A coumarin-based dual-channel fluorescent probe for discriminating Hcy and GSH in live cells
Xi-Xi
Wu
*a,
Chao-Wen
Guo
b,
Wen-Yu
Lu
b,
Hui-Jing
Li
b and
Yan-Chao
Wu
 *b
*b
aChengdu Women's and Children's Central Hospital, School of Medicine, University of Electronic Science and Technology of China, Chengdu 611731, China. E-mail: 946453461@qq.com
bWeihai Marine Organism and Medical Technology Research Institute, Harbin Institute of Technology, Weihai 264209, China. E-mail: yanchaowu@hit.edu.cn
First published on 8th December 2025
Abstract
Cysteine (Cys), homocysteine (Hcy), and glutathione (GSH) share similar functional groups, such as thiol (–SH) and amino (–NH2), but differ in their spatial structures. These structural differences can lead to distinct binding behaviors with fluorescent probes that possess multiple reactive sites, resulting in diverse reaction products and thereby producing distinct color and fluorescence responses. Based on this principle, a phenyl selenide-substituted coumarin probe (PSeC) capable of distinguishing GSH from Cys and Hcy is designed and synthesized in this work. The probe itself is non-fluorescent, but exhibits strong fluorescence upon reacting with biothiols. Within the PSeC structure, both the phenyl selenide and aldehyde groups serve as potential reaction sites for biothiol interaction, yielding characteristic fluorescence emissions—green for Cys and Hcy, and red for GSH. The probe displayed fluorescence enhancement factors of 90-fold for GSH, 142-fold for Hcy, and 29-fold for Cys. Moreover, the response time toward Cys was markedly longer than those toward Hcy and GSH, enabling discrimination of the three biothiols through distinct fluorescence channels and kinetic behaviors. Furthermore, the probe demonstrated the capability to differentiate Hcy and GSH in living cells based on their unique fluorescence signatures.
Introduction
Biothiols, including cysteine (Cys), homocysteine (Hcy), and glutathione (GSH), play crucial roles in biological systems as they participate in enzymatic and metabolic processes associated with proteins and transport mechanisms.1–3 They are closely related to numerous physiological functions and various diseases.4–6 Although these three biothiols share similar structural motifs, their biological functions are distinct.7 Cys regulates the rate of intracellular GSH synthesis in the active sulfur metabolic pathway and is the most abundant biogenic sulfur-containing amino acid in serum and plasma.8 Both deficiency and excess of Cys can lead to health problems: insufficient Cys may cause growth retardation, skin damage, neurotoxicity, edema, and liver injury, while excess levels are associated with Parkinson's and cardiovascular diseases.9–11 Structurally, Hcy differs from Cys by a single methylene group and typically exists in cells at concentrations of 5–15 µM. Abnormal Hcy levels are implicated in disorders such as osteoporosis, Alzheimer's disease, and cardiovascular diseases.12–15 Among the three, GSH is the most abundant intracellular biothiol (approximately 1–10 mM). It functions as a major antioxidant by converting into oxidized glutathione (GSSG) through disulfide bond formation when reacting with reactive oxygen species (ROS). The GSH/GSSG redox couple maintains cellular redox homeostasis and protects cells from oxidative stress.16,17 Depletion of GSH is one of the earliest indicators of apoptosis,18 and abnormal GSH levels are strongly associated with diseases such as Alzheimer's, leukemia, liver injury, and HIV/AIDS.11,19,20 Therefore, accurate detection and discrimination of biothiols are of great importance for disease diagnosis and prevention.Fluorescent probes have become powerful tools for studying biological thiols due to their advantages of simple synthesis, tunable photophysical properties, noninvasive detection, high sensitivity, and excellent spatiotemporal resolution.21–23 However, because Cys, Hcy, and GSH all contain amino and thiol groups, differentiating them using small-molecule fluorescent probes remains a major challenge.24–27 Current strategies for Cys detection include nucleophilic aromatic substitution and rearrangement reactions,28,29 Michael addition,30–33 or cyclization reaction with aldehyde groups.34,35 GSH can be distinguished from Cys/Hcy via deprotection of 2,4-dinitrobenzenesulfonyl fluorophores,36,37 selective Se–N bond cleavage,38,39 or GSH-induced substitution–cyclization reactions.40 However, because GSH and Cys metabolic pathways coexist and interact in living systems,41 simultaneous visualization of both remains difficult. Dual-probe systems can monitor these biothiols separately, but their use often introduces complications such as spectral overlap and reaction interference. Therefore, developing a single fluorescent probe with multifunctional reactivity is a more efficient strategy. Recently, multi-site fluorescent probes have been designed to exploit structural differences among biothiols, allowing them to react at different positions and produce distinct fluorescence emissions.42–44 Nevertheless, existing probes still face challenges such as small Stokes shifts, dependence on high organic solvent content, and limited capability to simultaneously discriminate among Cys, Hcy, and GSH.45–48 Hence, designing single-molecule fluorescent probes capable of distinguishing all three biothiols through multiple fluorescence channels with high selectivity is of great significance.
In this work, we report a coumarin-based fluorescent probe containing two reactive sites. The phenyl selenide moiety acts both as a fluorescence quencher and a leaving group in nucleophilic substitution with biothiols. Meanwhile, the neighboring aldehyde group enhances the electrophilicity of the coumarin Michael acceptor, facilitating cyclization with the amino group of biothiols. This dual-reactive design enables the probe to distinguish Cys/Hcy from GSH through different emission wavelengths and to differentiate Cys from Hcy based on reaction kinetics. The developed probe, PSeC, exhibits excellent performance in detecting and discriminating Hcy and GSH in living cells using dual-channel fluorescence imaging under laser scanning confocal microscopy.
Experimental
Materials and instruments
All reagents were bought from commercial suppliers and utilized without additional purification. A Bruker 400 MHz NMR spectrometer was used to conduct nuclear magnetic resonance spectroscopy. Mass spectrometry data were performed using LTQ Orbitrap XL. A PerkinElmer Lambda 365 spectrophotometer was used to measure UV-visible absorption spectra. The fluorescence spectrum was traced using a Hitachi FV-2700 fluorescence spectrometer. Cell fluorescence imaging was carried out using an Olympus FV-1000 confocal laser scanning microscope.Synthesis and characterization of probe PSeC
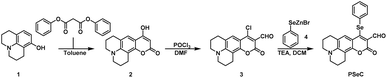
The synthesis route of probe PSeC is shown in Scheme 1. Compounds 2 and 3 were synthesized by using the procedure reported in the literature.44![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to afford compound 3 as a red solid (480 mg, 82%). 1H NMR (400 MHz, CDCl3): δ 10.22 (s, 1H), 7.39 (s, 1H), 3.41–3.37 (m, 4H), 2.86–2.77 (m, 4H), 2.01–1.97 (m, 4H). 13C NMR (100 MHz, CDCl3): δ 187.1, 160.1, 153.5, 151.3, 149.7, 124.9, 120.5, 109.4, 107.2, 105.5, 50.4, 50.0, 27.5, 20.9, 20.1, 19.9. HRMS (ESI) (C16H14ClNO3) m/z: calculated for [M + Na]+: 326.0555; found [M + Na]+: 326.0527.
1) to afford compound 3 as a red solid (480 mg, 82%). 1H NMR (400 MHz, CDCl3): δ 10.22 (s, 1H), 7.39 (s, 1H), 3.41–3.37 (m, 4H), 2.86–2.77 (m, 4H), 2.01–1.97 (m, 4H). 13C NMR (100 MHz, CDCl3): δ 187.1, 160.1, 153.5, 151.3, 149.7, 124.9, 120.5, 109.4, 107.2, 105.5, 50.4, 50.0, 27.5, 20.9, 20.1, 19.9. HRMS (ESI) (C16H14ClNO3) m/z: calculated for [M + Na]+: 326.0555; found [M + Na]+: 326.0527.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to yield probe PSeC as an orange solid (97 mg, 69%). The melting point of PSeC is 231–232 °C. 1H NMR (400 MHz, CDCl3): δ 10.35 (s, 1H), 7.52–7.49 (m, 2H), 7.30–7.27 (m, 3H), 7.03 (s, 1H), 3.33–3.24 (m, 4H), 2.87 (t, J = 6.4 Hz, 2H), 2.29 (t, J = 6.2 Hz, 2H), 2.00–1.93 (m, 2H), 1.85–1.79 (m, 2H). 13C NMR (100 MHz, CDCl3): δ 188.5, 160.9, 159.6, 149.6, 147.3, 132.3, 131.7, 128.6, 128.3, 127.1, 117.4, 113.5, 108.2, 104.4, 76.3, 76.0, 75.7, 49.1, 48.7, 26.1, 20.0, 19.2. HRMS (ESI) (C22H19NO3Se) m/z: calculated for [M + Na]+: 448.0423; found [M + Na]+: 448.0372.
1) to yield probe PSeC as an orange solid (97 mg, 69%). The melting point of PSeC is 231–232 °C. 1H NMR (400 MHz, CDCl3): δ 10.35 (s, 1H), 7.52–7.49 (m, 2H), 7.30–7.27 (m, 3H), 7.03 (s, 1H), 3.33–3.24 (m, 4H), 2.87 (t, J = 6.4 Hz, 2H), 2.29 (t, J = 6.2 Hz, 2H), 2.00–1.93 (m, 2H), 1.85–1.79 (m, 2H). 13C NMR (100 MHz, CDCl3): δ 188.5, 160.9, 159.6, 149.6, 147.3, 132.3, 131.7, 128.6, 128.3, 127.1, 117.4, 113.5, 108.2, 104.4, 76.3, 76.0, 75.7, 49.1, 48.7, 26.1, 20.0, 19.2. HRMS (ESI) (C22H19NO3Se) m/z: calculated for [M + Na]+: 448.0423; found [M + Na]+: 448.0372.
Preparation of test solution
The probe PSeC was dissolved in DMSO solvent to generate a 1 mM stock solution. Cys, Hcy, GSH, and other analytes were dissolved in deionized water to create a 10 mM stock solution.Cell imaging
HeLa cells were employed for fluorescence imaging studies. To evaluate the probe's ability to differentiate endogenous Hcy and GSH, cells were first incubated with PSeC (10 µM) for 30 min, followed by three washes with PBS (10 mM). For the detection of exogenous biothiols, endogenous thiols were removed by pretreating the cells with N-ethylmaleimide (NEM, 1 mM) for 30 min. The cells were then treated with Hcy or GSH for 30 min, washed three times with PBS, and incubated again with PSeC (10 µM) for 30 min. Fluorescence images were captured using a confocal laser scanning microscope.Results and discussion
Design of probe PSeC
Coumarin is widely used in the development of fluorescent probes owing to its excellent photophysical properties.49 Although many probes have been developed to discriminate Cys, Hcy, and GSH, most reported systems rely on high concentration organic solvent conditions for in vitro detection and generally fail to achieve clear discrimination, typically only distinguishing Cys/Hcy from GSH (Table S1).48,50–52 In this study, phenyl selenide was introduced as the recognition site due to its susceptibility to nucleophilic substitution with biothiols and its intrinsic fluorescence-quenching ability.42 Together with the aldehyde group, the probe features two distinct reactive sites, enabling it to generate unique fluorescence responses and reaction rates for different biothiols. Moreover, it operates effectively in PBS solution with only 0.1% DMSO, demonstrating high compatibility with aqueous biological environments. The synthetic route of probe PSeC is illustrated in Scheme 1. Compounds 2, 3, and PSeC were identified using 1H NMR, 13C NMR, and HRMS (Fig. S6–S11).Spectroscopic properties of PSeC
The spectroscopic response of PSeC toward Cys, Hcy, and GSH was investigated using UV-vis absorption and fluorescence spectroscopy. As shown in Fig. 1A, PSeC displayed an absorption peak at 500 nm and was almost non-fluorescent (ε = 2.94 × 104 L mol−1 cm−1, quantum yield (Φ) = 0.006), exhibiting an orange color under daylight. Upon addition of GSH, the solution turned pink, accompanied by a new absorption peak at 538 nm and a strong orange-red emission at 560 nm (Φ = 0.291), representing an approximately 90-fold enhancement compared to the free probe (Fig. 1B). In contrast, the addition of Cys or Hcy induced a color change to yellow-green, with the absorption peak blue-shifting to 406 nm and a strong green emission at 502 nm. The fluorescence quantum yields reached 0.057 for Cys and 0.284 for Hcy, corresponding to 29-fold and 142-fold enhancements relative to the probe alone. These results demonstrate that PSeC can distinguish between Cys/Hcy and GSH via dual excitation and emission channels.The probe PSeC exhibited distinctly different reaction kinetics toward the three biothiols (Fig. 2). While the fluorescence responses to Hcy and GSH reached equilibrium within 10–30 min, the reaction with Cys was significantly slower, requiring more than 1 h and resulting in a weaker signal. This kinetic disparity is quantified by the pseudo-first-order rate constants: 0.7652 min−1 for GSH, 0.2340 min−1 for Hcy, and 0.0363 min−1 for Cys, establishing the reactivity order of GSH > Hcy > Cys (Fig. S1). Due to its sluggish response, Cys was not emphasized in the following experiments.
Then, fluorescence titration tests were conducted using different channels. With the increase of Hcy concentration, the fluorescence intensity of the probe PSeC solution increased gradually at 502 nm under illumination at 406 nm (Fig. 3A). The saturation of the fluorescence intensity occurred gradually as the concentration of Hcy rose over 100 µM. In addition, as shown in Fig. 3B, there is a good linear correlation between the fluorescence intensity of the solution and Hcy concentration (ranging from 0 to 75 µM), and a low detection limit of 360 nM was calculated (3σ/k). After GSH was added, under illumination at 538 nm, the red channel's fluorescence intensity at 560 nm increased gradually as the GSH content increased (Fig. 3C). The solution's fluorescence intensity tended to achieve saturation at 35 µM. When the concentration is between 0 and 10 µM, there is a clear linear correlation between the fluorescence intensity of the solution at 560 nm and the GSH concentration (Fig. 3D), with a detection limit of 32 nM.
To evaluate the high selectivity of probe PSeC towards biothiols, the fluorescence changes of PSeC were observed after incubation with various analytes, including different amino acids (Ala, Asp, Glu, Phe, Gly, His, IIe, Leu, Lys, Met, Pro, Ser, Thr, Trp, Tyr, Val, Gln, Arg, Asn, Cys), common cations (Zn2+, Mg2+, Ca2+, Fe3+, NH4+, K+, Na+), common anions (Cl−, Br−, NO3−, SO42−, SO32−, CO32−, HCO3−), and oxidized species (HClO, H2O2). The probe PSeC solution has very little fluorescence on its own. However, biothiols (Hcy and GSH, 100 µM) can greatly increase the probe solution's fluorescence at 502 nm and 560 nm, respectively. However, after adding 200 µM of other interfering chemicals and collaborating for 1 h, the fluorescence intensity of the probe solution remained practically constant. After adding 100 µM biothiols and reacting for 30 min, the probe solution displayed strong fluorescence in the red channel at 560 nm and the green channel at 502 nm (Fig. 4, S2 and S3). Meanwhile, it was observed that the probe solution in the presence of Hcy did not show an increase in fluorescence intensity in the red channel, and the probe solution in the presence of GSH did not show any fluorescence intensity at all in the green channel. The inclusion of Cys resulted in only mild fluorescence being produced in the green channel. According to these findings, the probe PSeC can detect Hcy and GSH with great selectivity and is unaffected by other analytes, suggesting that it may be able to adapt to a variety of complicated settings found in living things. Additionally, the probe's dual channels allow it to distinguish between GSH and Hcy.
Two distinct excitation wavelengths were used to investigate the effects of changing pH values on biothiol detection in order to investigate the viability of employing PSeC in complicated environments. In the broad pH range of 1–14, the probe PSeC exhibited nearly negligible fluorescence when excited at a wavelength of 406 nm (Fig. 5). It demonstrated a notable increase in fluorescence in a pH 5–9 solution upon the addition of Hcy. The probe did not exhibit any fluorescence in the pH 1–14 solution when excited at 538 nm. However, in the broad pH range of 6–9, the probe demonstrated a notable fluorescence response to GSH. As a result, the PSeC probe can function well at pH values that are physiological.
 | ||
| Fig. 5 Fluorescence signal changes of the probe PSeC (10 µM) treated with 100 µM Hcy and GSH at different pH values for 30 min. (A) λex = 406 nm, λem = 502 nm; (B) λex = 538 nm, λem = 560 nm. | ||
To elucidate the response mechanism between the probe and Cys/Hcy/GSH, mass spectrometry (MS) analysis was performed on the reaction mixture of PSeC and biothiols in MeOH solution. According to previous studies, Cys and Hcy typically undergo an initial nucleophilic substitution followed by intramolecular rearrangement, attributed to the extremely short spatial distance between their NH2 and SH groups.42–44 In contrast, our system exhibited only nucleophilic substitution and subsequent cyclization. The proposed intermediates 2a and 2b were generated through nucleophilic substitution of PSeC by Cys and Hcy. Instead of proceeding through the rearrangement step, the NH2 group attacked the aldehyde functionality, yielding the cyclic products PSeC-Cys and PSeC-Hcy (Scheme 2), consistent with the mechanism reported by Cao et al.53 For GSH, which has greater spatial separation between –SH and –NH2 groups, rearrangement did not occur. Instead, the thiol group substituted the selenide at the coumarin 4-position to produce thiocoumarin proposed intermediate 1a, which subsequently formed an imine product (PSeC-GSH) exhibiting red fluorescence. The observed MS peaks at m/z 371.3, 385.5, 557.1, and 579.7 corresponded to [PSeC-Cys + H]+, [PSeC-Hcy + H]+, [PSeC-GSH + H]+, and [PSeC-GSH + Na]+, respectively (Fig. S4), confirming the proposed mechanisms. Future work will include fluorescence lifetime measurements to elucidate the detailed emission mechanisms.
Cell imaging
Encouraged by the above findings, the potential of PSeC for imaging endogenous and exogenous biothiols in living cells was evaluated. The cytotoxicity of PSeC was first assessed using a CCK-8 assay in HeLa cells after 24 h incubation, which revealed low cytotoxicity and good cell permeability (Fig. S5). Cells incubated without PSeC exhibited negligible fluorescence in either channel (Fig. 6). After being incubated with the PSeC probe for 30 min, the cells showed considerable fluorescence in the red channel for GSH detection and high fluorescence in the green channel for Hcy detection. When cells were pretreated with N-ethylmaleimide (NEM, 1 mM) to block endogenous thiols, no fluorescence was detected in either channel, confirming that PSeC successfully captured endogenous Hcy and GSH. To evaluate detection of exogenous biothiols, NEM-pretreated cells were incubated with Hcy or GSH followed by PSeC staining. As shown in Fig. 7, cells treated with Hcy exhibited strong green fluorescence but minimal red emission, whereas GSH-treated cells showed intense red fluorescence and negligible green signal. These results confirm that PSeC can selectively distinguish Hcy and GSH in living cells via dual-channel imaging.Time-dependent imaging further revealed that dual-channel fluorescence appeared as early as 2 min after probe incubation and increased gradually over time (Fig. S6). Moreover, the fluorescence intensities of both channels showed concentration-dependent enhancement (Fig. S7). Collectively, these results demonstrate that PSeC enables rapid, selective, and simultaneous visualization of intracellular Hcy and GSH through dual-excitation and dual-emission fluorescence imaging.
Conclusions
A coumarin-based probe, PSeC, has been developed for the selective detection of biothiols. The probe itself is non-fluorescent, but it can enable discrimination among Cys, Hcy, and GSH by emitting distinct fluorescence signals and exhibiting different reaction kinetics, owing to its dual recognition sites. PSeC demonstrates excellent selectivity for GSH and Hcy, along with a low detection limit. Cell-based assays confirmed its excellent biocompatibility and low cytotoxicity. Moreover, PSeC successfully differentiates between endogenous and exogenous Hcy and GSH in live cells through dual-channel fluorescence imaging.Author contributions
Xi-Xi Wu: writing – original draft, methodology, investigation, formal analysis, data curation. Chao-Wen Guo: validation, data curation. Wen-Yu Lu: supervision, conceptualization. Hui-Jing Li and Yan-Chao Wu: writing – review & editing, supervision, resources, funding acquisition, conceptualization. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The datasets supporting the conclusions of this article are included within the article and its additional files.Supplementary information (SI): data analysis methods, comparison of fluorescent probes based on coumarin for biothiols, time-dependent characteristics, selective fluorescence spectroscopy analysis, cell imaging research and compound characterization are included. See DOI: https://doi.org/10.1039/d5ay01742d.
Acknowledgements
This work was supported by Medical Research Project in Chengdu of Sichuan Province (2025268), and Shandong Provincial Natural Science Foundation of China (ZR2025LH85, ZR2025QC1335).References
- H. Han, F. Wang, J. J. Chen, X. X. Li, G. Q. Fu, J. W. Zhou, D. S. Zhou, W. Wu and H. M. Chen, J. Alzheimer's Dis., 2021, 82, 527–540 Search PubMed.
- A. P. A. dos Santos, J. K. da Silva, J. M. Neri, A. C. O. Neves, D. F. de Lima and F. G. Menezes, Org. Biomol. Chem., 2020, 18, 9398–9427 RSC.
- J. Raut and P. Sahoo, Mini-Rev. Org. Chem., 2021, 18, 867–884 CrossRef CAS.
- T. Rehman, M. A. Shabbir, M. Inam-Ur-Raheem, M. F. Manzoor, N. Ahmad, Z. W. Liu, M. H. Ahmad, A. Siddeeg, M. Abid and R. M. Aadil, Food Sci. Nutr., 2020, 8, 4696–4707 CrossRef CAS PubMed.
- J. H. Gao, Y. F. Tao, N. N. Wang, J. L. He, J. Zhang and W. L. Zhao, Spectrochim. Acta, Part A, 2018, 203, 77–84 CrossRef CAS.
- M. Prendecki, J. Florczak-Wyspianska, M. Kowalska, J. Ilkowski, T. Grzelak, K. Bialas, M. Wiszniewska, W. Kozubski and J. Dorszewska, Oncotarget, 2018, 9, 35207–35225 CrossRef.
- H. S. Jung, X. Q. Chen, J. S. Kim and J. Yoon, Chem. Soc. Rev., 2013, 42, 6019–6031 RSC.
- J. E. Dominy and M. H. Stipanuk, Nutr. Rev., 2004, 62, 348–353 Search PubMed.
- X. Li, M. Liu, Q. Y. Yi, M. Yang, X. Y. Zhang, Y. Shi, J. J. Huang, Q. Liu, J. M. Jiang, T. B. Wei, M. Wang and J. Y. Wang, Dyes Pigm., 2024, 224, 112031 CrossRef CAS.
- H. J. Deng, Z. T. Wu, Z. X. Zhao, L. Zhu, M. G. Tang, R. J. Yu and J. Y. Wang, Talanta, 2021, 231, 122331 CrossRef CAS.
- G. X. Yin, Y. B. Gan, H. M. Jiang, T. Yu, M. L. Liu, Y. Y. Zhang, H. T. Li, P. Yin and S. Z. Yao, Anal. Chem., 2021, 93, 9878–9886 CrossRef CAS PubMed.
- J. L. Guéant, R. M. Guéant-Rodriguez, A. Oussalah, S. Zuily and I. Rosenberg, Thromb. Haemostasis, 2023, 123, 270–282 CrossRef PubMed.
- A. Tanweer, A. Bano, W. Fatima and H. Javed, Adv. Life Sci., 2020, 8, 1–7 CAS.
- X. X. Liu, J. W. Zhang, M. R. Xia, J. R. Liu and S. Jiang, Exp. Ther. Med., 2019, 17, 1395–1399 CAS.
- M. A. Robbins, M. F. Elias, M. M. Budge, S. L. Brennan and P. K. Elias, Clin. Chem. Lab. Med., 2005, 43, 1101–1106 CAS.
- L. Flohé, Biochim. Biophys. Acta, 2013, 1830, 3139–3142 CrossRef.
- G. Bjorklund, M. D. Dosa, M. Maes, M. Dadar, R. E. Frye, M. Peana and S. Chirumbolo, Pharmacol. Res., 2021, 166, 105437 CrossRef.
- J. S. Stamler and A. Hausladen, Nat. Struct. Biol., 1998, 5, 247–249 Search PubMed.
- S. S. Pei, M. Minhajuddin, K. P. Callahan, M. Balys, J. M. Ashton, S. J. Neering, E. D. Lagadinou, C. Corbett, H. B. Ye, J. L. Liesveld, K. M. O'Dwyer, Z. Li, L. Shi, P. Greninger, J. Settleman, C. Benes, F. K. Hagen, J. Munger, P. A. Crooks, M. W. Becker and C. T. Jordan, J. Biol. Chem., 2013, 288, 33542–33558 Search PubMed.
- D. M. Townsend, K. D. Tew and H. Tapiero, Biomed. Pharmacother., 2003, 57, 145–155 CrossRef CAS.
- X. Chen, Y. Zhou, X. J. Peng and J. Yoon, Chem. Soc. Rev., 2010, 39, 2120–2135 Search PubMed.
- Y. Zhou and J. Yoon, Chem. Soc. Rev., 2012, 41, 52–67 Search PubMed.
- L. Y. Niu, Y. Z. Chen, H. R. Zheng, L. Z. Wu, C. H. Tung and Q. Z. Yang, Chem. Soc. Rev., 2015, 44, 6143–6160 RSC.
- X. Lin, Y. L. Hu, D. L. Yang and B. Chen, Dyes Pigm., 2020, 174, 107956 CrossRef CAS.
- Y. Yang, T. Zhou, M. Jin, K. Zhou, D. Liu, X. Li, F. Huo, W. Li and C. Yin, J. Am. Chem. Soc., 2020, 142, 1614–1620 Search PubMed.
- L. Wang, J. B. Wang, S. Xia, X. X. Wang, Y. T. Yu, H. W. Zhou and H. Y. Liu, Talanta, 2020, 219, 121296 Search PubMed.
- X. B. Wang and D. T. Zhang, Sens. Actuators, B, 2017, 241, 327–334 CrossRef CAS.
- J. B. Chao, J. M. Zhao, Y. B. Zhang, F. J. Huo and C. X. Yin, Sens. Actuators, B, 2021, 346, 130569 Search PubMed.
- Y. L. Zheng, Z. H. Chai, W. Tang, S. Yan, F. Dai and B. Zhou, Sens. Actuators, B, 2021, 330, 129343 Search PubMed.
- X. Zhang, L. W. Zhang, X. Y. Wang, X. Y. Han, Y. Huang, B. W. Li and L. X. Chen, J. Hazard. Mater., 2021, 419, 126476 Search PubMed.
- C. M. Han, H. R. Yang, M. Chen, Q. Q. Su, W. Feng and F. Y. Li, ACS Appl. Mater. Interfaces, 2015, 7, 27968–27975 Search PubMed.
- L. H. Zhao, X. He, D. Li, S. M. Xu, Y. B. Huang, X. L. Li, X. H. Wang, Y. Sun, P. Y. Ma and D. Q. Song, J. Mater. Chem. B, 2020, 8, 7652–7658 RSC.
- S. T. Cai, Q. C. Liu, C. Liu, S. He, L. C. Zhao, X. S. Zeng and J. Gong, J. Mater. Chem. B, 2022, 10, 1265–1271 Search PubMed.
- K. S. Lee, T. K. Kim, J. H. Lee, H. J. Kim and J. I. Hong, Chem. Commun., 2008, 6173–6175, 10.1039/b814581d.
- P. Wang, J. Liu, X. Lv, Y. L. Liu, Y. Zhao and W. Guo, Org. Lett., 2012, 14, 520–523 Search PubMed.
- X. Wang, J. Z. Lv, X. Y. Yao, Y. Li, F. Huang, M. M. Li, J. Yang, X. Y. Ruan and B. Tang, Chem. Commun., 2014, 50, 15439–15442 RSC.
- M. X. Jia, L. N. A. Wei, Y. X. Lu, R. Q. Zhang, Q. L. Chen, W. J. Xia, Y. Liu, F. Li and Y. Zhou, RSC Adv., 2022, 12, 2668–2674 RSC.
- B. Tang, Y. L. Xing, P. Li, N. Zhang, F. B. Yu and G. W. Yang, J. Am. Chem. Soc., 2007, 129, 11666–11667 Search PubMed.
- B. C. Zhu, X. L. Zhang, H. Y. Jia, Y. M. Li, S. T. Chen and S. C. Zhang, Dyes Pigm., 2010, 86, 87–92 Search PubMed.
- J. Liu, Y. Q. Sun, Y. Y. Huo, H. X. Zhang, L. F. Wang, P. Zhang, D. Song, Y. W. Shi and W. Guo, J. Am. Chem. Soc., 2014, 136, 574–577 Search PubMed.
- H. B. Liu, M. You, X. Y. Feng, J. L. Chen, B. Li, Z. G. Gao, H. Z. Xing, Z. J. Cong, B. J. Zhou, J. Zhu and M. J. Jin, Sens. Actuators, B, 2024, 400, 134870 Search PubMed.
- Y. Kim, S. V. Mulay, M. Choi, S. B. Yu, S. Jon and D. G. Churchill, Chem. Sci., 2015, 6, 5435–5439 Search PubMed.
- Y. F. Huang, Y. B. Zhang, F. J. Huo, J. B. Chao, F. Q. Cheng and C. X. Yin, J. Am. Chem. Soc., 2020, 142, 18706–18714 Search PubMed.
- X. B. Wang, H. J. Li, C. Liu, Y. X. Hu, M. C. Li and Y. C. Wu, Anal. Chem., 2021, 93, 2244–2253 Search PubMed.
- G. X. Yin, T. T. Niu, Y. B. Gan, T. Yu, P. Yin, H. M. Chen, Y. Y. Zhang, H. T. Li and S. Z. Yao, Angew. Chem., Int. Ed., 2018, 57, 4991–4994 Search PubMed.
- G. X. Yin, T. T. Niu, T. Yu, Y. B. Gan, X. Y. Sun, P. Yin, H. M. Chen, Y. Y. Zhang, H. T. Li and S. Z. Yao, Angew. Chem., Int. Ed., 2019, 58, 4557–4561 Search PubMed.
- K. M. Xiong, F. J. Huo, J. B. Chao, Y. B. Zhang and C. X. Yin, Anal. Chem., 2019, 91, 1472–1478 CrossRef CAS PubMed.
- X. G. Chen, Y. Mei and Q. H. Song, J. Mater. Chem. B, 2022, 10, 1272–1280 Search PubMed.
- D. Gupta, E. Guliani and K. Bajaj, Top. Curr. Chem., 2024, 382, 16 CrossRef CAS PubMed.
- S. V. Mulay, Y. Kim, M. Choi, D. Y. Lee, J. Choi, Y. Lee, S. Jon and D. G. Churchill, Anal. Chem., 2018, 90, 2648–2654 Search PubMed.
- G. Ding, X. C. Wang, D. R. Luo, S. Q. Meng, L. L. Zhou, Y. J. Fan, C. X. Ling-hu, J. P. Meng, W. T. Si, Q. Z. Chen and R. M. Bi, Dyes Pigm., 2023, 208, 110762 Search PubMed.
- X. L. Xue, Y. Wang, H. Zhang, S. J. Chen, S. Y. Niu, L. Y. Cui, K. P. Wang and Z. Q. Hu, Spectrochim. Acta, Part A, 2023, 292, 122410 CrossRef CAS PubMed.
- C. Cao, Y. Feng, H. Li, Y. Yang, X. R. Song, Y. Z. Wang, G. L. Zhang, W. Dou and W. S. Liu, Talanta, 2020, 219, 121353 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |