An–imidophosphorane (An = U–Pu) bond covalency and proton-coupled electron transfer thermodynamics driven by orbital energy matching
Chad M.
Studvick
 a,
Sourav
Dey
b,
Kaitlyn S.
Engle
c,
Julie E.
Niklas
a,
Sourav
Dey
b,
Kaitlyn S.
Engle
c,
Julie E.
Niklas
 c,
Jonah W.
Sprosty
a,
Henry S.
La Pierre
c,
Jonah W.
Sprosty
a,
Henry S.
La Pierre
 *cde and
Ivan A.
Popov
*cde and
Ivan A.
Popov
 *b
*b
aDepartment of Chemistry, University of Akron, Akron, Ohio 44325-3601, USA
bDepartment of Chemistry, Washington State University, Pullman, Washington 99164, USA. E-mail: ivan.popov@wsu.edu
cSchool of Chemistry and Biochemistry, Georgia Institute of Technology, Atlanta, Georgia 30332-0400, USA
dNuclear and Radiological Engineering and Medical Physics Program, School of Mechanical Engineering, Georgia Institute of Technology, Atlanta, Georgia 30332-0400, USA
ePhysical Sciences Division, Pacific Northwest National Laboratory, Richland, Washington 99352, USA
First published on 12th February 2026
Abstract
A series of mid-actinide (An = U–Pu) tetrahomoleptic complexes supported by highly electron-donating imidophosphorane ligands, NPC ([NPtBu(pyrr)2]−, where tBu = C(CH3)3; pyrr = pyrrolidinyl = N(C4H8)), are systematically investigated computationally and experimentally to elucidate the nature of actinide–ligand (An–L) covalency across the An3+/4+/5+ oxidation states. Trends in An–L bonding and redox properties for these complexes, together with their protonated counterparts, are examined using orbital-, electron density-, and energy-decomposition-based methods. This integrated approach reveals progressively improved energy matching between α-spin An 5f and Nim 2p orbitals with increasing atomic number and oxidation state, becoming particularly pronounced in the ligand-dominant π-bonding orbitals of An4+ and An5+. In contrast to the An3+ species, the enhanced An 5fπ contributions in the higher-valent counterparts drive the increase in An–Nim covalency for later An, thereby inverting the covalency trend to U < Np < Pu. Redistribution of electron density towards the An and Nim atomic basins due to the growing energy-matching assisted covalency correlates with higher pKa values and increased Nim–H bond dissociation free energies in protonated An4+ complexes. Electron density at Nim in An4+ shows a linear correlation with the pKa values calculated via the Bordwell equation. Calculations predict a cathodic shift of 0.84–1.00 V in the redox couples upon protonation, a trend validated when experimentally accessible. These findings demonstrate an increasing role of covalency driven by orbital energy matching from U to Pu in tuning the thermodynamic driving force for proton-coupled electron transfer in the An5+ species.
Introduction
Understanding the origin of metal–ligand (M–L) bonding interactions and their relationship to the thermodynamics of actinide (An) chemical reactivity is essential for advancing separation techniques.1–9 Consequently, the study of f-element M–L bond covalency10–23 has been a cornerstone of coordination chemistry and spectroscopy for both lanthanides and actinides since Seaborg's seminal work in 1954,24 driving the development of contemporary computational methods. A central goal has been to understand and leverage the An 5f orbitals, which are generally more diffuse and higher in energy than lanthanide 4f orbitals. The differences in the participation of the f orbitals in the M–L bonding, which define the degree of covalency, may also lead to differences in chemical reactivity.23,25–32 Higher-oxidation-state An complexes offer an ideal framework for examining the role of 5f orbitals in chemical bonding because their contributions become more pronounced. Ultimately, developing a fundamental understanding of M–L bond covalency in a systematic fashion across multiple oxidation states and the An series remains a crucial goal.33–39Previous studies have focused on applying theoretical methods to interpret spectroscopic and diffraction data, aiming to define the role of covalent bonding in various An complexes. In this context, methods such as X-ray diffraction (XRD),32,40–44 X-ray absorption spectroscopy,45–49 resonant inelastic X-ray scattering maps,45–49 Mössbauer spectroscopy,42 UV-vis-nIR spectroscopies,41,50 nuclear magnetic resonance (NMR),32,41,44,51 electron paramagnetic resonance,31,50 and magnetic measurements43,46,50,52 serve as valuable tools for experimental assessment of bond covalency, 4f/5f orbital contributions, and M oxidation states. These studies, though, are focused on complexes independent of their chemical reactivity. This limitation is largely derived from the absence of comparative reactions that are accessible experimentally across the An series.
Theoretically, covalency can be treated as a perturbation to the ionic limit, arising from orbital overlap and orbital energy matching in heteroatomic M–L bonds.6,36,53–75 The latter has garnered increasing attention in recent years and has been noted as a driving force for An–L covalency in borate and dipicolinate compounds.6,43,54,57,64,76,77 The interplay between orbital overlap and orbital energy matching varies depending on the oxidation state and identity of the An. These differences arise from changes in the radial extent and energy of the An 5f/6d orbitals relative to the L orbitals, which has been noted for a number of systematic studies of An species.37,39,60,61,65,78,79 Enhanced orbital mixing arising from improved orbital energy matching has been reported for Ln systems as well.80,81 For example, the increased 4f contributions in the Ln3+ complexes supported by the 3,5-di-tert-butyl-o-semiquinone ligands80 was attributed to the greater energetic accessibility of the 4f shell, leading to deviations from expected bonding trends across the Ln series (La–Gd, except Pm).
Understanding the factors that govern the M–L bonding may also be key to determining the thermodynamic aspects of a complex's reactivity. For example, it was suggested previously that the more covalent character of the U–C bonds in (η5-C5Me5)2U[η2-C2(SiMe3)2] makes it prone to react with alkynes, in contrast to the Th counterpart exhibiting more ionic bonding.82 Likewise, recent theoretical and inductively coupled plasma tandem mass spectrometer studies on the activation of methane by An cations suggest that there is a crossover in reactivity and bonding between Np+ and Pu+. It is described as a shift from An–L+ bonding defined by orbital overlap for the early An to bonding driven by orbital energy matching for the later An.83 These ties between M–L covalency and reactivity have previously been elucidated for other various An and transition metal complexes.84–91 Likewise, a recent combined theoretical and experimental study of MIV–cyclopropenyl complexes (M![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) =
= ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ti, Zr, Ce, Hf, Th) showed that the 4f-orbital covalency can give rise to the distinct chemical reactivity observed across a series of isostructural and isoelectronic d- and f-block complexes.92 Thus, establishing the origin of M–L covalency is indispensable for uncovering and rationalizing reactivity patterns in f-block species.
Ti, Zr, Ce, Hf, Th) showed that the 4f-orbital covalency can give rise to the distinct chemical reactivity observed across a series of isostructural and isoelectronic d- and f-block complexes.92 Thus, establishing the origin of M–L covalency is indispensable for uncovering and rationalizing reactivity patterns in f-block species.
Proton-coupled electron transfer (PCET) is a fundamental chemical transformation and has been studied extensively across the periodic table.93–95 Notably, the kinetics and thermodynamics of PCET reactions of transuranic complexes have only been recently defined,20,21,96 and a few reports of U complexes involved in PCET have been reported.97,98 Most existing studies of An PCET focus on the ubiquitous actinyl moiety in aqueous solution,99–101 where the fundamental PCET reaction step is often kinetically obscured by subsequent processes such as disproportionation, oligomerization, and hydrolysis. Recently, we quantified the kinetics and thermodynamics of PCET reactions of transuranic non-actinyl imidophosphorane compounds of Np and Pu20,21 and examined the redox chemistry of the An5+/4+ couple in [An(NPC)4] (An = Np, Pu) (NPC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) =
= ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [NPtBu(pyrr)2]−, tBu
[NPtBu(pyrr)2]−, tBu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) =
= ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) C(CH3)3, and pyrr
C(CH3)3, and pyrr![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) =
= ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) pyrrolidinyl = N(C4H8)). The calculations revealed increasing An–Nim covalent interactions across the U–Pu species (U < Np < Pu) attributed to enhanced energy matching between An 5f and Nim 2p orbitals. This trend correlates with greater electron density accumulation on the Nim atoms within the An5+ species, aligning with the observed PCET reaction rates: the Pu5+ complex undergoes PCET ∼5 orders of magnitude faster than Np5+, while the U5+ counterpart does not undergo a PCET reaction under the same conditions.
pyrrolidinyl = N(C4H8)). The calculations revealed increasing An–Nim covalent interactions across the U–Pu species (U < Np < Pu) attributed to enhanced energy matching between An 5f and Nim 2p orbitals. This trend correlates with greater electron density accumulation on the Nim atoms within the An5+ species, aligning with the observed PCET reaction rates: the Pu5+ complex undergoes PCET ∼5 orders of magnitude faster than Np5+, while the U5+ counterpart does not undergo a PCET reaction under the same conditions.
This homoleptic imidophosphorane ligand framework allows for a unique opportunity to study the PCET reaction and An–L covalency in a wide range of oxidation states across the mid-An (U–Pu). Since the An 5f orbitals progressively decrease in energy as the atomic number and oxidation state increase, systematic studies within a conserved ligand field are particularly relevant for understanding the trends in orbital overlap and orbital energy matching. Hence, in this work, a series of mid-An complexes are investigated employing the tetrahomoleptic [NPC]− ligand architecture (1-Anq+; 1-An = [Anq+(NPC)4]n, q = 3–5; n = −1, 0, +1) as well as their respective single ligand protonated congeners (2-Anq+; 2-An = [Anq+(HNPC)(NPC)3]n, q = 3, 4; n = +1), and higher-oxidation state 1-U6+, 2-U5+, 2-U6+, and 2-Np5+ complexes (Fig. 1). The origin of covalent interactions and covalency trends of the An–Nim bonds across U, Np, and Pu are evaluated as a function of the An oxidation state through orbital-based, electron density-based, and energy-decomposition approaches. Additionally, the redox chemistry of these complexes is modelled and the Nim–H bond dissociation free energies (BDFEs) and pKa values are determined. The thermodynamic drive for PCET in the An5+ species is rationalized through changes in the An–Nim covalency and electron density at the Nim atoms, arising in part from the enhanced An 5f–Nim 2p orbital energy matching and inherent redox properties of each complex. These computational insights are supported by experimental single-crystal XRD (SC-XRD) and electrochemical measurements on selected compounds, enabling a comparative analysis.
 | ||
| Fig. 1 Lewis structures of 1-Anq+ and 2-Anq+ (An = U–Pu). Full value range for each individual variable includes: An charge (q = 3, 4, 5, 6); complex charge (n = −1, 0, +1, +2). | ||
Experimental methods
The complexes of 2-U4+ and 2-Np4+ were prepared via protonation of 1-U4+ and 1-Np4+, respectively, with [H(OEt2)2][B(ArF5)4]; a mechanistically distinct preparation of 2-Np4+ compared to the previously published PCET reaction of 1-Np5+.20 This new synthetic method for 2-Np4+ increased the yield from a few crystals, only enough to obtain SC-XRD characterization, to near quantitative yield (99%) facilitating the UV-vis-nIR characterization and electrochemical analysis of this material.20 This high yield also enabled an attempt to reduce 2-Np4+ with KC8 to form the putative 2-Np3+. A colour change was observed, consistent with the colour change seen during the reduction of 1-Np4+ to 1-Np3+; however, 2-Np3+ could not be isolated (see the SI for further synthetic details).The synthesis of 2-U4+ was only possible via protonation with [H(OEt2)2][B(ArF5)4] (90%) as 1-U5+ is inactive for PCET. All compounds in this series retain a pseudo-tetrahedral core geometry around the An, with those containing a protonated ligand having slightly lower τ4 values due to the presence of the proton altering the inner coordination sphere (Table S73).102 In the 1-Anq+ series, all complexes within each oxidation state (q = 3–5) are isomorphic. Similarly, 2-Np4+ and 2-Pu4+ are isomorphic, whereas 2-U4+ is only isostructural with these two, crystallizing in a different space group. The unique protonated ligand in 2-U4+ is disordered over two positions, H1D and H1E, with 60% and 40% occupancies, respectively (Fig. S63). Due to this disorder and proximity to the heavy atom, U, refinement of the H atom positions was carried out using a riding model. This presents another key distinction between the U complex and its Np and Pu analogues, as in the latter cases the protons were defined in the electron difference map and refined as protons localized on N1D within an applied bond length parameter. Further crystallographic modelling details and selected structural metrics for the full series are highlighted in the SI and Table S73.
