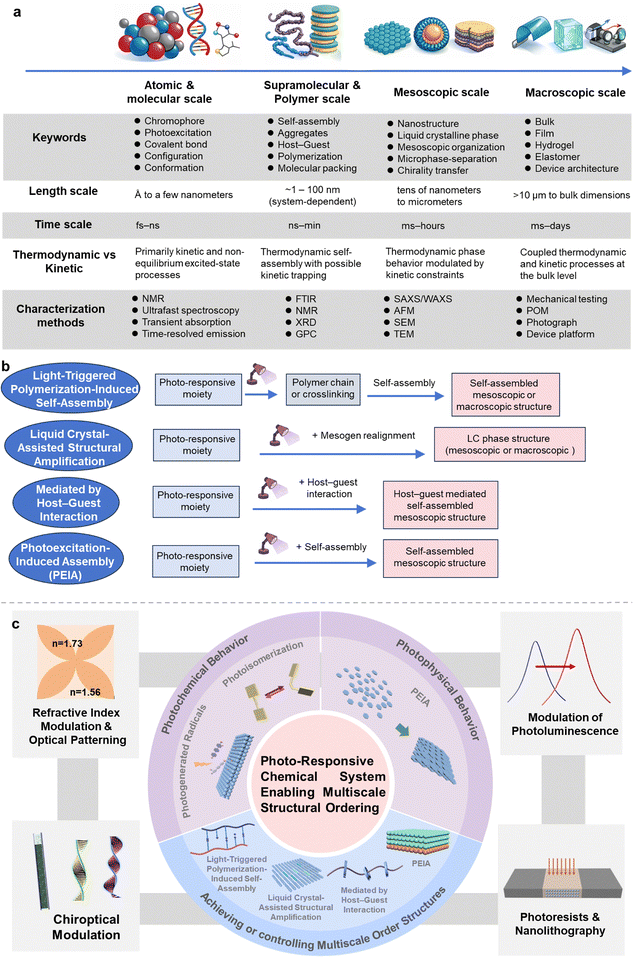
Open Access Article
Open Access ArticlePhoto-responsive chemical systems enabling multiscale structural ordering
Jianshuo
Cheng
,
Mouwei
Liu
and
Liangliang
Zhu
 *
*
State Key Laboratory of Molecular Engineering of Polymers, Department of Macromolecular Science, Fudan University, Shanghai 200438, China. E-mail: zhuliangliang@fudan.edu.cn
First published on 11th March 2026
Abstract
Beyond elemental composition and molecular structure, ordered multiscale architectures (self-assembled structure, crystal structure, phase structure, etc.) play a comparably important role in determining material functions, especially the optoelectronic properties. Light, a unique external stimulus with superior spatiotemporal resolution and non-invasive nature, provides unparalleled capabilities for dynamically constructing and manipulating such ordered structures. Since previous reviews on light-responsive materials have generally overlooked how light can govern multiscale structural ordering, this review comprehensively surveys the rapidly advancing field of photo-responsive chemical systems designed to achieve and control multiscale structural ordering. We begin by elucidating the fundamental photochemical and photophysical mechanisms at the molecular level, which serve as the primary triggers for molecular structural, configurational, or conformational transformations. Based on these events that initiate molecular perturbation, the core of the review critically examines several key light-mediated strategies for constructing and manipulating ordered hierarchical architectures, notably photo-triggered polymerization-induced self-assembly, liquid crystal-assisted phase structural amplification, photo-induced self-assembly mediated by host–guest interaction, and the emerging paradigm of photoexcitation-induced assembly (PEIA). We discuss how these strategies successfully translate molecular or nanoscale events into well-defined, macroscopic ordered states in real time and in situ. Finally, we provide a forward-looking perspective on the existing challenges and promising future research directions.
Key learning points(1) Fundamental principles and new mechanisms underlying photo-responsive materials.(2) Strategies for constructing and tuning ordered structures in photo-responsive systems. (3) Structure–property relationships: how different levels of structural ordering affect material performance. (4) Design guidelines and future directions for developing light-regulated materials with controllable structural ordering. |
1. Introduction
1.1. Structural order across scales
Ordered structures are ubiquitous in nature across multiple scales, from the atomic to the macroscopic level, representing spontaneous organizational forms that enable matter to adapt to environmental constraints and achieve optimal functionality.1–6 This inherent orderliness not only reflects the physico-chemical principles of nature but also provides principles for structural design in materials science, biology, and engineering.7–11 At the microscopic scale, thermodynamic driving forces (e.g., energy minimization, entropic effects) prompt the periodic arrangement of atoms, molecules, or colloidal particles.12–15 For instance, minerals like diamond and quartz acquire high strength and anisotropy through long-range atomic ordering,16–18 while biological molecules such as the DNA double helix achieve ordered encoding of genetic information via hydrogen bonding and base pairing.19–23 Amphiphilic molecules like phospholipids spontaneously form lipid bilayers in aqueous phases, where the ordered hydrophobic–hydrophilic interface underpins the selective permeability of cell membranes.24–27 At the mesoscopic scale, natural systems often integrate structures across different scales through hierarchical ordering to balance conflicting properties.28–31 Examples include the structural colors of peacock feathers and butterfly wings, which arise from coherent light scattering by periodic nanostructures rather than pigmental chemistry.24,32–34 On larger scales, space-filling and mechanical optimization drive the formation of regular geometric configurations, such as the hexagonal units in honeycombs, which maximize load-bearing efficiency with minimal material consumption, exhibiting strength significantly superior to foam materials.35–38 Across scales and systems, such ordered structures emerge as optimal solutions to the combined constraints of energy, information, and function. Their universality not only reveals the unity of natural laws but also provides the fundamental logic of “structure determines property” for the design of artificial materials.39–42Structural ordering is a universal principle for optimizing material properties.43–45 From atomic-scale crystalline arrangements (e.g., the sp3 hybridized carbon network in diamond conferring ultra-high hardness) to mesoscale biomineralized structures (e.g., the nacreous layer in abalone shells, a composite of aragonite and organic matter, synergizing strength and toughness), and further to macroscopic structures like honeycombs achieving lightweight and high strength, ordering dictates material characteristics across mechanical, optical, and electrical properties.46–49 This structure–property relationship offers crucial insights for material design. In materials science, in situ structural modulation refers to the real-time control of atomic/molecular order within a material under operational conditions via external stimuli (e.g., temperature, stress, electric/magnetic fields, chemical environment, light).50–52 Compared to traditional “synthesis-characterization-optimization” cycles, in situ modulation offers unique advantages such as dynamism, precision, and reversibility, serving as a key pathway for enhancing material order and optimizing performance.53,54
1.2. Tools for engineering structural order
To bridge the molecular events and macroscopic material responses discussed herein, a suite of complementary characterization tools is essential. At the molecular and nanoscale, transient absorption spectroscopy and time-resolved photoluminescence capture the kinetics of photoexcitation and the initial conformational changes. For mesoscale order, which is critical for self-assembled and liquid crystalline architectures, scattering techniques like small-/wide-angle X-ray scattering (SAXS/WAXS) provide statistically averaged information on periodic structures, phase symmetry, and lattice parameters, while atomic force microscopy (AFM) offers direct, real-space visualization of surface morphology and domain organization. At the macroscopic scale, the overall optical anisotropy and phase transitions are directly visualized through polarized optical microscopy (POM).3 This multi-technique framework forms the foundation for the mechanistic insights presented in the following sections, and is capable of effectively interpreting the structural data that underpins the progress of the field.1.3. Distinctive features and challenges of photo-responsive systems
Light-response stands out as a non-contact, high spatiotemporal resolution way for in situ modulation, demonstrating unique potential for dynamically manipulating ordered structures in materials.55–58 Unlike other external fields, like thermal, electric, magnetic, and so on, light control takes advantage of specific interactions between photon energy and matter (e.g., electronic excitation, molecular structural changes, localized heating), enabling high selectivity, rapid response, and multi-dimensional manipulation, thus becoming a pivotal tool for designing smart materials and functional devices.59–62However, a significant challenge persists in most photo-responsive chemical systems, namely, producing, maintaining, and enhancing structural order during photo-induced transformations. Even when the initially ordered arrangements are achieved, multi-scale structures often collapse due to misalignment of molecules or defects upon irradiation, originating from insufficient photoreaction efficiency and interference from side reactions.63 Addressing this requires precise molecular design, synergy of specific components or phases, and the development of novel photo-responsive mechanisms. In this way, structural orderliness is expected to evolve from molecule-scale photoresponsive moieties to larger scales under irradiation. This review aims to systematically summarize recent advances in this perspective, spanning chemical synthesis, material processing, and functional property control.
1.4. Scope and organization of this review
In this tutorial review, we first introduce the two primary types of photo-responsive behaviors (i.e., photochemical and photophysical), explain their fundamental principles at the molecular and microscopic scales, and summarize representative molecular systems. These fundamental response modes provide the molecular moiety-level upon which light can perturb molecular structure, configuration, conformation, interactions, or assembly equilibria. Based on these behaviors, we then discuss four distinct photo-responsive strategies for achieving or tuning multi-level hierarchical ordered structures (including self-assembled or phase-ordered systems), each of which leverages photochemical or photophysical events in different ways to induce structural transitions across scales. These strategies focus on how irradiation drives transformations between hierarchical levels or enhances structural order through controlled photo-perturbations. Key points and underlying mechanisms are elucidated through representative examples. Subsequently, we present typical material properties based on photo-responsive structural ordering, such as creating optical patterns with tuning refractive index, modulation of photoluminescence and chiroptical properties, lithography, and so forth. Finally, we offer perspectives on future directions for photo-responsive ordered systems, aiming to bridge light-stimulus, self-assembly, and material processing to advance this frontier towards practical applications (see also the outline in Fig. 1).To ensure conceptual clarity and pedagogical consistency throughout this Tutorial Review, we explicitly distinguish between photochemical processes, which involve ground-state chemical bond formation, cleavage, or isomerization, and photophysical processes, which primarily involve excited-state energy redistribution, conformational distortion, or intermolecular interaction modulation without permanent chemical transformation. In particular, the term photoexcitation-induced assembly (PEIA) is used here to describe emerging phenomena driven by excited-state intermolecular interactions or conformational dynamics, rather than by covalent bond reorganization. These definitions are applied consistently across all sections to avoid terminological ambiguity and to facilitate comparison among different light-responsive strategies.
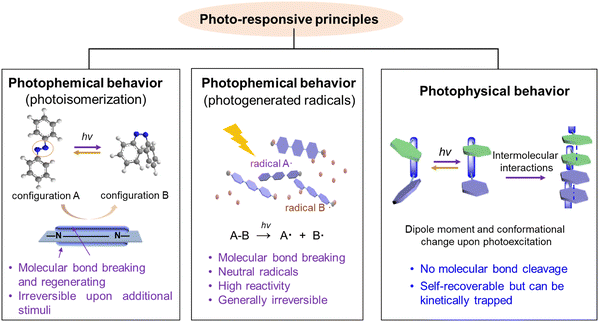
2. Photo-responsive principles: from molecular events to material responses
In this section, we aim to discuss fundamental principles and new mechanisms underlying photo-responsive materials. A clear understanding of photo-responsive behaviors requires examining how molecular systems evolve at the microscopic level upon photoirradiation. At this scale, light-mediated molecular control can be categorized into photochemical and photophysical processes (as shown in Fig. 2). The main criterion for distinguishing these two is whether it involves the breaking and reforming of chemical bonds. This section summarizes typical photo-responsive systems based on photochemical and photophysical processes, primarily analyzing molecular structural, configurational, or conformational changes during photo-responses from a theoretical and experimental standpoint, with the aim of providing a fundamental basis for enhancing material properties.2.1. Photochemical behavior
Photochemistry, a widely studied photo-responsive behavior, involves chemical reaction processes driven by photons under light irradiation.64–66 Its core principle involves electronic state transitions in molecules following light absorption, opening new reaction pathways.67–70 Research in photochemistry dates back to the 19th-century exploration of photosensitivity and photolysis. By the mid-20th century, advancements in laser technology and spectroscopy gradually revealed photochemical reaction mechanisms, particularly the dynamics of photoexcited states and ultrafast processes, laying the foundation for modern photochemistry.68,69,71–73 The past several decades have witnessed significant developments in light-initiated chemical reactions, utilizing typical photo-responsive molecular moieties such as azobenzene, stilbene, diaryl ethylene, and spiropyrane.74–83A distinguishing feature of photochemical reactions is their non-thermodynamic driving force: photon energy can directly excite molecules into high-energy excited states, overcoming energy barriers limiting thermal processes and opening new reaction pathways.59 Furthermore, photochemical processes offer high selectivity and controllability. By adjusting the wavelength, intensity, and polarization of the light source, spatial, temporal, and energy-resolved control of reactions can be achieved. These processes can even occur under mild conditions and frequently utilize visible light as the driver, offering the benefits of low energy consumption and environmental friendliness.84
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N π-bond and rotation around the N–N σ-bond, and the inversion (or concerted inversion) pathway, where one nitrogen atom inverts through a linear or nearly linear transition state while maintaining partial N
N π-bond and rotation around the N–N σ-bond, and the inversion (or concerted inversion) pathway, where one nitrogen atom inverts through a linear or nearly linear transition state while maintaining partial N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond character.87 Most photoisomerization processes are reversible, especially in cis–trans isomerization. By controlling light intensity, wavelength, or exposure duration, molecules can be switched between cis and trans configurations.88 For instance, azobenzene derivatives transition from the trans to the cis isomer under UV light irradiation and revert to the trans isomer under visible light or thermal treatment. This change results from electronic transitions upon photoexcitation, leading to the excited double bond breaking and reforming after rotation of the single bond. The reversibility makes photoisomerization widely applicable in smart materials, optical switches, and photo-responsive systems.89
N bond character.87 Most photoisomerization processes are reversible, especially in cis–trans isomerization. By controlling light intensity, wavelength, or exposure duration, molecules can be switched between cis and trans configurations.88 For instance, azobenzene derivatives transition from the trans to the cis isomer under UV light irradiation and revert to the trans isomer under visible light or thermal treatment. This change results from electronic transitions upon photoexcitation, leading to the excited double bond breaking and reforming after rotation of the single bond. The reversibility makes photoisomerization widely applicable in smart materials, optical switches, and photo-responsive systems.89
Photo-isomerization can achieve reversible single-crystal configurational changes if the efficiency of photoisomerization can be significantly improved, and the degrees of freedom of molecules can be well designed. Li et al. studied the reversible single crystal-to-single crystal (SCSC) photo-isomerization in azobenzene (Fig. 3).90 Using single-crystal X-ray diffraction techniques, the researchers demonstrated that under 365-nm light exposure (15 min, 112 mW cm−2), the large single crystals could undergo complete and reversible crystal transformation while maintaining long-range crystal order. The capture of the two intermediate states revealed a three-step mechanism, including the initial configurational change, followed by an isomerization process accompanied by minor, coordinated molecular motions (specifically interlayer sliding and subtle distance adjustments). This work redefines the understanding of azo conversions in crystals and opens up new avenues for the development of photo-responsive crystal materials.
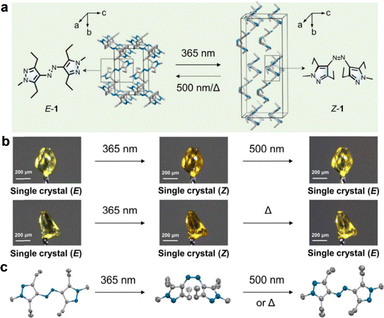 | ||
| Fig. 3 Photo-responsive systems enabling single crystal-to-single crystal conversion. (a) SCSC E ⇄ Z isomerization. (b) Photographs of single crystals with reversible SCSC isomerization. (c) Related X-ray molecular structures.90 Reproduced with permission.90 Copyright © 2025 American Chemical Society. | ||
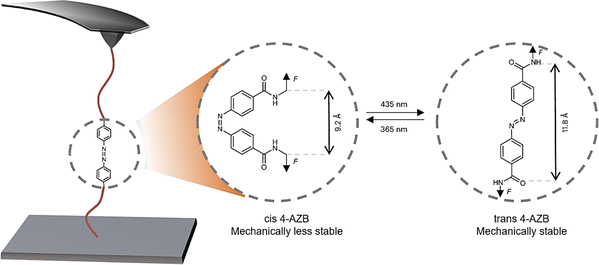
Photoisomerization can also regulate the mechanical properties of materials and establish the connection between molecules and macroscopic objects. Cao et al. demonstrated that azobenzene-based compounds exhibit photoswitchable mechanical strength, where light-induced structural changes alter their rupture forces, making them ideal photo-responsive mechanical elements (Fig. 4).91 Using single-molecule force spectroscopy (SMFS) and theoretical calculations, they found that the cis and trans 4,4′-azodibenzoic acid (4-AZB) isomers possess contrasting mechanical properties (600 pN for trans species and 200 pN for cis species). The different rupture forces of the two isomers were attributed to the pulling direction. These mechanical characteristics of azobenzene can be utilized to rationally control the macroscopic fracture behavior of polymer networks via light irradiation. These examples typically illustrate the photoisomerization portion in learning point (1).
In addition to radical-mediated synthesis, photogenerated radicals can also regulate the photoluminescent (PL) properties of materials. An et al. reported a dynamically tunable room-temperature phosphorescence (RTP) strategy based on the reversible transformation of radicals under external stimuli (Fig. 5).96 RTP has been a focus of scientific research due to its potential applications in optical materials, such as displays, memory devices, and bioimaging. However, achieving RTP often requires high molecular aggregation and strong intermolecular interactions, which aim to limit molecular flexibility and suppress non-radiative transitions in practical applications. Different from these traditional strategies, the researchers found that light irradiation can generate radicals in certain organic molecules (NDIA), which reversibly modulates the optical properties of the NDIA/PVA film. Specifically, light-induced radical generation can regulate RTP emission to become a radical emission, producing stronger PL signals under certain conditions within 90 s. The reversible transformation between the NDIA molecule and its anionic radical under light is related to excited-state bond weakening, where UV irradiation facilitates the cleavage of specific bonds, leading to radical formation and subsequent changes in emission properties. This example typically illustrates the photogenerated radicals portion in learning point (1).
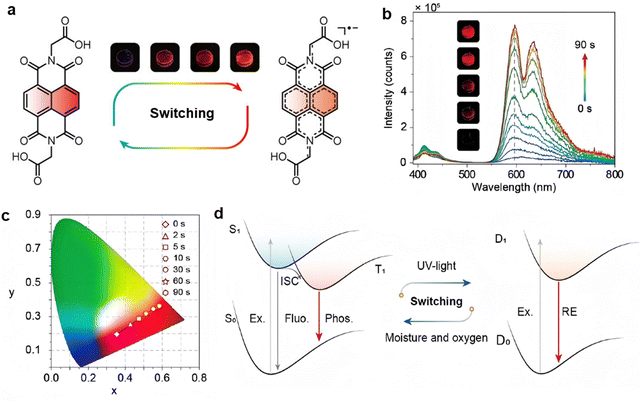 | ||
| Fig. 5 Photo-responsive systems with photogenerated radical transformation for dynamically tuning RTP. (a) Related chemical structures and photoluminescence images. (b) PL spectra of the NDIA/PVA film upon UV irradiation. (c) Corresponding emission color trajectory in the CIE coordinate diagram. (d) Corresponding energy transfer processes.96 Reproduced with permission.96 Copyright © 2022, Science China Press and Springer-Verlag GmbH Germany, part of Springer Nature. | ||
2.2. Photophysical behavior
Although many important photochemical systems have been thoroughly studied, the self-assembly process always has limitations. Firstly, the conversion rate of photochemical reactions is relatively low. Secondly, the presence of side reactions in photochemical reactions can have adverse effects on molecular assembly, potentially leading to defects and impacting their performance. Thirdly, most of these systems switch between two stable states, requiring additional energy consumption.63 In contrast to photochemical processes, photophysical processes involve a change only in the electronic states of molecules upon photon absorption, leading to conformational adjustments (e.g., change in spatial orientation due to single-bond rotation) or electron redistribution, without breaking chemical bonds.97,98 Photoinduced electron transfer (PET) is one of the earliest studied photophysical processes, where light absorption usually causes electron transfer from an electron-rich donor to an electron-deficient acceptor following photoexcitation.99 PET plays a vital role in many important photo-responsive processes, particularly in designing photo-responsive materials, photoelectric devices, and light-mediated electrochemical processes, providing the foundation for efficient energy conversion and information transfer. For instance, Liu et al. investigated the key role of molecular conformational folding in PET (Fig. 6).100 Their study revealed that conformational changes in naphthalimide-containing systems, particularly from an open to a folded state, can significantly enhance PET efficiency. Notably, although PET is generally classified as a photophysical process in the excited state without involving permanent chemical changes, it may also trigger subsequent photochemical reactions once the generated radical species proceed to further irreversible steps.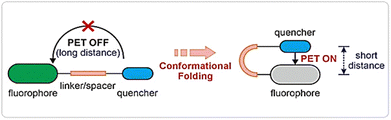 | ||
| Fig. 6 Photo-responsive systems with a conformation folding-activated PET.100 Reproduced with permission.100 Copyright © 2024 Chinese Chemical Society. This article is published under the terms of the Creative Commons Attribution-NonCommercial 3.0 Unported license (CC BY-NC 3.0, https://creativecommons.org/licenses/by-nc/3.0/). | ||
Besides PET, with advancements in photophysics, an increasing number of other photophysical mechanisms have been proposed, enriching the existing theories and strategies. For example, Tang et al. studied the mechanism of visualizing and regulating molecular motion via photoinduced clusteroluminescence in the solid state.101 They discovered that certain organic molecules in the solid state exhibit dynamic molecular motions upon photoexcitation, leading to changes in luminescence properties. Specifically, light-induced relative motions within molecular clusters trigger significant changes in clusteroluminescence, allowing real-time monitoring of molecular-level motions (Fig. 7). Li et al. investigated the photoinduced phosphorescence effect through 365-nm irradiation for 5 min.102 Their study revealed that photoinduced phosphorescence can be attributed to reversible changes in crystal packing. By adjusting the molecular packing structure, the intensity and lifetime of resonance phosphorescence can be significantly enhanced or regulated (Fig. 8). These reports provide molecular mechanisms and design principles for precisely regulating small-molecule crystal forms through photophysical processes.
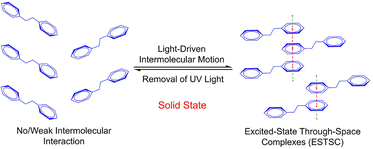 | ||
| Fig. 7 Photo-responsive systems with light-driven intermolecular motions in the solid state.101 Reproduced with permission.101 Copyright © 2019 American Chemical Society. | ||
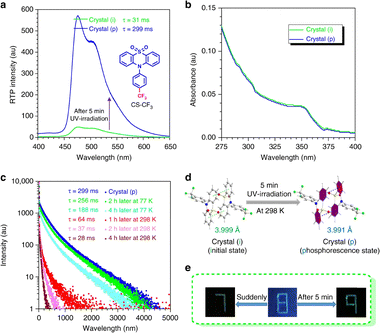 | ||
| Fig. 8 Photo-responsive systems bearing photophysical processes that affect crystal stacking and phosphorescence activation. (a) The RTP spectral change of CS-CF3 upon UV irradiation. (b)–(e) The corresponding change in UV-visible spectra, PL-decay curves, crystal structures, and a PL patterning.102 Reproduced under terms of the CC-BY license.102 Copyright © 2018, The Author(s), published by Springer Nature under a Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/). | ||
Recently, PEIA has been proposed as a novel photophysical behavior. By studying a class of persulfurated arene compounds, our group found that these compounds exhibited the ability to undergo aggregation upon photoirradiation, leading to the proposal of the PEIA effect.103 Compared to studying more AIE phenomena, the main difference between the two lies in the causality. AIE causes luminescence through aggregation, while PEIA leads to assembly through light excitation. Compared to standard excited-state aggregation, the main difference between the two lies in the driving force. The driving force of standard excited-state aggregation can be various methods such as electric and magnetic, while PEIA is driven by light, which gives PEIA advantages such as remote controllability and good biocompatibility. These molecules exhibit reversible aggregation upon photoexcitation (driven by their distinct excited-state conformation from the ground-state counterpart), and display flicker phosphorescence during the recovery process (Fig. 9). Accompanied by a long-lived dark state formed, the system has sufficient time to resist energy relaxation, ensuring that the molecules aggregate and exhibit PEIA. It is worth noting that most systems do not exhibit aggregation properties under light excitation due to their relatively rigid molecular conformations and short excited-state lifetimes. Currently, PEIA is mainly applicable to persulfurated arene systems. With the advancement of research, other photo-responsive skeletons have also emerged, demonstrating the universality of this strategy.104 The above examples typically illustrate the photophysical portion in learning point (1).
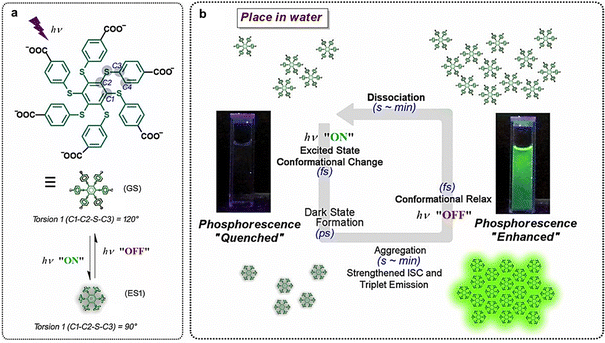 | ||
| Fig. 9 Photo-responsive systems rendering PEIA effect that can activate phosphorescence in solution. (a) Conformational change upon photoexcitation of the persulfurated arene. (b) Successive action of the photoactivation and self-recovery cycle with a flicker RTP effect of the main compound in aqueous media (ISC: intersystem crossing).103 Reproduced with permission.103 Copyright© 2019. Published under the PNAS license. | ||
3. Achieving or controlling multiscale order structures via photo-responsive processes
In this section, we turn to discuss strategies for constructing and tuning ordered structures in photo-responsive systems. The molecular-scale photochemical and photophysical events provide the fundamental “triggers” for structural change. However, the manifestation of these molecular perturbations at the mesoscopic and macroscopic scales relies on a critical amplification and organization process. To bridge this gap, photo-responsive strategies must effectively couple these primary molecular events (e.g., isomerization, electron transfer, conformational switching) with defined intermolecular interactions or collective dynamics. This coupling can propagate and amplify local changes through mechanisms such as cooperative alignment in liquid crystals, templated growth in host–guest systems, or polymerization-induced confinement, thereby translating discrete molecular actions into long-range, multiscale structural order.Before elaborating on the multi-scale orderliness, we first distinguish between the molecular, supramolecular, and macroscopic levels in order to better understand the process of multi-scale ordering. Molecular scale: orderliness is manifested in the spatial arrangement of atoms/molecules, chemical bonds, or coordination environments, as well as the resulting electronic states, molecular conformations, or dipole orientations.105,106 Supramolecular scale: orderliness stems from non-covalent interactions between molecules (such as hydrogen bonds, π–π stacking, hydrophobic interactions), forming assemblies with specific morphology, periodicity, or symmetry.107,108 Macroscopic scale: orderliness is reflected in the anisotropy of the material as a whole, regular surface morphology, macroscopic patterns, or functional structures at the device level.109,110
Upon the fundamental photochemical and photophysical events originate at the molecular level, a pivotal challenge lies in translating these photo-responsive molecular perturbations into controlled, long-range structural order from the microscopic to the macroscopic scale.111 Through photochemical or photophysical response processes, as well as with the help of precise molecular design, synergy of specific components or phases, or enrolment of new photo-responsive mechanisms, the structural changes occurring at the molecular scale can drive larger-scale self-assembly or phase transitions, thereby enabling adjustments in material order across multiple hierarchical levels.63,112 This multi-level ordering transformation not only provides new ideas for designing high-performance optical functional materials but also expands their application potential in micropatterning, structural color tuning, chiroptical modulation, luminescence control, photoresist, and so forth. In this section, we will explore in detail several strategies that utilize photo-responsive approaches to construct and regulate multi-level ordered structures, and analyse their strengths, weaknesses, and applicable scenarios through concrete examples (Table 1). Concurrently, we will delve into the mechanisms behind these photo-responsive processes, revealing the fundamental principles and influencing factors.
| Strategies | Photo-triggered polymerization-induced self-assembly | Liquid crystal-assisted structural amplification | Photo-induced ordered self-assembly mediated by host–guest interaction | Photoexcitation-induced assembly (PEIA) |
|---|---|---|---|---|
| Light-response type | Photochemical | Photochemical | Photochemical | Photophysical |
| Mechanism | Light-initiated polymerization drives in situ amphiphile formation, leading to spontaneous self-assembly via block incompatibility and phase separation | Photo-induced molecular changes are amplified through cooperative mesogen alignment and liquid crystalline phase transitions | Light modulates host–guest binding affinity, dynamically controlling supramolecular association and structural organization | Excited-state conformational distortion drives aggregation or non-equilibrium assembly, leading to ordered structural evolving |
| Molecular-level change | Photo-induced molecular structure change | Light-controlled orientation-induced collective reordering of liquid crystals | Light regulation of host–guest molecular recognition and encapsulation | Molecular excited-state conformational change |
| Primary driving force for ordering | Covalent bonds | Mesogenic cooperative orientation order | Changes in non-covalent interaction strength such as host–guest molecular recognition | Interactions between molecules driven by excited state conformations |
| Time scale | Seconds to hours, depending on polymerization kinetics and diffusion | Milliseconds to seconds for phase reorientation; longer if involving bulk relaxation | Milliseconds to minutes, depending on binding kinetics and diffusion | Ultrafast excitation (fs–ns) followed by assembly evolution (ns–min) |
| Reversibility | Generally irreversible due to covalent bond formation, except for dynamic covalent bonds | Typically reversible, depending on photoisomer stability and thermal back-relaxation | Highly reversible under appropriate binding equilibria | Potentially reversible but can be kinetically trapped. |
| Spatial resolution | Moderate to high; can be spatially patterned through localized light exposure, though diffusion may blur boundaries | High at the micro-scale; governed by liquid crystal elasticity and domain coupling | Moderate; limited by molecular diffusion and binding exchange rates | Mesoscale precision depends on diffusion and cooperative effects. |
| Advantages | Broadly applicable to various monomers and polymer systems; compatible with scalable synthesis | Effective in systems containing mesogenic units; particularly powerful in soft anisotropic materials | Versatile across diverse supramolecular chemistries; tunable via host–guest design | High photon utilization efficiency; suitable for large-scale and continuous morphology control |
| Limitations | Limited reversibility; structural precision depends on polymerization control; kinetic trapping and dispersity effects may arise | Requires mesogen incorporation; sensitive to temperature, alignment conditions, and phase stability. | Binding strength trade-off between stability and reversibility; may suffer from competitive interactions or environmental sensitivity | Requires chromophores with excited state conformational distortable and long lifetimes |
| Ordering product type | Nanocages, nanogels, nanovacuoles, nanofibers, etc. | Liquid crystal orientation pattern | Supramolecular polymers, nanofibers, hydrogels | Structural changes of assemblies or block copolymer phases |
3.1. Photo-triggered polymerization-induced self-assembly
Photo-triggered polymerization-induced self-assembly is a technique that utilizes light energy to initiate polymerization or photocrosslinking, and induce the self-assembly of leading polymers into ordered structures.113 It combines photo-responsiveness and polymerization reactions, thereby harnessing the controllability and non-invasiveness of light to regulate the assembly behavior of macromolecules or nanosystems. Photopolymerization-induced self-assembly is mainly applicable to block copolymers and surface-grafted polymer systems. The intensity, wavelength, and pattern of light irradiation directly determine the local initiation rate of polymerization. This in turn governs the gradual formation and in situ aggregation of the polymer blocks, allowing for the real-time, kinetically trapped fabrication of nanostructures with defined morphologies that are often inaccessible through traditional thermal equilibrium methods.Chen et al. proposed a simple and robust one-pot method to synthesize stilbene-containing block copolymer (BCP) nanowires with uniform morphology, combining photodimerization and polymerization-induced hierarchical self-assembly.114 Although the photodimerization reaction proceeded successfully, it resulted in a decrease in the overall structural order of the assembly. This demonstrates the critical importance of designing the dimerizable moieties attached to the polymer backbone. Thus, Russell et al. designed and synthesized a deuterated polystyrene (d8-PS, easy to track by NMR) and poly(methyl methacrylate) (PMMA) diblock copolymer PS-b-PMMA di-BCP (Fig. 10a and b).115 They further investigated the light-controlled ordering transition in PS-b-PMMA films via the photodimerization of an anthracene group. In this way, UV irradiation (365 nm, 35 mW cm−2) can regulate interactions between copolymer chains, transitioning the order state of the film (monitored by small-angle neutron scattering from 0.03796 Å to 0.03109 Å).
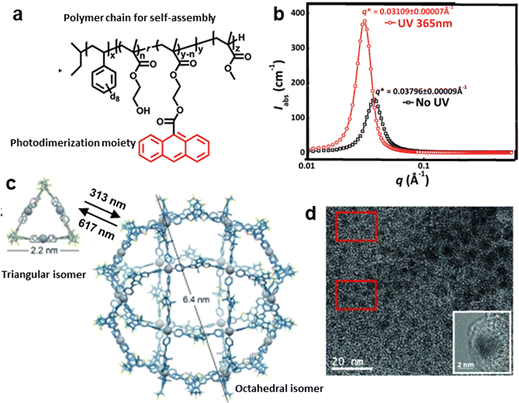 | ||
| Fig. 10 Photo-responsive systems enabling photo-triggered polymerization to construct ordered self-assembled structures. (a) PS-b-PMMA di-BCP with a anthracene-based dimerizable moiety and (b) corresponding small-angle neutron scattering profiles featured an ordered film morphology transfer.115 (c) Photoinduced cyclization enabling a switching between triangular and octahedral shapes of the molecular superstructure and (d) related TEM image featuring the yielded nanospheres.116 (a) and (b) Reproduced with permission.115 Copyright © 2011 American Chemical Society; (c) and (d) Reproduced with permission.116 Copyright © 2016 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
Photo-triggered polymerization-induced self-assembly can also be realized through photoinduced cyclization. Clever et al. demonstrated the reversible transformation of self-assembled structures controlled by the change in the geometry of photo-responsive molecules (Fig. 10c and d).116 They designed a photo-responsive self-assembly system where UV light regulates intramolecular cyclization, enabling reversible switching between triangular and octahedral shapes with a nonspherical morphology of 6.4 nm across. Such a light-induced geometric change drives the structural transformation from one regular morphology to another by altering intermolecular interactions. This study further shows that the light-induced geometric change can help achieve nanostructures with high tunability and dynamic response. These examples typically illustrate the photo-triggered polymerization-induced self-assembly strategy in learning point (2).
3.2. Liquid crystal-assisted structural amplification
The strategy of liquid crystal-assisted structural amplification is mainly applicable to small-molecule liquid crystal-doped systems. In liquid crystal (LC) environments, perturbations of photo-responsive molecules can readily induce ordered changes in the phase environment and amplify the molecular perturbation. LC molecules themselves possess anisotropy and long-range order characteristics. When photo-responsive moieties are embedded within an LC environment, light-induced changes in molecular configuration or dipole moment can disrupt the local orientational balance, thereby triggering the rearrangement of orientation domains within the LC phase. Due to the high fluidity and plasticity of LCs, this light-triggered perturbation diffuses successively along the orientation direction through molecular cooperative effects, manifesting as an evolution of ordered structures from local to global scales. The diffusion process achieves the translation of molecular-scale photo-responses to macroscopic-scale order regulation, providing a unique pathway for constructing light-controlled materials with multiscale order.117,118Katsonis et al. revealed how the ordered structure of LCs amplifies local molecular changes under light to produce macroscopic optical responses. They constructed a light-controlled chiral inversion cholesteric LC system, where a bisazo-photoactive dopant exhibits reversible three-state isomerization (EE, ZE, ZZ), with interconversion between configurations achievable via visible light irradiation and thermal relaxation (Fig. 11a). Throughout the process, the helix pitch can be regulated within the range of 2.2 µm to 30 µm. 117 Cheng et al. investigated the inversion of circularly polarized luminescence (CPL) signals in LCs.119 They introduced a photo-responsive achiral negative dichroism dye (KG) and a static achiral positive dichroism dye (NR) dopant with opposite dichroism to the CLC host, thus achieving the control of CPL chirality and intensity. By introducing a synergic dichroic modulation, the researchers achieved reversible CPL signal inversion in LC materials (Fig. 11b and c). Li et al. also studied chiral inversion in self-assembled helical superstructures based on a cyclic azobenzenophane (Fig. 11d).120 Their work demonstrated how LC-assisted self-assembly can achieve chiral inversion via light regulation with high reversibility. Similarly, the process of photoisomerization is also accompanied by extensive regulation of cholesterol pitch between 100 µm and 400 µm. Kosa et al. proposed a method of photoinduced liquid crystallization on naphthopyran-based materials, studying how light irradiation induces the formation of LC phases and its impact on material structure. Their work revealed a more fundamental phenomenon of “photoinduced liquid crystallization”, where light can trigger the transformation of a non-LC system into a highly ordered LC phase, thus achieving spontaneous ordering driven by the light field.121 The phase transition process occurred in seconds above the clear point of the liquid crystal (31.8 °C). This change reduces the system free energy in the ordered mesophase under isothermal conditions. POM images visually demonstrate the macroscopic manifestation of this light-controlled transition (Fig. 11e).
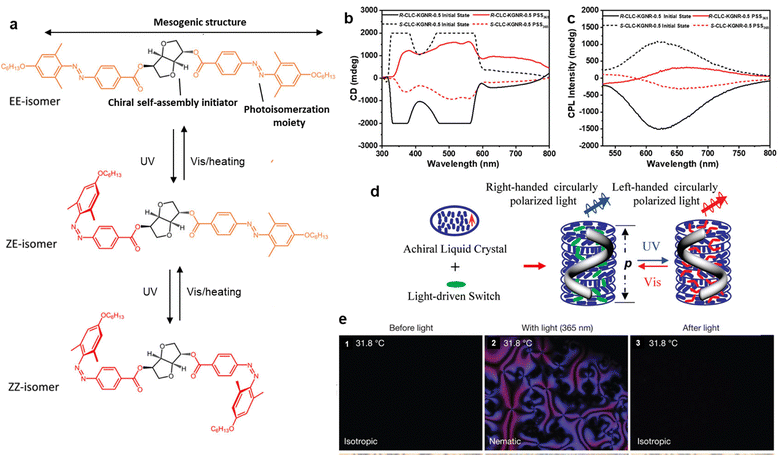 | ||
| Fig. 11 Mesogen-assisted photo-responsive systems enabling ordered phase structural regulation. (a) Photoisomerization of a bisazo-photoactive dopant, leading to a photoinduced chiral inversion in a photoactive cholesteric LC system.117 (b) CD and (c) CPL spectra of a liquid crystal system with an achiral negative dichroism dye (KG) and a static achiral dye (NR) as dopants, in its initial state and under light irradiation.119 (d) Schematic representation of light-driven reversible handedness inversion in LC-assisted self-organized helical superstructures based on a cyclic azobenzenophane.120 (e) POM images visualizing the macroscopic process of light-controlled LC phase before, during, and after UV exposure on a naphthopyran-based material.121 (a) Reproduced with permission.117 Copyright © 2019 American Chemical Society. This publication is licensed under CC-BY-NC-ND. (b) and (c) Reproduced with permission.119 Copyright © 2024 Wiley-VCH GmbH. (d) Reproduced with permission.120 Copyright © 2010 American Chemical Society. (e) Reproduced with permission.121 Copyright © 2012, Springer Nature Limited. | ||
Light can further drive multi-morphological and superficial phase transitions within LC media. Evans et al. studied light-controlled phase transitions in arylazopyrazole-based liquid crystals, focusing on the transition from hexagonal to cubic phases under light irradiation (Fig. 12a–c).122 This transition is evidenced by the appearance of a new series of peaks with a ratio of √6:√8:√14:√16:√20 and a reduction in the intensity of the hexagonal peaks (Fig. 12a). Mechanically, this transition is driven by light-induced packing frustration: photoisomerization of the azopyrazole moieties introduces geometric mismatch. The system resolves this frustration by reorganizing into a cubic phase, which accommodates the varied molecular geometries with lower curvature elastic energy. The study showed that the ordered structures of LC materials can undergo peculiar phase transitions under light, thereby increasing the rate of CO2 diffusion. Yu et al. studied the application of halogen bonding in block copolymer nanostructures, demonstrating how halogen bonds provide additional stability and control in LC molecular alignment (Fig. 12d).123 Through linearly polarized light (LPL) irradiation, the researchers achieved efficient photoalignment and photoreorientation of the nanostructures with a periodic distance of 21.7 nm. These examples typically illustrate the liquid crystal-assisted structural amplification strategy in learning point (2).
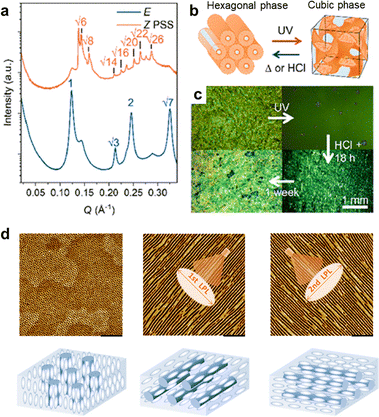 | ||
| Fig. 12 Mesogen-assisted photo-responsive systems enabling the modulation of morphology and orientation. (a) and (b) After UV irradiation, the SAXS patterns reveal a phase change from hexagonal to cubic in an arylazopyrazole-based LC system. (c) Corresponding POM micrographs showing birefringent, hexagonal-to-isotropic cubic transition.122 (d) AFM images of a halogen bonding mediated LC BCP film and its orientations upon polarized light irradiation. Scale bar: 200 nm.123 (a)–(c) Reproduced with permission.122 Copyright © 2024 The Authors. Published by American Chemical Society. This publication is licensed under CC-BY 4.0 (https://creativecommons.org/licenses/by/4.0/). (d) Reproduced with permission.123 Copyright © 2020, American Chemical Society. | ||
3.3. Photo-induced ordered self-assembly mediated by host–guest interaction
Host–guest interactions are a class of non-covalent forces based on molecular recognition, establishing selective binding between molecules through size matching, shape complementarity, and specific electrostatic, hydrogen bonding, or hydrophobic interactions. The host molecule typically possesses a regular cavity or channel structure (e.g., cyclodextrins, cucurbiturils, cyclic oligomers) capable of partially or fully encapsulating the guest molecule, thereby significantly altering the guest orientation and local environment.124,125 Macrocyclic structures like cyclodextrins and calixarenes are rich in non-covalent bonds such as hydrogen bonds and electrostatic interactions, and can form host–guest assemblies with small molecules via size effects.126,127 This interaction not only stabilizes the assembly units at the molecular level but can also extend local molecular recognition to larger-scale ordered structures through the arrangement of guest molecules. In this process, host–guest recognition provides both precise and directional control for construction, and ensures dynamic reversibility, enabling the systems to undergo controllable structural rearrangements under external stimuli (e.g., light, heat, solvent).128 Beyond simple binding, host–guest systems can also achieve sophisticated cooperative effects. The light-triggered encapsulation or release of a guest can alter intermolecular forces (e.g., π–π stacking, hydrophobic collapse) among the complexed units, thereby directing nanoscale morphology evolutions such as transitions between 1D fibers, 2D sheets, and 3D networks. Thus, host–guest recognition not only stabilizes assembly units but also translates finely tuned molecular responses into well-defined, higher-order architectures through synergistic non-covalent networks. This dual role makes host–guest chemistry a unique strategy for building adaptive, multi-scale ordered materials.As shown in Fig. 13a and b, Liu et al. reported a photo-controlled chiral self-assembly system based on cucurbit[8]uril (CB[8]).129 They present a multicomponent supramolecular heterodimer (Fig. 13a), which is constructed by photoswitchable DTE-bridged doubly charged bispyridinium (BP) salt (1), naphthalene (NP)-substituted chiral diphenylalanine (2), and cucurbit[8]uril (CB[8]). Diphenylalanine (l-Phe-l-Phe, FF) as a chiral source is introduced into the supramolecular system and provides the possibility of chiral transfer. Intermolecular hydrogen-bond interaction of FF and aromatic stacking effect of DTE supply a self-aggregated chance. These forces caused the formation of typical nanofibers with a diameter of 20–30 nm (Fig. 13b). Combining the ultra-stability of CB[8] complexes (the binding constant log K can reach over 10), the system realized the topological transformation from one-dimensional nanofibers to two-dimensional nanosheets (thickness: 7.8 nm), owing to the intervention of the rigid CB[8] host that blocked the hydrogen bonding between FFs. Photocontrollable and reversible conversion between two-dimensional nanosheets and one-dimensional nanorods with chiral modulation within 16 min was followed, because the photocyclization effect of DTE changed the π–π stacking and cooperated with the host–guest interaction to reconstruct the self-assembled morphology. The entire process is also accompanied by changes in electronic transitions and chiral self-assembly, allowing absorption and circular dichroism (CD) spectra to track these dynamic changes (Fig. 13c).
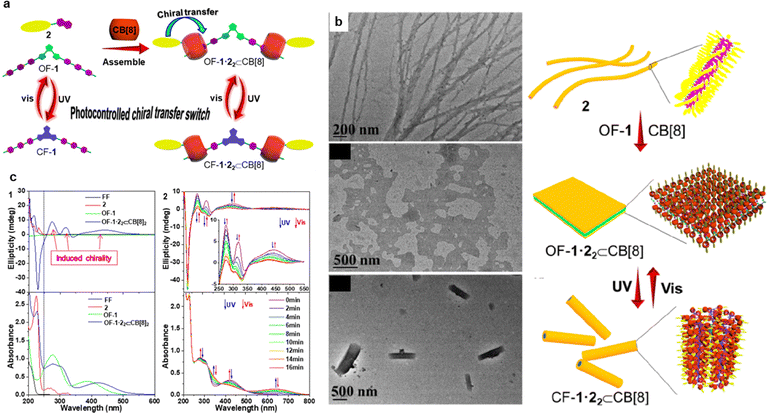 | ||
| Fig. 13 Photo-responsive systems with host–guest interactions for synergistically regulating the ordered structure of assemblies. (a) Photo-controlled chiral transfer driven by CB[8] host–guest recognition and (b) corresponding nano-morphological changes and cartoon illustrations.129 (c) Corresponding CD and absorption spectral changes.129 (a)–(c) Reproduced with permission.129 Copyright © 2023 The Authors. Published by American Chemical Society. This publication is licensed under CC-BY-NC-ND 4.0 (https://creativecommons.org/licenses/by-nc-nd/4.0/). | ||
Furthermore, Liu et al. constructed a controllable dimensionality transition system based on host–guest recognition between coumarin-diphenylalanine (CO-FF) and γ-cyclodextrin (γ-CD) (Fig. 14a and b).130 They constructed stable supramolecular assemblies using CO-FF and γ-CD (binding constant: 7.01 × 103 M−1). Through the collaboration of host–guest interactions, one-dimensional nanofibers with an average width of several nanometers and a length of tens of micrometers were initially formed, due to the similar peptide properties as in the previous example and the inclusion of cyclodextrin that enhanced the amphiphilicity of the guest molecule. Such nanofibers were then converted into two-dimensional films with a lateral dimension of tens of micrometers and a thickness of 4.5 nm via photo-dimerization of the coumarin groups after 365-nm irradiation for 8 h. This is because the host–guest complexes after photodimerization can grow along two perpendicular directions, and the resultant two-dimensional structure was stabilized by the hydrogen bonds and π–π interactions among adjacent di-CO-FF⊂γ-CD units (Fig. 14a). In another photo-switchable self-assembly system involving adamantanyl-modified diphenylalanine (Ada-FF) with azobenzene-bridged bis(β-CD) (H, Fig. 14a), high-constant complex (binding constant: 5.32 × 104 M−1) can be formed. On this basis, one-dimensional nanotubes (open-ended, curved tubular structures, formed by Ada-FF⊂H-cis) and two-dimensional nanosheets (formed by Ada-FF⊂H-trans), whose lengths and widths reached several micrometers with a height of approximately 7 nm, can be converted into each other through irradiation with different wavelengths of light.131 Typically, when the solution is exposed to ultraviolet light (365 nm) for 10 minutes, nearly all the two-dimensional nanosheets transform into one-dimensional nanotubes. Meanwhile, the resulting nanotubes can revert to their original two-dimensional nanosheets after being irradiated with visible light (450 nm) for 30 minutes (Fig. 14b). Here, the molecular length variation of H plays a crucial role (6.51 nm for the trans-species and 6.18 nm for the cis-one, accompanied by configurational bending, Fig. 14c). This, in turn, changes the self-assembly dynamics, achieving a morphological transition from two-dimensional to one-dimensional while weakening the rigidity of the system. These examples typically illustrate the strategy of photo-induced ordered self-assembly mediated by host–guest interaction in learning point (2).
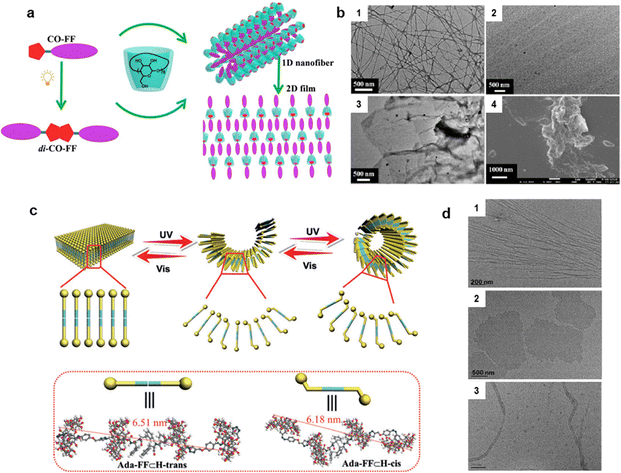 | ||
| Fig. 14 Photo-responsive systems with host–guest interactions for synergistically regulating the ordered structure of assemblies. (a) A coumarin-diphenylalanine (CO-FF) based guest system and schematic illustration of their assemblies with γ-CD. (b) TEM images of CO-FF⊂γ-CD irradiated for (1) 0, (2) 4, and (3) 8 h, and (4) SEM image for 8 h at 365 nm.130 (c) Ada-FF and H, and their assembly. (d) TEM images showed: (1) the one-dimensional structure of Ada-FF (scale =0.2 µm); (2) the two-dimensional structure of Ada-FF⊂H-trans (scale = 0.5 µm); and (3) the one-dimensional structure of Ada-FF⊂H-cis (scale = 0.2 µm).131 (a) and (b) Reproduced with permission.130 Copyright © 2018 American Chemical Society. (c) and (d) Reproduced with permission.131 Copyright © 2017 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
3.4. PEIA
PEIA can occur in situ to regulate molecular motion and drive self-assembly change of the photo-responsive groups themselves, or cooperatively drive the self-assembly change of a second component like polymer chains, thereby controlling the orderliness of the entire system.63 This strategy refers to the dual regulation of both self-assembled structures and phase structures. This photophysical process does not alter molecular structures but utilizes excited-state conformational change to drive the assembly and phase structures of the entire system.Building on the prior research, our group proposed a self-assembly strategy controlled by photoexcitation-mediated host–guest interactions to achieve a self-assembly transition from a linear nanostructure to a worm-like one (Fig. 15a).132 In detail, by introducing a unique photo-responsive group, persulfurated arene, into the host–guest system HB-CyD, we achieved a light-controlled transition from one ordered structure to another. Photoexcitation causes rearrangement of the host and the guest components, triggering a transformation in the self-assembled structure and enhancing the afterglow performance of the material. Based on a similar host–guest interaction, PEIA-caused 3D-to-2D exfoliation can also be attained on the ordered assembly of a persulfurated arene-based poly(pseudorotaxane) (PR) nanosheet (Fig. 15b).133 Light exposure induces interlayer separation of the 3D PR structure, forming individual nanosheets. These nanosheets subsequently reorganize into bilayer nanosheets upon solvent evaporation.
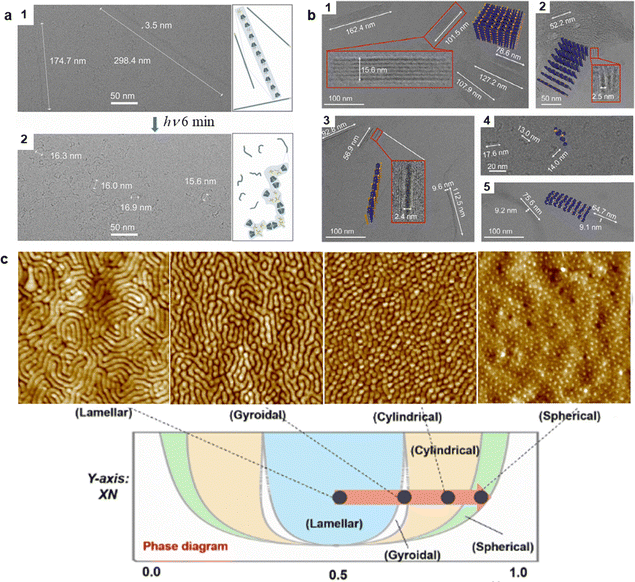 | ||
| Fig. 15 Photo-responsive systems rendering PEIA effect that can cause order-to-order structural transfer. (a) Cryo-TEM images of HB-CyD before and after UV irradiation and the corresponding structural transition diagram of HB-CyD assembly.132 (b) Cryo-TEM images of a persulfurated arene-based PR in pure water without light irradiation and after light irradiation with different periods.133 (c) AFM images and Schematic diagram of the PEIA-induced BCP self-assembly.135 (a) Reproduced with permission.132 This article is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported Licence (https://creativecommons.org/licenses/by-nc/3.0/). (b) Reproduced with permission.133 Copyright © 2022 American Chemical Society. (c) Reproduced with permission.135 Copyright © 2022 Wiley-VCH GmbH. | ||
Furthermore, by controlling molecular aggregation via photoexcitation, microphase separation in block copolymer materials can be regulated. Polystyrene-b-poly(L-lactide) (PS-b-PLLA) was chosen as the chiral BCP backbone, which could co-assemble with a six-carboxylate-group modified hexathiobenzene compound (referred to as M-1) through hydrogen bonding. The helical microphase-separation characteristic of PS-b-(M-1@PLLA) in a film state was observable within different co-assembly ratios. Due to the application of PEIA, driven by the excited-state conformation of M-1, the helical pitch could be regulated in situ.134 After the in situ irradiation for 20 s, the phase transitioned into an ordered helix, and the average pitch was approximately 25 nm. Through this PEIA-mediated phase control, we can also precisely adjust the phase volume ratio of the material (Fig. 15c).135 A typical PEIA molecule, hexa-(arylthio) benzene with noncovalent sites modified on the periphery (M-2), was non-covalently linked to a block copolymer (BCP), PS-b-PA, to form a supramolecular BCP ensemble M-2@PS-b-PA. The molecular framework of M-2 exerted a distinct conformational difference between its ground state and excited state, thus driving the self-assembly of the BCP chain synergically. The resulting system can access a full phase diagram across different irradiation periods, evolving from a lamellar structure to a gyroidal, cylindrical, and finally to a spherical one within 15 min. These examples typically illustrate the PEIA strategy in learning point (2).
3.5. Design considerations
The structural evolution of light-responsive systems is governed by the interplay between kinetic trapping and thermodynamic regulation, and this balance varies significantly across the four strategies discussed in this review. In photo-triggered polymerization-induced self-assembly, covalent bond formation often drives rapid structural fixation, unless dynamic covalent chemistry is introduced. In contrast, liquid crystal-assisted amplification and host–guest mediated assembly more commonly operate under thermodynamic or quasi-equilibrium conditions, where reversible molecular reorganization enables adaptive structural reconfiguration. PEIA occupies an intermediate case: while the primary photoevent occurs on ultrafast timescales, the subsequent ordering process may involve either reversible excited-state interaction modulation or non-equilibrium structural evolution, depending on relaxation pathways and cooperative effects. Across all four strategies, several common limitations emerge. Photofatigue and chromophore degradation may gradually reduce responsiveness under repeated cycling. Competitive side reactions or environmental perturbations (e.g., temperature, solvent polarity, oxygen) can alter interaction strength or phase stability. Diffusion-driven blurring may limit spatial precision at larger length scales. Furthermore, the trade-off between structural robustness and reversibility remains a fundamental constraint in many systems.4. Material properties enabled by light-triggered structural ordering
In this section, we will analyse structure–property relationships: how different levels of structural ordering affect material performance. Photo-responsive ordered materials have demonstrated broad application prospects in numerous fields due to their unique multi-structural changes and dynamic response characteristics.136 By introducing photo-responsive ability into address multiscale ordered structures, scientists have achieved precise control over material properties, enabling applications in micropatterning techniques, chiroptical modulation, luminescence control, photoresist, and so forth. This section will focus on several typical material properties, detailing how photo-responsive ordered materials play a unique role in these fields and analyzing their advantages and potential.4.1. Refractive index modulation and optical patterning
Due to changes in the refractive index of liquid crystal systems during photo-responsive processes, these materials can display different structural colors, enabling various visual patterning effects.137 In the process of photo-triggered structural ordering, light irradiation not only regulates molecular alignment but also significantly influences the material optical properties and structural manifestations. In recent years, refractive index modulation and optical patterning based on photo-responsive molecular materials have become research hotspots, with numerous studies focusing on exploring the effects of photo-triggered processes on materials such as liquid crystals and polymers.Katayama et al. revealed structural changes induced by light near topological defects through studying photoinduced molecular orientation changes.138 Using light-induced polarization/phase microscopy, they observed orientation changes of molecules around local topological defects, which can significantly alter the refractive index distribution. Osuji et al. studied the fast phase transition in liquid crystalline block co-oligomers (BCOs) under light irradiation.139 They found that UV and visible light could effectively control the alignment of BCO, enabling rapid switching of optical properties. Particularly, the light-driven phase transition affects the material anisotropy, resulting in significantly different refractive index characteristics under different irradiation conditions. Although the above systems involve a structural transition from order to disorder, they provide a new idea for the application of the system of light-responsive order transition in tunable optical devices.
To further optimize the relevant strategies, Li et al. found that under UV irradiation, the selective reflection spectrum of a LC material based on a cyclic azobenzenophane undergoes significant changes, thereby modulating its reflection behavior (Fig. 16). The regulation range of reflection wavelength can range from 450 nm to 800 nm. 120 Evans et al. explored light-driven phase transition phenomena in liquid crystal materials, where UV light induces a transition from a hexagonal to a cubic phase (Fig. 12c).122 This phase transition not only alters the material order but also significantly changes its optical properties, particularly in terms of refractive index and light scattering modulation. These examples typically illustrate the refractive index modulation portion in learning point (3).
 | ||
| Fig. 16 Photo-responsive systems enabling selective reflection tuning of LC materials. Polarized optical photomicrographs of a planar aligned film based on a cyclic azobenzenophane at room temperature, showing reversible phase transitions occurring by light irradiation of the sample inside a 5 µm cell.120 Reproduced with permission.120 Copyright © 2010 American Chemical Society. | ||
4.2. Chiroptical modulation
Chiral self-assembly systems based on photo-responsive materials show great potential for tuning chiroptical properties, particularly CPL.140,141 By introducing luminescent dyes into asymmetric systems, the system is likely to exhibit unique chiral luminescence, i.e., the CPL characteristics. Furthermore, by controlling the configuration or conformation changes of photo-responsive groups via light irradiation, the ordered structure and the chiroptical signal of the whole systems can be further regulated, achieving light-mediated tuning of CPL.As mentioned above, many research groups on light-induced LC phase transitions have discovered the phenomenon of chiroptical modulation and flipping. For example, Cheng et al. employed a cholesterol-based liquid crystal system with photo-responsive groups and achieved CPL signal inversion by adjusting the light intensity and wavelength (Fig. 11b and c).119 In this case, light-induced molecular configurational change caused chiral inversion in the system, thereby inverting the sign and intensity of its CPL signal. Light-tunable CPL characteristics can also be realized through PEIA effects. Our research found that light irradiation can induce molecular aggregation with boosting π–π stacking interactions within persulfurated arenes, subsequently regulating the structure of microphase separation in the chiral block copolymer, PS-b-(M-1@PLLA).134 This light-induced molecular aggregation allows the material to exhibit tunable CD and CPL responses under different lighting conditions. In this way, our study shows that light irradiation can not only regulate the order of molecular self-assembly but also affect the luminescence intensity and thus achieve an effective tuning of CPL from almost a silent level to a glum of 0.005 (Fig. 17). These examples typically illustrate the chiroptical modulation portion in learning point (3).
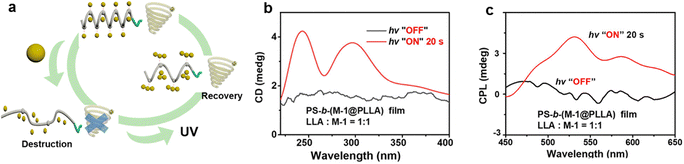 | ||
| Fig. 17 Photo-responsive systems enabling chiroptical amplification. (a) Schematic diagram of the in situ regulation of chirality and CPL by light irradiation in the PS-b-(M-1@PLLA) film. Corresponding (b) CD spectra and (c) CPL spectra before and after in situ photoirradiation for 20 s (λex = 365 nm).134 Reproduced with permission.134 Copyright © 2022 American Chemical Society. | ||
4.3. Modulation of photoluminescence properties
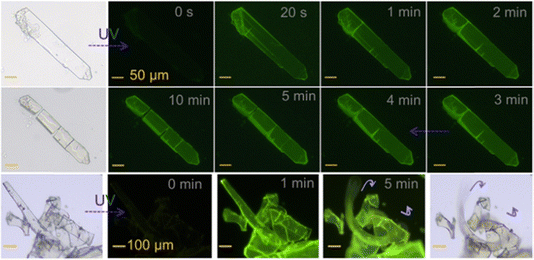
The modulation of PL properties is another important topic in photo-responsive materials research, especially concerning luminescence changes induced by the variations of ordered structures, with an application potential in smart light-emitting events. Through light-induced multiscale structural changes, not only can the fluorescence and RTP properties of materials be controlled, but also light-tunable afterglow (a long-lasting emission observable after ceasing excitation) can be achieved.142–145 This control over PL properties provides important pathways for designing novel optoelectronic devices and opens new avenues for applications of various smart optical materials.Our group investigated the effect of light on the luminescence behavior of tetrakis-(arylthio)benzene by studying the impact of photoinduced molecular rearrangement on its crystal structure.146 The PEIA strategy can effectively trigger molecular rearrangement, leading to changes of molecular arrangement in single crystals and consequently modulating its photoluminescence properties among yellow, blue, and violet luminescence. We also accessed fast light-regulated solid-state luminescent materials (phosphorescence activated within 20 s) with noticeable afterglow effect through the unsymmetric tetrakis-(arylthio)benzene-based single-luminophore engineering. With further continuous UV irradiation, macroscopic photomechanical behaviors such as directional cracking and bend ing could be observed in the crystalline state, clearly indicating the effect of PEIA in the solid state (Fig. 18).147 These examples typically illustrate the photoluminescent modulation portion in learning point (3).
 | ||
| Fig. 18 Photoinduced RTP enhancement and directional cracking behaviour of unsymmetric tetrakis-(arylthio)benzene crystals. Optical micrographs of crystals before and after UV irradiation showing photoinduced RTP enhancement and crystal bending behavior.147 Reproduced with permission.147 Copyright © 2024 Wiley-VCH GmbH. | ||
4.4. Photoresists and nanolithography
Photoresists and nanolithography technologies are widely used in micro-nano fabrication, particularly in the manufacturing of integrated circuits, optoelectronic devices, and ultra-high-density storage media. BCPs, as self-assembling polymeric materials, can guide the formation of nanostructures through their ordered transitions.148–150 With light-mediated self-assembly control, precise lithographic patterning can be achieved. This light-responsive nanostructure generation approach also shows great potential in those high-tech fields, such as high-precision photomask fabrication and magnetic storage media.151,152Watkins et al. demonstrated the application potential of photo-responsive materials in nanolithography by studying the light-induced ordering process with BCPs (Fig. 19a).153 They induced ordered alignment of polymer chains within a BCP film via UV irradiation (254 nm, 1 min) mediated by photoacid, forming highly regular nanostructures upon hydrogen bond remodeling between photoacid products and BCPs. Fig. 15 presents representative scanning force microscopy (SFM) images of both unexposed and exposed regions. The roughness of the unexposed area is attributed to crystallization (Fig. 19b and c). The UV-exposed area displays a fingerprint-like pattern, suggesting the formation of ordered cylindrical structures (Fig. 19d and e). Both height maps and phase maps reveal morphological features, clearly indicating that the photoinduced ordering process leads to the formation of topographical patterns. The self-assembly behavior of the photo-responsive BCPs allows control over material order at the nanoscale and points out some promising approaches for the development of photoresists and nanolithography technology. This example typically illustrates the lithographic property portion in learning point (3).
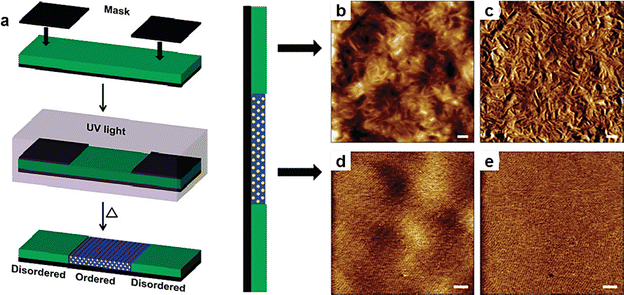 | ||
| Fig. 19 Photo-responsive systems enabling well-ordered cylindrical nanomorphology through region-selective irradiation. (a) Schematic of the photoinduced ordering process of a photoacid-mediated BCP film; SFM images of unexposed and exposed regions of the film: (b) height image of the unexposed region (z range = 10 nm); (c) phase image of the unexposed region (z range = 22°); (d) height image of the exposed region (z range = 4 nm); (e) phase image of the exposed region (z range = 9°). Scale bars = 150 nm.153 Reproduced with permission.153 Copyright © 2011, American Chemical Society. | ||
5. Summary and outlooks
Here, we attempt to present design guidelines and future directions for developing light-regulated materials with controllable structural ordering. This review has summarized the latest research progress in photo-responsive chemical systems enabling multiscale structural ordering. It focuses on the fundamental principles and emerging mechanisms underlying photo-responsive materials, as well as strategies for constructing and dynamically tuning ordered structures under light stimuli. We further discuss how structural ordering across different length scales governs material performance, and provide a comparative analysis of current approaches for achieving hierarchical ordering. A thorough understanding and control of the light-responsive arrangements at different scales is largely driven by their technological application value. From using the molecular isomerization in liquid crystals to achieve soft robotics technology, to utilizing the phase transition in block copolymers to realize the next-generation nano-lithography technology, these principles demonstrate that multi-scale arrangements are the key design parameters for achieving complex functions such as programmed movement, adaptive optics, and rewritable information storage.5.1. Summary of actionable design principles
Several generalizable design principles can be extracted from the four strategies discussed in this review. First, effective multiscale ordering requires a transduction mechanism that amplifies photoresponsive molecular events into cooperative intermolecular interactions. Without such amplification, structural reorganization remains localized. Second, the balance between kinetic trapping and thermodynamic equilibration fundamentally determines reversibility, spatial precision, and structural persistence. Third, spatial resolution is often limited not by the behaviors of the photoresponsive moieties but by diffusion and relaxation dynamics at larger length scales. Fourth, structural robustness and reversibility typically exhibit an intrinsic trade-off, requiring deliberate tuning of interaction strength, relaxation pathways, and environmental stability.At the same time, certain design constraints are strategy-specific. Covalent bond-forming systems often favor structural permanence but sacrifice reversibility; liquid-crystalline systems rely on mesogenic alignment and are sensitive to temperature and phase stability; host–guest systems depend critically on binding equilibria and competitive interactions; and photophysical strategies such as PEIA require chromophores with appropriate excited-state lifetimes and conformational flexibility. Recognizing which principles are broadly applicable and which are mechanism-dependent is essential for rational material design. These points collectively reflect the design guidelines in learning point (4).
5.2. Future challenges
Despite significant breakthroughs, several issues and challenges remain to be addressed, particularly at the application level. Specifically, the following aspects are key focus for future research.In summary, the research and development of photo-responsive ordered systems are progressing rapidly. The above paragraphs typically elaborate on the future directions for developing light-regulated materials with controllable structural ordering in learning point (4). With deepening theoretical research and continuous technological innovation, it is expected that photo-responsive materials will find practical applications in more fields, leading to transformative developments particularly in biomedicine, smart materials, and energy. In the future, we can anticipate the emergence of more efficient, stable, and easily scalable photo-responsive molecular materials, applied to a wider range of scenarios with practical needs.
Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work is supported by the National Natural Science Foundation of China (22275038 and 22575058).Notes and references
- H. Ma and J. Hao, Chem. Soc. Rev., 2011, 40, 5457–5471 RSC.
- J. S. Miller, Chem. Soc. Rev., 2011, 40, 3266–3296 RSC.
- H. K. Bisoyi and Q. Li, Acc. Chem. Res., 2014, 47, 3184–3195 CrossRef CAS PubMed.
- Q. Zhang, Y. Sun, C. He, F. Shi and M. Cheng, Adv. Sci., 2020, 7, 2002025 CrossRef CAS PubMed.
- T. Zhang, D. Wang and J. Liu, Adv. Mater., 2024, 36, 2408259 CrossRef CAS PubMed.
- H. Yin, K. Xing, Y. Zhang, D. M. A. S. Dissanayake, Z. Lu, H. Zhao, Z. Zeng, J.-H. Yun, D.-C. Qi and Z. Yin, Chem. Soc. Rev., 2021, 50, 6423–6482 RSC.
- R. Y. Liu, K. T. Tan, Y. F. Gong, Y. Z. Chen, Z. E. Li, S. L. Xie, T. He, Z. Lu, H. Yang and D. L. Jiang, Chem. Soc. Rev., 2021, 50, 120–242 RSC.
- Z. G. Zhao, R. C. Fang, Q. F. Rong and M. J. Liu, Adv. Mater., 2017, 29, 1703045 CrossRef PubMed.
- L. Xiang, Q. Li, C. Li, Q. Yang, F. Xu and Y. Mai, Adv. Mater., 2023, 35, 2207684 CrossRef CAS PubMed.
- G. Yang, H.-m Ding, Z. Kochovski, R. Hu, Y. Lu, Y.-q Ma, G. Chen and M. Jiang, Angew. Chem., Int. Ed., 2017, 56, 10691–10695 CrossRef CAS PubMed.
- L. Shao, J. Ma, J. L. Prelesnik, Y. Zhou, M. Nguyen, M. Zhao, S. A. Jenekhe, S. V. Kalinin, A. L. Ferguson, J. Pfaendtner, C. J. Mundy, J. J. De Yoreo, F. Baneyx and C.-L. Chen, Chem. Rev., 2022, 122, 17397–17478 CrossRef CAS PubMed.
- E. Potanina, C. Flindt, M. Moskalets and K. Brandner, Phys. Rev. X, 2021, 11, 021013 CAS.
- T. Liu, Z. He, J.-L. Wang, S.-Z. Sheng, Z.-Y. Xian, J.-W. Liu and S.-H. Yu, Angew. Chem., Int. Ed., 2025, 64, e202503613 CrossRef CAS PubMed.
- D. Guo, Y. Li, X. Zheng, F. Li, S. Chen, M. Li, Q. Yang, H. Li and Y. Song, J. Am. Chem. Soc., 2018, 140, 18–21 CrossRef CAS PubMed.
- Y. Yang, C.-C. Chen, M. C. Scott, C. Ophus, R. Xu, A. Pryor, L. Wu, F. Sun, W. Theis, J. Zhou, M. Eisenbach, P. R. C. Kent, R. F. Sabirianov, H. Zeng, P. Ercius and J. Miao, Nature, 2017, 542, 75–79 CrossRef CAS PubMed.
- A. M. Nie, Z. S. Zhao, B. Xu and Y. J. Tian, Nat. Mater., 2025, 24, 1172–1185 CrossRef CAS PubMed.
- Y. Wang, J. Chen, Y. Zhong, S. Jeong, R. Li and X. Ye, J. Am. Chem. Soc., 2022, 144, 13538–13546 CrossRef CAS PubMed.
- Y. Ku, K. Xu, L. Yan, K. Zhang, D. Song, X. Li, S. Li, S. Cheng and C. Shan, Carbon Energy, 2024, 6, e440 CrossRef CAS.
- F. Hong, F. Zhang, Y. Liu and H. Yan, Chem. Rev., 2017, 117, 12584–12640 CrossRef CAS PubMed.
- V. Kulikov, N. A. B. Johnson, A. J. Surman, M. Hutin, S. M. Kelly, M. Hezwani, D.-L. Long, G. Meyer and L. Cronin, Angew. Chem., Int. Ed., 2017, 56, 1141–1145 CrossRef CAS PubMed.
- F. Bošković, C. Maffeo, G. Patiño-Guillén, R. Tivony, A. Aksimentiev and U. F. Keyser, ACS Nano, 2024, 18, 15013–15024 CrossRef PubMed.
- S. I. S. Hendrikse, R. Contreras-Montoya, A. V. Ellis, P. Thordarson and J. W. Steed, Chem. Soc. Rev., 2022, 51, 28–42 RSC.
- Q. Luo, C. Hou, Y. Bai, R. Wang and J. Liu, Chem. Rev., 2016, 116, 13571–13632 CrossRef CAS PubMed.
- M. Xie, M. G. N. Derks, E. H. W. Koch, C. B. van Boven, M. Janlad, B. Bagheri, Z. Xu, D. Kovryzhenko, C. A. van Walree, A. Sobota, M. Weingarth, J. Wong-Ekkabut, M. Karttunen, E. Breukink, J. A. Killian, A. F. P. Sonnen and J. H. Lorent, J. Am. Chem. Soc., 2025, 147, 9175–9189 CrossRef CAS PubMed.
- S. MuhammedNazaar, J. Yao, M. R. Necelis, Y. C. Park, Z. Shen, M. D. Bridges, R. Guo, N. Swope, M. S. Rhee, M. Kim, K. H. Kim, W. L. Hubbell, K. G. Fleming, L. Columbus, S.-G. Kang and H. Hong, Sci. Adv., 2025, 11, eadv9568 CrossRef CAS PubMed.
- L. Jin, N. P. Kamat, S. Jena and J. W. Szostak, Small, 2018, 14, 1704077 CrossRef PubMed.
- B. Kwon, M. Lee, A. J. Waring and M. Hong, J. Am. Chem. Soc., 2018, 140, 8246–8259 CrossRef CAS PubMed.
- X. Fu, J.-M. Park, R. Liang, Y. Xue and M. Zhong, Prog. Polym. Sci., 2025, 170, 102027 CrossRef CAS.
- A. Xin, Y. Su, S. Feng, M. Yan, K. Yu, Z. Feng, K. Hoon Lee, L. Sun and Q. Wang, Adv. Mater., 2021, 33, 2006946 CrossRef CAS PubMed.
- C. Yuan, W. Ji, R. Xing, J. Li, E. Gazit and X. Yan, Nat. Rev. Chem., 2019, 3, 567–588 CrossRef CAS PubMed.
- C. Zhang, G. Chu, Z. Ruan, N. Tang, C. Song, Q. Li, W. Zhou, J. Jin, H. Haick, Y. Chen and D. Cui, ACS Nano, 2022, 16, 16584–16597 CrossRef CAS PubMed.
- P. Vukusic, J. R. Sambles and C. R. Lawrence, Nature, 2000, 404, 457 CrossRef CAS PubMed.
- Z. Chen, F. Fu, Y. Yu, H. Wang, Y. Shang and Y. Zhao, Adv. Mater., 2019, 31, 1805431 CrossRef PubMed.
- M. Kolle, P. M. Salgard-Cunha, M. R. J. Scherer, F. Huang, P. Vukusic, S. Mahajan, J. J. Baumberg and U. Steiner, Nat. Nanotechnol., 2010, 5, 511–515 CrossRef CAS PubMed.
- L. Zhang, S. Feih, S. Daynes, S. Chang, M. Y. Wang, J. Wei and W. F. Lu, Addit. Manuf., 2018, 23, 505–515 CAS.
- K. Chen and L. Li, Adv. Mater., 2019, 31, 1901115 CrossRef PubMed.
- L.-J. Chen and H.-B. Yang, Acc. Chem. Res., 2018, 51, 2699–2710 CrossRef CAS PubMed.
- K. L. Hvorecny, Nat. Struct. Mol. Biol., 2024, 31, 1147–1149 CrossRef CAS PubMed.
- H. J. Bae, S. Bae, J. Yoon, C. Park, K. Kim, S. Kwon and W. Park, Sci. Adv., 2017, 3, e1700071 CrossRef PubMed.
- Y. Deng, S. Gao, J. Liu, U. Gohs, E. Mäder and G. Heinrich, Mater. Horiz., 2017, 4, 389–395 RSC.
- G. Li, J. Fei, Y. Xu, Y. Li and J. Li, Adv. Funct. Mater., 2018, 28, 1706557 CrossRef.
- Z. Wang, X. Rong, D. Zhao, X. Zhang, C. He and N. Zhao, Mater. Horiz., 2025, 12, 10478–10497 RSC.
- K. Liu, H. Ding, S. Li, Y. Niu, Y. Zeng, J. Zhang, X. Du and Z. Gu, Nat. Commun., 2022, 13, 4563 CrossRef CAS PubMed.
- H.-T. Zhang, T. Zhang and X. Zhang, Adv. Sci., 2023, 10, 2300193 CrossRef PubMed.
- C. Li, X. Qin, Z. Zhang, Y. Lv, S. Zhang, Y. Fan, S. Liang, B. Guo, Z. Li, Y. Liu and D. Luo, Nano Today, 2022, 42, 101354 CrossRef CAS.
- T. Le, V. C. Epa, F. R. Burden and D. A. Winkler, Chem. Rev., 2012, 112, 2889–2919 CrossRef CAS PubMed.
- Y. Wang, I. C. Jenkins, J. T. McGinley, T. Sinno and J. C. Crocker, Nat. Commun., 2017, 8, 14173 CrossRef CAS PubMed.
- Y. Xiao, M. Zhou, M. Zeng and L. Fu, Adv. Sci., 2019, 6, 1801501 CrossRef PubMed.
- D.-G. Kang, Y.-B. Kook, I.-S. Kim, M. Rim, H. Ko, C.-H. Hsu, C.-L. Wang, K.-H. Ryu, D.-Y. Kim and K.-U. Jeong, Small, 2025, 21, 2404782 CrossRef CAS PubMed.
- T. Guo, L. Luo, L. Wang, F. Zhang, Y. Liu and J. Leng, ACS Nano, 2025, 19, 18003–18036 CrossRef CAS PubMed.
- S. Chandrasekaran, Q. Wang, Q. Liu, H. Wang, D. Qiu, H. Lu, Y. Liu, C. Bowen and H. Huang, Chem. Soc. Rev., 2025, 54, 2275–2343 RSC.
- Z. Meng, S. Miao, Y. Liu, Y. Li, Y. Ma, W. Luo and H. Huang, Mater. Horiz., 2025, 12, 8380–8408 RSC.
- H. Yin, X. Yu, Y. Zhu, Z. Hou, J. Cunha, Z. Liang and Z. Yu, Adv. Sci., 2025, 12, e03340 CrossRef CAS PubMed.
- M. Li, H. Hu, B. Liu, X. Liu, Z.-G. Zheng, H. Tian and W.-H. Zhu, J. Am. Chem. Soc., 2022, 144, 20773–20784 CrossRef CAS PubMed.
- N. P. Nazirkar, V. Tran, P. Bassène, A. Ndiaye, J. Barringer, J. Jiang, W. Cha, R. Harder, J. Shi, M. N'Gom and E. Fohtung, Adv. Mater., 2025, 37, 2415231 CrossRef CAS PubMed.
- A. S. Gliozzi, M. Miniaci, A. Chiappone, A. Bergamini, B. Morin and E. Descrovi, Nat. Commun., 2020, 11, 2576 CrossRef CAS PubMed.
- C. Wang, M. P. O’Hagan, Z. Li, J. Zhang, X. Ma, H. Tian and I. Willner, Chem. Soc. Rev., 2022, 51, 720–760 RSC.
- W. Xu, D. M. Sanchez, U. Raucci, H. Zhou, X. Dong, M. Hu, C. J. Bardeen, T. J. Martinez and R. C. Hayward, Nat. Mater., 2023, 22, 1152–1159 CrossRef CAS PubMed.
- O. P. Dimitriev, Chem. Rev., 2022, 122, 8487–8593 CrossRef CAS PubMed.
- Y. Deng, G. Long, Y. Zhang, W. Zhao, G. Zhou, B. L. Feringa and J. Chen, Light:Sci. Appl., 2024, 13, 63 CrossRef CAS PubMed.
- H. P. Lee and A. K. Gaharwar, Adv. Sci., 2020, 7, 2000863 CrossRef CAS PubMed.
- Y. Liu and P. Xing, Adv. Mater., 2023, 35, 2300968 CrossRef CAS PubMed.
- X. Jia and L. Zhu, Acc. Chem. Res., 2023, 56, 655–666 CrossRef CAS PubMed.
- J. Großkopf, T. Kratz, T. Rigotti and T. Bach, Chem. Rev., 2022, 122, 1626–1653 CrossRef PubMed.
- A. P. Demchenko, V. I. Tomin and P.-T. Chou, Chem. Rev., 2017, 117, 13353–13381 CrossRef CAS PubMed.
- I. M. Irshadeen, S. L. Walden, M. Wegener, V. X. Truong, H. Frisch, J. P. Blinco and C. Barner-Kowollik, J. Am. Chem. Soc., 2021, 143, 21113–21126 CrossRef CAS PubMed.
- A. Fihey, A. Perrier, W. R. Browne and D. Jacquemin, Chem. Soc. Rev., 2015, 44, 3719–3759 RSC.
- Y. Boeije and M. Olivucci, Chem. Soc. Rev., 2023, 52, 2643–2687 RSC.
- L. Salem, Science, 1976, 191, 822–830 CrossRef CAS PubMed.
- L. Huang and G. Han, Nat. Rev. Chem., 2024, 8, 238–255 CrossRef CAS PubMed.
- N. J. Turro and G. Schuster, Science, 1975, 187, 303–312 CrossRef CAS PubMed.
- T. Kumpulainen, B. Lang, A. Rosspeintner and E. Vauthey, Chem. Rev., 2017, 117, 10826–10939 CrossRef CAS PubMed.
- J.-Y. Xu, X. Tong, P. Yu, G. E. Wenya, T. McGrath, M. J. Fong, J. Wu and Z. M. Wang, Adv. Sci., 2018, 5, 1800221 CrossRef PubMed.
- H.-B. Cheng, S. Zhang, J. Qi, X.-J. Liang and J. Yoon, Adv. Mater., 2021, 33, 2007290 CrossRef CAS PubMed.
- M. Zhu, W. Wang, C. Zhang, L. Zhu and S. Yang, Adv. Fiber Mater., 2021, 3, 172–179 CrossRef CAS.
- H. M. D. Bandara and S. C. Burdette, Chem. Soc. Rev., 2012, 41, 1809–1825 RSC.
- C. Wang, M. D. J. Waters, P. Zhang, J. Suchan, V. Svoboda, T. T. Luu, C. Perry, Z. Yin, P. Slavíček and H. J. Wörner, Nat. Chem., 2022, 14, 1126–1132 CrossRef CAS PubMed.
- U. Raucci, H. Weir, C. Bannwarth, D. M. Sanchez and T. J. Martínez, Nat. Commun., 2022, 13, 2091 CrossRef CAS PubMed.
- F. D. Lewis, R. S. Kalgutkar and J.-S. Yang, J. Am. Chem. Soc., 1999, 121, 12045–12053 CrossRef CAS.
- M. Irie, T. Fukaminato, K. Matsuda and S. Kobatake, Chem. Rev., 2014, 114, 12174–12277 CrossRef CAS PubMed.
- S. Lin, K. G. Gutierrez-Cuevas, X. Zhang, J. Guo and Q. Li, Adv. Funct. Mater., 2021, 31, 2007957 CrossRef CAS.
- L. Hou, T. Leydecker, X. Zhang, W. Rekab, M. Herder, C. Cendra, S. Hecht, I. McCulloch, A. Salleo, E. Orgiu and P. Samorì, J. Am. Chem. Soc., 2020, 142, 11050–11059 CrossRef CAS PubMed.
- R. Klajn, Chem. Soc. Rev., 2014, 43, 148–184 RSC.
- V. M. Lechner, M. Nappi, P. J. Deneny, S. Folliet, J. C. K. Chu and M. J. Gaunt, Chem. Rev., 2022, 122, 1752–1829 CrossRef CAS PubMed.
- J. Zhu, X.-W. Sun, X. Yang, S.-N. Yu, L. Liang, Y.-Z. Chen, X. Zheng, M. Yu, L. Yan, J. Tang, W. Zhao, X.-J. Yang and B. Wu, Angew. Chem., Int. Ed., 2023, 62, e202314510 CrossRef CAS PubMed.
- J. Gemen, J. R. Church, T.-P. Ruoko, N. Durandin, M. J. Białek, M. Weißenfels, M. Feller, M. Kazes, M. Odaybat, V. A. Borin, R. Kalepu, Y. Diskin-Posner, D. Oron, M. J. Fuchter, A. Priimagi, I. Schapiro and R. Klajn, Science, 2023, 381, 1357–1363 CrossRef CAS PubMed.
- C.-W. Chang, Y.-C. Lu, T.-T. Wang and E. W.-G. Diau, J. Am. Chem. Soc., 2004, 126, 10109–10118 CrossRef CAS PubMed.
- W.-J. Li, W.-T. Xu, X.-Q. Wang, Y. Jiang, Y. Zhu, D.-Y. Zhang, X.-Q. Xu, L.-R. Hu, W. Wang and H.-B. Yang, J. Am. Chem. Soc., 2023, 145, 14498–14509 CrossRef CAS PubMed.
- K. P. Xie, Z. Y. Ruan, B. H. Lyu, X. X. Chen, X. W. Zhang, G. Z. Huang, Y. C. Chen, Z. P. Ni and M. L. Tong, Angew. Chem., Int. Ed., 2021, 60, 27144–27150 CrossRef CAS PubMed.
- T. Dang, Y. He, X. Yu, Z.-Y. Zhang, L. Li and T. Li, J. Am. Chem. Soc., 2025, 147, 38677–38683 CrossRef CAS PubMed.
- Y. Li, B. Xue, J. Yang, J. Jiang, J. Liu, Y. Zhou, J. Zhang, M. Wu, Y. Yuan, Z. Zhu, Z. J. Wang, Y. Chen, Y. Harabuchi, T. Nakajima, W. Wang, S. Maeda, J. P. Gong and Y. Cao, Nat. Chem., 2024, 16, 446–455 CrossRef CAS PubMed.
- J. Wang, S. Shen, X. Li, J. Li and F. Dong, Chem. Rev., 2025, 125, 7811–7917 CrossRef CAS PubMed.
- Z. Huang, N. Luo, C. Zhang and F. Wang, Nat. Rev. Chem., 2022, 6, 197–214 CrossRef PubMed.
- S. Crespi and M. Fagnoni, Chem. Rev., 2020, 120, 9790–9833 CrossRef CAS PubMed.
- C.-Y. Huang, J. Li and C.-J. Li, Chem. Sci., 2022, 13, 5465–5504 RSC.
- X. Yao, H. Shi, X. Wang, H. Wang, Q. Li, Y. Li, J. Liang, J. Li, Y. He, H. Ma, W. Huang and Z. An, Sci. China: Chem., 2022, 65, 1538–1543 CrossRef CAS.
- Y. N. Hong, J. W. Y. Lam and B. Z. Tang, Chem. Soc. Rev., 2011, 40, 5361–5388 RSC.
- M. Gao, R. Wu, Y. Zhang, Y. Meng, M. Fang, J. Yang and Z. Li, J. Am. Chem. Soc., 2025, 147, 2653–2663 CrossRef CAS PubMed.
- D. Escudero, Acc. Chem. Res., 2016, 49, 1816–1824 CrossRef CAS PubMed.
- S. Huang, A. Abedi Syed Ali, Z. Li, R. Huang, X. Yan, M. Izadyar, Q. Qiao, Y. Fang, Z. Xu and X. Liu, CCS Chem., 2024, 6, 2804–2813 CrossRef CAS.
- H. Zhang, L. Du, L. Wang, J. Liu, Q. Wan, R. T. K. Kwok, J. W. Y. Lam, D. L. Phillips and B. Z. Tang, J. Phys. Chem. Lett., 2019, 10, 7077–7085 CrossRef CAS PubMed.
- J. Yang, X. Zhen, B. Wang, X. Gao, Z. Ren, J. Wang, Y. Xie, J. Li, Q. Peng, K. Pu and Z. Li, Nat. Commun., 2018, 9, 840 CrossRef PubMed.
- X. Jia, C. Shao, X. Bai, Q. Zhou, B. Wu, L. Wang, B. Yue, H. Zhu and L. Zhu, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 4816–4821 CrossRef CAS PubMed.
- S. Wu, Z. Li, G. V. Baryshnikov, M. Zhang, B. Jiang and L. Zhu, Chem. Mater., 2026, 38, 1325–1332 CrossRef CAS.
- G. Xu, A. Chen, F. Feng, Y. Wu and X. Wang, ACS Nano, 2024, 18, 35347–35355 CrossRef CAS PubMed.
- Z. Li, C. Y. Gao, D. Su, C. Jia and X. Guo, ACS Mater. Lett., 2021, 3, 1484–1502 CrossRef CAS.
- S. Datta, H. Itabashi, T. Saito and S. Yagai, Nat. Chem., 2025, 17, 477–492 CrossRef CAS PubMed.
- C. S. Zehe, J. A. Hill, N. P. Funnell, K. Kreger, K. P. van der Zwan, A. L. Goodwin, H.-W. Schmidt and J. Senker, Angew. Chem., Int. Ed., 2017, 56, 4432–4437 CrossRef CAS PubMed.
- J. Neugebauer and T. Hickel, WIREs Comput. Mol. Sci., 2013, 3, 438–448 CrossRef CAS PubMed.
- P. J. Santos, P. A. Gabrys, L. Z. Zornberg, M. S. Lee and R. J. Macfarlane, Nature, 2021, 591, 586–591 CrossRef CAS PubMed.
- M. D. Ward and P. R. Raithby, Chem. Soc. Rev., 2013, 42, 1619–1636 RSC.
- H. K. Bisoyi and Q. Li, Chem. Rev., 2016, 116, 15089–15166 CrossRef CAS PubMed.
- Z.-X. Chang, R.-M. Zhu, C.-Y. Hong and W.-J. Zhang, Sci. China: Chem., 2024, 67, 390–397 CrossRef CAS.
- S. Guan and A. Chen, ACS Macro Lett., 2020, 9, 14–19 CrossRef CAS PubMed.
- W. Chen, J. Y. Wang, W. Zhao, L. Li, X. Wei, A. C. Balazs, K. Matyjaszewski and T. P. Russell, J. Am. Chem. Soc., 2011, 133, 17217–17224 CrossRef CAS PubMed.
- M. Han, Y. Luo, B. Damaschke, L. Gomez, X. Ribas, A. Jose, P. Peretzki, M. Seibt and G. H. Clever, Angew. Chem., Int. Ed., 2016, 55, 445–449 CrossRef CAS PubMed.
- A. Ryabchun, D. Yakovlev, A. Bobrovsky and N. Katsonis, ACS Appl. Mater. Interfaces, 2019, 11, 10895–10904 CrossRef CAS PubMed.
- S. Pan, M. F. Ni, B. Mu, Q. Li, X. Y. Hu, C. Lin, D. Z. Chen and L. Y. Wang, Adv. Funct. Mater., 2015, 25, 3571–3580 CrossRef CAS.
- Y. Li, Y. Chen, J. Luo, Y. Quan and Y. Cheng, Adv. Mater., 2024, 36, e2312331 CrossRef PubMed.
- M. Mathews, R. S. Zola, S. Hurley, D. K. Yang, T. J. White, T. J. Bunning and Q. Li, J. Am. Chem. Soc., 2010, 132, 18361–18366 CrossRef CAS PubMed.
- T. Kosa, L. Sukhomlinova, L. Su, B. Taheri, T. J. White and T. J. Bunning, Nature, 2012, 485, 347–349 CrossRef CAS PubMed.
- B. E. Jones, J. L. Greenfield, N. Cowieson, M. J. Fuchter and R. C. Evans, J. Am. Chem. Soc., 2024, 146, 12315–12319 CrossRef CAS PubMed.
- Y. X. Chen, S. Huang, T. J. Wang and H. F. Yu, Macromolecules, 2020, 53, 1486–1493 CrossRef CAS.
- B. V. K. J. Schmidt and C. Barner-Kowollik, Angew. Chem., Int. Ed., 2017, 56, 8350–8369 CrossRef CAS PubMed.
- X. Ma and H. Tian, Acc. Chem. Res., 2014, 47, 1971–1981 CrossRef CAS PubMed.
- X. J. Loh, Mater. Horiz., 2014, 1, 185–195 RSC.
- X. K. Ma, W. Zhang, Z. X. Liu, H. Y. Zhang, B. Zhang and Y. Liu, Adv. Mater., 2021, 33, 2007476 CrossRef CAS PubMed.
- H. Yang, B. Yuan, X. Zhang and O. A. Scherman, Acc. Chem. Res., 2014, 47, 2106–2115 CrossRef CAS PubMed.
- G. Liu, C. Tian, X. Fan, X. Xue, L. Feng, C. Wang and Y. Liu, JACS Au, 2023, 3, 2550–2556 CrossRef CAS PubMed.
- N. Cheng, Y. Chen, J. Yu, J. J. Li and Y. Liu, ACS Appl. Mater. Interfaces, 2018, 10, 6810–6814 CrossRef CAS PubMed.
- H. L. Sun, Y. Chen, X. Han and Y. Liu, Angew. Chem., Int. Ed., 2017, 56, 7062–7065 CrossRef CAS PubMed.
- M. Liu, B. Wu, G. V. Baryshnikov, S. Shen, H. Sun, X. Gu, H. Agren, Y. Xu, Q. Zou, D. H. Qu and L. Zhu, Chem. Sci., 2024, 15, 12569–12579 RSC.
- M. Liu, B. Wu, S. Shen, H. Sun, X. Gu, S. Li, Z. Tang, J. Cheng, X. Ma, M. Zhang, Y. Xu and L. Zhu, ACS Macro Lett., 2025, 14, 93–100 CrossRef CAS PubMed.
- B. Yue, X. Feng, C. Wang, M. Zhang, H. Lin, X. Jia and L. Zhu, ACS Nano, 2022, 16, 16201–16210 CrossRef CAS PubMed.
- B. Yue, X. Jia, G. V. Baryshnikov, X. Jin, X. Feng, Y. Lu, M. Luo, M. Zhang, S. Shen, H. Agren and L. Zhu, Angew. Chem., Int. Ed., 2022, 61, e202209777 CrossRef CAS PubMed.
- F. Xu and B. L. Feringa, Adv. Mater., 2023, 35, 2204413 Search PubMed.
- P. Zhang, L. T. de Haan, M. G. Debije and A. P. H. J. Schenning, Light:Sci. Appl., 2022, 11, 248 CrossRef CAS PubMed.
- H. Sakanoue, W. Y. Sohn and K. Katayama, ACS Omega, 2019, 4, 13936–13942 Search PubMed.
- C. Lee, D. Ndaya, R. Bosire, N. K. Kim, R. M. Kasi and C. O. Osuji, J. Am. Chem. Soc., 2022, 144, 390–399 Search PubMed.
- L. L. Zhu, X. Li, S. J. Wu, K. T. Nguyen, H. Yan, H. Ågren and Y. L. Zhao, J. Am. Chem. Soc., 2013, 135, 9174–9180 CrossRef CAS PubMed.
- S. E. Penty, G. R. F. Orton, D. J. Black, R. Pal, M. A. Zwijnenburg and T. A. Barendt, J. Am. Chem. Soc., 2024, 146, 5470–5479 CrossRef CAS PubMed.
- X. Chen, X. F. Hou, X. M. Chen and Q. Li, Nat. Commun., 2024, 15, 5401 CrossRef CAS PubMed.
- J. Huang, Y. Jiang, Q. Chen, H. Xie and S. Zhou, Nat. Commun., 2023, 14, 7131 CrossRef CAS PubMed.
- D.-X. Feng, Y. Mu, J. Li, S. De Han, J.-H. Li, H.-L. Sun, M. Pan, J.-X. Hu and G.-M. Wang, Adv. Funct. Mater., 2023, 33, 2305796 CrossRef CAS.
- M. Komura, H. Sotome, H. Miyasaka, T. Ogawa and Y. Tani, Chem. Sci., 2023, 14, 5302–5308 RSC.
- S. Shen, G. Baryshnikov, B. B. Yue, B. Wu, X. P. Li, M. Zhang, H. Ågren and L. L. Zhu, J. Mater. Chem. C, 2021, 9, 11707–11714 RSC.
- H. Sun, Z. Yu, C. Li, M. Zhang, S. Shen, M. Li, M. Liu, Z. Li, D. Wu and L. Zhu, Angew. Chem., Int. Ed., 2025, 64, e202413827 CrossRef CAS PubMed.
- G. G. Yang, H. J. Choi, K. H. Han, J. H. Kim, C. W. Lee, E. I. Jung, H. M. Jin and S. O. Kim, ACS Appl. Mater. Interfaces, 2022, 14, 12011–12037 CrossRef CAS PubMed.
- L. Yao and J. J. Watkins, ACS Nano, 2013, 7, 1513–1523 CrossRef CAS PubMed.
- L. L. Zhu, H. Tran, F. L. Beyer, S. D. Walck, X. Li, H. Ågren, K. L. Killops and L. M. Campos, J. Am. Chem. Soc., 2014, 136, 13381–13387 CrossRef CAS PubMed.
- C. E. Kim, J. W. Shim, S. J. Ham and S. H. Kim, ACS Omega, 2025, 10, 8579–8587 CrossRef CAS PubMed.
- W. C. Xu, C. W. Liu, S. F. Liang, D. C. Zhang, Y. Z. Liu and S. Wu, Adv. Mater., 2022, 34, 2202150 CrossRef CAS PubMed.
- V. K. Daga, E. L. Schwartz, C. M. Chandler, J. K. Lee, Y. Lin, C. K. Ober and J. J. Watkins, Nano Lett., 2011, 11, 1153–1160 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |