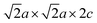Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Structure and magnetism of mixed-valent iron oxysulfides Sr3Fe4O6S2 (Sr3(Fe3+)2O5(Fe2+)2OS2) and Sr4Fe4O7S2 (Sr4(Fe3+)2O6(Fe2+)2OS2)
Alexis N.
Gillette
a,
Annabel R.
Lyon
 a,
Stanislav
Savvin
bc,
Ines
Puente-Orench
b,
Sepideh
Rahimisheikh
d,
Joke
Hadermann
a,
Stanislav
Savvin
bc,
Ines
Puente-Orench
b,
Sepideh
Rahimisheikh
d,
Joke
Hadermann
 d,
Magali
Gimeno
a,
Michael L.
Neidig
d,
Magali
Gimeno
a,
Michael L.
Neidig
 a and
Simon J.
Clarke
a and
Simon J.
Clarke
 *a
*a
aDepartment of Chemistry, University of Oxford, Inorganic Chemistry Laboratory, South Parks Rd, Oxford, OX1 3QR, UK. E-mail: simon.clarke@chem.ox.ac.uk
bInstitut Laue-Langevin, 71 Avenue des Martyrs, 38000, Grenoble, France
cInstituto de Nanociencia y Materiales de Aragón, CSIC – Universidad de Zaragoza, Facultad de Ciencias C/Pedro Cerbuna 12, Zaragoza, Spain
dElectron Microscopy for Materials Science (EMAT), University of Antwerp, Groenenborgerlaan 171, B-2020, Antwerp, Belgium
First published on 9th February 2026
Abstract
The crystal and magnetic structures of Sr3Fe4O6S2 (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Sr3Fe2O5Fe2OS2) and Sr4Fe4O7S2 (
Sr3Fe2O5Fe2OS2) and Sr4Fe4O7S2 (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Sr4Fe2O6Fe2OS2), designed using a building-block approach, are reported. They are fully charge-ordered with Fe2+ and Fe3+ ions in distinct layers showing independent long-range magnetic order. Complex microstructures in some regions suggest new targets.
Sr4Fe2O6Fe2OS2), designed using a building-block approach, are reported. They are fully charge-ordered with Fe2+ and Fe3+ ions in distinct layers showing independent long-range magnetic order. Complex microstructures in some regions suggest new targets.
Mixed-anion compounds are a growth area of solid-state chemistry. Layered structures are often adopted due to anion ordering and desirable properties include tuneable bandgap energies for semiconductors,1 promising thermoelectric properties,2 high-temperature superconductivity3 and complex magnetism.4 A fruitful design method for oxide chalcogenides5 is to use as building blocks individual layers or slabs found in known compounds, and combine them in new ways to target new structures and compositions. We proposed new targets containing the strontium iron oxide slabs from Sr3Fe2O5Cu2S2 or Sr2FeO3CuS6 combined with the iron oxide sulfide layer in Sr2F2Fe2OS2.7 The similar basal lattice parameters of these compounds suggested 1
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 intergrowths might form.
1 intergrowths might form.
These targets, conveniently formulated as Sr3Fe2O5Fe2OS2 (compound 1) and Sr4Fe2O6Fe2OS2 (2), were synthesized in high purity from SrO, Fe2O3, Fe, and S by ceramic solid-state synthesis in alumina crucibles within evacuated sealed fused silica tubes heated at 1 °C per minute to 900 °C (enabling sulfur to react before attaining a high vapour pressure) with a dwell time of 48 hours resulting in black powders stable to ambient air. Full synthetic details are in the SI. Structural models were devised by constructing 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 intergrowths as indicated above, with appropriate interatomic distances, and these accounted well for the powder diffraction patterns of both new products. Both phases contain iron in distinct Fe1 and Fe2 layers and in two different coordination environments: a square pyramidal [FeO5] unit (Fe1—initially assumed to contain Fe3+) and a distorted octahedral [FeO2S4] unit with O atoms arranged trans to one another (Fe2—initially assumed to contain Fe2+). 1 crystallizes in the primitive tetragonal space group P4/mmm with layers of basal-vertex-linked [FeO5] pyramids joined via apical oxide vertices to form double-thickness layers of [FeO5] pyramids (Fig. 1a). 2 (Fig. 1b) crystallizes in the body-centred tetragonal space group I4/mmm with adjacent layers of basal-vertex-linked [FeO5] pyramids separated and shifted by (a + b)/2 in the basal plane, effectively by the insertion of a rock salt layer SrO into the oxide slab present in 1 (Fig. 1c and d). In addition to the different Fe-sites within the phases, there are also different sites occupied by the Sr2+ cations, 12- and 8-coordinate sites in 1 and 9- and 8-coordinate sites in 2 (Fig. S2).
1 intergrowths as indicated above, with appropriate interatomic distances, and these accounted well for the powder diffraction patterns of both new products. Both phases contain iron in distinct Fe1 and Fe2 layers and in two different coordination environments: a square pyramidal [FeO5] unit (Fe1—initially assumed to contain Fe3+) and a distorted octahedral [FeO2S4] unit with O atoms arranged trans to one another (Fe2—initially assumed to contain Fe2+). 1 crystallizes in the primitive tetragonal space group P4/mmm with layers of basal-vertex-linked [FeO5] pyramids joined via apical oxide vertices to form double-thickness layers of [FeO5] pyramids (Fig. 1a). 2 (Fig. 1b) crystallizes in the body-centred tetragonal space group I4/mmm with adjacent layers of basal-vertex-linked [FeO5] pyramids separated and shifted by (a + b)/2 in the basal plane, effectively by the insertion of a rock salt layer SrO into the oxide slab present in 1 (Fig. 1c and d). In addition to the different Fe-sites within the phases, there are also different sites occupied by the Sr2+ cations, 12- and 8-coordinate sites in 1 and 9- and 8-coordinate sites in 2 (Fig. S2).
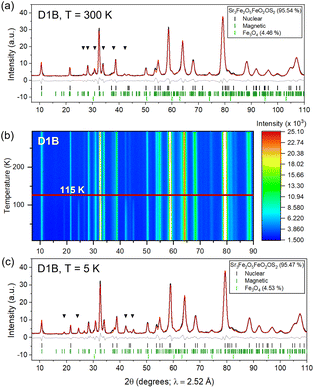
Fig. 2 shows Rietveld refinements against synchrotron X-ray powder diffraction (XRPD) patterns (beamline I11 (Diamond Light Source (DLS))) (Table 1).8 Neutron Powder Diffraction (NPD) used beamlines D1B, D2B, and XtremeD at the Institut Laue Langevin (ILL)9–11 to better locate O atoms (Fig. S1 and Tables S1b and S2). Refinements showed that all sites were fully occupied within the uncertainties of the refinements. Fe–O and Fe–S bond lengths indicated the more oxophilic Fe3+ in the [FeO5] square pyramids and Fe2+ ions in the [FeO2S4] octahedra.
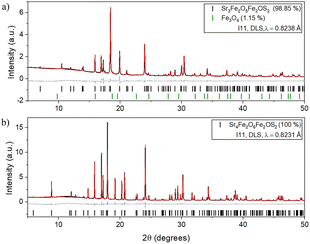 | ||
| Fig. 2 Rietveld refinements (300 K) against XRPD patterns (I11, DLS) of (a) 1: Sr3Fe2O5Fe2OS2 (Rwp = 3.956%) and (b) 2: Sr4Fe2O6Fe2OS2 (Rwp = 4.811%). Observed (black), calculated (red) and difference (grey) curves are shown. See also Table 1 and Table S1. | ||
| 1 – Sr3Fe2O5Fe2OS2 | 2 – Sr4Fe2O6Fe2OS2 | |
|---|---|---|
| Space group | P4/mmm (No. 123) | I4/mmm (No. 139) |
| a (Å) | 3.9615(6) | 3.945892(8) |
| c (Å) | 13.519(9) | 31.9730(1) |
| V (Å3) | 212.18(1) | 497.822(3) |
NPD data from D1B (Fig. 3) were used to deduce the magnetic structure of 1. Low-angle peaks of magnetic origin, present at 300 K, gain little intensity on cooling, indicating magnetic long-range order (LRO) well above room temperature. Further magnetic reflections below 115 K indicate further magnetic ordering (Fig. 3b). At 300 K, the magnetic peaks of 1 can be indexed on a 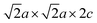 expansion of the nuclear cell and testing of possible antiferromagnetic models revealed that the intensities could be fully accounted for by LRO of only the Fe3+ ions on the Fe1 sites. Within each [Sr3Fe2O5]2+ slab, Fe3+ moments order antiferromagnetically via Fe–O–Fe 163.7° super-exchange interactions involving the bases of the square pyramids and are oriented within the basal plane. The Fe1 moments are also coupled antiferromagnetically via 180° Fe–O–Fe super-exchange through shared apical oxides. This ordering is similar to that in the related Ae3Fe2O5Cu2Ch2 (Ae = Ba, Sr, Ca; Ch = S, Se).12,13 The long-range ordered moment saturates at 4.07(5)µB per Fe1 in 1 below 160 K, diminished from the expected 5µB for a high-spin d5 Fe3+ cation due to covalency.
expansion of the nuclear cell and testing of possible antiferromagnetic models revealed that the intensities could be fully accounted for by LRO of only the Fe3+ ions on the Fe1 sites. Within each [Sr3Fe2O5]2+ slab, Fe3+ moments order antiferromagnetically via Fe–O–Fe 163.7° super-exchange interactions involving the bases of the square pyramids and are oriented within the basal plane. The Fe1 moments are also coupled antiferromagnetically via 180° Fe–O–Fe super-exchange through shared apical oxides. This ordering is similar to that in the related Ae3Fe2O5Cu2Ch2 (Ae = Ba, Sr, Ca; Ch = S, Se).12,13 The long-range ordered moment saturates at 4.07(5)µB per Fe1 in 1 below 160 K, diminished from the expected 5µB for a high-spin d5 Fe3+ cation due to covalency.
NPD may, in principle, not always provide a unique magnetic model.14 In 1 a single-phase magnetic model at 300 K has Fe1 moments collinear within a slab, but rotated in adjacent slabs by 73° relative to one other (Fig. 4b); this rotation angle increases to 85° at 5 K. A two-phase model indistinguishable by powder diffraction is discussed in the SI. The Fe2+ ions on the Fe2 sites in 1 show magnetic LRO below 115 K. New magnetic peaks saturated at 5 K were indexed on a 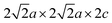 expansion of the nuclear cell. The ordering scheme (Fig. 4c) was deduced by comparison with related compounds and the magnitude of the long-range ordered Fe2+ moments (3.33(7)µB) are similar in phases containing exclusively these magnetic layers: La2O2Fe2OS2 (3.30(4)µB),15 Sr2F2Fe2OS2 (3.3(1)µB),16 and La2O2Fe2OSe2 (3.50(5)µB).17
expansion of the nuclear cell. The ordering scheme (Fig. 4c) was deduced by comparison with related compounds and the magnitude of the long-range ordered Fe2+ moments (3.33(7)µB) are similar in phases containing exclusively these magnetic layers: La2O2Fe2OS2 (3.30(4)µB),15 Sr2F2Fe2OS2 (3.3(1)µB),16 and La2O2Fe2OSe2 (3.50(5)µB).17
The magnetic structure of 2 was determined by NPD (300 K on GEM (ISIS facility) (Fig. S12) and on cooling on XtremeD (Fig. S13) and D2B at ILL (Fig. S1c, S1d and Table S3)). Like in 1, only Fe3+ cations (Fe1) are ordered at room temperature with magnetic peaks indexed on a 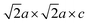 supercell. Antiferromagnetic ordering of Fe1 moments within the sheets of basal-vertex-sharing [FeO5] square pyramids was found to be similar to that in 1, but separation of the sheets by insertion of an SrO layer into the oxide slabs and the loss of the strong Fe–Oaxial-Fe superexchange means the ordered moment (2.14(2)µB) is unsaturated at 300 K, but it saturates at 3.7(2) per Fe3+ ion (comparable to in 1) at 5 K. The Fe1 moments in 2 are best modelled as tilted from the c axis by 56(1)° at 5 K. As in 1, other related models are indistinguishable in a powder measurement (see SI). Note that high-spin d5 moments, as on the Fe1 sites in 1 and 2, have very weak directional preference.18 The Fe2 site moments on Fe2+ cations in the Fe2O sheets of 2 order below 150 K in a similar pattern to that in 1 on a
supercell. Antiferromagnetic ordering of Fe1 moments within the sheets of basal-vertex-sharing [FeO5] square pyramids was found to be similar to that in 1, but separation of the sheets by insertion of an SrO layer into the oxide slabs and the loss of the strong Fe–Oaxial-Fe superexchange means the ordered moment (2.14(2)µB) is unsaturated at 300 K, but it saturates at 3.7(2) per Fe3+ ion (comparable to in 1) at 5 K. The Fe1 moments in 2 are best modelled as tilted from the c axis by 56(1)° at 5 K. As in 1, other related models are indistinguishable in a powder measurement (see SI). Note that high-spin d5 moments, as on the Fe1 sites in 1 and 2, have very weak directional preference.18 The Fe2 site moments on Fe2+ cations in the Fe2O sheets of 2 order below 150 K in a similar pattern to that in 1 on a 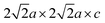 expansion of the nuclear cell (see Fig. S14). The Fe2+ moment refines to 2.1(1)µB at 5 K, somewhat lower than in 1. In 2, there is a mismatch in the Rietveld refinement at 5 K for an asymmetric magnetic peak around 40.6° in the XtremeD data (red triangle in Fig. 5b) arising from the Fe2 magnetic ordering. This also appears in related body-centred compounds containing [Fe2OS2] layers (e.g. La2O2Fe2OS2)15 and has been attributed to intrinsic stacking faults in the magnetic structure as explained in ref. 15.
expansion of the nuclear cell (see Fig. S14). The Fe2+ moment refines to 2.1(1)µB at 5 K, somewhat lower than in 1. In 2, there is a mismatch in the Rietveld refinement at 5 K for an asymmetric magnetic peak around 40.6° in the XtremeD data (red triangle in Fig. 5b) arising from the Fe2 magnetic ordering. This also appears in related body-centred compounds containing [Fe2OS2] layers (e.g. La2O2Fe2OS2)15 and has been attributed to intrinsic stacking faults in the magnetic structure as explained in ref. 15.
57Fe Mössbauer spectroscopy measurements (see SI for details) on Sr3Fe2O5Fe2OS2 (1) are consistent with the PND results, showing independent magnetic ordering on the Fe1 and Fe2 sites. At room temperature there is a sextet due to the ordered Fe1 moments (isomer shift (δ) of 0.34 mm s−1 suggesting Fe3+) and a doublet from the Fe2 moments (δ = 0.87 mm s−1 suggesting Fe2+)19 plus two minority doublet components of uncertain origin (but see below). At 80 K (Fig. 6b), the ordered Fe2 moments give a highly asymmetric sextet quantitatively similar to those in Ba2F2Fe2OSe2, Sr2F2Fe2OS27 and Nd2O2Fe2OSe219 with related [Fe2OS2] layers, and one of the minor doublets (δ = 0.51 mm s−1 suggesting Fe3+) remains (6%).
 | ||
| Fig. 6 57Fe Mössbauer spectra for tetragonal Sr3Fe2O5Fe2OS2 (1) at room temperature (RT) (a) and 80 K (b). Fe1 (Fe3+) (Brown) is magnetically ordered a both temperatures, producing sextets from the magnetic hyperfine interaction and Fe2 (Fe2+) (Blue) undergoes long-range magnetic ordering below 115 K (according to NPD), and in this case the sextet is highly asymmetric as found in compounds with related motifs.7,19 The origin of the lower population doublet (Green) required to fit the data at both temperatures, and the second minority orange doublet at RT is uncertain. Fitting parameters are in Table S4. | ||
Some samples of 1 were found to show broad additional features in the powder diffraction patterns (Fig. S3(a)) and others were found, using synchrotron X-ray powder data, to undergo subtle distortions which can be modelled by reducing the symmetry to orthorhombic below 500 K (Fig. S3(b) and S4), and this was sometimes induced by lengthy annealing. Preliminary transmission electron microscopy (TEM) measurements (Fig. 7) on a sample of 1 which was tetragonal at all temperatures and did not show any broad features in the powder diffraction patterns nevertheless show the presence of diverse stacking faults which may account for the minority Mössbauer components, and the occurrence of ubiquitous Fe3O4 impurities. These faults include translational faults along the stacking direction (Fig. 7b and d) and thicker Fe1-layer-related blocks (Fig. 7e), and may suggest new targets and options for compositional and structural tuning. Variation in such features between samples may be responsible for the broad peaks in the diffractograms of some samples and the reduction in symmetry in others (see Fig. S5 for images of a sample of 1 that is orthorhombic, or lower, by PXRD at RT) and requires further detailed investigation. Such stacking faults have been noted in related compounds such as Sr2FeO3CuSe.20 From preliminary TEM measurements (Fig. S7), compound 2 seems less prone to such defects or impurities.
In summary, Sr3Fe2O5Fe2OS2 (1) and Sr4Fe2O6Fe2OS2 (2) have been constructed using building blocks known from existing compounds. Fe3+ cations in [FeO5] square-based pyramids are antiferromagnetically ordered at room temperature and Fe2+ cations in trans-[FeO2S4] octahedra order antiferromagnetically below 115 K and 150 K in 1 and 2 respectively. Single crystal neutron diffraction experiments are required to fully resolve intrinsic ambiguity in the magnetic structures. We have previously identified21 new targets from analysis of high-resolution HAADF-STEM images, and in the case of 1, analysis of such images (Fig. 7 and Fig. S5) reveals the occurrence of stacking fault regions as well as perfectly ordered regions showing that these compounds have the capacity to adopt more complex structures. Future work will involve taking inspiration from these images to target new compositions as well as tuning the magnetic interactions and electron counts in 1 and 2 by iso- and aliovalent substitution and topochemical transformations.
ANG: synthesis/analysis for 1, ARL: synthesis/analysis for 2. SR and JH: Electron diffraction and TEM imaging/analysis. SS and IPO: NPD data collection at ILL. MG and MLN: Mössbauer data collection/analysis. SJC: materials and initial concepts. ANG drafted the manuscript with input from the other authors.
Conflicts of interest
There are no conflicts to declare.Data availability
PND data are available at doi:https://doi.org/10.5291/ILL-DATA.5-31-2917 and https://doi.org/10.5291/ILL-DATA.5-31-3008. Other data supporting this article have been included as part of the supplementary information (SI). Supplementary information: additional synthetic and analysis details. https://doi.org/10.1039/d6cc00150e.Acknowledgements
We thank the UK EPSRC for funding (EP/T027991/1, EP/R042594/1). For beam time we thank DLS (CY32893), ILL (5-31-2917 & 5-31-3008), and ISIS (XB2390151). A. N. G. was supported by the John and Daria Barry Scholarship. S. R. and J. H. received funding from the University of Antwerp through BOF TOP 38689.References
- K. Ueda and H. Hosono, J. Appl. Phys., 2002, 91, 4768–4770 CrossRef CAS.
- S. D. N. Luu and P. Vaqueiro, J. Mater., 2016, 2, 131–140 Search PubMed.
- Y. Kamihara, T. Watanabe, M. Hirano and H. Hosono, J. Am. Chem. Soc., 2008, 130, 3296–3297 CrossRef CAS PubMed.
- H. Kageyama, K. Hayashi, K. Maeda, J. P. Attfield, Z. Hiroi, J. M. Rondinelli and K. R. Poeppelmeier, Nat. Commun., 2018, 9, 772 CrossRef PubMed.
- L. Cario, H. Kabbour and A. Meerschaut, Chem. Mater., 2005, 17, 234–236 CrossRef CAS.
- W. J. Zhu and P. H. Hor, J. Solid State Chem., 1997, 134, 128–131 CrossRef CAS.
- H. Kabbour, E. Janod, B. Corraze, M. Danot, C. Lee, M.-H. Whangbo and L. Cario, J. Am. Chem. Soc., 2008, 130, 8261–8270 CrossRef CAS PubMed.
- S. P. Thompson, J. E. Parker, J. Potter, T. P. Hill, A. Birt, T. M. Cobb, F. Yuan and C. C. Tang, Rev. Sci. Instrum., 2009, 80, 879 CrossRef PubMed.
- I. Puente-Orench, J. F. Clergeau, S. Martínez, M. Olmos, O. Fabelo and J. Campo, J. Phys.: Conf. Ser., 2014, 549, 012003 CrossRef.
- E. Suard and A. Hewat, Neutron News, 2001, 12, 30–33 CrossRef.
- J. A. Rodríguez-Velamazán, J. Campo, J. Rodríguez-Carvajal and P. Noguera, J. Phys.: Conf. Ser., 2011, 325, 012010 CrossRef.
- M. Lü, O. Mentré, E. E. Gordon, M. H. Whangbo, A. Wattiaux, M. Duttine, N. Tiercelin and H. Kabbour, J. Magn. Magn. Mater., 2017, 444, 147–153 CrossRef.
- R. D. Smyth, J. A. D. Wilson, P. Manuel and S. J. Clarke, J. Solid State Chem., 2022, 307, 122841 CrossRef CAS.
- G. Shirane, Acta Crystallogr., 1959, 12, 282–285 CrossRef CAS.
- R. K. Oogarah, E. Suard and E. E. McCabe, J. Magn. Magn. Mater., 2018, 446, 101–107 CrossRef CAS.
- L. L. Zhao, S. Wu, J. K. Wang, J. P. Hodges, C. Broholm and E. Morosan, Phys. Rev. B: Condens. Matter Mater. Phys., 2013, 87, 020406(R) CrossRef.
- E. E. McCabe, C. Stock, E. E. Rodrigues, A. S. Wills, J. W. Taylor and J. S. O. Evans, Phys. Rev. B: Condens. Matter Mater. Phys., 2014, 89, 100402(R) CrossRef.
- M. H. Whangbo, E. E. Gordon, H. Xiang, H. J. Koo and C. Lee, Acc. Chem. Res., 2015, 48, 3080–3087 CrossRef CAS PubMed.
- Y. Fuwa, M. Wakeshima and Y. Hinatsu, J. Phys.: Condens. Matter, 2010, 22, 346003 CrossRef PubMed.
- D. Berthebaud, O. I. Levedev, D. Pelloquin and A. Maignan, Solid State Sci., 2014, 36, 94–100 CrossRef CAS.
- B. C. Sheath, X. Xu, P. Manuel, J. Hadermann, M. Batuk, J. O’Sullivan, R. S. Bonilla and S. J. Clarke, Inorg. Chem., 2022, 61, 12373–12385 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |