Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Catalyst-free insertion of carbon monoxide into terminal Mg–C(sp3) bonds
M.
Kubisz
a,
I.
Fernandez
 *b and
T. J.
Hadlington
*b and
T. J.
Hadlington
 *a
*a
aFakultät für Chemie, Technische Universität München, Lichtenberg Strasse 4, 85747, Garching, Germany. E-mail: terrance.hadlington@tum.de
bDepartamento de Química Orgánica I and Centro de Innovación en Química Avanzada, Facultad de Ciencias Químicas, Universidad Complutense de Madrid, 28040, Madrid, Spain. E-mail: israel@quim.ucm.es
First published on 9th March 2026
Abstract
We describe the catalyst-free Insertion of carbon monoxide (CO) into magnesium alkyl bonds, leading exclusively to enediolate compounds through C–C coupling. Computational investigations suggest a metallated ketene intermedate. Ultimately, the formed enedoliate fragment can be liberated on reaction with a silane, establishing a potential pathway for s-block metal alkyl formylation catalysis.
The functionalization of carbon monoxide (CO) as an abundant C1 building block forms the foundation of numerous industrially significant catalytic processes,1 such as hydroformylation,2–4 the Fischer–Tropsch process,5–7 and the Pauson–Khand reaction.8,9 These processes are reliant on transition metal catalysts, perhaps the most prominent example being the cobalt and ruthenium catalysts required for alkene hydroformylation. Contemporary chemistry looks towards displacing these often high-cost, toxic systems with more sustainable derivatives.10,11 As such, recent efforts have explored the utility of main group elements in CO valorisation, demonstrating numerous stoichiometric transformations such as reductive CO coupling, complete C–O bond scission, and M–H insertion.12,13 In this space, the alkaline earth metals have been notably prolific.14
In landmark simultaneous reports, the groups of Hill and Jones demonstrated that magnesium hydride systems readily insert CO into their Mg–H bonds, leading to dimerization in forming ethylene diolate complexes (Fig. 1(a)),15,16 and can catalytically silylate CO to methanol equivalents.16 This was later extended to calcium and insertion into M–N bonds (M = Mg, Ca) in formal CO reductive amination,17 to Ba,18 and by our group to Be.19 The Jones group has also demonstrated numerous examples of reductive CO homologation using MgI dimers, leading to e.g. the ethenediolate, deltate and benzenehexolate anions.20,21 More recently, CO activation has been extended to anionic alkaline earth metal(I) dimers22,23 and hydride complexes.24 These promising transformations demonstrate the utility of these abundant metals in functionalising CO. Yet, catalyst-free alkaline earth-mediated transfer of this C1 building block to hydrocarbon functional groups is limited, stymying progress in displacing transition metals in hydroformylation catalyses. Early work here demonstrated that Grignard reagents react with a CO atmosphere in the presence of Ni(CO)4,25 or simply with an excess of Ni(CO)4 leading to a range of CO-functionalised products, including acyloins via CO coupling (Fig. 1(b)).26 In 2025, Maron, Xu, and co-workers extended this chemistry to well-defined DipNacnac-stabilised magnesium alkyl species (DipNacnac = [HC{C(Me)Ndip}2]−, Dip = 2,6-iPr2C6H3), leading to alkyl-substituted diolate and triolate complexes through Ni-catalysed cabonylation (Fig. 1(c)).27 To date, only highly strained28 and/or activated Mg–C bonds are known to insert CO without need for a catalyst,29 with occurrences being very rare indeed, whilst Harder and co-workers reported a unique reactive pathway involving 1,1-addition of CO into intramolecular Mg-benzyl fragments.30
 | ||
| Fig. 1 (a): initial examples of CO reactivity with alkaline earth hydrides; (b): early reports on the Ni-aided reactivity of CO with magnesium organyls; (c): recently reported Ni-catalysed CO insertion in Mg–C bonds; (d): this work – catalyst-free insertion of CO into Mg–C(sp3) bonds. | ||
In this work (Fig. 1(d)), we report the facile insertion of carbon monoxide into the Mg–C bonds of terminal magnesium alkyl complexes, in the absence of a catalyst. This exclusively leads to alkyl-functionalised ethene diolate products, with no significant formation of side-products. We additionally demonstrate that these species undergo reaction with phenyl silane, in the formation of the corresponding magnesium hydride complex. Computational assessment of the reaction coordinate suggests a divergent mechanism to that observed in hydride insertion, forming a ketenyl intermediate prior to formation of the target products. These results move a step closer to achieving transition metal-free alkene carbonylation processes.
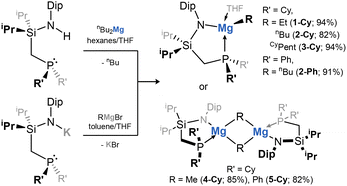
This work began with the synthesis of magnesium organyl complexes bearing our developed phosphine-appended amido ligands (viz.RL; RL = {[R2PCH2Si(iPr)2](Dip)N}; Dip = 2,6-iPr2C6H3; R = Ph, Cy), akin to reported beryllium hydride chemistry.19 This utilized two approaches: (i) the protonated ligands are reacted with nBu2Mg, in loss of butane and formation of RLMg(nBu), or (ii) the ligand potassium salts are reacted with Grignard reagents (i.e. RMgBr), in loss of KBr and formation of the target compounds (Scheme 1). This led to five complexes featuring the CyL ligand (R = Me, nBu, Et, CyPent, and Ph), and a single complex featuring the PhL ligand (R = nBu; Scheme 1).† Solid-state structures of complexes bearing Et (1-Cy), nBu (2-Cy and -Ph), and CyPent (3-Cy) groups, crystallise as 4-coordinate monomeric THF adducts (e.g.2-Ph, Fig. 2(a)), whilst Me and Ph complexes (4-Cy and 5-Cy, respectively) are isolated as THF-free dimeric species, bridged by their organyl groups. The range of P–Mg bond lengths (2.533–2.985 Å) lies within those of the relatively few phosphine-coordinated magnesium complexes reported.31–39 The shortest Mg–C distance of 2.130(6) Å, observed in 2-Cy, aligns with those in established monomeric magnesium alkyl species,40–42 as do those observed in dimeric 4-Cy and 5-Cy, which are somewhat longer (e.g. 2.654(1) Å in 4-Cy). The 31P{1H} NMR spectra display a singlet resonance for all compounds with shifts similar to those for RLK salts.43,44 Alkyl ligands are clearly observed in the 1H NMR spectra – notable high-field shifts are observed for the α-carbon of such species (e.g.2-Cy: −0.09 ppm).
Following isolation of the above magnesium organyls, their reactivity towards CO was investigated. Samples of nBu substituted 2-Cy or 2-Ph as solutions in C6D6 show a clear reaction with CO over the course of two days, in a Teflon-sealed NMR tube charged at atmospheric pressure. In addition to being sluggish, in situ31P{1H} NMR analyses were indicative of the formation of multiple products. To improve these points, the reactions were repeated on a preparative scale in a Teflon-sealed Schlenk flask (e.g. 200 mg) or Fischer Porter bottle (e.g. up to 1.8 g), with rapid stirring and 2 bar CO pressure.‡ After 24 h, NMR spectroscopic analysis indicates complete conversion of all starting material, and now formation of a single new species. 1H NMR spectra indicate a shift of the alkyl α-carbon C–H resonance away from those observed at −0.09 (2-Cy) and 0.20 (2-Ph) ppm, giving initial evidence for insertion into the Mg–C bonds in these species. Concentrated pentane solutions of these products stored at −30 °C overnight yield large colourless crystals – single-crystal X-ray diffraction (SC-XRD) analysis reveals these to be dimagnesium dec-5-enediolate complexes 6-Cy and 6-Ph (Fig. 3(a) and (b)), isolated in relatively low yields of 38 and 28% due to their high solubility. As observed in reported magnesium hydride chemistry, these species likely arise from initial insertion of CO into Mg–CBu bonds, followed by C–C coupling (vide infra). Importantly, this forms a new C10 chain through combining two C4 chains and two C1 building blocks, without an external catalyst or significantly activated Mg–C bonds. Importantly, such reactivity of metal-alkyls is only known for early transition metal systems (e.g. Zr, Hf),45–47 and lanthanide derivatives.48 Structurally, a clear C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond is found between C63 and C68 in the dec-5-ene chain (d = 1.362(2) Å), akin to distances observed in reported ethenediolate ligands. The 13C{1H} NMR spectra for both compounds are in keeping with this ligand motif: resonances relating to alkenyl-C centres are observed at δ = 139.7 (6-Cy) and 140.4 (6-Ph) ppm, with the α-C of the nBu chain observed at 34.5 (6-Cy) and 30.3 (6-Ph) ppm. A single ligand environment is observed for the two RL ligands, e.g. with a single resonance observed in their 31P{1H} NMR spectra (δ = −14.7 (6-Cy) and −23.6 (6-Ph) ppm), reflecting the solid-state structure of these species, whereby their phosphine chelating arms lie trans to the ethenyl unit (see Fig. 3(b)).
C bond is found between C63 and C68 in the dec-5-ene chain (d = 1.362(2) Å), akin to distances observed in reported ethenediolate ligands. The 13C{1H} NMR spectra for both compounds are in keeping with this ligand motif: resonances relating to alkenyl-C centres are observed at δ = 139.7 (6-Cy) and 140.4 (6-Ph) ppm, with the α-C of the nBu chain observed at 34.5 (6-Cy) and 30.3 (6-Ph) ppm. A single ligand environment is observed for the two RL ligands, e.g. with a single resonance observed in their 31P{1H} NMR spectra (δ = −14.7 (6-Cy) and −23.6 (6-Ph) ppm), reflecting the solid-state structure of these species, whereby their phosphine chelating arms lie trans to the ethenyl unit (see Fig. 3(b)).
Interestingly, this observed reactivity is divergent from recently reported Ni-catalysed insertion of CO into the Mg–CBu bond of [DipNacnacMgBu·(OEt2)], whereby three equiv. of CO react per two Mg centres, forming the unique [Bu3C3O3]−2 anion.27 Small R groups (e.g. Me) were found react akin to the above-described 2-Cy and 2-Ph. As such, we extended our reactivity studies to 1 and 3–5, to explore any differences. We are surprised to find that dimeric Me and Ph complexes (i.e.4-Cy and 5-Cy) in THF solutions do not react with CO, even after prolonged reaction times with heating, potentially due to their dimeric nature quenching reactivity. In contrast, monomeric Et and CyPent species 1-Cy and 3-Cy demonstrate clean reactivity towards CO (2 atm, RT), forming single species after 24 h. NMR spectroscopic data for the former is in keeping with that for 6-Cy, indicative of the formation of novel hex-3-enediolate magnesium complex, 7-Cy (Fig. 3(a)). In contrast, in situ1H NMR spectra for reactions with 3-Cy indicate the elimination of one equiv. cyclopentene per two Mg centres, and the presence of two new ligand environments (31P{1H} NMR: δ = −13.6 and −14.2 ppm). As per complexes 6 and 7, 13C NMR indicates the presence of a now unsymmetrical enediolate moiety (δ = 127.0, OC-H; 139.1, OC-CyPent ppm), whilst a new 1H singlet is observed at δ = 6.07 ppm in the 1H NMR which aligns with resonances for known ethenediolate C–H moieties (e.g. in Mg and Ca complexes in Fig. 1(a): 5.63 and 5.00 ppm, respectively), and in Xu's but-3-enediolate.15,17,27 Thus, reaction of CyPent compound 3-Cy with CO leads to the 2-cyclopentyl-ethenedoliate complex 8-Cy (Fig. 3(a)). This, again, is divergent from the reported Ni-catalysed reaction between [DipNacnacMg(CyPent)·(THF)] and CO, which rather leads to a methylenecyclopentane species through an intermediary 1,2-hydride shift. These observations highlight the effect of small changes in ligand on the course of a reaction in reactive CO chemistry, and indeed the effect of a catalytic transition metal on affecting the reaction coordinate.
Given the unique nature of the reaction between CO and the described magnesium alkyl complex, the mechanism for this process was investigated computationally by means of Density Functional Theory (DFT) calculations (see SI for details). As depicted in Fig. 3(b), the process begins with the slightly endergonic (ΔG = 2.7 kcal mol−1) coordination of CO to the magnesium center to produce INT1. From this species, insertion of the coordinated CO into the Mg–CEt bond takes place to produce the k2-C,O-propionyl complex INT2 in an exergonic process (ΔG = −3.6 kcal mol−1). This insertion reaction requires a low free activation barrier of 13.9 kcal mol−1 (from the separate reactants 1M and CO) viaTS1, a saddle point associated with the formation of the new C–C bond with concomitant Mg-CEt bond rupture. Coordination of a second molecule of CO leads to slightly endergonic (ΔG = 1.8 kcal mol−1) formation of intermediate INT3, from which a new, strongly exergonic (ΔG = −12.9 kcal mol−1) and barrierless insertion reaction takes place to produce the ketene intermediate INT4 (see SI, Fig. S91).
Once INT4 is formed, exergonic coordination of its ketene carbonyl group to a new molecule of 1M would lead to INT5, from which the final ethenediolate species 2M can be formed. This final, highly exergonic reaction (ΔG = −75.4 kcal mol−1) takes place viaTS2 with a rather low free activation barrier of only ΔG≠ = 5.1 kcal mol−1, which is fully consistent with a kinetically accessible process occurring at room temperature.
Finally, having observed the (catalyst-free) insertion of CO into Mg–C bonds, we sought to liberate the organic fragment in service of a magnesium-centred catalytic process akin to hydroformylation. No reaction is observed between complexes 6, 7, and 8 and dihydrogen (up to 2 bar, 70 °C). Reaction with a hydridic reagent, however, is more promising: addition of PhSiH3 to C6D6 solutions of 6-Cy leads to the slow formation of a new species (Scheme 2), with a clear triplet resonance observed in the 1H NMR spectrum at δ = 3.98 ppm – we attribute this to a dimeric Mg–H system, whereby the hydride ligand couples to two Cy2P-groups of the ligand scaffold. Storage of such samples overnight at ambient temperature leads to the formation of large colourless crystals. These are found to be hydride complex 9 (Fig. 4), formed through metathesis of Mg–O bonds in 6-Cy. Compound 9 is a relatively rare example of a phosphine-coordinated group 2 hydride complex, with previous examples being reported by Harder and co-workers.35,37 The Mg⋯Mg distance of 2.795(1) Å is similar to those observed in reported dimeric magnesium hydride complexes. The P-Mg distance is significantly contracted when compared with all other species reported here, at 2.556(1) Å, presumably due to the low-coordinate Mg centres in the absence of alkyl C–H⋯Mg stabilisation. We are surprised to find, in spite of this, that 9 does not readily coordinate THF, and seems to exclusively crystallise as the solvent-free dimer. This is in contrast to Nacnac-stabilised derivatives, whose reactivity in the solvent-free state is significantly amplified. This may suggest a high reactivity of group 2 complexes stabilised by our phosphine-appended ligand systems, e.g.RL, which we will report in due course.
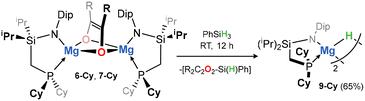 | ||
| Scheme 2 The reaction of complex 6-Cy with phenyl silane, forming magnesium hydride compound 9 (isolated yield in parentheses). | ||
MK: investigation, formalanalysis, data curation, writing – review and editing. IF: methodology (computational), formal analysis (computational), funding acquisition, writing – original draft and review and editing. TJH: conceptualisation, supervision, funding acquisition, project administration, writing – original draft.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cc00736h.CCDC 2528198–2528207 contain the supplementary crystallographic data for this paper.49a–j
Acknowledgements
T. J. H. thanks the ERC for a Starting grant (Project 101076897 – SINGAMBI), the DFG for funding (Project Nr. 523956566), and Prof. Thomas Fässler for his continued support. I. F. is grateful for financial support from the Spanish MICIU/AEI/10.13039/501100011033 (Grant PID2022-139318NB-I00).References
- Carbon Monoxide in Organic Synthesis: Carbonylation Chemistry, ed. B. Gabriele, Wiley, 1st edn, 2021 Search PubMed.
- A. Börner and R. Franke, Hydroformylation: fundamentals, processes, and applications in organic systhesis, Wiley-VCH Verlag GmbH & Co, Weinheim, 2016 Search PubMed.
- B. Zhang, D. Peña Fuentes and A. Börner, ChemTexts, 2022, 8, 2 CrossRef.
- R. Franke, D. Selent and A. Börner, Chem. Rev., 2012, 112, 5675–5732 CrossRef CAS PubMed.
- A. de Klerk, Fischer-Tropsch refining, Wiley-VCH Verlag & Co. KGaA, Weinheim, 2011 Search PubMed.
- K. T. Rommens and M. Saeys, Chem. Rev., 2023, 123, 5798–5858 CrossRef CAS PubMed.
- A. Yohannes and I. Gates, Coord. Chem. Rev., 2026, 547, 217096 CrossRef CAS.
- The Pauson-Khand Reaction: Scope, Variations and Applications, ed., R. Rios Torres, Wiley, 1st edn, 2012 Search PubMed.
- A. Lledó and J. Solà, Adv. Synth. Catal., 2024, 366, 574–592 CrossRef.
- R. Luque and F. Leung-Yuk Lam, Sustainable Catalysis: Energy-Efficient Reactions and Applications, Wiley, 1st edn, 2018 Search PubMed.
- K. M. P. Wheelhouse, R. L. Webster and G. L. Beutner, Org. Process Res. Dev., 2023, 27, 1157–1159 CrossRef CAS.
- S. Fujimori and S. Inoue, J. Am. Chem. Soc., 2022, 144, 2034–2050 CrossRef CAS PubMed.
- D. W. Stephan, Chem. Soc. Rev., 2023, 52, 4632–4643 RSC.
- M. J. Evans and C. Jones, Chem. Soc. Rev., 2024, 53, 5054–5082 RSC.
- R. Lalrempuia, C. E. Kefalidis, S. J. Bonyhady, B. Schwarze, L. Maron, A. Stasch and C. Jones, J. Am. Chem. Soc., 2015, 137, 8944–8947 CrossRef CAS PubMed.
- M. D. Anker, M. S. Hill, J. P. Lowe and M. F. Mahon, Angew. Chem., Int. Ed., 2015, 54, 10009–10011 CrossRef CAS PubMed.
- M. D. Anker, C. E. Kefalidis, Y. Yang, J. Fang, M. S. Hill, M. F. Mahon and L. Maron, J. Am. Chem. Soc., 2017, 139, 10036–10054 CrossRef CAS PubMed.
- X. Shi, C. Hou, C. Zhou, Y. Song and J. Cheng, Angew. Chem., Int. Ed., 2017, 56, 16650–16653 CrossRef CAS PubMed.
- T. J. Hadlington and T. Szilvási, Nat. Commun., 2022, 13, 461 CrossRef CAS PubMed.
- K. Yuvaraj, I. Douair, A. Paparo, L. Maron and C. Jones, J. Am. Chem. Soc., 2019, 141, 8764–8768 CrossRef CAS PubMed.
- A. Paparo, K. Yuvaraj, A. J. R. Matthews, I. Douair, L. Maron and C. Jones, Angew. Chem., Int. Ed., 2021, 60, 630–634 CrossRef CAS PubMed.
- H.-Y. Liu, R. J. Schwamm, S. E. Neale, M. S. Hill, C. L. McMullin and M. F. Mahon, J. Am. Chem. Soc., 2021, 143, 17851–17856 CrossRef CAS PubMed.
- D. T. Nguyen, R. Mondal, M. J. Evans, J. M. Parr and C. Jones, Angew. Chem., Int. Ed., 2025, 64, e202500264 CrossRef CAS PubMed.
- J. S. McMullen, R. Huo, P. Vasko, A. J. Edwards and J. Hicks, Angew. Chem., Int. Ed., 2023, 62, e202215218 CrossRef CAS PubMed.
- W. L. Gilliland and A. A. Blanchard, J. Am. Chem. Soc., 1926, 48, 410–420 CrossRef CAS.
- F. L. Benton, M. C. Voss and P. A. McCusker, J. Am. Chem. Soc., 1945, 67, 82–83 CrossRef CAS.
- M. Wang, Y. Cai, T. Rajeshkumar, L. Maron and X. Xu, Chem. – Eur. J., 2025, 31, e202404594 CrossRef CAS PubMed.
- K. Yuvaraj and C. Jones, Chem. Commun., 2021, 57, 9224–9227 RSC.
- A. J. Boutland, A. Carroll, C. Alvarez Lamsfus, A. Stasch, L. Maron and C. Jones, J. Am. Chem. Soc., 2017, 139, 18190–18193 CrossRef CAS PubMed.
- O. P. E. Townrow, T. Richter, J. Langer and S. Harder, Chem. – Eur. J., 2025, 31, e202404028 CrossRef CAS PubMed.
- J. Pahl, T. E. Stennett, M. Volland, D. M. Guldi and S. Harder, Chem. – Eur. J., 2019, 25, 2025–2034 CrossRef CAS PubMed.
- A. Pape, M. Lutz and G. Müller, Angew. Chem., Int. Ed. Engl., 1994, 33, 2281–2284 CrossRef.
- M. M. Meinholz, S. K. Pandey, S. M. Deuerlein and D. Stalke, Dalton Trans., 2011, 40, 1662 RSC.
- N. D. Contrella and R. F. Jordan, Organometallics, 2014, 33, 7199–7208 CrossRef CAS.
- J. Langer, I. Kosygin, R. Puchta, J. Pahl and S. Harder, Chem. – Eur. J., 2016, 22, 17425–17435 CrossRef CAS PubMed.
- A. Koch, S. Krieck, H. Görls and M. Westerhausen, Inorganics, 2016, 4, 39 CrossRef.
- J. Langer, B. Maitland, S. Grams, A. Ciucka, J. Pahl, H. Elsen and S. Harder, Angew. Chem., Int. Ed., 2017, 56, 5021–5025 CrossRef CAS PubMed.
- S. Bestgen, C. Schoo, B. L. Neumeier, T. J. Feuerstein, C. Zovko, R. Köppe, C. Feldmann and P. W. Roesky, Angew. Chem., Int. Ed., 2018, 57, 14265–14269 CrossRef CAS PubMed.
- L. Garcia, M. D. Anker, M. F. Mahon, L. Maron and M. S. Hill, Dalton Trans., 2018, 47, 12684–12693 RSC.
- J. Spielmann, D. F.-J. Piesik and S. Harder, Chem. – Eur. J., 2010, 16, 8307–8318 CrossRef CAS PubMed.
- M. H. Chisholm, K. Choojun, A. S. Chow, G. Fraenkel and J. C. Gallucci, Inorg. Chem., 2013, 52, 11302–11310 CrossRef CAS PubMed.
- M. S. Hill, D. J. MacDougall and M. F. Mahon, Dalton Trans., 2010, 39, 11129 RSC.
- P. M. Keil, T. Szilvási and T. J. Hadlington, Chem. Sci., 2021, 12, 5582–5590 Search PubMed.
- T. L. Kalkuhl, I. Fernández and T. J. Hadlington, Chemistry, 2025, 11, 102349 Search PubMed.
- P. Hofmann, M. Frede, P. Stauffert, W. Lasser and U. Thewalt, Angew. Chem., Int. Ed. Engl., 1985, 24, 712–713 CrossRef.
- G. Erker, P. Czisch, R. Schlund, K. Angermund and C. Krüger, Angew. Chem., Int. Ed. Engl., 1986, 25, 364–365 Search PubMed.
- L. P. Spencer and M. D. Fryzuk, J. Organomet. Chem., 2005, 690, 5788–5803 CrossRef CAS.
- M. R. Luevano, C. R. Stennett, J. W. Ziller and W. J. Evans, J. Am. Chem. Soc., 2025, 147(15), 12386–12391 CrossRef CAS PubMed.
- (a) CCDC 2528198: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qvss5; (b) CCDC 2528199: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qvst6; (c) CCDC 2528200: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qvsv7; (d) CCDC 2528201: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qvsw8; (e) CCDC 2528202: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qvsx9; (f) CCDC 2528203: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qvsyb; (g) CCDC 2528204: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qvszc; (h) CCDC 2528205: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qvt0f; (i) CCDC 2528206: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qvt1g; (j) CCDC 2528207: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qvt2h.
Footnotes |
| † On one occasion, a few crystals of the Et2Mg complex CyLMgEt(μ-MgEt2)EtMgCyL were found co-crystallised with 3. These could not be separated, and as such clean analytical data for that complex could not be attained. Nevertheless, the X-ray crystal structure is given in the Supporting Information (Fig. S90). |
| ‡ We presume that higher pressures promote more selective reactions due to the low solubility of CO in aliphatic solvents – that is, lower CO pressures perhaps lead to longer-lived intermediates, which undergo divergent reactivity. |
| This journal is © The Royal Society of Chemistry 2026 |