Efficient bifunctional water splitting catalysts enabled by crystalline–amorphous NixSy@NiFe LDH heterojunctions†
Shaobo
Ye
,
Yong
Xu
,
Xiaoyu
Bai
,
Zhao
Liang
,
Qiao
Liu
 ,
Qiliang
Wei
,
Qiliang
Wei
 ,
Dongjiang
Yang
,
Dongjiang
Yang
 ,
Weiyou
Yang
,
Weiyou
Yang
 ,
Fengmei
Gao
* and
Qing
Shi
,
Fengmei
Gao
* and
Qing
Shi
 *
*
Institute of Micro/Nano Materials and Devices, Ningbo University of Technology, Ningbo City, 315211, P. R. China. E-mail: sq@nbut.edu.cn
First published on 4th April 2025
Abstract
Developing crystalline–amorphous heterojunctions presents a promising pathway to enhance the electrocatalytic performance of both the hydrogen evolution reaction (HER) and the oxygen evolution reaction (OER). This study reports the exploration of novel crystalline–amorphous NixSy@NiFe LDH heterostructures for efficient HER and OER, which are synthesized via in situ growth of NiFe-LDH on nickel sulfide (NixSy) nanowires. The resultant three-dimensional core–shell structures remarkably increase the active sites, enhance the charge transfer, and facilitate the gas release during the catalytic process. In situ Raman spectroscopy and density functional theory (DFT) calculations verify that the introduced Fe could boost the OER activity by promoting the structural reconstruction to form the disorder of NiOOH@NiFeOOH species, and reducing the energy barrier for conversion of oxygen-containing intermediates. The heterojunction interface in NixSy@NiFe LDH modifies the electron distribution, thus significantly lowering the Gibbs free energy of hydrogen adsorption (ΔGH* = 0.1 eV) compared to that of NixSy. Correspondingly, the NixSy@NiFe LDH exhibits superior bifunctional performance for the HER and OER in alkaline solution, delivering high current densities of −100 and 200 mA cm−2 at low overpotentials of 159 and 250 mV for the HER and OER, respectively, as well as an excellent stability against operation over 250 h at 200 mA cm−2, implying its promise toward commercial applications.
1. Introduction
With the acceleration of industrialization and the widespread use of fossil fuels, the problem of the greenhouse effect has become increasingly severe. As a result, reducing carbon emissions has become one of the most urgent global challenges. Hydrogen, as a clean energy source, is regarded as an ideal substitute for fossil fuels, and the electrolysis of water to produce hydrogen is considered a practical method for achieving this transition.1–4 Over the years, extensive research has been conducted on electrochemical water splitting. The cathodic process involves a two-electron hydrogen evolution reaction, while the oxygen evolution reaction (OER) at the anode, which is a four-electron coupled proton transfer process, represents a significant barrier to efficient water splitting, ultimately limiting overall performance.5,6 Currently, commercially available catalysts predominantly consist of precious metal catalysts, such as Pt- and Ru/Ir-based catalysts. However, their limited availability and high costs pose significant challenges for large-scale application. Therefore, the development of resourceful, cost-effective and high-performance bifunctional catalysts is crucial for achieving large-scale water splitting.7,8 Consequently, researchers have explored various low-cost alternatives for the HER and OER including transition metal sulfides,9,10 phosphides,11,12 nitrides,13,14 carbides14,15 and metal hydroxides.16–20 To minimize material costs and improve economic viability, researchers are focusing on the development of bifunctional catalysts with high catalytic activity and exceptional durability for use in electrolytic cells.Layered double hydroxides (LDHs) are a class of materials with layered structures, where the main layers are composed of two different valence metal cations (such as Ni2+ and Fe3+) connected by oxygen bridges, and interlayer spaces are occupied by exchangeable anions (such as carbonate and nitrate) and water molecules.21 In 2013, Dai and his team first applied LDHs to the electrocatalytic oxygen evolution reaction (OER), sparking widespread attention in the field.22–24 However, the intrinsic low electrical conductivity and limited exposure of active sites in LDHs have limited their catalytic performance. To overcome these limitations, researchers have employed various strategies to optimize the performance of LDHs, for example, doping, exfoliation, morphology modulation, and construction of heterojunctions. Guo et al. developed an efficient NiFeOOH–VZn catalyst through in situ leaching of Zn from the NiFeZn LDH.25 This leaching process effectively lowers the formation potential of NiFeOOH active species, thereby significantly enhancing catalytic activity for the OER. Zhou et al. reported a defect-rich few-layered NiFe-LDH nanosheet with in situ borate modified by gas-exfoliation using NaBH4 as a tri-functional reductant. The introduction of borate elevated the oxidation state of Ni, narrowed the band gap, and upshifted the d band center of the catalyst, resulting in a reduced energy barrier for the OER.26 Compared with other structural adjustment strategies, the construction of conductive core and amorphous shell heterojunction interfaces is a particularly effective strategy for enhancing electrocatalytic performance.27,28 This strategy offers several notable advantages: (1) overcoming the inherently low conductivity of LDHs; (2) maximizing the exposure of active sites to improve catalytic efficiency; (3) regulating the interfacial charge distribution and optimizing the electron structure; and (4) integrating diverse active species onto a single electrode, thereby achieving a multifunctional catalyst. The synergistic interaction of these factors not only significantly enhances catalytic performance but also imparts multifunctional catalytic properties to the electrode. For example, Lv and his team fabricated a Co3O4@NiFe LDH heterojunction for the OER, whose interface provides abundant active sites, thereby facilitating rapid charge transfer.29 He et al. developed a NiTe@NiCo LDH heterojunction for the OER. The heterojunction significantly enhances the efficiency of electron transfer and catalytic activity through the significant interface effect, showing lower overpotential and superior stability compared to single-component materials.30
In parallel, transition metal sulfides such as Ni3S2 have garnered attention as promising HER catalysts due to their excellent metallic conductivity and stability. For example, Zhang et al. demonstrated that copper doping in Ni3S2 significantly enhanced its HER performance by enhancing water adsorption and dissociation.31 Peng and his colleagues developed porous Ni/Ni3S2 micro-sheets on nickel foam, achieving excellent HER and OER activity.32 Ni3S2 also exhibits intrinsic OER catalytic activity and excellent conductivity, making it an ideal candidate for constructing efficient heterojunctions to simultaneously enhance both the OER and HER performance. Consequently, integrating LDH and Ni3S2 into a single electrocatalyst represents an efficient strategy for constructing highly effective bifunctional catalysts for both the OER and the HER. However, while several studies have reported bifunctional electrocatalysts based on LDH and Ni3S2, the rational design of a structure that maximizes the exposure of active species and enhances conductivity remains an important yet challenging task. Furthermore, systematically uncovering the synergistic interactions between LDH and Ni3S2 is critical for the theoretical design and development of high-performance catalysts.
Herein, we developed a crystalline–amorphous NixSy@NiFe LDH heterojunction bifunctional catalyst, characterized by crystalline NixSy nanowires serving as the core and amorphous NiFe LDH nanosheets forming the shell. The dynamic surface reconstruction mechanism during the OER process was unveiled through in situ Raman spectroscopy. The results reveal that the NixSy@NiFe LDH heterojunction undergoes a surface reconstruction process, leading to the formation of a NixSy@NiOOH@NiFeOOH heterojunction with an increased degree of structural disorder. Theoretical calculations further demonstrated that the reconstructed NiOOH@NiFeOOH active species possesses lower Gibbs free energies for the rate-limiting step in the conversion from *OH to *O, enhancing the OER performance. For the HER, theoretical calculations were employed to investigate the active sites in NixSy@NiFe LDH, including H adsorption on Nix*Sy@NiFe LDH and NixSy@Ni*Fe LDH, as well as Nix*Sy. The calculations confirmed that the Ni* sites in Ni*xSy@NiFe LDH exhibit an extremely low adsorption Gibbs free energy (ΔGH* = 0.1 eV), significantly lower than those in Ni*xSy (0.32 eV) and NixSy@Ni*Fe LDH (0.71 eV). These findings indicate the heterojunction interface between NixSy and NiFe LDH efficiently regulates the electronic structure, thus improving the catalytic activity for the HER. As a result, the NixSy@NiFe LDH exhibited excellent catalytic activity and stability for both the OER and the HER. Specifically, it required an overpotential of 159 mV at −100 mA cm−2 for the HER and 250 mV at 200 mA cm−2 for the OER, while maintaining stable operation for over 250 hours. This innovative design not only achieves highly efficient and durable bifunctional electrocatalysts but also provides valuable insights into the synergistic catalytic mechanisms of heterojunction catalysts, paving the way for the development of advanced electrocatalysts for sustainable energy applications.
2. Results and discussion
2.1 Synthesis and characterization
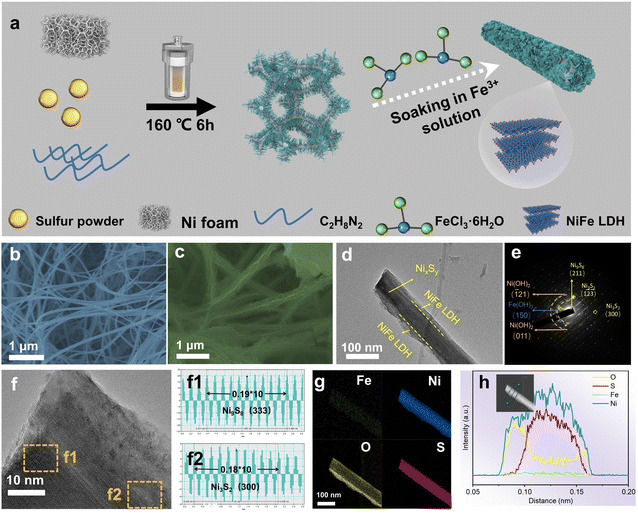
The synthesis route for NixSy@NiFe LDH is illustrated in Fig. 1a. NixSy nanowire arrays (Fig. 1b) with an average diameter of approximately 70 nm were first synthesized on smooth nickel foam (Fig. S1†) using a hydrothermal method. The control group produced NixSy precursor catalysts with varying morphologies by precisely controlling the amount of sulfur powder added, as shown in Fig. S2a–f.† Subsequently, NixSy@NiFe LDH nanowire heterojunctions were obtained through chemical soaking in a FeCl3·6H2O aqueous solution at room temperature for 30 min. Following soaking with FeCl3·6H2O solution, numerous nanosheets were clearly observed to grow uniformly and vertically on the nanowires, forming a unique 3D core–shell nanostructure with a very rough surface (Fig. 1c and d). In the control group, different morphologies of NixSy@NiFe LDH catalysts (Fig. S3a–j†) were achieved by carefully regulating the soaking time. The NixSy nanowire arrays on nickel foam provide plenty of active sites for the growth of NiFe LDH nanosheets. Furthermore, these nanowires also function as excellent electron-conducting pathways. This novel hierarchical three-dimensional core–shell nanostructure not only offers abundant catalytically active sites but also facilitates contact with the electrolyte and promotes rapid release of generated gas bubbles.33The TEM image in Fig. 1d further illustrates the core–shell nanostructures with a 50 nm core of NixSy nanorods and a 20 nm shell of NiFe LDH nanosheets. The interconnected NiFe LDH nanosheets provided abundant channels for rapid electrolyte diffusion and efficient gas release, and the interface between crystalline NixSy and amorphous NiFe LDH generates rich defects that modulate the electronic structure and facilitate charge redistribution at the boundary, thereby enhancing HER and OER performance. Fig. 1e displays the selected area electron diffraction (SAED) pattern of NixSy@NiFe LDH. The bright, evenly distributed diffraction spots are assigned to the (211) face of Ni9S8 and the (1![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif)
![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ) and (300) faces of Ni3S2, indicating that the NixSy core exhibits a high degree of crystallinity. And the diffraction rings without distinct bright spots are associated with the amorphous (
) and (300) faces of Ni3S2, indicating that the NixSy core exhibits a high degree of crystallinity. And the diffraction rings without distinct bright spots are associated with the amorphous (![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 21) and (011) faces of Ni(OH)2 and the (150) face of Fe(OH)3, confirming the amorphous nature of the NiFe-LDH shell. This combination of crystalline and amorphous structures highlights the unique core–shell architecture of NixSy@NiFe LDH. The HR-TEM images of NixSy@NiFe LDH (Fig. 1f) reveal lattice spacings of 0.19 nm for the (333) crystal face of Ni9S8 (Fig. 1f1) and 0.18 nm for the (300) crystal face of Ni3S2 (Fig. 1f2). The NiFe LDH nanosheets in NixSy@NiFe LDH exhibited numerous defects, as indicated by the arrows in Fig. S4.† These disordered defects distributed along the lattice edges of the NiFe LDH nanosheets, suggesting the presence of a large number of dangling bonds at the surface of these materials.34 This can increase the number of exposed active sites and alter the electronic properties of the interface, thereby significantly enhancing the catalytic activity. The successful synthesis of NixSy@NiFe LDH core–shell nanostructures was further confirmed using the elemental mapping image (Fig. 1g) and Energy Dispersive X-ray (EDX) line scanning results (Fig. S5 and S2h†), clearly indicating the presence of NixSy in the central region and NiFe LDH in the outer region. The Ni and Fe contents in the fabricated samples were quantitatively analyzed by inductively coupled plasma-optical emission spectrometry (ICP-OES), as summarized in Table S1.† The Ni contents in NixSy@NiFe LDH, NixSy and NiFe LDH are 67.3%, 68.7% and 65.9%, respectively, while the Fe contents in NixSy@NiFe LDH and NiFe LDH are 3.5% and 3.3%, respectively. Additionally, we investigated the morphological changes of NiFe LDH at varying soaking times. The SEM images are presented in Fig. S6.† As the soaking time increases, the soaking patterns on the smooth nickel foam surface become more pronounced.
21) and (011) faces of Ni(OH)2 and the (150) face of Fe(OH)3, confirming the amorphous nature of the NiFe-LDH shell. This combination of crystalline and amorphous structures highlights the unique core–shell architecture of NixSy@NiFe LDH. The HR-TEM images of NixSy@NiFe LDH (Fig. 1f) reveal lattice spacings of 0.19 nm for the (333) crystal face of Ni9S8 (Fig. 1f1) and 0.18 nm for the (300) crystal face of Ni3S2 (Fig. 1f2). The NiFe LDH nanosheets in NixSy@NiFe LDH exhibited numerous defects, as indicated by the arrows in Fig. S4.† These disordered defects distributed along the lattice edges of the NiFe LDH nanosheets, suggesting the presence of a large number of dangling bonds at the surface of these materials.34 This can increase the number of exposed active sites and alter the electronic properties of the interface, thereby significantly enhancing the catalytic activity. The successful synthesis of NixSy@NiFe LDH core–shell nanostructures was further confirmed using the elemental mapping image (Fig. 1g) and Energy Dispersive X-ray (EDX) line scanning results (Fig. S5 and S2h†), clearly indicating the presence of NixSy in the central region and NiFe LDH in the outer region. The Ni and Fe contents in the fabricated samples were quantitatively analyzed by inductively coupled plasma-optical emission spectrometry (ICP-OES), as summarized in Table S1.† The Ni contents in NixSy@NiFe LDH, NixSy and NiFe LDH are 67.3%, 68.7% and 65.9%, respectively, while the Fe contents in NixSy@NiFe LDH and NiFe LDH are 3.5% and 3.3%, respectively. Additionally, we investigated the morphological changes of NiFe LDH at varying soaking times. The SEM images are presented in Fig. S6.† As the soaking time increases, the soaking patterns on the smooth nickel foam surface become more pronounced.
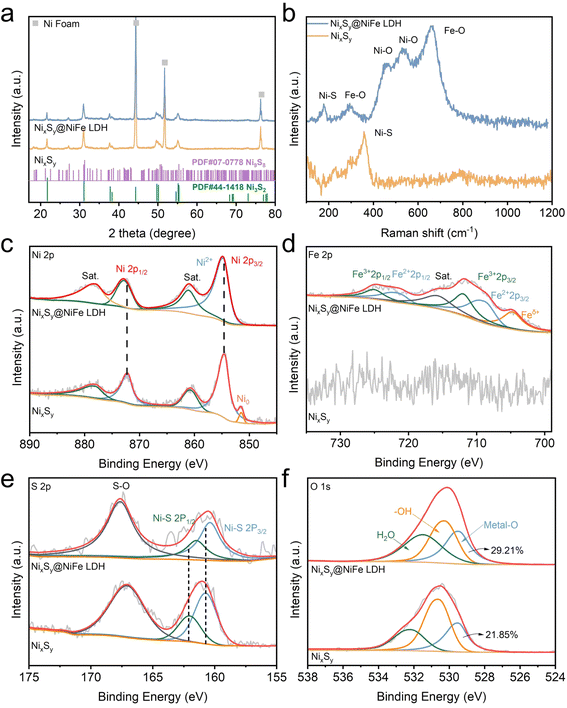
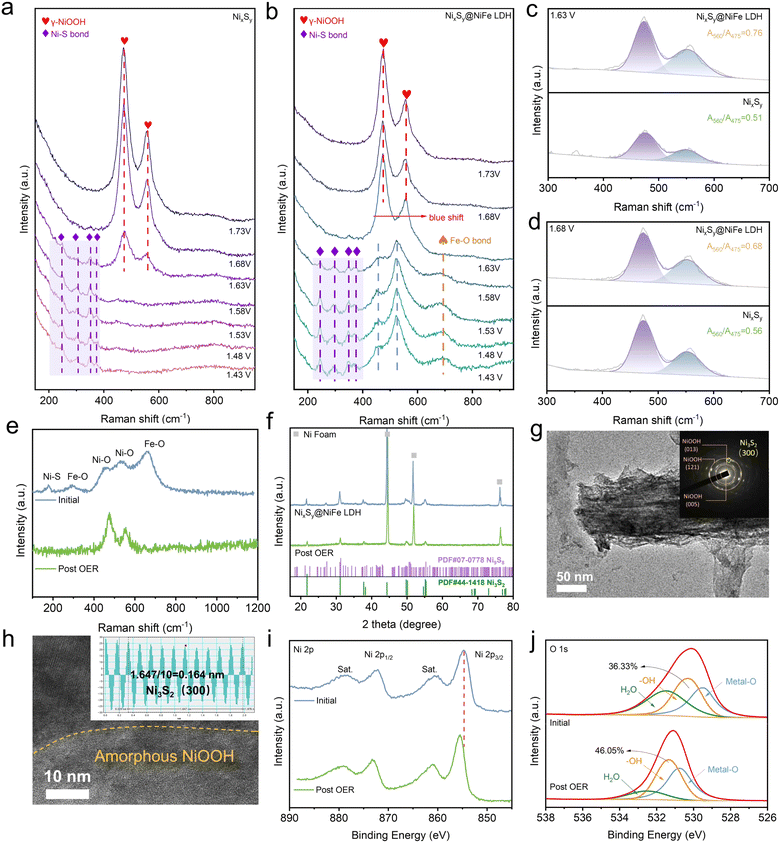
The structure of the as-prepared samples was further analyzed using X-ray diffraction (XRD). As shown in Fig. 2a, the diffraction peaks at 21.7°, 31.1°, 38.2°, 50.1° and 55.3° correspond to the (101), (110), (021), (211) and (300) crystal planes of Ni3S2 (JCPDS no. 44-1418), while the diffraction peaks at 18.3°, 27.3° and 51.3° belong to the (021), (131) and (423) crystal planes of Ni9S8 (JCPDS no. 07-0778). Following the modification of NiFe LDH nanosheets, no new diffraction peaks were detected, which can be attributed to the amorphous state of the NiFe LDH nanosheets consistent with the TEM results.35 The prepared samples were further characterized using Raman spectroscopy. As shown in Fig. 2b, a prominent characteristic peak at 377 cm−1 associated with a non-vulcanized Ni–S bond was observed in the NixSy sample.36 The Raman peaks at 178 and 282 cm−1 correspond to characteristic vibrations of Ni–S and Fe–O bonds, while the broad bands detected at 452 cm−1 and 535 cm−1 are attributed to the presence of disordered Ni(OH)2 clusters. Additionally, the peak at 658 cm−1 corresponds to the Fe–O bond within disordered FeOOH nanosheets, which were observed in NixSy@NiFe LDH. These results also verify the successful preparation of NixSy@NiFe LDH heterojunctions.37,38
X-ray photoelectron spectroscopy (XPS) was employed for further analyzing the elemental composition and chemical valence states of the as-fabricated samples. The XPS survey scan shown in Fig. S7† reveals the presence of O, S, Fe, and Ni elements in NixSy@NiFe LDH, while the characteristic signals associated with Fe are absent in the NixSy sample. In the high-resolution Ni 2p spectra depicted in Fig. 2c, the Ni0 characteristic peak at 851.5 eV is only observed in NixSy,39 and the Ni0 peak disappears in NixSy@NiFe LDH, possibly due to signal attenuation and oxidation of high-valence metal ions from the NiFe LDH layer. Nevertheless, the binding energies at 854.78 eV (Ni 2p1/2) and 872.83 eV (Ni 2p3/2) for NixSy@NiFe LDH are shifted toward higher values compared to those of NixSy, indicating a significant electron interaction between the NixSy core and the NiFe LDH shell.40 In the Fe 2p spectra presented in Fig. 2d, the Fe 2p3/2 and 2p1/2 peaks of Fe2+ are located at 708.7 and 722.3 eV, and Fe 2p3/2 and 2p1/2 peaks of Fe3+ are located at 711.2 and 724.5 eV, respectively. Fe2+ arises from the reduction of trivalent iron ions to bivalent ions during sulfide soaking with trivalent iron salts, while a peak at approximately 704.8 eV is attributed to Feδ+ associated with an Fe–S bond,41 suggesting that the Fe–S bonds are formed during the soaking process. Peak fitting of the S 2p spectrum of NixSy (Fig. 2e) reveals a pair of peaks at 160.73 and 162.03 eV, along with a broad peak at 167.07 eV. The former peaks correspond to the presence of a M–S bond, while the latter peak indicates the existence of a surface sulfur–oxygen bond.42 In the NixSy@NiFe LDH sample, the shift towards lower binding energy in the sulfur–nickel metallic bond signals confirms electron transfer between Ni–S–Fe. The peaks centered at 532.19, 530.60, and 529.50 eV in the O 1s spectrum correspond to surface-adsorbed molecular water (Fig. 2f), hydroxyl groups, and metal oxide species, respectively.43 The content of M–O in NixSy@NiFe LDH is significantly increased due to the formation of Ni–O and Fe–O bonds in the soaking process. These findings demonstrate that oxygen-containing nickel and iron species are successfully associated with binding NixSy during the soaking process, leading to alterations in electronic states and thus enhancing the catalytic activity.
2.2 Electrocatalytic performance
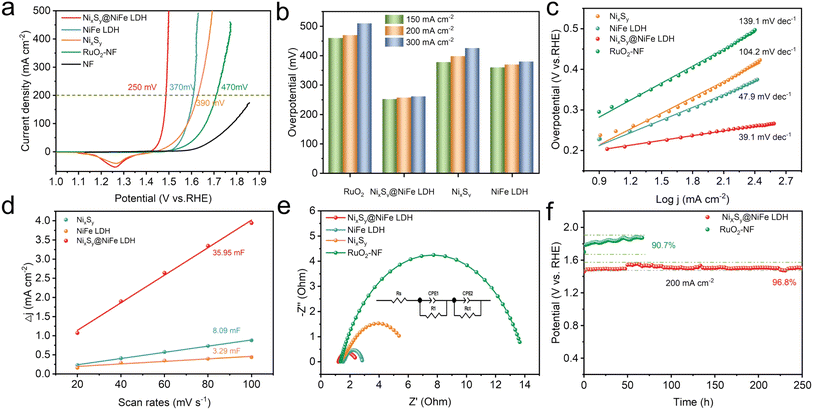
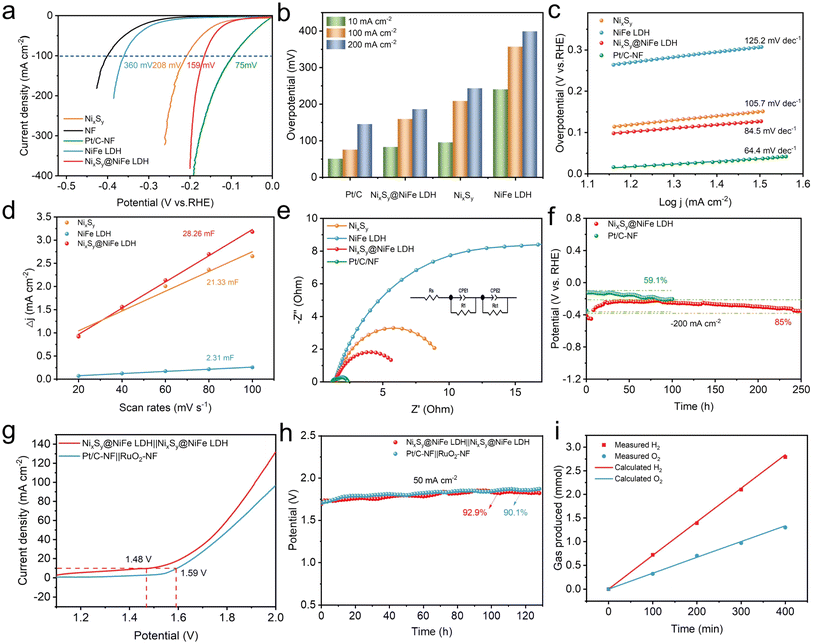
The OER and HER performances of the as-fabricated electrodes were investigated and compared with those of RuO2 and Pt/C coated nickel foam (RuO2/NF and Pt/C/NF) in 1.0 M KOH electrolyte. In order to obtain the best performance for the OER and HER, NixSy precursors and NixSy@NiFe LDH were evaluated by varying the amount of S powder (Fig. S8†) and the soaking time (Fig. S9†). It is worth noting that the NixSy precursor and NixSy@NiFe LDH electrode exhibit superior OER and HER performance when using a S powder dosage of 60 mg and a soaking time of 30 min, respectively.Turnover frequency (TOF) is one of the important parameters for evaluating the kinetics of electrocatalysts. According to previous studies, TOF was measured by electrochemical methods.45 Fig. S11† shows the linear relationship between the reduction peaks of the fabricated samples at different scan rates, with the detailed calculation process described in the TOF calculation section. As shown in Fig. S12a,† the NixSy@NiFe LDH catalyst exhibited the highest TOF value. And at an overpotential of 250 mV, the TOF value of NixSy@NiFe LDH (0.82) was significantly higher than that of NiFe LDH (0.01) and NixSy (0.02), as shown in Fig. S12b.† To investigate the intrinsic OER catalytic activity of the fabricated catalysts, the OER kinetic current was normalized using their corresponding ECSA and roughness factor (Rf). As shown in Fig. S13a,† the NixSy@NiFe LDH electrode exhibited an overpotential of 267 mV at a current density of 0.5 mA cm−2ECSA, which was 53 and 83 mV lower than those of NiFe LDH and NixSy electrodes, respectively. By standardizing the OER kinetic current using the Rf,19 the specific activity (js) of the electrode was evaluated at 1.48 V vs. RHE. As shown in Fig. S13b,† the js of NixSy@NiFe LDH was 0.21 mA cm−2Rf, significantly higher than those of NiFe LDH (0.064 mA cm−2Rf) and NixSy (0.0047 mA cm−2Rf).
The electrochemical impedance spectroscopy (EIS) results indicate that NixSy@NiFe LDH exhibits the lowest charge-transfer resistance among the catalysts (Rct = 0.91 Ω, Table. S3†). This highlights its superior ability to facilitate electron transfer and accelerate kinetics during the OER process, which is attributed to the crystalline NixSy core (Fig. 3e). In addition, durability is another critical parameter for OER catalysts. As demonstrated in Fig. 3f, the NixSy@NiFe LDH electrocatalyst shows excellent long-term stability by retaining 96.8% performance at a current density of 200 mA cm−2 over a duration of 250 h.
Based on the results derived from the aforementioned OER and HER analyses, it can be concluded that the bifunctional NixSy@NiFe LDH catalyst demonstrates exceptional efficiency in water electrocatalysis within alkaline solutions. Subsequently, we assembled an overall water electrolysis cell based on NixSy@NiFe LDH as both the anode and the cathode. Concurrently, the cell based on the noble metal Pt/C-NF‖RuO2-NF was tested under the same conditions. As depicted in Fig. 4g, the cell voltage for NixSy@NiFe LDH at a current density of 10 mA cm−2 was measured to be 1.48 V, which is lower than that observed for the cell based on commercial Pt/C-NF‖RuO2-NF (1.59 V). A comprehensive comparison of the overall water electrolysis activity of NixSy@NiFe LDH with other reported catalysts is presented in Table S6.† Notably, NixSy@NiFe LDH surpasses most of the existing overall water electrolysis catalysts. The long-term stability of overall water electrolysis at a high current density of 50 mA cm−2 was investigated (Fig. 4h). NixSy@NiFe LDH also exhibited excellent stability without obvious decay after 120 h of stability testing. Thus, the NixSy@NiFe LDH bifunctional catalyst exhibits large ECSA, excellent conductivity, and superior stability—factors that strongly support efficient water electrolysis. At a constant current density of 20 mA cm−2, gas products generated during water electrolysis were collected utilizing a self-constructed gas collection system (Fig. 4i and S16†). Following this collection period, it was determined that the ratio of H2 to O2 produced was maintained at approximately 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. These measured values align closely with theoretical expectations, indicating Faraday efficiency approaching nearly 100%. Fig. S17† reveals that a working voltage of 1.5 V can successfully drive an overall water splitting cell assembled with NixSy@NiFe LDH, suggesting that the NixSy@NiFe LDH exhibits broad prospects in practical applications.
1. These measured values align closely with theoretical expectations, indicating Faraday efficiency approaching nearly 100%. Fig. S17† reveals that a working voltage of 1.5 V can successfully drive an overall water splitting cell assembled with NixSy@NiFe LDH, suggesting that the NixSy@NiFe LDH exhibits broad prospects in practical applications.
2.3 Further analysis of the catalytic mechanism of NixSy@NiFe LDH
To gain a deeper understanding of the reaction mechanism, the systematic characterization of the NixSy@NiFe LDH electrode after the OER was carried out using Raman spectroscopy, XRD, SEM, TEM, and XPS. As shown in Fig. 5e, following the OER test, two distinct peaks at 472 and 556 cm−1 attributed to NiOOH emerged in the Raman spectrum, indicating that the surface of NixSy and NiFe LDH was converted to NiOOH and NiFeOOH, respectively. The XRD pattern (Fig. 5f) shows a reduction in the overall intensity of the NixSy diffraction peaks, while the SEM images demonstrate that the nanowire morphology of NixSy@NiFe LDH was well preserved (Fig. S18†). The TEM image shown in Fig. 5g further confirms the morphology of the nanowires after the OER test. The SAED results show the (013), (![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 21), and (005) crystal planes of γ-NiOOH with poor crystallinity, as well as the (300) face of Ni3S2 that retains its crystal structure, which is consistent with the results of HRTEM, as shown in Fig. 5h. During the long-term stability test, the lattice fringes of Ni3S2 were still clearly visible, with a 0.16 nm lattice fringe clearly corresponding to the (300) plane of Ni3S2. Following the OER stability assessment, the STEM image and corresponding element mapping of NixSy@NiFe LDH revealed the coexistence of Fe, Ni, O, and S elements, as demonstrated in Fig. S19a.† From the Ni 2p spectrum analysis, it was observed that the Ni 2p peak (Fig. 5i) exhibits an overall blue shift, indicating an increase in the nickel oxidation state, with high-valent nickel typically being considered the active species for the OER. This corresponds to the in situ Raman results. In the O1s spectrum shown in Fig. 5j, we identified metal hydroxide as the predominant oxygen component, which increased from an initial value of 36.33% to 46.05% after prolonged testing. Furthermore, according to the ESI data (Fig. S5 and S19b†), the oxygen content increased from 40.96% to 56.72%, indicating the formation of a thin metal hydroxide film on the heterogeneous surface. As shown in Fig. S20a,† there was an overall blue shift observed for the S 2p peak along with its transformation into a higher oxidation state. However, as depicted in Fig. 1h, S5, S19b and c,† following the OER stability test, the sulfur content in the NixSy@NiFe LDH nanowire core decreased significantly, which can be attributed to the desulfurization oxidation of sulfur-containing substances and the difference in depth of penetration between XPS and EDS mapping techniques.47 Additionally, a similar blue shift was noted in the Fe 2p spectra (Fig. S20b†).
21), and (005) crystal planes of γ-NiOOH with poor crystallinity, as well as the (300) face of Ni3S2 that retains its crystal structure, which is consistent with the results of HRTEM, as shown in Fig. 5h. During the long-term stability test, the lattice fringes of Ni3S2 were still clearly visible, with a 0.16 nm lattice fringe clearly corresponding to the (300) plane of Ni3S2. Following the OER stability assessment, the STEM image and corresponding element mapping of NixSy@NiFe LDH revealed the coexistence of Fe, Ni, O, and S elements, as demonstrated in Fig. S19a.† From the Ni 2p spectrum analysis, it was observed that the Ni 2p peak (Fig. 5i) exhibits an overall blue shift, indicating an increase in the nickel oxidation state, with high-valent nickel typically being considered the active species for the OER. This corresponds to the in situ Raman results. In the O1s spectrum shown in Fig. 5j, we identified metal hydroxide as the predominant oxygen component, which increased from an initial value of 36.33% to 46.05% after prolonged testing. Furthermore, according to the ESI data (Fig. S5 and S19b†), the oxygen content increased from 40.96% to 56.72%, indicating the formation of a thin metal hydroxide film on the heterogeneous surface. As shown in Fig. S20a,† there was an overall blue shift observed for the S 2p peak along with its transformation into a higher oxidation state. However, as depicted in Fig. 1h, S5, S19b and c,† following the OER stability test, the sulfur content in the NixSy@NiFe LDH nanowire core decreased significantly, which can be attributed to the desulfurization oxidation of sulfur-containing substances and the difference in depth of penetration between XPS and EDS mapping techniques.47 Additionally, a similar blue shift was noted in the Fe 2p spectra (Fig. S20b†).
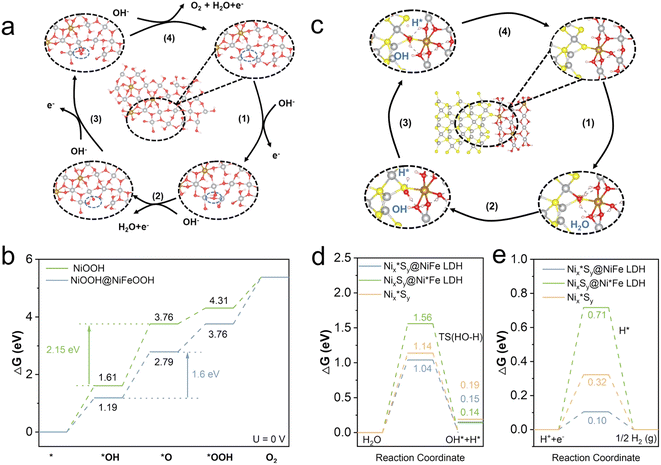
To further investigate the mechanism for the high performance of NixSy@NiFe LDH, DFT calculations were carried out separately for NiOOH and NiOOH@NiFeOOH, where the reconstructed structure was used as a computational model, as shown in Fig. S21a and b.† The alkaline OER consists of four basic steps: first, adsorption of OH− to form *OH, followed by the generation of *O and *OOH intermediates, and finally the desorption of O2, as demonstrated in Fig. 6a. The Gibbs free energies of the four steps shown in Fig. 6b suggest that the rate-determining step (RDS) for both NiOOH and NiOOH@NiFeOOH is the deprotonation process of *OH to form *O. Compared with NiOOH, the ΔG(*OH) of NiOOH@NiFeOOH is 1.6 eV, lower than that of NiOOH (2.15 eV)(Fig. S22†). The decrease of energy barriers in the RDS for NiOOH@NiFeOOH is beneficial to facilitate the desorption of OH and improve the OER catalytic activity. The above results confirmed that the reconstructed NiOOH@NiFeOOH is the real active species for the OER. The introduction of Fe into the materials promotes the structural transition and increases the degree of structural disorder. As a result, the NixSy@NiFe LDH exhibits remarkable OER catalytic activity.
Given that no redox reaction occurred during the HER process and Ni3S2 was still present after the reaction, we constructed a model similar to the Ni3S2@NiFe LDH and Ni3S2 structures (Fig. S21c and d†), and all possible active sites in both Ni3S2 and Ni3S2@NiFe LDH were screened for better explain the relationship between the activity and structure. Activity towards the HER is strongly correlated with the energy barrier of the transition state of water dissociation. Fig. 6c and d demonstrate that the energy barrier of the transition state of water dissociation on Ni3*S2@NiFe LDH is 1.04 eV, which is significantly lower than those of Ni3S2@Ni*Fe LDH (1.56 eV, Fig. S23†) and Ni3*S2 (1.14 eV, Fig. S24†). As for H adsorption, the corresponding adsorption model is shown in Fig. S25.† The calculation results showed that Ni3*S2@NiFe LDH exhibits the lowest hydrogen adsorption energy of 0.1 eV (Fig. 6e), which is much lower than the values for Ni3S2@Ni*Fe LDH (0.71 eV) and Ni3*S2 (0.32 eV). These results confirm that the active sites for water dissociation and hydrogen adsorption in Ni3S2@NiFe LDH are primarily located in the Ni3*S2@NiFe LDH rather than in the Ni3S2@Ni*Fe LDH, indicating that the interfacial interaction between NixSy and NiFe LDH endows the NixSy@NiFe LDH with excellent catalytic activity.
3. Conclusion
In summary, the NixSy@NiFe LDH bifunctional electrocatalyst, featuring a three-dimensional crystalline–amorphous core–shell structure, demonstrated excellent performance for both the OER and the HER in alkaline solutions. It exhibited impressive electrocatalytic performance, with overpotentials of 159 mV for the HER at −100 mA cm−2 and 250 mV for the OER at 200 mA cm−2, alongside outstanding long-term stability for over 250 h at 200 mA cm−2. Additionally, the electrolyzer utilizing this material as both the cathode and the anode operated at a low potential of 1.48 V at a current density of 10 mA cm−2 and retained robust performance in long-term stability tests. The exceptional bifunctional catalytic properties are attributed to its structural properties: (1) the crystalline–amorphous structure enhances the maximization of catalytically active sites and promotes efficient electron/proton transport, (2) the heterojunction interface changes interfacial charge distribution and optimizes the free energy of adsorption for reactive intermediates. As for the OER, the incorporation of iron into the structure enhances the catalytic activity by promoting structural transformation and increasing the degree of disorder of NiOOH active species. In terms of the HER, the unique interface between the NixSy core and NiFe LDH shell optimizes hydrogen adsorption, significantly improving HER activity.Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.Author contributions
Shaobo Ye: conceptualization, investigation, experimental analysis, methodology, and writing original draft. Yong Xu: conceptualization, methodology, and data curation. Xiaoyu Bai: methodology and experimental analysis. Zhao Liang: methodology. Qiao Liu: methodology. Qiliang Wei: experimental analysis. Dongjiang Yang: methodology. Weiyou Yang: conceptualization, funding, and review & editing. Fengmei Gao: conceptualization, funding, and review & editing. Qing Shi: conceptualization, funding, and review & editing.Conflicts of interest
The authors declare no conflict of interest.Acknowledgements
This work was supported by the National Natural Science Foundation of China (NSFC, Grant No. 52302144, 52372063, and 52202061), the Scientific Research Starting Foundation of Ningbo University of Technology (2022KQ39), the Natural Science Foundation of Shanxi Province (202101D121119), and the Natural Science Foundation of Hunan Province (Grant No. 2022JJ40068).References
- J. Luo, J.-H. Im, M. T. Mayer, M. Schreier, M. K. Nazeeruddin, N.-G. Park, S. D. Tilley, H. J. Fan and M. Grätzel, Science, 2014, 345, 1593–1596 Search PubMed.
- Y. Zhou, Z. Wang, Z. Pan, L. Liu, J. Xi, X. Luo and Y. Shen, Adv. Mater., 2019, 31, 1806769 Search PubMed.
- N. Logeshwaran, G. Kim, P. Thangavel, S. S. Jeon, K. Thiyagarajan, K. R. Kishore, H. Lee, I. Seo, H. Yun, S. Lee, B. H. Kim and Y. J. Lee, Adv. Sci., 2025, 12, e2413176 Search PubMed.
- P. Thangavel, M. Ha, S. Kumaraguru, A. Meena, A. N. Singh, A. M. Harzandi and K. S. Kim, Energy Environ. Sci., 2020, 13, 3447–3458 Search PubMed.
- M. Tahir, L. Pan, F. Idrees, X. Zhang, L. Wang, J.-J. Zou and Z. L. Wang, Nano Energy, 2017, 37, 136–157 Search PubMed.
- D. Y. Kim, D. S. Jeong, P. Thangavel, M. Ha, H. Hwang, Y.-J. Park, T. J. Shin, D.-H. Seo, H. S. Shin and K. S. Kim, Chem, 2023, 9, 3304–3318 Search PubMed.
- L. Yang, Z. Guo, J. Huang, Y. Xi, R. Gao, G. Su, W. Wang, L. Cao and B. Dong, Adv. Mater., 2017, 29, 1704574 Search PubMed.
- M. Li, Y. Zhu, H. Wang, C. Wang, N. Pinna and X. Lu, Adv. Energy Mater., 2019, 9, 1803185 Search PubMed.
- Z. Zheng, L. Yu, M. Gao, X. Chen, W. Zhou, C. Ma, L. Wu, J. Zhu, X. Meng, J. Hu, Y. Tu, S. Wu, J. Mao, Z. Tian and D. Deng, Nat. Commun., 2020, 11, 3315 Search PubMed.
- T. Sun, J. Wang, X. Chi, Y. Lin, Z. Chen, X. Ling, C. Qiu, Y. Xu, L. Song, W. Chen and C. Su, ACS Catal., 2018, 8, 7585–7592 Search PubMed.
- Y. Li, Z. Dong and L. Jiao, Adv. Energy Mater., 2020, 10, 1902104 Search PubMed.
- J. Tian, Q. Liu, A. M. Asiri and X. Sun, J. Am. Chem. Soc., 2014, 136, 7587–7590 Search PubMed.
- T. Liu, M. Li, C. Jiao, M. Hassan, X. Bo, M. Zhou and H.-L. Wang, J. Mater. Chem. A, 2017, 5, 9377–9390 Search PubMed.
- Z. Chen, Y. Song, J. Cai, X. Zheng, D. Han, Y. Wu, Y. Zang, S. Niu, Y. Liu and J. Zhu, Angew. Chem., Int. Ed., 2018, 57, 5076–5080 Search PubMed.
- H. Ang, H. Wang, B. Li, Y. Zong, X. Wang and Q. Yan, Small, 2016, 12, 2859–2865 Search PubMed.
- X. Zhang, H. Yi, M. Jin, Q. Lian, Y. Huang, Z. Ai, R. Huang, Z. Zuo, C. Tang, A. Amini, F. Jia, S. Song and C. Cheng, Small, 2022, 18, 2203710 Search PubMed.
- H. Xie, Y. Feng, X. He, Y. Zhu, Z. Li, H. Liu, S. Zeng, Q. Qian and G. Zhang, Small, 2023, 19, 2207425 Search PubMed.
- P. Thangavel, G. Kim and K. S. Kim, J. Mater. Chem. A, 2021, 9, 14043–14051 Search PubMed.
- P. Thangavel, H. Lee, T. H. Kong, S. Kwon, A. Tayyebi, J. h. Lee, S. M. Choi and Y. Kwon, Adv. Energy Mater., 2022, 13, 2203401 Search PubMed.
- S. Balasundari, S. Jayasubramaniyan, P. Thangavel, M. Vithiya, T. Rani, P. A. Rayjada, N. Satyanarayana and P. Muralidharan, ACS Appl. Eng. Mater., 2023, 1, 606–615 Search PubMed.
- G. Fan, F. Li, D. G. Evans and X. Duan, Chem. Soc. Rev., 2014, 43, 7040–7066 Search PubMed.
- M. Gong, Y. Li, H. Wang, Y. Liang, J. Z. Wu, J. Zhou, J. Wang, T. Regier, F. Wei and H. Dai, J. Am. Chem. Soc., 2013, 135, 8452–8455 Search PubMed.
- L. Tan, J. Yu, C. Wang, H. Wang, X. Liu, H. Gao, L. Xin, D. Liu, W. Hou and T. Zhan, Adv. Funct. Mater., 2022, 32, 2200951 Search PubMed.
- Y. Yang, Y. Xie, Z. Yu, S. Guo, M. Yuan, H. Yao, Z. Liang, Y. R. Lu, T.-S. Chan, C. Li, H. Dong and S. Ma, Chem. Eng. J., 2021, 419, 129512 Search PubMed.
- H. Guo, L. Zhang, D. Ou, Q. Liu, Z. Wu, W. Yang, Z. Fang and Q. Shi, Small, 2023, 12, e2307069 Search PubMed.
- S. Zhou, H. He, J. Li, Z. Ye, Z. Liu, J. Shi, Y. Hu and W. Cai, Adv. Funct. Mater., 2023, 34, 2313700 Search PubMed.
- L. Li, H. Sun, X. Xu, M. Humayun, X. Ao, M. F. Yuen, X. Xue, Y. Wu, Y. Yang and C. Wang, ACS Appl. Mater. Interfaces, 2022, 14, 50783–50793 Search PubMed.
- Y. Wang, X. Li, Z. Huang, H. Wang, Z. Chen, J. Zhang, X. Zheng, Y. Deng and W. Hu, Angew Chem. Int. Ed. Engl., 2023, 62, e202215256 Search PubMed.
- J. Lv, L. Wang, R. Li, K. Zhang, D. Zhao, Y. Li, X. Li, X. Huang and G. Wang, ACS Catal., 2021, 11, 14338–14351 Search PubMed.
- B. He, P. Zhao, G.-X. Pan, Q. Lu, H.-Q. Li, F. Ye, Y.-W. Tang, Q.-L. Hao and Z. Su, J. Alloys Compd., 2023, 938, 168673 Search PubMed.
- L. Zhang, X. Gao, Y. Zhu, A. Liu, H. Dong, D. Wu, Z. Han, W. Wang, Y. Fang, J. Zhang, Z. Kou, B. Qian and T. T. Wang, Nanoscale, 2021, 13, 2456–2464 Search PubMed.
- L. Peng, C. Wang, Q. Wang, R. Shi, T. Zhang and G. I. N. Waterhouse, Adv. Energy Sustainability Res., 2021, 2, 2100078 Search PubMed.
- L. Yu, H. Zhou, J. Sun, F. Qin, F. Yu, J. Bao, Y. Yu, S. Chen and Z. Ren, Energy Environ. Sci., 2017, 10, 1820–1827 Search PubMed.
- J. Zhou, L. Yu, Q. Zhu, C. Huang and Y. Yu, J. Mater. Chem. A, 2019, 7, 18118–18125 Search PubMed.
- Y. Wang, Y. Zhang, Z. Liu, C. Xie, S. Feng, D. Liu, M. Shao and S. Wang, Angew Chem. Int. Ed. Engl., 2017, 56, 5867–5871 Search PubMed.
- K. Zhang, Y. Duan, N. Graham and W. Yu, Appl. Catal., B, 2023, 323, 122144 Search PubMed.
- Y. Sang, G. Ding, Z. Guo, Y. Xue, G. Li and R. Zhang, J. Alloys Compd., 2022, 919, 165855 Search PubMed.
- Y. Li, J. Liu, S. Li and S. Peng, ACS Catal., 2024, 14, 4807–4819 Search PubMed.
- W. Zhong, W. Li, C. Yang, J. Wu, R. Zhao, M. Idrees, H. Xiang, Q. Zhang and X. Li, J. Energy Chem., 2021, 61, 236–242 Search PubMed.
- X. Wang, H. Song, S. Ma, M. Li, G. He, M. Xie and X. Guo, Chem. Eng. J., 2022, 432, 134319 Search PubMed.
- D. Li, W. Wan, Z. Wang, H. Wu, S. Wu, T. Jiang, G. Cai, C. Jiang and F. Ren, Adv. Energy Mater., 2022, 12, 2201913 Search PubMed.
- J. T. Ren, L. Chen, W. W. Tian, X. L. Song, Q. H. Kong, H. Y. Wang and Z. Y. Yuan, Small, 2023, 19, e2300194 Search PubMed.
- M. Chen, Y. Zhang, J. Chen, R. Wang, B. Zhang, B. Song and P. Xu, Small, 2023, 2309371 Search PubMed.
- X. Li, G. Q. Han, Y. R. Liu, B. Dong, W. H. Hu, X. Shang, Y. M. Chai and C. G. Liu, ACS Appl. Mater. Interfaces, 2016, 8, 20057–20066 Search PubMed.
- J. Zhang, Y. Fang, Y. Chen, X. Zhang, H. Xiao, M. Zhao, C. Zhao, X. Ma, T. Hu, E. Luo, J. Jia and H. Wu, J. Colloid Interface Sci., 2024, 653, 821–832 Search PubMed.
- S. Lee, L. Bai and X. Hu, Angew. Chem., Int. Ed., 2020, 59, 8072–8077 Search PubMed.
- Y. Wu, Y. Li, M. Yuan, H. Hao, X. San, Z. Lv, L. Xu and B. Wei, Chem. Eng. J., 2022, 427, 131944 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5ta00601e |
| This journal is © The Royal Society of Chemistry 2025 |