Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Accessing zinc(I) via reductive elimination of cyclopentadienes
Benjamin
Pitz
 a,
Sergi
Danés
a,
Sergi
Danés
 *b,
Bernd
Morgenstern
a and
André
Schäfer
*b,
Bernd
Morgenstern
a and
André
Schäfer
 *a
*a
aFaculty of Natural Sciences and Technology, Department of Chemistry, Saarland University, Campus Saarbrücken, 66123 Saarbrücken, Germany. E-mail: andre.schaefer@uni-saarland.de
bInstitute of Chemical Research of Catalonia (ICIQ), Barcelona Institute of Science and Technology (BIST), Avinguda Països Catalans 16, 43007 Tarragona, Spain. E-mail: sdanes@iciq.es
First published on 8th January 2026
Abstract
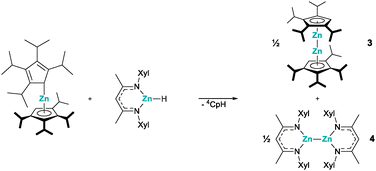
The reaction between zincocenes and β-diketiminate zinc hydrides was investigated, and the compounds were found to undergo reductive elimination reactions, giving the corresponding zinc(I) species and cyclopentadienes as the elimination product. The zinc(I) compounds underwent disproportionation reactions in several cases and a ligand exchange reaction in one instance, which gave rise to octaisopropyldizincocene.
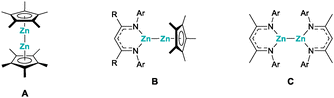
Organozinc compounds are among the first organometallic molecules ever described, dating back to the report of diethyl zinc, in the mid-19th century.1 However, in the large majority of organozinc compounds, zinc exhibits the +II oxidation state, due to its formal 4s2 valence electron configuration.2 Hence, the isolation of decamethyldizincocene, A, in 2004, was a major milestone in modern organometallic chemistry, as it not only represented a stable molecule with two zinc atoms in the +I oxidation state, but also introduced the structural motif of a dimetallocene (Fig. 1).3a,b Noteworthily, this is still rare, with just a handful of examples.3c–e While the initial synthesis did offer access to zinc(I), the exact mechanism by which this remarkable compound formed remained unclear for many years. Evidence that the key step is a reductive elimination of pentamethylcyclopentadiene from a transient zinc hydride species and decamethylzincocene, Cp*2Zn, was provided by Fischer et al., who were able to detect penta-methylcyclopentadiene (Cp*H), in the reaction mixture by NMR spectroscopy.4 At the same time, their research offered an improved access to decamethyldizincocene, A, which has become an important starting material for other zinc(I) compounds.5 For instance, when dizincocene A is treated with one or two equivalents of a suitable protic ligand, such as a β-diketiminate, protonation and substitution of one or both of the Cp* ligands take place, giving access to the corresponding β-diketiminate (NacNac) zinc(I) complexes, B or C (Fig. 1). Of course, this synthetic route requires the preparation of the dizincocene precursor, which is a multi-step synthesis itself. Thus, low-valent organozinc chemistry would greatly benefit from a more direct access to zinc(I), from easily prepared starting materials.
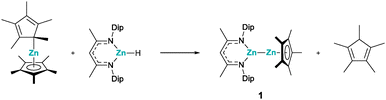
Noteworthily, Xu and Crimmin reported that Zn–Zn bonds can be formed by transition metal mediated dehydrocoupling, starting from β-diketiminate zinc hydrides, which presented an interesting synthetic strategy.6 With this in mind and expanding on Fischer's findings of a zinc centred reductive elimination of decamethylcyclopentadiene,4 we chose to investigate the reactions between zincocenes and β-diketiminate zinc hydrides. We started our investigation with the reaction of decamethyl-zincocene, Cp*2Zn, and an N-Dip-substituted β-diketiminate zinc hydride, DipNacNacZnH, as these compounds are easily accessible,7–9 and more importantly, the potential zinc(I) reaction product had already been described by Schulz et al., offering comprehensive NMR data for comparison.5a Indeed, stirring a benzene solution of Cp*2Zn and DipNacNacZnH at ambient temperature for 24 h results in the formation of the corresponding heteroleptic zinc(I) compound, 1, along with pentamethylcyclopentadiene (Cp*H) (Scheme 1). Both products were identified by their characteristic 1H and 13C NMR chemical shifts.5a This clearly shows that zinc(I) compounds can be accessed from zinc(II) precursors by reductive elimination of a cyclopentadiene.
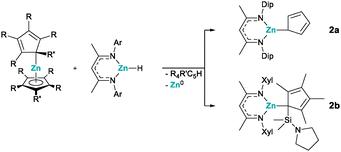
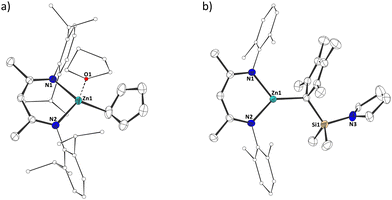
To probe how universal this route is, we prepared different zincocenes and β-diketiminate zinc hydrides. When unsubstituted zincocene, Cp2Zn, was reacted with the same DipNacNacZnH, we could again observe cyclopentadiene as an elimination product, along with a new set of signals in the 1H and 13C NMR spectra. On the other hand, a recently reported bis(pyrrolidinyldimethylsilyl)-octamethylzincocene, CpSi#2Zn,10 and DipNacNacZnH showed no reaction under identical conditions. We attribute this to steric hindrance, as this zincocene does undergo a reaction with N-xylyl-substituted derivative XylNacNacZnH.7,11 In both cases, in which we observed a reaction between the zincocene and the zinc hydride, we initially assigned the new sets of signals in the 1H and 13C NMR spectra to the corresponding heteroleptic zinc(I) compounds. Peculiarly, however, we always observed precipitation of a grey solid in these reactions. While it was easily removed by filtration, and NMR spectroscopy otherwise indicated the reactions to be highly selective, giving only one product, it made us suspicious about the exact nature of the products. It is noteworthy that the heteroleptic zinc(I) compounds of the type “NacNacZn(I)–Zn(I)(C5R5)” cannot easily be differentiated by NMR spectroscopy from their heteroleptic zinc(II) analogues of the type “NacNacZn(II)(C5R5)”, due to their identical ligand systems and thus close structural relationship. Fortunately, we were able to obtain single crystals of both products, suitable for SC-XRD analysis (in the case of 2a in the form of its thf adduct). This enabled us to structurally authenticate the compounds to be heteroleptic zinc(II) species 2a,b (Scheme 2). The solid-state structures clearly show one β-diketiminate and one cyclopentadienyl moiety bound to the zinc(II) centers (Fig. 2).
Accordingly, we believe that the reductive elimination does indeed take place and that the heteroleptic zinc(I) compounds formed initially, although we could not observe them spectroscopically. Apparently, these zinc(I) species are not persistent under ambient conditions, because of insufficient kinetic stabilization, due to the relatively small steric demand of the substituents, and consequently undergo disproportionation reactions. Although the zinc(I) species remained elusive in these cases, this method does present an elegant synthetic route to heteroleptic (cyclopentadienyl)(β-diketiminate)zinc(II) compounds. Following these results, it was evident that a certain steric demand is necessary to stabilize zinc(I) species. We therefore prepared the sterically much more demanding octaisopropyl-zincocene, 4Cp2Zn.12 Stirring a benzene or toluene solution of 4Cp2Zn and DipNacNacZnH at ambient temperature did not result in any reaction, presumably due to the steric hindrance. However, reacting 4Cp2Zn with the less steric demanding XylNacNacZnH resulted in the formation of tetraisopropyl-cyclopentadiene (4CpH), along with the appearance of new signals in the 1H and 13C NMR spectra. The absence of any grey precipitate led us to believe that the corresponding heteroleptic zinc(I) complex had formed. Much to our surprise, SC-XRD analysis of single crystals obtained from the reaction mixture revealed that the product was indeed the homoleptic octaisopropyldizincocene 3 (Scheme 3 and Fig. 3)!
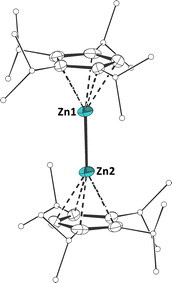 | ||
| Fig. 3 Molecular structure of 3 in the crystal (displacement ellipsoids at 50% probability level; isopropyl groups drawn as ball-and-stick models; and H atoms omitted for clarity). | ||
We propose that this results from a heteroleptic homoleptic ligand exchange, following the initial formation of the heteroleptic zinc(I) species “DipNacNacZn(I)–Zn(I)(C5HiPr4)”. Therefore, based on stochiometric considerations, we tentatively assign the remaining signals to the homoleptic bis(β-diketiminate)zinc(I) species 4.13 Although the exact mechanism remains unclear at this time, it is noteworthy that the synthesis of 3 is well reproducible. The isolation of dizincocene 3 is quite remarkable, as it is only the third example of such a compound, following Carmona's reports of decamethyldizincocene, A, and diethyloctamethyldizincocene, (Me4EtC5)2Zn2, some 20 years ago.3a,b Noteworthily, the two previously reported dizincocenes are structurally very closely related and 3 exhibits a substantially different substitution pattern. Furthermore, while all dizincocenes exhibit near linear/coplanar arrangements of the cyclopentadienide ligands (∢(ZnZn4Cpcent): 177.04(2)°), the dizincocenes reported by Carmona et al. possess eclipsed conformations in the solid state, while 3 shows a staggered conformation in the solid state. However, we attribute this to the packing effect in the crystal. The Zn–Zn bond lengths in the previously reported dizincocenes are 229.50(3) pm ((Me4EtC5)2Zn23b) and 230.49(4) pm (A3a), which is longer than that in 3, measuring 227.92(4) pm, despite its bulkier substitution pattern. This may be a result of attractive dispersion interactions between the isopropyl groups (vide infra), an effect that was previously observed in an isopropyl-substituted heterobimetallic dimetallocene.3e To investigate the electronic properties of dizincocene 3 and investigate the Zn–Zn bond, we performed DFT calculations.7 Inspection of the Kohn–Sham molecular orbitals revealed that the HOMO−4 corresponds to the Zn–Zn σ bond. On the other hand, the HOMO to HOMO−3 are associated with the ligand centred π systems, while the LUMO to LUMO+2 are located at the Zn2 moiety and justify its Lewis acidic nature (Fig. S14).14 To gain further insight into the nature of the Zn–Zn bond in 3, we performed a Natural Bond Orbital (NBO) analysis, which disclosed that the Zn–Zn bond exhibits a pronounced s character of 81.8%, originating from the formal 4s1 configuration of the zinc(I) centres (Fig. 4).
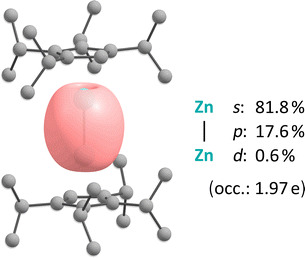 | ||
| Fig. 4 NBO contour plot illustrating the Zn–Zn σ bond in 3, calculated at the PBE0-D3(BJ)/def2-TZVP//PBE0-D3(BJ)/def2-SVP level of theory. | ||
This is consistent with the Zn–Zn bonds in other zinc(I) compounds stabilized by other ligands, such as β-diketiminates (Table S2). To understand why the Zn–Zn bond in 3 is so short, we performed Energy Decomposition Analysis combined with Natural Orbitals for Chemical Valence (EDA-NOCV).15 The overall dissociation energy of 3 is computed to be De = 307.8 kJ mol−1, which is slightly higher than that of Carmona's dizincocene A (De = 294.0 kJ mol−1),16 but lower than for some NacNac substituted systems (Table S3). This high dissociation energy is also reflected by our experimental observation of a remarkably high thermal stability of 3. More specifically, dizincocene 3 is stable for at least 4 days in boiling toluene (110 °C) under an atmosphere of argon. The σ-type NOCV bonding channel constitutes the most stabilizing orbital interaction in 3 (ΔEorb-σ = −163.4 kJ mol−1) and in A (ΔEorb-σ = −160.1 kJ mol−1) and is slightly stronger than in NacNac substituted systems (Table S3). This is consistent with the shorter Zn–Zn bonds in dizincocenes than in many other compounds exhibiting Zn–Zn bonds and matches the calculated bond orders (Table S1).6 Thus, dizincocenes exhibit a strong covalent Zn–Zn σ bond. Furthermore, as speculated (vide supra), attractive dispersion interactions also do play a role in the stabilization of the Zn–Zn bond in 3 (ΔEdisp = −30.8 kJ mol−1). This is the reason why 3 exhibits the shortest Zn–Zn bond of all dizincocenes, and this contribution is approximately two times larger than in Carmona's dizincocene A (ΔEdisp = −14.5 kJ mol−1). Noteworthily, this is substantially lower than in NacNac substituted compounds with Zn–Zn bonds (Table S3), which exhibit strong dispersion interactions between the flanking aryl groups. In general, our bonding analysis studies suggest that the Cp-substituted dizincocenes have shorter Zn–Zn bonds, due to stronger orbital and electrostatic interactions, while dispersion interactions play a secondary role. In contrast, NacNac-substituted complexes exhibit longer Zn–Zn bonds, with the orbital and electrostatic contributions being substantially weaker and attractive dispersion interactions stronger.
In summary, a new access to zinc(I)organyls, starting from β-diketiminate zinc(II)hydrides and zincocenes, via reductive elimination of cyclopentadienes, is reported. This route provides improved access to low-valent zinc(I) compounds and will be of interest for the preparation of other zinc(I)organyls, in the future. While the mechanism will be the subject of future investigations, we found that the reaction itself and the stability of the formed zinc(I) compounds are heavily influenced by steric factors. While the combination of too bulky substitution patterns prevented any reaction, low steric bulk resulted in disproportionation of the zinc(I) species due to insufficient kinetic stabilization. Most remarkably, in one case, the homoleptic dizincocene was reproducibly isolated, rather than the heteroleptic product. Octaisopropyldizincocene, 3, is the third example of such a species and the first derivative with a substantially different substitution pattern, more than twenty years after Carmona's initial report.3a
B. P. conducted most of the experimental work and assisted in writing the manuscript and SI. S. D. performed the bonding analysis of 3, interpreted the results and assisted in writing the manuscript and SI. B. M. performed the single-crystal X-ray diffractometry, solved and refined the crystal structures and assisted in the interpretation of the data. A. S. conceived and supervised the project, and wrote, reviewed and edited the manuscript and SI with input from the other authors.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article are available in the supplementary information (SI). Supplementary information: experimental details and NMR data, NMR spectra, XRD data, computational details, and additional references. See DOI: https://doi.org/10.1039/d5cc06448a.CCDC 2501219 ({XylNacNacZnI}2), 2501220 ({XylNacNacZnH}2), 2501222 (XylNacNac2Zn), 2501223 (2a·thf), 2501225 (2b) and 2501226 (3) contain the supplementary crystallographic data for this paper.17a–f
Acknowledgements
Jessica Lambert is acknowledged for performing preliminary experiments. Sergi Danés thanks Prof. Dr. Julio Lloret-Fillol for his generous support and insightful discussions, as well as the PHOTOSINT project for funding (101118129). Susanne Harling is acknowledged for elemental analysis. The German Research Foundation, DFG, is thanked for funding (SCHA1915/3-1/2). Instrumentation and technical assistance for this work were provided by the service centre mass spectrometry and the service centre X-ray diffraction, with financial support from Saarland University and German Research Foundation (INST 256/506-1 (D8 Venture); 256/582-1 (Synergy-S)).References
- (a) E. Frankland, Justus Liebigs Ann. Chem., 1849, 71, 171–213 CrossRef; (b) E. Frankland and B. F. Duppa, J. Chem. Soc., 1864, 17, 29–36 RSC.
- Z. Rappoport and I. Marek, The Chemistry of Organozinc Compounds in Patai Series: The Chemistry of Functional Groups, John Wiley & Sons, 2006, ISBN: 0-470-09337-4 Search PubMed.
- (a) I. Resa, E. Carmona, E. Gutierrez-Puebla and A. Monge, Science, 2004, 305, 1136–1138 CrossRef PubMed; (b) A. Grirrane, I. Resa, A. Rodriguez, E. Carmona, E. Alvarez, E. Gutierrez-Puebla, A. Monge, A. Galindo, D. del Río and R. A. Andersen, J. Am. Chem. Soc., 2007, 129, 693–703 CrossRef CAS PubMed (corrigendum: J. Am. Chem. Soc., 2007, 129, 14100–14100); (c) J. T. Boronski, A. E. Crumpton, L. L. Wales and S. Aldridge, Science, 2023, 380, 1147–1149 Search PubMed; (d) J. T. Boronski, A. E. Crumpton, A. F. Roper and S. Aldridge, Nat. Chem., 2024, 16, 1295–1300 CrossRef CAS PubMed; (e) I.-A. Bischoff, S. Danés, P. Thoni, B. Morgenstern, D. M. Andrada, C. Müller, J. Lambert, E. C. J. Gießelmann, M. Zimmer and A. Schäfer, Nat. Chem., 2024, 16, 1093–1100 Search PubMed.
- K. Freitag, H. Banh, C. Ganesamoorthy, C. Gemel, R. W. Seidel and R. A. Fischer, Dalton Trans., 2013, 42, 10540–10544 RSC.
- (a) S. Schulz, D. Schuchmann, U. Westphal and M. Bolte, Organometallics, 2009, 28, 1590–1592 CrossRef CAS; (b) S. Schulz, S. Gondzik, D. Schuchmann, U. Westphal, L. Dobrzycki, R. Boesea and S. Harder, Chem. Commun., 2010, 46, 7757–7759 RSC; (c) B. Li, K. Huse, C. Wölper and S. Schulz, Chem. Commun., 2021, 57, 13692–13695 Search PubMed; (d) R. K. Sahoo, S. Rajput, A. G. Patroa and S. Nembenna, Dalton Trans., 2022, 51, 16009–16016 Search PubMed.
- (a) M. Chen, S. Jiang, L. Maron and X. Xu, Dalton Trans., 2019, 48, 1931–1935 RSC; (b) S. Jiang, Y. Cai, A. Carpentier, I. del Rosal, L. Maron and X. Xu, Chem. Commun., 2021, 57, 13696–13699 RSC; (c) M. Perez-Jimenez, B. L. Geoghegan, A. Collauto, M. M. Rößler and M. R. Crimmin, Angew. Chem., Int. Ed., 2024, 63, e202411828 Search PubMed.
- See SI for further details and references.
- R. Blom, J. Boersma, P. H. M. Budzelaar, B. Fischer, A. Haaland, H. V. Volden and J. Weidlein, Acta Chem. Scand., Ser. A, 1986, 40, 113–120 CrossRef.
- J. Spielmann, D. Piesik, B. Wittkamp, G. Jansena and S. Harder, Chem. Commun., 2009, 3455–3456 Search PubMed.
- J. Lambert, B. Morgenstern and A. Schäfer, Z. Naturforsch., B, 2025, 80, 37–41 CrossRef CAS.
- H. Hao, C. Cui, H. W. Roesky, G. Bai, H.-G. Schmidt and M. Noltemeyer, Chem. Commun., 2001, 1118–1119 RSC.
- D. J. Burkey and T. P. Hanusa, J. Organomet. Chem., 1996, 512, 165–173 CrossRef CAS.
- The 1H and 13C NMR chemical shifts assigned to 4 do not correspond to the shifts of the homoleptic spiro-cyclic zinc(II) species XylNacNac2Zn(II).7.
- (a) S. Schulz, D. Schuchmann, I. Krossing, D. Himmel, D. Bläser and R. Boese, Angew. Chem., Int. Ed., 2009, 48, 5748–5751 CrossRef CAS PubMed; (b) P. Jochmann and D. W. Stephan, Chem. – Eur. J., 2014, 20, 8370–8378 CrossRef CAS PubMed.
- M. P. Mitoraj, A. Michalak and T. Ziegler, J. Chem. Theory Comput., 2009, 5, 962–975 CrossRef CAS PubMed.
- M. A. Gosch and D. J. D. Wilson, Organometallics, 2023, 42, 2185–2196 CrossRef CAS.
- (a) CCDC 2501219: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2pyqhw; (b) CCDC 2501220: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2pyqjx; (c) CCDC 2501222: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2pyqlz; (d) CCDC 2501223: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2pyqm0; (e) CCDC 2501225: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2pyqp2; (f) CCDC 2501226: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2pyqq3.
| This journal is © The Royal Society of Chemistry 2026 |