Open Access Article
Open Access ArticleCentral-metal-cation-based modulation of gas adsorption selectivity in porous tetrapyrrolic materials
Subrata
Maji
a,
Prachi
Gupta
 b,
Rob
Clowes
b,
Yoshitaka
Matsushita
b,
Rob
Clowes
b,
Yoshitaka
Matsushita
 c,
Lok Kumar
Shrestha
c,
Lok Kumar
Shrestha
 a,
Anna G.
Slater
a,
Anna G.
Slater
 b,
Jonathan P.
Hill
b,
Jonathan P.
Hill
 a and
Mandeep K.
Chahal
a and
Mandeep K.
Chahal
 *d
*d
aResearch Center for Materials Nanoarchitectonics, National Institute for Materials Science, 1-1 Namiki, Tsukuba, Ibaraki 305-0044, Japan
bDepartment of Chemistry and Materials Innovation Factory, University of Liverpool, Liverpool L69 7ZD, UK
cResearch Network and Facility Services Division, National Institute for Materials Science, 1-2-1 Sengen, Tsukuba, Ibaraki 305-0047, Japan
dChemistry and Forensic Science, School of Natural Sciences, University of Kent, CT2 7NH Canterbury, UK. E-mail: m.k.chahal@kent.ac.uk
First published on 17th February 2026
Abstract
We present a direct strategy to assemble porous tetrapyrrolic materials with tunable gas uptake selectivity by varying the coordinated cation. Co-OX1 shows improved CO2 uptake of 51.66 cm3 g−1 at 298 K, while free base-OX1 demonstrates a CO2/N2 selectivity of 202.9. This approach offers a viable route to CO2 capture technologies.
A critical driver of climate change is the rising level of atmospheric CO2, which threatens human health, ecosystems, food security and the global economy.1,2 Porous materials are suitable candidates for applications in gas capture technologies.3–5 Known porous solids such as zeolites, metal–organic frameworks (MOFs), and covalent organic frameworks (COFs), offer high gas adsorption capacity and selectivity but suffer from poor processability.6–8 However, once formed, these porous network materials are difficult to disperse in most media, making deposition on substrates challenging and obstructing applications. As an alternative, porous organic molecular materials such as hydrogen- and halogen-bonded organic frameworks (HOF, XOF, resp.), porous organic cages, supramolecular organic frameworks (SOFs), and π-organic frameworks, lack strong intermolecular bonds between building blocks9–12 enabling processing from solution as porous solids or liquids. These porous materials offer various advantages such as ease of structural characterisation, solution processability, tunability, modularity, and ease of regeneration.
Since the emergence of the processable porous materials field, porphyrin-based cages, HOFs, and SOFs have been reported based on intrinsic and/or extrinsic porous networks.13–18 Porphyrins are particularly attractive components of porous materials due to their unique tunable photophysical, photochemical, and electrochemical properties, which allow adaptation to specific functional roles. Topological and pore structure modulation without a requirement for de novo design is crucial in porous materials, enabling precise control over molecular diffusion, adsorption selectivity, and host–guest interactions.19,20 For porphyrinic HOFs, pore engineering has recently been achieved by introducing structure-directing agents (SDAs) such as solvents,16,21 or small cations22 or by varying metal cation coordination.23–25
In general, the metal cation in metalloporphyrins controls the macrocyclic geometry, axial coordination, electronic structure, and intermolecular interactions. Although variation of the coordinated metal cation is known to affect textural properties in metalloporphyrin-based HOFs,23–25 there are no reports so far showing that metalation of porphyrins can influence the selectivity of gas adsorption. Despite this, metalation remains a powerful strategy to generate novel porous materials from existing porphyrin building blocks. This raises the intriguing possibility that metalation might be used as a design tool not only to enhance structural stability and engineer pore structure, but also to enable effective access to new processable porous materials with selective gas storage and sensing capabilities.
We have recently reported a nickel-porphyrin-based porous processable framework material that enables highly sensitive and selective detection of acetone vapour under high humidity conditions.26 Furthermore, analyte specificity can be controlled by varying the molecular structure of the porphyrin.27 This extrinsic porous material is formed through multipoint supramolecular interactions including π–π stacking, metal coordination, and hydrogen bonding.
To build on this, we hypothesised that variation of metalation could influence supramolecular interactions and crystal packing due to conformational and coordinative changes at the porphyrin core, which might lead to gas uptake selectivities. Here, we report a series of metalloporphyrin compounds, Cu-OX1 (1), H2-OX1 (2), Ni-OX1 (3), Co-OX1 (4), Zn-OX1 (5) and Ni-OX3 (6), where the effect of the central metal atom (or its absence) on the porosity of the resulting processable porous materials (Fig. 1) and also the potential of these materials for selective CO2/N2 uptake, have been studied. Synthetic details are given in the SI. Co-OX1 (4) was synthesised by exchanging Ni(II) for Co(II) since the formylation route previously used for Ni-OX1 (3)28 is incompatible with Co(II), leading to mixtures of oxidised products. Cu-OX1 (1) was synthesised following the formylation route, followed by reaction with 5,6-diaminopyrazine-2,3-dicarbonitrile, then oxidation using DDQ or PbO2. The compounds were characterised using spectroscopic techniques (Fig. S1–S9) and single crystal X-ray diffraction studies.
 | ||
| Fig. 1 Chemical structures of M-OX1 and Ni-OX3 compounds synthesised to investigate the effect of metal cations on porous framework structure and gas adsorption selectivity. | ||
Single crystals of Cu-OX1 (1), Ni-OX1 (3), Co-OX1 (4) and Ni-OX3 (6) were grown from suitable solvents. Ni-OX1 (3) (CCDC number: 1920462)29 and Ni-OX3 (6) (CCDC numbers: 2107801–2107804)26 were reported previously.26 The complexes exhibit supramolecular structures formed through different non-covalent interactions (π–π stacking, hydrogen bonding, metal to ligand co-ordination); see Fig. S10–S27. For Cu-OX1 (1), there are four molecules per unit cell with π–π bonds (dimer shortest distance of 3.38(1) Å) and an H-bond, 3.49(8) Å between C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and phenol H. CHCl3 molecules are trapped inside the extended framework as shown in Fig. S11. Molecules of Ni-OX1 (3) form π–π dimers in the solid state with a short intermolecular closest approach of 3.28(4) Å and a C
O and phenol H. CHCl3 molecules are trapped inside the extended framework as shown in Fig. S11. Molecules of Ni-OX1 (3) form π–π dimers in the solid state with a short intermolecular closest approach of 3.28(4) Å and a C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N⋯Ni(II) distance of 3.52(9) Å (Fig. S17). Co-OX1 (4) forms an extended network similar to Cu-OX1 (1) with a π-stacking distance of 3.30(9) Å and a longer H-bond distance of 3.55(6) Å between C
N⋯Ni(II) distance of 3.52(9) Å (Fig. S17). Co-OX1 (4) forms an extended network similar to Cu-OX1 (1) with a π-stacking distance of 3.30(9) Å and a longer H-bond distance of 3.55(6) Å between C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and phenol H (Fig. S22). The magnitude of mean displacement of the 24-atom core (Δ24) and β-pyrrole carbons in Ni-OX1 (3) (Δ24 = 0.284 Å, ΔCβ = 0.365 Å) and Ni-OX3 (6) (Δ24 = 0.471 Å, ΔCβ = 0.330 Å) is much larger than in Cu-OX1 (1) (Δ24 = 0.070 Å, ΔCβ = 0.072 Å) or Co-OX1 (4) (Δ24 = 0.090 Å, ΔCβ = 0.135 Å) suggesting that both Cu-OX1 (1) and Co-OX1 (4) have quasi-planar conformations while the Ni-complexes in both OX1 and OX3 forms are nonplanar (Fig. S28).
O and phenol H (Fig. S22). The magnitude of mean displacement of the 24-atom core (Δ24) and β-pyrrole carbons in Ni-OX1 (3) (Δ24 = 0.284 Å, ΔCβ = 0.365 Å) and Ni-OX3 (6) (Δ24 = 0.471 Å, ΔCβ = 0.330 Å) is much larger than in Cu-OX1 (1) (Δ24 = 0.070 Å, ΔCβ = 0.072 Å) or Co-OX1 (4) (Δ24 = 0.090 Å, ΔCβ = 0.135 Å) suggesting that both Cu-OX1 (1) and Co-OX1 (4) have quasi-planar conformations while the Ni-complexes in both OX1 and OX3 forms are nonplanar (Fig. S28).
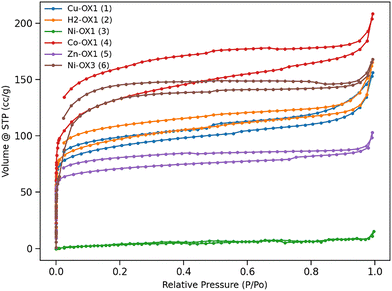
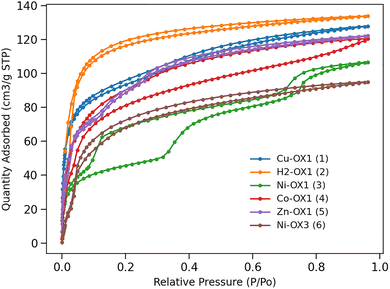
The crystalline nature of the compounds was confirmed by powder X-ray diffraction (PXRD) studies (Fig. S29), and their permanent porosities were investigated by measuring N2 sorption isotherms at 77 K (Fig. 2). To ensure removal of guest molecules, all samples were degassed under vacuum at 120 °C for 24 h prior to N2 sorption measurements. Important textural parameters derived from these measurements are shown in Table S1, indicating that each system exhibits characteristic N2 uptake, Brunauer–Emmett–Teller (BET) surface area and pore volume. Ni-OX1 (3) shows negligible N2 uptake (15.33 cm3 g−1) with a BET surface area of 15.12 m2 g−1. Previously, we proposed that a highly non-planar porphyrin core (Δ24 displacement of the porphyrin mean planes) was required for effective N2 adsorption.26 However, surprisingly, the planar derivatives Cu-OX1 (1) and Co-OX1 (4) exhibit some of the largest BET surface areas and N2 uptakes. This observation suggests that the porphyrin core structure is not the only factor influencing adsorption properties in these systems, but metalation and/or the identity of the coordinated metal atom also plays a significant role in influencing gas adsorption. The BET surface areas, N2 uptakes and corresponding pore volumes for each system are summarised in Table 1.
| Compound | BET surface areas | N2 uptakes | Pore volumes | CO2 uptakes |
|---|---|---|---|---|
| Cu-OX1 (1) | 450.57 | 156.14 | 0.304 | 127.96 |
| H2-OX1 (2) | 450.71 | 165.28 | 0.296 | 133.94 |
| Ni-OX1 (3) | 15.12 | 15.33 | 0.040 | 106.65 |
| Co-OX1 (4) | 509.91 | 208.51 | 0.338 | 120.41 |
| Zn-OX1 (5) | 346.58 | 103.10 | 0.162 | 122.15 |
| Ni-OX3 (6) | 343.56 | 167.95 | 0.249 | 95.07 |
Given the different N2 uptakes at 77 K under gas saturation conditions, we proceeded to investigate CO2 uptake at 195 K and also under saturation conditions to assess the maximum CO2 adsorption capacity of these complexes (Fig. 3). The CO2 uptake capacities shown in Table 1 indicate that Ni-OX3 (6) has the lowest affinity towards CO2. CO2 and N2 uptake data at 195 K and 77 K, respectively, reveal that Ni-OX1 (3) exhibits a substantially higher affinity for CO2 (106.65 cm3 g−1) than for N2 (15.33 cm3 g−1). This suggests an ultra-microporous structure since enhanced CO2 uptake is most likely due to the smaller kinetic diameter of CO2, which facilitates its diffusion into smaller pores. This observation is consistent with Ni-OX1 (3) having the lowest pore volume (0.040 cm3 g−1) of the systems studied. All the materials studied show significant CO2 uptakes making them suitable for applications involving efficient CO2 adsorption, such as gas separation or carbon capture. Both Ni-OX1 (3) and Co-OX1 (4) exhibit hysteresis in their adsorption/desorption curves which can be attributed to phase transition occurring during the process. These phenomena are also confirmed by the PXRD patterns obtained before and after CO2 adsorption as shown in Fig. S32 and S33 where the PXRD patterns differ especially in the low angle regions.
PXRD data obtained prior to and following CO2 uptake studies indicate that the crystalline structure is retained after activation at 110 °C, although some phase transitions in the materials are observed (Fig. S30–35). Thermogravimetric analyses (Fig. S36) of the porous frameworks indicate weight losses starting at 50 °C complete by 170 °C assigned to solvent losses. The materials then exhibit a steady plateaux up to 380 °C, followed by weight losses of 45–59% occurring up to 900 °C assigned to decomposition. These data indicate that both the free-base and metal complexes have excellent thermal stability.
Considering the excellent thermal stability and selective gas uptake by Ni-OX1 (3), the suitability of these porphyrin complexes for gas separation applications was studied. While processable porous materials have been studied for gas separation, the impact of metalation and central atom identity at the porphyrin core remains unexplored, although it might be used to identify a wide range of useful materials without the need for de novo design. For this purpose, we conducted N2 and CO2 sorption experiments both at 273 K and 298 K (Fig. 4). All the materials studied show higher CO2 uptakes at both 273 K and 298 K than their N2 adsorption capacities as shown in Table 2. H2-OX1 (2) demonstrated the highest CO2 uptake of 67.65 cm3 g−1 at 273 K when compared to metal-containing compounds. However, at 298 K, the CO2 uptake of the metal compounds exceeded that of H2-OX1 (2), with Co-OX1 (4) showing the highest uptake of 51.66 cm3 g−1. These uptake values at 298 K are important for practical applications, as direct CO2 capture from the atmosphere under ambient conditions is the key for industrial processes. These results highlight the role of metal ions in tuning the CO2 uptake behaviour. Additionally, these CO2 adsorption capacities are larger than those already reported for other processable porphyrinic porous organic molecular materials (see Table S2) making our materials a significant advance in terms of possible applications. In this study, CO2 sorption measurements were performed on the same samples at three different temperatures (195 K, 273 K, and 298 K), which indicates that the samples exhibit porosity over multiple measurement cycles. The N2 adsorption capacities of these materials are significantly lower, ranging from 0.38 to 8.95 cm3 g−1, highlighting their selectivity of adsorption. Also, Ni-OX3 (6) exhibits the lowest CO2 sorption. The significant differences in CO2 and N2 adsorption capacities suggest that these materials could be effective for CO2/N2 separation. CO2/N2 selectivity was determined using the ideal adsorbed solution theory (IAST),30,31 a reliable method for predicting selectivity in binary gas separations (Fig. S37–S48). For a 0.15/0.85 CO2/N2 mixture, the highest selectivity of 143.4 was calculated for H2-OX1 (2) at 298 K and 0.1 bar, but metal complexes also demonstrate good selectivity. For example, Cu-OX1 (1) has a selectivity of 44.0 and Ni-OX1 (3) showed a value of 34.8 at 0.1 bar. At 273 K, the IAST selectivity differs: Cu-OX1 (1) shows the highest CO2/N2 selectivity of 77.8 at 0.1 bar, followed by Ni-OX1 (3) with a selectivity of 72.7. In contrast, H2-OX1 (2) has a significantly lower selectivity of 9.2 at 0.1 bar. These high selectivity values indicate that varying the central atom in porphyrinic systems can be used to tune the properties of materials for gas separation applications.
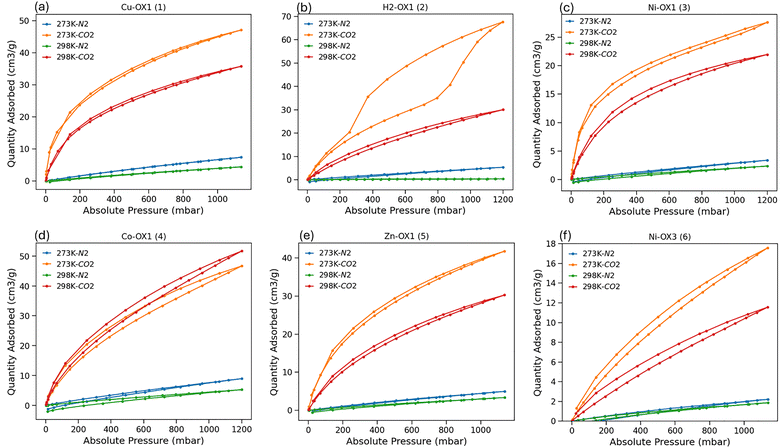 | ||
| Fig. 4 CO2 and N2 adsorption isotherms at 273 K and 298 K, respectively for (a) Cu-OX1 (1); (b) H2-OX1 (2); (c) Ni-OX1 (3); (d) Co-OX1 (4); (e) Zn-OX1 (5); and (f) Ni-OX3. (6). | ||
| Compound | CO2 uptakes at 273 K | CO2 uptakes at 298 K | N2 uptakes at 273 K | N2 uptakes at 298 K |
|---|---|---|---|---|
| Cu-OX1 (1) | 47.06 | 35.76 | 7.39 | 4.41 |
| H2-OX1 (2) | 67.65 | 30.04 | 5.36 | 0.38 |
| Ni-OX1 (3) | 27.61 | 21.96 | 3.39 | 2.37 |
| Co-OX1 (4) | 46.67 | 51.66 | 8.95 | 5.27 |
| Zn-OX1 (5) | 41.79 | 30.28 | 4.99 | 3.37 |
| Ni-OX3 (6) | 17.59 | 11.57 | 2.21 | 1.86 |
In conclusion, we report the modulation of the CO2/N2 gas adsorption capabilities of a family of supramolecular porous porphyrins by varying the coordinated metal cation. Under saturation conditions, Ni-OX1 (3) shows negligible N2 uptake (15.33 cm3 g−1) while Ni-OX3 (6) and other metal and free-base OX1 complexes show significant adsorption. OX1 systems show substantial CO2 uptake (up to 133.94 cm3 g−1 for H2-OX1) at 195 K. Further experiments at 278 K and 298 K, along with IAST calculations, confirm selective CO2 adsorption and separation. These materials are solution processable, and their solubility in common organic solvents offers an additional advantage by enabling applications beyond bulk sorption experiments, such as thin-film fabrication and device integration.
SM, PG, RC, YM, and LKS: investigation; validation; writing – review and editing. AGS, JPH: supervision; validation; writing – review and editing; funding acquisition. MKC: conceptualization; funding acquisition; synthesis; writing – original draft, review and editing.
MKC thanks the Royal Society of Chemistry (R23-0850952021) and the Royal Society (RG\R1\251071) for funding. AGS thanks the Royal Society for a University Research Fellowship (URF\R1\201168). This work made use of shared equipment at the Materials Innovation Factory (MIF) created as part of the UK Research Partnership Innovation Fund (Research England) and co-funded by the Sir Henry Royce Institute.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included in the supplementary information (SI). Supplementary information: experimental methods, crystallographic details, thermal analysis, PXRD patterns, and IAST plots for selectivity prediction. See DOI: https://doi.org/10.1039/d5cc06796k.CCDC 2501550 and 2501551 contain the supplementary crystallographic data for this paper.32a,b
References
- D. Wang, J. Penuelas, Y. Tao, I. Loladze, C. Cai, L. Song, J. Zhang, G. Zhang, Y. Wang, W. Zhou, Q. Li and C. Zhu, Global Change Biol., 2025, 31, e70299 CrossRef PubMed.
- L. J. R. Nunes, Environments, 2023, 10, 66 Search PubMed.
- L. Pan, K. M. Adams, H. E. Hernandez, X. Wang, C. Zheng, Y. Hattori and K. Kaneko, J. Am. Chem. Soc., 2003, 125, 3062–3067 CrossRef PubMed.
- L. Wang and R. Yang, J. Phys. Chem. C, 2011, 115, 21264–21272 Search PubMed.
- A. H. Farmahini, S. Krishnamurthy, D. Friedrich, S. Brandani and L. Sarkisov, Chem. Rev., 2021, 121, 10666–10741 Search PubMed.
- N. Stock and S. Biswas, Chem. Rev., 2012, 112, 933–969 Search PubMed.
- G. Zhang, B. Hua, A. Dey, M. Ghosh, B. A. Moosa and N. M. Khashab, Acc. Chem. Res., 2021, 54, 155–168 CrossRef PubMed.
- A. G. Slater and A. I. Cooper, Science, 2015, 348, aaa8075 Search PubMed.
- J. Tian, P. K. Thallapally and B. P. McGrail, CrystEngComm, 2012, 14, 1909–1919 Search PubMed.
- P. Sun, H. Dong, S. Lv, Y. Yin, G. Gong, L. Wang, J. Wang and S. Chen, J. Mater. Chem. A, 2024, 12, 1128–1134 Search PubMed.
- Z. J. Lin, S. A. R. Mahammed, T. F. Liu and R. Cao, ACS Cent. Sci., 2022, 8, 1589–1608 Search PubMed.
- M. Liu, M. A. Little, K. E. Jelfs, J. T. A. Jones, M. Schmidtmann, S. Y. Chong, T. Hasell and A. I. Cooper, J. Am. Chem. Soc., 2014, 136, 7583–7586 CrossRef PubMed.
- S. Hong, Md. R. Rohman, J. Jia, Y. Kim, D. Moon, Y. Kim, Y. H. Ko, E. Lee and K. Kim, Angew. Chem., Int. Ed., 2015, 127, 13439–13442 CrossRef.
- W. Yang, B. Li, H. Wang, O. Alduhaish, K. Alfooty, M. A. Zayed, P. Li, H. D. Arman and B. Chen, Cryst. Growth Des., 2015, 15, 2000–2004 Search PubMed.
- Y. N. Gong, D. C. Zhong and T. B. Lu, Angew. Chem., Int. Ed., 2025, 64, e202424452 CrossRef PubMed.
- L. Ma, H. Arman, Y. Xie, W. Zhou and B. Chen, Cryst. Growth Des., 2022, 22, 3808–3814 CrossRef.
- J. H. Lee, H. Park, Y. Kim, D. Yim, T. Kim, J. Choi, Y. Lee and W. D. Jang, ACS Appl. Mater. Interfaces, 2023, 15, 57507–57513 Search PubMed.
- W. Yang, F. Yang, T. L. Hu, S. C. King, H. Wang, H. Wu, W. Zhou, J. R. Li, H. D. Arman and B. Chen, Cryst. Growth Des., 2016, 16, 5831–5835 Search PubMed.
- A. G. Slater, P. S. Reiss, A. Pulido, M. A. Little, D. L. Holden, L. Chen, S. Y. Chong, B. M. Alston, R. Clowes, M. Haranczyk, M. E. Briggs, T. Hasell, G. M. Day and A. I. Cooper, ACS Cent. Sci., 2017, 3, 734–742 CrossRef PubMed.
- M. O’Shaughnessy, P. R. Spackman, M. A. Little, L. Catalano, A. James, G. M. Day and A. I. Cooper, Chem. Commun., 2022, 58, 13254–13257 RSC.
- X. T. He, Y. H. Luo, Z. Y. Zheng, C. Wang, J. Y. Wang, D. L. Hong, L. H. Zhai, L. H. Guo and B. W. Sun, ACS Appl. Nano Mater., 2019, 2, 7719–7727 Search PubMed.
- Y. Cai, J. Gao, J. H. Li, P. Liu, Y. Zheng, W. Zhou, H. Wu, L. Li, R. B. Lin and B. Chen, Angew. Chem., Int. Ed., 2023, 62, e202308579 Search PubMed.
- P. Tholen, C. A. Peeples, M. M. Ayhan, L. Wagner, H. Thomas, P. Imbrasas, Y. Zorlu, C. Baretzky, S. Reineke, G. Hanna and G. Yücesan, Small, 2022, 18, 2204578 CrossRef PubMed.
- Q. Yin, E. V. Alexandrov, D. H. Si, Q. Q. Huang, Z. Bin Fang, Y. Zhang, A. A. Zhang, W. K. Qin, Y. L. Li, T. F. Liu and D. M. Proserpio, Angew. Chem., Int. Ed., 2022, 61, e202115854 CrossRef PubMed.
- S. Huang, Y. Chang, Z. Li, J. Cao, Y. Song, J. Gao, L. Sun and J. Hou, Adv. Funct. Mater., 2023, 33, 2211631 Search PubMed.
- M. K. Chahal, S. Maji, A. Liyanage, Y. Matsushita, P. Tozman, D. T. Payne, W. Jevasuwan, N. Fukata, P. A. Karr, J. Labuta, L. K. Shrestha, S. Ishihara, K. Ariga, F. D’Souza, G. Yoshikawa, Y. Yamauchi and J. P. Hill, Mater. Chem. Front., 2023, 7, 325–332 RSC.
- S. Maji, J. Hynek, K. Nakada, A. Hori, K. Minami, J. Labuta, M. K. Chahal, D. T. Payne, G. J. Richards, L. K. Shrestha, K. Ariga, Y. Yamauchi, G. Yoshikawa and J. P. Hill, Adv. Mater. Technol., 2025, 10, e00294 CrossRef.
- M. K. Chahal and M. Sankar, Dalton Trans., 2016, 45, 16404–16412 Search PubMed.
- M. K. Chahal, J. Labuta, V. Březina, P. A. Karr, Y. Matsushita, W. A. Webre, D. T. Payne, K. Ariga, F. D'Souza and J. P. Hill, Dalton Trans., 2019, 48, 15583–15596 Search PubMed.
- C. M. Simon, B. Smit and M. Haranczyk, Comput. Phys. Commun., 2016, 200, 364–380 CrossRef.
- Mantid, Manipulation and Analysis Toolkit for Instrument Data, 2013, Mantid Project DOI:10.5286/SOFTWARE/MANTID6.14.
- (a) CCDC 2501550: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2pz25y; (b) CCDC 2501551: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2pz26z.
| This journal is © The Royal Society of Chemistry 2026 |