Multifunctional Bi2TeO5:Er3+ with dual-stimuli photochromism and switchable upconversion for optical security
Asad
Ullah
a,
Asif Ali
Haider
 b,
Jiayan
Liao
b,
Jiayan
Liao
 *c,
Xue
Bai
a,
Anjun
Huang
*c,
Xue
Bai
a,
Anjun
Huang
 a,
Imran
Khan
a,
Yangke
Cun
a,
Yuewei
Li
c,
Yingfan
Li
c,
Zhiguo
Song
a,
Imran
Khan
a,
Yangke
Cun
a,
Yuewei
Li
c,
Yingfan
Li
c,
Zhiguo
Song
 a,
Jianbei
Qiu
a,
Jianbei
Qiu
 ad and
Zhengwen
Yang
ad and
Zhengwen
Yang
 *ad
*ad
aCollege of Materials Science and Engineering, Kunming University of Science and Technology, Kunming, 650093, P. R. China. E-mail: yangzw@kust.edu.cn
bSchool of Materials and Energy, Yunnan University, Kunming, 650091, China
cInstitute for Biomedical Materials and Devices (IBMD), Faculty of Science, University of Technology Sydney, NSW 2007, Australia. E-mail: jiayan.liao@uts.edu.au
dSouthwest United Graduate School, Kunming, 650092, China
First published on 8th October 2025
Abstract
Photochromic luminescent materials are increasingly vital in optical data storage, anti-counterfeiting, and forensic technologies due to their reversible color change and emission tunability. However, most suffer from limited color contrast, single-mode transitions, and poor functional integration. Here, we report a dual-stimuli responsive Bi2TeO5:Er3+ ceramic that overcomes these limitations by combining dual-mode photochromism with wavelength-tunable upconversion luminescence (UCL) in a single system. Upon 365 nm UV exposure, the material exhibits a two-stage color change from light pink to yellow (3 min) and black (10 min) with a high contrast of 55.2%. X-ray irradiation induces a distinct light pink-to-yellow shift (33.3%) over 15 minutes. Both transitions are fully reversible under 532 nm laser bleaching with ∼99% recovery. The ceramic also displays efficient UCL: green emission under 980 nm excitation (96.4% modulation). Furthermore, the material enables rapid latent fingerprint visualization within 3 minutes and demonstrates excellent performance in powder-based forensic testing. This integrated system of dual-mode photochromism and reversible luminescence presents a versatile platform for multi-modal optical security technologies.
1. Introduction
Anti-counterfeiting technologies are critical to modern society, employing sophisticated methods such as fingerprint authentication systems to safeguard assets ranging from currency and identification documents to pharmaceuticals and food products.1 Beyond economic losses, counterfeit goods—particularly in the pharmaceutical sector—pose severe risks to public health and safety. The increasing demand for secure authentication has positioned optical information technologies at the forefront of anti-counterfeiting strategies.2,3 Photochromic materials, which reversibly change color under external stimuli,4–6 have emerged as promising candidates for optical data storage, dynamic displays, security labeling, and biometric identification.7–12 They offer unique advantages, including high data density, low power consumption, and long-term durability.13–16 While both organic and inorganic materials exhibit photochromism,17–19 early research focused primarily on organic molecules (e.g., spiropyrans,20 azobenzenes,21 and diarylethenes22,23) due to their structural tunability.24–26 However, inorganic photochromic systems have proven to be more suitable for long-term photonic applications, owing to their facile synthesis, superior thermal stability,27 environmental robustness,28,29 and particular suitability for long-term photonic applications.30 Despite these advances, a critical challenge remains: the development of single-phase materials that simultaneously exhibit dual-mode photochromism and tunable luminescence. Such multifunctional systems would enable more sophisticated security features and advanced optical data encoding, yet most existing materials still offer either photochromic or luminescence functionality in isolation, limiting their practical utility.31Inorganic photochromic materials are generally classified into three categories. The first includes transition metal oxides (e.g., WO3 and TiO2), which show single-color photochromism under UV or visible light.32–34 The second comprises dual-mode materials responsive to multiple stimuli, such as Ba2NaNb5O15:Er3+ (ref. 35) and CaAl2Si2O8:Tb3+ (ref. 36), capable of reversible changes under both UV and X-ray irradiation. Materials like SrBi4Ti4O15:Er3+ (ref. 37) and Sr2SnO4:Yb3+/Ho3+ (ref. 38) ceramics further integrate dual-luminescence features with photochromism. The third group includes multicolor systems like SrY2O4:Bi3+,Er3+ (ref. 39) and Ba5(PO4)3Cl:Eu2+,Ce3+,F− (ref. 40), enabling complex multi-state responses for advanced optical coding.
Despite these developments, most photochromic systems remain limited by single-functionality, poor color contrast, or lack of reversibility.41 Many require the combination of multiple materials to achieve multifunctional performance, which complicates device design and limits integration. Therefore, developing single-phase materials capable of exhibiting dual-mode photochromism and tunable luminescence remains an urgent need.42
At the core of photochromism in inorganic systems is the presence of crystal defects—particularly oxygen vacancies—which act as color centers.43 These defects trap and release charge carriers under irradiation, inducing changes in optical absorption and emission.44 Materials with stable crystal frameworks and high defect tolerance are well-suited to support multiple color-switching states while maintaining structural integrity across repeated cycles.
Beyond data storage, photoresponse materials are also highly relevant to forensic science. Latent fingerprint analysis is a foundational tool in law enforcement, typically conducted using metal or magnetic powders. However, these conventional techniques often struggle with poor contrast, surface contamination, or interference under ambient light.45 Recent innovations aim to improve fingerprint visualization using photochromic and luminescent nanomaterials that enable fast, high-contrast imaging on various surfaces.46–48 Our previous work on WO3 (ref. 49) and MoO3 (ref. 50) based ceramics achieved a reversible color change for fingerprint detection but required long activation times—30 and 10 minutes, respectively.
To address these shortcomings, we now report a new Bi2TeO5:Er3+ (BTO:Er) ceramic that offers significantly faster and multifunctional optical responses. The BTO:Er system introduced in this study is a multifunctional ceramic that exhibits dual-stimuli responsive photochromism triggered by either UV or X-ray radiation and switchable upconversion luminescence under near-infrared excitation. Under UV light (365 nm), it exhibits a two-step color transformation from light pink to yellow (3 minutes), and then to black (10 minutes), with a remarkable contrast of 55.2%. Under X-ray exposure, it transforms from light pink to yellow within 15 minutes (33.3% contrast). Both transitions are fully reversible upon exposure to 532 nm green laser light within 4 minutes, with near-complete recovery. Moreover, the material shows excitation-wavelength-dependent UCL: green emission under 980 nm with a modulation efficiency of 96.4%. These emissions remain robust in both UV and X-ray photochromic states, enabling integrated readout and security authentication. Notably, this material supports both direct ceramic-based fingerprint development and conventional powder-based techniques, offering dual-mode utility for practical forensic use. The BTO:Er ceramic allows rapid fingerprint detection through both direct surface imaging and powder-based application, achieving visualization within 3 minutes. The integration of dual-mode photochromism and switchable luminescence provides a multifunctional platform for applications in optical data storage, anti-counterfeiting, and forensic imaging.
2. Experimental
2.1 Sample preparation
Bi2TeO5:Er3+ ceramics were synthesized via a conventional solid-state reaction method using high-purity Bi2O3 (99.90%), TeO2 (99.90%), and Er2O3 (99.99%) as starting materials. Er3+ was introduced at varying molar concentrations (x = 0, 0.5, 1, 2, 3, and 4 mol%). The precursors were weighed stoichiometrically, mixed with ethanol, and ground in an agate mortar for 35 minutes to ensure homogeneity. After drying, the mixtures were calcined in alumina crucibles at 750 °C for 3 hours in air, followed by natural cooling to room temperature. The calcined powders were re-ground for 25 minutes and pressed into 18 mm diameter discs using an electric press (YLJ-60TAS), with the addition of 2–3 drops (0.1–0.15 mL) of polyvinyl alcohol (PVA) solution as a binder to 1.5 g of Bi2TeO5:Er3+ powder. The discs were then dried at 70 °C for 35 minutes to enhance rigidity. Photochromic properties were evaluated by irradiating the samples with 365 nm UV light (12 W) and X-rays. Photobleaching was performed using a 532 nm laser (2.2 W cm−2, 5 mm beam diameter), with observation under an optical microscope. The ceramic stage was adjustable to allow controlled variation of laser exposure times across different regions of the samples. To monitor thermal effects during bleaching, we used a thermal imaging camera (UNI-T Pro, UTi260B) to measure the ceramic's surface temperature during 532 nm laser (2.2 W) exposure.2.2 Crystal structure
The phase composition of Bi2TeO5:Er3+ ceramics was analyzed using an X’Pert3 powder X-ray diffractometer (Malvern Panalytical). X-ray-induced photochromic behavior was further examined with a Bruker D8 focusing diffractometer equipped with Cu-Kα radiation (λ = 0.15405 nm) and an Amptek X-ray tube. The morphology and elemental distribution were assessed via scanning electron microscopy (SEM) using both the Hitachi SU8000 and ZEISS GeminiSEM 300 systems. Diffuse reflectance spectra of pure and Er3+-doped Bi2TeO5 samples were recorded using a HITACHI U-4100 spectrophotometer. Upconversion luminescence (UCL) spectra were collected using a HITACHI F-7000 spectrophotometer under 980 nm excitation, while fluorescence decay measurements were performed with an Edinburgh FLS1000 spectrometer. Oxygen vacancy analysis was carried out using electron paramagnetic resonance (EPR, Bruker EMXplus-6/1). The surface chemical composition and oxidation states were examined using X-ray photoelectron spectroscopy (XPS, Thermo Scientific K-Alpha). High-resolution images of photochromic logos were captured with a Nikon D850 camera and filter glass, and long-exposure photography is used to highlight surface features and color contrast in photochromic states.Three key applications are demonstrated: optical storage, anti-counterfeiting, and a dual-mode fingerprint detection system. Detailed mechanisms for both fingerprint detection methods (ceramic-based and powder-based on glass) and the flower-pattern inscription for anti-counterfeiting are provided in the SI.
3. Results and discussion
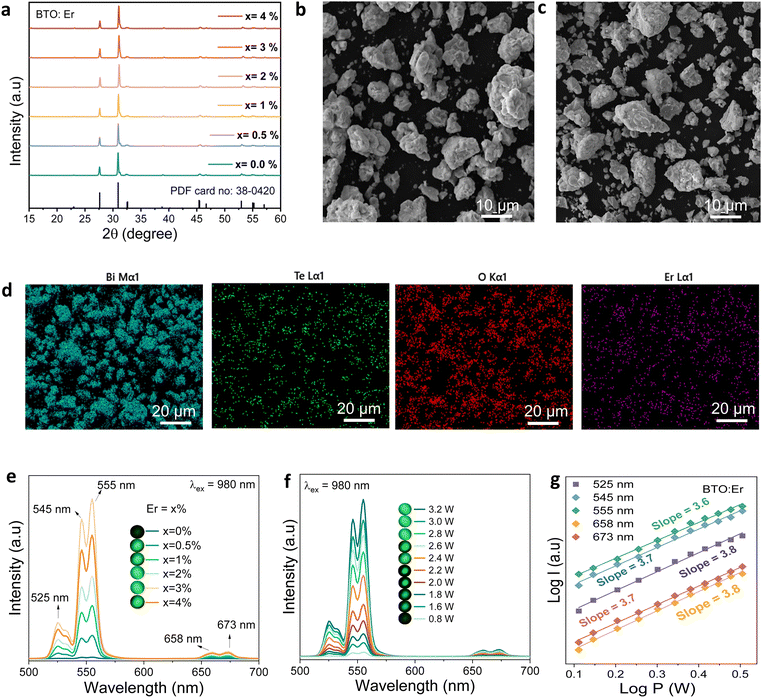
X-ray diffraction (XRD) analysis was conducted on both undoped and Er3+-doped Bi2TeO5 ceramics (Er3+ = 0, 0.5, 1, 2, 3, and 4 mol%) synthesized via a solid-state reaction at 750 °C for three hours. As shown in Fig. 1(a), all samples exhibit a single-phase crystalline structure matching the standard Bi2TeO5 pattern (PDF no. 38-0420), confirming phase purity across all doping levels. SEM micrographs in Fig. 1(b and c) reveal changes in the morphology with Er3+ doping, while EDS elemental mapping in Fig. 1(d) shows uniform distributions of Bi (cyan), Te (green), O (red), and Er (purple), confirming successful and homogeneous incorporation of Er3+ ions into the BTO lattice without detectable segregation or clustering.Photoluminescence (PL) describes light emission from materials following photon absorption. When the emitted radiation has a shorter wavelength (higher energy) than the excitation source, the process is termed down-conversion luminescence (or downshifting). Conversely, emission at longer wavelengths (lower energy) constitutes up-conversion luminescence (anti-Stokes PL), a nonlinear optical process requiring multiple photon absorption.51 The upconversion (UC) emission behavior of BTO:Er was examined under 980 nm excitation. A well-established four-photon process drives the observed green and red emissions. In this mechanism, sequential absorption of low-energy photons excites electrons to higher energy states, resulting in visible emission. As shown in Fig. 1(e), the green UC peaks at 525, 545, and 555 nm originate from the 2H11/2 → 4I15/2 and 4S3/2 → 4I15/2 transitions of Er3+. The red emissions at 658 and 673 nm are attributed to the 4F9/2 → 4I15/2 transition of Er3+.52 UCL intensity exhibits a concentration-dependent trend: it increases with Er3+ content up to 3 mol% and then declines due to concentration quenching. Therefore, Bi2TeO5:3 mol% Er3+ (BTO:Er) was selected for further optical investigations.
A power-dependent study under 980 nm excitation (Fig. 1(f)) shows that both green and red UCL bands vary consistently with laser power (0.8–3.2 W). The relationship follows the power law:53
| I ∝ Pn | (1) |
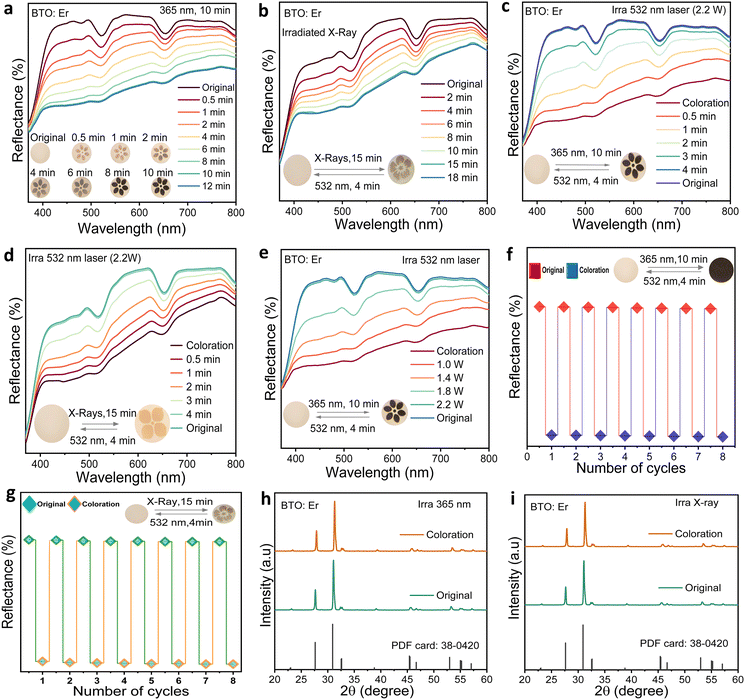
Both undoped BTO and BTO:Er ceramics exhibit distinct photochromic behavior when exposed to 365 nm UV light or X-ray irradiation (Fig. S1). Upon 10 minutes of UV exposure or 15 minutes of X-ray irradiation, both materials undergo observable color transformations, which are fully reversible upon 532 nm laser irradiation for 4 minutes. Diffuse reflectance spectra reveal significant decreases in reflection intensity across the visible range (370–800 nm) following both UV and X-ray exposure, confirming strong photochromic responses under both stimuli. To quantify the photo-induced changes, the absorption contrast (ΔRabs) was calculated using the following equation:55
| ΔRabs = [(T0 − Tc)/T0] × 100% | (2) |
Time-resolved reflectance spectra in Fig. 2(a) show that under 365 nm UV irradiation, BTO:Er transitions from light pink to yellow within 3 minutes and to black by 10 minutes. Two Er3+-related absorption bands at 521 nm and 653 nm emerge during this process. Under X-ray exposure (Fig. 3(b)), coloration saturates after 15 minutes, with a maximum ΔAbs of 33.3% at 460 nm, which is lower than that observed under UV—likely due to the reduced absorption cross-section and increased scattering effects of X-rays. The incorporation of Er3+ ions increases absorption efficiency by introducing intermediate energy levels, thereby facilitating more effective electron–hole pair separation and photo-induced carrier trapping under both stimuli.
The reversibility of photochromism in BTO:Er was demonstrated using 532 nm laser bleaching. Fig. 2(c and d) show a gradual increase in reflectance from 370 to 800 nm during bleaching, with full recovery of the original color state within 4 minutes. Increasing the laser power from 1.0 W to 2.2 W further accelerates this recovery, indicating that higher photon flux promotes a faster release of trapped electrons (Fig. 2(e)). After monitoring the surface temperature during the bleaching process with 532 nm laser irradiation (2.2 W), we observed a maximum of only 53 °C after a 5-minute exposure (Fig. S3). This relatively low temperature rise suggests that the bleaching process is not driven by thermal effects from the laser.
To assess durability, BTO:Er was subjected to repeated coloration–bleaching cycles using alternating 365 nm UV and X-ray exposures, followed by 532 nm laser bleaching. Reflectance at 460 nm remained stable across multiple cycles (Fig. 2(f and g)), confirming excellent photochromic repeatability. The color transformations also remained consistent: light pink ↔ black (UV) and light yellow ↔ full yellow (X-ray). XRD patterns before and after irradiation (Fig. 2(h and i)) show no peak shifts or new phases, demonstrating that the photochromic process does not alter the crystal structure. This stability confirms that the mechanism is electronic rather than structural.
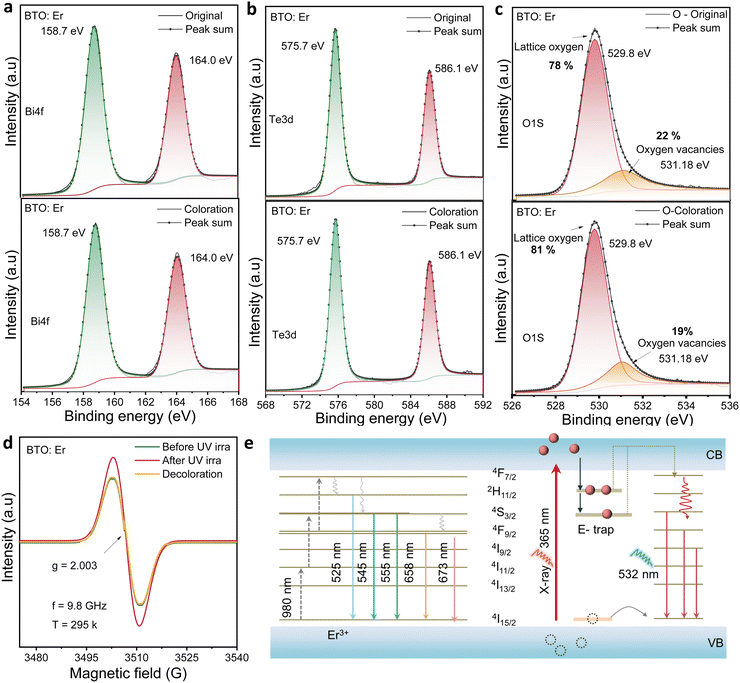
XPS survey spectra in Fig. S4 confirm the stable elemental composition (Bi, Te, O, and Er) in both BTO and BTO:Er after UV exposure. High-resolution XPS spectra of pure BTO in Fig. S5 show that Bi 4f and Te 3d peak positions remain unchanged, with the oxygen vacancy content decreasing slightly from 26% to 25% after 365 nm UV irradiation. In contrast, BTO:Er (Fig. 3(a–c) and Fig. S6) exhibits a more pronounced drop in the vacancy concentration—from 22% to 19%—while maintaining peak positions at 529.8 eV and 531.08 eV. A more pronounced decrease in the oxygen vacancy concentration is observed in the Er3+-doped BTO sample compared to the undoped sample after 365 nm UV irradiation for 10 min. This reduction in oxygen vacancies suggests that vacancy sites actively participate in electron trapping during the photochromic process. This supports the active participation and partial consumption of oxygen vacancies during photochromic transformation. EPR spectroscopy further validates this mechanism. Measurements taken before irradiation, after 10 minutes of UV exposure, and following 532 nm laser bleaching (Fig. 3d) reveal a distinct, reversible modulation in EPR signal intensity (g = 2.003), consistent with the formation and neutralization of electron-trapped oxygen vacancies. The increased EPR signal after UV irradiation indicates the generation of trapped electrons, while signal reduction after bleaching reflects electron release and vacancy state recovery. The apparent inconsistency between EPR and XPS results is resolved by understanding their different detection regions: EPR shows increased bulk oxygen vacancies from trapped photo-generated carriers. In contrast, XPS, being surface-sensitive, reveals a decrease in oxygen-deficient species because surface vacancies are partially compensated during the photochromic process. Thus, the two results are complementary rather than contradictory, consistent with previous studies distinguishing between bulk and surface vacancy behaviors.
The photochromic mechanism is summarized in Fig. 3e. Upon UV or X-ray exposure, electrons are excited from the valence band (VB) to the conduction band (CB). In the UV-induced dual-stage response, electrons are initially trapped in neutral vacancies (V0), generating a pink-to-yellow transition. Prolonged exposure induces additional electron trapping, forming F-color centers and leading to a yellow-to-black transformation. X-ray excitation, however, only triggers the initial transition due to its limited absorption efficiency. Simultaneously, upconversion luminescence (UCL) is suppressed due to reabsorption effects and energy transfer from Er3+ ions to defect sites.56 Subsequent 532 nm laser exposure releases trapped electrons, restoring both the optical absorption and luminescence properties. The enhanced photochromic performance of BTO:Er is attributed to synergistic effects: Er3+ ions act as electron traps, while oxygen vacancies mediate charge separation, resulting in more efficient and reversible color switching than in undoped BTO. The photochromic and bleaching behaviors of Er3+-doped Bi2TeO5 are attributed to a reversible color-center mechanism. Upon exposure to UV light or X-rays, electrons are excited from the valence band to the conduction band and are subsequently trapped at oxygen vacancy sites, forming color centers that produce a visible color change. Irradiation with a 532 nm laser then releases these trapped electrons, eliminating the color centers and restoring the material to its initial optical state.
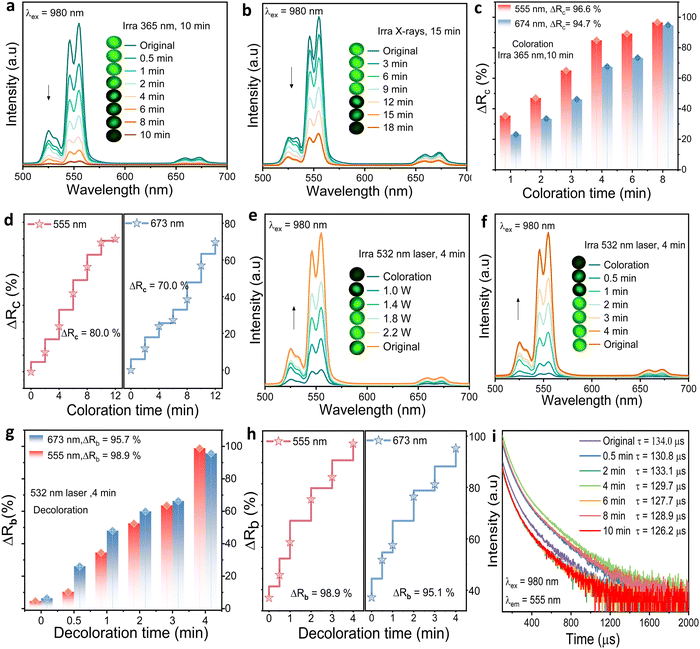
The BTO:Er ceramic demonstrates a strong correlation between its photochromic behavior and UCL properties, evidenced by the overlap between diffuse reflectance and UCL spectra. Under 980 nm excitation, both green and red UCL emissions are reversibly modulated due to reabsorption by photo-induced color centers. As shown in Fig. 4(a and b), prolonged exposure to 365 nm UV light or X-ray irradiation leads to a gradual decrease in UCL intensity, reflecting enhanced optical absorption.
The degree of UCL modulation is quantitatively expressed by the modulation rate (ΔRc), defined as
| ΔRc = [(I0 − Ic)/I0] × 100% | (3) |
During the bleaching process, UCL intensities recover as the reabsorption effect diminishes. Fig. 4(e) shows that increasing the 532 nm laser power (1.0–2.2 W) leads to enhanced recovery of both emission bands. After 4 minutes at 2.2 W, nearly complete UCL restoration is achieved, as shown in the spectral evolution (Fig. 4(f)). The bleaching efficiency (ΔRb) was calculated using the following expression:
| ΔRb = (Ib/I0) × 100% | (4) |
4. Multifunctional applications
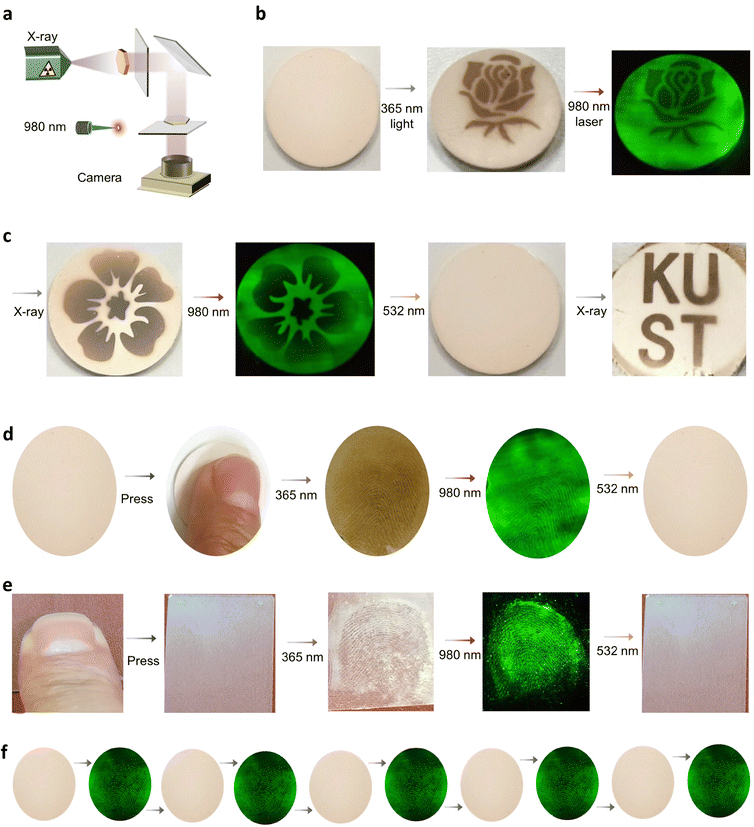
BTO:Er ceramics offer a multifunctional platform for advanced anti-counterfeiting, combining reversible photochromism with excitation-dependent UCL. This dual-mode authentication system operates via UV (365 nm) or X-ray exposure for pattern inscription, UCL-based readout, and controlled erasure using a 532 nm laser. As illustrated in Fig. 5(a), the protocol enables secure, rewritable pattern encoding through a mask-based approach. To demonstrate versatility, floral patterns were inscribed on the ceramic surface using either UV (10 min) or X-ray (15 min) exposure, as shown in Fig. 5(b). These patterns are visible under ambient light due to photochromism and further enhanced by green (980 nm) UCL emissions. All patterns are fully erasable within 4 minutes under 532 nm laser irradiation. This integration of multi-wavelength responsiveness, high UCL modulation, and full reversibility ensures layered security, making BTO:Er ceramics a robust and reusable solution for next-generation anti-counterfeiting technologies.The BTO:Er ceramic system holds significant application potential for advanced optical data storage, enabled by its dual-stimuli responsive photochromism and switchable UCL. As shown in Fig. 5(c), information can be written, read, and erased using UV (365 nm), X-ray, and 532 nm laser light, respectively. Photo-induced color transformations are driven by oxygen vacancy generation, allowing for dynamic and reversible encoding. In a practical demonstration, a flower pattern was inscribed on the ceramic surface using a hollow mask and irradiated with either UV or X-rays. Upon 980 nm laser excitation, the pattern exhibited vivid green UCL. Erasure was achieved in 4 minutes using 532 nm laser exposure. This reversible cycle showcases the material's suitability for rewritable optical memory.
The BTO:Er ceramic system enables dual-mode fingerprint detection, supporting both direct ceramic surface visualization and powder-based forensic analysis. The complete fingerprint acquisition, recognition, and erasure cycle are illustrated in Fig. 5d. The direct ceramic detection process follows three stages. First, finger contact leaves a latent print on the ceramic surface. Upon 365 nm UV light exposure (12 W, 3 min), the fingerprint becomes visible due to localized photochromic changes. The developed pattern remains stable after UV removal. A 980 nm laser scan enhances clarity within 20 seconds via UCL. The print is then erased by 532 nm laser irradiation (2.2 W, 4 min), fully resetting the surface.
In parallel, a powder-based method was developed for use on transparent glass substrates. A latent print is deposited, followed by the application of fine BTO:Er powder, which adheres selectively to moisture and oil residues on the ridges.57 After excess powder is removed, UV exposure (12 W, 3 min) reveals the print via the photochromic response of the particles. Pattern definition is further enhanced by 980 nm UCL imaging.
When a finger is pressed on a plain ceramic or glass surface without UV light, no visible fingerprint appears, indicating that the observed fingerprint phenomenon is not solely due to the presence of Er doping. The distinct color contrast in fingerprint regions arises from two synergistic mechanisms. First, organic components in fingerprint residues (such as amino acids, fatty acids, and urea) enhance localized thermal effects under 365 nm (12 W) UV irradiation, promoting the formation of oxygen vacancies in the BTO:Er ceramic matrix.58 Second, water molecules in sweat facilitate proton (H+) insertion into the BTO host lattice, further modifying its optical properties.49 The developed fingerprint can then be fully erased by exposure to a 532 nm laser for 4 minutes, completing the visualization–erasure cycle (Fig. 5e). Both detection approaches—direct ceramic-based and powder-based—enable high-contrast fingerprint visualization within 3 minutes, offering a significant speed advantage over previously reported lanthanide-based systems such as WO3 or MoO3, which typically require 10–30 minutes.
Our investigation provides a detailed examination of latent and UCL contrast fingerprints, revealing distinct hierarchical features. While Level-1 details primarily displayed loop patterns due to partial print quality, the analysis successfully resolved Level-2 minutiae such as ridge endings, bifurcations, and bridges, as well as Level-3 microscopic features like sweat pores (Fig. S8). The clear visualization of sweat pores—critical for personal identification—demonstrates the effectiveness of our imaging technique, despite some limitations in capturing complete Level-1 patterns.59
Additionally, pixel intensity profile analysis using ImageJ software confirmed a pronounced contrast between ridge and furrow regions in both ceramic-based and powder-based fingerprint methods (Fig. S9(a and b)). Ridges exhibited higher pixel values due to strong phosphor adhesion, resulting in intense green luminescence, whereas furrows appeared darker with lower pixel values due to minimal material adherence.60 When comparing substrates, fingerprints developed directly on ceramics showed higher ridge intensity than those on glass surfaces using the powder-dusting technique.
This speed advantage, combined with dual-mode flexibility, underscores the system's forensic utility. The durability and repeatability of BTO:Er for fingerprint detection were evaluated across five full cycles of writing and erasure. As shown in Fig. 5(f) and Fig. S10, both ceramic- and powder-based modes maintained reliable performance. Long-term stability testing further confirmed the durability of fingerprint visibility over extended periods. Fig. S11 confirms fingerprint visibility for over 240 days without significant degradation. Together, these results make BTO:Er ceramics a robust and practical solution for real-world forensic applications, offering a rapid response, multi-surface compatibility, and reusable detection capabilities.
5. Conclusions
In conclusion, we successfully synthesized a novel Bi2TeO5:Er3+ phosphor system via a simple solid-state reaction method, achieving integrated dual-mode photochromism and dual-wavelength upconversion luminescence within a single-phase ceramic. The material demonstrates distinct, reversible color transformations under both UV (365 nm) and X-ray irradiation, with high coloration contrasts of 55.2% and 33.3%, respectively, and excellent bleaching reversibility (up to 99% recovery). The green emission (modulation rate: 96.4%) under 980 nm excitation is sensitive to both photochromic switching and the excitation source. Furthermore, this system was evaluated using two modes of forensic detection: direct ceramic fingerprint visualization and powder-based fingerprint enhancement. Both methods achieved rapid detection within 3 minutes and demonstrated exceptional repeatability and long-term stability, outperforming traditional lanthanide-based systems in speed and versatility. Overall, this work highlights the synergistic interplay between defect-driven photochromism and Er3+-mediated luminescence, providing deeper insight into reversible optical modulation mechanisms. The combined features of easy fabrication, fast response, multicolor control, and reusability make Bi2TeO5:Er3+ a promising platform for next-generation applications in rewritable optical storage, anti-counterfeiting technologies, and high-resolution forensic imaging.Author contributions
Z. Y. and J. L. conceptualized, designed, and supervised the project. U. A. conducted the experiments with support from J. L. and Z. Y. Manuscript preparation and revisions were carried out collaboratively by A. U., J. L. and Z. Y., with all authors contributing to discussions and manuscript feedback.Conflicts of interest
The authors declare that they have no known competing financial interest or personal relationships that could have appeared to influence the work reported in this study.Data availability
All original data of this study are available from the corresponding authors upon reasonable request. The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5tc03067f.Acknowledgements
This work was supported by the Key Project of the National Natural Science Foundation of China-Yunnan Joint Fund (U2102215), the National Natural Science Foundation (52472002), the Science and Technology Project of Southwest Joint Graduate School of Yunnan Province (202302A0370008), the 2024 Industrial Innovation Talent Support Project (preparation of luminous materials, performance control and application in plateau agriculture) (YFGRC202407), the National Natural Science Foundation of High-End Foreign Expert Introduction Plan (G2022039008L), the Academician Workstation of Cherkasova Tatiana in Yunnan Province (202305AF150099), the Yunnan Province Major Science and Technology Special Plan (202302AB080005), the UTS Chancellor's Research Fellowship Program (J. L., PRO22-15457), and the National Health and Medical Research Council (J. L., 2025442).References
- J. Yadav, R. Mallick, S. K. Samal, M. Shankar, S. N. Achary and B. S. Naidu, J. Mater. Chem. C, 2025, 13, 9830 RSC.
- D. Gao, J. Gao, F. Gao, Q. Kuang, Y. Pan, Y. Chen and Z. Pan, J. Mater. Chem. C, 2021, 9, 16634 RSC.
- D. Gao, F. Gao, Q. Kuang, X. Zhang, Z. Zhang, Y. Pan, R. Chai and H. Jiao, ACS Appl. Nano Mater., 2022, 5, 9929 CrossRef CAS.
- Y. Hu, D. Gao, X. Zhang and S. Yun, Chem. Eng. J., 2025, 509, 161181 CrossRef CAS.
- Y. Niu, F. Wu, Y. Zhuo, J. Li, Q. Zhang, Y. Teng, X. Xie, H. Dong and Z. Mu, Ceram. Int., 2025, 51, 30597 CrossRef CAS.
- Pushpendra, S. Singh, S. Srinidhi, R. K. Kunchala, R. Kalia, S. N. Achary and B. S. Naidu, Cryst. Growth Des., 2021, 21, 4619 CrossRef CAS.
- Z. Zeng, B. Huang, X. Wang, L. Lu, Q. Lu, M. Sun, T. Wu, T. Ma, J. Xu and Y. J. Xu, Adv. Mater., 2020, 32, 2004506 CrossRef CAS PubMed.
- Z. Xu, P. Li, H. Sun and Q. Zhang, Ceram. Int., 2024, 50, 27661 CrossRef CAS.
- Q. Zhang, Y. Zhang, H. Sun, W. Geng, X. Wang, X. Hao and S. An, ACS Appl. Mater. Interfaces, 2016, 8, 34581–34589 CrossRef CAS PubMed.
- H. Zhao, J. Liao, S. Fu, Y. Zi, X. Bai, Y. Ci, Y. Zhang, X. Cai, Y. Li and Y. Cun, ACS Energy Lett., 2025, 10, 1235 CrossRef CAS.
- H. C. Byron, C. Swain, P. Paturi, P. Colinet, R. Rullan, V. Halava, T. Le Bahers and M. Lastusaari, Adv. Funct. Mater., 2023, 33, 2303398 CrossRef CAS.
- A. Ullah, A. A. Haider, I. Khan, Y. Cun, Y. Liu, P. Ren, Z. Song, J. Qiu, C. Tatiana and A. Huang, J. Am. Ceram. Soc., 2025, 108, e70079 CrossRef CAS.
- X. Yuan, E. Cui, K. Liu, F. Artizzu, X. Liao, J. Zhao, J. Tang, W. Sun, J. Liu and Y. Liu, J. Colloid Interface Sci., 2023, 641, 961 CrossRef CAS PubMed.
- Q. Zhang, X. Zheng, H. Sun, W. Li, X. Wang, X. Hao and S. An, ACS Appl. Mater. Interfaces, 2016, 8, 4789 CrossRef CAS PubMed.
- H. Zhao, Y. Cun, X. Bai, D. Xiao, J. Qiu, Z. Song, J. Liao and Z. Yang, ACS Energy Lett., 2022, 7, 2060 CrossRef CAS.
- F. Wang, R. Lyu, H. Xu, R. Gong and B. Ding, Responsive Mater., 2024, 2, e20240007 CrossRef CAS.
- S. Cao, Q. Chen, J. Liu, C. Wu, L. Li, J. Xu, G. Cheng and F. Gao, J. Eur. Ceram. Soc., 2020, 40, 6061 CrossRef CAS.
- J. Gong, P. Du, W. Li, G. Yuan, X. Mao and L. Luo, Laser Photonics Rev., 2022, 16, 2200170 CrossRef CAS.
- H. Al Sabea, L. Norel, O. Galangau, T. Roisnel, O. Maury, F. Riobé and S. Rigaut, Adv. Funct. Mater., 2020, 30, 2002943 CrossRef CAS.
- H. Wu, W. Wu, L. Hu, J. Zhu, Q. Li, Y. Gao, Y. Wei, G. Jiang and Y. Yang, J. Chem. Eng., 2023, 469, 143781 CrossRef CAS.
- A. Mukherjee, M. D. Seyfried and B. J. Ravoo, Angew. Chem., Int. Ed., 2023, 62, e202304437 CrossRef CAS PubMed.
- H. B. Cheng, S. Zhang, E. Bai, X. Cao, J. Wang, J. Qi, J. Liu, J. Zhao, L. Zhang and J. Yoon, Adv. Mater., 2022, 34, 2108289 CrossRef CAS PubMed.
- C. Wang, X. K. Ma, P. Guo, C. Jiang, Y. H. Liu, G. Liu, X. Xu and Y. Liu, Adv. Sci., 2022, 9, 2103041 CrossRef CAS PubMed.
- C. Li, H. Yan, L.-X. Zhao, G.-F. Zhang, Z. Hu, Z.-L. Huang and M.-Q. Zhu, Nat. Commun., 2014, 5, 5709 CrossRef CAS PubMed.
- J. Zhang, Q. Zou and H. Tian, Adv. Mater., 2013, 25, 378 CrossRef CAS PubMed.
- R. Pardo, M. Zayat and D. Levy, Chem. Soc. Rev., 2011, 40, 672 RSC.
- Q. Zhang, H. Sun, X. Wang, X. Hao and S. An, ACS Appl. Mater. Interfaces, 2015, 7, 25289 CrossRef CAS PubMed.
- Z. Yang, J. Du, L. I. Martin and D. Poelman, J. Eur. Ceram. Soc., 2021, 41, 1925 CrossRef CAS.
- T. Wei, F. Yang, B. Jia, C. Zhao, L. Liu, H. Zhang, Y. Zhang, J. Zhang, X. Yan and J. Yang, Ceram. Int., 2020, 46, 18507 CrossRef CAS.
- A. A. Haider, H. Zhao, Y. Zi, Z. Xu, X. Bai, Y. Cun, Y. Liu, H. Babeker, A. Saeed and Z. Song, Adv. Opt. Mater., 2023, 12, 2302265 CrossRef.
- Q. Du, J. Ueda, R. Zheng and S. Tanabe, Adv. Opt. Mater., 2023, 11, 2202612 CrossRef CAS.
- S. Wang, W. Fan, Z. Liu, A. Yu and X. Jiang, J. Mater. Chem. C, 2018, 6, 191 RSC.
- A. A. Haider, Y. Cun, X. Bai, Z. Xu, Y. Zi, J. Qiu, Z. Song, A. Huang and Z. Yang, J. Mater. Chem. C, 2022, 10, 6243 RSC.
- Y. Zhan, Z. Yang, Z. Xu, Z. Hu, X. Bai, Y. Ren, M. Li, A. Ullah, I. Khan and J. Qiu, Chem. Eng. J., 2020, 394, 124967 CrossRef CAS.
- Y. Hu, Y. Sun, S. Hou, D. Chen and Z. Yang, Chem. Eng. J., 2024, 495, 153600 CrossRef CAS.
- X. Bai, Y. Zhang, H. Zhao, Y. Zi, Z. Xu, A. Huang, Y. Cun, Y. Liu, Z. Song and J. Qiu, Adv. Sci., 2025, 2412986 CrossRef CAS PubMed.
- Y. Wu, X. Zhao, Z. Zhang, J. Xiang, H. Suo and C. Guo, Ceram. Int., 2021, 47, 15067 CrossRef CAS.
- Y. Mu, P. Du, W. Li and L. Luo, Ceram. Int., 2021, 47, 25037 CrossRef CAS.
- Q. Yu, F. Lu, L. Ma, P. Dai, F. Hu, H. Guo and R. Wei, Ceram. Int., 2024, 50, 15417 CrossRef CAS.
- W. Hu, Z. Qiu, Y. Bao, J. Du, J. Zhang, P. F. Smet, X.-D. Wen and S. Lian, ACS Energy Lett., 2024, 9, 2145 CrossRef CAS.
- F. Yang, B. Jia, T. Wei, C. Zhao, Q. Zhou, Z. Li, M. Du, M. Wang, Y. Liu and C. Xie, Inorg. Chem. Front., 2019, 6, 2756 RSC.
- Q. Zhang, X. Zheng, H. Sun, W. Li, X. Wang, X. Hao and S. An, ACS Appl. Mater. Interfaces, 2016, 8, 4789 CrossRef CAS PubMed.
- P. Li, L. Lu, X. Li, Z. Ma, W. Nie, L. Liu, X. Jian, W. Cao, B. Wang and Q. Zhang, J. Rare Earths, 2025 DOI:10.1016/j.jre.2025.01.010.
- H. Chen, Z. Xi, F. Guo, W. Long, X. Zhang, X. Li and P. Fang, Ceram. Int., 2024, 50, 4885 CrossRef CAS.
- P. Hazarika and D. A. Russell, Angew. Chem., Int. Ed., 2012, 51, 3524 CrossRef CAS PubMed.
- G. S. Sodhi and J. Kaur, Forensic Sci. Int., 2001, 120, 172 CrossRef CAS PubMed.
- H. Chen, R. Ma, Y. Chen and L.-J. Fan, ACS Appl. Mater. Interfaces, 2017, 9, 4908–4915 CrossRef CAS PubMed.
- M. Wang, M. Li, A. Yu, Y. Zhu, M. Yang and C. Mao, Adv. Funct. Mater., 2017, 27, 1606243 CrossRef PubMed.
- J. Ruan, Z. Yang, Y. Wen, M. Li, Y. Ren, J. Qiu, Z. Song and Y. Wang, Chem. Eng. J., 2020, 383, 123180 CrossRef CAS.
- M. Li, Z. Yang, Y. Wen, J. Ruan, Y. Ren, J. Qiu, Z. Song, Y. Wang and B. Liu, Adv. Mater. Technol., 2020, 5, 2070069 CrossRef CAS.
- Z. Huang, X. Li, T. Liang, B. Ren, X. Zhang, Y. Zheng, Q. Zhang, Z. Fang, M. Wu and M. Zulfiqar, Responsive Mater., 2024, 2, e20240019 CrossRef CAS.
- H. Sun, Q. Zhang, X. Wang and M. Gu, Ceram. Int., 2014, 40, 2581 CrossRef CAS.
- P. Pei, R. Wei, B. Wang, J. Su, Z. Zhang and W. Liu, Adv. Funct. Mater., 2021, 31, 2102479 CrossRef CAS.
- Y. Jiang, Y. Tong, S. Chen, W. Zhang, F. Hu, R. Wei and H. Guo, Chem. Eng. J., 2021, 413, 127470 CrossRef CAS.
- H. Zhao, Y. Li, C. Mi, Y. Zi, X. Bai, A. A. Haider, Y. Cun, A. Huang, Y. Liu and J. Qiu, InfoMat, 2024, 6, e12546 CrossRef CAS.
- K. Li, L. Luo, Y. Zhang, W. Li and Y. Hou, ACS Appl. Mater. Interfaces, 2018, 10, 41525 CrossRef CAS PubMed.
- A. A. Haider, X. Zhao, L. Liu, Z. Huang, J. Quan, Y. Tang, Y. Qin, X. Huang, Y. Shen and J. Zhu, J. Rare Earth, 2025 DOI:10.1016/j.jre.2025.02.001.
- A. Ullah, I. Khan, Y. Cun, Y. Liu, Z. Song, J. Qiu, T. G. Cherkasova, A. Huang, A. A. Haider and Z. Yang, Inorg. Chem. Front., 2024, 12, 144 RSC.
- J. Yadav, S. K. Samal, J. M. Saju, S. N. Achary and B. S. J. N. Naidu, Nanotechnol., 2025, 36, 195201 CrossRef CAS PubMed.
- A. A. Haider, X. Huang, J. Xie, Y. Qin, C. Jia, J. Jia, J. Chen, J. Zhang, W. Qian and J. Zhu, Mater. Today Chem., 2025, 45, 102684 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2025 |