Revolutionizing water splitting performance by probing the influence of electron transfer on the NiCr-LDH/VS2/NF heterostructure†
Aditi
De
 ab,
Pandiarajan
Devi
ab,
Pandiarajan
Devi
 ac,
Palanichamy
Murugan
ac,
Palanichamy
Murugan
 ac and
Subrata
Kundu
ac and
Subrata
Kundu
 *ab
*ab
aAcademy of Scientific and Innovative Research (AcSIR), Ghaziabad-201002, India
bElectrochemical Process Engineering (EPE) Division, CSIR-Central Electrochemical Research Institute (CECRI), Karaikudi, Tamil Nadu 630006, India. E-mail: skundu@cecri.res.in; kundu.subrata@gmail.com; Fax: +91 4565-241487; Tel: +91 4565-241487
cElectrochemical Power Sources (ECPS) Division, CSIR-Central Electrochemical Research Institute (CECRI), Karaikudi-630003, Tamil Nadu, India
First published on 2nd April 2025
Abstract
Electrocatalytic water splitting for the production of green hydrogen addresses the current energy crisis and potential energy storage. Herein, we have fabricated a low-cost, highly efficient transition metal-based heterostructure of NiCr-LDH over VS2 using three-dimensional (3D) nickel foam as a substrate. This self-supported NiCr-LDH/VS2/NF heterostructure catalyst works as an excellent bifunctional electrode to catalyze the oxygen evolution reaction (OER) and the hydrogen evolution reaction (HER) by exhibiting very low overpotential values of 209 mV and 116 mV, respectively, to attain 50 mA cm−2 current density in 1 M KOH solution. In addition, NiCr-LDH/VS2/NF shows a cell voltage of 1.545 V to attain 10 mA cm−2 current density and a 40-hour long-term static stability. The n-type semiconducting NiCr-LDH and p-type conducting VS2 enhance the electrocatalytic performance by their synergistic effect, changing the surface-modified electronic structure, and generation of improved thin-nanosheet-like mesoporous morphology with superhydrophilic surfaces. Density functional theory (DFT) calculations confirm an interesting charge repopulation observation in both the layers of NiCr-LDH and VS2 that increases the overall electrocatalytic reaction performance (as confirmed by charge density and Bader charge analysis). This material holds impressive application potential that can guide the design and screening of efficient earth-abundant bifunctional electrocatalysts.
1. Introduction
Fossil fuels being the basis of modern global economic development are now facing scrutiny owing to their significant and detrimental environmental effects and limited resources. Growth of the world population and advancement of standard of living, especially in developing countries has raised alarm about the rapid depletion of reserved crude oil. There are emerging reports that the depletion can happen within a few decades. Alternative renewable energy sources like solar, hydro, wind, biomass, etc. have garnered significant attention over the decades.1–3 Among all methods, electrochemical water splitting involving the oxygen evolution reaction (OER) at the anode and the hydrogen evolution reaction (HER) at the cathode is a hotspot research area as it manufactures green hydrogen with zero carbon emission and has a high energy density of ∼120 MJ kg−1, almost three times the energy density value of diesel/gasoline.4 However, its activity is disrupted by the OER, a complex four-electron transfer process. Generally, IrO2 and RuO2 are considered to be state-of-the-art catalysts for the OER, and Pt-based materials are well-known catalysts for the HER. Nevertheless, their use is limited due to high cost and scarcity in nature, hampering large-scale industrial applications. Therefore, as an alternative, non-noble transition metal-based oxide, hydroxide, nitride, and sulfide electrocatalysts are being highly explored in recent times as they are not only cost-effective but also with proper modification can sometimes act as bifunctional electrocatalysts with outstanding OER and HER performance. Among these entities, layered double hydroxides (LDHs), especially NiFe- and CoFe-LDHs are widely explored due to their synergistic effect, which make them favourable for electrocatalytic reactions.5–7 However, their catalytic activity was boosted, and noticeable catalytic performance was observed when these bare LDH components were further modified with different strategies like doping with noble metal ions, creating vacancies, heterostructure formation with conducting materials, fine-tuning of the intercalated position, etc.8–10On the other hand, transition metal-based sulfides (TMSs) like MoS2 and WS2 also exhibit potential electrochemical activity due to their highly conducting nature. Nevertheless, individual TMSs lack active sites due to insufficient catalytic efficiency for intermediate steps, electronic structure limitation, etc. Hence, fabricating cost-effective bifunctional electrocatalysts by constructing a multicomponent nanomaterial is vital for augmenting the electrochemical performance and practical large-scale application in the electrocatalytic water splitting field. Zhong et al. synthesized N-doped Ni3S2/VS2 nanosheets that exhibit high electrocatalytic OER performance (227 mV at 10 mA cm−2) and HER performance with a low HER overpotential (151 mV at 10 mA cm−2) owing to increased conductivity and catalytic active site after N-doping.11 Recently, Wang et al. synthesized an optimal performance NiFe-LDH/NiS2/VS2 by an electrodeposition process, showing excellent catalytic activity with a small overpotential of 380 mV at 10 mA cm−2 for TWS. This performance is facilitated by interface tuning and heterojunction creation, which help in synergistic interaction resulting in more exposed active sites and improvement of electron transfer across the electrode–electrolyte interface. However, these research studies lack a comprehensive mechanistic understanding with a proper explanation of the characterization processes observed. For instance, in XPS, the negative peak shift of Ni 2p and Fe 2p is not justified evidently by Wang’s group.12 Therefore, accurate characterization and investigation, ideally through density functional theory (DFT) or other in operando and in situ methods, are essential for advancing the research of large-scale electrocatalytic application of these materials. Herein, we have created a NiCr-LDH/VS2/NF heterostructure via a two-step hydrothermal method with a less explored NiCr-LDH material and VS2 as a source material for the OER, HER, and total water splitting (TWS) study. In 1 M KOH alkaline solution, NiCr-LDH/VS2/NF demands very low overpotentials of 209 mV and 116 mV for the OER and HER, respectively, to attain 50 mA cm−2 current density with >30-hour long durability. Additionally, NiCr-LDH/VS2/NF needs a lower cell potential of 1.545 V to achieve 10 mA cm−2 current density with 40-hour stability. Initially, Ni and Cr were chosen among the TMs by considering the prominent electrocatalytic activity of Ni2+, as it can easily oxidize to Ni3+, and Cr being well known for its stability in alkaline medium helps in low catalytic degradation that balances the structure with fine-tuning properties. VS2, on the contrary, is highly conducting in nature.13,14 So, the synergistic effect of n-type semiconductive NiCr-LDH/NF and p-type highly conducting bare VS2/NF (confirmed by Mott–Schottky analysis) can easily regulate the interface and increase the semiconducting nature of NiCr-LDH/NF, resulting in high electrocatalytic performance with an elevated active surface area for the overall NiCr-LDH/VS2/NF heterostructure. Interestingly, it is observed from the charge density difference (CDD) calculation that the two layers are perfectly connected through the electrostatic interaction with a stable energy (−0.91 eV), and the charge transfer of the interlayers does not occur much, rather than the charge repopulation that occurs within the layer of NiCr-LDH and bare VS2. The strong hybridization between the 3d states of Ni and Cr and the 2p states of oxygen improves the conductivity and electron mobility across the layer, which elevates the overall electrocatalytic property. Notably, for the NiCr-LDH, a fascinating indirect electron transfer occurs via the pathway Ni → O → Cr (when charge redistribution occurs near the Ni site) and vice versa due to charge repopulation, and it is further confirmed by partial density of states (PDOS) and partial charge analysis (PARCHG). Our work demonstrates an effective strategy to produce an excellent bifunctional electrocatalyst for total water splitting with a unique observation of the charge re-distribution occuring in the LDH layer as well as the VS2 layer.
2. Results and discussion
The optimized NiCr-LDH/VS2/NF electrocatalyst was synthesized by a two-step hydrothermal process. Initially, pristine NiCr LDH and VS2 were grown over Ni foam via a hydrothermal process. For heterostructure formation, the same process was followed, but instead of Ni foam, bare VS2/NF was used in the formation of NiCr-LDH/NF to obtain the NiCr-LDH/VS2/NF heterostructure, and NiCr-LDH/NF was used in the formation of VS2 to obtain the VS2/NiCr-LDH/NF heterostructure. The influence of the electrocatalytic efficiency of highly conductive VS2 over the NiCr-LDH/NF was investigated. Evidently, among these four prepared catalysts, NiCr-LDH/VS2/NF achieved remarkable catalytic outcomes. A visual representation of the full synthesis process of all fabricated catalysts is diagrammed in Scheme 1.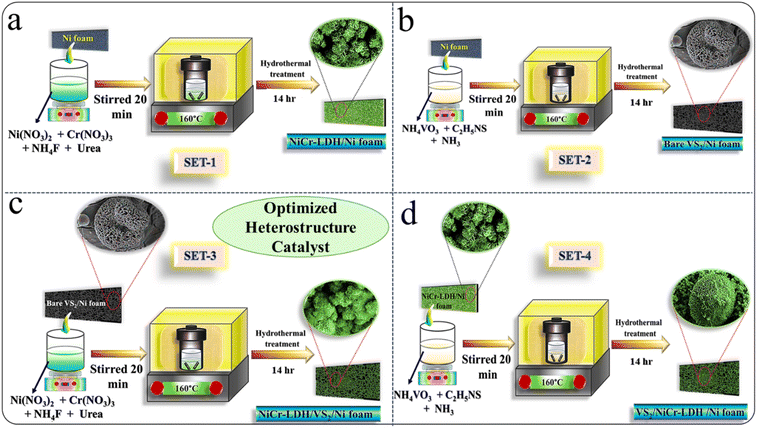 | ||
| Scheme 1 Systematic synthetic scheme for the synthesis of (a) NiCr-LDH/NF; (b) bare VS2/NF; (c) NiCr-LDH/VS2/NF; (d) VS2/NiCr-LDH/NF. | ||
A complete overview of the detailed fabrication technique of the synthesized catalysts and material used is presented in the ESI.† Following substantial synthesis of all four catalysts, the co-precipitated catalysts were initially validated through powder X-ray diffraction (PXRD) at a scan rate of 5° per minute to find out the crystalline phase and arrangement. Primarily, the working electrodes grown over Ni foam (NF) were directly chosen to investigate XRD outcomes. However, significant strong Ni peaks of NF at 2θ values of 44°, 52°, and 77° mask the other LDH peaks (Fig. S1a†). Consequently, respective powdered samples obtained by centrifuging solutions after hydrothermal synthesis were utilized for more accurate XRD analysis.In Fig. 1a, PXRD patterns of NiCr-LDH/NF, bare VS2/NF, NiCr-LDH/VS2/NF, and VS2/NiCr-LDH/NF are displayed.
The peaks of NiCr-LDH/NF are compared with the hexagonal β phase of the Ni(OH)2 plane (JCPDS: 01-073-1520), and the formation of bare VS2/NF is confirmed by the monoclinic VS2 structure (JCPDS: 01-080-0901). For NiCr-LDH/NF, the peak observed at 19.2° perfectly matches with the 001 planes of JCPDS: 01-073-1520.
Similarly, for bare VS2/NF, the peak observed at 11.2° and 22.5° perfectly matches the 003 and 006 planes of JCPDS: 01-080-0901. Interestingly, after the formation of NiCr-LDH/VS2/NF and VS2/NiCr-LDH/NF heterostructures, the above-mentioned peaks slightly shifted to a higher binding energy (∼0.2–0.3°), as portrayed in Fig. S1b,† highlighting the effective synergistic interaction between LDH and the sulfide layer of the VS2 structure, influenced by compression in the overall lattice structure. All other peaks detected in the PXRD analysis of all four synthesized catalysts strongly aligned with the JCPDS files mentioned above, confirming the formation of heterostructures. It is worth mentioning that, after the formation of the polycrystalline NiCr-LDH/VS2/NF heterostructure, the intense peaks mostly match with those of the NiCr-LDH/NF structure, confirming that the top layer of heterostructure is NiCr-LDH rather than bare VS2. Similarly, for VS2/NiCr-LDH/NF, the top layer is bare VS2 and bottom layer is NiCr-LDH. Next, to scrutinize the functional group present in the as-prepared electrocatalysts, Fourier Transform Infrared (FT-IR) spectroscopy was performed (Fig. 1b). IR absorption band occurs at 3428, 1645, 1386, and 1485 cm−1, representing stretching vibrations of the hydroxyl group (–OH) and asymmetric and symmetric stretching vibration modes of CO32− located in the intercalated position of LDH, and bending vibration of intercalated H2O molecules, respectively. Peaks that arise at 1100–500 cm−1 are generally due to M–O, M–O–M, and O–M–O bond vibration (M = Ni, Cr, V). For bare VS2/NF and the VS2/NiCr-LDH/NF heterostructure, one characteristic peak of V–S bond formation occurs at 990 cm−1.15
Following structural conformation, Raman spectroscopy was performed to identify the chemical composition and bond formation existing in the system. For NiCr-LDH/NF, characteristic peaks occur at 128, 311, 460, and 540 cm−1, among which the first low-frequency peak might be generated due to transitional vibration of the Ni–O bond (Fig. 1c). The prominent peaks at 311 and 460 cm−1 are attributed to the Eg and A1g lattice vibrational modes of Ni(OH)2, respectively. The presence of the bending mode of Cr–O–H of Cr(OH)3 at 530 cm−1 and the second-order acoustic mode of Ni(OH)2 at 540 cm−1 give the characteristic broad peak at 540 cm−1, confirming the presence of both Cr–O and Ni–O bonds in the NiCr LDH.16 For bare VS2/NF, characteristic peaks occur at 150, 220, 260, 348, 402, and 512 cm−1, among which the vibrational frequency at 260 cm−1 is attributed to the in-plane vibration E1g mode and that at 402 cm−1 to the out-of-plane vibration A1g mode in the S–V–S interlayer of the VS2 material.17 The peak arising at ∼150 cm−1 is due to the two-phonon process mode.18 After the formation of NiCr-LDH/VS2/NF and VS2/NiCr-LDH/NF heterostructures, Raman peak intensities are drastically reduced, and the broadening of Raman peaks occurs. This might be due to the reason that after the formation of the heterostructure, the elevated interface area offers more opportunities for phonons to localize at the interface and interact with the interface and scatter. The interference effects of the low energy phonons result in a complex spectrum, which reflects the unique vibrational properties of n-type NiCr-LDH/NF and p-type bare VS2/NF, and the overall outcome reveals the reduction and broadening of Raman peak signals for the heterostructure materials.19 These localized phonons may assist in improving the electrocatalytic performance by helping in the overall charge transport and providing thermal stability to the heterostructure during electrochemical reactions under harsh cathodic and anodic conditions. Additionally, the morphology of the optimized NiCr-LDH/VS2/NF heterostructure catalyst was explored using Field Emission-Scanning Electron Microscopy (FE-SEM); the low- and high-magnification images in Fig. 1d and e reveals the formation of a small-sized NiCr LDH structure over the bigger size VS2 layer. For a better understanding of the visual representation, we have analyzed the FESEM image of all four catalysts including the bare VS2/NF and VS2/NiCr-LDH/NF heterostructure in Fig. S2.† Fig. S2a and b† are low- and high-magnification FESEM image of NiCr-LDH/NF where we can observe the uniform distribution of flower-like structures all over Ni foam.20 Fig. S2c and d† depicts comparatively bigger sizes and different types of floral structures of conductive bare VS2/NF electrocatalysts. Notably, during the formation of the VS2/NiCr-LDH/NF heterostructure, bigger size floral VS2 was placed over the uniformly distributed smaller and denser NiCr LDH layer, which is clearly observed in the low- magnification image in Fig. S2e.† However, in Fig. S2f,† during the formation of the heterostructure, a negligible number of NiCr LDH floral parts was placed above the VS2 layer portrayed in high-magnification FESEM image of VS2/NiCr-LDH/NF, which helps in visibly differentiating both nanostructures. From the above observation, it is conclusive that while comparing NiCr-LDH/NF with bare VS2/NF, the nanosheets become denser, and the sheets become closer for NiCr-LDH/NF, which creates better channels providing more active sites that accelerate charge transfer resulting in better catalytic activity for NiCr-LDH/NF. The DFT study, charge density and Bader charge analyses proved that after heterostructure formation no charge transfer occurs between layers, while charges in the layer are redistributed, and therefore layers were bonded through the electrostatic interaction. Herein, highly conducting bigger size floral VS2 acts as an electron donor and helps in improving the charge repopulation in the NiCr-LDH layer that directly influences the electrocatalytic activity by intra-layer electronic transfer during the reaction, ensuring that the NiCr-LDH/VS2/NF catalyst stands out as the most potent catalyst. On the other hand, for the VS2/NiCr-LDH/NF catalyst, the most exposed site during the electrochemical reaction is VS2, as bulkier VS2 masks most of the NiCr LDH layer, as witnessed in Fig. S2f,† resulting in low activity compared to NiCr-LDH/VS2/NF, but still it shows enhanced activity compared to the bare materials NiCr-LDH/NF and bare VS2/NF considered for the synergistic interaction created upon heterostructure formation. The quantitative elemental distribution of all four catalysts acquired from FE-SEM analysis is displayed and tabulated in Fig. S3,† where the high nickel content for all the catalysts is attributed to the presence of Ni foam as the substrate used.
Following this, detailed microstructural analysis was performed by High-Resolution Transmission Electron Microscopy (HR-TEM) analysis, which allowed us to obtain more in-depth information about the morphology, lattice spacing, structure, crystallinity, and atomic distribution over the electrode surface. Fig. 1f and g displays low- and high-magnification HRTEM images of NiCr-LDH/VS2/NF where distinct sheets of flower-like NiCr LDH and the VS2 structure are visible that were acquired after sonication for a few minutes. Fig. 1h shows the SAED pattern, indicating the polycrystalline nature of NiCr-LDH/VS2/NF that perfectly aligns with XRD outcomes. The SAED pattern shows (404) and (113) planes of VS2 and (110) planes of LDH that confirm the presence of both compounds in the fabricated heterostructure. Furthermore, from lattice fringes analysis in Fig. 1i, a clear heterostructure interface marked by a red dotted line also confirms the heterostructure formation visually. To enhance the visibility and more accurate illustration of the crystallographic features, we have performed an inverse fast Fourier transform (IFFT) on the TEM image of the selected zones of the synthesized heterostructure and measured the d-spacing, allowing for a clearer visualization of the fringe lines. The d-spacing value of 0.232 nm (011) confirms the presence of NiCr LDH, while the interplanar distance of ∼0.39 nm (006) in the darker side separated by a red dotted line interface confirms the presence of the VS2 plane. Next, in Fig. 1j, the High Angle Annular Dark Field (HAADF) image of NiCr-LDH/VS2/NF is chosen for elemental colour mapping, and the corresponding results of the elemental mix, V, S, Ni, Cr, C, and O, respectively, in Fig. 1k–q portray the uniform distribution of elements over the catalytic surface. In addition, HRTEM images of NiCr-LDH/NF, bare VS2/NF, and VS2/NiCr-LDH/NF, including low- and high-resolution TEM images, SAED pattern, latice fringes, and HAADF colour mapping, are provided in Fig. S4(a–i), (j–r) and S5,† respectively.
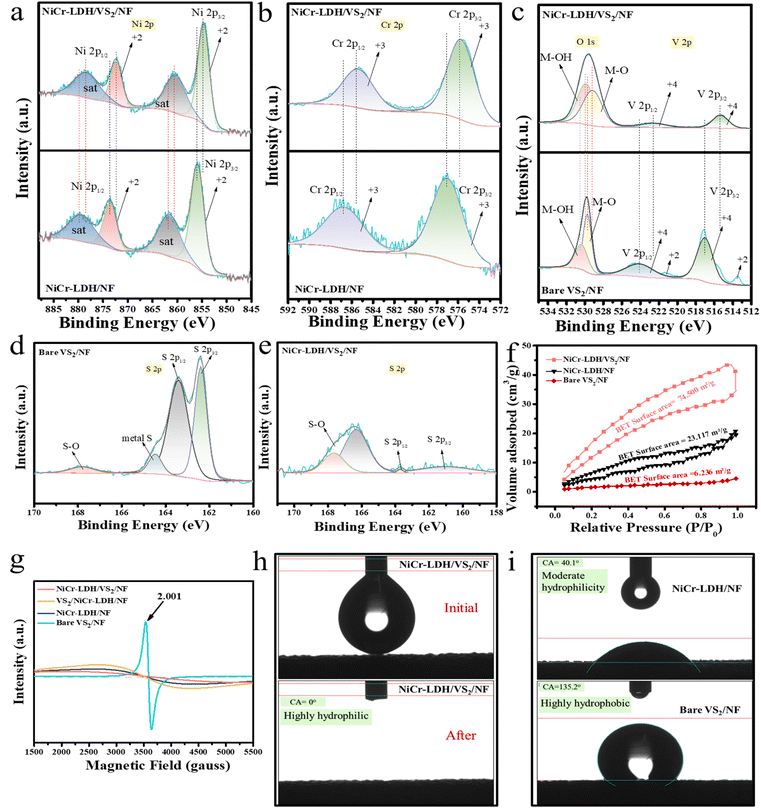
To explore oxidation states and the chemical nature of the prepared catalysts, an X-ray photoelectron spectroscopy (XPS) study was performed for pristine NiCr-LDH/NF, bare VS2/NF, and optimized high activity NiCr-LDH/VS2/NF electrocatalysts. The XPS survey spectrum for NiCr-LDH/NF in Fig. S6a† confirms the presence of all expected elements Ni, Cr, C, and O present in the system. Similarly, for bare VS2/NF (Fig. S6b†), the presence of V, S, C, and O is observed; for NiCr-LDH/VS2/NF (Fig. S6c†), all the expected elements Ni, Cr, V, O, C, and S are observed across the system. Fig. 2a shows the deconvoluted Ni 2p high-resolution XPS spectra for NiCr-LDH/NF and NiCr-LDH/VS2/NF, where two spin–orbit coupling peaks of NiCr-LDH/NF, Ni 2p3/2 and Ni 2p1/2, are observed at binding energies of 856.21 eV and 873.60 eV, respectively, along with the corresponding satellite peaks at 861.86 eV and 879.81 eV, verifying the +2 oxidation state of Ni. Interestingly, after heterostructure formation, all these characteristic peaks shift to lower binding energies (ΔBE = ∼1.22–1.28 eV), with binding energies of 854.99 and 872.33 for Ni 2p3/2 and Ni 2p1/2 and corresponding satellite peaks at 860.60, and 878.59, respectively. This >1 eV binding energy shift is in good agreement with the theoretical results, where CDD outcomes also suggest the charge repopulation within both NiCr-LDH and VS2 layers of the NiCr-LDH/VS2/NF heterostructure.
Due to this charge repopulation within the layers after heterostructure formation, not only Ni peaks but also deconvoluted XPS spectra of Cr 2p, V2p, and O 1s show significantly negative shifting. In Fig. 2b, deconvoluted Cr 2p XPS analysis of NiCr-LDH/NF and NiCr-LDH/VS2/NF suggests the +3 oxidation state of Cr having two spin–orbit coupling peaks of NiCr-LDH/NF; Cr 2p3/2 and Cr 2p1/2, at binding energies of 577.13 eV and 586.82 eV, respectively, shifted to lower binding energies of 575.84 eV and 585.57 eV, respectively, for NiCr-LDH/VS2/NF with ΔBE = ∼1.25–1.29 eV. Meanwhile, in Fig. 2c, deconvoluted XPS spectra of both V 2p and O 1s demonstrated that both stemmed from the close binding energy values. From the deconvoluted V 2p XPS pattern, the +4 oxidation state of vanadium with two spin–orbit coupling peaks for each V 2p3/2 and V 2p1/2, is confirmed for both bare VS2/NF and NiCr-LDH/VS2/N at peak positions of 517.05 eV, 524.08 eV and 515.44 eV, 522.67 eV, respectively (ΔBE = ∼1.41–1.61 eV). Here, significant negative shifting reveals a negative charge redistribution after heterostructure formation also occurs in VS2 layers. Although for the NiCr-LDH/VS2/NF heterostructure, the +4 oxidation state of vanadium is confirmed by two peaks of V 2p, for bare VS2/NF two extra marginally small low intense peaks at 513.54 eV and 521.52 eV suggest a very low concentration of the +2 oxidation state of vanadium. This might be attributed to the surface oxidation of some VS2 components or a highly conductive material charge transfer occurring in between the layers of VS2 that might lead to a partial reduction of some vanadium ions. Next, for bare VS2/NF, O 1s peaks at 529.68 eV and 530.57 eV indicate, respectively, the M–O and M–OH (M = Ni, V) bonds present in the system. The O 1s peaks of NiCr-LDH/VS2/NF are slightly shifted to negative binding energy values of 529.16 eV and 530.05 eV (ΔBE = ∼0.52 eV). Following this, deconvoluted XPS spectra of S 2p for bare VS2/NF and NiCr-LDH/VS2/NF are portrayed in Fig. 2d and e, respectively. Bare VS2/NF peaks at 162.3 eV, 163.53 eV, 164.4 eV, and 167.8 eV represent S 2p3/2, S 2p1/2, metallic S, and S–O bonds, respectively. This weak signal of metallic S arises due to charge redistribution in the VS2 layer, and the interaction of V and S in highly conducting VS2 leads to an exclusive chemical environment. Another weak signal of S–O in high binding energy occurred due to minimal surface oxidation. For NiCr-LDH/VS2/NF, due to a two-step hydrothermal process, prolonged exposure of the surface to air at high temperature and pressure leads to more surface oxidation, and the S–O peak intensity increases along with additional peaks at 167.64 eV and 166.31 eV. However, broad peaks of S 2p3/2 and S 2p1/2 at 161.0 eV and 163.50 eV, respectively, suggest that after NiCr-LDH/VS2/NF heterostructure formation VS2 layers are still intact. Furthermore, BET analysis was carried out using the nitrogen (N2) adsorption/desorption process, and from the measured adsorbed volume the average surface area was calculated. As shown in Fig. 2f, BET and BJH analysis curves of NiCr-LDH/NF, bare VS2/NF, and NiCr-LDH/VS2/NF have 6.236, 23.117, and 74.500 m2 g−1 surface area values, respectively, with a typical type-IV isotherm with a H3-type sorption hysteresis loop, indicating the microporous nature of surface. Additionally, average pore widths of 5.226, 4.474, and 2.858 nm for the above-mentioned respective catalysts were obtained using the BJH method and are tabulated in Fig. S7,† suggesting the mesoporosity of all the synthesized catalysts. The total pore volume and BET isotherm results determined during the experiment are also charted in Fig. S7.† Fascinatingly, these results perfectly match with the electrochemical experiment where bare VS2/NF is the low performer and NiCr-LDH/VS2/NF is the most active, which can be explained from the high surface area and elevated total pore volume of NiCr-LDH/VS2/NF, enabling more surface-active sites for electrochemical performance.21,22 The mesoporous nature of the optimized LDH heterostructure catalyst with a narrower pore width creates a smooth channel, boosting the performance of electroactive species in faradaic redox reactions. Moreover, EPR analysis of all four catalysts is depicted in Fig. 2g. A sharp peak occurs for VS2 at g = 2.001, suggesting the availability of unpaired electrons present in the system that results in high conductivity of VS2.23,24 In the case of NiCr-LDH, EPR peak intensity is low due to Cr3+ in octahedral symmetry with 3d3 electronic configuration, showing electron spin S = 3/2 occupying lower energy t2g orbital.25 The combined effect of NiCr-LDH and bare VS2 in NiCr-LDH/VS2 leads to the low intense EPR peaks of NiCr-LDH/VS2/NF and VS2/NiCr-LDH/NF heterostructures, suggesting low magnetism and lesser unpaired electrons compared to highly conducting VS2. However, after formation of the heterostructure, due to charge redistribution and repopulation near the layers of NiCr-LDH and bare VS2, very few lattice distorted vibrational phonons with repopulated electron interaction increase the charge transfer effect in interlayer positions that ultimately boosts the electrochemical performance.19,26 The surface behavior of heterogeneous catalysts is also crucial for water-splitting applications for boosting the affinity between the electrode and electrolyte via efficient diffusion. Static contact angle measurements were performed for the Nickel Foam (NF) substrate, NiCr-LDH/NF, bare VS2/NF, and NiCr-LDH/VS2/NF to examine the wettability of these solid surfaces, determined by its intrinsic nature and 3D skeleton structure. For NF, the contact angle is approximately 150° with a water droplet, indicating a highly hydrophobic nature (Fig. S8†). In contrast, the optimized NiCr-LDH/VS2/NF heterostructure shows a superhydrophilic nature as it immediately absorbs the water droplet within a second with a contact angle outcome of 0° (Fig. 2h). This phenomenon attributed to surface roughness was drastically increased by the 3D porous structure and nanosheet of the NiCr-LDH/VS2/NF heterostructure (Fig. 2i). NiCr-LDH/NF shows moderate hydrophilicity with a contact angle of approximately 40°. Again, the contact angle of bare VS2/NF is high (≈135°), suggesting hydrophobicity for this catalyst. This is the reason for the low electrochemical catalytic activity of bare VS2/NF even though EPR analysis suggests the highly conductive nature of our prepared bare VS2/NF as it finds itself difficult to adsorb the water molecule upon contact.27–30
3. Experimental section
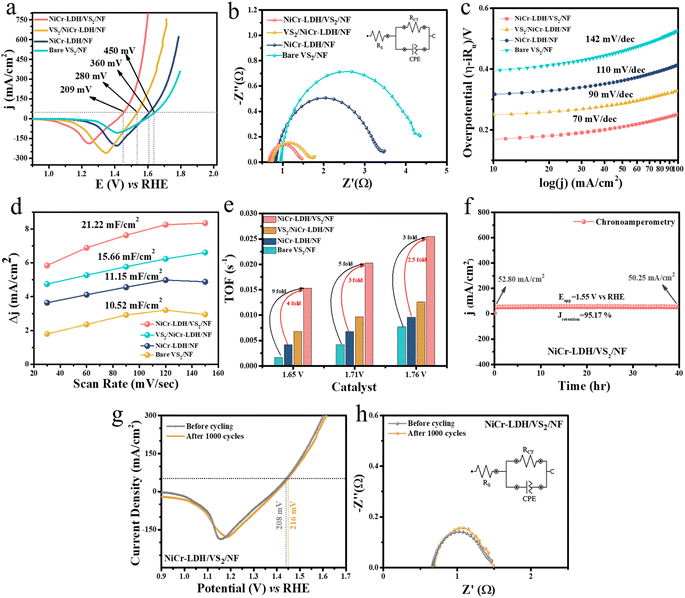
Once the successful fabrication of the NiCr-LDH/VS2/NF heterostructure was affirmed, we examined the electrochemical application of all the as-prepared catalysts, namely NiCr-LDH/NF, bare VS2/NF, NiCr-LDH/VS2/NF, and VS2/NiCr-LDH/NF. Among these, NiCr-LDH/VS2/NF showed remarkable catalytic activity in the OER, HER as well as the TWS reaction. Initially, the OER was carried out in a typical three-electrode system in a basic 1 M KOH solution. The above-mentioned prepared catalysts served as the working electrode with Pt/C as the counter electrode and Hg/HgO as the reference electrode. To gain insight into the activity of prepared catalysts, primarily a linear sweep voltammetry (LSV) study with a slow scan rate of 5 mV s−1 was performed (Fig. 3a). The 50% iR-corrected LSV curve reveals exceptional performance for NiCr-LDH/VS2/NF with a very low overpotential of 209 mV to attain 50 mA cm−2 current density, compared to VS2/NiCr-LDH/NF (280 mV), NiCr-LDH/NF (360 mV) and bare VS2/NF (450 mV). For better visualization, a bar diagram of overpotential values with an error bar at 50 mA cm−2 is displayed in Fig. S9.† This high performance of NiCr-LDH/VS2/NF heterostructure arises due to charge re-population near the Ni site and Cr site of NiCr-LDH layers that helps in stabilizing the intermediates generated during the whole process by lowering the activation energy of those steps. The corresponding electrochemical impedance spectroscopy (EIS) analysis (Fig. 3b) exhibited the lowest charge transfer resistance (Rct) value of 0.795 Ω for NiCr-LDH/VS2/NF, suggesting low resistance for electron transfer near the electrode–electrolyte interface. Rct values of NiCr-LDH/NF, bare VS2/NF, and VS2/NiCr-LDH/NF are 2.65, 3.75, and 1.02 Ω, respectively. The inset of Fig. 3b displays the corresponding circuit diagram. Furthermore, the Tafel slopes in Fig. 3c obtained from iR-corrected LSV analysis show values of 70, 90, 110, and 142 mV dec−1 for NiCr-LDH/VS2/NF, VS2/NiCr-LDH/NF, NiCr-LDH/NF, and bare VS2/NF, respectively, suggesting better charge transfer kinetics for the former one. Following this, the electrochemical double-layer capacitance (Cdl) analysis was performed to obtain data concerning the electrochemical active surface area (ECSA) as it directly correlates with the Cdl of a catalyst. Scan-rate-dependent CV within the potential range of 0.20 to 0.30 V vs. Hg/HgO (Fig. S10(a–d)†) and the difference in anodic and cathodic peak currents are plotted in Fig. 3d. From this figure, the obtained slope values give the ultimate Cdl values, which are 21.22, 15.66, 11.15, and 10.52 mF cm−2 for NiCr-LDH/VS2/NF, VS2/NiCr-LDH/NF, NiCr-LDH/NF, and bare VS2/NF, respectively. The large catalytic active site for NiCr-LDH/VS2/NF is again confirmed by the Cdl values. Later, a range of TOF values for all the catalysts were calculated as 1.65 V, 1.71 V, and 1.76 V, where NiCr-LDH/VS2/NF exhibits 9-fold, 5-fold and 3-fold increases of TOF value compared to bare VS2/NF and 4-fold, 3-fold, and 2.5-fold increases of TOF value compared to NiCr-LDH/NF, respectively, suggesting the optimized heterostructure releases very high concentration of oxygen molecules per unit active site per second than the remaining catalyst (Fig. 3e). The TOF values were calculated from the active surface area values from the reduction area obtained from backward LSV curves of all the catalysts (Fig. S11†).In Fig. 3f, static stability was evaluated using a chronoamperometric study at 1.55 V vs. RHE for the best active catalyst NiCr-LDH/VS2/NF that maintains 95.17% of its activity even after a long exposure of 40 hours under harsh anodic conditions. A dynamic stability, i.e., acceleration–degradation (AD) study was also performed with 1000 constant CV cycles at 150 mV s−1 (Fig. 3g and h), where minimal degradation of catalytic performance via LSV and EIS suggests advantageous robustness of the NiCr-LDH/VS2/NF heterostructure catalyst. A comparative table with previously reported related catalysts is displayed in Table S1,† which demonstrates the superior OER performance of NiCr-LDH/VS2/NF in the majority of instances. Additionally, in operando EIS analysis was carried out at different potentials from 1.02 V to 1.704 V to elucidate the enhanced electron flow for the optimized NiCr-LDH/VS2/NF heterostructure compared to another similar heterostructure and the source components. The Bode and Nyquist plots for all the catalysts are shown in Fig. S12(a–d) and (e–h),† respectively, and the obtained Rct outcomes at each potential are plotted for all catalysts in Fig. S12(i–l).† For better understanding and inspection, we have chosen a mid-voltage value of 1.564 V in the OER region. At 1.564 V, the gradual decrease of Rct values of bare VS2/NF, NiCr-LDH/NF, VS2/NiCr-LDH/NF, and NiCr-LDH/VS2/NF from 56.60, 53.65, 31.3, and 30, respectively, suggesting less hindrance and hence better charge transfer kinetics for the optimized NiCr-LDH/VS2/NF catalyst. Moreover, in Rct graphs, OER initiation starts from a very low resistance of 1.32 V, whereas for low-active bare VS2/NF, the Rct value is 1.37, suggesting higher resistance that prevents the smooth electrochemical reaction for bare VS2/NF. In the Bode plots, the high-frequency region and low-frequency region are shown by colour coding, where in the high-frequency region catalyst activation starts and in the low-frequency region electrooxidation of the catalysts occurs, i.e. the OER takes place. The phase angle values of bare VS2/NF, NiCr-LDH/NF, VS2/NiCr-LDH/NF, and NiCr-LDH/VS2/NF at 6 Hz frequency and 1.564 V are 42.2°, 40°, 31.3°, and 30°, respectively. The lowering of phase angle values of the above-mentioned respective catalysts at a particular frequency value and specific applied potential value in the low-frequency region suggests simplification of interface reaction charge transfer, resulting in boosted OER kinetics for NiCr-LDH/VS2/NF.
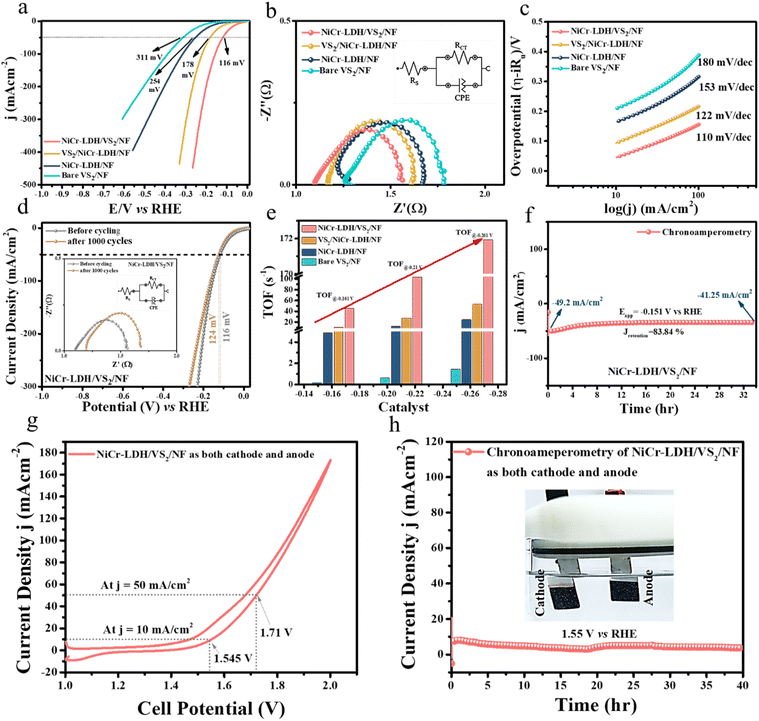
Following the OER, the HER was also performed in 1 M KOH with a three-electrode system for all the catalysts. For HER evaluation, all modified catalysts acted as a working electrode, Hg/HgO as the reference electrode, and a graphite electrode as the counter electrode. Likewise, NiCr-LDH/VS2/NF shows the highest activity followed by VS2/NiCr-LDH/NF, NiCr-LDH/NF, and bare VS2/NF. The 50% iR-corrected LSV curve in Fig. 4a demonstrates that NiCr-LDH/VS2/NF achieves the lowest overpotential of 116 mV to attain 50 mA cm−2 current density, while VS2/NiCr-LDH/NF, NiCr-LDH/NF, and bare VS2/NF show 178 mV, 254 mV, and 311 mV overpotential values, respectively. A bar diagram of HER overpotential values at 50 mA cm−2 with an error bar is displayed in Fig. S13† for better visualization. The corresponding impedance Rct values (0.45 Ω, 0.46 Ω, 0.47 Ω, and 0.49 Ω) and Tafel slope values (110, 122, 153, and 180 mV dec−1) signify low resistance and enhanced electron transfer efficiency across the electrode–electrolyte interface for the optimized NiCr-LDH/VS2/NF heterostructure (Fig. 4b and c). From the Tafel slope values, it is predictable that all the catalysts follow the Volmer–Heyrovsky pathway.31 The dynamic acceleration–degradation study stability over 1000 CV cycles (Fig. 4d) exhibited an overpotential upsurge of only 8 mV, confirming the catalyst's stability. In the inset, the corresponding fitted impedance spectra are included, which are consistent with the LSV findings. Next, TOF values at three different potential values were calculated, considering that the surface-active area is obtained from the non-faradic region of the scan rate-dependent CV curve at 30 mV s−1 scan rate. The TOF value increases in the order of bare VS2/NF → NiCr-LDH/NF → VS2/NiCr-LDH/NF → NiCr-LDH/VS2/NF in all the voltages chosen for analysis (Fig. 4e), suggesting a greater number of hydrogen molecule evolution per unit sec and per active site. Next, a static chronoamperometry study in Fig. 4f suggests ∼84% retention of the catalyst after 33-hour durability at a potential of −0.151 V vs. RHE. Moreover, the HER performance of our best active catalyst NiCr-LDH/VS2/NF was compared with a former reported comparable catalyst in Table S2,† where our catalyst stands consistent with those of the existing modified catalysts. Additionally, to gain insight into the conducting behaviour of prepared catalysts, an electrochemical Mott–Schottky analysis was carried out, and the results are portrayed in Fig. S14.† From Mott–Schottky (MS) analysis, negative slope of VS2 in Fig. S14b† suggests the p-type nature of bare VS2 and n-type structure of NiCr-LDH (Fig. S14a†).
After the formation of NiCr-LDH/VS2/NF, the semiconducting nature of NiCr-LDH immensely increases in the heterostructure owing to the synergistic effect of p-type bare VS2 and n-type NiCr-LDH. Moreover, as confirmed by the DFT study, charge repopulation occurs in both the layered structure of NiCr-LDH and bare VS2.
Building on the NiCr-LDH/VS2/NF heterostructure's excellent OER and HER performances, we created a two-electrode system in 1 M KOH alkaline media. The constructed bifunctional heterostructure exhibited excellent TWS performance, demanding cell voltages of 1.545 V and 1.71 V to attain 10 and 50 mA cm−2 current densities, respectively (Fig. 4g). The NiCr-LDH/VS2/NF‖NiCr-LDH/VS2/NF set-up (displayed in the inset of Fig. 4h) also exhibits 40-hour long-term durability at a constant potential of 1.55 V vs. RHE in the chronoamperometry experiment. The TWS results were compared with those of similar recently published catalysts (Table S3†), indicating that our synthesized NiCr-LDH/VS2/NF heterostructure is indeed an economical and effective bifunctional electrocatalyst with outstanding performance as well as steadiness for the water splitting reaction.
4. Computational insights from the DFT study
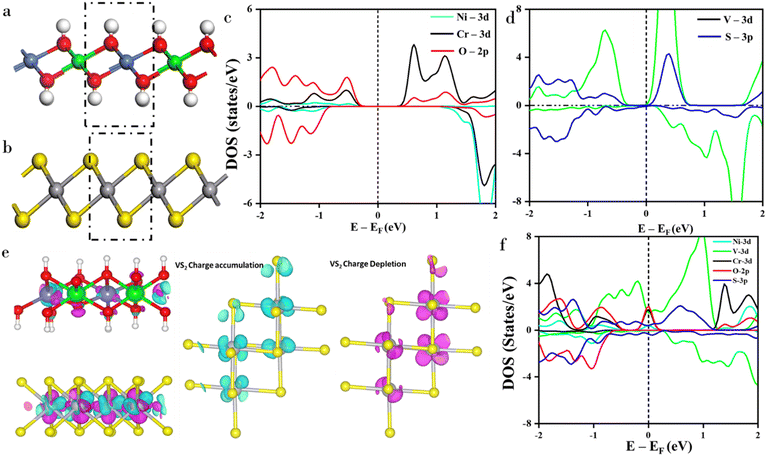
To understand the interaction between NiCr-LDH and VS2 systems, the structural and electronic properties were studied using first-principles calculations. Based on the experimental findings, we consider the system of NiCr-LDH to have a composition of 50% Ni and 50% Cr. To mimic the Ni2+ and Cr3+ ions in the LDH system, two hydrogen atoms were removed, and the selection of those atoms was done after considering various possible sites. The stable atomic structure is shown in Fig. 5. The calculated results revealed that the lattice parameters of NiCr-LDH and various bond distances are consistent with the previous theoretical as well as experimental reports,32–34 which are reported in Table 1. It is worth mentioning that compared to other LDH materials, NiCr-LDH exhibits a lower over-potential, and its performance can be plausibly improved further by doping, substitution, and heterostructuring with other catalysts.32–34 In this work, NiCr-LDH was combined with the conductive-rich VS2, to improve the electrocatalytic performance. Based on experimental findings, the underlying mechanism is investigated at the atomic level using first-principles calculations. The charge states of the metal atoms are assigned as Ni2+ and Cr3+, respectively, as confirmed by bond distance measurements and electronic density of states, following the elimination of hydrogen atoms. Average Ni–O and Cr–O bond distances are 2.09 and 2.02 Å, respectively.| System | Lattice parameter | Bond length |
|---|---|---|
| NiCr-LDH | a = b = 6.21; c = 20.95 Å | Cr–O = 2.04; Ni–O = 2.10 Å |
| VS2 | a = b = 6.58; c = 21.30 Å | V–S = 2.35 Å |
| NiCr-LDH/VS2 | a = 6.42; b = 6.36; c = 20.93 Å | Cr–O = 2.17; Ni–O = 2.38; V–S = 2.32–2.47 Å |
The electronic DOS (shown in Fig. 5c) clearly indicates that the electron occupancy in the 3d-orbital of Ni and Cr atoms is found to be t2g6e2g and t2g3e0g, respectively. In addition, the occupied and unoccupied energy levels are well separated, indicating the semiconducting nature of the NiCr-LDH system. On the other hand, very few states lie in the Fermi energy of the bare VS2 system (Fig. 5d), which facilitates the electron transfer during electrocatalytic reactions. To further understand the enhanced electrocatalytic performance of NiCr-LDH compared to VS2, a heterostructure of NiCr-LDH and VS2 is produced, and the optimized structure NiCr-LDH/VS2 is shown in Fig. 5e. The binding energy (EBE) is calculated as follows:
| EBE = ETotal − [[ENiCr-LDH] + [EVS2]] |
To understand the charge transfer between NiCr-LDH and VS2, we calculated the charge density difference (CDD) and analyzed the regions of charge accumulation and depletion, as depicted in Fig. 5e. It clearly demonstrates that the two layers interact primarily through electrostatic forces without significant charge transfer between the layers. Instead, the charge redistribution is pronounced within VS2 and NiCr-LDH layers; therefore, the energy gain during the formation of heterostructure is observed to be −0.91 eV. Here, we could not observe any significant charge present in the interface from CDD calculations, as it was formed by the hydroxyl groups (OH−) from one end and S2− ions from another end. Therefore, the interaction between two monolayers in the heterostructure is governed by electrostatic forces. Due to this type of interaction, the charge repopulation or charge redistribution is pronounced within the layers of VS2 as well as NiCr-LDH. To quantify the charge redistribution, we performed Bader charge analysis. The VS2 and NiCr-LDH layers exhibit charge re-populations of ∼0.21e− and 0.24e−, respectively, within the simulated cell. This redistributed charge improves the catalytic activity of the heterostructure. To provide deeper insights, we examined the charge accumulation and depletion specifically within the VS2 layer, as shown in Fig. 5e. The results indicate that the VS2 layer exhibits charge accumulation and depletion patterns, resembling the shapes of dxy or dyz orbitals. This phenomenon can be attributed to structural distortions in the VS2 layer due to the formation of the heterostructure. The originally symmetrical octahedral structure of VS2 transitions into a distorted configuration, with the bond distances varying from 2.32 to 2.47 Å. Notably, the axial bonds in the VS6 octahedra become more covalent due to decreasing bond lengths by 0.03 Å compared to its pristine form.
To further understand these effects, we calculated the PDOS and PARCHG, and the results are illustrated in Fig. 5f, S15 and S16.† These findings provide a comprehensive understanding of the electronic properties and local structural distortions that contribute to the enhanced electrocatalytic performance. Electronic properties of the heterostructure reveal that, closer to the Fermi level, Cr 3d and O 2p states are mainly presented as compared to Ni 3d states, which are responsible for enhanced electronic conductivity in the heterostructure. Furthermore, the small portion of O 2p states is unoccupied and that states are expected to activate the catalytic activity. It is also worth mentioning that the strong p–d hybridization between the Cr 3d orbitals and the O 2p orbitals is also observed after formation of the heterostructure, which enhances the overall catalytic activity. On the other hand, more amount of V 3d states are distributed at the Fermi level; therefore, the VS2 layer acts as an electron reservoir, where electrons are localized in dxy and dyz orbitals, as seen in Fig. S16.† In contrast, pristine VS2 (without NiCr-LDH) lacks hydroxyl groups, which reduces its electrocatalytic performance despite its intrinsic conductivity. The incorporation of NiCr-LDH into the system improves the performance, with the hydroxyl groups playing a pivotal role in promoting electron transfer and enhancing the overall electrocatalytic activity. To gain deeper insight into the contributions of individual orbitals, we deduced the angular momentum-decomposed DOS for 3d states of Cr, Ni, and V, as presented in Fig. S15.† The results reveal that in the case of VS2, the majority of states are dominated by the dxy and dyz orbitals. This phenomenon is attributed to charge localization predominantly within the axial plane, which further supports the understanding of the material’s electronic behavior and its impact on the electrocatalytic performance. The CDD results indicate that the VS2 layer exhibits significant charge accumulation in the x and y directions. Overall, our DFT reveals that the redistributed charges in the NiCr-LDH layers are directly involved in the catalytic activity owing to the presence of hydroxyl groups, whereas, such charges present in the VS2 layer act as the electron reservoir, which may be indirectly involved in the catalytic activity.
5. Post-characterization studies
The post-OER study of the working electrode after the electrochemical reaction was carried out using FE-SEM, HR-TEM, and XPS analyses, and the results are shown in Fig. S17.† The low- and high-magnification FESEM image confirm flower-like morphology and retention of all expected elements (Fig. S17a–c†); HRTEM images show that the sheet-like polycrystalline structure is retainedalong with all elements, including Ni, Cr, V, S, O, and K (arises due to 1 M KOH), as confirmed by elemental colour mapping (Fig. S17d–m†). Post-OER XPS spectra of Ni 2p, Cr 2p, and O 1s are presented in Fig. S17† for NiCr-LDH/VS2/NF, where additional peaks for the +3 oxidation state in Ni 2p, +6 oxidation state in Cr 2p, and surface adsorbate H2O molecules were observed, suggesting that all the elements undergo oxidation throughout the prolonged OER study. The low intensity Cr peak and the missing peak of V and S elements in the Fig. S18† survey spectrum of XPS along with the very low percentage of Cr, V, and S in the EDAX (Fig. S17c†) obtained because, as for post-study, powder sample acquired during synthesis of the materials was not analyzed; rather, directly substrate coated NF was taken for post-study. Therefore, a higher concentration of nickel from NF masks the other elements used as source elements for the formation of the heterostructure. However, in the post-OER colour mapping study performed by HR-TEM visually displays the presence of all the elements and their uniform elemental distribution, which confirm the stable and durable nature of the mesoporous nanostructured NiCr-LDH/VS2/NF catalyst despite the rigorous anodic potential sweep.6. Conclusion
To sum up, we have synthesized a novel heterostructure using the semiconducting NiCr LDH and highly conducting VS2, which shows high electrocatalytic performance. The optimized mesoporous NiCr-LDH/VS2/NF heterostructure, with its superhydrophilic nature, shows excellent bifunctional activity for the OER and HER with overpotentials of 209 mV and 116 mV, respectively, to attain 50 mA cm−2 current density in 1 M KOH solution. Moreover, NiCr-LDH/VS2/NF needs a cell voltage of 1.545 V to attain 10 mA cm−2 current density with 40-hour long-term exposure, demonstrating the stable nature of the catalyst. The elevated electrochemical performance of NiCr-LDH/NF after forming the NiCr-LDH/VS2/NF heterostructure is due to the charge repopulation in NiCr-LDH as well as the VS2 layer. This helps in electron transfer and facilitates the overall mechanism by ensuring the stability of the high oxidation state for the OER and enabling relaxed hydrogen abstraction from metal-absorbed H2O in the HER. In conclusion, combining NiCr-LDH with VS2 significantly enhances the electrocatalytic performance due to the conducting nature of VS2, electrostatic interaction between the layer, and hybridization between the 3d states of Cr and the 2p states of oxygen. In the heterostructure, the synergistic interaction between NiCr-LDH and VS2 provides a feasible route for improving the electrocatalytic efficiency. The detailed DFT theory study highlights an interesting charge repopulation observation in both the layers of NiCr-LDH and VS2, which increases the overall electrocatalytic reaction performance (confirmed by charge density and Bader charge analysis). The in operando EIS analysis helps in the understanding of the reaction kinetics. Overall, this work might help researchers for the fabrication and development of efficient bifunctional economically affordable heterostructure electrocatalysts using transition metal-based LDHs, oxides, chalcogenides etc. for water splitting studies for the future generation of green hydrogen technology.Data availability
The authors declare that all data from the submitted work are available upon request from the authors.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors wish to acknowledge Dr K. Ramesha, Director, CSIR-CECRI, for his constant support. Aditi De wishes to acknowledge support through a DST fellowship with project number CRG/2021/001089. Also, the authors would like to acknowledge V. Prabu, K. RanjithKumar, Krushna Chandra Nayak, and R. Ravishankar of the CIF division for their help in characterization studies. S. Kundu wishes to acknowledge the Department of Science and Technology (DST) for CRG research funding – funding number # CRG/2021/001089 dated 21 November 2021 with institute project number GAP 22/2021 dated 1 February 2022. CSIR-CECRI MS Reference Number: CECRI/PESVC/Pubs/2024-156.References
-
T. Nejat Veziroglu, in Energy Procedia, Elsevier Ltd, 2012, vol. 29, pp. 654–656 Search PubMed
.
- T. N. Veziroǧlu and S. Şahin, Energy Convers. Manage., 2008, 49, 1820–1831 CrossRef
.
- J. O. Abe, A. P. I. Popoola, E. Ajenifuja and O. M. Popoola, Int. J. Hydrogen Energy, 2019, 44, 15072–15086 CrossRef CAS
.
- W. Li, Y. Liu, A. Azam, Y. Liu, J. Yang, D. Wang, C. C. Sorrell, C. Zhao and S. Li, Adv. Mater., 2024, 36, 2404658 CrossRef CAS PubMed
.
- A. De, A. Karmakar and S. Kundu, ACS Appl. Energy Mater., 2023, 6, 5761–5773 CrossRef CAS
.
- K. Bera, R. Madhu, H. N. Dhandapani, S. Nagappan, A. De and S. Kundu, Inorg. Chem., 2022, 61, 16895–16904 CrossRef CAS PubMed
.
- S. Nagappan, A. Karmakar, R. Madhu, H. N Dhandapani, K. Bera, A. De and S. Kundu, ACS Appl. Energy Mater., 2022, 5, 12768–12781 CrossRef CAS
.
- R. Madhu, A. Karmakar, K. Bera, S. Nagappan, H. N. Dhandapani, A. De, S. S. Roy and S. Kundu, Mater. Chem. Front., 2023, 7, 2120–2152 RSC
.
- Y. Yang, S. Wei, Y. Li, D. Guo, H. Liu and L. Liu, Appl. Catal., B, 2022, 314, 121491 CrossRef CAS
.
- Y. Guo, X. Zou, X. Wei, W. Bao, J. Zhang, J. Han and F. Jia, Chin. J. Struct. Chem., 2024, 43, 100206 CrossRef CAS
.
- X. Zhong, J. Tang, J. Wang, M. Shao, J. Chai, S. Wang, M. Yang, Y. Yang, N. Wang, S. Wang, B. Xu and H. Pan, Electrochim. Acta, 2018, 269, 55–61 CrossRef CAS
.
- T. Wang, X. Zhang, X. Yu, J. Li, K. Wang and J. Niu, Molecules, 2024, 29, 951 CrossRef CAS PubMed
.
- J. Feng, X. Sun, C. Wu, L. Peng, C. Lin, S. Hu, J. Yang and Y. Xie, J. Am. Chem. Soc., 2011, 133, 17832–17838 CrossRef CAS PubMed
.
- Y. Cui, W. Fan, X. Liu, J. Ren and Y. Gao, Comput. Mater. Sci., 2021, 200, 110767 CrossRef CAS
.
- A. T. E. Vilian, S.-K. Hwang, M. J. Lee, M. Bagavathi, Y. S. Huh and Y.-K. Han, Ecotoxicol. Environ. Saf., 2021, 228, 113021 CrossRef CAS PubMed
.
- N. S. Padalkar, S. V. Sadavar, R. B. Shinde, A. S. Patil, U. M. Patil, D. S. Dhawale, R. N. Bulakhe, H. Kim, H. Im, A. Vinu, C. D. Lokhande and J. L. Gunjakar, J. Colloid Interface Sci., 2022, 616, 548–559 CrossRef CAS PubMed
.
- U. K. Ghorui, P. Mondal, B. Adhikary, A. Mondal and A. Sarkar, ChemElectroChem, 2022, 9, 202200526 CrossRef
.
- J. Yuan, J. Wu, W. J. Hardy, P. Loya, M. Lou, Y. Yang, S. Najmaei, M. Jiang, F. Qin, K. Keyshar, H. Ji, W. Gao, J. Bao, J. Kono, D. Natelson, P. M. Ajayan and J. Lou, Adv. Mater., 2015, 27, 5605–5609 CrossRef CAS PubMed
.
- X. Tian, X. Yan, G. Varnavides, Y. Yuan, D. S. Kim, C. J. Ciccarino, P. Anikeeva, M.-Y. Li, L.-J. Li, P. Narang, X. Pan and J. Miao, Sci. Adv., 2021, 7, eabi6699 CrossRef CAS PubMed
.
- S. Singha Roy, R. Madhu, K. Bera, S. Nagappan, H. N. Dhandapani, A. De and S. Kundu, ACS Appl. Mater. Interfaces, 2024, 16, 5965–5976 CrossRef CAS PubMed
.
- S. Cong, J. Yu, B. Liu, W. Teng and Y. Tang, New J. Chem., 2022, 46, 15227–15243 RSC
.
- A. Elsonbaty, M. Harb, M. Soliman, S. Ebrahim and A. Eltahan, Appl. Phys. Lett., 2021, 118, 023901 CrossRef CAS
.
- R. Weindl, R. Khare, L. Kovarik, A. Jentys, K. Reuter, H. Shi and J. A. Lercher, Angew. Chem., Int. Ed., 2021, 60, 9301–9305 CrossRef CAS PubMed
.
- R. Wang, Q. Chen, X. Liu, Y. Hu, L. Cao and B. Dong, Small, 2024, 20, 2311217 CrossRef CAS PubMed
.
- Y. Yang, L. Dang, M. J. Shearer, H. Sheng, W. Li, J. Chen, P. Xiao, Y. Zhang, R. J. Hamers and S. Jin, Adv. Energy Mater., 2018, 8, 1703189 CrossRef
.
- Y. Ni, H. Zhang, S. Hu, H. Wang, S. Volz and S. Xiong, Int. J. Heat Mass Transfer, 2019, 144, 118608 CrossRef CAS
.
- V. Pundir, A. Gaur, R. Kaur, Aashi and V. Bagchi, Energy Adv., 2023, 2, 321–327 RSC
.
- L. Huang, L. Xu, Y. Yang, H. Yu, H. Tao, D. Li and X. Dong, J. Mater. Sci.: Mater. Electron., 2020, 31, 6607–6617 CrossRef CAS
.
- J. Cao, J. Zhou, Y. Zhang, Y. Wang and X. Liu, ACS Appl. Mater. Interfaces, 2018, 10, 1752–1760 CrossRef CAS PubMed
.
- N. Chen, S. Che, H. Liu, N. Ta, G. Li, F. Chen, G. Ma, F. Yang and Y. Li, Catalysts, 2022, 12, 1319 CrossRef CAS
.
- A. De, R. Madhu, K. Bera, H. N. Dhandapani, S. Nagappan, S. Singha Roy and S. Kundu, J. Mater. Chem. A, 2023, 11, 25055–25071 RSC
.
- S. Biswal, N. Divya, B. Mishra, D. Pohl, B. Rellinghaus, D. Ghosh and B. P. Tripathi, J. Mater. Chem. A, 2023, 12, 2491–2500 RSC
.
- Q. Su, P. Wang, Q. Liu, R. Sheng, W. Cheng, J. Ding, Y. Lei and Y. Huang, Appl. Catal., B, 2024, 351, 123994 CrossRef CAS
.
- A. A. Lobinsky, D. S. Dmitriev and V. I. Popkov, Int. J. Hydrogen Energy, 2023, 48, 22495–22501 CrossRef CAS
.
Footnote |
| † Electronic supplementary information (ESI) available: A detailed scheme of synthesis with reagents used, electrode fabrication, information about instruments used, additional characterization results, and electrochemical study outcomes. See DOI: https://doi.org/10.1039/d5ta01733e |
| This journal is © The Royal Society of Chemistry 2025 |