Lattice doping and NiO synergistic decoration for enhanced CO2 electrolysis in solid oxide electrolysis cells
Maoyu
Kang
a,
Shu
Xiao
a,
Qiran
Zhang
a,
Jinhuang
Cai
 a,
Dunkui
Li
a,
Chang-An
Zhou
a,
Dunkui
Li
a,
Chang-An
Zhou
 *a,
Hongjiao
Li
*a,
Hongjiao
Li
 a,
Chao
Wang
a,
Lei
Song
a,
Chao
Wang
a,
Lei
Song
 a,
Kui
Ma
a,
Kui
Ma
 *a and
Hairong
Yue
*a and
Hairong
Yue
 abc
abc
aLow-Carbon Technology and Chemical Reaction Engineering Laboratory, School of Chemical Engineering, Sichuan University, Chengdu 610065, China. E-mail: zhouchangan@scu.edu.cn; kuima@scu.edu.cn
bInstitute of New Energy and Low-Carbon Technology, Sichuan University, Chengdu 610207, China
cCoal Mining Research Institute, China Coal Technology and Engineering Group, Beijing 100013, China
First published on 8th April 2026
Abstract
This work presents a NiO/Ni-SDC composite cathode with synergistic effect of lattice doping and surface oxide modification, which notably facilitates O2− transport and CO2 adsorption-activation, leading to boosted CO2 reduction in solid oxide electrolysis cells.
Excessive use of fossil fuels leads to the continuous accumulation of CO2 in the atmosphere. The electrochemical conversion of CO2 into energetic compounds such as CO is considered an effective strategy for both carbon emission reduction and renewable energy storage.1–3 Among the various CO2 electrolysis technologies, high-temperature solid oxide electrolysis cells (SOECs) have attracted considerable attention due to their capability to achieve relatively high current density, energy efficiency, and CO selectivity at temperatures in the range of 700–800 °C.3,4 Moreover, the CO produced by this technology can be combined with hydrogen sources for direct energy production or the synthesis of valuable chemicals.5–9 However, the commercialization of SOECs is largely hindered by the performance of key materials, particularly the activity and stability of the cathode materials. Consequently, the development of advanced cathode materials that combine high catalytic activity with exceptional durability has been a research hotspot in recent years.10
Ceria-based materials exhibit significant application potential in CO2 reduction reactions (CO2RRs) due to their excellent oxygen storage capacity, good redox stability, and abundant surface oxygen vacancies under reducing atmospheres, which facilitate CO2 adsorption and dissociation.11–13 However, their application in SOECs has been partially limited by the intrinsically low electronic conductivities and catalytic activity.14,15 Notably, under reducing conditions typical of the cathode, ceria-based materials (e.g., Sm-doped ceria and Gd-doped ceria) act as mixed ionic electronic conductors (MIECs) as a result of the Ce4+/Ce3+ redox coupling, thereby boosting their electrical conductivity and surface activity for cathode applications.16–19 Additionally, studies indicate that ceria-based supports can form favorable interfacial interactions with loaded metal species, which effectively promote the dispersion of active sites.20–22 It has been reported that the electrolysis performance of ceria-based materials can be improved by introducing metal species via routes such as metal–particle impregnation and cation doping.23–28 However, the coarsening, agglomeration, and oxidation of metal particles loaded onto a ceria-based surface at high temperatures readily lead to a decline in electrochemical activity. This problem also exists in the traditional Ni-YSZ materials with high catalytic activity.29
Herein, a Ni-decorated Sm-doped CeO2 (SDC) cathode was synthesized using a simple sol–gel method, constructing a NiO/Ni, Sm-codoped CeO2 composite structure that integrates nickel lattice doping with surface nickel oxide modification on SDC. The introduced Ni species are partly incorporated as Ni2+ into the fluorite SDC lattice, while the remaining fraction is present as highly dispersed NiO nanoparticles distributed on the SDC surface. Through the synergistic effect of these two forms of Ni, charge transport and catalytic activity are effectively optimized, thereby enhancing the CO2 electrolysis performance of SOECs. As a result, at 800 °C and 1.6 V, the CO2 electrolysis current density of the Ni-decorated SDC cathode reached 2.57 A cm−2, representing an approximately 60% increase compared to that of the unmodified SDC cathode.
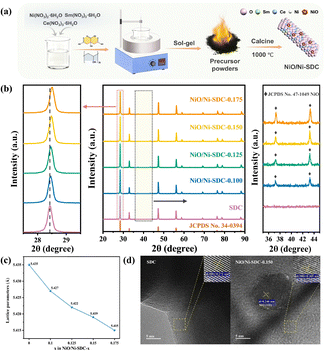
The synthetic route diagram of the NiO/Ni-SDC cathode is depicted in Fig. 1a. The XRD patterns of NiO/Ni-SDC-x powders (where x = 0.100, 0.125, 0.150 and 0.175, representing the molar amount of Ni species introduced into SDC) are shown in Fig. 1b. All samples display characteristic peaks of the CeO2 (JCPDS No. 34-0394) fluorite structure, corresponding to the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group. As the Ni content increases, the main diffraction peaks progressively shift toward higher 2θ angles, indicating a reduction in the lattice parameter upon Ni species incorporation. The contraction of the lattice is primarily attributed to the smaller ionic radius of Ni2+ (0.69 Å, VI) compared with those of Sm3+ (1.08 Å, VIII) and Ce4+ (0.97 Å, VIII).30,31 The crystal structures were analyzed by Rietveld refinement (Fig. S1). The lattice parameter exhibits a decreasing trend with increasing Ni content, declining from 5.427 Å for NiO/Ni-SDC-0.100 to 5.415 Å for NiO/Ni-SDC-0.175 (Fig. 1c), which is consistent with the observed shift in the XRD peak positions. Moreover, the successful doping of Ni2+ is further confirmed by the TEM results. The (200) interplanar spacing of SDC drops from 0.271 nm to 0.269 nm following Ni decoration (Fig. 1d).
m space group. As the Ni content increases, the main diffraction peaks progressively shift toward higher 2θ angles, indicating a reduction in the lattice parameter upon Ni species incorporation. The contraction of the lattice is primarily attributed to the smaller ionic radius of Ni2+ (0.69 Å, VI) compared with those of Sm3+ (1.08 Å, VIII) and Ce4+ (0.97 Å, VIII).30,31 The crystal structures were analyzed by Rietveld refinement (Fig. S1). The lattice parameter exhibits a decreasing trend with increasing Ni content, declining from 5.427 Å for NiO/Ni-SDC-0.100 to 5.415 Å for NiO/Ni-SDC-0.175 (Fig. 1c), which is consistent with the observed shift in the XRD peak positions. Moreover, the successful doping of Ni2+ is further confirmed by the TEM results. The (200) interplanar spacing of SDC drops from 0.271 nm to 0.269 nm following Ni decoration (Fig. 1d).
 | ||
| Fig. 1 (a) Synthesis scheme of NiO/Ni-SDC-x. (b) XRD patterns of SDC and NiO/Ni-SDC-x. (c) Curve of the lattice constant variation with Ni content. (d) HRTEM images of SDC and NiO/Ni-SDC-0.150. | ||
Additionally, weak peaks appeared at 2θ = 37.2° and 43.3° in the XRD patterns of all the NiO/Ni-SDC-x samples, which are the (111) and (200) diffraction peaks of NiO (JCPDS No. 47-1049) with the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group.32Fig. 1d displays nanoparticles on the sample surface with a lattice spacing of 0.240 nm, corresponding to the (111) plane of NiO. This further confirms the presence of a small amount of NiO phase distributed on the Ni-SDC surface, forming a NiO/Ni-SDC composite structure. As shown in Fig. S2, NiO is mainly present as fine, highly dispersed nanoparticles distributed on the surface of the backbone, which are beneficial for increasing the number of active sites.33,34 Additionally, the overall NiO/Ni-SDC-0.150 still retains a porous structure favorable for gas diffusion, similar to that of pristine SDC, with no continuous blocking layer observed.
m space group.32Fig. 1d displays nanoparticles on the sample surface with a lattice spacing of 0.240 nm, corresponding to the (111) plane of NiO. This further confirms the presence of a small amount of NiO phase distributed on the Ni-SDC surface, forming a NiO/Ni-SDC composite structure. As shown in Fig. S2, NiO is mainly present as fine, highly dispersed nanoparticles distributed on the surface of the backbone, which are beneficial for increasing the number of active sites.33,34 Additionally, the overall NiO/Ni-SDC-0.150 still retains a porous structure favorable for gas diffusion, similar to that of pristine SDC, with no continuous blocking layer observed.
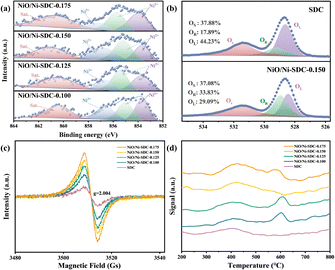
As shown in Fig. 2a, the Ni 2p X-ray photoelectron spectra (XPS) of all Ni-decorated samples can be fitted into Ni2+ and Ni3+ components.35,36 The introduced Ni species exist in a mixed valence state. This provides additional electron hopping pathways via Ni2+–O2−–Ni3+ → Ni3+–O−–Ni3+ → Ni3+–O2−–Ni2+, thereby enhancing electron conduction.37,38 The O 1s spectrum exhibits three distinct peaks at 528.5 eV, 529.2 eV, and 531.5 eV (Fig. 2b and Fig. S3), corresponding to lattice oxygen (OL: O2−), highly oxidative oxygen (OR: O22−/O−), and surface chemisorbed oxygen (OS: CO32−/OH−), respectively.15 Compared to SDC, NiO/Ni-SDC-x exhibits an increased percentage of OR, a decreased percentage of OL, and essentially unchanged OS (Table S1), with these trends intensifying at higher Ni contents. The OR content is closely related to the electrode catalytic activity, and its increase promotes CO2 activation.39,40 In electron paramagnetic resonance (EPR) spectra (Fig. 2c), Ni-decorated SDC exhibits a significantly stronger signal at g = 2.004 compared to pristine SDC, indicative of a higher density of oxygen vacancies,41 which play a pivotal role in oxygen ion transport capacity and CO2 chemisorption and activation.42 The trend of the oxygen vacancy content in NiO/Ni-SDC-x is consistent with the XPS results. Given that catalyst surface adsorption is a prerequisite for catalytic CO2 conversion, the CO2 adsorption behavior was evaluated via CO2 temperature-programmed desorption (CO2-TPD) (Fig. 2d). CO2 desorption peaks observed near 400 °C correspond to the decomposition of stable bidentate or bridged carbonates, which are closely associated with surface oxygen vacancies.43 Notably, a distinct desorption peak emerges at approximately 600 °C upon the incorporation of Ni species, which is absent for the pristine SDC sample. This peak may be associated with the adsorption of carbonate species at the NiO/Ni-SDC-x interfaces.44 Overall, the results indicate that the incorporation of Ni species enhances the CO2 adsorption capacity.
 | ||
| Fig. 2 XPS spectra of (a) Ni 2p for NiO/Ni-SDC-x and (b) O 1s for SDC and NiO/Ni-SDC-0.150. (c) EPR spectra of SDC and NiO/Ni-SDC-x. (d) CO2-TPD profiles of the SDC and NiO/Ni-SDC-x samples. | ||
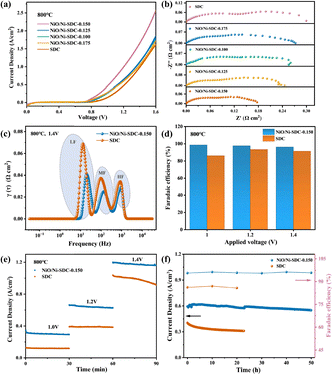
The catalytic activity of SDC and Ni-decorated SDC for the CO2RRs was systematically evaluated using a single-cell configuration. J–V curves indicate that the introduction of Ni species enhances the electrolytic current density across the entire temperature range (Fig. 3a and Fig. S4). At 800 °C and 1.6 V, the NiO/Ni-SDC-0.150 cathode exhibits a current density of 2.57 A cm−2, outperforming both the pristine SDC cathode (1.61 A cm−2) and the Ni/Ni-SDC-0.150 cathode (1.96 A cm−2) obtained by reducing NiO/Ni-SDC-0.150 under a H2 atmosphere (Fig. S5). This performance also surpasses those of several reported cathode materials, and even a few cathode materials modified with in situ exsolved nanoparticles, including Ni, NiFe, and NiCu alloys (Fig. S5). Electrochemical impedance spectroscopy (EIS) measurements were conducted at 800 °C with applied voltages of 1.0, 1.2, and 1.4 V. Subtracting the ohmic resistance (RO) corresponding to the electrolyte and current collection facilitates intuitive comparison of electrode performance. EIS reveals that Ni decoration significantly reduces the polarization resistance (Rp) at 800 °C under different voltages (Fig. 3b and Fig. S6). Notably, at 1.4 V, Rp decreased significantly by 41%, from 0.300 Ω cm2 (SDC) to 0.177 Ω cm2 (NiO/Ni-SDC-0.150), suggesting enhanced activity (Fig. 3b). NiO/Ni-SDC-0.150 also exhibits a lower polarization resistance than the reduced Ni/Ni-SDC-0.150 (0.218 Ω cm2) (Fig. S5). Given that all cells possess the same electrolyte and anode, the observed reduction in polarization resistance can be attributed solely to the cathode as a result of Ni decoration.
To further clarify electrode kinetics, distribution of relaxation times (DRT) analysis was performed based on the electrochemical impedance spectra, with the elementary process analysis shown in Fig. 3c. The DRT spectrum can be divided into three peaks: high-frequency (HF), medium-frequency (MF), and low-frequency (LF) response peaks, corresponding to charge transfer, O2− diffusion, and CO2 adsorption–dissociation, respectively.15,45 The values for NiO/Ni-SDC-0.150 showed a significant decrease across all three processes. Based on the DRT analysis results, the EIS spectra were subsequently fitted using the complex nonlinear least-squares (CNLS) method (Fig. S7). The introduction of Ni species led to a substantial resistance reduction of 25.0%, 42.7%, and 47.9% for the charge transfer, O2− diffusion, and CO2 adsorption–dissociation processes, respectively (Table S2). This suggests that the improved electrochemical performance of NiO/Ni-SDC-0.150 is primarily attributed to the enhanced CO2 adsorption–dissociation processes. Additionally, the faradaic efficiencies for CO exceeded 96% at the voltage range of 1.0–1.4 V of the NiO/Ni-SDC-0.150 cell (Fig. 3d).
Operation stability is also a critical criterion for evaluating the practical application of SOECs. Both the NiO/Ni-SDC-0.150 and SDC cells exhibited excellent short-term stability (Fig. 3e). Also, at a higher voltage (1.4 V), NiO/Ni-SDC-0.150 shows noticeably superior short-term stability and current density. This is due to the CO2RRs electron transport kinetics significantly enhanced at high voltage. Furthermore, the enhanced CO2 adsorption–dissociation ability of NiO/Ni-SDC-0.150 thus accounts for its reduced current density decay compared to SDC at high voltage.41 Importantly, the NiO/Ni-SDC-0.150 cell demonstrated exceptional long-term stability and structural integrity during a 50-h test at 800 °C and 1.2 V (Fig. 3f). The maintained current density of 600 mA cm−2 and faradaic efficiency of 96% confirms its stable catalysis towards CO2 to CO. In comparison, the pristine SDC cell exhibited a current density of less than 400 mA cm−2. Moreover, the NiO/Ni-SDC-0.150 cathode after testing retained its well-adhered and porous surface, with no detectable carbon deposition (Fig. S8).
To better understand the intrinsic effect of the NiO/Ni-SDC composite structure on CO2RRs activity, density functional theory (DFT) calculations were performed, and the structural models of SDC, Ni-SDC, NiO/Ni-SDC and Ni/Ni-SDC were constructed and described (Fig. S9). In this work, the CO2 adsorption and dissociation processes were primarily calculated to elucidate the CO2 activation mechanism upon the introduction of Ni species (Fig. 4). This reaction begins with CO2 adsorption, forming a carbonate intermediate (bidentate CO2* species). Enhanced CO2 adsorption increases the surface concentration of carbonate intermediates, and definitely accelerates the overall reaction.40 Compared to SDC (−0.91 eV), NiO/Ni-SDC (−2.43 eV) exhibits a more energetically favorable adsorption of CO2. Although Ni-SDC (−1.70 eV) and Ni/Ni-SDC (−2.11 eV) also showed improvement over the pristine SDC, the enhancement observed in NiO/Ni-SDC was the most pronounced. These CO2* species then dissociate through an electrochemical process to form CO*, accompanied by the transfer of 2e−, whereby one oxygen atom from the carbonate intermediate is incorporated into the surface, yielding adsorbed CO*. The dissociation activation energy of CO2 in SDC is 0.79 eV. NiO/Ni-SDC exhibits the lowest CO2 dissociation activation energy (0.66 eV), followed by Ni/Ni-SDC (0.71 eV) and Ni-SDC (0.77 eV). Compared to the purely lattice-doped Ni-SDC, NiO/Ni-SDC maintains a high oxygen vacancy concentration, introducing additional interface oxygen species, which further promotes CO2 adsorption and dissociation. However, after reducing the surface-dispersed NiO to metallic Ni, the CO2 activation ability decreases. This may be attributed to CO2 exhibiting a stronger tendency to adsorb stably as carbonates on NiO-covered surfaces than interacting with metallic Ni.46 Moreover, compared to the Ni/SDC interfaces, the NiO/SDC interfaces possess a more compatible crystal structure, which may provide a low-resistance channel for facilitated O2− transport.47,48 Overall, the results indicate that the unique NiO/Ni-SDC structure can effectively enhance CO2 adsorption and dissociation, thereby improving the overall electrochemical performance. This finding challenges the conventional strategy of decorating cathodes with metal particles to boost CO2 electrolysis.
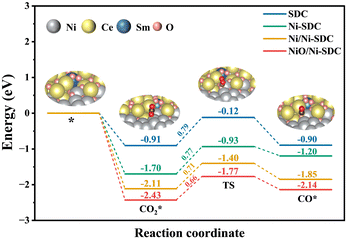 | ||
| Fig. 4 Energy profile of CO2 reduction on SDC, Ni-SDC, Ni/Ni-SDC and NiO/Ni-SDC; the inset shows the atomic structure model of NiO/Ni-SDC. | ||
In summary, this work presents a multi-dimensional catalytic design by synergistically integrating lattice doping with surface decoration. Through designing NiO/Ni-SDC structure, additional electron hopping pathways and abundant oxygen vacancies are created, thereby promoting electrode kinetics and significantly reducing the electrode polarization resistance to 0.177 Ω cm2. In addition, the NiO particles on NiO/Ni-SDC surface further enhance the CO2 electrolysis by strengthening CO2 adsorption and lowering the CO2 dissociation energy barrier from 0.77 eV to 0.66 eV. The modification in the form of stable oxide phase rather than metallic phase can alleviate metal agglomeration and oxidation, demonstrating a 50-h stable electrolysis performance. This work provides a feasible new approach for designing efficient cathodes for SOECs.
Maoyu Kang: investigation, data curation, and writing – original draft. Shu Xiao: investigation and software. Qiran Zhang: investigation and validation. Jinhuang Cai: validation and formal analysis. Dunkui Li: validation. Chang-An Zhou: methodology, writing – review and editing, and funding acquisition. Hongjiao Li: resources. Lei Song: formal analysis. Chao Wang: formal analysis. Kui Ma: methodology, funding acquisition, and project administration. Hairong Yue: funding acquisition and supervision.
Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cc01378c.
Acknowledgements
This work is financially supported by the National Natural Science Foundation of China (22578295, 22308234, 22378280 and U24A20538), the Sichuan Science and Technology Program (2025ZNSFSC0929 and 2024NSFSC1133), and the project funded by the China Postdoctoral Science Foundation (GZC20231775 and 2024M762219).References
- Y. Jiang, F. Chen and C. Xia, J. Power Sources, 2021, 493, 229713 CrossRef CAS.
- M. Xu, R. Cao, H. Qin, N. Zhang, W. Yan, L. Liu, J. T. S. Irvine and D. Chen, Mater. Rep. Energy, 2023, 3, 100198 CAS.
- P. Qiu, C. Li, B. Liu, D. Yan, J. Li and L. Jia, J. Adv. Ceram., 2023, 12, 1463–1510 CrossRef CAS.
- Y. Luo, T. Liu, Y. Wang and M. Ding, Chem. Catal., 2023, 3, 100815 CAS.
- S. Lee, M. Kim, K. T. Lee, J. T. S. Irvine and T. H. Shin, Adv. Energy Mater., 2021, 11, 2100339 CrossRef CAS.
- X. Li, P. Anderson, H.-R. M. Jhong, M. Paster, J. F. Stubbins and P. J. A. Kenis, Energy Fuels, 2016, 30, 5980–5989 CrossRef CAS.
- X. Hou, Y. Jiang, K. Wei, C. Jiang, T.-C. Jen, Y. Yao, X. Liu, J. Ma and J. T. S. Irvine, Chem. Rev., 2024, 124, 5119–5166 CrossRef CAS PubMed.
- L. Chen, F. Chen and C. Xia, Energy Environ. Sci., 2014, 7, 4018–4022 RSC.
- M. Kim, H. Kim, A. Akromjon, I. Jeong, S. Oh, G. Youn and K. T. Lee, Appl. Catal., B, 2025, 371, 125222 CrossRef CAS.
- W. Chen and C. Sun, Energy Mater., 2025, 5, 500045 CAS.
- P. Ebrahimi, A. Kumar and M. Khraisheh, Catalysts, 2022, 12, 1101 CrossRef CAS.
- K. Chang, H. Zhang, M. Cheng and Q. Lu, ACS Catal., 2020, 10, 613–631 CrossRef CAS.
- E. M. Sala, N. Mazzanti, M. B. Mogensen and C. Chatzichristodoulou, Solid State Ionics, 2022, 375, 115833 CrossRef CAS.
- Y. Zhou, F. Wei and H. Wu, Electrochim. Acta, 2022, 419, 140434 CrossRef CAS.
- L. Zhang, Y. Jiang, K. Zhu, N. Shi, Z. U. Rehman, R. Peng and C. Xia, Small Methods, 2024, 8, 2301686 CrossRef CAS PubMed.
- S. W. Lee, T. H. Nam, S. H. Lee, T. Ishihara, J. T. S. Irvine and T. H. Shin, Energy Environ. Sci., 2025, 18, 1205–1213 RSC.
- A. Shaur, M. Drazkowski, S. Zhu, B. Boukamp and H. J. M. Bouwmeester, J. Mater. Chem. A, 2023, 11, 25020–25030 RSC.
- A. Nenning, M. Holzmann, J. Fleig and A. K. Opitz, Mater. Adv., 2021, 2, 5422–5431 RSC.
- L. Zhang, Y. Jiang, K. Zhu, N. Shi, Z. U. Rehman, R. Peng and C. Xia, Small Methods, 2024, 8, 2301686 CrossRef CAS PubMed.
- A. Parastaev, V. Muravev, E. Huertas Osta, A. J. F. Van Hoof, T. F. Kimpel, N. Kosinov and E. J. M. Hensen, Nat. Catal., 2020, 3, 526–533 CrossRef CAS.
- Z. Li, S. Chen, W. Wang, J. Sun, X. Wang, D. Fu, Z.-J. Zhao, C. Pei and J. Gong, ACS Catal., 2025, 15, 6078–6087 CrossRef CAS.
- C.-A. Zhou, K. Ma, Z. Zhuang, M. Ran, G. Shu, C. Wang, L. Song, L. Zheng, H. Yue and D. Wang, J. Am. Chem. Soc., 2024, 146, 21453–21465 CrossRef PubMed.
- M. Khoshkalam, M. A. Faghihi-Sani, X. Tong, M. Chen and P. V. Hendriksen, J. Electrochem. Soc., 2020, 167, 024505 CrossRef CAS.
- N. W. Kwak, S. J. Jeong, H. G. Seo, S. Lee, Y. Kim, J. K. Kim, P. Byeon, S.-Y. Chung and W. Jung, Nat. Commun., 2018, 9, 4829 CrossRef PubMed.
- H. Jiang, L. Wang, H. Kaneko, R. Gu, G. Su, L. Li, J. Zhang, H. Song, F. Zhu, A. Yamaguchi, J. Xu, F. Liu, M. Miyauchi, W. Ding and M. Zhong, Nat. Catal., 2023, 6, 519–530 CrossRef CAS.
- H. Lv, X. Dong, R. Li, C. Zeng, X. Zhang, Y. Song, H. Liu, J. Shao, N. Ta, Q. Zhao, Q. Fu, J. Xiao, G. Wang and X. Bao, Nat. Chem., 2025, 17, 695–702 CrossRef CAS PubMed.
- I. D. Unachukwu, V. Vibhu, J. Uecker, I. C. Vinke, R.-A. Eichel and L. G. J. (Bert) De Haart, J. CO2 Util., 2023, 69, 102423 CrossRef CAS.
- T. Su, Y. Li, S. Xue, Z. Xu, M. Zheng and C. Xia, J. Mater. Chem. A, 2019, 7, 1598–1606 RSC.
- A. D. N. Kamkeng and M. Wang, Chem. Eng. J., 2022, 429, 132158 CrossRef CAS.
- S. R. Gawali, Bull. Math. Sci., 2024, 47, 48–54 CrossRef.
- L. Zhang, Y. Jiang, K. Zhu, N. Shi, Z. U. Rehman, R. Peng and C. Xia, Small Methods, 2024, 8, 2301686 CrossRef CAS PubMed.
- N. K. Hoa, Ceram. - Silik., 2017, 62, 41–49 Search PubMed.
- J. T. S. Irvine, D. Neagu, M. C. Verbraeken, C. Chatzichristodoulou, C. Graves and M. B. Mogensen, Nat. Energy, 2016, 1, 15014 CrossRef CAS.
- S. W. Lee, T. H. Nam, M. Kim, S. Lee, K. H. Lee, J. H. Park and T. H. Shin, Inorg. Chem. Front., 2023, 10, 3536–3543 RSC.
- T. Wang, N. Sun, R. Wang, D. Dong, T. Wei and Z. Wang, J. Power Sources, 2025, 626, 235821 CrossRef CAS.
- M. C. Biesinger, B. P. Payne, A. P. Grosvenor, L. W. M. Lau, A. R. Gerson and R. St. C. Smart, Appl. Surf. Sci., 2011, 257, 2717–2730 CrossRef CAS.
- Y. Zhou, F. Wei and H. Wu, Electrochim. Acta, 2022, 419, 140434 CrossRef CAS.
- Y. Zhou, T. Liu, Y. Song, H. Lv, Q. Liu, N. Ta, X. Zhang and G. Wang, Chin. J. Catal., 2022, 43, 1710–1718 CrossRef CAS.
- Q. Shao, X. Han, K. Li, W. Guan, Y. Ling and Z. Lv, J. Colloid Interface Sci., 2025, 693, 137564 CrossRef CAS PubMed.
- S. Zhang, X. Hu, T. Liu, H. Liu, Y. Guo, G. Zou, W. Zhang, X. Zhang, P. Zhang, R. Yu, Y. Song, C. Xia, G. Wang and X. Bao, Adv. Mater., 2025, e18116 Search PubMed.
- J. Feng, Y. Wang, H. Li, Z. Zhao, H. Zheng and X. Ding, ACS Sustainable Chem. Eng., 2025, 13, 9565–9575 CrossRef CAS.
- T. Wang, N. Sun, R. Wang, D. Dong, T. Wei and Z. Wang, J. Power Sources, 2025, 626, 235821 CrossRef CAS.
- Y. Zhou, Z. Zhou, Y. Song, X. Zhang, F. Guan, H. Lv, Q. Liu, S. Miao, G. Wang and X. Bao, Nano Energy, 2018, 50, 43–51 CrossRef CAS.
- B. Qian, D. Zhang, P. Shan, H. Ye and Y. Zheng, J. Mater. Chem. A, 2025, 13, 16092–16101 RSC.
- F. Hu, B. He, K. Chen, W. Ma, Y. Huang, S. Zhao, Y. Chen and L. Zhao, Adv. Mater., 2025, e12310 Search PubMed.
- D. E. A. Gordon and R. M. Lambert, Surf. Sci., 1993, 287–288, 114–118 CrossRef CAS.
- K. Liu, L. Liao, L. Li, M. A. Nawaz, G. Liao and X. Xu, Surf. Interfaces, 2025, 56, 105496 CrossRef CAS.
- Y. Zhang, F. Ye and W.-D. Z. Li, Chem. – Eur. J., 2021, 27, 3766–3771 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |