Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
The acetate anion promotes hydrolysis of poly(ethylene terephthalate) in ionic liquid–water mixtures
Maariyah Y.
Suleman†
a,
Harriet L.
Judah†
 a,
Panagiotis
Bexis
a,
Panagiotis
Bexis
 a,
Paul
Fennell
b,
Jason P.
Hallett
a,
Paul
Fennell
b,
Jason P.
Hallett
 b and
Agnieszka
Brandt-Talbot
b and
Agnieszka
Brandt-Talbot
 *a
*a
aDepartment of Chemistry, Imperial College London, London, W12 0BZ, UK. E-mail: agi@imperial.ac.uk
bDepartment of Chemical Engineering, Imperial College London, London, SW7 2AZ, UK
First published on 11th August 2025
Abstract
A circular plastic economy reduces raw material consumption and discourages pollution. Chemical recycling upgrades the quality of recyclate and is a complementary approach to thermomechanical recycling of plastic waste. This study investigated the use of aprotic and protic ionic liquids (ILs) as solvents for chemical recycling by the hydrolysis of the most common polyester plastic, poly(ethylene terephthalate) (PET). Combinations of three types of cations (aprotic 1-alkyl-3-methylimidazolium, protic 1-methylimidazolium and protic 1,5-biazocyclo-[4.3.0]non-5-enium) combined with a range of anions (acetate, chloride, methanesulfonate, hydrogen sulfate, methyl sulfate, trifluoromethanesulfonate and chlorozincate) were used to hydrolyse PET in the presence of 15 wt% water as the co-solvent and reagent. PET conversion under the screening conditions (180 °C, 3 h, 5% PET loading) varied between 1 and 100%, with ILs containing the acetate anion enabling >97% PET conversion irrespective of the cation. Acidification with aqueous HCl recovered crude crystallised terephthalic acid (TPA). Significant crude yields (46–93%) were only observed for the acetate ILs. The purity of the crude TPA was 34–98%, with 1-ethy-3-methylimidazolium acetate, [C2C1im][OAc], and 1-methylimidazolium acetate, [C1Him][OAc], yielding more and purer TPA than 1,5-biazocyclo-[4.3.0]non-5-enium acetate, [DBNH][OAc]. TPA solubility, PET conversion and TPA yield generally correlated well with increasing pKa and higher hydrogen bond acceptor strength of the IL anion, suggesting that the depolymerisation mechanism in the acetate IL water mixtures is base catalysed. The screening identifies aqueous mixtures of the (pseudo)-protic IL [C1Him][OAc] as promising catalytic solvent component for the chemical recycling of PET at an industrially feasible temperature, due to high isolated TPA yields and purity achieved at a low solvent cost ($1.74–2.15 per kg). However, an effective separation approach for the monomers TPA and ethylene glycol from the solvent remains to be developed.
Green foundation1. This work advances green chemistry by identifying water-containing ionic liquids, especially the low-cost protic ionic liquids, such as 1-methylimidazolium acetate, [C1Him][OAc], as effective media for PET depolymerisation. Using the solvent mixture offers milder reaction conditions compared to conventional methods. This approach supports circular economy goals by enabling chemical recycling of plastic waste.2. By employing [C1Him][OAc] with 15 wt% water, the study achieved 98.5% PET conversion and 82.4% crude TPA yield with 77.8% purity using moderate hydrolysis conditions (180 °C, 3 h). The IL is easy to prepare and low-cost (∼$2 per kg), which is >10 times lower than the cost of the analogous aprotic ionic liquid solvent. The study also introduced chromatographic purity analysis, improving the accuracy of performance evaluation over purely gravimetric methods. 3. Future work should develop monomer recovery methods that avoid acid addition to reduce waste generation during monomer isolation and enable IL reuse. Research into process intensification (higher loading), and application to mixed and degraded plastic waste will enhance the environmental and practical impact of this method. |
Introduction
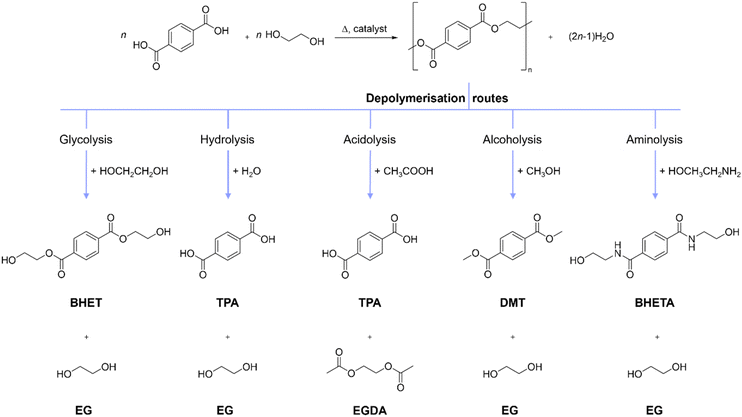
PET is a commodity plastic that is widely used in applications such as packaging, construction, textiles and electronics,1,2 accounting for 6.2% of global plastic production.3,4 The European PET bottle recycling rate was 50% in 2022, which was achieved through thermomechanical recycling, consisting of shredding waste PET to produce flakes that can be melted and re-extruded into recycled plastic pellets. Thermomechanical recycling is a well-developed technology; however, the recycled plastic typically has degraded material properties. For example, it is coloured or has reduced mechanical strength, caused by lower molecular weight and contamination with other polymers and materials.5,6 As a result, mechanically recycled PET from bottles and food packaging is often used in applications with less stringent specifications for mechanical strength and transparency, such as polyester textiles, while textile PET remain unrecycled.7 To encourage the use of recycled PET in food packaging and make coloured polyester textiles recyclable, methods to purify and upgrade PET-containing waste are required.8Chemical plastic recycling encompasses a variety of methods that employ heat or chemical agents to separate additives from the polymer or to break the chemical bonds in the polymer, which recovers chemical feedstocks, purified oligomers or monomers.9 Biological plastic recycling employs microorganisms or enzymes to break chemical bonds in polymers to facilitate recycling at the oligomer or monomer level.10 PET is a condensation polymer and can be chemically recycled by cleaving the ester bonds, typically using a solvent containing nucleophiles such as water, glycols, alcohols, and amines, yielding a range of depolymerisation products (Fig. 1).6,9,11–13 Challenges for chemical recycling via depolymerisation include avoiding the generation of by-products and chemical waste, and minimising the energy requirement for depolymerisation and monomer recovery.14 PET is sparingly soluble in most solvents due to its high molar weight and crystallinity.15 As a result, PET is typically depolymerised at the solid–liquid interface, including for approaches that employ ILs.16–18
Glycolysis of PET with ethylene glycol (EG) as the monomer, solvent and nucleophile is the most investigated approach, generating solubilised bis(2-hydroxyethyl) terephthalate (BHET) and EG. However, glycolysis faces challenges during purification, such as slow crystallisation of crude BHET at low temperatures, which requires cooling to 0–10 °C for up to 24 hours.19,20 Recently, depolymerisation of PET in anhydrous acetic acid has been reported as an approach called acetolysis, yielding TPA and acetylated EG after evaporating the solvent.13 While acetolysis results in rapid crystallisation of TPA, it requires anhydrous conditions and high temperatures (>280 °C).13
The depolymerisation of PET using water as the nucleophile (hydrolysis) is attractive, as it is a direct reversal of the industrial PET production method (Fig. 1). However, without a suitable catalyst, high temperatures and pressures or long reaction times are required to achieve depolymerisation.21 Catalysts for PET hydrolysis can be inorganic and organic hydroxides, Lewis acids, ILs and deep eutectic solvents. PET hydrolysis has been carried out using aqueous solutions of sodium hydroxide (NaOH) at low temperatures of 50–80 °C, yielding crystallised TPA.22 The TPA can only be isolated after reducing the pH by adding HCl or H2SO4 to protonate sodium terephthalate, which creates salt waste (NaCl or Na2SO4). Enzymes are effective at hydrolysing PET at low temperatures (55 °C), but reaction times are long (4 days) and addition of acid is required to precipitate the TPA.23 Recently, PET was hydrolysed at 80–120 °C within 1 h using mixtures of iron salt hydrates and organic sulfonic acids, achieving PET conversion and an isolated crude TPA yield up to 100% and 96.4%, respectively. TPA precipitation occurred without the need to adjust the pH, instead, with water used as an anti-solvent. However, the crude TPA was bright yellow.24
ILs have been used as catalysts or catalytic solvents for PET depolymerisation, with IL-assisted glycolysis being the most commonly reported approach. The IL is used as a catalyst (approx. 5 wt% loading relative to EG) to accelerate the reaction or to replace the inorganic Lewis acidic catalyst, Zn(OAc)2.16,25–27 A number of protic ILs have been reported for PET glycolysis, for example, triazabicylodecanium methanesulfonate, [TBD][CH3SO3],28 1,5-diazabicyclo[4.3.0]non-5-enium 4-methylphenoxide, [DBNH][4-CH3PhO],27 1,8-diazabicyclo[5.4.0]undec-7-enium imidazolate, [HDBU][Im],29 1,5,7-triazabicyclo[4.4.0]dec-5-enium acetate, [TBD][OAc],30 and 2-hydroxyethyl ammonium acetate, [2-HEAA][OAc],31 with complete PET conversion and BHET yields of at least 85%.
IL-assisted hydrolysis of PET has been less commonly reported. Hydrolysis of PET (polyester) fibre employing mixtures of 1-butyl-3-methylimidazolium hydrogen sulfate, [C4C1im][HSO4], or 1-methylimidazolium hydrogen sulfate, [C1Him][HSO4], with 40–60 wt% water was demonstrated,32 with PET conversion up to 80% at 185–195 °C after 2.5–3.5 h, while TPA yield and purity were not quantified. 1-Butyl-3-methylimidazolium chloride, [C4C1im]Cl, accelerated PET textile hydrolysis as 5% additive in aqueous and methanolic solutions of sodium hydroxide.33 Choline lysinate, [Ch][Lys], enabled PET hydrolysis after 2 h at 180 °C, achieving 55% conversion and 56% un-isolated TPA yield.34 Hydrolysis of PET in aqueous choline phosphate, [Ch]3[PO4], was employed at 120 °C, yielding crude crystallised TPA after 3 h upon acidification, with the TPA yield not reported.35 Liu et al. used a mixture of [C4C1im]Cl and 1-methyl-3-(3-sulfopropyl)-imidazolium hydrogen sulfate, [HSO3-pmim][HSO4], and 40 wt% water to hydrolyse PET at 170 °C for 4.5 h, reporting 89% crude isolated TPA yield after adding acid.36 A 94% isolated TPA yield was reported for 1-(3-propylsulfonic)-3-methylimidazolium chloride, [HSO3-pmim]Cl, mixed with 75 wt% water at 210 °C after 24 h, also employing acidification to isolate the TPA.37 PET hydrolysis was reported in the presence of “switchable ILs” generated from a range of amines, CO2 and water in a pressure reactor, with a reaction temperature of 150 °C and a reaction time of 6–24 h achieving up to ≥99% TPA yield upon acidification.38 Only small groups (2–6) of ILs were compared by individual studies.
The product purity is often not considered in studies that report isolated TPA yields, or methods are applied that assess product purity only qualitatively, such as differential scanning calorimetry, thermogravimetric analysis, X-ray diffraction, mass spectrometry, and spectroscopic techniques.16,27 Acid–base titration was used to assess purity quantitatively;13 however, it cannot distinguish TPA from acetic acid, 2-hydroxyethyl terephthalic acid (HETA), and other carboxylic acid impurities such as p-toluic acid and 4-carboxybenzaldehyde. While NMR spectroscopy can provide quantitative insights, chromatographic separation is superior in precision and accuracy, and should be used for quantitative assessment of product purity.
This study systematically investigated the influence of the IL structures (anion and cation) on PET conversion and TPA yield following PET hydrolysis, with over 20 IL compositions used and with a focus on easy-to-synthesise protic ILs, which are synthesised through a proton transfer from a Brønsted acid to an amine. The study also demonstrates that chromatographic quantification and NMR spectroscopy are important tools to correctly report TPA yield and monitor product purity and solvent stability.
Experimental
Materials
Reagents were used as received unless specified otherwise. PET beverage bottles were washed with deionised water and dried before cutting to 0.5 mm size particles (SM 2000, Retsch), followed by sieving through a 0.5 mm mesh. 1-Ethyl-3-methylimidazolium acetate, [C2C1im][OAc], (>95%, 0.48% H2O) and 1-butyl-3-methylimidazolium chloride, [C4C1im]Cl, (>98%, ≤1.0% H2O) were purchased from IoLiTec Technologies (Germany). 1-Ethyl-3-methylimidazolium hydrogen sulfate, [C2C1im][HSO4], (95%, 0.14% H2O), 1-butyl-3-methylimidazolium methyl sulfate, [C4C1im][CH3SO4], (95%), deuterated dimethyl sulfoxide (DMSO-d6, 99.96% atom% D), terephthalic acid (99%), 1-methylimidazole (99%), acetic acid (glacial, 99%), 1,3,5-trimethoxybenzene (≥99%) and HPLC-grade DMSO (≥99.7%) were purchased from Merck. Ethylene glycol (99.5%) was purchased from Acros Organics. 1,5-Diazabicyclo[4.3.0]non-5-ene (98%), trifluoromethanesulfonic acid (≥99%) and methanesulfonic acid (≥98%) were purchased from Fluorochem. Sulfuric acid (5 M) and hydrochloric acid (≥37%) were purchased from Honeywell Fluka. Zinc chloride (ZnCl2, ≥98%) was purchased from Alfa-Aesar. Sodium hydroxide (NaOH pellets, 98.5–100%) was purchased from AnalaR Normapur.NMR spectroscopy
NMR spectra were recorded on a Bruker 400 MHz spectrometer at room temperature and processed using MestreNova software version 12 (Mestrelab Research, S.L., Santiago de Compostela, Spain). Chemical shifts (δ) are reported in parts per million (ppm). All NMR spectra were recorded in DMSO-d6.Synthesis of protic ionic liquids
Equimolar amounts of acid and amine (unless otherwise specified) were charged into a round-bottom flask with a stir bar. The acid was added via dropwise addition through a dropping funnel. An ice bath was used to maintain the temperature below 5 °C. The mixture was stirred overnight at room temperature to ensure complete mixing. Deionised water was added as needed to produce ILs containing 15 wt% H2O. [C4C1im]Cl, [C4C1im][MeSO4], [DBNH][OAc], [DBNH][OAc] (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), [DBNH][OAc] (2
2), [DBNH][OAc] (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), [DBNH][MeSO3], [C1Him][OTf] and [C1Him][MeSO3] were solid at room temperature, so they were manually ground into powder and dried under vacuum before water was added. The water content of all synthesised ILs was adjusted to 15 wt%. If above this value, the water content was reduced using a rotary evaporator and measured using a volumetric Karl-Fischer titrator (Mettler Toledo titrator, EasyPlus Easy KFV). All ILs used in this work are shown in Fig. 2 and Table S1. The synthesised ILs were characterised with NMR spectroscopy and their detailed syntheses are described in the SI (Fig. S1–S12).
1), [DBNH][MeSO3], [C1Him][OTf] and [C1Him][MeSO3] were solid at room temperature, so they were manually ground into powder and dried under vacuum before water was added. The water content of all synthesised ILs was adjusted to 15 wt%. If above this value, the water content was reduced using a rotary evaporator and measured using a volumetric Karl-Fischer titrator (Mettler Toledo titrator, EasyPlus Easy KFV). All ILs used in this work are shown in Fig. 2 and Table S1. The synthesised ILs were characterised with NMR spectroscopy and their detailed syntheses are described in the SI (Fig. S1–S12).
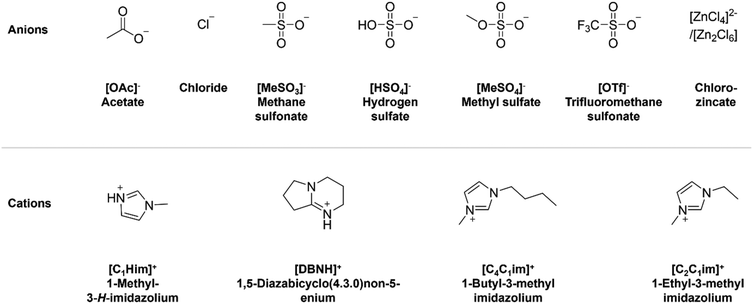 | ||
| Fig. 2 Structures of anions and cations used to construct the protic and aprotic ILs employed in this work. | ||
PET hydrolysis
0.100 g of PET powder and 2.0 g of IL–water mixtures (15 ± 0.5 wt% water) were added to a 15 mL pressure tube (Aceglass) and sealed with a Teflon cap and O-ring (silicon or FETFE), forming a suspension with 5% PET loading according to eqn (1), in which mPET, mIL and mwater are the weights of the components: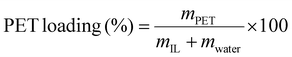 | (1) |
Samples were placed in a preheated oven (Binder ED 23, Binder GmbH) at the selected temperature (usually 180 °C) for a selected length of time. After hydrolysis, the pressure tube was cooled to room temperature for at least 30 min. If a solid residue was present, 2 M sodium hydroxide was added until pH 10–11 was reached to dissolve TPA, using pH strips for monitoring. The remaining residue was assumed to be PET and separated via vacuum filtration using a Buchner funnel and a Whatman™ 542 hardened ashless filter paper. The solid residue was washed with deionised water, dried in a vacuum oven at 60 °C for 24 h and the weight recorded. The weight was used to calculate PET conversion as follows (eqn (2)):
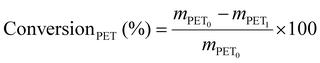 | (2) |
To recover crude TPA, the filtrate was acidified with 37% aqueous HCl until pH 2–3 was reached (monitored using pH strips) and the solution became cloudy. The suspension was refrigerated (4 °C) for 24 h to complete precipitation, followed by vacuum filtration and washing with deionised water. The precipitate was dried in a vacuum oven for 24 h at 60 °C on the filter paper before recording the weight of isolated TPA. The crude TPA yield was calculated as follows (eqn (3)):
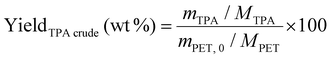 | (3) |
Determination of TPA purity in the crude product
The content of TPA in the crystallised product was determined using high performance liquid chromatography (HPLC) using a 1260 Infinity II Hybrid SFC/UHPLC (Agilent Technologies). The HPLC was equipped with an auto-sampler, a C-18 column (Raptor C18, 50 × 2.1 mm, 2.7 μm) and a UV-Vis diode array detector, with a wavelength of 254 nm used for detection. A gradient of acetonitrile and 0.1% formic acid (aq) was used as the mobile phase. HPLC samples were prepared in HPLC-grade DMSO using 5.1 mM 1,3,5-trimethoxybenzene (TMB) as the internal standard. Calibration curves were prepared using purchased commercial TPA (99% purity). Isolated crude TPA (4–6 mg) was dissolved in a solution of DMSO containing 5.1 mM TMB. The solution was filtered through a PTFE microfilter with a 0.2 μm pore size to remove undissolved impurities and dust particles prior to analysis. A calibration curve for TPA was prepared ranging from 0.2 mM to 7.5 mM, and the calculated mass of TPA in the sample, mTPA,calc, was determined via external calibration. TPA purity was calculated according to eqn (4):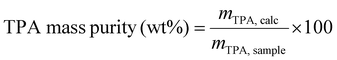 | (4) |
 | (5) |
 | (6) |
Monomer solubility screening
0.100 g of TPA mixed with 2.0 g of IL solution or solvent (water, acetic acid, 1-methylimidazole, or 1,5-diazabicyclo[4.3.0]non-5-ene) containing 15 wt% water was placed in a 15 mL pressure tube with a Teflon cap and silicon or FETFE O-ring. The tube was heated at 180 °C for 3 h in a pre-heated oven. The solutions were visually inspected while still in the oven to see if the TPA was solubilised, and once they had cooled down, a photograph was taken (in Fig. S22) and the qualitative observation was recorded.Results and discussion
Ionic liquid selection and synthesis of protic ILs
Two imidazolium cations were selected, the aprotic 1,3-dialkylimidazolium cation type and the protic 1-methylimidazolium cation type (Fig. 2). ILs with an imidazolium cation are well studied in the literature and relatively stable when heated.36,39,40 Due to the low pKa of 1-methylimidazole (pKa = 7.1),41 an amidinium cation, 1,5-diazabicyclo[4.3.0]non-5-enium ([DBNH]+), derived from an amine with a higher pKa (pKa = 12.8)42 was also studied. The higher pKa of the amidine nitrogen promotes preparation of protic ILs with a higher degree of proton transfer.43 An unprecedented variety of anions were chosen which covers a wide range of hydrogen bond acceptor strengths.39 In addition, an anion with Brønsted acidity ([HSO4]−) and one with the potential to generate Lewis acidic species ([ZnCl3]−) were included. The cation and anion selection was limited to ILs that are water miscible.PET hydrolysis in ionic liquid–water mixtures
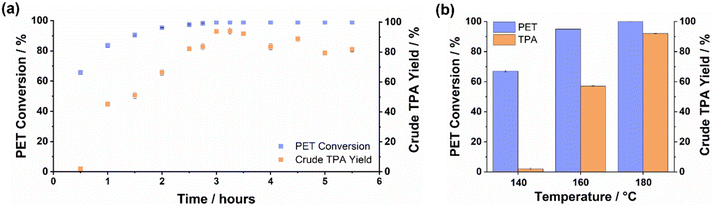
PET conversion was observed in the [C2C1im][OAc]–water mixture (Fig. 3) and depolymerisation was confirmed by detecting peaks for TPA and EG in the 1H-NMR spectrum of the post-hydrolysis solution (Fig. S23a). The reaction time was varied between 0.5 h and 5.5 h at the screening temperature (Fig. 3a), showing that PET conversion reached 100% at around 2.5 h for the [C2C1im][OAc]–water mixture. The reaction temperature was also varied between 140 and 180 °C, with PET conversion decreasing with lower reaction temperature as expected (Fig. 3b). The screening experiments confirmed that 180 °C is an appropriate temperature for the 3 h reaction time.
Interestingly, a lag was observed between PET conversion and TPA yield, the difference being more prominent at shorter reaction times and lower reaction temperatures (Fig. 3). A maximum crude TPA yield (92%) was observed at around 3.3 h, which occurred ∼50 min after achieving full PET conversion. The lag suggests that intermediate depolymerisation products form which are soluble in dilute aqueous acid, potentially glycol terminated oligomers and BHET. A gradual drop in yield at longer times suggests that slower reactions may have affected the purity of the crude product; however, the product purity was not determined for this set of experiments.
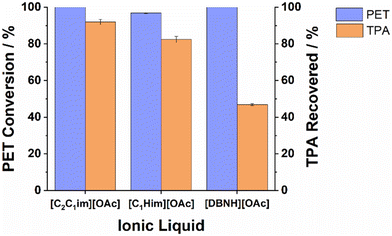 | ||
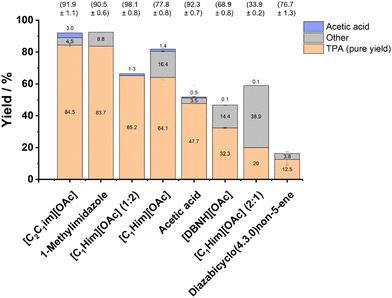
| Fig. 5 Conversion and recovered crude TPA yield obtained with various acetate ionic liquids. PET was hydrolysed at 180 °C for 3 h in the presence of 15 wt% water, with a PET loading of 5%. | ||
| Solvent name | Abbreviation | PET conversion (%) | Isolated crude TPA yield (wt%) |
|---|---|---|---|
| 1-Ethyl-3-methylimidazolium acetate | [C2C1im][OAc] | 100.0 ± 0.0 | 91.9 ± 1.2 |
| 1-Butyl-3-methylimidazolium methyl sulfate | [C4C1im][MeSO4] | 25.9 ± 1.0 | 0 |
| 1-Butyl-3-methylimidazolium methanesulfonate | [C4C1im][MeSO3] | 32.1 ± 1.2 | 2.6 ± 0.1 |
| 1-Ethyl-3-methylimidazolium hydrogen sulfate | [C2C1im][HSO4] | 11.2 ± 0.3 | 0.1 ± 0.0 |
| 1-Butyl-3-methylimidazolium hydrogen sulfate | [C4C1im][HSO4] | 6.7 ± 0.4 | 0.3 ± 0.1 |
| 1-Butyl-3-methylimidazolium chloride | [C4C1im]Cl | 0.1 ± 0.0 | 0 |
| 1-Methylimidazolium acetate | [C1Him][OAc] | 98.5 ± 0.4 | 82.4 ± 2.2 |
| 1-Methylimidazolium acetate | [C1Him][OAc] (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
98.4 ± 0.4 | 66.5 ± 0.1 |
| 1-Methylimidazolium acetate | [C1Him][OAc] (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
97.5 ± 0.3 | 59.0 ± 0.3 |
| 1-Methylimidazolium chlorozincate | [C1Him][ZnCl3] | 23.0 ± 0.4 | 0.1 ± 0.0 |
| 1-Methylimidazolium triflate | [C1Him][OTf] | 9.0 ± 1.2 | 2.8 ± 0.3 |
| 1-Methylimidazolium chloride | [C1Him]Cl | 4.9 ± 0.3 | 0 |
| 1-Methylimidazolium hydrogen sulfate | [C1Him][HSO4] | 3.5 ± 0.3 | 0 |
| 1-Methylimidazolium methanesulfonate | [C1Him][MeSO3] | 1.0 ± 0.3 | 1.5 ± 0.2 |
| Diazabicyclo(4.3.0)non-5-enium acetate | [DBNH][OAc] | 100.0 ± 0.0 | 46.8 ± 0.4 |
| Diazabicyclo(4.3.0)non-5-enium acetate | [DBNH][OAc] (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
100.0 ± 0.0 | 7.1 ± 0.2 |
| Diazabicyclo(4.3.0)non-5-enium acetate | [DBNH][OAc] (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
98.4 ± 0.0 | 9.2 ± 0.2 |
| Diazabicyclo(4.3.0)non-5-enium methanesulfonate | [DBNH][MeSO3] | 16.1 ± 1.0 | 0.2 ± 0.1 |
| Diazabicyclo(4.3.0)non-5-enium hydrogen sulfate | [DBNH][HSO4] | 12.3 ± 0.1 | 0.1 ± 0.0 |
| Diazabicyclo(4.3.0)non-5-enium chloride | [DBNH]Cl | 11.6 ± 1.1 | 0 |
| 1-Methylimidazole | 98.4 ± 0.6 | 92.5 ± 3.5 | |
| Diazabicyclo(4.3.0)non-5-ene | 100.0 ± 0.0 | 16.3 ± 4.4 | |
| Acetic acid | 62 ± 13 | 52 ± 12 | |
| Water (deionised) | 0 | 0 |
Conversion and crude TPA yield were also determined for the amines used to generate the protic acetate ILs and for acetic acid (each containing 15 wt% water), and for deionised water as a control. PET conversion was observed for the three aqueous mixtures in the order diazabicyclo(4.3.0)non-5-ene (DBN) > 1-methylimidazole > acetic acid, but not in deionised water, demonstrating the need for using a catalyst or catalytically active solvent. Successful conversion of PET with 1-methylimidazole and DBN shows that the unprotonated base could play a role in promoting PET depolymerisation.41,42 1-Methylimidazole is a known catalyst for ester hydrolysis and transesterifications via formation of an activated intermediate.46,47
Acetic acid generated a lower and more variable PET conversion (∼62 ± 13%), with a similar crude TPA yield (∼52 ± 12%). It is unclear why the standard errors for the measurements in acetic acid were large. A control experiment weighing the samples showed that solvent losses were not responsible for the variation in yield.
Low PET conversion was also observed for the methanesulfonate, methyl sulfate, hydrogen sulfate, triflate and chlorozincate ILs. It has been reported that 1-butyl-3-methylimidazolium chlorozincate, [C4C1im][ZnCl3], and 1-allyl-3-methylimidazolium chlorozincate, [AllC1im][ZnCl3], catalyse the transesterification of PET with EG with high conversions (98% and 100%, respectively) and yield BHET as the major product.50,51 The results for the PET hydrolysis with a chlorozincate IL demonstrates that ILs effective in catalysing PET glycolysis do not necessarily work for PET hydrolysis.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 were explored for PET hydrolysis with [C1Him][OAc] and [DBNH][OAc], one with a 50 mol% excess amine and one with 50 mol% excess acetic acid, while all other reaction parameters remained the same. Fig. 6 shows that PET conversion was high for all investigated ABRs, while the crude TPA yield was the highest for the 1
1 were explored for PET hydrolysis with [C1Him][OAc] and [DBNH][OAc], one with a 50 mol% excess amine and one with 50 mol% excess acetic acid, while all other reaction parameters remained the same. Fig. 6 shows that PET conversion was high for all investigated ABRs, while the crude TPA yield was the highest for the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ABR for both ILs, demonstrating the importance of ABR as an additional reaction and process parameter for protic ILs.
1 ABR for both ILs, demonstrating the importance of ABR as an additional reaction and process parameter for protic ILs.
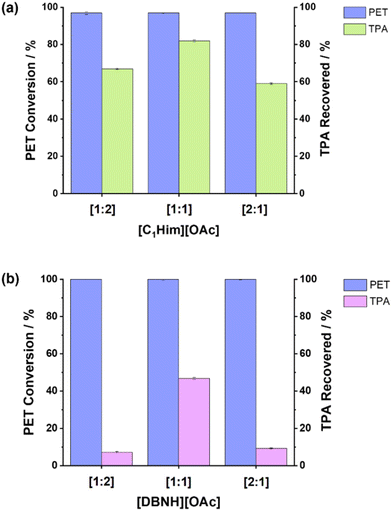 | ||
Fig. 6 Effect of changing the ABR (mol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mol) in the protic acetate ILs on PET conversion and crude TPA yield. (a) [C1Him][OAc] and (b) [DBNH][OAc]. mol) in the protic acetate ILs on PET conversion and crude TPA yield. (a) [C1Him][OAc] and (b) [DBNH][OAc]. | ||
It is currently unclear why the equimolar stoichiometry achieved the best crude product yield for the protic ILs; however, the solvent composition likely affects the hydrogen bonding network and proton transfer equilibrium,54 which would influence the catalysis in the solvent. Variation of the ABR should be investigated more thoroughly going forward.
Characterisation of crude TPA recovered from acetate ILs and protic IL precursors
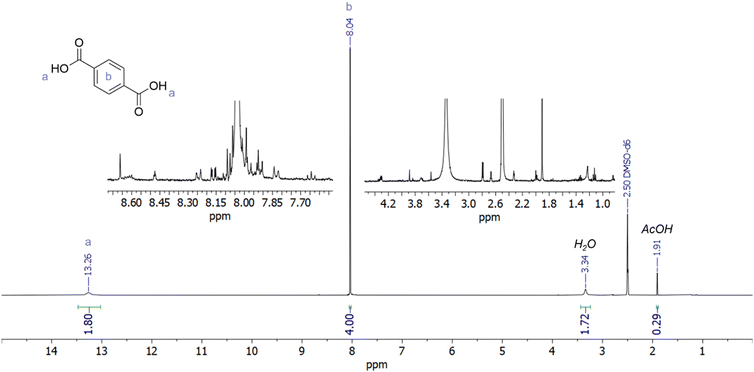
1H-NMR spectra of the crude precipitates (e.g.Fig. 4 and Fig. S27) show that the majority of the isolated precipitate was TPA, identified by a singlet peak at 8.0 ppm. The 1H-NMR spectra (Fig. 4 and Fig. S28) also indicate the presence of other compounds with aromatic (7.2–9.0 ppm) and aliphatic signals (0.1–5.0 ppm). These are likely depolymerisation products where TPA is esterified with EG (such as the dimers and trimers), or they may be due to the IL cation. Commercial TPA (Fig. S29) also contained impurities that gave 1H-NMR signals (7.8 and 8.2 ppm), which could be the ortho- and meta-dicarboxylic acids. Acetic acid (1.9 ppm) was also found in the precipitates generated with acetate ILs and with acetic acid. The amount of acetic acid present in the crude product varied between 0.2 and 3.2 wt% of the crude TPA (Table S3).While 1H-NMR spectroscopy can be used to detect and quantify TPA, EG, and acetic acid, HPLC is a more precise technique for quantification, as it separates TPA from other similar products. Hence, this study employed HPLC to analyse selected samples, with the method development discussed in the SI, and data on sample purity shown in Fig. 7 and Table S3.
This study shows product purity substantially varied with solvent composition. The purest TPA product (98 wt%) was obtained with [C1Him][OAc] with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ABR, followed by the benchmark IL [C2C1im][OAc] (92 wt%). Use of acetic acid and 1-methylimidazole also generated high TPA purities (>90%). For the acetate ILs with 1
2 ABR, followed by the benchmark IL [C2C1im][OAc] (92 wt%). Use of acetic acid and 1-methylimidazole also generated high TPA purities (>90%). For the acetate ILs with 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry, TPA purity followed the same trend as the crude TPA yield ([C2C1im][OAc] > [C1Him][OAc] > [DBNH][OAc]). To meet the requirements for polymer-grade TPA (>99%),55 the crude TPA could be purified via crystallisation, as performed in the Amoco process.56
1 stoichiometry, TPA purity followed the same trend as the crude TPA yield ([C2C1im][OAc] > [C1Him][OAc] > [DBNH][OAc]). To meet the requirements for polymer-grade TPA (>99%),55 the crude TPA could be purified via crystallisation, as performed in the Amoco process.56
The pure TPA yield was determined by combining TPA purity with the crude TPA yield. The highest pure TPA yield was obtained with [C2C1im][OAc] (85%) and 1-methylimidazole (84%), while the lowest pure yields were obtained with [DBNH][OAc] and DBN (32% and 13%), which is likely associated with the decomposition of [DBNH][OAc] and DBN (Fig. S26). The decomposition and reactivity of the decomposition product suggest that the performance of the DBN-based IL may not be improved meaningfully with further optimisation. While the equimolar composition of [C1Him][OAc] yielded the highest crude TPA yield, the pure TPA yield obtained with the excess acid composition was similar (65% and 64%, respectively), further demonstrating the importance of ABR as a reaction parameter.
The HPLC analysis detected up to 22 additional compounds in the isolated crude TPA (Fig. S30). Among them were the partially hydrolysed BHET and HETA products (Fig. S31), which were identified by mass spectrometry. 20 compounds remained unidentified due to the inability to obtain mass spectra with the LC-MS; however, it is likely that they include incompletely hydrolysed PET fragments, such as dimers, trimers and oligomers, which requires further investigation. Comparison of TPA purities determined by mass with the ones obtained by peak area confirmed that a significant proportion of impurities was not detected when employing HPLC-UV-vis analysis (Table S3), which is limited to compounds that are resolved by the eluent and column combination and are UV active. The difference between mass and area purity was particularly high for precipitates generated with [DBNH][OAc] (Δ = −13%), DBN (Δ = −12%) and [C1Him][OAc] 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (Δ = −18%), which also generated the lowest TPA yields (crude and pure). Overall, the 1
1 (Δ = −18%), which also generated the lowest TPA yields (crude and pure). Overall, the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and the excess acid composition of [C1Him][OAc] resulted in the highest pure TPA yield for protic ILs using the screening conditions, suggesting that these solvents may be preferable.
1 and the excess acid composition of [C1Him][OAc] resulted in the highest pure TPA yield for protic ILs using the screening conditions, suggesting that these solvents may be preferable.
Solubility of TPA in IL–water mixtures
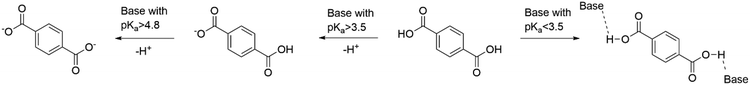
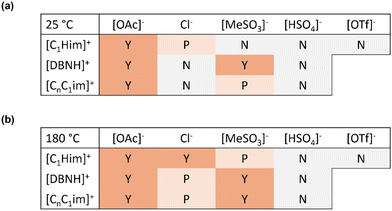
It was hypothesised that effective PET hydrolysis in IL–water mixtures may be linked to high TPA solubility, as removing the product from solid PET into the liquid phase could promote the hydrolysis reaction. Hence, monomer solubility was qualitatively observed at 5 wt% TPA loading for the IL–water mixtures investigated in this study (Table 2). Visual assessments were converted into the categories yes/no/partially. TPA solubility was found to be strongly anion dependent. All ILs with the acetate anion completely dissolved 5 wt% TPA at 25 °C and 180 °C, which is ascribed to the relatively high pKa of the acetate anion (4.8), leading to deprotonation of TPA (pKa1 3.5) (Fig. 8), including the IL solvents with excess acid and amine. TPA was also highly soluble in the presence of 1-methylimidazole and DBN, which can also be rationalised by the high pKas of these amines.41,42,57 TPA was not notably soluble in the acetic acid–water mixture and deionised water.| Temperature | |||
|---|---|---|---|
| 25 °C | 180 °C | ||
| Methylimidazolium | [C1Him][OAc] | Y | Y |
| [C1Him][OAc] excess AcOH | Y | Y | |
| [C1Him][OAc] excess C1im | Y | Y | |
| [C1Him]Cl | Partially | Y | |
| [C1Him][MeSO3] | N | Partially | |
| [C1Him][HSO4] | N | N | |
| [C1Him][OTf] | N | N | |
| [C1Him][ZnCl3] | N | N | |
| Diazabicyclo(4.3.0)non-5-enium | [DBNH][OAc] | Y | Y |
| [DBNH][OAc] excess AcOH | Y | Y | |
| [DBNH][OAc] excess DBN | Y | Y | |
| [DBNH]Cl | N | Partially | |
| [DBNH][MeSO3] | Y | Y | |
| [DBNH][HSO4] | N | N | |
| Dialkylimidazolium | [C2C1im][OAc] | Y | Y |
| [C2C1im][HSO4] | N | N | |
| [C4C1im][OAc] | Y | Y | |
| [C4C1im]Cl | N | Partially | |
| [C4C1im][MeSO3] | Partially | Y | |
| [C4C1im][HSO4] | N | N | |
| [C4C1im][MeSO4] | N | Partially | |
| Molecular solvents | Acetic acid | N | N |
| 1-Methylimidazole | Y | Y | |
| Diazabicyclo(4.3.0)non-5-ene | Y | Y | |
| De-ionised water | N | N | |
| Ethylene glycol | N | N | |
Full or partial solubilisation of 5 wt% TPA was observed for certain ILs whose anion has a lower pKa than that of TPA, for example 1-methylimidazolium chloride, [C1Him]Cl, [C4C1im]Cl and diazabicyclo(4.3.0)non-5-enium chloride, [DBNH]Cl. This is ascribed to the anion being a strong hydrogen bond acceptor (Kamlet–Taft β parameter = 0.83), as shown in Fig. 9. Solubility appeared to be temperature-dependent for these ILs, with better solubility observed at the screening reaction temperature (180 °C).
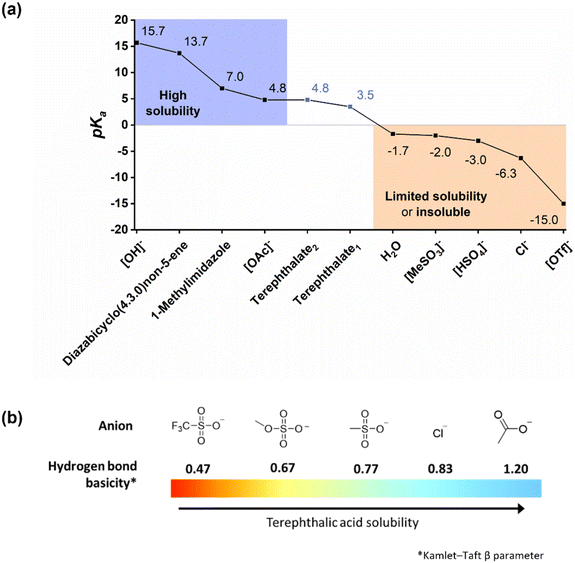 | ||
| Fig. 9 (a) Relationship between pKa of the corresponding acid of the IL or molecular solvent used in this study and TPA solubility. TPA exhibits high solubility in Brønsted basic solvents with a high pKa by generating terephthalate (TPA2−) salts. (b) Relationship between the Kamlet–Taft β parameter of ILs58 used in this study and TPA solubility. Solubility in solvents with lower pKa is nuanced and affected by the ability to form strong hydrogen bonds with TPA, of which the Kamlet–Taft β parameter is an indicator of. | ||
A substantial cation effect was observed for the ILs with chloride and methanesulfonate anions (Fig. 10). For methanesulfonate ILs, 5 wt% TPA was insoluble at 25 °C in protic 1-methylimidazolum methanesulfonate, [C1Him][MeSO3], completely soluble in aqueous diazabicyclo(4.3.0)non-5-enium methanesulfonate, [DBNH][MeSO3], and partially soluble in aprotic 1-butyl-3-methylimidazolium methanesulfonate, [C4C1im][MeSO3]. It is unclear why the methanesulfonate ILs are subject to a stronger cation effect than the other ILs; the lower pKa of methanesulfonic acid and the relatively high hydrogen bond basicity (β = 0.77) of the methanesulfonate anion may be playing a role.
5 wt% TPA was not soluble in ILs with a low hydrogen bond acceptor strength, including [OTf]− and [ZnCl3]−, nor the ILs with acidic anion and intermediate hydrogen bond basicity, [C1Him][HSO4], [C4C1im][HSO4] and diazabicyclo(4.3.0)non-5-enium hydrogen sulfate, [DBNH][HSO4]. This could be due to the acidic anion favouring TPA protonation and hence formation of crystallised TPA. However, 1-butyl-3-methylimidazolium methyl sulfate, [C4C1im][MeSO4], whose anion has a similar Kamlet–Taft β parameter without the acidity, did not promote TPA solubility at 25 °C, while only partial solubility was observed at 180 °C, suggesting that the hydrogen bonding of sulfate anions is generally insufficient. It is possible that transesterification and hydrolysis involving the methyl sulfate anion occur at 180 °C. Some of the qualitative TPA solubilities reported here are in good agreement with quantitative TPA solubilities reported in the literature for water-free ILs.59 The solubility of TPA in anhydrous ILs was reported as 25 wt%, 10 wt% and 0 wt% for [C1Him][OAc], [C1Him]Cl and [C1Him][HSO4], respectively.59
Fig. 11 suggests a correlation between TPA solubility and PET conversion; however, notable exceptions include [C1Him]Cl, [DBNH][MeSO3] and [C4C1im][MeSO3], which demonstrated substantial TPA solubility yet poor PET conversion (≤26%). At the same time, acetic acid provided good PET conversion despite its low TPA solubility.
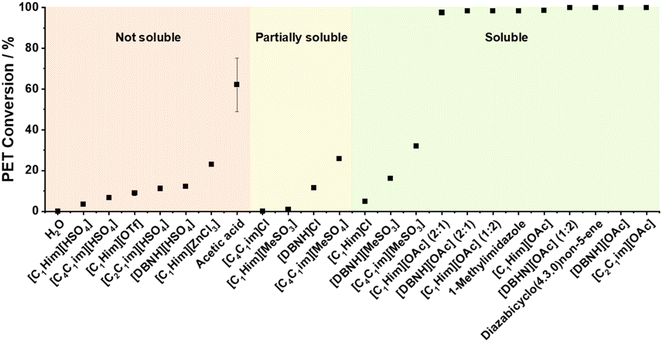 | ||
| Fig. 11 PET conversion in ILs or molecular solvents at 180 °C after 3 h with 15 wt% H2O present, categorised according to solubility of 5 wt% TPA in the aqueous solvent mixtures. | ||
Overall, it was observed that TPA solubility increased with the hydrogen bonding strength of the IL anion and the pKa of the anion's corresponding acid (Fig. 9). The observations suggest that the anion's hydrogen bond acceptor strength needs to be β > 0.67 to achieve good TPA solubility or the pKa of the corresponding acid of the anion needs to exceed the pKa1 of TPA. Incomplete proton transfer from acid to amine for some protic ILs may also enhance TPA solubility, with the unprotonated amine promoting solubility of the dicarboxylic acid through TPA deprotonation.
PET depolymerisation mechanism in IL–water mixtures
Using the data collected in this study, general features of the depolymerisation mechanism in IL–water mixtures can be discussed. The rate of organic reactions is determined by electronic and steric factors and mass transport. Since PET is insoluble in the IL–water mixtures, PET hydrolysis requires the cleavage of ester linkages at the solid–liquid interface. Formation of cracks confirms that depolymerisation occurs at the surface of PET particles,16,51,60 and the shrinking-core kinetic model is often assumed.17,18 Reactions at the solid–liquid interface are often slowed down due to mass transfer limitations, especially if the viscosity is high; however, differences in viscosity do not seem to explain the large variation in the observed PET conversion and TPA yield.Ester hydrolysis is known to proceed via a tetrahedral intermediate (Fig. 12),61 and its formation can be catalysed by acid or base. It is proposed that the reactive IL solvents enable a base catalysed mechanism, as the anion (acetate) can deprotonate water via an equilibrium, generating hydroxide ions which are strong nucleophiles. Hydroxide ions can also be generated via an equilibrium involving water and the amines, which could explain why 1-methylimidazole and DBN–water mixtures achieved high PET conversion. In addition, deprotonation of TPA drives conversion by shifting the equilibrium to the product side, which may also promote PET conversion.
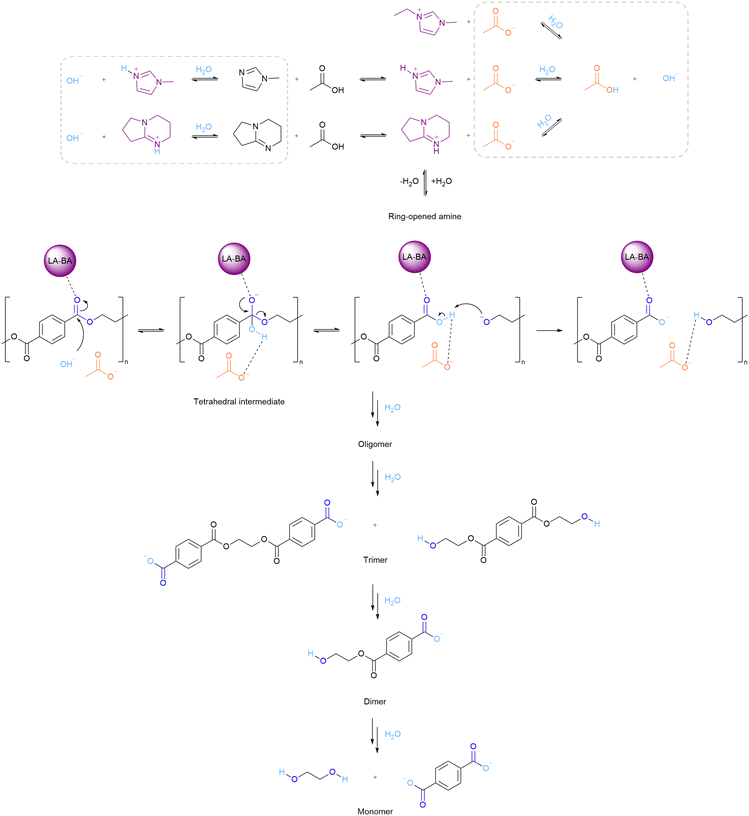 | ||
| Fig. 12 Proposed IL-catalysed PET hydrolysis reaction mechanism using aprotic and protic acetate ILs. | ||
It is proposed that of the two protic acetate ILs, only [C1Him][OAc] supplies noticeable quantities of amine without decomposition,54 due to the relatively low pKa difference between amine and acetic acid (ΔpKa < 3). The high PET conversion for acetate ILs, 1-methylimidazole and DBN can be explained by the (irreversible) deprotonation of TPA and carboxylic acid end groups of short oligomers; hence their reactivity is well-correlated with solubility. The data show that all tested ILs reduce the activation energy compared to water, which is a weak nucleophile. However, they require higher temperatures (at the same reaction time) compared to aqueous solutions of NaOH, due to the lower OH− concentration.
The cation has been assigned a stabilising role for the tetrahedral intermediate;18,38,62,63 however, our data do not provide evidence for specific interactions, especially since a number of Brønsted acidic sites are present, such as on the cation, hydronium ions and acetic acid. Generally, activation of the carbonyl group is more important for the acid catalysed ester hydrolysis mechanism than for the base catalysed mechanism. In support of the proposed mechanism, high proton affinity (correlated to the Kamlet–Taft parameter, β) of ILs was reported to lower the reaction rate of esterification (the reverse reaction of hydrolysis).64 It is proposed that in acetic acid–water mixtures, the acid catalysed ester hydrolysis mechanism is applicable. These mechanistic considerations are suggestions and require deeper investigation.
Consideration of process aspects, including solvent cost and hazards
Solvent recovery and cost are key for developing viable solvent-based chemical processes.65 IL stability is a concern in PET depolymerisation due to the elevated temperatures required to achieve good PET conversions and TPA yields at an acceptable reaction rate. Solvent and reagent stability provide an upper limit for reaction temperatures. Fig. 13 depicts a colour change for the [C2C1im][OAc]–water mixture, which started as a colourless solution and gradually turned dark brown. The discolouration was accompanied by the appearance of at least four new peaks (δ = 7.6, 7.2, 6.9 and 3.6 ppm) in the 1H-NMR spectrum at ∼2 mol% (Fig. S32 and Table S4), which suggests dealkylation of 1-ethyl-3-methylimidazolium by acetate to 1-alkylimidazoles and esters of acetic acid. This was expected, given the low stability of anhydrous [C2C1im][OAc] at 180 °C.66,67 Interestingly, this decomposition does not necessarily lower the IL's performance in this application, given that 1-methylimidazole also promoted PET hydrolysis. Varying degrees of discolouration were observed for the other ILs after PET depolymerisation (Fig. S33), except for [C4C1im][MeSO3]. The 1H-NMR spectrum of [DBNH][OAc], after applying the screening conditions, was examined and revealed the appearance of seven new peaks, suggesting again the presence of the ring-opened hydrolysis product (Fig. S25 and Table S2).49 This decomposition may be linked to the lower performance of the [DBNH][OAc]–water mixture, as discussed earlier. In other cases, notable decomposition products were not observed in the 1H-NMR spectra, despite the solvent becoming strongly coloured during heating, for example, the [C1Him][OAc]–water mixture (Fig. S34 and Table S5). A more thorough and quantitative study of the link between PET hydrolysis performance and IL stability under process conditions should be carried out in the future. | ||
| Fig. 13 Samples containing [C2C1im][OAc] with 15 wt% water and 5% shredded bottle PET heated at 180 °C as a function of time. Hydrolysis duration from 0.5 h (left) to 3 h (right) in 0.5 h increments. | ||
[C1Him][OAc] emerged from this study as a promising candidate, with good performance (PET conversion, TPA yield and purity) and stability. Hence, we estimated the cost of the IL using a process model developed by Chen et al.68 and compared it to the cost of the aprotic [C2C1im][OAc] (Table 3), which requires a more complex synthesis involving alkylation of the alkylimidazole and ion exchange. Details of the calculations can be found in the SI (Table S6). The cost estimate reveals that the production cost for the [C1Him][OAc]–water mixture is <10% of the cost of [C2C1im][OAc], whose synthesis was modelled as an alkylation of methylimidazole with ethyl chloride, followed by ion exchange.69 The model assumes that solvent costs are dominated by input cost for protic ILs at scale.68 The cost differences are due to additional synthetic steps needed for aprotic ILs.
| Solvent | Cost of amine ($ per kg of IL) | Cost of acid ($ per kg of IL) | Cost of water ($ per kg) | Solvent cost ($ per kg of IL–water mixture) |
|---|---|---|---|---|
| [C2C1im][OAc] + 15% water | N/A | N/A | $29.8 | |
| [C1Him][OAc] + 15% water | $1.84 | $0.27 | $1.77 × 10−4 | $2.11 |
[C1Him][OAc] (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) + 15% water 2) + 15% water |
$1.29 | $0.38 | $1.67 |
In our cost estimate, excess acid in the IL reduced the solvent cost, due to acetic acid being the cheaper component. A higher water content for hydrolysis may also reduce the cost further; hence increasing the water content should be investigated further. The estimated cost of the [C1Him][OAc]–water compositions is in the range of common organic solvents, such as acetone and ethyl acetate (1.30–1.40 $ per kg).68
When designing a sustainable chemical process, solvent hazards should also be considered, although use of a hazardous solvent can result in an overall more sustainable chemical process.71 ILs are currently produced at a small scale and hence limited commercial toxicity data are available. For example, the [OTf]− anion is undesirable, as it is fluorinated and hence likely persistent in the environment, while the Zn-containing anion is undesirable as Zn is a toxic heavy metal. 1-Methylimidazole has moderate toxicity, however, it has recently been classified as a teratogen under European REACH legislation. DBN is imported in the EU <1 tonne and hence does not have a REACH dossier. It is noted for its corrosiveness which is typical of amines. Acetic acid is also corrosive in its concentrated form and is classified as a flammable liquid but has otherwise low toxicity and is readily biobased. Decomposition products may modulate the solvent toxicity profile. The chemical hazards of the solvent need to be balanced with the benefit of a potential chemical recycling process and compared to the hazards and environmental impact of TPA and EG production from new (fossil) feedstocks.
The addition of an acid to precipitate the TPA from solution as carried out here changes the composition of the IL,23,57 making chemical PET recycling as carried out in this screening study uneconomical, while increasing environmental burdens by requiring fresh solvent. Recovery of TPA by antisolvent crystallisation (with water) and EG by distillation would be preferable and will be investigated for the lead solvent going forward.
Conclusions
This screening study found that hydrolysis of shredded bottle PET in IL–water mixtures strongly depends on anion selection. PET hydrolysis with the aprotic IL, [C2C1im][OAc], yielded screening conditions (180 °C, 3 h, 15 wt% H2O, 5% PET loading), which were successfully used to evaluate a library of aprotic and protic ILs. ILs with acetate anions gave the highest PET conversion and the highest isolated TPA yields (after dilution and acidification), regardless of whether they were protic or aprotic ILs. It was also found that measuring the percentage yield of the precipitated product typically overestimates TPA yield, as other hydrolysis products and solvent impurities are present in the crystallised product, shown by 1H-NMR spectroscopy and HPLC. It is hence strongly recommended that TPA purity is determined quantitatively to better evaluate solvent performance.The pseudo-protic IL, [C1Him][OAc], performed well in the screening, enabling high PET conversion (99%) and crude TPA yield (82%), and good TPA purity (78%) at acceptable temperatures, with 10 times lower solvent cost than the aprotic [C2C1im][OAc], which is encouraging for further study and process development. The IL–water mixtures based on [C1Him][OAc] at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ABRs generated similar TPA yields, showing that increasing the ABR can help lower solvent cost.
2 ABRs generated similar TPA yields, showing that increasing the ABR can help lower solvent cost.
Solubility of TPA could be correlated with the strength of interactions between the carboxylic acid groups in TPA and the anion of the IL, as found for other solutes containing hydroxyl groups such as cellulose or lignin.39 The pKa of the IL anion exceeding the pKa of terephthalate was a strong predictor of TPA solubility. Strong hydrogen bond acceptance could also promote solubilisation of TPA, even if the pKa of the anion was below the threshold. TPA solubility generally correlated with PET conversion, but some IL solvents with notable TPA solubility exhibited low PET conversion under the screening conditions. Based on the available data, it is proposed that acetate ILs promote depolymerisation of PET via the base catalysed ester hydrolysis mechanism, driven by TPA deprotonation. Based on the promising results observed for [C1Him][OAc]–water mixtures, a waste-free recovery method for the monomers TPA and EG should be developed, which is underway in our laboratory.
Author contributions
M. Y. S.: investigation, writing – original draft, formal analysis and visualisation. H. L. J.: methodology, investigation, formal data analysis and visualisation. P. B.: methodology and investigation. P. F.: conceptualisation, funding acquisition and supervision. J. P. H.: conceptualisation, funding acquisition and supervision. A. B.-T.: conceptualisation, funding acquisition, supervision and project administration. All authors contributed to writing – reviewing and editing.Conflicts of interest
The authors have no conflicts to declare.Data availability
Data supporting this article have been included as part of the SI file, which contains details relating to IL synthesis, HPLC method development, characterisation of the isolated TPA and IL solutions after use and details for solvent cost estimations. See DOI: https://doi.org/10.1039/d5gc02409a.Acknowledgements
We acknowledge funding of the work via EP/S025456/1, NE/W503198/1, EP/R513052/1 and EP/S023232/1. The authors thank Ruhi Patel and Reeya Patel for assistance in preliminary experiments, Tom Welton for advice, and Chris Roberts from the Centre for Rapid Online Analysis of Reactions (ROAR) facility for assisting with the development of the HPLC method, funded via EP/R008825/1 and EP/V029037/1.References
- R. Geyer, J. R. Jambeck and K. L. Law, Sci. Adv., 2017, 3, e1700782 CrossRef PubMed.
- A. Rahimi and J. M. García, Nat. Rev. Chem., 2017, 1, 0046 CrossRef.
- K. Kaiser, M. Schmid and M. Schlummer, Recycling, 2017, 3, 1 CrossRef.
- Plastics - the Facts 2022, Plastics Europe, 2022.
- D. K. Schneiderman and M. A. Hillmyer, Macromolecules, 2017, 50, 3733–3749 CrossRef CAS.
- G. P. Karayannidis and D. S. Achilias, Macromol. Mater. Eng., 2007, 292, 128–146 CrossRef CAS.
- A. C. Castillo, M. Malakkal, P. Bexis and K. Brophy, Enabling a greener plastic future through molecular science, Imprerial College London, Institute for Molecular Sciences and Engineering, 2020 Search PubMed.
- M. Babaei, M. Jalilian and K. Shahbaz, J. Environ. Chem. Eng., 2024, 12, 112507 CrossRef CAS.
- G. W. Coates and Y. D. Y. L. Getzler, Nat. Rev. Mater., 2020, 5, 501–516 CrossRef CAS.
- Y.-H. V. Soong, M. J. Sobkowicz and D. Xie, Bioengineering, 2022, 9, 98 CrossRef CAS PubMed.
- K. Ragaert, L. Delva and K. Van Geem, Waste Manage., 2017, 69, 24–58 CrossRef CAS PubMed.
- B. Geyer, G. Lorenz and A. Kandelbauer, eXPRESS Polym. Lett., 2016, 10, 559–586 CrossRef CAS.
- Y. Peng, J. Yang, C. Deng, J. Deng, L. Shen and Y. Fu, Nat. Commun., 2023, 14, 3249 CrossRef CAS PubMed.
- T. Uekert, J. S. DesVeaux, A. Singh, S. R. Nicholson, P. Lamers, T. Ghosh, J. E. McGeehan, A. C. Carpenter and G. T. Beckham, Green Chem., 2022, 24, 6531–6543 RSC.
- L. Cottam and R. P. Sheldon, Nature, 1965, 205, 1005–1005 CrossRef.
- A. M. Al-Sabagh, F. Z. Yehia, A.-M. M. F. Eissa, M. E. Moustafa, G. Eshaq, A.-R. M. Rabie and A. E. ElMetwally, Ind. Eng. Chem. Res., 2014, 53, 18443–18451 CrossRef CAS.
- Y. Liu, X. Yao, H. Yao, Q. Zhou, J. Xin, X. Lu and S. Zhang, Green Chem., 2020, 22, 3122–3131 RSC.
- R. Zhang, X. Zheng, X. Yao, K. Song, Q. Zhou, C. Shi, J. Xu, Y. Li, J. Xin, I. E.-T. El Sayed and X. Lu, Ind. Eng. Chem. Res., 2023, 62, 11851–11861 CrossRef CAS.
- G. Grause, J. Sutton, A. P. Dove, N. A. Mitchell and J. Wood, Cryst. Growth Des., 2024, 24, 7306–7321 CrossRef CAS PubMed.
- T. Wang, X. Gong, C. Shen, G. Yu and X. Chen, Polym. Degrad. Stab., 2021, 190, 109601 CrossRef CAS.
- C. N. Onwucha, C. O. Ehi-Eromosele, S. O. Ajayi, M. Schaefer, S. Indris and H. Ehrenberg, Ind. Eng. Chem. Res., 2023, 62, 6378–6385 CrossRef CAS.
- S. Ügdüler, K. M. Van Geem, R. Denolf, M. Roosen, N. Mys, K. Ragaert and S. De Meester, Green Chem., 2020, 22, 5376–5394 RSC.
- O. A. Attallah, M. Azeem, E. Nikolaivits, E. Topakas and M. B. Fournet, Polymers, 2022, 14, 109 CrossRef CAS PubMed.
- M. Rollo, F. Raffi, E. Rossi, M. Tiecco, E. Martinelli and G. Ciancaleoni, Chem. Eng. J., 2023, 456, 141092 CrossRef CAS.
- I. Cano, C. Martin, J. A. Fernandes, R. W. Lodge, J. Dupont, F. A. Casado-Carmona, R. Lucena, S. Cardenas, V. Sans and I. de Pedro, Appl. Catal., B, 2020, 260, 118110 CrossRef CAS.
- A. M. Al-Sabagh, F. Z. Yehia, A. M. F. Eissa, M. E. Moustafa, G. Eshaq, A. M. Rabie and A. E. ElMetwally, Polym. Degrad. Stab., 2014, 110, 364–377 CrossRef CAS.
- T. Wang, C. Shen, G. Yu and X. Chen, Polym. Degrad. Stab., 2022, 203, 110050 CrossRef CAS.
- C. Jehanno, I. Flores, A. P. Dove, A. J. Müller, F. Ruipérez and H. Sardon, Green Chem., 2018, 20, 1205–1212 RSC.
- R. Zhang, X. Zheng, X. Cheng, J. Xu, Y. Li, Q. Zhou, J. Xin, D. Yan and X. Lu, Materials, 2024, 17, 1583 CrossRef CAS PubMed.
- C. Zhu, L. Yang, C. Chen, G. Zeng and W. Jiang, Phys. Chem. Chem. Phys., 2023, 25, 27936–27941 RSC.
- J. D. Badia, R. Ballesteros-Garrido, A. Gamir-Cobacho, O. Gil-Castell and A. Cháfer, J. Environ. Chem. Eng., 2024, 12, 113134 CrossRef CAS.
- N. Liu, Y. S. Ma, K. W. Shu, B. Wu and D. Zhang, Adv. Mater. Res., 2014, 893, 23–26 Search PubMed.
- R. M. Musale and S. R. Shukla, J. Text. Inst., 2017, 108, 467–471 CrossRef CAS.
- C. Dou, H. Choudhary, Z. Wang, N. R. Baral, M. Mohan, R. A. Aguilar, S. Huang, A. Holiday, D. R. Banatao, S. Singh, C. D. Scown, J. D. Keasling, B. A. Simmons and N. Sun, One Earth, 2023, 6, 1576–1590 CrossRef.
- J. Sun, D. Liu, R. P. Young, A. G. Cruz, N. G. Isern, T. Schuerg, J. R. Cort, B. A. Simmons and S. Singh, ChemSusChem, 2018, 11, 781–792 CrossRef CAS PubMed.
- F. Liu, X. Cui, S. Yu, Z. Li and X. Ge, J. Appl. Polym. Sci., 2009, 114, 3561–3565 CrossRef CAS.
- A. S. Amarasekara, J. A. Gonzalez and V. C. Nwankwo, J. Ionic Liq., 2022, 2, 100021 CrossRef.
- Z. Zhao, J. Bai, H. Tao, S. Wang, K. Wang, W. Lin, L. Jiang, H. Li and C. Wang, Green Chem. Eng., 2024 DOI:10.1016/j.gce.2024.12.001.
- A. Brandt, J. Grasvik, J. P. Hallett and T. Welton, Green Chem., 2013, 15, 550–583 RSC.
- A. J. Greer, J. Jacquemin and C. Hardacre, Molecules, 2020, 25, 5207 CrossRef CAS PubMed.
- D. Sanna, V. Ugone, G. Micera, P. Buglyó, L. Bíró and E. Garribba, Dalton Trans., 2017, 46, 8950–8967 RSC.
- C. Wiles and P. Watts, Beilstein J. Org. Chem., 2011, 7, 1360–1371 CrossRef CAS PubMed.
- S. Kaiho, A. A. R. Hmayed, K. R. D. Chiaie, J. C. Worch and A. P. Dove, Macromolecules, 2022, 55, 10628–10639 CrossRef CAS.
- L. Cosimbescu, D. R. Merkel, J. Darsell and G. Petrossian, Ind. Eng. Chem. Res., 2021, 60, 12792–12797 CrossRef CAS.
- G. P. Karayannidis, A. P. Chatziavgoustis and D. S. Achilias, Adv. Polym. Technol., 2002, 21, 250–259 CrossRef CAS.
- G. O. Andrés, A. M. Granados and R. H. de Rossi, J. Org. Chem., 2001, 66, 7653–7657 CrossRef PubMed.
- P. A. R. Pires, N. I. Malek, T. C. Teixeira, T. A. Bioni, H. Nawaz and O. A. E. Seoud, Ind. Crops Prod., 2015, 77, 180–189 CrossRef CAS.
- F. S. Pereira, D. L. da S. Agostini, R. D. do E. Santo, E. R. deAzevedo, T. J. Bonagamba, A. E. Job and E. R. P. González, Green Chem., 2011, 13, 2146–2153 RSC.
- A. Parviainen, R. Wahlström, U. Liimatainen, T. Liitiä, S. Rovio, J. K. J. Helminen, U. Hyväkkö, A. W. T. King, A. Suurnäkki and I. Kilpeläinen, RSC Adv., 2015, 5, 69728–69737 RSC.
- Q. F. Yue, H. G. Yang, M. L. Zhang and X. F. Bai, Adv. Mater. Sci. Eng., 2014, 2014, 454756 CrossRef.
- Q. Wang, X. Lu, X. Zhou, M. Zhu, H. He and X. Zhang, J. Appl. Polym. Sci., 2013, 129, 3574–3581 CrossRef CAS.
- L. Wylie, M. Kéri, A. Udvardy, O. Hollóczki and B. Kirchner, ChemSusChem, 2023, 16, e202300535 CrossRef CAS PubMed.
- K. Chen, Y. Wang, J. Yao and H. Li, J. Phys. Chem. B, 2018, 122, 309–315 CrossRef CAS PubMed.
- H. Watanabe, N. Arai, H. Jihae, Y. Kawana and Y. Umebayashi, J. Mol. Liq., 2022, 352, 118705 CrossRef CAS.
- N. S. Allen, M. Edge, J. Daniels and D. Royall, Polym. Degrad. Stab., 1998, 62, 373–383 CrossRef CAS.
- R. A. F. Tomás, J. C. M. Bordado, J. F. P. Gomes and R. J. Sheehan, in Ullmann's Encyclopedia of Industrial Chemistry, 2024, pp. 1–17, DOI:10.1002/14356007.a26_193.pub3.
- J. P. Mészáros, W. Kandioller, G. Spengler, A. Prado-Roller, B. K. Keppler and É. A. Enyedy, Pharmaceutics, 2023, 15, 356 CrossRef PubMed.
- M. A. Ab Rani, A. Brant, L. Crowhurst, A. Dolan, M. Lui, N. H. Hassan, J. P. Hallett, P. A. Hunt, H. Niedermeyer, J. M. Perez-Arlandis, M. Schrems, T. Welton and R. Wilding, Phys. Chem. Chem. Phys., 2011, 13, 16831–16840 RSC.
- K. Matuszek, E. Pankalla, A. Grymel, P. Latos and A. Chrobok, Molecules, 2020, 25, 80 CrossRef CAS PubMed.
- X. Zhou, X. Lu, Q. Wang, M. Zhu and Z. Li, Pure Appl. Chem., 2012, 84, 789–801 CAS.
- J. Clayden, N. Greeves, S. Warren and P. Wothers, Organic Chemistry, Oxford University Press, Oxford, 1st edn, 2001 Search PubMed.
- H. Wang, Z. Li, Y. Liu, X. Zhang and S. Zhang, Green Chem., 2009, 11, 1568–1575 RSC.
- F. Scé, I. Cano, C. Martin, G. Beobide, Ó. Castillo and I. de Pedro, New J. Chem., 2019, 43, 3476–3485 RSC.
- T. P. Wells, J. P. Hallett, C. K. Williams and T. Welton, J. Org. Chem., 2008, 73, 5585–5588 CrossRef CAS PubMed.
- H. Baaqel, I. Díaz, V. Tulus, B. Chachuat, G. Guillén-Gosálbez and J. P. Hallett, Green Chem., 2020, 22, 3132–3140 RSC.
- J. D. Oxley, T. Prozorov and K. S. Suslick, J. Am. Chem. Soc., 2003, 125, 11138–11139 CrossRef CAS PubMed.
- M. T. Clough, K. Geyer, P. A. Hunt, J. Mertes and T. Welton, Phys. Chem. Chem. Phys., 2013, 15, 20480–20495 RSC.
- L. Chen, M. Sharifzadeh, N. Mac Dowell, T. Welton, N. Shah and J. P. Hallett, Green Chem., 2014, 16, 3098–3106 RSC.
- M. Morales, Process Design and Sustainability Assessment for Biorefinery Technologies, ETH, Zurich, 2016, DOI:10.3929/ethz-a-010797794.
- R. Turton, J. A. Shaeiwitz, D. Bhattacharyya and W. B. Whiting, Analysis, synthesis, and design of chemical processes, ed.R. Turton, J. A. Shaeiwitz, D. Bhattacharyya and W. B. WhitingPrentice Hall, Boston, 5th edn, 2018 Search PubMed.
- T. Welton, Proc. R. Soc. A, 2015, 471, 20150502 CrossRef PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2025 |