Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Continuous-flow electrochemical production of H2O2 in a gas diffusion electrode microreactor
Desislava Yordanova
Apostolova
a,
Ema
Gričar
a,
Maris Minna
Mathew
 a,
Tomaž
Gril
a,
Tomaž
Gril
 a,
Miha
Nosan
a,
Ivo
Bardarov
ab and
Boštjan
Genorio
a,
Miha
Nosan
a,
Ivo
Bardarov
ab and
Boštjan
Genorio
 *a
*a
aFaculty of Chemistry and Chemical Technology, University of Ljubljana, Večna pot 113, 1000 Ljubljana, Slovenia. E-mail: bostjan.genorio@fkkt.uni-lj.si
bDepartment of Chemistry, South-West University “Neofit Rilski”, 66 Ivan Mihajlov Str., 2700 Blagoevgrad, Bulgaria
First published on 17th April 2026
Abstract
A microfluidic electrochemical reactor equipped with a gas diffusion cathode continuously produced up to 22.3 mM H2O2, with ∼88% faradaic efficiency and an energy consumption of 5.1 kWh kg−1. This performance surpasses current membraneless electrochemical H2O2 production processes, eliminating the need for a membrane or separator while maintaining high efficiency.
Decentralized electrochemical production of hydrogen peroxide (H2O2) offers a sustainable, energy-efficient and green alternative to the anthraquinone process. However, the decentralized process is still limited by the oxygen mass transport and reactor design limitations. Here, we report a modified microfluidic electrochemical reactor that integrates a gas diffusion electrode (GDE) cathodic half-cell to mitigate oxygen transport limitations in the selective two-electron oxygen reduction reaction (2e− ORR). Direct comparison against a parallel-plate configuration1 reveals significantly enhanced H2O2 productivity and oxygen utilization. Coupled with in-line optical monitoring of H2O2 production and additional testing with an off-line electrochemical sensor, this work clarifies the decisive role of microreactor architecture in governing H2O2 selectivity in a metal-free system.
H2O2 is an environmentally benign oxidant widely used in bleaching, wastewater treatment, chemical synthesis, and energy storage/conversion,2–4 with steadily increasing global demand.5 Industrial production is dominated by the centralized anthraquinone process, which requires hydrogen, organic solvents, and platinum group metals (PGMs), and involves transportation of concentrated H2O2, posing safety and logistical challenges.6 Electrochemical synthesis offers a decentralized and potentially safer route via a two-electron oxygen reduction reaction in alkaline media (eqn (1)).
| O2 + H2O + 2e− → HO2− + OH− E0 = 0.74 V vs. RHE | (1) |
Beyond electrocatalyst composition, reactor design critically governs oxygen delivery and H2O2 production. Ion-exchange membrane systems and GDE-based electrolyzers improve oxygen utilization and product concentration.15–17 On the other hand, membraneless microfluidic electrochemical flow cells enhance mass transport by controlling flow rate and optimizing channel dimensions, while also helping to minimize reactant crossover.18,19 Nevertheless, systematic comparisons of different reactor architectures and their direct impact on H2O2 production remain limited. In addition, quantification is typically performed ex situ rather than through in situ measurements.20–22
By integrating a GDE cathodic half-cell into a microfluidic platform and comparing it against a parallel-plate microreactor, we demonstrate how gaseous oxygen supply directly governs H2O2 production and selectivity. The optimized, metal-free configuration establishes practical design principles for efficient, decentralized electrochemical H2O2 synthesis.1,23
The electrochemical microreactor housing was fabricated by CNC milling from PTFE blocks (Fig. 1(a)). The microreactor has an internal channel volume of 21.6 µL, corresponding to a retention time of 25.9 s at a flow rate of 50 µL min−1 (eqn (S1)). In the anode compartment, a glassy carbon (GC) electrode was embedded and served as the counter electrode. The cathode compartment was equipped with a gas inlet and an integrated gas flow channel to accommodate the gas diffusion electrode (GDE). The gas diffusion layer (GDL) consisted of carbon paper (Sigracet 22 BB, Fuel Cell Store). The catalyst ink, prepared using Ketjenblack EC-300J (KJB EC-300J), ultrapure water (resistivity of 18.2 MΩ cm), 2-propanol (IPA), and Nafion 117 solution, was deposited onto the GDL by spray-coating. Multiple GDEs were prepared with a target electrocatalyst loading of 300 µg cm−2 on a GDL working electrode (WE) with an area of 36 mm2 (inset of Fig. 1(a)). Full details of the ink formulation and the preparation procedure are provided in the SI. A reference electrode (Ag/AgCl/3 M NaCl, BASi Research) was placed in a separate sealed vial connected to the microchannel system that supplied electrolyte to the reactor (Fig. 1(b)). All potentials are reported versus the reversible hydrogen electrode (RHE). The electrolyte (O2-saturated 0.1 M KOH) was delivered through the microreactor using a syringe pump. H2O2 production was monitored using an in-line optical detection system,21 and, for high current chronopotentiometric (CP) measurements, an off-line electrochemical sensor20 was used.
Electrochemical measurements were performed using a potentiostat/galvanostat (BioLogic SP-300). Full details of the electrochemical measurements and data acquisition are provided in the SI.
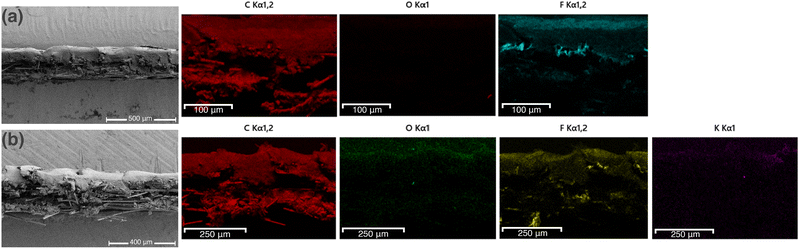
Before and after electrosynthesis experiments, characterization of the GDE electrodes was performed using scanning electron microscopy (SEM) coupled with energy-dispersive X-ray spectroscopy (EDS), Raman spectroscopy and X-ray photoelectron spectroscopy (XPS) (Fig. S3–S6). Cross-sectional SEM images (Fig. 2(a) and (b)) show that the spray-coated catalyst ink is deposited on the microporous layer (MPL), while the backing (support) layer of the GDE is clearly visible beneath it (shown in Fig. S3). Compared with the freshly coated sample, SEM analysis indicates no visible morphological changes; however, EDS analysis reveals an increased oxygen content on the catalyst surface after the electrochemical experiments.
This suggests surface oxidation during electrosynthesis of H2O2, which was further confirmed by the XPS results presented in Table 1.
| EDS [wt%] before EC | EDS [wt%] after EC | XPS [at%] before EC | XPS [at%] after EC | |
|---|---|---|---|---|
| WE | 1.7 | 4.1 | 5.26 | 9.56 |
The system (Fig. 1(b)) was designed to enable controlled CP for continuous-flow electrochemical H2O2 production in 0.1 M KOH and O2 flow to the cathode half-cell, while allowing real-time monitoring of H2O2 concentration using an integrated in-line optical sensor. Electrolyte flow was maintained at 50 µL min−1 using a syringe pump directly connected to the microreactor, and a mass flow controller regulated the O2 supply. The reference electrode was in electrolytic contact with the microreactor. Immediately downstream of the microreactor, an in-line bubble remover was installed to eliminate bubbles generated during the electrochemical measurements, likely originating from oxygen evolution at the counter electrode.1 This ensured uninterrupted flow to the optical sensor during H2O2 monitoring.
At the core of the system is the microreactor equipped with a GDE half-cell. KJB EC-300J powder was selected as the electrocatalyst due to its high selectivity and excellent processability, enabling the preparation of homogeneous catalyst inks. Before integration into the microfluidic setup, the electrocatalyst was evaluated using the rotating ring-disk electrode (RRDE) technique to determine H2O2 selectivity, which was found to be above 80% in the potential range 0.4–0.6 V vs. RHE (Fig. S7), a key requirement for this study.
Electrochemical impedance spectroscopy (EIS) measurements were performed before all electrochemical experiments, confirming an electrolyte resistance of 2–3 Ω for all measurements. The CP measurements were performed on different GDEs with similar loadings, using currents determined from cyclic voltammetry (CV), with current values selected based on specific potentials. As shown in Fig. S8, obtaining fully reproducible CVs at this stage was challenging. Nevertheless, the KJB EC-300J H2O2 selectivity at specific potentials on the GDE agrees with RRDE measurements.
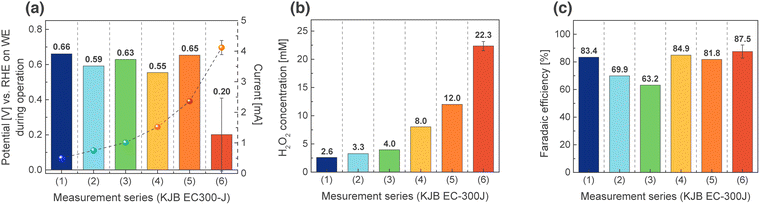
Recorded potentials of the WE during CP are shown in Fig. 3(a), with the corresponding H2O2 concentrations shown in Fig. 3(b). As expected, the measured H2O2 concentration increased with increasing applied current, reaching an average maximum of 22.3 mM at i = −4.11 mA (currents taken from CVs at E = 0.4 V vs. RHE). This represents approximately a 50-fold increase in H2O2 production compared with our previously published parallel-plate microreactor system,1 indicating that the implementation of a GDE and improved control over oxygen mass transport significantly enhance H2O2 production.
One limitation of applying high currents is the reduced reliable detection range of the in-line optical sensor. Up to reductive currents of 1.5 mA, the optical sensor functioned reliably, providing linear H2O2 determination shown in Fig. S2(a) and (b) with a correlation coefficient of 0.991. The H2O2 concentrations obtained from the in-line detection system are calculated using eqn (S2). However, at reductive currents exceeding 2 mA, reliable measurements were no longer possible due to the detection limit of the optical sensor. For these high-current measurements, an off-line electrochemical sensor was employed to determine reliable H2O2 concentrations. The calibration of the off-line electrochemical sensor is presented in Fig. S2(c). Detailed H2O2 concentrations from in-line and off-line detection are presented in Table S1.
The application of high currents also suggested possible electrocatalyst deactivation, as evidenced by the increased variability of the recorded WE potential. As shown in Fig. S9, WE and counter electrode (CE) potentials were stable at currents up to i = −2.4 mA, but at higher currents (Fig. S9(f)–(i)), the WE potential gradually dropped over time, indicating the need for higher overpotential to maintain H2O2 production. An exception was observed in Fig. S9(h), where, following a substantial potential drop, the measurement stabilized for the remainder of the experiment. We hypothesize that this behaviour may reflect in situ activation of the catalyst during electrochemical operation.
To investigate this possibility, consecutive CPs at various currents were performed on the same electrode (Fig. S10). The results indicate that a potential drop occurs at i = −1.5 mA, and subsequent repetitions suggest possible catalyst activation, as the system became more stable. While these observations are consistent with in situ activation (as indicated by the increase of oxygen content in Table 1), the exact mechanism remains unclear, and further studies are underway to confirm this phenomenon.
Faradaic efficiency (FE) was calculated using eqn (S3) for all electrochemical measurements. As shown in Fig. 3(c), the FE remained high, ranging from 63.2% to 87.5%, with the maximum observed at the highest H2O2 concentration. These results demonstrate the high efficiency of the system for the two-electron oxygen reduction reaction to H2O2.
The specific energy consumption of the system was calculated using eqn (S5) for the measurements that produced the highest H2O2 concentration based on the experimentally determined full-cell potential (eqn (S4) and Table S2). This resulted in an energy cost of approximately 5.12 kWh kg−1 H2O2, representing the average of four independent measurements performed under the best-performing operating conditions. For comparison, state-of-the-art electrochemical systems using a gas diffusion electrode with a membrane assembly consume 6.67 kWh kg−1 H2O2,24 indicating that the current membraneless configuration reduces energy consumption by about 23%. Although the conventional anthraquinone process requires 2.5–3.5 kWh kg−1 H2O2,25 these results show that the present system achieves competitive energy efficiency, even at this preliminary stage. The variation in energy efficiency observed between different measurement series (Table S2) can be attributed to minor fluctuations in cell voltage and H2O2 production, resulting from differences in oxygen mass transport, electrode wetting, and experimental uncertainties in product quantification.
In summary, a microfluidic electrochemical system with a gas diffusion electrode (GDE) enables efficient and controlled H2O2 production under alkaline conditions. Post-operation characterization showed increased surface oxygen content, indicating surface oxidation during catalysis that may contribute to catalyst activation. Real-time H2O2 monitoring with an in-line optical sensor, combined with continuous electrolyte flow and bubble management, ensures stable operation. H2O2 concentration increased with applied current, reaching 22.3 mM – approximately 50 times higher than a previously reported parallel-plate microreactor. High faradaic efficiencies (∼63–88%) confirm the dominance of the two-electron oxygen reduction reaction. Although optical sensor limitations and gradual catalyst deactivation occur at high currents, off-line measurements validate H2O2 profiles. The system achieves competitive energy consumption (∼5.12 kWh kg−1 H2O2), approaching the efficiency of the anthraquinone process and highlighting the potential of GDE-based membraneless microfluidic platforms for selective H2O2 electrosynthesis.
Author contributions
Desislava Yordanova Apostolova: writing – review & editing, writing – original draft, visualization, investigation, formal analysis, conceptualization. Ema Gričar: writing – review & editing, investigation, formal analysis. Maris Minna Mathew: writing – review & editing, investigation. Tomaž Gril: investigation, visualization. Miha Nosan: writing – review & editing, conceptualization. Ivo Bardarov: conceptualization. Boštjan Genorio: writing – review & editing, writing – original draft, visualization, supervision, project administration, methodology, investigation, funding acquisition, formal analysis, conceptualization.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data supporting this article have been included as part of the supplementary information (SI). The Supplementary information includes experimental protocols, characterization data, and extended experimental results, and is available at DOI: https://doi.org/10.1039/d6cc01364c.Raw data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgements
The financial support from the Slovenian Research and Innovation Agency (ARIS) through grants P2-0423, P1-0447, J7-4636, J2-50086, J7-50227, J2-60044, and L2-3161 and the infrastructure program I0-0022 is gratefully acknowledged. The authors would like to thank Torsten Mayr and Anders Ø. Tjell from TU Graz for providing the in-line optical H2O2 sensors. D. Y. Apostolova warmly thanks Jan Táborský for his assistance with the spray-coating procedure.References
- D. Y. Apostolova, I. Bardarov, A. Tjell, E. Gričar, M. Starin, P. F. B. D. Martins, M. Nosan, T. Mayr, D. Strmčnik, I. Plazl and B. Genorio, A parallel-plate electrochemical microreactor for the continuous production of hydrogen peroxide, Chem. Eng. J., 2025, 525, 170301 CrossRef CAS.
- S. C. Perry, D. Pangotra, L. Vieira, L. I. Csepei, V. Sieber, L. Wang, C. Ponce de León and F. C. Walsh, Electrochemical synthesis of hydrogen peroxide from water and oxygen, Nat. Rev. Chem., 2019, 3, 442–458 CrossRef CAS.
- R. Hage and A. Lienke, Applications of transition-metal catalysts to textile and wood-pulp bleaching, Angew. Chem., Int. Ed., 2005, 45, 206–222 CrossRef PubMed.
- S. Chen, Z. Chen, S. Siahrostami, T. R. Kim, D. Nordlund, D. Sokaras, S. Nowak, J. W. F. To, D. Higgins, R. Sinclair, J. K. Nørskov, T. F. Jaramillo and Z. Bao, Defective Carbon-Based Materials for the Electrochemical Synthesis of Hydrogen Peroxide, ACS Sustainable Chem. Eng., 2018, 6, 311–317 CrossRef CAS.
- Hydrogen Peroxide Market Size, Share|Growth Report, 2034, https://www.fortunebusinessinsights.com/hydrogen-peroxide-market-103920, accessed 24 February 2026.
- J. M. Campos-Martin, G. Blanco-Brieva and J. L. G. Fierro, Hydrogen peroxide synthesis: An outlook beyond the anthraquinone process, Angew. Chem., Int. Ed., 2006, 45, 6962–6984 CrossRef CAS PubMed.
- A. Verdaguer-Casadevall, D. Deiana, M. Karamad, S. Siahrostami, P. Malacrida, T. W. Hansen, J. Rossmeisl, I. Chorkendorff and I. E. L. Stephens, Trends in the electrochemical synthesis of H2O2: Enhancing activity and selectivity by electrocatalytic site engineering, Nano Lett., 2014, 14, 1603–1608 CrossRef CAS PubMed.
- X. Zhao and Y. Liu, Origin of Selective Production of Hydrogen Peroxide by Electrochemical Oxygen Reduction, J. Am. Chem. Soc., 2021, 143, 9423–9428 CrossRef CAS PubMed.
- Z. Lu, G. Chen, S. Siahrostami, Z. Chen, K. Liu, J. Xie, L. Liao, T. Wu, D. Lin, Y. Liu, T. F. Jaramillo, J. K. Nørskov and Y. Cui, High-efficiency oxygen reduction to hydrogen peroxide catalysed by oxidized carbon materials, Nat. Catal., 2018, 1, 156–162 CrossRef CAS.
- Z. Zheng, Y. H. Ng, D. W. Wang and R. Amal, Epitaxial Growth of Au–Pt–Ni Nanorods for Direct High Selectivity H2O2 Production, Adv. Mater., 2016, 28, 9949–9955 CrossRef CAS PubMed.
- H. Yang, W. Vogel, C. Lamy and N. Alonso-Vante, Structure and Electrocatalytic Activity of Carbon-Supported Pt-Ni Alloy Nanoparticles Toward the Oxygen Reduction Reaction, J. Phys. Chem. B, 2004, 108, 11024–11034 CrossRef CAS.
- M. Nosan, D. Strmčnik, V. Brusko, M. Kirsanova, M. Finšgar, A. M. Dimiev and B. Genorio, Correlating nickel functionalities to selectivity for hydrogen peroxide electrosynthesis, Sustainable Energy Fuels, 2023, 7, 2270–2278 RSC.
- I. Bardarov, D. Y. Apostolova, M. M. Mathew, M. Nosan, P. F. B. Dias Martins and B. Genorio, Flash Graphene: a Sustainable Prospect for Electrocatalysis, Acta Chim. Slov., 2024, 71, 541–557 CrossRef CAS.
- C. X. Zhao, B. Q. Li and Q. Zhang, Advanced electrosynthesis of hydrogen peroxide on oxidized carbon electrocatalyst, J. Energy Chem., 2019, 34, 10–11 CrossRef.
- X. Zhang, Y. Xia, C. Xia and H. Wang, Insights into Practical-Scale Electrochemical H2O2 Synthesis, Trends Chem., 2020, 2, 942–953 CrossRef CAS.
- C. Xia, Y. Xia, P. Zhu, L. Fan and H. Wang, Direct electrosynthesis of pure aqueous H2O2 solutions up to 20% by weight using a solid electrolyte, Science, 2019, 366, 226–231 CrossRef CAS PubMed.
- Z. Chen, S. Chen, S. Siahrostami, P. Chakthranont, C. Hahn, D. Nordlund, S. Dimosthenis, J. K. Nørskov, Z. Bao and T. F. Jaramillo, Development of a reactor with carbon catalysts for modular-scale, low-cost electrochemical generation of H2O2, React. Chem. Eng., 2017, 2, 239–245 RSC.
- P. Farinazzo Bergamo Dias Martins, I. Plazl, D. Strmcnik and B. Genorio, Prospect of microfluidic devices for on-site electrochemical production of hydrogen peroxide, Curr. Opin. Electrochem., 2023, 38, 101223 CrossRef CAS.
- R. Ferrigno, A. D. Stroock, T. D. Clark, M. Mayer and G. M. Whitesides, Membraneless vanadium redox fuel cell using laminar flow, J. Am. Chem. Soc., 2002, 124, 12930–12931 CrossRef CAS PubMed.
- E. Gričar, K. Kalcher, B. Genorio and M. Kolar, Highly sensitive amperometric detection of hydrogen peroxide in saliva based on n-doped graphene nanoribbons and MnO2 modified carbon paste electrodes, Sensors, 2021, 21, 8301 CrossRef PubMed.
- A. Tjell, B. Jud, R. Schaller-Ammann and T. Mayr, Optical hydrogen peroxide sensor for measurements in flow, Sens. Actuators, B, 2024, 400, 134904 CrossRef CAS.
- V. Lvovich and A. Scheeline, Amperometric Sensors for Simultaneous Superoxide and Hydrogen Peroxide Detection, J. Electroanal. Chem., 1987, 69, 454–462 Search PubMed.
- W. Xing, H. China and J. Zhang, Rotating electrode methods and oxygen reduction electrocatalysts, National Research Council of Canada, 2014 Search PubMed.
- Y. Zhao, N. Deng, Z. Fan, Z. T. Hu, L. Fan, J. Zhou and X. Huang, On-site H2O2 electro-generation process combined with ultraviolet: A promising approach for odorous compounds purification in drinking water system, Chem. Eng. J., 2022, 430, 132829 CrossRef CAS.
- P. Westbroek and E. Temmerman, Mechanism of hydrogen peroxide oxidation reaction at a glassy carbon electrode in alkaline solution, J. Electroanal. Chem., 2000, 482, 40–47 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |