of the aryl ring. Hydride transfer to the carbonyl carbon in TS-1 also appears less favourable for aryl substrates, due to π-conjugation between the aromatic ring with the carbonyl group. In contrast, smaller aliphatic aldehydes lack such π-stabilization and experience reduced steric congestion, which may contribute to their higher reactivity toward hydroboration.
Overall, the catalytic mechanism presented in Fig. 3 confirms a novel mode of metal–ligand cooperativity in 1: the transition metal activates the borane, while the Lewis acidic germylyne ligand activates the aldehyde. Importantly, this occurs without loss of the metal–ligand multiple-bond character. It should also be noted that Baik, Baker, and co-workers10a have recently proposed a relevant six-membered transition state for their Mn system bearing an SNS ligand. This activation is interpreted as metal-Lewis base cooperation (Fig. 5a), whereas our system operates through metal-Lewis acid cooperation (Fig. 5b).
 | ||
| Fig. 5 Two types of transition states for carbonyl hydroboration: (a) metal-Lewis base cooperation and (b) metal-Lewis acid cooperation. | ||
Conclusions
We have demonstrated that molybdenum–germanium triple-bonded complex 1 efficiently catalyses the hydroboration of aldehydes at room temperature and of ketones at elevated temperatures. The novel mode of metal–ligand cooperativity across the metal–germanium triple bond is deduced by kinetic experiments and KIE studies, supported by DFT calculations, further corroborating the proposed mechanism. This study represents a significant step toward further realising the catalytic potential of tetrylyne complexes and related polarized multiple-bonded systems.Author contributions
C. F. conducted experiments, performed theoretical studies and prepared the original manuscript. K. N. contributed to the theoretical studies and revision of the manuscript. S. I. and A. T. conducted kinetic experiments. Y. S. and S. M. performed theoretical studies. T. P. D. and S. I. synthesised the starting materials. H. H. conceived the idea of developing molybdenum germylyne complexes, directed the project, and finalised the manuscript. All authors contributed to the analysis and interpretation of the data and commented on the final draft of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article are included in the supplementary information (SI). Supplementary information: experimental procedures, characterisation data, kinetics, NMR spectra, DFT calculations, and atomic coordinates (xyz) for all optimised geometries. See DOI: https://doi.org/10.1039/d5dt02829a.Acknowledgements
This work was supported by JSPS KAKENHI grants JP_22H02088, 23K23356 (HH), 19H04687 and 21K14638 (KN) from the Japan Society for the Promotion of Science (JSPS). We would like to thank Prof. Yasuhiro Ohki, Mr Kanata Tanaka and Dr Hitoshi Izu of the Institute for Scientific Research, Kyoto University, for providing the DBpin reagent used in KIE experiments. Computation was performed using the Research Centre for Computational Science, Okazaki, Japan (project: 22-IMS-C102, 23-IMS-C089, 24-IMS-C041, 24-IMS-C084, 25-IMS-C042 and 25-IMS-C086).References
- M. L. Shegavi and S. K. Bose, Catal. Sci. Technol., 2019, 9, 3307–3336 RSC.
- K. Kuciński and G. Hreczycho, Green Chem., 2020, 22, 5210–5224 RSC.
- S. J. Geier, C. M. Vogels, J. A. Melanson and S. A. Westcott, Chem. Soc. Rev., 2022, 51, 8877–8922 RSC.
- C. C. Chong and R. Kinjo, ACS Catal., 2015, 5, 3238–3259 CrossRef CAS.
- S. Ramkumar, S. Reddappa, S. Mohan, K. Pramoda and S. K. Bose, ChemCatChem, 2025, 17(14), e00206 CrossRef CAS.
- S. R. Tamang and M. Findlater, Molecules, 2019, 24, 3194 CrossRef CAS PubMed.
- (a) S. K. Mandal and H. W. Roesky, Acc. Chem. Res., 2012, 45, 298–307 CrossRef CAS PubMed; (b) M. M. D. Roy, A. A. Omaña, A. S. S. Wilson, M. S. Hill, S. Aldridge and E. Rivard, Chem. Rev., 2021, 121, 12784–12965 CrossRef CAS PubMed.
- (a) J. Schneider, C. P. Sindlinger, S. M. Freitag, H. Schubert and L. Wesemann, Angew. Chem., Int. Ed., 2017, 56, 333–337 CrossRef CAS PubMed; (b) L. J. Bole, M. Uzelac, A. Hernán-Gómez, A. R. Kennedy, C. T. Óhara and E. Hevia, Inorg. Chem., 2021, 60, 13784–13796 CrossRef CAS PubMed.
- (a) Y. Wu, C. Shan, Y. Sun, P. Chen, J. Ying, J. Zhu, L. L. Liu and Y. Zhao, Chem. Commun., 2016, 52, 13799–13802 RSC; (b) V. A. Pollard, S. A. Orr, R. McLellan, A. R. Kennedy, E. Hevia and R. E. Mulvey, Chem. Commun., 2018, 54, 1233–1236 RSC.
- (a) M. R. Elsby, M. Son, C. Oh, J. Martin, M.-H. Baik and R. T. Baker, ACS Catal., 2021, 11, 9043–9051 CrossRef CAS; (b) S. Ataie, S. L. Dudra, E. R. Johnson and R. T. Baker, ACS Catal., 2023, 13, 10076–10084 CrossRef CAS; (c) C. Ekren, A. Kaithal, S. Sen, T. Weyhermüller, M. Hölscher, C. Werlé and W. Leitner, Nat. Chem., 2018, 9, 4521 Search PubMed.
- (a) J. R. Khusnutdinova and D. Milstein, Angew. Chem., Int. Ed., 2015, 54, 12236–12273 CrossRef CAS PubMed; (b) R. H. Morris, Acc. Chem. Res., 2015, 48, 1494–1502 CrossRef CAS PubMed; (c) T. Higashi, S. Kusumoto and K. Nozak, Chem. Rev., 2019, 119, 10393–10402 CrossRef CAS PubMed; (d) M. R. Elsby and R. T. Baker, Chem. Soc. Rev., 2020, 49, 8933–8987 RSC.
- (a) P. P. Power, Nature, 2010, 463, 171–177 CrossRef CAS PubMed; (b) Y. Mizuhata, T. Sasamori and N. Tokitoh, Chem. Rev., 2009, 109, 3479–3511 CrossRef CAS PubMed; (c) L. Wang, Y. Li, Z. Li and M. Kira, Coord. Chem. Rev., 2022, 457, 214413 CrossRef CAS.
- T. J. Hadlington, Chem. Soc. Rev., 2024, 53, 9738–9831 RSC.
- (a) M. Okazaki, H. Tobita and H. Ogino, Dalton Trans., 2003, 493–506 RSC; (b) R. Waterman, P. G. Hayes and T. D. Tilley, Acc. Chem. Res., 2007, 40, 712–721 CrossRef CAS PubMed; (c) L. Álvarez-Rodríguez, J. A. Cabeza, P. García-Álvarez and D. Polo, Coord. Chem. Rev., 2015, 300, 1–28 CrossRef; (d) R. J. Somerville and J. Campos, Eur. J. Inorg. Chem., 2021, 3488–3498 CrossRef CAS PubMed; (e) M. Ghosh, N. Sen and S. Khan, ACS Omega, 2022, 7, 6449–6454 CrossRef CAS PubMed.
- (a) T. J. Hadlington, M. Hermann, G. Frenking and C. Jones, J. Am. Chem. Soc., 2014, 136, 3028–3031 CrossRef CAS PubMed; (b) Y. Wu, C. Shan, Y. Sun, P. Chen, J. Ying, J. Zhu, L. L. Liu and Y. Zhao, Chem. Commun., 2016, 52, 13799–13802 RSC; (c) H. J. Robertson, M. N. Fujiwara and L. Liberman-Martin, Polyhedron, 2024, 250, 116837 CrossRef CAS; (d) A. P. Khuntia, N. Sarkar, A. G. Patro, R. K. Sahoo and S. Nembenna, Eur. J. Inorg. Chem., 2022, 20, e202200209 CrossRef; (e) M. M. D. Roy, S. Fujimori, M. J. Ferguson, R. McDonald, N. Tokitoh and E. Rivard, Chem. – Eur. J., 2018, 24, 14392–14399 CrossRef CAS PubMed; (f) S. Sinhababu, D. Singh, M. K. Sharma, R. K. Siwatch, P. Mahawar and S. Nagendran, Dalton Trans., 2019, 48, 4094–4100 RSC; (g) D. Sarkar, S. Dutta, C. Weetman, E. Schubert, D. Koley and S. Inoue, Chem. – Eur. J., 2021, 27, 13072–13078 CrossRef CAS PubMed; (h) K. V. Arsenyeva, K. I. Pashanova, O. Y. Trofimova, I. V. Ershova, M. G. Chegerev, A. A. Starikova, A. V. Cherkasov, M. A. Syroeshkin, A. Ya. Kozmenkova and A. V. Piskunov, New J. Chem., 2021, 45, 11758–11767 RSC.
- (a) K. Nakaya, S. Takahashi, A. Ishii, K. Boonpalit, P. Surawatanawong and N. Nakata, Dalton Trans., 2021, 50, 14810–14819 RSC; (b) R. Dasgupta, S. Das, S. Hiwase, S. K. Pati and S. Khan, Organometallics, 2019, 38, 1429–1435 CrossRef CAS; (c) Y. Liu, X. Liu, Y. Liu, W. Li, Y. Ding, M. Zhong, X. Ma and Z. Yang, Inorg. Chim. Acta, 2018, 471, 244–248 CrossRef CAS; (d) M. Zhong, Y. Ding, D. Jin, X. Ma, Y. Liu, B. Yan, Y. Yang, J. Peng and Z. Yang, Inorg. Chim. Acta, 2019, 486, 669–674 CrossRef CAS; (e) M. K. Sharma, M. Ansari, P. Mahawar, G. Rajaraman and S. Nagendran, Dalton Trans., 2019, 48, 664–672 RSC; (f) V. Nesterov, R. Baierl, F. Hanusch, A. E. Ferao and S. Inoue, J. Am. Chem. Soc., 2019, 141, 14576–14580 CrossRef CAS PubMed; (g) Y. Kobayashi, K. Nakaya, S. Takahashi, R. Watanabe and N. Nakata, Chem. – Eur. J., 2025, 31, e20250139 Search PubMed; (h) S. Pahar, V. Sharma, S. Tothadi and S. S. Sen, Dalton Trans., 2021, 50, 16678–16684 RSC.
- (a) J. Lee, J. Fan, A.-P. Koh, W.-J. J. Cheang, M.-C. Yang, M.-D. Su and C.-W. So, Eur. J. Inorg. Chem., 2022, 19, e202200129 CrossRef; (b) X. Qi, T. Zheng, J. Zhou, Y. Dong, X. Zuo, X. Li, H. Sun, O. Fuhr and D. Fenske, Organometallics, 2019, 38, 268–277 CrossRef CAS; (c) S. Khoo, J. Cao, F. Ng and C.-W. So, Inorg. Chem., 2018, 57, 12452–12455 CrossRef CAS PubMed; (d) L. Kapp, C. Wölper, H. Siera, G. Haberhauer and S. Schulz, Chem. Sci., 2024, 15, 4161–4170 RSC; (e) B.-X. Leong, J. Lee, Y. Li, M.-C. Yang, C.-K. Siu, M.-D. Su and C.-W. So, J. Am. Chem. Soc., 2019, 141, 17629–17636 CrossRef CAS PubMed; (f) G. Zhang, S. Li, H. Zeng, S. Zheng and M. C. Neary, RSC Adv., 2022, 12, 19086–19090 RSC; (g) K. Nakaya, S. Takahashi, A. Ishii and N. Nakata, Inorg. Chem., 2022, 61, 15510–15519 CrossRef CAS PubMed.
- (a) P. B. Glaser and T. D. Tilley, J. Am. Chem. Soc., 2003, 125, 13640–13641 CrossRef CAS PubMed; (b) E. Calimano and T. D. Tilley, J. Am. Chem. Soc., 2009, 131, 11161–11173 CrossRef CAS PubMed; (c) E. Calimano and T. D. Tilley, Organometallics, 2010, 29, 1680–1692 CrossRef CAS.
- T. Muraoka, Y. Shimizu, H. Kobayashi, K. Ueno and H. Ogino, Organometallics, 2010, 29, 5423–5426 CrossRef CAS.
- (a) H. Hashimoto and H. Tobita, Coord. Chem. Rev., 2018, 355, 362–379 CrossRef CAS; (b) H. Hashimoto and K. Nagata, Chem. Lett., 2021, 50, 778–787 CrossRef CAS; (c) L. R. Maurer, J. Rump and A. C. Filippou, Inorganics, 2023, 11, 129 CrossRef CAS; (d) N. Takagi, K. Yamazaki and S. Nagase, Bull. Korean Chem. Soc., 2003, 24, 832–836 CrossRef CAS; (e) K. K. Pandey, P. Patidar and P. P. Power, Inorg. Chem., 2011, 50, 7080–7089 CrossRef CAS PubMed.
- T. P. Dhungana, H. Hashimoto, M. Ray and H. Tobita, Organometallics, 2020, 39, 4350–4361 CrossRef CAS.
-
T. P. Dhungana, H. Hashimoto, K. Nagata and H. Tobita, Catalytic Hydrosilylation of Carbonyl Compounds by a Germylyne Complex with a Mo
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Ge Triple Bond, ChemRxiv, 2025, preprint, DOI:10.26434/chemrxiv-2025-1hc87, https://chemrxiv.org/engage/chemrxiv/article-details/6901bf08a482cba122a3eab5.
Ge Triple Bond, ChemRxiv, 2025, preprint, DOI:10.26434/chemrxiv-2025-1hc87, https://chemrxiv.org/engage/chemrxiv/article-details/6901bf08a482cba122a3eab5. - T. P. Dhungana, N. Takagi, B. Zhu, K. Nagata, S. Sakaki, H. Tobita and H. Hashimoto, Mechanistic Insights into Catalytic Hydrosilylation of Carbonyl Compounds versus [2 + 2] Cycloaddition Pathway Mediated by Germylyne Complexes, ChemRxiv, 2025, preprint, DOI:10.26434/chemrxiv-2025-8tqj5, https://chemrxiv.org/engage/chemrxiv/article-details/690c0d51113cc7cfff6c10ff.
- H. Stachowiak, J. Kaźmierczak, K. Kuciński and G. Hreczycho, Green Chem., 2018, 20, 1738–1742 RSC.
- The TOF of 2.1 h−1 was estimated by us using data reported in the literature (ref. 9a).
- (a) J. F. Hartwig, S. Bhandari and P. R. Rablen, J. Am. Chem. Soc., 1994, 116, 1839–1844 CrossRef CAS; (b) X. Gao, X.-Y. Yu and C.-R. Chang, Phys. Chem. Chem. Phys., 2022, 24, 15182–15194 RSC; (c) E. M. Simmons and J. F. Hartwig, Angew. Chem., Int. Ed., 2012, 51, 3066–3072 CrossRef CAS PubMed; (d) M. Gómez-Gallego and M. A. Sierra, Chem. Rev., 2011, 111, 4857–4963 CrossRef PubMed.
- A. S. Chang, K. E. Kawamura, H. S. Henness, V. M. Salpino, J. C. Greene, L. N. Zakharov and A. K. Cook, ACS Catal., 2022, 12, 11002–11014 CrossRef CAS.
- (a) H. Hashimoto, T. Fukuda, H. Tobita, M. Ray and S. Sakaki, Angew. Chem., Int. Ed., 2012, 51, 2930–2933 CrossRef CAS PubMed; (b) T. Fukuda, H. Hashimoto and H. Tobita, Chem. Commun., 2013, 49, 4232–4234 RSC; (c) T. Fukuda, H. Hashimoto and H. Tobita, J. Am. Chem. Soc., 2014, 136, 80–83 CrossRef CAS PubMed; (d) T. Yoshimoto, H. Hashimoto, N. Hayakawa, T. Matsuo and H. Tobita, Organometallics, 2016, 35, 3444–3447 CrossRef CAS; (e) T. Yoshimoto, H. Hashimoto, N. Takagi, S. Sakaki, N. Hayakawa, T. Matsuo and H. Tobita, Chem. – Eur. J., 2019, 25, 3795–3798 CrossRef CAS PubMed; (f) K. Nagata, H. Omura and H. Hashimoto, Chem. – Asian J., 2023, 18, e202300801 CrossRef CAS PubMed.
- The NPA charges (Mo: −1.44; Ge: +1.35) indicate that the Ge atom in complex 1 bears a significant positive charge, consistent with its role as the primary Lewis acidic site in the cooperative activation of HBpin and carbonyl substrates (Table S4).
- M. Iglesias, F. J. Fernández-Alvarez and L. A. Oro, Coord. Chem. Rev., 2019, 386, 240–266 CrossRef CAS.
- Based on a survey of the Cambridge Structural Database and CSD version 5.46 (November 2024).
| This journal is © The Royal Society of Chemistry 2026 |