A biomass derived jute carbon integrated FeCoNi alloy as a robust catalyst for alkaline water splitting†
Sobia
Dilpazir
 a,
Yuda Prima
Hardianto
b,
Muhammad
Imran
c,
Mohd. Yusuf
Khan
ad,
Md. Abdul
Aziz
a,
Abduljamiu
Amao
a,
Yuda Prima
Hardianto
b,
Muhammad
Imran
c,
Mohd. Yusuf
Khan
ad,
Md. Abdul
Aziz
a,
Abduljamiu
Amao
 e and
Abuzar
Khan
e and
Abuzar
Khan
 *a
*a
aInterdisciplinary Research Center for Hydrogen Technologies and Carbon Management (IRC-HTCM), Core Research Facilities, King Fahd University of Petroleum & Minerals, Box 5040, Dhahran 31261, Saudi Arabia. E-mail: abuzar@kfupm.edu.sa
bPhysics Department, King Fahd University of Petroleum & Minerals, KFUPM Box 5047, Dhahran 31261, Saudi Arabia
cDepartment of Chemistry, Government College University, Faisalabad, Pakpattan Road, Sahiwal, Punjab 57000, Pakistan
dMaterials Science and Engineering Department, King Fahd University of Petroleum & Minerals, Dhahran, 31261, Saudi Arabia
eCenter for Integrative Petroleum Research, College of Petroleum Engineering and Geosciences, King Fahd University of Petroleum & Minerals, Dhahran 31261, Saudi Arabia
First published on 14th July 2025
Abstract
Sustainable energy solutions demand efficient electrocatalysts for hydrogen and oxygen evolution reactions. This study presents an innovative approach of utilizing inexpensive biomass, specifically jute carbon incorporating a tri-metallic alloy of iron, cobalt and nickel (JCM) as a bifunctional catalyst for high-performance alkaline water splitting. The JCM catalyst demonstrates superior electrocatalytic activity, with significantly reduced overpotentials and excellent long-term stability. Its hierarchical porous structure facilitates the dispersion of alloyed metal nanoparticles, improving mass transport and charge transfer. Electrochemical testing reveals that the JCM catalyst has the lowest charge transfer resistance and higher double layer capacitance, contributing to its superior bifunctional performance. It achieves a low overpotential of 123 mV for the HER and 230 mV for the OER with promising HER and OER stability while maintaining consistent performance for over 100 hours, while in a real laboratory electrolyzer, it maintains a satisfactory operational stability for 24 hours. Density functional theory simulations indicate an optimal hydrogen adsorption energy (ΔG = −3.67 eV), supporting its promising HER performance. These findings suggest that jute-derived, metal alloy-integrated porous carbon is a robust, efficient, and cost-effective bifunctional electrocatalyst for alkaline water splitting, making it a promising candidate for sustainable and scalable renewable energy applications.
1. Introduction
The demand for sustainable energy solutions has intensified due to the escalating energy crisis driven by the depletion of traditional energy sources and the worsening environmental challenges posed by greenhouse gas emissions. The global community recognizes that the development of sustainable and renewable energy storage technologies is not only beneficial but also a necessity for addressing these multifaceted issues.1–3 Among various available renewable energy resources including hydroelectric, wind, tidal, and solar energy, hydrogen production through water splitting is regarded as one of the most promising options due to its environmental benignity and cost-effectiveness.4–7However, the efficiency of water splitting is fundamentally limited by the electrochemical reactions involved, specifically the oxygen evolution reaction (OER)8–11 and the hydrogen evolution reaction (HER).12–15 These reactions are crucial to renewable energy systems, yet they face significant activation losses and higher overpotentials. This is primarily due to the complex processes involving multi-electron transfers and the breaking of thermodynamically stable bonds of water H–O–H (248 kJ mol−1).16–18 Currently, noble metals such as platinum (Pt), ruthenium (Ru), and their oxides (RuO2 and IrO2) are considered the most advanced catalysts for these reactions. However, their high cost, scarcity, and poor stability, especially for alkaline or neutral electrocatalytic water splitting, restrict their large scale industrial application.13,19,20 In response to these limitations, there has been a significant shift in focus toward non-precious metal-based electrocatalysts that can deliver significant catalytic activity while being more economically viable for widespread use. As a result, rigorous efforts have been devoted to the exploration of advanced electrocatalytic materials that not only enhance the reaction kinetics but also demonstrate stability and efficiency under harsh chemical conditions. The development of these non-precious metal catalysts has opened new avenues for sustainable energy conversion, aligning with the global objectives of reducing reliance on fossil fuels and advancing clean energy technologies.21–25
The efficiency of electrochemical devices is significantly influenced by the nature of their electrode materials. Among the various materials employed in electrochemical systems, activated carbon (AC) has gained prominence due to its high surface area, excellent electrical conductivity, and tunable porosity.26,27 AC derived from biomass sources presents a sustainable and economically sound alternative to conventional carbon materials, aligning with the principles of a circular economy. Biomass-derived porous carbon materials have the potential to bridge the gap between performance and sustainability in electrocatalysis. Their environmental benefits are significant, as they utilize renewable biomass resources, reducing reliance on non-renewable materials and contributing to a circular economy.28–31 The carbon structure itself can be optimized through various synthesis techniques, such as pyrolysis, chemical activation, or templating methods, to enhance surface area, porosity, and conductivity. These enhancements make biomass-derived carbons particularly effective as a support for non-noble transition metal catalysts. Recent studies have shown that incorporating biomass-derived carbon materials with transition metals such as Fe, Ni, and Co can enhance their electrocatalytic activity for the OER and HER32,33 by improving the electrical conductivity of the carbon matrix and increasing the number of active sites available for catalysis. Specifically, tri-metallic alloys with transition metals including Fe, Co, and Ni improve electrocatalytic performance due to their distinct synergistic effects. In order to obtain outstanding bifunctional activity for the HER and OER, we focused on integrating an FeCoNi alloy into a biomass-derived porous carbon. Hoang et al. used cauliflower leaves to synthesize Ni/NiO nanoparticles (NiOx-AC-500) fixed in nitrogen-doped activated carbon which exhibited an overpotential of 346 mV for the OER and 180 mV for the HER.34 Lv et al. prepared a catalyst with a heterojunction (Co2N0.67-BHPC) by using mushrooms as N and P sources by a pot pyrolysis method which demonstrated an overpotential of 340 mV for the OER and 210 mV for the HER in 1 M KOH.35 The electrocatalyst derived from carrot co-doped with Ni and P (PC-Ni0.75) exhibited an overpotential of 297 mV for the HER in an alkaline medium while 290 mV for the OER in the same medium.36 Yang et al. reported a porous carbon electrocatalyst (FeNi-NC) using peanut shells as a precursor and a co-doped FeNi alloy and nitrogen with an overpotential of 380 mV for the OER in an alkaline medium.37 Chang et al. reported a bifunctional NiFe alloy-embedded N-doped porous carbon electrocatalyst (NiFe@NC) for the OER and HER with an overpotential of 270 mV and 206 mV respectively.38 Multimetallic metal–organic aerogel electrocatalysts (MG-FeNiCo) are assembled from 1,3,5-benzenetricarboxylic acid and Fe3+, Co2+, and Ni2+ nitrates via a facile one-pot method reported by Zhang et al.39 demonstrating an overpotential of 248 mV and 272 mV at a current density of 20 mA cm−2 and 50 mA cm−2. Additionally, literature reports suggest that incorporating a small amount of Fe (up to 10%) into metal based catalysts significantly enhances electrocatalytic performance by modifying the electronic structure and optimizing adsorption energies for oxygenated intermediates.40 Studies have demonstrated that Fe incorporation leads to improved OER and HER activity due to its ability to tune the d-band center of Co and Ni, thereby facilitating charge transfer and increasing active site exposure.41 Moreover, a lower Fe content minimizes the formation of inactive oxide species, which can hinder long-term catalytic stability. By keeping Fe below 10%, we ensure optimal synergy between transition metals while maintaining high conductivity and structural integrity.42
Corchorus capsularis (jute) is an abundant and low-cost (∼0.5 USD per kg) biomass feedstock, particularly in regions such as India and Bangladesh. This natural fiber is not only economically advantageous but also possesses unique structural features that facilitate efficient mass and charge transport. The application of jute-derived porous carbon materials, especially when modified with transition metals, represents a significant advancement in the field of electrochemical water splitting. By utilizing cost-effective and widely available biomasses such as jute, materials that are both sustainable and efficient for energy applications could be developed.
In this study, we present an innovative approach that integrates a tri-metallic alloy with jute-derived carbon, denoted as the jute carbon matrix (JCM), to enhance performance of electrochemical water splitting. The preparation process involves chemical exfoliation and activation of jute carbon followed by integrating metals as alloy/s, which affects the structural and electrochemical characteristics of the resultant AC. By systematically evaluating the performance of these jute-derived porous carbon materials for both the OER and HER in alkaline electrolytes, we aim to improve their efficiency and stability as sustainable electrocatalysts for water electrolysis. The incorporation of the transition metal alloy into jute-derived carbon considerably enhanced the electrochemical performance of these materials. The initial results indicated that the jute-derived carbon doped with transition metals exhibits significantly low overpotentials for both the HER and OER, suggesting its potential as a viable alternative to conventional noble metal catalysts.
2. Experimental
2.1. Chemicals
Potassium hydroxide (KOH), hydrogen peroxide (H2O2), sulfuric acid (H2SO4), nickel nitrate hexahydrate (Ni(NO3)2·6H2O), cobalt nitrate hexahydrate (Co(NO3)2·6H2O), iron nitrate nonahydrate (Fe(NO3)3·9H2O), and ammonium hydroxide (NH4OH) were utilized as received from Merck without any further treatment. Deionized water was used for making solutions and washing purposes.2.2. Synthesis
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 and left for 5 hours. The resulting powdered mixture was subjected to pyrolysis in a tube furnace at 850 °C, for 2 hours with a controlled ramp rate of 5 °C min−1 followed by cooling to room temperature. The resulting carbon was subjected to further heat treatment at 2700 °C for 5 hours under an argon atmosphere at a heating rate of 10 °C min−1 and a cooling rate of 5 °C min−1. The jute carbon powder obtained after carbonization was initially hydrophobic. It was treated with H2SO4 and H2O2 to exfoliate and introduce hydrophilicity, resulting in JCT.
4 and left for 5 hours. The resulting powdered mixture was subjected to pyrolysis in a tube furnace at 850 °C, for 2 hours with a controlled ramp rate of 5 °C min−1 followed by cooling to room temperature. The resulting carbon was subjected to further heat treatment at 2700 °C for 5 hours under an argon atmosphere at a heating rate of 10 °C min−1 and a cooling rate of 5 °C min−1. The jute carbon powder obtained after carbonization was initially hydrophobic. It was treated with H2SO4 and H2O2 to exfoliate and introduce hydrophilicity, resulting in JCT.
3. Results and discussion
3.1. Structural characterization
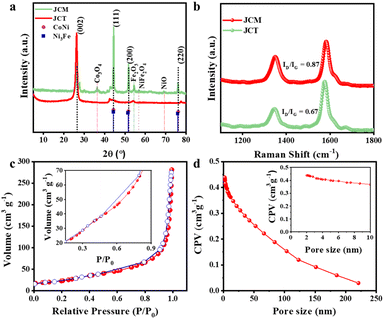
The development of the JCM from its precursor JCT employed a novel strategy for integrating a trimetallic alloy into the JCT matrix. The functional groups introduced into JCT interact synergistically with the metal ions inducing charge redistribution throughout the carbon framework to create an optimal electronic environment. Additionally, the pores of the carbon provide adsorption sites for these metal ions. Upon addition of aqueous ammonia, metal ions form complexes enhancing their adsorption onto the carbon matrix. The addition of ammonia raises the pH of the solution, potentially leading to the precipitation of insoluble metal hydroxides. At higher temperature and under an inert Ar atmosphere, which serve as reducing conditions suitable for alloy formation, excess ammonia decomposes, facilitating the formation of well-dispersed metal/metal alloy nanoparticles within the carbon matrix. The hierarchical porous structure of jute-derived carbon materials enhances mass transport and maximizes active site exposure. Additionally, incorporating trimetallic alloy nanoparticles improves electrical conductivity and introduces more active sites, significantly enhancing catalytic performance for both the HER and OER. This integrated approach exhibits superior stability compared to carbon-only or single-metal systems, positioning jute-derived trimetallic porous carbon materials as a sustainable and innovative option for high-performance electrocatalysts in advanced energy conversion technologies.21,44–46XRD analysis was performed to study the structural transformation and crystallographic structure of JCT to the JCM, focusing on phase purity and graphitization. The XRD analysis results for JCT and the JCM presented in Fig. 2a depict that the JCT exhibits a distinct diffraction peak at 2θ = 26.2° attributed to C (002) plane suggesting a reasonably high level of graphitization reported by Shah et al. from our group.43 The oxidative treatment of jute carbon, aimed at exfoliation and functionalization, introduces greater disorder, perhaps due to oxygen-containing functional groups. This is evidenced by the JCT exhibiting a similar but slightly broadened C (002) peak, indicating the conversion of reduced graphitic peaks to GO and subsequently enhancing its hydrophilicity that facilitates interaction with transition metals. The XRD patterns of the JCM exhibit distinct peaks corresponding to the transition metals, in addition to the typical carbon peaks. Interestingly, there is a slight shift in the peaks of Fe, Co and Ní due to alloy formation by FeCoNi; however, the FCC structure remains predominant in the JCM. The FeCoNi alloy exhibits the combined features of FeNi and CoNi and CoFe alloys. Corresponding to the FCC structure of the JCM distinct peaks at 2θ = 44.1°, 50.8°, 75.3° are attributed to the (111), (200), and (220) lattice planes of the FeCoNi alloy respectively (JCPDS card no. 01-074-5694, 47-1405 JCPDS card no. 47-1417 Co–Ni Alloy JCPDS card no. 04-085) while the peak at 2θ = 26.3° is indexed to the (002) graphitic plane of carbon (JCPDS card no. 75-1621). The peaks centered at 2θ = 36.7° and 54.1° correspond to the (311) plane of Co3O4 (JCPDS card no. 42-1467) and (116) plane of α-Fe2O3 (JCPDS card no. 19-0629) respectively47–49 while a minor peak for NiO corresponding to the 311 plane (JCPDS card no. 47-1049) is observed at 2θ = 69.2°. The relatively low intensities of these oxide peaks indicate minimal contribution from these oxides validating that the FeCoNi alloy is the predominant phase in the designed catalyst. In addition a small peak observed at 2θ = 56.9° corresponds to the 511 plane of NiFe2O4 (JCPDS card no. 01-074-2081). The XRD results demonstrate that while both materials have some graphitic properties, their structural properties are influenced by exfoliation and metal incorporation. The JCM was annealed in a reducing H2 environment to eliminate the possibility of oxide or hydroxide formation during carbonization under Ar. XRD patterns of the JCM carbonized in Ar and in 10% H2 with Ar balance were compared (Fig. S2†) revealing nearly identical peak positions with minor contribution from NiO at 2θ = 69.2° and NiFe2O4 at 2θ = 56.9°, validating the phase purity of the tri-metallic alloy. The structural characteristics of carbon-based materials, particularly their graphitic and defective nature, are analyzed using Raman spectroscopy (Fig. 2b). All samples exhibit two conspicuous peaks in their Raman spectra, positioned at about 1580 cm−2 (G-band) and 1320 cm−2 (D-band). The presence of graphitic carbon is indicated by the stretching of C–C bonds, which leads to the G-band associated with the in-plane E2g vibrational mode. Graphitic lattice defects reflecting disorder are indicated by the D-band, which is associated with the A1g vibrational mode.50,51 The defect density in carbon materials can be assessed using the intensity ratio of the D-band and G-band (ID/IG). The Raman analysis results demonstrate that the JCM exhibited a higher ID/IG ratio of 0.87 than the intensity ratio of 0.64 of JCT, indicating that more defects are introduced by alloy formation. These defects are known to improve the overall electrochemical performance and increase electrical conductivity of the material and thereby the JCM is anticipated to exhibit improved electrochemical performance for water electrolysis.
Porosity is one of the most important factors for the incorporation of transition metals into carbon materials since it directly influences the dispersion and accessibility of metal species on the carbon matrix. Interactions between graphitic structures and oxygen-containing functional groups may improve the integration of transition metals such as Fe, Co, and Ni into porous carbon materials. Hierarchical porosity also helps in preventing the agglomeration of metal nanoparticles, which ensures effective and stable catalytic activity. The porosity of JCT is analyzed using nitrogen adsorption–desorption studies. Fig. 2c illustrates the nitrogen adsorption/desorption isotherms of JCT as obtained before43 exhibiting a mesoporous structure characterized by a typical type IV isotherm and hysteresis loop (inset) in the relative pressure range of 0.15–0.99 P/Po. Moreover, the hierarchical porous nature of JCT is further confirmed by the sharp increase in the adsorption loop at higher pressures (P/Po > 0.9), indicating the presence of macropores. The specific surface area of JCT was calculated to be 1100 m2 g−1, according to the Brunauer–Emmett–Teller (BET) method. The Barrett–Joyner–Halenda (BJH) pore size distribution curves are shown in Fig. 2d demonstrating a wide range of pore sizes, from about 1.5 nm to ∼200 nm by JCT with an average BJH pore diameter of 15 nm. The functional properties of JCT are enhanced by its hierarchical structure, which combines micro-, meso-, and macropores. Ions can be captured by micropores, transported by mesopores, and diffused into the bulk by macropores. By facilitating effective ion diffusion, the porous structure improves the electrochemical efficiency of energy conversion devices while simultaneously decreasing resistance. The hierarchical porous structure enabled the effective incorporation of the metal alloy with different atomic radii, thereby providing junctures for synergy between the porous carbon and transition metals.
The morphology of JCT and the JCM was investigated using FESEM (Fig. S3–S8†). The FESEM micrographs of JCT and the JCM at different magnifications with distinct features are displayed in Fig. 3a–d. Chemical exfoliation with H2SO4 and H2O2 followed by high temperature annealing leads to exfoliation of carbon sheets. Both JCT and the JCM have nanosheet like structures which are arranged in a hierarchical structure with micropores, mesopores and macropores facilitating mass and charge transport. However, the JCM is distinguished by the presence of truncated octahedral shaped FeCoNi alloy nanoparticles (∼40–60 nm) dispersed within the graphitic sheets. This morphology is characteristic of FCC alloys. The porous graphitized carbon support provided a stable platform for nanoparticle growth and dispersion, facilitating efficient mass transfer and homogeneous distribution within the carbon sheets. The XRD analysis corroborated the formation of FCC-structured FeNi, CoNi, and CoFe alloy phases, consistent with the truncated octahedral morphology. Annealing the sample at 850 °C for 2 hours under an inert Ar atmosphere further promoted alloying of the metal components, contributing to uniform particle size and morphology.
Energy dispersive X-ray spectroscopy of the synthesized electrocatalyst showed successful formation of the metal alloy dispersed on jute carbon derived carbon with approximately equal concentrations of Co and Ni while Fe is in trace as displayed in Fig. 3e and f. The detailed elemental mapping confirmed the presence of C, Co, Ni and Fe in the JCM (Fig. S9†). The compositions obtained from the EDS analysis agree well with the results from ICP-MS analysis with relative atomic percentages of 9% Co, 7% Ni, and 0.8% Fe within the carbon framework (alongside 86% carbon). The consistency between these results confirms the uniform distribution of the metal alloy in the porous carbon matrix, as visualized through EDS mapping. Fe, though present in a smaller amount (<10% of the total metal content), acts as a key dopant, enhancing the electronic environment and catalytic activity. The detailed morphology and structure of JCT and the JCM were further studied using TEM analysis. Additional details of the structure of the JCM through TEM analysis at different magnifications are provided in Fig. S10–S12.† It can be seen that the JCT is composed of a very thin and porous uniform layered sheet like structure (Fig. 3g) while the prepared catalyst JCM is also composed of layers of extremely thin sheets but with evenly distributed metal nanoparticles, as illustrated in Fig. 3h and i. The metal alloy nanoparticles are embedded between the sheets of the JCM (Fig. S12†). This encapsulation highlights the integration of the metal alloy nanoparticles with the carbon structure, enhancing local electronic conductivity and providing more accessible electrochemical reaction sites. The HRTEM analysis reveals graphitized carbon sheets, showing distinct fringes that correspond to the (002) planes, with a d-spacing of approximately 0.33 nm evinced in Fig. 3j. These fringes indicate an ordered arrangement of carbon atoms, highlighting the crystalline nature of the material. Importantly, the metal alloy nanoparticles are encapsulated within the thin carbon sheets, which obscure their fringes, as illustrated in Fig. 3k. The SAED pattern presented in Fig. 3l displays clear, well-defined rings characteristic of a crystalline structure. These rings signify the presence of specific crystal planes within the sample. However, the diffraction spots appeared somewhat diffused due to the embedding of the metal alloy nanoparticles in the carbon sheets. This embedding likely caused scattering and overlapping, diminishing the brightness of the spots. Despite this, the clarity of the rings in the SAED pattern confirms the crystalline nature of the material.
XPS survey analysis of JCT shows the presence of carbon and oxygen, while for the JCM, it confirmed the presence of C, O, Fe, Co, and Ni elements (Fig. S13 and S14†). The C 1s XPS spectrum of JCT is presented in Fig. S15†, while for the JCM, C 1s can be deconvoluted into peaks for sp2-hydridized carbon (284.6 eV), C–O bonds (286.2 eV) and the O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group (288.1 eV) (Fig. 4a). The Fe 2p XPS spectrum is resolved into 4 peaks for Fe0 at 705.9 eV, while the peaks at 709.2 eV and 720.1 eV correspond to 2p1/2 and 2p3/2 for the Fe–O bond respectively. The peaks at 714.9 eV and 735 eV are attributed to shake up satellites (Fig. 4b). The Co 2p XPS spectrum is resolved into two pairs of 2p3/2/2p1/2 doublets corresponding to metallic Co2+, Co–O, metallic Co and shake up satellites centered at 780.2 eV, 781.7 eV, 795 eV 786 eV and 803.0 eV respectively (Fig. 4c). Ni 2p exhibited five peaks with two satellite peaks centered at 843.8 eV and 878 eV while three additional signals corresponding to metallic and oxidized Ni were observed at 850.9 eV, 858.6 eV and 863.9 eV, respectively (Fig. 4d). The high resolution oxygen XPS of JCT exhibits only an organic oxygen peak (Fig. S16†) while for the JCM the O 1s is deconvoluted to 2 major peaks corresponding to organic oxygen and metal bonded oxygen at 531.8 eV, and 533.6 eV respectively (Fig. S17†). The presence of Fe–C and metallic oxygen indicates the presence of M–O–C linkage which is the possible active site for electrocatalysis due to its polarity, effectively representing the synergistic effect between the alloy, carbon and oxygen.
O group (288.1 eV) (Fig. 4a). The Fe 2p XPS spectrum is resolved into 4 peaks for Fe0 at 705.9 eV, while the peaks at 709.2 eV and 720.1 eV correspond to 2p1/2 and 2p3/2 for the Fe–O bond respectively. The peaks at 714.9 eV and 735 eV are attributed to shake up satellites (Fig. 4b). The Co 2p XPS spectrum is resolved into two pairs of 2p3/2/2p1/2 doublets corresponding to metallic Co2+, Co–O, metallic Co and shake up satellites centered at 780.2 eV, 781.7 eV, 795 eV 786 eV and 803.0 eV respectively (Fig. 4c). Ni 2p exhibited five peaks with two satellite peaks centered at 843.8 eV and 878 eV while three additional signals corresponding to metallic and oxidized Ni were observed at 850.9 eV, 858.6 eV and 863.9 eV, respectively (Fig. 4d). The high resolution oxygen XPS of JCT exhibits only an organic oxygen peak (Fig. S16†) while for the JCM the O 1s is deconvoluted to 2 major peaks corresponding to organic oxygen and metal bonded oxygen at 531.8 eV, and 533.6 eV respectively (Fig. S17†). The presence of Fe–C and metallic oxygen indicates the presence of M–O–C linkage which is the possible active site for electrocatalysis due to its polarity, effectively representing the synergistic effect between the alloy, carbon and oxygen.
3.2. Electrochemical performance evaluation
The detailed characterization of the synthesized electrocatalysts revealed valuable information that indicated their desirable features for an electrocatalyst. Therefore, we evaluated the reaction kinetics for the OER and HER using our synthesized catalysts, JCT and JCM, through a range of electrochemical analysis techniques. The initial assessment of the synthesized catalysts' electrochemical performance was made by analyzing HER activity. The electrodes for testing were prepared by applying a homogeneous coating layer of the synthesized catalysts (JCT and JCM) on pretreated carbon cloth (ESI†). A conventional three-electrode setup and 1.0 M KOH electrolyte were used for this purpose. Polarization curves were recorded using linear sweep voltammetry (LSV), with a scan rate of 5 mV s−1, as illustrated in Fig. 5a. Incorporation of the FeCoNi alloy onto the porous carbon framework support significantly enhanced HER activity. The JCM catalyst exhibited the highest HER activity, with a minimal overpotential and required overpotential (η10) of only 123 mV to reach a current density of −10 mA cm−2. This represents a mere 90 mV difference compared to the benchmark Pt/C catalyst, highlighting the effectiveness of our developed electrodes over precious metal catalysts. The overpotential of the JCM is significantly improved as compared to the parent JCT matrix exhibiting an η10 of 430 mV at a current density of 10 mA cm−2 as shown in Fig. 5a. This indicates that it outperformed the parent JCT and other electrocatalysts reported in recent literature (Table S2†). Additionally, JCM's overpotential is superior to that of other transition metal (TM)-based and Fe, Co, and Ni based HER electrocatalysts. To compare other metal compositions using jute derived carbon we have tested a series of binary and ternary metal compositions as illustrated in Fig. S18 and S19† revealing that our catalyst JCM outperformed the other reference compositions and metal loadings. In addition to overpotential the kinetics of the reaction is also a crucial factor for assessing the electrochemical performance of the catalysts. The kinetics of the HER was evaluated through corresponding Tafel plots. The Tafel equation i.e. (η = b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) j + a), where η is the overpotential, b denotes the Tafel slope, j represents the current density, and a is the constant, was used to linearly fit the Tafel plots obtained from LSV curves to evaluate the OER kinetics of the electrocatalysts. A change in the mechanism/kinetics of the reaction on the catalyst surface is exclusively associated with a change in the Tafel slope. Therefore, Tafel slope values can be used to infer the reaction mechanism and kinetics. However, the interpretation of the aforementioned values is not necessarily straightforward, since higher Tafel slopes are typically reported since multiple reactions may occur at the same potential. Additionally, these values may be impacted by changes in the transfer coefficients. The strength of the M–H bonds is very important for the hydrogen evolution reaction kinetics of electrocatalysts. Sabatier's principle states that the M–H bonds on electrocatalysts should be optimally balanced i.e. they should be neither too weak nor too strong to promote the production of M–H intermediates and the subsequent release of H2. Consequently, the hydrogen binding energy has been recognized as a HER activity descriptor.
j + a), where η is the overpotential, b denotes the Tafel slope, j represents the current density, and a is the constant, was used to linearly fit the Tafel plots obtained from LSV curves to evaluate the OER kinetics of the electrocatalysts. A change in the mechanism/kinetics of the reaction on the catalyst surface is exclusively associated with a change in the Tafel slope. Therefore, Tafel slope values can be used to infer the reaction mechanism and kinetics. However, the interpretation of the aforementioned values is not necessarily straightforward, since higher Tafel slopes are typically reported since multiple reactions may occur at the same potential. Additionally, these values may be impacted by changes in the transfer coefficients. The strength of the M–H bonds is very important for the hydrogen evolution reaction kinetics of electrocatalysts. Sabatier's principle states that the M–H bonds on electrocatalysts should be optimally balanced i.e. they should be neither too weak nor too strong to promote the production of M–H intermediates and the subsequent release of H2. Consequently, the hydrogen binding energy has been recognized as a HER activity descriptor.
Although the overpotential of the JCM is slightly higher than the overpotential of Pt/C, the Tafel slope of JCM is smaller than that of Pt/C, indicating the faster kinetics realized using the JCM electrode. The Tafel slope of the JCM is 62 mV dec−1 lower than that of Pt/C's 70 mV dec−1 and JCT's at 130 mV dec−1 (Fig. 5b). This suggests favorable and faster desorption of adsorbed hydrogen at the catalyst surface. The smallest Tafel slope by the JCM implies that kinetics of the reaction is significantly improved through incorporation of a transition metal alloy into the carbon matrix and through the synergistic effect between electronic structures of these metals. As a result, the HER on the JCM is characterized by the adsorption of hydrogen atoms onto metal sites involving d orbitals of Fe, Co, and Ni, followed by the formation and desorption of hydrogen gas through the Volmer–Heyrovsky mechanism. This mechanism demonstrates the critical role of the catalyst's structural characteristics and electronic interactions in improving electrocatalytic performance.
The electrochemical active surface area (ECSA) measures the number of active sites available for the reaction, where a higher value indicates greater surface activity. The capacitive current density is directly related to ECSA which was measured by recording CVs at different scan rates (Fig. S20 and S21†). For the JCM, the slope derived from the capacitive current density at different scan rates is 207 mF cm−2, significantly higher than JCT's slope of 66 mF cm−2 (Fig. 5c), confirming JCM's superior electrochemical active surface area and active site accessibility. Charge transfer resistance (Rct) is evaluated using EIS which reflects the efficiency of electron transport at the electrode–electrolyte interface. The JCM exhibits an exceptionally low Rct of 2.9 Ω (Fig. 5d), indicating highly efficient electron transfer and rapid reaction kinetics compared to JCT. The equivalent circuit parameters obtained from fitting the EIS data are presented in Table S3.†
Stability is a crucial aspect to assess the commercial applicability of electrocatalysts for various reasons. For industrial application it is imperative to maintain good stability and durability as well since operational losses may lead to poor efficiency, low output, increased cost for frequent catalyst replacement and inability to achieve sustained efficiency. In addition to stability, durability is also an important factor for commercial application and cost effectiveness of the electrocatalyst. The stability of the catalyst was analyzed by chronoamperometry. The JCM catalysts exhibited exceptional stability for more than 100 hours without any noticeable current losses highlighting their potential for use in commercial applications (Fig. 5e and f). The remarkable stability of the JCM in terms of high current retention and stable overpotential supports its promise for extended use in the HER and other electrochemical applications, making it a strong contender for environmentally friendly, cost effective and facile power sources.
The growing need for sustainable energy solutions draws attention to the need to improve the efficiency of various electrochemical reactions in addition to the HER. Recognized for its comparatively large overpotential compared to the hydrogen evolution reaction, the oxygen evolution reaction is an important process in electrochemical water splitting controlling the cell potential for overall water splitting. Several factors contribute to the high overpotential of the OER, such as the inherent thermodynamic stability of water molecules, the multi-electron transfer process, and the need for overcoming the kinetic barriers associated with the reaction. A large amount of energy is often required to enable the formation of O–O bonds during the reaction in conventional OER systems requiring exceptionally remarkable catalysts. Depending on the importance of these crucial roles of the OER, new materials that can more effectively facilitate these reactions have been the focus of research and are extensively being explored. Our biomass derived electrocatalyst JCM is one such novel material and was assessed in an ambient atmosphere for its OER performance.
LSV was performed at different scan rates and performance was compared at a scan rate of 5 mV s−1 using a traditional three-electrode configuration with a 1.0 M KOH electrolyte to acquire polarization curves. The results revealed that the hybrid JCM exhibited remarkable OER performance, requiring a minimal overpotential (η10) of only 230 mV to achieve a current density of 10 mA cm−2 (Fig. 6a). Considering that it is 180 mV less than that of the standard IrO2 catalyst, this result is quite remarkable and demonstrates the superior performance of the JCM when compared to conventional precious metal catalysts. In contrast, the advantages of multi-metal alloy incorporation into the biomass derived carbon matrix were demonstrated by substantially higher overpotential of the parent JCT matrix for realizing the same current density at 410 mV.
The exceptional performance of the JCM can be ascribed to a number of important components and key factors throughout the framework. One important key factor is the additive impact that occurs when transition metals have been integrated within the porous and conductive carbon matrix. Tang et al. demonstrated that electrochemical activity of carbon-based materials is improved when metals such as cobalt, nickel, and iron are introduced into the matrix because these elements optimize electronic interactions.52 Adsorption and desorption, the two vital processes for efficient OER, are rendered more facile by this modification; thereby, a more balanced ratio of the strengths of metal–oxygen (M–O) bonds is achieved by electronic property tuning and is proved crucial for effective catalytic activity.53,54 Our biomass derived metal alloy integrated carbon exhibits an altered electronic structure as compared to JCT, resulting in active discerns which enhance hydroxide adsorption. Consequently, the JCM has an advantageous intermediate adsorption profile, which speeds up the OER and helps keep overpotential low. To validate the kinetics of the OER, the Tafel slopes of the prepared catalysts were evaluated. A much lower value of 58 mV dec−1 was observed for the Tafel slope for the JCM compared to the values recorded for IrO2 (83 mV dec−1) and JCT (103 mV dec−1). The reaction kinetics is improved by the smaller Tafel slope, implying that the JCM enables more rapid desorption of oxygen at the catalyst surface (Fig. 6b). The significance of improving kinetic parameters to enhance electrocatalyst performance has been previously demonstrated in the literature, and our results align with this improvement.
For the JCM, the slope calculated from CV at different scan rates (Fig. S22 and S23†) for the capacitive current density is 36 mF cm−2 is higher than JCT's slope of 32 mF cm−2 (Fig. 6c), reflecting improved active site availability for the JCM and higher ECSA attributed to its hierarchical porous structure and the incorporation of the transition metal alloy. EIS further evaluates the charge transfer resistance (Fig. 6d). A lower charge transfer resistance (Rct) indicates faster electron transfer and improved catalytic kinetics. The JCM exhibits a significantly lower Rct compared to JCT, confirming its superior charge transfer efficiency during the oxygen evolution reaction (OER). The equivalent circuit parameters derived from fitting the electrochemical impedance spectroscopy (EIS) data are provided in Table S4.†
The stability of catalysts is the most critical factor for evaluating the economic feasibility of electrocatalysts for practical and industrial applications particularly for complex OER electrocatalysis. We used chronoamperometry to comprehensively evaluate the stability of the JCM catalyst and an exceptional stability was demonstrated by the JCM as it maintained stable performance for more than 100 hours without any notable current losses with a current retention of >99% (Fig. 6e and f). The remarkable stability of the JCM highlights its potential for use in commercial settings over the long term. Stability of active sites and material degradation are two of the aspects that affect OER electrocatalyst stability. Challenges such as metal leaching and structural collapse because of extreme conditions are common. These concerns are mitigated by JCM's distinctive composition and structural characteristics. Using multi-metals (alloys) instead of a single metal in the carbon matrix improves catalytic activity and strengthens the structure, reducing the possibility of leaching and also preventing agglomeration. Furthermore, the carbon produced by jute possesses a large surface area and porosity, which allows for effective mass movement and reduces the production of inactive species during prolonged use. In practical scale up applications such as water electrolysis, where consistent performance is crucial for the overall efficiency of the system, such stability is imperative. Based on impressive OER and HER, water splitting in alkaline electrolyte was analyzed with a cell potential of only 260 mV (Fig. S24†).
In addition to promising electrochemical performance for the OER and HER in an alkaline medium, the JCM catalyst fairly retains its structure and morphology after 100 hours of operation both in the HER and OER as illustrated in (Fig. S25–S31†).
Keeping in view the promising stability of the JCM and to further evaluate the performance and durability of the JCM electrocatalyst in real systems an alkaline AEM electrolyzer was constructed with a membrane electrode assembly on the laboratory scale as illustrated in Fig. 7a. The alkaline AEM electrolyzer primarily comprises cathodic and anodic flow fields, gaskets, a JCM coated cathode, an anion exchange membrane, and JCM catalyst coated electrodes. The JCM is used as both the anode and cathode and acts as a bifunctional electrocatalyst in membrane electrode assembly (MEA) which was connected to an electrolyte reservoir, a temperature control unit and a potentiostat. Fig. 7b illustrates that the JCM made AEM electrolyzer maintained stable operation for 24 h at a current of 0.82 A at 70 °C by applying a voltage of only 1.8 V highlighting its robustness for industrial applications.
 | ||
| Fig. 7 (a) The photograph of a lab scale alkaline AEM electrolyzer built using the JCM as both the cathode and anode. (b) The durability of the AEM electrolyzer at 0.82 A. | ||
To confirm the electrochemical performance of the prepared electrode regarding its electrocatalytic properties for the HER, theoretical studies using density functional theory simulations and an electrical circuit model were conducted by structure optimization with details provided in the ESI (Fig. S32–S35).† The DFT simulations focused on evaluating the electrocatalytic properties by determining the hydrogen adsorption energy on the surface of the electrode material. The overall HER pathway can be described by the following equation, which serves as a reference for calculating the adsorption energy:
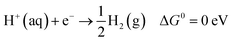 | (1) |
The HER is a multistep process that involves the adsorption of hydrogen ions and their reduction to form H2 molecules. This process is typically described by the Volmer–Tafel and Volmer–Heyrovsky mechanisms. As shown in eqn (1), there is no difference in the total Gibbs free energy between the initial state (H+ (aq) + e−) and the product state 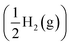 . Therefore, the total Gibbs free energy at the intermediate state, when hydrogen is adsorbed onto the catalyst, can be used to evaluate HER performance using the following eqn (2):
. Therefore, the total Gibbs free energy at the intermediate state, when hydrogen is adsorbed onto the catalyst, can be used to evaluate HER performance using the following eqn (2):
| ΔGH* = ΔEH* + ΔEZPE − TΔSH | (2) |
In eqn (2), ΔEH* represents the adsorption energy of hydrogen on the catalyst, ΔEZPE denotes the zero-point energy correction, and ΔSH signifies the entropy difference between adsorbed hydrogen and hydrogen in the gas phase. In this DFT simulation, the contributions of the catalyst to ΔEZPE and ΔSH are assumed to be negligible and can therefore be neglected. Additionally, the adsorption of hydrogen on the catalyst takes the following general form:
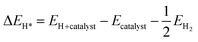 | (3) |
The total Gibbs free energy of each catalyst is calculated using eqn (2) and (3), incorporating corrections for zero-point energy and the entropy of hydrogen. This process involves constructing a two-layer slab of the catalyst material with an optimized vacuum of 20 Å. Structure optimization is performed both without hydrogen (Ecatalyst) and with hydrogen (EH+catalyst) before conducting the self-consistent field (SCF) calculation to determine the total energy for both scenarios. Additionally, the SCF calculation is applied to the hydrogen molecule to obtain its total energy (EH2).
Fig. S36(a–c)† illustrate the optimized structures of NiFe alloy/carbon, NiCo alloy/carbon, and NiCoFe alloy/carbon with adsorbed hydrogen, viewed from the top. This optimization process involved adjusting the atomic positions of the defined materials and calculating their corresponding energies. The optimization completes when the minimum energy is reached. Fig. S33–S35† show the total energy during the optimization process for all electrocatalysts. It is evident that the adsorbed hydrogen causes only minimal changes to the slab structure. By employing the optimized structure for both the catalyst with adsorbed hydrogen and the one without, the total Gibbs free energy was successfully determined. Specifically, NiFe-alloy/carbon demonstrated a total Gibbs free energy of −0.274 Rydberg (or −3.73 eV), which is comparable to that of NiCoFe-alloy/carbon and NiCo-alloy/carbon, both of which exhibited a total Gibbs free energy of −0.271 Rydberg (or −3.68 eV), as shown in Fig. S36(d).† Generally, a negative Gibbs free energy indicates that the catalyst can adsorb hydrogen; however, a value closer to zero suggests easier desorption, which is favorable for an effective electrocatalytic process. The strength of the M–H bonds is crucial for the hydrogen evolution reaction kinetics of electrocatalysts as discussed above. Consequently, the NiCoFe alloy supported on carbon is identified as the most effective electrocatalyst among all the electrodes tested. This finding further validates our systematic investigation, as illustrated in Fig. S18,† which demonstrates that while NiCo and FeNi exhibit comparable performance, the NiCoFe configuration shows a notably enhanced electrocatalytic activity. Additionally, the density of states for all electrocatalysts was successfully calculated, with the results illustrated in Fig. S36(e).† Notably, near the Fermi level energy (E − Ef = 0), NiCoFe-alloy/carbon and NiCo-alloy/carbon display a higher DOS compared to NiFe-alloy/carbon. In general, a greater density of states near the Fermi level suggests enhanced electrochemical adsorption capacity. These findings further reinforce the trends observed in Fig. S18,† where, although NiCo and FeNi exhibit comparable behavior, the NiCo alloy displays enhanced electrocatalytic performance at lower potentials due to a greater availability of electrons participating in the reaction. Additionally, a comparison of the electrocatalytic activity between NiCoFe-alloy/carbon and bare carbon reveals that NiCoFe-alloy/carbon exhibits a Gibbs free energy value much closer to zero, as shown in Fig. S36(f).† This shift indicates improved catalytic activity resulting from the successful integration of the NiCoFe alloy with carbon. The observed enhancement aligns well with our experimental findings illustrated in Fig. 5(a). To further investigate the origin of this improvement, the density of states of NiCoFe-alloy/carbon was analyzed in comparison with those of pristine NiCoFe alloy and bare carbon, as depicted in Fig. S36(g).† It is evident that carbon alone possesses a significantly lower DOS near the Fermi level, whereas NiCoFe-alloy/carbon shows a markedly higher DOS in this region. This suggests that the composite material has greater electron availability, leading to superior electrochemical adsorption characteristics and, consequently, enhanced electrocatalytic performance.
4. Discussion
The integration of dispersed trimetallic alloy nanoparticles within porous or layered structures of graphitized carbon presents a significant advancement in the development of efficient electrocatalysts for water electrolysis under harsh experimental conditions. This methodology enhances the overall activity of the electrocatalysts through various mechanisms such as charge redistribution and synergistic interactions between metals and metal-carbon layers. Specifically, trimetallic FeCoNi alloy integrated jute carbon exhibits great potential as a sustainable and cost-effective electrocatalyst for both the HER and OER, particularly in highly alkaline environments. The formation of trimetallic metal alloy nanoparticles is beneficial due to the distinct properties conferred by the synergistic effects of the combining metals. The alloying process adjusts the electronic structure, particularly the d-orbital configuration, which is critical for catalytic activity. For example, in the OER, the alloying of nickel, cobalt, and iron optimizes the d-orbital occupancy and the d-band center, enhancing the adsorption and activation of reaction intermediates such as hydroxyl (OH*), oxygen (O*), and peroxo (OOH*). This alteration leads to more efficient charge transfer during the OER, resulting in improved catalytic performance.55,56 However, in the HER, the d-orbital interactions within the trimetallic alloy facilitate the adsorption and activation of hydrogen-containing species (H* and H2O). The optimized configuration enhances the binding strength of these intermediates, promoting the HER.57,58 Additionally, the introduction of structural defects and coordination of unsaturated sites due to the alloying process increases the density of active sites available for both the HER and OER, improving electrocatalytic activity.The formation of truncated octahedral metal alloy nanoparticles can be linked to specific synthesis conditions, which involve a delicate balance of thermodynamic and kinetic factors. The addition of ammonia during sonication promotes instantaneous nucleation, leading to the formation of nanoparticles that adopt a truncated octahedral shape to minimize surface energy. This morphology which is characteristic of FCC alloys exposes a combination of low-index {111} and {100} facets known for their high surface energies59,60. Such high-energy surfaces favour the adsorption and activation of reactants, thereby enhancing catalytic activity. The porous graphitized carbon support which offers a stable platform for nanoparticle growth also promotes efficient mass transfer and homogeneous distribution within the carbon matrix. This intimate contact between the metal alloy nanoparticles and the carbon support facilitates efficient charge transfer and stabilizes active sites, contributing to the overall enhanced electrocatalytic performance. When compared to traditional single-metal systems, trimetallic alloys provide a broader range of active sites, improved electrical interactions, and enhanced catalytic kinetics. The jute carbon modified with the FeCoNi alloy demonstrates exceptional catalytic efficiency for both oxygen and hydrogen evolution, surpassing that of conventional metal and carbon-based catalysts. The alloy's ability to exploit the benefits of each constituent metal leads to superior adsorption–desorption equilibrium for both OER and HER intermediates, resulting in lower overpotentials.
The experimental results indicate that the JCM exhibits negligible degradation under harsh alkaline conditions, which is often a limitation of conventional electrocatalysts that are prone to activity loss due to dissolution. The presence of the metal alloy effectively prevents leaching and agglomeration, thereby maintaining catalytic performance over extended operational periods. The structural characteristics of the trimetallic alloy also play a crucial role in determining the electrocatalytic performance. DFT calculations reveal that the NiCoFe alloy exhibits a Gibbs free energy (ΔG) of −3.67 eV for hydrogen adsorption, which is closer to thermodynamic neutrality compared to bare carbon (ΔG of −9.93 eV). This indicates an optimal hydrogen adsorption/desorption equilibrium facilitated by the alloy formation which is corroborated by experimental observations of a low overpotential of 123 mV and small Tafel slope of 62 mV dec−1. The enhanced stability of the trimetallic alloy under harsh conditions is attributed to the synergistic effects among Ni, Co, and Fe, which prevents the dissolution of active sites, thus sustaining catalytic activity over time. This robust structure is particularly advantageous for practical applications in water splitting devices.
The sustainability and cost-effectiveness of using jute-derived carbon materials for development of electrocatalysts have a significant advantage. The conversion of jute, an abundant natural fibre, into carbon materials represents a greener method of utilizing agricultural waste, thereby reducing environmental impact and manufacturing costs compared to precious metal catalysts such as platinum or iridium. This approach aligns with sustainable economic practices by transforming renewable resources into high-value materials for energy applications. Hence, the incorporation of dispersed trimetallic alloy nanoparticles within graphitized carbon matrices represents a compelling strategy for developing highly active and durable electrocatalysts for alkaline water splitting. The synergistic effects of the alloying process, combined with the optimized structural characteristics and sustainability of the materials, offer significant advancements in the field of electrocatalysis. As research progresses, this hybrid nanostructure makes provision for practical, scalable solutions in renewable energy technologies.
5. Conclusion
In this study, we present an innovative approach to enhance the electrocatalytic performance of jute-derived carbon integrated with a tri-metallic alloy (Fe, Co, and Ni). The metal alloy integrated carbon catalyst i.e. JCM exhibits remarkable improvements in electrochemical performance for both hydrogen and oxygen evolution reactions. The incorporation of the transition metal alloy into the carbon matrix not only improves the electrical conductivity but also increases the number of active sites available for catalysis, significantly enhancing the efficiency of the reactions. The structural features of the JCM, including a hierarchical porous network and the dispersion of metal nanoparticles, play a crucial role in optimizing both mass transport and charge transfer. These characteristics are essential for achieving low overpotentials and high catalytic stability. The JCM demonstrated a low overpotential of 123 mV for the HER and showed exceptional stability, remaining efficient for over 100 hours of operation while achieving an overpotential of only 230 mV for the OER with the smallest Tafel slope and extraordinary stability, suggesting that the JCM has the potential to outperform traditional precious metal catalysts while being cost-effective and sustainable, making it highly attractive for large-scale industrial applications. DFT simulations supporting the experimental results revealed that the NiCoFe alloy has an optimal Gibbs free energy ensuring hydrogen adsorption and desorption and thereby improving the HER activity. Furthermore, this study affirms the potential of biomass-derived materials, such as jute, to bridge the gap between performance and sustainability. The utilization of jute biomass as a precursor for designing low-cost, high-performance electrocatalysts not only offers an environmentally friendly solution but also supports the principles of the circular economy. As such, the JCM represents a promising candidate for the development of efficient and cost-effective electrocatalysts for renewable energy technologies and promotes more accessible, sustainable energy solutions.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this article.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors gratefully acknowledge the support received through the MbSC2030 Direct Funded Research Grant (MbSC24203), provided by the Deanship of Research at King Fahd University of Petroleum & Minerals (KFUPM). Research support from the Interdisciplinary Research Centre for Hydrogen Technologies and Carbon Management (IRC-HTCM) at KFUPM is also sincerely appreciated.References
- S. Sun, Z. Liu, Z. J. Xu and T. Wu, Appl. Catal., B, 2024, 358, 124404 CrossRef CAS.
- I. Letchumanan, R. Mohamad Yunus, M. S. Mastar @ Masdar, M. Beygisangchin, S. K. Kamarudin and N. A. Karim, Int. J. Hydrogen Energy, 2024, 59, 1518–1539 CrossRef CAS.
- J. Masa, C. Andronescu and W. Schuhmann, Angew. Chem., Int. Ed., 2020, 59, 15298–15312 CrossRef CAS.
- H. Jia, L. Meng, Y. Lu, T. Liang, Y. Yuan, Y. Hu, Z. Dong, Y. Zhou, P. Guan, L. Zhou, C. Liu, M. Li, T. Wan, B.-J. Ni, Z. Han and D. Chu, J. Mater. Chem. A, 2024, 12, 28595–28617 RSC.
- P. C. Sherrell, M. Iesalnieks, Y. Ehrnst, A. R. Rezk and A. Šutka, Adv. Energy Sustainability Res., 2024, 5, 2400008 CrossRef.
- H. Y. Kim and S. H. Joo, J. Mater. Chem. A, 2020, 8, 8195–8217 RSC.
- S. E. Hosseini and M. A. Wahid, Renew. Sustain. Energy Rev., 2016, 57, 850–866 CrossRef CAS.
- T. Naito, T. Shinagawa, T. Nishimoto and K. Takanabe, Inorg. Chem. Front., 2021, 8, 2900–2917 RSC.
- H. Wang, Z. Yan, F. Cheng and J. Chen, Adv. Sci., 2024, 11, 1–33 Search PubMed.
- Y. Chen, Q. Li, Y. Lin, J. Liu, J. Pan, J. Hu and X. Xu, Nat. Commun., 2024, 15, 7278 CrossRef CAS.
- M. Xu, J. Wang, S.-G. Sun and Z. Chen, JACS Au, 2024, 4, 3964–3975 CrossRef CAS PubMed.
- K. Tran and Z. W. Ulissi, Nat. Catal., 2018, 1, 696–703 CrossRef CAS.
- R. Yao, K. Sun, K. Zhang, Y. Wu, Y. Du, Q. Zhao, G. Liu, C. Chen, Y. Sun and J. Li, Nat. Commun., 2024, 15, 1–12 Search PubMed.
- M. A. Qadeer, X. Zhang, M. A. Farid, M. Tanveer, Y. Yan, S. Du, Z. F. Huang, M. Tahir and J. J. Zou, J. Power Sources, 2024, 613, 234856 CrossRef CAS.
- S. Dilpazir, H. He, Z. Li, M. Wang, P. Lu, R. Liu, Z. Xie, D. Gao and G. Zhang, ACS Appl. Energy Mater., 2018, 1, 3283–3291 CrossRef CAS.
- S. Perumal, I. Pokhrel, U. Muhammad, X. Shao, Y. Han, M. Kim and H. Lee, ACS Mater. Lett., 2024, 6, 3625–3666 CrossRef CAS.
- H. Boumeriame, E. S. Da Silva, A. S. Cherevan, T. Chafik, J. L. Faria and D. Eder, J. Energy Chem., 2021, 64, 406–431 CrossRef.
- K. Zeng and D. Zhang, Prog. Energy Combust. Sci., 2010, 36, 307–326 CrossRef CAS.
- K. K. Joshi, P. M. Pataniya, G. R. Bhadu and C. K. Sumesh, Int. J. Hydrogen Energy, 2024, 49, 829–842 CrossRef CAS.
- Z. Li, L. Sun, Y. Zhang, Y. Han, W. Zhuang, L. Tian and W. Tan, Coord. Chem. Rev., 2024, 510, 215837 CrossRef CAS.
- E. Jiang, N. Song, S. Hong, M. Xiao, D. Zhu, Z. Yan, J. Sun, G. Chen, C. Li and H. Dong, Electrochim. Acta, 2022, 407, 139895 CrossRef CAS.
- Z. Zhang, P. Li, Q. Feng, B. Wei, C. Deng, J. Fan, H. Li and H. Wang, ACS Appl. Mater. Interfaces, 2018, 10, 32171–32179 CrossRef CAS.
- Q. Wang, R. Guo, Z. Wang, D. Shen, R. Yu, K. H. Luo, C. Wu and S. Gu, Fuel, 2021, 293, 120440 CrossRef CAS.
- S. Surendran, S. Shanmugapriya, S. Shanmugam, L. Vasylechko and R. Kalai Selvan, ACS Appl. Energy Mater., 2018, 1, 78–92 CrossRef CAS.
- S. Dilpazir, R. Liu, M. Yuan, M. Imran, Z. Liu, Y. Xie, H. Zhao and G. Zhang, J. Mater. Chem. A, 2020, 8, 10865–10874 RSC.
- X. Ma, C. Guo, J. Xiang, Y. Qi, J. Yu, K. Li, Z. Tao, J. Wu and Y. Lv, Int. J. Hydrogen Energy, 2024, 60, 845–866 CrossRef CAS.
- X. Li, C. Yang, S. Wang, X. Mao and K. Yu, Diamond Relat. Mater., 2022, 129, 109382 CrossRef CAS.
- N. Prabu, R. S. A. Saravanan, T. Kesavan, G. Maduraiveeran and M. Sasidharan, Carbon, 2019, 152, 188–197 CrossRef CAS.
- S. Sekar, D. H. Sim and S. Lee, Nanomaterials, 2022, 12, 531 CrossRef CAS PubMed.
- L. X. Zuo, W. J. Wang, R. Bin Song, J. J. Lv, L. P. Jiang and J. J. Zhu, ACS Sustain. Chem. Eng., 2017, 5, 10275–10282 CrossRef CAS.
- X. Liu, P. Cai, G. Wang and Z. Wen, Int. J. Hydrogen Energy, 2020, 45, 32940–32948 CrossRef CAS.
- W. Li, F. Guo, X. Wei, Y. Du and Y. Chen, RSC Adv., 2020, 10, 36644–36653 RSC.
- L. Xiong, B. Liu, H. Wang and X. Lin, Ind. Eng. Chem. Res., 2023, 62, 7040–7047 CrossRef CAS.
- Y. Liu, G. Mou, Y. Wang, F. He, N. Dong, Y. Lin, M. Zhong and B. Su, ACS Appl. Nano Mater., 2022, 5, 2953–2961 CrossRef CAS.
- X. Lv, Z. Xiao, H. Wang, X. Wang, L. Shan, F. Wang, C. Wei, X. Tang and Y. Chen, J. Energy Chem., 2021, 54, 626–638 CrossRef CAS.
- H. Gao, Y. Wang, S. Zhou, S. Song, X. Tian, W. Li, Y. Yuan and J. Zang, Chem. Eng. J., 2021, 426, 130854 CrossRef CAS.
- L. Yang, X. Zeng, D. Wang and D. Cao, Energy Storage Mater., 2018, 12, 277–283 CrossRef.
- J. Chang, W. Wang, L. Wang, D. Wu, F. Xu, K. Jiang, G. Wang and Z. Gao, Appl. Catal., A, 2023, 650, 118984 CrossRef CAS.
- Y. Xiao, Q. Wang, X. Feng, J. Wu, P. Liao, Y. Tong and J. Zhang, J. Mater. Chem. A, 2021, 9, 17451–17458 RSC.
- N. Li, D. K. Bediako, R. G. Hadt, D. Hayes, T. J. Kempa, F. von Cube, D. C. Bell, L. X. Chen and D. G. Nocera, Proc. Natl. Acad. Sci. U. S. A., 2017, 114, 1486–1491 CrossRef CAS PubMed.
- D. Luo, B. Yang, Z. Mei, Q. Kang, G. Chen, X. Liu and N. Zhang, ACS Appl. Mater. Interfaces, 2022, 14, 52857–52867 CrossRef CAS PubMed.
- Z. Sun, A. Curto, J. Rodríguez-Fernández, Z. Wang, A. Parikh, J. Fester, M. Dong, A. Vojvodic and J. V. Lauritsen, ACS Nano, 2021, 15, 18226–18236 CrossRef CAS PubMed.
- S. S. Shah, H. Yang, M. Ashraf, M. A. A. Qasem, A. S. Hakeem and M. A. Aziz, Chem.–Asian J., 2022, 17, e202200567 CrossRef CAS.
- P. Manasa, Z. J. Lei and F. Ran, J. Energy Storage, 2020, 30, 101494 CrossRef.
- B. Wang, Q. Liu, A. Yuan, Q. Shi, L. Jiang, W. Yang, T. Yang and X. Hou, J. Mater. Sci. Technol., 2024, 182, 1–11 CrossRef CAS.
- M. B. Poudel, S. Vijayapradeep, K. Sekar, J. S. Kim and D. J. Yoo, J. Mater. Chem. A, 2024, 12, 10185–10195 RSC.
- C. Wang, J. Zhang, Z. Zhang, G. Ren and D. Cai, RSC Adv., 2020, 10, 38906–38911 RSC.
- M. Goudarzi and M. Salavati-Niasari, J. Alloys Compd., 2019, 784, 676–685 CrossRef CAS.
- Y. Jiao, C. Wan, W. Bao, H. Gao, D. Liang and J. Li, Carbohydr. Polym., 2018, 189, 371–378 CrossRef CAS PubMed.
- B. Li, Y. Nan, P. Zhang and X. Song, RSC Adv., 2016, 06, 19797–19806 RSC.
- G. Wang, L. Yang, Y. Chen, W. Wang and Z. Gao, Diam. Relat. Mater., 2023, 136, 109957 CrossRef CAS.
- W. T. Tang, X. Liu, Y. Li, Y. Pu, Y. Lu, Z. Song, Q. Wang, R. Yu and J. Shui, Nano Res., 2020, 13, 447–454 CrossRef CAS.
- Z.-J. Zhang, H.-M. Xu, C.-J. Huang, T.-Y. Shuai, Q.-N. Zhan, Z. Song and G.-R. Li, Nanoscale, 2024, 16, 19970–19997 RSC.
- Y. Du, F. Xie, M. Lu, R. Lv, W. Liu, Y. Yan, S. Yan and Z. Zou, Nat. Commun., 2024, 15, 1780 CrossRef CAS.
- L. Cao, B. W. Zhang and S. Zhao, Chem.–Eur. J., 2023, 29, e202202000 CrossRef CAS PubMed.
- J. R. Swierk, S. Klaus, L. Trotochaud, A. T. Bell and T. D. Tilley, J. Phys. Chem. C, 2015, 119, 19022–19029 CrossRef CAS.
- M. Yu, E. Budiyanto and H. Tüysüz, Angew Chem. Int. Ed. Engl., 2022, 61, e202103824 CrossRef CAS PubMed.
- Y. Hu and C. Wang, J. Mater. Chem. A, 2024, 12, 16769–16779 RSC.
- H.-F. Zhao, J.-Q. Yao, Y.-S. Wang, N. Gao, T. Zhang, L. li, Y. Liu, Z.-J. Chen, J. Peng, X.-W. Liu and H.-B. Yu, Adv. Sci., 2024, 11, 2404095 CrossRef CAS.
- N. Tian, Z.-Y. Zhou and S.-G. Sun, J. Phys. Chem. C, 2008, 112, 19801–19817 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5ta02765a |
| This journal is © The Royal Society of Chemistry 2025 |