Open Access Article
Open Access ArticleSurface-initiated living crystallization-driven self-assembly: from precision nanofabrication to functional interfaces
Jiawei
Tao†
 *,
Yao
Lu†
,
Chenchen
Gao†
,
Peiwen
Wang
,
Zhenyan
Ji
,
Sen
Lv
,
Xiaomin
Cai
,
Jingwen
Zhang
and
Huibin
Qiu
*,
Yao
Lu†
,
Chenchen
Gao†
,
Peiwen
Wang
,
Zhenyan
Ji
,
Sen
Lv
,
Xiaomin
Cai
,
Jingwen
Zhang
and
Huibin
Qiu
 *
*
State Key Laboratory of Synergistic Chem-Bio Synthesis, School of Chemistry and Chemical Engineering, Frontiers Science Center for Transformative Molecules, State Key Laboratory of Micro-Nano Engineering Science, Shanghai Jiao Tong University, Shanghai 200240, China. E-mail: jiawei.tao@sjtu.edu.cn; hbqiu@sjtu.edu.cn
First published on 12th March 2026
Abstract
The precise fabrication of nanostructured materials on surfaces is paramount for advancing next-generation technologies in catalysis, electronics, biomedicine, etc. Recently, surface-initiated living crystallization-driven self-assembly (SIL-CDSA) has enabled unprecedented control over the architecture, orientation, and functionality of solid-state nanostructures on desired substrates. This review outlines key principles of SIL-CDSA, including seed immobilization, confined epitaxial growth and micellar brush formation, together with practical methods for dimension control and spatial ordering. Intrinsic core/corona chemistry and the incorporation of exogenous species allowing targeted functionalization are also discussed, covering catalytic, electronic and bioactive features. Case studies are introduced to demonstrate SIL-CDSA as a versatile platform for hierarchically ordered nanostructures, delivering exceptional control over size, periodicity, and composition. By directly translating precision self-assembly to surfaces, SIL-CDSA opens powerful routes to multifunctional, high-performance materials for catalysis, energy systems, sensing, and device fabrication, positioning it as a transformative tool for advanced nanomanufacturing.
1. Introduction
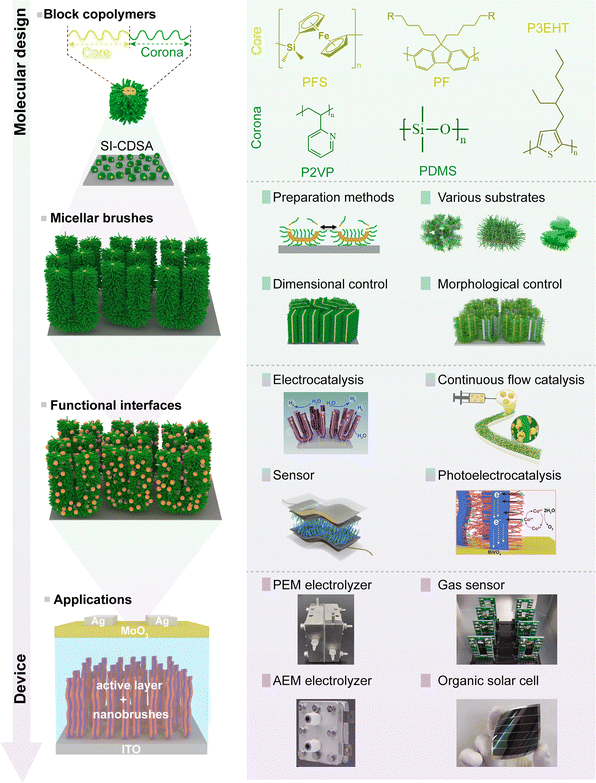
The quest for bottom-up fabrication of materials with molecular-level precision has been a central ambition in nanoscience, drawing inspiration from the exquisite control observed in biological systems.1–3 Such precision is essential for unlocking the full potential of functional materials in fields ranging from quantum electronics to targeted therapeutics.4–7 Conventional self-assembly processes, while powerful, often yield structures with broad size distributions and limited morphological complexity due to stochastic nucleation and growth events.8–10 This limitation spurred the development of “living” self-assembly methodologies, analogous to living polymerization, where the growth of nanostructures proceeds from a fixed number of active seed sites in the absence of termination and chain transfer.11,12 This paradigm shift enables the fabrication of nanostructures with precisely controlled dimensions, low dispersity, and complex, segmented architectures.4,13,14Among these living methods, crystallization-driven self-assembly (CDSA) has proven particularly versatile and powerful.15–19 CDSA leverages the crystallization of a core-forming block within a block copolymer (BCP) to drive the assembly process.20,21 The epitaxial crystallization of subsequently added unimers (soluble BCPs) at the termini of existing seed nanocrystals further leads to the growth of low-curvature nanostructures,22,23 such as one-dimensional (1D) cylindrical micelles,15,24–30 two-dimensional (2D) platelets31–38 and hierarchical micelles with anisotropic shapes.39–45 The “living” nature of this process allows the precise tuning of nanostructure length or area by simply adjusting the unimer-to-seed ratio.26,46 This approach was pioneered in the studies of poly(ferrocenylsilane) (PFS)-based block copolymers, where epitaxial growth enabled the fabrication of monodisperse nanofibers with tunable lengths.47 However, a key challenge appears during the translation of this solution-phase precision to solid substrates.48 While solution-based CDSA yields remarkable materials, their random deposition onto surfaces forfeits control over orientation and spatial organization, hindering their integration into functional devices.
Such limitation has recently been directly addressed by the advent of surface-initiated living CDSA (SIL-CDSA), a strategy that tethers the assembly process to a substrate.49 By immobilizing seed nanocrystals onto a surface, the subsequent epitaxial growth is spatially confined, enabling the fabrication of densely packed, vertically aligned nanostructure arrays, often termed “micellar brushes”.49–58 This approach bridges the molecular precision of living self-assembly with the macroscopic order required for device integration (Fig. 1). Core scientific objectives in this burgeoning field revolve around: (1) achieving robust and versatile control over seed immobilization on diverse substrates; (2) elucidating fundamental mechanisms governing growth kinetics and morphology at solid–liquid interfaces; (3) tailoring the dimensions, shape, and hierarchical complexity of surface-grafted nanostructures; (4) developing strategies for functionalization toward specific applications; (5) probing the performance limits and technological potential of SIL-CDSA-based devices. This review positions SIL-CDSA as a cornerstone of nanotechnology, offering viable insights for researchers pursuing multifunctional, scalable nanomaterials.
2. Overview of living CDSA
Traditional approaches relying on solvent compatibility [e.g., polystyrene-b-poly(ethylene oxide) (PS-b-PEO) in water/tetrahydrofuran (THF)], typically form spherical micelles or vesicles through entropy-driven separation.59–67 In contrast, living CDSA fundamentally differs by employing anisotropic crystallization of core-forming blocks to precisely yield low-curvature architectures. The watershed study in 2007 by Manners and co-workers demonstrated this by the self-assembly of PFS-b-polyisoprene (PFS-b-PI) in hexane, where epitaxial growth of PFS cores produced monodisperse nanocylinders with lengths tunable from 200 nm to 2 µm.46 Subsequent work extended living CDSA to diverse crystallizable polymers: e.g., poly(ε-caprolactone) (PCL) formed 2D platelets in THF/water,68 poly(3-hexylthiophene) (P3HT) generated semiconducting fibers.69 Unlike solvophobic systems limited by equilibrium thermodynamics, living CDSA operates under kinetic control governed by chain folding energetics and crystal lattice matching, and revolutionizes the fabrication of nanostructures by mimicking the temporal control of chain growth polymerization. Introducing pre-formed micelle seeds to unimer solutions initiates epitaxial crystallization at termini, with growth rates following first-order kinetics.34,35,50,70 Polymerization-induced CDSA further integrates synthesis and assembly. Ring-opening polymerization of ε-caprolactone directly generated crystalline cores templating uniform 2D platelets.71 Sequential addition of different unimers enables architectural complexity: segmented PDMS/P2VP nanofibers exhibit diverse, multidimensional assemblies in different solvents.40Two complementary strategies are widely used to control nucleation and growth in living CDSA. Self-seeding leverages the inherent crystallinity of the core-forming block: thermal or solvent-induced partial dissolution removes less ordered regions while preserving well-defined crystalline nuclei that govern uniform regrowth upon cooling.72,73 This three-step protocol—fragmentation of initial micelles (often by sonication), annealing above a partial dissolution temperature to eliminate defective crystallites, and controlled cooling to allow epitaxial growth—effectively decouples nucleation and growth and yields size control beyond what is possible in equilibrium-driven solvophobic assembly.74,75 Seeded growth, by contrast, employs stable, preformed micelle seeds added to unimer solutions at ambient conditions.76 Pioneered for PFS-based BCPs, this approach has expanded to diverse crystalline-coil systems, including π-conjugated polymers4,77–79 and biodegradable polyesters,80,81 favoring unprecedentedly high monodispersity in solution assembly.
3. Surface-initiated living CDSA: principles and fabrication
Both self-seeding and seeded-growth methods were developed and optimized in free solution, but translating these concepts to interfaces is essential for practical applications such as surface patterning and device integration. The growth behavior of SIL-CDSA differs fundamentally from that in solution because the micelle seeds are tethered to an immobilized substrate.17,48,82 In this configuration, the elongation of crystalline domains is no longer governed solely by unimer and self-nucleation in free solution, but instead results from the combined influence of restricted unimer diffusion through the surrounding medium and site-specific attachment at the surface-bound seed layer.20,57,83 The finite mobility of unimers, dictated by their concentration, solvent quality, and diffusional path length, directly influences the rate and uniformity of crystallite extension.36,84 At the same time, the chemistry and topology of the underlying substrate modulate the polymer–substrate interactions (e.g., hydrogen bonding, π–π stacking, electrostatic attraction). Additionally, surface coverage of initiating sites determines the lateral packing density of nanostructures. Variations in these parameters collectively regulate the attainable dimensions (height, density), spatial arrangement, and orientational order of the assembled nanostructures.8,37 By deliberately tuning unimer supply, initiation density, and interfacial chemistry, SIL-CDSA can achieve a structural precision in terms of uniformity, alignment, and pattern complexity, which is rarely accessible with conventional solution-based CDSA,26,85,86 thereby establishing a robust platform for the versatile fabrication strategies detailed in the sections that follow.87–893.1. Fabrication strategies
The fabrication of SIL-CDSA architectures is a multi-step process, beginning with the preparation of well-defined micelle seeds, followed by their immobilization on a substrate, and culminating in controlled directional growth. The precision and reproducibility at each stage are crucial for achieving uniform nanostructure arrays.3.1.1.1. Fragmentation. The most widely used method relies on first forming the desired micellar aggregate and then fragmenting it into smaller, uniform pieces, often through sonication (Fig. 2a).51,76 This route is broadly applicable because it does not require specialized equipment or polymer designs, and it is compatible with many semicrystalline core-forming blocks. Importantly, seed length and dispersity can be tuned by controlling the parent micelle morphology and the fragmentation conditions (e.g., sonication power/time, temperature, and polymer concentration). Despite being relatively time-consuming, its robustness and reproducibility make it the default choice in most studies (Table 1).
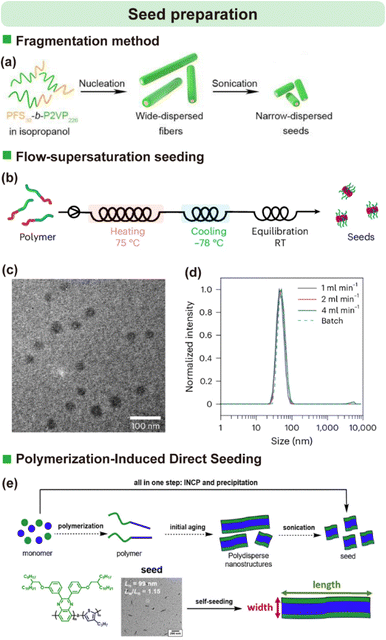 | ||
| Fig. 2 (a) Schematic illustration of the conventional fragmentation method used to prepare micelle seeds. Reproduced from ref. 51 with permission from American Chemical Society, copyright 2021. (b) Scheme for preparation of micelle seeds generated in-flow using diblock copolymers. (c) Transmission electron microscope (TEM) image of the resulting seeds. (d) Dynamic light scattering size distributions of flow-prepared seeds obtained at various flow rates. Reproduced from ref. 5 with permission from Springer Nature, copyright 2025. (e) Comparison of methods for producing uniform seeds: conventional CDSA (dashed arrow) versus polymerization-induced direct seeding (solid arrow) in a single step. Reproduced from ref. 68 with permission from American Chemical Society, copyright 2024. | ||
| Method | Principle | Time required | Typical applicability |
|---|---|---|---|
| Fragmentation51,76 | Induce self-assembly to form large aggregates, then break into short, uniform seeds | Days to a week | Default choice in most CDSA studies; general seed preparation for various block copolymers. |
| Flow-supersaturation seeding5 | Use a flow system to rapidly reach supersaturation, leading to uniform micelle nucleation in minutes. | Minutes | High-throughput or industrial CDSA processes; rapid prototyping. |
| Polymerization-induced direct seeding86 | Generate seeds directly during PI-CDSA of block copolymers, followed by simple purification. | Hours | When polymer synthesis and seed preparation can be integrated; tailored nanostructures. |
3.1.1.2. Flow supersaturation seeding. Recent innovations have focused on accelerating and simplifying seed production. For instance, Xiao et al. demonstrated a rapid seed preparation method using a flow system where supersaturation of a polymer solution drives uniform seed micelle formation in minutes, a significant reduction from the week-long processes previously reported (Fig. 2b–d).5 Mechanistically, rapid mixing under flow enables precise control over the nucleation window, helping suppress secondary nucleation and broad size distributions. Beyond speed, this strategy is attractive for scale-up because it improves throughput by orders of magnitude and is amenable to continuous, end-to-end production, enabling direct conversion from polymer feedstock to seeded nanostructure growth.
3.1.1.3. Polymerization induced direct seeding. Another strategy involves leveraging the polymer synthesis itself. Kim et al. showed that polymerization-induced CDSA (PI-CDSA) of fully conjugated block copolymers could produce nanoribbons, and simple purification by precipitation yielded short, uniform seed structures directly, bypassing the conventional aging and fragmentation steps entirely (Fig. 2e).86 However, PI-CDSA seed generation is currently more system-dependent, requiring compatibility between polymerization conditions, solubility evolution, and crystallization/assembly kinetics.
Collectively, these approaches substantially shorten seed preparation time and broaden the toolkit for living CDSA. Nevertheless, the conventional “self-assembly + fragmentation” route remains most commonly adopted because of its generality across polymer chemistries and morphologies, whereas newer methods (flow-based supersaturation and PI-CDSA-derived seeds) offer compelling advantages in speed, integration, and scalability but may require more stringent control of solution thermodynamics or polymerization/assembly coupling (Table 1).
3.1.2.1. Drop casting
A simple and versatile approach in which a micelle seed solution is dispensed directly onto the substrate surface, followed by solvent evaporation (Fig. 3a). This method is applicable to both smooth and rough surfaces, including silicon wafers, glass slides, and metal foils, and is particularly useful for rapid prototyping or small-area patterning.
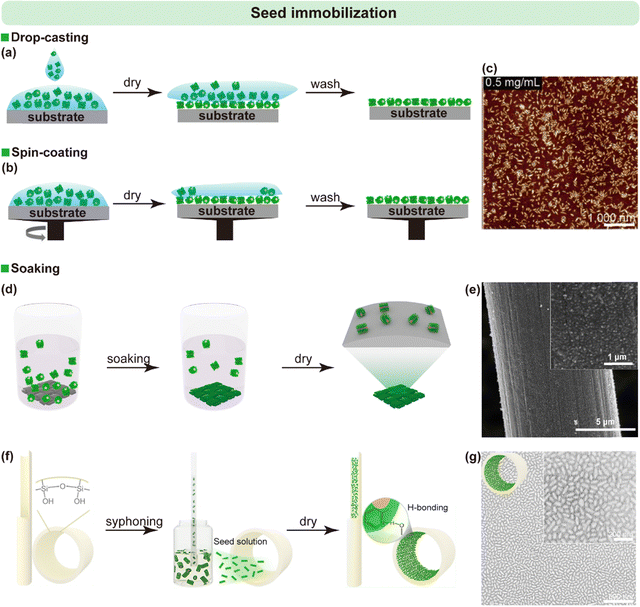 | ||
| Fig. 3 (a) Schematic of drop-casting used for micelle seed immobilization. (b) and (c) Schematic of spin-coating (b) and corresponding AFM image (c). (c) is reproduced from ref. 49 with permission from AAAS, copyright 2019. (d–g) Schematics of the soaking method, with corresponding SEM images shown in (e) and (g). (e) is reproduced from ref. 54 with permission from Spring Nature, copyright 2025. (g) is reproduced from ref. 51 with permission from Wiley, copyright 2021. | ||
3.1.2.2. Spin coating
Best suited for smooth, planar substrates. By applying centrifugal force, seeds are evenly distributed over the surface, enabling the formation of highly uniform and densely packed seed layers (Fig. 3b and c). This uniformity is critical for producing vertically aligned micellar brushes with controlled density.49
3.1.2.3. Soaking
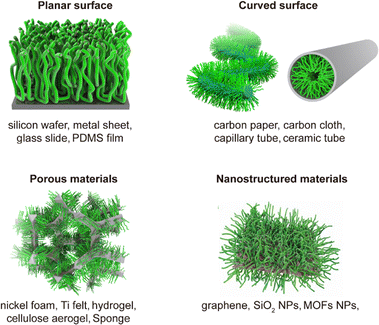
Ideal for porous or nanostructured substrates such as carbon cloth, nickel foam, or graphene nanosheets (Fig. 3d and e). In this approach, micelle seeds readily diffuse into and adsorb throughout the micro- and nano-scale pores, enabling high surface coverage even on complex, irregular geometries. The method can also be applied to tubular materials, such as glass capillaries, by simply immersing one end of the tube into the seed solution, allowing capillary action to draw the solution inward (Fig. 3f and g).
Seed–substrate adhesion in SIL-CDSA is typically achieved through either non-covalent or covalent interactions. Non-covalent approaches, including hydrogen bonding, electrostatic attraction, and coordination, offer simple and versatile immobilization routes that do not require chemical surface modification. For example, pyridine groups in a P2VP corona can hydrogen-bond with surface silanol groups on silica, or directly coordinate to gold through pyridine–Au interactions. Although these methods enable rapid and straightforward attachment across a variety of substrates, the binding strength is generally lower than that of covalent linkages. Such interactions are also sensitive to environmental conditions. For instance, under acidic conditions, protonation of the pyridine nitrogen diminishes its ability to hydrogen-bond or coordinate with metal surfaces, which may lead to seed detachment.
By contrast, covalent bonding provides the most robust form of immobilization, ensuring stability even under demanding processing conditions. This strategy typically involves pre-functionalizing the substrate with reactive moieties capable of forming covalent linkages with corona chain functional groups. For instance, micelle seeds bearing a P2VP corona can be anchored to otherwise inert interfaces via reaction with 3-iodopropyltrimethoxysilane (Fig. 4).49 The enhanced durability of such attachment was demonstrated by subjecting the immobilized seeds to quaternization with methyl iodide (MeI), a treatment known to disrupt hydrogen-bond-based immobilization. No detectable detachment was observed for covalently bound seeds. Covalent strategies are particularly advantageous for stabilizing seeds on chemically inert substrates such as polymers or elastomers.
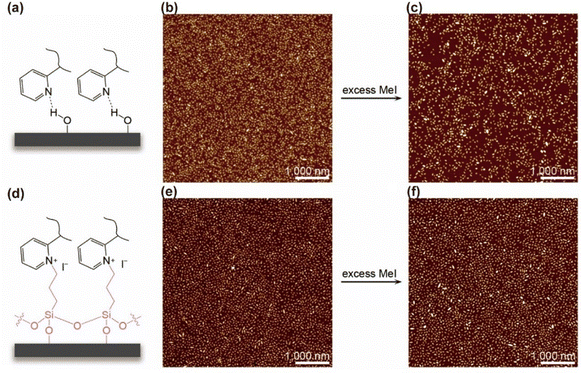 | ||
| Fig. 4 (a) Schematic of H-bond-based immobilization of PFS-b-P2VP seeds on silicon. (b) and (c) AFM height images of PFS36-b-P2VP502 seeds (0.5 mg mL−1 in isopropanol) immobilized via H-bonding before (b) and after (c) MeI treatment. Significant seed detachment was observed. (d) Schematic of covalent immobilization using 3-iodopropyltrimethoxysilane. (e) and (f) AFM height images of PFS36-b-P2VP502 seeds immobilized covalently before (e) and after (f) MeI treatment. Covalent bonding prevented noticeable detachment. Reproduced from ref. 49 with permission from AAAS, copyright 2019. | ||
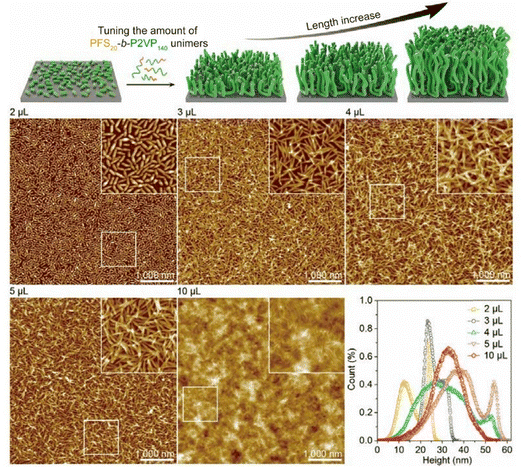 | ||
| Fig. 5 Top: schematic illustrating brush elongation and height increase with added unimers. Bottom: AFM height images and distributions of PFS20-b-P2VP140 brushes grown from seeds (Ln = 58 nm). Adding 2–5 µL unimer transformed lying micelles (∼20 nm height) into stacked (∼25 nm) and coral-like structures (10–60 nm height, average 35–40 nm). At 10 µL, brushes collapsed into a uniform layer (∼35 nm), with heights possibly underestimated due to probe inaccessibility. Reproduced from ref. 49 with permission from AAAS, copyright 2019. | ||
By tuning both factors in concert, SIL-CDSA enables predictable and reproducible brush growth with controlled length, alignment, and uniformity across the surface. Such precision is critical for engineering surface-bound nanostructures with tailored mechanical stability, optical anisotropy, or interfacial properties that match specific application needs. Notably, micellar brush growth via SIL-CDSA has been validated on a wide range of chemically and structurally diverse substrates—including carbon cloth, carbon paper,54 nickel foam,53 metal–organic-framework (MOF) particles,55 PDMS membranes52 and others (Fig. 6), demonstrating the adaptability and universality of this methodology. This broad substrate compatibility greatly facilitates integration of micellar brushes into complex architectures, such as catalytic electrodes, flexible coatings, or porous scaffolds, and opens opportunities for hybrid material systems that combine organic precision with inorganic robustness.
3.2. Mechanistic insights
Manners et al. employed atomic force microscopy (AFM) to conduct detailed, real-time investigations of the SIL-CDSA of individual micelle seed immobilized on silicon substrates.58 Their findings revealed that fiber-like micelle growth from single, surface-bound seed proceeds through four distinct kinetic regimes, evolving from an initial surface-confined mode to a surface-limited regime. This progression was reflected in changes in the growth rate with increasing length of the epitaxially grown segments extending from each seed terminus (Fig. 7a).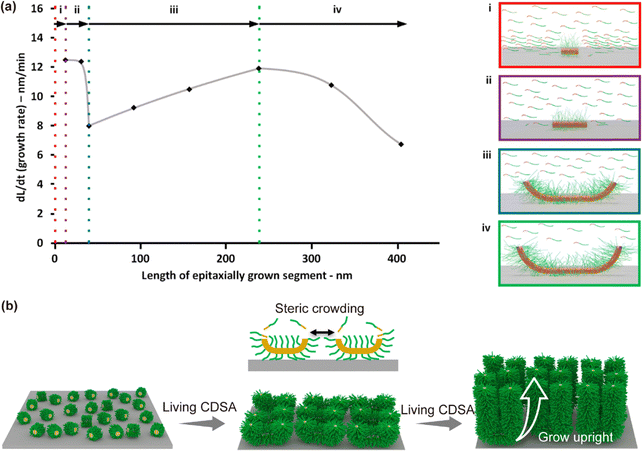 | ||
| Fig. 7 (a) Mathematical fit of the interfacial growth rate of PFS-b-P2VP micelles versus segment length formed by epitaxial growth, obtained by adding PFS-b-P2VP in THF (2.5 µL, 10 mg mL−1) to PFS-b-P2VP surface-bound seeds (SBSs) (5 µL, 0.01 mg mL−1, Ln = 31 nm) immobilized on Si wafers in 0.5 mL iPrOH. Dotted lines and arrows indicate four distinct regimes (i–iv). Schematic representations of these regimes: (i) SBSs show a high initial growth rate due to unimer near the surface. (ii) Growth rate declines exponentially as local unimer is rapidly consumed; growth remains diffusion-limited under strong surface influence. (iii) A slower exponential rate increase occurs as longer micelle segments extend further into solution, improving unimer access. (iv) Local unimer depletion leads to a decline similar to seeded growth in bulk solution. The peak and drop-off are fitted with a Boltzmann curve, reflecting steady-state, diffusion-limited growth. Reproduced from ref. 58 with permission from American Chemical Society, copyright 2022. (b) Schematic illustrating steric crowding between adjacent seeds, causing them to extend predominantly away from the surface. | ||
To illustrate, ex situ data were obtained from micelles grown by interfacial seeded growth of a PFS-b-P2VP unimer solution in THF from PFS-b-P2VP surface-bound seeds. The growth curves were fitted to mathematical models capturing the four kinetic stages:
– Initial regime – very rapid elongation (Fig. 7a(i)).
– First decay – exponential decrease in rate up to a turning point (Fig. 7a(ii)), attributed to depletion of unimer near the surface.
– Surface-dissociation acceleration – exponential rate increase (Fig. 7a(iii)) as the micelle termini lift away from the substrate, enhancing unimer access.
– Final steady state – Boltzmann-type rate decay (Fig. 7a(iv)) as bulk unimer depletion limits further growth.
When a high density of seeds is immobilized to form micellar brushes, the growth geometry changes markedly (Fig. 7b). Steric crowding between adjacent seeds and their expanding coronas limits lateral growth, forcing nascent micelles to extend predominantly away from the surface. This vertical or tilted orientation bypasses the prolonged surface-parallel regime seen for isolated seeds, leading to earlier onset of surface dissociation and a more uniform, upright brush-like architecture. In such densely packed systems, unimer delivery to growth fronts is further constrained by diffusion through the interstitial corona matrix, potentially generating heterogeneous growth rates among fibers.
4. Advanced structural control and modulation
4.1. Dimension control of micellar brush arrays
The axial dimensions of cylindrical micellar brushes generated by SIL-CDSA are governed primarily by the kinetic parameters of living crystallization, as outlined previously. In essence, systematic variation of the seed-to-unimer ratio enables precise regulation of brush height. This principle forms the basis for predictable one-dimensional growth, yet SIL-CDSA can also be adapted to create complex 2D micellar architectures by deliberately perturbing the lateral symmetry of the growth environment.A representative strategy for achieving such lateral growth anisotropy relies on the tilted immobilization of cylindrical micelle seeds, exemplified by PFS24-b-P2VP371, on silicon substrates (Fig. 8a).83 Hydrogen bonding between the pyridine moieties of the P2VP corona and surface silanol groups induces asymmetric conformations, in which one side of the seed is sterically shielded by collapsed P2VP chains, while the opposite side remains exposed and available for epitaxial extension. Subsequent introduction of a blended unimer feed comprising PFS-b-P2VP block copolymers together with PFS homopolymer, drives site-selective crystallization exclusively on the unshielded flank, producing unilateral platelet growth from the immobilized seeds. The extent of unilateral platelet formation is tunable: increasing the initial seed length from 129 to 535 nm enhances the yield from approximately 44% to 92% (Fig. 8b and c). Elevated growth temperatures (e.g., 30 °C) typically favor unilateral propagation with efficiencies of 70–90% (Fig. 8d and e). Manipulation of the corona chain length provides another control lever—shorter P2VP coronas (degree of polymerization 139) significantly reduce the yield relative to longer coronas (degree of polymerization 371), with values falling from ca. 90% to 47%.
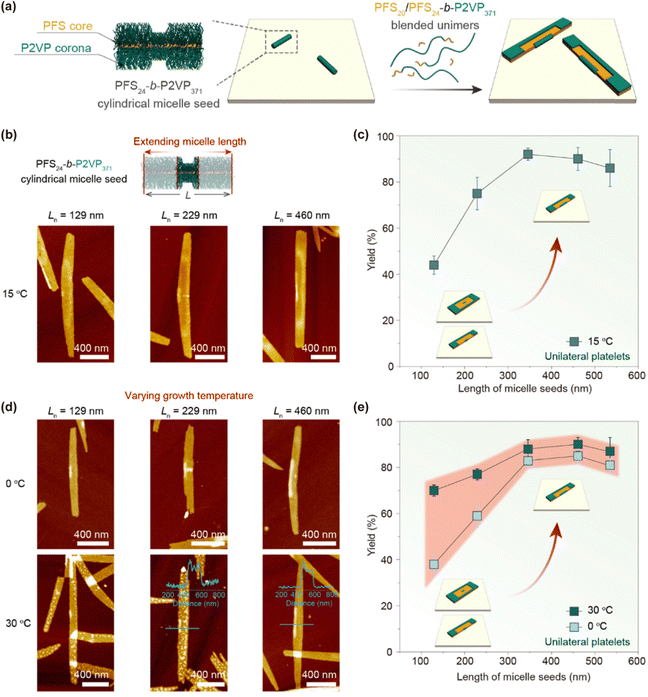 | ||
| Fig. 8 (a) Schematic of surface-initiated unilateral 2D living CDSA, with PFS24-b-P2VP371 cylindrical micelle seeds sparsely immobilized on silicon prior to addition of blended unimers. (b) AFM images of unilateral platelets grown from seeds of Ln = 129, 229, and 460 nm. (c) Unilateral platelet yield as a function of seed length. (d) AFM images of platelets obtained at 0 °C and 30 °C from immobilized seeds of the same lengths. (e) Yield dependence on growth temperature. Reproduced from ref. 83 with permission from American Chemical Society, copyright 2024. | ||
Ordered 2D micellar brush arrays can be fabricated through distinct approaches tailored to the desired surface architecture (Fig. 9). In one route, discrete slope-like platelet arrays are obtained by introducing spherical micelles (e.g., PS-b-P2VP) as inert spacers incapable of initiating CDSA. Co-immobilisation of such spheres with cylindrical seeds via static vertical deposition followed by exposure to blended unimers results in unilateral off-surface platelet growth from the cylinders, with the spheres dictating an ≈30° inclination in the platelet orientation. Alternatively, dense blade-like arrays can be realized by directionally depositing pure PFS cylindrical seeds in a compact side-by-side configuration, yielding ordered domains with a periodicity of ∼40 nm. Blended unimer addition in this case drives vertical unilateral platelet formation to heights of ≈90 nm, generating uniform, large-area arrays with a high degree of order. Beyond these single-corona structures, sequential addition of distinct blended unimers facilitates segmented unilateral platelets comprising, for example, P2VP and PDMS corona regions. Such segmented architectures expand post-assembly possibilities, including the site-selective incorporation of platinum nanoparticles into the P2VP segments, and can be rendered as freestanding arrays by attenuating surface adhesion through treatment with triethylamine.
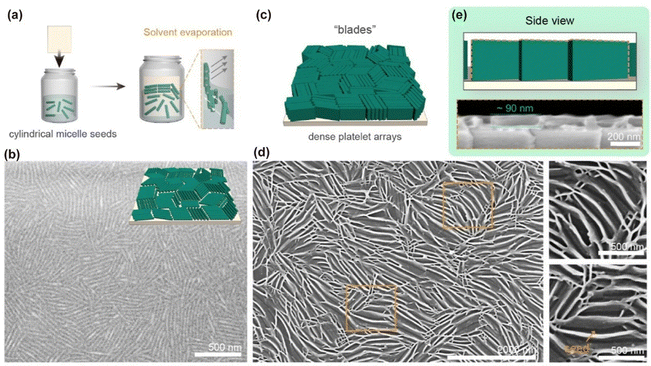 | ||
| Fig. 9 (a) Illustration for the static vertical deposition of cylindrical micelle seeds via solvent evaporation. (b) Schematic illustration and SEM image of the silicon surface that densely immobilized with PFS24-b-P2VP371 cylindrical micelle seeds (Ln = 460 nm). (c–e) Schematic illustration (c), top-view (d), and side-view (e) SEM images of the dense blade-like platelet arrays formed after the addition of blended unimers of PFS24-b-P2VP371 and PFS20. Reproduced from ref. 83 with permission from American Chemical Society, copyright 2024. | ||
4.2. Compositional diversification
The functional versatility of micellar brushes can be substantially expanded by modifying the chemical composition of the constituent block copolymers. By varying the crystalline core-forming segment, the corona-forming segment, or both, it becomes possible to engineer interfacial interactions, responsiveness to external stimuli, and tailored optoelectronic characteristics.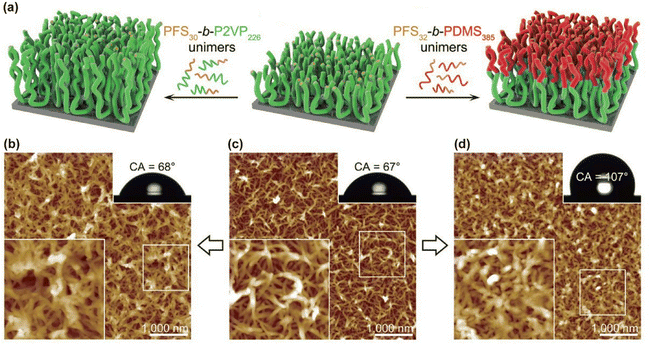 | ||
| Fig. 10 (a) Diagram illustrating the sequential growth mechanism of PFS30-b-P2VP226 micellar brushes and the formation of segmented micellar brushes. (b–d) AFM height images displaying: (b) short micellar brushes obtained by introducing 3 µL of PFS30-b-P2VP226 unimer solution; (c) elongated micellar brushes generated by an additional 3 µL of PFS30-b-P2VP226 unimer solution to the structures in (b); and (d) segmented micellar brushes produced by subsequently adding 3 µL of PFS32-b-PDMS385 unimer solution to the samples in (c). Lower-left insets present magnified views of the marked regions, while upper-right insets show photographs of water droplets on the corresponding surfaces together with the measured contact angles (CA). Reproduced from ref. 49 with permission from AAAS, Copyright 2019. | ||
 | ||
| Fig. 11 (a) Schematic showing the fabrication route: patterned immobilization of seed micelles (P3EHT22-b-P2VP80 and PFS21-b-P2VP139), followed by simultaneous, independent extension of both micellar brush types. Images depict the dual-brush patterns under daylight and UV light, and their subsequent vertical alignment upon Pt nanoparticle loading. (b) and (c) Optical photographs of patterned brushes under daylight (b) and 365 nm UV illumination (c), obtained after adding 3 µL PFS21-b-P2VP139 unimers and 6 µL P3EHT22-b-P2VP80 unimers (10 mg mL−1 in THF each). (d) and (e) AFM (d) and SEM (e) micrographs showing PFS21-b-P2VP139 brushes confined within the patterned regions. (f) SEM image of vertically aligned PFS21-b-P2VP139 brushes after treatment with 100 µL Na2PtCl6 aqueous solution. (g) and (h) AFM (g) and SEM (h) images displaying patterned P3EHT22-b-P2VP80 brushes. (i) SEM image of their vertical alignment following an identical Pt precursor treatment. Reproduced from ref. 57 with permission from Wiley, copyright 2024. | ||
5. Functionalization of micellar brushes
Micellar brushes can act not only as nanoscale templates but also as hierarchically organized functional platforms, either by exploiting the intrinsic physicochemical properties of the constituent block copolymers or by incorporating exogenous functional components. If the block copolymer itself is optoelectronically active, redox-active, semiconducting, or photoreactive, arranging it into a brush architecture can amplify such properties via enhanced crystallinity, improved orientational order of functional moieties, and facilitated charge or mass transport. These effects have been demonstrated in contexts such as organic photovoltaics, photoelectrochemical devices, and chemical/biological sensing.Beyond intrinsic functionality, a wide range of exogenous functionalization strategies can be implemented through chemical handles located in either the corona or the core domain, enabling the immobilization, transformation, or in situ generation of new materials without compromising spatial order.
5.1. Corona-mediated conjugation
The solvated corona of surface-immobilized cylindrical micellar brushes offers a highly versatile and accessible binding interface for the incorporation of exogenous functional entities. In particular, P2VP coronas present electron-donating pyridyl nitrogen atoms that are capable of forming stable coordination bonds with a wide range of metal centers. Owing to this property, Sun et al. exploited the highly solvated and permeable nature of the P2VP corona to immobilize up to 33 distinct metal species in the form of isolated single atoms, achieving remarkable loadings in the range of 8.3% to 40.9 wt% (Fig. 12a–d).53 The forest-like, open three-dimensional architecture of the brushes not only enhanced ion capture efficiency but also promoted rapid mass transport, a feature particularly beneficial during catalytically demanding processes.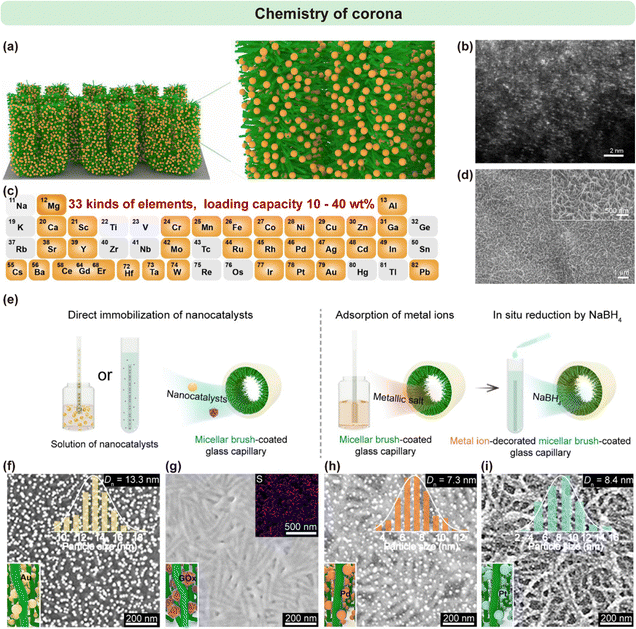 | ||
| Fig. 12 (a) Schematic representation illustrating the formation of a Pt single-atom nanoforest via mild coordination interactions within the micellar brush corona. (b) Aberration-corrected annular dark-field STEM (AC-ADF-STEM) image showing the dispersed Pt single atoms in the nanoforest architecture. (c) Overview of single-atom nanoforests incorporating 32 different metal species. (d) SEM image depicting the surface morphology of the Pt single-atom nanoforest. Reproduced from ref. 53 with permission from AAAS, Copyright 2025. (e) Schematic illustration of two approaches: direct immobilization, in which a solution of the desired nanocatalyst is introduced into a glass capillary coated with PFS30-b-P2VP226 micellar brushes, allowing spontaneous adsorption via coordination, hydrogen bonding, or electrostatic interactions; and in situ generation, in which metallic salt and NaBH4 solutions are sequentially introduced by siphoning and soaking. (f) and (g) SEM images of collapsed PFS30-b-P2VP226 brushes after direct immobilization of (f) Au NPs (inset: Size distribution) and (g) GOx (inset: EDX mapping of sulfur). (h) and (i) SEM images of collapsed brushes after in situ generation of (h) Pd NPs and (i) Pt NPs (insets: size distributions). Reproduced from ref. 51 with permission from Wiley, Copyright 2021. | ||
In their approach, in situ SI-CDSA was performed directly on nickel foam substrates, leading to the growth of PFS-b-P2VP brushes. These brushes were subsequently electrostatically complexed with PtCl42− ions, which were then stabilized as Pt single-atom “nanoforests” via strong coordination to the pyridyl groups in the corona. When the corona is engineered to carry additional charged or polar functional groups, a broader spectrum of noncovalent interactions, such as electrostatics and hydrogen bonding can be exploited for cargo immobilization.
Through these binding pathways, a diverse array of functional materials has been successfully integrated into micellar brush frameworks (Fig. 12e(i)).51 Examples include MOFs, TiO2 nanoparticles and enzymes, each imparting distinct physicochemical properties to the hybrid constructs. Such multifunctional architectures exhibit an expanded functional scope, with demonstrated utility in fields as varied as photocatalysis, molecular recognition, and chemical or biological sensing. The combination of structural tunability, high surface accessibility, and adaptable binding chemistry underscores the potential of solvated micellar brushes as a modular platform for advanced hybrid material design.
Beyond simple binding, the structural features of the polymer corona can be rationally exploited to control the spatial organization and packing density of nanoparticles with nanometer precision. In the work of Tao et al., the well-defined inter-coronal spacing of cylindrical PFS-b-P2VP micellar brushes (∼3.5 nm)91 was selected to closely match the diameter of pre-synthesized ruthenium nanoparticles (∼3.3 nm) (Fig. 13a).54 This geometric complementarity, combined with the reversible and dynamic nature of Ru–pyridine coordination chemistry, enabled the nanoparticles to infiltrate into the interstitial spaces between P2VP corona chains.
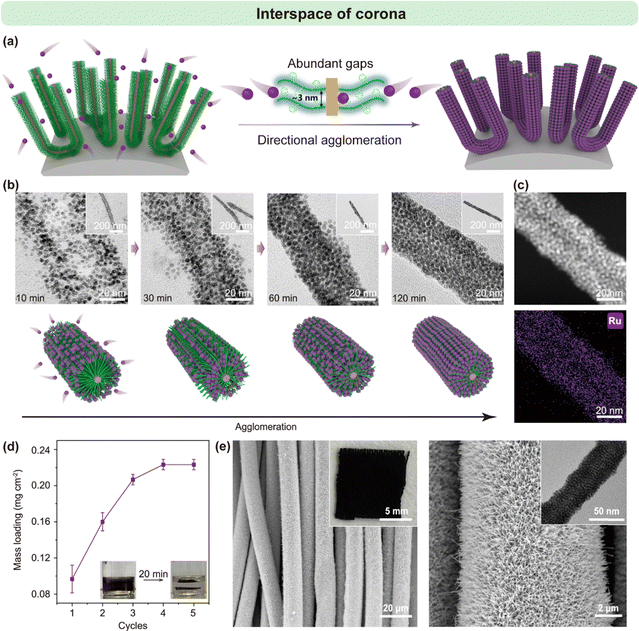 | ||
| Fig. 13 (a) Schematic representation of the fabrication process for Ru nanoparticle (Ru NP) nanoarrays on carbon cloth. (b) Illustration of Ru NP penetration into the P2VP corona of PFS29-b-P2VP230 cylindrical micelles, accompanied by TEM images of representative samples at various fabrication stages. (c) High-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) and energy-dispersive spectroscopy (EDS) mapping of a nanowire densely packed with Ru NPs. (d) Plot showing the dependence of Ru NP mass loading on the number of immersion cycles; inset photographs depict the color change in the Ru NP suspension when micellar brush-modified carbon cloth is introduced. (e) SEM images of Ru NP nanoarrays on carbon cloth. Insets include a photograph of a piece of carbon cloth uniformly coated with the nanoarray and a TEM image of a single nanowire composed of closely stacked Ru NPs. Reproduced from ref. 54 with permission from Spring Nature, copyright 2025. | ||
Upon coordination-driven infiltration, local rearrangements of the corona led to a dramatic contraction of the nanoparticle distance from 5.1 ± 1.9 nm in the initial loosely bound state to 0.5 ± 0.08 nm in the final densely packed configuration (Fig. 13b and c). Such close-proximity assembly goes well beyond conventional steric or electrostatic packing, as it requires both spatial matching and dynamic bonding to overcome entropic penalties and corona chain crowding.
The methodology offers several important advantages. First, it allows for collective particle positioning within the polymer template without the need for high-temperature annealing or solvent evaporation-induced aggregation. Second, it enables the translation of nanoscale spatial order into extended, substrate-scale architectures (Fig. 13d and e), manifested in this case as vertically aligned, high-density nanowire arrays of various metals (Ru, Ir, Pt, Rh). By selecting nanoparticles and coronas of appropriate dimensions and coordination chemistries, the approach would be generalized to a broad range of inorganic materials and polymer brush architectures.
In addition, the sub-nanometer interparticle gaps created via this strategy are highly relevant for applications in plasmonics, catalysis, and sensing, where strong near-field coupling or cooperative surface reactions are desired. Control over vertical alignment and periodicity across macroscopic substrates also opens potential pathways toward scalable fabrication of functional nanoscale devices. This study thus demonstrates that the corona is not merely a passive steric barrier, but a tunable, functional scaffold that can mediate structure-specific infiltration and drive the self-organization of nanoparticles into unprecedented dense and ordered arrays.
5.2. Core-driven transformation
The crystalline PFS core within micellar brushes provides a unique reactive scaffold owing to the organometallic nature of its ferrocene units, which exhibit highly reversible Fe(II)/Fe(III) redox chemistry.93–95 This intrinsic property can be strategically harnessed to in situ convert the polymeric core into ordered inorganic nanostructures without disrupting the overall spatial arrangement of the brush array. In the pioneering work of Lu et al.,92 oxidative treatments using strong oxidants such as potassium permanganate (KMnO4), potassium ferrate (K2FeO4), dipotassium tetrachloroplatinate (K2PtCl4), potassium tetrachloropalladate (K2PdCl4), potassium pentachlororuthenate (K2RuCl5), gold chloride (AuCl3), and rhodium chloride (RhCl3) were applied to PFS-containing cylindrical micelles anchored on surfaces. During oxidation, the Fe(II) centres in ferrocene are oxidised to Fe(III), triggering cleavage of the Si–C backbone and subsequent precipitation or deposition of the corresponding inorganic species directly within the confinement of the original micellar core domain. The choice of oxidant dictates the chemical identity of the resulting nanostructure: KMnO4 produces well-aligned manganese oxide nanowires (Fig. 14a and b), and noble-metal salts such as AuCl3 can be reduced in situ to generate metallic nanoparticles. Importantly, because the transformation proceeds under shape-preserving conditions defined by the crystalline micellar geometry, the resulting inorganic phase inherits the original nanoscale ordering, producing highly uniform, vertically aligned wire- or rod-like arrays (Fig. 14c and d). Such core-to-inorganic conversions open a direct route to integrate functional metal oxides or noble metals into micellar brush platforms, thereby extending their utility to applications in electrocatalysis, photocatalysis, sensing, and advanced energy-storage systems such as supercapacitors and rechargeable batteries. | ||
| Fig. 14 (a) Schematic illustration of the formation of nanorods densely coated with thin MnO2 nanosheets via a direct redox reaction between PFS-b-P2VPq cylindrical micelles and KMnO4. (b) TEM images depicting the progressive deposition and growth of manganese oxide on PFS-b-P2VPq cylindrical micelles during the redox process. (c) Schematic representation of MnO2 nanoarray fabrication, involving surface-initiated living growth of PFS-b-P2VPq micellar brushes followed by an in situ redox reaction with KMnO4 under ambient conditions. (d, e) Top-view and (f) cross-sectional SEM images of the MnO2 nanoarray on a silicon wafer. Reproduced from ref. 92 with permission from American Chemical Society, copyright 2025. | ||
5.3. In situ transformation and post-assembly processing
After the micellar brush arrays have been immobilized onto a substrate, their exogenous or guest components can be deliberately transformed or modified to access new material phases or functionalities. Such transformations are typically induced by thermal treatment, annealing under controlled atmospheres, or post-assembly chemical reactions, while relying on the pre-organized brush morphology to spatially confine the process and preserve long-range order. This approach enables the conversion from a soft polymeric/scaffolded state into functional inorganic or hybrid nanostructures without sacrificing the nanoscale precision imparted by the original self-assembly.A representative example was reported by Tao et al., in which metal nanoparticles distributed within the brush framework underwent thermally driven coalescence during high-temperature annealing.54 At sufficiently elevated temperatures, surface diffusion of the metal atoms caused discrete nanoparticles to merge preferentially along the brush axis, leading to the formation of continuous, nearly single-crystalline metallic nanowires while maintaining their spatial arrangement (Fig. 15a and b). This morphological consolidation had a profound impact on the electronic properties: the direct electrical pathways and reduced grain boundary scattering produced conductivity increases of up to four orders of magnitude compared to the as-prepared state, alongside a marked enhancement of intrinsic metallic behavior (Fig. 15c). By co-incorporating different elemental nanoparticles prior to annealing, Tao and co-workers further demonstrated the fabrication of compositionally controlled alloy nanowire arrays, thereby providing a general route to ordered multimetallic or alloyed architectures at the nanoscale (Fig. 15d and e). Such a strategy holds particular promise for nanoelectronic interconnects, plasmonic waveguides, and catalytically active surfaces.
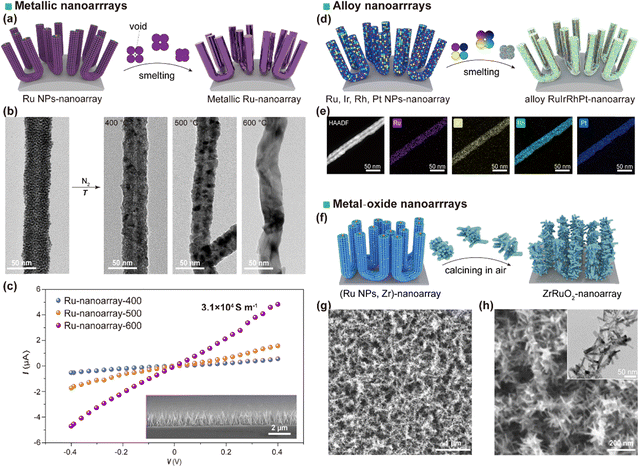 | ||
| Fig. 15 (a) Schematic representation depicting the transformation of a Ru NP-nanoarray into a continuous metallic Ru nanoarray via smelting. (b) TEM images comparing the as-prepared Ru NP-nanoarray with metallic Ru-nanoarrays treated at different temperatures (T = 400, 500, and 600 °C). (c) Electrical conduction measurements of metallic Ru-nanoarray-T. (d) Schematic illustrating the conversion of (Ru, Ir, Rh, Pt) NP-nanoarrays into an alloy RuIrRhPt-nanoarray. (e) HAADF-STEM and EDS elemental mapping images of the alloy nanoarray, showing uniform distributions of Ru, Ir, Rh, and Pt. Reproduced from ref. 54 with permission from Spring Nature, Copyright 2025. (f) Schematic illustrating the fabrication of a ZrRuO2 nanoarray using micellar brushes as a structural template. (g) and (h) SEM images of the ZrRuO2 nanoarray grown on a Ti fiber felt, and TEM image detailing its nanostructure. Reproduced from ref. 96 with permission from Wiley, copyright 2025. | ||
Thermal treatment can also drive oxidative transformations when performed in ambient air (Fig. 15f–h).96 In the case of brushes containing metals or metal-ion-loaded cores, controlled thermal oxidation yields ordered arrays of metal-doped metal-oxide nanostructures whose composition, crystallinity, and doping level can be tuned through the precursor loading and annealing profile. This pathway grants access to functional oxides including semiconducting, magnetic, or catalytic variants, whilst retaining the nanoscale order inherited from the micellar template.
Beyond purely thermal transformations, solution-phase post-assembly reactions have also been harnessed to impart new structural and chemical complexity. As demonstrated by Wang et al., micellar brushes can be sequentially immersed in metal-precursor solutions to load the desired cations into predefined regions, followed by treatment with organic linker solutions under mild conditions (Fig. 16).97 This stepwise coordination chemistry enables the in situ crystallization of MOF nanolayers within or atop the brush-templated sites. The resulting MOF coatings exhibit exceptionally high positional registry and uniform coverage across large substrate areas, demonstrating the ability of the brush scaffold to direct the assembly of porous crystalline materials. Such MOF–polymer hybrid arrays combine the chemical selectivity and sorption capacity of MOFs with the ordered architecture of CDSA-derived brushes, opening avenues in selective sensing, membrane separations, and host–guest catalysis.
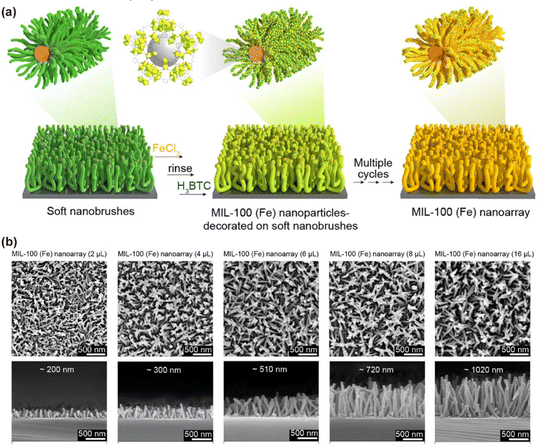 | ||
| Fig. 16 (a) Schematic representation of the fabrication process for MOF nanoarrays. (b) Top-view and cross-sectional SEM images of MIL-100(Fe) nanoarrays templated by the corresponding soft nanobrushes. Reproduced from ref. 97 with permission from Spring Nature, copyright 2022. | ||
By integrating in situ phase transformations with the positional fidelity of micellar brush arrays, these post-assembly strategies offer a powerful route to engineer nanoscale materials and heterostructures with precisely orchestrated composition, crystallinity, and functional performance.
6. Applications
The exquisite structural and functional control afforded by SIL-CDSA has positioned it as a compelling platform for a diverse range of applications, from energy conversion and storage to advanced biomedical interfaces and electronic devices.6.1. Catalytic interfaces
The ability to create high-density, vertically-aligned arrays of nanostructures with high surface area makes SIL-CDSA an ideal method for fabricating advanced catalytic interfaces. The resulting vertically-aligned micellar brush arrays offer a high surface area-to-volume ratio and rationally designed pathways for mass transport, addressing key challenges in heterogeneous catalysis.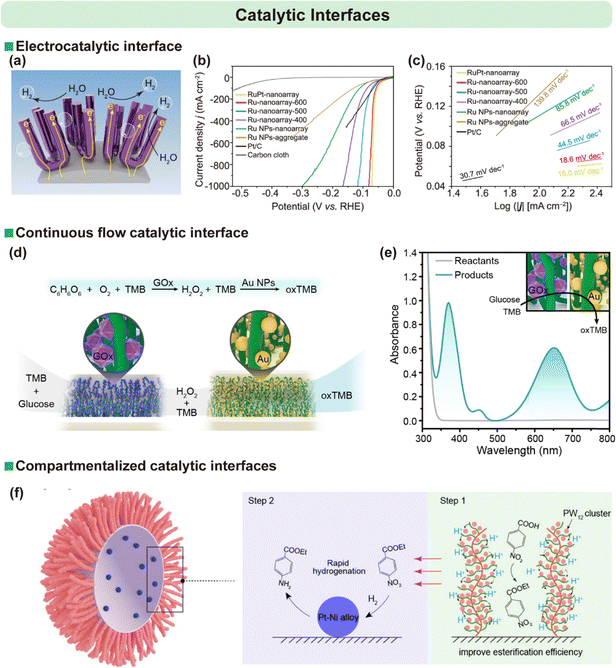 | ||
| Fig. 17 (a) Schematic representation of a metallic Ru nanoarray designed for the hydrogen evolution reaction (HER). (b) iR-corrected polarization curves in 0.5 M H2SO4 solution for alloy RuPt nanoarray, metallic Ru nanoarray-T, Ru NP nanoarray, Ru NP aggregates, commercial Pt/C-decorated carbon cloth, and pristine carbon cloth. (c) Tafel plots derived from the polarization curves shown in (b). Reproduced from ref. 54 with permission from Spring Nature, Copyright 2025. (d) and (e) Illustration and UV-vis spectra for a cascade reaction in PBS (pH 5.05) sequentially catalyzed by GOx-functionalized micellar brushes and Au NP-functionalized micellar brushes. Reproduced from ref. 51 with permission from Wiley, copyright 2021. (f) Schematic diagram of the sequential tandem conversion of p-nitrobenzoic acid within a microreactor engineered via surface modification strategies. Reproduced from ref. 55 with permission from Wiley, copyright 2025. | ||
6.2. Bioinspired interfaces
Micellar brushes are employed as core templates to engineer bioinspired interfaces for high-performance piezoresistive electronic skins (E-skins), effectively addressing the challenge of fabricating well-defined nanostructures on soft substrates under mild processing conditions.52 To prepare the substrate, PDMS–CB films featuring micrometer-scale wavy surfaces were obtained by casting a PDMS/CB mixture onto sandpaper molds, followed by thermal curing (Fig. 18a). The microdome dimensions within the PDMS–CB films were precisely tuned by selecting sandpaper with mesh sizes ranging from 200 to 1000. Subsequently, micelle seeds were uniformly deposited across the PDMS–CB surface. Due to their nanoscale size, the seeds were able to conformally coat the irregular topography, enabling the growth of a dense micellar brush upon addition of PFS32-b-P2VP251 unimers. Platinum nanoparticle (Pt NP)–based nanoarrays were then formed via in situ aggregation within the micellar domains, leading to large-area hierarchical architectures after freeze-drying (Fig. 18b).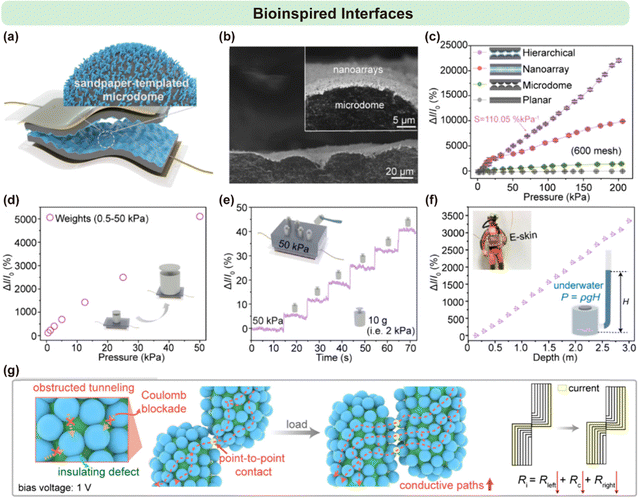 | ||
| Fig. 18 (a) Schematic representation of a hierarchically structured electronic skin (E-skin) featuring micelle/Pt nanoarrays on the surface of a microdome-patterned PDMS–CB film. (b) Cross-sectional SEM images of the hierarchically structured film prepared using 600-mesh sandpaper. (c) Relative current variations for different E-skin configurations within 200 kPa pressure range. Bias voltage = 1 V. (d) Static weight pressure sensing at applied pressures of 0.5, 1.25, 2.5, 5, 12.5, 25, and 50 kPa. (e) Detection of an additional small pressure of 2 kPa (weight: 10 g, applied area: 5 × 10−5 m2) under a preload pressure of 50 kPa (weight: 250 g, applied area: 5 × 10−5 m2). (f) Relative current change of the hierarchically structured E-skin under varying water levels. Inset: schematic diagram illustrating hydrostatic pressure detection. (g) Schematic illustrating electron transport and conductive pathways during loading between a pair of interlocked micelle/Pt nanoarrays composed of various nanoparticles. Ri: total resistance of the interlocked nanoarrays under load; Rleft and Rright: internal resistances of each nanoarray; Rc: contact resistance. Reproduced from ref. 52 with permission from American Chemical Society, copyright 2025. | ||
The resulting E-skins, incorporating these hierarchical structures, were assessed for their piezoresistive performance under applied pressures from 0 to 200 kPa. Devices with the smallest microdome features (1000- and 800-mesh) exhibited a characteristic two-stage increase in relative current change, with a limited linear sensing range. Increasing the microdome size (600–200 mesh) significantly enhanced linearity, with the E-skin fabricated from 600-mesh sandpaper achieving an outstanding linear response across the entire 0.1–200 kPa range and an impressive sensitivity of 110.05% kPa−1 (Fig. 18c).
To further validate the linear pressure detection capability, a sequence of static weights was applied individually to the surface of the hierarchically structured E-skin. The relative current varied linearly with increasing pressure in the 0.5–50 kPa range (Fig. 18d). Under a high base load of 50 kPa, the device was able to resolve successive 2 kPa increments with remarkable precision (Fig. 18e). Additionally, when immersed at various depths in water, the sensor accurately detected hydrostatic pressure variations (Fig. 18f). This combination of broad-range linearity, high sensitivity, and excellent resolution highlights the potential of such hierarchical E-skins for wearable sensing systems in next-generation aquatic or snorkeling applications.
6.3. Devices
The integration of SIL-CDSA architectures into functional devices is a rapidly advancing frontier. The oriented nature of the nanostructures is particularly beneficial for charge-based devices. Tao et al. applied SIL-CDSA to fabricate ruthenium-containing nanowire brush arrays directly on PEM for electrolytic water splitting (Fig. 19a and b).54,96 The oriented, high-surface-area arrangement facilitated efficient proton conduction and catalytic exposure, improving the performance of PEM electrolyzers. Similarly, Sun et al. developed dense “nanoforests” of cylindrical micellar brushes as electrode interfaces in AEM electrolyzers (Fig. 19c and d).53 By tailoring the corona chemistry to promote hydroxide ion transport and catalyst dispersion, these vertically aligned structures enhanced reaction kinetics and reduced polarization losses.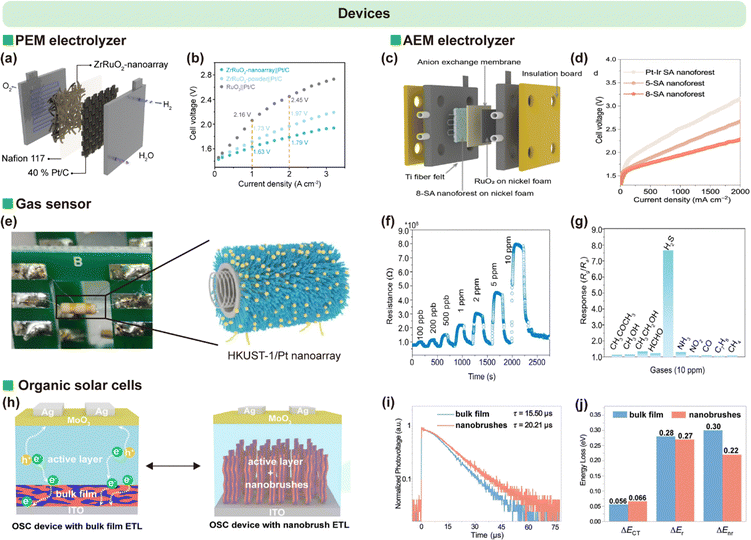 | ||
| Fig. 19 (a) Schematic diagram of a PEM electrolyzer. (b) Current–voltage (I–V) curves of electrolyzers employing ZrRuO2 nanoarray, ZrRuO2 powder, and commercial RuO2 as the anode, respectively. Reproduced from ref. 54 with permission from Spring Nature, Copyright 2025. (c) Schematic illustration of the AEM electrolyzer device. (d) I–V curves of the AEM electrolyzer using Pt–Ir SA, 5-SA, and 8-SA nanoforests as the cathodic catalyst and commercial RuO2 as the anodic catalyst at 60 °C. Reproduced from ref. 53 with permission from AAAS, Copyright 2025. (e) Schematic of a side-heated HKUST-1 nanoarray-based gas sensor. (f) Response–recovery curves of the HKUST-1 nanoarray/Pt-0.36 sensor to H2S at various concentrations at 200 °C. (g) Responses of the HKUST-1 nanoarray/Pt-0.36 sensor to different chemical vapors (10 ppm) at 200 °C. Reproduced from ref. 97 with permission from Spring Nature, Copyright 2022. (h) Schematic illustration of charge transport in inverted organic solar cells (OSCs) based on bulk films and nanobrushes. (i) Transient photovoltage decay curves of bulk film–based and nanobrush–based OSCs. (j) Detailed energy loss analysis in bulk film–based and nanobrush–based OSCs. Reproduced from ref. 56 with permission from American Chemical Society, Copyright 2025. | ||
Wang et al. exploited SIL-CDSA to grow MOF arrays on the surface of ceramic tubes (Fig. 19e–g).97 The oriented MOF coating provided uniform pore channels and high accessible surface area, enabling sensitive and selective gas detection with rapid response times. Yang et al. employed fully conjugated PF-b-QPT micellar brushes as structurally ordered, photoactive interlayers in organic solar cells (Fig. 19h–j).56 The brush orientation promoted directional charge transport, while the conjugated corona improved light absorption and exciton dissociation, resulting in enhanced power conversion efficiency relative to disordered control layers.
These examples collectively illustrate the versatility of SIL-CDSA in device engineering: the method offers nanostructural precision, orientation control, and compositional diversity in a single platform. Continued development of this approach is expected to open pathways to high-performance systems across energy conversion, sensing, and optoelectronic technologies.
7. Conclusion and perspectives
Surface-initiated living crystallization-driven self-assembly (SIL-CDSA) has evolved rapidly from a nascent concept into a robust and versatile strategy for precision nanofabrication on surfaces. As outlined in this review, the field has made significant advances in controlling the dimensions, morphology, and functionality of surface-grafted nanostructures. The ability to produce dense, vertically aligned arrays of micelles and platelets with tunable height, width, and area has provided unprecedented architectural control. Complex structures such as asymmetric platelets and patterned hierarchical assemblies, have been realized alongside the programmed integration of multi-component systems. By harnessing both intrinsic polymer functionalities and post-assembly modification of the micellar corona, these architectures have found application in catalysis, electronics, and biomedicine. Landmark achievements, including the fabrication of patterned dual-micellar brushes, and the construction of high-performance electrodes and microreactors, highlight the transformative potential of this bottom-up approach for next-generation materials and devices.Despite this progress, several fundamental challenges and exciting opportunities remain, which will define the future trajectory of the field.
1. Expanding the materials library: a significant portion of seminal SIL-CDSA work has relied on a relatively small set of well-behaved, crystallizable BCPs, most notably those based on PFS. While systems based on conjugated polymers like P3EHT and PF are emerging, there is a critical need to expand the library of materials amenable to SIL-CDSA. This involves designing new BCPs with diverse crystallizable cores (e.g., biodegradable polyesters, high-performance engineering polymers) and functional coronas. Success here will broaden the application space to areas like transient electronics and high-temperature materials.
2. Deepening mechanistic understanding: while the general principles of SIL-CDSA are understood, a deeper, quantitative understanding of the kinetics and thermodynamics at the solid–liquid interface is needed. How does the chemical and physical nature of the substrate (e.g., hydrophobicity, surface energy, crystallinity) precisely influence unimer diffusion, epitaxial addition rates, and the stability of the resulting structures? Advanced in situ characterization techniques, such as liquid-cell atomic force microscopy and surface-sensitive scattering methods, combined with sophisticated computational modeling, will be essential to unravel these complex interactions and develop predictive models for SIL-CDSA.
3. Achieving true three-dimensional complexity: current SIL-CDSA methods excel at creating dense 2.5D structures—essentially extruded 2D patterns. The next grand challenge is to achieve true 3D architectural control, enabling the fabrication of structures with programmed complexity not just on the surface but in the vertical dimension as well. This might involve techniques that can switch growth direction, alternate between 1D and 2D growth modes, or integrate orthogonal self-assembly processes. The CDSA-PISA concept is a step in this direction, but realizing complex, interconnected 3D networks remain an open frontier.
4. Scalability and manufacturing: to move from the laboratory to industrial application, fabrication methods must be scalable, cost-effective, and compatible with existing manufacturing platforms. While flow-based synthesis and rapid self-assembly protocols are promising, significant work is needed to translate the precise, multi-step process of seed immobilization and growth to large-area substrates (e.g., wafer-scale). Developing parallel, high-throughput techniques for surface patterning and growth will be paramount for the technological maturation of SIL-CDSA.
In conclusion, SIL-CDSA stands at a pivotal juncture. It has firmly established its credentials as a premier tool for bottom-up nanofabrication on surfaces. The future of the field lies in diversifying its material palette, deepening its mechanistic foundations, elevating its architectural complexity into the third dimension, and engineering scalable pathways to manufacturing. By addressing these challenges, SIL-CDSA is poised to deliver a new generation of smart, functional interfaces that will drive innovation across science and technology.
Author contributions
Jiawei Tao: conceptualization, writing – original draft; Chenchen Gao, Yao Lu: writing – original draft; Zhenyan Ji, Peiwen Wang, Sen Lv, Xiaomin Cai, Jingwen Zhang: writing – review & editing; Huibin Qiu: conceptualization, funding acquisition, supervision, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data was generated or analyzed as part of this review.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (22425203, 52403329, 22561160131), the Innovation Program of Shanghai Municipal Education Commission (202101070002E00084) and the Natural Science Foundation of Shanghai (25ZR1402249).Notes and references
- P. Zhang, X. Wang, Y. Zhang, Y. Wei, N. Shen, S. Chen and B. Xu, Adv. Funct. Mater., 2024, 34, 2402307 CrossRef CAS.
- C. Liu, H. Ma, S. Yuan, Y. Jin and W. Tian, ACS Nano, 2025, 19, 2047–2069 CrossRef CAS PubMed.
- L. Ma and H. Yang, Bio-Des. Manuf., 2023, 6, 735–741 CrossRef.
- J. Cai, C. Li and I. Manners, Angew. Chem., Int. Ed., 2025, 64, e202501552 CrossRef CAS PubMed.
- L. Xiao, T. Xia, J. Zhang, S. J. Parkinson, J. Y. Rho, A. P. Dove and R. K. O’. Reilly, Nat. Synth., 2025, 4, 808–815 CrossRef CAS.
- N. R. Glavin, P. M. Ajayan and S. Kar, Adv. Mater., 2023, 35, 2204928 CrossRef CAS PubMed.
- C. Mao, Adv. Mater., 2025, 37, 2418618 CrossRef CAS PubMed.
- S. Park, S.-Y. Kang, S. Yang and T.-L. Choi, J. Am. Chem. Soc., 2024, 146, 19369–19376 CrossRef CAS PubMed.
- F. A. Escobedo, A. Haji-Akbari and S. Sharma, J. Chem. Theory Comput., 2024, 20, 1503–1504 CrossRef CAS PubMed.
- S.-H. Yu, R. O’Reilly, L. Jiang and N. A. Kotov, Acc. Chem. Res., 2022, 55, 1783–1784 CrossRef CAS PubMed.
- Y. Song, Nat. Chem. Biol., 2024, 20, 1549 CrossRef CAS PubMed.
- J. Cornel Erik, J. Jiang, S. Chen and J. Du, CCS Chem, 2020, 3, 2104–2125 CrossRef.
- S. Pearce, H. K. MacKenzie, H. Shaikh, Z. Liu, R. Harniman, J. C. Eloi, S. Davis, R. M. Richardson, E. A. LaPierre, I. Manners and Y. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202506872 CrossRef CAS PubMed.
- J. Zhang, T. Xiao, Z. Liu, Y. Yin, C. Li, F. Xu and Y. Mai, J. Am. Chem. Soc., 2025, 147, 19148–19156 CrossRef CAS PubMed.
- R. A. Scanga, A. Shahrokhinia, J. Borges, S. H. Sarault, M. B. Ross and J. F. Reuther, J. Am. Chem. Soc., 2023, 145, 6319–6329 CrossRef CAS PubMed.
- J. Gu, Z. Chu, B. Zheng and Z. Tong, ACS Macro Lett., 2025, 14, 645–657 CrossRef CAS PubMed.
- J. Jiang and M. A. Winnik, Acc. Chem. Res., 2025, 58, 1683–1695 CrossRef CAS PubMed.
- S. Mahapatra, P. Dey and G. Ghosh, Polym. J., 2024, 56, 949–975 CrossRef CAS.
- A. Sahu, S. Mahapatra, P. Dey and G. Ghosh, Macromol. Chem. Phys., 2025, 226, 2400426 CrossRef CAS.
- Y. Guo, T. Xia, V. Walter, Y. Xie, J. Y. Rho, L. Xiao, R. K. O’Reilly and M. I. Wallace, Nat. Commun., 2025, 16, 2672 CrossRef CAS PubMed.
- T. Xia, L. Xiao, Y. Xie, A. P. Dove and R. K. O’Reilly, Chem. Rev., 2025, 125, 10105–10178 CrossRef CAS PubMed.
- L. Liu, L. Zhu, Z. Chu and Z. Tong, Macromolecules, 2023, 56, 5984–5992 CrossRef CAS.
- L. Zhu, L. Liu, S. Varlas, R.-Y. Wang, R. K. O’Reilly and Z. Tong, ACS Nano, 2023, 17, 24141–24153 CrossRef CAS PubMed.
- H. Qiu, G. Russo, P. A. Rupar, L. Chabanne, M. A. Winnik and I. Manners, Angew. Chem., Int. Ed., 2012, 51, 11882–11885 CrossRef CAS PubMed.
- Z. Deng, Y. Sun, Z. Chen, Y. Zhao and A. Chen, Macromolecules, 2025, 58, 2345–2356 CrossRef CAS.
- J. Nie, J. Zhao, L. Li, T. Zhou and G. Guerin, Angew. Chem., Int. Ed., 2025, 64, e202511515 CrossRef CAS PubMed.
- Y.-N. Pan, C.-C. Ye, S.-L. Huang, C. Wang, M.-Y. Han and L. Xu, Angew. Chem., Int. Ed., 2025, 64, e202418131 CrossRef CAS PubMed.
- Y. Gao, H. Qiu, H. Zhou, X. Li, R. Harniman, M. A. Winnik and I. Manners, J. Am. Chem. Soc., 2015, 137, 2203–2206 CrossRef CAS PubMed.
- J. Nie, Z. Wang, L. Xia, X. Huang, G. Lu and C. Feng, Eur. Polym. J., 2023, 197, 112384 CrossRef CAS.
- J. R. Finnegan, D. J. Lunn, O. E. C. Gould, Z. M. Hudson, G. R. Whittell, M. A. Winnik and I. Manners, J. Am. Chem. Soc., 2014, 136, 13835–13844 CrossRef CAS PubMed.
- F. Teng, B. Xiang, L. Liu, S. Varlas and Z. Tong, J. Am. Chem. Soc., 2023, 145, 28049–28060 CrossRef CAS PubMed.
- Z. Tong, Y. Xie, M. C. Arno, Y. Zhang, I. Manners, R. K. O’Reilly and A. P. Dove, Nat. Chem., 2023, 15, 824–831 CrossRef CAS PubMed.
- H. Qiu, Y. Gao, C. E. Boott, O. E. C. Gould, R. L. Harniman, M. J. Miles, S. E. D. Webb, M. A. Winnik and I. Manners, Science, 2016, 352, 697–701 CrossRef CAS PubMed.
- R. Deng, X. Mao, S. Pearce, J. Tian, Y. Zhang and I. Manners, J. Am. Chem. Soc., 2022, 144, 19051–19059 CrossRef CAS PubMed.
- A. Nazemi, X. He, L. R. MacFarlane, R. L. Harniman, M.-S. Hsiao, M. A. Winnik, C. F. J. Faul and I. Manners, J. Am. Chem. Soc., 2017, 139, 4409–4417 CrossRef CAS PubMed.
- Y. Su, Y. Jiang, L. Liu, Y. Xie, S. Chen, Y. Wang, R. K. O’Reilly and Z. Tong, Macromolecules, 2022, 55, 1067–1076 CrossRef CAS.
- T. Xia, L. Xiao, K. Sun, J. Y. Rho, Y. Xie, S. J. Parkinson, L. Sangroniz, J. Zhang, J. Lin, A. J. Müller, L. Gao, A. P. Dove and R. K. O’Reilly, Macromolecules, 2024, 57, 11210–11220 CrossRef CAS.
- F. Teng, J. Gu, Z. Chu and Z. Tong, Macromolecules, 2025, 58, 249–255 CrossRef CAS.
- Z. M. Hudson, C. E. Boott, M. E. Robinson, P. A. Rupar, M. A. Winnik and I. Manners, Nat. Chem., 2014, 6, 893–898 CrossRef CAS PubMed.
- H. Qiu, Z. M. Hudson, M. A. Winnik and I. Manners, Science, 2015, 347, 1329–1332 CrossRef CAS PubMed.
- X. Li, Y. Gao, C. E. Boott, D. W. Hayward, R. Harniman, G. R. Whittell, R. M. Richardson, M. A. Winnik and I. Manners, J. Am. Chem. Soc., 2016, 138, 4087–4095 CrossRef CAS PubMed.
- X. Li, Y. Gao, C. E. Boott, M. A. Winnik and I. Manners, Nat. Commun., 2015, 6, 8127 CrossRef PubMed.
- X. Li, Y. Gao, R. Harniman, M. Winnik and I. Manners, J. Am. Chem. Soc., 2016, 138, 12902–12912 CrossRef CAS PubMed.
- H. K. MacKenzie, Y. Zhang, W. Zheng, H. Shaikh, L. R. MacFarlane, R. A. Musgrave and I. Manners, J. Am. Chem. Soc., 2024, 146, 18504–18512 CrossRef CAS PubMed.
- B. Jin, Q. Li, L. Hu, Q. Liu, Y. Chen, Y. Luo, S. Chi and X. Li, Angew. Chem., Int. Ed., 2023, 62, e202219067 CrossRef CAS PubMed.
- C. Zhang, J. Lin, L. Wang and L. Gao, J. Phys. Chem. Lett., 2022, 13, 6215–6222 CrossRef CAS PubMed.
- X. Wang, K. Liu, A. C. Arsenault, D. A. Rider, G. A. Ozin, M. A. Winnik and I. Manners, J. Am. Chem. Soc., 2007, 129, 5630–5639 CrossRef CAS PubMed.
- J. Feng, Y. Qiu, H. Gao and Y. Wu, Acc. Chem. Res., 2024, 57, 222–233 CrossRef CAS PubMed.
- J. Cai, C. Li, N. Kong, Y. Lu, G. Lin, X. Wang, Y. Yao, I. Manners and H. Qiu, Science, 2019, 366, 1095–1098 CrossRef CAS PubMed.
- T. Gädt, N. S. Ieong, G. Cambridge, M. A. Winnik and I. Manners, Nat. Mater., 2009, 8, 144–150 CrossRef PubMed.
- G. Lin, J. Cai, Y. Sun, Y. Cui, Q. Liu, I. Manners and H. Qiu, Angew. Chem., Int. Ed., 2021, 60, 24637–24643 CrossRef CAS PubMed.
- Y. Lu, Y. Xing, J. Tao, S. Wang, G. Lin, H. Zhang and H. Qiu, Chem. Mater., 2025, 37, 3274–3283 CrossRef CAS.
- Y. Sun, Y. Zang, B. He, G. Lin, Z. Liu, L. Yang, L. Chen, L. Li, X. Liu, C. Shen and H. Qiu, Sci. Adv., 2025, 11, eadq2948 CrossRef CAS PubMed.
- J. Tao, R. Gao, G. Lin, C. Chu, Y. Sun, C. Yu, Y. Ma and H. Qiu, Nat. Commun., 2025, 16, 4996 CrossRef CAS PubMed.
- S. Wang, Y. Wang, B. Fang, S. Zhang, H. Zhou, Y. Lu, C. Shen and H. Qiu, Angew. Chem., Int. Ed., 2025, 64, e202508249 CrossRef CAS PubMed.
- L. Yang, C. Li, N. An, J. Gao, Y. Wei, J. Qiao, J. Dai, N. Yu, Y. Sun, Q. Lin, X. Zhang, J. Zhang, Z. Tang, X. Hao, G. Lu, Z. Wei, I. Manners, Y. Kuang, H. Huang, A. Facchetti and H. Qiu, J. Am. Chem. Soc., 2025, 147, 6763–6771 CrossRef CAS PubMed.
- Y. Zhang, L. Yang, Y. Sun, G. Lin, I. Manners and H. Qiu, Angew. Chem., Int. Ed., 2024, 63, e202315740 CrossRef CAS PubMed.
- R. L. Harniman, S. Pearce and I. Manners, J. Am. Chem. Soc., 2022, 144, 951–962 CrossRef CAS PubMed.
- Z. Sun, Z. Xu, M. Ding, L. Wang, L. Zhao, P. Sui, G. Li, H. Jin, Y. Zhou and S. Lin, Angew. Chem., Int. Ed., 2025, 64, e202503104 CrossRef CAS PubMed.
- J. Tan, J. Zhang, M. Chen, J. Cen, W. Wang, C. You, X. Hu, J. Hu and S. Liu, Angew. Chem., Int. Ed., 2025, e22563 Search PubMed.
- Z. Li, S. Wang, Y. Ma, J. C. M. van Hest and H. Che, Angew. Chem., Int. Ed., 2025, e22097 Search PubMed.
- J. Xiao and J. Du, J. Am. Chem. Soc., 2020, 142, 6569–6577 CrossRef CAS PubMed.
- T. Wang, Y. Li, E. J. Cornel, C. Li and J. Du, ACS Nano, 2021, 15, 9027–9038 CrossRef CAS PubMed.
- E. Equy, E. Ibarboure, E. Grelet and S. Lecommandoux, J. Am. Chem. Soc., 2025, 147, 9727–9738 CrossRef CAS PubMed.
- Z. Sun, Z. Xu, P. Wu, L. Wang, M. Ding, L. Zhao, Y. Zheng, Y. Zhou, S. Lin and H. Jin, Angew. Chem., Int. Ed., 2025, 64, e202507852 CrossRef CAS PubMed.
- Y. Luo, H. Wu, X. Zhou, J. Wang, S. Er, Y. Li, P. L. W. Welzen, R. A. J. F. Oerlemans, L. K. E. A. Abdelmohsen, J. Shao and J. C. M. van Hest, J. Am. Chem. Soc., 2023, 145, 20073–20080 CrossRef CAS PubMed.
- M.-Y. Li, L.-M. Wu, H.-Q. Xie and B. Shen, Sci. Adv., 2025, 11, eadv8999 CrossRef CAS PubMed.
- Y. Cha, C. Jarrett-Wilkins, M. A. Rahman, T. Zhu, Y. Sha, I. Manners and C. Tang, ACS Macro Lett., 2019, 8, 835–840 CrossRef CAS PubMed.
- J. B. Gilroy, D. J. Lunn, S. K. Patra, G. R. Whittell, M. A. Winnik and I. Manners, Macromolecules, 2012, 45, 5806–5815 CrossRef CAS.
- Y. Zhang, S. Pearce, J.-C. Eloi, R. L. Harniman, J. Tian, C. Cordoba, Y. Kang, T. Fukui, H. Qiu, A. Blackburn, R. M. Richardson and I. Manners, J. Am. Chem. Soc., 2021, 143, 5805–5814 CrossRef CAS PubMed.
- T. Xia, Z. Tong, Y. Xie, M. C. Arno, S. Lei, L. Xiao, J. Y. Rho, C. T. J. Ferguson, I. Manners, A. P. Dove and R. K. O’Reilly, J. Am. Chem. Soc., 2023, 145, 25274–25282 CrossRef CAS PubMed.
- J. Qian, Y. Lu, G. Cambridge, G. Guerin, I. Manners and M. A. Winnik, Macromolecules, 2012, 45, 8363–8372 CrossRef CAS.
- S. Ganda and M. H. Stenzel, Prog. Polym. Sci., 2020, 101, 101195 CrossRef CAS.
- J. Qian, G. Guerin, Y. Lu, G. Cambridge, I. Manners and M. A. Winnik, Angew. Chem., Int. Ed., 2011, 50, 1622–1625 CrossRef CAS PubMed.
- G. Guerin, G. Molev, P. A. Rupar, I. Manners and M. A. Winnik, Macromolecules, 2020, 53, 10198–10211 CrossRef CAS.
- X. Wang, G. Guerin, H. Wang, Y. Wang, I. Manners and M. A. Winnik, Science, 2007, 317, 644–647 CrossRef CAS PubMed.
- S. K. Patra, R. Ahmed, G. R. Whittell, D. J. Lunn, E. L. Dunphy, M. A. Winnik and I. Manners, J. Am. Chem. Soc., 2011, 133, 8842–8845 CrossRef CAS PubMed.
- J. Gwyther, J. B. Gilroy, P. A. Rupar, D. J. Lunn, E. Kynaston, S. K. Patra, G. R. Whittell, M. A. Winnik and I. Manners, Chem. – Eur. J., 2013, 19, 9186–9197 CrossRef CAS PubMed.
- J. Qian, X. Li, D. J. Lunn, J. Gwyther, Z. M. Hudson, E. Kynaston, P. A. Rupar, M. A. Winnik and I. Manners, J. Am. Chem. Soc., 2014, 136, 4121–4124 CrossRef CAS PubMed.
- W. Yu, M. Inam, J. R. Jones, A. P. Dove and R. K. O'Reilly, Polym. Chem., 2017, 8, 5504–5512 RSC.
- Y. He, J.-C. Eloi, R. L. Harniman, R. M. Richardson, G. R. Whittell, R. T. Mathers, A. P. Dove, R. K. O’Reilly and I. Manners, J. Am. Chem. Soc., 2019, 141, 19088–19098 CrossRef CAS PubMed.
- J. H. Hwang, J. Yoon, M. Seo, J. P. Patterson and E. Lee, Matter, 2025, 8, 102148 CrossRef CAS.
- G. Lin, J. Tao, Y. Sun, Y. Cui, I. Manners and H. Qiu, J. Am. Chem. Soc., 2024, 146, 14734–14744 CrossRef CAS PubMed.
- D. Chakraborty, W. Ma, P. G. Vekilov and J. D. Rimer, Chem. Mater., 2025, 37, 4158–4168 CrossRef CAS.
- Y. Li, X. Shan, S. Li, J. Wang, Z. Li, Z. Wang, X. Li, W. Hong, M. Li and Y. Ma, Angew. Chem., Int. Ed., 2023, 62, e202311778 CrossRef CAS PubMed.
- H. Kim, J. Lee, S.-H. Hwang, N. Yun, S. Park and T.-L. Choi, J. Am. Chem. Soc., 2024, 146, 20750–20757 CrossRef CAS PubMed.
- Z. Wang, Q. Liang, M. Li, G. Sun, S. Li, T. Zhu, Y. Han, H. Xia, Z. Ren, B. Yu, J. Zhang, R. Ma, H. Thachoth Chandran, L. Cheng, L. Zhang, D. Li, S. Chen, X. Lu, C. Yan, R. Azmi, K. Liu, J. Tang and G. Li, Adv. Mater., 2025, 37, 2418011 CrossRef CAS PubMed.
- G. Lu, H. A, Y. Zhao, Y. Zhao, H. Xu, W. Shang, X. Chen, J. Sun, H. Zhang, J. Wu, B. Dai, B. Van der Bruggen, R. Dewil, A. K. An and S. Zheng, Nat. Commun., 2025, 16, 2284 CrossRef CAS PubMed.
- X. Wu, W. Yuan, Y. Wu, C. Wang, L. Xue, Y. Zhou, X. Zhang, S. Jiang, B. Zhao, Y. Chen, C. Yang, L. Ding, Y. Tang and X. Ding, Adv. Energy Mater., 2024, 14, 2304204 CrossRef CAS.
- Y. Shen, S. Zhang, Z. Qin, A. Beuque, L. Pinard, S. Asahina, N. Asano, R. Zhang, J. Zhao, Y. Fan, X. Liu, Z. Yan and S. Mintova, ACS Catal., 2024, 14, 3766–3777 CrossRef CAS.
- J. Tian, S.-H. Xie, U. Borucu, S. Lei, Y. Zhang and I. Manners, Nat. Mater., 2023, 22, 786–792 CrossRef CAS PubMed.
- Y. Lu, J. Tao, Z. Li, S. Wang, X. Cai and H. Qiu, J. Am. Chem. Soc., 2026, 148, 3158–3166 CrossRef PubMed.
- H. Wang, X. Wang, M. A. Winnik and I. Manners, J. Am. Chem. Soc., 2008, 130, 12921–12930 CrossRef CAS PubMed.
- X.-S. Wang, H. Wang, N. Coombs, M. A. Winnik and I. Manners, J. Am. Chem. Soc., 2005, 127, 8924–8925 CrossRef CAS PubMed.
- H. Qiu, V. A. Du, M. A. Winnik and I. Manners, J. Am. Chem. Soc., 2013, 135, 17739–17742 CrossRef CAS PubMed.
- J. Tao, B. Fang, Z. Fang, G. Lin, Z. Ji, C. Gao, R. Gao and H. Qiu, Angew. Chem., Int. Ed., 2025, 64, e202512348 CrossRef CAS PubMed.
- S. Wang, W. Xie, P. Wu, G. Lin, Y. Cui, J. Tao, G. Zeng, Y. Deng and H. Qiu, Nat. Commun., 2022, 13, 6673 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |