Open Access Article
Open Access ArticleOne spiro to shift them all: tuning fluorescent organic nanoparticles emission via steric design
Eléonore
Kurek†
 a,
Ophélie
Dal Pra†
a,
Ophélie
Dal Pra†
 a,
Aliocha
Skrzypczak‡
a,
Aliocha
Skrzypczak‡
 a,
Stéphane
Massip
a,
Stéphane
Massip
 b,
Jonathan
Daniel
b,
Jonathan
Daniel
 a,
Mireille
Blanchard-Desce
a,
Mireille
Blanchard-Desce
 *a and
Chloé
Grazon
*a and
Chloé
Grazon
 *a
*a
aUniv. Bordeaux, CNRS, Bordeaux INP, ISM, UMR 5255, F-33400 Talence, France. E-mail: chloe.grazon@u-bordeaux.fr; mireille.blanchard-desce@u-bordeaux.fr
bUniv. Bordeaux, CNRS, INSERM, IECB, UAR 3033, F-33600 Pessac, France
First published on 10th February 2026
Abstract
Three quadrupolar fluorene-based dyes differing only by non-conjugated substituents form fluorescent organic nanoparticles (dFONs) with tunable emission from blue to yellow. A spirocyclic central substituent induces a marked red-shift via altered packing. When internalized into cells, dFONs evolve showing a characteristic blue-shift, enabling fluorescence-based monitoring of nanoparticle integrity and bioimaging performance.
Bioimaging applications require probes that not only emit brightly but also report on their physical and chemical state in complex environments. Conventional fluorescent nanoparticles (NPs) – whether inorganic quantum dots or dye-doped polymer/silica NPs – are designed for stability.1,2 However, in the case of NPs based on supramolecular organization, such as micelles or aggregates, assessing if their structural integrity is retained within cells is trickier. Monitoring such nanoparticle disassembly usually requires multi-dye strategies or labelling approaches, which complicate synthesis and interpretation.3–6
Fluorescent organic nanoparticles (FONs), and in particular dye-based FONs (dFONs) obtained by nanoprecipitation of dedicated π-conjugated dyes, provide a promising alternative.7,8 The spectral properties of dFONs are highly sensitive to the packing of the dye building blocks. Moreover, the fluorescence emission of dFONs made from polar and polarizable dyes is inherently sensitive to changes in the local environment, and supramolecular organization.9 This makes dFONs ideal candidates for developing self-reporting nanoprobes whose spectral response directly reflects their aggregation state.
Here, we report a family of quadrupolar dyes differing only in their non-conjugated substituents on a fluorene core, which form self-stabilized organic nanoparticles with distinct packing geometries and emission colours. The resulting dFONs display strong structure-dependent spectral signatures – from blue to yellow emission – originating from different degrees of excitonic coupling features. When internalized by eukaryotic cells, the dFONs with strong excitonic coupling undergo a characteristic blue-shift in their emission, indicating a change in packing and environment of the constituting dyes. This demonstrates that fluorescence can serve as a direct optical readout of nanoparticle integrity, without requiring additional labelling or ratiometric design.
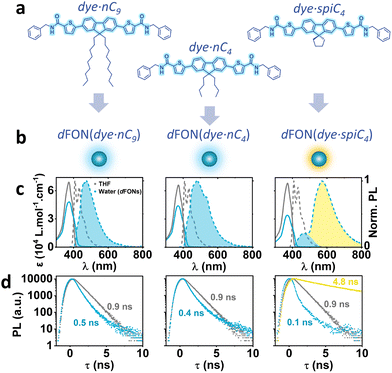
Three original quadrupolar dyes dye·nC9, dye·nC4 and dye·spiC4 (Fig. 1a) with identical π-conjugated systems were synthesized through a three- or four step synthesis. They possess a common conjugated part, constituting an electron-donating fluorene core, flanked by two thienyl moieties conjugated with two electron-withdrawing amide end-groups. The dyes differ only in the nature of the alkyl pending substituents in the fluorene core, with dye·nC9 bearing two nonyl chains, dye·nC4 two butyl chains and dye·spiC4 a spiro cycle (resulting in a spirofluorene core). The dyes are synthesized via a Pd-catalyzed Suzuki–Miyaura cross-coupling10–12 between home-made 2,7-diiodospiro[alkyl-9,9-fluorene] (Scheme S1) and commercially available 5-carboxythiophene-2-boronic acid pinacol ester to introduce a conjugated carboxylic acid function (Scheme S2). The di-acid is then activated (Scheme S3) and reacted with benzylamine (Schemes S4 and S5) to yield the purified desired dyes with overall yields of 12%, 9% and 29% respectively for dye·nC9, dye·nC4 and dye·spiC4 (1H and 13C NMR spectra provided in Fig. S2–S4).
The photophysical properties of the three dyes dye·nC9, dye·nC4 and dye·spiC4 were first examined in THF (Table 1 and Table S2). Considering their identical conjugated systems, all three dyes share similar properties in solution; an absorption band peaking at 373 nm associated with a molar absorption coefficient εmax at ca. 68 × 103 M−1 cm−1 and a fluorescence emission maximum of ca. 408 nm, associated with a fluorescence quantum yield ΦF of ca. 0.70 and a fluorescence lifetime τ of 0.9 ns. The ε-normalized absorption spectra and normalized emission spectra of the dyes in THF are represented in grey in Fig. 1c and the solvatochromism of the dyes is depicted in Fig. S5: the three dyes exhibit no dependence of the absorption and emission spectra on solvent polarity, while the well-defined vibronic structures of the emission spectra indicates low interaction of the excited state with the solvent. This indicates that no symmetry-breaking occurs in the excited state.
| Sample | Solvent | λ maxabs,a (nm) | ε max,b (×103 M−1 cm−1) | Δ ν ,c (cm−1) | λ maxem,d (nm) |
Φ
F![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e e |
τ amp,f (ns) | k r,g (ns−1) | k nr,h (ns−1) | B V,i (×103 M−1 cm−1 nm−3) |
|---|---|---|---|---|---|---|---|---|---|---|
A more comprehensive version of this table is provided in the SI (Table S1).a Absorption maximum wavelength.b Molar absorption coefficient of the dyes at λmaxabs (in solution in THF or as dFONs subunits), measured from three differently prepared solutions and subsequent dilutions (error bars correspond to the standard deviation on all points).c Stokes shift.d Emission maximum wavelength.e Relative fluorescence quantum yield, using references: quinine bisulfate in H2SO4 0.5 M (ΦF = 0.546)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13 and 4-(dicyanomethylene)-2-methyl-6-[p-(dimethyl-amino)styryl]-4H-pyran in EtOH (ΦF = 0.437).14 Error bars correspond to the standard error on the linear regression.f Amplitude-averaged fluorescence lifetime, using λexc = 370 nm and λem = 450 nm, except for (*): λem = 590 nm.g Radiative rate constant.h Non-radiative rate constant.i Brightness per volume. All steady-state fluorescence emission spectra were recorded using λexc = 370 nm. 13 and 4-(dicyanomethylene)-2-methyl-6-[p-(dimethyl-amino)styryl]-4H-pyran in EtOH (ΦF = 0.437).14 Error bars correspond to the standard error on the linear regression.f Amplitude-averaged fluorescence lifetime, using λexc = 370 nm and λem = 450 nm, except for (*): λem = 590 nm.g Radiative rate constant.h Non-radiative rate constant.i Brightness per volume. All steady-state fluorescence emission spectra were recorded using λexc = 370 nm. |
||||||||||
| dye·nC9 | THF | 373 | 69 ± 8 | 2300 | 408 | 0.72 ± 0.02 | 0.9 | 0.8 | 0.3 | 35 |
| dFON(dye·nC9) | H2O | 374 | 48 ± 8 | 5685 | 475 | 0.23 ± 0.01 | 0.5 | 0.5 | 1.5 | 8 |
| dye·nC4 | THF | 373 | 66 ± 2 | 2360 | 409 | 0.65 ± 0.03 | 0.9 | 0.7 | 0.4 | 36 |
| dFON(dye·nC4) | H2O | 370 | 44 ± 2 | 6237 | 481 | 0.15 ± 0.01 | 0.4 | 0.4 | 2.1 | 6 |
| dye·spiC4 | THF | 372 | 68 ± 2 | 2372 | 408 | 0.70 ± 0.03 | 0.9 | 0.8 | 0.3 | 44 |
| dFON(dye·spiC4) | H2O | 362 | 34 ± 2 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 050 050 |
569 (466) | 0.03 ± 0.01 | 0.1/4.8* | 0.1/0.01* | 10/0.2* | 0.9 |
The three dFONs were prepared from these three dyes using the nanoprecipitation method (Fig. S1): a 0.5 mM stock solution of dye in THF![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (9
DMSO (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was added dropwise to a large volume of distilled water under magnetic stirring, such that the final proportion of organic solvent did not surpass 1%. This process yielded colourless and non-turbid colloidal dispersions of dFONs.
1) was added dropwise to a large volume of distilled water under magnetic stirring, such that the final proportion of organic solvent did not surpass 1%. This process yielded colourless and non-turbid colloidal dispersions of dFONs.
The photophysical properties of the resulting nanoparticles dFON(dye·nC9), dFON(dye·nC4) and dFON(dye·spiC4) were then investigated (Table 1 and Table S2). The normalized absorption and emission spectra of the dFONs are represented in blue in Fig. 1c (full normalized spectra in Fig. S5). While the absorption spectra of dFON(dye·nC9) and dFON(dye·nC4) are quite similar to their molecular counterparts in THF, dFON(dye·spiC4) presents a 10 nm hypsochromic shift. Additionally, the molecular ε of dyes as building blocks of dFON(dye·nC9) and dFON(dye·nC4) was ca. 30% lower than that of the dyes in THF, while that for dFON(dye·spiC4) was 50% lower. This hypochromic effect, along with the slight broadening (Fig. 1c and Fig. S5) of the absorption band were reported before in closely related systems9,12 and were attributed to excitonic splitting.15,16 The most striking differences by far are found in the dFONs emissive properties. Compared to the molecular dyes in solution, all dFONs display a broadened and bathochromically shifted emission, with a loss of vibronic structure (Fig. 1c and Table 1). dFON(dye·nC9) and dFON(dye·nC4) emission spectra are not identical but close, with an emission at 475 nm and 481 nm, respectively. The spectral shape of dFON(dye·nC4) is slightly broader and displays a small additional shoulder around 540 nm. These observed changes in the shape, position, and intensity of the emission spectrum are a common occurrence in dFONs, due to intermolecular interactions. In addition, the fluorescence quantum yields (ΦF) of dFON(dye·nC9) (ΦF = 0.23) and dFON(dye·nC4) (ΦF = 0.15) are lower than that of the dyes in THF solutions. This trend arises from two key factors: a slight reduction in the radiative rate constant (kr) in relation to the interchromophoric interactions within the aggregated dFONs, and a marked increase in the non-radiative rate constant (knr) (Table 1 and Fig. S6), which might be ascribed to vibrational deactivation due to water molecules in close proximity to the surface of the nanoparticle.17 We also stress that the fluorescence emission of crystals of dye·nC9 and dye·nC4 does not overlap with the corresponding dFONs spectra (Fig. S8), suggesting the amorphous nature of these molecular aggregates.
In comparison, dFON(dye·spiC4) emissive properties are strikingly different: the spectrum comprises a main band at 569 nm, and a minor band (about 15% of the main band in height) at 466 nm, resembling that of dFON(dye·nC9) and dFON(dye·nC4). Concomitantly, the fluorescence quantum yield is drastically reduced, down to 0.03, and the fluorescence lifetime is significantly increased to 4.8 ns at 590 nm (Fig. 1d and Fig. S6, S7). We attribute this new band in the yellow region to strong excitonic coupling,16,18 which stabilizes a low-energy emissive state, thus reducing kr and enhancing knr and resulting in reduced brightness. The fact that only dFON(dye·spiC4) exhibit this band indicates that the rigid nature of the spiro substituent and its small size play a crucial role, by strongly affecting molecular packing within dFONs. This showcases the importance of the nature of the substituents in dye design for dFONs.9,19
For comparison, a dye bearing the same spiro substituent but with benzyl-amide groups replaced by diethyl-imide groups (dye·spiC4NEt2 (6), Scheme S6, Fig. S5–S7 and Tables S1, S2)12 exhibits only a weak red shoulder in its emission. In addition, the crystal structures of dye·spiC4 and dye·spiC9 (Fig. S11, S12 and Tables S3, S4) indicate that the benzyl-amide groups could play a role in the molecular arrangement of the dyes. This suggests that, beyond the spiro unit promoting closer dye proximity compared to extended alkyl chains, the benzyl-amide groups at the dye termini may also modulate the spatial organization of the dyes within dFONs, thereby influencing their optical properties. The effective Stokes Shift of dFON(dye·spiC4) is huge compared to that of dFON(dye·nC9) and dFON(dye·nC4) (10 × 103 cm−1 compared to 6 × 103 cm−1).
The morphological characteristics of dFONs were investigated using transmission electron microscopy (TEM), as illustrated in Fig. 2 and Fig. S9. dFON(dye·nC9) and dFON(dye·nC4) appeared as spherical nanoparticles with mean diameters of 35–40 nm. dFON(dye·spiC4) were found to exhibit polymorphism, with one population of spherical nanoparticles, averaging ca. 35 nm in diameter and a second population of “rod-like” particles, measuring roughly 20 nm in width and from 20 up to 150 nm in length.
The average number of dyes per nanoparticles (N, eqn (S1)) is derived from the dFONs dry diameter. The theoretical brightness (B, eqn (S4), Table S1 and Fig. S10) and brightness per volume (BV, eqn (S5), Table 1) of the NPs are then calculated. While dFON(dye·nC9) and dFON(dye·nC4) have a similar brightness around 2 × 108 M−1 cm−1, the brightness of dFON(dye·spiC4) is ten times lower (2 × 107 M−1 cm−1). This resulted in brightnesses per volume around 7 × 103 M−1 cm−1 nm−3 for dFON(dye·nC9) and dFON(dye·nC4) and 0.9 × 103 M−1 cm−1 nm−3 for dFON(dye·spiC4). The BV of dFONs containing dyes with alkyl chains is approximately ten times higher than that of PMMA nanoparticles doped with blue-emitting cyanine (BV = 7 × 102 M−1 cm−1 nm−3) for NPs of comparable size.20 In contrast, dFON(dye·spiC4) exhibit a 100-fold higher overall brightness compared to QD540 (B = 1.7 × 105 M−1 cm−1),21 while showing a similar brightness per unit volume (BV,QD540 = 1.5 × 103 M−1 cm−1 nm−3), which can be attributed to the smaller size of bare QDs.
Finally, before using these nanoparticles in biological applications, their colloidal stabilities in different media (water or cell culture medium) were evaluated (Fig. S13). Over 24 hours, neither a change in absorbance nor setting was observed. This confirms the good colloidal stability of the dFONs in such conditions and supports their potential use in cell culture medium. In addition, the emissive properties of the dFON(dye·spiC4) were evaluated in supplemented cell culture media (DMEM + FBS) (Fig. S14). Overall the emissive spectra remain the same, but a decrease of the band at 570 nm and an increase of the band at 466 nm is observed.
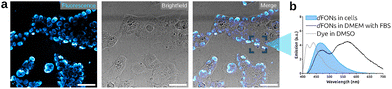
dFON(dye·spiC4) were then evaluated as bioimaging probes in fluorescence microscopy experiments on HEK and COS-7 cells (Fig. 3 and Fig. S15, S16). First, the dFONs were incubated with HEK cells in DMEM supplemented with 10% FBS. Live, wash-free imaging was then conducted over one hour to monitor the intracellular fate of the nanoparticles. Under these conditions, only the dFONs that had entered or were concentrated inside the cells could be detected, whereas dispersed nanoparticles in the culture medium remained below the detection threshold.
After approximately one hour, the dFONs accumulated inside the cells and displayed an emission band with a maximum around 460 nm (Fig. 3). This emission does not overlap with that of the dFONs in DMEM + FBS, nor with that of the isolated dye·spiC4 in a polar solvent such as DMSO. Instead, it closely resembles the emission of dFON(dye·nC4) or dFON(dye·nC9). To further investigate this unexpected behaviour, the intracellular accumulation of dFON(dye·spiC4) was monitored at ca. 30, 40, 50 and 90 min (Fig. S15). Remarkably, the nanoparticles entered the cells rapidly, within ca. 30 min, and continued to accumulate over the full duration of the experiment. Concomitantly, the fluorescence emission of dFON(dye·spiC4) underwent a pronounced hypsochromic shift.
We hypothesize that upon cell internalization, the dFONs undergo a structural reorganization within the cellular environment while remaining in an aggregate state. Such a reorganization would account for the loss of excitonic coupling signatures, while maintaining an emission profile characteristic of aggregated dyes rather than isolated chromophores.
To assess whether this behavior was cell-line dependent, we also incubated dFON(dye·spiC4) with COS-7 cells and imaged them after 2 h (Fig. S16). Strikingly, the fluorescence signal was again centered around 450 nm and the excitonic coupling signature was lost. These observations indicate that the dFONs exhibit the same intracellular behavior regardless of the cell line investigated (fibroblast or embryonic).
Our study demonstrates that a single substituent modification on a dye can drastically alter its solid-state properties, leading in our case to a pronounced red-shift in emission. This unique spectral signature provides an efficient means to monitor the aggregation state of fluorophores within nanoparticles without the need for mixed-dye systems – an important asset for applications such as biosensing or drug-delivery, where controlling nanoparticle behaviour is essential. In living cells, dFON(dye·spiC4) displays a markedly broadened and blue-shifted emission compared to dFONs in aqueous suspension, a behaviour that indicates a loss of excitonic coupling promoted either by the dissociation of the nanoaggregates (as previously observed for other dFONs3) or by reorganization of the dyes within the assemblies. What makes this system truly innovative is its reliance on a single dye component, which—despite its simplicity—achieves dramatic fluorescence signal modulation in cells. This modulation is driven entirely by the dynamic reorganization of the dyes within the nanoparticle, rather than the conventional “off/on” mechanisms seen in most systems (where fluorescence is quenched in aggregated states and only recovers upon nanoparticle dissolution in biological media)22 or solvatochromic effects, where the dissolution of the nanoparticles is followed by a change in the fluorescence signal.23 Importantly, the optical tuning is achieved here while preserving the nanoparticle features associated with high brightness. Such sensitivity to the cellular environment, combined with distinct staining profiles across cell lines, highlights that the nature of the dye substituents has a strong influence on the cellular fate of dFONs (as already demonstrated for other systems9,24) and further supports their potential for controlled sensing12 and drug-delivery applications.25
CRediT: Eleonore Kurek: investigation, validation, methodology, visualisation, writing – original draft preparation. Ophélie Dal Pra: investigation, visualisation, writing – review & editing. Aliocha Skrzypczak: investigation, writing – Review & Editing. Stéphane Massip: investigation, data curration. Jonathan Daniel: investigation, visualization, writing – review & editing. Mireille Blanchard-Desce: conceptualization, funding acquisition, supervision, writing – review & editing. Chloé Grazon: conceptualization, data curation, funding acquisition, methodology, project administration, supervision, visualisation, writing – original draft preparation.
Conflicts of interest
There are no conflicts to declare.Data availability
Data for this article are available at Zenodo repository 18324973.Supplementary information (SI): materials and methods, chemical characterizations of dyes, extra photophysical datas, bioimaging. See DOI: https://doi.org/10.1039/d5cc06995e.
CCDC 2524470 and 2524471 contain the supplementary crystallographic data for this paper.26a,b
Acknowledgements
The synthesis of dyes 5b and 6 were performed by J. Traisnel and A. Okba, respectively. This project has received funding from the European Research Council (ERC) under the European Union's Horizon Europe research and innovation program (Grant agreement no. 101077364). Financial support for ODP and EK was provided through the MENESR (Ministère de l’Education Nationale, de l’Enseignement Supérieur et de la Recherche) of France. This work has also benefited from the LIGHT S&T Graduate Program (PIA 3 Investment for Future Program, ANR-17-EURE-0027), support and facilities and expertise of IECB Biophysical and Structural Chemistry platform (BPCS), CNRS UAR3033, Inserm US001, Univ Bordeaux. Continuous support from the University of Bordeaux, Bordeaux-INP and CNRS is greatly acknowledged.References
- W. R. Algar, A. Szwarczewski and M. Massey, Anal. Chem., 2023, 95, 551–559 CAS.
- W. R. Algar, M. Massey, K. Rees, R. Higgins, K. D. Krause, G. H. Darwish, W. J. Peveler, Z. Xiao, H.-Y. Tsai, R. Gupta, K. Lix, M. V. Tran and H. Kim, Chem. Rev., 2021, 121, 9243–9358 CrossRef CAS PubMed.
- J. Boucard, T. Briolay, T. Blondy, M. Boujtita, S. Nedellec, P. Hulin, M. Grégoire, C. Blanquart and E. Ishow, ACS Appl. Mater. Interfaces, 2019, 11, 32808–32814 Search PubMed.
- N. Curtin, M. Garre, J.-B. Bodin, N. Solem, R. Méallet-Renault and D. F. O'Shea, RSC Adv., 2022, 12, 35655–35665 RSC.
- M. Zhang, C. Wang, C. Yang, H. Wu, H. Xu and G. Liang, Anal. Chem., 2021, 93, 5665–5669 CrossRef CAS PubMed.
- S. W. Morton, X. Zhao, M. A. Quadir and P. T. Hammond, Biomaterials, 2014, 35, 3489–3496 CrossRef CAS PubMed.
- V. Parthasarathy, S. Fery-Forgues, E. Campioli, G. Recher, F. Terenziani and M. Blanchard-Desce, Small, 2011, 7, 3219–3229 CrossRef CAS PubMed.
- S. Fery-Forgues, Nanoscale, 2013, 5, 8428–8442 RSC.
- J. Daniel, O. D. Pra, E. Kurek, C. Grazon and M. Blanchard-Desce, C. R. Chim, 2024, 27, 1–17 CrossRef CAS.
- J. Daniel, A. G. Godin, M. Palayret, B. Lounis, L. Cognet and M. Blanchard-Desce, J. Phys. D: Appl. Phys., 2016, 49, 084002 Search PubMed.
- H. Li, J. Daniel, J.-B. Verlhac, M. Blanchard-Desce and N. Sojic, Chem. – Eur. J., 2016, 22, 12702–12714 CrossRef CAS PubMed.
- O. Dal Pra, J. Daniel, G. Recher, M. Blanchard-Desce and C. Grazon, Small Methods, 2024, 8, 2400716 CrossRef CAS PubMed.
- D. F. Eaton, Pure Appl. Chem., 1988, 60, 1107–1114 CrossRef CAS.
- K. Rurack and M. Spieles, Anal. Chem., 2011, 83, 1232–1242 CrossRef CAS PubMed.
- M. Kasha, H. R. Rawls and M. Ashraf El-Bayoumi, Pure Appl. Chem., 1965, 11, 371–392 Search PubMed.
- A. S. Davydov, Phys.-Usp., 1964, 7, 145 Search PubMed.
- J. Maillard, K. Klehs, C. Rumble, E. Vauthey, M. Heilemann and A. Fürstenberg, Chem. Sci., 2021, 12, 1352–1362 RSC.
- J. Gierschner, J. Shi, B. Milián-Medina, D. Roca-Sanjuán, S. Varghese and S. Park, Adv. Opt. Mater., 2021, 9, 2002251 CrossRef CAS.
- J. Daniel, These de doctorat, Bordeaux, 2015 Search PubMed.
- P. Ashokkumar, N. Adarsh and A. S. Klymchenko, Small, 2020, 16, 2002494 CrossRef CAS PubMed.
- R. Gupta, W. J. Peveler, K. Lix and W. R. Algar, Anal. Chem., 2019, 91, 10955–10960 CrossRef CAS PubMed.
- J. Boucard, C. Linot, T. Blondy, S. Nedellec, P. Hulin, C. Blanquart, L. Lartigue and E. Ishow, Small, 2018, 14, 1802307 CrossRef PubMed.
- A. Faucon, H. Benhelli-Mokrani, L. A. Córdova, B. Brulin, D. Heymann, P. Hulin, S. Nedellec and E. Ishow, Adv. Healthcare Mater., 2015, 4, 2727–2734 CrossRef CAS PubMed.
- A. H. A. M. V. Onzen, L. Albertazzi, A. P. H. J. Schenning, L.-G. Milroy and L. Brunsveld, Chem. Commun., 2017, 53, 1626–1629 RSC.
- A. Jana, K. S. P. Devi, T. K. Maiti and N. D. P. Singh, J. Am. Chem. Soc., 2012, 134, 7656–7659 CrossRef CAS PubMed.
- (a) CCDC 2524470: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qqxjx; (b) CCDC 2524471: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2qqxky.
Footnotes |
| † These authors contributed equally to this work. |
| ‡ Present address: Univ Rennes, INSA Rennes, CNRS, ISCR (Institut des Sciences Chimiques de Rennes), UMR 6226, Université de Rennes, Rennes, France. |
| This journal is © The Royal Society of Chemistry 2026 |