Open Access Article
Open Access ArticleEnhancing catalytic amine regeneration in CO2 capture using ZrOxHy-coated mesoporous silica
Cheng
Zhou
 *a,
Yuewen
Shao
a,
Mostafa Torka
Beydokhti
*a,
Yuewen
Shao
a,
Mostafa Torka
Beydokhti
 a,
Jing
Ma
a,
Pierre
Eloy
a,
Jing
Ma
a,
Pierre
Eloy
 b,
Damien P.
Debecker
b,
Damien P.
Debecker
 b,
Michiel
Dusselier
b,
Michiel
Dusselier
 a,
Walter
Vermeiren
c and
Bert F.
Sels
a,
Walter
Vermeiren
c and
Bert F.
Sels
 *a
*a
aCenter for Sustainable Catalysis and Engineering, KU Leuven, Celestijnenlaan 200F, Heverlee 3001, Belgium. E-mail: cheng.zhou@kuleuven.be; bert.sels@kuleuven.be
bUniversité catholique de Louvain (UCLouvain), Institute of Condensed Matter and Nanosciences (IMCN), Place Louis Pasteur, 1, Louvain-la-Neuve 1348, Belgium
cTotalEnergies OneTech Belgium, Zone industrielle C, Feluy 7181, Belgium
First published on 19th March 2026
Abstract
Bulk ZrOxHy is an effective catalyst for amine regeneration in CO2 absorption, but its high cost and poor support stability may hinder application. Here, ZrOxHy is deposited as a theoratical monolayer within MCM-41 mesopores, achieving similar stable catalytic activity to bulk ZrOxHy with just 13 wt% Zr loading, offering a cost-effective path toward industrial use.
Post-combustion CO2 capture using aqueous amine solvents is widely regarded as the most practical short-term strategy for large-scale mitigation of greenhouse gas emissions from existing power plants and industrial facilities.1,2 Despite its maturity, conventional thermal regeneration of amine solutions remains highly energy-intensive.3 This is mainly due to the slow kinetics of CO2 desorption at moderate temperatures, while amine degradation at elevated temperatures further increases operating costs.4–6 To address this bottleneck, catalytic amine regeneration has emerged as an approach, offering the potential to accelerate desorption kinetics while reducing the energy consumption.7–14
Among the catalysts reported to date, zirconium oxo-hydroxide (ZrOxHy) materials have demonstrated very recently outstanding activity for promoting amine regeneration, with excellent performance confirmed under both batch and continuous reactor configurations.15,16 Despite their promise, the direct application of bulk ZrOxHy in industrial settings is hindered by the need for supported catalysts with stronger mechanical strength and the relatively high cost of zirconium compared to silicon.17,18 To date, achieving a uniform and accessible dispersion of ZrOxHy on a support, without compromising catalytic performance, has remained uncertain.
In this work, we demonstrate a cost-effective strategy to harness the exceptional catalytic properties of ZrOxHy through its controlled deposition within the mesoporous framework of MCM-41 (Zr-MCM-41). Remarkably, with only 13 wt% zirconium loading, the resulting supported catalyst delivers activity comparable to bulk ZrOxHy per total catalyst mass. This high efficiency is attributed to the uniform high dispersion of active ZrOxHy species on the mesopore walls, maintaining full accessibility of the porous network. Importantly, no detrimental interaction between MCM-41 and ZrOxHy was detected, highlighting the structural and mechanistic fidelity of the active phase and its promise for scalable amine regeneration catalysis.
The original bulk ZrOxHy catalyst was synthesized via a sol–gel method,15 during which the wet gel underwent more than a 80% volume reduction upon drying. Photographs depicting the shrinkage of the ZrOxHy gel before and after drying are shown in Fig. S1. This substantial contraction facilitates the deposition of the material within a porous SiO2 support without blocking its inherent pore network. While MCM-41 is a commercialized SiO2 support with ordered straight meso-pores. MCM-41, a well-established commercial mesoporous silica featuring a highly ordered channel structure, was chosen as the support material.
As illustrated in Fig. 1a, MCM-41 powder was mixed with a ZrO(NO3)2 precursor solution. The abundant surface Si–OH groups of MCM-41 provide anchoring sites for the Zr hydrogel at the gel–support interface. During drying at 100 °C, condensation of the wet gel induces significant volume contraction. Within the 2.5 nm mesopores, however, spatial confinement and capillary forces, together with strong interfacial Si–O(H)–Zr bonding, stabilize the gel–wall interface and prevent detachment or particle segregation. As a result, shrinkage occurs primarily along the pore walls, producing a uniform, conformal ZrOxHy coating. This is proved in Fig. 1b, N2 adsorption–desorption isotherms of pristine and ZrOxHy-coated MCM-41 retain the characteristic Type-IV profile, indicating that mesoporosity and pore ordering are largely preserved. The coated sample shows reduced adsorption volume and surface area (Table S1), reflecting partial thickening of the pore walls by ZrOxHy. Additionally, the coated sample displays a reduced high-pressure uptake, suggesting a decrease in interparticle voids and implying the presence of ZrOxHy on the external particle surfaces. Pore size distributions (Fig. 1c) show a narrow peak at 2.8 nm for MCM-41, which shifts slightly to 2.5 nm after ZrOxHy coating. The ∼0.3 nm wall thickness increase is consistent with the theoretical monolayer thickness of ZrOxHy, suggesting a near-monolayer coverage. The mesopore volume decreases from 0.77 to 0.54 cm3 g−1, corresponding to ∼28 wt% Zr (assuming all volume loss is due to ZrOxHy with a density of 3.25 g cm−3), in close agreement with ICP measurements (32 wt%), confirming that most ZrOxHy resides within the mesoporous channels.
Transmission electron microscopy (TEM) images of Zr-MCM-41(32 wt%) (Fig. 1d and Fig. S2) reveal well-defined mesoporous channels without distinct ZrOxHy particles, indicating a uniform, conformal in-pore coating. Energy Dispersive X-ray Spectroscopy (EDX-TEM) (Fig. 1f and g) confirms homogeneous Zr distribution throughout the MCM-41 particles except for the external surface, where an oligomerized from ZrOxHy particles and thicker ZrOxHy layer are witness (Fig. S3), and reflecting unrestricted gel growth outside the spatially confined mesopores. The high Zr content on the external surface is further quantified by X-ray photoelectron spectroscopy (XPS), which gives a Zr/Si ratio of 0.29, corresponding to ∼43 wt% Zr, which is higher than the bulk value. However, considering the small external surface area of MCM-41, this external Zr accounts for only ∼4.8% of the total Zr, meaning that over 95% of Zr remains well confined within the mesoporous channels without blocking the pores.
The catalytic activity of Zr-MCM-41 (32 wt%) for CO2 desorption was evaluated in a batch reactor (Fig. S4) using 30 wt% CO2-rich monoethanolamine (MEA), the standard industrial solvent. The mixture was heated from 25 °C to 88 °C and held isothermally for 3 hours. For comparison, desorption was also tested with bulk ZrOxHy, pristine MCM-41, and without any catalyst (blank).
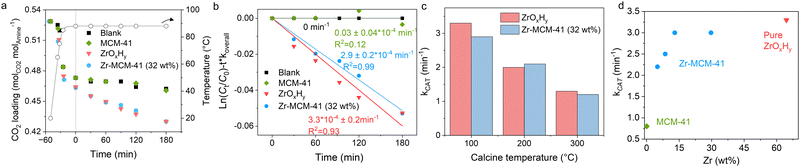
Fig. 2a shows the CO2 loading (molCO2 molamine−1) over time. Bulk ZrOxHy markedly accelerates desorption during both heating and isothermal stages, while MCM-41 shows negligible effect. Zr-MCM-41 (32 wt%) achieves nearly the same enhancement as bulk ZrOxHy. The isothermal catalytic rate constant (kCAT), calculated assuming first-order kinetics19 after subtracting the thermal background (Fig. 2b), is 2.9 × 10−4 min−1 for Zr-MCM-41, comparable to bulk ZrOxHy (3.3 × 10−4 min−1). MCM-41 itself exhibits a minimal kCAT of 0.02 × 10−4 min−1, confirming that the activity originates exclusively from the ZrOxHy coating. The overall kinetic (with thermal background) is shown in the Fig. S5, with good linearity.
ZrOxHy is known to be sensitive to thermal treatment, as dehydration reduces the surface hydroxyl groups responsible for its catalytic activity. In this study, Zr-MCM-41 (32 wt%) exhibited a similar trend of activity loss with increasing calcination temperature as bulk ZrOxHy (Fig. 2c), though the decline was noticeably less pronounced. After calcination at 200 °C, Zr-MCM-41(32 wt%) retained 72% of its initial activity, compared to 60% for bulk ZrOxHy; at 300 °C, the retained activity was 41% versus 39%, respectively. These results indicate that while some loss of active surface hydroxyls during calcination is unavoidable, supporting ZrOxHy on mesoporous silica such as MCM-41 can substantially mitigate deactivation, preserving a greater fraction of the catalytic performance.
Zr with different loading was carried out, as shown in Fig. 2d, although increasing Zr loading initially enhances the desorption rate, further increases beyond 13 wt% do not result in proportional kCAT enhancement. The Weisz–Prater criterion was applied to Zr-MCM-41 (32 wt%), yielding CWP ≪ 1 (see SI for detailed calculations), ruling out intraparticle diffusion limitations. The observed rate plateau therefore likely reflects reduced Zr utilization at higher loadings, suggesting that more dispersed Zr may be preferred. Since ZrOxHy was mostly coated on the pore wall for 32 wt% loading, the decrease on the activity should not relate to aggregation of the Zr. TEM images (Fig. S6) reveal well-defined mesoporous channels without discernible ZrOxHy nanoparticles, indicating the absence of segregated oxide domains. EDX mapping of Zr-MCM-41 (13 wt%) (Fig. S7) shows a dilute yet homogeneous Zr distribution that closely follows the silica framework, in contrast to the higher local intensity observed at 32 wt% loading. As calculated in previous, a full monolayer ZrOxHy coating on MCM-41 theoretically requires at least 28 wt% Zr, suggesting that at lower loadings a significant fraction of the SiO2 surface remains exposed. It is further supported by additional experiments using conventional amorphous SiO2 with lower surface area (254 m2 g−1), where Zr–SiO2 exhibits a much lower kCAT of 2.3 × 10−4 min−1, at similar Zr loading (13 wt%).
To further clarify the nature of the active sites in the supported catalysts, in situ diffuse reflectance infrared Fourier transform spectrometry (DRIFTS) measurements were performed on MCM-41 and Zr-MCM-41 samples with Zr loadings of 13 wt% and 32 wt% at different temperatures under N2 (Fig. 3a–c). The sharp band at ∼3740 cm−1, attributed to isolated Si–OH groups, progressively decreases with increasing Zr loading, while a new band near ∼3550 cm−1 and a broad feature centered around ∼3400 cm−1 emerge and intensify. These lower-frequency and broader bands indicate the formation of Zr-related hydroxyl species with stronger hydrogen-bonding interactions, which are absent in pristine MCM-41. Notably, no new hydroxyl species are observed when comparing the 13 wt% and 32 wt% Zr-loaded samples; the primary difference lies in the relative abundance of Zr-OH and Si-OH groups rather than their nature.
 | ||
| Fig. 3 In situ DRIFS under different temperature (a) MCM-41, (b) Zr-MCM-41 (13 wt%) (c) Zr-MCM-41 (32 wt%). (d) Zr mass normalized kCAT (min−1 g−1) versus Zr loading amount. (e) Zr-MCM-41 (13 wt%) catalyst regeneration performance. Reaction conditions: 150 mL CO2-saturated 30 wt% MEA, 3 g catalyst, N2 flow of 0.5 L min−1 from the top of the condenser, 88 °C. (f) CO2 desorption rate from 30 wt% MEA for a 16 ± 2% regeneration ratio in the continuous reactor set-up with shaped Zr-MCM-41 (13 wt%). Reaction condition: 88 °C, CO2 partial pressure of 25 ± 5 kPa as determined by MS, 6 g catalyst, particle diameter of 0.25 to 0.5 mm. The glass beads with similar sizes of catalyst are used in the blank to exclude the influence of bed porosity. The CO2 desorption rate of ZrOxHy and glass beads data was taken from reported data under same condition.15 | ||
When normalized by Zr loading mass, as shown in Fig. 3d, the Zr-specific activity decreases roughly logarithmically with increasing Zr content. This trend suggests that higher surface density reduces the effective turnover per Zr site, which can be up to 8.6 times higher than that of pure ZrOxHy. Additionally, the support effect may also impact the activity of individual Zr sites, specifically the influence of exposed Si–OH groups on Zr, as reported in previous studies,20 though further investigation is needed to fully clarify this effect.
Catalyst recyclability is a key factor in evaluating performance. For Zr-MCM-41 (13 wt%), the catalyst can be regenerated through both water-wash recycling and direct reuse (detailed in Fig. S8) without significant loss of activity (Fig. 3e).
The catalyst was evaluated in a continuous fixed-bed reactor for 5 h without noticeable deactivation, exhibiting performance comparable to that of pure ZrOxHy (Fig. 3f). During this period, the activity of Zr-MCM-41 (13 wt%) remained essentially constant, indicating stable intrinsic active sites under the applied desorption conditions. Prolonged operation, however, led to deterioration of the packed bed, as the shaped catalyst bodies disintegrated into fine particles and were washed out by the liquid flow. This behavior indicates that the observed stability limitation arises primarily from insufficient mechanical strength and bed integrity under high gas–liquid flux, rather than from chemical deactivation of the ZrOxHy active phase. Future work should therefore focus on improving catalyst shaping and mechanical robustness for long-term operation.
This study demonstrates the successful development of a high-performance ZrOxHy catalyst uniformly coated onto the mesopore walls of MCM-41. The resulting Zr-MCM-41 combines exceptional catalytic activity, comparable to bulk ZrOxHy, with a remarkably low Zr loading of just 13 wt%. While calcination inevitably reduces activity, the supported catalyst exhibits less deactivation than the unsupported counterpart, highlighting the stabilizing effect of the mesoporous support. Together, these results provide compelling evidence that Zr-MCM-41 is a cost-efficient, robust, and scalable catalyst, offering significant promise for practical implementation in industrial CO2 amine-regeneration processes. Future works may focusing on high mechanical strengthen shaped catalyst and investigation of lower-cost, high-surface-area commercial supports, such as activated carbon.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included in the main text and supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5cc06632h.Acknowledgements
C. Z. acknowledge Walter Vermandel for technical supports. B. F. S. acknowledges TotalEnergies for financial support. M. T. B. and M. D. thank the European Research Council (ERC) for funding: ERC Starting Grant 948449 named Z-EURECA, i.e., Zeolite synthesis in Unusual Reactors for Enhanced Catalysts to MD. The TEM-EDS in this research was supported by the FWO infrastructure projects (AKUL13/19 and I000920N). The N2 physisorption research was supported by KUL internal funding, KA/16/064. The DRIFTS was supported by FWO, G0A0321N.References
- S. Chu, Science, 2009, 325, 1599 CrossRef CAS PubMed.
- IEA, Energy Technology Perspectives 2020, https://www.iea.org/reports/energy-technology-perspectives-2020.
- C. Zou, Q. Zhao, G. Zhang and B. Xiong, Nat. Gas Ind. B, 2016, 3, 1–11 CrossRef.
- F. Raganati and P. Ammendola, Energy Fuels, 2024, 38, 13858–13905 CrossRef CAS.
- F. de Meyer and C. Bignaud, Chem. Eng. J., 2022, 428, 131264 CrossRef CAS.
- S. Nakao, K. Yogo, K. Goto, T. Kai and H. Yamada, Advanced CO2 capture technologies: Absorption, adsorption, and membrane separation methods, 2019 Search PubMed.
- X. Li, L. Xing, Z. Chen, Y. Wang, C. Wang, G. Zhan, H. Chang and J. Li, Adv. Funct. Mater., 2025, 2414293, 1–10 Search PubMed.
- X. Zhong, W. Kong, K. Yang, T. Song, Z. Dong, L. Liang, S. Zhang, W. Li and S. Li, Adv. Funct. Mater., 2025, 2422336, 1–16 Search PubMed.
- Y. Li, L. Xing, Z. Chen, G. Zhan, B. Yuan, Y. Peng, L. Wang and J. Li, Appl. Catal., B, 2024, 358, 124350 CrossRef CAS.
- Q. Sun, H. Gao, M. Xiao, T. Sema and Z. Liang, Environ. Sci. Technol., 2024, 58, 10052–10059 CrossRef CAS PubMed.
- C. Zhou, J. Shi, W. Zhou, K. Cheng, Q. Zhang, J. Kang and Y. Wang, ACS Catal., 2019, 10, 302–310 CrossRef.
- C. Zhou, I. Khalil, F. Rammal, M. Dusselier, P. Kumar, M. Lacroix, E. Makshina, Y. Liao and B. F. Sels, ACS Catal., 2022, 12, 11485–11493 CrossRef CAS.
- M. S. Alivand, O. Mazaheri, Y. Wu, A. Zavabeti, A. J. Christofferson, N. Meftahi, S. P. Russo, G. W. Stevens, C. A. Scholes and K. A. Mumford, Nat. Commun., 2022, 13, 1–11 Search PubMed.
- Q. Lai, S. Toan, M. A. Assiri, H. Cheng, A. G. Russell, H. Adidharma, M. Radosz and M. Fan, Nat. Commun., 2018, 9, 1–7 CAS.
- C. Zhou, M. T. Beydokhti, F. Rammal, P. Kumar, M. Lacroix, W. Vermeiren, M. Dusselier, Y. Liao and B. F. Sels, Nat. Catal., 2025, 8, 270–281 CrossRef CAS.
- L. Ji, J. Li, R. Zhai, J. Wang, X. Wang, S. Yan and M. Hua, ACS Omega, 2022, 7, 44620–44630 CrossRef CAS PubMed.
- R. J. Farrauto, Handbook of Industrial Chemistry and Biotechnology, 2017, pp. 1995–2035 Search PubMed.
- D. Leybo, U. J. Etim, M. Monai, S. R. Bare, Z. Zhong and C. Vogt, Chem. Soc. Rev., 2024, 53, 10450–10490 RSC.
- P. V. Danckwerts, Chem. Eng. Sci., 1979, 34, 443–446 CrossRef CAS.
- X. Zhang, Y. Huang, J. Yang, H. Gao, Y. Huang, X. Luo, Z. Liang and P. Tontiwachwuthikul, Chem. Eng. J., 2020, 383, 123077 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |