Open Access Article
Open Access ArticleIn situ transmission electron microscopy observations of CaCO3 crystallization onto polysaccharide-coated nanoparticles
Brenna M.
Knight
 ab,
Biao
Jin
ab,
Biao
Jin
 c,
Yuna
Bae
c,
Yuna
Bae
 c,
James J.
De Yoreo
c,
James J.
De Yoreo
 cd and
Patricia M.
Dove
cd and
Patricia M.
Dove
 *abef
*abef
aDepartment of Chemistry, Virginia Tech, Blacksburg, VA 24061, USA. E-mail: dove@vt.edu
bMacromolecules Innovation Institute, Virginia Tech, Blacksburg, VA 24061, USA
cPhysical Sciences Division, Pacific Northwest National Laboratory, Richland, WA 99354, USA
dDepartment of Materials Science and Engineering, University of Washington, Seattle, WA 98195, USA
eDepartment of Geosciences, Virginia Tech, Blacksburg, VA 24061, USA
fDepartment of Materials Science and Engineering, Virginia Tech, Blacksburg, VA 24061, USA
First published on 10th April 2026
Abstract
Polysaccharides and proteoglycans are widely associated with the organic matrix at sites of CaCO3 biomineralization, and previous studies indicate that these macromolecules may confer greater roles in mineral nucleation than previously recognized. This investigation uses in situ liquid-phase transmission electron microscopy (LP-TEM) to observe CaCO3 nucleation onto aminated silica (SiO2–NH3+) nanoparticles treated with a layer of chitosan (near-neutral derivative of chitin) or heparin (a carboxylated and highly sulfated glycosaminoglycan). In the absence of polysaccharides, few CaCO3 particles formed and exhibited mobility. However, the SiO2–NH3+ nanoparticles were enveloped in a region of higher mass density relative to the bulk solution, suggesting the development of a local solute-rich environment that surrounds the charged NH3+ groups. The heparin- or chitosan-coated silica particles also exhibited regions of higher mass density around the nanoparticles. In the presence of these polysaccharide coatings, we observed the nucleation of abundant CaCO3 particles whereby the polyanionic heparin promoted more nucleation than the weakly cationic chitosan. Many crystallites appeared to form at the polysaccharide–TEM cell membrane–solution interface, further indicating interfacial and macromolecule-specific control on crystallization. The combined results demonstrate that chitosan and heparin have an appreciable effect on the timing, size, and location of CaCO3 nucleation compared to the polysaccharide-free nanoparticles.
1. Introduction
During biological crystallization, the organic matrix modulates the timing and placement of biominerals to form unique biocomposites. Macromolecules confer much of this activity.1–6 In CaCO3-forming systems, the organic matrix often contains chitin and anionic biopolymers (e.g., SO3− and/or COO− functionalized) alongside specialized proteins.1,7–13 However, a mechanistic understanding of how macromolecular properties translate to control over CaCO3 nucleation and subsequent growth and phase transformation processes is not yet resolved. Insight into where and how CaCO3 nucleation occurs within organic interfacial environments is also relevant to subsurface and engineered systems, where nanoscale interphases associated with organic matter and mineral surfaces can influence carbonate scaling and the co-precipitation of critical rare earth elements.Investigations of CaCO3 nucleation onto chitosan materials with varied degrees of sulfation demonstrated a relationship between net charge, the position of functional groups, calcium binding, and interfacial free energy (γnet).14,15 Parallel molecular dynamics (MD) studies predict that Ca2+ interactions with sulfated chitosans are solvent-separated by distances that correlate with the degree of sulfation (0.5–1.2 nm).15–20 Model results also indicate that nucleation is favored to occur at a distance from the polysaccharide–water interface that correlates with the position of solvation waters around functional groups and Ca2+.15,16,18 The concept that crystal nucleation can occur away from a surface, but within the non-bulk diffuse layer, is supported by recent studies using liquid phase in situ transmission electron microscopy (LP-TEM). For example, an investigation of CaCO3 nucleation onto carboxyl-rich de novo protein templates reported an ≈1 nm separation between nanocrystals and the protein substrate.21 This distance is consistent with the thickness of the hydration layer around calcite,21,22 further implicating solvation as a control on nucleation. Also, an ≈1 nm separation was observed between new nuclei and existing particles in TEM studies of colloidal gold23 and hematite.24 These experimental and computational lines of evidence raise the question of where CaCO3 nucleation occurs within the substrate–solution interfacial environment for polysaccharides such as chitosan. Using insights from our previous experimental and modeling studies of calcite nucleation onto sulfated and uncharged polysaccharides, we hypothesize that CaCO3 nucleation takes place away from the polysaccharide surface at a separation distance that is dependent on macromolecular and solution properties in the interphase region/diffuse layer.
To test this hypothesis, we used in situ LP-TEM to image real-time nucleation of CaCO3 in the presence of the polysaccharides. The technique allows observations of crystallization processes at the nanometer to near-Å scale.25,26 However, biomacromolecules are difficult to image by this approach because of their low electron density,27–29 and therefore, they present little contrast between the molecule and the adjacent bulk solution. The low contrast is exacerbated by the variable interphase region between the bulk solution and the polysaccharide. The nature of this region is dependent on solution composition (e.g., pH and ionic strength) as well as biopolymer properties that include net charge, charge geometry/configuration, and persistence length (i.e., effective chain stiffness).30–32 We were able to address some of the limitations by utilizing 20 nm aminated silica nanoparticles (SiO2–NH3+) as substrates for the polysaccharides (Fig. 1A and inset). By this approach, the silica–PS interface was better resolved in the TEM cell.
