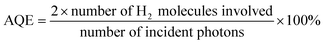Salt-assisted activation of n → π* electronic transition in orange carbon nitride for enhanced visible-light-driven H2 generation†
Lin
Lei
 a,
Yongbo
Fan
b,
Yuxin
Jia
c,
Huiqing
Fan
a,
Yongbo
Fan
b,
Yuxin
Jia
c,
Huiqing
Fan
 *a,
Weijia
Wang
*a,
Weijia
Wang
 *a and
Haitao
Huang
*a and
Haitao
Huang
 *b
*b
aState Key Laboratory of Solidification Processing, School of Materials Science and Engineering, Northwestern Polytechnical University, Xi'an 710072, China. E-mail: hqfan3@163.com; weijia.wang@nwpu.edu.cn
bDepartment of Applied Physics and Research Institute for Smart Energy, The Hong Kong Polytechnic University, Hong Kong, China. E-mail: aphhuang@polyu.edu.hk
cSchool of Electronics and Information, Northwestern Polytechnical University, Xi'an 710072, China
First published on 3rd December 2024
Abstract
Metal salts demonstrate significant advantages in promoting the polymerization of highly crystalline carbon nitrides for meeting the demands of green photocatalytic hydrogen production. However, the enhanced light absorption arising from n → π* electronic transition is difficult to achieve within symmetric and orderly planar heptazine-based structures, which are typically induced by molten-salt treatments. Herein, a novel orange carbon nitride (OCN) with activated n → π* electronic transition is synthesized via a NaCl-assisted melamine assembly of melem and a secondary calcination approach. Na+ ions are crucial for the formation of hexagonal melem flakes, which are exfoliated from rod-like structures connected by interlayer van der Waals forces. The cyano groups within OCN can widen the visible light harvesting ability and adjust the band structure. The introduction of nitrogen defects suppresses the radiative recombination of photogenerated charge carriers by creating a midgap energy level, thereby facilitating efficient electron–hole separation and migration. Benefiting from the n → π* electronic transition and optimized carrier dynamics, the OCN presents an enhanced hydrogen evolution rate of 1043 μmol g−1 h−1 under visible light (λ > 420 nm). Our low-temperature NaCl-assisted polymerization process not only reduces crystallinity but also endows unique optical features to carbon nitrides, which broadens the function of metal salts in synthesizing catalysts for solar energy applications.
1 Introduction
Achieving zero carbon emissions is a pivotal objective for sustainable development in the future.1 Photocatalysis stands out as a clean and renewable technology for harnessing solar energy and converting it into chemical energy. A prime application of this technology is the production of hydrogen (H2) via the photocatalytic splitting of water under illumination.2 The key point of such process is the selection and development of suitable catalysts with desired features, in which cost-effective, eco-friendly, and photocatalytic activity need to be fully considered. Polymeric carbon nitrides (PCNs), widely studied metal-free semiconductors in the last decade, have exhibited great potential in the photocatalytic field owing to their facile synthesis, non-toxicity, visible light-active capacity, and excellent stability under thermal and corrosive conditions.3 However, the practical application of PCNs faces significant challenges, primarily due to insufficient active sites, limited visible light absorption, and the slow kinetics of photo-induced electron–hole pair separation and migration.4Molten-salt (NaX/KX, where X = Cl, Br, or I) strategy has been reported extensively for synthesizing highly crystalline carbon nitride structures,5–8 enabling the efficient charge transfer/diffusion across the interlayer galleries. For example, a hydrophilic carbon nitride could be synthesized by a salt-assisted heating process using KCl and ammonium thiocyanate as precursors.9 The optimization of crystallinity and functional groups enable it to form a quasi-homogeneous system in water. Salt-assisted process is also facilitated to induce cyano (–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N) group structural defects and coordinate alkali metal ions into the interval between adjacent melon chains, which is crucial for tailoring the charge distribution and optical property.10–12 Typically, dark-colored carbon nitrides, such as red/orange carbon nitrides, with distinct lattice fringes and enhanced light response could be synthesized using eutectic salts and organic molecules.13,14 Notably, the broad optical harvest range is primarily attributed to the narrowed bandgap, intuitively, a red-shifted absorption edge towards the longer-wavelength region. However, the incompatibility between the intrinsic smaller bandgap and the robust redox kinetics in the PCNs remains a scientific dilemma, making it challenging to effectively proceed H2 production. Therefore, activating the n → π* electronic transition (a new shoulder absorption near 500 nm) may be much preferable for extending the visible light response range while preserving a large thermodynamic driving force for redox reactions.15 Such a transition can be realized under lower photon energy irradiation, which does not significantly affect the original bandgap. The n → π* electronic transition in PCN is partially associated with the lone-pair electrons of sp2-hybridized nitrogen atoms.16 An effective approach to achieve n → π* electronic transition is to distort the tri-s-triazine units and break the symmetry and planar structure of PCN.17 However, activating this transition in the highly crystalline PCN is a great challenge, possibly due to the molten-salt surfaces that continually orientate the polymerization and assembly of tri-s-triazine units. This process results in a relatively symmetrical and planar structure, which is not conducive to the emergence of the n → π* electronic transition.18 Consequently, the n → π* electronic transition is to some extent hardly accessible via most reported molten-salt treatments throughout the reactive process of precursors (e.g., melamine and urea), involving melting, adsorption, nucleation, and polymerization stages.19 Therefore, the strategic application of salts during the synthesis stages to facilitate the n → π* electronic transition in PCN is a relatively unexplored area that warrants further investigation.
N) group structural defects and coordinate alkali metal ions into the interval between adjacent melon chains, which is crucial for tailoring the charge distribution and optical property.10–12 Typically, dark-colored carbon nitrides, such as red/orange carbon nitrides, with distinct lattice fringes and enhanced light response could be synthesized using eutectic salts and organic molecules.13,14 Notably, the broad optical harvest range is primarily attributed to the narrowed bandgap, intuitively, a red-shifted absorption edge towards the longer-wavelength region. However, the incompatibility between the intrinsic smaller bandgap and the robust redox kinetics in the PCNs remains a scientific dilemma, making it challenging to effectively proceed H2 production. Therefore, activating the n → π* electronic transition (a new shoulder absorption near 500 nm) may be much preferable for extending the visible light response range while preserving a large thermodynamic driving force for redox reactions.15 Such a transition can be realized under lower photon energy irradiation, which does not significantly affect the original bandgap. The n → π* electronic transition in PCN is partially associated with the lone-pair electrons of sp2-hybridized nitrogen atoms.16 An effective approach to achieve n → π* electronic transition is to distort the tri-s-triazine units and break the symmetry and planar structure of PCN.17 However, activating this transition in the highly crystalline PCN is a great challenge, possibly due to the molten-salt surfaces that continually orientate the polymerization and assembly of tri-s-triazine units. This process results in a relatively symmetrical and planar structure, which is not conducive to the emergence of the n → π* electronic transition.18 Consequently, the n → π* electronic transition is to some extent hardly accessible via most reported molten-salt treatments throughout the reactive process of precursors (e.g., melamine and urea), involving melting, adsorption, nucleation, and polymerization stages.19 Therefore, the strategic application of salts during the synthesis stages to facilitate the n → π* electronic transition in PCN is a relatively unexplored area that warrants further investigation.
Melem (2,5,8-triamino-s-heptazine), an intermediate also called the pre-polymerized product of the precursors, is typically synthesized at relatively low temperatures (400–430 °C).20 Inspired by the nanostructures of melem that play a vital role in influencing the properties for further derived PCNs at higher temperatures (>450 °C), we synthesized a Na+-coordinated melem (Na-melem) hexagonal block with changes in phase and compositions based on a salt pre-heating process, which can be utilized to produce an orange carbon nitride (OCN) with an activated n → π* electronic transition. During the low-temperature stage, NaCl crystals serve to separate the melamine precursors, providing a large surface confinement to orientate the pre-polymerization of melem monomers. This designed low-temperature molten-salt treatment results in the morphology of melem tending to aggregate into hexagonal blocks rather than typical rod-like supramolecules after NaCl removal. Upon high-temperature calcination, the resulting OCN features large-surface lamellar stacking nanosheets with distinct edge steps, a result of the templating effect of NaCl crystals. Comprehensive characterizations and theoretical calculations confirm significant alterations in the physicochemical and photoelectric properties. Consistent with our expectations, the OCN demonstrates enhanced photocatalytic activity for H2 evolution under visible light irradiation, attributed to the synergistic effect of the enhanced n → π* electronic transition and optimized charge transfer dynamics.
2 Experimental
2.1. Material preparation
2.2. Characterization
X-ray diffraction (XRD) measurements were performed using a Shimadzu 7000 diffractometer with Cu Kα irradiation (λ = 1.5406 Å). Fourier transform infrared (FTIR) spectra were recorded using a Thermo Scientific Nicolet iS50 instrument. Transmission electron microscopy (TEM) was performed using an FEI Talos 200 microscope equipped with an energy-dispersive X-ray spectrometer (EDS). Scanning electron microscopic (SEM) images were acquired using an FEI Helios G4 CX instrument. X-ray photoelectron spectroscopy (XPS) spectra were recorded using a PHI 50000 Versa Probe III spectrometer. UV-vis diffuse reflectance spectroscopy (UV-vis DRS) spectra were recorded using a Lambda 1050+ spectrophotometer. An Edinburgh FLS1000 instrument (excitation wavelength: 375 nm) was used to record photoluminescence (PL) spectra. Nitrogen adsorption–desorption isotherms and porosity distribution were measured using an auto sorb-iQ analyzer machine at 77 K. Solid-state 13C nuclear magnetic resonance (NMR) spectra were recorded using a Bruker 400WB spectrometer. Electron paramagnetic resonance (EPR) spectra were recorded using a Bruker A300 spectrometer. Thermo-gravimetric (TG) analysis was evaluated under an Ar atmosphere at a heating rate of 10 °C min−1 using a NETZSCH STA 449F5 thermal analyzer. The contact angles of the as-synthesized samples were measured using an SZ-CAMC32 surface tension measuring instrument at ambient temperature. Photoelectrochemical experiments were conducted using an electrochemical workstation (CHI660E, Chenhua, Shanghai, China).2.3. Photocatalytic test
The photocatalytic H2 evolution performance of the prepared samples was assessed using a Pyrex top-irradiation reaction vessel (Beijing Perfectlight, Labsolar 6A). A quantity of 50 mg of each photocatalyst was suspended in an aqueous solution containing 90 mL of distilled water, 10 mL of triethanolamine (acting as a hole sacrificial agent), and 0.7 mL of chloroplatinic acid (1 wt% Pt, serving as a co-catalyst) within the reaction cell. The mixture was stirred magnetically at 10 °C. A 300 W xenon lamp, equipped with a 420 nm cut-off filter, was employed to simulate visible light conditions. Prior to light illumination, the system was purged with a vacuum pump to eliminate air from the reaction environment. The generated H2 was quantified using a gas chromatographic instrument (Fuli GC9790II, thermal conductivity detector, 5 Å molecular sieve column, with high-purity nitrogen as the carrier gas). The apparent quantum efficiency (AQE) was calculated using the following equation:2.4. Theoretical calculations
Density functional theory (DFT) calculations were done using the Vienna Ab initio Simulation Package (VASP). A generalized gradient approximation (GGA)-Perdew–Burke–Ernzerhof (PBE) functional was employed to treat electron exchange and correlation energy. An energy cutoff of 500 eV was specified for the all-electron plane-wave basis set. To insulate the interaction between adjacent layers, a vacuum layer of 20 Å was incorporated. A (2 × 2 × 1) Monkhorst–Pack k-point mesh was employed for sampling the Brillouin-zone integrations. The convergence criteria for energy convergent standard and force were established to 10−5 eV and 0.02 eV Å−1, respectively.3 Results and discussion
3.1. Characterization of the material structure
The novel orange carbon nitride (OCN) was synthesized via a NaCl-assisted pre-polymerization and secondary calcination method (Fig. 1a). As observed by the scanning electron microscopic (SEM) image, the size of the NaCl particle is larger than 5 μm (Fig. S1a†), which provides a large surface for the adsorption of melamine molecules and the assembly of melem intermediates. After washing out of NaCl with deionized water, the resulting Na-melem exhibits a large lamellar morphology with some hexagonal blocks assembled on its surface (Fig. 1b and c). In contrast, conventional melem synthesized at 400 °C consists of irregular bulks that are several micrometers in size (Fig. S1b†), and they easily rearrange into hexagonal rod-like melem supramolecules in water due to the presence of intermolecular hydrogen bond networks of heptazine units, characteristic of its R3c space group (Fig. 1d).21 Notably, the most significant morphological distinction between Na-melem and rod-like-melem lies in the longitudinal length perpendicular to the hexagonal sheet structures. This is because the phase structure of melem has been altered due to the on-surface polymerization process facilitated by NaCl treatment. As can be seen in the X-ray diffraction (XRD) patterns (Fig. 1e), the as-synthesized bulk melem is similar to an oligomer containing a melem-related structure that has been observed in the previous reports, which can be indexed to the space group P21/c.22,23 Two sharp diffraction peaks at 12.3° and 13.5° are mainly ascribed to the parallel arrangement of the C6N7(NH2)3 molecules with different intraplanar distances. The dominant reflection at 25.3° and 27.3° is observed that can be attributed to layered arrangement of melem units with interlayer distances of 0.352 and 0.326 nm, respectively.24 After undergoing a self-assembly process in water, both obtained rod-like-melem and Na-melem present more ordered crystal patterns. These changes in XRD patterns indicate that water can effectively regulate the exposure of different crystal facets and the formation of new phases of melem. The emergence of two sharp diffraction peaks at 6.1° and 12.3° indexed to the (110) and (220) planes correspond to the intraplanar periodic hydrogen bond interaction of melem hydrate.25 Almost no difference in the position and intensity of the two peaks can be observed in the samples. However, in comparison to the rod-like-melem, the dominant peak at 26.9° representing interlayer stacking of melem hydrate for Na-melem becomes lower, which can be attributed to the π–π interaction between adjacent hexagonal planar sheets of melem hydrate that is reduced due to Na+ coordination. Importantly, this peak position also shifts to a lower angle, meaning an increased interlayer spacing. These results indicate that Na+ is likely to exist in the interlayer rather than the intralayer of melem hydrate, leading to the expansion of the interlayer distance. X-ray photoelectron spectroscopy (XPS) spectra of Na 1s can be used to confirm the presence of Na+ in Na-melem (Fig. S2a and b†), and the NaCl salt is completely removed after washing because almost no Cl signal can be observed from the XPS Cl 1s spectra (Fig. S2c†). Schematic illustrations of the interlayer stacking structures of Na-melem and rod-like-melem are displayed in Fig. 1f. From previous reports, alkali metal ions such as Na+ and K+ could be intercalated between the triazine–heptazine units or melon chains of g-C3N4.26,27 The interaction between the bridge Na+ in the triangular vacancies and N in the heptazine backbone distorted the aromatic conjugated plane, which make Na+ not completely exist in the in-plane structure of g-C3N4, but occupy a certain position in the interlayer and affected interlayer spacing.28 Given the identical repeat heptazine units in Na-melem and PCN, the intercalation of Na+, with a larger atomic size than C and N, into the melem interlayer structure is able to enlarge the stacking distance. This enlargement effectively weakens the van der Waals force between the layers, leading to the peeling of hexagonal rods along the vertical direction to form hexagonal sheets. Despite the significant changes in the physicochemical properties of Na-melem, thermogravimetric analysis indicates no substantial difference in thermal decomposition behavior compared to melem below the synthesis temperature of 650 °C (Fig. S3†).To synthesize the orange OCN, a secondary calcination process at 650 °C was applied to Na-melem. From these SEM images (Fig. 2a–c), it can be observed that these large sheet morphologies with a lateral size of tens of microns are maintained after annealing. The original hexagonal morphology from Na-melem is destroyed and a large number of irregular pores appear on the surface of the OCN. In addition, a straight step can be observed at the edge of the sheet. Such step is formed by the on-surface polymerization effect of the NaCl crystal with a similar edge in appearance (Fig. 2d), effectively serving as a hard template to orientate the assembly of 2D melem sheets. Similarly, the transmission electron microscopic (TEM) image in Fig. 2e also clearly shows that the OCN has a large lamellar morphology with distinctive edge steps. However, the OCN displays a uniform non-crystalline structure with no lattice fringes appearing from the high-resolution TEM image (Fig. 2f), which is markedly different from the crystalline carbon nitride typically obtained through conventional molten-salt methods.29 Furthermore, the selected-area electron diffraction (SAED) pattern displays fuzzy diffraction rings of the typical (100) and (002) planes (Fig. 2g),30 respectively. An important reason is that no NaCl salt is involved in the second calcination stage, which avoids the formation of highly crystalline phase.18 The potential decreased crystalline degree in the OCN indicates that the presence of Na+ rather than the NaCl crystal in Na-melem is difficult to develop in-plane and interlayer ordered carbon nitride structures at temperatures above 600 °C. Elemental mapping demonstrates the uniform distribution of C, N, and Na across the entire selected area, implying successful incorporation of Na+ into the OCN structure (Fig. 2h). In contrast, the PCN synthesized via the thermal polycondensation of melamine displays a densely packed agglomerate morphology (Fig. S4a and c†), while the melem-derived MCN shows a wrinkled structure composed of continuously connected curved layers (Fig. S4b and d†). These findings underscore the pivotal role of phase and microstructural changes in melem intermediates in shaping the properties of the final carbon nitride products.
 | ||
| Fig. 2 (a–c) SEM images of OCN. (d) TEM image of NaCl. (e) TEM image, (f) high-resolution TEM image, (g) SAED pattern, and (h) corresponding elemental mappings of C, N, and Na distribution of OCN. | ||
Due to the robust correlation between reactive active sites and the morphology, nitrogen adsorption–desorption isotherm analysis demonstrates that the BET surface area of OCN (12.2 m2 g−1) does not largely exceed that of PCN (5.5 m2 g−1) and is even lower than that of MCN (16.4 m2 g−1). This is primarily due to the smaller curved nanosheets and the abundance of pores in MCN, as depicted in Fig. 3a. The pore size distribution, categorized as a type IV isotherm for OCN, also indicates a reduced pore volume compared to MCN profited from its large-area lamellar morphology (Fig. 3b). The phase structure and crystallinity of these products were recorded using XRD. As illustrated in Fig. 3c, the XRD pattern of the MCN closely resembles that of the PCN. Compared with the PCN and MCN, the (002) diffraction peak of the OCN, corresponding to the interplanar stacking of aromatic systems, exhibits a lower intensity and a broader full width at half maximum.31 Moreover, the intensity of the (100) peak, associated with the in-plane repeating heptazine motifs in the OCN, almost disappears, meaning its reduced crystallinity in both parallel and perpendicular directions to the nanosheet.32 The position of the (002) peak shifts from 27.4° in PCN and 27.6° in MCN to 27.3° in OCN, indicating the retention of a large interlayer distance after Na-melem annealing. The changes in molecular structures of OCN were identified by Fourier transform infrared (FTIR) spectroscopy. The emergence of a new sharp peak located at 2180 cm−1 in OCN originates from the asymmetric stretching vibration of –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N groups, resulting from the partial loss of terminal ammonia in heptazine rings (Fig. 3d).32 The peak at 808 cm−1 belongs to the breathing pattern of the heptazine unit, while the bands in the regions of 1200 to 1800 cm−1 and 3000 to 3400 cm−1 are assigned to the C
N groups, resulting from the partial loss of terminal ammonia in heptazine rings (Fig. 3d).32 The peak at 808 cm−1 belongs to the breathing pattern of the heptazine unit, while the bands in the regions of 1200 to 1800 cm−1 and 3000 to 3400 cm−1 are assigned to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N heterocycles and the stretching vibration of N–H,33 respectively. Compared with the PCN and MCN, the attenuated N–H stretching vibration in OCN also indicates that the formation of –C
N heterocycles and the stretching vibration of N–H,33 respectively. Compared with the PCN and MCN, the attenuated N–H stretching vibration in OCN also indicates that the formation of –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N groups destroys the intrinsic site of the terminal ammonia. Notably, the presence of new bands at 992 and 1145 cm−1 in the OCN is assigned to the symmetric and asymmetric vibrations of NC2 bonds and Na-NC2 groups (Fig. 3e),34 further indicating that metal ions have been introduced into the heptazine network structure. In addition, the left-shift of the C–N stretching vibration peak from 1540 cm−1 in the PCN and MCN to 1560 cm−1 in the OCN implies that Na+ coordination and the formation of –C
N groups destroys the intrinsic site of the terminal ammonia. Notably, the presence of new bands at 992 and 1145 cm−1 in the OCN is assigned to the symmetric and asymmetric vibrations of NC2 bonds and Na-NC2 groups (Fig. 3e),34 further indicating that metal ions have been introduced into the heptazine network structure. In addition, the left-shift of the C–N stretching vibration peak from 1540 cm−1 in the PCN and MCN to 1560 cm−1 in the OCN implies that Na+ coordination and the formation of –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N groups lead to a distorted and fluctuant conjugated structure,35 which is much conducive to make the n → π* electronic transition allowable in the OCN. Based on the above-mentioned results, the primary chemical framework of the OCN is maintained despite the incorporation of impurities and defects. Further insights into the molecular structure difference of the as-prepared samples were monitored by solid-state 13C nuclear magnetic resonance (NMR) spectroscopy. Two signals at approximately 156.5 and 164.4 ppm are detected in the 13C spectra of the PCN, MCN, and OCN (Fig. 3f), which are assigned to the chemical shifts of C3N (C2) and C2N–NHx (C3) in the heptazine units.36 Moreover, two weak new peaks emerged at 122.1 and 170.2 ppm, corresponding to the C atom (C1) in –C
N groups lead to a distorted and fluctuant conjugated structure,35 which is much conducive to make the n → π* electronic transition allowable in the OCN. Based on the above-mentioned results, the primary chemical framework of the OCN is maintained despite the incorporation of impurities and defects. Further insights into the molecular structure difference of the as-prepared samples were monitored by solid-state 13C nuclear magnetic resonance (NMR) spectroscopy. Two signals at approximately 156.5 and 164.4 ppm are detected in the 13C spectra of the PCN, MCN, and OCN (Fig. 3f), which are assigned to the chemical shifts of C3N (C2) and C2N–NHx (C3) in the heptazine units.36 Moreover, two weak new peaks emerged at 122.1 and 170.2 ppm, corresponding to the C atom (C1) in –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N groups and the C atom (C4) adjacent to –C
N groups and the C atom (C4) adjacent to –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N groups,37 suggesting that the NaCl-assisted pre-polymerization process readily deprotonates terminal C–NH2 groups and forms nitrogen defects, consistent well with the FTIR finding. The above analysis shows that the molecular structure of OCN does not belong to poly(heptazine imide) carbon nitride with high crystallinity and stored long-lived photoelectrons, which are usually synthesized in the presence of potassium thiocyanate.38,39
N groups,37 suggesting that the NaCl-assisted pre-polymerization process readily deprotonates terminal C–NH2 groups and forms nitrogen defects, consistent well with the FTIR finding. The above analysis shows that the molecular structure of OCN does not belong to poly(heptazine imide) carbon nitride with high crystallinity and stored long-lived photoelectrons, which are usually synthesized in the presence of potassium thiocyanate.38,39
 | ||
| Fig. 3 (a) N2 adsorption/desorption isotherms, (b) pore size distribution, (c) XRD patterns, (d and e) FTIR spectra, and (f) solid-state 13C NMR spectra of PCN, MCN, and OCN. | ||
XPS was employed to explore the difference in compositions and chemical bonding of the synthesized samples. The survey spectra show the existence of C, N, Na, and O elements in the OCN, as shown in Fig. 4a. Two Na signals at 495 and 1069 eV are attributed to the Na LK1 Auger peaks and Na 1s,40 respectively. The high-resolution Na 1s peak further confirms the formation of Na–N bond coordination (Fig. 4b), demonstrating that alkali metal Na is successfully incorporated between adjacent melon chains.10 Consistent with the Na-melem observations, no signal related to Cl element is detected in the Cl 1s spectra, as presented in Fig. S5.† In the XPS C 1s spectra (Fig. 4c), PCN and MCN can be deconvoluted into three peaks at 284.6, 286.1, and 288.0 eV, corresponding to reference C (C–C), C–NH bond, and sp2-hybridized C (N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) in the heptazine unit,41 respectively. For the OCN, the robust peak at 286.1 eV is attributed to the synergistic existence of C–NH and –C
N) in the heptazine unit,41 respectively. For the OCN, the robust peak at 286.1 eV is attributed to the synergistic existence of C–NH and –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N groups, aligning with the finding from FTIR and NMR analyses. Obviously, two new peaks emerge in the OCN, centered at 289.2 and 290.0 eV. The former faint peak is assigned to the O–C
N groups, aligning with the finding from FTIR and NMR analyses. Obviously, two new peaks emerge in the OCN, centered at 289.2 and 290.0 eV. The former faint peak is assigned to the O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O defect, arising from oxygen species induced by hydrogen bond interaction between Na-melem and water.25 The subsequent high-temperature calcination process at 650 °C results in the formation of a small number of oxygen-containing defects at the heptazine ring edges. The latter peak is ascribed to a shakeup satellite associated with the carbon π–π* transition.42 The N 1s spectra can be deconvolved into four peaks at 398.5, 399.7, 401.1, and 403.8 eV, which is assigned to sp2-hybridized N (C–N
O defect, arising from oxygen species induced by hydrogen bond interaction between Na-melem and water.25 The subsequent high-temperature calcination process at 650 °C results in the formation of a small number of oxygen-containing defects at the heptazine ring edges. The latter peak is ascribed to a shakeup satellite associated with the carbon π–π* transition.42 The N 1s spectra can be deconvolved into four peaks at 398.5, 399.7, 401.1, and 403.8 eV, which is assigned to sp2-hybridized N (C–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), tertiary N (N–(C)3), terminal C–NHx group, and π excitation,43 respectively (Fig. 4d). Compared to PCN and MCN, there is an apparent decrease in N–(C)3 and an increase in C–NHx bonds of OCN mainly caused by the formation of negatively charged –C
C), tertiary N (N–(C)3), terminal C–NHx group, and π excitation,43 respectively (Fig. 4d). Compared to PCN and MCN, there is an apparent decrease in N–(C)3 and an increase in C–NHx bonds of OCN mainly caused by the formation of negatively charged –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N groups. In addition, with the formation of the Na–N bond, the C–N
N groups. In addition, with the formation of the Na–N bond, the C–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C peak in OCN shifts to a higher binding energy, which is due to the fact that the introduced Na leads to a redistribution of electron density in the heptazine framework. The electron in heptazine unit tends to transfer to Na, resulting in the increased electron density on Na and decreased electron density on N. As the higher electron density corresponds to a lower XPS binding energy, the shift in the XPS peak of N matches the change in electron density. These results confirm that the incorporation of Na+ and –C
C peak in OCN shifts to a higher binding energy, which is due to the fact that the introduced Na leads to a redistribution of electron density in the heptazine framework. The electron in heptazine unit tends to transfer to Na, resulting in the increased electron density on Na and decreased electron density on N. As the higher electron density corresponds to a lower XPS binding energy, the shift in the XPS peak of N matches the change in electron density. These results confirm that the incorporation of Na+ and –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N groups significantly influence the composition and chemical state of the photocatalysts.
N groups significantly influence the composition and chemical state of the photocatalysts.
 | ||
| Fig. 4 (a) XPS survey spectra of PCN, MCN, and OCN. (b) High-resolution Na 1s spectra of OCN. High-resolution (c) C 1s and (d) N 1s spectra of PCN, MCN, and OCN. | ||
3.2. Optical absorption properties and band structure
Optimizations in the phase structure and chemical composition of OCN are supposed to result in changes in their optical absorption and electronic structure. The ultraviolet-visible diffuse reflectance spectra (UV-vis DRS) indicate that PCN shows poor light-harvesting ability with a typical absorption edge around 460 nm (Fig. 5a). As the MCN and OCN are synthesized at a high temperature of 650 °C, the intrinsic absorption band originating from the π → π* electron transition is enhanced. Further slightly enhanced optical absorption (460–630 nm) can be observed in the MCN, mainly due to the formation of N defect (vacancy) at high temperatures, leading to the generation of defect states below the conduction band (CB).20 Moreover, the n → π* electron transition arising from the broken symmetry and lattice vibration (out of plane) of the planar heptazine units is activated in OCN,44 in which an obvious absorption shoulder emerges at a wavelength of 500 nm in the visible light region. The absorption edges of both the MCN and the OCN also undergo a progressive redshift under these conditions. According to Kubelka–Munk transformations, the intrinsic bandgap narrows from 2.69 eV of PCN and 2.55 eV of MCN to 2.50 eV of OCN (Fig. 5b). The dominant merit of low crystalline or amorphous carbon nitride is the intrinsic smaller bandgap over its partially crystalline counterpart due to their band tail states, enabling the efficient utilization of a wide range of visible light.45 The previous research demonstrated a similar phenomenon in the N-deficient carbon nitride with low crystallinity synthesized by one-step alkali-assisted polymerization of KOH and urea at 550 °C.19 Specifically, the enhancement of light absorption is mainly attributed to the narrowing of the bandgap, rather than the appearance of n → π* electron transition. Therefore, our two-step salt-assisted calcination combined with a high polymerization temperature is the key to twist the planer structure of OCN, making the n → π* electron transition possible. The transition energy of defect level can be determined to be 1.94 eV and 1.98 eV for the MCN and OCN, respectively, meaning that electrons can transition from the valence band (VB) to the defect energy level under photon illumination with a long wavelength and low energy. The Mott–Schottky curves of the photocatalysts are tested at different frequencies of 800, 1000, and 1500 Hz, respectively, where the positive slopes indicate the n-type features of PCN, MCN, and OCN (Fig. 5c). The flat band potentials obtained by the confluence of tangents are about −1.25, −1.18, and −1.37 eV (vs. Ag/AgCl) for the PCN, MCN, and OCN, respectively. Based on the values of bandgaps and flat band potentials, the VB potentials of the PCN, MCN, and OCN were evaluated to be 1.54, 1.42, and 1.28 eV (vs. NHE), respectively, and the band structure arrangements are listed in Fig. 5d. Consistent with the markedly increased visible light harvesting, the OCN suffers a matching appearance variation from yellow of PCN and light orange of MCN to orange. The high CB potential of OCN indicates a large thermodynamic driving force for water reduction. The calculated potential of the defect energy level is −0.52 eV for the MCN and −0.70 eV for the OCN, which theoretically meets the requirement of H2 production.46 | ||
| Fig. 5 (a) UV-vis DRS spectra, (b) Kubelka–Munk curves, (c) Mott–Schottky curves under different frequencies, and (d) band structures with corresponding photographs of PCN, MCN, and OCN. | ||
3.3. Theoretical calculation
In order to gain a deeper understanding of the variation in the electronic characteristics and charge kinetics of OCN, influenced by the formation of defects from negatively charged functional groups and metal atomic coordination, density functional theory (DFT) calculations were performed. The in-plane molecular structure of PCN consists of periodic melon chains, as depicted in Fig. 6a. The structural model of OCN can be determined according to several aspects. Due to the larger radius of Na than C and N, alkali metals Na+ and K+ are usually intercalated in the interstitial region between amine-linked heptazine-based melon chains.5,47 The above-mentioned FTIR spectroscopy, NMR, and XPS results show that Na+ only accounts for a small part of the OCN, and it is stable by interacting with N atoms in the surrounding heptazine ring. To preserve the semiconductor property of OCN, a Na+ is embedded in the middle of adjacent melon chains, and a –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N group is formed at the termini of the melon chains in the computational structure model (Fig. 6d). It can clearly be found that the bandgap energy of OCN is smaller than that of PCN (Fig. 6b and e) in good agreement with the decreased trend observed in the UV-vis DRS data. Moreover, a midgap energy level appears below the CB bottom in the OCN, attributed to the defect state that induced a strong absorption tail. The distance between the midgap and the CB bottom is different from the experimental value, as it is influenced by various factors in DFT calculations, such as the approximation of functional, electron self-interaction and the choice of calculation parameters and methods. The above-mentioned results indicate that the introduction of Na atom and –C
N group is formed at the termini of the melon chains in the computational structure model (Fig. 6d). It can clearly be found that the bandgap energy of OCN is smaller than that of PCN (Fig. 6b and e) in good agreement with the decreased trend observed in the UV-vis DRS data. Moreover, a midgap energy level appears below the CB bottom in the OCN, attributed to the defect state that induced a strong absorption tail. The distance between the midgap and the CB bottom is different from the experimental value, as it is influenced by various factors in DFT calculations, such as the approximation of functional, electron self-interaction and the choice of calculation parameters and methods. The above-mentioned results indicate that the introduction of Na atom and –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N defect leads to decrease in the bandgap and the formation of midgap state in g-C3N4. The formation of a similar midgap state can be observed in the defect structural g-C3N4 based on DFT calculation.10,48 The midgap acts as the dominant center for light excitation and relaxation. An additional potential advantage of midgap is that it provides trapping sites for photogenerated charge carriers and prevents them from rapid recombination, thereby facilitating electron transfer and photocatalytic processes.49,50 The density of states (DOS) analysis shows that both C 2p and N 2p orbitals contribute to the lowest unoccupied molecular orbital (LUMO), while the highest occupied molecular orbital (HOMO) is predominantly composed of N 2p orbitals for both the PCN and the OCN, as shown in Fig. 6c and f. The introduction of Na does not participate in the orbital composition. In the PCN, due to the high symmetry of melon chains, the occupied and unoccupied orbits partially overlap around those C and N atoms, as shown in Fig. 6g. In contrast, the charge density in the OCN is redistributed across different locations without any overlap (Fig. 6h), indicating better spatial separation of charge carriers. Such spatial separation effectively isolates the redox sites, thereby inhibiting the recombination of photogenerated carriers.
N defect leads to decrease in the bandgap and the formation of midgap state in g-C3N4. The formation of a similar midgap state can be observed in the defect structural g-C3N4 based on DFT calculation.10,48 The midgap acts as the dominant center for light excitation and relaxation. An additional potential advantage of midgap is that it provides trapping sites for photogenerated charge carriers and prevents them from rapid recombination, thereby facilitating electron transfer and photocatalytic processes.49,50 The density of states (DOS) analysis shows that both C 2p and N 2p orbitals contribute to the lowest unoccupied molecular orbital (LUMO), while the highest occupied molecular orbital (HOMO) is predominantly composed of N 2p orbitals for both the PCN and the OCN, as shown in Fig. 6c and f. The introduction of Na does not participate in the orbital composition. In the PCN, due to the high symmetry of melon chains, the occupied and unoccupied orbits partially overlap around those C and N atoms, as shown in Fig. 6g. In contrast, the charge density in the OCN is redistributed across different locations without any overlap (Fig. 6h), indicating better spatial separation of charge carriers. Such spatial separation effectively isolates the redox sites, thereby inhibiting the recombination of photogenerated carriers.
3.4. Photoexcitation charge separation
Photoluminescence (PL) spectra were recorded to explore the recombination dynamics of photoinduced charge carriers. As depicted in Fig. 7a, under a 365 nm excitation source, a monitored strong emission peak centered at 470 nm means the severe recombination of charge carriers in the PCN.51 The MCN shows a markedly reduced peak intensity, and the PL peak in the OCN almost disappears. The quenching of the PL peak in the MCN and OCN is primarily attributed to the inhibited radiative recombination pathway due to the formation of a defect energy level below the CB.52 The synergistic contribution of the –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N group-related N defect and Na+ doping structure to the decay of PL strength in OCN is greater than the N vacancy alone to this process in MCN. Notably, the amplified PL curve of OCN can be fitted to two peaks. The former peak (P1) relates to the band-to-band recombination of the photoinduced electron–hole pair, and the peak position is red-shifted to 487 nm, consistent with the decreased Eg in the UV-vis DRS spectra. The latter one (P2) is associated with the excitation and recombination between the VB and the defect energy level. Time-resolved PL spectra clearly show the reduced average PL lifetime of OCN (1.80 ns) compared to the PCN (6.11 ns) and MCN (3.68 ns) (Fig. 7b). The significantly reduced average PL lifetime clearly suggests a higher charge separation and transfer efficiency in OCN, which are favorable for facilitating the charge carrier diffusion rapidly to the surface for subsequent photocatalytic redox reactions.53,54 The remarkable restrained radiative recombination and shortening PL lifetime in the OCN are ascribed to the emergence of new energy level-induced rapid electron–hole recombination processes.55 The detailed lifetime data and applied biexponential fitting are shown in Table S1.† The shorter lifetime τ1 stems from the radiative decay of exciton from CB or defect states to VB, while the longer lifetime τ2 is due to nonradiative decay of photogenerated carriers.56 In the MCN and OCN, their near-band-edge trap states receive and accumulate the photogenerated electrons transferred from the CB bottom in a bi-exponential relaxation manner, and the type and number of defects affect the trap depths.57 Shallow and deep trap states can be distinguished according to the energy difference between the band edge and trap state, and generally the shallow trap state electrons have good reactivity.58,59 The above-mentioned DFT result indicates that the midgap state in the OCN belongs to the shallow trap state. It is worth noting that the lifetimes τ1 and τ2 in the OCN are relatively shorter than those in MCN (τ1, 0.45 vs. 0.77 ns; τ2, 5.14 vs. 5.25 ns), implying relatively shallow depths of the trap state in the OCN.57 It means that the shallow trap state closer to the CB edge of OCN has a higher thermodynamic driving force for reduction reactions than the MCN. We therefore speculate that the OCN may have better electrochemical and photocatalytic properties than MCN. As depicted in Fig. 7c, the PCN exhibits typical band-to-band recombination (Path I), where photoexcited electrons return from the CB to the VB. However, in the OCN, the defect energy level acts as an electron trap state, capturing photogenerated electrons from the CB bottom and occurring carrier's recombination mainly via Path II (photoexcited electrons from trap states back to the VB). This is evidenced by the lower P1 peak intensity relative to P2 in the PL spectra. In addition, the smallest impedance arc radius in the electrochemical impedance spectra (EIS) and enhanced transient photocurrent response when under light illumination verify the high charge separation and transfer dynamics in the OCN, as shown in Fig. 7d and e. Electron paramagnetic resonance (EPR) was used to analyze the spin state of unpaired electrons, as illustrated in Fig. 7f. Compared with the PCN exhibiting no EPR signal, the MCN shows a high signal (g = 2.003). This is ascribed to the MCN synthesized at a high temperature that results in a more extended π-conjugated aromatic system with enhanced delocalization. Similar to the MCN, almost no change in g-value is observed in the OCN, but the slightly improved EPR intensity suggests the presence of more unpaired electrons and facilitates mobility within π-conjugated aromatic rings,60 which is mainly caused by the formation of a structure rich in –C
N group-related N defect and Na+ doping structure to the decay of PL strength in OCN is greater than the N vacancy alone to this process in MCN. Notably, the amplified PL curve of OCN can be fitted to two peaks. The former peak (P1) relates to the band-to-band recombination of the photoinduced electron–hole pair, and the peak position is red-shifted to 487 nm, consistent with the decreased Eg in the UV-vis DRS spectra. The latter one (P2) is associated with the excitation and recombination between the VB and the defect energy level. Time-resolved PL spectra clearly show the reduced average PL lifetime of OCN (1.80 ns) compared to the PCN (6.11 ns) and MCN (3.68 ns) (Fig. 7b). The significantly reduced average PL lifetime clearly suggests a higher charge separation and transfer efficiency in OCN, which are favorable for facilitating the charge carrier diffusion rapidly to the surface for subsequent photocatalytic redox reactions.53,54 The remarkable restrained radiative recombination and shortening PL lifetime in the OCN are ascribed to the emergence of new energy level-induced rapid electron–hole recombination processes.55 The detailed lifetime data and applied biexponential fitting are shown in Table S1.† The shorter lifetime τ1 stems from the radiative decay of exciton from CB or defect states to VB, while the longer lifetime τ2 is due to nonradiative decay of photogenerated carriers.56 In the MCN and OCN, their near-band-edge trap states receive and accumulate the photogenerated electrons transferred from the CB bottom in a bi-exponential relaxation manner, and the type and number of defects affect the trap depths.57 Shallow and deep trap states can be distinguished according to the energy difference between the band edge and trap state, and generally the shallow trap state electrons have good reactivity.58,59 The above-mentioned DFT result indicates that the midgap state in the OCN belongs to the shallow trap state. It is worth noting that the lifetimes τ1 and τ2 in the OCN are relatively shorter than those in MCN (τ1, 0.45 vs. 0.77 ns; τ2, 5.14 vs. 5.25 ns), implying relatively shallow depths of the trap state in the OCN.57 It means that the shallow trap state closer to the CB edge of OCN has a higher thermodynamic driving force for reduction reactions than the MCN. We therefore speculate that the OCN may have better electrochemical and photocatalytic properties than MCN. As depicted in Fig. 7c, the PCN exhibits typical band-to-band recombination (Path I), where photoexcited electrons return from the CB to the VB. However, in the OCN, the defect energy level acts as an electron trap state, capturing photogenerated electrons from the CB bottom and occurring carrier's recombination mainly via Path II (photoexcited electrons from trap states back to the VB). This is evidenced by the lower P1 peak intensity relative to P2 in the PL spectra. In addition, the smallest impedance arc radius in the electrochemical impedance spectra (EIS) and enhanced transient photocurrent response when under light illumination verify the high charge separation and transfer dynamics in the OCN, as shown in Fig. 7d and e. Electron paramagnetic resonance (EPR) was used to analyze the spin state of unpaired electrons, as illustrated in Fig. 7f. Compared with the PCN exhibiting no EPR signal, the MCN shows a high signal (g = 2.003). This is ascribed to the MCN synthesized at a high temperature that results in a more extended π-conjugated aromatic system with enhanced delocalization. Similar to the MCN, almost no change in g-value is observed in the OCN, but the slightly improved EPR intensity suggests the presence of more unpaired electrons and facilitates mobility within π-conjugated aromatic rings,60 which is mainly caused by the formation of a structure rich in –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N groups and Na+ coordination in the OCN. Taken together, the activated n → π* electron transition in the OCN greatly promotes the separation of photoinduced electron–hole pairs.
N groups and Na+ coordination in the OCN. Taken together, the activated n → π* electron transition in the OCN greatly promotes the separation of photoinduced electron–hole pairs.
3.5. Evaluation of the photocatalytic performance
Defect-induced broadening of light harvesting ability and midgap states are essential factors in determining the performance of photocatalysts. The photocatalytic behaviors were evaluated by photocatalytic H2 evolution under visible light irradiation (λ > 420 nm) in the presence of 1 wt% Pt and triethanolamine as the co-catalyst and hole sacrificial agent, respectively. As shown in Fig. 8a, the OCN exhibits the highest H2 production, reaching approximately 208 μmol after 4 h of irradiation. Cycling H2 generation tests were also performed, and only a slight decrease in H2 evolution is observed over five consecutive runs (20 h), indicating the relatively high stability of the OCN in the catalytic process (Fig. 8b). After the stability test, the structure, morphology, and compositions of the used OCN catalyst show negligible changes compared to the fresh sample (Fig. S6–8†), suggesting the good stability of OCN in photocatalytic H2 production. It can be noted that the BET-specific surface area of the OCN is not the highest and is even lower than that of the MCN. Thus, the surface area-normalized activity of those catalysts should be compared. The HER per unit surface area (Fig. 8c, red columns) was calculated by the average HER (Fig. 8c, gray columns) over the BET surface area of the PCN, MCN, and OCN. The normalized HER value for OCN is 85.5 μmol h−1 m−2, which is also higher than that of PCN (34.1 μmol h−1 m−2) and MCN (33.7 μmol h−1 m−2). This means that the performance improvement of OCN is mainly due to its enhanced optical features and other optimized charge carrier recombination and separation processes. The surface tension of the samples was investigated by the water contact angle test (Fig. S9†). Compared with the PCN, the MCN and OCN exhibit a smaller water contact angle, suggesting that their surface becomes hydrophilic and tends to adsorb water molecules. Moreover, we found that the performance of OCN can be tailored by calcining Na-melem at different temperatures, that is, when the temperature is 650 °C, the maximum HER reaches 1043 μmol h−1 g−1 (Fig. 8d). The UV-vis DRS spectra of OCN synthesized at different temperatures show that the change regularity of n → π* electronic transition is highly positive correlated with the increased tendency of temperature (Fig. S10†). This indicates that a reasonable polymerization temperature is the key to achieving the optimal relationship between the structure and the performance. To explore the effect of the activated n → π* electronic transition on photocatalytic activity, a wavelength-dependent H2 evolution production experiment was conducted using different monochromatic light irradiation (Fig. 8e). The decrease in photocatalytic activity in the OCN corresponds to the declining optical absorption curve with the increase in the wavelength, suggesting that H2 generation comes from energy band excitation. The apparent quantum efficiency (AQE) value of OCN reaches 3.17% at 420 nm, which is remarkably higher than that of previously reported Na+-doped carbon nitride photocatalysts (Table S2†). The lack of H2 evolution above 450 nm for PCN is mainly caused by its poor visible light absorption ability. OCN shows higher activity than the PCN and MCN at a wavelength greater than 450 nm, and even weak H2 production is detectable at 600 nm. These results demonstrate that the enhanced light harvesting corresponding to n → π* electronic transition can achieve processes of electron photoexcitation and participate in the subsequent reduction reaction.4 Conclusions
In summary, a novel orange carbon nitride has been successfully synthesized via a NaCl-assisted melamine assembly of melem, followed by a secondary calcination treatment, which exhibits enhanced photocatalytic H2 production performance. The Na-melem intermediates, obtained at low temperatures, present a hexagonal sheet rather than the typical rod-like morphology, mainly due to the insertion of Na+ that destroys periodic stacking between the interlayers. This strong correlation between the intermediate and the final product endows the derived carbon nitride with unique physicochemical and photoelectric properties. Remarkably, the n → π* electronic transition is activated in the carbon nitride, endowing this material with an extended light absorption edge of nearly 650 nm. Such activated n → π* electronic transition mainly originates from Na+ coordination and –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N groups that result in distorted and fluctuant conjugated structures. Experiments and theoretical calculations further demonstrate that the induced structural defects enable a midgap energy level that emerged below the CB, which effectively inhibits the band-to-band recombination of photogenerated charge carriers and realizes a rapid separation process.
N groups that result in distorted and fluctuant conjugated structures. Experiments and theoretical calculations further demonstrate that the induced structural defects enable a midgap energy level that emerged below the CB, which effectively inhibits the band-to-band recombination of photogenerated charge carriers and realizes a rapid separation process.
Data availability
The data supporting this article have been included as part of the ESI.†Author contributions
Lin Lei: methodology, formal analysis, software, writing – original draft. Yongbo Fan: investigation. Yuxin Jia: validation. Huiqing Fan: resources, funding acquisition. Weijia Wang: supervision, writing – review & editing. Haitao Huang: writing – review & editing.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by the National Nature Science Foundation (52372125 and 52333009), the Shaanxi Provincial Science Foundation (2021GXLH-01–11), the Yulin Project (2022-19-11), the High-Quality Patent Cultivation Project (XGD2021–04), the Fundamental Research Funds for the Central Universities (D5000230071), and the 111 Program of MOE of China (B08040). We would also like to thank the Analytical & Testing Center of Northwestern Polytechnical University.Notes and references
- M. G. Darmayanti, K. L. Tuck and S. H. Thang, Adv. Mater., 2024, 36, 2403324 CrossRef CAS PubMed.
- S. Qi, K. Zhu, T. Xu, H. Zhang, X. Guo, J. Wang, F. Zhang and X. Zong, Adv. Mater., 2024, 36, 202403328 Search PubMed.
- N. P. Dharmarajan, D. Vidyasagar, J. H. Yang, S. N. Talapaneni, J. Lee, K. Ramadass, G. Singh, M. Fawaz, P. Kumar and A. Vinu, Adv. Mater., 2024, 36, 2306895 CrossRef CAS PubMed.
- S. Hou, X. Gao, X. Lv, Y. Zhao, X. Yin, Y. Liu, J. Fang, X. Yu, X. Ma, T. Ma and D. Su, Nanomicro Lett., 2024, 16, 70 Search PubMed.
- C. Xu, H. Liu, D. Wang, D. Li, Y. Zhang, X. Liu, J. Huang, S. Wu, D. Fan, H. Liu and H. Pan, Appl. Catal., B, 2023, 334, 122835 CrossRef CAS.
- Y. Zhang, Q. Cao, A. Meng, X. Wu, Y. Xiao, C. Su and Q. Zhang, Adv. Mater., 2023, 35, e2306831 CrossRef PubMed.
- F. He, Y. Lu, Y. Wu, S. Wang, Y. Zhang, P. Dong, Y. Wang, C. Zhao, S. Wang, J. Zhang and S. Wang, Adv. Mater., 2024, 36, e2307490 CrossRef PubMed.
- J. Zhang, X. Liang, C. Zhang, L. Lin, W. Xing, Z. Yu, G. Zhang and X. Wang, Angew Chem. Int. Ed. Engl., 2022, 61, e202210849 CrossRef CAS PubMed.
- C. Wang, Y. Lu, Z. Wang, H. Liao, W. Zhou, Y. He, S. M. Osman, M. An, Y. Asakura, Y. Yamauchi, L. Wang and Z. Yuan, Appl. Catal. B Environ., 2024, 350, 123902 CrossRef CAS.
- L. Chen, C. Chen, Z. Yang, S. Li, C. Chu and B. Chen, Adv. Funct. Mater., 2021, 31, 2105731 CrossRef CAS.
- Y. Shao, X. Hao, S. Lu and Z. Jin, Chem. Eng. J., 2023, 454, 140123 CrossRef CAS.
- L. L. Liu, F. Chen, J. H. Wu, J. J. Chen and H. Q. Yu, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2215305120 CrossRef CAS PubMed.
- Y. Xu, M. Fan, W. Yang, Y. Xiao, L. Zeng, X. Wu, Q. Xu, C. Su and Q. He, Adv. Mater., 2021, 33, e2101455 CrossRef PubMed.
- G. Zhang, Y. Xu, C. He, P. Zhang and H. Mi, Appl. Catal., B, 2021, 283, 119636 CrossRef CAS.
- S. An, G. Zhang, K. Li, Z. Huang, X. Wang, Y. Guo, J. Hou, C. Song and X. Guo, Adv. Mater., 2021, 33, e2104361 CrossRef PubMed.
- X. Lin, H. Du, D. Jiang, P. Zhang, Z. Yu, H. Bi and Y. Yuan, J. Energy Chem., 2022, 65, 541–547 CrossRef CAS.
- S. Hu, D. Jiang, L. Gu, G. Xu, Z. Li and Y. Yuan, Chem. Eng. J., 2020, 399, 125847 CrossRef CAS.
- F. Guo, B. Hu, C. Yang, J. Zhang, Y. Hou and X. Wang, Adv. Mater., 2021, 33, e2101466 CrossRef PubMed.
- H. Yu, R. Shi, Y. Zhao, T. Bian, Y. Zhao, C. Zhou, G. I. N. Waterhouse, L. Z. Wu, C. H. Tung and T. Zhang, Adv. Mater., 2017, 29, 1605148 CrossRef PubMed.
- W. Li, Z. Wei, K. Zhu, W. Wei, J. Yang, J. Jing, D. L. Phillips and Y. Zhu, Appl. Catal., B, 2022, 306, 121142 CrossRef CAS.
- G. Zhao, G. Liu, H. Pang, H. Liu, H. Zhang, K. Chang, X. Meng, X. Wang and J. Ye, Small, 2016, 12, 6160–6166 CrossRef CAS PubMed.
- V. W. Lau, M. B. Mesch, V. Duppel, V. Blum, J. Senker and B. V. Lotsch, J. Am. Chem. Soc., 2015, 137, 1064–1072 CrossRef CAS PubMed.
- S. Chu, C. Wang, J. Feng, Y. Wang and Z. Zou, Int. J. Hydrogen Energy, 2014, 39, 13519–13526 CrossRef CAS.
- S. J. Makowski, P. Kostler and W. Schnick, Chemistry, 2012, 18, 3248–3257 CrossRef CAS PubMed.
- Z. Huang, F. W. Yan and G. Q. Yuan, ACS Sustain. Chem. Eng., 2018, 6, 3187–3195 CrossRef CAS.
- G. Zhang, L. Lin, G. Li, Y. Zhang, A. Savateev, S. Zafeiratos, X. Wang and M. Antonietti, Angew Chem. Int. Ed. Engl., 2018, 57, 9372–9376 CrossRef CAS PubMed.
- Y. Yang, S. Wang, Y. Jiao, Z. Wang, M. Xiao, A. Du, Y. Li, J. Wang and L. Wang, Adv. Funct. Mater., 2018, 28, 1805698 CrossRef.
- H. Gao, S. Yan, J. Wang, Y. A. Huang, P. Wang, Z. Li and Z. Zou, Phys. Chem. Chem. Phys., 2013, 15, 18077–18084 RSC.
- H. Li, B. Cheng, J. Xu, J. Yu and S. Cao, EES Catal., 2024, 2, 411–447 RSC.
- W. Wang, L. Du, R. Xia, R. Liang, T. Zhou, H. K. Lee, Z. Yan, H. Luo, C. Shang, D. L. Phillips and Z. Guo, Energy Environ. Sci., 2023, 16, 460–472 RSC.
- X. Wu, D. Li, B. Luo, B. Chen, Y. Huang, T. Yu, N. Shen, L. Li and W. Shi, Appl. Catal., B, 2023, 325, 122292 CrossRef CAS.
- Y. Wang, M. Sun, W. Tang, Q. Li, Z. Ren, Y. Liu, Y. Zhang, C. Zeng, Z. Wang, Y. Wu, J. Hao, X. Wu and R. Yang, Mater. Today Phys., 2022, 23, 100634 CrossRef CAS.
- Z. Li, Y. Zhou, Y. Zhou, K. Wang, Y. Yun, S. Chen, W. Jiao, L. Chen, B. Zou and M. Zhu, Nat. Commun., 2023, 14, 5742 CrossRef CAS PubMed.
- Y. Li, P. Li, J. Wang, Y. Yang, W. Yao, Z. Wei, J. Wu, X. Yan, X. Xu, Y. Liu and Y. Zhu, Appl. Catal., B, 2018, 225, 519–529 CrossRef CAS.
- B. Zhai, H. Li, G. Gao, Y. Wang, P. Niu, S. Wang and L. Li, Adv. Funct. Mater., 2022, 32, 2207375 CrossRef CAS.
- Q. Li, Y. Tong, Y. Zeng, X.-K. Gu and M. Ding, Chem. Eng. J., 2022, 450, 138010 CrossRef CAS.
- K. Zhang, C. Liu, Q. Liu, Z. Mo and D. Zhang, Catalysts, 2023, 13, 717 CrossRef CAS.
- J. Kröger, A. Jiménez-Solano, G. Savasci, P. Rovó, I. Moudrakovski, K. Küster, H. Schlomberg, H. A. Vignolo-González, V. Duppel, L. Grunenberg, C. B. Dayan, M. Sitti, F. Podjaski, C. Ochsenfeld and B. V. Lotsch, Adv. Energy Mater., 2020, 11, 2003016 CrossRef.
- D. B. Nimbalkar, V.-C. Nguyen, C.-Y. Shih and H. Teng, Appl. Catal. B Environ., 2022, 316, 121601 CrossRef CAS.
- R. L. Calabro, D. S. Yang and D. Y. Kim, J. Colloid Interface Sci., 2018, 527, 132–140 CrossRef CAS PubMed.
- S. Gao, X. Wang, C. Song, S. Zhou, F. Yang and Y. Kong, Appl. Catal., B, 2021, 295, 120272 CrossRef CAS.
- H. Wang, X. Sun, D. Li, X. Zhang, S. Chen, W. Shao, Y. Tian and Y. Xie, J. Am. Chem. Soc., 2017, 139, 2468–2473 CrossRef CAS PubMed.
- S. Tong, X. Zhang and P. Yang, Environ. Res., 2023, 236, 116805 CrossRef CAS PubMed.
- D. Jiang, S. Hu, Y. Qu, X. Tian, H. Du, C. Zhu, Z. Li, L. Yin, Y. Yuan and G. Liu, Adv. Funct. Mater., 2024, 34, 2311803 CrossRef CAS.
- Y. Kang, Y. Yang, L. C. Yin, X. Kang, G. Liu and H. M. Cheng, Adv. Mater., 2015, 27, 4572–4577 CrossRef CAS PubMed.
- J. Ran, T. Y. Ma, G. Gao, X.-W. Du and S. Z. Qiao, Energy Environ. Sci., 2015, 8, 3708–3717 RSC.
- H. Liu, X. Liu, C. Xu, D. Wang, D. Li, J. Huang, S. Wu, Z. Wang and H. Pan, J. Mater. Chem. A, 2024, 12, 9200–9211 RSC.
- Y. Jiang, L. Zeng, C. Cao, W. Yang, L. Huang and J. Mate, Sci. Technol., 2022, 111, 9–16 CAS.
- X. B. Chen, L. Liu, P. Y. Yu and S. S. Mao, Science, 2011, 331, 746–750 CrossRef CAS PubMed.
- C. Xu, P. Qiu, H. Chen and F. Jiang, Appl. Surf. Sci., 2020, 529, 147088 CrossRef CAS.
- H. Sun, Y. Shi, W. Shi and F. Guo, Appl. Surf. Sci., 2022, 593, 153281 CrossRef CAS.
- Y. Kang, Y. Yang, L. C. Yin, X. Kang, L. Wang, G. Liu and H. M. Cheng, Adv. Mater., 2016, 28, 6471–6477 CrossRef CAS PubMed.
- Y. Xu, X. He, H. Zhong, D. J. Singh, L. Zhang and R. Wang, Appl. Catal. B Environ., 2019, 246, 349–355 CrossRef CAS.
- S. Liu, Z. Wang, Y. Lu, H. Li, X. Chen, G. Wei, T. Wu, D.-J. Maguire, G. Ye and J. Chen, Appl. Catal. B Environ., 2021, 282, 119523 CrossRef CAS.
- J. Yuan, X. Liu, Y. Tang, Y. Zeng, L. Wang, S. Zhang, T. Cai, Y. Liu, S. Luo, Y. Pei and C. Liu, Appl. Catal. B Environ., 2018, 237, 24–31 CrossRef CAS.
- N. S. Han, H. S. Shim, J. H. Seo, S. Y. Kim, S. M. Park and J. K. Song, J. Appl. Phys., 2010, 107, 084306 CrossRef.
- H. Q. Xu, J. Hu, D. Wang, Z. Li, Q. Zhang, Y. Luo, S. H. Yu and H. L. Jiang, J. Am. Chem. Soc., 2015, 137, 13440–13443 CrossRef CAS PubMed.
- J. Jiang, C. Ling, T. Xu, W. Wang, X. Niu, A. Zafar, Z. Yan, X. Wang, Y. You, L. Sun, J. Lu, J. Wang and Z. Ni, Adv. Mater., 2018, 30, 1804332 CrossRef PubMed.
- R. Godin, Y. Wang, M. A. Zwijnenburg, J. Tang and J. R. Durrant, J. Am. Chem. Soc., 2017, 139, 5216–5224 CrossRef CAS PubMed.
- Y. Zheng, Y. Luo, Q. Ruan, S. Wang, J. Yu, X. Guo, W. Zhang, H. Xie, Z. Zhang and Y. Huang, Appl. Catal., B, 2022, 311, 121372 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4ta07246d |
| This journal is © The Royal Society of Chemistry 2025 |