Open Access Article
Open Access ArticleA triazatruxene decorated multi-resonance emitter with hybrid short- and long-range charge-transfer excited states enables efficient narrowband deep-blue electroluminescence
Ying
Gao†
ab,
Xiaoyu
Guo†
a,
Bingjie
Xie
a,
Jingsheng
Miao
a,
Yang
Zou
 *a and
Chuluo
Yang
*a and
Chuluo
Yang
 *a
*a
aShenzhen Key Laboratory of New Information Display and Storage Materials, College of Materials Science and Engineering, Shenzhen University, Shenzhen 518060, PR China. E-mail: yangzou@szu.edu.cn, clyang@szu.edu.cn
bCollege of Physics and Optoelectronic Engineering, Shenzhen University, Shenzhen 518060, People's Republic of China
First published on 31st March 2026
Abstract
A TADF emitter with hybrid short- and long-range charge-transfer excited states is designed by grafting a triazatruxene donor onto a multi-resonance framework. The emitter exhibits narrowband deep-blue emission with enhanced photophysical properties and enables efficient deep-blue electroluminescence.
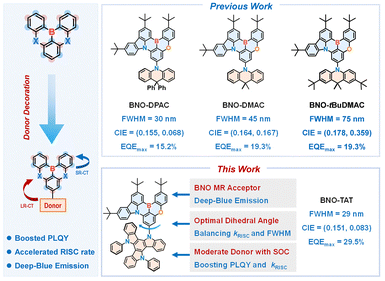
Thermally activated delayed fluorescence (TADF) materials enable high electroluminescence efficiency without noble metals.1 Conventional donor–acceptor (D–A) TADF emitters effectively reduce the singlet–triplet energy gap (ΔEST) to promote reverse intersystem crossing (RISC), but their long-range charge-transfer (LR-CT) character often leads to broad emission and poor color purity, limiting deep-blue OLED applications. To address this issue, the multiple-resonance (MR) strategy introduced by Hatakeyama et al. employs rigid, planar frameworks with atomically defined donor and acceptor units, producing short-range charge-transfer (SR-CT) excited states and narrowband emission.2,3 However, the highly localized electronic structures of MR-TADF emitters generally result in slow RISC rates (kRISC), causing incomplete triplet harvesting and efficiency loss.4,5 According to Fermi's golden rule, kRISC can be enhanced by reducing ΔEST or increasing spin–orbit coupling (SOC),6 yet ΔEST reduction in MR systems is intrinsically limited. While heavy-atom incorporation enhances SOC,7–9 it may compromise stability.10,11 Alternatively, boron-based MR frameworks can introduce a “hidden” LR-CT state, providing a heavy-atom-free route to accelerate kRISC and achieve high-efficiency, color-pure OLEDs.12–16
As one of the primary colors, deep-blue emission plays a critical role in next-generation display technologies.17 The MR framework BNO, composed of carbazole and phenol as resonant partners, is a well-established building block for deep-blue emitters.18,19 Despite its intrinsic narrowband deep-blue emission, BNO-based MR emitters typically suffer from moderate photoluminescence quantum yields (PLQYs) and slow kRISC, which result in limited device efficiency and pronounced efficiency roll-off in electroluminescent devices. To address these limitations, we sought to accelerate kRISC in BNO-based MR emitters by introducing additional LR-CT excited states. Accordingly, a series of MR-TADF emitters—BNO-DPAC, BNO-DMAC, and BNO-tBuDMAC—were designed by decorating the BNO MR moiety with acridine-based donors (Scheme 1).20 Systematic investigation revealed that the donor structure exerts a profound influence on the photophysical properties. BNO-DPAC, featuring a quasi-axial acridine conformation, exhibits the weakest LR-CT character, thereby retaining narrowband deep-blue emission but suffering from low PLQY and slow kRISC. In contrast, the stronger donors in BNO-DMAC and BNO-tBuDMAC significantly enhance PLQY and kRISC but induce pronounced red shifts and spectral broadening, deviating from the desired deep-blue region. These results highlight the critical yet challenging requirement of precisely balancing donor strength to activate LR-CT-assisted RISC while preserving narrowband deep-blue emission originating from SR-CT states.
Triazatruxene (TAT) is a well-established donor motif in D–A-type TADF emitters, known for its high PLQY and fast kRISC.21–23 We proposed TAT as an ideal donor for BNO-based emitters with coexisting short- and long-range charge-transfer (SR/LR-CT) excited states, aiming to achieve narrowband deep-blue emission, high PLQY, and accelerated kRISC simultaneously. This design is supported by the moderate electron-donating strength of TAT, comparable to that of DMAC, and its unique C3h-symmetric double indolo[3,2-b]carbazole framework, which reduces the D–A twisting angle and enables well-regulated LR-CT character. Moreover, the triarylamine-rich structure facilitates the formation of multiple CT excited states with enhanced spin–orbit coupling, providing additional pathways for triplet harvesting. Based on this strategy, a BNO-TAT emitter was synthesized, exhibiting a significantly accelerated kRISC. Sensitizer-free OLEDs based on BNO-TAT achieved efficient deep-blue emission with preserved color purity and an EQE approaching 30%, nearly double that of the BNO-DPAC-based control device.
The chemical structure of the designed emitter BNO-TAT is shown in Scheme 1, along with BNO-DPAC as the control emitter. The BNO acceptor moiety was prepared via sequential nucleophilic substitution and borylation reactions. Subsequently, the TAT and the acceptor fragment were coupled through a Buchwald–Hartwig amination to afford the target product BNO-TAT (Scheme S1). Its chemical structure was unambiguously confirmed by 1H NMR, 13C NMR, 2D COSY and ROESY NMR spectroscopy and high-resolution mass spectrometry (Fig. S1–S6, SI). Moreover, thermogravimetric analysis (TGA, Fig. S7) revealed a thermal decomposition temperature of 490 °C for BNO-TAT, demonstrating excellent thermal stability and confirming its suitability for vacuum thermal evaporation in device fabrication.
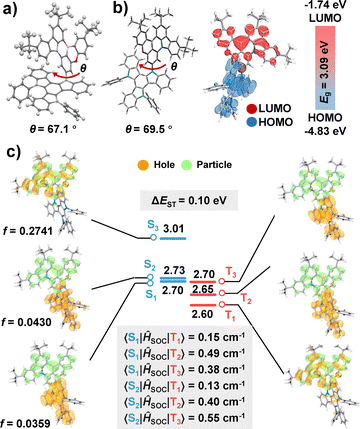
To further elucidate the rationality of the molecular design, density functional theory (DFT) and time-dependent DFT (TD-DFT) calculations were carried out using the Gaussian 16 software package with the B3LYP functional. As depicted in Fig. 1 and Fig. S9–S11, the highest occupied molecular orbital (HOMO) is primarily localized on the TAT donor, while the lowest unoccupied molecular orbital (LUMO) resides on the MR-type acceptor fragment, confirming the intended D–A electronic separation. The calculated dihedral angle between the donor and acceptor units is 69.5°, which is close to that of the single crystal structure, and consistent with the design objective of achieving a well-regulated LR-CT character. Natural transition orbital (NTO) analysis reveals that both BNO-TAT and BNO-DPAC exhibit pronounced LR-CT character in their S1 states, accompanied by several closely lying higher singlet (Sn) states with SR-CT character within an energy window of 0.30 eV.
Although the two emitters possess comparable T1 energies, the stronger electron-donating ability of TAT stabilizes the S1 state of BNO-TAT, resulting in a reduced singlet–triplet energy gap (ΔEST) of 0.10 eV, approximately half that of BNO-DPAC (0.24 eV), indicative of enhanced TADF behavior. Notably, BNO-TAT exhibits a significantly denser manifold of singlet and triplet excited states than BNO-DPAC (Fig. S11), with four nearly degenerate singlet and triplet states distributed within 0.08 eV. This dense excited-state landscape facilitates multiple triplet exciton upconversion pathways, thereby enhancing exciton utilization efficiency in the electroluminescence (EL) devices. Furthermore, NTO analysis of the T1–T3 states reveals hybrid SR/LR-CT character, with greater spatial overlap between hole and particle distributions in the Sn and Tn states of BNO-TAT compared to BNO-DPAC. Such NTOs lead to markedly enhanced spin–orbit coupling matrix elements (SOCMEs) of 0.49 and 0.38 cm−1 for 〈S1|ĤSOC|T2〉 and 〈S1|ĤSOC|T3〉, respectively. These values are substantially higher than those of BNO-DPAC (0.04 and 0.09 cm−1, Fig. S5). In addition, the oscillator strength of the S1 state in BNO-TAT (f = 0.0359) is nearly an order of magnitude larger than that of BNO-DPAC (f = 0.0032), consistent with its higher PLQY. Collectively, these computational results provide strong theoretical support for the superior photophysical and electroluminescence performance of BNO-TAT.
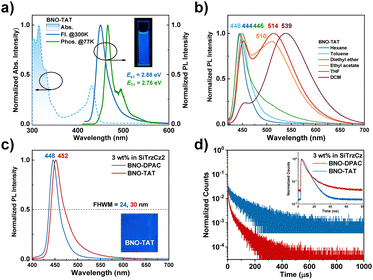
The absorption, fluorescence (Fl.), and phosphorescence (Phos.) spectra of BNO-TAT were measured in a dilute toluene solution. As shown in Fig. 2a and Fig. S12, BNO-TAT exhibits strong broadband absorption in the range of 300–370 nm, primarily attributed to π–π* and n–π* transitions of the arylamine and BNO moieties. The intense primary absorption band centered at 430 nm is assigned to the SR-CT-type transitions from the MR-type acceptor. In a dilute toluene solution, BNO-TAT exhibits an emission maximum at 450 nm with a full width at half maximum (FWHM) of only 26 nm, indicating a negligible spectral shift compared to BNO-DPAC. This spectral similarity underlies achieving high-purity deep-blue electroluminescence in OLEDs. Moreover, based on the onset of fluorescence and phosphorescence spectra, the ΔEST for BNO-TAT is calculated to be only 0.12 eV, indicating its TADF character.
Beyond the intrinsic narrowband deep-blue emission arising from the SR-CT excited state, solvatochromic studies were performed to elucidate the presence of a hidden LR-CT character. As shown in Fig. 2b, BNO-TAT exhibits a dominant sharp emission peak at approximately 450 nm in low- to medium-polarity solvents (hexane and toluene), which is characteristic of SR-CT fluorescence. Upon increasing the solvent polarity, a weak tailing emission beyond 500 nm emerges in diethyl ether. Notably, in more polar solvents, BNO-TAT displays a clear dual-emission behavior: the narrow emission centered at ∼450 nm remains essentially unchanged with increasing polarity, confirming its SR-CT origin, while a broad emission band undergoes a pronounced bathochromic shift from 510 nm in ethyl acetate to 514 nm in tetrahydrofuran and ultimately becomes the dominant peak at 539 nm in dichloromethane. This pronounced solvatochromic response provides direct evidence for the involvement of LR-CT excited states in BNO-TAT, thereby validating the proposed molecular design strategy. In contrast, the reference compound BNO-DPAC exhibits only a weak shoulder around 530 nm even in highly polar dichloromethane,20 indicating a much less pronounced LR-CT contribution. This comparison highlights the strengthened LR-CT character in BNO-TAT, which is expected to underpin its enhanced photophysical performance.
When doped into 9,9′-(6-(3-(triphenylsilyl)phenyl)-1,3,5-triazine-2,4-diyl)bis(9H-carbazole) (SiTrzCz2) at a concentration of 3 wt%, both emitters exhibit strong photoluminescence, achieving PLQYs of 99% for BNO-TAT and 92% for BNO-DPAC. As shown in Fig. 2c, the doped films display emission maxima at 448 nm for BNO-TAT and 452 nm for BNO-DPAC. Compared to their emissions in dilute toluene solutions, both materials show slight red shifts and moderate spectral broadening, which can be attributed to π–π interactions with the host matrix. Notably, in contrast to the previously reported BNO-DMAC, which exhibits a significantly broadened emission (FWHM = 56 nm) due to excessive LR-CT character, BNO-TAT maintains a much narrower emission bandwidth (FWHM = 30 nm) due to ideal hybrid SR/LR-CT character. This well-preserved narrowband deep-blue emission is critical for achieving high color purity in display applications.
Transient photoluminescence decay measurements were conducted to evaluate the TADF characteristics of both emitters. As illustrated in Fig. 2d, BNO-TAT exhibits a longer prompt fluorescence lifetime (τp = 10.0 ns) than BNO-DPAC (τp = 6.1 ns), consistent with its enhanced LR-CT character. BNO-TAT shows a pronounced temperature dependence (Fig. S13), with a delayed fluorescence lifetime of 28.4 ± 0.2 µs and a delayed component contribution of approximately 68%, indicative of dominant TADF behavior. In contrast, BNO-DPAC exhibits a longer delayed lifetime of 66.2 ± 0.2 µs and a lower delayed component contribution of 52%. Benefiting from its well-regulated LR-CT character, BNO-TAT not only achieves near-unity PLQY but also exhibits an accelerated kRISC of 1.1 × 105 s−1, over three times that of BNO-DPAC (Table S1). These results provide compelling evidence for more efficient triplet exciton utilization, underscoring the advantages of this design strategy for high-performance OLED applications.
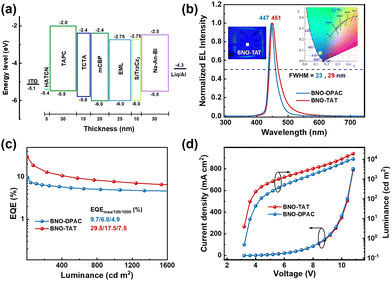
OLEDs were fabricated to evaluate the electroluminescence performance of the two emitters, employing the following layer structure: indium tin oxide (ITO)/1,4,5,8,9-hexaazatriphenylene-hexacarbonitrile (HATCN, 5 nm)/1,1-bis[(di-4-tolylamino)-phenyl]cyclohexane (TAPC, 30 nm)/tris(4-carbazol-9-ylphenyl)amine (TCTA, 15 nm)/1,3-di(9H-carbazol-9-yl)benzene (mCBP, 20 nm)/emissive layer (EML, 25 nm)/9,9′-(6-(4-(triphenylsilyl)phenyl)-1,3,5-triazine-2,4-diyl)bis(9H-carbazole) (SiTrzCz2, 10 nm)/2-ethyl-1-(4-(10-(naphthalen-2-yl)anthracen-9-yl)phenyl)-1H-benzo[d]imidazole (Na-An-BI, 30 nm)/lithium quinolate (Liq, 2 nm)/aluminum (Al, 100 nm). In this structure, TAPC and Na-An-BI function as hole-transport and electron-transport layers, respectively, while mCBP serves as both a hole-blocking and an exciton-confining layer. HATCN acts as an anode interfacial layer, and Liq serves as a cathode interfacial layer. Given the HOMO energy level of the emitter at −5.12 eV, the EML was adopted with a doping system of SiTrzCz2: 1 wt%, 3 wt%, 5 wt%, and 7 wt% BNO-TAT and 3 wt% BNO-DPAC, where SiTrzCz2 was used as the host material. The chemical structures of the materials used in the EL device are shown in Fig. S14.
As shown in Fig. 3 and Fig. S15–S17 and Table S2, at a doping concentration of 1 wt%, the BNO-TAT-based device displayed a narrowband deep-blue emission peaking at 448 nm and a FWHM of 28 nm, corresponding to the Commission Internationale de l'Éclairage (CIE) chromaticity coordinates of (0.156, 0.069). Aside from the well-maintained narrowband deep-blue emission, the maximum external quantum efficiency (EQEmax) and maximum brightness (Lmax) reached 25.8% and 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 780 cd m−2, which are significantly superior to those of other BNO-based emitters reported in the literature.18–20 At a doping concentration of 3 wt%, BNO-TAT delivered further optimized performance, with stable emission peaking at 446 nm and a FWHM of 29 nm. The EL peak positions closely match those of the corresponding PL spectra of the doped films. The EQEmax reaches up to 30%, representing a twofold enhancement compared to the control device using BNO-DPAC as the emitter, while the peak brightness reaches 18916 cd m−2, which is nearly double that of the BNO-DPAC-based device. These much superior electroluminescent characteristics can be attributed to the intrinsically high PLQY of BNO-TAT and the efficient utilization of triplet excitons enabled by its TADF mechanism.
780 cd m−2, which are significantly superior to those of other BNO-based emitters reported in the literature.18–20 At a doping concentration of 3 wt%, BNO-TAT delivered further optimized performance, with stable emission peaking at 446 nm and a FWHM of 29 nm. The EL peak positions closely match those of the corresponding PL spectra of the doped films. The EQEmax reaches up to 30%, representing a twofold enhancement compared to the control device using BNO-DPAC as the emitter, while the peak brightness reaches 18916 cd m−2, which is nearly double that of the BNO-DPAC-based device. These much superior electroluminescent characteristics can be attributed to the intrinsically high PLQY of BNO-TAT and the efficient utilization of triplet excitons enabled by its TADF mechanism.
In conclusion, by decorating a triazatruxene donor onto a BNO MR framework, we have developed an emitter that features hybrid SR/LR-CT excited states. The well-regulated LR-CT character introduced by the triazatruxene unit preserves the intrinsic narrowband deep-blue emission and high PLQY of the MR moiety, while enabling enhanced photophysical properties through a dense manifold of excited states that facilitates more efficient triplet exciton utilization. As a result, the emitter exhibits a high PLQY together with accelerated kRISC without sacrificing spectral narrowness in the deep-blue region. Consequently, doped OLEDs based on this emitter achieve an EQEmax of 29.5% while maintaining narrowband deep-blue emission, representing a highly competitive performance among state-of-the-art deep-blue emitters.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental section, related spectra of the materials; TGA and electrochemical curves, and electroluminescence data. See DOI: https://doi.org/10.1039/d6cc00501b.CCDC 2536739 contains the supplementary crystallographic data for this paper.24
Acknowledgements
We gratefully acknowledge financial support from the National Natural Science Foundation of China (52573203 and 52130308), the Guangdong Basic and Applied Basic Research Foundation (2025A1515010777), the Shenzhen Science and Technology Program (ZDSYS20210623091813040 and JCYJ20250604182224032), and the Research Team Cultivation Program of Shenzhen University (2023DFT004). We also thank the Instrumental Analysis Center of Shenzhen University for analytical support.References
- Y. Liu, C. Li, Z. Ren, S. Yan and M. R. Bryce, Nat. Rev. Mater., 2018, 3, 18020 CrossRef CAS.
- T. Hatakeyama, K. Shiren, K. Nakajima, S. Nomura, S. Nakatsuka, K. Kinoshita, J. Ni, Y. Ono and T. Ikuta, Adv. Mater., 2016, 28, 2777–2781 CrossRef CAS PubMed.
- Y. Kondo, K. Yoshiura, S. Kitera, H. Nishi, S. Oda, H. Gotoh, Y. Sasada, M. Yanai and T. Hatakeyama, Nat.Photonics, 2019, 13, 678–682 CrossRef CAS.
- X. Wu, S. Ni, C. H. Wang, W. Zhu and P. T. Chou, Chem. Rev., 2025, 125, 6685–6752 CrossRef CAS PubMed.
- M. Mamada, M. Hayakawa, J. Ochi and T. Hatakeyama, Chem. Soc. Rev., 2024, 53, 1624–1692 RSC.
- P. K. Samanta, D. Kim, V. Coropceanu and J.-L. Brédas, J. Am. Chem. Soc., 2017, 139, 4042–4051 CrossRef CAS PubMed.
- Y. X. Hu, J. Miao, T. Hua, Z. Huang, Y. Qi, Y. Zou, Y. Qiu, H. Xia, H. Liu, X. Cao and C. Yang, Nat. Photonics, 2022, 16, 803–810 CrossRef CAS.
- I. S. Park, H. Min and T. Yasuda, Angew. Chem., Int. Ed., 2022, 61, e202205684 Search PubMed.
- H. Lin, Z. Ye, S. Xian, Z. Chen, J. Miao, Z. Huang, C. Zhong, S. Gong, X. Cao and C. Yang, Adv. Mater., 2025, 37, e2502459 CrossRef PubMed.
- J. Liu, J. Miao, J. Dong, Z. Chen, Z. Huang and C. Yang, Adv. Mater., 2026, 38, e13987 CrossRef CAS PubMed.
- Y. Zou, M. Yu, Y. Xu, Z. Xiao, X. Song, Y. Hu, Z. Xu, C. Zhong, J. He, X. Cao, K. Li, J. Miao and C. Yang, Chemistry, 2024, 10, 1485–1501 Search PubMed.
- J. Han, Y. Chen, N. Li, Z. Huang and C. Yang, Aggregate, 2022, 3, e182 CrossRef CAS.
- Y. Xu, C. Li, Z. Li, Q. Wang, X. Cai, J. Wei and Y. Wang, Angew. Chem., Int. Ed., 2020, 59, 17442–17446 CrossRef CAS PubMed.
- S. Liao, Z. Zhang, Y. Gao, Z. Chen, J. Miao, Y. Zou and C. Yang, Chin. J. Chem., 2025, 44, 303–310 CrossRef.
- Y.-H. He, J.-Y. Liu, Z. Zhang, G.-W. Chen, Y.-C. Wang, G. Yuan, F.-M. Xie, J.-X. Tang and Y.-Q. Li, Matter, 2025, 8, 102188 CrossRef CAS.
- Z. Huang, H. Xie, J. Miao, Y. Wei, Y. Zou, T. Hua, X. Cao and C. Yang, J. Am. Chem. Soc, 2023, 145, 12550–12560 Search PubMed.
- A. Monkman, ACS Appl. Mater. Interfaces, 2022, 14, 20463–20467 Search PubMed.
- J. Han, Z. Huang, X. Lv, J. Miao, Y. Qiu, X. Cao and C. Yang, Adv. Opt. Mater., 2021, 10, 2102092 Search PubMed.
- J. Park, J. Lim, J. H. Lee, B. Jang, J. H. Han, S. S. Yoon and J. Y. Lee, ACS Appl. Mater. Interfaces, 2021, 13, 45798–45805 Search PubMed.
- X. Huang, Y. Xu, J. Miao, Y.-Y. Jing, S. Wang, Z. Ye, Z. Huang, X. Cao and C. Yang, J. Mater. Chem. C, 2023, 11, 11885–11894 Search PubMed.
- P. L. Dos Santos, J. S. Ward, D. G. Congrave, A. S. Batsanov, J. Eng, J. E. Stacey, T. J. Penfold, A. P. Monkman and M. R. Bryce, Adv. Sci., 2018, 5, 1700989 Search PubMed.
- D. H. Ahn, S. W. Kim, H. Lee, I. J. Ko, D. Karthik, J. Y. Lee and J. H. Kwon, Nat. Photonics, 2019, 13, 540 Search PubMed.
- K. R. Naveen, H. Lee, R. Braveenth, D. Karthik, K. J. Yang, S. J. Hwang and J. H. Kwon, Adv. Funct. Mater., 2021, 32, 2110356 Search PubMed.
- CCDC 2536739: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2r4p9x.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |