Enhanced breakdown strength and polarization behavior in relaxor ferroelectric films via bidirectional design of defect engineering and heterogeneous interface construction
Shuo
Zhang
a,
Hua
Hao
 b,
Rui
Huang
b,
Minghe
Cao
b,
Rui
Huang
b,
Minghe
Cao
 b,
Zhonghua
Yao
b,
Zhonghua
Yao
 b and
Hanxing
Liu
b and
Hanxing
Liu
 *ab
*ab
aState Key Laboratory of Advanced Technology for Materials Synthesis and Processing, International School of Material Science and Engineering, Wuhan University of Technology, Wuhan, 430070, China. E-mail: lhxhp@whut.edu.cn
bState Key Laboratory of Advanced Technology for Materials Synthesis and Processing, School of Material Science and Engineering, Wuhan University of Technology, Wuhan, 430070, China
First published on 1st February 2025
Abstract
Electronic power systems require the development of dielectric capacitors that combine high energy storage density and high efficiency. However, there are some unavoidable defects such as oxygen vacancies in dielectrics, which seriously deteriorate the energy storage performance. Here, a bidirectional design of defect engineering and heterogeneous interfacial construction is conducted in Fe-doped 0.8Bi0.5Na0.5TiO3–0.2Bi3.25La0.75Ti3O12 relaxor ferroelectric films to reduce leakage current and improve polarization. Simultaneously, the heterogeneous interfacial construction hinders the mass transfer process and forms more interfaces, which impede the formation of electronic pathways. The synergistic action of bidirectional design provides a substantial increase in breakdown strength, combined with excellent dielectric properties and high temperature, frequency and cycle stability. Finally, a great balance between energy storage density (83.6 J cm−3) and efficiency (74.1%) is achieved for 2 mol% Fe-doped films. This work provides a strategy for designing advanced electrostatic capacitors through defect and interface regulation.
1. Introduction
Innovations in energy storage technologies are crucial for the efficient utilization of renewable energy sources and the mass production of electric vehicles. In recent years, dielectric capacitors with high power density and fast charge–discharge rates have received tremendous attention, which has a significance for advanced electronic power systems.1–3 However, the current energy density of dielectrics is far from adequate to meet the growing demand for electrical energy. Improvement in energy density will enhance the competitiveness of dielectric capacitors with other energy storage devices. In principle, the recoverable energy storage density (Wrec) and the efficiency (η) reflect the energy storage capacity of materials, which can be determined using the following equation: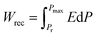 . The η is then obtained using Wrec/(Wrec + Wloss). Therefore, the high breakdown strength, large maximum polarization (Pmax) and low remnant polarization (Pr) are pivotal factors to achieve high energy storage density.4,5 The dielectric capacitors with superior performance are strongly demanded in electronic power systems. As an environmentally friendly perovskite with high polarization, Bi0.5Na0.5TiO3 (NBT) possesses immeasurable value and position in dielectric materials and has a high Curie temperature (TC = 320 °C) and typical ferroelectric square hysteresis loop with large remanent polarization (Pr = 38 μC cm−2) at room temperature.6 However, due to its element volatilization during the high temperature annealing process,7 there are some inevitable defects, which are considered to be detrimental to the material's properties in general. Indeed, the study about point defects in ferroelectric film capacitors has received increasing attention, especially the vacancies, which are most relevant to the dielectric properties.8 These defects may act as carriers to form electronic pathways, thus increasing the leakage conductance and reducing the resistivity, resulting in the decrease in breakdown strength, which hinders the improvement of the energy storage properties.9
. The η is then obtained using Wrec/(Wrec + Wloss). Therefore, the high breakdown strength, large maximum polarization (Pmax) and low remnant polarization (Pr) are pivotal factors to achieve high energy storage density.4,5 The dielectric capacitors with superior performance are strongly demanded in electronic power systems. As an environmentally friendly perovskite with high polarization, Bi0.5Na0.5TiO3 (NBT) possesses immeasurable value and position in dielectric materials and has a high Curie temperature (TC = 320 °C) and typical ferroelectric square hysteresis loop with large remanent polarization (Pr = 38 μC cm−2) at room temperature.6 However, due to its element volatilization during the high temperature annealing process,7 there are some inevitable defects, which are considered to be detrimental to the material's properties in general. Indeed, the study about point defects in ferroelectric film capacitors has received increasing attention, especially the vacancies, which are most relevant to the dielectric properties.8 These defects may act as carriers to form electronic pathways, thus increasing the leakage conductance and reducing the resistivity, resulting in the decrease in breakdown strength, which hinders the improvement of the energy storage properties.9
However, contrary to conventional intuition, the existence of defects in materials need not always constitute a disadvantage. In several cases, the modulation of defects contributes to improving the performance. For example, by doping aliovalent ions, specific defects and charge carriers can be obtained in the semiconductor substrate, enabling the construction of N/P-type semiconductors.10 In the field of energy storage ceramics, the modulation of ceramic properties by defects is not yet widely recognized. This is mainly due to the low defect formation energy of ceramic materials, as well as the coexistence of multiple defects, which include vacancies, interstitials, antisite defects, and other extrinsic defects and so on. The modulation achieved by defects in the material can effectively avoid the degradation of the properties by the defects, but also can reduce the manufacturing cost to a certain extent. Consequently, it is of great significance to design defect engineering to weaken the negative effects of defects in ceramic films and achieve improvement in performance. Up to now, a number of experiments have been conducted to investigate the design of defects. Pan et al. controlled the oxygen partial pressure in a pulsed laser deposition method to enable incorporation of oxygen vacancies and bismuth vacancies to form deep level vacancy complexes with high charge carrier activation energies, resulting in a significant reduction of the conductivity.9 The addition of some dopants to ceramic materials helps to control defects in terms of type, concentration and location. The typical effective method is aliovalent doping. Song et al. reported the formation of a defect complex by the combination of Mn2+ ions and oxygen vacancies, which reduced the concentration of leakage current density, resulting in the high energy storage density of 47.6 J cm−3 at 4100 kV cm−1 in the SBT-BFO system.11
Further enhancement of energy storage performance can be achieved by controlling the crystallization process to achieve an increase in breakdown strength while resulting in a reduction in leakage current density. The construction of heterogeneous interfaces by component modulation may be a simple and effective strategy. In polycrystalline materials, the dopant ions will tend to aggregate at the grain boundary and form heterogeneous interfaces due to the interaction potential between solutes, thus inhibiting the growth of grains and generating more interfaces, which can effectively improve the breakdown strength.12–14 It is reported that the doped Zn in KNN–BZN–Zn ceramics will aggregate at the grain boundary and inhibit grain growth during the sintering process.13 Another reported study also mentioned that the lanthanum ion–cation vacancy pairs at the grain boundary reduce the mobility rate of the grain boundary and thus inhibit the grain growth.14
Based on the above discussion, we proposed a novel strategy as shown in Fig. 1 to realize the bidirectional design of heterogeneous interface construction and defect engineering, so as to drastically enhance the dielectric properties and breakdown strength in relaxor ferroelectric films and ultimately to obtain excellent energy storage performance. To realize this strategy, a lead-free perovskite-structured x mol% Fe-doped 0.8Bi0.5Na0.5TiO3–0.2Bi3.25La0.75Ti3O12 (x = 0, 2, 4, and 6, abbreviated as 0F, 2F, 4F, and 6F) relaxor ferroelectric was employed. Bi and Na share a different local environment in the lattice, resulting in NBT losing its original high symmetry and appearance of spontaneous polarization, thus leading to the outstanding ferroelectricity.15 Bi3.25La0.75Ti3O12 (BLT) provides excellent fatigue resistance for NBT-based films, due to its unique bismuth–oxygen layer structure acting as a barrier layer. Furthermore, BLT has a high Curie temperature (∼675 °C), which is also conducive to achieving stable energy storage performance at high temperatures, significantly enhancing the stability and lifespan of the films.16 The doped Fe ions enter the lattice as a substitute for Ti ions, leading to an increase in the relative displacement between the Bi and TiO6 octahedra due to the larger radius, thus resulting in a larger polarization. In addition, an appropriate amount of Fe effectively suppresses the valence change of Ti ions and reduces the volatilization of Na and Bi during high temperature annealing, as well as combines oxygen vacancies to form defect dipoles 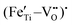 and
and 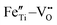 . These factors jointly reduce the concentration of oxygen vacancies, thus reducing the leakage current density. Simultaneously, the doped Fe aggregates at the grain boundary and forms a heterogeneous interface, preventing the mass transfer process and inducing the generation of additional interfaces, which impede the formation of electron paths and greatly increase the breakdown strength. Ultimately, with the synergistic action of bidirectional design, an excellent energy storage density of 83.6 J cm−3 with a great efficiency of 74.1% is obtained for 2F thin films. This finding provides a feasible and efficient strategy to develop high-performance materials for energy storage applications.
. These factors jointly reduce the concentration of oxygen vacancies, thus reducing the leakage current density. Simultaneously, the doped Fe aggregates at the grain boundary and forms a heterogeneous interface, preventing the mass transfer process and inducing the generation of additional interfaces, which impede the formation of electron paths and greatly increase the breakdown strength. Ultimately, with the synergistic action of bidirectional design, an excellent energy storage density of 83.6 J cm−3 with a great efficiency of 74.1% is obtained for 2F thin films. This finding provides a feasible and efficient strategy to develop high-performance materials for energy storage applications.
 | ||
| Fig. 1 Schematic diagram showing improvement in the energy storage properties of NBT-based films via bidirectional design. | ||
2. Experimental section
The x mol% Fe-doped NBT-BLT thin films are deposited on Pt/Ti/SiO2/Si (100) by a sol–gel method. Firstly, tetrabutyl titanium (C16H36O4Ti, 99.0%, Aladdin), bismuth nitrate pentahydrate [Bi(NO3)3·5H2O, 99.0%, Aladdin], lanthanum nitrate [La(NO3)3·6H2O, 98.0%, Aladdin], iron(III) nitrate nonahydrate [FeN3O9·9H2O, 99%, Aladdin], and sodium acetate [CH3COONa, 99.0%, Aladdin] are dissolved in a mixture of acetic acid solvent and 2-methoxyethanol in a stoichiometric ratio, and an appropriate amount of acetylacetone is added as a stabilizer to stabilize the solution. The final concentration of precursor solutions is 0.1 mol L−1. In addition, additional 10% Bi source and Na source are added to compensate for the volatilization during the high temperature annealing process. The precursor solutions were left to rest at 20 °C for 24 h to form stable solutions. Then the solutions are spin-coated onto the substrates at a preliminary rotation speed of 600 rpm−1 for 5 s and a high speed of 4500 rpm−1 for 40 s, respectively. Finally, the films are dried at 450 °C and then annealed at 625 °C for 4 min. The above steps were repeated to achieve the films with a thickness of ∼160 nm.The crystalline structure of the films is observed using a PANalytical X'Pert PRO X-ray diffractometer (XRD, X’Pert Pro, The Netherlands). The surface roughness is measured by scanning probe microscopy (AFM, Nanoscope IV, USA). The element valence state and 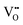 content fluctuations are calculated from the results of X-ray photoelectron spectroscopy (XPS, ESCALAB 250Xi). Microstructure analysis and elemental mapping of the films are performed by scanning electron microscopy (SEM, Zeiss Ultra Plus, Germany). The amorphous matrix and microcrystalline regions are confirmed by field emission transmission electron microscopy (TEM, Talos F200X, USA). To test the electrical properties, platinum electrodes with an area of 3.14 × 10−4 cm−2 are sputter deposited on top of the films by means of a shadow mask. Electrical hysteresis loops (P–E loops) are studied by means of the PolyK polarisation loop test system. Dielectric measurements are performed using an impedance analyser (Agilent E4294, USA). Current–voltage (I–V) tests are carried out with a Keithley 6517A electrodynamic tester. The dielectric constants and dielectric losses of the composite films are measured with an impedance analyser (4292A, USA). BDS and hysteresis loops were tested using a ferroelectric workstation (RT66A, USA).
content fluctuations are calculated from the results of X-ray photoelectron spectroscopy (XPS, ESCALAB 250Xi). Microstructure analysis and elemental mapping of the films are performed by scanning electron microscopy (SEM, Zeiss Ultra Plus, Germany). The amorphous matrix and microcrystalline regions are confirmed by field emission transmission electron microscopy (TEM, Talos F200X, USA). To test the electrical properties, platinum electrodes with an area of 3.14 × 10−4 cm−2 are sputter deposited on top of the films by means of a shadow mask. Electrical hysteresis loops (P–E loops) are studied by means of the PolyK polarisation loop test system. Dielectric measurements are performed using an impedance analyser (Agilent E4294, USA). Current–voltage (I–V) tests are carried out with a Keithley 6517A electrodynamic tester. The dielectric constants and dielectric losses of the composite films are measured with an impedance analyser (4292A, USA). BDS and hysteresis loops were tested using a ferroelectric workstation (RT66A, USA).
3. Results and discussion
The XRD results are shown in Fig. 2(a). The peaks of the film match with those of other similar standard perovskite structures, and the narrow and sharp morphology indicates that the film is highly crystalline. The films maintain a highly similar lattice arrangement with different Fe contents, which can be ascribed to the highly stable perovskite structure. In the partial enlarged patterns of Fig. 2(b), the peak of (200) gradually shifts to a lower angle with the Fe content. This shift can be explained by a larger ionic radius of Fe ions than Ti ions (RTi4+ = 0.605 Å, RFe3+ = 0.645 Å, and RFe2+ = 0.780 Å).17 The doped Fe ions enter the lattice, resulting in lattice distortion and increasing the lattice constant. | ||
| Fig. 2 (a) X-ray diffraction patterns in the 2θ range of 20–60°. (b) 45.5–47° of the films; atomic force microscopy (AFM) phase images. (c) 0F; (d) 2F; and (e) EDS mapping images of the 2F films. | ||
Fig. 2(c) and (d) display the surface roughness of the 0F films and 2F films. The AFM images show that 2F films exhibit more uniform surface features as well as lower roughness than 0F films, i.e. 1.6 nm rms vs. 2.0 nm rms, respectively. Fig. 2(e) shows the element distribution of 2F films recorded by energy dispersive spectroscopy (EDS). The homogeneous distribution of the dominating elements confirms the uniform chemical structure of the films.
The SEM morphologies of NBT–BLT composite thin films with different Fe contents are shown in Fig. 3(a)–(f). There are some tiny holes on the surface, which can be ascribed to the volatilization of Bi and Na. The equations are shown below:7
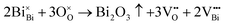 | (1) |
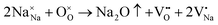 | (2) |
Clearly, the holes of 2F films are significantly reduced compared with those of 0F films, implying that the appropriate amount of Fe content effectively inhibits the volatilization of Bi and Na. Moreover, as the Fe content increases, the mean grain size initially decreases from 47.6 nm to 30.3 nm in 2F films and then increases slightly to 41.6 nm. These findings demonstrate that the moderate Fe content can effectively hinder the growth of grains, thus forming more grain boundaries and improving the densification of the film.18,19 Generally, the grain growth is attributed to the mass transfer process in the sintering process.2 The decrease in the concentration of oxygen vacancies in Fe-doped films may lead to a reduction in the grain size. Fig. 3(f) shows the SEM cross-sections of the 2F film. The surface of the film is flat and closely adhered to the bottom electrode with a thickness of about 160 nm.
As mentioned above, in the case of the polycrystalline materials, the dopant ions may undergo segregation during crystallization, which has a significant impact on the grain size. Thus, the microstructural analysis is conducted using a bright-field transmission electron microscope (TEM) to clarify this issue. Distinct lattice fringes are evident in a high-resolution transmission electron microscopy (HRTEM) image, as shown in Fig. 4(a). The measured interplanar spacing is about 0.383 nm, corresponding to the NBT-based films. Fig. 4(b) shows the selected area electron diffraction (SAED) image of the 2F sample. The bright and uniform diffraction spots demonstrate the good crystallinity of the film. We measured the distribution of Fe ions and Ti ions at grain boundaries (Fig. 4(c) and (d)). As observed in the TEM image and corresponding EDS line-scan of the 2F sample, there is an inhomogeneous distribution of elements at grain boundaries. The Fe content at grain boundaries is significantly higher than in the middle, while Ti content shows the opposite tendency. Moreover, the diffusion behavior of Fe was also investigated in the area labeled by ①–③ in Fig. 4(c). The spectral intensity of Fe at grain boundaries is much higher than that in other parts, and the Fe content first increases from 0.03 to 0.06 and then decreases to 0.03, which suggests that the Fe element aggregates at grain boundaries. In polycrystalline materials, when a small amount of dopant ions enters into the solid solution, the dopant ions will be attracted to or repelled from the grain boundaries due to the interaction potential between the solutes and the solute ions will tend to be unevenly distributed in the grain boundaries (as shown in Fig. 4(e)), resulting in a heterogeneous interface, where the ion mobility is reduced, consequently inhibiting the grain growth.13,20,21
Therefore, the inhibitory effect on the overflow of oxygen ions and the heterogeneous interfaces lead to the smaller grain sizes and more grain boundaries. However, an excessive Fe content will lead to the creation of cationic vacancies, so that the grain size tends to grow slightly, which results in a decrease in densification. In general, the decrease in grain size implies an increase in grain boundaries, which can effectively hinder the formation of breakdown paths, leading to an increase in breakdown strength. Based on the relationship between grain size and breakdown strength proposed by Tunkasiri:14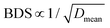 , the 2F films with heterogeneous interfaces have smaller grains with higher densification, resulting in an enhanced breakdown strength.
, the 2F films with heterogeneous interfaces have smaller grains with higher densification, resulting in an enhanced breakdown strength.
X-ray photoelectron spectroscopy (XPS) was carried out on the films with different Fe contents to verify the consistency of the elemental content with the chemical formula as well as to detect the valences of Fe ions and oxygen vacancies. Fig. 5(a) and (b) show the full spectrum of 2F films and the comparison between the actual and theoretical contents of elements in 0F and 2F films, respectively. Despite the slight discrepancy between the actual and theoretical ratios, the reason for which could be the volatilization of Bi and Na elements during the high temperature annealing process22 and the generation of oxygen vacancies, the results still show that the films are prepared as designed. In addition, it is worth noting that the actual ratios of elements in 2F films are closer to the theoretical ratios than that in 0F films. Fig. 5(c) shows the XPS spectra of Fe in 2F films. The Fe 2p3/2 peaks are fitted using a built-in program for XPS. Two peaks whose positions are located at 709.7 eV and 710.9 eV are obtained, corresponding to Fe2+ and Fe3+, which agrees with the reported literature.23 These findings demonstrate that Fe exists in the valence states of Fe3+ and Fe2+ in the films. The Ti 2p spectra of the 0F and 2F films are shown in Fig. 5(d) and (e). The Ti4+ ions are detected, accompanied by a signal from Ti3+ ions, which indicates that partial Ti4+ ions have been reduced.14 The valence change of Ti ions may lead to the generation of oxygen vacancies, the process of which is shown in formula (3).7
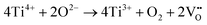 | (3) |
The peak ratio (ATi3+/ATi4+), representing the degree of Ti reduction, decreases from 0.74 to 0.52 for the 2F film. Moreover, the peak position of Ti 2p3/2 shifts from 457.9 eV to 458.3 eV. Since the binding energy is positively correlated with ion valence and electronegativity, the displacement of the peak can be attributed to the greater binding energy of Ti4+ ions than Ti3+ ions, indicating that the valence change of Ti was inhibited in the film with Fe content.24
Fig. 5(f) and (g) show the O 1s spectra of the films. Through Gaussian–Lorentz fitting of O (1s) peaks in the 0F and 2F films, it is found that the main peak located at 530.0 eV can be assigned to lattice oxygen, and the satellite peak around 531.6 eV is mainly derived from oxygen vacancies.25,26 In general, the oxygen vacancy content is characterized semi-quantitatively by the peak area ratio of OV/OL, which is 30.7% and 16.0% for samples with 0 mol% and 2 mol% Fe-doped, respectively. The concentration change of the oxygen vacancy implies that the moderate amount of Fe played an important role in reducing the oxygen vacancy concentration in the film. This can be ascribed to the fact that the Fe contributes to inhibiting the valence change of Ti ions and the volatilization of Bi and Na,27 leading to less oxygen vacancy generation. Moreover, it is believed that the introduced Fe2+ and Fe3+ ions form defect dipoles such as 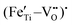 and
and 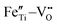 with the oxygen vacancy28,29 and hence efficiently decrease oxygen vacancy concentration, the process of which is shown in formulas (4) and (5):16
with the oxygen vacancy28,29 and hence efficiently decrease oxygen vacancy concentration, the process of which is shown in formulas (4) and (5):16
 | (4) |
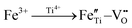 | (5) |
Fig. 6(a) shows the dielectric properties of the samples, which show a continuous increasing trend with the Fe content. As the Ti4+ ions are substituted by the larger Fe2+ and Fe3+ ions, the relative displacement between A-site ions and the TiO6 octahedron increases (Fig. 6(b)), which creates the tensile chemical pressure in the lattices leading to a larger dielectric constant.16 Moreover, compared with the pure NBT–BLT films, the films with Fe content exhibit a lower dielectric loss. This phenomenon may be ascribed to the fact that the introduction of Fe refines the grains, which strengthens the interface effect, and the resulting more barrier layers block the long-range shift of electrons.30
Fig. 6(c)–(f) show the temperature dependence of dielectric constant and dielectric loss at 1–100 k Hz. In general, the dielectric loss of the films tends to increase due to the thermal activation of the VO-related space charges.9 In this work, there are no apparent variations of the dielectric loss as the temperature is increased from 20 to 200 °C, benefiting from the high film quality of the samples. As a comparison, the films with Fe content retain a stable dielectric constant and low loss tangent (<0.08) at temperatures of up to 200 °C. Such a low loss factor is highly essential for solving the problems of self-heating and thermal runaway in dielectric capacitors.
The electrical performances of the NBT–BLT films with different Fe contents were further characterized. The I–V result and the Weibull distribution analysis of the breakdown strength are depicted in Fig. 7(a) and (b). The leakage current increases with the electric field. This can be ascribed to the fact that the force acting on the charges intensifies with the electric field, leading to an acceleration in the movement, thus resulting in an increase in leakage current. Furthermore, the defects in the film will become pathways for charge transport, further amplifying the leakage current. Moreover, the leakage current density shows a decreasing trend and then increases with the Fe content. The variation in oxygen vacancy concentration should be responsible for the result. As a typical defect, oxygen vacancies usually act as shallow charge carrier traps and severely degrade the electrical resistivity of dielectrics, consequentially contributing to large leakage current and low breakdown strength.9 In this work, an appropriate amount of Fe effectively inhibits the volatilization of the Bi and Na elements, as well as the valence change of Ti4+ ions, leading to a reduction in the concentration of oxygen vacancies. Moreover, the oxygen vacancies are easily trapped by Fe ions to form defect dipoles, which efficiently limit the movement of oxygen vacancies and the generation of the breakdown path, thus resulting in a high resistivity. Nevertheless, the excess Fe will increase the oxygen vacancy concentration, which induces an increase in the leakage current. The minimum leakage current density was obtained for 2F films, which contributes to the enhancement of breakdown strength. The characteristic of breakdown strength can be estimated by Weibull distribution. The Weibull distribution follows the equations:31
| Xi = In(Ei) | (6) |
| Yi = In(−In(1 − i/(n + 1))) | (7) |
| E1 ≤ E2 ≤ ⋯ Ei ≤ ⋯ En | (8) |
The energy storage properties of xF films are evaluated in accordance with the P–E loops measured at 1000 Hz (Fig. 7(c) and (d)). It is clear that the breakdown strength shows a tendency to increase and then decrease with the addition of Fe, which is compatible with the results of leakage current and Weibull distribution analysis. In addition, the polarization is continuously increasing with the Fe content, which is in accord with the trend of the dielectric constant. The substitution of Fe2+ and Fe3+ for Ti4+ should be responsible for the result, which enlarges the cell volume of NBT and creates the tensile chemical pressure in the lattices, thus leading to a larger polarization.16,32,33
Fig. 7(e) and (f) show the calculated values of Wrec and η plotted as a function of the external electric field and the comparison of energy storage performance between different samples. Generally speaking, there is a negative correlation between electromechanical breakdown and permittivity in dielectrics, which is shown in the formula below:34
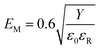 | (9) |
As the important index of energy storage capacitors, the performance reliability with respect to the switching cycles and working temperatures was also characterized. Fig. 8(a) and (b) present the temperature dependent P–E loops of the 2F film at 1250 kV cm−1, which are slim over a wide temperature range from 25 °C to 200 °C. The Wrec and η decrease slightly from 41.8 J cm−3 and 78.4% to 38.5 J cm−3 and 70.3%, demonstrating a favorable temperature stability. Furthermore, the 2F sample undergoes a charge–discharge process over 1 × 105 cycles with negligible degradation of energy performance. As shown in Fig. 8(c) and (d), a series of typically slender P–E loops could be obtained and the change rate of energy storage is below 5%. These results indicate that the films still retain high energy storage performance at high temperature and possess good cycle stability. A comparison between the 2F films reported in this work and typical lead-free films that have been reported in recent years was made in order to further evaluate the energy storage performance (Fig. 8(e)).7,35–53 The results illustrated that a superior balance between energy storage density and efficiency has been attained for 2F films reported in this work, which is an ideal feature for high-performance dielectric capacitors.
4. Conclusion
In summary, we demonstrate the effect of the bidirectional design of heterogeneous interface construction and defect engineering on the enhancement of energy storage in dielectrics. The heterogeneous interface design realizes the regulation of microstructure and effectively reduces the grain size, consequentially improving the breakdown strength. Meanwhile, the defect engineering reduces the oxygen vacancy concentration, substantially improves the leakage current, and further realizes the improvement of the electrical properties. As a result, we achieved a high overall energy storage performance, including a high Wrec of 83.6 J cm−3 and a high efficiency of 74.1%, for the 2 mol% Fe-doped 0.8NBT–0.2BLT film. This work reveals the significance of rational design and precise control of defects for high-performance dielectric energy storage.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Acknowledgements
This work was supported by the National Key Research and Development Program of China (No. 2023YFB3812200).References
- Z. M. Dang, J. K. Yuan, S. H. Yao and R. J. Liao, Adv. Mater., 2013, 25, 6334–6365 CAS.
- Z. B. Pan, D. Hu, Y. Zhang, J. J. Liu, B. Shen and J. W. Zhai, J. Mater. Chem. C, 2019, 7, 4072–4078 RSC.
- C. L. Diao, H. Wang, B. Y. Wang, Y. Q. He, Y. B. Hou and H. W. Zheng, J. Mater. Sci.: Mater. Electron., 2022, 33, 21199–21222 CrossRef CAS.
- Z. H. Yao, Z. Song, H. Hao, Z. Y. Yu, M. H. Cao, S. J. Zhang, M. T. Lanagan and H. X. Liu, Adv. Mater., 2017, 29, 15 Search PubMed.
- H. Wang, Y. Wang, B. Y. Wang, M. Q. Li, M. T. Li, F. Wang, C. L. Li, C. L. Diao, H. Luo and H. W. Zheng, ACS Appl. Mater. Interfaces, 2022, 14, 55130–55142 CrossRef CAS PubMed.
- W. Zhu, Z. Y. Shen, W. Deng, K. Li, W. Q. Luo, F. S. Song, X. J. Zeng, Z. M. Wang and Y. M. Li, J. Materiomics, 2024, 10, 86–123 CrossRef.
- J. Wang, G. X. Qiu, H. Qian, Y. F. Liu, J. Luo and Y. N. Lyu, Appl. Surf. Sci., 2022, 571, 9 Search PubMed.
- W. Abbas, M. S. Ibrahim, M. Waseem, C. Lu, H. H. Lee, S. Fazal, K. H. Loo and A. Pramanick, Chem. Eng. J., 2024, 482, 28 CrossRef.
- H. Pan, N. Feng, X. Xu, W. W. Li, Q. H. Zhang, S. Lan, Y. Q. Liu, H. Z. Sha, K. Bi, B. Xu, J. Ma, L. Gu, R. Yu, Y. Shen, X. R. Wang, J. L. MacManus-Driscoll, C. L. Chen, C. W. Nan and Y. H. Lin, Energy Storage Mater., 2021, 42, 836–844 CrossRef.
- Y. Feng, J. G. Wu, Q. G. Chi, W. L. Li, Y. Yu and W. D. Fei, Chem. Rev., 2020, 120, 1710–1787 CrossRef CAS PubMed.
- B. J. Song, S. H. Wu, H. Yan, K. Zhu, L. X. Xu, B. Shen and J. W. Zhai, J. Mater. Sci. Technol., 2021, 77, 178–186 CrossRef CAS.
- J. Y. Yi, J. K. Lee and K. S. Hong, J. Am. Ceram. Soc., 2002, 85, 3004–3010 CrossRef CAS.
- Y. Huan, T. Wei, X. Z. Wang, X. M. Liu, P. Y. Zhao and X. H. Wang, Chem. Eng. J., 2021, 425, 9 CrossRef.
- X. T. Zhang, L. L. Zhao, L. W. Liu, Z. A. Zhang and B. Cui, Chem. Eng. J., 2022, 435, 9 Search PubMed.
- J. Yin, H. Tao, Y. X. Zhang, J. H. Han, Y. L. Huang, Z. T. Li, X. M. Zhang and J. G. Wu, J. Mater. Chem. A, 2020, 8, 9209–9217 RSC.
- Z. B. Pan, P. Wang, X. Hou, L. M. Yao, G. Z. Zhang, J. Wang, J. J. Liu, M. Shen, Y. J. Zhang, S. L. Jiang, J. W. Zhai and Q. Wang, Adv. Energy Mater., 2020, 10, 9 Search PubMed.
- Y. J. Xie, H. Hao, Z. T. Huang, S. Zhang, M. H. Cao, Z. H. Yao and H. X. Liu, J. Alloys Compd., 2021, 884, 7 Search PubMed.
- M. Gao, W. Ge, X. Li, H. Yuan, C. Liu, H. Zhao, Y. Ma and Y. Chang, Phys. Status Solidi A, 2020, 217, 2000253 CrossRef CAS.
- V. Porokhonskyy, L. Jin and D. Damjanovic, Appl. Phys. Lett., 2009, 94, 212906 CrossRef.
- M. Acosta, L. A. Schmitt, L. Molina-Luna, M. C. Scherrer, M. Brilz, K. G. Webber, M. Deluca, H. J. Kleebe, J. Rödel and W. Donner, J. Am. Ceram. Soc., 2015, 98, 3405–3422 CrossRef CAS.
- X. F. Zhou, G. L. Xue, Y. C. Su, H. Luo, Y. Zhang, D. W. Wang and D. Zhang, Chem. Eng. J., 2023, 458, 13 Search PubMed.
- Q. Xu, Z. Song, W. L. Tang, H. Hao, L. Zhang, M. Appiah, M. H. Cao, Z. H. Yao, Z. C. He and H. X. Liu, J. Am. Ceram. Soc., 2015, 98, 3119–3126 CrossRef CAS.
- A. Kursumovic, W. W. Li, S. Cho, P. J. Curran, D. H. L. Tjhe and J. L. MacManus-Driscoll, Nano Energy, 2020, 71, 8 CrossRef.
- F. D. Zhang, X. S. Qiao, Q. Q. Shi, X. L. Chao, Z. P. Yang and D. Wu, J. Eur. Ceram. Soc., 2021, 41, 368–375 CrossRef CAS.
- K. H. Ye, K. S. Li, Y. R. Lu, Z. J. Guo, N. Ni, H. Liu, Y. C. Huang, H. B. Ji and P. S. Wang, TrAC, Trends Anal. Chem., 2019, 116, 102–108 CrossRef CAS.
- Z. Wei, W. C. Wang, W. L. Li, X. Q. Bai, J. F. Zhao, E. C. M. Tse, D. L. Phillips and Y. F. Zhu, Angew. Chem., Int. Ed., 2021, 60, 8236–8242 CrossRef CAS PubMed.
- Z. B. Pan, J. Ding, X. Hou, S. H. Shi, L. M. Yao, J. J. Liu, P. Li, J. W. Chen, J. W. Zhai and H. Pan, J. Mater. Chem. A, 2021, 9, 9281–9290 RSC.
- C. H. Yang, Y. J. Han, J. Qian, P. P. Lv, X. J. Lin, S. F. Huang and Z. X. Cheng, ACS Appl. Mater. Interfaces, 2019, 11, 12647–12655 CrossRef CAS PubMed.
- H. Pan, F. Li, Y. Liu, Q. H. Zhang, M. Wang, S. Lan, Y. P. Zheng, J. Ma, L. Gu, Y. Shen, P. Yu, S. J. Zhang, L. Q. Chen, Y. H. Lin and C. W. Nan, Science, 2019, 365, 578–582 CrossRef CAS PubMed.
- H. D. Z. Yang, S. Qu, Y. Hou, H. Ma, J. Wang, J. Wang, X. Wei and Z. Xu, J. Mater. Chem. A, 2016, 4, 13778–13785 RSC.
- X. Zhang, Y. Shen, B. Xu, Q. H. Zhang, L. Gu, J. Y. Jiang, J. Ma, Y. H. Lin and C. W. Nan, Adv. Mater., 2016, 28, 2055 CrossRef CAS PubMed.
- J. J. Wang, P. P. Wu, X. Q. Ma and L. Q. Chen, J. Appl. Phys., 2010, 108, 114105 CrossRef.
- Y. H. Huang, J. J. Wang, T. N. Yang, Y. J. Wu, X. M. Chen and L. Q. Chen, Appl. Phys. Lett., 2018, 112, 102901 CrossRef.
- L. Yang, X. Kong, F. Li, H. Hao, Z. Cheng, H. Liu, J.-F. Li and S. Zhang, Prog. Mater. Sci., 2019, 102(107), 102–108 Search PubMed.
- B. D. Qingguo Chi, C. Yin, X. Zhang, Z. Xu, C. Zhang, Y. Zhang and T. Zhang, Ceram. Int., 2024, 35128–35136 Search PubMed.
- J. H. Wang, N. N. Sun, Y. Li, Q. W. Zhang, X. H. Hao and X. J. Chou, Ceram. Int., 2017, 43, 7804–7809 CrossRef CAS.
- S. H. Yu, C. M. Zhang, M. Y. Wu, H. L. Dong, Z. Sun and L. X. Li, Ceram. Int., 2021, 47, 1238–1243 CrossRef CAS.
- M. Liu, C. Z. Gong, R. H. Wei, L. Hu, J. M. Dai, X. B. Zhu and Y. P. Sun, J. Alloys Compd., 2023, 967, 7 Search PubMed.
- X. X. Liu, Y. Yao, X. F. Wang, L. Zhao and X. Y. San, Coatings, 2024, 14, 8 CAS.
- B. B. Yang, M. Y. Guo, D. P. Song, X. W. Tang, R. H. Wei, L. Hu, J. Yang, W. H. Song, J. M. Dai, X. J. Lou, X. B. Zhu and Y. P. Sun, Appl. Phys. Lett., 2018, 113, 5 Search PubMed.
- L. Zhang, B. B. Yang, J. Y. Yu, Y. Deng, K. Huang, M. Zhang and X. B. Zhu, J. Alloys Compd., 2019, 799, 66–70 CrossRef CAS.
- J. Ding, Z. B. Pan, P. X. Chen, D. Hu, F. Yang, P. Li, J. J. Liu and J. W. Zhai, Ceram. Int., 2020, 46, 14816–14821 CrossRef CAS.
- W. F. Yue, Y. L. Cai, H. Y. Zhao, Q. S. Guo, D. W. Wang and T. T. Jia, Ceram. Int., 2024, 50, 13644–13651 CrossRef CAS.
- C. W. Bin, X. Hou, Z. Q. Yu, L. C. Liao, H. Yang, Y. Y. Liu and J. Wang, ACS Appl. Mater. Interfaces, 2024, 16, 2231–2239 CrossRef CAS PubMed.
- Z. Li, S. J. Chen, H. Biao and Y. Q. Gong, J. Mater. Sci.: Mater. Electron., 2024, 35, 12 CrossRef.
- F. H. Dashuang Fu, H. Tian, J. Li, J. Zhang, Z. Kang, Y. Wu and X. Wang, Ceram. Int., 2024, 0272–8842 Search PubMed.
- M. Kim and J. Y. Son, J. Energy Storage, 2024, 84, 8 Search PubMed.
- C. Y. Yue, H. J. Sun, C. Yan, X. H. Huang, H. T. Sui and Y. L. Hu, J. Mater. Chem. C, 2022, 10, 10356–10364 RSC.
- J. Xie, H. X. Liu, Z. H. Yao, H. Hao, Y. J. Xie, Z. X. Li and M. H. Cao, J. Eur. Ceram. Soc., 2020, 40, 1243–1249 CrossRef CAS.
- C. L. Diao, H. X. Liu, Z. X. Li, Z. H. Yao, H. Hao and M. H. Cao, J. Alloys Compd., 2020, 845, 8 CrossRef.
- Z. S. Liang, J. W. Wang, X. Liu, C. Li and X. F. Du, Appl. Phys. Lett., 2024, 124, 6 Search PubMed.
- Y. Q. Hu, A. Mattursun, M. Feng, N. T. Liu, H. N. Wang, K. Qu, X. Deng, Z. Guan, Z. Z. Yang, B. B. Chen, N. Zhong, C. G. Duan and P. H. Xiang, Small Methods, 2024, 8 Search PubMed.
- X. Zhang, L. Shu, Z. Q. Yang, L. S. Liu, F. Y. Zhu, H. L. Wang, Y. Y. S. Cheng, Y. Huang and J. F. Li, Nano Energy, 2024, 121, 8 Search PubMed.
| This journal is © The Royal Society of Chemistry 2025 |






