Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
2-Pyridone ligands as a design platform for palladium-catalyzed C(sp2)–H functionalization reactions
Namrata
Kotwal
,
Deepak
Sharma
and
Claire
Empel
 *
*
Martin-Luther-University Halle-Wittenberg Institute of Chemistry, Kurt-Mothes-Str. 2, 06120 Halle (Saale), Germany. E-mail: claire.empel@chemie.uni-halle.de
First published on 10th April 2026
Abstract
2-Pyridone ligands have emerged as uniquely powerful promoters in transition-metal-catalyzed C–H functionalization, enabling transformations that are often inaccessible with conventional ligand classes. Despite their growing impact, their role is frequently treated only implicitly within broader ligand surveys. This review provides a focused analysis of 2-pyridone ligands as a distinct ligand design platform, emphasizing how they actively participate in C–H activation. We highlight the mechanistic roles of 2-pyridone ligands in key elementary steps, including concerted metalation–deprotonation, weak and dynamic metal coordination, and stabilization of reactive catalytic intermediates. Particular attention is devoted to structure–function relationships, illustrating how substitution patterns on the 2-pyridone scaffold govern basicity, coordination behavior, reactivity, and selectivity in C(sp2)–H functionalization. By extracting unifying design principles from diverse catalytic systems, this review aims to provide practical guidelines for the rational development of next-generation 2-pyridone ligands, with relevance to C–H functionalization.
1. Introduction
Transition-metal-catalyzed C–H functionalization has emerged as a powerful strategy for molecular diversification, enabling the selective transformation of otherwise inert C–H bonds into C–C and C–X (heteroatom) bonds.1–4 Two general approaches have been developed to control site selectivity: directed and non-directed C–H functionalization. Directed strategies employ strongly coordinating directing groups to position the metal catalyst proximal to a specific C–H bond, thereby enabling selective activation. However, this approach often requires additional steps for installation and removal of the directing group.5–12 More recently, the research area of directed C–H functionalization was further expanded towards visible-light-mediated approaches (Scheme 1).3,13 In contrast, non-directed C–H functionalization avoids permanent directing groups and thus offers a more step-economical alternative. Nevertheless, these reactions remain challenging due to weak substrate–metal interactions, the high intrinsic barrier associated with C–H bond cleavage, and the difficulty of achieving high site selectivity. Consequently, overcoming these limitations represents a central objective in the development of non-directed C(sp2)–H activation chemistry (Scheme 1).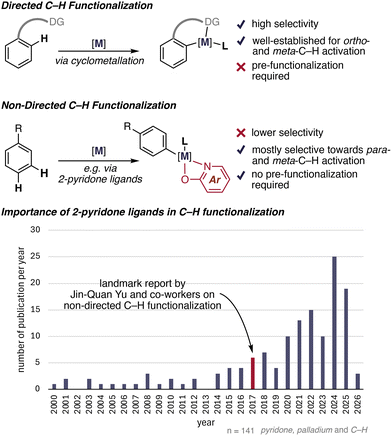 | ||
| Scheme 1 Strategic approaches to site-selective Pd-catalyzed C(sp2)–H activation and the importance of 2-pyridone ligands in non-directed C–H functionalization. | ||
A key conceptual advance in the area of non-directed C–H activation was the recognition that specialized ligands can participate in accelerating C–H bond cleavage rather than merely serving as spectator ligands.14–17 In a seminal contribution, Yu and co-workers demonstrated ligand-accelerated non-directed C(sp2)–H functionalization enabled by electron-deficient 2-pyridone ligands, establishing ligand assistance as an efficient strategy for lowering the C–H activation barrier without relying on permanent directing groups.18 These findings highlighted that carefully designed, weakly coordinating yet functionally active ligands can directly participate in elementary steps of the catalytic cycle, thereby determining the feasibility of arene-limited reactions.
Therefore, in recent years, ligand design has emerged as a key strategy to address the reactivity and selectivity challenges associated with non-directed C–H functionalization. Apart from 2-pyridone ligands, the use of dual-ligand catalytic systems has proven highly effective in enabling palladium-catalyzed non-directed C(sp2)–H activation. Pioneering studies by the van Gemmeren group demonstrated that combinations of pyridine ligands in combination with N-acetylglycine and derivatives thereof can significantly enhance catalytic activity and enable efficient non-directed arene functionalization.19–22 Related advances from the Fernández-Ibáñez23–25 and Čorić26,27 groups further underpin the power of dual-ligand approaches to finetune the steric and electronic properties needed for high selectivity and reactivity. While these pioneering dual-ligand studies have significantly advanced non-directed C–H functionalization, this review will focus exclusively on the development and application of 2-pyridone ligands and for a broader perspective on non-directed C–H functionalization previous reviews could be of interest to the reader.14–17
2-Pyridone ligands have proven particularly compelling as enabling design elements in palladium-catalyzed C–H activation. The unique structural properties of 2-pyridone scaffold namely tautomerism, tunable basicity, and weak but flexible coordination ability within a compact heterocycle, distinguishes it from classical neutral donors and permanently anionic ligand frameworks. Importantly, 2-pyridone-based ligands frequently enable reactivity that is inaccessible with conventional ligand classes, underscoring their unique capacity to promote challenging non-directed C(sp2)–H activation processes through cooperative ligand participation.28–30
Despite the growing number of transformations employing 2-pyridone ligands, a rational design principle remains shaded. In many studies, 2-pyridones are identified through empirical optimization as optimal ligands, while questions regarding coordination mode, proton-transfer capability, and the influence of substitution patterns are addressed only implicitly. Existing literature studies mainly rely on C–H functionalization across multiple ligand families, leaving the structure–function relationships that govern the distinctive behavior of 2-pyridone ligands not yet fully elucidated.
In this review, we focus on 2-pyridone ligands as a distinct and mechanistically active ligand design platform for transition-metal-catalyzed C(sp2)–H functionalization reactions. Emphasis is primarily laid on understanding how the intrinsic features associated with 2-pyridone scaffold govern coordination behavior, proton transfer, and catalytic turnover in non-directed C(sp2)–H functionalization. By consolidating insights from studies that explicitly use 2-pyridone ligands, this review aims to distill how structure–function relationships and ligand design principles can guide the rational development of pyridone-enabled catalytic systems for C(sp2)–H functionalization reactions.
2. Structural and electronic features of 2-pyridone ligands
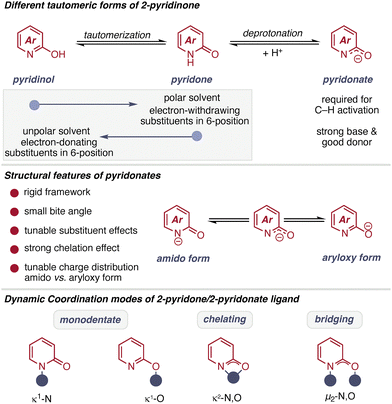
2-Pyridone ligands represent a highly versatile class of N,O-coordinating scaffolds that are particularly well suited for C–H activation and functionalization chemistry. Their attractiveness arises from a unique combination of electronic tunability, rigid backbone geometry, defined bite angle, and adaptable charge distribution. The heteroaromatic core provides a rigid backbone, which enforces a well-defined spatial arrangement of the nitrogen and oxygen donor atoms. This rigidity typically results in a relatively tight bite angle upon chelation to a metal center, a feature that can strongly influence metal coordination geometry and, consequently, catalytic reactivity. Compared to more flexible bidentate ligands, 2-pyridone-derived systems often impose greater geometric constraint, which can facilitate key steps such as concerted metalation–deprotonation (CMD) in C–H activation processes.30,31A central advantage of the 2-pyridone scaffold is its modular substituent pattern. Substituents can be introduced at various ring positions to systematically tune steric and electronic properties. Electron-withdrawing groups decrease electron density at the donor atoms and increase ligand acidity, whereas electron-donating substituents enhance donor strength. Substitution at the 6-position is particularly impactful, as it is proximal to the coordinating heteroatoms and therefore directly influences both steric environment and metal–ligand bond strength.32
Beyond these geometric and electronic features, a defining feature that distinguishes 2-pyridone ligands from many other ligand classes, is its ability to possess an additional defining characteristic that sets them apart from many conventional ligand frameworks: their ability to undergo tautomeric interconversion with the corresponding 2-hydroxypyridine form (Scheme 2).33–35 This tautomerism introduces a dynamic acid–base element into the ligand scaffold, enabling reversible protonation and internal-base behavior that can directly assist key elementary steps such as proton transfer during CMD-type C–H activation.
The reactivity of 2-pyridone ligands originates from the interplay between tautomerism, deprotonation, and coordination flexibility. The scaffold exists in equilibrium between the 2-hydroxypyridine and 2-pyridone forms, while deprotonation of the 2-pyridone form generates the corresponding pyridonate species. This proton-responsive manifold directly modulates donor properties, charge distribution, and binding mode. In the neutral pyridone form, coordination typically proceeds via N-donation, whereas deprotonation enhances O-donor character and introduces anionic X-type behavior. The resulting pyridonate exhibits increased chelation strength and altered electronic communication with the metal center.
The rigid heteroaromatic backbone enforces a defined spatial arrangement of the N and O donor atoms, leading to a relatively small bite angle in the chelating mode (Scheme 2). This geometric constraint, combined with strong N,O-chelation and tunable substituent effects, enables precise control over metal coordination geometry. Beyond classical bidentate chelation, 2-pyridone ligands can also adopt monodentate, chelating, or bridging coordination modes. Such adaptability allows the ligand to stabilize different catalytic intermediates and, where required, to mediate metal–metal interactions in multinuclear assemblies.36,37
The N,O-donor set confers hemilabile character, enabling reversible donor dissociation to generate transient open coordination sites while preserving complex stability. This dynamic flexibility promotes substrate binding and turnover without full ligand displacement. Moreover, the pyridone/pyridonate unit enables metal–ligand cooperativity (MLC), functioning as a Lewis base through coordination and as a Brønsted base upon deprotonation. This dual reactivity facilitates proton transfer and charge redistribution, directly assisting CMD-type C–H activation and underscoring the ligand's active role in C–H bond activation. Collectively, these interrelated features—tautomerism, dynamic coordination modes, hemilability, and electronic programmability—establish 2-pyridone ligands as adaptable, cooperative frameworks ideally suited for C–H functionalization catalysis.
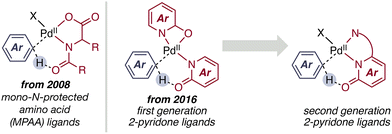
Mechanistic understanding of these cooperative features has driven the evolution from first-Generation pyridone ligands—largely demonstrating great activity in C–H activation. Following rational design principles second-generation systems relying on additional coordinating groups have been developed (Scheme 3). Strategic incorporation of electron-withdrawing substituents enhances ligand acidity and CMD competence, while substitution proximal to the coordinating heteroatoms fine-tunes steric environment, bite angle, and metal–ligand bond strength. This progression marks a shift from empirical ligand optimization toward mechanism-guided design, establishing 2-pyridones as electronically programmable and cooperatively active platforms for C–H activation catalysis.4,14,38
Having delineated the evolution of first- and second-generation 2-pyridone ligands, attention now turns to their impact across specific C–H functionalization manifolds. Examination of individual reaction classes enables a more detailed assessment of how electronic and steric modulation of the pyridone scaffold translates into reactivity and selectivity outcomes. The following sections therefore contextualize 2-pyridone ligands within key transformations, highlighting recurring mechanistic features and transformation-specific effects that collectively define their role in palladium-catalyzed C(sp2)–H activation.
3. Non-directed C(sp2)–H olefination reactions
3.1. C(sp2)–H olefination with activated olefins
C(sp2)–H olefination constitutes the reaction manifold in which 2-pyridone ligands most convincingly emerged as determining elements of palladium catalysis. The landmark report in 2017 by Jin-Quan Yu and co-workers fundamentally altered the landscape of non-directed arene functionalization by demonstrating that electron-deficient 2-pyridone ligands were not simply beneficial but essential for productive turnover under arene-limited conditions.18 In the absence of suitably tuned pyridones, catalysis either stalled or proceeded with dramatically diminished efficiency, whereas conventional neutral donors and simple carboxylate systems proved ineffective (Scheme 4). Such binary ligand dependence provides compelling evidence that the 2-pyridone scaffold directly engages the turnover-limiting C–H cleavage step.Mechanistic interrogation of these systems strongly supports a ligand-assisted CMD pathway in which the pyridone unit operates as an internal, proton-responsive base. Electron-withdrawing substituents enhance ligand acidity and reinforce transition-state organization for proton abstraction (TS1), thereby lowering the energetic barrier for C–H cleavage. The correlation between increasing ligand electron deficiency and enhanced reactivity is strikingly systematic, underscoring that C–H activation is not merely influenced—but orchestrated—by the electronic character of the pyridone scaffold. The preservation of the N–H motif across highly active systems further substantiates its participation as a proton shuttle within a cooperative metal–ligand framework.
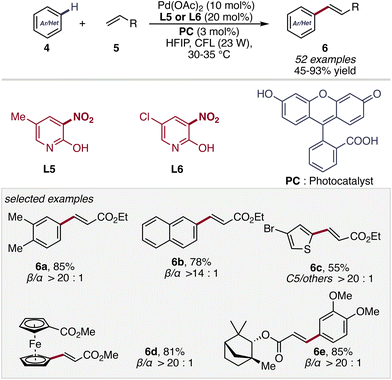
To enable milder reaction conditions the C(sp2)-H olefination was further developed by Maiti and co-workers applying a photocatalyst.39
In this work, the substitution of the 2-pyridone core on the 3-position with a nitro group is crucial to enable a high β-selectivity (>25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 β/α for o-xylene) and good yielding reaction (Scheme 5). Ligand omission leads to marked erosion of both reactivity and selectivity. Mechanistically, the pyridone operates as a proton-responsive internal base within a ligand-assisted CMD manifold, transiently engaging the C–H bond through cooperative metal–ligand interaction. Its electron-deficient character enhances acidity and stabilizes the developing charge in the transition state, while the preserved N–H motif enables dynamic proton shuttling during C–H scission. By lowering the intrinsic barrier to metalation and organizing the transition state, pyridones shift control from substrate bias to catalyst design—establishing ligand-enabled C–H activation as a fundamentally catalyst-governed process.
1 β/α for o-xylene) and good yielding reaction (Scheme 5). Ligand omission leads to marked erosion of both reactivity and selectivity. Mechanistically, the pyridone operates as a proton-responsive internal base within a ligand-assisted CMD manifold, transiently engaging the C–H bond through cooperative metal–ligand interaction. Its electron-deficient character enhances acidity and stabilizes the developing charge in the transition state, while the preserved N–H motif enables dynamic proton shuttling during C–H scission. By lowering the intrinsic barrier to metalation and organizing the transition state, pyridones shift control from substrate bias to catalyst design—establishing ligand-enabled C–H activation as a fundamentally catalyst-governed process.
Building upon these advances in non-directed C(sp2)–H olefination, in 2024, the Yu group further demonstrated ligand-controlled switchable site selectivity in the olefination of silyl-protected phenols (Scheme 6).14 By modulating palladium engagement with competing C–H bonds, they demonstrated that electron-deficient monodentate 2-pyridone ligands promote conventional para-selectivity, largely reflecting intrinsic substrate bias. In striking contrast, a dual-ligand system a bidentate pyridine–pyridone (second generation of 2-pyridone ligands) and a monodentate pyridine framework inverts this preference, enabling meta-selective functionalization in the absence of a directing group.
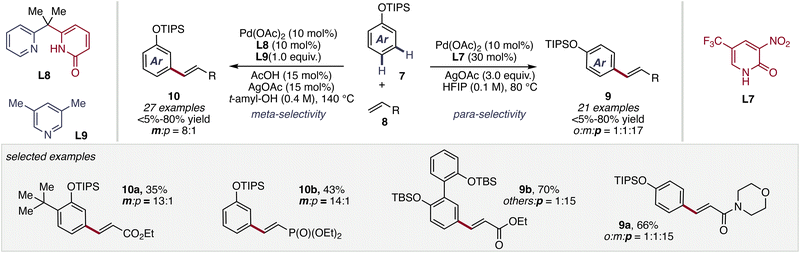 | ||
| Scheme 6 Selective para- and meta-C–H activation of silyl-protected phenols by ligand design by Yu et al. | ||
This switchable selectivity highlights that the pyridone scaffold actively reorganizes the metal–substrate interaction during the C–H activation step. The capacity to dictate both reactivity and regioselectivity through ligand design represents a level of catalyst control previously achievable primarily through covalently attached directing groups. Collectively, these studies define a coherent evolution: 2-pyridone ligands function as mechanistic regulators that modulate transition-state organization, proton abstraction dynamics, and palladium coordination geometry. In doing so, they transform non-directed C–H activation from a substrate-governed process into a catalyst-programmed event, firmly establishing ligand-enabled CMD as a central paradigm in modern C–H functionalization chemistry.
3.2. C(sp2)–H olefination using fluorinated substrates
Fluorinated motifs play a central role in medicinal chemistry and materials science due to their pronounced effects on molecular properties.40–42 Accordingly, C(sp2)–H olefination with fluorinated olefins offers a direct route to valuable fluorinated alkenyl arenes. However, these transformations are challenging, as electron-deficient fluorinated olefins display reduced reactivity in migratory insertion and can perturb catalyst coordination and turnover. Despite these limitations, their distinct electronic properties can enable complementary reactivity and provide streamlined access to functionalized fluorinated scaffolds.In 2019, Yu and co-workers demonstrated the application of ethenesulfonyl fluoride (11) in the palladium-catalyzed synthesis of β-arylethenesulfonyl fluorides 12 (Scheme 7).43 Their study revealed that catalyst systems lacking pyridone ligands displayed severely diminished reactivity yielding product in <10%, whereas the introduction of carefully tuned pyridones (electron-deficient, L10) restored productive turnover and enabled the formation of desired product 12. Interestingly ligand L1 proved less selective and provided the desired product 12 in reduced yield underlining the importance of minuscule differences of the electronic structure of the 2-pyridine ligands.
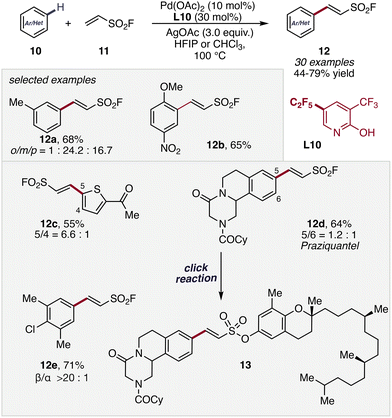 | ||
| Scheme 7 Synthesis of β-arylethenesulfonyl fluorides via non-directed C–H functionalization by Yu et al. | ||
Interestingly, the resulting β-arylethenesulfonyl fluoride products (12d) serve as versatile SuFEx click chemistry handles, enabling rapid assembly of complex bioactive conjugates such as praziquantel–tocopherol hybrids (13), thereby highlighting the potential of this pyridone-enabled C–H olefination strategy for late-stage diversification of pharmaceutically relevant molecules.44–46
A complementary advance was reported by the Empel group in 2025. In their study α-trifluoromethyl substituted styrenes were explored in the reaction with naphthalene and other simple arenes broadening the applicability of non-directed C–H functionalization towards 1,1-disubstituted olefines (Scheme 8).47
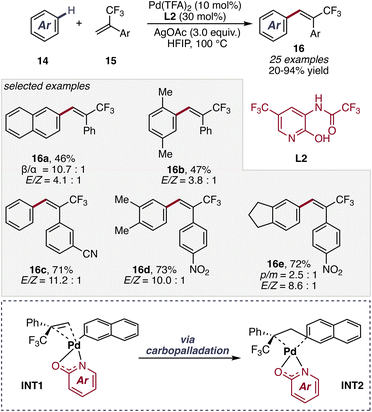 | ||
| Scheme 8 α-Trifluoromethyl substituted styrenes in non-directed C–H functionalization reactions of simple arenes by Empel et al. | ||
Ligand variation experiments revealed a marked decrease in β/α selectivity when the pyridone coordination motif was disrupted, highlighting the critical role of 2-pyridone coordination in organizing the C–H activation transition state and governing regioselectivity. Further mechanistic investigations, including kinetic studies and isotope labeling, support a catalytic cycle initiated by dissociation of the trimeric palladium precursor to a monomeric Pd(II) species, followed by coordination of substrate (14) and selectivity-determining C–H activation via a ligand-assisted CMD transition state to give INT1. Subsequent coordination of the olefin enables carbopalladation to generate a Pd–alkyl intermediate INT2, which undergoes β-hydride elimination to furnish the desired product 16 predominantly in the E-configuration, likely stabilized by favorable π–π interactions between the aryl fragments in the transition state (Scheme 8). Finally, AgOAc-mediated reoxidation of Pd(0) regenerates the active Pd(II) catalyst, completing the catalytic cycle.
3.3. 2-Pyridone ligands in C(sp2)–H olefination of N-heterocycles
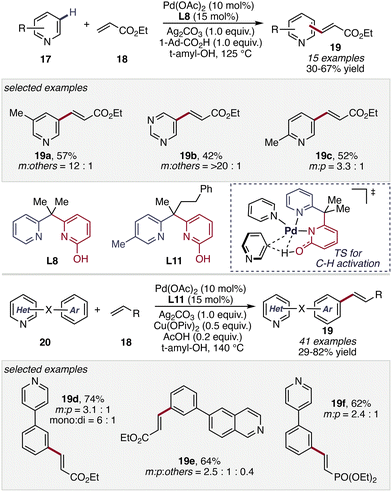
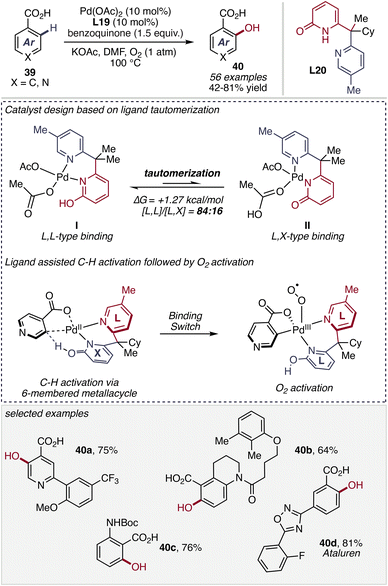
The non-directed C–H activation of N-heterocycles remains a significant challenge despite its considerable synthetic importance. Compared to simple arenes, N-heterocycles are often intrinsically less reactive due to their electron-deficient character. The presence of Lewis basic nitrogen atoms can result in strong coordination to transition metal catalysts, leading to catalyst deactivation or unproductive binding modes.48 In the absence of directing groups, these issues are compounded by the inherent difficulty of achieving both sufficient reactivity and regioselectivity, often requiring highly active catalysts or forcing conditions and frequently resulting in low efficiency or poor selectivity. Nevertheless, the development of non-directed strategies is highly desirable, as they enable direct functionalization of heterocycles without pre-installed directing groups, thereby improving step economy and providing access to substitution patterns inaccessible by conventional directed approaches.Overcoming these challenges, a significant advance was reported by Yu and co-workers in 2023, who demonstrated the power of pyridone-based ligand design in non-directed C–H activation. In this study, electron-deficient pyridine–2-pyridone ligands (L8 and L11) enabled efficient C(sp2)–H olefination of diverse N-heteroarenes (17, 20) under arene-limited conditions without requiring excess substrate (Scheme 9).49 The method displayed a broad substrate scope, tolerating diverse substitution patterns on pyridine rings as well as heterocyclic biaryl frameworks, affording the corresponding olefinated products 19 with useful levels of regioselectivity. Mechanistic and control experiments revealed that the reactivity arises from a cooperative dual-ligand catalytic system. In this framework, the bidentate pyridine–pyridone ligand promotes the key C–H cleavage step through a ligand-assisted CMD pathway. In this concerted metalation–deprotonation transition state, the pyridone ligand functions as an internal base in a six-membered cyclic arrangement, enabling simultaneous proton transfer and Pd–C bond formation, while the N-heteroarene substrate itself transiently acts as a monodentate ligand, forming a cationic Pd(II) complex with enhanced affinity for arene coordination. This synergistic interaction between the designed ligand scaffold and the coordinating substrate effectively balances catalyst stability and reactivity, enabling selective C–H activation in strongly coordinating heteroarene systems.
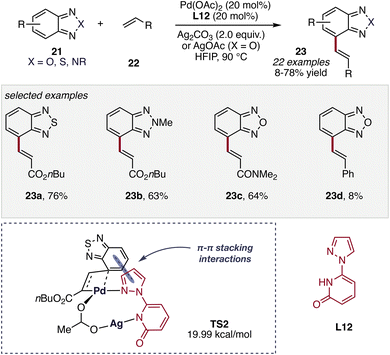
Further expanding the role of pyridone-derived ligands in heteroarene functionalization, the Joo group introduced a pyrazolopyridone (PzPyOH) ligand system (L12) within a Pd–Ag bimetallic catalytic framework for dehydrogenative C(sp2)–H alkenylation of benzodiazole-type heterocycles 21.50 In contrast to conventional protocols where Ag(I) salts function primarily as terminal oxidants or halide scavengers, this study highlights a cooperative Pd–Ag catalytic manifold in which both metals actively participate in the catalytic cycle (TS2, Scheme 10). The PzPyOH ligand provides a bifunctional coordination environment, where the pyridone moiety promotes C–H activation through ligand-assisted CMD, while the pyrazole donor stabilizes the palladium center and facilitates interaction with the silver species. Mechanistic studies suggest that the Ag center assists in substrate activation and migratory insertion steps, forming a heterobimetallic environment that lowers the barrier for C–H cleavage and subsequent C–C bond formation to afford the desired products 23. This Pd–Ag synergy, combined with the cooperative N,O-donor framework of the PzPyOH ligand, enables efficient C4-selective dehydrogenative alkenylation of 2,1,3-benzothiadiazole, benzoxadiazole, and benzotriazole derivatives, underscoring how strategic ligand design can unlock new catalytic reactivity when integrated with heterobimetallic activation modes.
4. 2-Pyridone ligands in C(sp2)–H (pseudo) halogenation reactions
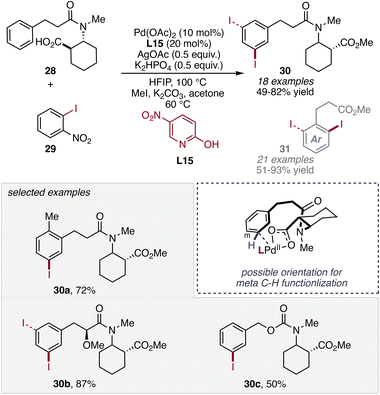
Beyond C–C bond-forming transformations, the 2-pyridone ligand platform has also proven highly effective in C–H (pseudo)halogenation reactions, where installation of halogens provides versatile handles for subsequent cross-coupling and late-stage functionalization. In particular, Pd-catalyzed C(sp2)–H iodination offers a direct route to aryl iodides but often suffers from competing iodide coordination and inefficient C–H activation under non-directed conditions. In this context, Li and co-workers introduced strategy for ligand L14 mediated arene iodination based on formal C(sp2)–H/C(sp2)–I functional group metathesis.51 Using 2-nitrophenyl iodides 25 as mild iodine-transfer reagents, they were able to achieve meta- and ortho-selective iodination of aniline derivatives 24 under mild conditions (Scheme 11). This strategy avoids the use of highly electrophilic iodinating agents and demonstrates how metathesis-type halogen transfer can be leveraged to achieve selective C–H iodination of electron-rich arenes. The work highlights the broader potential of functional group metathesis (as in N-acetyl-N-arylglycines) as a mild and selective platform for C–H halogenation reactions.Inspired by their previous work,51 where a minor amount (≈10%) of the double C–H iodination product was observed, Li and co-workers further expanded this metathesis-based iodination strategy, demonstrating the broader applicability of formal C(sp2)–H/C(sp2)–I functional group metathesis for site-selective arene iodination to afford corresponding products 30 and 31 (Scheme 12).52 In their developed protocol, simple aryl iodides were employed as mild iodinating reagents, representing a mechanistically distinct alternative to conventional electrophilic iodination protocols.
Using commercially available 2-nitrophenyl iodides 29, they were able to achieve Pd(II)-catalyzed C–H iodination of hydrocinnamic acids 28 and related arenes, assisted by chelating ability of aliphatic carboxyl directing groups to enable site-selective meta-C–H functionalization.
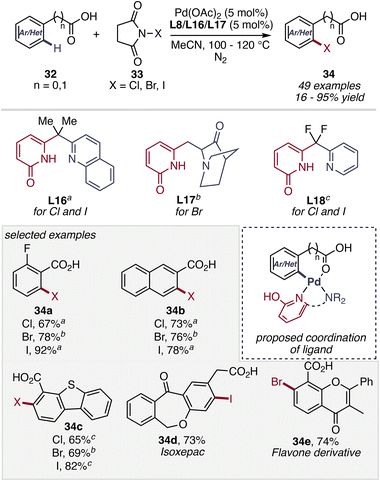
In 2025, in this context, a notable contribution from the Yu group demonstrated a practical C–H halogenation of (hetero)benzoic and (hetero)aryl acetic acids 32, enabled by carefully designed 2-pyridone ligands. By employing three distinct ligands distinct ligands L16, L17, L18, the method enables efficient C–H halogenation to afford desired class of products 34 across arenes with diverse reactivity profiles using NXS reagents 33 (Scheme 13). This protocol provides a direct and scalable route to synthetically valuable aryl halides, operating under mild conditions with broad substrate scope and compatibility for late-stage functionalization. Importantly, ligand–substrate coordination governs the transformation, allowing high levels of site-selectivity even in heteroaromatic and structurally complex systems. This coordination enforces a well-defined ligand-assisted CMD manifold, wherein the 2-pyridone motif serves as an internal base in a cyclic transition state, enabling selective C–H cleavage and positioning the aryl–Pd intermediate for efficient electrophilic halogenation. The methodology accommodates multiple halogen sources and delivers halogenated products efficiently, highlighting its operational simplicity and synthetic utility. Overall, this work exemplifies how systematic ligand design can translate mechanistic insight into practical and broadly applicable C–H halogenation strategies.53
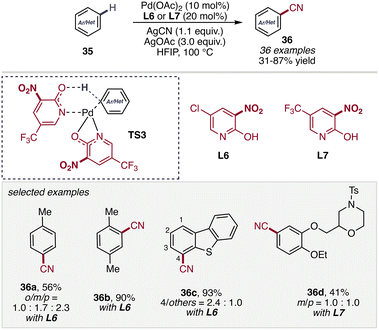
Extending the scope of 2-pyridone-enabled C–H (pseudo) halogenation chemistry, the same ligand platform has also been successfully applied to C(sp2)–H cyanation reactions, enabling the direct installation of synthetically valuable nitrile functionalities onto aromatic frameworks. In this context, Yu and co-workers reported a 2-pyridone-ligand-enabled (L6/L7) Pd-catalyzed non-directed C(sp2)–H cyanation of arenes 35 using AgCN as the cyanide source (Scheme 14).54 The transformation proceeds efficiently under arene-limited conditions and exhibits broad functional group tolerance, enabling cyanation across a range of simple arenes as well as structurally complex molecules. Mechanistic and substrate scope studies revealed that electronic effects primarily govern the regioselectivity of C–H activation, with more electron-rich arene positions preferentially undergoing cyanation, while steric effects play a comparatively minor role. This work further highlights the ability of electron-deficient 2-pyridone ligands to enable challenging non-directed C–H functionalization processes, even in the presence of strongly coordinating nucleophiles such as cyanide.
5. 2-Pyridone ligands in C(sp2)–H deuteration, hydroxylation and acyloxylation reactions
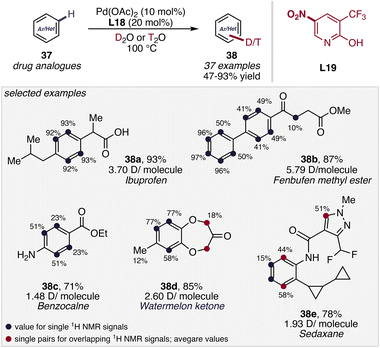
Building on the ability of pyridone frameworks to regulate catalyst speciation and promote ligand-assisted C–H activation, recent efforts have extended their application to C–H isotope exchange reactions, particularly arene deuteration, where similar challenges related to catalyst stability and selective C–H activation must be addressed. Following this, Werz, Maiti and co-workers reported a Pd-catalyzed C(sp2)–H deuteration and tritiation protocol enabled by an electron-deficient 2-pyridone ligand (L19), which proved superior to electron-rich analogues18 Using D2O or T2O as the isotopic source, the method enables efficient isotope incorporation under mild conditions (Scheme 15).55 Notably, the transformation exhibits broad electronic tolerance, accommodating both electron-rich and electron-deficient arenes, and is particularly effective for late-stage isotopic labelling of structurally complex molecules, including drug-like scaffolds and natural product derivatives.Beyond isotope exchange processes, the cooperative reactivity of pyridone-based ligands has also enabled challenging C–H oxygenation reactions, particularly the direct hydroxylation of aromatic C–H bonds. In a landmark study from 2021, Yu and co-workers reported a Pd-catalyzed C(sp2)–H hydroxylation of (hetero)aromatic carboxylic acids (39) using molecular oxygen as the terminal oxidant, enabled by a modified bidentate pyridine–pyridone ligand (L20) framework to access hydroxylated arenes (Scheme 16). A distinctive feature of this catalytic system is the tautomeric flexibility of the pyridine–pyridone ligand, which allows interconversion between neutral L,L-type coordination and an anionic L,X-type coordination mode. This dynamic behavior plays a crucial role in the catalytic cycle: the L,X configuration (I) allows the formation of a six-membered palladacycle adopting a puckered geometry that facilitates ligand-assisted C–H activation via a CMD pathway, whereas the L,L coordination mode (II) stabilizes key palladium intermediates during the aerobic oxidation steps involving O2. In combination with the carboxylate directing group, this cooperative ligand environment enables highly site-selective ortho-C–H activation, effectively suppressing competing pathways even in complex substrates. Consequently, the catalytic system delivers site-selective phenol formation across a broad substrate scope, including the late-stage hydroxylation of pharmaceutically relevant molecules, highlighting how switchable ligand coordination modes can orchestrate both reactivity and selectivity in demanding C–H oxygenation reactions.56
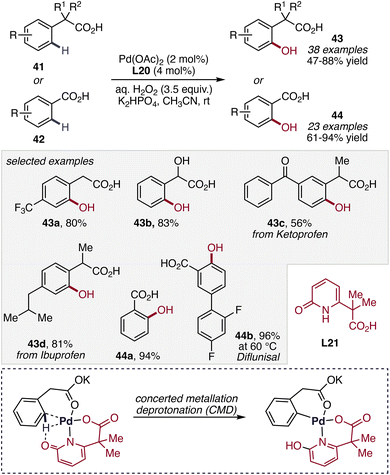
Although the pyridine–pyridone ligand framework effectively promotes C–H hydroxylation using molecular oxygen as the terminal oxidant, the use of O2 presents challenges related to scalability and operational practicality. To address these challenges, Yu and co-workers developed a Pd-catalyzed C(sp2)–H hydroxylation protocol employing aqueous H2O2 as a practical and environmentally benign oxygen source in 2022.57 While attractive for large-scale oxidation chemistry,58 the application of H2O2 in Pd-catalyzed C–H activation is complicated by its tendency to decompose under the elevated temperatures required for C–H cleavage, necessitating careful ligand design. The CMD-active 2-pyridone motif in the bifunctional ligand facilitates rapid C–H activation and stabilizes the palladacycle intermediate, enabling efficient oxidation with H2O2. Using a bifunctional carboxyl–pyridone ligand (L21), the optimized Pd catalyst enabled site-selective ortho-C(sp2)–H hydroxylation of aryl carboxylic acid derivatives (41) and benzoic acids (42), furnishing a broad range of phenolic products (43, 44). The method exhibits broad functional-group tolerance and accommodates both electron-rich and electron-deficient substrates, with the carboxylate directing group cooperating with the ligand framework to facilitate C–H activation and oxygen incorporation (Scheme 17). Remarkably, the practicality of this methodology was demonstrated through the large-scale hydroxylation of ibuprofen (43d), highlighting its potential applicability in industrially relevant phenol synthesis.
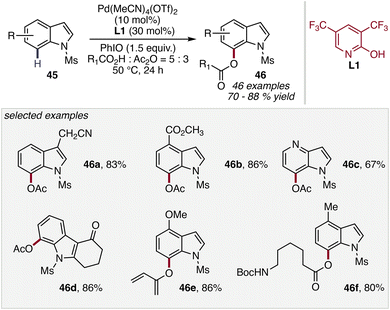
Extending pyridone-enabled C–H functionalization to heterocyclic frameworks, in 2026, Yang and co-workers reported a Pd(II)-catalyzed direct C7-acyloxylation of indoles 45, enabling selective installation of acyloxy groups at a position that is traditionally difficult to access. The transformation relies on pyridone ligand L1 to promote site-selective C–H activation, overriding the inherent reactivity bias of the indole scaffold. Notably, the method achieves high C7-selectivity without the need for pre-installed directing groups, providing a streamlined approach to functionalized indole derivatives (Scheme 18).59
A range of indole substrates and carboxylic acid coupling partners were found to be compatible, affording the corresponding C7-acyloxylated products 46 in good yields. The ability to selectively functionalize the remote C7 position highlights the efficiency of ligand-controlled C–H activation in steering regioselectivity within heteroaromatic systems, offering a valuable strategy for the synthesis of biologically relevant indole derivatives.
6. 2-Pyridone Ligands in C(sp2)–H Chalcogenation of arenes and (Hetero)arenes
In 2022, Maiti and co-workers expanded the scope of non-directed C(sp2)–H functionalization by developing a dual-ligand-enabled nondirected C(sp2)–H chalcogenation of (hetero)arenes (47), enabling the direct synthesis of diaryl sulfides 49 through C–H thioarylation (Scheme 19).60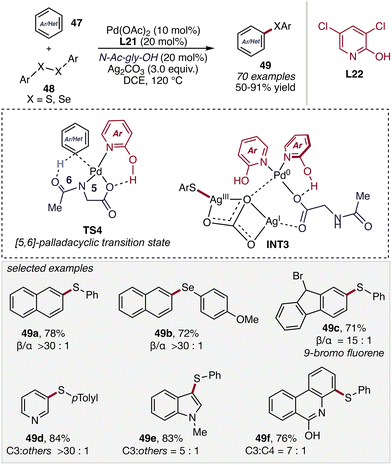 | ||
| Scheme 19 Dual-ligand-assisted Pd-catalyzed C(sp2)–H thioarylation of (hetero)arenes by Maiti et al. | ||
The catalytic system employs a complementary ligand combination in which a pyridone-type ligand (L22) promotes C–H activation via a concerted metalation–deprotonation pathway, while N-acetyl glycine, acting as the second ligand, simultaneously stabilizes the active palladium species and facilitates selective product release. This cooperative ligand environment allows the reaction to proceed under arene-limited conditions. Furthermore, a broad range of electron-rich and electron-deficient arenes as well as heteroarenes were well tolerated, affording structurally diverse diaryl sulfide products. Notably, the protocol also enabled the facile synthesis of pharmaceutically relevant molecules such as Promazine and Mequitazine. Mechanistic investigations and computational studies suggested that C–H activation is both the rate- and regio-determining step, proceeding through a characteristic [5,6]-palladacyclic transition state (TS4). The pyridone ligand was found to lower the C–H activation barrier compared to conventional carboxylate-type ligands. Subsequently, C–S bond formation likely occurs via a transmetalation-like pathway involving a Pd–Ag heterometallic intermediate (INT3).
7. Conclusions and outlook
In conclusions, this review highlights 2-pyridone ligands as an effective and versatile platform for transition-metal-catalyzed C(sp2)–H functionalization, particularly in palladium catalysis. Across diverse transformations—including olefination, halogenation, cyanation, isotope exchange, and oxygenation—pyridone ligands consistently facilitate challenging C–H activation processes through ligand-assisted CMD and metal–ligand cooperativity. Their ability to function as weakly coordinating yet proton-responsive ligands enabled efficient C–H cleavage while maintaining catalyst turnover under arene-limited conditions. The structural and electronic tunability of the pyridone scaffold plays a crucial role in controlling catalyst reactivity and selectivity. Additionally, it has been realized that subtle modifications of ligand substitution patterns can influence acidity, coordination behavior, and transition-state organization, allowing pyridone ligands to address challenging substrates such as heteroarenes, and complex bioactive molecules.Looking forward, deeper structure–function investigations, and the development of bifunctional or cooperative second generation 2-pyridone ligand systems are likely to further advance pyridone-enabled C–H activation. Continued progress in ligand-centered catalyst design will broaden the scope and practical applicability of this platform in modern C–H functionalization chemistry.
Conflicts of interest
There are no conflicts to declare.Data availability
The data for this review article are available in the references cited in the manuscript.References
- T. Dalton, T. Faber and F. Glorius, ACS Cent. Sci., 2021, 7, 245–261 CrossRef CAS PubMed.
- J. Wencel-Delord and F. Glorius, Nat. Chem., 2013, 5, 369–375 CrossRef CAS PubMed.
- C. Pei, C. Empel and R. M. Koenigs, Angew. Chem., Int. Ed., 2022, 31, e202201743 Search PubMed.
- Y.-H. Li, Y. Ouyang, J.-L. Yan, N. Chekshin and J.-Q. Yu, Acc. Chem. Res., 2025, 58, 2910–2926 CrossRef CAS PubMed.
- J. Grover, G. Prakash, A. Mandal, D. Ghosh, S. Maiti, C. Empel and D. Maiti, Adv. Sci., 2026, 13, e19731 CrossRef CAS.
- P. Gandeepan and L. Ackermann, Chem, 2018, 4, 199–222 CAS.
- C. Sambiagio, D. Schönbauer, R. Blieck, T. Dao-Huy, G. Pototschnig, P. Schaaf, T. Wiesinger, M. F. Zia, J. Wencel-Delord, T. Besset, B. U. W. Maes and M. Schnürch, Chem. Soc. Rev., 2018, 47, 6603–6743 RSC.
- S. Rej, Y. Ano and N. Chatani, Chem. Rev., 2020, 120, 1788–1887 CrossRef CAS PubMed.
- S. R. Neufeldt and M. S. Sanford, Acc. Chem. Res., 2012, 45, 936–946 CrossRef CAS.
- T. W. Lyons and M. S. Sanford, Chem. Rev., 2010, 110, 1147–1169 CrossRef CAS PubMed.
- C. Empel, S. Jana, T. Langletz and R. M. Koenigs, Chem. – Eur. J., 2022, 28, e202104321 CrossRef CAS PubMed.
- Q. Shao, K. Wu, Z. Zhuang, S. Qian and J.-Q. Yu, Acc. Chem. Res., 2020, 53, 833–851 CrossRef CAS PubMed.
- C. Empel, S. Jana, A. Koodan and R. M. Koenigs, ACS Catal., 2022, 12, 8229–8236 CrossRef CAS.
- G. Meng, J.-L. Yan, N. Chekshin, D. A. Strassfeld and J.-Q. Yu, ACS Catal., 2024, 14, 12806–12813 CrossRef CAS PubMed.
- J. Moradell, C. Bohan, A. Pop and E. P. Urriolabeitia, ChemCatChem, 2025, 17, e00664 CrossRef CAS.
- S. K. Agrawal, S. Porey, Y. Bairagi, S. Maiti, A. C. Bissember and D. Maiti, Chem. Soc. Rev., 2025, 54, 6122–6174 RSC.
- P. Wedi and M. van Gemmeren, Angew. Chem., Int. Ed., 2018, 57, 13016–13027 CrossRef CAS PubMed.
- P. Wang, P. Verma, G. Xia, J. Shi, J. X. Qiao, S. Tao, P. T. W. Cheng, M. A. Poss, M. E. Farmer, K.-S. Yeung and J.-Q. Yu, Nature, 2017, 551, 489–493 CrossRef CAS.
- F. Deufel and M. Van Gemmeren, J. Chem. Educ., 2024, 101, 3410–3417 CrossRef CAS PubMed.
- H. Chen, A. Mondal, P. Wedi and M. Van Gemmeren, ACS Catal., 2019, 9, 1979–1984 CrossRef CAS.
- H. Chen, P. Wedi, T. Meyer, G. Tavakoli and M. van Gemmeren, Angew. Chem., Int. Ed., 2018, 57, 2497–2501 CrossRef CAS PubMed.
- P. Wedi, M. Farizyan, K. Bergander, C. Mück-Lichtenfeld and M. Van Gemmeren, Angew. Chem., Int. Ed., 2021, 60, 15641–15649 CrossRef CAS PubMed.
- K. Naksomboon, E. Gómez-Bengoa, J. Mehara, J. Roithová, E. Otten and M. Á. Fernández-Ibáñez, Chem. Sci., 2023, 14, 2943–2953 RSC.
- K. Deng, V. Sukowski and M. Á. Fernández-Ibáñez, Angew. Chem., Int. Ed., 2024, 63, e202400689 CrossRef CAS PubMed.
- K. Naksomboon, C. Valderas, M. Gómez-Martínez, Y. Álvarez-Casao and M. Á. Fernández-Ibáñez, ACS Catal., 2017, 7, 6342–6346 CrossRef CAS PubMed.
- J. Dhankhar, E. González-Fernández, C.-C. Dong, T. K. Mukhopadhyay, A. Linden and I. Čorić, J. Am. Chem. Soc., 2020, 142, 19040–19046 CrossRef CAS PubMed.
- J. Dhankhar, M. D. Hofer, A. Linden and I. Čorić, Angew. Chem., Int. Ed., 2022, 61, e202205470 CrossRef CAS PubMed.
- A. Fedulin and A. Jacobi Von Wangelin, Catal. Sci. Technol., 2024, 14, 26–42 RSC.
- N. Mandal and A. Datta, J. Org. Chem., 2020, 85, 13228–13238 CrossRef CAS PubMed.
- J. M. Rawson and R. E. P. Winpenny, Coord. Chem. Rev., 1995, 139, 313–374 CrossRef CAS.
- M. W. Drover, J. A. Love and L. L. Schafer, Chem. Soc. Rev., 2017, 46, 2913–2940 RSC.
- C. Smith, C. Hirschhäuser, G. Malcolm, D. Nasrallah and T. Gallagher, Synlett, 2014, 1904–1908 CAS.
- M. J. Cook and A. R. Katritzky, Tetrahedron Lett., 1976, 31, 2685–2688 CrossRef.
- R. H. Cox and A. A. Bothner-By, J. Phys. Chem., 1969, 73, 2465–2468 CrossRef CAS.
- J. Frank and A. R. Katritzky, J. Chem. Soc., Perkin Trans. 2, 1976, 1428–1431 RSC.
- J. C. Jeffrey and T. B. Rauchfuss, Inorg. Chem., 1979, 18, 2658–2666 CrossRef CAS.
- J. M. Clarkson and L. L. Schafer, Inorg. Chem., 2017, 56, 5553–5566 CrossRef CAS PubMed.
- G. Chen, Z. Zhuang, G. Li, T. G. Saint-Denis, Y. Hsiao, C. L. Joe and J. Yu, Angew. Chem., Int. Ed., 2017, 56, 1506–1509 CrossRef CAS PubMed.
- A. Saha, S. Guin, W. Ali, T. Bhattacharya, S. Sasmal, N. Goswami, G. Prakash, S. K. Sinha, H. B. Chandrashekar, S. Panda, S. S. Anjana and D. Maiti, J. Am. Chem. Soc., 2022, 144, 1929–1940 CrossRef CAS PubMed.
- S. Mishra, C. Jadala, S. Cardoza, G. R. Potuganti and G. R. Velma, RSC Adv., 2026, 16, 13830–13850 RSC.
- N. A. Meanwell, J. Med. Chem., 2018, 61, 5822–5880 CrossRef CAS PubMed.
- E. P. Gillis, K. J. Eastman, M. D. Hill, D. J. Donnelly and N. A. Meanwell, J. Med. Chem., 2015, 58, 8315–8359 CrossRef CAS PubMed.
- X.-Y. Chen, Y. Wu, J. Zhou, P. Wang and J.-Q. Yu, Org. Lett., 2019, 21, 1426–1429 CrossRef CAS PubMed.
- A. Scozzafava, T. Owa, A. Mastrolorenzo and C. Supuran, Curr. Med. Chem., 2003, 10, 925–953 CrossRef CAS.
- L. Guillemard, N. Kaplaneris, L. Ackermann and M. J. Johansson, Nat. Rev. Chem., 2021, 5, 522–545 CrossRef CAS PubMed.
- R. Jana, H. M. Begam and E. Dinda, Chem. Commun., 2021, 57, 10842–10866 RSC.
- L. Stockmann and C. Empel, Chem. – Eur. J., 2025, 31, e01973 CrossRef CAS PubMed.
- Y.-J. Liu, H. Xu, W.-J. Kong, M. Shang, H.-X. Dai and J.-Q. Yu, Nature, 2014, 515, 389–393 CrossRef CAS PubMed.
- G. Meng, Z. Wang, H. S. Sam Chan, N. Chekshin, Z. Li, P. Wang and J.-Q. Yu, Am. Chem. Soc., 2023, 145, 8198–8208 CrossRef CAS PubMed.
- S. Jeong, C. Lee and J. M. Joo, Adv. Synth. Catal., 2025, 367, e202401139 CrossRef CAS.
- N. Wang, Z. Chi, X. Wang, Z. Gao, S. Li and G. Li, Org. Lett., 2022, 24, 3657–3662 CrossRef CAS PubMed.
- S. Li, C. Zhang, L. Fu, H. Wang, L. Cai, X. Chen, X. Wang and G. Li, CCS Chem, 2022, 4, 1889–1900 CrossRef CAS.
- H. Zhao, Z. Li, X. Zhu and J.-Q. Yu, J. Am. Chem. Soc., 2025, 147, 31570–31577 CrossRef CAS PubMed.
- L. Liu, K. Yeung and J. Yu, Chem. – Eur. J., 2019, 25, 2199–2202 CrossRef CAS PubMed.
- C. Teja, S. Kolb, P. Colonna, J. Grover, S. Garcia-Argote, G. K. Lahiri, G. Pieters, D. B. Werz and D. Maiti, Angew. Chem., Int. Ed., 2024, 63, e202410162 CrossRef CAS PubMed.
- Z. Li, Z. Wang, N. Chekshin, S. Qian, J. X. Qiao, P. T. Cheng, K.-S. Yeung, W. R. Ewing and J.-Q. Yu, Science, 2021, 372, 1452–1457 CrossRef CAS PubMed.
- Z. Li, H. S. Park, J. X. Qiao, K.-S. Yeung and J.-Q. Yu, J. Am. Chem. Soc., 2022, 144, 18109–18116 CrossRef CAS PubMed.
- M. G. Clerici and O. A. Kholdeeva, in Liquid Phase Oxidation via Heterogeneous Catalysis – Organic Synthesis and Industrial Applications, John Wiley & Sons, Inc, Hoboken, New Jersy, 2013 Search PubMed.
- X. Zhang, J. Wang, S. Chen, J. Wu, H. Zhu, P. Guo and X. Yang, Org. Lett., 2026, 28, 1159–1163 CrossRef CAS PubMed.
- S. K. Sinha, S. Panja, J. Grover, P. S. Hazra, S. Pandit, Y. Bairagi, X. Zhang and D. Maiti, J. Am. Chem. Soc., 2022, 144, 12032–12042 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |