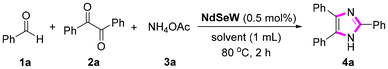A one-dimensional selenotungstate based on neodymium and {SeO3} groups for catalytic synthesis of imidazoles
Hao-Zhe
Wang
a,
Yu-Feng
Liu
*a,
Zhou-Fu
Lin
a,
Shi-Xiong
Li
 *b and
Guo-Ping
Yang
*b and
Guo-Ping
Yang
 *a
*a
aJiangxi Province Key Laboratory of Functional Organic Polymers, Jiangxi Key Laboratory for Mass Spectrometry and Instrumentation, East China University of Technology, Nanchang 330013, China. E-mail: erick@ecut.edu.cn; yfliu@ecut.edu.cn
bSchool of Mechanical and Resource Engineering, Wuzhou University, Wuzhou, Guangxi 543003, P. R. China. E-mail: lsx1324@163.com
First published on 5th January 2026
Abstract
A novel neodymium-{SeO3}-bridged selenotungstate, [Na16Nd2.5(SeO3)(SeO3)(mal)(W4O9)(SeW8O32)(SeW9O33)2(H2O)4]·ca.26H2O (NdSeW, mal = malic acid), has been successfully synthesized and structurally characterized. This compound is constructed from both {SeW9} and {SeW8} building units, which are linked through an organometallic {Nd2Se2(mal)W4} cluster, forming a distinctive triangular arrangement. Two triangular trimers are connected via Nd–O–W to form a hexameric unit, which is subsequently extended into a one-dimensional chain structure through bridging by two {SeO3} groups. This type of one-dimensional chain architecture, constructed through alternating connections involving neodymium and {SeO3} groups, is extremely rare in polyoxometalate chemistry. In addition, NdSeW exhibits excellent catalytic activity for the synthesis of 2,4,5-trisubstituted imidazoles through the three-component condensation of aldehydes, benzils, and ammonium acetate under environmentally benign conditions.
Introduction
Polyoxometalates (POMs) represent a class of nanoscale metal–oxygen clusters with diverse structural architectures and functional properties, garnering significant interest in the fields of materials science and catalysis.1–9 These clusters are predominantly composed of early transition metals such as tungsten, molybdenum, and vanadium in high oxidation states and exhibit remarkable versatility due to their tunable acidity, redox behavior, and ability to incorporate various heteroatoms and metal cations.10–14 Among these, lanthanide-containing POMs (Ln-POMs) have emerged as promising materials owing to the unique coordination flexibility and Lewis acidity of lanthanide ions, which facilitate the construction of novel structures and enhance catalytic performance.6,15–17The incorporation of selenium as a heteroatom in POM chemistry has enabled significant advances in structural diversification. Selenium functions not only as a templating agent but also as a bridging unit, facilitating the construction of novel architectures that are uncommon in conventional POM systems.18–21 Notably, lanthanide-functionalized selenotungstates provide a versatile platform for designing multifunctional materials with promising applications in catalysis, sensing, and molecular magnetism.22–24
Despite significant advancements, the integration of multiple building blocks within a single POM framework remains challenging due to variations in stability and reactivity among precursor fragments.25–27 Utilizing lanthanide ions as structural linkers provides a viable strategy to mitigate these limitations, enabling the assembly of complex structures incorporating heterogeneous building units.13,20 In this context, neodymium, characterized by its high coordination number and pronounced Lewis acidity, represents an interesting candidate for constructing novel POMs.
Herein, we report the synthesis and structural characterization of a novel neodymium–{SeO3}-bridged selenotungstate, [Na16Nd2.5(SeO3)(SeO3)(mal)(W4O9)(SeW8O32)(SeW9O33)2(H2O)4]·ca.26H2O (NdSeW), which incorporates both {SeW9} and {SeW8} building units stabilized by an organometallic {Nd2Se2(mal)W4} cluster. This compound features a unique triangular arrangement and further self-assembles into a one-dimensional chain through Nd–O–W and {SeO3} linkages. Moreover, NdSeW exhibits excellent catalytic performance in the synthesis of 2,4,5-trisubstituted imidazoles through the three-component condensation of aldehydes, benzils, and ammonium acetate. This work not only expands the structural diversity of Ln-containing selenotungstates but also highlights their potential as efficient catalysts under mild and environmentally benign conditions.
Results and discussion
Crystal structural description of NdSeW
NdSeW crystallizes in the monoclinic space group C2/c (Table S1) and its molecular skeleton comprises a trimeric polyoxoanion [Nd2.5(SeO3)(SeO3)(mal)(W4O9)(SeW8O32) (SeW9O33)2(H2O)4]16−. Bond-valence-sum (BVS) analysis indicated that all Nd and W atoms exhibit oxidation states of +3 and +6, respectively (Table S2). As exhibited in Fig. 1a, the structure of compound NdSeW features one {Nd0.5SeW8} building unit capping a dimeric {Nd2Se2(mal)W4(SeW9)2} fragment (Fig. 1a and b). This configuration arises through substitution of a tungsten position in the {SeW9} unit by an Nd3 atom, generating the {Nd0.5SeW8} cluster (Fig. 1h). Concurrently, the organometallic linker cluster {Nd2Se2(mal)W4} bridges two {SeW9} units, thus constituting the {Nd2Se2(mal)W4(SeW9)2} framework (Fig. 1b–d). NdSeW also displays a triangular arrangement of two trivacant Keggin {SeW9} anionic units and one {SeW8} anionic unit connected by an {Nd2.5Se2W4(mal)} cluster, thereby forming a distinct triangular configuration (Fig. 1e). This linker stabilizes the trimeric assembly through the formation of W–O–W, W–O–Nd, and W–O–Se bonds between the cluster and the {SeW9} or {SeW8} polyoxoanions (Fig. 1a and e–g). Within the {Nd2.5Se2W4(mal)} cluster, the oxygen atoms (O82 and O83) of the DL-malic acid ligand coordinate to W21, generating a five-membered chelate ring (Fig. 1i). The three Nd centers (Nd1, Nd2, and Nd3) connect to peripheral tungsten and selenium atoms via metal–oxygen–metal (M–O–M) linkages. All three Nd3+ ions adopt a distorted octacoordinate geometry (Fig. S1). Nd1 is coordinated through oxygen-sharing to six μ2-O atoms from {WO6} octahedra (W9, W19, W22, W26, W28, and W30), one μ2-O atom from a {SeO3} unit (Se5), one μ2-O atom from a sodium center (Na1), and one μ-O atom from a water molecule (Fig. 1j). Nd2 is coordinated through oxygen-sharing to five μ2-O atoms from {WO6} octahedra (W9, W12, W13, W19, and W30), one μ2-O atom from a {SeO3} unit (Se4), one μ2-O atom from a sodium center (Na4), and one μ-O atom from a water molecule (Fig. 1k). Notably, the Nd3 site exhibits positional disorder over two symmetry-equivalent positions (occupancy 0.5 each), with each Nd3+ ion coordinated via four μ3-O bridges linking to Na2, Se5, and W6, and four μ2-O bridges connecting to W2 and W5 (Fig. 1l). The bond distances of Nd–O range from 2.330 to 2.566 Å (Table S2).Dimerization occurs between two adjacent trimeric clusters via key structural linkers comprising two Na+ ions (Na5), the fractionally occupied Nd3 site, and two DL-malic acid ligands (Fig. S2). This distinctive dimer adopts a distorted configuration characterized by an ideal dihedral angle of precisely 78.783° between the planes defined by the constituent trimeric clusters (Fig. 2a and c). The Nd3 center bridges the dimeric units through coordination to four μ2-O atoms originating from W6 and W28. Each DL-malic acid ligand exhibits dual-bridging functionality: one carboxylate terminus coordinates Na5, which is simultaneously bound to the μ2-O donor from W6, while the opposing carboxylate group chelates W28 via two μ2-O atoms. Sodium atoms occupy peripheral positions around the dimeric units of NdSeW, playing critical roles in stabilizing the nanocluster assembly. This structural motif extends into a 1D chain (Fig. 2d) through alternating the vertical connectivity of dimeric clusters, where adjacent triangular clusters maintain parallel orientation (Fig. S3). Within this architecture, two Se4 atoms bridge neighboring clusters via coordination to two Nd2 atoms (through O atoms) and two W30 atoms from triangular subunits. Simultaneously, two Na4 atoms consolidate the chain by dual-bridging interactions: they coordinate two Nd2 atoms while linking two DL-malic acid ligands through oxygen bridges, thereby providing essential stabilization to the 1D framework (Fig. S4). In Fig. 2e, the 1D chain architecture is visualized with displaced 1a dimeric units represented as pink triangles and conventional NdSeW units as blue triangles. This color-coded scheme distinctly reveals the alternating vertical stacking pattern of dimeric clusters, where all triangular units maintain strict parallel alignment along the chain axis.
Furthermore, Fourier transform infrared (FT-IR) spectroscopy and X-ray diffraction (XRD) were employed to further verify the structure of NdSeW. XRD measurements (Fig. S5) confirmed the phase purity of NdSeW, as evidenced by the consistency between the experimental diffraction peaks and those in the simulated pattern. Meanwhile, the FT-IR analysis (Fig. S6) revealed vibrational modes commensurate with a lacunary Keggin-type architecture. The spectrum showed ν(W–O–W) bridges at 780 and 832 cm−1, terminal ν(W![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) bonds at 957 cm−1, and ν(Se–O) vibrations at 1052 cm−1. Features associated with the DL-malic acid ligand were also discernible, with C
O) bonds at 957 cm−1, and ν(Se–O) vibrations at 1052 cm−1. Features associated with the DL-malic acid ligand were also discernible, with C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and C–O stretching vibrations occurring at 1621 and 1416 cm−1, respectively, validating its coordination. Finally, a broad band centered at 3407 cm−1 was indicative of O–H stretches from water molecules.
O and C–O stretching vibrations occurring at 1621 and 1416 cm−1, respectively, validating its coordination. Finally, a broad band centered at 3407 cm−1 was indicative of O–H stretches from water molecules.
Catalytic performance
Imidazole, a significant heterocyclic compound, is extensively utilized in various fields including biomedicine, materials science, and organic synthesis.28–30 The conventional synthesis of imidazole derivatives typically involves the acid-catalyzed condensation reaction of aldehydes, benzils, and ammonium acetate.31–35 However, existing catalytic systems are associated with several limitations, such as environmental contamination caused by metal catalysts and organic solvents, long reaction times, harsh reaction conditions, and complicated operational procedures.36,37 Therefore, the development of an efficient, practical, and environmentally friendly catalytic system remains highly desirable and urgently needed. Considering the unique Lewis acid catalytic activity of rare-earth (RE) elements and our experience in RE-POM catalysis, the application of NdSeW as a catalyst for the condensation reaction of aldehydes, benzils, and ammonium acetate presents a promising approach for the synthesis of imidazoles.Therefore, we selected the three-component condensation reaction of benzaldehyde (1a, 0.2 mmol), benzil (2a, 0.2 mmol), and ammonium acetate (3a, 0.6 mmol) to evaluate the catalytic performance of NdSeW (Table 1). Under solvent-free conditions at 80 °C for 2 h, the yields of the desired product 4a with and without NdSeW were 57% and 10%, respectively (Table 1, entries 1 and 2). These results indicate that NdSeW indeed exhibits significant catalytic activity for this reaction. Subsequently, the influence of a series of green solvents, including dimethyl carbonate (DMC), ethyl acetate (EA), water, and EtOH, on the reaction yield was investigated. The addition of DMC, EA, and water did not have a positive effect; however, when EtOH was used as the solvent, the yield increased to 72% (Table 1, entries 3–6). When the reaction temperature was raised to 90 °C, the yield increased to 85%, and further increasing the temperature did not result in a significant improvement in yield. When the reaction was carried out for 3 h, the yield of 4a reached 97%. When the catalyst loading was reduced to 0.4 mol%, the yield significantly decreased to 88%. Further increasing the catalyst loading did not lead to a better result. Thus, the optimal catalytic conditions were determined as follows: benzaldehyde (1a, 0.2 mmol), benzil (2a, 0.2 mmol), ammonium acetate (3a, 0.6 mmol), NdSeW (0.5 mol%), and ethanol (1 mL), reacted at 90 °C for 3 h. Furthermore, we summarized and compared this catalytic system with those previously reported for constructing imidazole compounds (Table S3). The results demonstrate that NdSeW offers advantages in this reaction, including a low catalyst loading and high product yield.
| Entry | Solvent | Temperature (°C) | Time (h) | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: benzaldehyde (1a, 0.2 mmol), benzil (2a, 0.2 mmol), ammonium acetate (3a, 0.6 mmol), solvent (1 mL), and NdSeW (0.5 mol%) for 2 h. b The yields were determined by GC with biphenyl as the internal standard. c Without a catalyst. d Catalyst loading: 0.4 mol%. e Catalyst loading: 0.6 mol%. | ||||
| 1c | Solvent-free | 80 | 2 | 10 |
| 2 | Solvent-free | 80 | 2 | 57 |
| 3 | DMC | 80 | 2 | 32 |
| 4 | EA | 80 | 2 | 41 |
| 5 | H2O | 80 | 2 | 9 |
| 6 | EtOH | 80 | 2 | 72 |
| 7 | EtOH | 90 | 2 | 85 |
| 8 | EtOH | 100 | 2 | 86 |
| 9 | EtOH | 90 | 2.5 | 92 |
| 10 | EtOH | 90 | 3 | 97 |
| 11d | EtOH | 90 | 3 | 88 |
| 12e | EtOH | 90 | 3 | 96 |
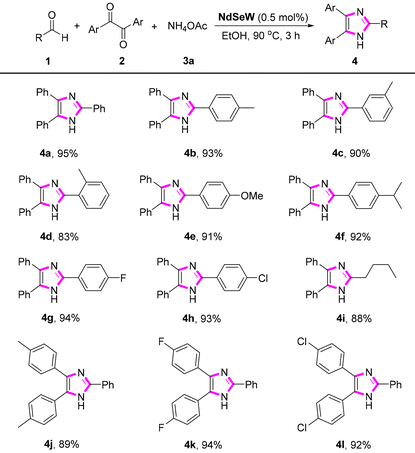
Subsequently, to evaluate the compatibility of this catalytic system, various aldehydes and benzils were used to prepare 2,4,5-trisubstituted imidazole derivatives (Table 2). First, the reactivity of a series of aldehydes containing electron-donating and electron-withdrawing groups was investigated. Benzaldehydes with electron-donating groups (such as –Me, –OMe, and –iPr) reacted smoothly with benzaldehyde and ammonium acetate, affording the corresponding imidazole products in 83%–93% yields (4a–4f). The steric hindrance of the benzaldehyde had a certain influence on the yield, as evidenced by the gradually decreasing yields observed for p-, m-, and o-methyl-substituted benzaldehydes. Benzaldehydes containing electron-withdrawing groups (such as –F and –Cl) were also effectively converted into the corresponding desired products, with yields of 94% and 93%, respectively (4g–4h). Furthermore, aliphatic aldehyde was well tolerated under this catalytic system; for instance, n-pentanal was converted into the desired product 4i in 88% yield. A series of 4,4′-disubstituted benzil compounds, such as 4,4′-dimethylbenzil, 4,4′-dichlorobenzil and 4,4′-dibromobenzil, also reacted efficiently with benzaldehyde and ammonium acetate, affording the corresponding products in 89%–94% yields (4j–4l).
Finally, we investigated the stability and recyclability of NdSeW. PXRD analysis of the catalyst before and after the reaction revealed no significant differences between the recovered and as-synthesized NdSeW (Fig. S5). Furthermore, catalytic cycling experiments were conducted to evaluate the reusability of NdSeW. There was no notable decrease in product yield after five consecutive cycles (Fig. 3a). This preliminary result indicates that NdSeW exhibits basic stability and recoverability under the tested conditions. To identify the active sites of NdSeW and explore the reaction mechanism, a series of control experiments was performed. As shown in Fig. 3b, both NdCl3·6H2O and (NH4)6Na18[Se6W39O141(H2O)3]·60H2O ({Se6W39}) were able to promote the reaction, but the yield obtained with NdCl3·6H2O was higher than that with {Se6W39}, suggesting that Nd3+ plays a dominant role in the catalysis. Notably, the catalytic efficiencies of NdCl3·6H2O, {Se6W39}, and their mixture were all lower than that of NdSeW, indicating that the well-defined crystalline structure has a positive effect on the catalytic activity. Based on relevant reports and the above results,38,39 a plausible reaction mechanism was proposed in Fig. S8. NdSeW activated the carbonyl groups of the aldehyde and benzil and then reacted with ammonia (generated in situ from ammonium acetate) to form imine intermediates A and B, respectively. The intermediate A condensed with the carbonyl group of B to give intermediate C, which finally undergoes dehydration and cyclization to afford the product 4a.
Conclusions
In conclusion, we have successfully synthesized and characterized a novel neodymium-{SeO3}-bridged selenotungstate (NdSeW). This compound features a distinctive architecture, in which {SeW9} and {SeW8} units are linked by an organometallic {Nd2Se2(mal)W4} cluster to form a triangular trimeric structure. Moreover, these trimers further self-assemble into an extended one-dimensional chain through alternating connections of neodymium and {SeO3} groups. Notably, NdSeW demonstrates high catalytic efficiency and versatility in the one-pot synthesis of 2,4,5-trisubstituted imidazoles via a three-component condensation of aldehydes, benzils, and ammonium acetate. The catalytic system proceeds under mild and environmentally friendly conditions, employing EtOH as a green solvent, with water as the sole by-product. This work not only enriches the structural diversity and synthetic methodology of Ln-POMs but also highlights their potential as sustainable catalysts for facilitating valuable organic transformations.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5dt02801a.CCDC 2489933 (NdSeW) contains the supplementary crystallographic data for this paper.40
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (22301033) and the Jiangxi Provincial Natural Science Foundation (20212BAB213001).References
- H.-X. Bi, X.-Y. Yin, J.-Y. He, H. Song, S.-J. Lu, Y.-Y. Ma and Z.-G. Han, Conjugated organic component-functionalized hourglass-type phosphomolybdates for visible-light photocatalytic Cr(VI) reduction in wide pH range, Rare Met., 2023, 42, 3638–3650 CrossRef CAS.
- A. Bijelic, M. Aureliano and A. Rompel, Polyoxometalates as Potential Next-Generation Metallodrugs in the Combat Against Cancer, Angew. Chem., Int. Ed., 2019, 58, 2980–2999 CrossRef CAS PubMed.
- Y. Chen, Z.-X. Zhang, P.-W. Cai, Z.-W. Guo, Z.-W. Lu, C. Sun, X.-X. Li, J.-X. Chen, Z.-H. Wen and S.-T. Zheng, Polyoxotungstate Featuring Zinc-Ion-Triggered Structural Transformation as An Efficient Electrolyte Additive for Aqueous Zinc-Ion Batteries, Angew. Chem., Int. Ed., 2025, 64, e202420284 CrossRef CAS PubMed.
- J. Lei, Z. Yu, S. Ru, Z. Wang, S. Li, N. Luo, J. Huang, X. Peng, F. Jiang and Y. Wei, Decavanadate-Catalyzed Oxidative Cross-Dehydrogenative Coupling: an Efficient and Sustainable Approach toward P(O)─S Bond Formation, Angew. Chem., Int. Ed., 2025, 64, e202507245 CrossRef CAS PubMed.
- H. Li, M. Yang, Z. Yuan, Y. Sun, P. Ma, J. Niu and J. Wang, Construction of one Ru2W12-cluster and six lacunary Keggin tungstoarsenate leading to the larger Ru-containing polyoxometalate photocatalyst, Chin. Chem. Lett., 2022, 33, 4664–4668 CrossRef CAS.
- S.-R. Li, W.-D. Liu, L.-S. Long, L.-S. Zheng and X.-J. Kong, Recent advances in polyoxometalate-based lanthanide–oxo clusters, Polyoxometalates, 2023, 2, 9140022 CrossRef.
- Y.-F. Liu, C.-W. Hu and G.-P. Yang, Recent advances in polyoxometalates acid-catalyzed organic reactions, Chin. Chem. Lett., 2023, 34, 108097 CrossRef CAS.
- Z.-Y. Liu, Y.-D. Lin, Y. Hao, H.-N. Chen, Z.-W. Guo, X.-X. Li and S.-T. Zheng, Recent advances in polyoxoniobate-catalyzed reactions, Tungsten, 2022, 4, 81–98 CrossRef.
- G. Yang, Y. Liu and Y. Wei, Application of polyoxometalates in biomass conversion, Coord. Chem. Rev., 2024, 521, 216172 CrossRef CAS.
- X. Huang, S. Liu, G. Liu, Y. Tao, C. Wang, Y. Zhang, Z. Li, H. Wang, Z. Zhou, G. Shen, Z. Xue and D. Sun, An unprecedented 2-fold interpenetrated lvt open framework built from Zn6 ring seamed trivacant polyoxotungstates used for photocatalytic synthesis of pyridine derivatives, Appl. Catal., B, 2023, 323, 122134 CrossRef CAS.
- C. Lian and G.-Y. Yang, A tetrameric polyoxometalate with two orderly and dense double-layered {Ni12W8/3} cluster centers, Chem. Commun., 2025, 61, 14402–14405 RSC.
- G. Liu, Y. Chen, Y. Chen, Y. Shi, M. Zhang, G. Shen, P. Qi, J. Li, D. Ma, F. Yu and X. Huang, Indirect Electrocatalysis S─N/S─S Bond Construction by Robust Polyoxometalate Based Foams, Adv. Mater., 2023, 35, 2304716 CrossRef CAS PubMed.
- L. Liu, J. Jiang, X. Liu, G. Liu, D. Wang, L. Chen and J. Zhao, First series of mixed (PIII, SeIV)-heteroatomoriented rare-earth-embedded polyoxotungstates containing distinct building blocks, Inorg. Chem. Front., 2020, 7, 4640–4651 RSC.
- X. Zhang, H. Li, N. Xu, X. Liu and X.-L. Wang, Engineering geometric metamorphosis in {P6M2Mo16O73}-based high-nuclearity metal superclusters: from tetrahedral to square assemblies via topological transformations, Inorg. Chem. Front., 2025, 12, 3823–3832 RSC.
- C. Boskovic, Rare Earth Polyoxometalates, Acc. Chem. Res., 2017, 50, 2205–2214 CrossRef CAS PubMed.
- Y.-C. Dai, S.-Y. Zhang, X.-X. Xiao, M.-J. Li, J.-C. Liu, L.-J. Chen and J.-W. Zhao, A double-tartrate-bridged deca-nuclearity europium-tungsten cluster embedded selenotungstate and its selective optical sensing of o-nitrophenol, Polyoxometalates, 2023, 2, 9140041 CrossRef.
- D. Zhou, B. Li, Q. Zhao, X. Tang, T. Lan, H. Su, G. Yang and W. Xuan, Solvent-modulated assembly of peptide and cerium functionalized gigantic {Mo120Ce6}2 dimers for high-efficiency photocatalytic oxidation, Inorg. Chem. Front., 2024, 11, 2355–2364 RSC.
- J. M. Cameron, J. Gao, L. Vilà-Nadal, D.-L. Long and L. Cronin, Formation, self-assembly and transformation of a transient selenotungstate building block into clusters, chains and macrocycles, Chem. Commun., 2014, 50, 2155–2157 RSC.
- J. Jiang, L. Liu, G. Liu, D. Wang, Y. Zhang, L. Chen and J. Zhao, Organic–Inorganic Hybrid Cerium-Encapsulated Selenotungstate Including Three Building Blocks and Its Electrochemical Detection of Dopamine and Paracetamol, Inorg. Chem., 2020, 59, 15355–15364 CrossRef CAS.
- L. Liu, J. Jiang, L. Cui, J. Zhao, X. Cao and L. Chen, Double Trigonal Pyramidal {SeO3} Groups Bridged 2-Picolinic Acid Modified Cerium-Inlaid Polyoxometalate Including Mixed Selenotungstate Subunits for Electrochemically Sensing Ochratoxin A, Inorg. Chem., 2022, 61, 1949–1960 CrossRef CAS PubMed.
- J. Yan, D.-L. Long and L. Cronin, Development of a Building Block Strategy To Access Gigantic Nanoscale Heteropolyoxotungstates by Using SeO32− as a Template Linker, Angew. Chem., Int. Ed., 2010, 49, 4117–4120 CrossRef CAS.
- W.-C. Chen, H.-L. Li, X.-L. Wang, K.-Z. Shao, Z.-M. Su and E.-B. Wang, Assembly of Cerium(III)-Stabilized Polyoxotungstate Nanoclusters with SeO32−/TeO32− Templates: From Single Polyoxoanions to Inorganic Hollow Spheres in Dilute Solution, Chem. – Eur. J., 2013, 19, 11007–11015 CrossRef CAS PubMed.
- H.-L. Li, Y.-J. Liu, Y.-M. Li, L.-J. Chen, J.-W. Zhao and G.-Y. Yang, Unprecedented Selenium and Lanthanide Simultaneously Bridging Selenotungstate Aggregates Stabilized by Four Tetra-vacant Dawson-like {Se2W14} Units, Chem. – Asian J., 2018, 13, 2897–2907 CrossRef CAS PubMed.
- H.-L. Li, S.-H. Zhao, N.-H. Wang, Y.-L. Ma, C. Lian and X. Cao, Se-Rich Multinuclear Er-Containing Dawson-type Poly(selenotungstate), Inorg. Chem., 2024, 63, 21645–21651 CrossRef CAS.
- W.-C. Chen, L.-K. Yan, C.-X. Wu, X.-L. Wang, K.-Z. Shao, Z.-M. Su and E.-B. Wang, Assembly of Keggin-/Dawson-type Polyoxotungstate Clusters with Different Metal Units and SeO32− Heteroanion Templates, Cryst. Growth Des., 2014, 14, 5099–5110 CrossRef CAS.
- G. Liu, L. Liu, T. Gong, Y. Li, L. Chen and J. Zhao, Nicotinic-Acid-Ornamented Tetrameric Rare-Earth-Substituted Phospho(III)tungstates with the Coexistence of Mixed Keggin/Dawson Building Blocks and Its Honeycomb Nanofilm for Detecting Toxins, Inorg. Chem., 2021, 60, 14457–14466 CrossRef CAS PubMed.
- J.-C. Liu, J.-F. Wang, Q. Han, P. Shangguan, L.-L. Liu, L.-J. Chen, J.-W. Zhao, C. Streb and Y.-F. Song, Multicomponent Self-Assembly of a Giant Heterometallic Polyoxotungstate Supercluster with Antitumor Activity, Angew. Chem., Int. Ed., 2021, 60, 11153–11157 CrossRef CAS PubMed.
- Y. Gao, B. Mei, Y. Wu, Q. Zhao, Z. Bao, H. Qin, Y. Xu, H. Lv, X. Peng, Y. He, T. Luo, R. Yao, W. Zhang, H. Lei and R. Cao, A Cobalt(III) Corrole with a Tethered Imidazole for Boosted Electrocatalytic Oxygen Reduction Reaction, Chin. J. Chem., 2023, 41, 2866–2872 CrossRef CAS.
- K. Ravikumar and M. S. Dangate, Imidazole and thiazole derivatives: Synthesis, characterization, and nonlinear optical properties, J. Mol. Struct., 2025, 1338, 142287 CrossRef CAS.
- L. Zhang, X.-M. Peng, G. L. V. Damu, R.-X. Geng and C.-H. Zhou, Comprehensive Review in Current Developments of Imidazole-Based Medicinal Chemistry, Med. Res. Rev., 2014, 34, 340–437 CrossRef CAS PubMed.
- A. Alanthadka, S. D. Elango, P. Thangavel, N. Subbiah, S. Vellaisamy and U. M. Chockalingam, Construction of substituted imidazoles from aryl methyl ketones and benzylamines via N-heterocyclic carbene-catalysis, Catal. Commun., 2019, 125, 26–31 CrossRef CAS.
- J.-R. Chang, L. Cao, S. Jia, F. Fang, Z.-H. Fan, Q.-J. Pan, Y.-N. Ma and X. Chen, Synthesis of Imidazole Adducts of BH3, BF3, and B3H7 and Comparison of Their Nucleophilic Substitution Reaction Activity, Chin. J. Chem., 2025, 43, 925–930 CrossRef CAS.
- J. Jayram and V. Jeena, Copper-catalyzed aerobic benzylic sp3 C–H oxidation mediated synthesis of 2,4,5-trisubstituted imidazoles via a domino multi-component reaction, Green Chem., 2017, 19, 5841–5845 RSC.
- D. Kumar, D. N. Kommi, N. Bollineni, A. R. Patel and A. K. Chakraborti, Catalytic procedures for multicomponent synthesis of imidazoles: selectivity control during the competitive formation of tri- and tetrasubstituted imidazoles, Green Chem., 2012, 14, 2038–2049 RSC.
- X. Tian, L. Song, M. Rudolph, F. Rominger, T. Oeser and A. S. K. Hashmi, Sulfilimines as Versatile Nitrene Transfer Reagents: Facile Access to Diverse Aza-Heterocycles, Angew. Chem., Int. Ed., 2019, 58, 3589–3593 CrossRef CAS PubMed.
- C. T. F. Salfeena, R. Jalaja, R. Davis, E. Suresh and S. B. Somappa, Synthesis of 1,2,4-Trisubstituted-(1H)-imidazoles through Cu(OTf)2/I2-Catalyzed C–C Bond Cleavage of Chalcones and Benzylamines, ACS Omega, 2018, 3, 8074–8082 CrossRef CAS PubMed.
- J. O. Strelnikova, N. V. Rostovskii, G. L. Starova, A. F. Khlebnikov and M. S. Novikov, Rh(II)-Catalyzed Transannulation of 1,2,4-Oxadiazole Derivatives with 1-Sulfonyl-1,2,3-triazoles: Regioselective Synthesis of 5-Sulfonamidoimidazoles, J. Org. Chem., 2018, 83, 11232–11244 CrossRef CAS.
- N. Hosseini Mohtasham and M. Gholizadeh, Magnetic horsetail plant ash (Fe3O4@HA): a novel, natural and highly efficient heterogeneous nanocatalyst for the green synthesis of 2,4,5-trisubstituted imidazoles, Res. Chem. Intermed., 2021, 47, 2507–2525 CrossRef CAS.
- M. Masteri-Farahani, A. Ezabadi, R. Mazarei, P. Ataeinia, S. Shahsavarifar and F. Mousavi, A new nanocomposite catalyst based on clay-supported heteropolyacid for the green synthesis of 2,4,5-trisubstituted imidazoles, Appl. Organomet. Chem., 2020, 34, e5727 CrossRef CAS.
- CCDC 2489933: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pkzfp.
| This journal is © The Royal Society of Chemistry 2026 |