Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Quantifying static capacity losses in solid-state battery composites via coulometric titration comparison
Kilian
Vettori†
 *a,
Maximilian
Kissel†
*a,
Maximilian
Kissel†
 a,
Daniel
Wagner
a,
Steffen
Schröder
b and
Jürgen
Janek
a,
Daniel
Wagner
a,
Steffen
Schröder
b and
Jürgen
Janek
 *a
*a
aInstitute of Physical Chemistry & Center for Materials Research (ZfM/LaMa), Justus-Liebig-University Giessen, Heinrich-Buff-Ring 17, 35392, Giessen, Germany. E-mail: kilian.vettori@uni-giessen.de; juergen.janek@phys.chemie.uni-giessen.de
bInstitute of Experimental Physics I & Center for Materials Research (ZfM/LaMa), Justus Liebig-University Giessen, Heinrich-Buff-Ring 16, 35392, Giessen, Germany
First published on 23rd February 2026
Abstract
An electrochemical method is discussed to quantify in situ the static CAM utilization, i.e., the fraction of electrochemically active mass in solid-state battery composite cathodes. For that, coulometric titration curves of the same active material in cells with solid and liquid electrolytes are compared. The results are contrasted with an ex situ method based on X-ray diffraction.
Solid-state batteries (SSBs) have attracted substantial attention in recent years due to their potential to deliver higher energy and power density, along with expected improved safety compared to conventional lithium-ion batteries (LIBs) that use liquid electrolytes (LE).1,2 Despite the potential benefits, several challenges must be overcome before SSBs can be widely commercialized.3 This includes issues such as interfacial instability between the electrodes and the solid electrolyte (SE), as well as chemo-mechanical degradation, which currently limits performance and long-term stability.4 While the goal for the negative side is a reservoir-free lithium metal electrode, the energy density of the cell is limited by the positive electrode, which is designed as a composite of cathode active material (CAM), SE and conductive additives.5
As a figure of merit, CAM-specific capacities and capacity losses are often calculated with respect to the mass of CAM in the cathode, mCAM,tot, and used to compare the performance of different composites or cells.6 In a recent study, we discussed the importance of differentiating between static and kinetic capacity losses in SSB composite cathodes and revealed that different composite mixing can be a main reason for variations in mass-specific capacities with respect to mCAM,tot.7 Thereby, a central challenge of SSB composites is to properly connect all CAM particles electronically. If this is not achieved, lower mass-specific capacities are observed in SSBs compared to LIBs. We recommend to consider these as static capacity losses.7,8 The accurate evaluation of mass-specific capacities accounting for static capacity loss of SSBs is critical to avoid data misinterpretation at an early stage of research and for quality control during scale-up.
In the present work, we discuss a reliable and simple electrochemical method to estimate in situ the actual electrochemically active mass, mCAM,act, for a given SSB (half) cell with respect to a reference LIB measurement. The methodology is further called coulometric titration comparison (CTC) and has been successfully employed in several of our previous publications.7–11 CTC shall be highlighted as well as critically discussed here in depth to be more easily implemented by other scientists testing active materials in different electrolyte systems. First, we sketch the differences between LIB and SSB cathode microstructures and their consequences for the capacities during cycling. Then, we illustrate the working principle and the experimental approach of CTC and discuss underlying assumptions as well as natural limitations of the method, including the numerical uncertainty of the method. Finally, we show the results of CTC from an exemplary measurement with LiNi0.82Mn0.07Co0.11O2 (NCM82) in an SSB composite cathode with Li6PS5Cl as the SE, and in a porous cathode in organic LE. The determined active mass mCAM,act and the resulting CAM utilization ΘCAM,SSB are compared with Rietveld-refined ex situ XRD measurements on composite cathodes, revealing the fraction of electronically unconnected, inactive CAM.
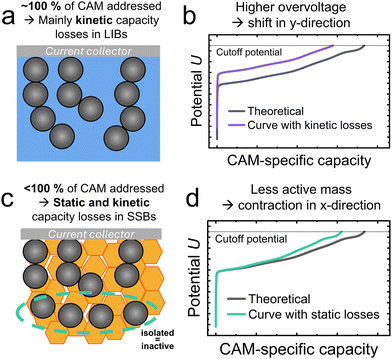
Let us first picture the difference in connectivity of CAM particles in the cathodes of LIBs and SSBs (Fig. 1). In conventional LIBs a porous, electron-conducting network of CAM particles, binder, and carbon additives is infiltrated by LE.12,13 In this porous electrode design, it can be assumed that nearly all CAM particles are ionically and electronically connected and thus electrochemically active (Fig. 1a). For discussion of this assumption the reader is referred to SI Section S2.1. Differences in measurable specific capacity q during the first cycles can then be mainly attributed to kinetic effects, which increase the overpotential (Fig. 1b), so that the cutoff potential is reached earlier.
The situation is different in composite cathodes in SSB cells (Fig. 1c). In this case, the CAM particles need to be mixed with a SE, which comprises particles with a specific size distribution, morphology, and mechanical properties that should be adapted to the properties of the CAM.14,15 As shown in previous studies also by our group,7,16 it can happen that a substantial amount of CAM particles is already from the beginning not incorporated in the electronic network, e.g. due to poor mixing. In both, LIB and SSB, carbon additives are used to electronically connect the CAM particles. For LIBs more porous carbons are used, whereas SSBs often employ carbon nanofibers (CNF) for long range connection.
The unconnected CAM particles do not take part in charging/discharging, and must be considered as electrochemically inactive, lowering mCAM,act. Thus, in addition to kinetic capacity losses, substantial static capacity losses7 need to be considered, which are visible by a horizontal compression of the charging curve (Fig. 1d). To accurately evaluate the specific capacity and avoid data misinterpretation, precise knowledge of the actual electrochemically active mass mCAM,act in SSB composite cathodes is required.7
Here, CTC represents a straightforward approach to quantify mCAM,act and ΘCAM,SSB. The corresponding procedure is sketched in Fig. 2 and will be elaborated in the following for NCM82. Experimental details are given in the SI Section S1.
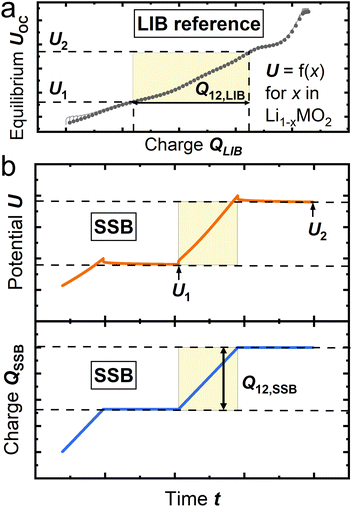 | ||
| Fig. 2 Working principle of the CTC. (a) Coulometric titration data in LIB serving as reference, (b) potential and accumulated charge of SSB during charge and relaxation steps. | ||
First, a LIB cell is built as reference, and a coulometric titration curve, e.g. based on the galvanostatic intermittent titration technique (GITT),17 is recorded (Fig. 2a). The open circuit potential (OCP) follows a characteristic function vs. the lithium content x in Li1−xNCMO2, which can also be expressed as state-of-charge (SoC) or mass specific capacity q.18,19 These thermodynamic data serve as the reference curve. Coulometric titration measurements contain inherent errors which are discussed below and estimated in detail in SI Section S2.
To calculate mCAM,act in the SSB composite cathode, two equilibrated potentials U1 and U2, and the charge Q12,SSB that was accumulated to reach U2 need to be known (Fig. 2b). Q12,SSB is then compared to the corresponding Q12,LIB from LIB data.
Together with mCAM,LIB, i.e., the total mass of CAM in the LIB cathode, the actual electrochemically active mass of the SSB cathode can be calculated with eqn (1):
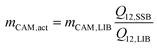 | (1) |
If less CAM is statically active, after reaching U2 (or the corresponding SoC), the SSB has accumulated less charge Q12,SSB compared to the LIB. The fraction of active CAM in the SSB, i.e. the CAM utilization ΘCAM,SSB, can then be calculated viaeqn (2):
| θCAM,SSB = mCAM,act/mCAM,tot | (2) |
The following fundamental assumptions need to be considered when calculating the active mass via CTC:
First, it is assumed that in the LIB reference cell, 100% of the CAM is electrochemically active and thus all particles contribute to the coulometric titration curve (See SI Section S2.1). Furthermore, CAM utilization is understood as constant during the experiment.
Second, the potentials obtained for LIB and SSB need to be in equilibrium and follow the same titration curve of the pristine CAM. OCP values need to be in a region, where the titration curve is a well-defined and monotonically increasing function vs. the lithium content x.
Third, the capacity/charge measured is purely from de-/lithiation of the CAM, meaning a faradaic efficiency FE of 1 without contributions from other reactions, as e.g., electrolyte decomposition.
Furthermore, to ensure a meaningful result for mCAM,act, the parameters entering eqn (1) have to be carefully extracted, leading to the following experimental considerations:
The mass of CAM, which is used during the preparation of electrodes (LIB and SSB) is essential for a reliable application of CTC. This means that both mCAM,tot and mCAM,LIB enter the CAM utilization ΘCAM,SSB with the weighing error. Furthermore, it is known that the CAM utilization in SSBs can be dynamic within the SoC, especially due to interparticle contact issues after volume contraction of NCM-type layered oxides above ≈4.2 V.20 CTC is recommended at potentials where negligible chemo-mechanical changes in utilization occur in SSBs.21,22
The potentials need to be equilibrated. Thus, it is recommended to measure at potentials/SoCs where electrode kinetics is fast. For NCM, this is again at medium potentials/SoCs,23–25 where diffusion of lithium is fast, allowing shorter relaxation times and avoiding concentration gradients.26,27
Obviously, SSB and LIB potentials need to be compared against the same reference potential. Since SSBs often employ other counter electrodes, their potentials need to be known and stable, as the In-InLi anode, with +620 mV vs. Li+/Li (SI Section S3.1).28
Another consideration regards the OCP fading due to parasitic reactions, e.g. electrolyte decomposition re-lithiating the NCM. To quantify this, dU/dt at the end of relaxation, often called “OCV fading” or “self-discharge”, at different potentials can be extracted and should be minimized experimentally.29 The SI Section S2.3.1.2 shows such evaluation for the LIB reference cell. In general, it can be useful to use low loadings to reduce lithium concentration gradients in the cathode, coming with the cost of less precision for mCAM,LIB. SI Section S2 provides data and recommendations on optimal experimental design. Potentials should be unique for a specific charge, meaning that two-phase regions/potential plateaus should be avoided, posing challenges for two-phase materials like LiFePO4 (SI Section S2.2).30 Finally the potential curve should be as close as possible to the pristine CAM, since extended processing is known to change the titration curve.31
The charge: We further recommend applying formation cycles to the cells to obtain stable titration curves for the active material, where the faradaic Efficiency (FE) of CAM de-/lithiation is close to 1 and no other processes like CEI formation are expected. Of course, cycling induced changes should be minimized. To identify voltage regions dominated by CAM de-/lithiation, we suggest comparing titration curves in both electrolytes.
We applied the CTC to two SSB cathode composite batches, which are expected to exhibit varying degrees of CAM utilization due to different electrode compositions.16 One batch with a CAM:SE:CNF ratio of 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30
30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (further called “good SSB” and shown in blue) and one with CAM:SE of 60
1 (further called “good SSB” and shown in blue) and one with CAM:SE of 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 (“bad SSB”, shown in orange).
40 (“bad SSB”, shown in orange).
We obtained multiple relaxed potentials for both SSBs by repeating a simple charge and relaxation protocol, shown by highlighted points in Fig. 3a. The SSB charge procedures (orange, blue data points) deviate from the coulometric titration data of the LIB reference (grey). The inlet showcases how a CAM utilization is calculated for two OCP values of the “bad” SSB. This procedure (eqn (2)) was applied for all neighboring OCPs and both cells as shown in SI Section S3.2 via a Python script.32 From the obtained CAM utilizations we calculated a selected average (SI section S3.2), resulting in ΘCAM,SSB of 76.4 ± 5.8% and 101.2 ± 2.6% for bad and good SSB, respectively.
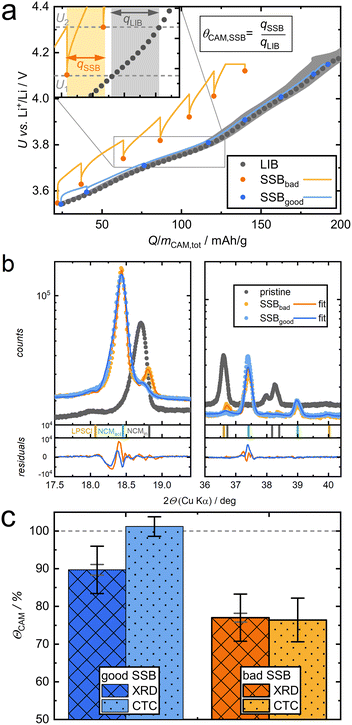 | ||
| Fig. 3 Experimental results on good and bad SSB cathode composites. (a) Electrochemical data during application of CTC with LIB reference. The inlet shows an exemplary calculation of ΘCAM,SSB for two OCPs of the bad SSB. (b) XRD spectra and Rietveld refinements with indicated reflex positions. (c) Resulting CAM utilization (eqn (2)) from XRD and CTC method. Grey error bars for XRD are from Rietveld refinement and black from an estimated error of 5 wt% per phase. Error bars for CTC are from application to multiple potential steps (SI Section S3.2). | ||
Additionally, we performed XRD on the “good” and “bad” SSB composites after extracting them in the U2 state after holding U = 4.15 V for 12 h. While the major part of the signal corresponds to active, delithiated NCM (NCMact, green indicator) in H2 phase, distinct reflexes of inactive NCM (NCMinact, grey indicator) in H1 phase are observed for both SSBs (Fig. 3b).33 For comparison, also a pristine composite, containing only NCMinact, is shown in grey. Full diffractograms are given in SI section S4. Shifting of reflexes due to changes in lattice parameters is well understood for NCM and can be compared for the used NCM82 with an in situ XRD in a liquid cell reference (SI Section S4). Following the approach by Bartsch et al.34 and Strauss et al.35 the XRD data were refined by the Rietveld method with two NCM phases and the SE phase, allowing an estimation of ΘCAM,SSB. The results of both methods, CTC and XRD, are compared in Fig. 3c. The XRD data validate the CTC method, delivering 89.7% CAM utilization for the good SSB in contrast to only 77.0% for the bad SSB. In SI Section S5 the charge curves of good and bad SSB are compared when being normalized to mCAM,act instead of mCAM,tot.
We observe a systematically lower CAM utilization when quantified by XRD for SSBs that perform very well (as measured by CTC). This could be due to a LIB reference that suffers from similar capacity losses as the SSBs, thus overestimating ΘCAM,SSB. Another reason could be LPSCl contributing to capacity while the actual mCAM,act is lower. Most plausible is that Rietveld refinement of a phase with a minor volume fraction and correspondingly small signal in XRD data inherently leads to error-prone results, e.g. overinterpretation of device-related shoulders of reflexes.
We believe CTC offers multiple advantages over the approach based on XRD while allowing comparable accuracy. First, it requires no additional equipment and enables an in situ determination of mCAM,act instead of ex situ and post mortem measurements, which is typically done for XRD on SSBs. Furthermore, XRD data refinement as quantification of small mass fractions is inherently inaccurate.
In contrast, coulometric titration comparison (CTC) allows reliable in situ access to mCAM,act. The procedure is simple to implement and allows an estimation of errors, providing a useful tool. We have applied it successfully to SSB composite cathodes and verified the results with an ex situ XRD method, but the procedure can be used for any suitable active material under the assumptions discussed in this work. Furthermore, best-practice is described regarding experimental design and the evaluation via Python script facilitates the implementation of CTC. In general, coulometric titration has proven to be highly useful within our working group as multiple publications employing CTC show.7–11 Applied to SSBs, the possibility to uncover the presence of electrochemically inactive electrode material, may help to develop and optimize advanced cathode composites for next-generation applications.
Author contributions
Kilian Vettori: investigation, writing – original draft, software. Maximilian Kissel: investigation, writing – original draft. Daniel Wagner: formal analysis (XRD Rietveld refinements). Steffen Schröder: writing – review & editing. Jürgen Janek: supervision, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
Coulometric titration data and measurements and the Python code used for evaluation are available in the CTC-data-analysis repository at https://doi.org/10.5281/zenodo.17967963.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5cc07213a.
Acknowledgements
This work has been funded by the Federal Ministry of Research, Technology and Space under the project HIPOBAT (FKZ: 03XP0611E) and by Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) through the Priority Program 2289 (Project 462470125).References
- A. M. Bates, Y. Preger, L. Torres-Castro, K. L. Harrison, S. J. Harris and J. Hewson, Joule, 2022, 6, 742–755 CrossRef CAS.
- J. Janek and W. G. Zeier, Nat. Energy, 2016, 1, 16141 Search PubMed.
- J. Janek and W. G. Zeier, Nat. Energy, 2023, 8, 230–240 CrossRef.
- A. Banerjee, X. Wang, C. Fang, E. A. Wu and Y. S. Meng, Chem. Rev., 2020, 120, 6878–6933 CrossRef CAS PubMed.
- S. Randau, D. A. Weber, O. Kötz, R. Koerver, P. Braun, A. Weber, E. Ivers-Tiffée, T. Adermann, J. Kulisch, W. G. Zeier, F. H. Richter and J. Janek, Nat. Energy, 2020, 5, 259–270 Search PubMed.
- S. Puls, E. Nazmutdinova, F. Kalyk, H. M. Woolley, J. F. Thomsen, Z. Cheng, A. Fauchier-Magnan, A. Gautam, M. Gockeln, S.-Y. Ham, M. T. Hasan, M.-G. Jeong, D. Hiraoka, J. S. Kim, T. Kutsch, B. Lelotte, P. Minnmann, V. Miß, K. Motohashi, D. L. Nelson, F. Ooms, F. Piccolo, C. Plank, M. Rosner, S. E. Sandoval, E. Schlautmann, R. Schuster, D. Spencer-Jolly, Y. Sun, B. S. Vishnugopi, R. Zhang, H. Zheng, P. Adelhelm, T. Brezesinski, P. G. Bruce, M. Danzer, M. El Kazzi, H. Gasteiger, K. B. Hatzell, A. Hayashi, F. Hippauf, J. Janek, Y. S. Jung, M. T. McDowell, Y. S. Meng, P. P. Mukherjee, S. Ohno, B. Roling, A. Sakuda, J. Schwenzel, X. Sun, C. Villevieille, M. Wagemaker, W. G. Zeier and N. M. Vargas-Barbosa, Nat. Energy, 2024, 9, 1310–1320 CrossRef CAS.
- M. Kissel, M. Schosland, J. Töws, D. Kalita, Y. Schneider, J. Kessler-Kühn, S. Schröder, J. Schubert, F. Frankenberg, A. Kwade, A. Bielefeld, F. H. Richter and J. Janek, Adv. Energy Mater., 2025, 15, 2405405 CrossRef CAS.
- G. Conforto, R. Ruess, D. Schröder, E. Trevisanello, R. Fantin, F. H. Richter and J. Janek, J. Electrochem. Soc., 2021, 168, 70546 CrossRef CAS.
- P. Minnmann, J. Schubert, S. Kremer, R. Rekers, S. Burkhardt, R. Ruess, A. Bielefeld, F. H. Richter and J. Janek, J. Electrochem. Soc., 2024, 171, 60514 Search PubMed.
- B.-X. Shi, Y. Yusim, S. Sen, T. Demuth, R. Ruess, K. Volz, A. Henss and F. H. Richter, Adv. Energy Mater., 2023, 13, 2300310 CrossRef CAS.
- B.-X. Shi, F. Weber, Y. Yusim, T. Demuth, K. Vettori, A. Münchinger, G. Titvinidze, K. Volz, A. Henss, R. Berger and F. H. Richter, J. Mater. Chem. A, 2025, 13, 2600–2614 RSC.
- A. Kwade, W. Haselrieder, R. Leithoff, A. Modlinger, F. Dietrich and K. Droeder, Nat. Energy, 2018, 3, 290–300 CrossRef.
- A. Shodiev, F. M. Zanotto, J. Yu, M. Chouchane, J. Li and A. A. Franco, Energy Storage Mater., 2022, 49, 268–277 CrossRef.
- P. Minnmann, F. Strauss, A. Bielefeld, R. Ruess, P. Adelhelm, S. Burkhardt, S. L. Dreyer, E. Trevisanello, H. Ehrenberg, T. Brezesinski, F. H. Richter and J. Janek, Adv. Energy Mater., 2022, 12, 2201425 CrossRef CAS.
- A. Bielefeld, D. A. Weber and J. Janek, ACS Appl. Mater. Interfaces, 2020, 12, 12821–12833 CrossRef CAS PubMed.
- A. Bielefeld, D. A. Weber and J. Janek, J. Phys. Chem. C, 2019, 123, 1626–1634 CrossRef CAS.
- W. Weppner and R. A. Huggins, J. Electrochem. Soc., 1977, 124, 1569–1578 CrossRef CAS.
- M. Levi and D. Aurbach, Electrochim. Acta, 1999, 45, 167–185 CrossRef CAS.
- M. D. Radin, S. Hy, M. Sina, C. Fang, H. Liu, J. Vinckeviciute, M. Zhang, M. S. Whittingham, Y. S. Meng and A. van der Ven, Adv. Energy Mater., 2017, 7, 1602888 CrossRef.
- R. Koerver, W. Zhang, L. de Biasi, S. Schweidler, A. O. Kondrakov, S. Kolling, T. Brezesinski, P. Hartmann, W. G. Zeier and J. Janek, Energy Environ. Sci., 2018, 11, 2142–2158 RSC.
- X. Liu, B. Zheng, J. Zhao, W. Zhao, Z. Liang, Y. Su, C. Xie, K. Zhou, Y. Xiang, J. Zhu, H. Wang, G. Zhong, Z. Gong, J. Huang and Y. Yang, Adv. Energy Mater., 2021, 11, 2003583 CrossRef CAS.
- S. H. Park, K. G. Naik, B. S. Vishnugopi, X. Xiao, M. Drakopoulos, N. T. Vo, Z. Zhong, P. P. Mukherjee and K. B. Hatzell, ACS Nano, 2025, 22262–22269 CrossRef.
- A. Liu, N. Phattharasupakun, M. M. E. Cormier, E. Zsoldos, N. Zhang, E. Lyle, P. Arab, M. Sawangphruk and J. R. Dahn, J. Electrochem. Soc., 2021, 168, 70503 CrossRef CAS.
- R. Weber, A. J. Louli, K. P. Plucknett and J. R. Dahn, J. Electrochem. Soc., 2019, 166, A1779–A1784 CrossRef CAS.
- R. Morasch, H. A. Gasteiger and B. Suthar, J. Electrochem. Soc., 2023, 170, 80522 Search PubMed.
- M. D. Levi, K. Gamolsky, D. Aurbach, U. Heider and R. Oesten, J. Electroanal. Chem., 1999, 477, 32–40 CrossRef CAS.
- K. Vettori, S. Schröder, L. Ahrens, R. Wilhelm, S. Kremer, J. K. Eckhardt, T. Brezesinski, A. Kondrakov, J. Mayer, A. Henss and J. Janek, Adv. Energy Mater., 2025, 2502148 CrossRef CAS.
- A. L. Santhosha, L. Medenbach, J. R. Buchheim and P. Adelhelm, Batteries Supercaps, 2019, 2, 524–529 CrossRef CAS.
- T. Roth, L. Streck, A. Graule, P. Niehoff and A. Jossen, J. Electrochem. Soc., 2023, 170, 20502 Search PubMed.
- D. Di Lecce and J. Hassoun, J. Phys. Chem. C, 2015, 119, 20855–20863 CrossRef CAS.
- L. Zheng, C. Wei, M. D. L. Garayt, J. MacInnis and M. N. Obrovac, J. Electrochem. Soc., 2019, 166, A2924–A2927 CrossRef CAS.
- K. Vettori, CTC-data-analysis, 2026, Zenodo10.5281/zenodo.17967963.
- C. Ghanty, B. Markovsky, E. M. Erickson, M. Talianker, O. Haik, Y. Tal-Yossef, A. Mor, D. Aurbach, J. Lampert, A. Volkov, J.-Y. Shin, A. Garsuch, F. F. Chesneau and C. Erk, ChemElectroChem, 2015, 2, 1479–1486 CrossRef CAS.
- T. Bartsch, A.-Y. Kim, F. Strauss, L. de Biasi, J. H. Teo, J. Janek, P. Hartmann and T. Brezesinski, Chem. Commun., 2019, 55, 11223–11226 RSC.
- F. Strauss, T. Bartsch, L. de Biasi, A.-Y. Kim, J. Janek, P. Hartmann and T. Brezesinski, ACS Energy Lett., 2018, 3, 992–996 CrossRef CAS.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |