Synthesis of dynamic N-acylhydrazone-based macrocycles†
Nicoleta
Sandu‡
a,
Anamaria
Hanganu‡
ab,
Codruţa
Popescu‡
a,
Alexandra M.
Demeter
c,
Anca G.
Mirea
d,
Andrei
Kuncser
d,
Cristina
Tăblet
e,
Niculina D.
Hădade
 f,
Mihaela
Florea
dg,
Daniel P.
Funeriu
*a and
Mihaela
Matache
f,
Mihaela
Florea
dg,
Daniel P.
Funeriu
*a and
Mihaela
Matache
 *a
*a
aUniversity of Bucharest, Faculty of Chemistry, Research Centre of Applied Organic Chemistry, 90 Panduri Street, Bucharest, RO-050663, Romania. E-mail: mihaela.matache@g.unibuc.ro
b“C. D. Nenitzescu” Institute of Organic and Supramolecular Chemistry of the Romanian Academy202B Spl, Independenţei, Bucharest, 060023, Romania
cSC Microsin SRL, 51-63 Pericle Papahagi Street, Bucharest, 032364, Romania
dNational Institute of Material Physics, 405A Atomistilor Street, Magurele, 077125, Romania
eTitu Maiorescu University, Faculty of Pharmacy, Gh. Sincai Bd. 16, Bucharest, 040317, Romania
fFaculty of Chemistry and Chemical Engineering, Supramolecular Organic and Organometallic Chemistry Centre, “Babes-Bolyai” University, 11 Arany Janos Str., Cluj-Napoca, 400028, Romania
gUniversity of Bucharest, International School of Doctoral Studies, 90 Panduri Street, Bucharest, RO-050663, Romania
First published on 24th February 2025
Abstract
We present herein the synthesis of novel [2 + 2] and [3 + 3] N-acylhydrazone-based macrocycles using a pool of dialdehydes and dihydrazides, under thermodynamic control. The resulting macrocycles, which were assigned triangular and rectangular shapes, were characterised in the solid state. The rectangular macrocycle was further investigated in solution by absorption and emission spectroscopy. Additionally, the rectangular macrocycle was used in various assays to investigate the hosting capacity towards various guests using NMR, HRMS, UV-visible and fluorescence spectroscopy. Theoretical calculations regarding structure and properties of the novel compounds were in agreement with the experimental details. The experimental data showed intriguing results toward tetra-n-butylammonium fluoride.
Introduction
N-Acylhydrazones (Fig. 1) are versatile molecules that have attracted growing interest in the search for biologically active molecules,1 effective photoswitches under light irradiation2 or usefulness for convenient synthesis of heterocycles, such as 1,3,4-oxadiazoles.3 An increase in the number of hydrazone units provides an increase of the application range; for example, hydroxy-bis-N-acylhydrazones (i.e., structure I, Fig. 1) have been reported as ligands for metal ions,4 gelators5 or switchable light-emissive molecules.6 Furthermore, thanks to the reversibility of the resulting hydrazone bonds, reaction between polyaldehydes and polyhydrazides has been increasingly encountered. During the past few decades, we have witnessed a wide range of polyfunctionalized building blocks being used, under dynamic covalent chemistry (DCC) conditions, for the preparation of efficient macrocyclic hosts for various organic molecules (i.e., structure II, Fig. 1).7 Dynamic polymers (often called dynamers)8 or crystalline polymers (often called covalent organic frameworks, COFs)9 have also been developed using the N-acylhydrazone bond. Unlike hydrazones, imines were more intensively screened to find macrocycles as receptors for metal ions (i.e. structure III, Fig. 1) or small-molecules7 and, to a lesser extent, for anions.10During our work6 involving N-acylhydrazones, we recognized dialdehydes 1 (Scheme 1) as interesting building blocks and we used these as starting materials for preparation of the corresponding hydrazides 2a and 2b (Scheme 1). A literature survey showed that imine-based macrocycles obtained starting from similar phenol-derived aldehyde and amino building blocks were not able to form a cyclic cavity under thermodynamic control.11 However, tetranuclear Ni(II) complexes12 have been reported to occur from aldehydes of type 1, and the corresponding aminomethyl phenols when the metal ions were used as templates (often called Robson-type complexes). Dialdehydes of type 1 were also used as precursors13 in preparation of imine macrocycles with a wide variety of amines, resulting in compartmental [1 + 1] and [2 + 2] macrocycles and the corresponding polynuclear metal-ion complexes.
Unlike imines, a major drawback in the use of N-acylhydrazones is their poor solubility in water and most organic solvents. This can sometimes be overcome by grafting solubilising functional groups such as hydroxyl. These groups may act as chelating sites for metal ions and, more importantly, enhance the solubility of the hydrazones, concomitantly affording interplay between discrete molecules and polymeric structures, by control of the reaction conditions.14
In this context, we report herein our findings regarding reactions of a pool of dialdehydes 1 and dihydrazides 2 (Scheme 1), under thermodynamic control, with scaling up and isolation of novel N-acylhydrazone-based macrocycles. We further performed experiments to investigate the hosting capacity of the resulting macrocycles using NMR, HRMS, UV-visible and fluorescence spectroscopy. We also performed theoretical calculations regarding structures and properties of the novel compounds which were in agreement with the experimental details.
Results and discussion
Synthesis of the precursors
The precursor aldehydes were synthesized by MnO2-mediated oxidation of commercial 2,6-bis(hydroxymethyl)-p-cresol12 and Duff reaction of p-tert-butylphenol with urotropine and trifluoroacetic acid.15 Aldehyde 1a was used to prepare aldehyde 1c, by acetylation with acetyl chloride. Aldehydes 1a and 1b constituted starting materials (see ESI, Scheme S1†) for preparation of hydrazides 2a and 2b, through oxidation, esterification and subsequent reaction with hydrazine hydrate (Scheme 1). The oxidation reagent for transformation of aldehydes into carboxylic acid was silver oxide,16 which was very well tolerated by the phenol group, providing the acids in excellent yields. Target hydrazides 2a and 2b were obtained in very good to excellent yields, although purification was rather tedious involving acid–base treatments with careful control of pH, due to the presence of multiple acidic and basic functional groups.Macrocyclisation experiments
Once we had synthesized the dialdehydes 1 and dihydrazides 2, we performed our initial experimental screenings, using NMR as monitoring tool. Formation of macrocycles under thermodynamic control in competition with polymerization is favoured by dilution.17 In addition, previous reports using N-acylhydrazones indicate that the choice of reaction conditions is crucial for directing the product formations toward a macrocycle or a dynamic polymer and even switching between the two macrocyclic and polymeric states.14,18We performed reactions between dialdehydes 1 and dihydrazides 2 in the NMR tube, in DMSO-d6, and formation of the products was monitored by recording the 1H NMR spectra in time. Our preliminary experiments focused on finding the appropriate working concentration so that all members of the reaction mixture could be dissolved at equilibrium. For example, 25 mM stock solutions of 1b and 2b in DMSO-d6 were used to assay final concentrations between 1 mM and 10 mM of the reactants, yielding the best results for 5 mM and 7.5 mM. We further decided to use 5 mM final concentrations of the precursors. Spectra of reaction between 1b and 2b in stoichiometric ratio indicated, after approximately 2 hours, the formation of a symmetrical major species (compound 6, Fig. 2A). More accurately, in Fig. 2A, notable changes could be observed between the 1H-NMR spectra recorded initially and after 30 minutes, when the proton signals corresponding to the initial aldehyde (δ = 10.26 ppm, respectively δ = 8.09 ppm) disappeared. However, small changes in the NMR spectra still occurred between 30 minutes and 120 minutes, while no more changes were observed after 120 minutes, suggesting that equilibrium was completely reached. Use of catalytic amounts of TFA (trifluoroacetic acid) did not yield significant time decrease for formation of the major species. Similar spectral profiles were also obtained for mixtures of 1a and 2a in stoichiometric amounts (see ESI, Fig. S1†). We further focused on reaction between 1b and 2b to establish the nature of the resulting major species. Previous work involving dialdehyde 1b in reaction with amines reported a [3 + 3] macrocycle useful to bind Zn2+ metal ions in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 complex stoichiometry.19 ESI(−)-HRMS analysis (Fig. 2B) indicated a [3 + 3] structure of the resulting compound 6, as the spectrum revealed two peaks at m/z = 1307.6225 and m/z = 653.3076 which we assigned to [M − H]− and [M − 2H]2−. To elucidate the macrocyclic shape, we recorded 2D NMR (NOESY, Fig. 2C) indicating a triangular shape, according to the space interactions of protons within the molecule. The spectrum revealed NOE connectivity between
3 complex stoichiometry.19 ESI(−)-HRMS analysis (Fig. 2B) indicated a [3 + 3] structure of the resulting compound 6, as the spectrum revealed two peaks at m/z = 1307.6225 and m/z = 653.3076 which we assigned to [M − H]− and [M − 2H]2−. To elucidate the macrocyclic shape, we recorded 2D NMR (NOESY, Fig. 2C) indicating a triangular shape, according to the space interactions of protons within the molecule. The spectrum revealed NOE connectivity between 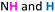 (hydrazide moieties) protons, as well as
(hydrazide moieties) protons, as well as 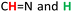 (aldehyde moieties) protons. This was also supported by theoretical calculations. The optimized geometry is shown in ESI (Fig. S7†).
(aldehyde moieties) protons. This was also supported by theoretical calculations. The optimized geometry is shown in ESI (Fig. S7†).
Similar experiments were performed for all possible stoichiometric combinations between dialdehydes 1 and dihydrazides 2 shown in Scheme 1: 1b + 2c, 1c + 2a, 3 + 2c, 3 + 2b (see ESI, Fig. S4–S6†). Interestingly, among all these possible combinations, the macrocycle resulting from reaction of 1b and 2c (Fig. 3) yielded the most promising results, as the single combination that afforded one major species at equilibrium. Thus, we performed the same set of NMR experiments to set the optimum concentration of the monomers and the time required to reach thermodynamic equilibrium, without any acid catalyst. We found two optimal concentrations, namely 7.5 mM and 10 mM. As shown in Fig. 3, a major species was formed over an extended period (days), while addition of catalytic TFA significantly shortened the equilibration time to 15 minutes (see ESI, Fig. S2†). ESI(+)-HRMS spectra indicated a [2 + 2] macrocycle, while the 2D-NMR spectrum (DMSO-d6, 500 MHz) confirmed a rectangular shape for compound 7 by NOE connectivity between  (hydrazide moieties),
(hydrazide moieties), 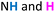 (hydrazide moieties),
(hydrazide moieties), 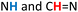 as well as
as well as 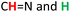 (aldehyde moieties) protons. The DFT calculations are in in agreement with the experimental results and the optimized geometry resulting from DFT calculation is shown in ESI (Fig. S7†).
(aldehyde moieties) protons. The DFT calculations are in in agreement with the experimental results and the optimized geometry resulting from DFT calculation is shown in ESI (Fig. S7†).
Although we were unable to obtain a suitable single crystal, the NMR and HRMS analyses provide strong evidence supporting the proposed structures.
Solid-state analysis
Upscaling and isolation of 6 and 7 allowed us to perform solid-state analysis of the resulting compounds. FTIR, thermal analysis, powder X-ray diffraction (PXRD), scanning electron microscopy (SEM), and transmission electron microscopy (TEM) results are available in ESI (Fig. S11–S14†). Briefly, FTIR spectra of 6 and 7 confirmed the presence of the hydrazone linkages,20 while thermal analysis profiles indicated a higher stability for compound 6.SEM images of compound 7 show a layered morphology, while macrocycle 6 has a texture that is much more compact, consistent with the thermogravimetric analysis indicating higher stability (ESI, Fig. S13A†). Moreover, TEM images of 7 revealed once again the layered morphology of this compound (ESI, Fig. S13B†). PXRD of 6 and 7 (ESI, Fig. S14†) indicated a higher crystallinity for compound 6, while the interlayer d-spacings of 3.4 Å (for 6) and 3.5 Å (for 7) were assigned to the distance of π–π stacking in agreement with the DFT calculations and literature data.16,21,22
Optical properties and hosting capacity
Compound 7 was synthesized in larger amounts, providing reproducible structural analysis data with the product obtained in the NMR tube (see ESI, Fig. S3†). It is a yellow-orange compound that showed solubility in DMSO, and we characterised it by UV-visible spectroscopy (5 μM): it exhibited two absorption maxima at λmax = 310 nm and λmax = 371 nm, corresponding to π–π* transitions in the aromatic rings. We conducted experiments for the determination of the molar extinction coefficient (see ESI, Fig. S15†) and we obtained a value of ε = 39![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 581 L mol−1 cm−1 for λmax = 366 nm. The calculated absorption spectrum of compound 7 in DMSO is shown in ESI (Fig. S16†), indicating a very good agreement with the experimental spectrum. The long-wavelength absorption band was assigned mainly due to the HOMO-1 → LUMO transition. This is a π → π* transition with a significant charge transfer characteristic from p-tert-butylphenol to the N-acylhydrazone fragment. The HOMO has a small contribution from phenolic –OH, so we expect this band to be affected by the removal of the proton. Upon addition of TBAF to a solution of compound 7 in DMSO, a new band was visible in the experimental spectrum at around λ = 450 nm. Supported by DFT calculation (ESI, Fig. S9†), we assigned this band to the formation of the corresponding monoanion species. When the PBE0 functional was used, the computed spectrum of the monoanion was red-shifted compared to the experiment, whereas when the CAM-B3LYP functional was used, the spectrum was blue-shifted. However, both functionals indicated that the monoanion had a longer absorption wavelength than the neutral species.
581 L mol−1 cm−1 for λmax = 366 nm. The calculated absorption spectrum of compound 7 in DMSO is shown in ESI (Fig. S16†), indicating a very good agreement with the experimental spectrum. The long-wavelength absorption band was assigned mainly due to the HOMO-1 → LUMO transition. This is a π → π* transition with a significant charge transfer characteristic from p-tert-butylphenol to the N-acylhydrazone fragment. The HOMO has a small contribution from phenolic –OH, so we expect this band to be affected by the removal of the proton. Upon addition of TBAF to a solution of compound 7 in DMSO, a new band was visible in the experimental spectrum at around λ = 450 nm. Supported by DFT calculation (ESI, Fig. S9†), we assigned this band to the formation of the corresponding monoanion species. When the PBE0 functional was used, the computed spectrum of the monoanion was red-shifted compared to the experiment, whereas when the CAM-B3LYP functional was used, the spectrum was blue-shifted. However, both functionals indicated that the monoanion had a longer absorption wavelength than the neutral species.
We have also noted naked-eye emission of yellow-orange light for DMSO solutions of compound 7 under a UV lamp (λexc = 365 nm) and we conducted fluorescence experiments (Fig. 4). The emission maxima depended on the excitation wavelength and we rationalized that the structure of the N-acylhydrazone permits the occurrence of ESIPT (excited-state intramolecular proton transfer),6 as evidenced also by superposition of the UV-visible spectra and excitation spectra (see ESI, Fig. S16†). The excitation and emission spectra displayed maxima at λexc = 480 nm and λem = 540 nm, respectively.
 | ||
| Fig. 4 Emission spectra for titration of 7 with TBAF (DMSO) and plot of the emission intensity vs. no. of equivalents of TBAF. | ||
Compound 7 was further assayed for hosting ability23 using salts of tetra-n-butylammonium fluoride (TBAF), chloride (TBAC), bromide (TBAB), and hydrogen sulphate (TBAS). Mixtures of 7 with the salts and recording of the UV-visible spectra indicated a response only in the case of the fluoride (see ESI, Fig. S17 and S18†) in terms of a decrease of the band at λmax = 371 nm and increase of a new band at λmax = 445 nm. We further performed titration experiments of macrocycle 7 (final concentration 10−5 M) with increasing amounts of TBAF solution in DMSO in order to find the stoichiometry of the host–guest system. We used BindFit24 to fit the spectroscopic data and we obtained a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry (see ESI, Fig. S18†).
1 stoichiometry (see ESI, Fig. S18†).
We also followed using NMR the interaction between 7 and TBAF and TBAB, which indicated a response only in the case of the fluoride (see ESI, Fig. S22† for NMR spectra for compound 7 and TBAB). Stock solutions in DMSO-d6 of 7 (5 mM) and TBAF (50 mM) were used to perform titration. Addition of increasing amounts of TBAF from 0.1 eq. to 2 eq. and recording of the spectra of the resulting mixtures led to the results shown in Fig. 5 (see also ESI, Fig. S20 and S21†). We could observe several modifications: disappearance of the phenol and amide signals, shielding of the signals corresponding to the aromatic protons in the structure of 7 (Δδ for Hald between 6 and 72 Hz; Δδ for Hhidr between 2 and 24 Hz), as well as the signal corresponding to the imine group (Δδ for C![[H with combining macron]](https://www.rsc.org/images/entities/char_0048_0304.gif) N between δ = 2 and δ = 35 Hz). Thus, the NMR analysis suggested formation of a host–guest complex for the fluoride-containing compound. Intriguingly, we could also observe changes in the signals corresponding to the protons in the vicinity of the nitrogen atom in TBAF (Δδ for H2C–N between δ = 1 Hz and δ = 12 Hz), which has been previously observed as ion-pair complexes (i.e. TBACl, TBAHF2) with amide-based macrocyclic hosts and the presence of multiple species in solution. These data might indicate an initial deprotonation of 7 and further formation of an ion-pair complex of 7 with TBA (1
N between δ = 2 and δ = 35 Hz). Thus, the NMR analysis suggested formation of a host–guest complex for the fluoride-containing compound. Intriguingly, we could also observe changes in the signals corresponding to the protons in the vicinity of the nitrogen atom in TBAF (Δδ for H2C–N between δ = 1 Hz and δ = 12 Hz), which has been previously observed as ion-pair complexes (i.e. TBACl, TBAHF2) with amide-based macrocyclic hosts and the presence of multiple species in solution. These data might indicate an initial deprotonation of 7 and further formation of an ion-pair complex of 7 with TBA (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1).25 Use of Bindfit24 for the data obtained from the NMR studies also confirmed 1
1).25 Use of Bindfit24 for the data obtained from the NMR studies also confirmed 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry (see ESI, Fig. S24†).
1 stoichiometry (see ESI, Fig. S24†).
During our studies, we noticed an enhancement of the fluorescence when 7 was mixed with TBAF, and we used fluorescence spectroscopy to study their interaction. Thus, a stock solution of compound 7 in DMSO (5 mM) was used for titration against TBAF (50 mM) up to 2.5 equivalents (Fig. 4). We found a red-shift of the emission maximum from λem = 528 nm to λem = 565 nm, concomitant with an enhancement in the emission intensity. Fig. 4 shows a plot of the emission intensity against number of TBAF equivalents. The resulting experimental data were rather intriguing (see also ESI, Fig. S19†) and one probable explanation could be the presence of multiple species that contribute to the emission. One experimental observation to support this is that the excitation spectra of 7 and mixtures of 7 and TBAF are red-shifted compared to the corresponding absorption spectra. Therefore, 7 could exist as a dianion in the excited state in the presence of TBAF. The absorption spectrum of the dianion calculated with DFT confirms these findings (ESI, Fig. S10†). Moreover, previous studies on fluoride interactions with N-acylhydrazone in COFs10c report the formation of negative nitrogen from the amide bond which causes emission of light. The interaction of 7 with TBAF may either lead to deprotonation of the hydroxyl and amide10c groups or cause hydrogen bonding between the fluoride and the two groups.25 We performed some testing experiments in NMR on model N-acylhydrazones similar to our systems to assign the structural changes that are contributing to the emissions upon addition of TBAF. We performed 1H NMR experiments with the N-acylhydrazones in the presence of TBAF and tBuOK. In both cases, we could assign the disappearance of both signals corresponding to the NH and OH groups to complete deprotonation of the hydroxyl and amide groups (see ESI, Fig. S25 and S26†). Thus, deprotonation occurs most probably also in the case of macrocycle 7 and several species existing in solution10c could contribute to even more species in the excited state.
Finally, to get a further insight into these results, we also performed HRMS experiments of compound 7 treated with TBAF. Although we could observe the molecular ion peak in positive ionization (Fig. 3B), the experiments involving tetra-n-butylammonium salts could not be performed similarly, but rather in negative mode.
Fig. 6A shows the high-resolution mass spectrum of compound 7 which resulted from mixing for 72 hours equal amounts of 1b and 2c and further isolated. We occasionally observed that heating a freshly prepared solution of compound 7 in DMSO may lead to new signals in the mass spectrum recorded immediately after, suggesting the existence of more species (see ESI, Fig. S30†). Additions of TBAF to such a solution produced the spectra shown in Fig. 6 that were recorded at different time intervals after initial mixing. The spectrum recorded after two days displayed several signals that at first sight could be assigned to association of molecules in negative mode. However, signals at m/z = 1091.4546 or m/z = 1820.7698 are indicative of formation of larger macrocycles like [3 + 3] and [5 + 5], respectively (Fig. 6B), which are very unlikely to be treated as instrumental artefacts. Moreover, the mass spectrum recorded after five days brings new notable signals: that at m/z = 727.3010 has a different isotopic distribution, which might correspond to [2 + 2], [4 + 4] and [6 + 6] macrocycles. This is supported also by the signal at m/z = 1091.4559, assigned to double-charged [6 + 6]. Recording the sample after 7 days showed almost no change in the spectrum profile.
These results prompted us to perform more experiments in order to understand the behavior of the compound. Firstly, we mixed aldehyde 1b and hydrazide 2c in stoichiometric amounts in the absence and presence of TBAF (1 eq), left the mixture heating for 3 days, then isolated the products by addition of water and filtration of the resulting solids. Negative ESI mass spectra of these are shown in Fig. 7. For the reaction performed in the absence of TBAF, the spectrum showed a clear signal at m/z = 727.3079 corresponding to monocharged [2 + 2] macrocycle, while the reaction performed in the presence of TBAF resulted in a mixture of [2 + 2], [3 + 3], and [4 + 4] species.
We also performed experiments with other ammonium salts. While mixing macrocycle 7 with TBAC resulted in a mixture of species (see ESI, Fig. S31†), the mass spectrum of the solution containing macrocycle 7 and TBAB (Fig. 7, bottom) suggested association of the resulting species with a bromide anion. For example, the spectrum showed signals at m/z = 807.2252, m/z = 1171.3763, m/z = 1537.5303, which could be assigned to monocharged complexes of [2 + 2], [3 + 3] and [4 + 4] with the bromide anion (see also ESI, Fig. S32†).
Detailed analysis of all the HRMS experiments revealed that macrocycle 7 could be formed exclusively in the presence or absence of TFA, while use of TBAF led to a mixture of species. However, the addition of TBAF to a solution of the isolated macrocycle 7 led to a mixture of species that evolved in time toward formation of a major species, most likely a [6 + 6] macrocycle. These results could explain the difficulties we encountered in fitting data for the complexation experiments performed using UV-visible, emission and NMR spectroscopies, because of the existence in the solution of several species.
Experimental
General information
All reagents and solvents were purchased from commercial suppliers and used without further purification. Thin-layer chromatography (TLC) was performed on silica gel-coated aluminium Merck Silica 60 F254 plates with fluorescent indicator. All plates were visualized by UV irradiation at 254 nm. The aldehydes were also visualized with DNFH (2,4-dinitrophenylhydrazine). The chromatography columns used for the purification of the compounds were hand-packed in the lab using the same type of silica gel. Dichloromethane (DCM), methanol (MeOH), petroleum ether (PE) and ethyl acetate (AcOEt) were used as solvents, PE and AcOEt for column chromatography, methanol (MeOH) and water (H2O) for recrystallisation. Proton and carbon nuclear magnetic resonance (NMR) spectra were recorded on Bruker FOURIER (300 MHz) and Bruker Avance III (500 MHz) spectrometers using deuterated solvents: dimethyl sulfoxide-d6 (degree of deuteration: 99.8%) and chloroform-d3. Chemical shifts (δ) are reported in parts per million from internal reference peak values of residual solvent. The signals of the compounds studied had multiplicity equal to 1 and are abbreviated with s for singlet. High-resolution mass spectra were recorded with a Thermo Scientific (LTQ XL Orbitrap) spectrometer using the ESI technique in positive and negative ion modes. The samples were introduced into the spectrometer by direct infusion. Ionization conditions were for ESI(−): spray voltage, 3.23 kV; gas flows (sheath, auxiliary, sweep), 15, 5, 5 arbitrary units; capillary temperature, 250 °C; capillary voltage, −36 V; tube lens voltage, −164 V. For ESI(+): spray voltage, 5 kV; gas flows, 15, 5, 5 arbitrary units; capillary temperature, 275 °C; capillary voltage, 34 V; tube lens voltage, 207 V. Microscans were set to three for all ESI(−)-HRMS and ESI(+)-HRMS experiments.Melting points were determined in open capillary tubes using a STUART SMP3 electric melting point apparatus and were uncorrected. Absorption spectra were recorded with a Jasco V-630 spectrophotometer using a 10 mm quartz cell. Stock solutions were prepared in DMSO (5 × 10−5 mol L−1) and diluted to 5 × 10−6 mol L−1 using DMSO. Fluorescence spectra were recorded with a multimode spectral scanning reader (Thermo Scientific Varioskan Flash). The spectra were recorded in proper plates using 5 nm excitation and emission slits for all measurements. Stock solution was prepared in DMSO (5 × 10−3 mol L−1), and diluted to 5 × 10−4 mol L−1 using DMSO.
Powder X-ray diffraction (XRD) was performed using a D8 ADVANCE diffractometer (Bruker-57AXS GmbH, Germany) with Ni-filtered Cu radiation (λ = 1.54184 Å), at room temperature, in Bragg–Brentano geometry, in the 2θ range from 5 to 60° at a scanning speed of 0.02° min−1. Thermogravimetric analysis of macrocycles was carried out with a Themys One 1150 TGA in nitrogen atmosphere. The analysis was performed in Pt crucibles with heating from 30 °C up to 500 °C at a rate of 10 °C min−1. The morphology was investigated using a Zeiss Evo 50 XVP scanning electron microscope (SEM). The operational parameters used for the SEM images were: accelerating voltage of 20 kV, measuring time of 300 s, and working distance of around 14 mm. The morphology was also studied with a JEOL2100 transmission electron microscope (TEM). The presence of functional groups was investigated by Fourier transform infrared (FTIR) spectroscopy using a PerkinElmer spectrometer in attenuated total reflectance (ATR) mode with a diamond crystal plate (Pike Technologies, Madison, WI) device. The spectra were recorded in the spectral range of 500–4000 cm−1, at a nominal resolution of 4 cm−1 and 32 scans per acquisition.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH = 4
MeOH = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). 1H NMR (500 MHz, DMSO-d6): δ = 7.76 (s, 2H, Haromatic); 2.25 (s, 3H, CH3) ppm. 13C NMR (125 MHz, DMSO-d6): δ = 169.1; 160.0; 135.7; 126.4; 116.9; 19.6 ppm.
1). 1H NMR (500 MHz, DMSO-d6): δ = 7.76 (s, 2H, Haromatic); 2.25 (s, 3H, CH3) ppm. 13C NMR (125 MHz, DMSO-d6): δ = 169.1; 160.0; 135.7; 126.4; 116.9; 19.6 ppm.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH = 4
MeOH = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). 1H NMR (500 MHz, DMSO-d6): δ = 7.94 (s, 2H, Haromatic); 1.26 (s, 9H, t-Bu) ppm. 13C NMR (125 MHz, DMSO-d6): δ = 169.0; 163.3; 137.8; 132.0; 116.7; 33.6; 31.1 ppm.
1). 1H NMR (500 MHz, DMSO-d6): δ = 7.94 (s, 2H, Haromatic); 1.26 (s, 9H, t-Bu) ppm. 13C NMR (125 MHz, DMSO-d6): δ = 169.0; 163.3; 137.8; 132.0; 116.7; 33.6; 31.1 ppm.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PE = 1
PE = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4). 1H NMR (500 MHz, CDCl3): δ = 11.64 (s, 1H, OH); 8.07 (s, 2H, Haromatic); 3.96 (s, 6H, CH3O); 1.32 (s, 9H, t-Bu) ppm. 13C NMR (125 MHz, CDCl3): δ = 168.4; 159.4; 141.3; 133.3; 115.9; 52.4; 34.2; 31.2 ppm.
4). 1H NMR (500 MHz, CDCl3): δ = 11.64 (s, 1H, OH); 8.07 (s, 2H, Haromatic); 3.96 (s, 6H, CH3O); 1.32 (s, 9H, t-Bu) ppm. 13C NMR (125 MHz, CDCl3): δ = 168.4; 159.4; 141.3; 133.3; 115.9; 52.4; 34.2; 31.2 ppm.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DCM = 1
DCM = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5).1H NMR (500 MHz, DMSO-d6): δ = 9.92 (bs, 2H, NH); 7.78 (s, 2H, Haromatic); 5.04 (bs, 4H, NH2); 2.24 (s, 3H, Me) ppm. 13C NMR (125 MHz, DMSO-d6): δ = 166.2; 157.7; 132.3; 126.1; 117.4; 20.0 ppm.
5).1H NMR (500 MHz, DMSO-d6): δ = 9.92 (bs, 2H, NH); 7.78 (s, 2H, Haromatic); 5.04 (bs, 4H, NH2); 2.24 (s, 3H, Me) ppm. 13C NMR (125 MHz, DMSO-d6): δ = 166.2; 157.7; 132.3; 126.1; 117.4; 20.0 ppm.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DCM = 1
DCM = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4). 1H NMR (500 MHz, DMSO-d6): δ = 10.07 (bs, 2H, NH); 7.96 (s, 2H, Haromatic); 5.01 (bs, 4H, NH2); 1.28 (s, 9H, t-Bu) ppm. 13C NMR (125 MHz, DMSO-d6): δ = 166.4; 156.9; 140.3; 128.6; 117.0; 34.1; 31.1 ppm.
4). 1H NMR (500 MHz, DMSO-d6): δ = 10.07 (bs, 2H, NH); 7.96 (s, 2H, Haromatic); 5.01 (bs, 4H, NH2); 1.28 (s, 9H, t-Bu) ppm. 13C NMR (125 MHz, DMSO-d6): δ = 166.4; 156.9; 140.3; 128.6; 117.0; 34.1; 31.1 ppm.
Compound 6
1H NMR (500 MHz, DMSO-d6): δ = 12.36 (s, 6H, NH); 12.18 (s, 6H, OH); 8.81 (s, 6H, CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N); 8.06 (s, 6H, Haromatic aldehyde); 7.81 (s, 6H, Haromatic hydrazide); 1.37 (s, 27H, t-Bualdehyde); 1.35 (s, 27H, t-Buhydrazide) ppm. HRMS (ESI, −), m/z: calculated for C72H83N12O12 [M − H]−: 1307.6248; found: 1307.6225 [M − H]−, 653.3076 [M − 2H]2−.
N); 8.06 (s, 6H, Haromatic aldehyde); 7.81 (s, 6H, Haromatic hydrazide); 1.37 (s, 27H, t-Bualdehyde); 1.35 (s, 27H, t-Buhydrazide) ppm. HRMS (ESI, −), m/z: calculated for C72H83N12O12 [M − H]−: 1307.6248; found: 1307.6225 [M − H]−, 653.3076 [M − 2H]2−.
Compound 7
Compound 1b (300 mg, 10 mmol, 1 eq.) and 125 mL DMSO were added to a round bottom flask (flask 1). In another round bottom flask (flask 2) compound 3 (282.5 mg, 10 mmol, 1 eq.) and 36 mL DMSO were added. After compounds 1b and 3 had completely dissolved, the solution from flask 2 was poured over the solution in flask 1. The mixture was heated at 60 °C for 3 days. After the reaction was finished, 50 mL H2O was added. The resulting yellow precipitate was isolated through filtration under reduced pressure, rinsed with H2O and ethylic ether. A pure compound was obtained. 1H NMR (500 MHz, DMSO-d6): δ = 12.41 (s, 2H, OH); 12.31 (s, 4H, NH); 8.82 (s, 4H, CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N); 8.12 (s, 8H, Haromatic hydrazide); 7.80 (s, 4H, Haromatic aldehyde); 1.35 (s, 18H, t-Bu) ppm. 13C NMR (125 MHz, DMSO-d6): δ = 162.3; 154.9; 147.1; 141.7; 135.8; 127.9; 127.1; 119.6; 34.0; 31.2 ppm. HRMS (ESI, +), m/z: calculated for C40H41N8O6 [M + H]+ 729.3065; found: 729.3146 [M + H]+, 751.2966 [M + Na]+.
N); 8.12 (s, 8H, Haromatic hydrazide); 7.80 (s, 4H, Haromatic aldehyde); 1.35 (s, 18H, t-Bu) ppm. 13C NMR (125 MHz, DMSO-d6): δ = 162.3; 154.9; 147.1; 141.7; 135.8; 127.9; 127.1; 119.6; 34.0; 31.2 ppm. HRMS (ESI, +), m/z: calculated for C40H41N8O6 [M + H]+ 729.3065; found: 729.3146 [M + H]+, 751.2966 [M + Na]+.
Conclusions
In conclusion, we have described the synthesis of novel [2 + 2] and [3 + 3] N-acylhydrazone-based macrocycles using a pool of dialdehydes and dihydrazides, under thermodynamic control. Formation of the macrocycles was monitored by 1H NMR, until equilibrium was reached, with and without any acid catalyst loading. Geometry of the macrocycles was assigned using 2D NMR and HRMS experiments as triangular and rectangular. The resulting compounds were also characterised using solid-state FTIR and thermal analysis, as well as using SEM, TEM and PXRD. The rectangular macrocycle was synthesized on a larger scale and the optical properties were investigated to show yellow emission under light irradiation. Moreover, hosting capacity assays indicated response for fluoride, using tetra-n-butylammonium as a counter anion in a stoichiometry of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 by UV-visible and NMR spectroscopy. Interestingly, recording fluorescence spectra of mixtures of the macrocycle and TBAF yielded intriguing results compared with the other spectroscopic techniques, suggesting the existence of multiple species in the excited state. Another notable observation regarded the 1H NMR spectra of mixtures of macrocycle and TBAF which indicated changes in chemical shifts for both residues. Difficulties of data fitting in all complexation experiments are very likely due to the existence of more species in solution. This is also supported by HRMS spectra that suggested amplification of several species, with notable formation of a [6 + 6] macrocycle as the major species in the presence of TBAF. In addition, bromide hosting was also observed in the HRMS experiments.
1 by UV-visible and NMR spectroscopy. Interestingly, recording fluorescence spectra of mixtures of the macrocycle and TBAF yielded intriguing results compared with the other spectroscopic techniques, suggesting the existence of multiple species in the excited state. Another notable observation regarded the 1H NMR spectra of mixtures of macrocycle and TBAF which indicated changes in chemical shifts for both residues. Difficulties of data fitting in all complexation experiments are very likely due to the existence of more species in solution. This is also supported by HRMS spectra that suggested amplification of several species, with notable formation of a [6 + 6] macrocycle as the major species in the presence of TBAF. In addition, bromide hosting was also observed in the HRMS experiments.
Data availability
The data supporting this article have been included as part of the ESI.† All raw data will be made available on request.Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript. Conceptualization – MM, DF. Data curation – NS, AH, CP, MF, NH. Formal analysis – NS, CT, MM. Investigation – NS, AH, CP, AM, AK, NH. Methodology – MM, DF, MF, NH. Project administration – MM. Supervision – MM, DF. Validation – NS, AH, MF, NH, MM. Visualization – NS, MM. Writing – original draft – NS, AH, AM, CT, MF, NH, MM. Writing – review & editing – all authors.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was partially supported by grants of the Romanian Ministry of Research, Innovation and Digitization, PNRR/2022/C9/MCID/I8, project number CF 760056/23.05.2023, code 235/29.11.2022. MF and AM acknowledge financial support from the Romanian Ministry of Research, Innovation and Digitization through the Core Program 2023–2026 (contract PC3-PN23080303). University of Bucharest – UniRem project no. 244 is also gratefully acknowledged.References
- S. Thota, D. A. Rodrigues, P. S. M. de Pinheiro, L. M. Lima, C. A. M. Fraga and E. J. Barreiro, Bioorg. Med. Chem. Lett., 2018, 28, 2797–2806 CrossRef CAS PubMed.
- (a) D. J. van Dijken, P. Kovaříček, S. P. Ihrig and S. Hecht, J. Am. Chem. Soc., 2015, 137, 14982–14991 CrossRef CAS PubMed; (b) P. Gupta, T. Panda, S. Allu, S. Borah, A. Baishya, A. Gunnam, A. Nangia, P. Naumov and N. K. Nath, Cryst. Growth Des., 2019, 19, 3039–3044 CrossRef CAS.
- (a) C. C. Paraschivescu, N. D. Hădade, A. G. Coman, A. Gautier, F. Cisnetti and M. Matache, Tetrahedron Lett., 2015, 56, 3961–3964 CrossRef CAS; (b) C. C. Paraschivescu, M. Matache, C. Dobrotă, A. Nicolescu, C. Maxim, C. Deleanu, I. C. Fărcăşanu and N. D. Hădade, J. Org. Chem., 2013, 78, 2670–2679 CrossRef CAS PubMed; (c) C. C. Anghel, A. G. Mirea, C. C. Popescu, A. M. Mădălan, A. Hanganu, A. Bende, N. D. Hădade, M. Matache and M. Andruh, Dyes Pigm., 2023, 210, 111023 CrossRef CAS.
- (a) S. Anbu, S. Kamalraj, C. Jayabaskaran and P. S. Mukherjee, Inorg. Chem., 2013, 52, 8294–8296 CrossRef CAS PubMed; (b) J.-C. Qin, B.-D. Wang, Z.-Y. Yang and K.-C. Yu, Sens. Actuators, B Chem., 2016, 224, 892–898 CrossRef CAS; (c) S. Joshi, S. Agarwal, A. Panjla, S. Valiyaveettil, S. Ganesh and S. Verma, ChemBioChem, 2022, 23, e202100654 CrossRef CAS PubMed.
- A. Maity, F. Ali, H. Agarwalla, B. Anothumakkool and A. Das, Chem. Commun., 2015, 51, 2130–2133 RSC.
- (a) A. G. Coman, A. Paun, C. C. Popescu, N. D. Hădade, A. Hanganu, G. Chiritoiu, I. C. Farcasanu and M. Matache, Bioorg. Chem., 2019, 92, 103295 CrossRef CAS PubMed; (b) A. G. Coman, A. Paun, C. C. Popescu, N. D. Hădade, C. C. Anghel, A. M. Mădălan, P. Ioniţă and M. Matache, New J. Chem., 2018, 42, 14111–14119 RSC.
- (a) F. B. L. Cougnon, A. R. Stefankiewicz and S. Ulrich, Chem. Sci., 2024, 15, 879–895 RSC; (b) M. Matache, E. Bogdan and N. D. Hădade, Chem.–Eur. J., 2014, 20, 2106–2131 CrossRef CAS PubMed; (c) T. Jiao, G. Wu, Y. Zhang, L. Shen, Y. Lei, C.-Y. Wang, A. C. Fahrenbach and H. Li, Angew. Chem., Int. Ed. Engl., 2020, 59, 18350–18367 CrossRef CAS PubMed.
- Y. Zhang and M. Barboiu, Chem. Rev., 2016, 116, 809–834 CrossRef CAS PubMed.
- (a) F. J. Uribe-Romo, C. J. Doonan, H. Furukawa, K. Oisaki and O. M. Yaghi, J. Am. Chem. Soc., 2011, 133, 11478–11481 CrossRef CAS PubMed; (b) Z. Zhuang, H. Shi, J. Kang and D. Liu, Mater. Today Chem., 2021, 22, 100573 CrossRef CAS.
- (a) Z. Shu, S. Sun, N. Gu, Z. Yang, Y. Shang, Y. Yang, M. Xia, B. Lin and P. Yang, Anal. Chim. Acta, 2023, 1253, 341093 CrossRef CAS PubMed; (b) J. Jose, A. Sreekanth, A. M. John, S. M. Basheer and P. B. Sreeja, Res. Chem. Intermed., 2019, 45, 425–435 CrossRef CAS; (c) Z. Li, N. Huang, K. H. Lee, Y. Feng, S. Tao, Q. Jiang, Y. Nagao, S. Irle and D. Jiang, J. Am. Chem. Soc., 2018, 140, 12374–12377 CrossRef CAS PubMed; (d) Y. Zhou, J. F. Zhang and J. Yoon, Chem. Rev., 2014, 114, 5511–5571 CrossRef CAS PubMed.
- (a) S. Hazra, S. Majumder, M. Fleck, R. Koner and S. Mohanta, Polyhedron, 2009, 28, 2871–2878 CrossRef CAS; (b) B. Dutta, P. Bag, B. Adhikary, U. Flörke and K. Nag, J. Org. Chem., 2004, 69, 5419–5427 CrossRef CAS PubMed; (c) A. Aguiari, E. Bullita, U. Casellato, P. Guerriero, S. Tamburini and P. A. Vigato, Inorganica Chim. Acta, 1992, 202, 157–171 CrossRef CAS.
- M. Bell, A. J. Edwards, B. F. Hoskins, E. H. K Chab and R. Robson, J. Am. Chem. Soc., 1989, 111, 3603–3610 CrossRef CAS.
- P. Vigato, S. Tamburini and L. Bertolo, Coord. Chem. Rev., 2007, 251, 1311–1492 CrossRef CAS.
- S. Ulrich, E. Buhler ang and J.-M. Lehn, New J. Chem., 2009, 33, 271–292 RSC.
- (a) N. Grimblat, A. M. Sarotti, T. S. Kaufman and S. O. Simonetti, Org. Biomol. Chem., 2016, 14, 10496–10501 RSC; (b) L. F. Lindoy, Synthesis, 1998, 07, 1029–1032 CrossRef.
- W. E. Benet, G. S. Lewis, L. Z. Yang and D. E. P. Hughes, J. Chem. Res., 2011, 35, 675–677 CrossRef CAS.
- (a) S. Di Stefano and G. Ercolani, Adv. Phys. Org. Chem., 2016, 50, 1–76 CrossRef; (b) G. Ercolani, L. Mandolini, P. Mencarelli and S. Roelens, J. Am. Chem. Soc., 1993, 115, 3901–3908 CrossRef CAS.
- (a) S. Ulrich and J.-M. Lehn, Angew. Chem., Int. Ed. Engl., 2008, 47, 2240–2243 CrossRef CAS PubMed; (b) Y. Choi, S. S. Lee and J. H. Jung, J. Mater. Chem. C, 2016, 4, 9646–9650 RSC; (c) W. G. Skene and J.-M. Lehn, Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 8270–8275 CrossRef CAS PubMed.
- Y. Fu, Z. Xing, C. Zhu, H. Yang, W. He, C. Zhu and Y. Cheng, Tetrahedron Lett., 2012, 53, 804–807 CrossRef CAS.
- F. J. Uribe-Romo, J. R. Hunt, H. Furukawa, C. Klöck, M. O'Keeffe and O. M. Yaghi, J. Am. Chem. Soc., 2009, 131, 4570–4571 CrossRef CAS PubMed.
- Y. Yang, W. Feng, J. Hu, S. Zou, R. Gao, K. Yamato, M. Kline, Z. Cai, Y. Gao, Y. Wang, Y. Li, Y. Yang, L. Yuan, X. C. Zeng and B. Gong, J. Am. Chem. Soc., 2011, 133, 18590–18593 CrossRef CAS PubMed.
- B. D. Cullity and S. R. Stock, Elements of X-Ray Diffraction, 3 edn, Prentice-Hall, Englewood Cliffs, NJ, 2001 Search PubMed.
- V. Martí-Centelles, M. D. Pandey, M. I. Burguete and S. V. Luis, Chem. Rev., 2015, 115, 8736–8834 CrossRef PubMed.
- D. Brynn Hibbert and P. Thordarson, Chem. Commun., 2016, 52, 12792–12805 RSC.
- (a) R. O. Ramabhadran, Y. Liu, Y. Hua, M. Ciardi, A. H. Flood and K. Raghavachari, J. Am. Chem. Soc., 2014, 136, 5078–5089 CrossRef CAS PubMed; (b) Y. Hua, R. O. Ramabhadran, E. O. Uduehi, J. A. Karty, K. Raghavachari and A. H. Flood, Chem. Eur. J., 2011, 17, 312–321 CrossRef CAS PubMed; (c) U. Manna, G. Das and M. A. Hossain, Coord. Chem. Rev., 2022, 455, 214357 CrossRef CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, GAUSSIAN 09, Revision C.01, Gaussian, Inc., Wallingford CT, 2009 Search PubMed.
- J. Tomasi, B. Mennucci and R. Cammi, Chem. Rev., 2005, 105, 2999–3093 CrossRef CAS PubMed.
- H. Okawa, M. Honda and S. Kida, Chem. Lett., 1972, 1, 1027–1030 CrossRef.
- A. A. Morton and C. E. Claff, J. Am. Chem. Soc., 1954, 76, 4935–4938 CrossRef.
- C. J. Fahrni and A. Pfaltz, Helv. Chim. Acta, 1998, 81, 491–506 CrossRef CAS.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4ta09035g |
| ‡ These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2025 |







