Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Controls and evidence for assigning europium emission in Eu-treated CsPbCl3 nanocrystals
Yanfei
Wang
,
Martin
Vacha
 and
Shun
Omagari
and
Shun
Omagari
 *
*
School of Materials and Chemical Technology, Institute of Science Tokyo, 2-12-1 Ookayama, Meguro, Tokyo 152-8552, Japan. E-mail: omagari.s.aa@m.titech.ac.jp; Tel: +81 3 5734 3643
First published on 24th February 2026
Abstract
Lanthanide doping of semiconductor nanocrystals is widely pursued to enhance luminescence via photosensitization, and lead halide perovskite nanocrystals have been frequently reported as effective hosts due to their large absorption cross sections. Here we synthesized cubic-CsPbCl3 nanocrystals in the presence of Eu3+ precursors, which have been reported to exhibit strong photosensitized Eu3+ emission, and reproduce the emission motifs reported for Eu-treated CsPbCl3 nanocrystals. Using centrifugation fractionation together with control experiments and spectral comparisons, we find that the ensemble Eu3+ line emission is dominated by Eu–ligand complex byproducts that persist through standard processing. Transmission electron microscopy confirms the presence of cubic CsPbCl3 nanocrystals, and spot energy-dispersive X-ray spectroscopy detects Eu on individual cubic particles. Single-particle spectroscopy further reveals dilute emissive particles exhibiting anticorrelated host emission and an anomalous broad band, which we assign to a lanthanide-centered charge-transfer state (CTS), indicating a competing pathway that disfavors Eu3+ f–f emission from the perovskite host. Overall, these results decouple evidence for Eu-containing CsPbCl3 nanocrystals from the origin of ensemble Eu3+ line emission, motivate purification and identification protocols, and demonstrate the utility of single-particle spectroscopy for assessing lanthanide-doped semiconductor nanocrystals.
1. Introduction
Trivalent lanthanide (Ln3+) ions are known for their high monochromaticity and long emission lifetimes and have been used in lighting, displays, lasers, and bioimaging.1–4 A disadvantage of Ln3+ ions is their small absorption cross-section, which is 3–4 orders of magnitude lower than that of typical organic phosphors, leading to low emission intensity even at unity quantum yield. One strategy to obtain intense luminescence from Ln3+ ions is photosensitization, in which an electronic state with a large absorption cross-section acts as an antenna and transfers energy to the emitting states of the Ln3+ ion. For example, Ce3+, Eu3+, and Tb3+ ions are used in fluorescent lamps, where 4f–5d, ligand-to-metal charge transfer, and Ce3+ → Tb3+ energy transfer, respectively, serve as antennae that absorb light from the UV discharge lamp.5–7 In Ln3+ complexes, excited states of organic ligands play the role of antennae and transfer energy to the Ln3+ ion mainly via the triplet excited state of the ligands.8A similar approach has been applied to inorganic semiconductor nanoparticles.9 The first report of Ln3+-doped inorganic semiconductor nanoparticles with photosensitized emission dates back to 2006, when Eu was incorporated into CdS quantum dots.10 Other host materials soon followed, including II-VI semiconductors (e.g., CdSe,11,12 ZnS13,14), perovskites (e.g., CsPbCl3,15–17 CsPbBr315,18–20), and others (e.g., CaZnOS,21 In2O3,22 and SnO223). In these materials, the host semiconductor is proposed to absorb light and transfer energy to Ln3+ ions through electron and/or energy transfer with proper host design, leading in some cases to intense Ln3+ luminescence.24–26
There was a controversy regarding whether, and how, Ln3+ ions were actually doped into cesium lead halide nanocrystals18–20,26–30 until the report by Liu et al. in 2023.31 Using HAADF-STEM and EDS imaging of a single ∼20 nm CsPbCl3 nanoparticle, they unambiguously proved that Yb3+ ions were successfully doped, mainly by occupying lattice interstitial sites and substituting Pb2+ cations. Atom-probe tomography of a sub-micron CsPbCl3 single crystal showed homogeneous doping at ∼5 at%. The finding represents a major milestone for single-particle detection of Ln3+-doped nanocrystals. The low absorption cross-section (low absorption rate) combined with millisecond-scale emission lifetimes (low emission rate) of Ln3+ ions means a low excitation–emission cycle rate per unit time, so single-particle detection of Ln3+-doped nanocrystals has traditionally required 103–105 W cm−2 excitation—far above the ∼10–102 W cm−2 typically used for single-molecule spectroscopy.32–34 Although photosensitization addresses the low absorption rate, the low emission rate cannot be compensated beyond a threshold where the excitation rate exceeds the emission rate (as in Ln3+ complexes). In nanocrystals, however, a few hundred Ln3+ ions confined in a small volume can yield higher total emission per unit time. For example, a 10 nm CsPbCl3 nanocrystal doped at 5 at% contains ∼280 Ln3+ ions. If each Ln3+ has an intrinsic 20% quantum yield in the CsPbCl3 host, reliable single-particle detection is achievable in a standard setup (∼50 W cm−2 excitation, 10% detection efficiency) with host → Ln3+ energy-transfer efficiency as low as ∼6%. For comparison, a typical organic fluorophore with ∼90% quantum yield reaches a similar photon output under the same conditions.
The next potential ambiguity to resolve for Ln3+-doped CsPbCl3 nanocrystals is how Ln3+ emission is observed from these systems. For instance, prior reports on CsPbCl3:Eu3+ nanocrystals from different groups clearly show the characteristic Eu3+ luminescence peaks, with a main peak at 612 nm,15,18,35–38 but the emission is usually accompanied by radiative excitonic recombination of the CsPbCl3 host (∼410 nm) and/or an anomalous broad band in between, implying at least three types of emitting centers. The combinations and relative intensities of these emissions vary across reports. In many reports, the Eu3+ line emission is implicitly assigned to host-sensitized Eu3+ dopants under the assumption that the measured ensemble is a sufficiently purified population of CsPbCl3:Eu3+ nanocrystals, an assumption often supported mainly by TEM/STEM-EDS evidence for Eu in nanocrystals. However, without purification steps that explicitly exclude Eu-containing byproducts, the sources of the emission remain ambiguous at the ensemble level; the Eu3+ ions that are doped in the CsPbCl3 nanocrystal host may be dark, while impurities containing Eu3+ could actually be the source of the Eu3+ emission. In systems like CsPbCl3 nanocrystals where the synthesized products are purified through centrifugation, there is no guarantee of full purification. Common identification techniques such as TEM (and STEM-EDS), XRD, solid-state NMR, or IR can miss impurities if they are insensitive to the measurement method. Single-particle spectroscopy is a possible workaround to address this ambiguity since they spatially separate impurities even at very low concentration.
Herein, we revisit the origin of the Eu3+ line-emission (and other accompanied emission) motifs commonly observed in Eu-treated CsPbCl3 nanocrystal syntheses, with the specific goal of separating evidence for Eu-containing perovskite nanocrystals from evidence for the emitting Eu3+ species. Our approach combines centrifugation-based fractionation with control syntheses and comparative optical spectroscopy to track how Eu3+ emission features partition across the products of synthesis and processing. In parallel, we employ electron microscopy and spot energy-dispersive X-ray spectroscopy to confirm the presence of cubic CsPbCl3 nanocrystals and to evaluate whether Eu is detectable on individual particles. Finally, we use single-particle spectroscopy to probe dilute, localized emitters that can be obscured in ensemble measurements. Together, these measurements enable a more direct assignment of Eu3+ emission in Eu-treated perovskite nanocrystal preparations and clarify what additional evidence is required to connect Eu3+ line emission to the nanocrystal host.
2. Experimental section
2.1 Materials
Cs2CO3 (99.9%), PbCl2 (99.999%), Eu(NO3)3·6H2O (>99.95%), oleic acid (OA), oleyl amine (OAm), and PMMA (Mw ∼ 350![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) were purchased from Sigma-Aldrich Japan. 1-Octadecene (ODE) was purchased from Tokyo Chemical Industries. Toluene was purchased from Kanto Chemical Co., Inc. All reagents were used without further purification.
000) were purchased from Sigma-Aldrich Japan. 1-Octadecene (ODE) was purchased from Tokyo Chemical Industries. Toluene was purchased from Kanto Chemical Co., Inc. All reagents were used without further purification.
2.2 Apparatus
2.3 Synthesis
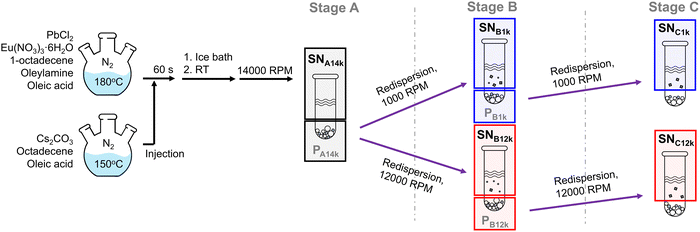
The nanocrystals were synthesized using the hot injection method, as previously reported.37 The selection of this particular report was based on how reliably and consistently the properties can be reproduced. The detailed procedure is as described below (Scheme 1).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 RPM (16
000 RPM (16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 RCF) for 5 minutes. The precipitate was redispersed in 0.5 mL toluene, sonicated, and separated into two 0.25 mL portions for further centrifugation. One portion was centrifuged at 1000 RPM (80 RCF), and the other at 12
500 RCF) for 5 minutes. The precipitate was redispersed in 0.5 mL toluene, sonicated, and separated into two 0.25 mL portions for further centrifugation. One portion was centrifuged at 1000 RPM (80 RCF), and the other at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 RPM (11
000 RPM (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 RCF). Each of these precipitates was redispersed in 0.5 mL toluene and sonicated. Both redispersed precipitates were then centrifuged at the same speeds as in the previous step, and their supernatants were collected. The samples were stored in dark and measured on the next day of the synthesis to minimize potential degradation and exposures to atmosphere. All samples were diluted by tenfold before the measurements.
700 RCF). Each of these precipitates was redispersed in 0.5 mL toluene and sonicated. Both redispersed precipitates were then centrifuged at the same speeds as in the previous step, and their supernatants were collected. The samples were stored in dark and measured on the next day of the synthesis to minimize potential degradation and exposures to atmosphere. All samples were diluted by tenfold before the measurements.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 RPM. The corresponding precipitate is PA14k.
000 RPM. The corresponding precipitate is PA14k.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 RPM). Precipitate: PB12k.
000 RPM). Precipitate: PB12k.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 RPM).
000 RPM).
3. Results and discussion
We prepared a total of five samples corresponding to different purification stages and centrifugation speeds (Scheme 1), and their compositions and supporting evidence are summarized in Table 1. Stage A used the maximum speed of our centrifuge (14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 RPM; 16
000 RPM; 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 RCF for our rotor) to pellet as much particulate material as possible and to facilitate removal of a supernatant enriched in molecular/ligand species. Stages B and C then used two widely separated speeds—1000 RPM (80 RCF) and 12
500 RCF for our rotor) to pellet as much particulate material as possible and to facilitate removal of a supernatant enriched in molecular/ligand species. Stages B and C then used two widely separated speeds—1000 RPM (80 RCF) and 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 RPM (11
000 RPM (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 RCF)—to create a strong sedimentation contrast for differential fractionation. We report both RPM and RCF because RCF is the physically relevant quantity but many prior reports provide only RPM, which depends on rotor radius. The 1000 RPM condition was chosen as the lowest speed that still produced a clean, collectable pellet/supernatant boundary, whereas 12
700 RCF)—to create a strong sedimentation contrast for differential fractionation. We report both RPM and RCF because RCF is the physically relevant quantity but many prior reports provide only RPM, which depends on rotor radius. The 1000 RPM condition was chosen as the lowest speed that still produced a clean, collectable pellet/supernatant boundary, whereas 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 RPM reflects a commonly used upper-end condition. Under this design, the Stage B supernatant at 1000 RPM (SNB1k) is expected to retain the largest overall population of dispersible nanocrystals, while the Stage B supernatant at 12
000 RPM reflects a commonly used upper-end condition. Under this design, the Stage B supernatant at 1000 RPM (SNB1k) is expected to retain the largest overall population of dispersible nanocrystals, while the Stage B supernatant at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 RPM (SNB12k) should be comparatively enriched in the smallest/most dispersible species. The Stage C supernatants (SNC1k and SNC12k) represent the residual components that remain dispersible in toluene after further processing. The following sections describe the structural and spectroscopic characterization that underpins these assignments; deviations from the expected fractionation behavior are discussed as mechanistically informative rather than treated as experimental noise.
000 RPM (SNB12k) should be comparatively enriched in the smallest/most dispersible species. The Stage C supernatants (SNC1k and SNC12k) represent the residual components that remain dispersible in toluene after further processing. The following sections describe the structural and spectroscopic characterization that underpins these assignments; deviations from the expected fractionation behavior are discussed as mechanistically informative rather than treated as experimental noise.
| Stage | Fraction | Main component | CsPbCl3 | Component CsPbCl3:Eu3+ | OA + OAm + Eu3+ complexes |
|---|---|---|---|---|---|
| The “main component” is assigned based on the dominant luminescent contribution within each fraction. Top rows indicate primary evidence (direct observation in the data). Bottom rows in parentheses indicate secondary evidence (assignments deduced from comparison with other measurements). A dash indicates that the component was not observed. | |||||
| Stage A | SNA14k | OA + OAm + Eu3+ complexes | — | — | Fig. 2(a) |
| — | — | (Fig. 3(a) and 4(a)) | |||
| Stage B | SNB1k | OA + OAm + Eu3+ complexes | Fig. 1(a–c), 4(b) and Fig. S8 | Fig. 1(a–c), 4(a) and Fig. S8, Table S1: | Fig. 1(a and d) |
| (Fig. 2(c)) | (Fig. 3a) | ||||
| SNB12k | OA + OAm + Eu3+ complexes | Fig. S6(a) and S9 | Fig. S6(a) and Fig. 4(c and d) | Fig. 2(b) | |
| (Fig. 1(d)) | (Fig. 3(a)) | ||||
| Stage C | SNC1k | CsPbCl3 | Fig. 2(c) and 4(f) | Fig. 4(e) | — |
| (Fig. S2(d)) | (Fig. 4(a)) | — | |||
| SNC12k | CsPbCl3 | Fig. 2(d) and 4(h) | Fig. 4(g) | — | |
| (Fig. S2(e)) | (Fig. 4(c and d)) | — | |||
3.1 Reproduction of CsPbCl3:Eu3+ and their properties
According to the previous report,37 the target sample is the supernatant after second centrifugation (Stage B). In order to extract as many target CsPbCl3:Eu3+ nanocrystals as possible, the speed of the second centrifugation was set to 1000 RPM (80 RCF). The obtained sample will be denoted SNB1k. The absorption and emission spectrum (369 nm excitation) of SNB1k recorded 1 day after synthesis are shown in Fig. 1(a). The absorption spectrum is a featureless rise in absorbance toward shorter wavelengths, beginning below ∼500 nm (named Type-A1). The absence of any defined absorption peak (in contrast to the ∼400 nm band-edge seen in typical CsPbCl3 nanocrystals, i.e. Type-A2 in Fig. S2(b)) suggests that the absorption of SNB1k is dominated by species other than CsPbCl3 nanocrystals. Meanwhile, the emission spectrum consisted of three distinct components: Type-E1, a broad band from 400–600 nm peaking near ∼450 nm; Type-E2, the relatively sharp excitonic emission of CsPbCl3 near ∼405 nm;39 and Type-E3, Eu3+ lines with a peak at 615 nm (parity-forbidden 4f–4f transitions: 5D0 → 7F2).40 Consequently, the presence of Eu3+ line emission in an ensemble spectrum establishes that Eu3+ is being sensitized, but it does not uniquely identify the sensitizing species. Both lattice-incorporated Eu3+ and Eu-containing molecular byproducts can, in principle, produce similar Eu3+ emission signatures.41,42 Previous reports on CsPbCl3:Eu3+ nanocrystals report Type-E3 spectrum (Eu3+ emission) along with Type-E1 or Type-E2 spectra,15,18,35–38,43–46 so we were able to reproduce most of the spectral features reported for these systems. We note that the Type-E3 spectrum was weak compared to the other peaks, also in alignment with the synthetic protocol we followed for this study.37The temporal evolution in Fig. S3(b) further supports that the emission components originate from different species. Specifically, Type-E2 becomes undetectable after 11 days, while the other emission components remain largely unchanged. If all features arose from a single emitter population, their relative contributions would be expected to change in a correlated manner; instead, the selective disappearance of Type-E2 supports decoupled emission origins. This also highlights that the mere coexistence of Eu3+ line emission and CsPbCl3-related emission in an ensemble spectrum (e.g., on day 1 after synthesis) is not sufficient to conclude host-sensitized Eu3+ emission, because distinct emissive species can contribute simultaneously to the same ensemble measurement. Further experimental evidence and discussion will be described in Section 3.2. Low emission intensity of Type-E2 spectrum in SNB1k in comparison to the undoped samples (Fig. S2(b)) indicates either inefficient nanocrystal emission or low nanocrystal population, especially considering that the centrifugation speed was set low to collect as many nanoparticles as possible. Based on the absorption spectrum (which showed Type-A1), a large population of weakly emissive nanocrystals is unlikely; their strong absorption would remain visible.47 We therefore conclude that only a small quantity of nanocrystals was present in SNB1k.
We also obtained transmission electron microscope (TEM) images of SNB1k (Fig. 1(b–d)). Cubic nanocrystals (∼25–35 nm, Fig. 1(b and c)) and medium-sized spherical nanoparticles (∼20 nm, Fig. 1(d)) were found. Zoomed-in TEM images of these particles are provided in Fig. S4(a–c). As a reference, Fig. S4(d and e) shows the undoped CsPbCl3 nanocrystals under the same purification procedure, where only ∼7 nm cubes were observed. Based on prior reports18–20,26–29,31 and our undoped control, the cubic nanocrystals are the target CsPbCl3:Eu3+ material, with the remaining particles attributed to byproducts. Furthermore, energy dispersive X-ray spectroscopy (EDS) was performed on a single cubic nanocrystal and surrounding regions (Fig. S5 and Table S1). All expected elements of CsPbCl3:Eu3+—Cs, Pb, Cl, and Eu—were detected, indicating that the cubic CsPbCl3 nanocrystals indeed contained Eu3+ ions. A high carbon signal was also observed at the nanocrystal location, which cannot be fully attributed to the carbon support film of the TEM grid and likely originates from surface ligands (OA/OAm), consistent with standard colloidal syntheses. In contrast, surrounding areas without visible particles showed high carbon but no detectable Eu, suggesting free ligands or organic byproducts. Overall, we were able to reproduce most properties reported previously.
3.2 Comparison with other samples and attribution of the emission types
In order to separate the emission spectral components in the mixture of the product, we also measured the absorption and emission spectrum of the sample in Stage B at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 RPM (16
000 RPM (16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 RCF). The sample will be denoted SNB12k. The absorption spectrum of SNB12k was identical (Type-A1) to that of SNB1k (both showed Type-A1 absorption), whereas the emission lacked Type-E2 spectrum (Fig. 2(b)). This is likely because of the higher centrifugation speed that removes more CsPbCl3 nanocrystals from the supernatant, thus removing most host emission. It should be noted, however, that TEM images still showed that the cubic nanocrystals (Fig. S6(a)) and medium-sized spherical nanoparticle (Fig. S6(b)) were still present in the sample, and also included large spherical nanoparticles (∼40 nm, Fig. S6(c)), and small spherical nanoparticles (∼5 nm, Fig. S6(d)) in addition to those observed in SNB1k. The lack of Type-E2 spectrum without a major difference in the Type-E3 emission intensity indicates that CsPbCl3 nanocrystal host emission and Eu3+ emission are not necessarily correlated at ensemble level.
500 RCF). The sample will be denoted SNB12k. The absorption spectrum of SNB12k was identical (Type-A1) to that of SNB1k (both showed Type-A1 absorption), whereas the emission lacked Type-E2 spectrum (Fig. 2(b)). This is likely because of the higher centrifugation speed that removes more CsPbCl3 nanocrystals from the supernatant, thus removing most host emission. It should be noted, however, that TEM images still showed that the cubic nanocrystals (Fig. S6(a)) and medium-sized spherical nanoparticle (Fig. S6(b)) were still present in the sample, and also included large spherical nanoparticles (∼40 nm, Fig. S6(c)), and small spherical nanoparticles (∼5 nm, Fig. S6(d)) in addition to those observed in SNB1k. The lack of Type-E2 spectrum without a major difference in the Type-E3 emission intensity indicates that CsPbCl3 nanocrystal host emission and Eu3+ emission are not necessarily correlated at ensemble level.
 | ||
| Fig. 2 Ensemble absorption and emission spectra (excitation at 369 nm) of (a) SNa14k, (b) SNb12k, (c) SNc1k, and (d) SNc12k. The absorption type (Type-A1, 2) and emission type (Type-E1–3) are indicated. The emission intensities can be qualitatively compared between the samples here and with that in Fig. 1(a) since the measurements were under the same setup. | ||
Further centrifugation (Stage C) yielded samples (SNC1k and SNC12k) with absorption and emission spectra typically observed in CsPbCl3 nanocrystals, namely Type-A1 absorption and Type-E2 emission (Fig. 2(c and d)). Minor or pronounced onsets in the absorption indicate the presence of smaller nanocrystals. The undoped control samples showed similar spectra (Fig. S2(d and e)). The centrifugation speed did not have a significant impact on the absorption and emission spectra or their intensity. Type-E1 or -E3 emission were not observed, which suggests that the nanocrystals may not exhibit any substantial Eu3+ emission. This raises two possibilities: Eu3+ ions were not incorporated into most nanocrystals, and/or Eu3+ ions are not emissive in this host. We further investigate these two possibilities in Section 3.3.
We also measured the absorption and emission spectra of the sample at Stage A (SNA14k, Fig. 2(a)), which would provide insight into the components at the upstream of the purification process. Here, we observed Type-A1 absorption, and Type-E1 and -E3 emission. No Type-E2 emission (CsPbCl3 nanocrystal host emission) was observed, implying low content of CsPbCl3 nanocrystals. We note that the intensity ratio between Type-E1 and -E3 among SNA14k, SNB1k and SNB12k is roughly similar at 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, suggesting that these samples share a common origin as well. Our spectrometer has sufficient spectral resolution to resolve Stark splitting of Eu3+ transitions (see Section 2.2 apparatus and Fig. S7), where the maximum number of Stark lines is 2J + 1 and their relative intensities depend on the crystal field. If emission occurs from Eu3+ ions doped in the Pb2+ sites of CsPbCl3 nanocrystals, then a well-resolved Stark splitting intrinsic to the crystal field of the Pb2+ site should be observed. Thus, 5D0 → 7F1 (≈ 584 nm) can split into up to 3 lines and 5D0 → 7F2 into up to 5 lines. No splitting was observed; instead, the lines were broadened into smooth peaks, indicating a distribution of crystal-field environments (i.e. no single, discrete Eu3+ site).
1, suggesting that these samples share a common origin as well. Our spectrometer has sufficient spectral resolution to resolve Stark splitting of Eu3+ transitions (see Section 2.2 apparatus and Fig. S7), where the maximum number of Stark lines is 2J + 1 and their relative intensities depend on the crystal field. If emission occurs from Eu3+ ions doped in the Pb2+ sites of CsPbCl3 nanocrystals, then a well-resolved Stark splitting intrinsic to the crystal field of the Pb2+ site should be observed. Thus, 5D0 → 7F1 (≈ 584 nm) can split into up to 3 lines and 5D0 → 7F2 into up to 5 lines. No splitting was observed; instead, the lines were broadened into smooth peaks, indicating a distribution of crystal-field environments (i.e. no single, discrete Eu3+ site).
We initially hypothesized that emission spectroscopy with high wavelength resolution could provide a simple, reliable check for impurities and byproducts (or their absence). If Eu3+ ions are successfully doped into Pb2+ sites in the nanocrystals without impurities, then well-resolved Stark splitting should be observed. However, a theoretical study suggested that Eu3+ emission in CsPbCl3 nanocrystals may arise from oxygen-related surface defects, which implies that emission occurs mainly at the surface of the nanocrystals.48 There is also some experimental evidence that suggests Ln3+ ions are primarily doped near the surface.18 Because surface defects tend to be structurally heterogeneous, Eu3+ ions would not experience a single crystal-field environment, leading to broadened, featureless 4f-transition lines. Therefore, the spectral features (or lack thereof) are not necessarily a reliable indicator for the absence of impurities.
To identify the source of Type-E3 emission, we prepared mixtures of OA, OAm, and Eu(NO3)3 in ODE without any Cs or Pb precursors and measured their spectra (Fig. 3). Eu(NO3)3 did not dissolve in ODE by itself and was therefore unmeasurable. OAm + Eu yielded a strongly scattering dispersion with artificially high apparent absorbance (Fig. S8), so its relative emission intensity is unreliable. Type-E3 (Eu3+) emission was observed for OA + Eu and OA + OAm + Eu, suggesting that Eu3+ emission mainly arises from OA-bound Eu3+ complexes. The lack of Stark resolution indicates that these species are also microheterogeneous. The integrated intensity ratio I01:I02 (emission from 5D0 → 7F1versus5D0 → 7F2) depends on the crystal-field symmetry49 and can be used to compare the complexes with the doped samples. After baseline subtraction of the Type-E1 emission, the ratios were ∼1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.1 for SNA14k and ∼1
7.1 for SNA14k and ∼1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6.6 for OA + OAm + Eu mixture, possibly suggesting a common origin. And as previously mentioned, the spectral similarities between SNA14k, SNB1k and SNB12k further suggests that SNB1k and SNB12k also share a common origin with OA + OAm + Eu mixture.
6.6 for OA + OAm + Eu mixture, possibly suggesting a common origin. And as previously mentioned, the spectral similarities between SNA14k, SNB1k and SNB12k further suggests that SNB1k and SNB12k also share a common origin with OA + OAm + Eu mixture.
Type-E1 broad emission was observed in the OAm + Eu and OA + OAm + Eu mixtures as well. Notably, the peak wavelength of this emission shifted depending on the reactant combination: ∼415 nm for OA alone, ∼425 nm for OAm alone; adding Eu(NO3)3 causes a red-shift to ∼430 nm (for OA + Eu) and ∼440 nm (for OA + OAm). Three conclusions follow. First, relative to the ligands alone, Eu(NO3)3 tends to redshift the spectra, indicating chemical interaction between Eu3+ and the ligands. Second, because Type-E3 appears only when OA and Eu salt are both present, Eu3+ emission is likely photosensitized by chemically bound OA. Indeed, previous report on Eu-oleate showed that they are emissive.50 We note that our system differs in regards to the solvent used (ODE) to synthesize them as well as the presence of OAm, which may have added the microheterogeneity owing to their long alkyl chain. Third, OA and OAm interact chemically, producing a much higher emission intensity than either ligand alone. OA and OAm are known to form an ammonium-oleate, and OAm is known to assist OA binding to perovskite surfaces.51,52 Note: a broad blue emission (Type-E1) is also observed in undoped CsPbCl3 samples in Stage-A (Fig. S2(a)), likely due to emissive ligand-related species, consistent with the emissions from OA + OAm. Thus, the spectral changes observed are consistent with these known interactions. We infer that OA + OAm + Eu forms an Eu-complex that reproduces the SNA14k, SNB1k, and SNB12k emission spectra (neglecting host emission), suggesting that Eu3+ emission likely originates from this complex rather than from CsPbCl3 host sensitization. The OA/OAm ligand environment is inherent to the colloidal synthesis and that any Eu not incorporated into the lattice would naturally remain as such complexes in solution. This interpretation may also explain the lower nanocrystal yield compared with undoped syntheses: the doped SNA14k samples showed Type-A1 absorption, whereas undoped SNA14k showed Type-A2 (Fig. S2(a)). Formation of Eu3+ complexes may inhibit CsPbCl3 nanocrystal formation by sequestering ligands during synthesis.
3.3 Photophysical properties at single-particle level
Ensemble data on CsPbCl3:Eu3+ suggested that Eu3+ emission mainly arises from complexes. However, this does not automatically exclude the possibility of Eu3+ emission via CsPbCl3 host sensitization, nor does it address whether Eu3+ is actually doped into the host. As noted in the Introduction, Ln3+ complexes have intrinsically long emission lifetimes (hundreds of µs to ms) compared to typical emitters like perovskite nanocrystals (few to tens of ns), so under continuous excitation emission per unit time (photon flux) in Ln3+ complexes is far lower than that of nanocrystal exciton emitters. In a mixture of Eu3+ complexes and CsPbCl3:Eu3+ nanocrystals, fluorescence microscopy will largely eliminate signals from complexes while directly detecting any emissive CsPbCl3:Eu3+ nanocrystals that are present. This means that single-particle fluorescence provides both spatial isolation of emitters and selective sensitivity to fast emitters (nanocrystals) over slow emitters (complexes).Representative wide-field images and spectra for doped samples in Stage B (SNB1k and SNB12k) and C (SNC1k and SNC12k) are shown in Fig. 4. Samples were dispersed in 2 wt% PMMA films at the same concentration used for solution spectra and excited at 360 nm. Particles were very dilute in SNB1k and SNB12k; often only one particle—or none—was observed per field of view, consistent with the ensemble results. Overall, Eu3+ emission (Type-E3) was not observed, and particles appeared either as large, blurred (non-circular) spots or as diffraction-limited points. The majority of particles showed Type-E2 (host) emission (see Fig. S9–S12 for more examples), even for SNB12k samples where host emission was undetectable at the ensemble level. The appearance of Type-E2 at the single-particle level but not in ensemble spectra likely reflects the very low number of emitting nanocrystals, which kept the ensemble signal below the detection threshold. Conversely, the ensemble Type-E1 band likely arises from a weakly emissive but abundant species—consistent with the OA/OAm salt and Eu3+ complexes.
Many particles also showed a broad and featureless anomalous band from 500–650 nm peaking inhomogeneously at 580–590 nm (Type-E4), irrespective of the observed particle size. Type-E4 signals were generally weak (low S/N) and were accompanied by relatively weak Type-E2 emission (e.g., Fig. 4(a, c, e and g)). In contrast, particles without Type-E4 exhibited intense, high-S/N Type-E2 emission (e.g., Fig. 4(b, d, f and h)). Type-E4 was not observed for undoped CsPbCl3, implying that it is exclusive to doped samples. The fraction of particles exhibiting Type-E4 emission was not markedly different from that of particles without Type-E4; however, because Type-E4 is very weak while Type-E2 is substantially stronger, the Type-E4 component is likely averaged out or buried below the noise floor in ensemble measurements. This observation underscores the utility of single-particle fluorescence microscopy for resolving emissive components that are rare and/or weak yet spatially localized. Three attributions are possible for Type-E4: (1) defect emission28,35 (especially under charge imbalance from Ln3+ incorporation35), (2) a defect-assisted charge-transfer state (CTS, a dopant-centered charge-transfer state, see below),44,46 and (3) Eu2+ emission.53,54 Eu2+ 4f–5d emission in chloride perovskites occurs at much higher energy (bluer) than 580–590 nm; thus, the Eu2+ assignment is less consistent with our observed band position.54 All three scenarios, however, require the presence of Eu in or near the lattice, which—along with the EDS evidence of Eu—indicates that Type-E4 is a consequence of successful Eu incorporation. Furthermore, Type-E1 and Type-E4 are distinct broad emissions: E1 originates from abundant ligand-bound species (appearing in ensemble spectra), whereas E4 is an entirely different, dopant-associated emission that is only discernible at the single-particle level.
Irrespective of whether Type-E4 arises from a defect state or a CTS, the presence of an intermediate level near 580–590 nm (∼17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm−1) suggests that host-sensitized Eu3+ emission is disfavored because the Eu3+ emitting level 5D0 lies at a comparable energy.40 Population reaching 5D0 can undergo rapid nonradiative relaxation (e.g., back-transfer) via this intermediate rather than yield Eu3+ 4f–4f emission.
000 cm−1) suggests that host-sensitized Eu3+ emission is disfavored because the Eu3+ emitting level 5D0 lies at a comparable energy.40 Population reaching 5D0 can undergo rapid nonradiative relaxation (e.g., back-transfer) via this intermediate rather than yield Eu3+ 4f–4f emission.
Under the defect-assisted sensitization model proposed for Yb-doped CsPbCl3 (Fig. 5(a)), the photophysical pathway involves a host-derived defect state feeding a lanthanide-centered charge-transfer state (CTS), i.e. [Ln3+–Cl−–VPb2−–Cl−–Ln3+]− to [Ln2+–Cl−–VPb2−–Cl−–Ln3+], which then populates the dopant's 4f excited manifold.44,46 A key consequence is that the defect level is primarily host-derived, whereas the CTS energy is dopant-dependent (through local structure and the fixed relationship between lanthanides in Ln3+/Ln2+ redox potentials55,56). A Yb-centered CTS is expected at higher energy than the Eu-centered CTS by approximately 6450 cm−1.55,56 If we were to assign Type-E4 emission to defect emission, then (because the defect is host-set) the Eu-centered CTS would have to lie far below ∼17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm−1, i.e., in the near-IR, which has not been reported for any host material up to date. Conversely, assigning the 580–590 nm band to the CTS (which is Eu-linked and therefore dopant-dependent) resolves this conflict. We therefore favor a CTS assignment for Type-E4 emission, which is in agreement with the previous report.41 As illustrated in Fig. 5(b), after the excitation of the CsPbCl3 nanocrystal host, the relaxation process is a charge separation into clustered host-derived defect state (state (1)), and then a relaxation to the CTS state (state (2)) directly or through the 5D0 state of Eu3+ ion (state (3)). Emitter state of Eu3+ ions (5D0 level) lies at ∼17
000 cm−1, i.e., in the near-IR, which has not been reported for any host material up to date. Conversely, assigning the 580–590 nm band to the CTS (which is Eu-linked and therefore dopant-dependent) resolves this conflict. We therefore favor a CTS assignment for Type-E4 emission, which is in agreement with the previous report.41 As illustrated in Fig. 5(b), after the excitation of the CsPbCl3 nanocrystal host, the relaxation process is a charge separation into clustered host-derived defect state (state (1)), and then a relaxation to the CTS state (state (2)) directly or through the 5D0 state of Eu3+ ion (state (3)). Emitter state of Eu3+ ions (5D0 level) lies at ∼17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 cm−1—comparable to the CTS state—and cannot be efficiently populated. Taken together, this analysis underscores why Yb3+ remains viable in CsPbCl3: the Yb3+ 4f excited manifold (2F5/2, ∼10
300 cm−1—comparable to the CTS state—and cannot be efficiently populated. Taken together, this analysis underscores why Yb3+ remains viable in CsPbCl3: the Yb3+ 4f excited manifold (2F5/2, ∼10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 cm−1) lies well below the intermediate, which suppresses back-transfer and agrees with reports of host-sensitized Yb3+ emission in CsPbCl3.16,18,44,57–64
200 cm−1) lies well below the intermediate, which suppresses back-transfer and agrees with reports of host-sensitized Yb3+ emission in CsPbCl3.16,18,44,57–64
 | ||
| Fig. 5 Energy diagram of (a) CsPbCl3:Yb3+ (based on the report by W. Chang et al.44), and (b) CsPbCl3:Eu3+ in this work. The star represents neutrally excited state. The color on the states represents positive charge (blue) and negative charge (red) and their degree of localization. State (1) is a clustered host-derived defect state, state (2) is the CTS, and state (3) is the localized 4f-excited state of the lanthanide. The energy difference between state (2) of CsPbCl3:Yb3+ and CsPbCl3CsPbCl3:Eu3+ is based on the ln3+/ln2+ redox potential and is practically fixed at ∼6450−1 under the same host. The energy difference between state (3) of the two compounds are intrinsically fixed at ∼7100−1 regardless of the host material. | ||
These considerations also point to an engineering route: if the CTS corresponds to a dopant-vacancy cluster (e.g. [Ln3+–Cl−–VPb2−–Cl−–Ln3+]− to [Ln2+–Cl−–VPb2−–Cl−–Ln3+] transition44,57), then strategies that reduce deep vacancy formation and strengthen halide passivation (i.e., using X-type, Cl-donating ligands)65 could destabilize energy or decouple the CTS, potentially making Eu3+ emission more favorable. Overall, lanthanide emission in CsPbCl3 cannot be rationalized by simple energy-gap matching alone; one must account for the existence, energy, and coupling of intermediate states and assess their compatibility with the lanthanide in question.
We cannot determine whether the absence of Eu3+ f–f lines is due to insufficient dopant concentration (below our detection threshold) or an intrinsic inability of CsPbCl3:Eu3+ to sensitize Eu3+ under the mechanism described above. Furthermore, our results pertain to the specific synthetic method we employed; minor to major differences may be present in other systems. Indeed, across reports on CsPbCl3:Eu3+ nanocrystals, one or more of Type-E1, -E2, and -E3 are often missing.15,18,35–38 Consequently, Eu3+ emission requires careful assignment—particularly at the ensemble level—before attributing it to host-sensitized dopants. Reliable ensemble measurements further require verified purification, because impurities and by-products generated during synthesis can obscure the true emission origin.
4. Conclusions
We investigated ensemble and single-particle emission from Eu-treated CsPbCl3 nanocrystals purified at different centrifugation speeds. The main points are:• Conventional hot-injection synthesis of CsPbCl3:Eu3+ yields mixed NCs and molecular/inorganic species; Stage-A centrifugation at 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 RPM (16
000 RPM (16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 RCF) should partition them, yet the supernatant fractions of the Stage-B centrifugation (SNB1k and SNB12k) that are often used in prior reports continued to exhibit molecular-origin emission.
500 RCF) should partition them, yet the supernatant fractions of the Stage-B centrifugation (SNB1k and SNB12k) that are often used in prior reports continued to exhibit molecular-origin emission.
• In ensemble spectra, the dominant source of Eu3+ lines was microheterogeneous Eu–ligand complexes formed with OA/OAm (as evidenced by Eu3+ emission in OA/OAm![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) +
+ ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Eu salt without Cs/Pb).
Eu salt without Cs/Pb).
• Eu3+ ions may be incorporated into the CsPbCl3 host, but an additional broad 500–650 nm band appears (Type-E4) with no accompanying Eu3+ lines, even from individual particles.
• The weak band near 580–590 nm (∼17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm−1) is most consistently interpreted as a dopant-linked intermediate (defect-assisted CTS) lying close to the Eu3+ 5D0 energy and therefore in a strong competition with Eu3+ f–f emission via back-transfer.
000 cm−1) is most consistently interpreted as a dopant-linked intermediate (defect-assisted CTS) lying close to the Eu3+ 5D0 energy and therefore in a strong competition with Eu3+ f–f emission via back-transfer.
We emphasize the distinction between successful doping and dopant-origin emission under typical syntheses. Reliable attribution requires verified purification and corroborating single-particle data—ensemble Eu3+ lines alone are insufficient when Eu–ligand complexes and intermediate states may contribute because both lattice-incorporated Eu3+ and Eu-complex byproducts can produce similar Eu3+ emission lines. More broadly, band-gap alignment by itself is inadequate; one must also evaluate whether defect/CTS intermediates exist, where they lie energetically, and how strongly they couple to the lanthanide levels. Adopting such stringent criteria will sharpen the basis on which lanthanide emission is assigned in perovskite nanocrystal studies going forward.
Author contributions
S. O. and M. V. contributed to the conceptualization of the study. S. O. contributed to the design of the experiments and wrote the manuscript. S. O. and mainly Y. W. contributed to the synthesis, materials measurements, and data analysis. S. O., Y. W., and M. V. contributed to the discussion of the data. All authors contributed to the manuscript revision and read and approved the submitted version.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: Single-particle spectroscopy setup (Fig. S1). Ensemble absorption and emission spectra of non-doped CsPbCl3 nanocrystal samples (Fig. S2). Ensemble absorption and emission spectra of CsPbCl3:Eu3+ nanocrystal samples 11 days after synthesis (Fig. S3). Zoomed images of TEM images of SNB1k (Fig. S4). Spot selected for EDS measurements (Fig. S5). EDS results (Table S1). TEM images of SNB12k (Fig. S6). Emission spectra of a fluorescent lamp measured under the same fluorescence spectrometer (Fig. S7). Ensemble absorption spectra of reactant combination mixture without Pb and Cs source (Fig. S8). More representative fluorescence microscope images of the particles (Fig. S9–S12). See DOI: https://doi.org/10.1039/d6cp00181e.Acknowledgements
The research was financially supported by the JSPS KAKENHI grant number 23K13629, “Grant-in-Aid for Early-Career Scientists”, 24K01449, “Grant-in-Aid for Scientific Research (B)”, and by The SATOMI Scholarship Foundation “Satomi Research Proposal Award”. We also thank Dr Akira Genseki of Core Facility Center in Institute of Science Tokyo for the TEM measurements.References
- J.-C. G. Bünzli, Coord. Chem. Rev., 2015, 293–294, 19–47 CrossRef.
- J. Kido and Y. Okamoto, Chem. Rev., 2002, 102, 2357–2368 CrossRef CAS PubMed.
- A. J. Amoroso and S. J. Pope, Chem. Soc. Rev., 2015, 44, 4723–4742 RSC.
- W. Zheng, P. Huang, D. Tu, E. Ma, H. Zhu and X. Chen, Chem. Soc. Rev., 2015, 44, 1379–1415 RSC.
- T. Jüstel, H. Nikol and C. Ronda, Angew. Chem., Int. Ed., 1998, 37, 3084–3103 CrossRef.
- V. Pankratov, A. I. Popov, S. A. Chernov, A. Zharkouskaya and C. Feldmann, Phys. Status Solidi B, 2010, 247, 2252–2257 CrossRef CAS.
- V. Dubey, J. Kaur and S. Agrawal, Res. Chem. Intermed., 2014, 41, 4727–4739 CrossRef.
- J. C. Bünzli and C. Piguet, Chem. Soc. Rev., 2005, 34, 1048–1077 RSC.
- A. Tew, L. van Turnhout, Y. Deng, R. Arul, J. Ye, T. Liu, Z. Jiang, L. Dai, H. Zhu, Y. Zhang, A. Rao and Z. Yu, Appl. Phys. Rev, 2024, 11, 021329 CAS.
- B. Julián, J. Planelles, E. Cordoncillo, P. Escribano, P. Aschehoug, C. Sanchez, B. Viana and F. Pellé, J. Mater. Chem., 2006, 16, 4612–4618 RSC.
- R. Martin-Rodriguez, R. Geitenbeek and A. Meijerink, J. Am. Chem. Soc., 2013, 135, 13668–13671 CrossRef CAS PubMed.
- Z. Chen, Y. Li and L. Wu, Nanoscale Adv., 2023, 5, 1397–1404 RSC.
- J. Planelles-Aragó, B. Julián-López, E. Cordoncillo, P. Escribano, F. Pellé, B. Viana and C. Sanchez, J. Mater. Chem., 2008, 18, 5193–5199 RSC.
- Y. Wang, X. Liang, E. Liu, X. Hu and J. Fan, Nanotechnol., 2015, 26, 375601 CrossRef PubMed.
- Q. Li, Y. Liu, P. Chen, J. Hou, Y. Sun, G. Zhao, N. Zhang, J. Zou, J. Xu, Y. Fang and N. Dai, J. Phys. Chem. C, 2018, 122, 29044–29050 CrossRef CAS.
- T. J. Milstein, D. M. Kroupa and D. R. Gamelin, Nano Lett., 2018, 18, 3792–3799 CrossRef CAS PubMed.
- R. Sun, P. Lu, D. Zhou, W. Xu, N. Ding, H. Shao, Y. Zhang, D. Li, N. Wang, X. Zhuang, B. Dong, X. Bai and H. Song, ACS Energy Lett., 2020, 5, 2131–2139 CrossRef CAS.
- G. Pan, X. Bai, D. Yang, X. Chen, P. Jing, S. Qu, L. Zhang, D. Zhou, J. Zhu, W. Xu, B. Dong and H. Song, Nano Lett., 2017, 17, 8005–8011 CrossRef CAS PubMed.
- D. Zhou, D. Liu, G. Pan, X. Chen, D. Li, W. Xu, X. Bai and H. Song, Adv. Mater., 2017, 29, 1704149 Search PubMed.
- J. Duan, Y. Zhao, X. Yang, Y. Wang, B. He and Q. Tang, Adv. Energy Mater., 2018, 8, 1802346 CrossRef.
- X. Zhang, Q. Zhu, B. Chen, S. Wang, A. L. Rogach and F. Wang, Adv. Photonics Res., 2020, 2, 2000089 CrossRef.
- Q. Xiao, Y. Liu, L. Liu, R. Li, W. Luo and X. Chen, J. Phys. Chem. C, 2010, 114, 9314–9321 CrossRef CAS.
- J. Kong, H. Zhu, R. Li, W. Luo and X. Chen, Opt. Lett., 2009, 34, 1873–1875 Search PubMed.
- H. Zhang, Z.-H. Chen, X. Liu and F. Zhang, Nano Res., 2020, 13, 1795–1809 CrossRef CAS.
- G. H. Debnath, P. Mukherjee and D. H. Waldeck, J. Phys. Chem. C, 2020, 124, 26495–26517 CrossRef CAS.
- R. Marin and D. Jaque, Chem. Rev., 2021, 121, 1425–1462 Search PubMed.
- Y. Xie, B. Peng, I. Bravic, Y. Yu, Y. Dong, R. Liang, Q. Ou, B. Monserrat and S. Zhang, Adv. Sci., 2020, 7, 2001698 Search PubMed.
- J. Y. D. Roh, M. D. Smith, M. J. Crane, D. Biner, T. J. Milstein, K. W. Krämer and D. R. Gamelin, Phys. Rev. Mater., 2020, 4, 105405 CrossRef CAS.
- J.-P. Ma, Y.-M. Chen, L.-M. Zhang, S.-Q. Guo, J.-D. Liu, H. Li, B.-J. Ye, Z.-Y. Li, Y. Zhou, B.-B. Zhang, O. M. Bakr, J.-Y. Zhang and H.-T. Sun, J. Mater. Chem. C, 2019, 7, 3037–3048 RSC.
- W. J. Mir, T. Sheikh, H. Arfin, Z. Xia and A. Nag, NPG Asia Mater., 2020, 12, 9 CrossRef CAS.
- W. Xu, J. Liu, B. Dong, J. Huang, H. Shi, X. Xue and M. Liu, Sci. Adv., 2023, 9, eadi7931 CrossRef CAS PubMed.
- D. J. Gargas, E. M. Chan, A. D. Ostrowski, S. Aloni, M. V. Altoe, E. S. Barnard, B. Sanii, J. J. Urban, D. J. Milliron, B. E. Cohen and P. J. Schuck, Nat. Nanotechnol., 2014, 9, 300–305 CrossRef CAS PubMed.
- A. D. Ostrowski, E. M. Chan, D. J. Gargas, E. M. Katz, G. Han, P. J. Schuck, D. J. Milliron and B. E. Cohen, ACS Nano, 2012, 6, 2686–2692 CrossRef CAS PubMed.
- J. A. O. Galindo, A. R. Pessoa, A. M. Amaral and L. D. S. Menezes, Nanoscale Adv., 2021, 3, 6231–6241 RSC.
- R. Sun, D. Zhou, Y. Ding, Y. Wang, Y. Wang, X. Zhuang, S. Liu, N. Ding, T. Wang, W. Xu and H. Song, Light: Sci. Appl., 2022, 11, 340 Search PubMed.
- X. Jing, D. Zhou, R. Sun, Y. Zhang, Y. Li, X. Li, Q. Li, H. Song and B. Liu, Adv. Funct. Mater., 2021, 31, 2100930 CrossRef CAS.
- W. Wang, S. Song, P. Lv, J. Li, B. Cao and Z. Liu, J. Lumin., 2023, 260, 119894 Search PubMed.
- G. H. Debnath, B. P. Bloom, S. Tan and D. H. Waldeck, Nanoscale, 2022, 14, 6037–6051 Search PubMed.
- L. Protesescu, S. Yakunin, M. I. Bodnarchuk, F. Krieg, R. Caputo, C. H. Hendon, R. X. Yang, A. Walsh and M. V. Kovalenko, Nano Lett., 2015, 15, 3692–3696 CrossRef CAS PubMed.
- K. Binnemans, R. Van Deun, C. Görller-Walrand and J. L. Adam, J. Non-Cryst. Solids, 1998, 238, 11–29 CrossRef CAS.
- S. Miyazaki, M. Gotanda, Y. Kitagawa, Y. Hasegawa, K. Miyata and K. Onda, J. Phys. Chem. Lett., 2024, 15, 10718–10724 CrossRef CAS PubMed.
- J. Sun, H. Fu, H. Jing, X. Hu, D. Chen, F. Li, Y. Liu, X. Qin and W. Huang, Adv. Mater., 2025, 37, e2417397 Search PubMed.
- Y. Liu, G. Pan, R. Wang, H. Shao, H. Wang, W. Xu, H. Cui and H. Song, Nanoscale, 2018, 10, 14067–14072 Search PubMed.
- W. J. Chang, S. Irgen-Gioro, S. Padgaonkar, R. López-Arteaga and E. A. Weiss, J. Phys. Chem. C, 2021, 125, 25634–25642 Search PubMed.
- W. Wang, S. Song, B. Cao and J. Li, J. Lumin., 2022, 247, 118901 CrossRef CAS.
- A. Shukla, G. Kaur, K. J. Babu, A. Kaur, D. K. Yadav and H. N. Ghosh, J. Phys. Chem. Lett., 2022, 13, 83–90 CrossRef CAS PubMed.
- S. Mannar, P. Mandal, A. Roy and R. Viswanatha, J. Phys. Chem. Lett., 2022, 13, 6290–6297 CrossRef CAS PubMed.
- A. Bala and V. Kumar, ACS Appl. Nano Mater., 2020, 3, 4437–4444 CrossRef CAS.
- P. A. Tanner, Chem. Soc. Rev., 2013, 42, 5090–5101 RSC.
- G. Liu, C. E. Conn and C. J. Drummond, J. Phys. Chem. B, 2009, 113, 15949–15959 CrossRef CAS PubMed.
- G. Almeida, L. Goldoni, Q. Akkerman, Z. Dang, A. H. Khan, S. Marras, I. Moreels and L. Manna, ACS Nano, 2018, 12, 1704–1711 CrossRef CAS PubMed.
- J. De Roo, M. Ibanez, P. Geiregat, G. Nedelcu, W. Walravens, J. Maes, J. C. Martins, I. Van Driessche, M. V. Kovalenko and Z. Hens, ACS Nano, 2016, 10, 2071–2081 CrossRef CAS PubMed.
- H. Yuan, S. Jin, S. Zheng, R. Xie, T. Pang, B. Zhuang, F. Huang and D. Chen, J. Mater. Chem. C, 2024, 12, 1430–1439 RSC.
- F. Alam, K. D. Wegner, S. Pouget, L. Amidani, K. Kvashnina, D. Aldakov and P. Reiss, J. Chem. Phys., 2019, 151, 231101 CrossRef PubMed.
- P. Dorenbos, J. Electrochem. Soc., 2005, 152, H107 CrossRef CAS.
- P. Dorenbos, J. Lumin., 2007, 122–123, 315–317 CrossRef CAS.
- X. Li, S. Duan, H. Liu, G. Chen, Y. Luo and H. Agren, J. Phys. Chem. Lett., 2019, 10, 487–492 CrossRef CAS PubMed.
- X. Zhang, Y. Zhang, X. Zhang, W. Yin, Y. Wang, H. Wang, M. Lu, Z. Li, Z. Gu and W. W. Yu, J. Mater. Chem. C, 2018, 6, 10101–10105 RSC.
- T. Cai, J. Wang, W. Li, K. Hills-Kimball, H. Yang, Y. Nagaoka, Y. Yuan, R. Zia and O. Chen, Adv. Sci., 2020, 7, 2001317 CrossRef CAS PubMed.
- W. J. Mir, Y. Mahor, A. Lohar, M. Jagadeeswararao, S. Das, S. Mahamuni and A. Nag, Chem. Mater., 2018, 30, 8170–8178 CrossRef CAS.
- M. A. Padhiar, S. Zhang, M. Wang, N. Zamin Khan, S. Iqbal, Y. Ji, N. Muhammad, S. A. Khan and S. Pan, Nanomaterials, 2023, 13, 2703 CrossRef CAS PubMed.
- X. Luo, T. Ding, X. Liu, Y. Liu and K. Wu, Nano Lett., 2019, 19, 338–341 CrossRef CAS PubMed.
- D. Li and G. Chen, J. Phys. Chem. Lett., 2023, 14, 2837–2844 CrossRef CAS PubMed.
- S. M. Loh, Y. Jing, T. C. Sum, A. Bruno, S. G. Mhaisalkar and S. A. Blundell, J. Phys. Chem. Lett., 2025, 16, 2295–2300 CrossRef CAS PubMed.
- N. C. Anderson, M. P. Hendricks, J. J. Choi and J. S. Owen, J. Am. Chem. Soc., 2013, 135, 18536–18548 CrossRef CAS PubMed.
| This journal is © the Owner Societies 2026 |