Open Access Article
Open Access ArticleInvestigating trends in actinide covalency and magnetism with 35/37Cl SSNMR spectroscopy and first-principles calculations
Adam R. Altenhof†
 *a,
Karla A. Erickson
*a,
Karla A. Erickson b,
Daniel A. Rehn
b,
Daniel A. Rehn c,
Taylor V. Fetrowd,
Ann E. Mattssonc,
Marisa J. Monreale and
Harris E. Mason
c,
Taylor V. Fetrowd,
Ann E. Mattssonc,
Marisa J. Monreale and
Harris E. Mason *e
*e
aMPA-Q, Los Alamos National Laboratory, Los Alamos, NM 87545, USA. E-mail: adam.altenhof@eaps.ethz.ch
bC-CDE, Los Alamos National Laboratory, Los Alamos, NM 87545, USA
cXCP-5, Los Alamos National Laboratory, Los Alamos, NM 87545, USA
dSigma-1, Los Alamos National Laboratory, Los Alamos, NM 87545, USA
eC-IIAC, Los Alamos National Laboratory, Los Alamos, NM 87545, USA. E-mail: hmason@lanl.gov
First published on 31st March 2026
Abstract
Solid-state NMR (SSNMR) spectroscopy is a powerful technique for studying actinide chemistry but has been significantly limited due to the complex paramagnetism, and radiological hazards presented by these materials. Lanthanide and actinide salts often feature magnetic ordering and can be paramagnetic, ferromagnetic, or antiferromagnetic depending on temperature and electronic structure. Paramagnetic interactions can manifest in SSNMR both as secular spectral shifts and/or couplings as well as contributions from non-secular relaxation. Both effects can be directly measured with NMR and used to extrapolate rich chemical information such as coordination environments, bonding characteristics, local molecular dynamics, and correlation times. Typically, these studies are carried out on high-γ and highly abundant NMR-active isotopes (e.g., 1H, 6/7Li, 19F, 23Na, etc.) or on enriched rare isotopes (e.g., 2H and 17O), which can be expensive. Herein, we present a facile methodology to measure the 35/37Cl electric-field gradient (EFG) and paramagnetic shift anisotropy (SA) tensor components using static wideline SSNMR measurements of LaCl3, NdCl3, UCl3, and UCl4. The static powder spectra were measured with both 35Cl and 37Cl SSNMR to increase the fidelity of the extracted tensor parameters. Variable temperature NMR of a select case confirms the Curie–Weiss paramagnetism. Relaxation measurements of both nuclei further corroborate observations owing to the paramagnetic relaxation enhancement and reveal simultaneous quadrupolar relaxation mechanisms. Density functional theory (DFT) calculations using Hubbard U corrections to the uranium valence orbitals show excellent agreement with experimental EFG tensor parameters and help describe the bonding characteristics in these lanthanide and actinide systems.
1. Introduction
Actinide salts are a crucial component of molten-salt reactors (MSRs) where chloride salts are used as fuel sources for fissile isotopes (e.g., 235U). The preparation of actinide chloride salts for MSRs currently lacks reliable and quantitative characterization techniques.1–3 This limitation is largely due to (i) the hazards presented by radioactive actinide materials and (ii) the complicated physics of d and f electron orbitals involved in bonding that are reflected in available spectroscopies. The larger radial extent of d and f orbitals in actinides compared to the lanthanides has also been suggested to allow greater electron sharing with the valence electronic orbitals of ligands. Extensive studies combining X-ray based spectroscopies and ab initio modeling have explored these interactions, but direct modeling of these spectra is complex and open to interpretation.4–8 NMR spectroscopy is one of the best available techniques for routine characterization and can provide information on local and longer-range structure and provides a direct measurement of the local electron density calculated by density functional theory (DFT). The measured NMR parameters are highly correlated to coordination environments and local bonding phenomena and can provide an insight to the complex valence systems of d and f orbitals in lanthanides and actinides.Paramagnetic solid-state NMR (SSNMR) spectroscopy is a growing field that can provide rich information about coordination environments, bonding characteristics, and local molecular dynamics. Paramagnetism manifests in NMR due to the coupling of unpaired electron spins to nuclear spins, referred to as the hyperfine coupling interaction as described by the hyperfine tensor, A, with the characteristic coupling constant, A. The interaction consists of an isotropic Fermi-contact component, Aiso, which causes isotropic shifts in the resonance frequency of NMR spins, and a pseudo-contact anisotropic electron-nuclear spin dipolar coupling component, AD, which causes inhomogeneous broadening in these resonances. Both of these components can experience further inhomogeneous broadening when bulk magnetic susceptibility (BMS) effects are present.9 The shifts and inhomogeneous broadening in the resulting NMR spectra can be quantified by the hyperfine tensor parameters, which are often summarized with shift anisotropy (SA) tensor parameters that reflect the sum of diamagnetic and paramagnetic contributions.9–15 Pell et al. have provided a comprehensive overview of solution- and solid-state paramagnetic NMR spectra with many case studies.16 Most of these studies are carried out with favorable NMR handles, i.e., spin-1/2 and highly abundant nuclei, where studies with quadrupolar (spin > 1/2) NMR handles are less common by comparison. Furthermore, there are many SSNMR studies of lanthanide materials but only limited reports for actinide materials.17–30 Part of the limitation in the actinide studies is the hazard presented by radioactivity.31
Over the past 20 years there has been an increasing interest in the use of NMR-active quadrupolar halogens as SSNMR handles (i.e., 35/37Cl, 79/81Br, and 127I). They are extremely sensitive to local structural changes and dynamics, as reflected in their measured electric-field gradient (EFG) tensor parameters. Of these handles, 35Cl is by far the most accessible for SSNMR studies. Although it has a low gyromagnetic ratio, it has a high natural abundance (75.78%) and a significantly smaller quadrupole moment, Q, than 79/81Br or 127I, causing the powder spectra to span reasonable breadths at high magnetic field strengths. 35/37Cl SSNMR studies have been carried out on a plethora of organic and inorganic materials as highlighted in several review articles by Bryce and coworkers.32–35 Methodologies for studying 35/37Cl and similar NMR handles are continuing to improve with advances in static wideline and ultra-wideline NMR,36–44 fast-MAS and indirect-detection techniques,45,46 multiple-quantum approaches under MAS,47–49 among others.50 There have been previous 35Cl SSNMR studies of single crystal metal chloride salts with reported EFG and hyperfine tensor parameters, where the metals include paramagnetic Ni2+, Cu2+, Mn2+, Co2+, and Fe2+ (depending on temperature).51–55 However, there have been comparatively few reports of 35Cl SSNMR of crystalline powder materials that are paramagnetic,56–58 with the exception of numerous perchlorate and chlorate samples.33 Bauder and Wu recently reported 35/37Cl SSNMR of paramagnetic Co2+ chlorides and demonstrated substantial shifts observed in 35/37Cl spectra.59 Recently there have also been nuclear quadrupole resonance (NQR) studies of uranium complexes using 85/87Rb and 35Cl as spectroscopic handles, where only the influence of the EFG tensors can be measured in those cases.60,61 There have been considerable efforts in the analysis of 2H SSNMR as simultaneously influenced by the first-order quadrupolar interaction (FOQI) and SA primarily using modified echo and Car-Purcell/Meiboom-Gill (CPMG) type pulse sequences.62–69 The only comparable approaches of separating second-order quadrupolar interactions (SOQI) and SA interactions in half-integer spin nuclei are with the COASTER pulse sequence from Grandinetti et al., which needs to be performed under MAS with a rotor angle of 70.12°,70 and the TOP-STMAS/MQMAS method from Carvalho et al., which also requires MAS.71
When studying half-integer-spin quadrupolar nuclei such as 35/37Cl (I = 3/2), researchers often focus on the acquisition of the central transition (CT) powder pattern, which spans a far narrower frequency breadth comparted to the satellite transitions (STs). CT powder patterns can be simultaneously influenced by the SOQI, SA interactions, and/or dipole–dipole interactions; herein, we focus on simultaneous SOQI and shielding interactions. The complicated orientation dependence of the SOQI convolved with the effects of SA results in static powder spectra that are difficult to analyze and deconvolve their individual contributions.72 The problem in these cases is that eight parameters are needed to accurately model the EFG and SA tensor parameters (two for the EFG, three for the SA, and three Euler angles relating the orientations of the two tensors). To accurately determine all eight parameters, multiple NMR spectra are required. Typically, a pattern should be recorded under MAS, in which the manifestation of the SA parameters is averaged, and a static pattern should be measured. Then both should be acquired at a second magnetic field such that the manifestation of the EFG and SA parameters will change. One problem with this strategy is for cases where the inhomogeneous broadening (from EFG, SA, or both) is too large for sufficient averaging of under MAS, where complex spinning sideband manifolds can hamper analysis. Furthermore, access to a second magnetic field is not a ubiquitous option for researchers and in the current work, radioactive samples are measured where limited exposure to multiple pieces of equipment and spectrometers is desirable.
Instead, the use of 37Cl NMR as a compliment to 35Cl measurements can mimic the effects of measuring 35Cl at a second higher-magnetic field. A higher field will cause a decrease in pattern broadening from the SOQI (with a νQ2/B0 dependence, where νQ is the quadrupolar frequency) and a linear increase in broadening from magnetic shielding (linear B0 dependence). 37Cl will experience a smaller CQ as compared to 35Cl (as scaled by the ratio of their quadrupolar moments, Q(37Cl)/Q(35Cl) = 0.78812) and the same ηQ resulting in less inhomogeneous broadening from the SOQI and will be influenced by nearly identical SA tensor parameters.73–75 Using 35/37Cl together can provide a much simpler experimental strategy for determining multiple tensor parameters since the Larmor frequencies are close together (i.e., a single broadband probe can measure both) and limit exposure for radioactive materials.
Herein, we discuss the use of 35/37Cl SSNMR to study lanthanide and actinide chloride salts. The central strategy for this study is to use static 35Cl SSNMR measurements to establish the EFG and paramagnetic SA tensor parameters and then confirm them with 37Cl SSNMR measurements. In every case 1D spectra, T1's, and Teff2's are measured and compared between the compounds studied. We also calculate local molecular orbitals (LMOs) and bond order parameters from periodic DFT calculations to help constrain the origins of the SA and EFG tensors to provide insight into the relative covalency of the metal–chloride bonds.
2. Methods
2.1. Samples
Lanthanum trichloride [Strem Chemicals Inc., LaCl3] and neodymium trichloride [Strem Chemicals Inc, NdCl3] were purchased. Depleted uranium trichloride [UCl3]76 and uranium tetrachloride [UCl4]2,3,77 were prepared according to literature procedures. Radioactive UCl3 and UCl4 samples were transferred into polychlorotrifluoroethylene (PCTFE) inserts within 4 mm zirconia NMR rotors and non-radioactive samples were packed directly into the rotor sleeves; therefore, measurements of radioactive samples contain less material. Sample purity was confirmed by powder Xray diffraction (XRD) patterns (see SI).2.2. SSNMR Spectroscopy
SSNMR spectra were acquired using a Bruker Avance NEO console and a 9.4 T (υ0(1H) = 399.94 MHz) Bruker wide-bore magnet at resonance frequencies of υ0(35Cl) = 39.186 MHz and υ0(37Cl) = 32.618 MHz. Two Bruker broadband 4 mm HX magic-angle spinning (MAS) probes were used under static conditions, where one was used for radioactive samples and the other was used for non-radioactive samples to limit the potential for equipment contamination. 35/37Cl RF pulse powers and chemical-shift reference frequencies were calibrated using NaCl (s) with δiso = 0.0 ppm. All spectra were recorded with the WURST-CPMG pulse sequence with varied parameters (see SI).36,37,78–80 The temperature reading for variable temperature (VT) experiments was calibrated using the 207Pb SSNMR of PbNO3.81 All other measurements were made under ambient conditions around 20 °C. 35/37Cl effective T2's (Teff2) were measured using the WURST-CPMG time-domain data and T1's were measured with a WURST-CPMG/Saturation-Recovery (WCPMG-SR) type pulse sequence using 16 relaxation delays.822.3. Processing and simulations
All data were processed with Python using DESPERATE and NMRGLUE.83,84 WURST-CPMG echo trains were coadded into a single echo, multiplied by a Gaussian function, Fourier transformed, then phase corrected with zeroth, first, and second-order phasing85 in every case. Subsequent simulations and fitting were conducted in ssNake version 1.3.86 Additional SIMPSON simulations are presented in the SI.87,882.4. Density functional theory
Plane wave DFT calculations were carried out using the Vienna Ab initio Simulation Package (VASP) version 5.4.4.89–92 The Revised Perdew–Burke–Ernzerhof (PBE) functional93 with both the Hubbard U correction (DFT+U) was used for calculations of the paramagnetic NdCl3, UCl3, and UCl4 materials. The rotationally invariant method of Dudarev et al. was used where Ueff = U − J,94 where J is the on-site exchange interaction and in all cases was set to 0 eV. These materials were calculated in the ferromagnetic state and scaled to the paramagnetic regime using a Curie–Weiss model as described in Kim et al.95 The optimal Ueff value was obtained by running calculations for different Ueff values and determining the best match to the measured EFG tensors (Fig. S9–S12). Calculations were also performed including spin–orbit coupling but provided qualitatively inaccurate results for NdCl3 and had minor effects on those for UCl3 and UCl4 (Table S3). Thus, all calculations reported in the main text were performed in the absence of spin–orbit coupling for consistency. All calculations were performed on the unrelaxed XRD structures drawn from the literature. A plane-wave kinetic energy cutoff of 500 eV and convergence energy difference of 10−8 eV were used for EFG and chemical shift calculations. The local orbital basis suite towards electronic-structure reconstruction (LOBSTER) software package was used to perform crystal orbital Hamiltonian population (COHP), crystal bond order index (COBI), and reconstructed local molecular orbital (RLMO) analyses.96–98 The bond-weighted values of the COHP and COBI are reported as the integrated crystal Hamiltonian population (ICOHP) and integrated crystal bond order index (ICOBI).3. Results and discussion
3.1. Overview
In the following sections, 35Cl and 37Cl SSNMR measurements are conducted on LaCl3, NdCl3, UCl3, and UCl4, which all feature a single chlorine site. Diamagnetic LaCl3 is used as a control, since it is expected to exhibit CT powder spectra only influenced by the SOQI, which will serve to baseline the EFG tensor parameters for structurally similar analogues. All subsequent samples are paramagnetic at room temperature and are expected to be influenced by paramagnetic SA interactions. 35Cl spectra are used to establish both the EFG and SA parameters using fits in ssNake and 37Cl spectra are used to confirm the fitted parameters. Variable temperature (VT) 35Cl NMR is used to verify the paramagnetic SA parameters in a select case for NdCl3. In every case the 35/37Cl T1's and Teff2's are measured and compared. DFT calculations help constrain the fitted EFG and SA tensor parameters and provide insight to the type of bonding in these lanthanide and actinide chloride compounds.3.2. 35/37Cl SSNMR
35Cl SSNMR was conducted on all samples outlined above (Fig. 1). The first compound considered is LaCl3 (Fig. 1a), which features a single chlorine site that is in a 9-coordinate geometry around La3+.99 The static powder pattern is fit in ssNake where the EFG and SA tensor parameters are extracted. The fitted parameters for this and all compounds are summarized in Table 1, where LaCl3 has CQ = 7.91 MHz, ηQ = 0.49, δiso = 302.3 ppm, and all other SA parameters are negligible. There is no discernable influence of SA on the static powder pattern within the precision of our measurements. The δiso reported is well within the range of diamagnetic shielding that is often reported for 35Cl SSNMR of metal complexes and iconic compounds.32–34 These tensor parameters and the visual appearance of the spectrum and its discontinuities are highly commensurate with a typical static SOQI CT powder pattern, which is consistent with the material being diamagnetic.| Compound | Ueff (eV) | CQ (35Cl) (MHz) | ηQ | δiso (ppm) | Ω (ppm) | κ (ppm) | α (°) | β (°) | γ (°) | ICOHP (eV) | ICOBId | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a The principal components of the EFG tensors are ranked |V33| ≥ |V22| ≥ |V11|. The quadrupolar coupling constant and asymmetry parameter are given by CQ = eQV33/h, and ηQ = (V11 − V22)/V33, respectively. The sign of CQ cannot be determined from the experimental 35/37Cl spectra.b The principal components of the SA tensors are defined such that δ11 ≥ δ22 ≥ δ33. The isotropic shift, span, and skew are given by δiso = (δ11 + δ22 + δ33)/3, Ω = δ11 − δ33, and κ = 3(δ22 − δiso)/Ω, respectively. Calculated SA parameters use the full A tensor according to Pigliapochi et al.115c The Euler angles α, β, and γ define the relative orientation of the EFG and SA tensors. Euler angles are reported using the ZX′Z″ convention.129,130d The ICOHP and ICOBI values represent bond-weighted averages. | ||||||||||||
| LaCl3 | Exp. | 7.91 | 0.49 | 302.3 | 0 | 0.00 | 0.0 | 0.0 | 0.0 | |||
| DFT | 4.0 | 7.87 | 0.53 | 731 | −1.13 | 0.19 | ||||||
| NdCl3 | Exp. | 8.99 | 0.48 | 758.5 | 2004 | 0.52 | 9.2 | 4.5 | 0.0 | |||
| DFT | 1.0 | 8.98 | 0.49 | 1738 | 2549 | 1.00 | −1.17 | 0.20 | ||||
| UCl3 | Exp. | 8.68 | 0.52 | 1014.0 | 2255 | 1.00 | 20.4 | 2.0 | 0.0 | |||
| DFT | 6.0 | 9.25 | 0.58 | 1071 | 2038 | 0.99 | −1.10 | 0.19 | ||||
| UCl4 | Exp. | 12.00 | 0.35 | 600.0 | 5200 | −0.01 | 0.0 | 8.5 | 0.0 | |||
| DFT | 5.0 | −11.99 | 0.31 | −393 | 3283 | −0.43 | −1.65 | 0.29 | ||||
NdCl3 was then investigated, which is structurally similar to LaCl3 since both compounds occupy a P63/m space group, are both 9-coordinate to the metal center, and have very similar M–Cl bond distances (2.95 and 2.97 Å for LaCl3; 2.90 and 2.95 Å for NdCl3).99 The main difference is the unpaired 4f electrons of Nd3+. The resulting 35Cl pattern (Fig. 1b) is significantly different from that of LaCl3 where similar EFG parameters are found with CQ = 8.99 MHz, ηQ = 0.48, but significant SA parameters are necessary to fit the spectrum with δiso = 785.5 ppm, Ω = 2004 ppm, and κ = 0.52. For comparison, the pattern was fitted without SA parameters, which causes a significant mismatch in the central discontinuity and the broadening of the “horn” features in the powder pattern (Fig. S5). This δiso and Ω are amongst the largest reported for 35Cl NMR and are unlikely to arise from diamagnetic shielding. Since NdCl3 is paramagnetic, it is very likely that the SA tensor parameters are primarily from the manifestation of paramagnetic shielding, especially as compared to the diamagnetic LaCl3, which is almost identical in structure.
UCl3 is also structurally similar to LaCl3 and NdCl3 (i.e., they have the same space group, coordination environment, and similar bond distances)100 but like NdCl3, U3+ features unpaired f electrons (5f in this case) and is paramagnetic at room temperature. The 35Cl pattern of UCl3 (Fig. 1c) is visually much more similar to that of NdCl3 rather than LaCl3 and has similar EFG tensor parameters to both with CQ = 8.68 MHz and ηQ = 0.52. The SA parameters are significantly larger than both LaCl3 and NdCl3 with δiso = 1014 ppm, Ω = 2255 ppm, and κ = 1.00. UCl3 has a slightly higher paramagnetic susceptibility value than NdCl3, which could be one source for the larger SA tensor components, and it is structurally very similar to NdCl3, which is reflected in the EFG tensor parameters.
UCl4 is structurally unique from the trichloride compounds analyzed above, occupying an l41/amd space group and having an 8-coordinate geometry around U4+.101 The 35Cl powder pattern (Fig. 1d) is substantially broader than the others, owing to both the EFG parameters of CQ = 12.0 MHz and ηQ = 0.35 and the SA parameters with δiso = 600 ppm, Ω = 5200 ppm, and κ = –0.01. The larger CQ and substantial Ω cause the large frequency dispersion in this case. The δiso is less than that of NdCl3 and UCl3, but still much larger that of LaCl3, which matches the relative magnetic susceptibility between all the compounds (vide infra).
As mentioned earlier, it is difficult to isolate the EFG and SA parameters simultaneously using only one spectrum at one field and only acquired under static conditions as there are eight free variables involved in the fit. The 35Cl powder patterns presented herein are far too broad to garner useful information from MAS experiments at moderate speeds and may still be difficult with ultrafast MAS (Fig. S6). The issue of using MAS would not be alleviated at separate magnetic fields, since the net pattern breadth will increase at lower fields, marginally decrease at some higher fields, and increase at significantly higher fields (Fig. S7). Static measurements at a second magnetic field would be a useful check and constraint on the parameters; however, our laboratory (like many others) is equipped with only a single SSNMR system, and even with a second system, additional equipment would be exposed to radiological hazards. Therefore, using the 37Cl isotope as a handle for a second reference for the tensor parameters is an ideal alternative for simplicity and safety. Any shift differences experienced by the nuclei should be far less than the precision of our measurements.74
The 37Cl NMR spectra were acquired for all the compounds described above (Fig. 1e–h). In every case, the tensor parameters from the fits of the 35Cl data were used to simulate the 37Cl spectra (expect for the CQ, which was scaled accordingly). In each case, the simulated spectra match the experimental well. The main drawbacks of 37Cl are the slightly lower gyromagnetic ratio and its natural abundance being about one third of 35Cl causing a lower receptivity; therefore, the experimental 37Cl are slightly lower in signal-to-noise ratios and in some cases require slightly more Gaussian broadening. Nonetheless, the main discontinuities between all spectra align well, with the exception of the UCl4 pattern (Fig. 1h). This pattern appears to experience a bandpass-type depletion of signal, especially to the low-frequency part of the pattern, making it harder to clearly resolve the lower frequency horn (and its splitting) as well as the low frequency pattern edge, but is still discernable. There is also an anomalous feature in both 35/37Cl patterns of UCl3 where the high-frequency edge shows a peak-like feature (Fig. 1c and g). This may be due to T1 or T2 anisotropy, which would require much more sensitive measurements to confirm.82,102 It is important to note that 37Cl spectra are not used for fitting but serve as a check against the fitted parameters from the 35Cl data, increasing our confidence in both the EFG and SA tensor parameters across all compounds.
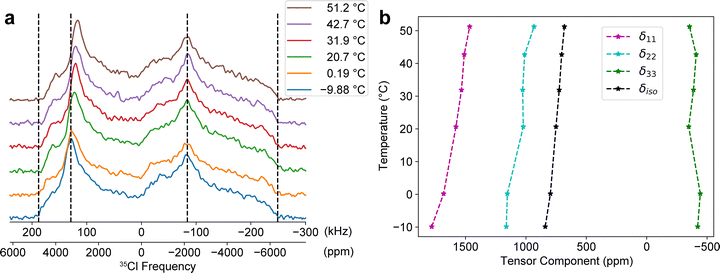
Having established that chloride compounds containing elements with non-negligible paramagnetic susceptibilities cause the onset of the effects of SA interactions, it is important to rule out that nuclear magnetic shielding (i.e., diamagnetic shift or chemical shift) is causing these effects. The paramagnetic susceptibility and therefore the SA tensor parameters should exhibit a 1/(T − Θ) temperature dependance, provided the temperature exceeds the Weiss constant, Θ.9,16 The VT 35Cl SSNMR experiments were carried out for NdCl3 since its lack of radioactivity allows us to measure more sample (due to our sample-packing technique).
The 35Cl SSNMR of NdCl3 was measured between ca. –10 °C and +50 °C (Fig. 2a). The 35Cl SSNMR spectra were recorded at each temperature using the same acquisition parameters as in Fig. 1b for the same amount of time (about 1 hour per spectrum). As the temperature increases, a noticeable shift in the high-frequency discontinuities of the 35Cl powder patterns are discernable (i.e., the edge and “horn” discontinuities as marked with dashed lines). However, there is no shift observed in the lower-frequency discontinuities. If the EFG parameters were changing with temperature, which can happen in dynamical systems,103–105 then both the high- and low-frequency features of the pattern would be expected to change (i.e., if a larger or smaller effective CQ was observed due to motional averaging, then the entire breadth of the pattern should grow or shrink, respectively – Fig. S8). Therefore, it is safe to conclude that only the SA tensor parameters could be changing in this case. The subsequent spectra were fit in ssNake using the established EFG tensor parameters from Fig. 1b and Table 1 as a starting point then the SA tensor parameters alone were fitted at each temperature. A summary of the fitted SA tensor parameters in standard convention are summarized in Fig. 2b. δiso is found to shift from 840.2 to 681.4 ppm over the temperature range. The fitted δiso's were fit with a linear function with R2 = 0.989. Therefore, the δiso exhibits a 1/(T − Θ) temperature dependence characteristic of the Curie–Weiss paramagnetic shielding. UCl3 is nearly identical in structure to NdCl3 and has a slightly higher paramagnetic susceptibility; therefore, it is safe to assume that UCl3 also experiences the Curie–Weiss paramagnetic shielding.
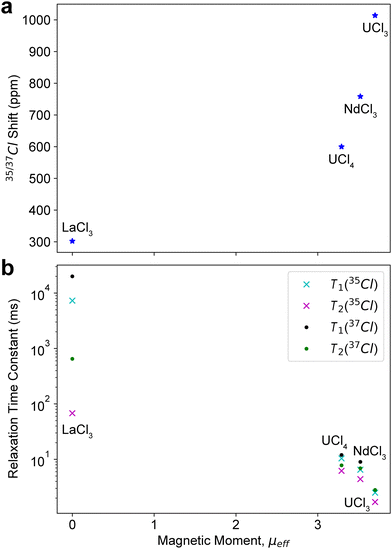
The relative 35/37Cl isotropic shift is compared across all samples (Fig. 3a). The shifts are plotted against the effective magnetic moment, µeff, of each compound. The room temperature µeff is zero for LaCl3 and is known for UCl3 (3.70) and UCl4 (3.29),106 but not for NdCl3; therefore a µeff of 3.52 µB was measured at 298 K using a Gouy balance. The shifts generally increase with the µeff's where the hyperfine constant, A ∝ µeff. However, the origin of the shifts in each sample can vary greatly with the changing electronic structure and is considered further in DFT calculations (vide infra). This same µeff trend is not observed in Ω, specifically with the large Ω = 5200 ppm for UCl4 (Table 1), which may suggest a significant dipolar contribution to the hyperfine coupling for this compound.
3.3. Relaxation measurements
Relaxation measurements were carried out for the 35/37Cl NMR data described above. The T1 is measured for both nuclei using a saturation-recovery (SR) type pulse sequence with WURST-CPMG signal detection.82,107 Inversion recovery experiments are more accurate in part due to the higher variation in range, but SR offers significantly faster measurements.108,109 Since every 1D spectrum in this work is measured with the WURST-CPMG pulse sequence, the effective-T2, Teff2, is obtained by default in every case, which also aids in signal detection by increasing the signal-to-noise of the spectra. It is important to note that multiple aspects of CPMG acquisition affect the measured Teff2 and this is not necessarily a reliable metric when ascribing physical differences between measurements and samples. Many experimental WURST-CPMG or standard (Q)CPMG parameters will impact the measured Teff2 including the echo delay times, RF fields, pulse widths, flip angles, pulse phase, and the accumulated errors in refocusing pulses.41,110–114 T1's are not susceptible to these aforementioned effects and should be considered a far more reliable relaxation measurement, but since the Teff2 is measured by default herein, it is also analyzed.The measured 35/37Cl relaxation constants are summarized in Table 2 and Fig. 3b. As mentioned above, LaCl3 is diamagnetic and features no influence from SA and has T1(35Cl) = 7.3 ± 0.2 s. This is compared to NdCl3 with T1(35Cl) = 6.5 ± 0.3 ms, which is approximately three-orders of magnitude smaller than LaCl3. Based on the 1D NMR spectra across all the compounds, the only contributing NMR interactions are the quadrupolar and SA interactions; therefore, these are also likely the only contributors to the non-secular relaxation. If quadrupolar relaxation was dominant, such a dramatic decrease in T1(35Cl) would not be expected for the CQ only differing by about 1 MHz between samples (i.e., for quadrupolar relaxation, 1/T1 = R1 ∝ CQ2). Therefore, the T1(35Cl) difference between the two samples is likely characteristic of the paramagnetic relaxation enhancement (PRE), which has R1 ∝ A2 ∝ µeff2. UCl3 and UCl4 likewise feature T1(35Cl) values that are ca. 1000-fold smaller than LaCl3. The T1(35Cl) differences between NdCl3, UCl3, and UCl4 all correlate well with their µeff's (Fig. 3b) commensurate with PRE. Although not as reliable as the T1 measurements, the same relaxation trends across these samples are also observed in the Teff2(35Cl) values. The difference between the Teff2(35Cl) of LaCl3 and the other samples is not as stark as the T1(35Cl)'s, likely because of the added secular factors impacting Teff2.
| Compound | T1 (35Cl) (ms) | T1 (37Cl) (ms) | Teff2 (35Cl)a (ms) | Teff2 (37Cl)a (ms) |
|---|---|---|---|---|
| a Teff2's are measured with varying echo delays, RF amplitudes, and WURST sweepwidths (see SI). | ||||
| LaCl3 | (7.3 ± 0.2) × 103 | (20 ± 2) × 103 | 68 ± 5 | 650 ± 10 |
| NdCl3 | 6.5 ± 0.3 | 9.0 ± 0.5 | 4.4 ± 0.1 | 6.9 ± 0.3 |
| UCl3 | 2.5 ± 0.4 | 2.8 ± 0.3 | 2.3 ± 0.3 | 2.8 ± 0.2 |
| UCl4 | 10.4 ± 0.9 | 12 ± 0.1 | 6.2 ± 0.1 | 7.8 ± 0.2 |
37Cl relaxation measurements were also made in every case and reflect the same relative differences between the samples. An important aspect to note is the comparison of the T1 values between 35Cl and 37Cl (Table 2). In every case, the T1(37Cl) > T1(35Cl). The dramatic decrease in T1(37Cl) between LaCl3 and the other samples, as well as the T1(37Cl)'s of the paramagnetic samples correlating with their magnetic moments (Fig. 3b), suggests the same PRE effects as 35Cl. Similar to 35Cl, the Teff2(37Cl) values match the µeff trends (Fig. 3b). As with T1 measurements it is also observed that Teff2(37Cl) > Teff2(35Cl). The differences between 37Cl and 35Cl are the gyromagnetic ratios, γ, and the measured CQ's. The former could cause an additional PRE contribution for each spin isotope (i.e., a PRE originating from scaling γ rather than µeff as discussed above). Whereas the latter could indicate the influence of a quadrupolar relaxation mechanism in these spin systems. This facet is clear in the case of diamagnetic LaCl3 where the 37Cl relaxation times are longer in the absence of any PRE. Therefore, both the PRE and quadrupolar relaxation mechanisms are likely simultaneously contributing to the total rate of relaxation in these spin systems.
3.4. Density functional theory
DFT calculations are first used to unravel the various contributions to the observed 35/37Cl NMR spectra. In all cases, reasonable agreement to the experimentally observed EFG tensor parameters are obtained with the DFT+U calculations with close agreement for LaCl3, NdCl3 and UCl4 (Table 1). Agreement with the experimental tensor values for UCl3 can be obtained by a +U correction to the 6d-orbitals in lieu of the 5f-orbitals but at the consequence of the incorrect electronic structures (see discussion in the SI). Therefore, +U corrections can only be applied to the 5f-orbitals for UCl3, which yields a slightly higher deviation from the experimental values.We also probe the effects of the electron hyperfine field from the paramagnetic metals on the 35/37Cl SA using experimental and calculated results. The paramagnetic shielding tensor, σS, is obtained from the calculated hyperfine tensor, A, and the g tensor, g, with the following relationship,115
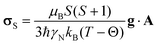 | (1) |
The calculated SA results for NdCl3, UCl3 and UCl4 are given in Table 1. With the overwhelming influence of A and the lack of experimental values, diamagnetic contributions to the SA are not reported, which yields no SA for the pure diamagnetic LaCl3 case. For NdCl3, the DFT calculations substantially overestimate the δiso using the literature g values. However, the results for UCl3 and UCl4 show relatively higher agreement to the measured 35/37Cl δiso's. The calculated A contribution to the span, Ω, is in good agreement for UCl3, and NdCl3, and underestimated for UCl4. Investigating the relative contributions to the SA (Table S6) reveal that the contact shift contributions substantially contribute to both the isotropic and anisotropic portion of the SA with the sign and magnitude of the g-tensor having the largest impact on the anisotropy.
These calculations also do not explicitly model the effects of thermally accessible excited electronic states that arise from zero-field splitting (ZFS) or crystal-field splitting (CFS) present in f-block metals. ZFS is known to impact chemical shifts in solution-state NMR of heavy element containing compounds and has been extensively described theoretically.119,120 Promising efforts have been made to incorporate these effects into solid-state NMR paramagnetic chemical shift calculations.121 However, the contributions of electron–electron repulsion, spin–orbit coupling, and CFS have been shown to contribute nearly equally to the magnetic properties of the actinide elements and can only be disentangled through extensive computational efforts.122 Although application of these methods to actinide and lanthanide-containing materials could improve the inaccuracies present in the current study, such developments remain a challenge.
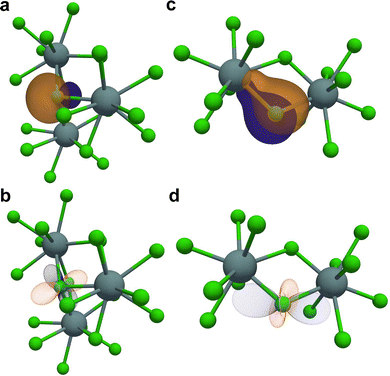
The DFT results can be further used to investigate the differences in the M–Cl bonds in these materials. In all the samples studied, the single Cl site adopts a bridging environment between two metal atoms. In the trichloride compounds, the three bridging M–Cl distances differ by at most 0.3% for NdCl3. However, in UCl4 the two bridging U–Cl bonds can be characterized as a short and a long bond with lengths of 2.64 Å and 2.89 Å, respectively. The differences in these bonding environments are investigated using the results from the LOBSTER analysis.96–98 This analysis provides measurements of bond covalency by calculating the overlap in the orbital energies and provides the simple numerical values in the form of the COHP, and a unitless parameter denoted the COBI which is a unitless parameter scaled from 0 to 1 where 0 is a fully ionic bonding interaction and 1 is completely covalent. This program was also recently updated to calculate local molecular orbitals (LMO) from periodic DFT calculations, which they denote as RLMO.98 Both UCl3 bonds and the longer UCl4 bonds have similar COBI values of 0.18 and 0.21, respectively which indicate a similar ionicity of these bonds. The COBI value of 0.38 for the shorter UCl4 bond indicates a slight increase in the covalency of this bond relative to the longer bonds. These differ slightly from the bond-weighted averages (ICHOP and ICOBI) in Table 1. This increased covalency results from the energy overlap between the Cl 3p and U 5f orbitals as revealed in the calculated density of states (DOS, Fig. S16).
Investigation of the RLMO for the Cl atom reveal a potential source of the increased covalency in UCl4. Looking at the RLMOs corresponding to one of the Cl π lone pair orbitals for these two compounds (Fig. 4) indicates that the electron density for this orbital is highly localized close to the Cl center in UCl3 whereas in UCl4 the density extends slightly along the longer U–Cl bond. Similar results have been observed in calculations of actinide hexachloride salts and have been invoked to explain relative covalency differences in this series.6 Further, in UCl3 Vzz is negative and points directly at the σ lone-pair orbital (Fig. 4b), which is characteristic of a more ionic bond.123 Contrastingly, Vzz for the UCl4 EFG tensor is positive and lies nearly parallel to the vector between the two U metal centers (Fig. 4d), which could suggest potential contributions from dative bonding between the U and Cl like that observed for previously in heavy transition-metal chlorides.33,34 The increased covalency in UCl4 likely explains the large Ω, as there will be a larger AD contribution when the U f electrons are closer to the 35/37Cl spins. It is unclear why the DFT calculations of A do not reflect this increase, but it could be linked to absence of spin–orbit paramagnetic effects and ZFS not being adequately captured. However, these results must be interpreted with care since VASP and LOBSTER project onto the electron density predicted onto lm-decomposed orbitals and although spin–orbit coupling is included in calculations, some differences with respect to the full Dirac equation could change the electron density, including the 5p1/2 and 6p1/2 states that overlap significantly with the nucleus.124–126 More work incorporating relativistic calculations based on the Dirac equation are likely required to fully understand these bonding interactions. Taken together, the results from the RLMO and DFT analysis suggest the increase in bond covalency for the U–Cl bond in UCl4 is responsible for both the increase in the 35/37Cl CQ and the dipolar contributions to the SA.
4. Conclusions
35/37Cl SSNMR of lanthanide and actinide chloride salts measured under static conditions offers a simple and sensitive measure of the paramagnetic SA effects without needing isotopic enrichment or specialized equipment. Some of the largest 35/37Cl SA parameters are reported for these materials, which is onset from the paramagnetic shielding resulting from hyperfine fields in uranium. The effects of bulk magnetic susceptibility likely introduce less inhomogeneous broadening than what can be resolved experimentally. The interplay of EFG and SA tensors offers a high-level of structural detail in a single experiment and offers an extremely unique spectral fingerprint of the analyte, which will be beneficial for future product characterization and identification. These measurements benchmark 35/37Cl shift ranges for U3+- and U4+-containing chloride compounds, which will further aid in future product characterization. All materials studied herein feature one chlorine site, but compounds or mixtures with multiple sites can still be resolved under static conditions.107,127 Static measurements at a single field alleviate safety concerns when measuring radioactive actinide materials. Measurements presented herein are relatively quick and simple to execute (i.e., <4 hours for the longest 35Cl measurement) and can be improved upon with static probes and larger coils, which may enable more advanced 35Cl–37Cl correlation experiments.128 37Cl measurements are approximately three-times slower but are ultimately not necessary to perform depending on the application. The size of the SA contribution hinders the CPMG-type acquisition presented herein, since the PRE shortens the T2; however, later actinides such as Pu and Am should exhibit less paramagnetic susceptibility than U3+, which would make the respective measurements of PuCl3 and AmCl3 easier than UCl3.DFT calculations using Hubbard+U corrections can help constrain the EFG and A tensor parameters and can enable easier independent identification of SA tensor parameters. Furthermore, these calculations provide an insight to the relative contributions of ionic and covalent bonding in the U–Cl systems, where experiments and calculations indicate a higher degree of bond covalency in UCl4 than UCl3. Our strategies for fast and reliable static measurements of actinide chloride salts will pave the way for high-throughput analysis useful to the molten salt reactor community and to researchers pursuing synthetic and fundamental advancements in actinide chemistry.
Conflicts of interest
There are no conflicts to declare.Data availability
All simulation input files, processing functions and scripts, and NMR datasets are available from the authors by request.Supplementary information (SI): additional simulations, experiments, and experimental details. See DOI: https://doi.org/10.1039/d5cp03944d.
Acknowledgements
Research presented in this report was supported by the Laboratory Directed Research and Development program of Los Alamos National Laboratory under project number 20230728DI. Los Alamos National Laboratory is operated by Triad National Security, LLC, for the National Nuclear Security Administration of US Department of Energy (contract no. 89233218CNA000001). The authors thank Professor Rob Schurko for helpful discussions.References
- T. V. Fetrow, J. P. Grabow, J. Leddy and S. R. Daly, Inorg. Chem., 2021, 60, 7593–7601 CrossRef CAS PubMed.
- D. Patel, A. J. Wooles, E. Hashem, H. Omorodion, R. J. Baker and S. T. Liddle, New J. Chem., 2015, 39, 7559–7562 RSC.
- J. L. Kiplinger, D. E. Morris, B. L. Scott and C. J. Burns, Organometallics, 2002, 21, 5978–5982 CrossRef CAS.
- D.-C. Sergentu and J. Autschbach, Chem. Sci., 2022, 13, 3194–3207 RSC.
- S. A. Kozimor, P. Yang, E. R. Batista, K. S. Boland, C. J. Burns, D. L. Clark, S. D. Conradson, R. L. Martin, M. P. Wilkerson and L. E. Wolfsberg, J. Am. Chem. Soc., 2009, 131, 12125–12136 CrossRef CAS PubMed.
- C. L. Silva, L. Amidani, M. Retegan, S. Weiss, E. F. Bazarkina, T. Graubner, F. Kraus and K. O. Kvashnina, Nat. Commun., 2024, 15, 6861 CrossRef CAS PubMed.
- J. N. Cross, J. Su, E. R. Batista, S. K. Cary, W. J. Evans, S. A. Kozimor, V. Mocko, B. L. Scott, B. W. Stein and C. J. Windorff, et al., J. Am. Chem. Soc., 2017, 139, 8667–8677 CrossRef CAS PubMed.
- S. G. Minasian, J. M. Keith, E. R. Batista, K. S. Boland, D. L. Clark, S. D. Conradson, S. A. Kozimor, R. L. Martin, D. E. Schwarz and D. K. Shuh, et al., J. Am. Chem. Soc., 2012, 134, 5586–5597 CrossRef CAS PubMed.
- L. Peng; R. J. Clément; M. Lin and Y. Yang, CHAPTER 1: NMR Principles of Paramagnetic Materials. In New Developments in NMR, The Royal Society of Chemistry, 2021, Vol. 2021, pp. 3–70 Search PubMed.
- N. F. Ramsey, Phys. Rev., 1950, 78, 695–699 CrossRef CAS.
- N. F. Ramsey, Phys. Rev., 1950, 78, 699–703 CrossRef CAS.
- N. F. Ramsey, Phys. Rev., 1950, 77, 567 CrossRef CAS.
- N. F. Ramsey, Phys. Rev., 1951, 83, 540–541 CrossRef CAS.
- N. F. Ramsey, Phys. Rev., 1952, 86, 243–246 CrossRef CAS.
- J. P. Carvalho, W. Papawassiliou and A. J. Pell, J. Magn. Reson., 2022, 340, 107235 CrossRef CAS PubMed.
- A. J. Pell, G. Pintacuda and C. P. Grey, Prog. Nucl. Magn. Reson. Spectrosc., 2019, 111, 1–271 CrossRef CAS PubMed.
- H. Yasuoka, G. Koutroulakis, H. Chudo, S. Richmond, D. K. Veirs, A. I. Smith, E. D. Bauer, J. D. Thompson, G. D. Jarvinen and D. L. Clark, Science, 2012, 336, 901–904 CrossRef CAS PubMed.
- A. P. Dioguardi, H. Yasuoka, S. M. Thomas, H. Sakai, S. K. Cary, S. A. Kozimor, T. E. Albrecht-Schmitt, H. C. Choi, J.-X. Zhu and J. D. Thompson, et al., Phys. Rev. B: Condens. Matter Mater. Phys., 2019, 99, 035104 CrossRef CAS.
- N. J. Curro, T. Caldwell, E. D. Bauer, L. A. Morales, M. J. Graf, Y. Bang, A. V. Balatsky, J. D. Thompson and J. L. Sarrao, Phys. B, 2006, 378–380, 915–919 CrossRef CAS.
- K. Ikushima, S. Tsutsui, Y. Haga, H. Yasuoka, R. E. Walstedt, N. M. Masaki, A. Nakamura, S. Nasu and Y. Ōnuki, Phys. Rev. B, 2001, 63, 104404 CrossRef.
- R. E. Walstedt, Y. Tokunaga, H. Kato, H. Sakai, T. Fujimoto, S. Kambe and H. Yasuoka, J. Phys. Soc. Jpn., 2006, 75, 77–81 CrossRef.
- K. D. Morrison, M. Zavarin, A. B. Kersting, J. D. Begg, H. E. Mason, E. Balboni and Y. Jiao, Environ. Sci. Technol., 2021, 55, 1626–1636 CrossRef CAS PubMed.
- E. C. Uribe, H. E. Mason, J. A. Shusterman, A. Bruchet and H. Nitsche, Dalton Trans., 2016, 45, 10447–10458 RSC.
- E. C. Uribe, H. E. Mason, J. A. Shusterman and W. W. Lukens, Dalton Trans., 2017, 46, 5441–5456 RSC.
- Y. Tokunaga, Y. Homma, S. Kambe, D. Aoki, H. Sakai, E. Yamamoto, A. Nakamura, Y. Shiokawa, R. E. Walstedt and H. Yasuoka, Phys. Rev. Lett., 2005, 94, 137209 CrossRef CAS PubMed.
- Y. Tokunaga, D. Aoki, Y. Homma, S. Kambe, H. Sakai, S. Ikeda, T. Fujimoto, R. E. Walstedt, H. Yasuoka and E. Yamamoto, et al., Phys. Rev. Lett., 2006, 97, 17–20 CrossRef PubMed.
- Y. Tokunaga, Y. Homma, S. Kambe, D. Aoki, H. Sakai, E. Yamamoto, A. Nakamura, Y. Shiokawa, R. E. Walstedt and H. Yasuoka, Phys. B, 2006, 378–380, 929–930 CrossRef CAS.
- Y. Tokunaga, D. Aoki, Y. Homma, S. Kambe, H. Sakai, S. Ikeda, T. Fujimoto, R. E. Walstedt, H. Yasuoka and Y. Shiokawa, et al., J. Magn. Magn. Mater., 2007, 310, 735–737 CrossRef CAS.
- Y. Tokunaga, T. Nishi, S. Kambe, M. Nakada, Y. Homma, H. Sakai and H. Chudo, J. Phys. Soc. Jpn., 2011, 80, 2010–2012 CrossRef.
- L. Martel, N. Magnani, J.-F. Vigier, J. Boshoven, C. Selfslag, I. Farnan, J.-C. Griveau, J. Somers and T. Fanghänel, Inorg. Chem., 2014, 53, 6928–6933 CrossRef CAS PubMed.
- I. Farnan, H. Cho, W. J. Weber, R. D. Scheele, N. R. Johnson and A. E. Kozelisky, Rev. Sci. Instrum., 2004, 75, 5232–5236 CrossRef CAS.
- D. L. Bryce and G. D. Sward, Magn. Reson. Chem., 2006, 44, 409–450 CrossRef CAS PubMed.
- C. M. Widdifield; R. P. Chapman and D. L. Bryce, Chapter 5 Chlorine, Bromine, and Iodine Solid-State NMR Spectroscopy. In Annual Reports on NMR Spectroscopy, Elsevier Ltd., 2009, Vol. 66, pp. 195–326 Search PubMed.
- P. M. J. Szell and D. L. Bryce, Recent Advances in Chlorine, Bromine, and Iodine Solid-State NMR Spectroscopy, In Annual Reports on NMR Spectroscopy, Elsevier Ltd., 2015, Vol. 84, pp. 115–162 Search PubMed.
- P. M. J. Szell and D. L. Bryce, Concepts Magn. Reson., Part A, 2016, 45A, e21412 CrossRef.
- L. A. O’Dell and R. W. Schurko, Chem. Phys. Lett., 2008, 464, 97–102 CrossRef.
- L. A. O’Dell, A. J. Rossini and R. W. Schurko, Chem. Phys. Lett., 2009, 468, 330–335 CrossRef.
- A. J. Rossini, H. Hamaed and R. W. Schurko, J. Magn. Reson., 2010, 206, 32–40 CrossRef CAS PubMed.
- D. A. Hirsh, A. J. Rossini, L. Emsley and R. W. Schurko, Phys. Chem. Chem. Phys., 2016, 18, 25893–25904 RSC.
- J. Koppe and M. R. Hansen, J. Phys. Chem. A, 2020, 124, 4314–4321 CrossRef CAS PubMed.
- I. Hung and Z. Gan, J. Magn. Reson., 2010, 204, 256–265 CrossRef CAS PubMed.
- M. J. Jaroszewicz, A. R. Altenhof, R. W. Schurko and L. Frydman, J. Am. Chem. Soc., 2021, 143, 19778–19784 CrossRef CAS PubMed.
- T. Wolf, A. Eden-Kossoy and L. Frydman, Solid State Nucl. Magn. Reson., 2023, 101862 CrossRef CAS PubMed.
- J. J. Kimball, A. R. Altenhof, M. J. Jaroszewicz and R. W. Schurko, J. Phys. Chem. A, 2023, 127, 9621–9634 CrossRef CAS PubMed.
- A. Venkatesh, M. P. Hanrahan and A. J. Rossini, Solid State Nucl. Magn. Reson., 2017, 84, 171–181 CrossRef CAS PubMed.
- A. V. Wijesekara, A. Venkatesh, B. J. Lampkin, B. VanVeller, J. W. Lubach, K. Nagapudi, I. Hung, P. L. Gor’kov, Z. Gan and A. J. Rossini, Chem. – Eur. J., 2020, 26, 7881–7888 CrossRef CAS PubMed.
- I. Hung and Z. Gan, J. Phys. Chem. Lett., 2020, 11, 4734–4740 CrossRef CAS PubMed.
- R. Bayzou, J. Trébosc, I. Hung, Z. Gan, O. Lafon and J.-P. Amoureux, J. Chem. Phys., 2022, 156, 064202 CrossRef CAS PubMed.
- I. Hung and Z. Gan, J. Magn. Reson., 2023, 350, 107423 CrossRef CAS PubMed.
- D. L. Bryce, Solid-state NMR of quadrupolar nuclei: Selected new methods and applications. In Annual Reports on NMR Spectroscopy, Elsevier Ltd., 2022, pp. 1–46 Search PubMed.
- H. Rinneberg, H. Haas and H. Hartmann, J. Chem. Phys., 1969, 50, 3064–3071 CrossRef CAS.
- H. Rinneberg and H. Hartmann, J. Chem. Phys., 1970, 52, 5814–5820 CrossRef CAS.
- A. Narath, Phys. Rev., 1965, 140, A552–A568 CrossRef.
- W. J. O’Sullivan, W. W. Simmons and W. A. Robinson, Phys. Rev., 1965, 140, A1759–A1771 CrossRef.
- W. H. Jones and S. L. Segel, Phys. Rev. Lett., 1964, 13, 528–530 CrossRef CAS.
- F. Engelke, D. Michel, W. Windsch, P. Sarv and E. Lippmaa, Ferroelectrics, 1990, 108, 337–340 CrossRef CAS.
- H. Trill, H. Eckert and V. I. Srdanov, Phys. Rev. B: Condens. Matter Mater. Phys., 2005, 71, 014412 CrossRef.
- D. Michel, U. Hacker, T. Erge and J. Petersson, Phys. Status Solidi, 1994, 185, 257–264 CrossRef CAS.
- L. Bauder and G. Wu, Magn. Reson. Chem., 2024, 62, 145–155 CrossRef CAS PubMed.
- S. Park, E. D. Walter, C. Z. Soderquist, S. I. Sinkov and H. Cho, J. Phys. Chem. A, 2023, 8347–8353 CrossRef CAS PubMed.
- S. Park, R. G. Surbella, S. I. Sinkov, A. B. Philips, J. Autschbach and H. Cho, Chem. Phys., 2023, 576, 112094 CrossRef.
- S. Antonijevic and S. Wimperis, J. Chem. Phys., 2005, 122, 044312 CrossRef PubMed.
- S. Antonijevic and S. Wimperis, J. Magn. Reson., 2003, 164, 343–350 CrossRef CAS PubMed.
- T. Iijima and K. Nishimura, Chem. Phys. Lett., 2011, 514, 181–186 CrossRef CAS.
- T. Iijima, T. Shimizu and K. Nishimura, J. Magn. Reson., 2015, 251, 57–64 CrossRef CAS PubMed.
- M. Mizuno, N. Itakura and K. Endo, Chem. Phys. Lett., 2005, 416, 358–363 CrossRef CAS.
- T. Iijima, S. Ohki and M. Tansho, Solid State Nucl. Magn. Reson., 2021, 112, 101709 CrossRef CAS PubMed.
- R. Aleksis, J. P. Carvalho, A. Jaworski and A. J. Pell, Solid State Nucl. Magn. Reson., 2019, 51–62 CrossRef CAS PubMed.
- R. Aleksis and A. J. Pell, J. Chem. Phys., 2021, 155, 094202 CrossRef CAS PubMed.
- J. T. Ash, N. M. Trease and P. J. Grandinetti, J. Am. Chem. Soc., 2008, 130, 10858–10859 CrossRef CAS PubMed.
- J. P. Carvalho, A. Jaworski, M. J. Brady and A. J. Pell, Magn. Reson. Chem., 2020, 58, 1055–1070 CrossRef CAS PubMed.
- D. L. Bryce, Tensor Interplay, In Encyclopedia of Magnetic Resonance, 2009 Search PubMed.
- F. A. Perras, C. M. Widdifield and D. L. Bryce, Solid State Nucl. Magn. Reson., 2012, 45–46, 36–44 CrossRef CAS PubMed.
- D. L. Bryce, G. D. Sward and S. Adiga, J. Am. Chem. Soc., 2006, 128, 2121–2134 CrossRef CAS PubMed.
- I. Hung, A. R. Altenhof, R. W. Schurko, D. L. Bryce, O. H. Han and Z. Gan, Magn. Reson. Chem., 2021, 59, 951–960 CrossRef CAS PubMed.
- K. A. Erickson, S. S. Parker and M. J. Monreal, Chem.: Methods, 2024, 4, 3–5 Search PubMed.
- J. A. Hermann; J. F. Suttle and H. R. Hoekstra, Uranium(IV) Chloride, In Inorganic Syntheses, John Wiley & Sons Ltd, Hoboken, New Jersey, 1957, pp. 143–145 Search PubMed.
- L. A. O’Dell, Solid State Nucl. Magn. Reson., 2013, 55–56, 28–41 CrossRef PubMed.
- R. W. Schurko, Acquisition of Wideline Solid-State NMR Spectra of Quadrupolar Nuclei, In Encyclopedia of Magnetic Resonance, John Wiley & Sons, Ltd, Chichester, UK, 2011, pp. 77–93 Search PubMed.
- R. W. Schurko, Acc. Chem. Res., 2013, 46, 1985–1995 CrossRef CAS PubMed.
- X. Guan and R. E. Stark, Solid State Nucl. Magn. Reson., 2010, 38, 74–76 CrossRef CAS PubMed.
- A. R. Altenhof, M. J. Jaroszewicz, K. J. Harris and R. W. Schurko, J. Chem. Phys., 2021, 154, 034202 CrossRef CAS PubMed.
- A. R. Altenhof, H. Mason and R. W. Schurko, J. Magn. Reson., 2023, 346, 107320 CrossRef CAS PubMed.
- J. J. Helmus and C. P. Jaroniec, J. Biomol. NMR, 2013, 55, 355–367 CrossRef CAS PubMed.
- M. J. Jaroszewicz, A. R. Altenhof, R. W. Schurko and L. Frydman, J. Magn. Reson., 2023, 354, 107528 CrossRef CAS PubMed.
- S. G. J. van Meerten, W. M. J. Franssen and A. P. M. Kentgens, J. Magn. Reson., 2019, 301, 56–66 CrossRef CAS PubMed.
- M. Bak, J. T. Rasmussen and N. C. Nielsen, J. Magn. Reson., 2011, 213, 366–400 CrossRef CAS PubMed.
- Z. Tošner, R. Andersen, B. Stevensson, M. Edén, N. C. Nielsen and T. Vosegaard, J. Magn. Reson., 2014, 246, 79–93 CrossRef PubMed.
- G. Kresse and J. Hafner, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 47, 558–561 CrossRef CAS PubMed.
- G. Kresse and J. Hafner, J. Phys.: Condens. Matter, 1994, 6, 8245–8257 CrossRef CAS.
- G. Kresse and J. Furthmüller, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- J. P. Perdew, A. Ruzsinszky, G. I. Csonka, O. A. Vydrov, G. E. Scuseria, L. A. Constantin, X. Zhou and K. Burke, Phys. Rev. Lett., 2008, 100, 136406 CrossRef PubMed.
- S. L. Dudarev, G. A. Botton, S. Y. Savrasov, C. J. Humphreys and A. P. Sutton, Phys. Rev. B: Condens. Matter Mater. Phys., 1998, 57, 1505–1509 CrossRef CAS.
- J. Kim, D. S. Middlemiss, N. A. Chernova, B. Y. X. Zhu, C. Masquelier and C. P. Grey, J. Am. Chem. Soc., 2010, 132, 16825–16840 CrossRef CAS PubMed.
- P. C. Müller, C. Ertural, J. Hempelmann and R. Dronskowski, J. Phys. Chem. C, 2021, 125, 7959–7970 CrossRef.
- R. Nelson, C. Ertural, J. George, V. L. Deringer, G. Hautier and R. Dronskowski, J. Comput. Chem., 2020, 41, 1931–1940 CrossRef CAS PubMed.
- M. Pauls, D. Schnieders and R. Dronskowski, J. Phys. Chem. A, 2023, 127, 6541–6551 CrossRef CAS PubMed.
- B. Morosin, J. Chem. Phys., 1968, 49, 3007–3012 CrossRef CAS.
- J. C. Taylor and P. W. Wilson, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1974, 30, 2803–2805 CrossRef.
- J. C. Taylor and P. W. Wilson, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1973, 29, 1942–1944 CrossRef CAS.
- C. A. O’Keefe, K. E. Johnston, K. Sutter, J. Autschbach, L. Delevoye, N. Popo, M. Taou, K. Oudatchin and R. W. Schurko, Inorg. Chem., 2014, 53, 9581–9597 CrossRef PubMed.
- J. H. Kristensen, G. L. Hoatson and R. L. Vold, Solid State Nucl. Magn. Reson., 1998, 13, 1–37 CrossRef CAS PubMed.
- R. L. Vold and G. L. Hoatson, J. Magn. Reson., 2009, 198, 57–72 CrossRef CAS PubMed.
- C. S. Vojvodin, S. T. Holmes, L. K. Watanabe, J. M. Rawson and R. W. Schurko, CrystEngComm, 2022, 24, 2626–2641 RSC.
- D. R. Kindra and W. J. Evans, Chem. Rev., 2014, 114, 8865–8882 CrossRef CAS PubMed.
- M. J. Jaroszewicz, L. Frydman and R. W. Schurko, J. Phys. Chem. A, 2017, 121, 51–65 CrossRef CAS PubMed.
- J. S. Frye, Concepts Magn. Reson., 1989, 1, 27–33 CrossRef.
- E. D. Becker, J. A. Ferretti, R. K. Gupta and G. H. Weiss, J. Magn. Reson., 1980, 37, 381–394 CAS.
- E. D. Ostroff and J. S. Waugh, Phys. Rev. Lett., 1966, 16, 1097–1098 CrossRef CAS.
- J. S. Waugh, C. H. Wang, L. M. Huber and R. L. Vold, J. Chem. Phys., 1968, 48, 662–670 CrossRef CAS.
- M. Hürlimann and D. Griffin, J. Magn. Reson., 2000, 143, 120–135 CrossRef PubMed.
- S. L. Veinberg, A. W. Lindquist, M. J. Jaroszewicz and R. W. Schurko, Solid State Nucl. Magn. Reson., 2016, 1–14 Search PubMed.
- A. R. Altenhof, Z. Gan and R. W. Schurko, J. Magn. Reson., 2022, 337 Search PubMed.
- R. Pigliapochi, A. J. Pell, I. D. Seymour, C. P. Grey, D. Ceresoli and M. Kaupp, Phys. Rev. B, 2017, 95, 054412 CrossRef.
- C. A. Hutchison, P. M. Llewellyn, E. Wong and P. Dorain, Phys. Rev., 1956, 102, 292 CrossRef CAS.
- J. D. Riley, J. M. Baker and R. J. Birgeneau, Proc. R. Soc. London. A. Math. Phys. Sci., 1970, 320, 369–395 CAS.
- G. Garton, M. T. Hutchings, R. Shore and W. P. Wolf, J. Chem. Phys., 1964, 41, 1970–1974 CrossRef CAS.
- V. Lupei and C. Stoicescu, J. Phys. C:Solid State Phys., 1979, 12, 4585–4590 CrossRef CAS.
- F. Gendron and J. Autschbach, J. Chem. Theory Comput., 2016, 12, 5309–5321 CrossRef CAS PubMed.
- A. Mondal and M. Kaupp, J. Phys. Chem. C, 2019, 123, 8387–8405 CrossRef CAS.
- K. Georgiadis, Computational Modelling of Molecular Nanomagnets, ed G. Rajaraman, Challenges and Advances in Computational Chemistry and Physics, Springer International Publishing, Cham, 2023, Vol. 34 Search PubMed.
- J. Autschbach, S. Zheng and R. W. Schurko, Concepts Magn. Reson., Part A, 2010, 36A, 84–126 CrossRef CAS.
- D. A. Rehn, J. M. Wills, T. E. Battelle and A. E. Mattsson, Phys. Rev. B, 2020, 101, 085114 CrossRef CAS.
- J. Autschbach, Relativistic Effects on NMR Parameters. In Science and Technology of Atomic, Molecular, Condensed Matter and Biological Systems, Elsevier Inc., 2013, Vol. 3, pp. 69–117 Search PubMed.
- J. Vicha, J. Novotný, S. Komorovsky, M. Straka, M. Kaupp and R. Marek, Chem. Rev., 2020, 120, 7065–7103 CrossRef CAS PubMed.
- A. R. Altenhof, M. J. Jaroszewicz, L. Frydman and R. Schurko, Phys. Chem. Chem. Phys., 2022, 22792–22805 RSC.
- T. Wolf, M. J. Jaroszewicz and L. Frydman, J. Phys. Chem. C, 2022, 126, 9386–9395 CrossRef CAS PubMed.
- M. Mehring, Principles of High Resolution NMR in Solids, Springer Berlin Heidelberg, Berlin, Heidelberg, 1983 Search PubMed.
- S. Adiga, D. Aebi and D. L. Bryce, Can. J. Chem., 2007, 85, 496–505 CrossRef CAS.
Footnote |
| † Current Address: Geothermal Energy and Geofluids Group, Department of Earth and Planetary Sciences, ETH Zürich, Zürich 8001, Switzerland. |
| This journal is © the Owner Societies 2026 |