Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Water-compatible acylation reactions with acid chlorides using a flow microreactor
Hiromichi V.
Miyagishi†
 ,
Daito
Takahashi†
,
Yiyuan
Jiang
,
Masahiro
Takumi
,
Daito
Takahashi†
,
Yiyuan
Jiang
,
Masahiro
Takumi
 and
Aiichiro
Nagaki
and
Aiichiro
Nagaki
 *
*
Department of Chemistry, Faculty of Science, Hokkaido University, Kita-10 Nishi-8 Kita-ku, Sapporo 060-0810, Japan. E-mail: nagaki@sci.hokudai.ac.jp
First published on 10th March 2026
Abstract
In this study, we used a flow microreactor for acylation with acid chlorides in a homogeneous aqueous medium, suppressing hydrolysis and providing up to 98% yield. Quenched-flow analysis indicated an associative transition state. This method applies to amines, phenols, thiols, and pharmaceuticals. Acylation of oseltamivir proceeds with a throughput of 2.1 g h−1, demonstrating scalability and practicality.
Condensation reactions that form amide bonds are essential in medicinal chemistry and peptide synthesis (Fig. 1A).1–3 According to recent analyses, amide bond formation reactions account for approximately 16% of all the transformations used in the synthesis of new drug candidates, which indicates their importance in drug discovery.4 Although amidation is usually carried out in organic solvents, interest in aqueous media has been increasing, especially for reactions with water-soluble compounds such as pharmaceuticals and biomolecules.5–14 From the viewpoint of green chemistry, aqueous systems are considered more environmentally friendly and practical. Activated esters and condensation reagents are often used for amidation in water; however, many of them have low reactivity and require long reaction times.12,15,16 Therefore, the development of highly reactive reagents for rapid amide bond formation in aqueous media is strongly desired.
 | ||
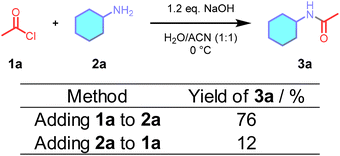
| Fig. 1 (A) Examples of pharmaceuticals containing an amide bond, (B) amidation with acid chloride and competing hydrolysis, and (C) amide synthesis using a flow microreactor (FMR). | ||
Among the various acylating reagents, acid chlorides are known for their extremely high reactivity, enabling rapid amidation reactions.15,17,18 However, under aqueous or water-containing conditions, they are inevitably hydrolyzed, making it difficult to efficiently promote the desired transformation (Fig. 1B).18,19 Although the Schotten–Baumann reaction, conducted in a biphasic system of water and organic solvent, is widely used to suppress hydrolysis,20 its scale-up is often limited by the small interfacial area and inefficient mass transfer.21,22 Therefore, the development of efficient acylation reactions in homogeneous water-containing solvents, remains an important challenge.
In this study, we focused on flow microreactor (FMR) technology to achieve efficient amide bond formation in homogeneous water-containing systems (Fig. 1C).14,15,23–26 FMR enables rapid mixing on a millisecond timescale, allowing reactions involving unstable species to proceed efficiently.27–34 In addition, FMR systems exhibit high durability, making them well-suited for integration into continuous chemical production processes.35,36 Positing that acid chlorides, which are typically unstable in aqueous media, can be utilized for amidation reactions using FMR, we demonstrated that FMR enhances the yield of amide formation reactions and enables kinetic analysis using quenched-flow techniques using the same setup. Furthermore, we successfully applied this method to the acylation of pharmaceutical compounds.
First, we attempted the N-acylation reaction under aqueous conditions in a batch reactor. A 0.1 M solution of acetyl chloride 1 in acetonitrile was mixed with a 0.1 M aqueous solution of cyclohexylamine containing 1.2 eq. of NaOH, following the procedures in Fig. 2. Addition of the amine solution to the acetyl chloride solution produced the desired amide product in only 12% yield, likely because of the hydrolysis of acetyl chloride by excess water in the system. In contrast, the addition of acetyl chloride solution to the aqueous amine solution resulted in 76% yield. These results indicate that although the reaction proceeded with a reasonable yield, the mixing method still had a significant influence on the yield, possibly due to local differences in water concentration in the reactor.
Next, we investigated the reaction using FMR (Fig. 3). To optimize the reaction conditions, a series of experiments were conducted by varying the inner diameter of the micromixer (∅: 250 and 500 µm) and total flow rate (Q: 1.5–12 mL min−1). Consequently, increasing the flow rate led to higher yields, with a maximum of 86% at Q = 6 mL min−1. The micromixer with a 250 µm inner diameter provided higher yields than the micromixer with 500 µm. This is presumably because insufficient mixing at lower flow rates leads to the hydrolysis of acetyl chloride. Based on these investigations, the optimal conditions were determined to be a micromixer with a 250 µm inner diameter and a flow rate of 6 mL min−1. These results demonstrate that the FMR-based system achieves higher yields than the corresponding batch process.
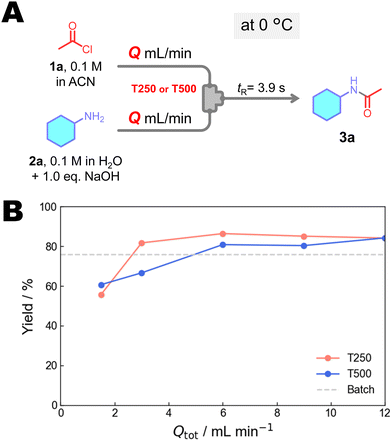 | ||
| Fig. 3 (A) Amidation of 2a under various mixers and flow rates and (B) effect of total flow rate on the yield of 3a. | ||
Subsequently, the effect of the base on reaction was investigated (Fig. 4). No significant improvement in yield was observed among the different bases tested. We examined the effect of NaOH loading by varying the number of equivalents from 0 to 5. The results showed that the yield reached 1.2 eq. with increasing NaOH equivalents. Based on these findings, NaOH, a commonly used base, was selected as the optimal base, and the optimal amount was determined to be 1.2 equivalents. Under these conditions, the desired amide produced 87% yield.
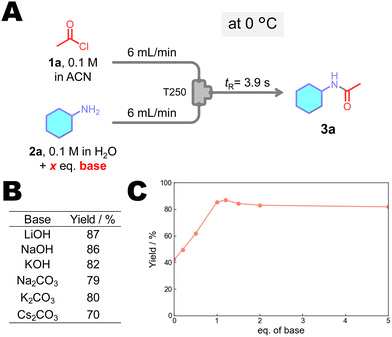 | ||
| Fig. 4 (A) Amidation of 2a using various bases, and effect of (B) base type and (C) base equivalents on the yield of 3a. | ||
The reaction kinetics was also investigated using the quenched-flow (QF) method (Fig. 5). The QF method enables monitoring of fast reactions by rapidly quenching them.37 Here, the amidation reaction was quenched with an excess amount of nPrNH2 following the acid chloride reaction with CyNH2 (Fig. 5A). Reaction progress was monitored by varying the residence time (tR), and a fitting analysis was performed to determine the kinetic parameters. The activation enthalpies (ΔH‡) for condensation and hydrolysis were 25.0 and 49.4 kJ mol−1, respectively, and the activation entropies (ΔS‡) were −81.3 and −41.9 J mol−1 K−1, respectively. These results demonstrate that amidation proceeds via a highly ordered associative transition state, as evidenced by the large negative ΔS‡ value.38
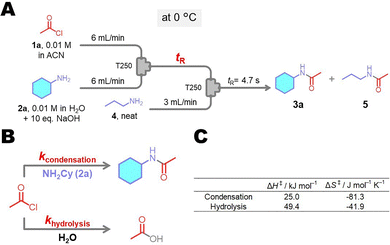 | ||
| Fig. 5 (A) Quenched-flow experiment of amidation of 2a, (B) side reaction of amidation, and (C) ΔH‡ and ΔS‡ values determined for the condensation and hydrolysis reactions. | ||
The substrate scope of amidation is shown in (Scheme 1). Reactions with primary amines (3a–e) yielded 83–90%. Acylation of 2-aminoethanol yielded 59% of N-acylated product (3f). This method, although also applicable to anilines (3g), reactions with secondary amines produced low yields, except for piperidine with 68% yield (3h–j). Although N-acylation of glucosamine resulted in low yield (3k), corresponding products were obtained in moderate-to-good yields with amino acid methyl esters (3l–o). Extension of this method to other acid chlorides revealed its applicability to various acyl chlorides (3p–r) and sulfonyl chlorides (3u–w), although methanesulfonyl chloride (3u) gave a lower yield likely due to its rapid hydrolysis. The protocol was also successfully applied to S-acylation of an aliphatic thiol and O-acylation of phenols (3s and 3t), whereas O-acylation of ethanol (3x) resulted in a low yield even with 3 equivalents of acid chloride. The results demonstrate the versatility of the flow method for amidation reactions involving a wide range of nucleophiles and acyl donors.
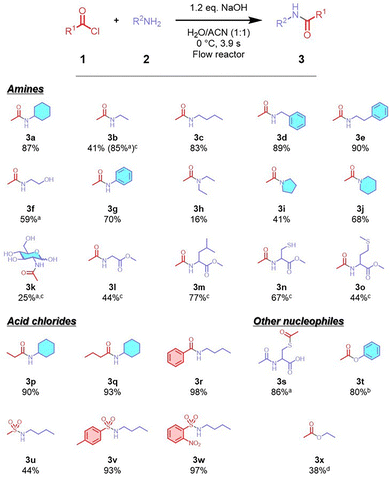 | ||
Scheme 1 Scope of amidation using an FMR system. a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Determined by 1H NMR. b Determined by 1H NMR. b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Determined by GC. c Determined by GC. c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.2 eq. NaOH. d 2.2 eq. NaOH. d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.0 eq. AcCl and 3.2 eq. NaOH. 3.0 eq. AcCl and 3.2 eq. NaOH. | ||
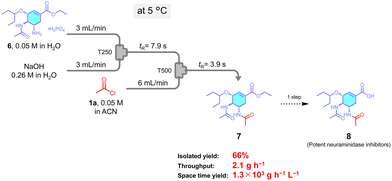
Finally, the flow method was applied to the synthesis of a pharmacological compound. Oseltamivir (6), an antiviral agent,39,40 was subjected to N-acylation using the flow setup in (Fig. 6). Product 7 was obtained in 66% yield, with a throughput of 2.1 g h−1 and a space-time yield of 1.3 × 104 g h−1 L−1. Product 7 was converted via ester hydrolysis to compound 8, a neuraminidase inhibitor active against new variants of the virus, via ester hydrolysis.41 These results demonstrate the applicability of the developed flow method for the efficient synthesis of pharmaceutically relevant molecules.
In conclusion, we developed an efficient FMR method for amide bond formation in homogeneous water-containing systems using acid chlorides as highly reactive acyl donors. Rapid mixing in FMR suppresses competitive hydrolysis and enables amidation under mild conditions. This method covers a broad range of nucleophiles and acyl donors, allowing kinetic analysis via quenched-flow experiments conducted in a similar setup. This approach was successfully applied to the synthesis of pharmaceutically relevant molecules. The results demonstrated that the developed FMR-based amidation strategy provides a powerful and scalable platform for the rapid, selective, and sustainable formation of amide bonds in aqueous media.
Conceptualization, AN; investigation, DT, YJ, MT; resources, HVM, AN; data curation, HVM, DT; writing – original draft preparation, HVM; writing – review and editing, HVM, DT, AN; visualization, HVM, DT; supervision, AN; project administration, HVM, AN; funding acquisition, HVM, AN. All the authors have read and agreed to the published version of the manuscript.
Conflicts of interest
There are no conflicts to declare.Data availability
Data supporting this article are included as part of the supplementary information (SI). supplementary information is available. See DOI: https://doi.org/10.1039/d5cc06943b.Acknowledgements
This work was supported by JSPS KAKENHI (Grant Numbers JP20KK0121, JP21H01936, JP23K17984, JP23H01961, JP23K04744, JP24K01515, JP25K21715, JP23K19232, JP25K18015), Japan Agency for Medical Research and Development (AMED-BINDS, AMED-GAPFREE7.2), New Energy and Industrial Technology Development Organization (NEDO, JPNP19004, JPNP14004), JST ACT-X (JPMJAX23DK), the Japan Keirin Autorace Foundation, the Takahashi Industrial and Economic Foundation, and Hokkaido Startup Future Creation development by mutual support networks (HSFC). We thank the Global Research Facility Alliance Center (Hokkaido University) for ESI-MS measurements using Thermo Scientific Orbitrap Elite. We also thank Mr Yusuke Takata (Hokkaido University) for FD-MS measurements using the JMS-T2000GCalpha and for his expert technical support. Part of analytical instrument was provided through the support of the JSPS Regional Core and Distinctive Research University Strengthening Promotion Project JPJS00420230001.References
- E. Valeur and M. Bradley, Chem. Soc. Rev., 2009, 38, 606–631 Search PubMed.
- V. R. Pattabiraman and J. W. Bode, Nature, 2011, 480, 471–479 CrossRef CAS PubMed.
- P. Acosta-Guzmán, A. Ojeda-Porras and D. Gamba-Sánchez, Adv. Synth. Catal., 2023, 365, 4359–4391 CrossRef.
- S. D. Roughley and A. M. Jordan, J. Med. Chem., 2011, 54, 3451–3479 CrossRef CAS PubMed.
- K. Hojo, H. Ichikawa, M. Maeda, S. Kida, Y. Fukumori and K. Kawasaki, J. Pept. Sci., 2007, 13, 493–497 Search PubMed.
- K. Hojo, H. Ichikawa, M. Onishi, Y. Fukumori and K. Kawasaki, J. Pept. Sci., 2011, 17, 487–492 CrossRef CAS PubMed.
- A. S. Galanis, F. Albericio and M. Grøtli, Org. Lett., 2009, 11, 4488–4491 CrossRef CAS PubMed.
- Q. Wang, Y. Wang and M. Kurosu, Org. Lett., 2012, 14, 3372–3375 CrossRef CAS PubMed.
- C. M. Gabriel, M. Keener, F. Gallou and B. H. Lipshutz, Org. Lett., 2015, 17, 3968–3971 CrossRef CAS PubMed.
- B. H. Lipshutz, F. Gallou and S. Handa, ACS Sustainable Chem. Eng., 2016, 4, 5838–5849 CrossRef CAS.
- D. Procopio, C. Siciliano, S. Trombino, D. E. Dumitrescu, F. Suciu and M. L. Di Gioia, Org. Biomol. Chem., 2022, 20, 1137–1149 RSC.
- S. Hazra, F. Gallou and S. Handa, ACS Sustainable Chem. Eng., 2022, 10, 5299–5306 CrossRef CAS.
- N. Soumanou, D. Lybye, T. Hjelmgaard and S. Faure, Green Chem., 2023, 25, 3615–3623 RSC.
- S. H. A. Rajendran, S. Kogler, P. Kögl, W. M. Braje, S. B. Ötvös and C. O. Kappe, ACS Sustainable Chem. Eng., 2025, 13, 6423–6432 CrossRef CAS.
- S. Fuse, Y. Mifune and T. Takahashi, Angew. Chem., Int. Ed., 2014, 53, 851–855 CrossRef CAS PubMed.
- M. Badland, R. Crook, B. Delayre, S. J. Fussell, I. Gladwell, M. Hawksworth, R. M. Howard, R. Walton and G. A. Weisenburger, Tetrahedron Lett., 2017, 58, 4391–4394 CrossRef CAS.
- N. O. V. Sonntag, Chem. Rev., 1953, 52, 237–416 CrossRef CAS.
- G. Zimmerman and C. Yuan, J. Am. Chem. Soc., 1955, 77, 332–333 CrossRef CAS.
- A. Queen, Can. J. Chem., 1967, 45, 1619–1629 CrossRef CAS.
- Comprehensive Organic Name Reactions and Reagents, 2010, 2536–2539.
- K. J. Carpenter, Chem. Eng. Sci., 2001, 56, 305–322 CrossRef CAS.
- G. Caygill, M. Zanfir and A. Gavriilidis, Org. Process Res. Dev., 2006, 10, 539–552 CrossRef CAS.
- A. I. Alfano, H. Lange and M. Brindisi, ChemSusChem, 2022, 15, e202102708 CrossRef CAS PubMed.
- L. S. de, M. Miranda, R. O. M. A. de Souza, R. A. C. Leão, P. F. Carneiro, S. F. Pedraza, O. V. de Carvalho, S. P. de Souza and R. V. Neves, Org. Process Res. Dev., 2019, 23, 2516–2520 CrossRef.
- M. Magosso, M. van den Berg and J. van der Schaaf, React. Chem. Eng., 2021, 6, 1574–1590 RSC.
- J. B. Wolf, J. W. Lee, M. B. Plutschack, D. Cambié, A. Seidel-Morgenstern and P. H. Seeberger, React. Chem. Eng., 2024, 9, 959–966 RSC.
- A. Nagaki, Tetrahedron Lett., 2019, 60, 150923 CrossRef CAS.
- A. Nagaki, Y. Ashikari, M. Takumi and T. Tamaki, Chem. Lett., 2021, 50, 485–492 CrossRef CAS.
- Y. Ashikari, T. Kawaguchi, K. Mandai, Y. Aizawa and A. Nagaki, J. Am. Chem. Soc., 2020, 142, 17039–17047 CrossRef CAS PubMed.
- K. Okamoto, R. Higuma, K. Muta, K. Fukumoto, Y. Tsuchihashi, Y. Ashikari and A. Nagaki, Chem. – Eur. J., 2023, e202301738 CrossRef CAS PubMed.
- H. V. Miyagishi, Y. Kimuro, Y. Ashikari and A. Nagaki, Org. Lett., 2024, 26, 5032–5036 CrossRef CAS PubMed.
- H. Soutome, H. Yamashita, Y. Shimizu, M. Takumi, Y. Ashikari and A. Nagaki, Nat. Commun., 2024, 15, 4873 CrossRef CAS PubMed.
- K. Muta, K. Okamoto, H. Nakayama, S. Wada and A. Nagaki, Nat. Commun., 2025, 16, 416 CrossRef CAS PubMed.
- Y. Ishii, T. Ogawa, K. Onda, H. Miyagishi, Y. Ashikari, A. Nagaki, S. Asano and K. Miyata, ChemRxiv, 2025, preprint DOI:10.26434/chemrxiv-2025-llskv.
- Y. Nakahara, M. Furusawa, Y. Endo, T. Shimazaki, K. Ohtsuka, Y. Takahashi, Y. Jiang and A. Nagaki, Chem. Eng. Technol., 2019, 42, 2154–2163 CrossRef CAS.
- H. Soutome, K. Maekawa, Y. Ashikari and A. Nagaki, Org. Process Res. Dev., 2024, 28, 2006–2012 CrossRef CAS.
- H. Hartridge and F. J. W. Roughton, Proc. R. Soc. London, Ser. A, 1923, 104, 376–394 CAS.
- T. W. Bentley, G. Llewellyn and J. A. McAlister, J. Org. Chem., 1996, 61, 7927–7932 CrossRef CAS PubMed.
- C. U. Kim, W. Lew, M. A. Williams, H. Liu, L. Zhang, S. Swaminathan, N. Bischofberger, M. S. Chen, D. B. Mendel, C. Y. Tai, W. G. Laver and R. C. Stevens, J. Am. Chem. Soc., 1997, 119, 681–690 CrossRef CAS PubMed.
- J. C. Rohloff, K. M. Kent, M. J. Postich, M. W. Becker, H. H. Chapman, D. E. Kelly, W. Lew, M. S. Louie, L. R. McGee, E. J. Prisbe, L. M. Schultze, R. H. Yu and L. Zhang, J. Org. Chem., 1998, 63, 4545–4550 CrossRef CAS.
- K. Wang, F. Yang, L. Wang, K. Liu, L. Sun, B. Lin, Y. Hu, B. Wang, M. Cheng and Y. Tian, Eur. J. Med. Chem., 2017, 141, 648–656 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |