Mediating the carbon black–natural rubber interface with thioamide-functionalized polysulfide for energy-saving composites†
Ruoyan
Huang
a,
Dong
Wang
a,
Zhenghai
Tang
*a,
Baochun
Guo
 *a and
Liqun
Zhang
*a and
Liqun
Zhang
 *abc
*abc
aDepartment of Polymer Materials and Engineering, South China University of Technology, Guangzhou 510640, P. R. China. E-mail: mszhtang@scut.edu.cn; psbcguo@scut.edu.cn
bCollege of Materials Science and Engineering, Beijing University of Chemical Technology, China. E-mail: zhanglq@mail.buct.edu.cn
cSchool of Chemical Engineering and Technology, Xi'an Jiaotong University, China
First published on 20th June 2025
Abstract
Interfacial modification of carbon black (CB)-filled natural rubber (NR) composites is crucial for achieving uniform CB dispersion and robust interfacial interaction, with the goals of improving mechanical properties and reducing hysteresis loss. In this study, we reported a one-step synthesis of thioamide-functionalized polysulfide (SCA) via inverse vulcanization of sulfur with cyclohexylamine and the simultaneous conversion of amine to thioamide groups. SCA could serve as a novel interfacial modifier for NR/CB composites, in which thioamide groups form robust hydrogen bonds with the oxygen-containing groups on the CB surface, and the polysulfide fragments cleave and covalently couple with NR, enabling the establishment of SCA-mediated bridges between NR and CB. Consequently, the incorporation of SCA remarkably suppresses CB aggregation and enhances interfacial interaction, resulting in a substantial decrease in the hysteresis loss of the composites. More importantly, the effects of SCA molecular structure on the composite structure and properties are systematically investigated. The thioamide content is found to be critical for improving CB dispersion within the composites, while the reactivity between NR and polysulfide fragments in SCA is the dominant factor governing interfacial interaction.
Introduction
Rubbers, renowned for their exceptional elasticity and extensibility, find widespread applications as tires, seals, flexible devices, and damping components across diverse industries.1,2 However, the practical applications of neat rubbers are greatly constrained by their inherently weak mechanical properties and poor fatigue resistance.3–5 To overcome these limitations, reinforcing fillers such as carbon black (CB),6–8 silica,9,10 and graphene11 are universally incorporated to improve comprehensive properties. Particularly, CB is the predominant reinforcement due to its cost-effectiveness and high ability to improve modulus, abrasion resistance, and durability.7,12,13 Nevertheless, CB particles tend to aggregate within rubber matrices, inevitably increasing the hysteresis loss of the composites due to micro-scale friction among CB agglomerates and at the CB–rubber interface under dynamic loading. An increased hysteresis loss signifies that more mechanical energy is converted into internal energy during service, which not only increases energy consumption but also causes heat accumulation within composites to accelerate aging and compromise performance.14To resolve the trade-off between reinforcement and hysteresis loss in CB-filled rubber composites, it is crucial to improve CB dispersion and strengthen interfacial interaction between CB and rubber. Thus far, three primary strategies including surface modification of CB,15 functionalization of rubber,16 and incorporation of interfacial modifiers (IMs)17 have been explored. The first two methods involve the introduction of specific groups onto the CB surface or rubber chains, which necessitates meticulous molecular engineering and solvent consumption, limiting their practicality. Comparatively, the incorporation of IMs emerges as the most practical and effective solution by establishing molecular linkages between CB and the rubber matrix. IMs typically feature bifunctional motifs: one group capable of reacting with the rubber matrix, and the other group interacting with CB. For example, Gao et al. engineered a polyether chain into a sulfur-containing silane coupling agent for modifying CB-filled natural rubber (NR/CB) composites.18 The polyether chain adsorbs onto the CB surface through hydrogen bonding, and the polysulfide moiety participates in the crosslinking reaction of NR, leading to improved CB dispersion. Compared with the unmodified composite, the composite modified with 10 phr (parts per hundred rubber) silane exhibited an 8% decrease in hysteresis loss, however, the tensile strength and modulus decreased due to polyether-induced plasticization. Fang et al. synthesized an adamantyl-based imine DADI as an IM for NR/CB composites.19 The adamantane functionality can stabilize rubber macroradicals during processing and facilitate their transfer to the CB surface to improve interfacial interaction. With the incorporation of 1 phr DADI, the composite demonstrated a 6 °C decrease in heat generation and a 21% reduction in hysteresis loss. Despite these advances, the influences of IM molecular structure on the coupling mechanism or composite properties have rarely been reported.
The concept of inverse vulcanization, introduced by Pyun and co-workers in 2013, offers an innovative strategy for synthesizing sulfur-rich copolymers by stabilizing polysulfide diradicals generated from ring-opening polymerization of element sulfur (S8) at elevated temperatures with olefinic comonomers.20 Inverse vulcanized polysulfides (IVPs), typically containing 50–90 wt% sulfur, exhibit exceptional optical transparency, electrochemical activity, and thermo-mechanical properties, making them highly promising for applications as electrode materials, infrared optical devices, and heavy metal adsorbents.21–23 Recently, significant efforts have been focused on regulating the molecular structure and functionality of IVPs by exploring new monomers24,25 and polymerization methods,26,27 with the goals of tuning their properties and expanding the application scopes.28,29 Particularly, the synthesis of IVPs containing functional groups is highly desirable, as the functional groups can endow IVPs with novel properties and serve as versatile chemical platforms for post-modification and polymerization. However, this endeavor is often fraught with challenges owing to poor compatibility between functional comonomers with S8. Based on the catalytic strategy and dynamic covalent polymerization approach, diverse functional groups including epoxy,9,30 carboxyl,31 hydroxyl,32 alkoxy,10,33 and benzyl chloride groups34 have been successfully incorporated into IVPs. Very recently, leveraging the reactivity of polysulfide fragments in IVPs with rubbers, our group pioneered the use of functional IVPs as highly efficient IMs for rubber composites.9,10,17
In this study, we synthesized a thioamide-functionalized polysulfide (SCA) and utilized it as an IM for NR/CB composites. The interfacial coupling mechanism was elucidated, and CB dispersion and interfacial interaction in the composites were meticulously characterized and correlated with composites macroscopic properties. More importantly, by regulating the chemical compositions of SCA, the effects of SCA molecular structure on the composite structure and properties were systematically investigated.
Results and discussion
Characterization of synthesized SCA
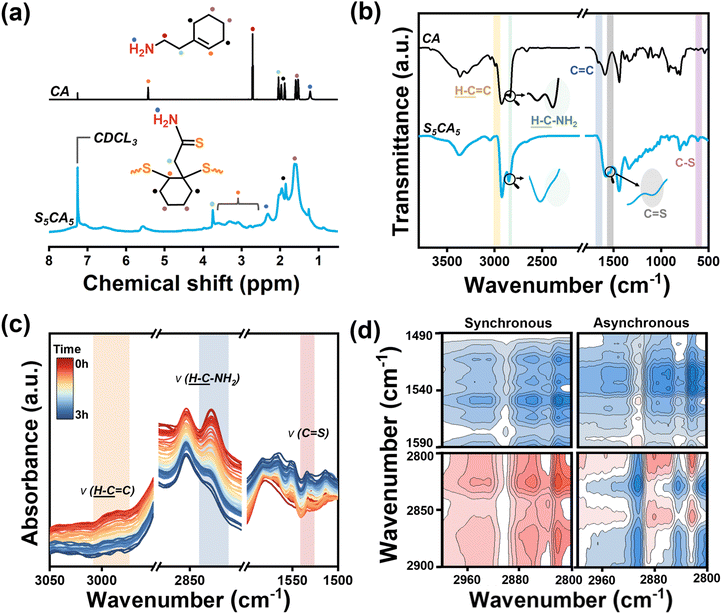
The successful copolymerization between 2-(1-cyclohexenyl) ethylamine (CA) and S8 was first confirmed by 1H NMR measurement. As shown in Fig. 1a, the 1H NMR spectrum of CA displays resonance signals at 5.4, 2.7, 2.0, and 1.2 ppm, corresponding to the protons in –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–, –CH2–NH2, –CH2– CH2–NH2, and –NH2, respectively.35 Upon copolymerization, taking S5CA5 (the mass feeding ratio of S8 and CA is 5
C–, –CH2–NH2, –CH2– CH2–NH2, and –NH2, respectively.35 Upon copolymerization, taking S5CA5 (the mass feeding ratio of S8 and CA is 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) as an example, the signal for –CH
5) as an example, the signal for –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C– at 5.4 ppm disappears, while new signals ranging from 2.9 to 3.5 ppm are observed, which are due to the protons in –S–CH–fragments.36 These observations indicate the addition of the sulfur atom onto the C
C– at 5.4 ppm disappears, while new signals ranging from 2.9 to 3.5 ppm are observed, which are due to the protons in –S–CH–fragments.36 These observations indicate the addition of the sulfur atom onto the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond. In addition, compared with CA, the signal for –CH2–NH2 at 2.7 ppm completely vanishes in S5CA5, and the signals for –CH2–CH2–NH2 and –NH2 shift to 3.7 and 2.3 ppm, respectively. Such shifts indicate the conversion of –CH2–NH2 to –C(
C bond. In addition, compared with CA, the signal for –CH2–NH2 at 2.7 ppm completely vanishes in S5CA5, and the signals for –CH2–CH2–NH2 and –NH2 shift to 3.7 and 2.3 ppm, respectively. Such shifts indicate the conversion of –CH2–NH2 to –C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S)–NH2, which has been documented in a previous study on S8 and alkylamine solutions.37 At a higher S8 feeding ratio, S7CA3 (the mass feeding ratio of S8 and CA is 7
S)–NH2, which has been documented in a previous study on S8 and alkylamine solutions.37 At a higher S8 feeding ratio, S7CA3 (the mass feeding ratio of S8 and CA is 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) exhibits similar spectral features to S5CA5. However, in the case of S3CA7 (the mass feeding ratio of S8 and CA is 3
3) exhibits similar spectral features to S5CA5. However, in the case of S3CA7 (the mass feeding ratio of S8 and CA is 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7) having a lower S8 feeding ratio, the peak related to –CH2–NH2 at 2.7 ppm is still discernible (Fig. S1†), indicating the incomplete conversion of –CH2–NH2 into the thioamide group due to the decreased availability of S8. According to the area integral, the molar ratio between –CH2–NH2 and –C(
7) having a lower S8 feeding ratio, the peak related to –CH2–NH2 at 2.7 ppm is still discernible (Fig. S1†), indicating the incomplete conversion of –CH2–NH2 into the thioamide group due to the decreased availability of S8. According to the area integral, the molar ratio between –CH2–NH2 and –C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S)–NH2 in S3CA7 is determined to be 1.
S)–NH2 in S3CA7 is determined to be 1.
In the FTIR spectrum of CA (Fig. 1b), the absorptions at 3366 and 3288 cm−1 are due to the N–H stretching vibration in the amine group, and the absorptions at 2995, 2835, and 1660 cm−1 correspond to the stretching vibrations of 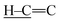 ,
, 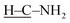 , and C
, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, respectively.35 In the FTIR spectrum of S5CA5, both
C, respectively.35 In the FTIR spectrum of S5CA5, both 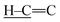 and C
and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations disappear, while a new peak at 690 cm−1 characteristic of C–S stretching vibration is observed, further confirming sulfur addition to the C
C stretching vibrations disappear, while a new peak at 690 cm−1 characteristic of C–S stretching vibration is observed, further confirming sulfur addition to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond.6 Additionally, the absorption of the
C bond.6 Additionally, the absorption of the 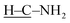 stretching vibration at 2835 cm−1 disappears, and a new absorption at 1539 cm−1 corresponding to the C
stretching vibration at 2835 cm−1 disappears, and a new absorption at 1539 cm−1 corresponding to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S stretching vibration emerges, accompanied by the N–H stretching vibration shift to 3374 cm−1, all of which collectively verify the conversion of –CH2–NH2 to –C(
S stretching vibration emerges, accompanied by the N–H stretching vibration shift to 3374 cm−1, all of which collectively verify the conversion of –CH2–NH2 to –C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S)–NH2.38,39
S)–NH2.38,39
To gain insights into the reaction pathway, time-dependent FTIR spectra were recorded for the mixture of equimass CA and S8 at 130 °C under argon. As depicted in Fig. 1c, as the reaction proceeds, the absorptions for the stretching vibrations of 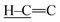 and
and 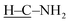 gradually diminish, while the absorption for the C
gradually diminish, while the absorption for the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S stretching vibration intensifies. Further, two-dimensional correlation analysis is conducted based on the time-dependent FTIR spectra (Fig. 1d). In the synchronous maps, three cross-peaks [Φ (V1, V2)] are observed at off-diagonal positions: a positive peak Φ (2995, 2835) and two negative peaks Φ (2995, 1539) and Φ (2835, 1539), indicating strong coupling among the affiliated groups, i.e. C
S stretching vibration intensifies. Further, two-dimensional correlation analysis is conducted based on the time-dependent FTIR spectra (Fig. 1d). In the synchronous maps, three cross-peaks [Φ (V1, V2)] are observed at off-diagonal positions: a positive peak Φ (2995, 2835) and two negative peaks Φ (2995, 1539) and Φ (2835, 1539), indicating strong coupling among the affiliated groups, i.e. C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, –CH2–NH2, and C
C, –CH2–NH2, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S. In the asynchronous maps, the cross-peak Ψ (2835, 1539) is negative. According to Noda's rule,40 the intensity change of –CH2–NH2 precedes that of C
S. In the asynchronous maps, the cross-peak Ψ (2835, 1539) is negative. According to Noda's rule,40 the intensity change of –CH2–NH2 precedes that of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S, indicating that the formation of thioamide is not a single-step process. Furthermore, the negative peaks of both Ψ (2995, 2835) and Ψ (2995, 1539) indicate a sequential response: –CH2–NH2 undergoes an intensity change first, followed by C
S, indicating that the formation of thioamide is not a single-step process. Furthermore, the negative peaks of both Ψ (2995, 2835) and Ψ (2995, 1539) indicate a sequential response: –CH2–NH2 undergoes an intensity change first, followed by C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, while C
C, while C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S responds last.
S responds last.
Based on the above analysis, a reaction mechanism between CA and S8 is proposed in Fig. 2a. At 130 °C, the S8 ring undergoes nucleophilic attack by nitrogen in CA, leading to the heterogeneous cleavage of the ring and formation of polysulfide intermediates composed of thiolate anions and amine salt cations. The thiolate anions subsequently attack the α-CH2 protons adjacent to amine groups, oxidizing them to imines—a transformation well-documented in previous studies.41,42 Concurrently, homolytic cleavage of polysulfide intermediates generates sulfur radicals that add to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds via inverse vulcanization pathways. Finally, the thiolate anions react with the imines to liberate H2S and form thioamide groups.37
C bonds via inverse vulcanization pathways. Finally, the thiolate anions react with the imines to liberate H2S and form thioamide groups.37
 | ||
| Fig. 2 (a) Possible reaction pathways between CA and S8. (b) Proposed coupling mechanism between CB and rubber by SCA. | ||
Coupling of CB and NR with SCA
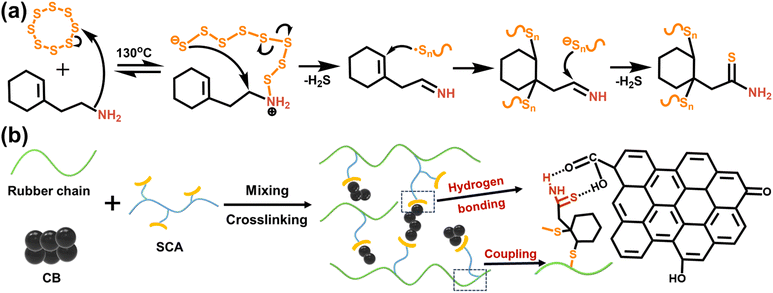
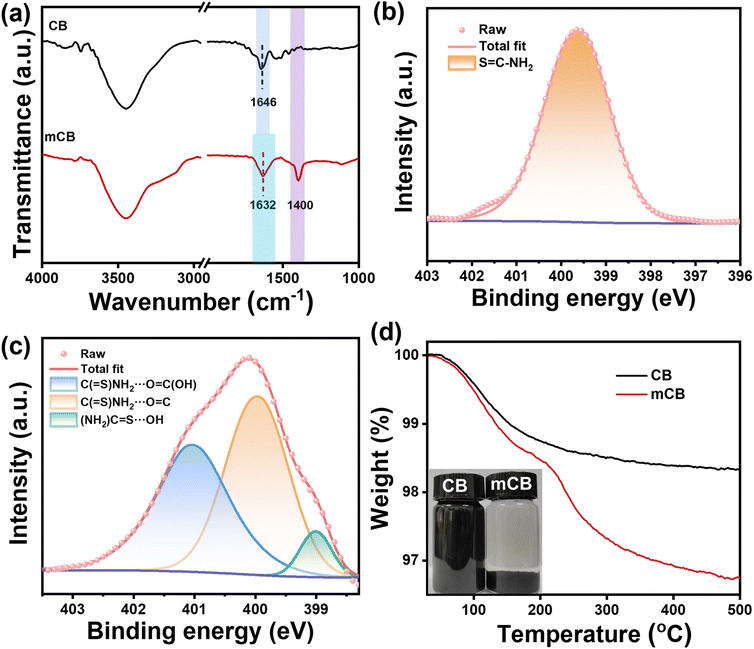
The CB surface features oxygenic groups, including carboxyl groups, phenolic hydroxyl groups, and quinone groups, which can serve as active sites for CB modification. We envision that the as-synthesized SCA can be used as a novel IM for the CB-filled NR composite: thioamide groups in SCA form robust hydrogen bonding interactions with the oxygenic groups on the CB surface, and the polysulfide fragments in SCA cleave and graft onto NR chains.6,16 These dual interactions establish molecular bridges between NR and CB, as schematically illustrated in Fig. 2b.To explore the interactions between SCA and CB, a model compound mCB was prepared by thermally treating CB with S5CA5 followed by Soxhlet extraction. Fig. 3a compares the FTIR spectra of pristine CB and mCB. In the FTIR spectrum of CB, the absorptions at 3447 and 1646 cm−1 are assigned to the stretching vibrations of –OH and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, respectively. Upon modification, mCB exhibits a new absorption at about 1400 cm−1, which is attributed to the –CH2– deformation vibration from the grafted S5CA5.35 Additionally, the C
O, respectively. Upon modification, mCB exhibits a new absorption at about 1400 cm−1, which is attributed to the –CH2– deformation vibration from the grafted S5CA5.35 Additionally, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching band in mCB broadens and shifts to 1632 cm−1, which is on account of the hydrogen bonding interactions between C
O stretching band in mCB broadens and shifts to 1632 cm−1, which is on account of the hydrogen bonding interactions between C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and thioamide groups.43,44
O and thioamide groups.43,44
 | ||
| Fig. 3 (a) FTIR spectra of CB and mCB. Deconvoluted N 1s spectra of (b) S5CA5 and (c) mCB. (d) TGA curves of CB and mCB. Inset is the photographs of CB and mCB dispersed in ethanol after 7 days. | ||
Further, XPS full spectra show that mCB exhibits S 2p and N 1s signals when compared to CB, confirming the grafting of S5CA5 onto the CB surface (Fig. S2†). In the deconvoluted high-resolution N 1s spectrum (Fig. 3b), S5CA5 exhibits a distinct peak at 399.6 eV, characteristic of the thioamide group. For mCB (Fig. 3c), the N 1s binding energy splits into three components located at 399.0, 400.0, and 401.0 eV, indicative of interactions between S5CA5 and CB. Specifically, the N–H (hydrogen-bond donor) and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S (hydrogen-bond acceptor) in thioamide groups interact with the oxygenic groups on the CB surface, modulating the electron cloud density around the atoms involved in the hydrogen bonds and thus elevating or lowering the binding energy of the N atom.44–47 Notably, the sulfur atom in thioamides is an exceptionally potent donor for the n–π* interaction, enabling thioamides to form stronger hydrogen bonds with C
S (hydrogen-bond acceptor) in thioamide groups interact with the oxygenic groups on the CB surface, modulating the electron cloud density around the atoms involved in the hydrogen bonds and thus elevating or lowering the binding energy of the N atom.44–47 Notably, the sulfur atom in thioamides is an exceptionally potent donor for the n–π* interaction, enabling thioamides to form stronger hydrogen bonds with C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O compared to amides and hydrazides.46
O compared to amides and hydrazides.46
The grafting efficiency of S5CA5 on the CB surface is evaluated using TGA. As shown in Fig. 3d, pristine CB exhibits a mass loss of 1.7% below 500 °C, corresponding to the elimination of oxygenic groups on the CB surface. In contrast, mCB shows a higher mass loss of 3.3% below 500 °C, attributed to the decomposition of the grafted S5CA5. Based on the residual mass at 500 °C, the grafting amount of S5CA5 on mCB is quantified as about 2.4%, and the grafting efficiency is further calculated to be 50% (Fig. S3†). To visually demonstrate the grafting of S5CA5 onto CB, dispersibility experiments were performed by suspending CB and mCB in ethanol at 1 mg mL−1 and then settling them for 7 days. As shown in the inset in Fig. 3d, CB remains stably dispersed, whereas mCB completely sediments from ethanol, manifesting that the surface chemistry of mCB is tailored into hydrophobicity upon S5CA5 grafting.
CB dispersion state and interfacial interaction in composites
In rubber composites, the filler dispersion state and interfacial adhesion are two critical factors in determining the comprehensive performance. TEM measurement visually reveals the CB dispersion state within NR/CB composites. As shown in Fig. 4a, NR/Blank (unmodified NR/CB composite) exhibits pronounced CB aggregates due to strong van der Waals forces among CB particles. Remarkably, as displayed in Fig. 4b and c, the incorporation of S5CA5 suppresses CB aggregation in NR/S5CA5-y (NR/CB composite modified with y phr of S5CA5), because the SCA-mediated interface improves the compatibility between CB and the NR matrix and mitigates thermodynamically driven self-agglomeration of CB. | ||
| Fig. 4 TEM images of (a) NR/Blank, (b) NR/S5CA5-0.5, and (c) NR/S5CA5-1.5. SEM images of (d) NR/Blank, (e) NR/S5CA5-0.5, and (f) NR/S5CA5-1.5. | ||
Morphologies of the composites were further evaluated by SEM measurement. NR/Blank displays an uneven distribution of CB with obvious aggregates exposed on the fracture surface (Fig. 4d), indicative of weak interfacial interaction between CB and the NR matrix. In contrast, SEM images of NR/S5CA5-y composites reveal that CB aggregates are broken into smaller ones and embedded within the composites with blurry interfacial boundaries (Fig. 4e and f). These findings provide convincing evidence that the interfacial adhesion is strengthened by incorporating SCA.
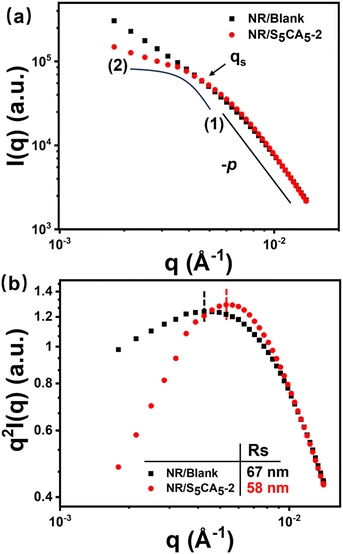
The hierarchical structures of CB in the composites were explored using ultra-small angle X-ray scattering (USAXS) measurement. As shown in Fig. 5a, taking NR/Blank and NR/S5CA5-2 as examples, the scattering profiles can be segmented by a cutoff qs into one Guinier regime and one power-law regime (I(q)–q−p). Here, qs relates to the gyration radius (Rs, Rs = π/qs) of the smallest structure elements of CB aggregates in the composites.48 In the power-law regime, the slope p of the two scattering curves is determined to be 3.4. Accordingly, the surface fractal dimension Ds of CB particles is determined to be 2.6 based on p = 6 − Ds, indicating that the surface of CB particles is rough.9 Further, to accurately identify Rs, the scattering profiles are analyzed using the Kratky presentation (Iq2vs. q) to transform the cutoffs into well-identified maxima.49 As shown in Fig. 5b, the calculated Rs value of NR/S5CA5-2 is declined from 67 to 58 nm when compared to NR/Blank, highlighting the suppressed CB aggregation upon S5CA5 incorporation.
 | ||
| Fig. 5 (a) Scattering curves and (b) Kratky representations of the scattering curves for NR/Blank and NR/S5CA5-2. | ||
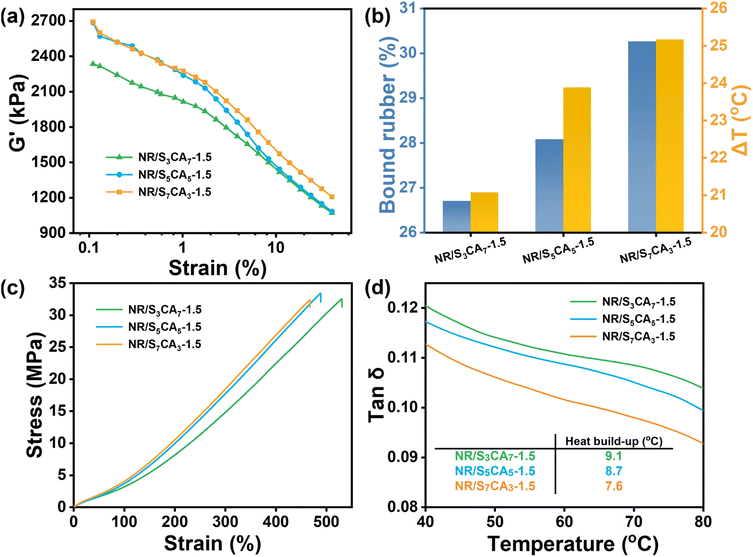
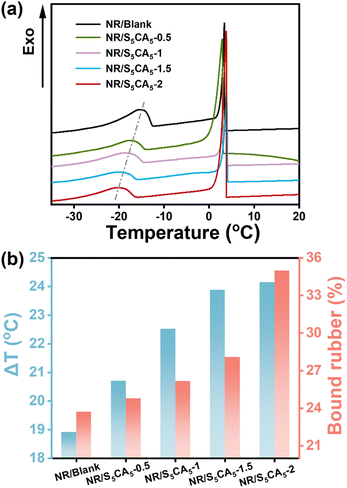
The interfacial adhesion between CB and the NR matrix was evaluated by measuring the freezing point of cyclohexane in the swollen composites. As displayed in Fig. 6a, DSC cooling curves of the swollen composites exhibit two distinct exothermic peaks: one at approximately 3.0 °C is due to the crystallization of free cyclohexane on the composite surface, and the other peak at a lower temperature corresponds to the crystallization of confined cyclohexane within the composite. The decreased freezing point of the confined cyclohexane originates from restricted chain mobility at the interface, which reduces the mesh size available for cyclohexane nucleation and thereby inhibits its crystallization.16 Consequently, the temperature difference between these two peaks (ΔT) is an indicator of interfacial adhesion.17 Remarkably, NR/S5CA5-y composites exhibit higher ΔT values than NR/Blank, with increasing ΔT values as S5CA5 dosage increases. These results demonstrate that the interfacial interaction in the composites is improved when SCA is incorporated.
 | ||
| Fig. 6 (a) DSC cooling curves for NR/Blank and NR/S5CA5-y swollen in cyclohexane. (b) BR content of NR/Blank and NR/S5CA5-y. | ||
Bound rubber (BR) is defined as the rubber tightly interacting with the filler surface and cannot be extracted by its good solvents anymore. The higher the BR content, the stronger the interfacial interaction. As shown in Fig. 6b, the BR content of NR/S5CA5-y composites consistently increases with S5CA5 dosage. Specifically, compared to NR/Blank, the BR content in NR/S5CA5-2 rises from 24% to 35%, exhibiting a 50% increment. The enhanced interfacial interaction stems from two synergic mechanisms as follows: (1) SCA-mediated bridging improves compatibility between the NR matrix and CB; (2) the improved CB dispersion allows a greater CB surface area to be accessible for interaction with NR chains.
Quasi-static and dynamic mechanical properties of composites
The quasi-static machinal characteristics of the composites obtained from tensile tests are summarized in Table S2.† Compared with NR/Blank, the moduli (stress at 300% strain) of NR/S5CA5-y composites gradually improve with the increase of S5CA5 dosage, and the ultimate strength and breaking strain remain comparable. This is because the improved CB dispersion and enhanced interfacial interaction facilitate stress transfer and promote finite extensibility of rubber chains between neighboring CB particles, thereby preventing premature failure and improving the modulus.50 In addition, the polysulfide fragments in S5CA5 can react with NR chains and increase network crosslink density (Fig. S4†), which is also responsible for the improved modulus.Rubber composites are frequently subjected to dynamic loading during servicing, where the viscoelastic properties are crucial as they determine energy storage and dissipation. Tires, as the primary application of rubber composites, have rolling resistance as a key performance metric, which represents the amount of mechanical energy converted into heat per unit distance. Rolling resistance is commonly quantified by the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ value of rubber composites at 7% strain, and a 20% decrease in rolling resistance corresponds to a 3–5% fuel saving.51 The dependence of tan
δ value of rubber composites at 7% strain, and a 20% decrease in rolling resistance corresponds to a 3–5% fuel saving.51 The dependence of tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ on strain for the composites is illustrated in Fig. 7a. Incorporating S5CA5 leads to a remarkable reduction in the tan
δ on strain for the composites is illustrated in Fig. 7a. Incorporating S5CA5 leads to a remarkable reduction in the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ value at 7% strain, with NR/S5CA5-2 exhibiting a 33% decrease compared to NR/Blank, indicating higher elasticity and lower hysteresis loss in the composites modified by S5CA5.
δ value at 7% strain, with NR/S5CA5-2 exhibiting a 33% decrease compared to NR/Blank, indicating higher elasticity and lower hysteresis loss in the composites modified by S5CA5.
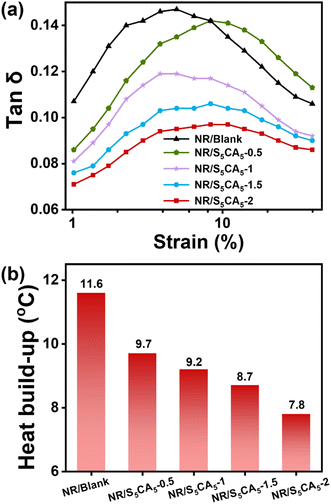 | ||
Fig. 7 (a) Dependence of tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ on strain and (b) heat build-up of NR/Blank and NR/S5CA5-y composites. δ on strain and (b) heat build-up of NR/Blank and NR/S5CA5-y composites. | ||
Furthermore, the heat built-up, manifested as heat accumulation within the composites during repeated compressive deformation, can directly mirror the hysteresis loss. As shown in Fig. 7b, the heat build-up value of the composites consistently decreases with increasing S5CA5 dosage, manifesting that less mechanical energy is dissipated. The reduction in the hysteresis loss mainly stems from two aspects: (1) improved CB dispersion suppresses the self-aggregation of CB, thereby reducing the interparticle friction during deformations; (2) enhanced interfacial adhesion restricts the slippage of macromolecular chains on the CB surface, thereby alleviating interfacial friction. In contrast to NR/CB composites modified with commercially available IMs and newly synthesized IMs in reported literature (Table S3†), the S5CA5-modified composite in this work exhibits a more pronounced reduction in hysteresis loss and heat generation. This is likely due to the robust interactions between the thioamide groups in S5CA5 and CB.
To simulate the service environment of tires, solid rubber wheels were fabricated from NR/Blank and NR/S5CA5-2, and the energy loss and temperature increment on the wheel surface were measured on a rotary power loss apparatus. As presented in Fig. 8a, compared with the NR/Blank wheel, the NR/S5CA5-2 wheel reveals a 30% reduction in energy loss (from 3.1 to 2.18 J/r) and a 7.3 °C decrease in surface temperature increment (from 72.5 to 65.2 °C). After the test, infrared thermography visually confirms a lower temperature on the NR/S5CA5-2 wheel, consistent with reduced energy dissipation (Fig. 8b). These outcomes indicate great advantages of S5CA5-modified composites in improving tire fuel economy and safety.
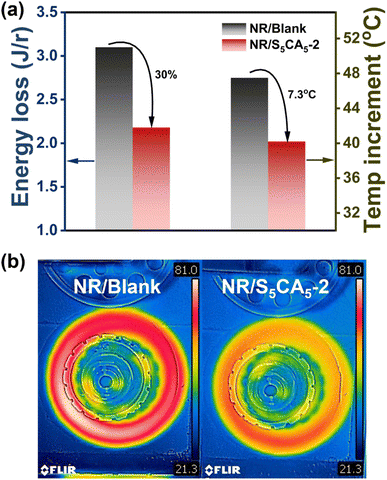 | ||
| Fig. 8 (a) Energy loss and temperature increment, and (b) infrared thermal images of NR/Blank and NR/S5CA5-2 wheels after the test. | ||
Insights into the coupling mechanism and structure–property relationship
To systematically investigate the effects of SCA molecular structure on the coupling mechanism and composite properties, SCAs with distinct chemical compositions were synthesized by varying the feeding ratio of S8 to CA and utilized as IMs. XRD patterns (Fig. S5†) and DSC curves (Fig. S6†) confirm the successful synthesis of SCAs. As the mass ratio of S8 to CA increases from 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 (S3CA7) to 7
7 (S3CA7) to 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 (S7CA3), the fraction of polysulfide fragments capable of reacting with rubber increases, while the content of thioamide groups interacting with CB conversely decreases (Table S4†).
3 (S7CA3), the fraction of polysulfide fragments capable of reacting with rubber increases, while the content of thioamide groups interacting with CB conversely decreases (Table S4†).
Fig. 9a illustrates the dependence of shear storage modulus (G′) on strain for the composites modified with SCA of varied chemical compositions. G′ of all the composites exhibits a nonlinear decrease with increasing strain, attributed to the collapse of CB agglomerates under large oscillatory shear.52 Thus, the difference in G′ between small and large strains (ΔG′) can serve as a quantitative indicator of CB dispersion, where a smaller ΔG′ indicates more uniform CB dispersion. Among the three composites, the incorporation of S3CA7 having the highest thioamide content leads to the lowest ΔG′, highlighting the pivotal role of the thioamide group in improving CB dispersion within the NR matrix.
As substantiated above, the incorporation of S5CA5 enhances interfacial interaction in the composites by forming molecular bridges across the interface through respective reaction with NR and interaction with CB. As demonstrated by the BR content and ΔT result (Fig. 9b), the interfacial interaction progressively enhances with increasing polysulfide fragment fraction in SCA, despite the reduction in thioamide group content. It is likely because, at 1.5 phr loading, SCA with distinct molecular structures can furnish sufficient amine sites to saturate CB surface interaction across all three composites. Consequently, the reaction between NR and polysulfide fragments in SCA is the dominant factor governing interfacial interaction. This finding provides fundamental guidance that the reactivity of the interfacial modifier with rubber should be rationally considered in the molecular design.
As the interfacial interaction enhances, the modulus of the composites consistently improves, and the breaking strain slightly decreases (Fig. 9c). This is because stronger interfacial interaction can immobilize more rubber chains onto the CB surface, forming a more constrained network that increases the modulus while reducing the breaking strain. Additionally, the enhanced interfacial interaction lowers energy dissipation by restricting rubber chain slippage on the CB surface under dynamic loading, leading to a progressive decrease in the hysteresis loss and fatigue-induced heat generation from S3CA7 to S7CA3 (Fig. 9d).
Conclusions
Thioamide-functionalized polysulfide SCA was successfully synthesized and utilized as an intelligent IM for NR/CB composites. 1H NMR, FTIR and time-dependent FTIR results verified the inverse vulcanization of S8 with CA and the conversion of –CH2–NH2 to –C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S)–NH2. Model compound studies demonstrated that the thioamide moieties in SCA interact with the oxygenic groups on the CB surface via robust hydrogen bonding interactions. The incorporation of SCA establishes bridges between NR and CB, leading to improved CB dispersion and enhanced interfacial interaction in the composites, as evidenced by morphological analysis and quantified using rheological, USAXS, BR and freezing point measurements. Consequently, SCA-modified composites exhibit a remarkable decrease in hysteresis loss and heat build-up. In addition, the effects of SCA molecular structure on the composite structure and properties were systematically investigated. The thioamide content plays a pivotal role in improving CB dispersion within the composites, while the reactivity of SCA with NR is the dominant factor governing the interfacial interaction. These findings establish SCA as a promising strategy for modifying NR/CB composites with low hysteresis loss and provide fundamental guidelines for rational interfacial engineering.
S)–NH2. Model compound studies demonstrated that the thioamide moieties in SCA interact with the oxygenic groups on the CB surface via robust hydrogen bonding interactions. The incorporation of SCA establishes bridges between NR and CB, leading to improved CB dispersion and enhanced interfacial interaction in the composites, as evidenced by morphological analysis and quantified using rheological, USAXS, BR and freezing point measurements. Consequently, SCA-modified composites exhibit a remarkable decrease in hysteresis loss and heat build-up. In addition, the effects of SCA molecular structure on the composite structure and properties were systematically investigated. The thioamide content plays a pivotal role in improving CB dispersion within the composites, while the reactivity of SCA with NR is the dominant factor governing the interfacial interaction. These findings establish SCA as a promising strategy for modifying NR/CB composites with low hysteresis loss and provide fundamental guidelines for rational interfacial engineering.
Data availability
The experimental procedures and analytical data are available within the manuscript and its ESl.†Author contributions
Ruoyan Huang: writing – review & editing, data curation, methodology, investigation. Dong Wang: investigation, data curation, methodology. Zhenghai Tang: writing – review & editing, visualization, conceptualization, supervision. Baochun Guo: supervision, conceptualization. Liqun Zhang: supervision.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The work was funded by the National Natural Science Foundation of China (52322303 and 52130305), and Guangdong Basic and Applied Basic Research Foundation (2023B1515020085). We appreciate USAXS Beamline BL10U1 at SSRF (Shanghai Synchrotron Radiation Facility, China) for providing the beamtime.Notes and references
- S. Fang, F. Li, J. Liu, L. Zhang, D. Wang, B. Liu, S. Wu, Z. Tang and B. Guo, Nano Energy, 2021, 83, 105822 CrossRef CAS.
- Y. Liu, Z. Tang, D. Wang, S. Wu and B. Guo, J. Mater. Chem. A, 2019, 7, 26867–26876 RSC.
- S. Wolff, Rubber Chem. Technol., 1996, 69, 325–346 CAS.
- L. Strohmeier, S. Bernd and S. Schlögl, Polym. Rev., 2022, 62, 142–174 CrossRef CAS.
- X. An, S. Wu, S. Xiong, S. Yu, Z. Tang, B. Guo and L. Zhang, J. Mater. Chem. A, 2023, 11, 8647–8655 RSC.
- D. Wang, Z. Tang, S. Fang, S. Wu, H. Zeng, A. Wang and B. Guo, Carbon, 2021, 184, 409–417 CrossRef CAS.
- S. Yu, Z. Tang, S. Fang, S. Wu and B. Guo, Composites, Part A, 2021, 149, 106589 CrossRef CAS.
- S. K. Srivastava and K. Manna, J. Mater. Chem. A, 2022, 10, 7431–7496 RSC.
- D. Wang, J. Chen, Z. Tang, R. Huang, Y. Xiao, J. Chen, P. Yin, B. Guo and L. Zhang, Macromolecules, 2024, 57, 470–480 CrossRef CAS.
- D. Wang, Z. Tang, R. Huang, H. Li, C. Zhang and B. Guo, Macromolecules, 2022, 55, 8485–8494 CrossRef CAS.
- Z. Tang, L. Zhang, W. Feng, B. Guo, F. Liu and D. Jia, Macromolecules, 2014, 47, 8663–8673 CrossRef CAS.
- Y. Fan, G. D. Fowler and M. Zhao, J. Cleaner Prod., 2020, 247, 119115 CrossRef CAS.
- M. M. Harussani, S. M. Sapuan, G. Nadeem, T. Rafin and W. Kirubaanand, Def. Technol., 2022, 18, 1281–1300 CrossRef.
- A. M. Kucherskii, Polym. Test., 2005, 24, 733–738 CrossRef CAS.
- S. Khodabakhshi, P. F. Fulvio and E. Andreoli, Carbon, 2020, 162, 604–649 CrossRef CAS.
- S. Fang, S. Wu, J. Huang, D. Wang, Z. Tang, B. Guo and L. Zhang, Ind. Eng. Chem. Res., 2020, 59, 21047–21057 CrossRef CAS.
- D. Wang, Z. Tang, R. Huang, Y. Duan, S. Wu, B. Guo and L. Zhang, Chem. Mater., 2023, 35, 764–772 CrossRef CAS.
- J. Gao, Y. Xing, L. An, X. Ye, H. Yao, H. Wang, X. Wu and L. Zhang, Polym. Compos., 2023, 44, 7997–8009 CrossRef CAS.
- S. Fang, S. Yu, S. Wu, J. Wang, Z. Tang and B. Guo, Compos. Sci. Technol., 2022, 225, 109482 CrossRef.
- W. J. Chung, J. J. Griebel, E. T. Kim, H. Yoon, A. G. Simmonds, H. J. Ji, P. T. Dirlam, R. S. Glass, J. J. Wie, N. A. Nguyen, B. W. Guralnick, J. Park, A. Somogyi, P. Theato, M. E. Mackay, Y. E. Sung, K. Char and J. Pyun, Nat. Chem., 2013, 5, 518–524 CrossRef CAS PubMed.
- H. Kim, J. Lee, H. Ahn, O. Kim and M. J. Park, Nat. Commun., 2015, 6, 7278 CrossRef CAS PubMed.
- T. S. Kleine, N. A. Nguyen, L. E. Anderson, S. Namnabat, E. A. LaVilla, S. A. Showghi, P. T. Dirlam, C. B. Arrington, M. S. Manchester, J. Schwiegerling, R. S. Glass, K. Char, R. A. Norwood, M. E. Mackay and J. Pyun, ACS Macro Lett., 2016, 5, 1152–1156 CrossRef CAS PubMed.
- F. Wu, S. Chen, V. Srot, Y. Huang, S. K. Sinha, P. A. van Aken, J. Maier and Y. Yu, Adv. Mater., 2018, 30, 1706643 CrossRef PubMed.
- T. Lee, P. T. Dirlam, J. T. Njardarson, R. S. Glass and J. Pyun, J. Am. Chem. Soc., 2021, 144, 5–22 CrossRef PubMed.
- S. Park, M. Chung, A. Lamprou, K. Seidel, S. Song, C. Schade, J. Lim and K. Char, Chem. Sci., 2022, 13, 566–572 RSC.
- J. Jia, J. Liu, Z.-Q. Wang, T. Liu, P. Yan, X.-Q. Gong, C. Zhao, L. Chen, C. Miao, W. Zhao, S. Cai, X.-C. Wang, A. I. Cooper, X. Wu, T. Hasell and Z.-J. Quan, Nat. Chem., 2022, 14, 1249–1257 CrossRef CAS PubMed.
- X. Wu, J. A. Smith, S. Petcher, B. Zhang, D. J. Parker, J. M. Griffin and T. Hasell, Nat. Commun., 2019, 10, 647 CrossRef PubMed.
- H. Berk, B. Balci, S. Ertan, M. Kaya and A. Cihaner, Mater. Today Commun., 2019, 19, 336–341 CrossRef CAS.
- Y. Zhang, T. S. Kleine, K. J. Carothers, D. D. Phan, R. S. Glass, M. E. Mackay, K. Char and J. Pyun, Polym. Chem., 2018, 9, 2290–2294 RSC.
- Y. Jin, C. Hu, Z. Wang, Z. Xia, R. Li, S. Shi, S. Xu and L. Yuan, ACS Sustain. Chem. Eng., 2023, 11, 11259–11268 CrossRef CAS.
- Y. Huang, Y. Liu, G. Si and C. Tan, ACS Sustain. Chem. Eng., 2024, 12, 2212–2224 CrossRef CAS.
- P. Yan, W. Zhao, B. Zhang, L. Jiang, S. Petcher, J. A. Smith, D. J. Parker, A. I. Cooper, J. Lei and T. Hasell, Angew. Chem., Int. Ed., 2020, 59, 13371–13378 CrossRef CAS PubMed.
- J. M. Scheiger, C. Direksilp, P. Falkenstein, A. Welle, M. Koenig, S. Heissler, J. Matysik, P. A. Levkin and P. Theato, Angew. Chem., Int. Ed., 2020, 59, 18639–18645 CrossRef CAS PubMed.
- I. Gomez, A. F. De Anastro, O. Leonet, J. A. Blazquez, H. J. Grande, J. Pyun and D. Mecerreyes, Macromol. Rapid Commun., 2018, 39, 1800529 CrossRef PubMed.
- T. Izgi, Ö. Alver, C. Parlak, M. T. Aytekin and M. Şenyel, Spectrochim. Acta, Part A, 2007, 68, 55–62 CrossRef CAS PubMed.
- Y. Zhang, J. J. Griebel, P. T. Dirlam, N. A. Nguyen, R. S. Glass, M. E. Mackay, K. Char and J. Pyun, Polym. Sci. Polym. Chem., 2017, 55, 107–116 CrossRef CAS.
- J. W. Thomson, K. Nagashima, P. M. Macdonald and G. A. Ozin, J. Am. Chem. Soc., 2011, 133, 5036–5041 CrossRef CAS PubMed.
- V. S. Wadi, K. K. Jena, K. Halique and S. M. Alhassan, ACS Appl. Polym. Mater., 2019, 2, 198–208 CrossRef.
- K. P. Patel, E. M. Gayakwad, V. V. Patil and G. S. Shankarling, Adv. Synth. Catal., 2019, 361, 2107–2116 CrossRef CAS.
- W. Li, H. Liu, H. Wang, Y. Chen, Y. Peng, H. Wu, Y. Hou, Y. Huang, Z. Yuan, B. Ye, H. Zhang and J. Wu, Chem. Mater., 2023, 35, 682–691 CrossRef CAS.
- T. B. Nguyen, L. Ermolenko and A. Al-Mourabit, Org. Lett., 2012, 14, 4274–4277 CrossRef CAS PubMed.
- Z. Sun, H. Huang, L. Li, L. Liu and Y. Chen, Macromolecules, 2017, 50, 8505–8511 CrossRef CAS.
- X. N. Zhang, Y. J. Wang, S. Sun, L. Hou, P. Wu, Z. L. Wu and Q. Zheng, Macromolecules, 2018, 51, 8136–8146 CrossRef CAS.
- Y. J. Wang, X. N. Zhang, Y. Song, Y. Zhao, L. Chen, F. Su, L. Li, Z. L. Wu and Q. Zheng, Chem. Mater., 2019, 31, 1430–1440 CrossRef CAS.
- S. Kumari, A. V. Carmona, A. K. Tiwari and P. C. Trippier, J. Med. Chem., 2020, 63, 12290–12358 CrossRef CAS PubMed.
- A. Choudhary and R. T. Raines, ChemBioChem, 2011, 12, 1801–1807 CrossRef CAS PubMed.
- S. Zhou, X. Zheng, X. Yu, J. Wang, J. Weng, X. Li, B. Feng and M. Yin, Chem. Mater., 2007, 19, 247–253 CrossRef CAS.
- T. Koga, T. Hashimoto, M. Takenaka, K. Aizawa, N. Amino, M. Nakamura, D. Yamaguchi and S. Koizumi, Macromolecules, 2008, 41, 453–464 CrossRef CAS.
- G. P. Baeza, A.-C. Genix, C. Degrandcourt, L. Petitjean, J. Gummel, M. Couty and J. Oberdisse, Macromolecules, 2013, 46, 317–329 CrossRef CAS.
- S. Wu, M. Qiu, Z. Tang, J. Liu and B. Guo, Macromolecules, 2017, 50, 3244–3253 CrossRef CAS.
- P. Zhang, M. Morris and D. Doshi, Rubber Chem. Technol., 2016, 89, 79–116 CAS.
- X. Shi, S. Sun, A. Zhao, H. Zhang, M. Zuo, Y. Song and Q. Zheng, Compos. Sci. Technol., 2021, 203, 108586 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5ta04129e |
| This journal is © The Royal Society of Chemistry 2025 |