Synthesis of pure-red-emission perovskite quantum dots with high color purity by A-site cation exchange with lecithin at room temperature
Kenshin
Yoshida
 a,
Taisei
Kimura
a,
Taisei
Kimura
 a,
Takuro
Iizuka
a,
Takashi
Nagata
a,
Shun
Hirashima
a,
Yuka
Horie
b,
Satoshi
Asakura
c,
Motofumi
Kashiwagi
d and
Akito
Masuhara
a,
Takuro
Iizuka
a,
Takashi
Nagata
a,
Shun
Hirashima
a,
Yuka
Horie
b,
Satoshi
Asakura
c,
Motofumi
Kashiwagi
d and
Akito
Masuhara
 *ae
*ae
aGraduate School of Science and Engineering, Yamagata University, 4-3-16 Jonan, Yonezawa, Yamagata, 992-8510, Japan
bFaculty of Engineering, Yamagata University, 4-3-16 Jonan, Yonezawa, Yamagata 992-8510, Japan
cISE CHEMICALS Corporation, 1-3-1 Kyobashi, Chuo-ku, Tokyo 104-0031, Japan
dZEON Corporation, Marunouchi, Chiyoda-ku, Tokyo 100-0005, Japan
eFrontier Center for Organic Materials, Yamagata University, 4-3-16 Jonan, Yonezawa, Yamagata 992-8510, Japan
First published on 30th January 2026
Abstract
The achievement of a full-width at half-maximum (FWHM) below 30 nm in pure-red PeQDs synthesized at room temperature, which is essential for ultra-high-definition television applications, has rarely been reported. Herein, we report the narrowest FWHM of 27 nm reported to date at room temperature, enabled by a stable A-site cation exchange employing lecithin.
Lead halide perovskite quantum dots1 (PeQDs, APbX3; A = methylammonium: CH3NH3+ (MA), formamidinium: CH(NH2)2+ (FA), Cs+; X = Cl−, Br−, I−) are promising candidates as phosphors for ultra-high-definition-television (UHDTV) applications due to their excellent optical properties, including narrow full width at half maximum (FWHM < 30 nm),2,3 high photoluminescence quantum yield (PLQY ≈ 100%),3–5 and wide color tunability (λem: 450–730 nm).6 In addition to perovskite-based emitters, various narrow-band red phosphors, particularly lanthanide-based materials such as Eu3+- and Mn4+-activated compounds, have been extensively investigated for display applications owing to their sharp emission lines and high color purity. However, these phosphors generally suffer from low absorption cross-sections arising from parity-forbidden f–f transitions, which limit their excitation efficiency under practical display backlight conditions. Furthermore, the emission wavelength tunability of lanthanide-based phosphors is inherently restricted by their fixed electronic transitions, making fine color adjustment challenging.7–9 In contrast, PeQDs offer high absorption coefficients, near-unity PLQYs, and versatile wavelength tunability through compositional and structural control, rendering them particularly attractive as solution-processable narrow-band red emitters for next-generation UHDTV displays. At room temperature, PeQDs were mainly synthesized using the ligand-assisted reprecipitation (LARP) method.10,11 LARP is a synthesis method that utilizes the solubility differences of PeQD precursors in polar and nonpolar solvents. However, polar solvents destabilize the ionic crystal structures of PeQDs.10 This issue is particularly critical for red-emission PeQDs, which inherently exhibit low structural stability owing to their unfavorable tolerance factors.12–14 The successful synthesis of high-performance red-emission PeQDs via the LARP method is infrequently reported. Polar solvents are known to accelerate Ostwald ripening,15 resulting in inhomogeneous crystal growth and poor crystallinity of the crystals. To date, PeQDs with pure-red emission (λem: 620–650 nm) and narrow FWHM, which meet UHDTV standards, have not been achieved via LARP. In contrast, the polar solvent-free room temperature (PFRT)4 synthesis method enables the dissolution of perovskite precursors in non-polar solvents by forming complexes with ligands. PeQDs are synthesized through ion-exchange reactions between these complex solutions, allowing for synthesis at room temperature while avoiding the use of polar solvents. This approach facilitates the formation of PeQDs with a relatively narrow FWHM.16,17 However, precise control over excessive ligand quantities is more challenging in the PFRT method because of the pre-formation of complexes, making emission wavelength tuning via the quantum size effect difficult. Previous reports on red-emission PeQDs synthesized via the PFRT method have targeted electroluminescence applications, with emission wavelengths of 599 and 710 nm, neither of which falls within the pure-red range.16,17 In contrast, prior efforts to tune the emission wavelength within the pure-red region have focused on halide composition tuning, which is independent of quantum confinement. However, halide-mixed PeQDs exhibit emission wavelength instability under light or electrical biases.18,19
Herein, we propose a simple strategy for fabricating pure-red-emission PeQDs by tuning the emission wavelength through A-site cation exchange20 of red-emission PeQDs synthesized by the PFRT method. Furthermore, the introduction of lecithin during the A-site cation exchange enabled precise emission wavelength control while preserving the PL spectral shape and optical properties. Ultimately, we successfully fabricated high-performance pure-red PeQDs (λPL = 625 nm, FWHM = 27 nm) via a simple room-temperature process.
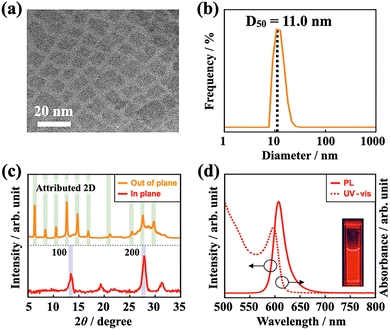
PeQDs were synthesized by the PFRT method employing ion-exchange reactions of complexes based on the HSAB principle. Transmission electron microscopy (TEM) analyses of the synthesized PeQDs demonstrated low contrast in the TEM images (Fig. 1a), suggesting inhibited crystal growth and the formation of plate-like nanocrystals with minimal thickness. Dynamic light scattering (DLS) measurements confirmed monodispersity, with a median particle size (D50) of 11.0 nm (Fig. 1b). The out-of-plane X-ray diffraction (XRD) patterns exhibited periodic low-angle diffraction peaks, indicating the face-on stacking of two-dimensional (2D) nanoplatelets.21 While in-plane XRD patterns did not exhibit peaks corresponding to the nanoplatelets, distinct diffraction from the PeQD was observed, corroborating the plate-like structure (Fig. 1c).
The UV-vis absorption and photoluminescence (PL) of the as-synthesized PeQDs demonstrated distinct absorption peaks attributable to band edge discretization, indicative of strong quantum confinement.22 The PL spectrum exhibited a narrow FWHM of 25 nm and a high PLQY of 87% (Fig. 1d). However, the emission wavelength exhibited a significant blue shift to 607 nm, positioning it outside the pure-red range (λem: 620–650 nm). This observation suggests the necessity for an A-site cation exchange.
In the following section, we elucidate the mechanism and procedure of A-site cation exchange. Formamidinium acetate (FAAc) was employed as the source of formamidinium (FA) for the A-site cation exchange. FAAc powder was introduced into the PeQDs dispersion to initiate the exchange process. During A-site cation exchange, oleic acid was incorporated to form oleic acid–FA complexes, facilitating a gradual ion-exchange reaction through the formation of these complexes. In the lecithin-containing system, lecithin was added immediately prior to the injection of oleic acid. Herein, a slower exchange process was employed to mitigate the abrupt formation of lattice strain due to rapid A-site cation exchange.
The presence of lecithin in the PeQDs after A-site cation exchange was confirmed by 31P-NMR spectroscopy (Fig. S1). The detection of phosphorus signals attributable to lecithin, even after multiple washes to remove excess ligands, clearly indicates that lecithin remained coordinated to the PeQDs.
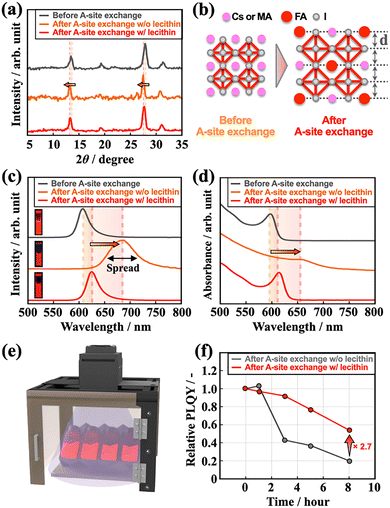
In both the w/o and w/lecithin systems, X-ray photoelectron spectroscopy (XPS) analysis of Pb and I before and after the A-site cation exchange indicated a shift in the binding energy to lower values (Fig. S2a–d), which is consistent with the incorporation of FA cations with larger ionic radii rather than Cs+ or MA+. The in-plane XRD analysis showed that the (100) and (200) diffraction peaks shifted toward lower angles from their initial positions in both the w/o and w/lecithin systems (Fig. 2a). These results indicate MA or Cs to FA exchange. This observation suggests lattice expansion due to the presence of a larger A-site cation (Fig. 2b), in accordance with Bragg's law. These findings confirm the successful integration of cations into the lattice. Furthermore, in the w/o lecithin system, the shift width suggested that more FA could be incorporated.
In the w/o lecithin system, the PL spectra observed after the A-site cation exchange exhibited a shift in the emission wavelength, accompanied by a broadening of the FWHM and a reduction in the PLQY (Fig. 2c). The UV-vis spectra indicated the disappearance of sharp quantum confinement features following the exchange, suggesting coarsening of the crystal structure (Fig. 2d). In contrast, PL measurements of lecithin-assisted A-site cation exchange confirmed the successful modulation of the emission wavelength from 607 to 625 nm, within the pure-red range, while maintaining a high PLQY of 87% and a narrow FWHM of 27 nm (Fig. 2c), which is the narrowest in the pure-red region reported to date (Fig. S3).23–29 UV-vis spectra indicated that the characteristic absorption peaks were preserved with lecithin, in contrast to the loss observed in its absence (Fig. 2d). These contrasting behaviors suggest that lecithin plays a dual role in stabilizing PeQDs during the A-site cation exchange process by both suppressing crystal distortion and moderating the exchange kinetics. Surface ligands are critical for governing A-site cation exchange in PeQDs, and previous studies have shown that the exchange kinetics strongly depend on the protonation state of the surface A-site cations.20,30 In this regard, lecithin, which can form strong interactions with surface A-site cations and iodide ions in PeQDs,25 is expected to inhibit protonation of the A-site. Therefore, such surface stabilization is likely to retard the kinetics of A-site cation exchange, preventing abrupt structural rearrangements. Consequently, the combined suppression of crystal distortion and inhibition of rapid A-site cation exchange enables stable and controlled A-site substitution, thereby preserving the optical quality of PeQDs. To evaluate the photostability of the PeQDs after A-site cation exchange, a light irradiation test was performed under blue-light irradiation (λem = 450 nm), simulating a display backlight with an intensity of 30 mW cm−2 (Fig. 2e). These results clearly demonstrate the superior stability of the lecithin-assisted A-site cation exchange system compared to that of the w/o lecithin counterpart (Fig. 2f).
In both the w/o and w/lecithin systems, SEM and energy-dispersive X-ray spectroscopy (EDX) mapping of the samples, both before and after A-site cation exchange, revealed a uniform distribution of Pb and I, with a molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.1, which is slightly above the theoretical ratio of 1
3.1, which is slightly above the theoretical ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.0 (Fig. S4a–d). This indicates an iodine-rich composition and minimal iodide loss, suggesting that structural defects did not form during the exchange process. Consequently, the reduction in PLQY is primarily attributed to grain coarsening.
3.0 (Fig. S4a–d). This indicates an iodine-rich composition and minimal iodide loss, suggesting that structural defects did not form during the exchange process. Consequently, the reduction in PLQY is primarily attributed to grain coarsening.
Fig. S5a and b present the TEM images of PeQDs after A-site cation exchange w/o and w/lecithin. TEM analysis indicated that after A-site cation exchange w/o lecithin, the PeQDs exhibited high contrast compared to before A-site cation exchange. However, the TEM images following lecithin-assisted exchange demonstrated no notable increase in crystal size, suggesting a stable exchange. Fig. S6 presents the DLS measurement results of the PeQDs after the A-site cation exchange. The PeQD particle size after A-site cation exchange w/o lecithin increased significantly to > 50 nm. DLS measurements consistently revealed a significant increase in particle size, with a D50 value of 86.9 nm. These findings suggest that the crystal structure may have partially collapsed during the A-site cation exchange, leading to non-uniform crystal growth through Ostwald ripening. The consequent enlargement in crystal size and size distribution likely contributed to the broadening of the FWHM, as well as to the reduction of exciton binding energy and the enhancement of non-radiative recombination. Given these challenges, stabilizing the surface of PeQDs with lecithin is essential to prevent phase transitions induced by A-site cation exchange. DLS analysis of lecithin-assisted A-site cation exchange demonstrated no notable increase in crystal size, suggesting a stable exchange process and a moderate size distribution of D50 = 19.0 nm, which aligns with A-site cation exchange (Fig. S6). These results confirm that lecithin plays a crucial role in achieving a stable A-site cation exchange. Furthermore, the lattice plane spacings were estimated from fast Fourier transform (FFT) patterns obtained from the TEM images. The (100) lattice spacing of the PeQDs prepared without lecithin was determined to be 0.644 nm (Fig. S5c), whereas a larger spacing of 0.661 nm was observed for the lecithin-assisted system (Fig. S5d). These results suggest that the lecithin-containing system likely incorporates a larger amount of FA with a larger ionic radius, indicating an enhanced tolerance of the lattice to the distortion induced by FA incorporation. These differences in structural tolerance are likely responsible for the observed variations in the aggregation behavior and crystal morphology following A-site exchange.
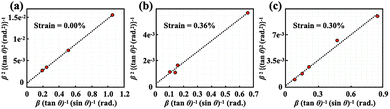
XRD is a well-established technique for elucidating crystal structures. The broadening of diffraction peaks in XRD patterns is primarily attributed to crystallite size effects and lattice strain. Consequently, the quantitative analysis of the peak broadening facilitates the estimation of lattice strain. In this study, the lattice strain of the PeQDs was quantitatively evaluated using the Halder–Wagner method,31 which enables the deconvolution of size- and strain-induced broadening. This approach estimates the integral breadth of the diffraction peaks by employing a Voigt function, which is a convolution of the Gaussian and Lorentzian profiles, to account for both contributions. The XRD data were fitted using eqn (1),
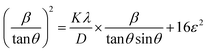 | (1) |
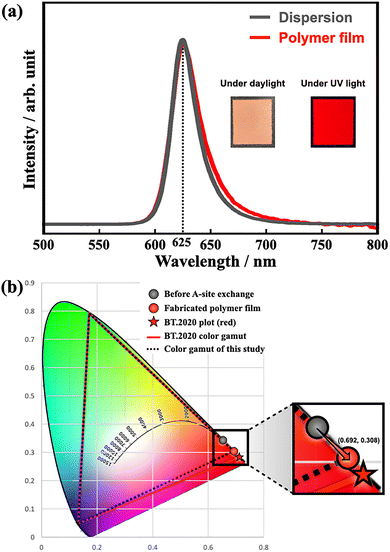
Finally, a polymer film suitable for display applications was fabricated using PeQDs subjected to lecithin-assisted A-site cation exchange reaction. The polymer film almost completely preserved the shapes of both the photoluminescence and absorption spectra observed in dispersion (Fig. 4a). When these data were plotted on the CIE 1931 chromaticity diagram, the chromaticity coordinates of the dispersion and film were perfectly aligned. Furthermore, the color gamut constructed by connecting the chromaticity coordinates of the PeQDs with the ideal green and blue coordinates defined by the BT.2020 standard exhibited excellent coverage, reaching 96.9% of the BT.2020 color space (Fig. 4b).
In summary, this study demonstrates that precise control of subtle lattice distortions significantly enhances the optical properties of A-site cation exchange. A facile room-temperature strategy has been developed for red-emission PeQDs, achieving the highest reported color purity. This approach demonstrates that incorporating lecithin during A-site cation exchange effectively suppresses lattice distortion, thereby facilitating a stable and controlled exchange process. To the best of our knowledge, the results obtained in this study represent the narrowest FWHM reported for pure-red emission PeQDs in a dispersion state.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article are included in the supplementary information (SI). Supplementary information: materials and methods, synthesis of PeQDs, fabrication of film, 31P-NMR spectroscopy, XPS spectra, Comparison of FWHM with previous works, SEM-EDX mapping, TEM images and FFT patterns and DLS spectrum. See DOI: https://doi.org/10.1039/d5cc06683b.Acknowledgements
This research was supported by the Adaptable and Seamless Technology Transfer Program through Target-driven R&D (A-STEP) from the Japan Science and Technology Agency (JST), Grant Number JPMJTR223B, Japan Society for Promotion of Science (JSPS) Research Fellowship for Young Scientists (DC1), the JSPS Program for Promoting Japan's Peak Research Universities (J-PEAKS) Grant Number JPJS00420240014, and Scholarship of Fuji Seal Foundation.References
- J. Shamsi, A. S. Urban, M. Imran, L. De Trizio and L. Manna, Chem. Rev., 2019, 119, 3296–3348 CrossRef CAS PubMed.
- Y. Morikawa, R. Sato, N. Oshita, T. Kimura, K. Yoshida, M. Goto, S. Asakura, M. Kashiwagi and A. Masuhara, Appl. Phys. Express, 2023, 16, 055001 Search PubMed.
- A. Liu, C. Bi, J. Li, M. Zhang, C. Cheng, D. Binks and J. Tian, Nano Lett., 2023, 23, 2405–2411 CrossRef CAS PubMed.
- K. Yoshida, N. Oshita, T. Kimura, M. Goto, R. Oikawa, M. Uwano, S. Asakura, T. Chiba and A. Masuhara, Appl. Phys. Express, 2024, 17, 025002 CrossRef CAS.
- S. W. Dai, B. W. Hsu, C. Y. Chen, C. A. Lee, H. Y. Liu, H. F. Wang, Y. C. Huang, T. L. Wu, A. Manikandan, R. M. Ho, C. S. Tsao, C. H. Cheng, Y. L. Chueh and H. W. Lin, Adv. Mater., 2018, 30, 1705532 CrossRef PubMed.
- I. Levchuk, P. Herre, M. Brandl, A. Osvet, R. Hock, W. Peukert, P. Schweizer, E. Spiecker, M. Batentschuk and C. J. Brabec, Chem. Commun., 2016, 53, 244–247 RSC.
- K. Singh, P. Pradhan, S. Priya, S. Mund and S. Vaidyanathan, Dalton Trans., 2023, 52, 13027–13057 RSC.
- B. Rajamouli, P. Sood, S. Giri, V. Krishnan and V. Sivakumar, Eur. J. Inorg. Chem., 2016, 3900–3911 CrossRef CAS.
- M. M. Rodríguez-García, A. Ciric, Z. Ristic, J. A. G. Williams, M. D. Dramićanin and I. R. Evans, J. Mater. Chem. C, 2021, 9, 7474–7484 RSC.
- F. Zhang, S. Huang, P. Wang, X. Chen, S. Zhao, Y. Dong and H. Zhong, Chem. Mater., 2017, 29, 3793–3799 CrossRef CAS.
- H. Z. F. Zhang, C. Chen, X.-G. Wu, X. Hu, H. Huang, J. Han, B. Zou and Y. Dong, ACS Nano, 2015, 9, 4533–4542 CrossRef PubMed.
- C. H. Lu, G. V. Biesold-McGee, Y. Liu, Z. Kang and Z. Lin, Chem. Soc. Rev., 2020, 49, 4953–5007 RSC.
- M. Ma, X. Zhang, L. Xu, X. Chen, L. Wang, T. Cheng, F. Wei, J. Yuan and B. Shen, Adv. Mater., 2023, 35, e2300653 CrossRef PubMed.
- Y. Tang, A. Lesage and P. Schall, J. Mater. Chem. C, 2020, 8, 17139–17156 RSC.
- J. T. DuBose, A. Christy, J. Chakkamalayath and P. V. Kamat, ACS Mater. Lett., 2021, 4, 93–101 CrossRef.
- J. Hu, C. Bi, X. Zhang, Y. Lu, W. Zhou, Z. Zheng, Y. Tang, F. Lu, Z. Yao, B. Tian, X. Wang, K. Selvakumar, J. Tian and M. Sui, Appl. Mater. Today, 2022, 29, 101592 CrossRef.
- T. Wang, X. Li, T. Fang, S. Wang and J. Song, Chem. Eng. J., 2021, 418, 129361 CrossRef CAS.
- H. Zhang, X. Fu, Y. Tang, H. Wang, C. Zhang, W. W. Yu, X. Wang, Y. Zhang and M. Xiao, Nat. Commun., 2019, 10, 1088 CrossRef CAS PubMed.
- P. V. Kamat and M. Kuno, Acc. Chem. Res., 2021, 54, 520–531 CrossRef CAS PubMed.
- C. Otero-Martinez, M. Imran, N. J. Schrenker, J. Ye, K. Ji, A. Rao, S. D. Stranks, R. L. Z. Hoye, S. Bals, L. Manna, J. Perez-Juste and L. Polavarapu, Angew. Chem., Int. Ed., 2022, 61, e202205617 CrossRef CAS PubMed.
- C. J. Krajewska, A. E. K. Kaplan, M. Kick, D. B. Berkinsky, H. Zhu, T. Sverko, T. Van Voorhis and M. G. Bawendi, Nano Lett., 2023, 23, 2148–2157 Search PubMed.
- J. Ye, D. Gaur, C. Mi, Z. Chen, I. L. Fernandez, H. Zhao, Y. Dong, L. Polavarapu and R. L. Z. Hoye, Chem. Soc. Rev., 2024, 53, 8095–8122 Search PubMed.
- Y. Li, M. Deng, X. Zhang, T. Xu, X. Wang, Z. Yao, Q. Wang, L. Qian and C. Xiang, Nat. Commun., 2024, 15, 5696 Search PubMed.
- Y. Li, J. Qian, D. Zhao and R. Song, RSC Adv., 2021, 11, 18432–18439 RSC.
- W. J. Mir, A. Alamoudi, J. Yin, K. E. Yorov, P. Maity, R. Naphade, B. Shao, J. Wang, M. N. Lintangpradipto, S. Nematulloev, A. H. Emwas, A. Genovese, O. F. Mohammed and O. M. Bakr, J. Am. Chem. Soc., 2022, 144, 13302–13310 CrossRef CAS PubMed.
- Y. F. Lan, J. S. Yao, J. N. Yang, Y. H. Song, X. C. Ru, Q. Zhang, L. Z. Feng, T. Chen, K. H. Song and H. B. Yao, Nano Lett., 2021, 21, 8756–8763 CrossRef CAS.
- X. Ren, H.-L. Hu, Z.-Y. Chen, L.-X. Gao, H. Hao, Y. Liu and F.-L. Jiang, ACS Appl. Nano Mater., 2023, 6, 6092–6102 CrossRef CAS.
- H. M. Lai, Z. Lu, C. K. K. Choi, W. Zhou, C. Ngo Yau, B. Z. Tang and H. Ko, ACS Appl. Nano Mater., 2022, 5, 12366–12373 CrossRef CAS.
- Y. Lou, H. Wang, Z. Zhou, M. Zeng, Z. Yan, X. Yang, P. Fu, A. Huang, D. Li, T. Wu, Y. Bai and H. M. Cheng, ACS Nano, 2025, 19, 23151–23161 CrossRef CAS PubMed.
- C. Zhao, J. Shi, H. Huang, Q. Zhao, X. Zhang and J. Yuan, Small Sci., 2024, 4, 2300132 Search PubMed.
- T. Kimura, K. Yoshida, K. Narazaki, K. Yanagihashi, S. Hirashima, Y. Oyama, K. S. Thakuri, Y. Ito, S. Asakura, M. Kashiwagi, M. S. White, T. Chiba and A. Masuhara, J. Mater. Chem. C, 2025, 13, 14202–14210 RSC.
| This journal is © The Royal Society of Chemistry 2026 |