 Open Access Article
Open Access ArticleDeveloping point-of-need (PON) cancer biosensors using microfluidic paper-based analytical devices: recent advances
Farnaz Bahavarnia†
a,
Fatemeh Nami†b,
Mohammad Hasanzadeh *c and
Nasrin Shadjou
*c and
Nasrin Shadjou d
d
aNutrition Research Center, Tabriz University of Medical Sciences, Tabriz, Iran
bAsian Nano Ink Co., Tabriz University of Medical Sciences, Tabriz, Iran
cPharmaceutical Analysis Research Center, Tabriz University of Medical Sciences, Tabriz, Iran. E-mail: hasanzadehm@tbzmed.ac.ir
dDepartment of Nanotechnology, Faculty of Chemistry, Urmia University, Urmia, Iran
First published on 9th December 2025
Abstract
Microfluidic paper-based analytical devices (µPADs) are considered a key solution as they offer a low-cost platform for developing point-of-need (PON) biosensors for cancer biomarker/cell detection. Nanomaterials utilized in the structures of µPADs and PADs provide powerful tools for the early-stage diagnosis of cancer. This review aims to summarize the recent advances in a variety of µPADs and PADs used for cancer diagnosis based on different types of nanomaterials, such as metal/metal oxide nanoparticles, magnetic nanoparticles, quantum dots, and 1–3 dimensional materials. This review also discusses the properties and functions of various nanomaterials, highlighting their recent applications in PAD- and µPAD-based diagnosis of cancer biomarkers and cells based on immune-, apta-, geno-, peptide-, and enzymatic-biosensing. Additionally, the critical role of detection techniques, like electrochemical, optical (UV-Vis spectrophotometry, SPR, SERS, and fluorometry), electrochemiluminescent, photoelectrochemical, and piezoelectric methods, in label-free and labeled bio-assays of cancer biomarkers are surveyed. The advantages and limitations of various sensing methods, techniques, types of materials and fabrication strategies are surveyed to explore the best cases for cancer diagnosis using PADs and µPADs. The roles of novel technologies such as AI and IOT, in the development of PAD- and µPAD-based cancer diagnosis are investigated. Finally, a comparison of the advantages and limitations of PADs and µPADs in the early-stage diagnosis of various cancers is reviewed towards exploring the research/technological gaps.
1. Introduction
Cancer is a disease in which cells grow abnormally and spread to other parts of the body. This disease can start in any part of the body and gradually affect other organs over time. Nanotechnology has revolutionized the diagnosis and treatment of cancer. Using nanoparticles, early detection, more precise targeting of tumors, and reduced side effects of treatment have become possible. Nanoparticles can act as drug carriers and deliver drugs directly to cancer cells without harming healthy tissues. Moreover, these particles are used in medical imaging to detect tumors in their early stages.1Nanoparticles can be used as contrast agents in imaging methods, such as MRI and CT scans, revealing tumors in the early stages when they are still small and treatable. They can identify specific biomarkers present on the surface of cancer cells, thus providing a more accurate diagnosis of cancer. Nanoparticles can interact with specific molecules present in cancer cells and identify the molecular changes associated with cancer.2 Nanomaterials have properties such as very large surface areas, specific chemical traits, and stability against biological modifications. The combination of nanomaterials with paper microfluidics provides many capabilities and efficiencies, including increased sensitivity and accuracy, design of advanced sensors and biomarkers, reduction in the required sample amount, development of multiplexed detection systems, finding targeted drugs, and drug control. For example, nanomaterials like gold nanoparticles, silver nanoparticles, carbon nanotubes, and nanofibers can identify biomarkers and reactions with high sensitivity. Gold nanoparticles are often used for labeling and color detection in rapid tests. Additionally, silver nanoparticles can be used to write sensitive and clear lines and as diagnostic markers.3
Paper microfluidics is an emerging technology that uses paper sheets to create microfluidic pathways for conducting biological, chemical, or biochemical reactions on a small scale with minimal sample volumes. These devices typically include channels, reservoirs, and place-to-place reagents where fluids move via gravity, capillarity, or pressure. The characteristics of paper microfluidics include ease of construction and use (requiring no special equipment and being easy to manufacture), low cost (with low production costs, making it suitable for mass use and in remote areas), sensitivity and rapid detection capability (able to identify biomarkers and chemicals in the shortest time), and portability due to their small size (lightweight and requiring no electricity or complex devices).4
Therefore, paper microfluidics is a simple, inexpensive, and fast technology that, when combined with nanomaterials, enhances sensitivity, accuracy, and multifunctionality. Its applications include rapid disease detection, biomarker detection, biosensor development, and multiplex tests, which have extensive uses in health, biotechnology, and medicine. In the context of cancer detection, the roles of these PADs and µPADs are identification for the detection of biomarkers/cells in low volumes of samples, early prediction and screening of these diseases, as well as application in personalized medicine and POCT. For example, PADs and µPADs can detect cancer-related biomarkers, such as proteins, DNA, RNA, or nanoparticles, in biological samples, like blood, saliva, urine, or other bodily fluids.5–7 This rapid and low-cost detection is particularly valuable in resource-limited environments. Additionally, by developing sensitive and specific tests based on this technology, cancer can be accurately detected in the early stages and before the appearance of obvious symptoms, leading to an increase in the success rate of treatment. Interestingly, these technologies enable rapid testing on-site, for example, in health centers in remote areas or in emergency situations, without the need for complex equipment and advanced laboratories. Therefore, PADs and µPADs led to a reduction in clinical and laboratory costs, ease of use and execution of tests by non-specialist staff, acceleration of the diagnosis process and rapid start of treatment. Therefore, PADs and µPADs have great potential for improving cancer detection and monitoring processes and can play significant roles as effective tools in the development of fast and affordable diagnostic systems.8–14 Methods for making paper-based sensors and paper-based microfluidic sensors are very popular due to their simplicity, low cost, and portability. Hereafter, the processes for manufacturing these types of sensors and related technologies are explained.
The detection mechanisms of PADs are based on several principles, including colorimetry, electrochemistry, and fluorescence. In a typical colorimetric biosensor, a paper strip is coated with a receptor that specifically binds to the target molecule of interest. The integration of PADs into POC settings has the potential to revolutionize cancer diagnosis and management, especially in resource-limited areas where traditional diagnostic tools may not be available. Rapid and accurate detection of cancer biomarkers using low-cost, portable devices can facilitate the early detection and monitoring of cancer in remote or underserved communities. However, challenges such as sensitivity, specificity, and reproducibility must be addressed before this technology can be widely adopted in clinical practice.15
It is important to point out that the detection of cancer biomarkers/cells using microfluidic paper-based sensors has primarily relied on several key methods, each leveraging different physical and chemical principles to achieve sensitive, specific, and rapid analyses. The main detection methods used in the bioassay of cancer biomarkers are summarized in Table 1, and their principle, advantages and common use cases were compared.
| Detection method | Principle | Advantages | Common use cases |
|---|---|---|---|
| Electrochemical | Charge transfer during biorecognition | Sensitive, portable, low cost, low detection limits, rapid response, cost-effective | Proteins, DNA, small molecules |
| Measures electrical signals, such as current, voltage, or impedance, resulting from biorecognition events (e.g., antibody-antigen binding) | |||
| Colorimetric | Visual color change, often enzyme-assisted | No electronic equipment, easy visualization | Tumor markers, like PSA and CEA |
| Simple, inexpensive, no need for external instruments, ideal for resource-limited settings | |||
| Fluorescent | Fluorescence emission quenching or enhancement | High sensitivity and specificity; suitable for multiplexed detection | Nucleic acids, exosomes, proteins |
| Biomarker binding induces or quenches the fluorescence of labeled probes, like quantum dots, carbon dots, or dyed molecules | |||
| Chemiluminescent | Light generated from chemical reactions | Quantitative biomarker analysis | |
| Electrochemiluminescent | Light emission from electrochemical reactions | Very sensitive, quantitative, multiplexing | DNA, proteins, tumor markers |
| Light emission from electrochemical excitation during biomolecular interactions | |||
| Photoelectrochemical | Photocurrent generated upon light irradiation | Low background, high sensitivity | Nucleic acids, proteins, metabolites |
| Light-induced charge transfer processes produce measurable photocurrents related to biomarker concentrations |
2. Methods for creating PADs and µPADs
2.1. Designing and cutting paper
• Choosing the type of paper: typically, special papers such as laboratory papers, foams, nanotubes, or other papers with suitable microfluidic properties are used.11,12• Cutting and designing: using scissors or laser cutting, channels, cavities, and reaction areas are shaped on the paper. This process is usually based on pre-drawn designs in design software.
2.2. Printing and imaging
• Printing methods such as inkjet printing with active materials, micro printing, or resin printing are used to create microfluidic channels.• Thermal or thermochemical printing is employed to create pressed or heat-sensitive pathways.
2.3. Coating and surface modification
• To control the flow of liquids, the surface of the paper may be coated with materials such as hydrophobic barriers. This is done by:• Typing resin or magnetic strips on paper.
• Using waterproof inks containing paraffin or silica, which create specific pathways.
2.4. Adding reagents and biomarkers
• In specific areas, reagents, enzymes, or nanoparticles are placed.• Sampling and rapid tests are conducted in these sections.
The fabrication of PADs and µPADs relies on methods designed to define microfluidic pathways, control liquid flow, and incorporate sensing on paper substrates. Cutting-based approaches, such as scissor cutting, craft plotting, and laser micromachining, physically shape paper into channels, reservoirs, and reaction zones by removing the material to form defined fluidic patterns. These methods are simple, rapid, and accessible, making them suitable for prototyping or low-cost laboratory settings. Printing-based techniques, including wax printing, inkjet printing, screen printing, and resin micro-printing, deposit hydrophobic barriers or functional materials directly onto the paper to generate flow-confined channels or reagent zones. Wax printing, for example, creates hydrophobic walls that penetrate through the paper when melted, while inkjet printing can deliver enzymes, nanoparticles, or DNA probes to specific sensing regions with high precision. Surface-modification methods apply polymers, silanes, nanoparticles, or paraffin coatings to adjust wettability, improve analyte capture, or prevent unwanted spreading of reagents, thereby enabling directional flow and enhanced analytical performance. Finally, reagent-loading strategies integrate biological recognition elements, such as antibodies, enzymes, aptamers, or colorimetric indicators, into predefined test pads, where they are dried and later rehydrated by the sample to trigger visible or electrochemical signals. Together, these fabrication approaches enable scalable, low-cost production of paper-based biosensors tailored for diverse diagnostic applications.
In essence, the fabrication of PADs and µPADs involves the design and cutting of paper, construction of channel pathways, addition of reagents and dyes, and stabilization of the structure (Fig. 1). These technologies are highly flexible, cost-effective, and scalable, making them very useful in medical, biotechnological, and environmental applications. Paper-based analytical devices (PADs) have emerged as promising diagnostic tools for cancer detection using biomarkers.7 Early cancer diagnosis through the development of accessible test platforms can significantly contribute to the prevention of disease progression. The fabrication of paper-based biosensors is relatively straightforward and can be performed using standard laboratory equipment.13 The use of paper as a substrate offers several advantages, including low cost, disposability, and biocompatibility. Additionally, paper is readily available and can be easily modified to incorporate various chemical or biological agents.14
Comprehensive reviews of various cancer biomarkers are available.16 Herein, we focus only on the identification of biomarkers based on paper microfluidics. This review first introduces nanomaterials and then analyzes their use in cancer diagnosis; it has covered many topics, including their electrochemistry, electrochemiluminescence, commercialization, manufacturing techniques, and microfluidic chips.17–20 As a departure from existing articles providing an overview of paper-based biosensors, our review examines studies in the field of microfluidic paper-based biosensors and discusses their public health implications, particularly regarding POC, point-of-need (PON), and telemedicine applications. This review highlights their potential benefits for patients and healthcare professionals, focusing on target biomarkers, production techniques, and measurement methods.
3. Road MAP
Biosensors encompass a wide range of platforms that convert biological interactions into measurable signals, enabling the sensitive and selective detection of analytes in medical, environmental, and industrial applications. Common types include electrochemical biosensors, which monitor current, potential, or impedance changes and are widely used for glucose monitoring and other point-of-care diagnostics; optical biosensors, such as surface plasmon resonance (SPR), fluorescence, Raman spectroscopy, and interferometric systems, which detect changes in light absorption, emission, or scattering and offer label-free, real-time analysis; piezoelectric and acoustic biosensors, like quartz crystal microbalance (QCM) devices, which measure mass or viscoelastic changes from biomolecular binding events; and thermal biosensors, which sense heat generated from biochemical reactions. Detection strategies vary from label-based approaches, which rely on fluorescent dyes, nanoparticles, or enzyme tags to amplify signals, to label-free techniques, which monitor intrinsic biomolecular interactions for faster, more direct analysis. Emerging trends, including nanomaterial-enhanced sensing, microfluidic integration, and AI-assisted data processing, have continued to expand biosensor sensitivity, multiplexing capability, and real-world clinical and environmental applicability.Beyond diagnostic naming, current efforts are increasingly focused on the following:
• Antibody- and biomarker-based targeting to enhance specificity and enable precision optical interrogation at cellular and molecular levels.
• Photon–matter interaction optimization, including multiphoton excitation, plasmonic enhancement, and nanoscale light delivery, to improve signal strength and biological compatibility.
• Bio-photonics integration, combining optical techniques with biological labeling, microfluidics, and advanced computational models.
• Instrument development that aligns with clinical workflow constraints, such as compact systems, fiber-based probes, real-time imaging, and AI-assisted interpretation.
These strategies are essential for advancing from laboratory demonstrations to tangible clinical translation. We intend to emphasize that the diagnoses are not stand-alone targets; rather, they represent clinically relevant contexts in which such combined innovations—instrumentation, molecular specificity, and advanced light–tissue interaction—are being actively developed and tested.
Scheme 1 illustrates the road map of this review article and the survey pathway of the investigation.
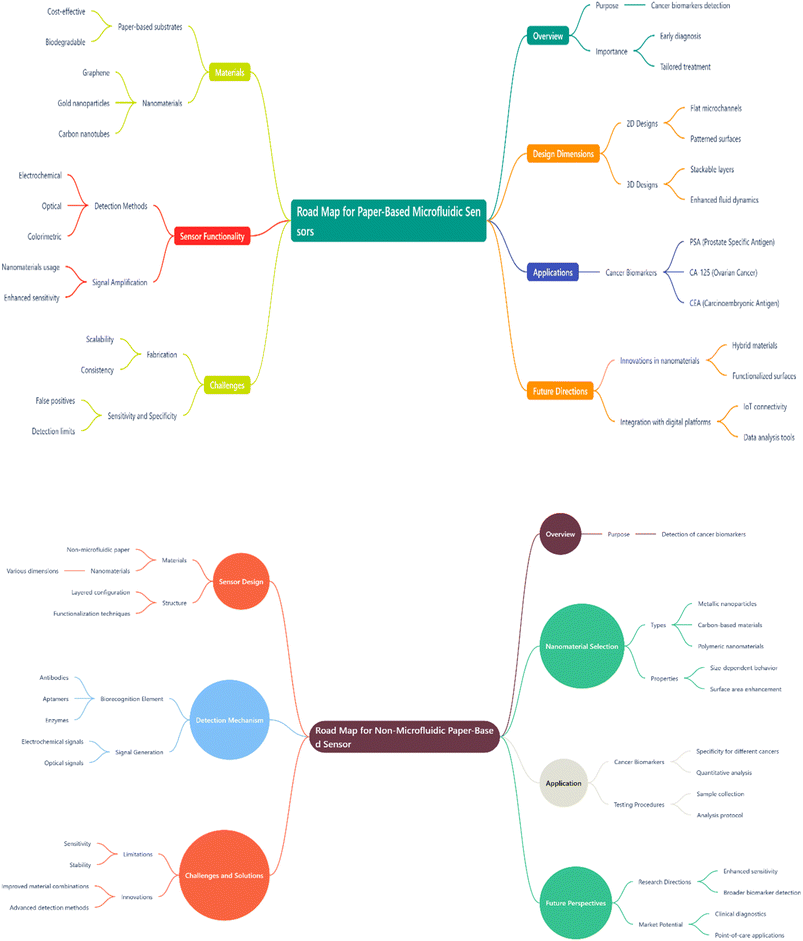 | ||
| Scheme 1 Roadmap of µPADs and PADs decorated with nanomaterials towards the early-stage diagnosis of cancer. | ||
4. Applications of nanomaterials in PADs and µPADs for cancer biomarker/cell detection
Tumor-associated biomarkers are crucial indicators of cancer presence and progression and are highly relevant to paper-based diagnostic platforms. Circulating tumor cells (CTCs), which disseminate from primary or metastatic tumors into the bloodstream, reflect the tumor burden and metastatic potential and can be captured using antibody-functionalized microfluidic interfaces. Tumor-derived extracellular vesicles, particularly exosomes, are nanoscale vesicles that transport tumor-specific proteins, lipids, and nucleic acids, and their stability and abundance make them attractive targets for affinity-based detection on paper substrates. Circulating nucleic acids—including circulating tumor DNA (ctDNA), which contains tumor-specific genetic and epigenetic alterations, and dysregulated microRNAs (miRNAs)—offer sensitive molecular signatures suitable for hybridization-based or nanoparticle-enhanced assays on paper devices. Additionally, overexpressed cell-surface receptors, such as HER2, EGFR, and folate, serve as direct recognition targets for aptamers or antibodies, enabling selective cancer cell identification. Collectively, these biomarkers enable minimally invasive, highly informative cancer detection strategies that align well with the low-cost, portable, and rapid analysis capabilities of paper-based analytical devices, strengthening their potential use in point-of-care oncology diagnostics.PADs and µPADs, as alternative platforms for POCT testing, have gained tremendous attention since they were first reported by Whiteside's group.21 Microfluidic tools have become important models for cancer investigation over the past decade.22 This technology has indicated a great potential for the design of novel methodologies for cancer diagnosis, monitoring, treatment, and disease follow-up. A variety of in vivo, ex vivo, and in vitro experimental models have traditionally been used to discover therapeutic targets and test novel drugs for cancer.23 As a result, microfluidic tools have the potential as a novel platform to carry out cancer research, monitoring and diagnosis.24 Therefore, the sensitive detection and selective identification of circulating cancer biomarkers are crucial steps in cancer clinical diagnostics. Microfluidic technology has been used in the past decade as a noteworthy device for cancer research.25 The technology of microfluidic systems provides a significant possibility for using sensor devices for a wide range of applications, such as clinical diagnosis, biological detection, and environmental monitoring.25–27 Microfluidic technologies combined with biosensors are more sensitive and precise in the detection of cancer biomarkers than classic platforms. In what follows, various nanomaterials that have been utilized for cancer biomarker detection are discussed.13,28 Due to the small sample volume used by µPADs for the detection of various target analytes, it is necessary to adopt methods for signal amplification. In µPADs, signal detection techniques are typically categorized into two main types: electrochemical and optical. Electrochemical devices utilize electrodes for measurement, while optical sensors employ visible measurement methods, including colorimetry, fluorescence, and chemiluminescence. This section of the review article summarizes the various techniques used for detecting different types of cancer with paper-based microfluidic sensors.7
Table 2 shows the broader applications of PADs and µPADs in different aspects of cancer biosensing. Additionally, Table 3 illustrates various aspects of µPADs and PADs.
| Application | Type of device | Biomarkers detected | Detection principles | Advantages |
|---|---|---|---|---|
| Tumor marker detection | Microfluidic paper-based electrochemical sensors | CA125 (ovarian cancer), PSA (prostate cancer), HER2 (breast cancer) | Electrochemical measurement based on immunoreactions | Portable, low-cost, rapid results, suitable for POCT |
| Nucleic acid detection | Paper-based nucleic acid sensors | Circulating tumor DNA (ctDNA), microRNAs | Isothermal amplification + colorimetric or electrochemical readout | High sensitivity, minimal equipment |
| Enzyme-linked assays | Colorimetric paper strips | Enzymes or metabolites associated with tumors | Enzyme–substrate reactions producing a color change | Simple, user-friendly, visual detection |
| Multiplexed detection | Paper microarrays | Multiple tumor markers simultaneously | Fluorescent or colorimetric signals | Comprehensive cancer profiling on a single platform |
| Aspect | µPADs | PADs |
|---|---|---|
| Control of fluid flow | Precise, programmable microchannels | Capillary-driven, passive flow |
| Detection types | Electrochemical, colorimetric, multiplexing | Primarily colorimetric, basic detection |
| Reagent use | Very low and controlled | Slightly higher, but still minimal |
| Sensitivity and specificity | Higher, due to controlled reactions | Usually lower, more variable |
| Ease of use | Moderate (requires preparation/handling) | Very easy, ideal for the rapid testing |
| Manufacturing complexity | High, requires advanced fabrication tools | Low, simple production techniques |
| Cost | Higher, especially for disposable microfluidics | Very low, suitable for mass production |
| Suitability for POC | Moderate to high, but a more complex setup | Excellent, especially in resource-limited settings |
| Multiplexing | Possible, integrated on a single chip | Limited, mostly single analyte |
Comparing PADs and µPADs based on their detection methods involves examining how each modality works, along with their respective strengths and weaknesses, which is important to select the best case for immunosensing of cancer biomarkers/cells. Therefore, we compare the advantages and limitations of detection methods used by PADs and µPADs for the detection of various types of cancer biomarkers/cells (Table 4).
| Type of detection method | Advantages | Limitation |
|---|---|---|
| Electrochemical | • High sensitivity and low detection limits: capable of detecting minute analyte concentrations (e.g., picomolar) | • Electrode fouling: nonspecific binding or biofouling can impair sensor performance over time |
| • Quantitative and selective: can be tailored with specific recognition elements | • Requirement for external electronics: though portable, needs a potentiostat or similar device | |
| • Rapid response time: usually provides real-time detection | • Environmental sensitivity: pH, ionic strength, and temperature can affect measurements | |
| • Low power consumption: suitable for portable, battery-powered devices | • Limited multiplexing: usually requires multiple electrodes for simultaneous detection | |
| • Compatibility with miniaturization: easy integration with microfabrication techniques | ||
| Optical detection (UV-Vis, fluorescence, and chemiluminescence) | • Simple visual readouts: UV-Vis allows for naked-eye detection, ideal for low-resource settings | • Interference from the sample matrix: turbidity, autofluorescence, or ambient light can affect accuracy |
| • High sensitivity (especially fluorescence and chemiluminescence): can detect very low analyte levels | • Need for dedicated optical devices: smartphones or portable detectors are often required for fluorescence or chemiluminescence | |
| • Multiplexing potential: multiple signals can be read using different wavelengths or fluorescence tags | • Photobleaching or signal decay: fluorescent signals can diminish over time | |
| • No external power for detection (especially for colorimetric): suitable for point-of-care use | • Limited quantitative accuracy: especially for colorimetric visual detection without proper calibration | |
| Electrochemiluminescence (ECL) | • High sensitivity: combines electrochemical control with luminescent detection, reducing background noise | • Requires specialized equipment: ECL detectors and power supplies can be bulky or expensive |
| • Low background interference: light is generated at the electrode surface | • Complex fabrication: incorporation of ECL reagents and electrodes is more challenging | |
| • Quantitative: offers good reproducibility and a broad dynamic range | • Limited portability: less suited for truly handheld, low-cost applications compared to pure electrochemical or colorimetric methods | |
| Photoelectrochemical (PEC) | • High sensitivity: combines optical and electrochemical detection modes for enhanced signals | • Complex device fabrication: requires integration of photoactive semiconducting materials |
| • Low background signal: light-controlled processes minimize noise | • Dependence on light source stability: needs consistent illumination sources | |
| • Potential for miniaturization: compatible with portable devices | • Environmental sensitivity: light intensity and sample conditions influence detection | |
| Piezoelectric | • Label-free detection: no need for labels or reporters | • Environmental sensitivity: temperature and humidity affect measurements |
| • Real-time monitoring: immediate response to analyte binding | • Limited miniaturization and integration: more complex than paper-based colorimetric or electrochemical sensors | |
| • High specificity: when combined with selective recognition elements (e.g., antibodies) | • Lower sensitivity for small molecules: better suited for larger analytes like cells or proteins |
In this review article, a general classification was made based on the dimensions of the types of nanomaterials used in the construction of both microfluidic and non-microfluidic paper sensors, as well as based on the type of bioreceptor, to achieve a roadmap. Then, practical examples of the application of various nanomaterials in the identification of different cancer biomarkers are discussed. After reviewing the examples, the performance of different sensors in this research area was analyzed. After examining the advantages and disadvantages, research gaps were identified, and necessary solutions were provided so that researchers and technologists could become familiar with the future of this technology in the field of early cancer detection.
4.1. Zero-dimensional nanomaterials
Zero-dimensional nanomaterials, which consist of small spherical particles at the nanoscale (typically less than 100 nanometers), play a very important and effective role in the development of cancer detection sensors. These nanomaterials are used in the design of sensitive and precise sensors to identify biological markers related to cancer due to their unique properties, such as high specific surface area, high chemical reactivity, and ability for physical and chemical manipulation. This leads to increased sensitivity and reduced false responses from the sensors. These nanoparticles can be combined with fluorescent, optical, or electrochemical labels, which enhance the signal when identifying biological markers related to cancer. For example, gold or silica nanoparticles may be used as labels in diagnostic systems. By utilizing the special properties of zero-dimensional nanomaterials, the amount of sample needed to identify cancer markers can be reduced, which is very important in sensitive and rapid diagnostic processes. These nanomaterials can provide results in a short time due to their quick reactions and higher detection power, which are crucial for early cancer diagnosis. Zero-dimensional nanomaterials are used in colorimetric, fluorescent, electrochemical, and optical sensors, each having specific features for detecting and measuring biological markers.29–31A recent study introduced an innovative origami-based electrochemical paper microfluidic device designed for enhanced specificity in vascular endothelial growth factor C (VEGF-C) diagnosis, an essential tumor biomarker for assessing cancer prognosis. Utilizing an indirect quantitative ELISA with a goat polyclonal antibody targeting human VEGF-C, the device demonstrated remarkable sensitivity, detecting VEGF-C within 0.01–100 ng mL−1, and the limit of detection was 10 pg mL−1. Although alternative methods, like pulsed resistive biosensors leveraging antibody-modified silver nanoparticles (AuNPs) and nanopipettes, provide simple POCT options, they face notable performance limitations. This highlights the demand for cost-effective, rapid devices for VEGF-C detection. The immunosensor mechanism involves the nanocomposite formation of VEGF-C with single-walled carbon nanotubes (SWCNT) and new methylene blue (NMB), followed by AuNP modification on the NMB/SWCNT nanocomposites, as detailed in Scheme 2.32
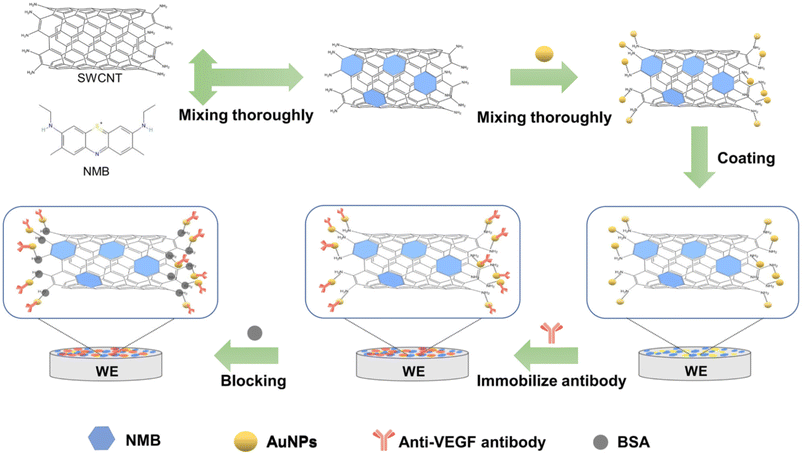 | ||
| Scheme 2 VEGF-C immunosensor based on gold nanoparticles (AuNPs) and NMB/SWCNT.32 | ||
Moreover, a study developed a sensitive, paper-based electrochemical immunosensor for carcinoembryonic antigen (CEA) detection using a screen-printed electrode (SPWE). Enhanced by NH2-functionalized graphene, thionine (Thi), and gold nanoparticles (AuNPs), it immobilizes anti-CEA antibodies and operates via a reduced thionine current in response to antibody-antigen binding (Scheme 3). With a linear detection ranging from 50 pg mL−1 to 500 ng mL−1 and a limit of detection of 10 pg mL−1, the sensor was validated with clinical serums, offering a cost-effective, accurate tool for cancer diagnosis at POC.33
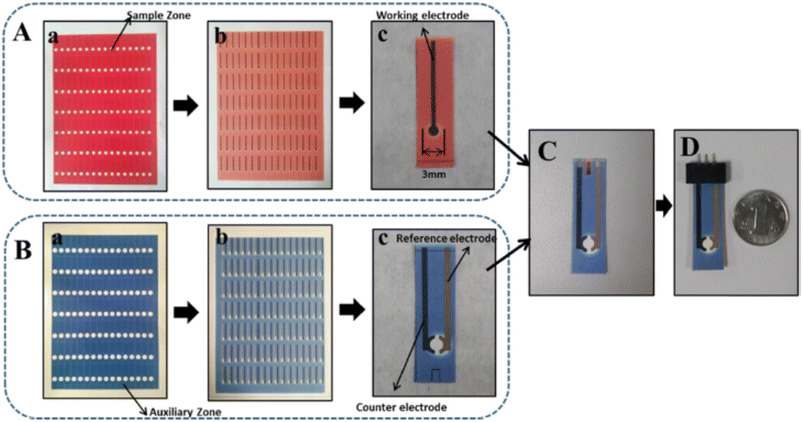 | ||
| Scheme 3 (A–D) Fabrication procedure of the paper-based device for sensing CEA.33 | ||
Researchers have developed a highly sensitive cancer biomarker detection method using gold nanoparticles (AuNPs) conjugated with biotinylated poly(adenine) ssDNA and streptavidin-horseradish peroxidase to enhance enzymatic signaling. This approach eliminates the need for complex, costly thiol-based AuNP modification and employs electrostatic interactions for antibody attachment, creating precise biomolecular recognition sites. Integrated into paper-based devices compatible with smartphones, this platform enables fast, simultaneous tests with minimal sample use, achieving a detection limit of 10 pg mL−1 for the biomarker in just 15 minutes. This innovative method holds promise for affordable, high-throughput diagnostics in resource-constrained settings.34
In a study, a three-dimensional paper-based analytical device, called the 3D-osPAD, was developed for the rapid and sensitive detection of anti-IFN-γ antibodies. This device employs an in situ gold signal amplification strategy, where Au3+ ions from HAuCl4 are reduced and deposited onto gold nanoparticles (AuNPs) to enhance the colorimetric signal. Diagnosing adult-onset immunodeficiency (AOID) related to anti-interferon-γ autoantibodies is challenging due to a lack of specific symptoms and detection methods. The 3D-osPAD simplifies the process by pre-drying the probe (AuNPs@IFN-γ) and reagents in separate cellulose layers. Using vertical microfluidic channels and patterned paper zones, reactions are automatically initiated with a single sample loading. This approach reduces the detection limit of anti-IFN-γ Abs from 0.1 µg mL−1 to 0.01 µg mL−1, achieving a tenfold increase in sensitivity. The entire process can be completed in 10 minutes, with clinical validation showing 100% sensitivity and specificity in distinguishing AOID patients from healthy controls. The 3D-osPAD effectively facilitates the monitoring of anti-IFN-γ antibody titers, enhancing clinical management and patient care.35
4.2. Magnetic nanoparticles
Zero-dimensional magnetic nanomaterials, which typically consist of micro or nanometer-sized particles with strong magnetic properties, play a very important and effective role in various sensing applications, including PADs and µPADs. PADs have become very popular for the quick and inexpensive detection of diseases, including cancer, due to their low cost, simplicity, and portability. Adding magnetic nanomaterials to the structures of PADs and µPADs significantly improves their performance by increasing sensitivity and selectivity, expanding the detection range, reducing sampling time, and simplifying processes. Magnetic nanoparticles act as targeting labels (such as binding to antibodies or antigens) due to their absorption capability and easy separation by magnetic fields. This allows these magnetic particles to be easily and accurately separated and identified when a sample contains cancer markers. These particles provide easy and rapid separation in detection processes, which in turn improves the overall efficiency of sensitive systems in detecting cancer markers.However, microfluidic-based sensors are based on small channels made from various materials and are highly suitable for biological detection and testing. The presence of magnetic nanomaterials in these systems has significant advantages. For example, by labeling biological markers with MNPs, cancer samples can be quickly and effectively separated within microfluidic channels before analysis occurs. In addition, MNMs enable detection systems to identify very small amounts of cancer markers due to their ability to absorb and concentrate biological markers, even in the presence of interfering substances. Interestingly, these particles can be controlled and guided by magnetic fields, allowing for the design of reactivated and highly sensitive systems in the form of small portable devices.
Therefore, zero-dimensional magnetic nanomaterials with strong magnetic properties, alongside biological and chemical properties, are very valuable tools for developing paper-based sensors and microfluidics in the early detection of cancer and other diseases. These materials increase sensitivity, speed, and efficiency and reduce testing costs, significantly improving the accuracy of diagnostic processes. In this section of the review, we survey the application of MNPs in PADs/µPADs for cancer diagnosis. Then, their advantages and limitations are discussed.36–38
For example, a study developed SERS paper chips by loading Ag/Mn3O4, Ag3PO4, and Ag3Cit onto Whatman filter paper (WFP), harnessing their strong oxidase mimicry and enhancing SERS properties. These chips generate SERS-active reporters (TMBox) via TMB oxidation, yielding robust SERS signals. Importantly, biothiols inhibit TMBox formation, diminishing the SERS signal, a phenomenon tied to their role in oxidative stress regulation and cell health. Each sensing channel produces distinct fingerprint variations reflecting the oxidative inhibition capacities of biothiols, enabling the accurate identification of six biothiols at concentrations as low as 1 µM through techniques like LDA and HCA. The array stands out because of its ability to determine intracellular GSH levels, discern normal cells from cancerous cells, identify tumor types, and broaden applications in diagnostics. Accordingly, designing biothiol-specific sensors using materials like Ag/Mn3O4, Ag3PO4, and Ag3Cit, with superior properties, offers exceptional significance for medical technology advancements.39
Researchers have developed a microfluidic paper-based fluorescent sensing device for the quantitative detection of microRNA (miRNA) and folate receptor (FR) expression on cell surfaces. Using a T-shaped double structure that dynamically self-assembles in response to stimuli, the device simultaneously detects two cancer biomarkers, proving its potential for biosensor development (Scheme 4). Additionally, layered MnO2 nanosheets exhibit superior quenching of fluorophore-labeled single-stranded DNA (ssDNA) due to lower adsorption by double-stranded DNA (dsDNA). This biosensor achieves miR-21 detection from 0.01 to 5.0 fM with a limit of 0.0033 fM and FR detection from 2.0 to 30.0 ng mL−1 with a 0.667 ng mL−1 threshold. These capabilities open new avenues for early disease diagnosis and therapeutic tools.40
 | ||
| Scheme 4 Microfluidic paper-based fluorescent sensing of microRNA (miRNA), and the assessment of folate receptor (FR) expression on the cell surface.40 | ||
A non-toxic method was developed to synthesize palladium nanoparticles (PdNPs) on Fe3O4@C, which act as peroxidase mimetics. These mimetics were employed in a µPAD-based multiplex colorimetric immunodevice, termed the α-sheet, for the site-specific immobilization of multiple antigens. The device uses flower-like gold nanoparticles to capture primary antibodies on paper zones, while Pd/Fe3O4@C labels secondary antibodies. In the presence of substrates like 3,3′,5,5′-tetramethylbenzidine and o-phenylenediamine, catalyzed reactions yield detectable colors (Scheme 5). A black wax-printed β-sheet with three observation windows overlays the α-sheet for precise color intensity analysis. The device, with a detection range of 0.005–30 ng mL−1 and a 1.7 pg mL−1 detection limit for CEA and α-fetoprotein (α-AFP), demonstrates potential as an affordable, effective tool for point-of-care diagnostics.41
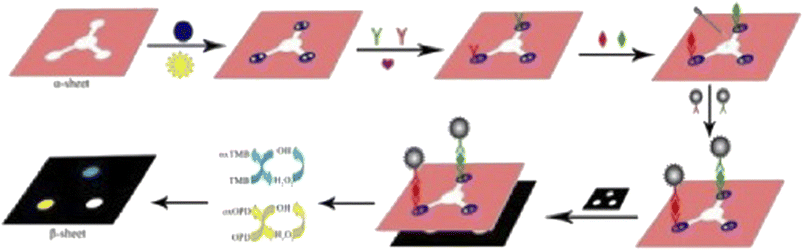 | ||
| Scheme 5 Microfluidic paper-based multiplex colorimetric immunodevice for CEA recognition.41 | ||
Microfluidic electrochemical arrays are revolutionizing cancer biomarker detection by offering rapid, cost-effective solutions with minimal instrumentation. A novel system features a 32-microelectrode array, which is enhanced by an 8-port manifold to support 256 sensors. Gold electrodes are crafted from commercial gold CD-Rs using wet-etching and patterned insulation. Using a print-and-peel method, sub-microliter hydrophobic wells around each sensor prevent cross-contamination during antibody immobilization. This setup (Scheme 6) enables 256 measurements in an hour by connecting eight 32-sensor immune-arrays to the manifold. Critical prostate cancer biomarkers (PSA, PSMA, IL-6, and PF-4) were detected in serum samples at clinically useful limits (0.05–2 pg mL; dynamic range over five decades). This innovative platform delivers fast and highly sensitive biomarker detection, advancing cancer diagnostics.42
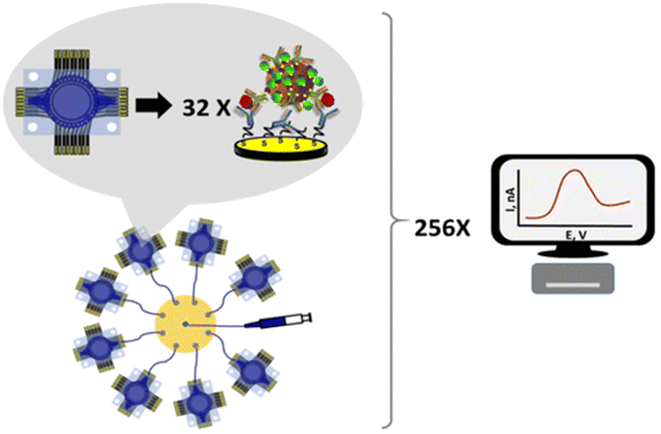 | ||
| Scheme 6 Microchip-based microfluidic electrochemical arrays for the detection of cancer biomarkers.42 | ||
A recent study introduced chemiluminescence (CL) immunoassays for the simultaneous detection of early biomarkers of acute myocardial infarction (AMI), including copeptin, heart-type fatty acid binding protein (h-FABP), and cardiac troponin I (cTnI). The system employed a Co2+-ABEI-functionalized magnetic carbon composite (Co2+-ABEI-Fe3O4@void@C) as its sensing interface and utilized a three-dimensional microfluidic paper-based device (3D µPAD) for detection. The platform (Co2+-ABEI-Fe3O4@void@C-CS/Au-Ab) combined the composite with chitosan and gold nanoparticle-loaded antibodies to form immuno complexes upon antigen binding, enhancing the CL signal through −COO– group catalysis. The 3D µPAD featured zones for the discrete detection of biomarkers, and time-resolved CL signals were triggered via the delayed delivery of hydrogen peroxide (H2O2) to each zone. This allowed for precise, simultaneous quantification of copeptin, h-FABP, and cTnI, with detection limits of 0.40, 0.32, and 0.50 pg mL−1, respectively, markedly exceeding prior detection efficiencies. Capable of use with human serum samples, the proposed method offers a straightforward, sensitive, and selective approach for early AMI diagnosis and management.5
In conclusion, MNPs have been widely utilized in PADs and µPADs for cancer biomarker detection due to several key advantages that enhance the performance, sensitivity, and functionality of these diagnostic platforms. The main reasons are as follows.
(I) Selective and efficient capture of biomarkers: magnetic nanoparticles can be functionalized with specific ligands, antibodies, or aptamers that selectively bind to target cancer biomarkers. This facilitates efficient and selective capture of the biomarkers from complex biological samples, such as blood, serum, or urine.
(II) Ease of separation and concentration: the magnetic properties of MNPs allow for the rapid and straightforward separation of bound biomarkers from the sample matrix using an external magnetic field. This reduces background noise, enhances detection sensitivity, and simplifies sample preparation.
(III) Reduced sample volume and time: magnetic separation minimizes the need for extensive washing and processing steps, enabling detection using smaller sample volumes and shorter assay times, which are crucial factors for point-of-care applications.
(IV) Improved sensitivity and signal amplification: magnetic nanoparticles can be used to carry signaling molecules or enzymes that generate measurable signals. Their magnetic nature also allows for the concentration of target biomarkers at specific locations on the paper device, boosting detection sensitivity.
(V) Compatibility with PAD-based platforms: PAD-based sensors are inherently low-cost, lightweight, and easy to handle. MNPs integrate well into these platforms because they can be manipulated with external magnets without disrupting the paper matrix, allowing for flexible assay designs and multiplexing.
(VI) Facilitating label-free and amplification strategies: MNPs enable label-free detection methods, like magnetic relaxation or impedimetric sensing, or can be used in conjunction with other amplification techniques to improve the limit of detection for cancer biomarkers.
(VII) Stability and storage: magnetic nanoparticles are relatively stable under various storage conditions, making them suitable for portable, on-site testing kits that are crucial for early cancer detection in resource-limited settings.
In summary, the combination of magnetic manipulation, high specificity, ease of use, and compatibility with PADs and µPADS makes magnetic nanoparticles versatile tools for developing efficient, sensitive, and low-cost point-of-care diagnostic tests for cancer biomarkers (Scheme 7).
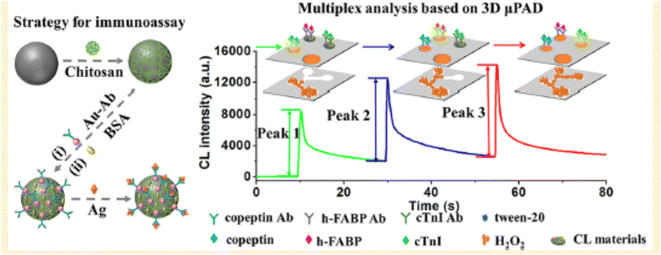 | ||
| Scheme 7 CL-based immunoassays of AMI biomarkers [h-FABP, cTnI].5 | ||
4.2.1.1. Carbon nanotubes (CNTs). CNTs have very important applications in various fields, including PADs and µPADs in cancer diagnosis. They have excellent electrical properties, so their use in the construction of channels in paper microfluidics can improve electrical conductivity and allow for better control of fluid flow. However, CNTs can contribute to the strength and integrity of paper structures, thereby increasing the durability and lifespan of microfluidics. Additionally, they act as catalytic agents and absorbers in paper of PADs and µPADs and facilitate the chemical processes necessary for detecting biomolecules or target substances. Therefore, CNTs, with their very large surface area and excellent electrochemical properties, enhance the sensitivity of PADs for detecting chemical and biological substances.43–45 CNTs can serve as carriers for the enzymes and biomolecules needed in biological sensors, leading to the increased stability and efficiency of the sensors.46,47 The presence of these nanomaterials accelerates chemical reactions in the sensors, reducing the response time. In addition, the use of CNTs in the manufacturing processes of paper sensors can simplify and make the production process more flexible while simultaneously reducing costs.48–50 Overall, CNTs play a key role in enhancing the performance of paper microfluidics and paper sensors due to their unique physical and chemical properties, making these technologies much more attractive and efficient for applications in the PAD- and µPAD-based diagnosis of cancer. In this section of the review, some examples of the application of PAD- and µPAD-based detection systems/strategies were surveyed.51–57
For example, a novel µPAD was developed for the rapid and sensitive quantification of colorectal cancer biomarkers, carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA 19-9) in human blood and serum. Anti-CEA and anti-CA 19-9 antibodies were covalently linked to fluorescent polystyrene particles, loaded into µPAD channels, and dried (Scheme 8). Upon sample introduction, immunoagglutination changed fluorescence scattering under ultraviolet (UV) light, enabling detection within one minute. CEA and CA 19-9 detection limits were 1 and 0.1 U mL−1, respectively. UV excitation improved double-normalized intensities (1.28–3.51) over Mie elastic scattering (1.067), ensuring high sensitivity even amidst serum or blood interference and allowing for multi-biomarker assays with minimal cross-reactivity. Serum stability enhanced accuracy and dynamic ranges due to protein stabilizing effects on embedded particles.58
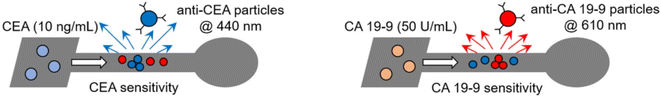 | ||
| Scheme 8 µPAD for the quantification of colorectal cancer biomarkers, carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA 19-9) in human blood and serum.58 | ||
Moreover, researchers have designed a novel electrochemical immunoassay leveraging a microfluidic paper-based device (µPAD) enhanced by multi-walled carbon nanotubes (MWCNTs). This three-dimensional electrochemical device (3D-µPED) enables sensitive dual detection of tumor markers in clinical serum, with linear ranges of 0.001–75.0 U mL−1 for cancer antigen 125 and 0.05–50.0 ng mL−1 for CEA (Scheme 9). Integrating with paper electronics, the 3D-µPED supports the development of affordable, disposable, and portable µPADs for point-of-care testing, public health, and environmental applications.59
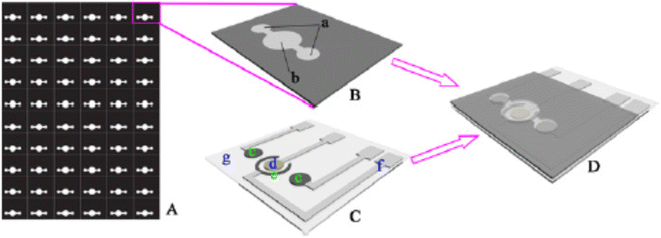 | ||
| Scheme 9 (A–D) 3D-µPED decorated with MVCNTs for the sensitive dual detection of tumor markers in clinical serum.59 | ||
Interestingly, scientists have developed an innovative micro-zone plate immunosensor using screen printing techniques to detect carcinoembryonic antigen (CEA). They immobilized a primary antibody on a paper zone with chitosan and porous gold for efficient antigen capture. A ZnFe2O4-multiwalled carbon nanotube-labeled antibody was employed to enhance detection, which facilitated the catalytic oxidation of TMB by H2O2, resulting in a distinct blue color change (Scheme 10). This simple method achieved a detection range of 0.005–30 ng mL−1 and a limit as low as 2.6 pg mL−1, demonstrating its potential to transform diagnostic applications.60
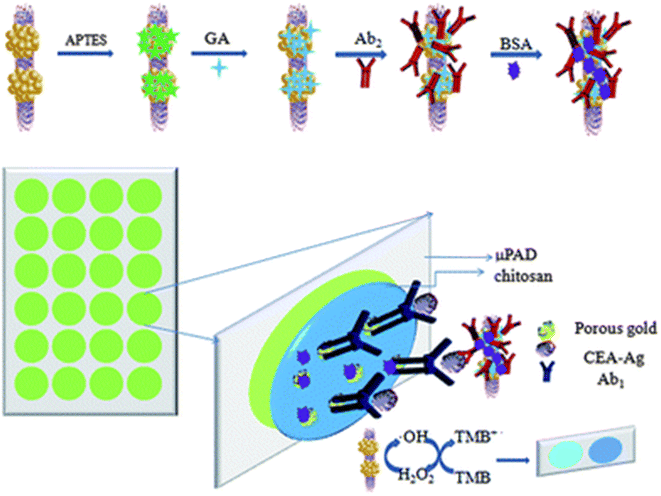 | ||
| Scheme 10 Micro-zone colorimetric immunosensor for the detection of CEA.60 | ||
Interestingly, a chemiluminescence (CL) immunoassay has been integrated into a cost-effective µ-PAD, creating a simple CL immunodevice, µ-PCLI. The device features a chitosan membrane-modified µ-PAD, where a captured antibody is covalently immobilized using a wax printing method. To boost assay performance, TiO2 nanoparticles on multiwalled carbon nanotubes (TiO2/MWCNTs) were synthesized as signal antibody (Ab2) catalysts. Following sandwich immunoreactions, TiO2/MWCNTs catalyzed the luminol-p-iodophenol-H2O2 CL system, enhancing emission (Scheme 11). Using prostate-specific antigen as a model analyte, this method achieved a linear response in the range of 0.001–20 ng mL−1 and a detection limit of 0.8 pg mL−1.61
 | ||
| Scheme 11 CL immunodevice based on µ-PCLI decorated with TiO2/MWCNTs towards cancer biomarker detection.61 | ||
Additionally, a study presents a cost-effective biosensor for early prostate cancer detection. It leverages bio-activated multi-walled carbon nanotubes (MWCNTs) with a micro-pore filter paper to target the PSA biomarker. Activated with anti-PSA monoclonal antibodies using advanced techniques (Scheme 12), the MWCNTs detect PSA levels ranging from 0 to 500 ng mL−1 in just 2 hours, achieving an impressive detection limit of 1.18 ng mL−1 through resistance changes. Capable of identifying early-stage cancer (above 4 ng mL−1), the biosensor is over 10 times faster than ELISA tests and boasts a detection sensitivity of nearly 50 times greater, positioning it as a transformative tool for early prostate cancer diagnosis.62
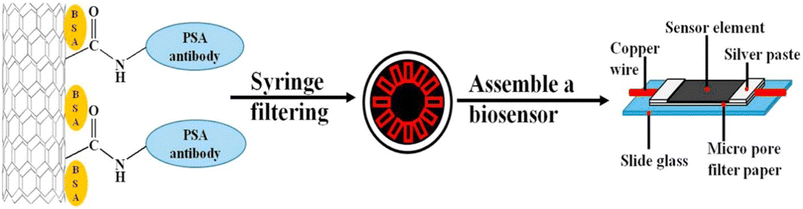 | ||
| Scheme 12 Micro-pore filter paper decorated with MWCNTs towards the sensing of PSA.62 | ||
Studies conducted on the utilization of CNT-modified PADs and µPADs in cancer biomarkers/cell detection have mainly focused on the following aspects:
(I) Increased sensitivity and reduced detection limit: investigating the effect of carbon nanotubes on the improvement of sensitivity in biological and chemical sensors, especially in detecting low-concentration substances.
(II) Enhanced electrical and catalytic properties: studying how to increase the electrical conductivity and catalytic capabilities of sensors, as nanotubes have superior properties in these areas.
(III) Stability and lifespan of sensors: examining how carbon nanotubes influence the compatibility, stability, and longer lifespan of paper sensors.
(IV) Applications in biomolecules and biosensors: developing and evaluating biological and platinized sensors for the detection of viruses, bacteria, and other biomolecules with the help of nanotubes.
However, why were minor reports published on the application of CNTs in PADs and µPADs?
The main reasons for the minor reports on the application of CNTs in PAD and µPAD structures for cancer diagnosis are as follows.
(I) Issues related to the production process and integration: incorporating carbon nanotubes into paper structures requires complex and controlled processes, which are more challenging compared to other materials (such as nanoparticles or simple conductive materials).
(II) Cost and scalability considerations: producing high-quality carbon nanotubes at commercial volumes is still costly and may have limitations in scalability.
(III) Safety and bioengineering limitations: there are concerns about the safety and toxicity of carbon nanotubes in biological and environmental applications, necessitating further studies.
(IV) Unknown responses to paper structures: the interaction of carbon nanotubes with paper structures and fluids in paper microfluidics is not fully understood and requires fundamental research.
Focus on other materials: most studies focus on nanoparticles, graphene, or other conductive materials that provide easier solutions or that are more widely used in the market. Overall, carbon nanotubes have great potential in the development of paper sensors, but challenges in manufacturing processes, cost, safety, and interaction with paper structures have limited research in this area. Technological advancements and reductions in production costs may fill this gap in the future, providing broader applications for CNTs in PADs and µPADs.
4.2.2.1. Graphene-based materials. The application of graphene compounds in the construction of paper sensors and paper microfluidic sensors is being developed and expanded because these materials possess unique properties that can significantly enhance the performance of these sensors.24,63–66 Graphene and its derivatives have very good electrical conductivity properties, which play a vital role in the development of electrochemical sensors, increasing sensitivity and measurement accuracy. Additionally, these materials have a very large surface area, which allows for surface modification and the occurrence of specific biological and chemical reactions, thereby enhancing sensor efficiency. In addition, graphene compounds possess good strength and stability, thus providing greater compatibility and durability in the structures of paper sensors. Finally, the ability to modify the surfaces of these materials for the transport of biomolecules, enzymes, or other sensing agents is very effective in improving the performance and specific characteristics of sensors.
Therefore, by adding graphene nanoparticles, paper sensors can detect target substances with high accuracy and sensitivity, even at very low concentrations. Interestingly, in electrochemical and photo electrochemical sensors, graphene compounds act as active electrodes that can enhance chemical and electrical processes. Moreover, the addition of graphene compounds allows for better transport, stabilization, and activity of biomolecules, resulting in more efficient and stable biological sensors. Due to their multiple properties, these materials enable the design of multifunctional and multi-purpose sensors that provide diverse responses to various substances. Therefore, graphene-based compounds, especially graphene and its derivatives, provide unparalleled opportunities to enhance the performance of PADs and µPADs, including increased sensitivity, reduced detection limits, and improved stability and lifespan of the sensors. The development of manufacturing technologies and surface modification of these materials could lead to broader applications in the fields of medicine, biotechnology, environment, and industry in the future, playing a significant role in the advancement of PAD- and µPAD-based sensing of cancer biomarkers/cells. Carbon-based materials are mainly used in PADs and µPADs for cancer biomarkers because of their exceptional electrical, chemical, and surface properties, which enable highly sensitive, stable, and low-cost detection. Their ease of functionalization and compatibility with diverse detection strategies make them ideal candidates for innovative point-of-care diagnostic devices aimed at early cancer detection.67–73 In this section of the review, some examples of PADs and µPADs decorated with graphene-based materials were surveyed.
For example, a study evaluated the application of phage-displayed recombinant antibody fragments as bio-detection elements in microfluidic paper-based devices (µPADs). Variable light (VL) and soluble VL antibody fragments were immobilized on chitosan-modified µPAD surfaces to detect glycine-linked gastrin 17 (G17-Gly), a key colorectal cancer biomarker. Detection probes consisted of phage shafts displaying single-chain variable fragments (scFvs) conjugated to AuNPs. A microfluidic sandwich immunoassay quantitated spot intensity using image analysis, yielding peptide calibration curves with linear signal-log concentration correlations. The detection ranges were 10−6–0.5 µM for phage-VL and 10−4–1 µM for soluble VL, with the limits of detection at 0.9 and 29 picomolar, respectively. This µPAD-based immunoassay provides a simple, amplification-free method for detecting biomarkers and pathogens with strong sensitivity.74
Researchers created an innovative paper-based biodevice using pen-on-paper technology, where Ag/rGO nanoink (silver nanoparticles/reduced graphene oxide) was handwritten onto flexible paper to detect the CA 125 protein in human fluids. Ag/rGO was produced by depositing silver nanoparticles on graphene oxide while simultaneously reducing it to rGO. To boost the device's performance, electrochemical methods were employed to modify the paper electrode with cystamine-coated gold nanoparticles (CysA/Au NPs), where cystamine bonds through sulfur atoms and attaches to Ag/rGO via amine interactions (Scheme 13). Anti-CA 125 antibodies were then immobilized on this surface. The device achieved a lower limit of quantitation (LLOQ) of 0.78 U mL−1 and a detection range of 0.78–400 units per mL, highlighting its sensitivity for early ovarian cancer detection.23
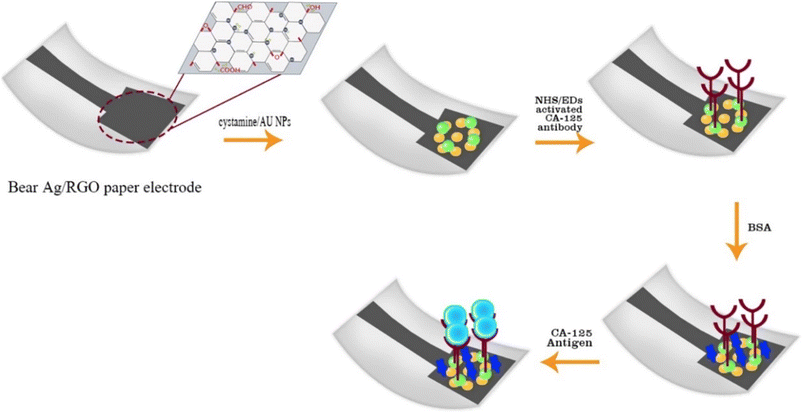 | ||
| Scheme 13 Electrochemical-based PADs for the detection of the CA 125 cancer biomarker.23 | ||
Additionally, a sensitive electrochemical immunoassay was developed using reduced graphene oxide (Ag/RGO) and silver nanoparticles to fabricate a paper-based immunosensor for detecting the CA15-3 tumor marker. This system, which is designed using an electrochemical approach, provides a high surface area for CA15-3 antibody immobilization (Scheme 14). The Ag-RGO/Cys-Au NP immunosensor showed optimal specificity and sensitivity, with a detection range of 15–125 U mL−1 and a quantification limit of 15 U mL−1. It effectively measured CA15-3 in raw plasma samples by leveraging Ag/RGO nano-ink and cysteamine-modified gold nanoparticles for signal enhancement and antibody binding.75
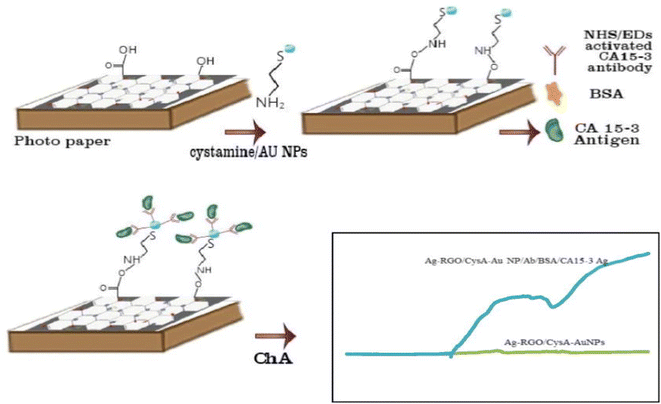 | ||
| Scheme 14 Paper-based electrochemical immunosensor for the detection of the CA 15-3 cancer biomarker.75 | ||
An innovative study improved immunoassay performance through graphene modification, leveraging its electrical conductivity for enhanced electron transfer and signal sensitivity. Silica nanoparticles served as labels for signal antibodies, reducing physical adsorption and boosting detection precision. This advanced method enables ultra-sensitive, multiplex detection of cancer biomarkers, like AFP, CA125, CA153, and CEA, directly at the point of care (Scheme 15). Using microfluidics, four capture antibodies were immobilized on electrodes, with differential pulse voltammetry (DPV) confirming assay specificity and minimal cross-reactivity. Serum tests revealed clear detection of biomarkers at concentrations as low as 0.001 ng mL−1 and 0.005 ng mL−1.76
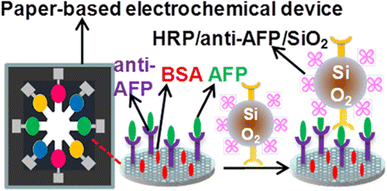 | ||
| Scheme 15 Multiplex detection of cancer biomarkers like AFP, CA125, CA153, and CEA using a paper-based non-microfluidic electrochemical sensor.76 | ||
Additionally, a novel signal-enhanced label-free electrochemical immunosensor using a gold nanoparticle/reduced graphene oxide (AuNPs-RGO) composite has been developed for neuron-specific enolase (NSE) detection. The composite is prepared using an eco-friendly method with chitosan (CS) serving as a reducing and stabilizing agent. Enhanced sensitivity is achieved through the AuNP-RGO nanostructure's multiple active sites (Scheme 16). The immunosensor demonstrates a linear response to NSE in the range of 0.1–2000 ng mL−1 and a detection limit of 0.05 ng mL−1. Optimized for rapid, selective, and sensitive detection, this technology facilitates advancements in clinical diagnostics, physiological studies, and analytical testing across diverse fields.77
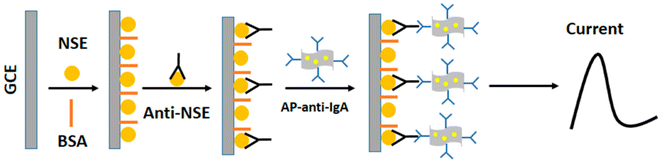 | ||
| Scheme 16 PADs for the electrochemical immunosensing of NSE.77 | ||
Interestingly, a study presented a paper-based electrochemical aptasensor for detecting CEA and NSE in clinical samples with high sensitivity and specificity. Utilizing wax and screen-printing techniques, the device enabled sample filtration and automatic injection, supporting the early diagnosis of lung cancer through simultaneous tumor biomarker detection. Amino-functionalized graphene-thionine-AuNPs and Prussian blue-poly(3,4-ethylenedioxythiophene)-AuNP nanocomposites were synthesized to enhance electron transfer and aptamer immobilization on modified electrodes. The label-free electrochemical method ensured rapid, simple point-of-care testing, with excellent linearity and detection ranges of 0.01–500 ng mL−1 for CEA and 0.05–500 ng mL−1 for NSE (Scheme 17). Limits of detection reached 2 pg mL−1 for CEA and 10 pg mL−1 for NSE, and tests using serum samples correlated well with large-scale ECL equipment. This platform offers potential for early cancer detection, especially in resource-constrained environments.78
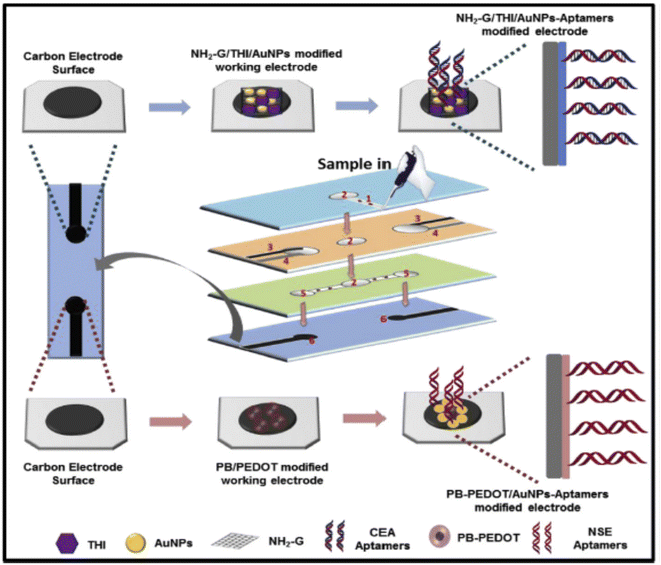 | ||
| Scheme 17 Multi-parameter electrochemical paper-based aptasensor for the detection of CEA and NSE biomarkers.78 | ||
Furthermore, a disposable and highly sensitive µPAD-based electrochemical immunosensor (µ-PEI) has been developed using reduced graphene oxide-tetra ethylene pentamine (rGO-TEPA) decorated with Au NPs as a functional electrode material. The rGO-TEPA enhances the current response due to its excellent conductivity and large surface area, while Au NPs improve biocompatibility and stability. This nanocomposite (rGO-TEPA/Au) provides an efficient platform for capturing antibodies (Ab1) and facilitating electron transfer through SPEs. A simple paper-based microfluidic channel integrated onto the SPEs/rGO-TEPA/Au device merges immunochromatography and immunofiltration (Scheme 18). For detection via square wave voltammetry, horseradish peroxidase-labeled signal antibodies with gold nanorods (HRP-GNRs-Ab2) act as tracers. When tested with AFP, the µ-PEI exhibited outstanding sensitivity, selectivity, stability, and a broad detection range (0.01–100.0 ng mL−1) with a remarkably low detection limit of 0.005 ng mL−1. Moreover, its application to human serum samples delivered highly satisfactory results.79
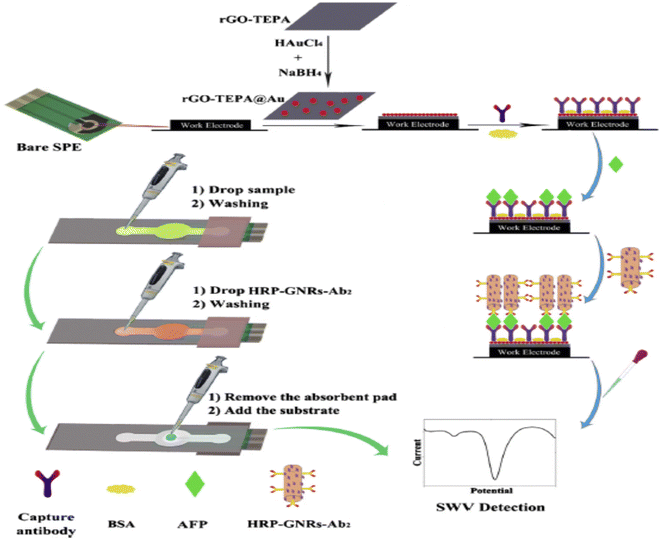 | ||
| Scheme 18 µ-PEI decorated with rGO-TEPA/Au towards the immunosensing of AFP.79 | ||
Interestingly, a PAD-based electrochemical immunosensor for detecting CA125 was developed using screen-printing technology. The working electrode was modified with reduced graphene oxide/thionine/gold nanoparticle (rGO/Thi/AuNP) nanocomposites to enable CA125 antibody immobilization and enhance signal sensitivity (Scheme 19). Detection was based on the reduction of thionine's current response caused by the antigen–antibody binding, which correlated to CA125 concentration. The sensor demonstrated a linear detection range of 0.1–200 U mL−1 with a limit of detection at 0.01 U mL−1. Showing high precision, stability, and accuracy, the immunosensor is promising for CA125 screening and potential point-of-care testing (POCT) for other tumor markers.80
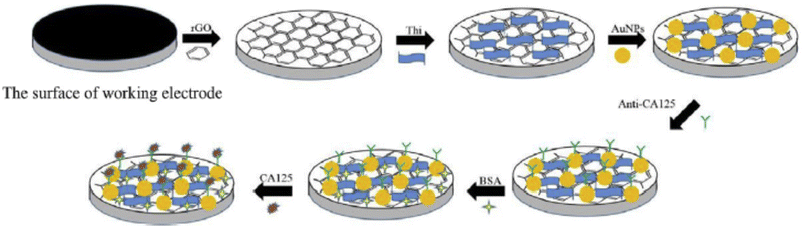 | ||
| Scheme 19 PADs modified with the rGO/Thi/AuNP nanocomposites towards the immunosensing of CA 125.80 | ||
The two-dimensional nature and high surface area of graphene and CNTs allow for abundant functionalization with recognition elements (antibodies, aptamers, and enzymes). In addition, this property provides more active sites for biomolecular interactions, thereby increasing sensitivity. The surface chemistry of carbon-based materials (e.g., carboxyl, hydroxyl, and epoxide groups) allows for the straightforward attachment of bio recognition molecules. Therefore, it facilitates the development of selective biosensors. These materials are generally inert, chemically stable, and biocompatible, reducing non-specific interactions and biofouling. However, they are easily available, inexpensive, and suitable for large-scale production. Carbon-based electrodes exhibit excellent stability over multiple cycles, making them reliable for repeated assays. Interestingly, carbon dots (C-dots) are especially useful in fluorescent sensing owing to their tunable emission properties. These materials are compatible with low-cost paper substrates, which are necessary for the construction of PADs. Finally, the high conductivity of these materials facilitates the sensitive electrochemical detection of biomarkers. Therefore, it enables small changes in charge or potential to be accurately measured, thereby enhancing detection limits. In conclusion, carbon-based materials, such as CNTs, graphene, GO, and rGO, are predominantly used in paper-based microfluidic and non-microfluidic sensors for the detection of cancer biomarkers because of their unique combination of properties, making them highly suitable for sensitive, flexible, and cost-effective detection platforms.
A recent study developed innovative µPADs using wax-printing for simultaneous fluorometric detection of lung cancer biomarkers, CEA and NSE. These devices incorporate two sensing zones linked to a sample area via microfluidic channels. Key advancements involved phase-engineering CsPbI3 perovskite quantum dots (IPQDs) modified with streptavidin and specific antibodies, enabling multiplexed sandwich immunoassays (Scheme 20). Offering benefits like low cost, portability, and simplicity, the µPADs achieved detection limits of 0.095 ng mL−1 for CEA and 30.0 ng mL−1 for NSE, with simultaneous detection limits of 0.12 and 32 ng mL−1, respectively. With low toxicity and high sensitivity from perovskite quantum dots, these devices present significant potential for detecting other biomarkers in diagnostics, environmental, and food safety research.87
 | ||
| Scheme 20 µPAD-based immunosensor for the detection of lung cancer biomarkers, CEA and NSE.87 | ||
Additionally, a novel 3D µPAD leveraging a GQD-based electrochemiluminescence (ECL) system was developed for the real-time visual detection of hydrogen sulfide (H2S) released from cancer cells. This innovative 3D origami cyto-device with hollow channels marks the first application for visual H2S quantification. The paper was functionalized with GQDs and integrated with Cu2+ and S2O82−. A macroporous Au@Pt-PWE modified with GQDs and aptamers enabled enhanced cell capture and signal amplification. The system exhibited fast response, robust stability, repeatability, a broad linear range, and low detection limits, successfully measuring H2S flux with cell concentrations ranging from 103 to 5.0 × 107 cells mL−1. Notably, after incubation with 1.0 × 105 cells mL−1 (10 µL), the ECL intensity reached 1518 a.u., estimating (1.07 ± 0.2) × 10−7 M H2S released per cell. In the proposed mechanism, Cu2+ initially inhibits GQD-S2O82− ECL activity, but H2S released by cancer cells binds Cu2+, enabling ECL signal generation through GQD-S2O82− interactions, which allows visual H2S sensing from cellular emissions.88
A study developed a PAD for the visual fluorescence detection of CEA by utilizing CdTe/CdSe QDs and a DNA-guided bioresponsive release system from mesoporous silica nanocontainers (MSNs). In the assay, glucose-loaded MSNs with CEA aptamers were combined with QD-enzyme-impregnated paper attached to a centrifuge tube lid. Complementary DNA was covalently conjugated to the aminated MSN, followed by aptamer-guided glucose encapsulation. Upon exposure to CEA, specific interactions opened MSN pores to release glucose, which was oxidized by glucose oxidase (GOD) on the QD-laced paper. This produced gluconic acid and hydrogen peroxide, leading to quenched QD fluorescence detectable by eye, smartphone, or fluorospectrometer (Scheme 21). The PAD achieved a linear detection range of 0.05–20 ng mL−1 and a sensitivity of 6.7 pg mL−1 (ppt) for CEA discrimination.89
 | ||
| Scheme 21 Fabrication and modification processes of the PAD for the fluorescence-based assay of glucose using CdTe/CdSe QDs.89 | ||
Although CQDs and GQDs hold substantial promise in biosensing due to their superior optical properties and biocompatibility, their application in µPADs for cancer biomarkers is still in the nascent stages. The key challenges include their stable and uniform integration onto paper substrates, maintaining signal clarity amidst biological matrices, achieving high sensitivity and specificity, and scaling up fabrication for widespread use. As ongoing research addresses these hurdles through improved nanomaterial synthesis, surface functionalization, and device engineering, we can expect an increase in reports and, eventually, practical applications in clinical diagnostics. The relatively few reports on the application of µPADs decorated with CQDs and GQDs for cancer biomarker detection stem from several interconnected scientific, technical, and practical challenges.
4.2.3.1. Novelty and emerging nature of CQDs and GQDs in biomedical sensing. CQDs and GQDs are relatively new materials in the domain of biosensing, especially for integration into paper-based microfluidic platforms. Although they have shown great promise due to their excellent photoluminescence, biocompatibility, and ease of functionalization, their application in this specific context is still emerging. Moreover, most studies have initially concentrated on their use in solution-based sensors, bio imaging, or electrochemical sensors, with fewer studies exploring their integration into paper microfluidic systems for complex biological samples.
4.2.3.2. Challenges in material integration and device fabrication. A consistent, stable, and reproducible decoration of paper substrates with CQDs or GQDs is technically challenging. Uniformly distributing these nanomaterials within microfluidic channels without aggregation or loss of activity requires advanced fabrication techniques. In addition, ensuring that QDs are stably embedded or coated on the paper without leaching or quenching their fluorescence during operation is difficult, especially in complex biological samples.
4.2.3.3. Photostability and signal interference. CQDs and GQDs are generally photostable compared to traditional fluorophores, but environmental factors (pH and ionic strength) in biological fluids can still affect their fluorescence. Furthermore, biological fluids and paper substrates can exhibit autofluorescence, which may interfere with quantum dot signals, complicating detection and quantification.
4.2.3.4. Sensitivity and selectivity challenges. Although CQDs and GQDs have excellent fluorescence properties, achieving the detection sensitivity needed for low-abundance cancer biomarkers in complex biological matrices remains difficult. Additionally, functionalizing CQDs or GQDs with selective recognition elements (antibodies and aptamers) that retain activity when integrated into the paper-microfluidic platform is yet to be extensively optimized for clinical relevance.
4.2.3.5. Biocompatibility and fouling. When used in biological samples, like blood or serum, non-specific adsorption of proteins and other biomolecules can quench or interfere with the fluorescence signals from CQDs/GQDs. In addition, although generally considered biocompatible, the safety and stability of certain CQD/GQD derivatives require thorough validation before clinical or widespread use.
4.2.3.6. Analytical and validation limitations. Most studies are preliminary and focus on proof-of-concept demonstrations rather than comprehensive clinical validation. Likewise, the standardization of nanomaterial fabrication and immobilization techniques is still evolving, limiting the reproducibility of the results across different labs.
4.2.3.7. Manufacturing and scalability constraints. Producing CQDs and GQDs with uniform quality at a scale and integrating them reliably into paper-based platforms remains a hurdle, affecting commercial viability. Although CQDs and GQDs can be synthesized relatively inexpensively, optimization and integration into complex device architectures increase overall costs.
4.3. Three-dimensional materials
Overall, MOFs are recognized as innovative and promising technologies in the field of cancer diagnosis and treatment due to their organized structure, ability to carry drugs and diagnostic materials, and customizable design. Ongoing research aims to improve the stability, wireless capabilities, and efficiency of these materials, opening new horizons in medicine and the treatment of cancer patients. In this section of this review, we surveyed various applications of MOFs for PAD and µPAD-based diagnosis of cancer biomarkers/cells.92
For example, a novel visible fluorescence immunoassay leverages a wet ammonia (NH3)-induced structural shift in NH2-MIL-125(Ti) coated on paper to detect a CEA biomarker. Gold nanoparticles functionalized with glutamate dehydrogenase (GDH) and a secondary antibody generate wet NH3 in a sandwich immunoassay setup. The NH2-MIL-125(Ti)-based PAD exhibits strong fluorescence triggered by the structural change, ensuring high accuracy and reproducibility (Scheme 22). This PAD allows for dual visible fluorescence detection, color- or physical-based, both discernible to the naked eye, enabling CEA detection at exceptionally low concentrations (0.041 ng mL−1). Promising for scalable production, this approach holds significant potential for advancing protein diagnostics and biosecurity.93
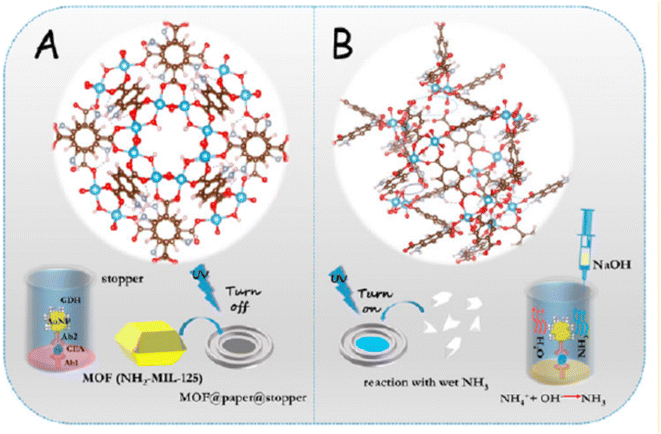 | ||
| Scheme 22 (A and B) Fluorescence immunoassay using NH2-MIL-125(Ti)-based metal–organic framework (MOF) impregnated on PADs towards the immunosensing of CEA.93 | ||
Additionally, a study developed a paper-based chemiluminescence (CL) device to assess the total phenolic content in food using a CL reaction with H2O2, rhodamine B, and a cobalt metal–organic framework (CoMOF). The CoMOF enhanced catalysis and stabilized the CL system for days. According to this report, adding 5 µL of phenolic compounds reduced CL intensity, enabling a straightforward analytical assay (Scheme 23). The device showed high sensitivity for gallic acid, quercetin, catechin, kaempferol, and caffeic acid, with detection limits of 0.98–2.55 ng mL−1.94
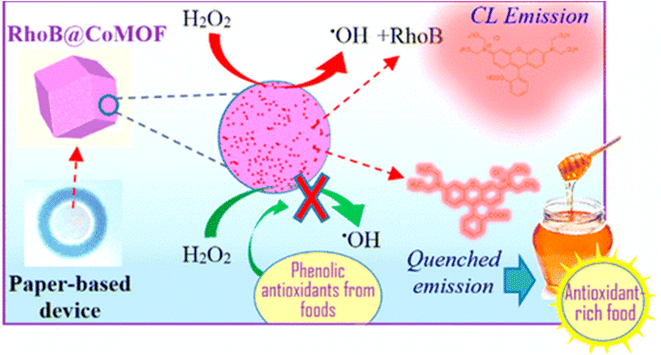 | ||
| Scheme 23 Paper-based CL device for the determination of phenolic compounds using the CL mechanism.94 | ||
Interestingly, a novel chromogenic reaction-free distance-based (CRFD) µPAD strategy was developed for the sensitive and accurate detection of microRNAs (miRNAs) via viscosity amplification and surface hydrophobicity modulation. Comprising sampling, indicator (red ink), and detection zones scaled with reference patterns, the µPAD quantifies miRNAs by pre-amplifying targets with rolling circle amplification and increasing viscosity. Flow differentiation between target and control samples is enhanced by modulating surface hydrophobicity using in situ MOF modification, thereby improving repeatability and precision (Scheme 24). As a proof-of-concept, miR-221 and miR-222 were detected directly by eye in liver cell lysates, with detection limits as low as 0.33 and 0.37 pM, respectively.95
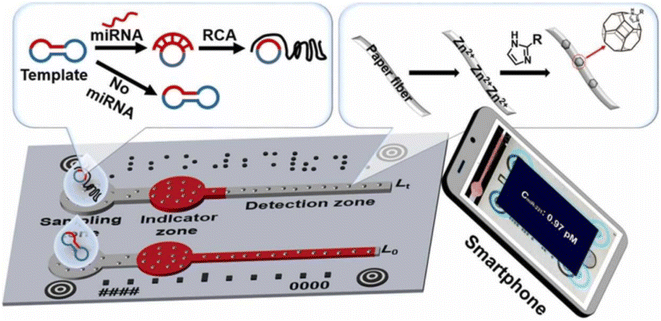 | ||
| Scheme 24 Chromogenic reaction-free distance-based (CRFD) µPAD for the sensitive and accurate quantitation of microRNAs (miRNAs).95 | ||
Although the potential of MOF-based PADs and µPADs in cancer detection is immense, they face critical challenges. Ensuring the chemical and structural stability of MOFs in biologically relevant conditions, such as varying pH levels and complex physiological media, is essential, as these factors can lead to hydrolysis and degradation. Additionally, biofouling from the non-specific adsorption of proteins and cells can undermine device performance, affecting sensitivity and reliability. Equally important is the assessment of MOF biocompatibility and potential toxicity, as leaching of metal ions may pose safety risks in clinical settings. The integration of MOFs into microscale channels also presents technical hurdles, including achieving uniform distribution and stable adhesion. Scalability and reproducibility in device fabrication remain significant obstacles compounded by the need for regulatory evaluation protocols for safety and efficacy. These challenges highlight the necessity for ongoing interdisciplinary research aimed at creating robust, biocompatible MOFs and standardized methods. Despite these issues, the integration of MOFs into microfluidic platforms signifies a transformative step in biomedical innovation. We are shifting from static materials to responsive, multifunctional systems that interact dynamically with biological environments. Advances in smart materials, additive manufacturing, artificial intelligence, and regulatory alignment promise to elevate MOF-based microfluidics, enhancing their sophistication and real-world applicability, ultimately leading to significant advancements in patient care.
For example, a study developed a portable and sensitive device for diagnosing pheochromocytoma (PCC) by detecting circulating tumor cells (CTCs). It integrates dual-mode microfluidic technology for electrochemical and visual analysis using a chip for automated cell sampling and a smartphone-based accessory for signal processing. PCC-CTCs are isolated via receptors specific to PCC cells, with targeted magnetic particles capturing them from blood samples. Nanozyme-enhanced covalent organic frameworks (COF@Pt) amplify the electrochemical signal and enable visual quantification. The device achieves a detection limit of 1 cell mL−1 and operates within a linear range of 2–105 cells per mL, offering a rapid, precise, and point-of-care solution for PCC diagnosis.100
In conclusion, there are no reports on the applications of MOFs, ZIFs, and COFs in the paper-based biosensing of cancer biomarkers. The relatively minor reports on the application of PADs and µPADs decorated with MOFs, ZIFs, and COPs for cancer biomarker detection can be attributed to several intertwined scientific, technical, and practical parameters, such as the complexity of material integration, synthetic challenges and cost, and limited understanding and optimization for biological samples.
Material compatibility: integrating MOFs, ZIFs, and COPs onto paper substrates, especially within microfluidic channels, requires careful surface modification and control of deposition techniques. This adds an extra layer of technical difficulty. In addition, maintaining the structural integrity and activity of these delicate or highly porous materials within a microfluidic and paper-based matrix remains challenging, particularly under operational conditions. Producing MOFs, ZIFs, and COPs with specific functional groups or pore structures suitable for selective biomarker detection can be laborious, often involving multi-step synthesis and precise control over crystallinity.
Cost considerations: the synthesis and functionalization of these advanced materials are usually expensive, which limits large-scale production and widespread application, especially in resource-constrained settings where paper-based sensors are the most advantageous. Additionally, MOFs and related frameworks may face issues with biofouling or nonspecific adsorption when exposed to complex biological fluids (blood, serum), which can hinder their effectiveness and reproducibility. Pore size and functionalization: tailoring these frameworks to selectively recognize cancer biomarkers at clinically relevant concentrations involves complex surface chemistry, which is still under active research.
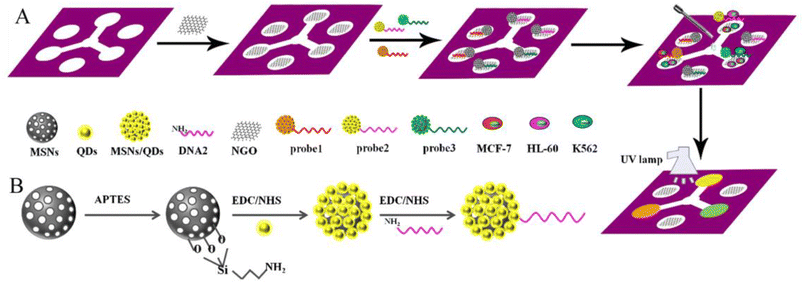 | ||
| Scheme 25 (A and B) MSN-GQs modified µ-PADs towards the biosensing of MCF-7, HL-60, and K562 cancer cells.105 | ||
Unfortunately, there are minor reports on the application of SMMs for the PAD- and µPAD-based sensing of cancer biomarkers and cells. The relatively limited number of reports on PADs and µPADs decorated with SMMs for detecting cancer biomarkers can be attributed to several interconnected scientific, technical, and practical factors, such as synthesis and functionalization challenges, complex fabrication, integration difficulty, material stability and compatibility, structural stability, and biocompatibility issues. Producing silica mesoporous materials with uniform pore size, high surface area, and suitable functionalization for biomarker recognition is synthetically demanding. Achieving consistent and stable deposition onto paper substrates within microfluidic channels adds further complexity. Incorporating SMMs into paper microfluidic platforms requires careful control to prevent pore clogging, uneven coating, or detachment, which can compromise sensor performance. Although silica is generally stable, mesoporous silica can be susceptible to collapse or pore blocking under certain conditions (e.g., in aqueous biological fluids), affecting its ability to serve as an effective sensor. Although silica is considered biocompatible, the surface chemistry of mesoporous silica needs to be tailored to prevent non-specific binding or biofouling in complex clinical samples, like blood or serum. However, functionalizing silica surfaces with specific recognition elements (antibodies and aptamers) while maintaining their accessibility within mesoporous channels requires precise chemistry, which can be challenging. Although pore size can enhance selectivity, it also restricts the diffusion of larger biomarkers or complexes, potentially reducing sensitivity or causing slower responses. It is important to point out that standard methods for integrating SMMs into paper-based microfluidics are less developed than those used for other nanomaterials, like CQDs or metal nanoparticles. Additionally, ensuring the long-term stability and reproducibility of these sensors in diverse conditions remains a challenge, slowing down extensive research and deployment. Therefore, the use of MSNs in PADs for biomarker detection is still an emerging research field. Many studies are at the proof-of-concept levels, focusing on fundamental properties rather than practical clinical applications. However, few studies have validated the sensitivity, selectivity, and reproducibility of these systems in real biological samples, which are critical steps before widespread adoption and reporting. Importantly, the synthesis and functionalization processes for mesoporous silica are generally more expensive than traditional detection materials. Producing large quantities of uniformly functionalized mesoporous silica at a low cost for paper-based devices is still an ongoing challenge.
In essence, although SMNs have highly attractive properties, such as high surface area, tunable pore sizes, and excellent functionalization potential, their integration into PADs and µPADs for cancer biomarker detection currently faces several synthetic, stability, and fabrication hurdles. These challenges have resulted in fewer reported studies so far. As research in nanomaterial synthesis, surface chemistry, and device engineering progresses, we may observe an increase in such applications, leveraging the unique advantages of SMNs for highly sensitive and selective cancer biomarker detection.
5. Utilization of smartphones for PAD- and µPAD-based cancer diagnosis
Smartphone-based PADs and µPADs for cancer diagnosis, particularly those with 3D designs incorporating folding and rotating structures, offer significant advantages, such as enhanced detection efficiency and reduced equipment costs. Typically, measurements are conducted using precision instruments to capture images, which are then analyzed using image processing software to assess the intensity of the color signals. Microfluidic paper-based analytical devices are often combined with scanners as detectors. The resulting images can be converted into various color spaces, including RGB (red, green, blue), CMYK (cyan, magenta, yellow, black), grayscale, HSV (hue, saturation, value), or CIE L*a*b. Among these color spaces, the RGB is the most commonly used due to its capability to produce a wide range of colors by superimposing three color channels. This makes its hardware implementation straightforward and simplifies subsequent image processing with software. The RGB color model has the advantage of not requiring any additional mathematical manipulation of the received data. This is because the software directly provides the average values of the components: red, green, and blue. Using the unmodified RGB model (without altering the raw data before analysis), the time required for analysis is reduced, and data processing becomes easier. Additionally, it simplifies the development of programs that can automatically convert RGB data into useful analytical information. Another commonly used color space for describing colors in digital images is HSV. This model is based on three components: hue (the color itself, measured in degrees from 0 to 360), saturation (the intensity or depth of the color), and value (the brightness of the color), all of which are expressed on a scale from 0 to 100%. The HSV model tends to be more intuitive for describing colors as it differentiates between chroma (pure color) and luma (brightness). However, it is more time-consuming and labor-intensive to use because it requires recalculating values from RGB to HSV. There is also a third option for image analysis: grayscale. However, it is rarely employed since it considers only luminance.A custom-made smartphone-based fluorescence microscope, automated image processing and particle counter software were developed to enumerate particles on paper, with a limit of detection of 1 cell per µL. This study introduced a two-layer paper microfluidic chip that offers a quicker, more cost-effective, and portable way to detect ROR1+ cancer cells in untreated buffy coat blood samples. Traditional leukemia diagnosis involves lengthy processes, like white blood cell counts, flow cytometry, and cytological analysis, requiring specialized training. The chip's first layer uses a GF/D glass fiber substrate with anti-ROR1 fluorescent particles for cell adsorption and imaging via a smartphone. The second layer includes grade 1 cellulose paper with wax-printed channels for capillary flow, enabling antigen detection and healthy cell analysis. Integrated with a smartphone-based fluorescence microscope and automated software, the device detects as few as one cell per microliter, achieving ultra-sensitive measurements of 0.1 cells per microliter in six seconds (Scheme 26). This enables the efficient imaging and quantification of ROR1+ cancer cells in buffy coat samples.106 Flow velocity analysis showed even greater sensitivity, with a limit of detection of 0.1 cells per µL in the first 6 s of the assay.
 | ||
| Scheme 26 (A–D) Smartphone-based fluorescence sensing of cancer cells.106 | ||
Table 5 compares the analytical performance of various types of paper-based non-microfluidic and microfluidic biosensors for cancer biomarker recognition using different analytical methods like electrochemical, optical, photoelectrochemical, and electrochemiluminescent methods.
| Detection method | Target analyte | Nanomaterial | Detection range | Detection limit | Advantages | Limitations | Ref |
|---|---|---|---|---|---|---|---|
| Chemiluminescence | Glycine-linked gastrin 17 (G17-Gly) | AuNPs | 10−6 to 5 × 10−1 µM for the phage-VL fragment and 10−4 to 1 µM for the soluble VL fragment | 0.9 and 29 picomolar | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 74 |
| Immunosensor | CA 125 | Ag/rGO, CysA/Au NPs | 0.78–400 units per mL | 0.78 U mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 23 |
| Fluorescence | MCF-7, HL-60 | Graphene oxide (GO)/MSNs | 180 to 8 × 107 cells per mL for MCF-7, 210 to 7 × 107 cells per mL for HL-60, and 200 to 7 × 107 cells per mL for K562 | 6270 cells per mL for MCF-7 and 65 cells per mL for HL-60 | The instrument has high sensitivity and selectivity, along with a low detection limit | Additional instruments are necessary to obtain quantitative results | 105 |
| Electrochemiluminescence | CA 125 protein | CysA/Au NPs/Ag-rGO | 0.78–400 units per mL | 0.78 units per mL | The device features high sensitivity and selectivity, a wide linear range, excellent reproducibility, and easy control | The process of manufacturing complicated devices and preparing materials | 23 |
| Electrochemistry | CA15-3 | Ag-RGO/Cys A-Au NPs | 15–125 U mL−1 | 15 U mL−1 | Quick sensor response, along with high sensitivity, selectivity, and stability | The fabrication process for the complicated device requires an electrochemical workstation | 75 |
| Surface-enhanced Raman scattering (SERS) | Biothiols | Ag/Mn3O4, Ag3PO4, and Ag3Cit | Not reported | 1 µM | Increased sensitivity, accurate molecular identification, and no need for bulky samples | Sensitivity to surface conditions, limitations in sample type, cost and complexity | 39 |
| Electrochemistry | AFP, CA125, CA153, and CEA | Silica nanoparticles | 0.001 ng mL−1 and 0.005 ng mL−1 | 0.1 M | Quick sensor response, along with high sensitivity, selectivity, and stability | The fabrication process for the complicated device requires an electrochemical workstation | 76 |
| Fluorescence | Carcinoembryonic antigen (CEA) and neuron-specific enolase (NSE) | (IPQDs) | 0.12 ng mL−1 and 32 ng mL−1 | 0.095 ng mL−1 for CEA and 30.0 ng mL−1 for NSE | The instrument has high sensitivity and selectivity, along with a low detection limit | Additional instruments are necessary to obtain quantitative results | 87 |
| Electrochemiluminescence | Hydrogen sulfide (H2S) | Au@Pt-PWE/GQDs | 1.0 × 103 cells per mL to 5.0 × 107 cells per mL | 1.0 × 105 cells per mL | The device features high sensitivity and selectivity, a wide linear range, excellent reproducibility, and easy control | The process of manufacturing complicated devices and preparing materials | 88 |
| Chemiluminescence | Carcinoembryonic antigen (CEA)and 19-9 (CA 19-9) | Not reported | Not reported | CEA was 1 pg mL−1, while for CA 19-9, it was 0.1 U mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 58 |
| Electrochemical immunosensor | Carcinoembryonic antigen (CEA) | MWCNTs | 0.001–75.0 U mL−1 for cancer antigen 125 and 0.05–50.0 ng mL−1 for CEA | Not reported | The device features high sensitivity and selectivity, a wide linear range, excellent reproducibility, and easy control | The process of manufacturing complicated devices and preparing materials | 59 |
| Immunosensor | Carcinoembryonic antigen (CEA) | ZnFe2O4 | 0.005–30 ng mL−1 | 2.6 pg mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 60 |
| Chemiluminescence | Cancer biomarker | TiO2/MWCNTs | 0.001–20 ng mL−1 | 0.8 pg mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 61 |
| Chemiluminescence | PSA | MWCNTs | 0–500 ng mL−1 | 1.18 ng mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 62 |
| Electrochemiluminescence | (NSE) | AuNP-RGO | 0.1–2000 ng mL−1 | 0.05 ng mL−1 | The device features high sensitivity and selectivity, a wide linear range, excellent reproducibility, and easy control | Additional instruments are necessary to obtain quantitative results | 77 |
| Fluorescence | Folate receptor (FR)/miR-21 | MnO2 nanosheets | miR-21 from 0.01 to 5.0 fM, FR 2.0 and 30.0 ng mL−1 | 0.0033 fM for miR-21, 0.667 ng/for FR | The instrument has high sensitivity and selectivity, along with a low detection limit | Additional instruments are necessary to obtain quantitative results | 40 |
| Immunosensor | Carcinoembryonic antigen (CEA) | (PdNPs) on Fe3O4@C | 0.005–30 ng mL−1 | 1.7 pg mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 41 |
| Electrochemiluminescence | PSA, PSMA, IL-6, and PF-4 | AuNPs | 0.05–2 pg mL−1 | 0.05 pg mL−1 | The device features high sensitivity and selectivity, a wide linear range, excellent reproducibility, and easy control | The process of manufacturing complicated devices and preparing materials | 42 |
| Chemiluminescence | AMI | Co2+-ABEI-Fe3O4@void@C | 0.40, 0.32 pg mL−1 | 0.50 pg mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 5 |
| Electrochemiluminescence | Growth factor C (VEGF-C) | Silver nanoparticles (AuNPs) and nanopipettes | 0.01–100 ng mL−1 | 10 pg mL−1 | The device features high sensitivity and selectivity, a wide linear range, excellent reproducibility, and easy control | The process of manufacturing complicated devices and preparing materials | 32 |
| Electrochemiluminescence | Carcinoembryonic antigen (CEA) | AuNPs | 50 pg mL−1 to 500 ng mL−1 | 10 pg mL−1 | The device features high sensitivity and selectivity, a wide linear range, excellent reproducibility, and easy control | The process of manufacturing complicated devices and preparing materials | 33 |
| Immunosensor | Cancer biomarker | (AuNPs) conjugated with biotinylated poly(adenine) ssDNA and streptavidin-horseradish peroxidase | Not reported | 10 pg mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 34 |
| Immunosensor | Anti-IFN-γ antibodies | AuNPs@IFN-γ | 0.1 µg mL−1 to 0.01 µg mL−1 | 0.01 µg mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 35 |
| Electrochemistry | Carcinoembryonic antigen (CEA) and neuron-specific enolase (NSE) | Graphene-thionine-gold nanoparticle (AuNP) nanocomposites and Prussian blue-poly(3,4-ethylenedioxythiophene)-AuNP nanocomposites | 0.01–500 ng mL−1 for CEA and 0.05–500 ng mL−1 for NSE | 2 pg mL−1 for CEA and 10 pg mL−1 for NSE | Quick sensor response, along with high sensitivity, selectivity, and stability | The fabrication process for the complicated device requires an electrochemical workstation | 78 |
| Immunosensor | Alpha-foetoprotein (AFP) | rGO-TEPA/Au | 0.01–100.0 ng mL−1 | 0.005 ng mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 79 |
| Immunosensor | CA125 | rGO/Thi/AuNPs | 0.1–200 U mL−1 | 0.01 U mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 80 |
| Fluorescence | Carcinoembryonic antigen (CEA) | CdTe/CdSe QDs | 0.05–20 ng mL−1 | 6.7 pg mL−1 (ppt) | The instrument has high sensitivity and selectivity, along with a low detection limit | Additional instruments are necessary to obtain quantitative results | 89 |
| Fluorescence | Carcinoembryonic antigen (CEA) | NH2-MIL-125(Ti)/Gold nanoparticles | Not reported | 0.041 ng mL−1 | The instrument has high sensitivity and selectivity, along with a low detection limit | Additional instruments are necessary to obtain quantitative results | 93 |
| Chemiluminescence | Gallic acid, quercetin, catechin, kaempferol, and caffeic acid | CoMOF | 0.98–2.55 ng mL−1 | 0.98 ng mL−1 | This instrument is simple to use, has a low background signal, offers high sensitivity and selectivity, and does not require an external excitation light source | A portable chemiluminescence reader is necessary for this process. Complicated device manufacturing (needs to be performed in the dark) | 94 |
| Chromogenic reaction-free distance-based (CRFD) | miR-221 and miR-222 | MOF | Not reported | miR-221 0.33 and miR-222 0.37 pM | Enhanced sensitivity and accuracy, elimination of chromogenic reactions, and versatility | Color fading and indistinct terminals. Differences in surface tension at the edges of the µPAD channels can cause the liquid to flow unevenly | 95 |
| Electrochemical | Circulating tumor cells (CTCs) | COF@Pt | 2–105 cells per mL | 1 cell per mL | Quick sensor response, along with high sensitivity, selectivity, and stability | The fabrication process for the complicated device requires an electrochemical workstation | 100 |
| Fluorescence | ROR1+ | Anti-ROR1-conjugated fluorescent particles | Not reported | 0.1 cells per microliter | The instrument has high sensitivity and selectivity, along with a low detection limit | Additional instruments are necessary to obtain quantitative results | 106 |
As shown in Table 5, various detection methods have been used in µPADs and PADs for cancer diagnosis, each possessing distinct characteristics. Among these methods, colorimetric techniques are the easiest to handle. Qualitative results can be detected with the naked eye, while semi-quantitative or quantitative results can be obtained using a colorimetric card or a smartphone. For electrochemical detection, a compact integrated electrochemical workstation can provide immediate results. Chemiluminescence and fluorescence methods have specific requirements concerning the detection environment. These methods necessitate addressing the effects of external ambient light and using specialized light-collecting equipment to gather and analyze optical signals. Although these methods are more complex to operate, they offer better sensitivity and higher detection accuracy. Additionally, other methods such as surface-enhanced Raman scattering, mass spectrometry, and magnetic detection have been applied in PADs and µPADs; however, these less common techniques are not the focus of this review. Table 5 lists some of the newly developed PADs and µPADs that have employed various detection methods in recent years. It is important to note that the progress of PADs and µPADs coincides with advancements in nanomaterials, primarily gold, silver, and carbon nanoparticles. Nanomaterials play a significant role across various detection methods due to their exceptional properties. In colorimetric methods, the distinctive color properties of nanomaterials can be utilized by modifying them on the analyte, resulting in color changes due to the accumulation of nanomaterials in the detection zone. For electrochemical methods, the excellent conductivity of these nanoparticles can be utilized by modifying the electrode surface and enhancing electrochemical performance. In chemiluminescence methods, nanomaterials can enhance signals through their catalytic properties. In fluorescence methods, nanomaterials may induce fluorescence quenching. Consequently, the development of nanomaterials has significantly advanced the capabilities of PADs and µPADs. Despite numerous teams devising various PADs and µPADs using different strategies to improve detection accuracy, only a limited number have been commercialized. The primary challenges include insufficient fluid control and the limitations of various detection methods. Thanks to the good biocompatibility of paper-based materials, PADs and µPADs have greatly reduced detection and sample attachment times compared to traditional microfluidic devices. However, since fluid transport relies on the capillary forces of paper-based materials, controlling the movement of the solution remains a significant drawback. It is important to point out that electrochemical and colorimetric detection modalities are the most commonly used in microfluidic paper-based cancer biomarker sensors due to their simplicity, rapid response, and suitability for point-of-care applications. Fluorescence and chemiluminescence methods are also popular in research settings owing to their high sensitivity and multiplexing capabilities.
Current µPADs utilize various approaches to construct hydrophilic or hydrophobic channels that transport solutions via capillary action. Paper-based sensors serve not only as the final reaction zone but also as the solution channel. Traditional microfluidic devices can effectively manage sample liquid transfer, but connecting the reactants with the substrate in the reaction zone often requires special measures. Therefore, combining traditional microfluidic equipment to establish a sample liquid transfer channel with paper materials as the final reaction zone can exploit the advantages of both device types.
Although numerous materials have been explored for constructing µPADs and PADs for cancer detection, including nanomaterials like nanoparticles, quantum dots, MOFs, and mesoporous silica, the field still has some gaps in the types of materials that have not been extensively used or incorporated. Here are some notable categories of materials that are generally underused or not yet reported in this specific context.
5.1. Conductive polymers (e.g., PEDOT, polyaniline, and PPy)
• Although conductive polymers are widely used in printed electronics and biosensors, their integration into paper-based microfluidic platforms for cancer biomarkers remains limited. They could offer advantages in electrochemical detection modes but are seldom combined with paper microfluidics for this purpose.5.2. 2D materials beyond graphene and transition metal dichalcogenides
• Materials like black phosphorus, phosphorene, or black arsenic have exceptional electronic and optical properties but have rarely been explored in paper-based systems for biomarker detection.5.3. Polymeric nanomaterials (e.g., polymeric microparticles and dendrimers)
• Although some polymeric nanoparticles are used in biosensing, their integration into paper microfluidic devices specifically for cancer biomarkers is limited. They can be functionalized for targeted recognition or signal amplification.5.4. MOFs besides traditional ones
• Although some MOFs have been explored, the use of newer MOF variants with tailored pore sizes and functionalities (e.g., zirconium-based Zr-MOFs and bio-MOFs) for paper-based diagnostics remains limited.5.5. Biopolymers (e.g., chitosan and cellulose derivatives)
• Although cellulose is the primary substrate, other biopolymers like chitosan or agarose are rarely used as functional coatings or layers within paper microfluidics for enhanced biorecognition or signal amplification.5.6. Inorganic nanostructures (e.g., metal oxide nanostructures)
• Materials like TiO2, ZnO, or CeO2 nanoparticles are well-known in sensing but are scarcely incorporated into paper-based platforms for direct cancer biomarker detection.5.7. MNPs
• Magnetic materials such as Fe3O4 nanoparticles can be useful for sample pre-concentration or separation, but their application in direct detection on paper microfluidic devices for cancer biomarkers remains underexplored.5.8. Biomimetic materials and peptide-based coatings
• These materials, mimicking biological receptors or enzymes, are not yet widely implemented in paper microfluidic settings for cancer biomarker detection but hold great potential for high specificity.In essence, while the field has explored a range of nanomaterials and coatings, several promising classes, including conductive polymers, advanced 2D materials beyond graphene, certain biopolymers, and inorganic nanostructures, are underrepresented or not yet widely used in the construction of paper-based microfluidic devices for cancer biomarker detection. Finally, Table 6 compares the advantages and limitations of µPAD and PAD sensors for the detection of cancer biomarkers.
| Type of sensor | Advantages | Limitations |
|---|---|---|
| µPADs | 1. Precise fluid control | 1. Manufacturing complexity: fabrication of microchannels and integration with detection elements require advanced techniques (e.g., photolithography) |
| 2. Microfluidic channels enable the controlled movement and mixing of small sample volumes | 2. Cost and scalability: higher production costs compared to simple paper strips | |
| 3. Enhances the accuracy and reproducibility of the assay | 3. Operational complexity: may need specialized equipment or user training for operation | |
| 4. Multiplexing capability | 4. Environmental sensitivity: microfluidic devices could be more susceptible to clogging or damage due to handling and environmental factors | |
| 5. Multiple detection zones can be integrated on a single device for the simultaneous analysis of various biomarkers | ||
| 6. Reduced reagent consumption | ||
| 7. Minimal reagent and sample volumes are needed, lowering costs | ||
| 8. Enhanced sensitivity and specificity | ||
| 9. Precise control over reaction conditions can improve detection limits | ||
| 10. Integration with electronic readouts | ||
| 11. Can incorporate miniaturized electronics for quantitative detection, such as electrochemical sensors | ||
| 12. Automation potential | ||
| 13. Allows for complex assay steps (e.g., fluid mixing, incubation) on a single platform | ||
| PADs | 1. Simplicity and ease of use: straightforward dipping or placement procedures, suitable for point-of-care settings | 1. Lower precision: less control over fluid flow and mixing, which can affect reproducibility and accuracy |
| 2. Low cost and rapid results: very inexpensive to produce and provide quick visual results | 2. Limited multi-analyte detection: usually designed for single analyte detection; multiplexing is challenging | |
| 3. Minimal equipment required: no need for external pumps, valves, or intricate fabrication techniques | 3. Sensitivity constraints: typically lower detection limits compared to microfluidic sensors | |
| 4. Robustness: less sensitive to environmental conditions, durable in field settings | 4. Qualitative or semi-quantitative data: often rely on visual interpretation; quantitative data require additional readout devices | |
| 5. Scalability: easy to mass-produce using simple printing or coating techniques |
6. Evaluation of applying PADs/µPADs to implantable devices, real-time monitoring, remote sensing and precision medicine
Paper-based analytical devices (PADs and µPADs) are well established as low-cost, disposable platforms for point-of-care testing and rapid biomarker assays, and there is increasing interest in extending their reach toward continuous, real-time, and precision-medicine applications. The current state-of-the-art suggests three realistic development tracks and several key limitations.6.1. Wearable/minimally invasive real-time monitoring (near-term and feasible)
Paper microfluidics have already been adapted into wearable formats for sampling sweat and other accessible biofluids and can perform continuous or repeated measurements with simple colorimetric or electrochemical readouts; hybrid devices that combine paper channels with flexible substrates and printed electronics can deliver near-real-time data streams suitable for monitoring physiological markers and patient adherence. This approach is supported by recent demonstrations of paper-integrated wearable sensors and molecularly imprinted paper devices for sweat analysis.6.2. Remote sensing/decentralized precision diagnostics (mid-term and promising)
PADs are already highly suited to remote, resource-limited screening (e.g., decentralized ctDNA/biomarker screening and antigen tests) because of low cost, simple operation, and compatibility with mobile-phone imaging or low-cost readers. Coupling PADs to cloud data pipelines and AI for pattern recognition could enable remote triage and longitudinal monitoring that feed into precision-oncology workflows (molecular tumor boards and therapy response monitoring). Recent reviews on paper-based cancer diagnostics and precision oncology underscore this translational pathway.6.3. Fully implantable continuous monitoring (longer-term and currently limited)
True implantable, continuous PADs for tumor monitoring face substantial technical and biological barriers. Implantable sensors demonstrated in the literature typically rely on miniaturized optical, electrochemical or MEMS technologies designed for biocompatibility, chronic stability, power supply, and tissue integration; proof-of-concept implantable systems for tumor-cavity monitoring have been reported, but these systems use robust implantable materials and optics rather than paper substrates. Translating paper-based chemistries into chronic implants requires solving biodegradation/sterility, fouling, immunogenicity, sealing against biofluids, and reliable in vivo readout/power, which are challenges that currently put implantable PADs at a low technology-readiness level compared with implantable BioMEMS and optical probes.7. Conclusion and future prospects
PADs and µPADs play a crucial role in miniaturized sensing systems, enabling large-scale laboratory operations to be performed on a single chip. Microfluidic systems are miniaturized devices where fluid behavior and control can be manipulated on a small platform, with surface forces often surpassing volumetric forces depending on the testing method used. In recent years, PADs and µPADs have been developed for use in POC technologies. PADs and µPADs offer numerous advantages, including ease of use, low cost, capillary action for liquid transfer without the need for power, the ability to store reagents in an active form within the fiber network, and the capacity to conduct multiple tests using various measurement techniques. These benefits are critical for advancing paper-based microfluidics in the fields of disease diagnosis, drug application, and environmental and food safety. Cancer is one of the most significant diseases requiring early detection worldwide. Identifying cancer-specific biomarkers offers essential data for both the early diagnosis and monitoring of disease progression. PADs and µPADs designed for cancer biomarker detection hold great promise for improving cure rates, enhancing quality of life, and reducing treatment costs. Although various bioanalytical platforms exist for detecting cancer biomarkers, there is a scarcity of studies and critical reviews focused on paper-based microfluidic platforms in the literature. Cancer is the second most prominent cause of death in developing countries. At present, due to the increasing number of cancer patients, cancer has attracted a lot of attention. Cancer nanotechnology holds promise in providing novel techniques for cancer detection in the initial stages, resulting in improved diagnosis and treatment. Conventional imaging techniques are exceptionally intrusive and non-specific and are frequently associated with toxicity to both tumor and solid cells. The advancement of novel nanomaterials has enabled the identification of cancer biomarkers with higher sensitivity and accuracy, which was not feasible previously. Steady and coordinated exploration endeavors should be embraced to utilize tremendous resources. Ultrasensitive biomarker sensing has been used to develop novel strategies for detecting early-stage cancer. Due to their unique characteristics, i.e., ease of synthesis, conductivity and bio-compatibility, MNPs are exceptional nanomaterials in theranostics. NPs have shown promise in cancer diagnosis and treatment due to their capacity to target specific cells and tissues. However, their cell specificity, ideal pharmacokinetics and bio distribution, and drug release management are obstacles. The way nanoparticles interact with biological systems can be influenced by their size, shape, surface charge, and chemistry. Toxicology, clearance, scalability, stability, control, and bio distribution require further investigation. Long-term safety, efficacy, reproducibility, and cost-effectiveness are also essential for nanoparticle-based therapies.In the future, PADs and µPADs structures and modified nanocomponents will undoubtedly have more innovative breakthroughs and novel PADs and µPADs will be developed in conjunction with traditional materials to establish sample liquid transmission channels that evolve new design functions.
Smartphone-based PADs and µPADs for cancer biomarker detection have gained considerable interest due to their potential for portability, low cost, and ease of use. However, they also have certain limitations. Table 7 compares the advantages and limitations of these smartphone-based PADs and µPADs for cancer biomarkers/cell sensing.
| Advantages | Limitations |
|---|---|
| Portability and POC accessibility | Limited sensitivity and quantitative accuracy |
| • Smartphones are ubiquitous and portable, enabling on-site testing outside traditional laboratories | • Smartphone cameras may be affected by lighting variations, image stability, and sensor quality, leading to inconsistent or less accurate measurements |
| • Facilitates rapid decision-making in clinical, remote, or resource-limited settings | Quantitative analysis can be challenging, especially at very low biomarker concentrations |
| Cost-effectiveness | Environmental and lighting dependence |
| • Utilizes existing smartphone hardware (camera, light source, processing power), reducing the need for expensive laboratory equipment | • Variations in ambient light, shadows, or camera focus can impact image quality and detection accuracy |
| • Paper-based platforms are inexpensive, making the overall system affordable | • Requires standardized imaging conditions or auxiliary accessories (e.g., boxes and lenses) |
| Ease of use | Hardware variability |
| • Simplifies operations through user-friendly apps that guide sample handling and data acquisition | • Differences in smartphone models (camera resolution, sensor sensitivity, and processing power) result in variability across devices |
| • Suitable for non-specialist users, including patients, community health workers, or clinicians in resource-limited areas | • Standardization and calibration are required for consistent results |
| Real-time data acquisition and analysis | Limited capabilities for complex assays |
| • Enables immediate digital acquisition of images or signals | • Certain assays (e.g., requiring fluorescence detection, microfluidic control, or complex spectral analysis) may be difficult to fully implement solely through smartphone imaging |
| • Facilitates instant processing, analysis, and storage of results | • Additional external components (filters and light sources) may be needed |
| Data sharing and remote monitoring | Sample preparation and integration |
| • Data can be wirelessly transmitted to healthcare providers | • Sample handling, preparation, and reagent delivery might still require manual steps outside the smartphone platform, potentially complicating point-of-care use |
| • Supports telemedicine and remote diagnostics | • The integration of complex fluidic manipulations is limited compared to laboratory automation |
| Multiplexing capability | Data security and privacy concerns |
| • Wireless data transmission raises issues related to patient privacy and data security | |
| • Potential to analyze multiple biomarkers simultaneously via multi-channel detection or advanced imaging and image analysis algorithms within the app | Regulatory and validation challenges |
| Required extensive validation, regulatory approval, and standardization to ensure reliability and accuracy comparable to laboratory standards |
In conclusion, smartphone-based PADs and µPADs for cancer biomarkers offer significant potential for accessible, real-time, and low-cost diagnostics, especially in resource-limited settings. However, challenges related to sensitivity, standardization, environmental sensitivity, and assay complexity need to be addressed to realize their full clinical utility.
Based on the studies discussed above, it is anticipated that the integration of smart devices into our daily lives can significantly reduce the burden on the healthcare system. Microfluidic paper-based analytical devices (µPADs) are expected to lower mortality rates by facilitating early cancer diagnosis. These devices are recyclable, portable, inexpensive, user-friendly, and suitable for use in resource-limited settings. Despite numerous studies focusing on cancer biomarkers, there are currently no widely used paper-based microfluidic kits in practice. Further research is needed to identify the advantages and disadvantages of paper-based µPADs, as well as to develop them into commercially viable kits. Large-scale studies involving real patient samples should be conducted to enhance the reproducibility and response times of these tests. Moreover, the interpretation of the results must align better with the needs of on-site diagnostic tests. Device designs should be simplified to ensure that they are more understandable and user-friendly for end users.
In light of the studies mentioned above, it is predicted that the health burden can be reduced if smart devices are more integrated into our lives. µPADs that are reusable, portable, inexpensive, field deployable, deployable in places with limited resources and user-friendly are expected to reduce mortality rates by enabling early diagnosis of cancer.
Currently, despite numerous studies on cancer biomarkers, no paper-based microfluidic kits are widely available in the field. Further research on the advantages and disadvantages of µPADs and their design as commercial kits is needed to understand their limitations and determine solutions. Large-scale studies with real patient samples should be conducted to improve the reproducibility and response times of the tests. In addition, the interpretation of results should be more in line with the requirements of on-site diagnostic tests, and device designs that are more understandable and simpler for the end user should be favored.
The remote diagnosis and treatment of patients using telecommunications technology (Telemedicine) is a technological development that promotes and ensures remote medical care through electronic platforms that enable disease detection and patient monitoring. The data collected using a wearable biosensor or other biosensors is captured by a smartphone/laptop and sent to a cloud, where it can be stored, processed and retrieved. Eventually, the data can be stored on another device with special applications. Its application on smartphones makes it an important object of study, as it enables the transfer of data from patient to doctor or doctor to patient in a portable, easy-to-use way. The field of telemedicine is on the rise, particularly in the area of cancer, in countries where access to healthcare for technologically disadvantaged populations is patchy. Given the cancers that have significantly impacted the health of the world's population in recent years, the presence of an application such as telemedicine has been of great importance.
Given the recent advances in paper-based technology, two other aspects deserve attention. Most devices provide only probabilistic results due to a general lack of selectivity. Finally, most approaches require the collection of blood or other biological fluids and thus the expertise of medical professionals.
Conflicts of interest
There is no conflict to declare.Data availability
Access to the data used in this study is available upon request and may be subject to approval by the data provider. Restrictions may apply to the availability of these data, which were used under the license for this study. Interested parties are encouraged to contact the corresponding author for further information on accessing the data.Acknowledgements
We appreciate the financial support provided for this work by the Pharmaceutical Analysis Research Center at Tabriz University of Medical Sciences, Tabriz, Iran (75827).References
- H. Sung, J. Ferlay, R. L. Siegel, M. Laversanne, I. Soerjomataram, A. Jemal and F. Bray, Ca-Cancer J. Clin., 2021, 71, 209–249 CrossRef.
- S. Sriharikrishnaa, P. S. Suresh and S. Prasada K, in Optical Polarimetric Modalities for Biomedical Research, Springer, 2023, pp. 307–330 Search PubMed.
- W. R. Algar, M. Massey, K. Rees, R. Higgins, K. D. Krause, G. H. Darwish, W. J. Peveler, Z. Xiao, H.-Y. Tsai and R. Gupta, Chem. Rev., 2021, 121, 9243–9358 CrossRef CAS.
- A. Singh and M. M. Amiji, Curr. Opin. Biotechnol., 2022, 74, 241–246 CrossRef CAS.
- R. Yang, F. Li, W. Zhang, W. Shen, D. Yang, Z. Bian and H. Cui, Anal. Chem., 2019, 91, 13006–13013 CrossRef CAS PubMed.
- K. Wisniewska, E. Marku, M. V. Ugurbas, I. Hartmane and M. Shukurova, Healthcare in Low-resource Settings, 2024, 12(s2), 12831 Search PubMed.
- B. Asci Erkocyigit, O. Ozufuklar, A. Yardim, E. Guler Celik and S. Timur, Biosensors, 2023, 13, 387 CrossRef CAS.
- E. Noviana, T. Ozer, C. S. Carrell, J. S. Link, C. McMahon, I. Jang and C. S. Henry, Chem. Rev., 2021, 121, 11835–11885 CrossRef CAS.
- R. K. Kankala, H. Zhang, C. G. Liu, K. R. Kanubaddi, C. H. Lee, S. B. Wang, W. Cui, H. A. Santos, K. Lin and A. Z. Chen, Adv. Funct. Mater., 2019, 29, 1902652 CrossRef CAS.
- S. Hassanpour and M. Hasanzadeh, Microchem. J., 2021, 168, 106424 CrossRef CAS.
- F. Precazzini, S. Detassis, A. S. Imperatori, M. A. Denti and P. Campomenosi, Int. J. Mol. Sci., 2021, 22, 1176 CrossRef CAS.
- R. V. Devi, M. Doble and R. S. Verma, Biosens. Bioelectron., 2015, 68, 688–698 CrossRef.
- Z. Liu, Y. Zhou, J. Lu, T. Gong, E. Ibáñez, A. Cifuentes and W. Lu, Biomark. Res., 2024, 12, 1–29 CrossRef.
- M. Costa, B. Veigas, J. Jacob, D. Santos, J. Gomes, P. Baptista, R. Martins, J. Inácio and E. Fortunato, Nanotechnology, 2014, 25, 094006 CrossRef CAS.
- V. B. C. Lee, N. F. Mohd-Naim, E. Tamiya and M. U. Ahmed, Anal. Sci., 2018, 34, 7–18 CrossRef CAS.
- A.-S. Bats, P. Mathevet, A. Buenerd, I. Orliaguet, E. Mery, S. Zerdoud, M.-A. Le Frère-Belda, M. Froissart, D. Querleu and A. Martinez, Ann. Surg. Oncol., 2013, 20, 413–422 CrossRef PubMed.
- A. Mondal, A. K. Nayak, P. Chakraborty, S. Banerjee and B. C. Nandy, Pharmaceutics, 2023, 15, 2064 CrossRef CAS PubMed.
- C. Kher and S. Kumar, Cureus, 2022, 14(9), e29059 Search PubMed.
- A. Khajuria, H. Alajangi, J. Singh, G. Passi, R. P. Barnwal, G. Singh and I. P. Kaur, in Handbook of Oncobiology: from Basic to Clinical Sciences, Springer, 2023, pp. 1–31 Search PubMed.
- F. Usman, K. H. Ghazali, R. Muda, N. H. Johari, J. O. Dennis, N. Tamam, A. Sulieman and Y. Ji, Sens. Actuators Rep., 2023, 6, 100172 CrossRef.
- A. W. Martinez, S. T. Phillips and G. M. Whitesides, Proc. Natl. Acad. Sci. U. S. A., 2008, 105, 19606–19611 CrossRef CAS.
- E. B. Strong, C. Knutsen, J. T. Wells, A. R. Jangid, M. L. Mitchell, N. W. Martinez and A. W. Martinez, Inventions, 2019, 4, 20 CrossRef.
- F. Bahavarnia, A. Saadati, S. Hassanpour, M. Hasanzadeh, N. Shadjou and A. Hassanzadeh, Int. J. Biol. Macromol., 2019, 138, 744–754 CrossRef CAS.
- F. Cui, J. Ji, J. Sun, J. Wang, H. Wang, Y. Zhang, H. Ding, Y. Lu, D. Xu and X. Sun, Anal. Bioanal. Chem., 2019, 411, 985–995 CrossRef CAS.
- B. V. Saghezi, K. Mansouri and R. Hallaj, Nano Sel., 2025, e202400108 CrossRef CAS.
- B. Alhalaili, I. N. Popescu, C. O. Rusanescu and R. Vidu, Sustainability, 2022, 14, 12844 CrossRef CAS.
- M. B. Behyar, F. Bahavarnia, A. Nilghaz, M. Hasanzadeh and N. Shadjou, RSC Adv., 2025, 15, 12321–12330 RSC.
- J. L. Garcia-Cordero and S. J. Maerkl, Curr. Opin. Biotechnol., 2020, 65, 37–44 CrossRef CAS.
- P. Iranpour, M. Ajamian, A. Safavi, N. Iranpoor, A. Abbaspour and S. Javanmardi, J. Mater. Sci.: Mater. Med., 2018, 29, 1–9 CrossRef CAS.
- N. Chattopadhyay, Z. Cai, J.-P. Pignol, B. Keller, E. Lechtman, R. Bendayan and R. M. Reilly, Mol. Pharmaceutics, 2010, 7, 2194–2206 CrossRef CAS.
- P. L. Mok, S. N. Leow, A. E.-H. Koh, H. H. Mohd Nizam, S. L. S. Ding, C. Luu, R. Ruhaslizan, H. S. Wong, W. H. W. A. Halim and M. H. Ng, Int. J. Mol. Sci., 2017, 18, 345 CrossRef PubMed.
- S. Sun, Y. Wang, T. Ming, J. Luo, Y. Xing, J. Liu, Y. Xiong, Y. Ma, S. Yan and Y. Yang, Commun. Biol., 2021, 4, 121 CrossRef CAS.
- Y. Wang, H. Xu, J. Luo, J. Liu, L. Wang, Y. Fan, S. Yan, Y. Yang and X. Cai, Biosens. Bioelectron., 2016, 83, 319–326 CrossRef CAS.
- J.-Y. Huang, H.-T. Lin, T.-H. Chen, C.-A. Chen, H.-T. Chang and C.-F. Chen, ACS Sens., 2018, 3, 174–182 CrossRef CAS PubMed.
- Y.-S. Chien, U.-I. Wu, Y.-H. Pan and C.-F. Chen, Biosens. Bioelectron., 2025, 117724 CrossRef CAS.
- A. Kumar, P. Shinde, L. Mohan, P. S. Mahapatra and T. S. Santra, in Microfluidics and Bio-MEMS, Jenny Stanford Publishing, 2020, pp. 1–47 Search PubMed.
- N. Prang, S. Preithner, K. Brischwein, A. Wöppel, J. Müller, C. Steiger, M. Peters, P. Baeuerle and A. da Silva, Br. J. Cancer, 2005, 92, 342–349 CrossRef CAS.
- S. Khizar, H. Ben Halima, N. M. Ahmad, N. Zine, A. Errachid and A. Elaissari, Electrophoresis, 2020, 41, 1206–1224 CrossRef CAS PubMed.
- J. L. Chen, D. I. Njoku, C. Tang, Y. Gao, J. Chen, Y. K. Peng, H. Sun, G. Mao, M. Pan and N. F. Y. Tam, Small Methods, 2024, 8, 2400155 CrossRef CAS.
- T. Tian, L. Li, Y. Zhang, H. Liu, L. Zhang, M. Yan and J. Yu, Sens. Actuators, B, 2018, 261, 44–50 CrossRef CAS.
- L. Liang, S. Ge, L. Li, F. Liu and J. Yu, Anal. Chim. Acta, 2015, 862, 70–76 CrossRef CAS PubMed.
- C. K. Tang, A. Vaze, M. Shen and J. F. Rusling, ACS Sens., 2016, 1, 1036–1043 CrossRef CAS.
- F. Gu, C. Hu, Q. Xia, C. Gong, S. Gao and Z. Chen, J. Nanopart. Res., 2018, 20, 1–10 CrossRef CAS.
- X. Zhang, S. Li, B. Pan, D. Pan, L. Liu, X. Hou, M. Chu, K. Kondoh and M. Zhao, Carbon, 2019, 155, 686–696 CrossRef CAS.
- M. He, S. Zhang and J. Zhang, Chem. Rev., 2020, 120, 12592–12684 CrossRef CAS.
- A. Sanginario, B. Miccoli and D. Demarchi, Biosensors, 2017, 7, 9 CrossRef PubMed.
- G.-H. Lu, W.-T. Shang, H. Deng, Z.-Y. Han, M. Hu, X.-Y. Liang, C.-H. Fang, X.-H. Zhu, Y.-F. Fan and J. Tian, Biomaterials, 2019, 195, 13–22 CrossRef CAS.
- P. Xu, M. Chen, G. Zeng, D. Huang, C. Lai, Z. Wang, M. Yan, Z. Huang, X. Gong and B. Song, J. Hazard. Mater., 2019, 374, 459–468 CrossRef CAS PubMed.
- B. Anis, W. K. Khalil, N. A. Kamel and S. L. Abd El-Messieh, Adv. Nat. Sci.:Nanosci. Nanotechnol., 2022, 12, 045017 Search PubMed.
- A. Antonucci, J. Kupis-Rozmysłowicz and A. A. Boghossian, ACS Appl. Mater. Interfaces, 2017, 9, 11321–11331 CrossRef CAS.
- A. V. Ravi Kiran, G. Kusuma Kumari, P. T. Krishnamurthy, P. K. Chintamaneni and S. K. S. Pindiprolu, in Handbook of Carbon Nanotubes, Springer, 2022, pp. 1739–1771 Search PubMed.
- B. K. Panigrahi and A. K. Nayak, Curr. Drug Delivery, 2020, 17, 558–576 CrossRef.
- R. Raj, P. Mongia, S. Kumar Sahu and A. Ram, Curr. Drug Targets, 2016, 17, 206–228 CrossRef CAS PubMed.
- H. Hashemzadeh and H. Raissi, J. Mol. Model., 2017, 23, 1–10 CrossRef CAS.
- S. Boncel, A. Pluta, M. Skonieczna, A. Gondela, B. Maciejewska, A. P. Herman, R. G. Jędrysiak, S. Budniok, K. Komędera and A. Błachowski, J. Nanomater., 2017, 2017, 1262309 Search PubMed.
- M. Kumar, P. Sharma, R. Maheshwari, M. Tekade, S. K. Shrivastava and R. K. Tekade, Nanotechnology-Based Targeted Drug Delivery Systems for Brain Tumors, 2018, pp. 397–437 Search PubMed.
- E. Gallardo-Toledo, C. Velasco-Aguirre and M. J. Kogan, in Nanomedicines for Brain Drug Delivery, Springer, 2020, pp. 149–172 Search PubMed.
- C. Baynes and J.-Y. Yoon, SLAS Technol., 2018, 23, 30–43 CrossRef CAS.
- P. Wang, L. Ge, M. Yan, X. Song, S. Ge and J. Yu, Biosens. Bioelectron., 2012, 32, 238–243 CrossRef CAS.
- W. Liu, H. Yang, Y. Ding, S. Ge, J. Yu, M. Yan and X. Song, Analyst, 2014, 139, 251–258 RSC.
- W. Li, S. Ge, S. Wang, M. Yan, L. Ge and J. Yu, Luminescence, 2013, 28, 496–502 CrossRef CAS.
- S. Ji, M. Lee and D. Kim, Biosens. Bioelectron., 2018, 102, 345–350 CrossRef CAS PubMed.
- P. Ratre, B. Jain, R. Kumari, S. Thareja, R. Tiwari, R. K. Srivastava, I. Y. Goryacheva and P. K. Mishra, ACS Omega, 2022, 7, 39586–39602 CrossRef CAS PubMed.
- S.-Y. Kang, H. Yin, K.-Q. Zhang, X. Chen and K.-Z. Wang, Mater. Sci. Eng., C, 2019, 99, 657–668 CrossRef CAS.
- H. M. Osorio, F. Castillo-Solís, S. Y. Barragán, C. Rodríguez-Pólit and R. Gonzalez-Pastor, Int. J. Mol. Sci., 2024, 25, 10539 CrossRef CAS.
- R. S. Tade, M. P. More, S. N. Nangare and P. O. Patil, J. Drug Targeting, 2022, 30, 269–286 CrossRef CAS PubMed.
- J. Dong, K. Wang, L. Sun, B. Sun, M. Yang, H. Chen, Y. Wang, J. Sun and L. Dong, Sens. Actuators, B, 2018, 256, 616–623 CrossRef CAS.
- S. Chung, R. A. Revia and M. Zhang, Adv. Mater., 2021, 33, 1904362 CrossRef CAS.
- X. Hai, J. Feng, X. Chen and J. Wang, J. Mater. Chem. B, 2018, 6, 3219–3234 RSC.
- H.-y. Fan, X.-h. Yu, K. Wang, Y.-j. Yin, Y.-j. Tang, Y.-l. Tang and X.-h. Liang, Eur. J. Med. Chem., 2019, 182, 111620 CrossRef CAS PubMed.
- F. Khodadadei, S. Safarian and N. Ghanbari, Mater. Sci. Eng., C, 2017, 79, 280–285 CrossRef CAS.
- M. K. Shukla, A. Parihar, C. Karthikeyan, D. Kumar and R. Khan, Nanoscale, 2023, 15, 14698–14716 RSC.
- P. Das, A. Sedighi and U. J. Krull, Anal. Chim. Acta, 2018, 1041, 1–24 CrossRef CAS.
- S. Jahedi, M. R. Tohidkia, M. Esmaeili, F. Bani and S. Kaboli, Microfluid. Nanofluid., 2025, 29, 16 CrossRef CAS.
- S. Hassanpour, M. Hasanzadeh, A. Saadati, N. Shadjou, J. Soleymani and A. Jouyban, Microchem. J., 2019, 146, 345–358 CrossRef CAS.
- Y. Wu, P. Xue, Y. Kang and K. M. Hui, Anal. Chem., 2013, 85, 8661–8668 CrossRef.
- Z. Wei, J. Zhang, A. Zhang, Y. Wang and X. Cai, Molecules, 2017, 22, 392 CrossRef.
- Y. Wang, J. Luo, J. Liu, S. Sun, Y. Xiong, Y. Ma, S. Yan, Y. Yang, H. Yin and X. Cai, Biosens. Bioelectron., 2019, 136, 84–90 CrossRef CAS PubMed.
- L. Cao, C. Fang, R. Zeng, X. Zhao, F. Zhao, Y. Jiang and Z. Chen, Sens. Actuators, B, 2017, 252, 44–54 CrossRef CAS.
- Y. Fan, S. Shi, J. Ma and Y. Guo, Biosens. Bioelectron., 2019, 135, 1–7 CrossRef CAS.
- S. Palencia, S. Vera, A. M. Díez-Pascual and M. P. San Andrés, Anal. Bioanal. Chem., 2015, 407, 4671–4682 CrossRef CAS PubMed.
- H. E. H. Ahmed and M. SOYLAK, TrAC, Trends Anal. Chem., 2024, 117939 CrossRef.
- U. Latief, S. ul Islam, Z. M. Khan and M. S. Khan, Spectrochim. Acta, Part A, 2021, 262, 120132 CrossRef CAS.
- S. Sunil and B. K. Mandal, J. Cluster Sci., 2023, 34, 2591–2607 CrossRef CAS.
- D. Chen, B. Li, T. Lei, D. Na, M. Nie, Y. Yang, C. Xie, Z. He and J. Wang, J. Nanobiotechnol., 2021, 19, 1–17 CrossRef.
- S. Mishra, K. Das, S. Chatterjee, P. Sahoo, S. Kundu, M. Pal, A. Bhaumik and C. K. Ghosh, ACS Omega, 2023, 8, 4566–4577 CrossRef CAS.
- C. Sanjayan, C. H. Ravikumar and R. G. Balakrishna, Chem. Eng. J., 2023, 464, 142581 CrossRef.
- L. Li, Y. Zhang, F. Liu, M. Su, L. Liang, S. Ge and J. Yu, Chem. Commun., 2015, 51, 14030–14033 RSC.
- Z. Qiu, J. Shu and D. Tang, Anal. Chem., 2017, 89, 5152–5160 CrossRef CAS.
- F. D. Duman and R. S. Forgan, J. Mater. Chem. B, 2021, 9, 3423–3449 RSC.
- K. Lu, T. Aung, N. Guo, R. Weichselbaum and W. Lin, Adv. Mater., 2018, 30, 1707634 CrossRef PubMed.
- J. Pantwalawalkar, P. Mhettar, S. Nangare, R. Mali, A. Ghule, P. Patil, S. Mohite, H. More and N. Jadhav, ACS Biomater. Sci. Eng., 2023, 9, 4497–4526 CrossRef CAS PubMed.
- S. Lv, Y. Tang, K. Zhang and D. Tang, Anal. Chem., 2018, 90, 14121–14125 CrossRef CAS.
- J. Hassanzadeh, H. A. Al Lawati and I. Al Lawati, Anal. Chem., 2019, 91, 10631–10639 CrossRef CAS PubMed.
- L. Zhang, J. Chen, H. Yang, Z. Yu, Y. Xu, S.-Y. Liu, Z. Dai and X. Zou, Sens. Actuators, B, 2022, 359, 131570 CrossRef CAS.
- R.-J. Wei, X. Luo, G.-H. Ning and D. Li, Acc. Chem. Res., 2025, 58, 746–761 CrossRef CAS.
- R. K. Sharma, P. Yadav, M. Yadav, R. Gupta, P. Rana, A. Srivastava, R. Zbořil, R. S. Varma, M. Antonietti and M. B. Gawande, Mater. Horiz., 2020, 7, 411–454 RSC.
- Q. Zheng, X. Liu, Y. Zheng, K. W. Yeung, Z. Cui, Y. Liang, Z. Li, S. Zhu, X. Wang and S. Wu, Chem. Soc. Rev., 2021, 50, 5086–5125 RSC.
- H. V. Babu, M. M. Bai and M. Rajeswara Rao, ACS Appl. Mater. Interfaces, 2019, 11, 11029–11060 CrossRef CAS.
- X. Liu, F. Wang, Y. Meng, L. Zhao, W. Shi, X. Wang, Z. He, J. Chao and C. Li, Biosens. Bioelectron., 2022, 207, 114208 CrossRef CAS PubMed.
- S. Jafari, H. Derakhshankhah, L. Alaei, A. Fattahi, B. S. Varnamkhasti and A. A. Saboury, Biomed. Pharmacother., 2019, 109, 1100–1111 CrossRef CAS.
- N. Pal, J.-H. Lee and E.-B. Cho, Nanomaterials, 2020, 10, 2122 CrossRef CAS PubMed.
- R. Narayan, U. Y. Nayak, A. M. Raichur and S. Garg, Pharmaceutics, 2018, 10, 118 CrossRef CAS.
- M. Mladenović, S. Jarić, M. Mundžić, A. Pavlović, I. Bobrinetskiy and N. Ž. Knežević, Biosensors, 2024, 14, 326 CrossRef.
- L. Liang, M. Su, L. Li, F. Lan, G. Yang, S. Ge, J. Yu and X. Song, Sens. Actuators, B, 2016, 229, 347–354 CrossRef CAS.
- T.-H. Ulep, R. Zenhausern, A. Gonzales, D. S. Knoff, P. A. L. Diaz, J. E. Castro and J.-Y. Yoon, Biosens. Bioelectron., 2020, 153, 112042 CrossRef CAS PubMed.
Footnote |
| † These authors were co-first author. |
| This journal is © The Royal Society of Chemistry 2025 |