The solution NMR of 2-U4+ (heteronuclear and 2D) agrees with the SC-XRD assignment. The 31P{1H} spectrum shows two signals in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio for their respective intensities at 381.0 ppm (NPC) and –90.9 ppm (HNPC). Each ligand exhibits a substantial paramagnetic shift relative to the free ligand HNPC (47.5 ppm), with shifts greater than those observed for either the Np or Pu analogues. Comparative stacked spectra are presented in Fig. S57.
1 ratio for their respective intensities at 381.0 ppm (NPC) and –90.9 ppm (HNPC). Each ligand exhibits a substantial paramagnetic shift relative to the free ligand HNPC (47.5 ppm), with shifts greater than those observed for either the Np or Pu analogues. Comparative stacked spectra are presented in Fig. S57.
Computational methods
Density functional theory (DFT) calculations
The geometries of all complexes are optimized in the gas phase using the PBE0 hybrid DFT functional103 as implemented in Gaussian 16 (Version A.03).104 The ECP60MWB105 small core quasi-relativistic pseudopotential and ECP60MWB_ANO106 basis sets are employed to describe U–Pu. All remaining atoms are treated with the all-electron Pople basis set 6-311G(d).107 For clarity, this level of theory will hereafter be referred to as PBE0 ECP. This computational protocol has been successfully applied in our previous studies of An-based imidophosphorane complexes,20–23 accurately reproducing geometries, electronic structures, and thermodynamic trends, and is therefore adopted in the present work. Geometrical analysis shows good agreement between the calculated and experimentally available structures, with An–Nim and P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Nim bond lengths deviating less than 1.7% and 1.4%, respectively, and all complexes adopting a pseudo-tetrahedral geometry (τ4 = 0.88–0.99) (Tables S3 and S4).
Nim bond lengths deviating less than 1.7% and 1.4%, respectively, and all complexes adopting a pseudo-tetrahedral geometry (τ4 = 0.88–0.99) (Tables S3 and S4).
An–Nim bonding interactions were characterized using the Quantum Theory of Atoms in Molecules (QTAIM)108 by evaluating the electron density ρ(r) and delocalization index δ(An,Nim) with Multiwfn.109 Complementary orbital-based insight was obtained from the Adaptive Natural Density Partitioning (AdNDP) analysis110 using the Natural Bond Orbital (NBO7) method.111 The interacting quantum atoms (IQA) energy decomposition method,112,113 as implemented in AIMAll,114 was used to partition the An–Nim interaction energy into classical electrostatic (Vcl) and exchange–correlation (Vxc) terms, with Vxc reflecting electron sharing and treated as the covalent component. IQA is a real-space, topology-based scheme that has proven effective for quantifying chemical bonding in An.55,115,116
All electrochemical calculations are referenced to the Fc+/0 couple, which reliably reproduces experimental redox potentials in related An systems.20,21,23 Gas-phase enthalpic and entropic contributions were omitted because they have little effect on computed redox potentials.117 Covalency metrics and redox potentials were also evaluated using (1) the PBE functional and (2) an all-electron basis set with relativistic DKH corrections. Both approaches yielded qualitatively similar trends to those obtained with the PBE0 ECP protocol (see SI for details).
BDFEs are calculated using total energies from the single point energy calculations in THF using the Douglas–Kroll–Hess fourth order (DKH4) relativistic Hamiltonian including spin–orbit coupling (SOC) and zero-point energy and thermal energy corrections from the corresponding gas phase optimizations as implemented in Gaussian 16. The procedure for calculating N–H BDFEs 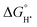 is based on the experimentally determined free energy of H atom transfer from 1-hydroxy-2,2,6,6-tetramethyl-piperidine (TEMPO-H) in THF
is based on the experimentally determined free energy of H atom transfer from 1-hydroxy-2,2,6,6-tetramethyl-piperidine (TEMPO-H) in THF 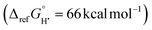 118 (see the SI for details). The pKa values are calculated from the Bordwell equation, BDFE = 1.37 pKa + 23.06E0 + CG, where CG is the solvent-specific H+/H˙ standard reduction potential94 (66 kcal mol−1 in THF119), E0 is the experimentally determined redox potential, and BDFE is the calculated value described above. To minimize uncertainty from computational approximations, pKa were determined only for species with experimentally measured redox potentials. Molecular electrostatic potentials at the Nim atoms were computed with Multiwfn, given their established correlation with pKa values.
118 (see the SI for details). The pKa values are calculated from the Bordwell equation, BDFE = 1.37 pKa + 23.06E0 + CG, where CG is the solvent-specific H+/H˙ standard reduction potential94 (66 kcal mol−1 in THF119), E0 is the experimentally determined redox potential, and BDFE is the calculated value described above. To minimize uncertainty from computational approximations, pKa were determined only for species with experimentally measured redox potentials. Molecular electrostatic potentials at the Nim atoms were computed with Multiwfn, given their established correlation with pKa values.
Multireference calculations
To account for the static correlation arising from the near degeneracy of the 5f orbitals, multireference Complete Active Space Self-consistent Field (CASSCF)120 calculations were performed on the DFT-optimized geometries of 1-An3+, 1-An4+, and 1-An5+ using ORCA 6.1.0.121,122 The DKH2 Hamiltonian was employed to account for scalar relativistic effects.123 The relativistically contracted SARC-DKH-TZVP basis set was employed for An,124 the DKH-def2-TZVP(-f) basis set for N atoms in the first coordination sphere, and the DKH-def2-SVP basis set for the rest of the atoms.125,126 The RIJCOSX approximation was used to reduce the computational cost. For U, the autoaux127 feature was used to generate the auxiliary basis set. Dynamic correlation was added through the N-Electron Valence State Second-Order Perturbation Theory (NEVPT2) method for the calculations of spin-state energetics.128 For 1-U4+ and 1-Np5+, the ground state is three and two-fold degenerate, respectively. Therefore, the CASSCF calculations were carried out with three triplets for 1-U4+ and two triplets for 1-Np5+. For other complexes and other spin states, only one root was considered. Restricted Active Space Self-Consistent Field (RASSCF)129 calculations were performed to quantify An orbital populations in the 3c–2e An–N–P π bonds within the 1-An4+ and 1-An5+ series (see the SI for details). These multireference results were used to benchmark and contextualize the pronounced An 5fπ contributions obtained at the DFT level for later An.Results and discussion
Molecular orbitals and density of states
Across all examined oxidation states, the An-dominant orbitals corresponding to the An unpaired 5f electrons in the α-spin manifold shift to progressively lower energies as the An series is traversed, which is captured in both DFT and CASSCF methods (Fig. 2 and Fig. S1–S8). In the 1-An3+ series, the 5f orbitals are well separated from the frontier L-based π bonding MOs, allowing for a straightforward qualitative assignment of the 5f3, 5f4, and 5f5 configurations in the U, Np, and Pu species, respectively. In 1-An4+, these orbitals gradually approach the L-dominant π bonding MOs, which remain relatively consistent regardless of the An identity (Fig. 2). Consequently, orbitals with considerable 5f contributions in 1-Pu4+ become ∼0.6–0.9 eV lower in energy than the L-dominant highest occupied molecular orbital, HOMO (Pu 6.3%; L 93.7%). Their average An contribution drops to 51.3% compared to 96.1% in 1-U4+ and 72.1% in 1-Np4+ (Table S6). The energy matching of M and L orbitals becomes even more pronounced in 1-An5+, as all orbitals shift to lower energies, most significantly in 1-Pu5+ (Fig. 2 and Fig. S3). While the more core-like nature of the 5f atomic orbitals (AOs) in later An is anticipated due to the incomplete screening of the additional nuclear charge,54,57,67,70 the extensive spread of Pu 5f AOs among L-dominant MOs in 1-Pu5+ makes assigning the 5f3 configuration more challenging based solely on canonical MOs. As was discussed before,20,21 in such cases, it is critical to delineate M contributions to the localized non-bonding elements from those describing M–L bonding by means of the electron localization methods, such as NBO/AdNDP (vide infra). However, it is important to note that M/L orbital energies and orbital mixing depend on the amount of Hartree–Fock exchange in the chosen DFT functional. Therefore, only relative orbital energy ordering and composition analyses are meaningful for comparing different An systems, which serves as the basis for the analysis in this study.A similar effect is observed upon a single L protonation of 1-An4+ to form 2-An4+, e.g., HOMOs of 1-U4+/1-Np4+ drop in energy by 0.75–0.81 eV. While the An 5f-dominant orbitals of 2-U4+/2-Np4+ can still be identified as the top frontier singly occupied MOs in the α-spin manifold, the Pu 5f atomic orbitals in 2-Pu4+ evince a considerable intermixing with the L-dominant orbitals. They are spread over ten MOs (HOMO–8 to HOMO–17) with the Pu contributions ranging from 16.6% to 65.8% (Fig. S5 and Table S9). As the overall charge increases due to the protonation of 1-An4+, the lowest unoccupied molecular orbitals (LUMOs) in 2-An4+ also stabilize and shift down in energy by 0.78–0.84 eV. It is noteworthy that the energy of the LUMO in 2-U4+ (–0.82 eV) is substantially lower than its non-protonated counterpart, 1-U4+ (0.02 eV). Our previous joint experimental and theoretical work23 revealed that the reduction of 1-U4+ to 1-U3+ could not be achieved either chemically or electrochemically, which was rationalized by the relatively high energy of the LUMO in 1-U4+. In contrast, 1-Np4+ could be chemically reduced to 1-Np3+, consistent with its lower-energy LUMO (–0.56 eV) calculated at the same level.
To more specifically pinpoint the energy matching between certain orbitals, the density of states130 (DOS) was calculated for all the studied complexes (see the SI for details). This analysis provides an alternative perspective to the MO diagrams for comparing the energies of the An 5f and ligand π bonding orbitals as a function of the An identity and oxidation state. The DOS plots have proven to be a valuable tool in the electronic structure analysis of a variety of other An-based molecular systems.59,70,75,131–133 The projected density of states (PDOS) plots, shown in Fig. S9–S15, evince a substantial energetic gap between the An 5f and Nim 2p states in the +3-oxidation state, irrespective of the An. This contrasts with the +4-oxidation state, where the Np and Pu complexes display an appreciable An 5f–Nim 2p energetic overlap, with the Pu 5f states being much closer to the Nim 2p states than Np 5f. Meanwhile, the U 5f states remain distinguished from Nim 2p, emphasizing the increased role of energy matching in the later An species. As the An oxidation state increases, the energy of the 5f bands decreases at a faster rate than the Nim 2p bands, leading to even greater energy matching as the An series is traversed. Thus, in 1-Np5+ and 1-Pu5+, the occupied An 5f states are completely energetically mixed with the Nim 2p manifold, whereas in 1-U5+ one 5f state is slightly offset in energy, corresponding to the singly occupied 5f orbital.
Natural atomic orbitals
In addition to the MO diagrams and the PDOS plots, analysis of the natural atomic orbitals (NAOs) can be employed to track how the energy difference between the An 5f and Nim 2p NAOs changes in both α- and β-spin manifolds (Fig. 3). First, the general trends show that the energy difference between the An 5f and Nim 2p NAOs decreases from U to Pu regardless of the An oxidation state, in line with the improved energy matching for later An. The average of the α- and β-NAO energies shows that the An 5f–Nim 2p NAO energy gap in the Np complexes decreases by 0.69–1.00 eV compared to the respective U species. This gap is further reduced by 0.64–0.90 eV in the Pu counterparts relative to their Np counterparts (Table S12). Second, as the oxidation state increases, the orbital energy difference becomes progressively smaller. For instance, the averaged An 5f–Nim 2p NAO gap decreases from 4.81 eV in 1-Pu3+ to 2.98 eV in 1-Pu4+ and further to 2.22 eV in 1-Pu5+. This result is in accordance with previously reported studies on the An2+Cl2 and An3+Cl3 complexes,60,61 which showed that the An3+Cl3 species generally had a greater orbital energy matching than An2+Cl2. A similar effect is observed upon ligand protonation, although less abrupt, e.g., the NAO energy gap decreases from 4.81 eV in 1-Pu3+ to 4.59 eV in 2-Pu3+ and from 2.98 eV in 1-Pu4+ to 2.88 eV in 2-Pu4+. Third, in the open shell species, the α-orbitals are found to be the driving force of energy matching as the Nim 2p NAOs energy gap with α-spin An NAOs is always smaller than it is with the respective β-spin An NAOs, owing to the presence of unpaired electron(s) on the An in the α-spin manifold. Across all species, 1-Pu5+ has the lowest energy α-An 5f NAOs, creating the only negative energy gap from the Nim 2p NAOs (–0.34 eV), with an α/β average energy gap (2.22 eV) even smaller than that of 1-U6+ (3.22 eV).AdNDP localized orbitals: population and composition
NAOs transformed to the natural bonding orbitals (NBOs) are further employed for the detailed orbital analysis of the An–Nim bonding interactions via the AdNDP method.110 This approach enables a detailed evaluation of the An–Nim bonding interactions, providing insight into the An contributions and how the orbital composition evolves with the An identity and oxidation state. In all open-shell species, AdNDP recovered non-bonding one-centre one-electron (1c–1e) element(s), corresponding to the An unpaired electron(s). In such cases, they are very localized, with occupation number (ON) values spanning a narrow range, 0.95–1.00 |e|, and with dominant 5f-orbital character (90.2–100.0%, Table S13). The corresponding 5fn configurations deciphered from the AdNDP analysis are instrumental for the validation of the An oxidation state, which is particularly useful for the higher-oxidation state An species exhibiting a significant spread of the An AOs in the canonical MOs. The assignments of the An oxidation state deduced from AdNDP also fall in line with the computed Npyramidalization (the sum of Npyrr valence angles vs. P–Npyrr bond length) for each studied complex (Fig. S16 and S17), supporting its use as a geometric indicator of M oxidation state in imidophosphorane complexes, as was suggested previously.20,22,23The covalency of the An–Nim interactions can be tracked through the An orbital contributions in the two-centre two-electron (2c–2e) An–Nim σ and three-centre two-electron (3c–2e) An–Nim–P π bonds shown in Fig. 4A and Fig. S18 and S19. A similar characteristic bonding pattern has previously been found in other An compounds featuring an imido-type ligand framework.134–136 The dominant 6d and 5f An orbital contributions are analysed for the σ- and π-bonding interactions in both α- and β-spin manifolds (Tables S14 and S15), as some differences are noted between them across the considered oxidation states. For clarity, the α-spin manifold will be discussed first. The 6d orbital contributions slightly diminish or remain relatively the same across the An of the same oxidation state. However, they steadily become larger as the An oxidation state increases, i.e., from ∼0.11–0.12 |e| in 1-An3+ to ∼0.19 |e| in 1-An5+. In contrast, the 5f contributions display a divergent trend in the lower-oxidation state (+3) vs. higher-oxidation states (+4, +5). Specifically, in the An3+ complexes, the contributions slightly decrease as the An series is traversed, whereas the An4+ and An5+ 5f contributions increase progressively from U to Pu. In fact, their magnitude becomes comparable or even surpasses the 6d contributions in 1-An5+, e.g., the 6d/5f values change from 0.19/0.18 |e| in 1-U5+ to 0.19/0.29 |e| in 1-Pu5+. Separate analyses of the σ (6dσ/5fσ) and π (6dπ/5fπ) bonding interactions in 1-An5+ further show that the growth in the overall An orbital population is driven predominantly by the increasing An 5fπ contributions in the α-spin manifold (Fig. 4B and Fig. S20).
The trend observed for the α-spin An orbital populations sharply contrasts with the β-spin counterparts, where both 6d and 5f contributions slightly decrease within the An series of the same oxidation state, e.g., An 6d/5f: 0.18/0.16 |e| 1-U5+vs. 0.15/0.13 |e| 1-Pu5+. The 6d/5f contributions still gradually increase from An3+ to An5+, as is the case for the α-spin manifold, although to a lesser extent. However, in contrast to the α-spin manifold, the β-spin 5f contributions do not exceed the 6d counterparts at higher-oxidation state An, and the An 5fπ contributions do not grow as the An series is traversed within a given oxidation state. It is also worthy to note that the α-spin An 5fσ+π orbital contributions start to outweigh the β-counterparts in the higher-oxidation state An4+ and An5+ species, especially for the later An. Also, the overall (α + β) 6d/5f An contributions echo the trends elucidated across all oxidation states for the α-spin manifold alone (Fig. S21 and Table S16). Hence, the overall (α + β) AdNDP data suggest that the increased An–Nim covalent interactions in the An4+ and An5+ species are driven by the greater α-spin An 5fπ orbital contributions, which increase with the growing prominence of orbital energy matching.
The greatest An 5f–Nim 2p energy matching found in 1-An5+ across all considered series correlates with the most covalent An–Nim interactions. As the orbital energy gap between M and L orbitals shrinks upon oxidation of 1-An4+ to form 1-An5+, the overall (α + β) An 5fσ+π orbital population increases by 0.18–0.23 |e| while the An 6dσ+π increases by only 0.05–0.08 |e|. The increase of the An 5fπ contributions in the 3c–2e An–Nim–P π bonds (0.13–0.21 |e|) is more pronounced compared to the 5fσ contributions in the 2c–2e An–Nim σ bonds (0.03–0.05 |e|). In contrast, while the 6d orbitals are more diffuse than 5f, the An 6dπ and 6dσ contributions increase by only 0.03–0.04 |e| (Fig. S21). The greater increase of the α-spin An 5fπ contributions can be demonstrated by plotting the An–Nim–P π bonds at the same isovalue across various systems. For instance, Fig. S22 shows that the An centres in 1-An4+ and 1-An5+ exhibit larger α-spin An 5fπ population when the An oxidation state increases from +4 to +5 as well as when the An series is traversed. This trend aligns with progressively improved An 5f–Nim 2p energy matching for the later An in these high-valent species. A similar picture was reported previously for the heavier An systems: AmCl3 and FmCl3 compounds, wherein the π α-spin orbital of the former and the π β-spin orbital of the latter also showed considerable signs of energy matching driven covalency.60
In accordance with the MO/NAO and PDOS energy plots, it is found that the protonated species 2-An4+ exhibit greater An–N orbital energy matching than the 1-An4+ species, but smaller than 1-An5+. From the geometric standpoint, with the elongation of one bond, the remaining three bonds slightly shrink in 2-An4+, becoming intermediate to their lengths in the 1-An4+ and 1-An5+ counterparts (e.g., 2.127 Å 2-U4+vs. 2.178 Å 1-U4+ and 2.094 Å 1-U5+). AdNDP analysis suggests that the covalency of the An–Nim interactions in 2-An4+ (across four An–Nim bonds) is closer to the 1-An4+ species than 1-An5+, e.g., An 6d + 5f: 1.85 |e| in 2-U4+vs. 1.83 |e| in 1-U4+ and 2.85 |e| in 1-U5+ (Tables S16 and S17). This is explained by the disruption of one of the π bonds due to the formation of the 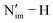 bond in 2-An4+ (Fig. S19). The An–N’im bond of the protonated ligand is much weaker, with a lower contribution from the An centre in the bonding interactions compared to the other An–Nim interactions, e.g., 10.8%/9.6% vs. 6.4%/3.4% in the σ/π components of the α-orbitals of 2-U4+ (Table S15). A similar effect was previously noted in the computational studies of the terminal UIV imido complex [U(TrenTIPS)(NH)], where analysis of the protonated precursor [U(TrenTIPS)(NH2)] revealed a marked reduction in the U contribution to the π components of the relevant NBOs, i.e., 17.2% vs. 10.4%.134
bond in 2-An4+ (Fig. S19). The An–N’im bond of the protonated ligand is much weaker, with a lower contribution from the An centre in the bonding interactions compared to the other An–Nim interactions, e.g., 10.8%/9.6% vs. 6.4%/3.4% in the σ/π components of the α-orbitals of 2-U4+ (Table S15). A similar effect was previously noted in the computational studies of the terminal UIV imido complex [U(TrenTIPS)(NH)], where analysis of the protonated precursor [U(TrenTIPS)(NH2)] revealed a marked reduction in the U contribution to the π components of the relevant NBOs, i.e., 17.2% vs. 10.4%.134
To corroborate the DFT-predicted increase in the An 5fπ orbital contributions from U to Pu in the An4+ and An5+ series, multireference calculations were carried out. CAS(n,7)SCF calculations on the 1-An3+, 1-An4+, and 1-An5+ complexes reveal pronounced multireference character (except for 1-U5+) as the leading electronic configuration contributes less than 85% to the total wavefunction (Fig. S23–S31 and Tables S18–S34). More extensive RASSCF calculations including the ligand-dominant An–Nim–P π bonding orbitals and their corresponding antibonding orbitals were performed for the 1-An4+ and 1-An5+ series, where the An 5fπ contributions are most pronounced (see the SI for details). Analysis of the RASSCF natural orbitals for these species shows ON values of 1.97–1.98 |e| for the bonding orbitals and 0.02–0.03 |e| for the corresponding antibonding orbitals (Fig. S32–S37 and Tables S35–S40). This suggests that the multireference character originates primarily from distributing the unpaired electrons among the largely nonbonding An 5f orbitals rather than from the Nim-based π system. As these nonbonding 5f orbitals drive the multireference behaviour, the RASSCF treatment is not expected to qualitatively alter the bonding trends obtained from the single-reference DFT-based AdNDP analysis. Consistent with the DFT results, RASSCF combined (6dπ + 5fπ) An contributions increase from 1-An4+ to 1-An5+ by ∼0.14 |e| on average, compared with 0.18–0.22 |e| at DFT. Although RASSCF calculations show slightly larger or comparable absolute 6dπ contributions relative to 5fπ in both series (Fig. S38), oxidation from 1-An4+ to 1-An5+ also leads to a larger increase in 5fπ (0.09–0.15 |e|) than in 6dπ (0.05–0.08 |e|).
QTAIM topological analysis of electron density
To complement the orbital-based bonding analysis, a topological analysis of the electron density was performed using QTAIM. Within this framework, orbital overlap can be evaluated through the electron density, ρ(r), at the An–Nim bond critical point (BCP). Additionally, the delocalization index, δ(An,Nim), serves as a quantitative measure of bond strength, reflecting the combined effects of orbital overlap and orbital energy matching between the interacting atoms (Fig. 5A and B and Table S41). Across all studied complexes, ρ(r) at the An–Nim BCP decreases from U to Pu in each oxidation state. In contrast, ρ(r) increases upon oxidation, from 1-An3+ (0.083–0.089 |e| Bohr−3) to 1-An4+ (0.112–0.115 |e| Bohr−3) and 1-An5+ (0.131–0.136 |e| Bohr−3). These trends indicate that the An–Nim orbital overlap is enhanced upon oxidation as the two atoms get closer, but diminishes along the An series from U to Pu. However, the δ(An,Nim) values reveal two distinct trends: one for the An3+ species, and another for An4+ and An5+. For 1-An3+ and 2-An3+, δ(An,Nim) decreases across the series in parallel with the ρ(r) trends (e.g., from 0.70 in 1-U3+ to 0.65 in 1-Pu3+). In contrast, for the higher-valent species the trend reverses, with δ(An,Nim) increasing from 0.90/1.14 in 1-U4+/1-U5+ to 0.92/1.19 in 1-Pu4+/1-Pu5+. While the absolute magnitude of these changes (i.e., 0.02 for 1-An4+ and 0.05 for 1-An5+) is small, it is the relative changes and systematic trends across the series that are chemically significant. Comparable relative variations have been demonstrated to be meaningful in prior QTAIM-based studies of actinide bonding.26,137–140 The divergent behaviour of ρ(r) and δ(An,Nim) suggests that the An 5f–Nim 2p energy matching becomes increasingly important for the higher-valent and later An complexes, compensating for the reduced orbital overlap along the series. These findings are consistent with high energy resolution fluorescence detected X-ray studies combined with multiplet theory of actinyl An–Oyl bonds,45 which revealed decreasing overlap-driven but increasing energy degeneracy-driven covalency from U to Pu without a concomitant build-up of bond-path electron density. A similar effect was reported for Ce(IV) halides, where, despite the smaller orbital overlap, covalent mixing between Ce 4f and X (X = F, Cl) 2p orbitals was found to be ∼25% greater in CeCl62− than in CeF62− due to the substantially improved orbital energy matching in the former.81 Overall, the δ(An,Nim) trends obtained from QTAIM across all considered species mirror the evolution of An–Nim covalency obtained from AdNDP, decreasing from U to Pu in 1-An3+ and 2-An3+, and slightly increasing in 1-An4+, 2-An4+, and 1-An5+.Spin decomposition of the delocalization indices into α- and β-components shows that δβ(An,Nim) decreases from U to Pu in each oxidation state for all species (Fig. S39). In contrast, δα(An,Nim) mirrors the behaviour of the total δ(An,Nim), decreasing across the An3+ series but progressively increasing in An4+ and An5+. Notably, the increase in δα(An,Nim) for the tetra- and pentavalent species parallels the increasingly larger α-spin An 5fπ contributions along the An series, identified by AdNDP as the dominant driver of the enhanced An orbital population. Thus, the QTAIM data support the AdNDP results, indicating that the reversal in An–Nim covalency trends in An4+ and An5+ compared to An3+ is driven by the α-spin manifold. Taken together, the AdNDP and QTAIM analyses suggest that, in the 1-An3+ and 2-An3+ complexes, An–Nim covalency is governed predominantly by orbital overlap with U > Np > Pu. In contrast, in 1-An4+, 2-An4+, and 1-An5+, although orbital overlap remains the primary factor, improved α-spin An 5f–Nim 2p energy matching leads to larger An 5fπ contributions for later An, thereby inverting the covalency trend to U < Np < Pu. Although this analysis cannot quantitatively disentangle the relative contributions of the enhanced An 5fπ population to orbital overlap vs. orbital energy matching, it nevertheless reveals a shift in their balance that becomes particularly important for the higher-valent species and later An.
IQA energy decomposition
The delocalization index δ(An,Nim) represents the number of electron pairs shared between the An and Nim basins and is often regarded as a real-space analogue of bond order, but it does not itself provide an energetic description of the An–Nim interaction. However, δ is approximately proportional to the exchange–correlation energy, Vxc, and in a zeroth-order approximation the two quantities are related by Vxc ≈ –δ/2R, where R is the internuclear distance.141–143 Consistent with this relationship, the overall Vxc(An–Nim) trends (Fig. 5C) qualitatively track those of δ(An,Nim). In 1-An3+, Vxc becomes less stabilizing from U (–91.5 kcal mol−1) to Pu (–82.1 kcal mol−1). In 1-An4+ it is nearly invariant (–117.1 to –117.4 kcal mol−1), whereas in 2-An4+ it becomes progressively more stabilizing from U (–135.3 kcal mol−1) to Pu (–138.8 kcal mol−1). In 1-An5+, Vxc is most stabilizing at Np (–152.7 kcal mol−1) and slightly less so at U (–150.2 kcal mol−1) and Pu (–150.7 kcal mol−1). The relative gain in exchange–correlation stabilization upon oxidation tends to be larger for higher-valent or protonated species. For example, ΔVxc(An–Nim) is 32.3 kcal mol−1 for 1-Np3+ → 1-Np4+ but increases to 35.6 and 38.4 kcal mol−1 for 1-Np4+ → 1-Np5+ and 2-Np3+ → 2-Np4+, respectively. More broadly, this gain in ΔVxc(An–Nim) upon oxidation generally increases along the An series: for 1-An3+ → 1-An4+, ΔVxc(An–Nim) grows from 25.9 kcal mol−1 (U) to 32.3 kcal mol−1 (Np) and 35.0 kcal mol−1 (Pu).Within the An3+ series, the 9.4–9.7 kcal mol−1 destabilizing change in Vxc(An–Nim) from U to Pu is mainly attributed to decreasing orbital overlap, as An 5f–Nim 2p energy matching is not yet developed. Consistent with this, the An 5fσ, 6dσ, 6dπ, and 5fπ populations either slightly decrease or remain constant across the An series in both 1-An3+ and 2-An3+. In contrast, variations in Vxc(An–Nim) driven by changes in orbital overlap upon oxidation are much larger. This can be assessed cleanly only for the U series, because Np- and Pu-based complexes already display additional stabilization from orbital energy matching in the An4+ and An5+ species, which obscures a clear separation of the two contributions. For example, the stabilizing gain in Vxc(An–Nim) is 25.9 kcal mol−1 for 1-U3+ → 1-U4+, 30.2 kcal mol−1 for 2-U3+ → 2-U4+, and 32.8 kcal mol−1 for 1-U4+ → 1-U5+.
The 2-An4+ series provides a particularly clear case for estimating the effect of orbital energy matching on Vxc(An–Nim). 2-U4+ and 2-Np4+ have identical ρ(r) values at the An–Nim BCP (0.128 |e| Bohr−3), indicating similar overlap, yet the slightly larger δ(An,Nim) in 2-Np4+ (1.08 vs. 1.05) yields 2.0 kcal mol−1 of additional exchange–correlation stabilization relative to 2-U4+. This extra stabilization is attributed to improved α-spin Np 5f–Nim 2p energy matching in 2-Np4+. Thus, the overlap-driven gain in Vxc(An–Nim) upon the 2-U3+ → 2-U4+ oxidation is much larger than the stabilization arising from orbital energy matching in going from 2-U4+ to 2-Np4+. It aligns with prior works showing that the orbital energy matching provides only modest stabilization relative to overlap-driven covalency and does not produce a substantial build-up of electron density in the internuclear region.45,53,55,73 In 2-Pu4+, where the orbital energy matching is even more favourable, Vxc is further stabilized by 1.5 and 3.5 kcal mol−1 relative to 2-Np4+ and 2-U4+, respectively, despite a slightly lower ρ(r) at the Pu–Nim BCP (0.127 |e| Bohr−3). Across this series, 5fσ, 6dσ, and 6dπ populations remain essentially constant or slightly decrease from U to Pu, whereas only the An 5fπ contributions increase, indicating that this “excess” in 5fπ in the later An is primarily responsible for the additional stabilization. This observation is consistent with the study of hydroxypyridinone complexes of Am–Cf, where the experimentally determined greater stability of An4+ over An3+ was reproduced theoretically and attributed to enhanced orbital mixing arising from improved An 5f–ligand π orbital energy matching.36
A similar picture emerges for the 1-An5+ series. From 1-U5+ to 1-Np5+, ρ(r) at the An–Nim BCP decreases slightly (0.136 → 0.135 |e| Bohr−3), yet Vxc is stabilized by 2.5 kcal mol−1, again consistent with enhanced Np 5f–Nim 2p energy matching relative to U. In 1-Pu5+, Vxc becomes 2.0 kcal mol−1 less stabilizing than in 1-Np5+, even though the 5fπ contribution continues to increase. This reversal reflects a more pronounced decrease in ρ(r) at the An–Nim BCP (0.004 |e| Bohr−3 from 1-Np5+ to 1-Pu5+vs. 0.001 |e| Bohr−3 from 1-U5+ to 1-Np5+), consistent with a slight An–Nim bond elongation from Np to Pu (by 0.007 Å). In going from 1-Np5+ to 1-Pu5+, this change is accompanied by reduced populations in 5fσ (–0.04 |e|), 6dσ (–0.02 |e|), and 6dπ (–0.01 |e|), and a compensating increase in 5fπ (+0.06 |e|). Taken together, these observations suggest that, although the effects of orbital overlap and energy matching cannot be rigorously disentangled in this case, the loss of overlap in 1-Pu5+ exerts a slightly stronger destabilizing effect on Vxc than the stabilizing contribution from improved energy matching relative to 1-Np5+. Conversely, comparison of 1-U5+ and 1-Pu5+ shows that the appreciably better orbital energy matching in 1-Pu5+ slightly outweighs the reduction in orbital overlap, thereby rendering Vxc marginally more negative than in 1-U5+.
In contrast to the increasing An–Nim covalency in the higher-valent species inferred from AdNDP/QTAIM and its impact on the exchange–correlation energy, the classical electrostatic term Vcl of the An–Nim interaction becomes progressively less stabilizing from U to Pu in each oxidation state (Fig. 5D). On an absolute scale, Vcl is substantially more negative than Vxc for a given oxidation state, indicating that the overall An–Nim interaction is dominated by electrostatics. For example, in 1-An5+, Vcl ranges from –714.3 to –607.7 kcal mol−1, compared with only –152.7 to –150.6 kcal mol−1 for Vxc. Consequently, the net An–Nim interaction energy becomes less stabilizing along the An series in all oxidation states, despite the nuanced evolution of covalency.
pKa and BDFE
As demonstrated above, the electronic charge accumulation in the bonding region, described by the electron density ρ(r) at the BCP, must be considered alongside the energy separation between the M and L orbitals, which can modulate the extent of M–L orbital mixing and impact the trends in covalency. The covalent mixing parameter, given as λ = HML/ΔEML (where HML is the Hamiltonian matrix element describing the interaction between the M and L orbitals, which in the extended Hückel theory, is approximated as proportional to the orbital overlap integral, and ΔEML is the energy gap between these orbitals), may increase as ΔEML decreases with improved orbital energy matching.6,53,54,58,74Based on the simultaneous decrease in ρ(r) and increase in δ(An,Nim) along the An series for 1-An4+, 2-An4+, and 1-An5+, the enhanced M–L covalency associated with increasingly larger An 5fπ contributions due to the better An 5f–Nim 2p energy matching appears to redistribute electron density toward the atomic centres rather than building up between the nuclei.45,53,73 This behaviour parallels that reported for An(η-C5H5)3 and An(η-C5H5)4, where stronger 5f mixing across the series was not accompanied by increased charge at the bond midpoint.67,68 Consistently, QTAIM reveals progressively increasing spin density at the M centre as the An series is traversed in 1-An4+, 2-An4+, and 1-An5+, counterbalanced by the spin density delocalized over the four Nim atoms (Tables S42 and S43). Within the 1-An5+ series, 1-Np5+ and 1-Pu5+ stand out, as their Nim spin densities (–0.27 and –0.65) are substantially larger in magnitude than that of 1-U5+ (–0.09) (Fig. S40). Similar conclusions are drawn from the NPA data for all the An4+ and An5+ complexes, while the An3+ counterparts show only minor changes in spin distribution (Tables S44 and S45).
The increase in the Nim spin density along the 1-An4+ and 1-An5+ series is also reflected in the increase in the Nimρ(r) at the nuclear critical points (NCPs) (Table S46). In 1-An4+, the average ρ(r) values at the Nim NCPs increase from 194.522 |e| Bohr−3 in 1-U4+ to 194.531 |e| Bohr−3 in 1-Np4+, and further to 194.551 |e| Bohr−3 in 1-Pu4+ (Fig. 6A). The calculated pKa values for 2-An4+ describing a heterolytic cleavage of the 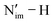 bond of the protonated ligand grow in tandem with the increasing electron density at the Nim NCPs in 1-An4+, from 23.7 for 1-U4+/2-U4+ to 27.4 for 1-Pu4+/2-Pu4+ (Table S49), evincing a linear correlation with R2 = 0.9986 (Fig. 6B). However, this correlation does not hold across multiple oxidation states. For a broader analysis of the trends in pKa across multiple oxidation states, molecular electrostatic potentials (MEPs) and the sum of 2p NAOs on the Nim atoms (Tables S47 and S48) were used, which were previously employed as descriptors for molecular acidity and basicity.144–147 In general, more negative MEP values and more positive energy valence NAOs are associated with a higher proton affinity.148 It is important to note that pKa was only calculated for the species with experimentally determined E1/2 values; therefore, the data in Fig. 6C and D only depict the 1-An4+ species, 1-Pu3+, and 1-U5+. The MEP on the Nim atoms shows an inverse linear correlation (R2 = 0.9723) with the calculated pKa values, becoming more negative in going from 1-U5+ (–18.3969 Eh/|e|) to 1-Pu3+ (–18.6125 Eh/|e|) (Fig. 6C). The sum of the energy of the Nim 2p NAOs computed for these complexes also linearly correlate (R2 = 0.9792) with the calculated pKa (Fig. 6D). Overall, the correlation of ρ(r) at the Nim NCPs, MEP on the Nim atoms, and the Nim 2p NAO values with pKa suggest their use for the assessment of acidity/basicity.
bond of the protonated ligand grow in tandem with the increasing electron density at the Nim NCPs in 1-An4+, from 23.7 for 1-U4+/2-U4+ to 27.4 for 1-Pu4+/2-Pu4+ (Table S49), evincing a linear correlation with R2 = 0.9986 (Fig. 6B). However, this correlation does not hold across multiple oxidation states. For a broader analysis of the trends in pKa across multiple oxidation states, molecular electrostatic potentials (MEPs) and the sum of 2p NAOs on the Nim atoms (Tables S47 and S48) were used, which were previously employed as descriptors for molecular acidity and basicity.144–147 In general, more negative MEP values and more positive energy valence NAOs are associated with a higher proton affinity.148 It is important to note that pKa was only calculated for the species with experimentally determined E1/2 values; therefore, the data in Fig. 6C and D only depict the 1-An4+ species, 1-Pu3+, and 1-U5+. The MEP on the Nim atoms shows an inverse linear correlation (R2 = 0.9723) with the calculated pKa values, becoming more negative in going from 1-U5+ (–18.3969 Eh/|e|) to 1-Pu3+ (–18.6125 Eh/|e|) (Fig. 6C). The sum of the energy of the Nim 2p NAOs computed for these complexes also linearly correlate (R2 = 0.9792) with the calculated pKa (Fig. 6D). Overall, the correlation of ρ(r) at the Nim NCPs, MEP on the Nim atoms, and the Nim 2p NAO values with pKa suggest their use for the assessment of acidity/basicity.
The computed SOC-corrected 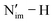 BDFE describing the homolytic cleavage of the
BDFE describing the homolytic cleavage of the 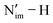 bond in 2-An4+ increases across the An series, e.g., from 62.3 kcal mol−1 in 2-U4+ to 96.1 kcal mol−1 in 2-Pu4+ (Table S49). In accordance, the smallest pKa calculated from the Bordwell equation for 2-U4+ indicates that it has the most acidic proton in the
bond in 2-An4+ increases across the An series, e.g., from 62.3 kcal mol−1 in 2-U4+ to 96.1 kcal mol−1 in 2-Pu4+ (Table S49). In accordance, the smallest pKa calculated from the Bordwell equation for 2-U4+ indicates that it has the most acidic proton in the 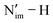 bond. This enhanced acidity correlates with the smallest accumulation of the electron density on Nim in 1-U4+ (194.522 |e| Bohr−3) compared to 1-Np4+ (194.531 |e| Bohr−3) and 1-Pu4+ (194.551 |e| Bohr−3). Likewise, in 1-An5+, ρ(r) at the Nim NCPs also increases across An, but more steeply: from 1-U5+ (194.547 |e| Bohr−3) to 1-Np5+ (194.574 |e| Bohr−3) and 1-Pu5+ (194.623 |e| Bohr−3). Notably, the value for 1-U5+ is even smaller than that in the lower-valent 1-Pu4+ (194.551 |e| Bohr−3), which may be related to the divergent PCET reactivity in these complexes: only 1-Np5+ and 1-Pu5+ were experimentally shown to undergo PCET,20,21 whereas 1-U5+ remained inactive under the same conditions. However, since this PCET reaction is a M–L-based process involving two centres (reduction of An and protonation of Nim) and is highly dependent on the ligand architecture, this correlation requires additional data points to be verified. It would be necessary to consider the net thermodynamics of the reaction including the fate of the THF radical to completely understand the reaction pathway. Combined experimental and computational studies focusing on the covalency and reactivity relations for other high-valent An systems employing modified imidophosphorane ligands are underway.
bond. This enhanced acidity correlates with the smallest accumulation of the electron density on Nim in 1-U4+ (194.522 |e| Bohr−3) compared to 1-Np4+ (194.531 |e| Bohr−3) and 1-Pu4+ (194.551 |e| Bohr−3). Likewise, in 1-An5+, ρ(r) at the Nim NCPs also increases across An, but more steeply: from 1-U5+ (194.547 |e| Bohr−3) to 1-Np5+ (194.574 |e| Bohr−3) and 1-Pu5+ (194.623 |e| Bohr−3). Notably, the value for 1-U5+ is even smaller than that in the lower-valent 1-Pu4+ (194.551 |e| Bohr−3), which may be related to the divergent PCET reactivity in these complexes: only 1-Np5+ and 1-Pu5+ were experimentally shown to undergo PCET,20,21 whereas 1-U5+ remained inactive under the same conditions. However, since this PCET reaction is a M–L-based process involving two centres (reduction of An and protonation of Nim) and is highly dependent on the ligand architecture, this correlation requires additional data points to be verified. It would be necessary to consider the net thermodynamics of the reaction including the fate of the THF radical to completely understand the reaction pathway. Combined experimental and computational studies focusing on the covalency and reactivity relations for other high-valent An systems employing modified imidophosphorane ligands are underway.
Redox properties
While trends in the Nim spin and electron densities can be instrumental for predicting thermodynamics of the hetero- and homolytic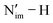 bond cleavage in these imidophosphorane ligands bound to An4+, it is the M centre that loses/gains electrons during the oxidation/reduction in these PCET reactions.10–12,20,21,23 However, as the M and L orbitals become closer in energy, the likelihood for a ligand-based oxidation increases. As was shown previously for the similar [Ln3+(NP*)4]− (Ln = Pr, Nd, Dy) complexes,12 oxidation of the Ln centre becomes unattainable for the Nd and Dy counterparts since their M-dominant MOs reside much lower in energy than the L-dominant orbitals compared to Pr, which can be oxidized to the 4+ state. In the present work, we also find that as the Nim 2p and An 5f orbitals get closer in energy in the studied An4+ and An5+ complexes, the relative thermodynamic energy difference between the structures resulting from An- and Nim-based oxidations becomes progressively smaller, e.g., from 75.4 kcal mol−1 for 1-U4+ to 43.7 kcal mol−1 in 1-U5+, and from 43.2 kcal mol−1 for 1-Pu4+ to 5.7 kcal mol−1 in 1-Pu5+ (Fig. S41). However, in all the considered cases, the calculated redox potentials for the thermodynamically favourable M-based oxidations align much more closely with experimental values and are indicative of An-based oxidations (Table S50).
bond cleavage in these imidophosphorane ligands bound to An4+, it is the M centre that loses/gains electrons during the oxidation/reduction in these PCET reactions.10–12,20,21,23 However, as the M and L orbitals become closer in energy, the likelihood for a ligand-based oxidation increases. As was shown previously for the similar [Ln3+(NP*)4]− (Ln = Pr, Nd, Dy) complexes,12 oxidation of the Ln centre becomes unattainable for the Nd and Dy counterparts since their M-dominant MOs reside much lower in energy than the L-dominant orbitals compared to Pr, which can be oxidized to the 4+ state. In the present work, we also find that as the Nim 2p and An 5f orbitals get closer in energy in the studied An4+ and An5+ complexes, the relative thermodynamic energy difference between the structures resulting from An- and Nim-based oxidations becomes progressively smaller, e.g., from 75.4 kcal mol−1 for 1-U4+ to 43.7 kcal mol−1 in 1-U5+, and from 43.2 kcal mol−1 for 1-Pu4+ to 5.7 kcal mol−1 in 1-Pu5+ (Fig. S41). However, in all the considered cases, the calculated redox potentials for the thermodynamically favourable M-based oxidations align much more closely with experimental values and are indicative of An-based oxidations (Table S50).
As the An 5f α-orbitals are stabilized across the An series, the calculated redox potentials exhibit a cathodic shift, e.g., from –1.72 V in 1-U5+/4+ to –0.16 V in 1-Pu5+/4+. Protonation induces a similar effect by substantially stabilizing their orbitals, leading to cathodic shifts of 0.84–1.00 V relative to the corresponding non-protonated species, e.g., the potential of the U5+/4+ couple shifts from –1.72 V in 1-U5+/4+ to –0.72 V in 2-U5+/4+. This result opens an avenue for achieving lower M oxidation states via chemical reduction of the protonated species. The substantially stabilized LUMO in 2-U4+ (–0.82 eV) compared to the one in the non-protonated counterpart, 1-U4+ (0.02 eV), suggests a possible route to access the 2-U3+ complex. The calculated 2-U4+/3+ potential of –2.96 V is at the border of the THF electrochemical window but is more positive than for the 1-Np4+/3+ couple (–3.23 V), which was only able to be observed chemically and not electrochemically. Encouragingly, the calculated 2-Np4+/3+ potential is even more positive, at –2.32 V, suggesting that this couple may be experimentally accessible, as was observed visually but not chemically isolable (vide supra). Conversely, this cathodic shift afforded by the ligand protonation hinders the ability for M oxidation, which precludes the observation of the 2-Pu5+/4+ couple experimentally.
The experimentally available redox potentials subsequently validate what is predicted by the DFT calculations for several complexes (Fig. 7 and Fig. S72). Cyclic voltammetry (CV) studies of 2-U4+ and 2-Np4+ were performed in 0.05 M [N(nBu)4][BPh4] in THF and referenced to Fc+/0 (see the SI for full details). The CV of 2-Np4+ shows a quasi-reversible wave with E1/2 = −2.60 V, assigned to the 2-Np4+/3+ couple (–2.32 V theor.), an irreversible wave with E1/2 = −0.16 V, assigned to 2-Np5+/4+ couple (0.10 V theor.), and an additional small third feature present with E1/2 = −0.70 V, identical to the 1-Np5+/4+ couple (−0.75 V theor.). A CV of 2-U4+ also boasts three features, two mirroring the waves of the 5+/4+ and 6+/5+ couples seen in 1-U4+, and a third new feature with E1/2 = −0.92 V assigned to 2-U5+/4+ (−0.72 V theor.) via an electrochemical titration. During this titration, both features assigned to 1-U4+ gradually disappear with higher acid concentrations which is followed by a notable increase in reversibility of the 2-U5+/4+ couple, confirming these assignments. Details on the electrochemical titration of 2-U4+ with sequential equivalents of [H(OEt2)2][B(ArF5)4] are shown in Fig. S73.
Conclusions
The highly versatile NPC ligand provides a unique platform to interrogate M–L bond covalency over multiple oxidation states for the mid-actinides (U–Pu). Our results demonstrate that the relative energies of the An 5f and Nim 2p orbitals control not only the magnitude, but also the mechanism of An–Nim covalency and thereby impact molecular properties such as pKa,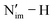 BDFEs, and redox potentials. While An–Nim covalency decreases from U to Pu in the +3 oxidation state, it increases across the An series in the +4 and +5 oxidation states, where improved orbital energy matching plays an increasingly important role. In the higher-valent 1-An4+, 2-An4+, and 1-An5+ complexes, MO/PDOS and NAO analyses, together with AdNDP/QTAIM data, reveal that enhanced α-spin An 5f–Nim 2p energy matching drives the growth in An–Nim covalency for later An. This is manifested by increasing An 5fπ contributions and larger spin densities at both the An and Nim centres due to the redistribution of electron density toward the atomic basins rather than the build-up of density in the internuclear region. These changes in the nature of covalency correlate with the divergent PCET behaviour observed for the An5+ complexes, where enhanced An 5f–Nim 2p mixing and growing density at Nim modulate the thermodynamic driving force for the PCET reactivity. IQA energy decomposition analysis further sheds light on the shift in balance between overlap-driven and energy-matching-driven covalency by evaluating exchange–correlation energy Vxc of the An–Nim bond in the An4+ and An5+ species. While changes in orbital overlap primarily control covalency upon oxidation from An3+ to An4+ and from An4+ to An5+, progressively improved An 5f–Nim 2p energy matching within the An series imparts an additional, subtler stabilization of the An–Nim interaction in the later, higher-valent An.
BDFEs, and redox potentials. While An–Nim covalency decreases from U to Pu in the +3 oxidation state, it increases across the An series in the +4 and +5 oxidation states, where improved orbital energy matching plays an increasingly important role. In the higher-valent 1-An4+, 2-An4+, and 1-An5+ complexes, MO/PDOS and NAO analyses, together with AdNDP/QTAIM data, reveal that enhanced α-spin An 5f–Nim 2p energy matching drives the growth in An–Nim covalency for later An. This is manifested by increasing An 5fπ contributions and larger spin densities at both the An and Nim centres due to the redistribution of electron density toward the atomic basins rather than the build-up of density in the internuclear region. These changes in the nature of covalency correlate with the divergent PCET behaviour observed for the An5+ complexes, where enhanced An 5f–Nim 2p mixing and growing density at Nim modulate the thermodynamic driving force for the PCET reactivity. IQA energy decomposition analysis further sheds light on the shift in balance between overlap-driven and energy-matching-driven covalency by evaluating exchange–correlation energy Vxc of the An–Nim bond in the An4+ and An5+ species. While changes in orbital overlap primarily control covalency upon oxidation from An3+ to An4+ and from An4+ to An5+, progressively improved An 5f–Nim 2p energy matching within the An series imparts an additional, subtler stabilization of the An–Nim interaction in the later, higher-valent An.
Protonation of the 1-An4+ and 1-An5+ complexes substantially stabilizes the An 5f α-orbitals in 2-An4+ and 2-An5+, leading to pronounced cathodic shifts in the An4+/3+ and An5+/4+ redox potentials and thereby altering the accessibility of these couples. The calculated pKa values of the 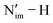 bond in 2-An4+ strongly correlate with the electron density at Nim in the corresponding 1-An4+ complexes, suggesting that this QTAIM descriptor may serve as a predictive proxy for protonation in other tetravalent systems. More broadly, both the MEP at Nim and the summed energies of Nim 2p NAOs correlate with pKa across multiple oxidation states.
bond in 2-An4+ strongly correlate with the electron density at Nim in the corresponding 1-An4+ complexes, suggesting that this QTAIM descriptor may serve as a predictive proxy for protonation in other tetravalent systems. More broadly, both the MEP at Nim and the summed energies of Nim 2p NAOs correlate with pKa across multiple oxidation states.
Overall, tuning the energies of An 5f orbitals through ligand design is shown to be vital for controlling An covalency, PCET thermodynamics, and redox accessibility. The strongly electron-donating NPC ligand can support An in the pentavalent state up to Pu. Modifying the NPC framework to systematically adjust electronic and steric properties, while correlating these changes with the An–L orbital overlap and energy matching, offers a promising route to rationally control An oxidation states and reactivity. Future combined theoretical and experimental studies employing tailored NPC-type ligands capable of stabilizing even higher An oxidation states within this framework are underway.
Author contributions
The study was conceived by I. A. P. and H. S. L. The theoretical calculations and analysis of the results were performed by C. M. S., S. D., J. W. S., and I. A. P. The experimental portion of the work was performed by K. S. E., J. E. N., and H. S. L. C. M. S. wrote the first draft and all the authors contributed to the writing, editing, and proofreading of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: computed structures, extended MO diagrams, orbital composition data, extended PDOS plots, AdNDP/QTAIM results, detailed redox potentials, complete experimental and methodological details, spectroscopic data (NMR, UV/vis, IR), selected crystallographic tables, and electrochemical characterization. See DOI: https://doi.org/10.1039/d6cp00281a.Figshare repository contains the XYZ coordinates of the optimized complexes at https://doi.org/10.6084/m9.figshare.29042807.
CCDC 2445923 contains the supplementary crystallographic data for this paper.149
Acknowledgements
I. A. P., C. M. S., and S. D. acknowledge the NNSA grant DE-NA0004151 as part of the TRUCoRE consortium funded by the NNSA Stewardship Science Academic Alliance program. Also, I. A. P. acknowledges startup funds and the Meyer Early Career Launch Fellowship at Washington State University (WSU). Computational resources at the ARCC HPC cluster at the University of Akron and at the Kamiak HPC cluster at the Center for Institutional Research Computing at WSU are gratefully acknowledged. This material is based upon work supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Heavy Element Chemistry program under Award Number DE-SC0019385 (K. S. E., J. E. N., H. S. L.).Notes and references
- M. Salvatores, Nucl. Eng. Des., 2005, 235, 805–816 CrossRef CAS.
- Implications of Partitioning and Transmutation in Radioactive Waste Management, International Atomic Energy Agency, Vienna, 2004 Search PubMed.
- F. W. Lewis, M. J. Hudson and L. M. Harwood, Synlett, 2011, 2609–2632 Search PubMed.
- M. J. Hudson, L. M. Harwood, D. M. Laventine and F. W. Lewis, Inorg. Chem., 2013, 52, 3414–3428 CrossRef CAS.
- S. F. Wolf, in The Chemistry of the Actinide and Transactinide Elements, ed L. R. Morss, N. M. Edelstein and J. Fuger, Springer Netherlands, Dordrecht, 2006, pp. 3273–3338 Search PubMed.
- M. L. Neidig, D. L. Clark and R. L. Martin, Coord. Chem. Rev., 2013, 257, 394–406 CrossRef CAS.
- K. L. Nash, Solvent Extr. Ion Exch., 1993, 11, 729–768 CrossRef CAS.
- Z. Wang, X. Dong, Q. Yan, J. Chen and C. Xu, Anal. Chem., 2022, 94, 7743–7746 CrossRef CAS.
- J.-C. G. Bünzli, Acc. Chem. Res., 2006, 39, 53–61 CrossRef.
- N. T. Rice, I. A. Popov, D. R. Russo, J. Bacsa, E. R. Batista, P. Yang, J. Telser and H. S. La Pierre, J. Am. Chem. Soc., 2019, 141, 13222–13233 CrossRef CAS PubMed.
- N. T. Rice, I. A. Popov, D. R. Russo, T. P. Gompa, A. Ramanathan, J. Bacsa, E. R. Batista, P. Yang and H. S. La Pierre, Chem. Sci., 2020, 11, 6149–6159 RSC.
- N. T. Rice, I. A. Popov, R. K. Carlson, S. M. Greer, A. C. Boggiano, B. W. Stein, J. Bacsa, E. R. Batista, P. Yang and H. S. La Pierre, Dalton Trans., 2022, 51, 6696–6706 RSC.
- A. R. Willauer, C. T. Palumbo, F. Fadaei-Tirani, I. Zivkovic, I. Douair, L. Maron and M. Mazzanti, J. Am. Chem. Soc., 2020, 142, 5538–5542 Search PubMed.
- A. R. Willauer, I. Douair, A.-S. Chauvin, F. Fadaei-Tirani, J.-C. G. Bünzli, L. Maron and M. Mazzanti, Chem. Sci., 2022, 13, 681–691 RSC.
- M. Tricoire, F.-C. Hsueh, M. Keener, T. Rajeshkumar, R. Scopelliti, I. Zivkovic, L. Maron and M. Mazzanti, Chem. Sci., 2024, 15, 6874–6883 RSC.
- P. S. Bagus, C. J. Nelin, K. M. Rosso, B. Schacherl and T. Vitova, Inorg. Chem., 2024, 63, 1793–1802 CrossRef CAS.
- P. S. Bagus, C. J. Nelin, B. Schacherl and T. Vitova, Inorg. Chem., 2024, 63, 13202–13213 CrossRef CAS.
- D. V. Kravchuk, L. J. Augustine, H. Rajapaksha, G. C. Benthin, E. R. Batista, P. Yang and T. Z. Forbes, Chem. – Eur. J., 2024, 30, e202304049 CrossRef CAS PubMed.
- J. E. C. Wren and G. Schreckenbach, Can. J. Chem., 2009, 87, 1436–1443 CrossRef CAS.
- J. E. Niklas, K. S. Otte, C. M. Studvick, S. Roy Chowdhury, B. Vlaisavljevich, J. Bacsa, F. Kleemiss, I. A. Popov and H. S. La Pierre, Nat. Chem., 2024, 16, 1490–1495 CrossRef CAS.
- K. S. Otte, J. E. Niklas, C. M. Studvick, C. L. Montgomery, A. R. C. Bredar, I. A. Popov and H. S. La Pierre, J. Am. Chem. Soc., 2024, 146, 21859–21867 CrossRef PubMed.
- J. E. Niklas, C. M. Studvick, J. Bacsa, I. A. Popov and H. S. La Pierre, Inorg. Chem., 2023, 62, 2304–2316 CrossRef PubMed.
- K. S. Otte, J. E. Niklas, C. M. Studvick, A. C. Boggiano, J. Bacsa, I. A. Popov and H. S. La Pierre, Angew. Chem., Int. Ed., 2023, 62, e202306580 CrossRef CAS.
- R. M. Diamond, K. Street Jr. and G. T. Seaborg, J. Am. Chem. Soc., 1954, 76, 1461–1469 CrossRef CAS.
- R. Beekmeyer and A. Kerridge, Inorganics, 2015, 3, 482–499 CrossRef CAS.
- Q.-R. Huang, J. R. Kingham and N. Kaltsoyannis, Dalton Trans., 2015, 44, 2554–2566 Search PubMed.
- D. E. Smiles, E. R. Batista, C. H. Booth, D. L. Clark, J. M. Keith, S. A. Kozimor, R. L. Martin, S. G. Minasian, D. K. Shuh, S. C. E. Stieber and T. Tyliszczak, Chem. Sci., 2020, 11, 2796–2809 Search PubMed.
- J. Murillo, J. A. Seed, A. J. Wooles, M. S. Oakley, C. A. P. Goodwin, M. Gregson, D. Dan, N. F. Chilton, A. J. Gaunt, S. A. Kozimor, S. T. Liddle and B. L. Scott, J. Am. Chem. Soc., 2024, 146, 4098–4111 Search PubMed.
- C. A. P. Goodwin, A. W. Schlimgen, T. E. Albrecht-Schönzart, E. R. Batista, A. J. Gaunt, M. T. Janicke, S. A. Kozimor, B. L. Scott, L. M. Stevens, F. D. White and P. Yang, Angew. Chem., Int. Ed., 2021, 60, 9459–9466 CrossRef CAS.
- T. G. Burrow, N. M. Alcock, M. S. Huzan, M. A. Dunstan, J. A. Seed, B. Detlefs, P. Glatzel, M. O. J. Y. Hunault, J. Bendix, K. S. Pedersen and M. L. Baker, J. Am. Chem. Soc., 2024, 146, 22570–22582 Search PubMed.
- A. Formanuik, A.-M. Ariciu, F. Ortu, R. Beekmeyer, A. Kerridge, F. Tuna, E. J. L. Mcinnes and D. P. Mills, Nat. Chem., 2017, 9, 578–583 Search PubMed.
- B. Hong, A. Näder, T. Sawallisch, T. Bode, S. Fichter, R. Gericke, P. Kaden, M. Patzschke, T. Stumpf, M. Schmidt and J. März, Inorg. Chem., 2024, 63, 17488–17501 CrossRef CAS.
- M. A. Islam, C. Berthon, J. Jung and H. Bolvin, Inorg. Chem., 2023, 62, 17254–17264 CrossRef CAS.
- N. Kaltsoyannis, Inorg. Chem., 2013, 52, 3407–3413 CrossRef CAS.
- J. Jung, M. Atanasov and F. Neese, Inorg. Chem., 2017, 56, 8802–8816 CrossRef CAS PubMed.
- M. P. Kelley, G. J. P. Deblonde, J. Su, C. H. Booth, R. J. Abergel, E. R. Batista and P. Yang, Inorg. Chem., 2018, 57, 5352–5363 Search PubMed.
- J.-B. Liu, G. P. Chen, W. Huang, D. L. Clark, W. H. E. Schwarz and J. Li, Dalton Trans., 2017, 46, 2542–2550 RSC.
- A. Jennifer G, Y. Gao, G. Schreckenbach and E. Varathan, Dalton Trans., 2022, 51, 10006–10019 RSC.
- S.-X. Hu, J.-J. Liu, J. K. Gibson and J. Li, Inorg. Chem., 2018, 57, 2899–2907 CrossRef CAS PubMed.
- S. P. Kelley, V. Smetana, J. S. Nuss, D. A. Dixon, M. Vasiliu, A.-V. Mudring and R. D. Rogers, Inorg. Chem., 2020, 59, 2861–2869 CrossRef CAS.
- C. J. Windorff, C. Celis-Barros, J. M. Sperling, N. C. McKinnon and T. E. Albrecht-Schmitt, Chem. Sci., 2020, 11, 2770–2782 RSC.
- A. L. Smith, P. E. Raison, A. Hen, D. Bykov, E. Colineau, J. P. Sanchez, R. J. M. Konings and A. K. Cheetham, Dalton Trans., 2015, 44, 18370–18377 RSC.
- S. K. Cary, M. Vasiliu, R. E. Baumbach, J. T. Stritzinger, T. D. Green, K. Diefenbach, J. N. Cross, K. L. Knappenberger, G. Liu and M. A. Silver, Nat. Commun., 2015, 6, 6827 CrossRef CAS.
- D. E. Smiles, G. Wu, P. Hrobárik and T. W. Hayton, J. Am. Chem. Soc., 2016, 138, 814–825 CrossRef CAS.
- T. Vitova, I. Pidchenko, D. Fellhauer, P. S. Bagus, Y. Joly, T. Pruessmann, S. Bahl, E. Gonzalez-Robles, J. Rothe, M. Altmaier, M. A. Denecke and H. Geckeis, Nat. Commun., 2017, 8, 16053 Search PubMed.
- M. Zegke, X. Zhang, I. Pidchenko, J. A. Hlina, R. M. Lord, J. Purkis, G. S. Nichol, N. Magnani, G. Schreckenbach, T. Vitova, J. B. Love and P. L. Arnold, Chem. Sci., 2019, 10, 9740–9751 Search PubMed.
- R. Polly, B. Schacherl, J. Rothe and T. Vitova, Inorg. Chem., 2021, 60, 18764–18776 Search PubMed.
- C. L. Silva, L. Amidani, M. Retegan, S. Weiss, E. F. Bazarkina, T. Graubner, F. Kraus and K. O. Kvashnina, Nat. Commun., 2024, 15, 6861 Search PubMed.
- G. Leinders, R. Bes, J. Pakarinen, K. Kvashnina and M. Verwerft, Inorg. Chem., 2017, 56, 6784–6787 CrossRef CAS PubMed.
- N. J. Wolford, X. Yu, S. C. Bart, J. Autschbach and M. L. Neidig, Dalton Trans., 2020, 49, 14401–14410 RSC.
- D.-C. Sergentu, G. T. Kent, S. L. Staun, X. Yu, H. Cho, J. Autschbach and T. W. Hayton, Inorg. Chem., 2020, 59, 10138–10145 Search PubMed.
- D. R. Kindra and W. J. Evans, Chem. Rev., 2014, 114, 8865–8882 CrossRef CAS PubMed.
- A. Kerridge, Chem. Commun., 2017, 53, 6685–6695 RSC.
- J. Su, E. R. Batista, K. S. Boland, S. E. Bone, J. A. Bradley, S. K. Cary, D. L. Clark, S. D. Conradson, A. S. Ditter, N. Kaltsoyannis, J. M. Keith, A. Kerridge, S. A. Kozimor, M. W. Löble, R. L. Martin, S. G. Minasian, V. Mocko, H. S. La Pierre, G. T. Seidler, D. K. Shuh, M. P. Wilkerson, L. E. Wolfsberg and P. Yang, J. Am. Chem. Soc., 2018, 140, 17977–17984 CrossRef CAS PubMed.
- J. A. Platts and R. J. Baker, Dalton Trans., 2020, 49, 1077–1088 RSC.
- S. Chattaraj and A. Bhattacharyya, Struct. Chem., 2023, 34, 307–316 CrossRef CAS.
- M. P. Kelley, J. Su, M. Urban, M. Luckey, E. R. Batista, P. Yang, J. C. Shafer, M. P. Kelley, E. R. Batista and J. C. Shafer, J. Am. Chem. Soc., 2017, 139, 9901–9908 CrossRef CAS PubMed.
- K. A. Pace, V. V. Klepov, A. A. Berseneva and H.-C. zur Loye, Chem. – Eur. J., 2021, 27, 5835–5841 CrossRef CAS PubMed.
- B. Sadhu and M. Dolg, Inorg. Chem., 2019, 58, 9738–9748 Search PubMed.
- S. Cooper and N. Kaltsoyannis, Dalton Trans., 2021, 50, 1478–1485 RSC.
- S. Cooper and N. Kaltsoyannis, Dalton Trans., 2022, 51, 5929–5937 RSC.
- J. Tanti, M. Lincoln and A. Kerridge, Inorganics, 2018, 6, 88 Search PubMed.
- F. A. Pereiro, S. S. Galley, J. A. Jackson and J. C. Shafer, Inorg. Chem., 2024, 63, 9687–9700 CrossRef CAS PubMed.
- S. G. Minasian, J. M. Keith, E. R. Batista, K. S. Boland, D. L. Clark, S. D. Conradson, S. A. Kozimor, R. L. Martin, D. E. Schwarz, D. K. Shuh, G. L. Wagner, M. P. Wilkerson, L. E. Wolfsberg and P. Yang, J. Am. Chem. Soc., 2012, 134, 5586–5597 CrossRef CAS.
- A. Parimi, E. Mosher and G. Schreckenbach, Dalton Trans., 2023, 52, 18035–18044 RSC.
- A. Jennifer G, Y. Gao, G. Schreckenbach and E. Varathan, Inorg. Chem., 2023, 62, 6920–6933 CrossRef CAS.
- M. J. Tassell and N. Kaltsoyannis, Dalton Trans., 2010, 39, 6719 RSC.
- I. Kirker and N. Kaltsoyannis, Dalton Trans., 2011, 40, 124–131 RSC.
- E. Lu, S. Sajjad, V. E. J. Berryman, A. J. Wooles, N. Kaltsoyannis, S. T. Liddle, E. Lu, S. Sajjad, V. E. J. Berryman, A. J. Wooles, N. Kaltsoyannis and S. T. Liddle, Nat. Commun., 2019, 10, 634 CrossRef CAS PubMed.
- I. D. Prodan, G. E. Scuseria and R. L. Martin, Phys. Rev. B, 2007, 76, 33101 CrossRef.
- R. E. Wilson, S. De Sio and V. Vallet, Nat. Commun., 2018, 9, 622 CrossRef PubMed.
- S. N. Kelly, D. R. Russo, E. T. Ouellette, D. Roy, A. J. Swift, M. A. Boreen, P. W. Smith, L. M. Moreau, J. Arnold and S. G. Minasian, Chem. Sci., 2024, 15, 13325–13332 RSC.
- N. Kaltsoyannis and A. Kerridge, Nat. Rev. Chem., 2024, 8, 701–712 CrossRef CAS PubMed.
- S. Armstrong, T. Malcomson and A. Kerridge, Phys. Chem. Chem. Phys., 2022, 24, 26692–26700 RSC.
- T. S. Neill, H. Ramanantoanina, N. Palina, T. Prüßmann, J. Rothe, X. Gaona, D. Fellhauer, D. Schild, C. Wansorra, D. Hauschild, R. Steininger, B. Schacherl, C. Heske, L. Weinhardt, O. Walter, F. Weigend and T. Vitova, J. Am. Chem. Soc., 2025, 147, 35401–35412 CrossRef CAS PubMed.
- M. J. Polinski, E. B. Garner Iii, R. Maurice, N. Planas, J. T. Stritzinger, T. G. Parker, J. N. Cross, T. D. Green, E. V. Alekseev and S. M. Van Cleve, Nat. Chem., 2014, 6, 387–392 CrossRef CAS PubMed.
- M. A. Silver, S. K. Cary, J. A. Johnson, R. E. Baumbach, A. A. Arico, M. Luckey, M. Urban, J. C. Wang, M. J. Polinski and A. Chemey, Science, 2016, 353, aaf3762 CrossRef PubMed.
- N. Kaltsoyannis, Chem. – Eur. J., 2018, 24, 2815–2825 Search PubMed.
- B. Sadhu, M. Dolg and M. S. Kulkarni, J. Comput. Chem., 2020, 41, 1427–1435 CrossRef CAS PubMed.
- D. Fiszbein, M. Beltran-Leiva, G. Rao, R. D. Britt, E. Batista, W. Lukens, P. Yang and P. Arnold, ChemRxiv, preprint, 2025 DOI:10.26434/chemrxiv-2025-89g00.
- J. A. Branson, P. W. Smith, D.-C. Sergentu, D. R. Russo, H. Gupta, C. H. Booth, J. Arnold, E. J. Schelter, J. Autschbach and S. G. Minasian, J. Am. Chem. Soc., 2024, 146, 25640–25655 CrossRef CAS.
- L. Zhang, G. Hou, G. Zi, W. Ding and M. D. Walter, J. Am. Chem. Soc., 2016, 138, 5130–5142 CrossRef CAS PubMed.
- A. R. Bubas, A. D. French, K. M. Melby, M. J. Rodriguez and R. M. Cox, Inorg. Chem. Front., 2025, 12, 1503–1516 Search PubMed.
- B. Fang, W. Ren, G. Hou, G. Zi, D.-C. Fang, L. Maron and M. D. Walter, J. Am. Chem. Soc., 2014, 136, 17249–17261 CrossRef CAS PubMed.
- L. Zhang, B. Fang, G. Hou, L. Ai, W. Ding, M. D. Walter and G. Zi, Dalton Trans., 2016, 45, 16441–16452 RSC.
- G. Zi, Chem. Commun., 2018, 54, 7412–7430 Search PubMed.
- S. Wang, D. Wang, Y. Heng, T. Li, W. Ding, G. Zi and M. D. Walter, Inorg. Chem., 2024, 63, 7473–7492 CrossRef CAS PubMed.
- D. Wang, W. Ding, G. Hou, G. Zi and M. D. Walter, Chem. – Eur. J., 2020, 26, 16888–16899 CrossRef CAS PubMed.
- L. Grunwald, D. F. Abbott and V. Mougel, JACS Au, 2024, 4, 1315–1322 CrossRef CAS PubMed.
- R. S. C. Charman and D. J. Liptrot, ChemPlusChem, 2024, 89, e202400439 CrossRef CAS PubMed.
- D. Munz, in Advances in Inorganic Chemistry, ed K. Meyer and R. van Eldik, Academic Press, 2023, vol. 82, pp. 189–236 Search PubMed.
- B. D. Vincenzini, X. Yu, S. Paloc, P. W. Smith, H. Gupta, P. Pandey, G. T. Kent, O. Ordonez, T. Keller, M. R. Gau, A. M. Bacon, S. G. Minasian, T. W. Hayton, J. Autschbach and E. J. Schelter, Nat. Chem., 2025, 17, 961–967 CrossRef CAS PubMed.
- R. G. Agarwal, S. C. Coste, B. D. Groff, A. M. Heuer, H. Noh, G. A. Parada, C. F. Wise, E. M. Nichols, J. J. Warren and J. M. Mayer, Chem. Rev., 2021, 122, 1–49 CrossRef PubMed.
- J. J. Warren, T. A. Tronic and J. M. Mayer, Chem. Rev., 2010, 110, 6961–7001 Search PubMed.
- D. G. Nocera, J. Am. Chem. Soc., 2022, 144, 1069–1081 CrossRef CAS PubMed.
- E. R. Mikeska, R. E. Wilson and J. D. Blakemore, Chem. – Eur. J., 2025, 31, e202402963 CrossRef CAS PubMed.
- D. Perales, N. J. Lin, M. R. Bronstetter, S. A. Ford, M. Zeller and S. C. Bart, Organometallics, 2022, 41, 606–616 CrossRef CAS.
- L. R. Valerio, B. M. Hakey, W. W. Brennessel and E. M. Matson, Chem. Commun., 2022, 58, 11244–11247 RSC.
- X. Hou, J. R. McLachlan and C. J. Dares, Chem. Commun., 2021, 57, 10891–10894 RSC.
- C. J. Dares, A. M. Lapides, B. J. Mincher and T. J. Meyer, Science, 2015, 350, 652–655 CrossRef CAS PubMed.
- R. Faizova, F. Fadaei-Tirani, A. S. Chauvin and M. Mazzanti, Angew. Chem., 2021, 133, 8308–8316 CrossRef.
- L. Yang, D. R. Powell and R. P. Houser, Dalton Trans., 2007, 955–964 Search PubMed.
- J. P. Perdew, M. Ernzerhof and K. Burke, J. Chem. Phys., 1996, 105, 9982–9985 CrossRef CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 16, Revision A.03, Wallingford, CT, 2016 Search PubMed.
- X. Cao, M. Dolg and H. Stoll, J. Chem. Phys., 2002, 118, 487–496 CrossRef.
- X. Cao and M. Dolg, J. Mol. Struct. THEOCHEM, 2004, 673, 203–209 CrossRef CAS.
- R. Krishnan, J. S. Binkley, R. Seeger and J. A. Pople, J. Chem. Phys., 1980, 72, 650–654 CrossRef CAS.
- R. F. W. Bader, Acc. Chem. Res., 2002, 18, 9–15 CrossRef.
- T. Lu and F. Chen, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS PubMed.
- D. Y. Zubarev and A. I. Boldyrev, Phys. Chem. Chem. Phys., 2008, 10, 5207–5217 RSC.
- J. E. D. Glendening, K. Badenhoop, A. E. Reed, J. E. Carpenter, J. A. Bohmann, C. M. Morales, P. Karafiloglou, C. R. Landis and F. Weinhold, NBO 7.0, Theoretical Chemistry Institute, University of Wisconsin, Madison, 2018 Search PubMed.
- M. A. Blanco, A. Martín Pendás and E. Francisco, J. Chem. Theory Comput., 2005, 1, 1096–1109 CrossRef CAS PubMed.
- A. M. Pendás, J. L. Casals-Sainz and E. Francisco, Chem. – Eur. J., 2019, 25, 309–314 CrossRef PubMed.
- T. A. Keith, AIMAll (Version 19.10.12), TK Gristmill Software, Overland Park, KS, USA, 2019 Search PubMed.
- V. E. J. Berryman, J. J. Shephard, T. Ochiai, A. N. Price, P. L. Arnold, S. Parsons and N. Kaltsoyannis, Phys. Chem. Chem. Phys., 2020, 22, 16804–16812 RSC.
- L. Yang, S. Cooper and N. Kaltsoyannis, Phys. Chem. Chem. Phys., 2021, 23, 4167–4177 RSC.
- J. P. Janet, S. Ramesh, C. Duan and H. J. Kulik, ACS Cent. Sci., 2020, 6, 513–524 CrossRef CAS PubMed.
- C. F. Wise, R. G. Agarwal and J. M. Mayer, J. Am. Chem. Soc., 2020, 142, 10681–10691 CrossRef CAS.
- E. P. Cappellani, S. D. Drouin, G. Jia, P. A. Maltby, R. H. Morris and C. T. Schweitzer, J. Am. Chem. Soc., 1994, 116, 3375–3388 CrossRef CAS.
- B. O. Roos, P. R. Taylor and P. E. M. Sigbahn, Chem. Phys., 1980, 48, 157–173 CrossRef CAS.
- F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2012, 2, 73–78 CAS.
- F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2025, 15, e70019 Search PubMed.
- T. Nakajima and K. Hirao, Chem. Rev., 2012, 112, 385–402 CrossRef CAS PubMed.
- D. A. Pantazis and F. Neese, J. Chem. Theory Comput., 2011, 7, 677–684 CrossRef CAS.
- F. Weigend, F. Furche and R. Ahlrichs, J. Chem. Phys., 2003, 119, 12753–12762 Search PubMed.
- F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- G. L. Stoychev, A. A. Auer and F. Neese, J. Chem. Theory Comput., 2017, 13, 554–562 CrossRef CAS PubMed.
- C. Angeli, R. Cimiraglia and J.-P. Malrieu, J. Chem. Phys., 2002, 117, 9138–9153 CrossRef CAS.
- P. A. Malmqvist, A. Rendell and B. O. Roos, J. Phys. Chem., 1990, 94, 5477–5482 CrossRef CAS.
- M. Y. Toriyama, A. M. Ganose, M. Dylla, S. Anand, J. Park, M. K. Brod, J. M. Munro, K. A. Persson, A. Jain and G. J. Snyder, Mater. Today Electron., 2022, 1, 100002 CrossRef.
- J. R. Walensky, R. L. Martin, J. W. Ziller and W. J. Evans, Inorg. Chem., 2010, 49, 10007–10012 CrossRef CAS PubMed.
- S. Niu, H.-X. Cai, H.-B. Zhao, L. Li and Q.-J. Pan, RSC Adv., 2020, 10, 26880–26887 RSC.
- J. M. Lozano, D. L. Clark, S. D. Conradson, C. Den Auwer, C. Fillaux, D. Guilaumont, D. Webster Keogh, J. Mustre de Leon, P. D. Palmer and E. Simoni, Phys. Chem. Chem. Phys., 2009, 11, 10396–10402 RSC.
- D. M. King, J. McMaster, F. Tuna, E. J. L. McInnes, W. Lewis, A. J. Blake and S. T. Liddle, J. Am. Chem. Soc., 2014, 136, 5619–5622 CrossRef CAS PubMed.
- T. S. Collins, C. Celis-Barros, M. J. Beltrán-Leiva, N. H. Anderson, M. Zeller, T. Albrecht-Schönzart and S. C. Bart, Inorg. Chem., 2020, 59, 18461–18468 CrossRef CAS.
- T. Li, Y. Heng, D. Wang, G. Hou, G. Zi, W. Ding and M. D. Walter, Inorg. Chem., 2024, 63, 9487–9510 Search PubMed.
- S. Roy Chowdhury, N. Rehberg and B. Vlaisavljevich, Inorg. Chem., 2025, 64, 5866–5877 CrossRef CAS PubMed.
- A. Kerridge, RSC Adv., 2014, 4, 12078–12086 RSC.
- X. Yu, D.-C. Sergentu, R. Feng and J. Autschbach, Inorg. Chem., 2021, 60, 17744–17757 CrossRef CAS PubMed.
- J. Du, X. Cheng, X. Zhou, M. Shi, G. Jiang and D. Chen, J. Phys. Chem. A, 2024, 128, 9587–9595 CrossRef CAS PubMed.
- E. Francisco, D. Menéndez Crespo, A. Costales and Á. Martín Pendás, J. Comput. Chem., 2017, 38, 816–829 CrossRef CAS PubMed.
- J. Jara-Cortés and J. Hernández-Trujillo, J. Comput. Chem., 2018, 39, 1103–1111 CrossRef PubMed.
- Z. Badri and C. Foroutan-Nejad, Phys. Chem. Chem. Phys., 2016, 18, 11693–11699 RSC.
- S. Liu, C. K. Schauer and L. G. Pedersen, J. Chem. Phys., 2009, 131, 164107 CrossRef PubMed.
- S. K. Burger, S. Liu and P. W. Ayers, J. Phys. Chem. A, 2011, 115, 1293–1304 CrossRef CAS.
- F. De Proft, S. Amira, K. Choho and P. Geerlings, J. Phys. Chem., 1994, 98, 5227–5233 Search PubMed.
- X. Xiao, X. Cao, D. B. Zhao, C. Y. Rong and S. B. Liu, Acta Phys.-Chim. Sin., 2020, 36, 1906034 Search PubMed.
- Y. Huang, L. Liu and S. Liu, Chem. Phys. Lett., 2012, 527, 73–78 CrossRef CAS.
- CCDC 2445923: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2n35rq.
| This journal is © the Owner Societies 2026 |