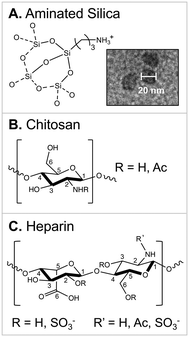 | ||
| Fig. 1 Materials used in this study. (A) Aminated silica nanoparticles (20 nm) were used as a control and subsequently coated with polysaccharides; (B) chitosan; (C) heparin. | ||
The experiments were designed to observe nucleation in calcite-supersaturated solutions containing: (1) SiO2–NH3+ nanoparticles (nps); (2) SiO2–NH3+ nps treated to have a chitosan coating; and (3) SiO2–NH3+ nps treated with heparin. The chitosan and heparin polysaccharides were chosen as models to represent macromolecules in the organic matrix that present near-neutral (to slightly cationic) and high net charge functional groups, respectively.
2. Experimental
2.1. Materials
The untreated SiO2–NH3+ nps (Fig. 1A, 20 nm, nanoComposix) were used as a control relative to the polysaccharide-coated materials. Each silica amine has a +1 charge (pKa ≈9–11) at the experimental pH (6.9–7.6) of this study.33 This high net-positive charge corresponds to approximately 2 NH3+ groups per nm2.34The polysaccharide compositions were as follows. Chitosan (Fig. 1B, sourced from Millipore Sigma, medium molecular weight, 270 kDa (ref. 15)) is a chitin derivative in which a fraction of the acetyl groups is converted to free amines. Chitosan presents an interfacial environment with a relatively low charge density, some hydrogen bonding, and thus a relatively short persistence length (Kuhn length).30 Given that these properties govern how polymers occupy the interfacial region between the solution and substrate, this material has a greater conformational freedom (relative to heparin) and can be considered a weakly flexible biopolymer. The material had a degree of deacetylation/degree of amination = 0.76 (by 1H NMR15) and an amine charge = +0.06 to +0.22 at pH 6.9–7.6 (pKa,chitosan ≈6.5).35,36 Therefore, the average charge per monosaccharide on chitosan was ≈+0.05 to +0.17. Assuming a surface area that is approximately equal to a glucose repeat unit, we estimate that chitosan presents a low net positive charge of ≈+0.04–0.13 charged amines per nm2.
In contrast, heparin (Fig. 1C, sourced from Millipore Sigma, one COO− and 2.4 SO3− groups per disaccharide, 18 kDa (ref. 37)) is a highly charged, polyanionic polysaccharide containing both carboxyl and sulfate groups. Each group has a −1 charge at the experimental pH (pKa,heparin ≈3–5),37–39 corresponding to an average charge of −1.7 per monosaccharide for our material, or approximately 1.3 anionic groups per nm2 (based on a glucose repeat unit). Heparin thereby presents a stronger electrostatic repulsion between chains and thus a larger persistence length to behave as a semi-rigid polyelectrolyte.30
2.2. Nanoparticle coating
For each experiment, SiO2–NH3+ nanoparticles were coated with chitosan or heparin using crosslinking (XL) methods adapted for sulfated chitosan derivatives.15,40,41 Briefly, heparin and chitosan solutions (2 mg mL−1) were prepared in deionized (DI) water and DI water containing 1% v/v acetic acid, respectively. Glutaraldehyde (1% in DI water, 0.25 mL) was added to the polysaccharide solutions (0.5 mL). After 20 min, aminated silica nanoparticles (0.25 mL, 2 mg mL−1 in DI water) were added. The mixtures were sonicated for 15 min and then centrifuged (10 min, 7000 rpm) and washed with DI water 3 times. Solutions were sonicated for 5 min prior to each use. Attenuated total reflectance (ATR, Bruker Alpha II, resolution 2 cm−1, 32 scans, 4000–6000 cm−1) was used to confirm that polysaccharides were attached to the aminated silica nanoparticles (Fig. S1).2.3. Liquid-phase transmission electron microscopy (LP-TEM)
The in situ crystallization observations used an experimental setup that was modeled after the method employed by Davila-Hernandez et al.21 The liquid-cell contained two square silicon chips (Hummingbird Scientific) measuring 2.6 × 2.6 mm2 each containing 50 nm-thick silicon nitride (Si3N4) membranes with 50 × 200 μm2 imaging windows. Prior to use, the chips underwent a 2 min plasma cleaning process with a Harrick Plasma Cleaner. For a typical experiment, 0.5 μL of 10 mM CaCl2 and 0.1 μL 2% nanoparticle solution were applied to the spacer chip with a spacer thickness of 100 nm. Next, 0.5 μL of a 10 mM NaHCO3 solution was added to produce a supersaturated CaCO3 solution (supersaturation (σ) with respect to calcite (σcalcite) ≈1.6),42,43 and the reaction solution was sealed using a window chip to form a liquid-cell. The liquid-cell was then placed inside a Hummingbird Scientific sample holder. A leak check was conducted by inserting the assembled holder into a Hummingbird Scientific high-vacuum leak checking station for 2 min. This station features a low base pressure (<1 × 10−6 mbar) and short pumping and venting times, and allows the liquid-cell window to be imaged with optical microscopy. The holder was then inserted into an FEI (a subsidiary of Thermo Fisher Scientific) Titan Environmental TEM operated at 300 kV with a Gatan UltraScan 1000 camera.A series of TEM images were captured beginning ≈10 min after mixing using an Eagle CCD with a resolution of 1024 × 1024 pixels, a condenser aperture size of 50 μm, and a spot size of 3 nm. In situ movies were recorded using the free software Gatan DigitalMicrograph and post-processing of images from the movies was performed using the open-source software ImageJ.44 A low electron dose rate of ≈100 e− nm−2 s−1 was employed to minimize beam effects. A schematic of the TEM setup is shown in Fig. S2.
For additional control experiments of 2% aminated SiO2 nanoparticles in pure water (without solute ions) and at pH 9 (σcalcite ≈1.6, 2.5 mM CaCl2 and 1 mM Na2CO3), LP-TEM was performed using an Insight Chips® nanochannel holder. This holder featured a 130 nm channel height, with inner surfaces encapsulated by 25 nm-thick silicon nitride and coated with Al2O3. The solution was loaded into the nanochannel via a flow line. Imaging was conducted with a Gatan Metro 300 camera.
3. Results and discussion
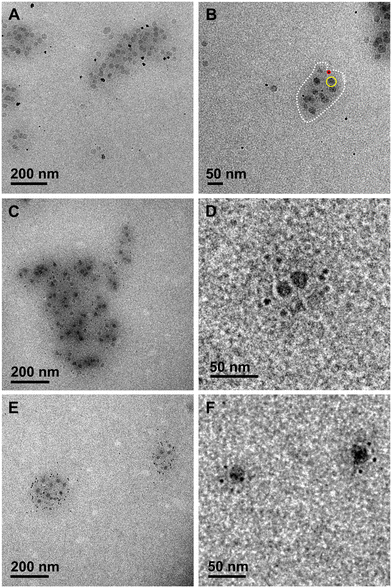
For all in situ systems, the CaCO3 products that formed are expected to be crystalline. Selected area electron diffraction (SAED) could not discern if the particles were crystalline or amorphous materials (Fig. S3). However, the nucleating particles were significantly more electron dense (i.e., darker) than would be expected for a hydrated cluster or amorphous material.45–47Particles were observed in regions that were different from the initial location of the electron beam, indicating that beam effects were not driving the nucleation process.21 Moreover, as discussed below, each of the three types of particles used in the study gave material-specific results. Thus, the progression of nucleation, both the location and rate, was not determined by solution radiolysis. Representative images from each experiment are shown in Fig. 2.
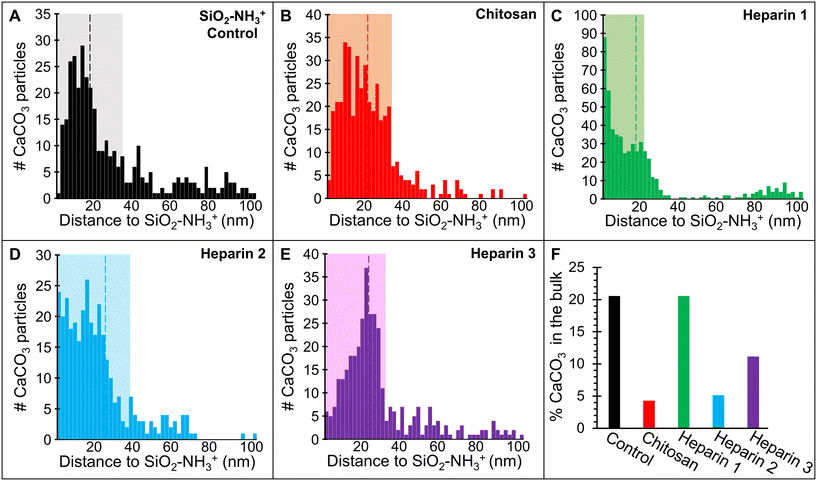
In CaCO3 supersaturated solutions, the aminated SiO2 nanoparticles (control) were enveloped in a region of solution that was darker than that of the surrounding bulk solution (outlined in white in Fig. 2B). This region presented an average width of 16 nm as measured from the nanoparticle surface to its perimeter (e.g., Fig. 2B; the distance between the yellow and white lines) with a maximum lateral extent of 34 nm (e.g., shaded area in Fig. 4A). All distances were determined by applying the measurement tool in ImageJ44 to the collected images. The increased grayscale value in this region reflects a greater mass density relative to the bulk solution composition and, thus, is indicative of ion accumulation around the charged NH3+ groups, creating a solute-rich environment. To confirm this interpretation, we conducted additional control experiments in water (without solute ions) (Fig. 3A) and did not find a darker region around the nanoparticle. This was also true for another experiment using calcite-supersaturated solutions at pH 9 (where the amines are uncharged) (Fig. 3B). In these solutions, CaCO3 crystallites formed in the bulk solution without evidence of np association (Fig. 3C). An implication of these observations is that substrate charge, including simple aminated SiO2 nanoparticles without a biopolymer treatment, can facilitate locally higher ion concentrations, and thus a potentially greater driving force for CaCO3 nucleation.
Returning to the polysaccharide-free control experiments (Fig. 2A and B), the induction time to the first appearance of CaCO3 crystals was ≈40 min. The induction time was measured as the period of time between the addition of NaHCO3 to the TEM cell (i.e., when the solution was first supersaturated, sect. 2.3) and observation of the initial CaCO3 particles. Crystals formed in the bulk solution as well as near the SiO2 nanoparticles. At ≈10 min post-induction, aggregates and larger individual crystals of CaCO3 were observed near the nanoparticles, and smaller crystals formed in the bulk solution (Fig. 2A and B). By measuring the distance from the outer edge of each SiO2–NH3+ nanoparticle to the nearest edge of each CaCO3 crystal (e.g., Fig. 2B; between the yellow and red circles), we determined that most nucleation occurred within 30 nm of the SiO2–NH3+ surface, which was similar to the estimated thickness of the electron-dense zone (Fig. 4A). Away from the nanoparticles, a significant number of CaCO3 particles (≈21%) formed in the bulk solution. However, many of these particles exhibited mobility during the first stages of nucleation.
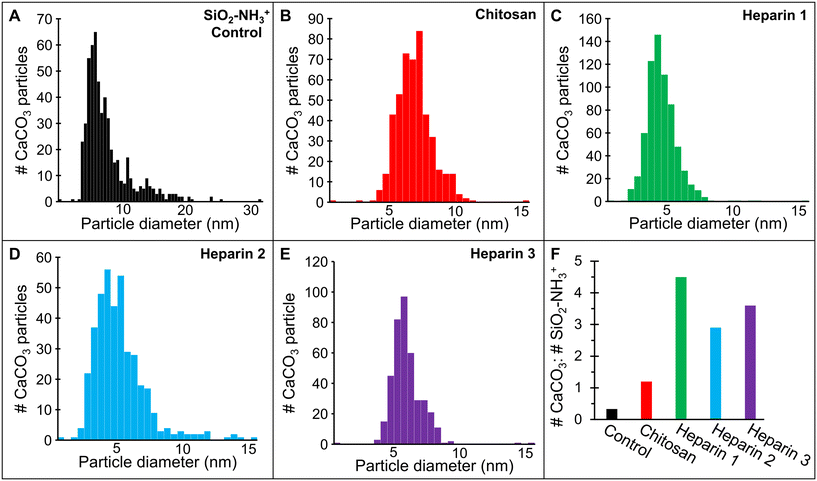
The average diameter of particles formed in the SiO2–NH3+ control experiment was 7.7 ± 3.8 nm; however, the crystal size distribution was asymmetric with a significant tail extending to larger sizes (Fig. 5A). Many particles grew to a diameter >15 nm (and up to 31 nm). There was no apparent association between particle size and distance from the nanoparticle edge. The low ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) of the total number of CaCO3 crystals to the number of silica nanoparticles within a representative viewing area indicates that the control SiO2–NH3+ particles and the associated zone do not have a strong direct nucleation ability relative to the bulk solution (Fig. 5F). This is further supported by in situ observations that the CaCO3 crystals were in motion during and after their formation, indicating that they were not bound to the aminated silica nanoparticles.
4) of the total number of CaCO3 crystals to the number of silica nanoparticles within a representative viewing area indicates that the control SiO2–NH3+ particles and the associated zone do not have a strong direct nucleation ability relative to the bulk solution (Fig. 5F). This is further supported by in situ observations that the CaCO3 crystals were in motion during and after their formation, indicating that they were not bound to the aminated silica nanoparticles.
Quantifying the position of CaCO3 crystals relative to the polysaccharide coating in the TEM liquid-cell presented challenges because the polysaccharides have a low mass density, which limited our ability to directly measure the thickness of the coating. Hence, we could not readily resolve the exact position of the crystallites relative to the polysaccharide–solution interface. The difficulty was amplified because TEM images are 2-dimensional projections of 3-dimensional features such that: 1) some distance measurements will be biased to apparent values that are smaller than the actual; 2) the measurements cannot discern the true distance of particles with positions above or overlapping the SiO2–NH3+ nanoparticles. However, the images of the polysaccharide-coated nanoparticle systems (Fig. 2C–F) show significant contrast between the CaCO3 particle-dense regions near nanoparticles and the particle-free bulk relative to the control (Fig. 2A and B). By assuming that this higher mass density region corresponds to the polysaccharide coating or interphase region and the associated ions (shaded regions in Fig. 4B–E), we were able to obtain insight into where crystals nucleated. We measured the distance from the nearest edge of each CaCO3 crystal to the outer edge of each SiO2–NH3+ nanoparticle. Most nucleation occurred <30 nm from the SiO2–NH3+ surface (Fig. 4B), which was within the estimated width of the electron-dense region and is comparable to that seen for the control.
In contrast to the control, with the chitosan-coated nanoparticles, most (96%) CaCO3 particles nucleated in close association with the polysaccharide interfacial region while very few crystals formed in the bulk solution (Fig. 2C and D and 4B and F). Moreover, the induction time to crystallization (≈25–30 min) was significantly shorter than for the control experiments. Also, the resulting CaCO3 particles were stationary, indicating they were at least weakly bound to the polysaccharide. Under these conditions, CaCO3 crystals formed with a higher number density, averaging 1.2 CaCO3 per SiO2–NH3+ nanoparticle (Fig. 5F) and a smaller average particle size with a narrower range of diameters (6.4 ± 1.3 nm, Fig. 5B) compared to the control, though the peak in the size distribution was similar for both. Few to no aggregates of CaCO3 crystals were observed.
Like chitosan, most CaCO3 crystals preferentially formed near the heparin-coated nanoparticles (80–95%) with a few in the bulk solution and they appeared at still-shorter incubation times (≈20 min) (Fig. 2E and F and 4C–F and S4). Compared to the chitosan-coated and control experiments, the number of heparin-associated CaCO3 crystals was the greatest with an average of 2.9 crystallites per SiO2–NH3+ nanoparticle (Fig. 5F). The average crystal size was smaller (diameters = 4.0 ± 1.2, 4.7 ± 2.0, and 5.4 ± 1.1 nm (Fig. 5C–E, respectively)) with an overall average of 4.6 ± 1.5 nm. These diameters are near or within estimates for the calcite critical nucleus size (1–5 nm) for the estimated supersaturation conditions of these experiments.22,48
A relationship between the particle size and distance from the SiO2–NH3+ particle edge was not observed for the heparin or chitosan experiments. Note that the three heparin trials resulted in ‘clouds’ of variable thicknesses around the nps, which roughly correlated with the distributions of the CaCO3 crystals. This suggests that heterogeneities in the thickness of heparin coating and/or the interphase environment were the primary influence on measured CaCO3 distances (e.g., Fig. 4C–E).
The smaller particle sizes, higher particle numbers, and shorter induction times measured for the chitosan and heparin systems are consistent with previous optical observations of CaCO3 crystallization in the presence of polymer additives, particularly with the addition of anionic molecules (e.g., carboxymethyl chitosan).49–51 The results are also consistent with rate measurements that determined that the kinetic term of the rate expression increases with the net charge of the polysaccharide (Fig. S5).14,15 However, it is important to remember that crystal size differences could be rooted in nucleation and/or extent of growth. Observations from this study cannot fully resolve the relative contributions between polysaccharide effects on growth rate versus nucleation. Hence, an alternative explanation is that the smaller particles may be a consequence of slower growth due to a post-nucleation reduction in supersaturation.
Recognizing the limitations of our measurements for the distance of separation between the CaCO3 crystals and SiO2–NH3+ nanoparticles, we propose that nucleation occurs in two types of environments. First, the interfacial environment within 0–40 nm from the nanoparticle surface has the primary control on crystal nucleation. Second, at distances beyond ≈40 nm, nucleation is dominated by bulk solution properties.
We acknowledge it is unlikely that the aminated silica nanoparticles alone (control) could regulate crystallization at distances of up to ≈20 nm into the solution. The Debye length for interfaces around these materials is expected to be <5 nm,52 suggesting that observed long-range effects may be a result of collective properties of the materials and solution environment rather than individual molecular level forces. This is consistent with the idea that CaCO3 nucleation is driven by water restructuring during substrate–ion interactions (i.e., entropically dominated) as opposed to coulombic forces alone.53–55 However, we observed a maximum number of CaCO3 particles at distances of 12–14 nm from a nanoparticle edge, which is well beyond the length that surfaces generate structured hydration layers (typically <2 nm as seen by 3D atomic force microscopy).56–59 Moreover, our value is much larger than those reported or predicted by molecular dynamics for other nucleating systems (1–2 nm).15,16,21,23,24
Consequently, appealing to a decrease in the interfacial energy due to such structuring as a source of enhanced nucleation is unfounded. Similarly, although electric fields can introduce an additional term in the work of cluster formation that reduces the critical size and thus increases nucleation rates,60 the distances from the functionalized particle surfaces and large ion concentrations would seem to preclude this effect as a source of enhanced nucleation. Further investigation will be required, but even if the heterogeneous distributions of Ca2+ and CO32− are such that their individual chemical potentials are uniform throughout the solution, perhaps small increases in the product of Ca2+ and CO32− activities lead to a consequent increase in local supersaturation that promotes nucleation in the solution near these nanoparticles (i.e., homogeneous nucleation).13,61–65
Irrespective of the mechanism, the larger number density of CaCO3 crystals that form in the region of the polysaccharide-coated nps compared to the bulk solution and the control experiments indicates the biopolymer-specific interactions with ions to promote nucleation. It is plausible that the greater persistence length of the heparin, which behaves as a semi-rigid polyelectrolyte, particularly under the low ionic strength conditions of these experiments,32,66 presents greater spatial organization with extended chains and thus more stable binding sites. The result is local environments that favor Ca2+ and HCO3− ion-rich regions and increase the probability of forming CaCO3 critical nuclei (e.g., ref. 15). This interpretation is consistent with our observations of shorter induction times, greater nucleation density, and similar distances between the crystallites and np surface. However, the variable CaCO3 particle distributions between samples (e.g., Fig. 4C–E) prevent a firm conclusion.
Chitosan, with its low charge and thus greater flexibility and shorter effective persistence length,31 presents a less-organized interphase region. Although this study could not resolve the structure and properties of the region, it is plausible that binding sites are weaker with greater fluctuations, resulting in fewer nucleation events compared to heparin. However, our study cannot assess if the origins of these effects on CaCO3 nucleation are kinetic (e.g., the number of possible nucleation sites,67 attachment rates, and barriers to ion binding, such as the desolvation barrier64) and/or thermodynamic (e.g., the supersaturation and interfacial free energy of forming a new crystal (γnet)). Both polysaccharides, however, yield favorable environments for nucleation near the polysaccharide–solution interface relative to the bulk solution.
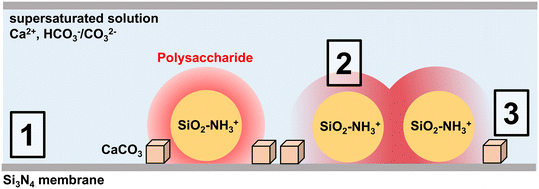
The in situ TEM images also indicate that CaCO3 crystals in the polysaccharide systems did not form directly on (e.g., separation distance = 0 nm; Fig. 5) or above the SiO2–NH3+ nps in the chitosan system, nor in two of the heparin replications. Rather, CaCO3 appears to form in definitive rings around coated nanoparticles (Fig. 2C and F). This is notable because if CaCO3 nucleates in an approximate hemisphere around the coated silica nps, the 2D projections would yield images of CaCO3 crystals overlapping with the silica substrates at all distances. A plausible explanation for this observation is that CaCO3 crystals nucleate near the boundary between the solution, the interphase region of polysaccharide coating, and the silicon nitride (Si3N4) membrane(s) that comprise the TEM liquid-cell. That is, crystals preferentially form at the polysaccharide–TEM membrane–solution interface. This scenario is supported by the fact that the particles do not undergo Brownian motion, implying that, like most objects imaged in liquid phase TEM, they exhibit greatly reduced mobility as a consequence of being in the vicinity of the membrane. Previous measurements show that γnet for nucleating calcite is the largest for highly charged polysaccharides (e.g., heparin).14,15 This insight, combined with observations of the preferred crystal placements, would seem to suggest that introducing the TEM cell membrane to the polysaccharide–solution system reduces γnet and/or increases the activity product or kinetic factors of the nucleation rate expression at the boundary of the materials (i.e., at the polysaccharide–membrane–solution interface).
Observations in this study suggest a model for how polysaccharides influence the placement of CaCO3 crystals during nucleation. Fig. 6 illustrates three types of interfaces in the TEM liquid-cell for our polysaccharide systems: 1) the Si3N4 membrane–solution interface that comprises the fluid in situ TEM cell, 2) polysaccharide–solution interface, and 3) polysaccharide–TEM membrane–solution interface. The γnet, ion distributions, and kinetic factors for each system are thus a collective outcome of the associated interfaces.
Conclusions
The findings from this in situ experimental study show that polysaccharide–water interfaces promote CaCO3 nucleation when compared to the aminated SiO2 nanoparticle control. We were unable to resolve detailed spatial relationships between the interfacial structure and crystal placement, but the measurements indicate that the number and position of the resulting crystals is likely related to polysaccharide-specific properties near the solution-interphase region. Heparin and chitosan present differences in net charge to the solution environment, and heparin is the most effective at controlling the placement and abundance of CaCO3 crystallites. This result is consistent with heparin's larger effective persistence length that stabilizes ion-rich regions.For both macromolecules, chitosan and heparin, heterogeneous nucleation is most-favored at the polysaccharide–TEM Si3N4 membrane contact, possibly through an additional influence of the TEM membrane on interfacial energy, ion concentration, and/or kinetic factors. The properties of the polysaccharides—through their interactions with local waters of solvation in these disrupted settings—directly influence where CaCO3 nucleation occurs. The charged interface of the amine-functionalized silica nps leads to surrounding ‘clouds’ of high ion concentration, but this environment does not appear to have a structuring influence on the placement of CaCO3 nuclei or the resulting crystal size relative to our observations for the polysaccharide interfaces. Thus, the evidence suggests that CaCO3 nucleation in the control system is best described as a homogeneous process. The findings reiterate the need for a stronger mechanistic understanding of how the functional groups influence local polysaccharide properties and structuring to control the timing and placement of crystal nuclei during mineralization.
Author contributions
B. M. K., B. J., Y. B., J. J. D. Y., and P. M. D. designed the research; B. M. K., B. J., and Y. B. performed the research; B. M. K., B. J., Y. B., J. J. D. Y., and P. M. D. analyzed the data; and B. M. K., J. J. D. Y., and P. M. D. wrote the paper.Conflicts of interest
There are no conflicts of interest to declare.Data availability
Data for this article, including TEM images and CaCO3 particle analysis, are available at Open Science Framework (OSF) at https://doi.org/10.17605/OSF.IO/KHJX6. Supplementary information (SI): which includes ATR data of polysaccharide coated nanoparticles, a schematic of the TEM setup, SAED pattern and ex situ images, TEM images for three heparin experiments, and reported measurments of calcite nucleation kinetics. Supplementary experiments using different solution conditions (crosslinked polysaccharides (no nanoparticles) and high ionic strength (IS = 0.6 M)) are also included in the SI. See DOI: https://doi.org/10.1039/d6ce00012f.Acknowledgements
This material is based on the work supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Geosciences program, with material preparation performed at Virginia Tech under Award Number DE FG02-00ER15112 (to P. M. D.) and in situ TEM performed at Pacific Northwest National Laboratory (PNNL) (to J. J. D. Y.) under FWP 56674. TEM experiments were performed on a project award (60753) from the Environmental Molecular Sciences Laboratory (EMSL), a DOE Office of Science User Facility sponsored by the Biological and Environmental Research program under Contract No. DE-AC05-76RL01830. PNNL is a multiprogram national laboratory operated for the DOE by Battelle Memorial Institute under Contract DE-AC05-76RL01830. We thank Alan Esker and Kevin Edgar for valuable discussions of polysaccharide behavior in aqueous solution.References
- L. Addadi, J. Moradian, E. Shay, N. G. Maroudas and S. Weiner, Proc. Natl. Acad. Sci. U. S. A., 1987, 84, 2732–2736 CrossRef CAS PubMed.
- L. Addadi, D. Joester, F. Nudelman and S. Weiner, Chem. – Eur. J., 2006, 12, 981–987 CrossRef PubMed.
- R. Fried and Y. Mastai, J. Cryst. Growth, 2012, 338, 147–151 CrossRef CAS.
- A. Gal, R. Wirth, Z. Barkay, N. Eliaz, A. Scheffel and D. Faivre, Chem. Commun., 2017, 53, 7740–7743 RSC.
- H. Pyles, S. Zhang, J. J. De Yoreo and D. Baker, Nature, 2019, 571, 251–256 CrossRef CAS PubMed.
- S. E. Gleeson, S. Kim, T. Yu, M. Marcolongo and C. Y. Li, ACS Appl. Bio Mater., 2022, 5, 4493–4503 CrossRef CAS PubMed.
- H. A. Lowenstam and S. Weiner, On Biomineralization, Oxford University Press, 1989 Search PubMed.
- J. L. Arias, A. Neira-Carrillo, J. I. Arias, C. Escobar, M. Bodero, M. David and M. S. Fernandez, J. Mater. Chem., 2004, 14, 2154–2160 RSC.
- D. Raabe, C. Sachs and P. Romano, Acta Mater., 2005, 53, 4281–4292 CrossRef CAS.
- J. L. Arias and M. S. Fernandez, Chem. Rev., 2008, 108, 4475–4482 CrossRef CAS PubMed.
- A. Gal, R. Wirth, J. Kopka, P. Fratzl, D. Faivre and A. Scheffel, Science, 2016, 353, 590–593 CrossRef CAS PubMed.
- J. L. Huang, Y. J. Liu, C. Liu, L. P. Xie and R. Q. Zhang, Int. J. Biol. Macromol., 2021, 189, 641–648 CrossRef CAS PubMed.
- B. M. Knight, K. J. Edgar, J. J. De Yoreo and P. M. Dove, Biomacromolecules, 2023, 24, 1078–1102 CrossRef CAS PubMed.
- A. J. Giuffre, L. M. Hamm, N. Han, J. J. De Yoreo and P. M. Dove, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 9261–9266 CrossRef CAS PubMed.
- B. M. Knight, R. Mondal, N. Han, N. F. Pietra, B. A. Hall, K. J. Edgar, V. Vaissier Welborn, L. A. Madsen, J. J. De Yoreo and P. M. Dove, Cryst. Growth Des., 2024, 24, 6338–6353 CrossRef CAS PubMed.
- H. J. Li, D. Yan, H. Q. Cai, H. B. Yi, X. B. Min and F. F. Xia, Phys. Chem. Chem. Phys., 2017, 19, 11390–11403 RSC.
- X. W. Wang, D. Toroz, S. Kim, S. L. Clegg, G. S. Park and D. Di Tommaso, Phys. Chem. Chem. Phys., 2020, 22, 16301–16313 RSC.
- R. G. Alberstein, J. L. Prelesnik, E. Nakouzi, S. Zhang, J. J. De Yoreo, J. Pfaendtner, F. A. Tezcan and C. J. Mundy, J. Phys. Chem. Lett., 2023, 14, 80–87 CrossRef CAS PubMed.
- J.-N. Boyn and E. A. Carter, J. Am. Chem. Soc., 2023, 145, 20462–20472 CrossRef CAS PubMed.
- J. N. Boyn and E. A. Carter, J. Phys. Chem. B, 2023, 127, 10824–10832 CrossRef CAS PubMed.
- F. A. Davila-Hernandez, B. Jin, H. Pyles, S. Zhang, Z. Wang, T. F. Huddy, A. K. Bera, A. Kang, C. L. Chen, J. J. De Yoreo and D. Baker, Nat. Commun., 2023, 14, 8191 CrossRef PubMed.
- H. Söngen, S. J. Schlegel, Y. Morais Jaques, J. Tracey, S. Hosseinpour, D. Hwang, R. Bechstein, M. Bonn, A. S. Foster, A. Kühnle and E. H. G. Backus, J. Phys. Chem. Lett., 2021, 12, 7605–7611 CrossRef PubMed.
- Y. W. Cheng, J. H. Tao, G. M. Zhu, J. A. Soltis, B. A. Legg, E. Nakouzi, J. J. De Yoreo, M. L. Sushko and J. Liu, Nanoscale, 2018, 10, 11907–11912 RSC.
- G. M. Zhu, M. L. Sushko, J. S. Loring, B. A. Legg, M. Song, J. A. Soltis, X. P. Huang, K. M. Rosso and J. J. De Yoreo, Nature, 2021, 590, 416–422 CrossRef CAS PubMed.
- C. Zeng, C. Vitale-Sullivan and X. Ma, Minerals, 2017, 7, 158 CrossRef.
- S. Pu, C. Gong and A. W. Robertson, R. Soc. Open Sci., 2020, 7, 191204 CrossRef CAS PubMed.
- M. V. Shapovalov and R. L. Dunbrack Jr, Proteins: Struct., Funct., Bioinf., 2007, 66, 279–303 CrossRef CAS PubMed.
- M. Kurudirek and T. Onaran, Radiat. Phys. Chem., 2015, 112, 125–138 CrossRef CAS.
- B. Cuevas-Zuviría and L. F. Pacios, J. Chem. Inf. Model., 2020, 60, 3831–3842 CrossRef PubMed.
- P. J. Flory, Principles of polymer chemistry, Cornell University Press, Ithaca, N.Y., 1953 Search PubMed.
- G. A. Morris, J. Castile, A. Smith, G. G. Adams and S. E. Harding, Carbohydr. Polym., 2009, 76, 616–621 CrossRef CAS.
- G. Pavlov, S. Finet, K. Tatarenko, E. Korneeva and C. Ebel, Eur. Biophys. J., 2003, 32, 437–449 CrossRef CAS PubMed.
- H. C. Brown, D. H. McDaniel and O. HÄFliger, in Determination of Organic Structures by Physical Methods, ed. E. A. Braude and F. C. Nachod, Academic Press, 1955, pp. 567–662 Search PubMed.
- T. Schiestel, H. Brunner and G. E. M. Tovar, J. Nanosci. Nanotechnol., 2004, 4, 504–511 CrossRef CAS PubMed.
- M. Rinaudo, Prog. Polym. Sci., 2006, 31, 603–632 CrossRef CAS.
- Q. Wang, X. Chen, N. Liu, S. Wang, C. Liu, X. Meng and C.-G. Liu, Carbohydr. Polym., 2006, 65, 194–201 CrossRef CAS.
- B. M. Knight, C. M. B. Gallagher, M. D. Schulz, K. J. Edgar, C. D. McNaul, C. A. McCutchin and P. M. Dove, Proc. Natl. Acad. Sci. U. S. A., 2025, 122, e2504348122 CrossRef CAS PubMed.
- J. W. Park and B. Chakrabarti, Biochem. Biophys. Res. Commun., 1977, 78, 604–608 CrossRef CAS PubMed.
- H. M. Wang, D. Loganathan and R. J. Linhardt, Biochem. J., 1991, 278, 689–695 CrossRef CAS PubMed.
- V. R. Sinha, A. K. Singla, S. Wadhawan, R. Kaushik, R. Kumria, K. Bansal and S. Dhawan, Int. J. Pharm., 2004, 274, 1–33 CrossRef CAS PubMed.
- N. Reddy, R. Reddy and Q. R. Jiang, Trends Biotechnol., 2015, 33, 362–369 CrossRef CAS PubMed.
- L. N. Plummer and E. Busenberg, Geochim. Cosmochim. Acta, 1982, 46, 1011–1040 CrossRef CAS.
- C. M. Bethke, The Geochemist's Workbench, Release 3.0 edn, 1998 Search PubMed.
- C. A. Schneider, W. S. Rasband and K. W. Eliceiri, Nat. Methods, 2012, 9, 671–675 CrossRef CAS PubMed.
- B. Jin, Y. Chen, H. Pyles, M. Baer, B. Legg, Z. Wang, N. Washton, K. Mueller, D. Baker, G. Schenter, C. Mundy and J. De Yoreo, Nat. Mater., 2025, 24, 125–132 CrossRef CAS PubMed.
- Z. Liu, Z. Zhang, Z. Wang, B. Jin, D. Li, J. Tao, R. Tang and J. De Yoreo, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 3397–3404 CrossRef CAS PubMed.
- P. Smeets, A. Finney, W. Habraken, F. Nudelman, H. Friedrich, J. Laven, J. De Yoreo, P. Rodger and N. Sommerdijk, Proc. Natl. Acad. Sci. U. S. A., 2017, 114, E7882–E7890 CrossRef CAS PubMed.
- Q. Hu, M. H. Nielsen, C. L. Freeman, L. M. Hamm, J. Tao, J. R. I. Lee, T. Y. J. Han, U. Becker, J. H. Harding, P. M. Dove and J. J. De Yoreo, Faraday Discuss., 2012, 159, 509–523 RSC.
- P. Liang, Y. Zhao, Q. Shen, D. J. Wang and D. F. Xu, J. Cryst. Growth, 2004, 261, 571–576 CrossRef CAS.
- Y. P. Huang, Q. Shen, W. P. Sui, M. L. Guo, Y. Zhao, D. J. Wang and D. F. Xu, Colloid Polym. Sci., 2007, 285, 641–647 CrossRef CAS.
- M. E. Fortuna, E. Ungureanu and C. D. Jitareanu, Materials, 2021, 14, 3336 CrossRef CAS PubMed.
- J. N. Israelachvili, Intermolecular and Surface Forces, Elsevier, 3rd edn, 2011 Search PubMed.
- C. G. Sinn, R. Dimova and M. Antonietti, Macromolecules, 2004, 37, 3444–3450 CrossRef CAS.
- M. Kellermeier, P. Raiteri, J. K. Berg, A. Kempter, J. D. Gale and D. Gebauer, ChemPhysChem, 2016, 17, 3535–3541 CrossRef CAS PubMed.
- V. V. Welborn, W. R. Archer and M. D. Schulz, J. Chem. Inf. Model., 2023, 63, 2030–2036 CrossRef PubMed.
- D. Martin-Jimenez, E. Chacon, P. Tarazona and R. Garcia, Nat. Commun., 2016, 7, 12164 CrossRef CAS PubMed.
- H. Söngen, R. Bechstein and A. Kühnle, J. Phys.: Condens. Matter, 2017, 29, 274001 CrossRef PubMed.
- E. Nakouzi, S. Kerisit, B. A. Legg, S. Yadav, D. Li, A. G. Stack, C. J. Mundy, J. Chun, G. K. Schenter and J. J. De Yoreo, J. Phys. Chem. C, 2023, 127, 2741–2752 CrossRef CAS.
- M. Zhang, B. A. Legg, B. A. Helfrecht, Y. Zhang, S. Tan, Y. Xia, R. K. Yodong, M. Iepure, V. Prabhakaran, P. J. Pauzauskie, Y. Min, C. J. Mundy and J. J. De Yoreo, Nat. Mater., 2026, 25, 487–494 CrossRef CAS PubMed.
- L. F. Alexander and N. Radacsi, CrystEngComm, 2019, 21, 5014–5031 RSC.
- A. Chernov, Phys.-Usp., 1961, 4, 116–148 CrossRef.
- A. Chernov, Modern Crystallography III: Crystal Growth, Springer, Berlin, 1984 Search PubMed.
- C. A. DiTusa, T. Christensen, K. A. McCall, C. A. Fierke and E. J. Toone, Biochemistry, 2001, 40, 5338–5344 CrossRef CAS PubMed.
- J. J. De Yoreo and P. G. Vekilov, Rev. Mineral. Geochem., 2003, 54, 57–93 CrossRef CAS.
- D. N. Petsev, K. Chen, O. Gliko and P. G. Vekilov, Proc. Natl. Acad. Sci. U. S. A., 2003, 100, 792–796 CrossRef CAS PubMed.
- B. A. Khorramian and S. S. Stivala, Arch. Biochem. Biophys., 1986, 247, 384–392 CrossRef CAS PubMed.
- D. Kashchiev and G. M. van Rosmalen, Cryst. Res. Technol., 2003, 38, 555–574 CrossRef CAS.
- D. Laage, T. Elsaesser and J. T. Hynes, Chem. Rev., 2017, 117, 10694–10725 CrossRef CAS PubMed.
- A. F. Wallace, J. J. DeYoreo and P. M. Dove, J. Am. Chem. Soc., 2009, 131, 5244–5250 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |